Abstract
Intravenous leiomyomatosis is a histologically benign smooth muscle tumor that arises either by direct extension of a uterine leiomyoma into the adjacent veins or by vascular intimal smooth muscle proliferation. Herein, we report the case of a 60-year-old female who was noted to have suspected cardiac mass on elective echocardiography done electively for abnormal electrocardiographic findings. Computed tomography and cardiac magnetic resonance indicated the presence of an intravenous leiomyoma originating from the uterus and extending to the inferior vena cava and right atrium. The patient was managed with a single-stage surgery involving cardiopulmonary bypass and excision of the right atrial mass, excision of the inferior vena cava tumor thrombus, and total abdominal hysterectomy and bilateral salpingo-oophorectomy.
Keywords: Intravenous leiomyomatosis, Uterine leiomyoma, IVC tumor thrombus, Right atrial mass
Introduction
Intravenous leiomyomatosis (IVL) is a rare benign smooth muscle tumor with a propensity to involve the pelvic veins and extend into the inferior vena cava (IVC) and the right-sided cardiac chambers. Intracardiac involvement can mimic a thrombus which can result in a delay in diagnosis. Herein, we describe a case of IVL with cardiac extension in a 60-year-old female patient, successfully removed with surgery. Written informed consent was obtained from the patient for publication of this case report, including accompanying images.
Case report
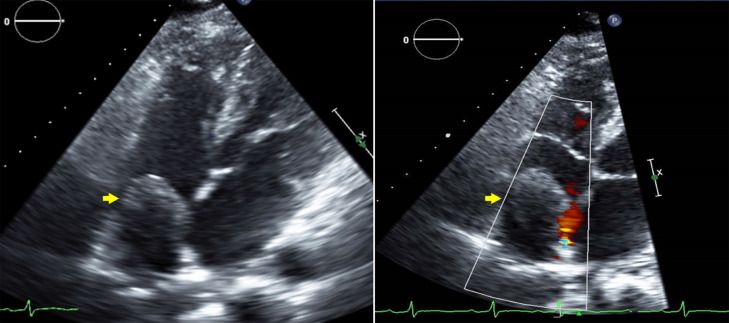
A 60-year-old female with history hypertension and diabetes was referred to the emergency department (ED) for evaluation of a suspected cardiac mass noted on elective echocardiography (ECHO) done for abnormal electrocardiographic findings, which was noted incidentally on wellness exam about 2-months ago. The ECHO (Fig. 1) showed a mobile echogenic mass in the right atrium and partially extending into the right ventricle, concerning for a thrombus or mass. In the emergency department, the patient was only complaining of mild lower extremity swelling and pain. She denied having chest pain, palpitations, shortness of breath, dyspnea on exertion, or abdominal pain. The physical examination was also within normal limits. Initial lab workup was normal, including white cell count, hemoglobin, and renal profile.
Fig.1.
Echocardiogram showed a mobile echogenic mass with perilesional vascularity (arrows) in the right atrium, partially extending through the tricuspid valve into the right ventricle.
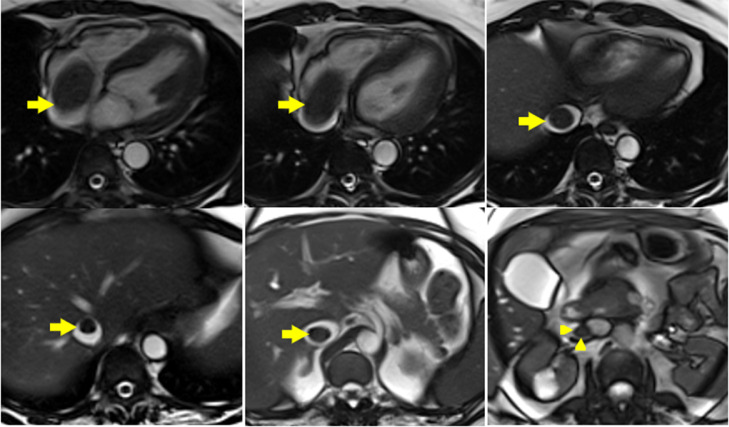
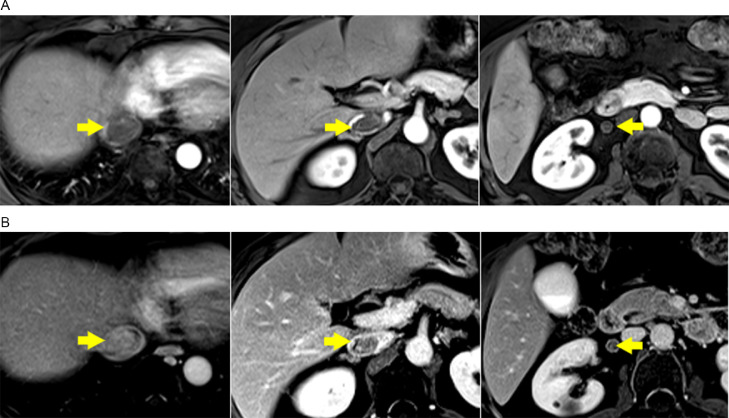
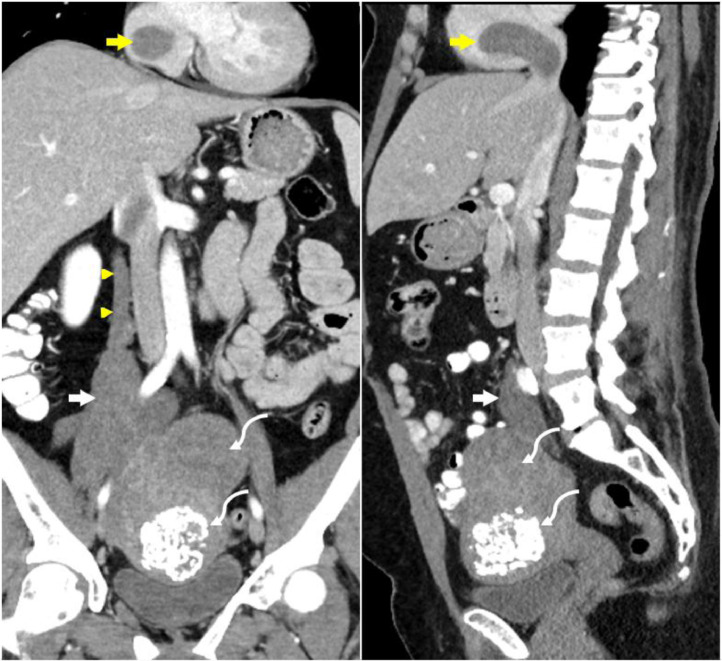
The patient was admitted and underwent cardiac magnetic resonance imaging which showed an enhancing right atrial mass measuring 4.5 × 3.2 cm that extended down into the IVC and the right ovarian vein (Figs. 2 and 3). To evaluate the full extent of the disease a computed tomography (CT) scan of the abdomen and pelvis was obtained, which showed a lobulated right adnexal mass measuring 9.1 cm and inseparable from the uterus. Filling defect and expansion of the right ovarian vein was present, which extended superiorly into the IVC and the right atrium. In addition, multiple calcified uterine leiomyomas were also present Figs. 4A and 4B.
Fig. 2.
Sequential axial steady-state free precision (SSFP) images from the level of the right atrium/ventricle to the kidneys show a large mass in the right atrium, extending into the inferior vena cava (arrows) and into the right ovarian vein (arrowheads).
Fig. 3 (.
A and B) Early (A) and delayed phase (B) postcontrast axial MR images show no early arterial enhancement but heterogeneous delayed enhancement of the mass.
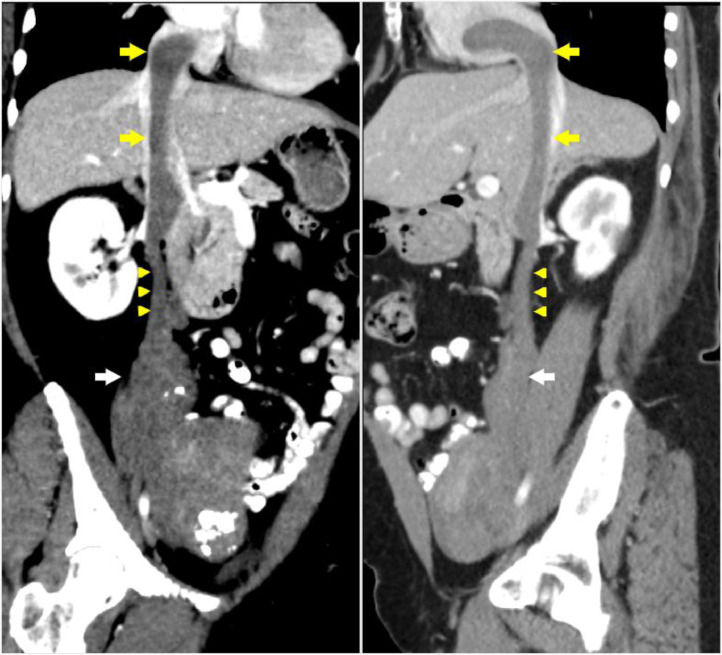
Fig. 4(A).
Coronal and sagittal contrast-enhanced CT of the abdomen and pelvis shows multiple uterine leiomyomas (curved white arrows) and a right adnexal mass (white arrow) that is inseparable from the uterus and contiguous with the expanded right ovarian vein (arrowheads). A right atrial hypodense mass is partially visualized (yellow arrow). (B) Coronal and sagittal curved multiplanar reformation (MPR) images of the abdomen and pelvis show the right adnexal mass (white arrow) extending and expanding the right ovarian vein (arrowheads), inferior vena cava, and the right atrium (yellow arrows). (Color version available online)
Fig. (4B).
Coronal and sagittal curved multiplanar reformation (MPR) images of the abdomen and pelvis show the right adnexal mass (white arrow) extending and expanding the right ovarian vein (arrowheads), inferior vena cava, and the right atrium (yellow arrows).
The patient underwent a single-stage surgical excision of the lesion by relevant surgical teams, involving cardiopulmonary bypass and excision of right atrial and right ventricular mass by cardiothoracic surgery, total abdominal hysterectomy with bilateral salpingo-oophorectomy by obstetrics and gynecology, and excision of IVC thrombus by vascular surgery. Intraoperatively the right renal vein was injured while excising the thrombus requiring urology consultation, who after assessment decided to proceed with right-sided nephrectomy. Histopathology of the uterus, ovaries, and the fallopian tubes showed multiple uterine leiomyomas with no leiomyomatous involvement of the ovaries or the fallopian tubes. However, smooth muscle proliferation was present within the right ovarian vein, consistent with intravenous leiomyomatosis. This smooth muscle proliferation extended into the inferior vena cava, right atrium, and right ventricle. The right renal vein was not involved by leiomyomata. The immunostains were positive for Desmin, which is positive in smooth muscle neoplastic cells, and CD31 which highlights vascular endothelium. These markers further supported the histopathologic diagnosis of intravenous leiomyomatosis.
The patient has had a long and protracted postoperative course, complicated by bowel ischemia and necrosis, requiring multiple exploratory laparotomies and bowel resections, and subsequent enterocutaneous fistula formations.
Discussion
IVL is a histologically benign smooth muscle tumor, developing within the pelvic and systemic veins. It is clinically aggressive extending into the gonadal veins, IVC, and the right-sided cardiac chambers [1]. Cardiac involvement is seen in approximately 10%-40% of cases [2,3]. The etiology of IVL is unclear; however, 2 theories have been proposed. One theory suggests that IVL originates de-novo from the vessel wall, whereas the other theory suggests intravenous extension from a preexisting uterine leiomyoma [4].
The age of presentation is highly variable, but most commonly affects women in the fifth decade. It can present with abnormal uterine bleeding, pelvic fullness, and pelvic pain due to concomitant presence of uterine leiomyoma. Patients with cardiac involvement may present with right-sided congestive symptoms, such as lower limb swelling, dyspnea, and congestive heart failure. Obstruction at tricuspid valve may result in syncope and even sudden death [5], [6], [7]. In a few cases, pulmonary embolism has also been reported [8,9]. In our case, the patient presented with mild bilateral lower extremity swelling, and the initial ECHO showed mild tricuspid regurgitation.
Echocardiography is frequently first to identify the cardiac mass with a nonspecific appearance that could mimic a thrombus or a primary cardiac tumor. CT and/or magnetic resoance imaging is then utilized to evaluate the morphology and delineate the anatomy and burden of the disease. Cross-sectional imaging is commonly characterized by a large, leiomyomatous uterus with variable intravenous growth into the myometrial veins and pelvic/systemic veins. The venous spread can occur via the uterine vein into the internal iliac veins, common iliac veins, and then the IVC and the right heart. Alternatively, as in our case, the tumor can involve the ovarian vein and extend directly into the IVC [10]. IVL involving the IVC can be differentiated from bland thrombus by a lack of enhancement of bland thrombus on postcontrast images. Moreover, a bland thrombus adheres to the wall of the vessel unlike IVL [11].
The definitive treatment for IVL is complete surgical excision [12,13]. The extent of surgery depends on the burden of the disease. Single-stage surgery is preferred, especially in low-risk patients, but is associated with longer operative time [14,15]. Our patient also underwent a single-stage cardiopulmonary bypass with resection of the right atrial mass and IVC along with total abdominal hysterectomy and bilateral salpingo-oophorectomy. The presence of estrogen and progesterone receptor is a good prognostic sign, adding the option of hormonal therapy in patients who are either unfit for surgery or those at high-risk for recurrence due to incomplete surgical resection. Antiestrogen therapy such as selective estrogen receptor modulators, gonadotrophin-releasing hormone agonists, and aromatase inhibitors, have been used in the management of patients with IVL with mixed results [13].
The true rate of recurrence of completely resected IVL is unknown; regrowth of tumor has been reported in as many as 30% of patients [16], [17], [18]. This may be due to incomplete resection or the presence of microscopic metastasis. The most common site of subsequent spread is to the lung [19,20]. Therefore, these patients require long-term follow-up imaging at 3- to 6-month intervals [18,21]. Valdes et al. suggested a baseline CT scan at 6-months after surgery with follow-up imaging every 2-5 years depending on the surgical excision and the initial tumor burden [1].
Footnotes
Disclosures: The authors have no conflicts of interest to disclose.
Contributor Information
Irfan Masood, Email: irmasood@utmb.edu.
Cihan Duran, Email: ciduran@utmb.edu.
Komal Malik, Email: komal.m2016@gmail.com.
Luba Frank, Email: lufrank@utmb.edu.
References
- 1.Valdés Devesa V., Conley C.R., Stone W.M., Collins J.M., Magrina J.F. Update on intravenous leiomyomatosis: report of five patients and literature review. Eur J Obstet Gynecol Reprod Biol. 2013;171(2):209–213. doi: 10.1016/j.ejogrb.2013.09.031. [DOI] [PubMed] [Google Scholar]
- 2.Kocica M.J, Vranes M.R., Kostic D., Kovacevic-Kostic N., Lackovic V., Bozic-Mihajlovic V. Intravenous leiomyomatosis with extension to the heart: rare or underestimated. J Thorac Cardiovasc Surg. 2005;130(6):1724–1726. doi: 10.1016/j.jtcvs.2005.08.021. [DOI] [PubMed] [Google Scholar]
- 3.Andrade L.A., Torresan R.Z, Sales Jr. J.F., Vicentini R, De Souza G.A. Intravenous leiomyomatosis of the uterus. A report of three cases. Pathol Oncol Res. 1998;4(1):44–47. doi: 10.1007/BF02904695. [DOI] [PubMed] [Google Scholar]
- 4.Konrad P., Mellblom L. Intravenous leiomyomatosis. Acta Obstet Gynecol Scand. 1989;68(4):371–376. doi: 10.3109/00016348909028675. [DOI] [PubMed] [Google Scholar]
- 5.Ling F.T., David T.E., Merchant N., Yu E., Butany J.W. Intracardiac extension of intravenous leiomyomatosis in a pregnant woman: A case report and review of the literature. Can J Cardiol. 2000;16(1):73–79. [PubMed] [Google Scholar]
- 6.Butler M.W., Sanders A. Obstructive shock in a 47 year old female with a deep venous thrombosis due to intravascular leiomyomatosis: a case report. Cases J. 2009;2:8159. doi: 10.4076/1757-1626-2-8159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee V.S., Thompson N.W., Cho K.J., Goldblum J.R. High-output cardiac failure: an unusual manifestation of intravenous leiomyomatosis. Surgery. 1993;113(4):466–470. [PubMed] [Google Scholar]
- 8.Andrade D., Vinck E.E., Torres L.N., Citarella D. Surgical removal of intravascular leiomyomas with intracardiac extension: A two-case report. J Card Surg. 2020;35(5):1094–1097. doi: 10.1111/jocs.14525. [DOI] [PubMed] [Google Scholar]
- 9.Ribeiro V., Almeida J., Madureira A.J., Lopez E., Machado L., Albuquerque R. Intracardiac leiomyomatosis complicated by pulmonary embolism: a multimodality imaging case of a rare entity. Can J Cardiol. 2013;29(12) doi: 10.1016/j.cjca.2013.09.008. 1743.e1-3. [DOI] [PubMed] [Google Scholar]
- 10.Lam P.M., Lo K.W., Yu M.Y., Wong W.S., Lau J.Y., Arifi A.A. Intravenous leiomyomatosis: two cases with different routes of tumor extension. J Vasc Surg. 2004;39(2):465–469. doi: 10.1016/j.jvs.2003.08.012. [DOI] [PubMed] [Google Scholar]
- 11.Xu Z.F., Yong F., Chen Y.Y., Pan A.Z. Uterine intravenous leiomyomatosis with cardiac extension: Imaging characteristics and literature review. World J Clin Oncol. 2013;4(1):25–28. doi: 10.5306/wjco.v4.i1.25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Buckley O., Madan R., Kwong R., Rybicki F.J., Hunsaker A. Cardiac masses, part 2: key imaging features for diagnosis and surgical planning. AJR Am J Roentgenol. 2011;197(5):W842–W851. doi: 10.2214/AJR.11.6903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Doyle M.P., Li A., Villanueva C.I., Peeceeyen S.C., Cooper M.G., Hanel K.C. Treatment of Intravenous Leiomyomatosis with Cardiac Extension following Incomplete Resection. Int J Vasc Med. 2015;2015 doi: 10.1155/2015/756141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rispoli P., Santovito D., Tallia C., Varetto G., Conforti M., Rinaldi M. A one-stage approach to the treatment of intravenous leiomyomatosis extending to the right heart. J Vasc Surg. 2010;52(1):212–215. doi: 10.1016/j.jvs.2010.02.018. [DOI] [PubMed] [Google Scholar]
- 15.Topcuoglu M.S., Yaliniz H., Poyrazoglu H., Tokcan A., Demir S.C., Bozkurt A. Intravenous leiomyomatosis extending into the right ventricle after subtotal hysterectomy. Ann Thorac Surg. 2004;78(1):330–332. doi: 10.1016/S0003-4975(03)01371-7. [DOI] [PubMed] [Google Scholar]
- 16.Clement P.B., Young R.H., Scully R.E. Intravenous leiomyomatosis of the uterus. A clinicopathological analysis of 16 cases with unusual histologic features. Am J Surg Pathol. 1988;12(12):932–945. [PubMed] [Google Scholar]
- 17.Du J., Zhao X., Guo D., Li H., Sun B. Intravenous leiomyomatosis of the uterus: a clinicopathologic study of 18 cases, with emphasis on early diagnosis and appropriate treatment strategies. Hum Pathol. 2011;42(9):1240–1246. doi: 10.1016/j.humpath.2010.10.015. [DOI] [PubMed] [Google Scholar]
- 18.Grella L., Arnold T.E., Kvilekval K.H., Giron F. Intravenous leiomyomatosis. J Vasc Surg. 1994;20(6):987–994. doi: 10.1016/0741-5214(94)90237-2. [DOI] [PubMed] [Google Scholar]
- 19.Lo K.W., Lau T.K. Intracardiac leiomyomatosis. Case report and literature review. Arch Gynecol Obstet. 2001;264(4):209–210. doi: 10.1007/s004040000115. [DOI] [PubMed] [Google Scholar]
- 20.Matsumoto K., Yamamoto T., Hisayoshi T., Asano G. Intravenous leiomyomatosis of the uterus with multiple pulmonary metastases associated with large bullae-like cyst formation. Pathol Int. 2001;51(5):396–401. doi: 10.1046/j.1440-1827.2001.01205.x. [DOI] [PubMed] [Google Scholar]
- 21.Evans A.T., Symmonds R.E., Gaffey T.A. Recurrent pelvic intravenous leiomyomatosis. Obstet Gynecol. 1981;57(2):260–264. [PubMed] [Google Scholar]