Introduction
Survival from infection and altered self requires an effective and tightly controlled immune response. Disorders of immunodeficiency or autoimmunity can be directly attributed to imbalances in the immune response [1–3]. It is now evident that changes in the metabolic status of cells and tissues have important and long-overlooked impacts on immunity [4, 5] and immune-related abnormalities [6, 7]. The reprogramming of immune cell metabolism is a regulatory event that governs the nature of the immune response in both health and disease. Studies of the metabolic signals that regulate host defense responses provide new insight into the determinants of immunity and the extent by which metabolic diseases, such as obesity and diabetes, are caused by immune dysfunctions.
Immunometabolic studies so far, focus primarily on glycolysis and oxidative phosphorylation, and have strongly affirmed the importance of these central energy supply pathways to support innate and adaptive immune cell activity and survival [4] [8, 9]. These findings opened avenues of investigation into additional metabolic networks that operate in the immune system, which have yet to be fully explored [4].
Peroxisomes are essential metabolic organelles present in virtually every eukaryotic cell and are important sites of distinct metabolic reactions essential for survival. Recent evidence demonstrated that peroxisomes are required for immune cell development and function [10], regulating key immune response pathways, such as the activation of nuclear factor kappa-light-chain-enhancer of activated B cells (NF-𝜅B) during bacterial infection [11] and mitochondrial antiviral signaling adaptor (MAVS)-mediated antiviral responses [12].
Here, we will summarize the last 10 years of discoveries that led to the recognition that peroxisomes are metabolic mediators of immunity. We will focus on findings that revealed a peroxisome requirement for immunity as well as evidence that defined the functional roles peroxisomes play in immune cell development and activity.
The peroxisome
Peroxisomes are ubiquitous organelles that regulate the synthesis and turnover of complex lipids. Peroxisomes are the only cellular compartment in eukaryotic cells where the β-oxidation of very-long chain fatty acids (VLCFAs), the synthesis of ether lipids, and the α-oxidation of branch-chained fatty acids takes place. Moreover, peroxisomes contribute to the detoxification of reactive anionic species and the metabolism of polyamines, amino acids, and carbohydrates [13, 14]. Delimited by a single membrane, peroxisomes arise either de novo from the endoplasmic reticulum (ER) [15] or by growth and fission of existing organelles [16]. Conserved Peroxin (Pex) genes encode proteins required for the formation and maintenance of the cellular peroxisome population [16–18].
Peroxisome metabolic functions are closely related with mitochondrial metabolism. Specifically, both organelles tightly cooperate to control fatty acids β-oxidation [19] and to maintain the oxidative homeostasis [20] in the cell. Until a few years ago, most studies pointed solely to the mitochondrion as a central hub of the immune and inflammatory response [21] against a variety of pathogens. Mitochondria act as a transmission site for different immune signaling events, such as the initiation of the MAVS-proteins–mediated interferon response [22] and the activation of the inflammasome [23]. Also, mitochondria contribute to the metabolic changes that affect immune cell behaviour [24, 25]. For instance, during the early or active phase of an immune response, the cellular metabolic shift from a catabolic to an anabolic state, such as the switch from fatty acids β-oxidation to fatty acid synthesis, is essential to drive the transformation of immune cells from metabolically quiescent (inactive) to a highly active metabolic state (activated) [24]. Although peroxisomes share many features with mitochondria, such as fatty acids β-oxidation, which is important for immune cell regulation (e.g., memory T-cells activation) [26], their unique role as pivotal regulators of cellular and systemic immune responses emerged only recently.
Peroxisomes regulate host–pathogen interactions
Peroxisomes were first described by a Swedish doctoral student, Johannes Rhodin, in 1954[27]. They were classified as organelles by the cytologist Christian de Duve in 1966 [28]. In 1978, de Duve and Lazarow realized that dysfunction in peroxisome activities affected VLCFA metabolism [29]. In 1989, Lazarow and Moser linked peroxisomal lipid metabolic defects to the insurgence of the cerebro-hepato-renal Zellweger syndrome that is now classified as severe form of peroxisome biogenesis disorder (PBD) [30], a genetic and metabolic condition caused by the deficiency or functional impairment of peroxisomes [31]. Since then, multiple studies of patients with PBD have illuminated the critical role of peroxisomes in human health and development, which revealed further connections between peroxisome dysfunction and other pathologies, such as Alzheimer’s and Parkinson’s diseases, aging, cancer, type 2 diabetes, and heart failure [32–36]. These findings suggested that peroxisomes contribute in different ways to the function, development and survival of different tissues. For instance, peroxisomes are increasingly recognized as producers of distal neurotrophic factors for the survival and function of central and peripheral neurons, responsible for the production of primary bile acids in liver hepatocytes and mediators of muscle function in myocytes [37]. Only in the past 10 years, peroxisomes have been described as regulators of the immune cell functions in response to viral and bacterial elicitors [10–12].
The first report that linked peroxisomes to the immune system was in 1974, by Gilchrist and colleagues, in a study which reported defects in differentiation and function of T cells in clinical cases of cerebro-hepato-renal Zellweger syndrome [38]. A few years later, in 1979, a report by Euguchi and colleagues described that peroxisomes of rat peritoneal macrophages were located proximal to phagosomes during phagocytosis, suggesting a role for the organelle in the phagocytic clearance of pathogens [39]. Despite these early observations, a role for peroxisomes in immunity was long overlooked.
In the past decade, multiple studies demonstrated that peroxisomes, like mitochondria, control immune pathways by producing bioactive metabolites important to drive immune signaling and also by recruiting signaling proteins to their membrane to promote immune pathways activation in response to a stimulus.
The first report was by Dixit and colleagues in 2010 where the authors showed that peroxisomes are essential to trigger interferon-mediated antiviral signaling. This study demonstrating that peroxisomes constitute an antiviral signaling platform and thus contribute to innate immunity was a real breakthrough in the peroxisomal field [12].
The main antiviral signaling pathways depend on the detection of viral proteins, lipids and nucleic acids by pattern recognition receptors (PRRs). Viral proteins are recognized by PRRs such as toll-like receptors (TLR)2/1, TLR2/6, and TLR4, while viral DNA, single-stranded RNA, and double-stranded RNA are recognized by TLR9, TLR7/8, and TLR3. Respectively [40, 41]. Cytosolic receptors such as RIG-I-like receptors (RLRs) are also involved in detecting viral nucleic acids [42]. Binding to viral RNA induces conformational changes in RLRs, which trigger their interaction with MAVS proteins. MAVS is a tail-anchored protein first described on the outer membrane of mitochondria [22]. Downstream signaling pathways activate transcription factors such as NF-𝜅B and interferon regulatory factors (IRFs), leading to production of proinflammatory cytokines and type I or III interferons. Dixit and colleagues reported that the MAVS localize in multiple cellular sites including mitochondria, a specific region of the ER membrane called mitochondrial-associated ER membrane, and peroxisomes [12, 43, 44] to mount an antiviral response. The localization of MAVS to peroxisomes and mitochondria drives different antiviral signaling programs, and peroxisome-associated MAVS seem to activate type III interferon response [12]. Moreover, the same group established that RLR-mediated type III interferon expression can be induced by various viruses, including reoviruses, Sendai viruses, and dengue viruses [45]. More recently, proteomic analysis of peroxisome-enriched fractions from Sendai virus–infected HepG2 cells identified 25 proteins that were previously linked to immune response signaling in virally infected cells compared to noninfected control cells [46], suggesting that peroxisomes are essential signaling platforms that regulate diverse antiviral responses.
Peroxisomes turn on/off pivotal cellular immune signaling
Multiple studies have further recognized the importance of peroxisomes in cellular innate immune signaling and inflammation. In a recent work, we defined mechanisms by which peroxisomes might control immune cell activities, such as phagocytosis and modulation of immune pathways in response to pathogens [11]. Using the genetic model system Drosophila melanogaster, we observed that peroxisomes control phagosome formation and maturation in macrophages and that their intervention is required for elimination of bacterial and fungal pathogens and host survival from the infection. We also demonstrated that the requirement for peroxisome metabolites in phagocytosis is conserved in the murine system. Phagocytosis is a complex process that involves a massive reorganization of the plasma membrane structure and composition. It is well known that phagocytosis is affected by lipid membrane composition and is dependent on the extracellular and intracellular lipid environment. Peroxisome lipid metabolism has an extensive impact on lipid membrane composition, and peroxisome dysfunction leads to an unbalanced pool of cellular lipids necessary to support changes in immune cell membranes, which in turn affect the phagocytic capacity of a cell [10, 47–49]. Membrane properties are indeed modified by changes in their fatty acid and cholesterol content [50]. Peroxisomes regulate intracellular and membrane fatty acid and cholesterol levels [51]. Of notice, the metabolism of polyunsaturated fatty acids (PUFAs) is partly dependent on peroxisomal β-oxidation, and PUFAs are direct modulators of phagocytosis in different phagocytic cells [52–56]. Using genetic and biochemical approaches, we demonstrated that peroxisomes regulate phagocytosis by providing fatty acids such as the PUFA docosahexaenoic acid (DHA) and reactive oxygen species (ROS) to promote the phagosome formation.
Changes in membrane lipid composition during an infection links the process of phagocytosis to the activation of other immune response strategies, such as the activation of inflammatory pathways. The incorporation of DHA into the cell membrane not only affects phagosome formation but, at the same time, alters the composition of lipid nanodomains that control the assembly or expulsion of a variety of transmembrane receptors. This effect impacts the ability of PRRs such as toll-like receptors, TLR2 and TLR4, to activate or inhibit, respectively, canonical proinflammatory signaling [57]. Therefore, the role of peroxisome-derived lipids can directly impact innate immune signaling that regulates differentiation, activation, and inhibition of multiple immune cells. Evidence from studies on the Drosophila melanogaster model system also unravelled the involvement of peroxisome metabolism in the regulation of innate immune signaling. Defects in immune signaling through the mitogen-activated protein kinases (MAPKs) cascade and NF-𝜅B were associated with peroxisome dysfunction. Additionally, subsequent evidence in Drosophila with dysfunctional peroxisomes in the intestinal epithelium showed heightened susceptibility to enteric bacterial infection and a pronounced intestinal dysbiosis due to an accumulation of cellular free fatty acids [58].
In a study carried out in a mouse model, Vijayan and colleagues probed the immunomodulatory properties of peroxisomes in macrophages [59]. In RAW 264.7 murine macrophage cell lines and in primary alveolar and peritoneal murine macrophages, the induction of peroxisome proliferation by treatments with 4-phenyl butyric acid, a noncanonical peroxisome proliferator, can reduce the expression of lipopolysaccharide (LPS)-induced proinflammatory proteins such as cyclooxygenase (COX-2), tumor necrosis factor alpha (TNF-α), and interleukins 6 (IL-6) and 12 (IL-12). Conversely, a macrophage cell line lacking functional peroxisomes, due to a mutation in Peroxin14 (Pex14), a gene that encodes for a peroxisomal membrane anchor protein required for peroxisome biogenesis, did not show this reduction in COX2 or any other inflammatory cytokines. The antiinflammatory effect was found to be dependent on peroxisomal β-oxidation activity because the deletions of key peroxisomal β-oxidation enzymes cause hyperexpression of COX2 and TNF-α proteins. The authors also suggested that the peroxisomal product necessary for this antiinflammatory effect in LPS-stimulated macrophages is DHA, leaving to speculation whether peroxisomes produce biolipids to initiate the resolution of inflammation. Of note, DHA has antiinflammatory properties on human primary monocytes and T-helper lymphocytes [60]. Interestingly, in this process, the activity of NF-𝜅B is not affected, suggesting that peroxisomes can regulate the immune cell activation with different strategies that are NF-𝜅B dependent or independent.
The immune regulatory properties of peroxisomes have also been associated to their role in the production of ether lipids that are exclusively produced by peroxisomes in mammals. Ether lipids are particularly abundant in white blood cells; in macrophages and neutrophils, they represent up to 46% of the total phospholipids [61]. Lodhi and colleagues reported that peroxisome-derived phosphatidylcholine and ether lipids are required for neutrophil survival in mice [62]. In another study, Facciotti and colleagues described the requirement of ether lipids for the education, differentiation, and maturation of invariant natural killer (iNKT) cells in the thymus, extending the importance of peroxisomes not only to the innate, but also to the adaptive immune cell differentiation processes. The development and maturation iNKT relies on the recognition of lipid self-antigens presented by the cell-surface molecule CD1d in the thymus [63]. The authors found that mice deficient in the peroxisomal enzyme glycerophosphate O-acyltransferase (GNPAT), essential for the synthesis of ether lipids, showed a significant alteration in the thymic maturation of iNKT cells and fewer iNKT cells in both the thymus and peripheral organs, which confirmed the role of ether-bonded lipids as iNKT cell antigens. Thus, peroxisome-derived lipids are nonredundant self-antigens required for the generation of a full iNKT cell repertoire [63] and essential for cells of the adaptive immune system.
Peroxisome–pathogen interactions
Peroxisomes can also be targeted by some bacteria and viruses to escape immune responses. West Nile and dengue virus (flaviviruses) infections were shown to trigger peroxisomal biogenesis inhibition [64]. The peroxisome loss is caused by the capsid protein-dependent sequestration and degradation of the peroxisomal biogenesis factor PEX19, which explains why the induction of type III interferon is impaired in cells infected by these viruses. The N-terminal protease of pestivirus localizes to peroxisomes, and this localization inactivates the transcription factor IRF3, one of the main regulators of interferon production [65]. Likewise, the interaction between the human immunodeficiency virus protein, negative regulatory factor, and the peroxisomal enzyme Acyl-CoA Thioesterase 8 (ACOT8), led to a down-regulation of the major histocompatibility complex I, limiting T-cell activation necessary to eliminate infected cells [66–68].
Another study reported by Boncompain and colleagues demonstrated that the bacterium Chlamydia trachomatis, an obligate intracellular pathogen responsible for millions of cases of sexually transmitted infections, relies on peroxisomes of the cells to support its metabolism. The study demonstrated that peroxisomes are imported into the Chlamydia-contained phagosome in infected cells, and, although the organelle is dispensable for bacterial replication, it seems to be essential for the production of exclusive metabolites, such as plasmalogens, for this bacterium [69]. These emerging cases of pathogens exploiting peroxisomes open new avenues of investigation of peroxisomes as potential therapeutic targets to manipulate host–pathogen interactions for the survival of the host.
Conclusions
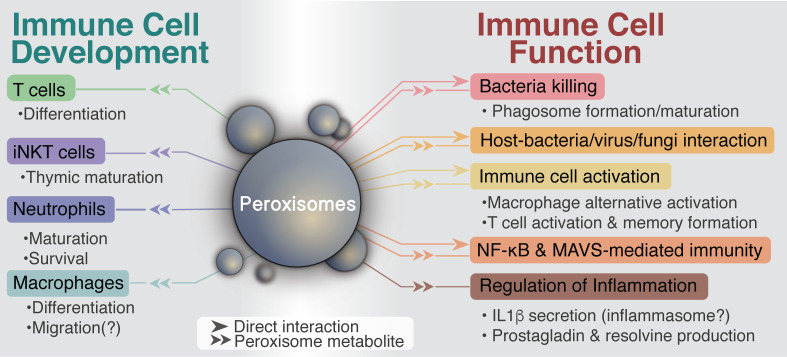
Immune disorders encompass a wide spectrum of human diseases with an ever-increasing impact on health. In 1970, the World Health Organization classified for the first time “primary immune deficiencies” as a small group of diseases characterized by recurrent or chronic infections, autoimmunity, allergy, inflammation, or cancer as a consequence of genetic alterations affecting the immune system [70]. To date, over 400 characterized immune deficiencies present a worldwide incidence of 1 in 10,000 [71]. There is therefore a pressing need to further characterize the underlying networks that govern immune cell functions in both health and disease to understand and correct immune disorders. The importance of peroxisomes in immunity and inflammation has become clear in the past 10 years (Fig 1). The role of peroxisomes in modulating cellular and extracellular fatty acids appears to explain their role as metabolic regulators of various immune functions. However, several peripheral metabolites and pathways are also required to shape the complexity of immune cell development, activation, and inhibition [4], and some of these rely, at least in part, on peroxisomal metabolism. For instance, polyamines, which are catabolized by peroxisomes [14], have been reported to be important for T-cell clonal expansion, macrophage alternative activation, and dendritic cell modulation [4].
Fig 1. Diagram summarizing the requirements for peroxisomes in modulating host–pathogen interactions.
Future studies to define how peroxisomes regulate discrete immune processes in innate and adaptive immune cells will be an important step towards understanding how cellular systems as a whole operate to generate and control effective immune responses. Also, these types of investigations will be critical to define how peroxisomal immunometabolic activities contribute to the development of immune disorders, the onset of metabolic diseases and chronic inflammation and elucidate the specific role peroxisomes play in host–pathogen interactions. We speculate that future investigations of peroxisomes in immunity will unveil alternative therapeutic targets to treat infections, inborn errors of immunity, and chronic inflammatory diseases.
Acknowledgments
The author acknowledge the Dalhousie Medical Research Foundation, the IWK Foundation, the Natural Sciences and Engineering Research Council of Canada (NSERC) and Research Nova Scotia.
The author thanks Drs Richard Rachubinski and Brendon Parsons for the critical reading of the manuscript.
Funding Statement
We want to thank the funding agencies the Natural Sciences and Engineering Research Council of Canada Discovery Grant # DGECR-2019-00106, The Dalhousie Medical Research Foundation and the Canadian Institute for Health Research operative grant #RN398695 - 426383. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Gupta S, Agrawal A, Agrawal S, Su H, Gollapudi S. A paradox of immunodeficiency and inflammation in human aging: lessons learned from apoptosis. Immun Ageing. 2006;3:5 10.1186/1742-4933-3-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Raje N, Dinakar C. Overview of Immunodeficiency Disorders. Immunol Allergy Clin North Am. 2015;35(4):599–623. 10.1016/j.iac.2015.07.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang L, Wang FS, Gershwin ME. Human autoimmune diseases: a comprehensive update. J Intern Med. 2015;278(4):369–95. 10.1111/joim.12395 . [DOI] [PubMed] [Google Scholar]
- 4.Puleston DJ, Villa M, Pearce EL. Ancillary Activity: Beyond Core Metabolism in Immune Cells. Cell Metab. 2017;26(1):131–41. 10.1016/j.cmet.2017.06.019 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.O'Neill LA, Pearce EJ. Immunometabolism governs dendritic cell and macrophage function. J Exp Med. 2016;213(1):15–23. 10.1084/jem.20151570 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Perl A. Review: Metabolic Control of Immune System Activation in Rheumatic Diseases. Arthritis Rheumatol. 2017;69(12):2259–70. 10.1002/art.40223 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Weyand CM, Zeisbrich M, Goronzy JJ. Metabolic signatures of T-cells and macrophages in rheumatoid arthritis. Curr Opin Immunol. 2017;46:112–20. 10.1016/j.coi.2017.04.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Thompson CB. Rethinking the regulation of cellular metabolism. Cold Spring Harb Symp Quant Biol. 2011;76:23–9. 10.1101/sqb.2012.76.010496 . [DOI] [PubMed] [Google Scholar]
- 9.Loftus RM, Finlay DK. Immunometabolism: Cellular Metabolism Turns Immune Regulator. J Biol Chem. 2016;291(1):1–10. 10.1074/jbc.R115.693903 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Di Cara F, Andreoletti P, Trompier D, Vejux A, Bulow MH, Sellin J, et al. Peroxisomes in Immune Response and Inflammation. Int J Mol Sci. 2019;20(16). 10.3390/ijms20163877 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Di Cara F, Sheshachalam A, Braverman NE, Rachubinski RA, Simmonds AJ. Peroxisome-Mediated Metabolism Is Required for Immune Response to Microbial Infection. Immunity. 2017;47(1):93–106 e7. 10.1016/j.immuni.2017.06.016 . [DOI] [PubMed] [Google Scholar]
- 12.Dixit E, Boulant S, Zhang Y, Lee AS, Odendall C, Shum B, et al. Peroxisomes are signaling platforms for antiviral innate immunity. Cell. 2010;141(4):668–81. 10.1016/j.cell.2010.04.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Schrader M, Fahimi HD. Peroxisomes and oxidative stress. Biochim Biophys Acta. 2006;1763(12):1755–66. 10.1016/j.bbamcr.2006.09.006 . [DOI] [PubMed] [Google Scholar]
- 14.Wanders RJ, Waterham HR. Biochemistry of mammalian peroxisomes revisited. Annu Rev Biochem. 2006;75:295–332. 10.1146/annurev.biochem.74.082803.133329 . [DOI] [PubMed] [Google Scholar]
- 15.Titorenko VI, Rachubinski RA. The endoplasmic reticulum plays an essential role in peroxisome biogenesis. Trends Biochem Sci. 1998;23(7):231–3. 10.1016/s0968-0004(98)01226-2 . [DOI] [PubMed] [Google Scholar]
- 16.Mast FD, Fagarasanu A, Knoblach B, Rachubinski RA. Peroxisome biogenesis: something old, something new, something borrowed. Physiology. 2010;25(6):347–56. Epub 2010/12/28. 10.1152/physiol.00025.2010 . [DOI] [PubMed] [Google Scholar]
- 17.Faust JE, Verma A, Peng C, McNew JA. An inventory of peroxisomal proteins and pathways in Drosophila melanogaster. Traffic. 2012;13(10):1378–92. 10.1111/j.1600-0854.2012.01393.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Smith JJ, Aitchison JD. Peroxisomes take shape. Nat Rev Mol Cell Biol. 2013;14(12):803–17. 10.1038/nrm3700 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shai N, Yifrach E, van Roermund CWT, Cohen N, Bibi C, L IJ, et al. Systematic mapping of contact sites reveals tethers and a function for the peroxisome-mitochondria contact. Nat Commun. 2018;9(1):1761 10.1038/s41467-018-03957-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fransen M, Lismont C, Walton P. The Peroxisome-Mitochondria Connection: How and Why? Int J Mol Sci. 2017;18(6). 10.3390/ijms18061126 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mohanty A, Tiwari-Pandey R, Pandey NR. Mitochondria: the indispensable players in innate immunity and guardians of the inflammatory response. J Cell Commun Signal. 2019;13(3):303–18. 10.1007/s12079-019-00507-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Seth RB, Sun L, Ea CK, Chen ZJ. Identification and characterization of MAVS, a mitochondrial antiviral signaling protein that activates NF-kappaB and IRF 3. Cell. 2005;122(5):669–82. 10.1016/j.cell.2005.08.012 . [DOI] [PubMed] [Google Scholar]
- 23.Martinon F. Inflammation initiated by stressed organelles. Joint Bone Spine. 2018;85(4):423–8. 10.1016/j.jbspin.2017.06.005 . [DOI] [PubMed] [Google Scholar]
- 24.Weinberg SE, Sena LA, Chandel NS. Mitochondria in the regulation of innate and adaptive immunity. Immunity. 2015;42(3):406–17. 10.1016/j.immuni.2015.02.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Sandhir R, Halder A, Sunkaria A. Mitochondria as a centrally positioned hub in the innate immune response. Biochim Biophys Acta Mol Basis Dis. 2017;1863(5):1090–7. 10.1016/j.bbadis.2016.10.020 . [DOI] [PubMed] [Google Scholar]
- 26.van der Windt GJ, O'Sullivan D, Everts B, Huang SC, Buck MD, Curtis JD, et al. CD8 memory T cells have a bioenergetic advantage that underlies their rapid recall ability. Proc Natl Acad Sci U S A. 2013;110(35):14336–41. 10.1073/pnas.1221740110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bernhard W, Rouiller C. Microbodies and the problem of mitochondrial regeneration in liver cells. J Biophys Biochem Cytol. 1956;2(4 Suppl):355–60. 10.1083/jcb.2.4.355 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.De Duve C, Baudhuin P. Peroxisomes (microbodies and related particles). Physiol Rev. 1966;46(2):323–57. 10.1152/physrev.1966.46.2.323 . [DOI] [PubMed] [Google Scholar]
- 29.Lazarow PB. Rat liver peroxisomes catalyze the beta oxidation of fatty acids. J Biol Chem. 1978;253(5):1522–8. . [PubMed] [Google Scholar]
- 30.Leighton F, Poole B, Beaufay H, Baudhuin P, Coffey JW, Fowler S, et al. The large-scale separation of peroxisomes, mitochondria, and lysosomes from the livers of rats injected with triton WR-1339. Improved isolation procedures, automated analysis, biochemical and morphological properties of fractions. J Cell Biol. 1968;37(2):482–513. 10.1083/jcb.37.2.482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Waterham HR, Ferdinandusse S, Wanders RJ. Human disorders of peroxisome metabolism and biogenesis. Biochim Biophys Acta. 2016;1863(5):922–33. 10.1016/j.bbamcr.2015.11.015 . [DOI] [PubMed] [Google Scholar]
- 32.Beach A, Burstein MT, Richard VR, Leonov A, Levy S, Titorenko VI. Integration of peroxisomes into an endomembrane system that governs cellular aging. Front Physiol. 2012;3:283 10.3389/fphys.2012.00283 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Colasante C, Chen J, Ahlemeyer B, Baumgart-Vogt E. Peroxisomes in cardiomyocytes and the peroxisome / peroxisome proliferator-activated receptor-loop. Thromb Haemost. 2015;113(3):452–63. 10.1160/TH14-06-0497 . [DOI] [PubMed] [Google Scholar]
- 34.Fransen M, Nordgren M, Wang B, Apanasets O, Van Veldhoven PP. Aging, age-related diseases and peroxisomes. Subcell Biochem. 2013;69:45–65. 10.1007/978-94-007-6889-5_3 . [DOI] [PubMed] [Google Scholar]
- 35.Trompier D, Vejux A, Zarrouk A, Gondcaille C, Geillon F, Nury T, et al. Brain peroxisomes. Biochimie. 2014;98:102–10. 10.1016/j.biochi.2013.09.009 . [DOI] [PubMed] [Google Scholar]
- 36.Merkling SH, Riahi H, Overheul GJ, Schenck A, van Rij RP. Peroxisome-associated Sgroppino links fat metabolism with survival after RNA virus infection in Drosophila. Sci Rep. 2019;9(1):2065 10.1038/s41598-019-38559-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Braverman NE, D'Agostino MD, Maclean GE. Peroxisome biogenesis disorders: Biological, clinical and pathophysiological perspectives. Dev Disabil Res Rev. 2013;17(3):187–96. 10.1002/ddrr.1113 . [DOI] [PubMed] [Google Scholar]
- 38.Gilchrist KW, Opitz JM, Gilbert EF, Tsang W, Miller P. Letter: Immunodeficiency in the cerebro-hepato-renal syndrome of Zellweger. Lancet. 1974;1(7849):164–5. . [PubMed] [Google Scholar]
- 39.Eguchi M, Sannes PL, Spicer SS. Peroxisomes of rat peritoneal macrophages during phagocytosis. Am J Pathol. 1979;95(2):281–94. [PMC free article] [PubMed] [Google Scholar]
- 40.Miyake K, Shibata T, Ohto U, Shimizu T, Saitoh SI, Fukui R, et al. Mechanisms controlling nucleic acid-sensing Toll-like receptors. Int Immunol. 2018;30(2):43–51. Epub 2018/02/17. 4857348 [pii] 10.1093/intimm/dxy016 . [DOI] [PubMed] [Google Scholar]
- 41.Lester SN, Li K. Toll-like receptors in antiviral innate immunity. J Mol Biol. 2014;426(6):1246–64. Epub 2013/12/10. S0022-2836(13)00735-3 [pii] 10.1016/j.jmb.2013.11.024 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Kell AM, Gale M Jr. RIG-I in RNA virus recognition. Virology. 2015;479–480:110–21. Epub 2015/03/10. S0042-6822(15)00063-X [pii] 10.1016/j.virol.2015.02.017 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Horner SM, Liu HM, Park HS, Briley J, Gale M Jr. Mitochondrial-associated endoplasmic reticulum membranes (MAM) form innate immune synapses and are targeted by hepatitis C virus. Proc Natl Acad Sci U S A. 2011;108(35):14590–5. Epub 2011/08/17. 1110133108 [pii] 10.1073/pnas.1110133108 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Vazquez C, Beachboard DC, Horner SM. Methods to Visualize MAVS Subcellular Localization. Methods Mol Biol. 2017;1656:131–42. Epub 2017/08/16. 10.1007/978-1-4939-7237-1_7 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Odendall C, Dixit E, Stavru F, Bierne H, Franz KM, Durbin AF, et al. Diverse intracellular pathogens activate type III interferon expression from peroxisomes. Nat Immunol. 2014;15(8):717–26. Epub 2014/06/24. ni.2915 [pii] 10.1038/ni.2915 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhou MT, Qin Y, Li M, Chen C, Chen X, Shu HB, et al. Quantitative Proteomics Reveals the Roles of Peroxisome-associated Proteins in Antiviral Innate Immune Responses. Mol Cell Proteomics. 2015;14(9):2535–49. Epub 2015/07/01. M115.048413 [pii] 10.1074/mcp.M115.048413 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Raas Q, Gondcaille C, Hamon Y, Leoni V, Caccia C, Menetrier F, et al. CRISPR/Cas9-mediated knockout of Abcd1 and Abcd2 genes in BV-2 cells: novel microglial models for X-linked Adrenoleukodystrophy. Biochim Biophys Acta Mol Cell Biol Lipids. 2019;1864(5):704–14. 10.1016/j.bbalip.2019.02.006 . [DOI] [PubMed] [Google Scholar]
- 48.Raas Q, Saih FE, Gondcaille C, Trompier D, Hamon Y, Leoni V, et al. A microglial cell model for acyl-CoA oxidase 1 deficiency. Biochim Biophys Acta Mol Cell Biol Lipids. 2018. 10.1016/j.bbalip.2018.10.005 . [DOI] [PubMed] [Google Scholar]
- 49.Levin R, Hammond GR, Balla T, De Camilli P, Fairn GD, Grinstein S. Multiphasic dynamics of phosphatidylinositol 4-phosphate during phagocytosis. Mol Biol Cell. 2017;28(1):128–40. 10.1091/mbc.E16-06-0451 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Williams MR, Cauvi DM, Rivera I, Hawisher D, De Maio A. Changes in macrophage function modulated by the lipid environment. Innate Immun. 2016;22(3):141–51. Epub 2016/03/10. 1753425916633886 [pii] 10.1177/1753425916633886 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chu BB, Liao YC, Qi W, Xie C, Du X, Wang J, et al. Cholesterol transport through lysosome-peroxisome membrane contacts. Cell. 2015;161(2):291–306. Epub 2015/04/11. 10.1016/j.cell.2015.02.019 . [DOI] [PubMed] [Google Scholar]
- 52.Hjorth E, Freund-Levi Y. Immunomodulation of microglia by docosahexaenoic acid and eicosapentaenoic acid. Curr Opin Clin Nutr Metab Care. 2012;15(2):134–43. Epub 2012/02/10. 10.1097/MCO.0b013e32835017cc . [DOI] [PubMed] [Google Scholar]
- 53.Adolph S, Fuhrmann H, Schumann J. Unsaturated fatty acids promote the phagocytosis of P. aeruginosa and R. equi by RAW264.7 macrophages. Curr Microbiol. 2012;65(6):649–55. Epub 2012/08/21. 10.1007/s00284-012-0207-3 . [DOI] [PubMed] [Google Scholar]
- 54.Gorjao R, Verlengia R, Lima TM, Soriano FG, Boaventura MF, Kanunfre CC, et al. Effect of docosahexaenoic acid-rich fish oil supplementation on human leukocyte function. Clin Nutr. 2006;25(6):923–38. Epub 2006/05/16. S0261-5614(06)00075-6 [pii] 10.1016/j.clnu.2006.03.004 . [DOI] [PubMed] [Google Scholar]
- 55.Pisani LF, Lecchi C, Invernizzi G, Sartorelli P, Savoini G, Ceciliani F. In vitro modulatory effect of omega-3 polyunsaturated fatty acid (EPA and DHA) on phagocytosis and ROS production of goat neutrophils. Vet Immunol Immunopathol. 2009;131(1–2):79–85. Epub 2009/04/28. S0165-2427(09)00127-5 [pii] 10.1016/j.vetimm.2009.03.018 . [DOI] [PubMed] [Google Scholar]
- 56.Lecchi C, Invernizzi G, Agazzi A, Ferroni M, Pisani LF, Savoini G, et al. In vitro modulation of caprine monocyte immune functions by omega-3 polyunsaturated fatty acids. Vet J. 2011;189(3):353–5. Epub 2010/10/05. S1090-0233(10)00292-3 [pii] 10.1016/j.tvjl.2010.09.001 . [DOI] [PubMed] [Google Scholar]
- 57.Hwang DH, Kim JA, Lee JY. Mechanisms for the activation of Toll-like receptor 2/4 by saturated fatty acids and inhibition by docosahexaenoic acid. Eur J Pharmacol. 2016;785:24–35. Epub 2016/04/18. S0014-2999(16)30237-0 [pii] 10.1016/j.ejphar.2016.04.024 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Di Cara F BlM, Simmonds AJ and Rachubinski R. Peroxisomes integrate metabolic and stress signaling to maintain enteric homeostasis and promote immune response from the gut. MBoC. 2018;29(22):2766–83. Epub 2018 Sep 6. 10.1091/mbc.E18-07-0434 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Vijayan V, Srinu T, Karnati S, Garikapati V, Linke M, Kamalyan L, et al. A New Immunomodulatory Role for Peroxisomes in Macrophages Activated by the TLR4 Ligand Lipopolysaccharide. J Immunol. 2017;198(6):2414–25. 10.4049/jimmunol.1601596 . [DOI] [PubMed] [Google Scholar]
- 60.Jaudszus A, Gruen M, Watzl B, Ness C, Roth A, Lochner A, et al. Evaluation of suppressive and pro-resolving effects of EPA and DHA in human primary monocytes and T-helper cells. J Lipid Res. 2013;54(4):923–35. 10.1194/jlr.P031260 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Brautigam C, Engelmann B, Reiss D, Reinhardt U, Thiery J, Richter WO, et al. Plasmalogen phospholipids in plasma lipoproteins of normolipidemic donors and patients with hypercholesterolemia treated by LDL apheresis. Atherosclerosis. 1996;119(1):77–88. 10.1016/0021-9150(95)05632-7 . [DOI] [PubMed] [Google Scholar]
- 62.Immunological asects of Rheumatology. US-Canada_Europe: Elsevier; 1981. [Google Scholar]
- 63.Facciotti F, Ramanjaneyulu GS, Lepore M, Sansano S, Cavallari M, Kistowska M, et al. Peroxisome-derived lipids are self antigens that stimulate invariant natural killer T cells in the thymus. Nat Immunol. 2012;13(5):474–80. 10.1038/ni.2245 . [DOI] [PubMed] [Google Scholar]
- 64.You J, Hou S, Malik-Soni N, Xu Z, Kumar A, Rachubinski RA, et al. Flavivirus Infection Impairs Peroxisome Biogenesis and Early Antiviral Signaling. J Virol. 2015;89(24):12349–61. Epub 2015/10/02. JVI.01365-15 [pii] 10.1128/JVI.01365-15 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Jefferson M, Whelband M, Mohorianu I, Powell PP. The pestivirus N terminal protease N(pro) redistributes to mitochondria and peroxisomes suggesting new sites for regulation of IRF3 by N(pro.). PLoS ONE. 2014;9(2):e88838 Epub 2014/02/20. 10.1371/journal.pone.0088838 PONE-D-13-25410 [pii]. . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Cohen GB, Rangan VS, Chen BK, Smith S, Baltimore D. The human thioesterase II protein binds to a site on HIV-1 Nef critical for CD4 down-regulation. J Biol Chem. 2000;275(30):23097–105. Epub 2000/05/16. 10.1074/jbc.M000536200 . [DOI] [PubMed] [Google Scholar]
- 67.Liu LX, Margottin F, Le Gall S, Schwartz O, Selig L, Benarous R, et al. Binding of HIV-1 Nef to a novel thioesterase enzyme correlates with Nef-mediated CD4 down-regulation. J Biol Chem. 1997;272(21):13779–85. Epub 1997/05/23. 10.1074/jbc.272.21.13779 . [DOI] [PubMed] [Google Scholar]
- 68.Watanabe H, Shiratori T, Shoji H, Miyatake S, Okazaki Y, Ikuta K, et al. A novel acyl-CoA thioesterase enhances its enzymatic activity by direct binding with HIV Nef. Biochem Biophys Res Commun. 1997;238(1):234–9. Epub 1997/09/23. S0006-291X(97)97217-X [pii] 10.1006/bbrc.1997.7217 . [DOI] [PubMed] [Google Scholar]
- 69.Boncompain G, Muller C, Meas-Yedid V, Schmitt-Kopplin P, Lazarow PB, Subtil A. The intracellular bacteria Chlamydia hijack peroxisomes and utilize their enzymatic capacity to produce bacteria-specific phospholipids. PLoS ONE. 2014;9(1):e86196 10.1371/journal.pone.0086196 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Picard C, Bobby Gaspar H, Al-Herz W, Bousfiha A, Casanova JL, Chatila T, et al. International Union of Immunological Societies: 2017 Primary Immunodeficiency Diseases Committee Report on Inborn Errors of Immunity. J Clin Immunol. 2018;38(1):96–128. 10.1007/s10875-017-0464-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.van Zelm MC, Condino-Neto A, Barbouche MR. Editorial: Primary Immunodeficiencies Worldwide. Front Immunol. 2019;10:3148 10.3389/fimmu.2019.03148 [DOI] [PMC free article] [PubMed] [Google Scholar]