Abstract
Since its migration from the Asian honey bee (Apis cerana) to the European honey bee (Apis mellifera), the ectoparasitic mite Varroa destructor has emerged as a major issue for beekeeping worldwide. Due to a short history of coevolution, the host–parasite relationship between A. mellifera and V. destructor is unbalanced, with honey bees suffering infestation effects at the individual, colony and population levels. Several control solutions have been developed to tackle the colony and production losses due to Varroa, but the burden caused by the mite in combination with other biotic and abiotic factors continues to increase, weakening the beekeeping industry. In this synthetic review, we highlight the main advances made between 2015 and 2020 on V. destructor biology and its impact on the health of the honey bee, A. mellifera. We also describe the main control solutions that are currently available to fight the mite and place a special focus on new methodological developments, which point to integrated pest management strategies for the control of Varroa in honey bee colonies.
Keywords: control methods, honey bee, IPM, resistance, selection, Varroa
Introduction
The mite, Varroa destructor, has been the subject of thorough investigations, since the 1980s after its introduction in Europe to Apis mellifera honey bee populations [1]. V. destructor continues its worldwide expansion and has now been reported in most countries. In the last 10 years, new invasions were noted in Hawaii [2], Reunion Island [3], Madagascar [4], Mauritius Island [5] and also African countries such as Uganda [6] and Ethiopia [7]. In early 2020, only Australia, several countries in Africa and a few islands have yet to report the presence of V. destructor in their A. mellifera populations [8,9]. Despite the extensive literature on the mite (see [10–12] for reviews), its biology and impact on its honey bee host remain partially unknown. The scientific community has also put many efforts into the development and validation of control methods to fight the deadly mite in A. mellifera colonies. This review highlights the main findings revealed by the rather large corpus of literature published between 2015 and early 2020 on V. destructor biology, the mite's pathogenic effects, and solutions developed to fight the mite.
Biology of Varroa destructor
Between 2015 and 2020, the behaviour of V. destructor, its genetics and physiology, have become more precisely understood. Its life cycle can be separated into two phases: the phoretic phase and the reproductive phase. The phoretic phase only concerns the female mite which uses the adult bee as a ‘transportation vector' and food source. During this phase, bees unintentionally take part in the spreading of Varroa within and between honey bee colonies. The reproductive phase begins when the mite enters into an unsealed brood cell containing a 5th stage bee larva, to lay eggs. Recently, Häußermann et al. revealed the possibility of a virgin mite beginning the phoretic phase. In that case, the mite invades a brood cell, lays an unfertilised egg in a new comb, and mates with its male offspring [13]. This finding confirms that female Varroa do not need to mate to lay eggs, and thus, have an arrhenotokous parthenogenetic reproduction system. While it was first thought that Varroa feeds on honey bee haemolymph, recent research shows that the parasite also feeds on the honey bee fat body [14]. Analyses of the whole-life transcriptome and proteome of the mite have helped to understand the gene expression and protein variations during its different life stages and have provided data for further investigations of mite physiology [15,16]. The morphology of Varroa’s heart has been described, its heartbeats have been recorded [17] and the sensory pit organs of the male have been scanned by electron microscopy [18], complementing the morphological research on female pit organs [19]. The identification of the chemosensory receptors on Varroa forelegs confirms the importance of their front legs in understanding their chemical environment [20]. Iovinella and collaborators performed the proteomic analysis of the chemosensory organs of the mites. This study identified novel semiochemical proteins, including odorant-binding proteins which are particularly abundant in mouths and forelegs [21]. Further chemical ecology investigations have indicated that the male Varroa detects female pheromones with its sensory pit organs present on their front legs [18]. All these findings are important to better understand V. destructor mating behaviour, detection and invasion of brood cells close to be capped. We know that V. destructor has the ability to mimic the cuticular hydrocarbons of their A. mellifera hosts at different stages [22]. It seems that this mimicry can rapidly adapt as Varroa is able to harbour the cuticular hydrocarbon profile of a new, artificially-given host [23,24]. Furthermore, V. destructor from A. cerana are better in mimicking new hosts than V. destructor from A. mellifera [23]. To do so, Varroa adapts its n-alkane:alkene ratio to fit with the honey bee's chemical profile [25]. This mimicry stands as a passive ability, because even when dead, Varroa continues to imitate host cuticular hydrocarbons [24]. Nevertheless, the mite needs to have access to the cuticular hydrocarbon of the host in order to adapt its own cuticular hydrocarbons [24].
Varroa destructor genetics
V. destructor infestation of A. mellifera was described as two partially-isolated clones in early 2000 [26], but it now seems that the genetic population of Varroa is more variable than expected [27]. Different haplotypes of V. destructor can be found within a given apiary or colony, ensuring the genetic flow in the mite population [28,29]. V. jacobsoni and V. destructor show very different evolutionary trajectories since their divergence [30]. Despite these differences, it seems that the two subspecies have a potential to hybridise [31]. If that is the case, hybridisation events could result in a new species of Varroa, possibly more detrimental, that would become a greater risk for honey bees.
Varroa-virus duo
The host-parasite relationship between honey bees and Varroa should actually be considered as a three-way relationship, as Varroa presence is closely associated with several bee viruses in colonies [32]. However, clear vectoring by Varroa has only been described for two viral species, deformed wing virus (DWV) and acute bee paralysis virus complex (ABPV) [33–35]. Bee viruses have several routes of vertical and horizontal transmission within the colony, but the vectoring ability of Varroa opens up new, very effective horizontal routes of transmission [36,37]. In the last 5 years, new viruses and variants linked to Varroa have been discovered in honey bees infested by the mite [38–42], and the association between DWV and the mite has been confirmed, but only for specific DWV variants [2,32]. New variants and viral species have also been described specifically in the mite, as demonstrated by VDV replication in the mite [43], in which, VDV-5, VDV-3 and VDV-2 can replicate in V. destuctor and not in A. mellifera, suggesting that their presence in honey bees is due to the Varroa’s feeding behaviour [38].
It is thought that some of the viruses infesting mites can change Varroa behaviour, which presents a new perspective for finding targets to kill the mite [43]. Similarly to its impact on the virus community infecting bees, the mite also alters the honey bee's bacteriome [44,45], but the impact of such an association remains unknown.
What can it do?
Impact of the mite at the individual level
The parasitism of honey bees by V. destructor decreases the body weight and water content of young emerging bees [46]. The lowered weight of the future adult bee increases with the number of mite foundresses [47,48]. In honey bees, the number of spermatozoids is correlated with drone body size [49]. By decreasing the size of drones, Varroa induce a deficit in sperm production, and thus, in reproductive fitness [50]. Varroa also alters flying, homing and orientation abilities in foragers [51], which in turn, limits efficiency in their ability to collect resources needed for colony development. Non-returning to the colony can also be considered as a mechanism of defence from parasited bees. The impact of Varroa on bee behaviour may be explained by its ability to alter neural processes [52], disrupting the bee-host's non-associative learning abilities. Indeed, parasited bees have a lower sugar responsiveness and a faster habituation to olfactory stimulation [52]. Moreover, Varroa provokes the down-regulation of immune gene expression in emerging infested adults [45], as well as proteomic changes in the honey bee's immune response [53–55]. It disrupts the bee's immune response by interfering in the cascade immune response [56]. For instance, Varroa reduces the number of haemocytes circulating in the haemolymph, and lowers the expression of prophenol oxidase involved in the synthesis of melanin [57,58]. Both haemocytes and melanin permit the encapsulation of pathogens during infection or wounding, and thus play a role in insect immune response and healing mechanisms [59]. The weakening of honey bee immunity [60,61] may be linked to the finding that mites feed on fat bodies, given that these organs play a major role in immunity [62].
The impact of Varroa coupled with other stressors of the colony
Varroa can interact with other biotic and abiotic stressors, such as environmental factors, other parasites and pathogens, pesticides or viruses. Climate change induces longer periods of brood rearing in honey bee colonies and foraging because of longer warm seasons. Longer brood period means more Varroa reproduction cycles and may lead to an increase in mite populations [63]. Nosema infection reduces the effectiveness of bee defences against the mite. In addition, neonicotinoid pesticides and Varroa both contribute to the decrease of winter honey bee population of the colony [64,65]. Together with another neonicotinoid, imidacloprid, Varroa decreases the bee's flying ability [66]. Monchanin et al. [67] demonstrate the negative impact of Varroa coupled with the neonicotinoid insecticide thiamethoxam on honey bee homing behaviour. Also, the Varroa-virus duo plays a key role in the weakening of the colony [68]. Emerging adult bees that were parasitised during the pupal stage show a higher infection rate of DWV compared with non-parasitised individuals [45]. DWV infection induces pathological effects such as crumpled wings and reduced body size leading to behavioural impairment. The DWV titer in honey bees increases as the immunity of the bee decreases [69]. DWV virus can also immunocompromise bees, which may result in a beneficial effect on Varroa reproduction [70].
As colonies are constantly exposed to different stressors at the same time, further research needs to focus on the interaction between two or more stressors at the same time to investigate their combined impact on honey bee fitness. For example, to determine interactions between co-occurring viruses and Varroa on honey bee health.
Honey bee colony dynamics favours Varroa population growth
The population of V. destructor in a colony is directly related to the amount of brood and, by extension, the colony's population size. One way for a colony to decrease the mite population is to create a broodless period. To do so, an increase in the frequency of the reproductive behaviour of swarming can be an adaptive defence mechanism of the honey bee colony against the overpopulation of mites. [71] (Figure 1).
Figure 1. Impact of Varroa destructor parasitism on Apis mellifera honey bees.
Varroa’s impact can be described at individual, colony and population levels. Bold terms correspond to findings published between 2015 and 2019.
Moreover, the number of colonies within an apiary can be advantageous for mite populations. Indeed, colonies of high-density apiaries have a greater infestation rate than that of low-density apiaries [72]. Colonies treated for Varroa can be reinfested when foragers rob food stores from a dying colony or drift into another colony [73], and when drones rest in foreign colonies during the mating period [74].
What can be done?
Different approaches are used by beekeepers to control Varroa infestation of their colonies. Synthetic and organic acaricides as well as essential oils provide good results to prevent colony losses and the latter, represent interesting tools for organic beekeeping [75]. Depending on the country, methods may differ due to variations in the laws governing the use of chemicals.
Chemical control
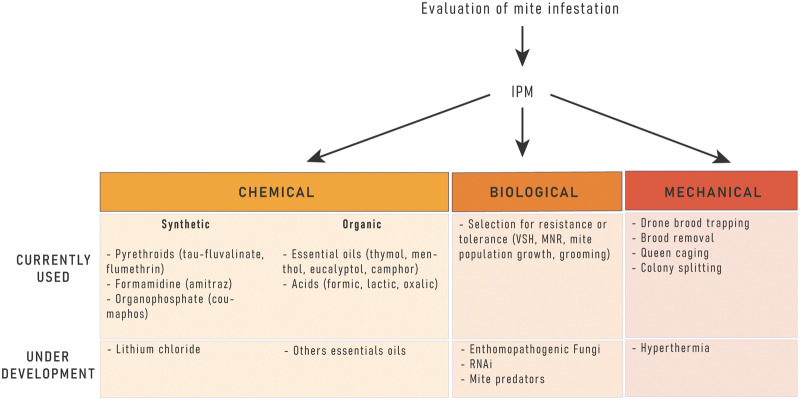
Conventional control using synthetic miticides has been used for more than 40 years against V. destructor [76]. However, only a limited number of molecules are available (Figure 2). They include the pyrethroids tau-fluvalinate (e.g. Apistan®, Klartan®, Mavrik®) and flumethrin (e.g. Bayvarol®, Polyvar yellow®), the formamidine amitraz (e.g. Apivar®, Apitraz®) or the organophosphorus coumaphos (e.g. Checkmite®, Asuntol®, Perizin®) [10,77]. All miticide products kill only Varroa on adult bees, as they cannot target the reproducing mites hidden in the capped brood. To overcome this shortcoming, products made of strips releasing the acaricide compound over time have been successfully developed and mites are killed when they emerge from the brood cells. These strips are easy to use and efficient but have important limitations, like the resistance developed by the mite and the drawback of accumulating as residues in bee products [78].
Figure 2. Methods currently used or under development to treat honey bee colonies against Varroa destructor parasitism.
Methods can be coupled within an integrated pest management scheme (IPM). VSH: Varroa-sensitive hygiene; MNR: Mite non-reproduction.
Mites are becoming increasingly resistant to acaricides [79–81]. Varroa resistance to fluvalinate is now widespread [82], due to DNA mutations [83,84]. Molecular tools have been developed to detect this resistance in Varroa populations [85,86]. A provisional solution to limit and temporally bypass Varroa resistance to miticides is to switch between molecules with different molecular targets. Synthetic acaricides could then provide good results for preventing colony losses [75]. Growing data have been published on acaricide residues in honey bee colony matrices, particularly in wax, which have the chemical properties to store the used lipophilic acaricides at concentrations that could even be toxic to the bees [78,87,88].
Because of the adverse impact that conventional synthetic acaricides have on bees and bee products worldwide, beekeepers are increasingly using organic control methods. Organic methods are usually less efficient compared with conventional synthetic acaricide treatment, but still effectively control mite populations [89]. The most common are essential oils such as thymol, and organic acids like oxalic acid and formic acid [77] (Figure 2). Organic acids are naturally found in bee products and have lower risk for triggering resistance in mites [77], but can still have some negative effects on bees, such as decreasing worker populations, increasing capped brood removal or decreasing drone sperm quality [90].
The use of organic control mostly involves flash treatments and thus needs a broodless colony to be efficient in killing phoretic Varroa. As such, organic product applications coupled with mechanical methods to exclude the brood can provide a good Varroa control solution [91,92]. Queen caging or brood removal can artificially create a broodless colony that keeps the mites on adult bees, making them accessible to acaricides [91,93]. A recent study suggests that while brood removal may result in a decrease in honey production, this loss can be compensated for by avoiding the use of acaricides, enabling a better price of hive products to be achieved [94]. Recently, a new formulation, to be used when brood is present and based on strips releasing oxalic acid, was tested successfully for Varroa control, creating a real opportunity in the organic control of Varroa [95].
Need for new active compounds
Although several products that efficiently control the mite are available, there is an urgent need for new active compounds because of the risk of Varroa resistance. A screening approach combining in silico screening (virtual screening of a chemolibrary of homology sequence models) with in vitro experiments to search for selectively inhibiting Varroa acetylcholinesterase was successfully developed leading to the discovery of new compounds that have the potential to become new treatments against the mite [96]. Lithium chloride has also been demonstrated as a potential compound against Varroa [97], as well as other essential oils and their combinations [98]. Recently, Bendifallah et al. [99] demonstrated the biological activity of sage essential oil as a Varroa control. However, currently there are no registered products available based on these new bioactive sources. The difficult part about the search of new acaricides to control the mite is that the compounds need to be safe for the bees. Development of in vitro rearing of Varroa under laboratory conditions will provide an efficient platform for rapidly screening activity of new compounds which are potentially useful in Varroa control.
Alternative approaches to the use of chemical treatments
Different techniques have been previously described to limit Varroa infestation, such as the ‘trapping comb technique’ or the use of screen bottom boards to trap the mites. A recent study showed that splitting colonies, which mimic swarming events that can control Varroa growth, could be an effective method for decreasing the mite populations [71].
Biological control methods using parasitic fungi have been developed successfully in controlled experiments [100,101]. Hamiduzzama et al. [100] showed varying virulence of entomopathogenic fungi on V. destructor. Unfortunately, most field experiments on honey bee colonies were not yet successful as the fungi can cause detrimental effects on brood development, queens and worker mortality as well as decreasing of weight in newly emerged adult bees [100]. Another experiment using a Beauveria strain showed an effect on Varroa mortality in the field but no visible negative effect on honey bee health [101]. To date, there is no available method on the market for beekeeping using this technique.
The use of predators has shown mixed results and was unsuccessful when applied to honey bee colonies, as was recently the case in an evaluation of the predatory mite Stratiolaelaps scimitus, which demonstrated interesting results in vials but no effect on honey bee colonies [102].
Hyperthermia has been used since the 1970s, and is based on the better heat resistance of the bees compared with Varroa [103]. Artificially heating the hive stops Varroa reproduction and kills the parasite, without harming the bees, because Varroa reproduction is significantly compromised at 36.5°C and mites die at 38°C [104,105]. Heating systems have been proposed to control the mites in different parts of the world, and there are a few systems available on the market at the moment [103,106].
One other option is to use RNA interference to knock down specific genes of Varroa, which has been studied since 2012 with successful results [43,107,108]. To date, there is no product available for beekeepers [109] (Figure 2).
We have the tools for integrated pest management (IPM)!
A variety of tools available for beekeepers make the development and use of the IPM concept in Varroa control possible (Figure 2). IPM is partly based on limiting pesticide use to when it is only necessary. This requires regular monitoring of the Varroa population levels, in order to detect critical infestations and decide on a treatment. As critical infestation levels differ worldwide, these must be defined for specific regions and biotopes.
Efficient but time-consuming techniques exist to diagnose a Varroa infestation [110]. Mites can be counted in the brood, on adults or on debris at the bottom of the hive. Brood examination consists of opening capped cells to verify Varroa infestation by removing the pupae and counting mites [77,111]. Mite count on adult bees is better documented and increasingly used. It consists of collecting 200–300 bees, separating the mites from bees with a surfactant substance, such as chloroform, alcohol, icing sugar or acaricide treatment, and counting the removed mites [77]. Debris examination can be operated with a sticky sheet placed on the floor of the hive with a thin wire mesh on top of it to prevent bees from cleaning out the fallen mites. Fallen mites stick to the board and honey bees are not able to remove the parasite from the hive [77]. As it is a time consuming and tedious method, a stratified method has been proposed to make an accurate estimation of the mites [112].
New techniques, using technological developments such as gas sensors or computer vision systems, are being tested to estimate Varroa infestation [113,114]. They have yet to be transferred to the field and to the beekeeping community.
Selective breeding of naturally resistant or tolerant honey bee populations
In parallel to the development of control solutions to fight the mite, quantitative geneticists and bee breeders have started to search for a longer-term, sustainable solution: selecting honey bee populations that can survive mite infestation without treatments. Several surviving honey bee populations have been identified or bred throughout the world (for a review of these efforts, see [115–117]). Examples of large-scale use of such populations in beekeeping are scarce [118,119] and currently limited by the lack of tools that allow selection of surviving honey bee colonies in the field.
Such tool development relies on the identification of specific phenotypes that characterise these populations. To do so, a better understanding of the mechanisms that undergird the ability to survive is necessary. Surviving can occur through the expression of resistance or tolerance traits, with resistance involving a reduction in Varroa growth, while tolerance reduces parasitic burden despite similar levels of Varroa growth [120]. A wide range of traits involved in honey bee survival to Varroa have been identified, and mainly relate to resistance mechanisms [121]. Tolerance has so far been suggested only in cases (e.g. in the Gotland population in Sweden) where colonies are able to support Varroa burden due to mechanisms of tolerance or resistance to the viruses that are associated with Varroa infestations [42,122].
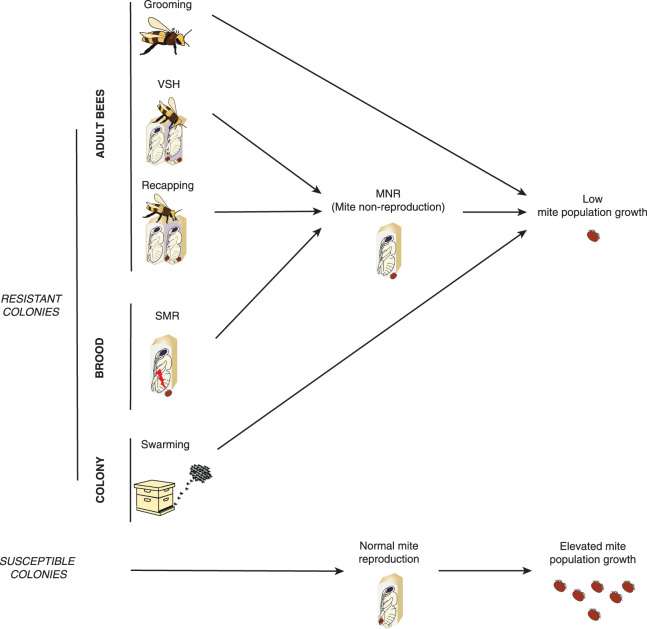
Recent investigations highlight the importance of behavioural defences displayed by Varroa resistant honey bee populations (Figure 3). Hygienic behaviour specifically targeting Varroa-infested capped brood cells (VSH, Varroa-sensitive hygiene) has been confirmed as a major trait contributing to reduced mite population growth in European and African bee populations [123–125]. Two other adult bee traits, grooming [126] and recapping [127,128], have been confirmed as important mechanisms for Varroa resistance. At the colony level, swarming can enhance resistance in surviving populations living in the wild [71,129]. Brood traits could also be involved in resistance abilities, if they confer hypersensibility of the brood that leads to the accelerated death or an increased rate of removal, thereby preventing the spread and reproduction of Varroa [110,130]. Altogether, these traits participate in limiting mite population growth as characterised by high levels of mite non-reproduction within the brood.
Figure 3. Main behavioural and physiological traits involved in the natural resistance of honey bees to the parasite Varroa.
VSH (Varroa-sensitive hygiene), recapping and SMR (suppressed mite reproduction) contribute to MNR (mite non-reproduction). Together with grooming and swarming these traits lead to colony resistant through low mite population growth.
Research has mostly focused on host traits to explain the survival of untreated colonies, but the parasite itself may play a central role too. Indeed host fitness can directly be affected by parasite fitness, as suggested by studies showing that Varroa infesting surviving colonies are genetically distinct from Varroa infesting neighbouring susceptible colonies [131,132].
Progress in understanding the mechanisms that underlie resistance abilities along with the recent boost in genomic tool development has opened the possibility of devising a diagnostic tool of resistance based on phenotype, directly accessible to beekeepers. So far, available methods are very tedious and difficult to apply in the field on large numbers of colonies. This is true for measures of mite population growth, mite non-reproduction or hygienic behaviours. The recent identifications of molecular and protein markers of several traits related to resistance (grooming, hygiene, VSH and mite non-reproduction), although very limited in overlap between different studies [121], opens up the possibility for marker-assisted selection [133–140]. It would allow beekeepers to easily select their colonies for on the basis of interesting resistance traits when the phenotype are difficult to characterise. To date, there are no products available on the market and research is continuing in this area.
Another perspective for the development of selection tools originates specifically from a detailed understanding of the mechanisms of VSH behaviour, a trait in which honey bees are able to specifically detect Varroa-infested brood. Strong evidence suggests that the recognition step involves the detection of Varroa infestation-associated semiochemicals [16,141–144]. Evaluation of the bee response following application of such candidate compounds in colonies, by acting as a reliable proxy of the VSH activity of the colony, could result in a practical field tool to phenotype resistant colonies.
Conclusion
Despite the amount of research done on the A. mellifera–V. destructor host-parasite model, Varroa remains a major issue for beekeeping throughout the world. Future developments, both in fundamental and applied research, are necessary to generate sustainable control solutions for this deadly parasite.
Summary
The latest findings on the physiology of Varroa and its behaviour provide a better understanding of its negative impact on bee health.
IPM methods can be used to limit the use of acaricides to control Varroa. The selection of resistant or tolerant honey bee populations could bring a sustainable mite control solution for beekeeping and wild honey bee populations.
Research needs to focus on the development of control methods, especially new active compounds to counter the mite's resistance against acaricides and to efficiently fight V. destructor.
Abbreviations
- ABPV
acute bee paralysis virus complex
- DWV
deformed wing virus
- IPM
integrated pest management
- VSH
Varroa-sensitive hygiene
Competing Interests
The authors declare that there are no competing interests associated with the manuscript.
Funding
Our work was supported by a PRIMA grant (EU H2020, #1812, ‘Plant-B') and by The Persephone Charitable and Environmental Trust.
Author Contributions
A.N., F.M. and Y.L.-C. designed and wrote the manuscript.
References
- 1.Oldroyd B.P. (1999) Coevolution while you wait: Varroa jacobsoni, a new parasite of western honeybees. Trends Ecol. Evol. 14, 312–315 10.1016/S0169-5347(99)01613-4 [DOI] [PubMed] [Google Scholar]
- 2.Santamaria J., Villalobos E.M., Brettell L.E., Nikaido S., Graham J.R. and Martin S. (2018) Evidence of Varroa-mediated deformed wing virus spillover in Hawaii. J. Invertebr. Pathol. 151, 126–130 10.1016/j.jip.2017.11.008 [DOI] [PubMed] [Google Scholar]
- 3.Esnault O. (2018) Diversité des agents pathogènes de l'abeille dans le Sud-Ouest de l'Océan Indien dans un contexte d'invasion récente de Varroa destructor et mortalités associées. [Doctoral thesis] La Réunion: Université de la Réunion
- 4.Rasolofoarivao H., Clémencet J., Ravaomanarivo L.H.R., Razafindrazaka D., Reynaud B. and Delatte H. (2013) Spread and strain determination of Varroa destructor (Acari: Varroidae) in Madagascar since its first report in 2010. Exp. Appl. Acarol. 60, 521–530 10.1007/s10493-013-9658-x [DOI] [PubMed] [Google Scholar]
- 5.Esnault O., Meenowa D., Sookar P., Chauzat M.-P. and Delatte H. (2019) Spread and strain determination of Varroa destructor following its introduction to Mauritius and interactions with the bee louse Braula pretoriensis in honey bee colonies. J. Apic. Res. 58, 75–83 10.1080/00218839.2018.1517987 [DOI] [Google Scholar]
- 6.Kasangaki P., Sarah Otim A., P'Odyek Abila P., Angiro C., Chemurot M. and Kajobe R. (2015) The presence of Varroa in Uganda and knowledge about it by the beekeeping industry. J. Apic. Res. 54, 373–377 10.1080/00218839.2016.1159858 [DOI] [Google Scholar]
- 7.Begna D. (2014) Occurrences and distributions of honeybee (Apis mellifera jemenetica) varroa mite (Varroa destructor) in Tigray Region, Ethiopia. J. Fisheries Livest. Prod. 03, 1000126 10.4172/2332-2608.1000126 [DOI] [Google Scholar]
- 8.Roberts J.M.K., Anderson D.L. and Durr P.A. (2017) Absence of Deformed Wing Virus and Varroa destructor in Australia provides unique perspectives on honeybee viral landscapes and colony losses. Sci. Rep. 7, 6925 10.1038/s41598-017-07290-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Muli E., Okwaro L.A., Kilonzo J., Ali N. and Monthy G.T. (2018) Varroa destructor – free islands in the south west Indian ocean. Bee World 95, 122–123 10.1080/0005772X.2018.1522835 [DOI] [Google Scholar]
- 10.Rosenkranz P., Aumeier P. and Ziegelmann B. (2010) Biology and control of Varroa destructor. J. Invertebr. Pathol. 103, 96–S119 10.1016/j.jip.2009.07.016 [DOI] [PubMed] [Google Scholar]
- 11.Sammataro D., Gerson U. and Needham G. (2000) Parasitic mites of honey bees: life history, implications, and impact. Annu. Rev. Entomol. 45, 519–548 10.1146/annurev.ento.45.1.519 [DOI] [PubMed] [Google Scholar]
- 12.Roth M.A., Wilson J.M., Tignor K.R. and Gross A.D. (2020) Biology and management of Varroa destructor (Mesostigmata: Varroidae) in Apis mellifera (Hymenoptera: Apidae) Colonies. J. Integr. Pest Manag. 11, 1 10.1093/jipm/pmz036 [DOI] [Google Scholar]
- 13.Häußermann C.K., Giacobino A., Munz R., Ziegelmann B., Palacio M.A. and Rosenkranz P. (2019) Reproductive parameters of female Varroa destructor and the impact of mating in worker brood of Apis mellifera. Apidologie 10.1007/s13592-019-00713-9 [DOI] [Google Scholar]
- 14.Ramsey S.D., Ochoa R., Bauchan G., Gulbronson C., Mowery J.D., Cohen A. et al. (2019) Varroa destructor feeds primarily on honey bee fat body tissue and not hemolymph. Proc. Natl. Acad. Sci. U.S.A. 116, 1792–1801 10.1073/pnas.1818371116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Mondet F., Rau A., Klopp C., Rohmer M., Severac D., Le Conte Y. et al. (2018) Transcriptome profiling of the honeybee parasite Varroa destructor provides new biological insights into the mite adult life cycle. BMC Genom. 19, 328 10.1186/s12864-018-4668-z [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.McAfee A., Chan Q.W.T., Evans J. and Foster L.J. (2017) A Varroa destructor protein atlas reveals molecular underpinnings of developmental transitions and sexual differentiation. Mol. Cell. Proteom. 16, 2125–2137 10.1074/mcp.RA117.000104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Koutouvela E. and Papachristoforou A. (2019) The heart of Varroa destructor: description, function and inhibition following acaricide application. Syst. Appl. Acarol. 24, 638–644 10.11158/saa.24.4.9 [DOI] [Google Scholar]
- 18.Häußermann C.K., Ziegelmann B., Bergmann P. and Rosenkranz P. (2015) Male mites (Varroa destructor) perceive the female sex pheromone with the sensory pit organ on the front leg tarsi. Apidologie 46, 771–778 10.1007/s13592-015-0367-9 [DOI] [Google Scholar]
- 19.Liu T.P. (1988) The morphology of the tarsal sensilla in the female mite Varroa jacobsoni. Pan-Pac. Entomol. 64, 261–265 [Google Scholar]
- 20.Eliash N., Singh N.K., Thangarajan S., Sela N., Leshkowitz D., Kamer Y. et al. (2017) Chemosensing of honeybee parasite, Varroa destructor: transcriptomic analysis. Sci. Rep. 7, 13091 10.1038/s41598-017-13167-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Iovinella I., McAfee A., Mastrobuoni G., Kempa S., Foster L.J., Pelosi P. et al. (2018) Proteomic analysis of chemosensory organs in the honey bee parasite Varroa destructor: a comprehensive examination of the potential carriers for semiochemicals. J. Proteom. 181, 131–141 10.1016/j.jprot.2018.04.009 [DOI] [PubMed] [Google Scholar]
- 22.Martin C., Salvy M., Provost E., Bagnères A.-G., Roux M., Crauser D. et al. (2001) Variations in chemical mimicry by the ectoparasitic mite Varroa jacobsoni according to the developmental stage of the host honey-bee Apis mellifera. Insect Biochem. Mol. Biol. 31, 365–379 10.1016/S0965-1748(00)00130-2 [DOI] [PubMed] [Google Scholar]
- 23.Le Conte Y., Huang Z.Y., Roux M., Zeng Z.J., Christidès J.-P. and Bagnères A.-G. (2015) Varroa destructor changes its cuticular hydrocarbons to mimic new hosts. Biol. Lett. 11, 20150233 10.1098/rsbl.2015.0233 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Kather R., Drijfhout F.P., Shemilt S. and Martin S.J. (2015) Evidence for passive chemical camouflage in the parasitic mite Varroa destructor. J. Chem. Ecol. 41, 178–186 10.1007/s10886-015-0548-z [DOI] [PubMed] [Google Scholar]
- 25.Kather R., Drijfhout F.P. and Martin S.J. (2015) Evidence for colony-specific differences in chemical mimicry in the parasitic mite Varroa destructor. Chemoecology 25, 215–222 10.1007/s00049-015-0191-8 [DOI] [PubMed] [Google Scholar]
- 26.Solignac M., Cornuet J., Vautrin D., Le Conte Y., Anderson D., Evans J. et al. (2005) The invasive Korea and Japan types of Varroa destructor, ectoparasitic mites of the Western honeybee (Apis mellifera), are two partly isolated clones. Proc. R. Soc. B 272, 411–419 10.1098/rspb.2004.2853 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Beaurepaire A.L., Krieger K.J. and Moritz R.F.A. (2017) Seasonal cycle of inbreeding and recombination of the parasitic mite Varroa destructor in honeybee colonies and its implications for the selection of acaricide resistance. Infect. Genet. Evol. 50, 49–54 10.1016/j.meegid.2017.02.011 [DOI] [PubMed] [Google Scholar]
- 28.Gajić B., Muñoz I., De La Rúa P., Stevanović J., Lakić N., Kulišić Z. et al. (2019) Coexistence of genetically different Varroa destructor in Apis mellifera colonies. Exp. Appl. Acarol. 78, 315–326 10.1007/s10493-019-00395-z [DOI] [PubMed] [Google Scholar]
- 29.Dynes T.L., De Roode J.C., Lyons J.I., Berry J.A., Delaplane K.S. and Brosi B.J. (2017) Fine scale population genetic structure of Varroa destructor, an ectoparasitic mite of the honey bee (Apis mellifera). Apidologie 48, 93–101 10.1007/s13592-016-0453-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Techer M.A., Rane R.V., Grau M.L., Roberts J.M.K., Sullivan S.T., Liachko I. et al. (2019) Divergent evolutionary trajectories following speciation in two ectoparasitic honey bee mites. Commun. Biol. 2, 357 10.1038/s42003-019-0606-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Dietemann V., Beaurepaire A., Page P., Yañez O., Buawangpong N., Chantawannakul P. et al. (2019) Population genetics of ectoparasitic mites Varroa spp. in Eastern and Western honey bees. Parasitology 146, 1429–1439 10.1017/S003118201900091X [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mondet F., de Miranda J.R., Kretzschmar A., Le Conte Y. and Mercer A.R. (2014) On the front fine: quantitative virus dynamics in honeybee (Apis mellifera L.) colonies along a new expansion front of the parasite Varroa destructor. PLoS Pathog. 10, e1004323 10.1371/journal.ppat.1004323 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.de Miranda J.R. and Genersch E. (2010) Deformed wing virus. J. Invertebr. Pathol. 103, S48–S61 10.1016/j.jip.2009.06.012 [DOI] [PubMed] [Google Scholar]
- 34.Martin S.J. and Brettell L.E. (2019) Deformed wing virus in honeybees and other insects. Annu. Rev. Virol. 6, 49–69 10.1146/annurev-virology-092818-015700 [DOI] [PubMed] [Google Scholar]
- 35.de Miranda J.R., Cordoni G. and Budge G. (2010) The Acute bee paralysis virus–Kashmir bee virus–Israeli acute paralysis virus complex. J. Invertebr. Pathol. 103, S30–S47 10.1016/j.jip.2009.06.014 [DOI] [PubMed] [Google Scholar]
- 36.de Miranda J.R., Bailey L., Ball B.V., Blanchard P., Budge G.E., Chejanovsky N. et al. (2013) Standard methods for virus research in Apis mellifera. J. Apic. Res. 52, 1–56 10.3896/IBRA.1.52.4.22 [DOI] [Google Scholar]
- 37.Amiri E., Kryger P., Meixner M.D., Strand M.K., Tarpy D.R. and Rueppell O. (2018) Quantitative patterns of vertical transmission of deformed wing virus in honey bees. PLoS One 13, e0195283 10.1371/journal.pone.0195283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Herrero S., Millán-Leiva A., Coll S., González-Martínez R.M., Parenti S. and González-Cabrera J. (2019) Identification of new viral variants specific to the honey bee mite Varroa destructor. Exp. Appl. Acarol. 79, 157–168 10.1007/s10493-019-00425-w [DOI] [PubMed] [Google Scholar]
- 39.Levin S., Sela N. and Chejanovsky N. (2016) Two novel viruses associated with the Apis mellifera pathogenic mite Varroa destructor. Sci. Rep. 6, 37710 10.1038/srep37710 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Levin S., Sela N., Erez T., Nestel D., Pettis J., Neumann P. et al. (2019) New viruses from the ectoparasite mite Varroa destructor infesting Apis mellifera and Apis cerana. Viruses 11, 94 10.3390/v11020094 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.de Miranda J., Cornman R., Evans J., Semberg E., Haddad N., Neumann P. et al. (2015) Genome characterization, prevalence and distribution of a macula-like virus from Apis mellifera and Varroa destructor. Viruses 7, 3586–3602 10.3390/v7072789 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mordecai G.J., Brettell L.E., Pachori P., Villalobos E.M., Martin S.J., Jones I.M. et al. (2016) Moku virus; a new Iflavirus found in wasps, honey bees and Varroa. Sci. Rep. 6, 34983 10.1038/srep34983 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Campbell E.M., Budge G.E., Watkins M. and Bowman A.S. (2016) Transcriptome analysis of the synganglion from the honey bee mite, Varroa destructor and RNAi knockdown of neural peptide targets. Insect Biochem. Mol. Biol. 70, 116–126 10.1016/j.ibmb.2015.12.007 [DOI] [PubMed] [Google Scholar]
- 44.Hubert J., Bicianova M., Ledvinka O., Kamler M., Lester P.J., Nesvorna M. et al. (2017) Changes in the bacteriome of honey bees associated with the parasite Varroa destructor, and pathogens Nosema and Lotmaria passim. Microb. Ecol. 73, 685–698 10.1007/s00248-016-0869-7 [DOI] [PubMed] [Google Scholar]
- 45.Marche M.G., Satta A., Floris I., Pusceddu M., Buffa F. and Ruiu L. (2019) Quantitative variation in the core bacterial community associated with honey bees from Varroa- infested colonies. J. Apic. Res. 58, 444–454 10.1080/00218839.2019.1589669 [DOI] [Google Scholar]
- 46.Bowen-Walker P.L. and Gunn A. (2001) The effect of the ectoparasitic mite, Varroa destructor on adult worker honeybee (Apis mellifera) emergence weights, water, protein, carbohydrate, and lipid levels. Entomol. Exp. Appl. 101, 207–217 10.1046/j.1570-7458.2001.00905.x [DOI] [Google Scholar]
- 47.Duay P., Jong D.D. and Engels W. (2003) Weight loss in drone pupae (Apis mellifera) multiply infested by Varroa destructor mites. Apidologie 34, 61–65 10.1051/apido:2002052 [DOI] [Google Scholar]
- 48.Strauss U., Dietemann V., Human H., Crewe R.M. and Pirk C.W.W. (2016) Resistance rather than tolerance explains survival of savannah honeybees (Apis mellifera scutellata) to infestation by the parasitic mite Varroa destructor. Parasitology 143, 374–387 10.1017/S0031182015001754 [DOI] [PubMed] [Google Scholar]
- 49.Schlüns H., Schlüns E.A., van Praagh J. and Moritz R.F.A. (2003) Sperm numbers in drone honeybees (Apis mellifera) depend on body size. Apidologie 34, 577–584 10.1051/apido:2003051 [DOI] [Google Scholar]
- 50.Duay P., Jong D.D. and Engels W. (2002) Decreased flight performance and sperm production in drones of the honey bee (Apis mellifera) slightly infested by Varroa destructor mites during pupal development. Genet. Mol. Res. 1, 227–232 PMID: [PubMed] [Google Scholar]
- 51.Kralj J. and Fuchs S. (2006) Parasitic Varroa destructor mites influence flight duration and homing ability of infested Apis mellifera foragers. Apidologie 37, 577–587 10.1051/apido:2006040 [DOI] [Google Scholar]
- 52.Kralj J., Brockmann A., Fuchs S. and Tautz J. (2007) The parasitic mite Varroa destructor affects non-associative learning in honey bee foragers, Apis mellifera L. J. Comp. Physiol. A. 193, 363–370 10.1007/s00359-006-0192-8 [DOI] [PubMed] [Google Scholar]
- 53.Erban T., Sopko B., Kadlikova K., Talacko P. and Harant K. (2019) Varroa destructor parasitism has a greater effect on proteome changes than the Deformed Wing Virus and activates TGF-β signaling pathways. Sci. Rep. 9, 9400 10.1038/s41598-019-45764-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Surlis C., Carolan J.C., Coffey M. and Kavanagh K. (2018) Quantitative proteomics reveals divergent responses in Apis mellifera worker and drone pupae to parasitization by Varroa destructor. J. Insect. Physiol. 107, 291–301 10.1016/j.jinsphys.2017.12.004 [DOI] [PubMed] [Google Scholar]
- 55.Słowińska M., Nynca J., Bąk B., Wilde J., Siuda M. and Ciereszko A. (2019) 2D-DIGE proteomic analysis reveals changes in haemolymph proteome of 1-day-old honey bee (Apis mellifera) workers in response to infection with Varroa destructor mites. Apidologie 50, 632–656 10.1007/s13592-019-00674-z [DOI] [Google Scholar]
- 56.Zaobidna E.A., Żółtowska K. and Łopieńska-Biernat E. (2017) Varroa destructor induces changes in the expression of immunity-related genes during the development of Apis mellifera worker and drone broods. Acta Parasitol. 62, 779–789 10.1515/ap-2017-0094 [DOI] [PubMed] [Google Scholar]
- 57.Koleoglu G., Goodwin P.H., Reyes-Quintana M., Hamiduzzaman M.M. and Guzman-Novoa E. (2018) Varroa destructor parasitism reduces hemocyte concentrations and prophenol oxidase gene expression in bees from two populations. Parasitol. Res. 117, 1175–1183 10.1007/s00436-018-5796-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wegener J., Ruhnke H., Scheller K., Mispagel S., Knollmann U., Kamp G. et al. (2016) Pathogenesis of varroosis at the level of the honey bee (Apis mellifera) colony. J. Insect.Physiol. 91–92, 1–9 10.1016/j.jinsphys.2016.06.004 [DOI] [PubMed] [Google Scholar]
- 59.Kanost M.R. and Gorman M.J (2008) Phenoloxidases in Insect Immunity In Insect Immunology, 1st edn (Beckage N.E., ed.), pp. 69–96, Elsevier/Academic Press, Amsterdam [Google Scholar]
- 60.Annoscia D., Brown S.P., Di Prisco G., De Paoli E., Del Fabbro S., Frizzera D. et al. (2019) Haemolymph removal by Varroa mite destabilizes the dynamical interaction between immune effectors and virus in bees, as predicted by Volterra's model. Proc. R. Soc. B 286, 20190331 10.1098/rspb.2019.0331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Yang X. and Cox-Foster D.L. (2005) Impact of an ectoparasite on the immunity and pathology of an invertebrate: evidence for host immunosuppression and viral amplification. Proc. Natl. Acad. Sci. U.S.A. 102, 7470–7475 10.1073/pnas.0501860102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Arrese E.L. and Soulages J.L. (2010) Insect fat body: energy, metabolism, and regulation. Annu. Rev. Entomol. 55, 207–225 10.1146/annurev-ento-112408-085356 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Le Conte Y., Ellis M. and Ritter W. (2010) Varroa mites and honey bee health: can Varroa explain part of the colony losses? Apidologie 41, 353–363 10.1051/apido/2010017 [DOI] [Google Scholar]
- 64.Straub L., Williams G.R., Vidondo B., Khongphinitbunjong K., Retschnig G., Schneeberger A. et al. (2019) Neonicotinoids and ectoparasitic mites synergistically impact honeybees. Sci. Rep. 9, 8159 10.1038/s41598-019-44207-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.van der Zee R., Gray A., Pisa L. and de Rijk T. (2015) An observational study of honey bee colony winter losses and their association with Varroa destructor, neonicotinoids and other risk factors. PLoS One 10, e0131611 10.1371/journal.pone.0131611 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Blanken L.J., van Langevelde F. and van Dooremalen C. (2015) Interaction between Varroa destructor and imidacloprid reduces flight capacity of honeybees. Proc. R. Soc. B 282, 20151738 10.1098/rspb.2015.1738 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Monchanin C., Henry M., Decourtye A., Dalmon A., Fortini D., Bœuf E. et al. (2019) Hazard of a neonicotinoid insecticide on the homing flight of the honeybee depends on climatic conditions and Varroa infestation. Chemosphere 224, 360–368 10.1016/j.chemosphere.2019.02.129 [DOI] [PubMed] [Google Scholar]
- 68.Barroso-Arévalo S., Fernández-Carrión E., Goyache J., Molero F., Puerta F. and Sánchez-Vizcaíno J.M. (2019) High load of deformed wing virus and Varroa destructor infestation are related to weakness of honey bee colonies in southern Spain. Front. Microbiol. 10, 1331 10.3389/fmicb.2019.01331 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Zhao Y., Heerman M., Peng W., Evans J.D., Rose R., DeGrandi-Hoffman G. et al. (2019) The dynamics of deformed wing virus concentration and host defensive gene expression after Varroa mite parasitism in honey bees, Apis mellifera. Insects 10, 16 10.3390/insects10010016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Di Prisco G., Annoscia D., Margiotta M., Ferrara R., Varricchio P., Zanni V. et al. (2016) A mutualistic symbiosis between a parasitic mite and a pathogenic virus undermines honey bee immunity and health. Proc. Natl. Acad. Sci. U.S.A. 113, 3203–3208 10.1073/pnas.1523515113 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Loftus J.C., Smith M.L. and Seeley T.D. (2016) How honey bee colonies survive in the wild: testing the importance of small nests and frequent swarming. PLoS One 11, e0150362 10.1371/journal.pone.0150362 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Frey E. and Rosenkranz P. (2014) Autumn invasion rates of Varroa destructor (Mesostigmata: Varroidae) into honey bee (Hymenoptera: Apidae) colonies and the resulting increase in mite populations. J. Econ. Entomol. 107, 508–515 10.1603/EC13381 [DOI] [PubMed] [Google Scholar]
- 73.Peck D.T. and Seeley T.D. (2019) Mite bombs or robber lures? The roles of drifting and robbing in Varroa destructor transmission from collapsing honey bee colonies to their neighbors. PLoS One 14, e0218392 10.1371/journal.pone.0218392 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Mortensen A.N., Jack C.J. and Ellis J.D. (2018) The discovery of Varroa destructor on drone honey bees, Apis mellifera, at drone congregation areas. Parasitol. Res. 117, 3337–3339 10.1007/s00436-018-6035-z [DOI] [PubMed] [Google Scholar]
- 75.Haber A.I., Steinhauer N.A. and vanEngelsdorp D. (2019) Use of chemical and nonchemical methods for the control of Varroa destructor (Acari: Varroidae) and associated winter colony losses in US beekeeping operations. J. Econ. Entomol. 112, 1509–1525 10.1093/jee/toz088 [DOI] [PubMed] [Google Scholar]
- 76.Koeniger N. and Fuchs S. (1989) Eleven years with Varroa—experiences, retrospects and prospects. Bee World 70, 148–159 10.1080/0005772X.1989.11099011 [DOI] [Google Scholar]
- 77.Mondet F. and Le Conte Y (2014) Parasites In Bee Health and Veterinarians, pp. 131–141. OIE, World Organisation for Animal Health, Paris [Google Scholar]
- 78.Bajuk P., Babnik B., Snoj K., Milčinski T., Pislak Ocepek L., Škof M. (2017) Coumaphos residues in honey, bee brood, and beeswax after Varroa treatment. Apidologie 48, 588–598 10.1007/s13592-017-0501-y [DOI] [Google Scholar]
- 79.Milani N. (1999) The resistance of Varroa jacobsoni Oud. to acaricides. Apidologie 30, 229–234 10.1051/apido:19990211 [DOI] [Google Scholar]
- 80.Martin S.J. (2004) Acaricide (pyrethroid) resistance in Varroa destructor. Bee World 85, 67–69 10.1080/0005772X.2004.11099632 [DOI] [Google Scholar]
- 81.Colin M.E., Vandame R., Jourdam P. and Di Pasquale S. (1997) Fluvalinate resistance of Varroa jacobsoni Oudemans (Acari: Varroidae) in mediterranean apiaries of France. Apidologie 28, 375–384 10.1051/apido:19970605 [DOI] [Google Scholar]
- 82.Panini M., Reguzzi M.C., Chiesa O., Cominelli F., Lupi D. and Mazzoni E. (2019) Pyrethroid resistance in Italian populations of the mite Varroa destructor: a focus on the Lombardy region. Bull. Insectol. 72, 221–232 [Google Scholar]
- 83.González-Cabrera J., Bumann H., Rodríguez-Vargas S., Kennedy P.J., Krieger K., Altreuther G. et al. (2018) A single mutation is driving resistance to pyrethroids in European populations of the parasitic mite, Varroa destructor. J. Pest. Sci. 91, 1137–1144 10.1007/s10340-018-0968-y [DOI] [Google Scholar]
- 84.Stara J., Pekar S., Nesvorna M., Kamler M., Doskocil I. and Hubert J. (2019) Spatio-temporal dynamics of Varroa destructor resistance to tau-fluvalinate in Czechia, associated with L925V sodium channel point mutation. Pest. Manag. Sci. 75, 1287–1294 10.1002/ps.5242 [DOI] [PubMed] [Google Scholar]
- 85.Stara J., Pekar S., Nesvorna M., Erban T., Vinsova H., Kopecky J. et al. (2019) Detection of tau-fluvalinate resistance in the mite Varroa destructor based on the comparison of vial test and PCR–RFLP of kdr mutation in sodium channel gene. Exp. Appl. Acarol. 77, 161–171 10.1007/s10493-019-00353-9 [DOI] [PubMed] [Google Scholar]
- 86.Millán-Leiva A., Hernández-Rodríguez C.S. and González-Cabrera J. (2018) New PCR–RFLP diagnostics methodology for detecting Varroa destructor resistant to synthetic pyrethroids. J. Pest. Sci. 91, 937–941 10.1007/s10340-018-0964-2 [DOI] [Google Scholar]
- 87.Ostiguy N., Drummond F.A., Aronstein K., Eitzer B., Ellis J.D., Spivak M. et al. (2019) Honey bee exposure to pesticides: a four-year nationwide study. Insects 10, 1–34 10.3390/insects10010013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Kast C., Kilchenmann V. and Droz B. (2019) Distribution of coumaphos in beeswax after treatment of honeybee colonies with CheckMite® against the parasitical mite Varroa destructor. Apidologie 51, 112–122 10.1007/s13592-019-00724-6 [DOI] [Google Scholar]
- 89.Pietropaoli M. and Formato G. (2019) Acaricide efficacy and honey bee toxicity of three new formic acid-based products to control Varroa destructor. J. Apic. Res. 58, 824–830 10.1080/00218839.2019.1656788 [DOI] [Google Scholar]
- 90.Tihelka E. (2018) Effects of synthetic and organic acaricides on honey bee health: a review. Slov. Vet. Res. 55, 119–140 10.26873/SVR-422-2017 [DOI] [Google Scholar]
- 91.Giacomelli A., Pietropaoli M., Carvelli A., Iacoponi F. and Formato G. (2016) Combination of thymol treatment (Apiguard®) and caging the queen technique to fight Varroa destructor. Apidologie 47, 606–616 10.1007/s13592-015-0408-4 [DOI] [Google Scholar]
- 92.Rademacher E. and Harz M. (2006) Oxalic acid for the control of varroosis in honey bee colonies – a review. Apidologie 37, 98–120 10.1051/apido:2005063 [DOI] [Google Scholar]
- 93.Gregorc A., Alburaki M., Werle C., Knight P.R. and Adamczyk J. (2017) Brood removal or queen caging combined with oxalic acid treatment to control Varroa mites (Varroa destructor) in honey bee colonies (Apis mellifera). Apidologie 48, 821–832 10.1007/s13592-017-0526-2 [DOI] [Google Scholar]
- 94.Mancuso T., Croce L. and Vercelli M. (2020) Total brood removal and other biotechniques for the sustainable control of Varroa mites in honey bee colonies: economic impact in beekeeping farm case studies in northwestern Italy. Sustainability 12, 2302 10.3390/su12062302 [DOI] [Google Scholar]
- 95.Maggi M., Tourn E., Negri P., Szawarski N., Marconi A., Gallez L. et al. (2016) A new formulation of oxalic acid for Varroa destructor control applied in Apis mellifera colonies in the presence of brood. Apidologie 47, 596–605 10.1007/s13592-015-0405-7 [DOI] [Google Scholar]
- 96.Riva C., Suzanne P., Charpentier G., Dulin F., Halm-Lemeille M.-P. and Sopkova-de Oliveira Santos J. (2019) In silico chemical library screening and experimental validation of novel compounds with potential varroacide activities. Pest. Biochem. Physiol. 160, 11–19 10.1016/j.pestbp.2019.05.012 [DOI] [PubMed] [Google Scholar]
- 97.Ziegelmann B., Abele E., Hannus S., Beitzinger M., Berg S. and Rosenkranz P. (2018) Lithium chloride effectively kills the honey bee parasite Varroa destructor by a systemic mode of action. Sci. Rep. 8, 683 10.1038/s41598-017-19137-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Brasesco C., Gende L., Negri P., Szawarski N., Iglesias A., Eguaras M. et al. (2017) Assessing in vitro acaricidal effect and joint action of a binary mixture between essential oil compounds (thymol, phellandrene, eucalyptol, cinnamaldehyde, myrcene, carvacrol) over ectoparasitic mite Varroa destructor (Acari: Varroidae). J. Apic. Sci. 61, 203–215 10.1515/jas-2017-0008 [DOI] [Google Scholar]
- 99.Bendifallah L., Belguendouz R., Hamoudi L. and Arab K. (2018) Biological activity of the Salvia officinalis L. (Lamiaceae) essential oil on Varroa destructor infested honeybees. Plants 7, 44 10.3390/plants7020044 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Hamiduzzaman M.Md., Sinia A., Guzman-Novoa E. and Goodwin P.H. (2012) Entomopathogenic fungi as potential biocontrol agents of the ecto-parasitic mite, Varroa destructor, and their effect on the immune response of honey bees (Apis mellifera L.). J. Invertebr. Pathol. 111, 237–243 10.1016/j.jip.2012.09.001 [DOI] [PubMed] [Google Scholar]
- 101.Sewify G.H., Ibrahim Y.Y. and Salah El-Deen M. (2015) Beauveria bassiana, a potential mycopesticide for efficient control of the Honey Bee Ectoparasitic Mite, Varroa destructor Anderson and Trueman. Egypt. J. Pest. Control 25, 333–337 [Google Scholar]
- 102.Rangel J. and Ward L. (2018) Evaluation of the predatory mite Stratiolaelaps scimitus for the biological control of the honey bee ectoparasitic mite Varroa destructor. J. Apic. Res. 57, 425–432 10.1080/00218839.2018.1457864 [DOI] [Google Scholar]
- 103.Goras G., Tananaki C.H., Gounari S., Dimou M., Lazaridou E., Karazafiris E. et al. (2018) Hyperthermia -a non-chemical control strategy against Varroa. J. Hellenic. Vet. Med. Soc. 66, 249 10.12681/jhvms.15869 [DOI] [Google Scholar]
- 104.Le Conte Y., Arnold G. and Desenfant P. (1990) Influence of brood temperature and hygrometry variations on the development of the honey bee ectoparasite Varroa jacobsoni (Mesostigmata: Varroidae). Environ. Entomol. 19, 1780–1785 10.1093/ee/19.6.1780 [DOI] [Google Scholar]
- 105.Kablau A., Berg S., Härtel S. and Scheiner R. (2019) Hyperthermia treatment can kill immature and adult Varroa destructor mites without reducing drone fertility. Apidologie 10.1007/s13592-019-00715-7 [DOI] [Google Scholar]
- 106.Tihelka E. (2016) History of Varroa heat treatment in Central Europe (1981–2013). Bee World 93, 4–6 10.1080/0005772X.2016.1204826 [DOI] [Google Scholar]
- 107.Huang Z.Y., Bian G., Xi Z. and Xie X. (2019) Genes important for survival or reproduction in Varroa destructor identified by RNAi: Varroa destructor RNAi. Insect Sci. 26, 68–75 10.1111/1744-7917.12513 [DOI] [PubMed] [Google Scholar]
- 108.Leonard S.P., Powell J.E., Perutka J., Geng P., Heckmann L.C., Horak R.D. et al. (2020) Engineered symbionts activate honey bee immunity and limit pathogens. Science 367, 573–576 10.1126/science.aax9039 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Garbian Y., Maori E., Kalev H., Shafir S. and Sela I. (2012) Bidirectional transfer of RNAi between honey bee and Varroa destructor: varroa gene silencing reduces Varroa population. PLoS Pathog. 8, e1003035 10.1371/journal.ppat.1003035 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Mondet F., Maisonnasse A., Kretzschmar A., Alaux C., Vallon J., Basso B. et al. (2016) Varroa son impact, les méthodes d’évaluation de l'infestation et les moyens de lutte. Innov. Agron. 53, 63–80 [Google Scholar]
- 111.Dietemann V., Nazzi F., Martin S.J., Anderson D.L., Locke B., Delaplane K.S. et al. (2013) Standard methods for Varroa research. J. Apic. Res. 52, 1–54 10.3896/IBRA.1.52.1.09 [DOI] [Google Scholar]
- 112.Kretzschmar A., Durand E., Maisonnasse A., Vallon J. and Conte Y.L. (2015) A new stratified sampling procedure which decreases error estimation of Varroa mite number on sticky boards. J. Econ. Entomol. 108, 1435–1443 10.1093/jee/tov077 [DOI] [PubMed] [Google Scholar]
- 113.Szczurek A., Maciejewska M., Bąk B., Wilk J., Wilde J. and Siuda M. (2019) Gas sensor array and classifiers as a means of varroosis detection. Sensors 20, 117 10.3390/s20010117 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Bjerge K., Frigaard C.E., Mikkelsen P.H., Nielsen T.H., Misbih M. and Kryger P. (2019) A computer vision system to monitor the infestation level of Varroa destructor in a honeybee colony. Comput. Electron. Agr. 164, 104898 10.1016/j.compag.2019.104898 [DOI] [Google Scholar]
- 115.Locke B. (2016) Natural Varroa mite-surviving Apis mellifera honeybee populations. Apidologie 47, 467–482 10.1007/s13592-015-0412-8 [DOI] [Google Scholar]
- 116.Rinderer T.E., Harris J.W., Hunt G.J. and de Guzman L.I. (2010) Breeding for resistance to Varroa destructor in North America. Apidologie 41, 409–424 10.1051/apido/2010015 [DOI] [Google Scholar]
- 117.Büchler R., Berg S. and Le Conte Y. (2010) Breeding for resistance to Varroa destructor in Europe. Apidologie 41, 393–408 10.1051/apido/2010011 [DOI] [Google Scholar]
- 118.Danka R.G., Harris J.W. and Dodds G.E. (2016) Selection of VSH-derived “Pol-line” honey bees and evaluation of their Varroa-resistance characteristics. Apidologie 47, 483–490 10.1007/s13592-015-0413-7 [DOI] [Google Scholar]
- 119.Kefuss J., Vanpoucke J., Bolt M. and Kefuss C. (2015) Selection for resistance to Varroa destructor under commercial beekeeping conditions. J. Apic. Res. 54, 563–576 10.1080/00218839.2016.1160709 [DOI] [Google Scholar]
- 120.Schneider D.S. and Ayres J.S. (2008) Two ways to survive infection: what resistance and tolerance can teach us about treating infectious diseases. Nat. Rev. Immunol. 8, 889–895 10.1038/nri2432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mondet F., Beaurepaire A., McAfee A., Locke B., Alaux C., Blanchard S. et al. (2020) Honey bee survival mechanisms against the parasite Varroa destructor: a systematic review of phenotypic and genomic research efforts. Int. J. Parasitol. 10.1016/j.ijpara.2020.03.005 [DOI] [PubMed] [Google Scholar]
- 122.Thaduri S., Stephan J.G., de Miranda J.R. and Locke B. (2019) Disentangling host-parasite-pathogen interactions in a Varroa-resistant honeybee population reveals virus tolerance as an independent, naturally adapted survival mechanism. Sci. Rep. 9, 6221 10.1038/s41598-019-42741-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Cheruiyot S.K., Lattorff H.M.G., Kahuthia-Gathu R., Mbugi J.P. and Muli E. (2018) Varroa-specific hygienic behavior of Apis mellifera scutellata in Kenya. Apidologie 49, 439–449 10.1007/s13592-018-0570-6 [DOI] [Google Scholar]
- 124.Kirrane M.J., de Guzman L.I., Whelan P.M., Frake A.M. and Rinderer T.E. (2018) Evaluations of the removal of Varroa destructor in Russian honey bee colonies that display different levels of Varroa sensitive hygienic activities. J. Insect. Behav. 31, 283–297 10.1007/s10905-018-9672-2 [DOI] [Google Scholar]
- 125.Panziera D., van Langevelde F. and Blacquière T. (2017) Varroa sensitive hygiene contributes to naturally selected Varroa resistance in honey bees. J. Apic. Res. 56, 635–642 10.1080/00218839.2017.1351860 [DOI] [Google Scholar]
- 126.Bahreini R. and Currie R.W. (2015) The influence of Nosema (Microspora: Nosematidae) infection on honey bee (Hymenoptera: Apidae) defense against Varroa destructor (Mesostigmata: Varroidae). J. Invertebr. Pathol. 132, 57–65 10.1016/j.jip.2015.07.019 [DOI] [PubMed] [Google Scholar]
- 127.Oddie M.A.Y., Neumann P. and Dahle B. (2019) Cell size and Varroa destructor mite infestations in susceptible and naturally-surviving honeybee (Apis mellifera) colonies. Apidologie 50, 1–10 10.1007/s13592-018-0610-2 [DOI] [Google Scholar]
- 128.Martin S.J., Hawkins G.P., Brettell L.E., Reece N., Correia-Oliveira M.E. and Allsopp M.H. (2019) Varroa destructor reproduction and cell re-capping in mite-resistant Apis mellifera populations. Apidologie 10.1007/s13592-019-00721-9 [DOI] [Google Scholar]
- 129.Ratti V., Kevan P.G. and Eberl H.J (2016) A discrete-continuous modeling framework to study the role of swarming in a honeybee-Varroa destrutor-virus system In Mathematical and Computational Approaches in Advancing Modern Science and Engineering (Bélair J., Frigaard I.A., Kunze H., Makarov R., Melnik R. and Spiteri R.J. eds.), pp. 299–308, Springer International Publishing, Cham [Google Scholar]
- 130.Page P., Lin Z., Buawangpong N., Zheng H., Hu F., Neumann P. et al. (2016) Social apoptosis in honey bee superorganisms. Sci. Rep. 6, 27210 10.1038/srep27210 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Beaurepaire A.L., Sann C., Arredondo D., Mondet F. and Le Conte Y. (2019) Behavioral genetics of the interactions between Apis mellifera and Varroa destructor. Insects 10, 299 10.3390/insects10090299 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Beaurepaire A.L., Moro A., Mondet F., Le Conte Y., Neumann P. and Locke B. (2019) Population genetics of ectoparasitic mites suggest arms race with honeybee hosts. Sci. Rep. 9, 11355 10.1038/s41598-019-47801-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Boutin S., Alburaki M., Mercier P.-L., Giovenazzo P. and Derome N. (2015) Differential gene expression between hygienic and non-hygienic honeybee (Apis mellifera L.) hives. BMC Genom 16, 500 10.1186/s12864-015-1714-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Gempe T., Stach S., Bienefeld K., Otte M. and Beye M. (2016) Behavioral and molecular studies of quantitative differences in hygienic behavior in honeybees. BMC Res. Notes 9, 474 10.1186/s13104-016-2269-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Mondet F., Alaux C., Severac D., Rohmer M., Mercer A.R. and Le Conte Y. (2015) Antennae hold a key to Varroa-sensitive hygiene behaviour in honey bees. Sci. Rep. 5, 10454 10.1038/srep10454 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Hu H., Bienefeld K., Wegener J., Zautke F., Hao Y., Feng M. et al. (2016) Proteome analysis of the hemolymph, mushroom body, and antenna provides novel insight into honeybee resistance against Varroa infestation. J. Proteome Res. 15, 2841–2854 10.1021/acs.jproteome.6b00423 [DOI] [PubMed] [Google Scholar]
- 137.Conlon B.H., Frey E., Rosenkranz P., Locke B., Moritz R.F.A. and Routtu J. (2018) The role of epistatic interactions underpinning resistance to parasitic Varroa mites in haploid honey bee (Apis mellifera) drones. J. Evol. Biol. 31, 801–809 10.1111/jeb.13271 [DOI] [PubMed] [Google Scholar]
- 138.Kirrane M.J., de Guzman L.I., Holloway B., Frake A.M., Rinderer T.E. and Whelan P.M. (2015) Phenotypic and genetic analyses of the Varroa sensitive hygienic trait in Russian honey bee (Hymenoptera: Apidae) colonies. PLoS One 10, e0116672 10.1371/journal.pone.0116672 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Scannapieco A.C., Mannino M.C., Soto G., Palacio M.A., Cladera J.L. and Lanzavecchia S.B. (2017) Expression analysis of genes putatively associated with hygienic behavior in selected stocks of Apis mellifera L. from Argentina. Insect. Soc. 64, 485–494 10.1007/s00040-017-0567-6 [DOI] [Google Scholar]
- 140.Spötter A., Gupta P., Mayer M., Reinsch N. and Bienefeld K. (2016) Genome-wide association study of a Varroa-specific defense behavior in honeybees (Apis mellifera). J. Hered. 107, 220–227 10.1093/jhered/esw005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Wagoner K., Spivak M., Hefetz A., Reams T. and Rueppell O. (2019) Stock-specific chemical brood signals are induced by Varroa and deformed wing virus, and elicit hygienic response in the honey bee. Sci. Rep. 9, 8753 10.1038/s41598-019-45008-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Nazzi F., Della Vedova G. and D'Agaro M. (2004) A semiochemical from brood cells infested by Varroa destructor triggers hygienic behaviour in Apis mellifera. Apidologie 35, 65–70 10.1051/apido:2003065 [DOI] [Google Scholar]
- 143.Mondet F., Kim S.H., de Miranda J.R., Beslay D., Le Conte Y. and Mercer A.R. (2016) Specific cues associated with honey bee social defence against Varroa destructor infested brood. Sci. Rep. 6, 25444 10.1038/srep25444 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.McAfee A., Chapman A., Iovinella I., Gallagher-Kurtzke Y., Collins T.F., Higo H. et al. (2018) A death pheromone, oleic acid, triggers hygienic behavior in honey bees (Apis mellifera L.). Sci. Rep. 8, 5719 10.1038/s41598-018-24054-2 [DOI] [PMC free article] [PubMed] [Google Scholar]