Abstract
The ongoing pandemic has stimulated study of the Renin Angiotensin Aldosterone System (RAAS), and how it can be manipulated to treat COVID-19. Studies are examining whether drugs that act on the RAAS system might be useful to treat COVID-19. COVID-19 and the RAAS are closely linked both in infection and in possible post-infection inflammatory cascades. We detail the Physiology and Pharmacology of the RAAS including the effects of aldosterone and atrial natriuretic peptide. It is appropriate that the theoretical benefits of modulation of the RAAS should be considered based on available knowledge of the complexity of the system. In this short review we have tried to explain the actions of the angiotensin family of peptides and produce a relatively simple model and diagrammatic summary of the RAAS and the possible sites of intervention.
Keywords: Angiotensin, COVID-19, RAAS
1. Introduction
The ongoing pandemic has stimulated study of the Renin Angiotensin Aldosterone System (RAAS), and how it can be manipulated to treat COVID-19. Researchers are examining whether drugs that act on the RAAS system might be useful to treat COVID-19. Possible benefits against COVID-19 are as yet purely theoretical, but pharmacological intervention may possibly be useful to (a) reduce binding of COVID-19 to cells, and/or (b) to counteract the RAAS pro-inflammatory cascade.
Severe Acute Respiratory Syndrome (SARS) emerged in 2002 and caused hundreds of deaths. The corona virus responsible (SARS-CoV-1) was shown to bind to cells via a critical receptor that was found to be the enzyme angiotensin converting enzyme type 2 (ACE2) (Kuba et al., 2005). SARS-CoV-2 (CoV-2) has recently emerged resulting in the current pandemic, and phylogenetic analysis indicates a bat origin of CoV-2, but with possible intermediate hosts (Wan et al., 2020). Like SARS-CoV-1, CoV-2 virus binds to the ACE2 enzyme through its trimeric spike glycoprotein and uses ACE2 to enter cells (Walls et al., 2020) and can target ACE2 in various cell types. ACE2 binds to CoV-2 with 15 nM affinity, about 10–20-fold higher affinity than to SARS-CoV-1 (Wrapp et al., 2020), and this may explain its greater virulence. ACE2 is a monocarboxypeptidase present on the surface of lung alveolar epithelial cells and enterocytes of the small intestine, endothelial cells and arterial smooth muscle cells (Hamming et al., 2004), and in a variety of tissues, including the heart and kidney (Tipnis et al., 2000). Once bound to the ACE2 enzyme, the virus infects the cell. The major targets of COVID-19 can to a large extent be explained by the distribution of ACE2, with major actions on the lungs, heart, GIT, kidney and vasculature. Infection usually starts in the respiratory tract, causing mild symptoms in 81%, severe in 14% and critical in 5%, of patients (Wu & McGoogan, 2020), but the risk of morbidity and mortality is increased by underlying health conditions, age (2.3% fatality overall, but 14.8% of those 80 years and over: Wu & McGoogan, 2020) and male sex (males represent 70% of COVID-19 deaths in Italy: Remuzzi and Remuzzi, 2020), among other factors. Lung thrombosis is being commonly found in patients with COVID-19 and disseminated intravascular coagulation is linked to high mortality (Tang et al., 2020). It has been proposed that ACE1 is linked to thrombosis in COVID-19 (Henry et al., 2020).
Gastrointestinal luminal ACE2 is a secondary site for CoV-2 infection and this may lead to damage to gut barrier function particularly in patients with diabetes mellitus and obesity, which themselves may affect the barrier function, resulting in systemic bacterial infection (Gheblawi et al., 2020).
Meta-analysis of six studies of patients with COVID-19 showed that 9.7% had diabetes, and the incidence of diabetes was about twofold higher in ICU/severe than in non-ICU/ non-severe patients (Li et al., 2020). There is a high frequency of obesity among patients admitted in intensive care for CoV-2, and disease severity increased with BMI (Simonnet et al. 2020). In rats a high fat diet induced obesity, comorbidities, and cardiac collagen overexpression, with an increase in angiotensin II levels in both adipose tissue and plasma, and increased AT1-receptor expression in cardiac tissue (da Silva-Bertani et al., 2020). The excess adipose tissue may be the source of the increased angiotensin II, with detrimental effects.
2. Reducing infection by CoV-2
Since CoV-2 enters cells via the ACE2 enzyme, it might seem that reducing the number of ACE2 molecules available for the virus would be a useful strategy for reducing infection. However, blockade of ACE2 may result in diminished binding of CoV-2 and diminish its entry into target cells, but at the cost of reducing levels of the enzyme. Since ACE2 produces mediators that are anti-inflammatory (see below), reducing the number of enzyme molecules might be detrimental. However, recombinant human ACE2 has the advantage of increasing ACE2 activity in the plasma (Arendse et al., 2019) while providing a decoy target for the virus, diverting the virus from target cells. Human recombinant soluble ACE2 can inhibit CoV-2 infection of engineered human blood vessel and kidney organoids and reduce viral load 1000–5000 fold (Monteil et al., 2020).
There is evidence from mainly animal studies (Soler et al., 2008) that angiotensin converting enzyme type 1 (ACE1) inhibitors, AT1-receptor antagonists (angiotensin type 1 receptor blocker: ARB) and aldosterone antagonists (mineralocorticoid receptor antagonist: MRA), commonly used clinically, could theoretically up-regulate ACE2 by diverting precursor molecules to ACE2, and so increase the number of binding targets for CoV-2. This has led to concerns about their use as medicines in patients contracting COVID-19. However, there is no clear evidence to suggest that the use of these medicines is linked to either increased risk of infection or to more severe symptoms of COVID-19 (see Jarcho et al., 2020). Data on the use of ACE1 inhibitors or ARB’ are too limited to suggest discontinuation in CoV-2 infection, and indeed withdrawal may be harmful in high risk patients (Vaduganathan et al., 2020).
Plasma ACE2 activity is low in healthy subjects, but elevated in patients with cardiovascular risk factors or cardiovascular disease (Patel et al., 2014). The strongest predictor of elevated plasma concentrations of ACE2 in two cohorts of heart failure patients was male sex, and this might at least partly explain the higher mortality rate from COVID-19 in men (Sama et al., 2020). In one of two heart failure cohorts, ACE1 inhibitor and ARB use in heart failure patients were independent predictors of lower plasma ACE2 (no predictor in other cohort) (Sama et al., 2020). Hence there is no evidence that ACE1 inhibitors or ARB’s increase plasma ACE2 and may even inconsistently lower ACE2 levels. Use of an MRA in heart failure patients was a weak predictor of higher plasma ACE2 concentrations in one of two cohorts (Sama et al., 2020). Hence, effects of MRAs on ACE2 were small and inconsistent, and even if this is consistently shown, evidence would be required that this is associated with more severe viral infection, and even then this has to be balanced against their usefulness in the treatment of heart failure (Sama et al., 2020).
3. The renin angiotensin aldosterone system (RAAS)
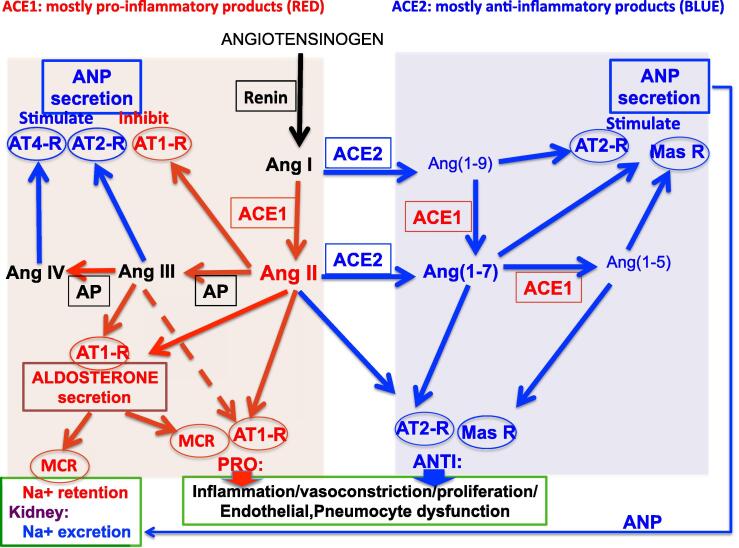
The diagram of Fig. 1 illustrates the complex interactions in the RAAS. The large polypeptide precursor angiotensinogen is converted by the enzyme renin to the decapeptide angiotensin I (Ang I). Fig. 2 shows the structures and the relationship between the major peptides in the angiotensin family.
Fig. 1.
The RAAS under normal conditions is a complex balance of pro- and anti-inflammatory mediators. For simplicity, some actions of agents are not indicated. The enzymes Renin, ACE1, ACE2 and aminopeptidases (AP) are displayed in small rectangles and receptors are displayed in small circles. Colour coding: red for generally pro-inflammatory pathways (arrows) and mediators, blue for generally anti-inflammatory pathways (arrows) and mediators. PRO indicates pro-inflammatory etc, ANTI indicates anti-inflammatory. Large rectangles show target tissues. Actions on cardiac atria are shown as stimulation or inhibition of atrial natriuretic peptide (ANP) secretion (due to the complexity of the diagram, ANP secretion is shown both top left and right). Actions on the zona glomerulosa of adrenal cortex are shown as aldosterone secretion. Actions shown on renal tubules are sodium (Na+) retention (aldosterone) and sodium (Na+) excretion (ANP). Widespread actions (PRO or ANTI) on multiple target tissues are Inflammation, vasoconstriction, proliferation, endothelial and pneumocyte dysfunction, etc. Background shading indicates: ACE1 products (pink); products requiring ACE2 alone or ACE2 and ACE1 (blue). Abbreviations: ANP, atrial natriuretic peptide; AP: aminopeptidases; AT1-R: AT1-receptor; AT2-R: AT2-receptor; AT4-R: AT4-receptor; mas-R: mas-receptor; MCR: mineralocorticoid receptor.
Fig. 2.
The relationship between the various angiotensin peptides.
Ang I is converted to Ang II by the enzyme ACE1 (often called simply ACE), cleaving the 2 C-terminal amino acids to produce the octopeptide Ang II (Fig. 1, Fig. 2). Ang II is thus the major ACE1 product and the most active hormone in the system, acting in a pro-inflammatory manner, involving especially AT1-receptors. Ang II also acts the zona glomerulosa of the adrenal cortex to release aldosterone (Fig. 1). Aldosterone itself has pro-inflammatory actions and causes sodium retention in the renal tubules, both involving mineralocorticoid receptors (MCR) (Fig. 1).
Ang II acts at AT1-receptors (usually pro-inflammatory) and AT2-receptors (usually anti-inflammatory). The AT1-receptor has a wide distribution in the body, but the AT2-receptor is more localised (de Gasparo et al., 2000). In human lung, AT1-receptors were reported in the vascular smooth muscle and endothelium, macrophages and fibroblasts, whereas AT2-receptors were found in the epithelium, and the proportion of AT1-receptors has been reported to increase in lung disease (Bullock et al., 2001).
Ang III and Ang IV are produced by the action of aminopeptidases to cleave the N-terminal amino acids forming Ang(2–8) (Ang III) and Ang (3–8) (Ang IV) (Fig. 1, Fig. 2). Ang IV may act mainly on AT4-receptors.
However, both Ang I and Ang II can be converted to alternative products by the enzyme ACE2 (see Fig. 1). The decapeptide Ang I is converted to the nonapeptide Ang(1–9) and the octapeptide Ang II is converted to the heptapeptide Ang(1–7), both by ACE-2 by cleaving the C-terminal amino acid (see Fig. 2). ACE1 converts Ang(1–9) to Ang(1–7), again cleaving 2 C-terminal amino acids. ACE-1 can also cleave a further 2 amino acids resulting in Ang(1–5). The major active agent formed involving ACE2 is thought to be Ang(1–7). Ang(1–7) has an anti-inflammatory profile of action, perhaps mainly by action at mas-receptors, although Ang(1–9) may also be of some importance, acting at the AT2-receptor (Ocaranza et al., 2014).
Gene knockout mice studies have informed on the importance of ACE2. Deficiency in ACE1 or addition of recombinant ACE2 can protect mice from severe acute lung injury induced by acid aspiration or sepsis (Imai et al., 2005). Targeted disruption of ACE2 in mice results in a severe cardiac contractility defect and increased angiotensin II levels, and this is reversed by genetic ablation of ACE1 (double knockout of both ACE1 and ACE2) (Crackower et al., 2002) or AT1-receptor antagonists (Nakamura et al., 2008).
3.1. The angiotensin family of peptides
The exact function of some of the angiotensin peptides has not been fully established, but the major reported actions of each of the peptides are listed below.
Ang I is thought to be mainly a precursor of active angiotensin peptides with limited, if any, activity.
Ang II is the most active hormone in the system and acts in a pro-inflammatory manner through actions at the AT1-receptor, resulting in increased blood pressure, increased endothelial permeability, fibrosis, apoptosis and pneumocyte dysfunction (de Gasparo et al., 2000). Macrophages and neutrophils that overexpress ACE1 have increased abilities to fight infections and kill bacteria by increased production of superoxide, although these effects may be independent of angiotensin II (Bernstein et al., 2018). Ang II inhibits ANP secretion by action at the AT1-receptor (Park et al., 2015). Ang II raises blood pressure in rats by peripheral (Yatabe et al., 2011), and central actions (Wright et al., 2003) and stimulates aldosterone secretion possibly by action at both AT1- and AT2-receptors (Yatabe et al., 2011). Ang II also has some counterbalancing anti-inflammatory actions involving the AT2-receptor, which is less widely located than the AT1-receptor, including vasodilatation by release of NO from the vascular endothelium (de Gasparo et al., 2000).
Ang III, given peripherally does not raise blood pressure in rats (Yatabe et al., 2011), but when given centrally, has similar pressor effects to Ang II, although less prolonged (Wright et al., 2003). Ang III stimulates aldosterone secretion by an action only partly involving AT2-receptors in rats (Yatabe et al., 2011), and ANP secretion via AT2-receptors (Park et al., 2015)
Ang IV stimulates ANP secretion by an action not involving AT1- or AT2-receptors (Park et al., 2015) and inhibits apoptosis and inflammation by action at the putative AT4 receptors (Park et al., 2016). Ang IV was found not to raise blood pressure or stimulate aldosterone secretion in rats (Yatabe et al., 2011). Ang IV may have beneficial actions, at least in the cerebro-vascular system, acting at the putative AT4 receptor (Royea et al., 2017).
Ang(1–9) inhibits apoptosis, inflammation (Cha et al., 2018), vasodilatation and cardiac remodelling (Ocaranza et al., 2014) by actions at the AT2-receptor. Incidentally, Ang(1–9), unlike the other anti-inflammatory products involving ACE2, does not need ACE1 for its formation (see Fig. 1).
Ang(1–7) may act on a distinct receptor, the mas receptor to produce effects opposing the actions of Ang II. Ang(1–7) stimulates ANP by action at the mas receptor (Park et al., 2015). Ang(1–7) was found not to raise blood pressure or stimulate aldosterone secretion in rats (Yatabe et al., 2011), although it is reported to share the anti-diuretic actions of Ang II (Bader et al., 2018). Mas has been identified as the receptor linked to the beneficial anti-inflammatory actions of Ang(1–7) (Santos et al., 2003), but other studies have failed to show an interaction between Ang(1–7) and Mas (Gaidarov et al., 2018), bringing into question whether Ang(1–7) is indeed the endogenous agonist of this receptor. Ang(1–7) may act as an antagonist or partial agonist at the AT1-receptor (Gaidarov et al., 2018). Other studies indicate that Ang(1–7) mediates at least part of its cardioprotective effects by binding to the AT1-receptor without activating Gq but acting as a β-arrestin-biased agonist (Teixeira et al., 2017).
Ang(1–5) stimulates ANP secretion by action involving the mas receptor (Park et al., 2015).
4. Effects of CoV-2 virus on the RAAS
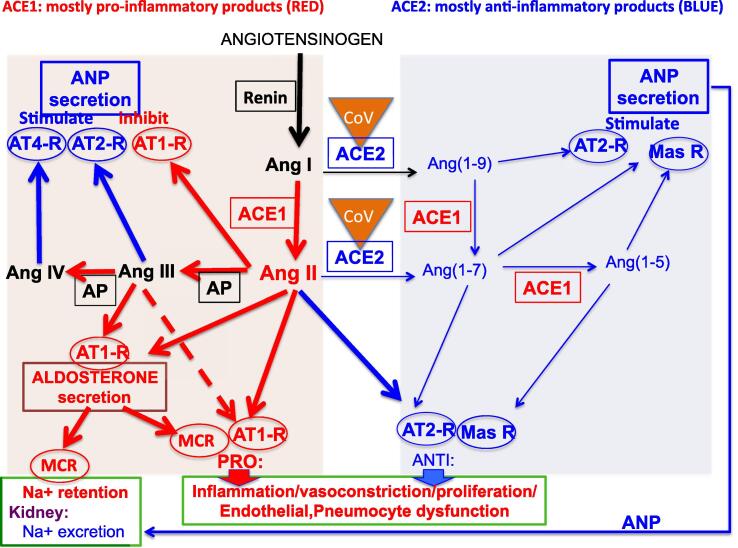
Not only is ACE2 the target of CoV-19 for penetration of cells, it is also inactivated by CoV-19. The diagram of Fig. 3 attempts to explain how binding of CoV-2 to ACE2 reduces availability of ACE2 to result in diminished production of anti-inflammatory products of ACE2, especially Ang(1–7), as shown by thin blue arrows in Fig. 3. This results in a dominance of the pro-inflammatory products are shown by thick red arrows in Fig. 3. Hence, the disruption of the RAAS by infection per se may lead to hyperstimulation and a cytokine storm through ACE1 product signalling pathways due to the loss of the counterbalancing ACE2 mechanisms.
Fig. 3.
The effects of Cov-2 virus on the RAAS. Cov-2 (CoV in yellow triangle) binds to ACE2, reducing the number of active ACE2 molecules. Thick arrows show diversion of precursors into the ACE1 product signalling pathways; thin arrows show diminished ACE2 product signalling pathways. The system is shifted towards pro-inflammatory pathways. Colour coding: red for generally pro-inflammatory pathways (arrows) and agents, blue for generally anti-inflammatory pathways (arrows) and agents. Abbreviations: ANP, atrial natriuretic peptide; AT1-R: AT1-receptor; AT2-R: AT2-receptor; AT4-R: AT4-receptor; CoV: Cov-2 virus bound to ACE2; mas-R: mas-receptor; MCR: mineralocorticoid receptor. For general description, see Fig. 1.
5. Manipulation of the RAAS system
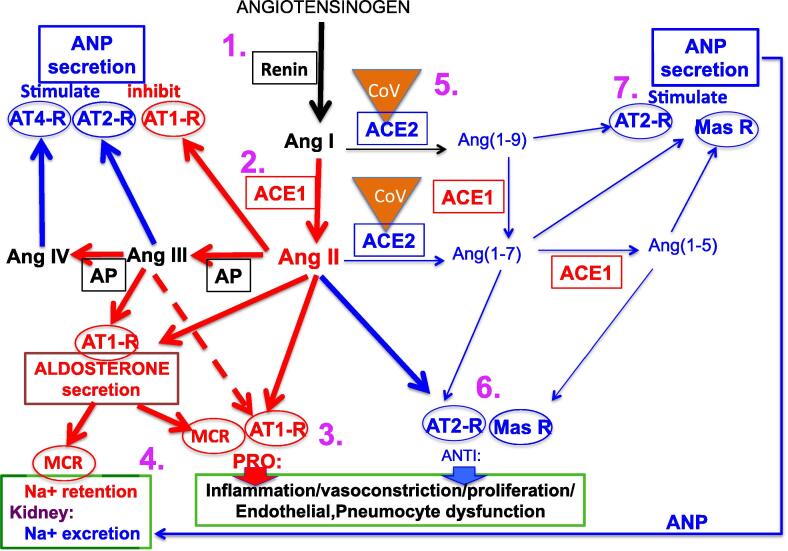
Fig. 4 shows possible sites of drug action in the RAAS. The object of modifying the RAAS system in COVID-19 is to reduce the formation of proinflammatory mediators, while increasing the formation of anti-inflammatory mediators. Counterbalance of the pro-inflammatory cascade could be achieved by strengthening the ACE2 product signalling pathways or weakening the ACE1 product signalling pathways, or both. The proinflammatory cascade may be weakened by inhibition of ACE1, preventing formation of Ang II, or by AT1-receptor antagonism/blockade of AT1-receptor, reducing the actions of Ang II at AT1-receptors and diverting Ang II to some anti-inflammatory actions involving the AT2-receptors. However, since ACE1, as well as ACE2, is required for the formation of Ang(1–7), blockade of AT1-receptors is the theoretically better pharmacological option, diverting angiotensin II, III & IV to anti-inflammatory actions, and allowing formation of the anti-inflammatory agent Ang(1–7). In contrast, ACE1 inhibition, while preventing formation of angiotensin II, also prevents formation of Ang(1–7). However, it must be noted that the overall balance between pro- and anti-inflammatory mediators achieved in the two situations (ARB and ACE1 inhibition) is not well established. Strengthening the ACE2 product signalling pathways can be achieved by increasing ACE2 levels in the plasma by recombinant enzyme, which in addition provides false targets for the virus, or by agonists mimicking products of ACE2, particularly Ang(1–7). Potential pharmacological strategies targeting the RAAS for treatment of COVID-19 have been discussed in recent publications (Zhang et al., 2020, Sriram and Insel, 2020), although it must be cautioned that, at present, these are only of unsubstantiated theoretical benefit.
Fig. 4.
Possible sites of drug action in manpulating the RAAS system that may potentially reduce the effects of Cov-2. Numbers 1–7 in purple refer to numbered sections (5.1-5.7) of the text concerning potential sites of drug action. Abbreviations: ANP, atrial natriuretic peptide (ANP); AT1-R: AT1-receptor; AT2-R: AT2-receptor; AT4-R: AT4-receptor; CoV: Cov-2 virus bound to ACE2; mas-R: mas-receptor; MCR: mineralocorticoid receptor. For general description, see Fig. 1.
5.1. Actions to prevent formation of Ang I
Gene therapy to suppress angiotensinogen at molecular level will result in diminished formation of both Ang II and Ang(1–7) (see Arendse et al., 2019). Renin inhibition will have similar actions but major adverse effects have been reported for the renin inhibitor aliskirin particularly in combination with ACE1 inhibitor or AT1-receptors antagonist (McMurray et al., 2016).
5.2. ACE1 inhibition
Inhibitors of ACE1 not only reduce formation of Ang II, but may divert the system to the formation of ACE2 products. ACE1 inhibitors and ARBs are widely used clinically. Heart failure patients receive ACE1 inhibitors or ARBs to lower their blood pressure and to slow down adverse cardiac remodelling (Yancy et al., 2017). There is limited evidence that ACE1 inhibitors and ARB’s have efficacy for migraine prevention (Rau & Dodick, 2019) and ACE1 inhibitor are used as chronic therapy in scleroderma (Lynch et al., 2016). Diabetes is linked, due to hyperglycemia-induced oxidative stress, to an increased mortality risk due to cardiovascular complications, and ACE inhibitors are employed to ameliorate this (Velagic et la., 2020). However cough is a common side effect of all ACE1 inhibitors, probably by prevention of breakdown of bradykinin, which may limit usefulness in COVID-19, and AT1-receptor blockers may be preferable (Sanchis-Gomar et al., 2020)
5.3. Antagonist for AT-1 receptors
Sartans such as telmisartan are selective antagonists of the AT1-receptor and reduce actions of Ang II at this receptor, not only diverting its actions more towards the AT2-receptor, but also leaving the ACE2 pathway intact.
ACE1 inhibitors and ARBs are used to treat hypertension, but, since ACE inhibitors have side effects of cough and uncommonly angioedema, risk-to-benefit analysis might suggest ARBs as the preferred choice (Messerli et al., 2018). In addition, there is the theoretical benefit of ARB’s in allowing formation of ACE1 dependent anti-inflammatory mediators (see Section 5, above). In essential hypertension, the ARB Losartan increased plasma renin activity and decreased plasma aldosterone (since AT1-receptor activation is required for aldosterone secretion) (Grossman et al., 1994), presumably resulting in increased angiotensin II levels.
Neprilysin is an endopeptidase than breaks down ANP and the vasodilator bradykinin: inhibition of the enzyme prevents the breakdown of these anti-inflammatory peptides. Combined neprilysin inhibition and ARB (AT1-receptor and neprilysin inhibitors: ARNIs) has been shown to be more effective than ARB alone in heart failure (Leong et al., 2019) and superior in mediating reverse cardiac remodelling (Wang et al., 2019). ARNIs cause a large rise in plasma angiotensin II levels but, perhaps surprisingly, were found not to increase plasma Ang(1–7) (Arendse et al., 2019).
Plasma angiotensin II levels are reported to be higher than of normal range in the majority of COVID-19 cases (90.2%), and were higher in critically ill COVID-19 patients than in those with mild COVID-19 symptoms (Wu et al., 2020). This may suggest that ARB’s or ACE1 inhibitors may prove useful in treating COVID-19. The high plasma angiotensin II levels may have to be considered in dosing with ARB’s, whereas ACE1 inhibitors directly lower angiotensin II levels, but admittedly with more side effects and with loss of some anti-inflammatory mediators.
5.4. Aldosterone antagonists
Often overlooked in the consideration of the more limited Renin Angiotensin System (RAS), is aldosterone, truly a component of the wider RAAS. Antagonists for mineralocorticoid receptors such as the aldosterone analogue spironolactone prevent actions of aldosterone including sodium retention. The combination of mineralocorticoid receptor antagonists with other antihypertensives improve blood pressure control in patients with resistant hypertension (Morimoto & Ichihara, 2020). Antagonists of aldosterone have also been found to increase ACE2 levels in human macrophages (Keidar et al., 2005). Aldosterone could theoretically be detrimental in COVID-19 infection by renal tubular actions to produce sodium retention, but also by tissue actions including endothelial alterations and immune system activation, resulting in pro-inflammatory actions (Davel et al., 2018).
5.5. ACE2 modulation
Increase in ACE2 may theoretically be detrimental or beneficial, but this has already been discussed in Section 2 above, particularly in terms of reducing infection.
Plasma angiotensin II, but not renin, was significantly higher in critically ill COVID-19 patients than in control or those with mild COVID-19 symptoms (Wu et al., 2020). Hence, elevated plasma angiotensin II elevation in severe COVID-19 infection may be due to loss of ACE2 by binding of CoV-2 rather than to increased renin activity, and this elevated angiotensin II may be a major pathological factor in critically ill COVID-19 patients (Wu et al., 2020). Hence, restoring ACE2 or employing agonists for AT2- or mas-receptors may be of potential therapeutic benefit in the later stage of infection.
5.6. AT2-receptor, AT4-receptor or mas receptor activation
Stimulation of either of the above receptors is likely to prove beneficial. Ang II acts on the AT2-receptor to produce counterbalancing anti-inflammatory actions. Ang(1–7) may act on a distinct receptor, the mas receptor to produce effects such as vasodilatation to oppose the actions of Ang II. Ang IV may also act on a specific AT-4 receptor which has been shown to have cardioprotective effect by inhibition of apoptosis (Park et al., 2016)
5.7. ANP-like actions
Stimulation of the particulate guanylate cyclase-A (pGC-A)/cGMP by atrial natriuretic peptide (ANP) results in a number of actions, including natriuresis, diuresis, blood pressure lowering, inhibition of cardiomyocyte hypertrophy and fibroblast proliferation, suppression of inflammatory cytokines and T cells and inhibition of aldosterone (Kuhn, 2016). Novel designer peptides that target the pGC-A receptor have been reported (Meems et al., 2019).
6. Sex differences
The greater mortality from COVID-19 reported in males worldwide (e.g. China: Parohan et al., 2020; Iran: Nikpouraghdam et al., 2020; Italy: Remuzzi and Remuzzi, 2020) may be partly explained by factors such as cardiovascular risk and increased incidence of smoking, alcohol consumption etc. Certainly, incidence of smoking is usually higher in males and nicotine has been reported to upregulate ACE1 and downregulate ACE2 (Oakes et al., 2018). However, there may also be a direct sex-related link, as male mice are more susceptible to SARS-CoV-1 infection than age-matched females (Channappanavar et al., 2017).
Modulation of the activity of the RAAS by sex steroids may markedly contribute to the gender differences observed in the pathophysiology of progressive kidney disease (Sandberg & Ji, 2003). In healthy individuals, circulating ACE2 levels are low, but elevated in, and a predictor of adverse events in, coronary artery disease (Ramchand et al., 2018). ACE2 levels are higher in male than female heart failure patients (Sama et al., 2020). In apparently healthy subjects, women exhibited a higher circulating concentration of the anti-inflammatory factor Ang(1–7) compared with men, whereas values of angiotensin II were similar (Sullivan et al., 2015).
Animal studies provide some evidence for increased ACE2 in males. Male mouse renal ACE2 is higher than female, and ACE2 activity was increased by ovariectomy in females (Liu et al., 2010). Male spontaneously hypertensive rats have higher cardiac ACE1 and ACE2 (Dalpiaz et al., 2015), and male hypertensive mice have higher renal ACE2 (Gupte et al., 2012), than female. Oestrogen deficiency in ovariectomised rats has been shown to activate the RAAS, increasing plasma angiotensin II and expression of pro-fibrotic factors (Hussien et al., 2019).
These results may suggest that there may be sex differences in ACE2 levels that may be linked to increased susceptibility to COVID-19, but further studies are required to elucidate the effects of sex on the function of the RAAS.
7. Conclusion
COVID-19 and the RAAS are closely linked both in infection and in possible post-infection inflammatory cascades. It is appropriate that the theoretical benefits of modulation of the RAAS should be considered based on available clinical evidence. In addition, in this short review we have tried to explain the actions of the angiotensin family of peptides and produce a relatively simple model and diagrammatic summary of the RAAS and the possible sites of intervention.
Declaration of Competing Interest
The authors declared that there is no conflict of interest.
Footnotes
Peer review under responsibility of King Saud University.
References
- Arendse L.B., Danser A.H.J., Poglitsch M., Touyz R.M., Burnett J.C., Jr., Llorens-Cortes C.1, Ehlers M.R., Sturrock E.D. Novel, therapeutic approaches targeting the renin-angiotensin system and associated peptides in hypertension and heart failure. Pharmacol. Rev. 2019;71(4):539–570. doi: 10.1124/pr.118.017129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bader M., Alenina N., Young D., Santos R.A.S., Touyz R.M. The meaning of mas. Hypertension. 2018;72:1072–1075. doi: 10.1161/HYPERTENSIONAHA.118.10918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bernstein K.E., Khan Z., Giani J.F., Cao D.Y., Bernstein E.A., Shen X.Z. Angiotensin-converting enzyme in innate and adaptive immunity. Nat. Rev. Nephrol. 2018;14(5):325–336. doi: 10.1038/nrneph.2018.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bullock G.R., Steyaert I., Bilbe G., Carey R.M., Kips J., De Paepe J.B., Pauwels R., Praet M., Siragy H.M., de Gasparo M. Distribution of type-1 and type-2 angiotensin receptors in the normal human lung and in lungs from patients with chronic obstructive pulmonary disease. Histochem. Cell Biol. 2001;115(2):117–124. doi: 10.1007/s004180000235. [DOI] [PubMed] [Google Scholar]
- Channappanavar R., Fett C., Mack M., Ten Eyck P.P., Meyerholz D.K., Perlman S. Sex-based differences in susceptibility to severe acute respiratory syndrome coronavirus infection. J. Immunol. 2017;198:4046–4053. doi: 10.4049/jimmunol.1601896. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cha S.A., Park B.M., Kim S.H. Angiotensin-(1–9) ameliorates pulmonary arterial hypertension via angiotensin type II receptor. Korean J. Physiol. Pharmacol. 2018;22(4):447–456. doi: 10.4196/kjpp.2018.22.4.447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Crackower M.A., Sarao R., Oudit G.Y., Yagil C., Kozieradzki I., Scanga S.E., Oliveira-dos-Santos A.J., da Costa J., Zhang L., Pei Y., Scholey J., Ferrario C.M., Manoukian A.S., Chappell M.C., Backx P.H., Yagil Y., Penninger J.M. Angiotensin-converting enzyme 2 is an essential regulator of heart function. Nature. 2002;417:822–828. doi: 10.1038/nature00786. [DOI] [PubMed] [Google Scholar]
- Dalpiaz P.L., Lamas A.Z., Caliman I.F., Ribeiro R.F.J., Abreu G.R., Moyses M.R., Andrade T.U., Gouvea S.A., Alves M.F., Carmona A.K. Sex hormones promote opposite effects on ACE and ACE2 activity, hypertrophy and cardiac contractility in spontaneously hypertensive rats. PLoS ONE. 2015;10(5):e0127515. doi: 10.1371/journal.pone.0127515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- da Silva-Bertani D.C.T., Vileigas D.F., Mota G.A.F., de Souza S.L.B., Sant'Ana P.G., Freire P.P., de Tomasi L.C., Corrêa C.R., Padovani C.R., Fernandes T., de Oliveira E.M., Cicogna A.C. Increased angiotensin II from adipose tissue modulates myocardial collagen I and III in obese rats. Life Sci. 2020;1(252):117650. doi: 10.1016/j.lfs.2020.117650. [DOI] [PubMed] [Google Scholar]
- Davel A.P., Jaffe I.Z., Tostes R.C., Jaisser F., Belin de Chantemèle E.J. New roles of aldosterone and mineralocorticoid receptors in cardiovascular disease: translational and sex-specific effects. Am. J. Physiol. Heart Circ. Physiol. 2018;315(4):H989–H999. doi: 10.1152/ajpheart.00073.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Gasparo M., Catt K.J., Inagami T., Wright J.W., Unger T. International union of pharmacology. XXIII. The angiotensin II receptors. Pharmacol. Rev. 2000;52(3):415–472. [PubMed] [Google Scholar]
- Gaidarov I., Adams J., Frazer J., Anthony T., Chen X., Gatlin J., Semple G., Unett D.J. Angiotensin (1–7) does not interact directly with MAS1, but can potently antagonize signaling from the AT1 receptor. Cell. Signal. 2018;50:9–24. doi: 10.1016/j.cellsig.2018.06.007. [DOI] [PubMed] [Google Scholar]
- Gheblawi M., Wang K., Viveiros A., Nguyen Q., Zhong J.C., Turner A.J., Raizada M.K., Grant M.B., Oudit G.Y. Angiotensin-converting enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system. Celebrating the 20th anniversary of the discovery of ACE2. Circ. Res. 2020;126:1456–1474. doi: 10.1161/CIRCRESAHA.120.317015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grossman E., Peleg E., Carroll J., Shamiss A., Rosenthal T. Hemodynamic and humoral effects of the angiotensin II antagonist losartan in essential hypertension. Am. J. Hypertension. 1994;12:1041–1044. doi: 10.1093/ajh/7.12.1041. [DOI] [PubMed] [Google Scholar]
- Gupte M., Thatcher S.E., Boustany-Kari C.M., Shoemaker R., Yiannikouris F., Zhang X., Karounos M., Cassis L.A. Angiotensin converting enzyme 2 contributes to sex differences in the development of obesity hypertension in C57BL/6 mice. Arterioscler. Thromb. Vasc. Biol. 2012;32:1392–1399. doi: 10.1161/ATVBAHA.112.248559. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamming I., Timens W., Bulthuis M.L. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203:631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Henry B.M., Vikse J., Benoit S., Favaloro E.J., Lippi G. Hyperinflammation and derangement of renin-angiotensin-aldosterone system in COVID-19: A novel hypothesis for clinically suspected hypercoagulopathy and microvascular immunothrombosis. Clin. Chim. Acta. 2020;507:167–173. doi: 10.1016/j.cca.2020.04.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hussien N.I., El-Kerdasy H.I., Sorour S.M., Shoman A.A. Chronic oestrogen deficiency induced by ovariectomy may cause lung fibrosis through activation of the renin-angiotensin system in rats. Arch. Physiol. Biochem. 2019;12:1–10. doi: 10.1080/13813455.2019.1676262. [DOI] [PubMed] [Google Scholar]
- Imai Y., Kuba K., Rao S., Huan Y., Guo F., Guan B., Yang P., Sarao R., Wada T., Leong-Poi H., Crackower M.A., Fukamizu A., Hui C.C., Hein L., Uhlig S., Slutsky A.S., Jiang C., Penninger J.M. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436:112–116. doi: 10.1038/nature03712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jarcho J.A., Ingelfinger J.R., Hamel M.B., D'Agostino R.B., Sr, Harrington D.P. Inhibitors of the renin-angiotensin-aldosterone system and COVID-19. N. Engl. J. Med. 2020 doi: 10.1056/NEJMe2012924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keidar S., Gamliel-Lazarovich A., Kaplan M. Mineralocorticoid receptor blocker increases angiotensin- converting enzyme 2 activity in congestive heart failure patients. Circ. Res. 2005;97:946–953. doi: 10.1161/01.RES.0000187500.24964.7A. [DOI] [PubMed] [Google Scholar]
- Kuba K., Imai Y., Rao S., Gao H., Guo F., Guan B., Huan Y., Yang P., Zhang Y., Deng W. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus-induced lung injury. Nat. Med. 2005;11:875–879. doi: 10.1038/nm1267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuhn M. Molecular physiology of membrane guanylyl cyclase receptors. Physiol. Rev. 2016;96(2):751–804. doi: 10.1152/physrev.00022.2015. [DOI] [PubMed] [Google Scholar]
- Leong D.P., McMurray J.J.V., Joseph P.G., Yusuf S. From ACE inhibitors/ARBs to ARNIs in coronary artery disease and heart failure. J. Am. Coll. Cardiol. 2019;74:683–698. doi: 10.1016/j.jacc.2019.04.068. [DOI] [PubMed] [Google Scholar]
- Li B., Yang J., Zhao F., Zhi L., Wang X., Liu L., Bi Z., Zhao Y. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clin. Res. Cardiol. 2020;109:531–538. doi: 10.1007/s00392-020-01626-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J., Ji H., Zheng W. Sex differences in renal angiotensin converting enzyme 2 (ACE2) activity are 17β-oestradiol-dependent and sex chromosome-independent. Biol. Sex Differ. 2010;1(1):6. doi: 10.1186/2042-6410-1-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lynch B.M., Edward P Stern, Voon Ong, Mark Harber, Aine Burns, Christopher P. Denton. UK Scleroderma Study Group (UKSSG) guidelines on the diagnosis and management of scleroderma renal crisis. Clin. Exp. Rheumatol. 2016;34(Suppl 100):106–109. [PubMed] [Google Scholar]
- McMurray J.J.V., Krum H., Abraham W.T., Dickstein K., Køber L.V. Aliskiren, enalapril, or aliskiren and enalapril in heart failure. N. Engl. J. Med. 2016;374:1521–1532. doi: 10.1056/NEJMoa1514859. [DOI] [PubMed] [Google Scholar]
- Meems L.M.G., Andersen I.A., Pan S., Harty G., Chen Y., Zheng Y., Harders G.E., Ichiki T., Heublein D.M., Iyer S.R., Sangaralingham S.J., McCormick D.J., Burnett J.C., Jr. Design, synthesis, and actions of an innovative bispecific designer peptide. Hypertension. 2019;73:900–909. doi: 10.1161/HYPERTENSIONAHA.118.12012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Messerli F.H., Bangalore S., Bavishi C., Rimoldi S.F. Angiotensin-converting enzyme inhibitors in hypertension: to use or not to use? J. Am. Coll. Cardiol. 2018;71:1474–1482. doi: 10.1016/j.jacc.2018.01.058. [DOI] [PubMed] [Google Scholar]
- Monteil V., Kwon H., Prado P., Hagelkruys A., Wimmer R.A., Stahl M., Leopoldi A., Garreta E., Hurtado del Pozo C., Prosper F., Romero J.P., Wirnsberger G., Zhang H., Slutsky A.S., Conder R., Montserrat N., Mirazimi A., Penninger J.M. Inhibition of SARS-CoV-2 infections in engineered human tissues using clinical-grade soluble human ACE2. Cell. 2020;181:905–913. doi: 10.1016/j.cell.2020.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morimoto S., Ichihara A. Management of primary aldosteronism and mineralocorticoid receptor-associated hypertension. Hypertens. Res. 2020 doi: 10.1038/s41440-020-0468-3. [DOI] [PubMed] [Google Scholar]
- Nakamura K., Koibuchi N., Nishimatsu H., Higashikuni Y., Hirata Y., Kugiyama K., Nagai R., Sata M. Candesartan ameliorates cardiac dysfunction observed in angiotensin-converting enzyme 2-deficient mice. Hypertens. Res. 2008;31:1953–1961. doi: 10.1291/hypres.31.1953. [DOI] [PubMed] [Google Scholar]
- Nikpouraghdam M., Farahani A.J., Alishiri G.H., Heydari S., Ebrahimnia M., Samadinia H. Epidemiological Characteristics of Coronavirus Disease 2019 (COVID-19) patients in IRAN: a single center study. J. Clin. Virol. 2020;127:104378. doi: 10.1016/j.jcv.2020.104378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oakes J.M., Fuchs R.M., Gardner J.D., Lazartigues E., Yue X. Nicotine and the renin-angiotensin system. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2018;315(5):R895–R906. doi: 10.1152/ajpregu.00099.2018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ocaranza M.P., Michea L., Chiong M., Lagos C.F., Lavandero S., Jalil J.E. Recent insights and therapeutic perspectives of angiotensin-(1–9) in the cardiovascular system. Clin. Sci. (Lond.) 2014;127(9):549–557. doi: 10.1042/CS20130449. [DOI] [PubMed] [Google Scholar]
- Park B.M., Cha S.A., Han B.R., Kim S.H. Angiotensin IV stimulates high atrial stretch-induced ANP secretion via insulin regulated aminopeptidase. Peptides. 2015;63:30–37. doi: 10.1016/j.peptides.2014.10.016. [DOI] [PubMed] [Google Scholar]
- Park B.M., Cha S.A., Lee S.H., Kim S.H. Angiotensin IV protects cardiac reperfusion injury by inhibiting apoptosis and inflammation via AT4R in rats. Peptides. 2016;79:66–74. doi: 10.1016/j.peptides.2016.03.017. [DOI] [PubMed] [Google Scholar]
- Parohan M., Yaghoubi S., Seraji A., Javanbakht M.H., Sarraf P., Djalali M. Risk factors for mortality in patients with Coronavirus disease 2019 (COVID-19) infection: a systematic review and meta-analysis of observational studies. Aging Male. 2020;8:1–9. doi: 10.1080/13685538.2020.1774748. [DOI] [PubMed] [Google Scholar]
- Patel S.K., Velkoska E., Freeman M., Wai B., Lancefield T.F., Burrell L.M. From gene to protein—experimental and clinical studies of ACE2 in blood pressure control and arterial hypertension. Front. Physiol. 2014;5:227. doi: 10.3389/fphys.2014.00227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramchand J., Patel S.K., Srivastava P.M., Farouque O., Burrell L.M. Elevated plasma angiotensin converting enzyme 2 activity is an independent predictor of major adverse cardiac events in patients with obstructive coronary artery disease. PLoS ONE. 2018;13(6):e0198144. doi: 10.1371/journal.pone.0198144. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rau J.C., Dodick D.W. Other preventive anti-migraine treatments: ACE inhibitors, ARBs, calcium channel blockers, serotonin antagonists, and NMDA receptor antagonists. Curr. Treat. Options Neurol. 2019;21:17. doi: 10.1007/s11940-019-0559-0. [DOI] [PubMed] [Google Scholar]
- Remuzzi A., Remuzzi G. COVID-19 and Italy: what next? Lancet. 2020 doi: 10.1016/S0140-6736(20)30627-9. S0140-6736(20)30627-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Royea J., Zhang L., Tong X.K., Hamel E. Angiotensin IV receptors mediate the cognitive and cerebrovascular benefits of losartan in a mouse model of Alzheimer's disease. J. Neurosci. 2017;37(22):5562–5573. doi: 10.1523/JNEUROSCI.0329-17.2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sama I.E., Ravera A., Santema B.T., van Goor H., Ter Maaten J.M., Cleland J.G.F., Rienstra M., Friedrich A.W., Samani N.J., Ng L.L., Dickstein K., Lang C.C., Filippatos G., Anker S.D., Ponikowski P., Metra M., van Veldhuisen D.J., Voors A.A. Circulating plasma concentrations of angiotensin-converting enzyme 2 in men and women with heart failure and effects of renin-angiotensin-aldosterone inhibitors. Eur. Heart J. 2020;41:1810–1817. doi: 10.1093/eurheartj/ehaa373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sanchis-Gomar F., Lavie C.J., Perez-Quilis C., Henry B.M., Lippi G. Angiotensin-converting enzyme 2 and antihypertensives (angiotensin receptor blockers and angiotensin-converting enzyme inhibitors) in coronavirus disease mayo. Clin. Proc. 2020 doi: 10.1016/j.mayocp.2020.03.026. pii: S0025-6196(20)30315-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sandberg K., Ji H. Sex and the renin angiotensin system: implications for gender differences in the progression of kidney disease. Adv. Ren. Replace Ther. 2003;10(1):15–23. doi: 10.1053/jarr.2003.50006. [DOI] [PubMed] [Google Scholar]
- Santos R.A., Simoes e Silva A.C., Maric C., Silva D.M.R., Machado R.P., de Buhr I., Heringer-Walther S., Pinheiro S.V.B., Lopes M.T., Bader M., Mendes E.P. Angiotensin-(1–7) is an endogenous ligand for the G-protein coupled receptor Mas. Proc. Natl. Acad. Sci. U. S. A. 2003;100:8258–8263. doi: 10.1073/pnas.1432869100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simonnet A., Chetboun M., Poissy J., Raverdy V., Noulette J., Duhamel A., Labreuche J., Mathieu D., Pattou F., Jourdain M. High prevalence of obesity in severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) requiring invasive mechanical ventilation. Obesity (Silver Spring) 2020 doi: 10.1002/oby.22831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soler M.J., Barrios C., Oliva R., Batlle D. Pharmacologic modulation of ACE2 expression. Curr. Hypertens. Rep. 2008;10(5):410–414. doi: 10.1007/s11906-008-0076-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sriram K., Insel P.A. A hypothesis for pathobiology and treatment of COVID-19: the centrality of ACE1/ACE2 imbalance. Br. J. Pharmacol. 2020 doi: 10.1111/bph.15082. (Epub ahead of print) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sullivan J.C., Rodriguez-Miguelez P., Zimmerman M.A., Harris R.A. Differences in angiotensin(1–7) between men and women. Am. J. Physiol. Heart Circ. Physiol. 2015;308:H1171–H1176. doi: 10.1152/ajpheart.00897.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang N., Li D., Wang X., Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020;18(4):844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Teixeira L.B., Parreiras-e-Silva L.T., Bruder-Nascimento T., Duarte D.A., Simões S.C., Costa R.M., Rodríguez D.Y., Ferreira P.A.B., Silva C.A.A., Abrao E.P., Oliveira E.B., Bouvier M., Tostes R.C., Costa-Neto C.M. Ang-(1–7) is an endogenous β-arrestin-biased agonist of the AT1 receptor with protective action in cardiac hypertrophy. Sci. Rep. 2017;7:11903. doi: 10.1038/s41598-017-12074-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tipnis S.R., Hooper N.M., Hyde R., Karran E., Christie G., Turner A.J. A human homolog of angiotensin-converting enzyme. Cloning and functional expression as a captopril-insensitive carboxypeptidase. J. Biol. Chem. 2000;275(43):33238–33243. doi: 10.1074/jbc.M002615200. [DOI] [PubMed] [Google Scholar]
- Vaduganathan M., Vardeny O., Michel T., McMurray J.J.V., Pfeffer M.A., Solomon S.D. Renin–angiotensin–aldosterone system inhibitors in patients with Covid-19. N. Engl. J. Med. 2020;382:1653–1659. doi: 10.1056/NEJMsr2005760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Velagic A., Qin C., Woodman O.L., Horowitz J.D., Ritchie R.H., Kemp-Harper B.K. Nitroxyl: a novel strategy to circumvent diabetes associated impairments in nitric oxide signaling. Front. Pharmacol. 2020;11:727. doi: 10.3389/fphar.2020.00727. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walls A.C., Park Y.-J., Tortorici M.A., Wall A., McGuire A.T., Veesler D. Structure, function, and antigenicity of the SARS-CoV-2 spike glyco-protein. Cell. 2020;181:281–292. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wan Y., Shang J., Graham R., Baric R.S., Li F. Receptor recog- nition by novel coronavirus from Wuhan: An analysis based on decade-long structural studies of SARS. J. Virol. 2020;94:e00127–e220. doi: 10.1128/JVI.00127-20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang Y., Zhou R., Lu C. Effects of the angiotensin-receptor neprilysin inhibitor on cardiac reverse remodeling: meta-analysis. J. Am. Heart Assoc. 2019;8:e012272. doi: 10.1161/JAHA.119.012272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wrapp D., Wang N., Corbett K.S., Goldsmith J.A., Hsieh C.L., Abiona O., Graham B.S., McLellan J.S. Cryo-EM structure of the 2019- nCoV spike in the prefusion conformation. Science. 2020;367:1260–1263. doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright J.R., Elizabeth Tamura-Myers, Wilson Wendy L., Roques Bernard P., Llorens-Cortes Catherine, Speth Robert C., Harding Joseph W. Conversion of brain angiotensin II to angiotensin III is critical for pressor response in rats. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2003;284:R725–R733. doi: 10.1152/ajpregu.00326.2002. [DOI] [PubMed] [Google Scholar]
- Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72 314 Cases from the Chinese center for disease control and prevention. JAMA. 2020 doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- Wu Z., Hu R., Zhang C., Ren W., Yu A., Zhou X. Elevation of plasma angiotensin II level is a potential pathogenesis for the critically ill COVID-19 patients. Version 2. Crit. Care. 2020;24:290. doi: 10.1186/s13054-020-03015-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yancy C.W., Jessup M., Bozkurt B. ACC/AHA/HFSA focused update of the 2013 ACCF/AHA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Failure Society of America. Circulation. 2017;2017(136):e137–e161. doi: 10.1161/CIR.0000000000000509. [DOI] [PubMed] [Google Scholar]
- Yatabe J., Yoneda M., Yatabe M.S., Watanabe T., Felder R.A., Jose P.A., Sanada H. Angiotensin III stimulates aldosterone secretion from adrenal gland partially via angiotensin II type 2 receptor but not angiotensin II type 1 receptor. Endocrinology. 2011;152:1582–1588. doi: 10.1210/en.2010-1070. [DOI] [PubMed] [Google Scholar]
- Zhang H., Penninger J.M., Li Y., Zhong N., Slutsky A.S. Angiotensin-converting enzyme 2 (ACE2) as a SARS-CoV-2 receptor: molecular mechanisms and potential therapeutic target. Intensive Care Med. 2020;46:586–590. doi: 10.1007/s00134-020-05985-9. [DOI] [PMC free article] [PubMed] [Google Scholar]