Abstract
Antibody-based interventions against SARS-CoV-2 could limit morbidity, mortality, and possibly transmission. An anticipated correlate of such countermeasures is the level of neutralizing antibodies against the SARS-CoV-2 spike protein, which engages with host ACE2 receptor for entry. Using an infectious molecular clone of vesicular stomatitis virus (VSV) expressing eGFP as a marker of infection, we replaced the glycoprotein gene (G) with the spike protein of SARS-CoV-2 (VSV-eGFP-SARS-CoV-2) and developed a high-throughput-imaging-based neutralization assay at biosafety level 2. We also developed a focus-reduction neutralization test with a clinical isolate of SARS-CoV-2 at biosafety level 3. Comparing the neutralizing activities of various antibodies and ACE2-Fc soluble decoy protein in both assays revealed a high degree of concordance. These assays will help define correlates of protection for antibody-based countermeasures and vaccines against SARS-CoV-2. Additionally, replication-competent VSV-eGFP-SARS-CoV-2 provides a tool for testing inhibitors of SARS-CoV-2 mediated entry under reduced biosafety containment.
Keywords: SARS-CoV-2, VSV, coronavirus, COVID19, neutralizing, antibody, ACE2, serum, surrogate assay
Graphical Abstract
Highlights
-
•
Vesicular stomatitis virus encoding the SARS-CoV-2 spike replicates to high titers
-
•
Virus propagation is enhanced by a truncation in the cytoplasmic tail of the spike
-
•
Neutralization can be assessed by BSL2 and BSL3 high-throughput assays
-
•
SARS-CoV-2- and VSV-SARS-CoV-2-based neutralization assays correlate
Case, Rothlauf et al. generate a replication-competent vesicular stomatitis virus (VSV) expressing the SARS-CoV-2 spike and compare the neutralizing activity of antibodies with VSV-SARS-CoV-2 to fully infectious SARS-CoV-2. They show that VSV-SARS-CoV-2 is a useful BSL2 surrogate virus, as neutralization profiles strongly correlate with focus-reduction neutralization tests using SARS-CoV-2.
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a positive-sense, single-stranded, enveloped RNA virus that was first isolated in Wuhan, China in December, 2019 from a cluster of acute respiratory illness cases (Guan et al., 2020). SARS-CoV-2 is the etiologic agent of coronavirus disease 2019 (COVID-19), which as of June 17, 2020 has more than 8.2 million confirmed cases causing 445,000 deaths. Virtually all countries and territories have been affected, with major epidemics in Central China, Italy, Spain, France, Iran, Russia, Brazil, India, Peru, the United Kingdom, and the United States. SARS-CoV-2 is thought to be of zoonotic origin and is closely related to the original SARS-CoV (Zhang et al., 2020; Zhou et al., 2020). Most cases are spread by direct human-to-human transmission, with community transmission occurring from both symptomatic and asymptomatic individuals (Bai et al., 2020). This has resulted in a global pandemic with severe economic, political, and social consequences. The development, characterization, and deployment of an effective vaccine or antibody prophylaxis or treatment against SARS-CoV-2 could prevent morbidity and mortality and curtail its epidemic spread.
The viral spike protein (S) mediates all steps of coronavirus entry into target cells, including receptor binding and membrane fusion (Tortorici and Veesler, 2019). During viral biogenesis, the S protein undergoes furin-dependent proteolytic processing as it transits through the trans-Golgi network and is cleaved into S1 and S2 subunits that function in receptor binding and membrane fusion, respectively (Walls et al., 2020). Angiotensin-converting enzyme 2 (ACE2) serves as a cell surface receptor (Letko et al., 2020; Wrapp et al., 2020) for SARS-CoV-2, and productive infection is facilitated by additional processing of S2 by the host cell serine protease TMPRSS2 (Hoffmann et al., 2020).
Laboratory studies of SARS-CoV-2 require biosafety level 3 (BSL3) containment with positive-pressure respirators. Single-round pseudotyped viruses complemented by expression of the SARS-CoV-2 S protein in trans serve as biosafety level 2 (BSL2) surrogates that can facilitate studies of viral entry and the inhibition of infection by neutralizing antibodies and other inhibitors (Hoffmann et al., 2020; Lei et al., 2020; Nie et al., 2020; Ou et al., 2020). Such pseudotyping approaches are used routinely by many laboratories for other highly pathogenic coronaviruses, including SARS-CoV and MERS-CoV (Fukushi et al., 2005, 2006; Giroglou et al., 2004; Kobinger et al., 2007). Viral pseudotyping assays are limited by the need to express the glycoprotein in trans and preclude forward genetic studies of the viral envelope protein. Expression of the glycoprotein is often accomplished by plasmid transfection, which requires optimization to minimize batch variation. Assays performed with such pseudotyped viruses rely on relative levels of infectivity as measured by a reporter assay without correlation to an infectious titer. It also is unknown how the display of S proteins on a heterologous virus impacts viral entry, antibody recognition, and antibody neutralization compared to infectious coronavirus. This question is important because neutralization assays are used to establish correlates of protection for vaccine and antibody-based countermeasures, and most manufacturers lack access to high-containment laboratories to test antibody responses against highly pathogenic coronaviruses such as SARS-CoV-2.
Here, we developed a simple and robust BSL2 assay for evaluating SARS-CoV-2 entry and its inhibition by antibodies. We engineered an infectious molecular clone of vesicular stomatitis virus (VSV) to encode the SARS-CoV-2 S protein in place of the native envelope glycoprotein (G) and rescued an autonomously replication-competent virus bearing the spike. Through passage of VSV-eGFP-SARS-CoV-2, we selected a gain-of-function mutation in S that allowed more efficient viral propagation yielding titers of >1 × 108 plaque-forming units (PFU)/mL. We characterized this variant with respect to inhibition by soluble human ACE2-Fc and monoclonal and polyclonal antibodies from humans and compared those results to neutralization tests with a clinical isolate of SARS-CoV-2. These studies demonstrate that a recombinant VSV expressing SARS-CoV-2 S behaves analogously to a clinical isolate of SARS-CoV-2, providing a useful high-throughput BSL2 assay for studying antibody neutralization or inhibition of viral spike-mediated entry.
Results
A Replication-Competent, Infectious VSV Chimera with SARS-CoV-2 S Protein
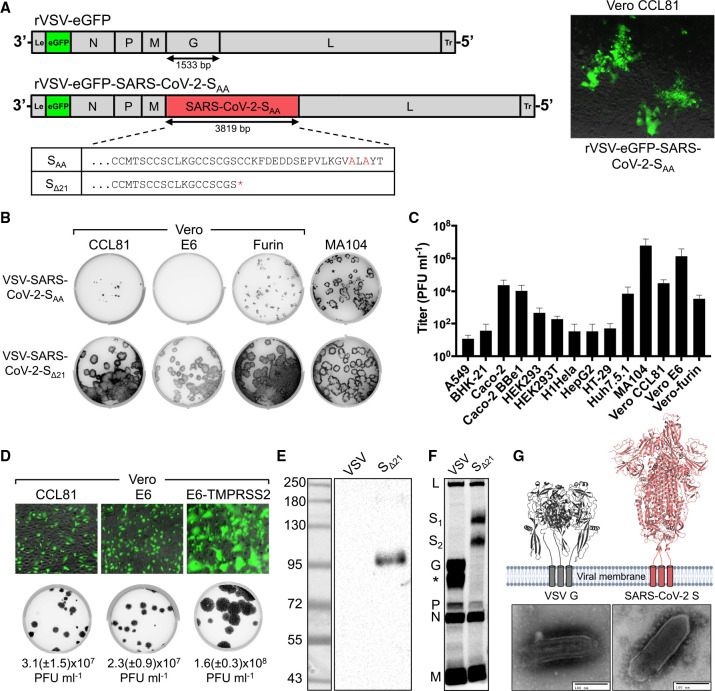
To generate a replication-competent virus to study entry and neutralization of SARS-CoV-2 at BSL2, we engineered an infectious molecular clone of VSV by replacing the endogenous glycoprotein (G) with SARS-CoV-2 S (Figure 1 A). SARS-CoV-2 S protein contains an endoplasmic reticulum (ER) retention sequence in the cytoplasmic tail (KxHxx-COOH) because virion assembly occurs in ER-Golgi intermediate compartments (Lontok et al., 2004; McBride et al., 2007; Ruch and Machamer, 2012). We pre-emptively altered that sequence to AxAxx to facilitate retargeting of S to the plasma membrane, the site of VSV assembly. Using established approaches (Figure S1A) (Whelan et al., 1995), we recovered infectious VSV-eGFP-SARS-CoV-2-SAA as determined by expression of the virus-encoded eGFP reporter ( Figure 1A, right). However, VSV-eGFP-SARS-CoV-2-SAA propagation was inefficient on Vero CCL81 cells. This result prompted us to test additional modifications of the cytoplasmic tail of S, which were also defective in autonomous amplification (Figure S1B). To overcome this limitation, we used a forward genetic approach to isolate two adaptive variants of VSV-eGFP-SARS-CoV-2-SAA ( Figure S1C). Virus was plaque-purified from the transfected cell supernatants, and one variant was passaged twice on Vero CCL81 cells. Following subsequent plaque isolation and serial amplification, we sequenced the viral RNA in infected cells at the seventh passage. A second, independent plaque from transfected cell supernatants was passaged an additional five times on a rhesus monkey MA104 cell line. Both approaches led to the emergence of a virus that contained a single mutation, a cysteine to stop mutation at residue 1253 (TGC to TGA at nucleotide 3759), which truncates the cytoplasmic tail of SARS-CoV-2 S by 21 residues (Figure 1A). This virus, hereafter referred to as VSV-SARS-CoV-2-SΔ21, was passed 12 times in total to assess genetic stability by next generation sequencing, which revealed no additional mutations in the spike (SRA: SRR11878607; BioProject: PRJNA635934). Comparison of plaque morphology of VSV-SARS-CoV-2-SΔ21 and VSV-eGFP-SARS-CoV-2-SAA on three Vero cell subtypes and MA104 cells demonstrates that the selected variant spreads more efficiently (Figure 1B). Screening of a larger panel of cell types (Figure 1C) identified MA104 and Vero E6 cells as supporting the highest levels of virus production. Ectopic expression of TMPRSS2 led to a further ∼10-fold increase in viral titer and larger plaque size (Figure 1D). VSV-SARS-CoV-2-SΔ21 also was capable of infecting Calu-3 cells, a human epithelial lung adenocarcinoma cell line (Figure S2).
Figure 1.
Generation and Characterization of an Infectious VSV-SARS-CoV-2 Chimera
(A) A schematic diagram depicting the genomic organization of the VSV recombinants. Shown 3′ to 5′ are the leader region (Le), eGFP, nucleocapsid (N), phosphoprotein (P), matrix (M), glycoprotein (G) or SARS-CoV-2 S, large polymerase (L), and trailer region (Tr). On right, infection of Vero CCL81 cells with supernatant from cells transfected with the eGFP reporter VSV-SARS-CoV-2-SAA. Images were acquired 44 h post-infection (hpi) using a fluorescence microscope, and GFP and transmitted light images were merged using ImageJ. Shown at bottom, the alignment of the cytoplasmic tail of the VSV-SARS-CoV-2-SAA and the sequence resulting from forward genetic selection of a mutant, which truncated the cytoplasmic tail by 21 amino acids. Mutations deviating from the wild-type spike are indicated in red, and an asterisk signifies a mutation to a stop codon.
(B) Plaque assays were performed to compare the spread of VSV-SARS-CoV-2-SAA rescue supernatant and VSV-SARS-CoV-2-SΔ21 on Vero CCL81, Vero E6, Vero-furin, and MA104 cells. Plates were scanned on a biomolecular imager and expression of eGFP is shown 92 hpi (representative images are shown; n > 3 except for SAA on Vero E6, Vero-furin, and MA104 cells).
(C) The indicated cell types were infected with VSV-SARS-CoV-2-SΔ21 at an MOI of 0.5. Cells and supernatants were harvested at 24 hpi and titrated on MA104 cells (data are pooled from three or more independent experiments; error bars indicate standard deviation of the mean.
(D Top: the indicated cells were infected with VSV-SARS-CoV-2-SΔ21 at an MOI of 2. Images were acquired 7.5 hpi using a fluorescence microscope and GFP, and transmitted light images were processed and merged using ImageJ (data are representative of two independent experiments). Bottom: Plaque assays were performed on the indicated cell types using VSV-SARS-CoV-2-SΔ21. Images showing GFP expression were acquired 48 hpi using a biomolecular imager (data are representative of at least three independent experiments; standard deviations of the mean are shown).
(E) Western blotting was performed on concentrated VSV-SARS-CoV-2-SΔ21 and wild-type VSV particles on an 8% non-reducing SDS-PAGE gel. S1 was detected using a cross-reactive anti-SARS-CoV mAb (CR3022) (data are representative of two independent experiments).
(F) BSRT7/5 cells were inoculated at an MOI of 10 with VSV-eGFP, G-complemented VSV-SARS-CoV-2-SΔ21, or mock infected (not shown), and were metabolically labeled with [35S] methionine and cysteine for 20 h starting at 5 hpi in the presence of actinomycin D. Viral supernatants were analyzed by SDS-PAGE. A representative phosphor-image is shown from two independent experiments. An asterisk indicates a band that also was detected in the mock lane (not shown).
(G) Purified VSV-WT and VSV-SARS-CoV-2-SΔ21 particles were subjected to negative stain electron microscopy; scale bars are equivalent to 100 nm. Prefusion structures of each respective glycoprotein are modeled above each EM image (PDB: 5I2S and 6VSB). See also Figures S1 and S2.
SARS-CoV-2-SΔ21 Is Incorporated into Infectious VSV Particles
To confirm incorporation of SARS-CoV-2 S into particles, we first amplified the virus in the presence of VSV G to allow infection of cell types independently of the S protein. The VSV G trans-complemented VSV-SARS-CoV-2-SΔ21 efficiently infects HEK293T cells, which then serve as a source of production of virus particles containing SARS-CoV-2 S protein. Western blotting of supernatants with CR3022, a cross-reactive anti-S monoclonal antibody (mAb) (ter Meulen et al., 2006; Yuan et al., 2020), established the presence of SΔ21 in VSV-SARS-CoV-2-SΔ21 particles, but not in the parental VSV (Figure 1E). The protein detected migrated at ∼100 kilodaltons, a band that corresponds to the cleaved S1 subunit of the glycoprotein (Watanabe et al., 2020). To examine whether the SΔ21 incorporated into VSV particles is processed to S1 and S2, we performed [35S] cysteine-methionine metabolic labeling in BSRT7 cells, which support robust VSV replication, and analyzed released particles by SDS-PAGE and phosphorimaging. In addition to the VSV structural proteins (N, P, M, and L), two additional bands that correspond in size to glycosylated S1 (107 kDa) and S2Δ21 (85 kDa) were observed for VSV-SARS-CoV-2-SΔ21 (Figure 1F). Negative-stain electron microscopy of sucrose-gradient purified virus particles revealed that the membrane protein projecting from VSV-SARS-CoV-2-SΔ21 is larger than observed on wild-type VSV particles (Figure 1G), which reflects the larger size of the coronavirus spike.
A High-Throughput Focus-Forming Assay with a Clinical Isolate of SARS-CoV-2
VSV-SARS-CoV-2-SΔ21 has several advantages for detection and measuring of neutralizing antibodies, including lower biosafety containment level, ease of production and use, and rapid reporter gene readout. Nonetheless, the difference in virus morphology (spherical CoV versus bullet-shaped VSV) and possible effects on the conformational display of S on the virion surface raise questions of whether the accessibility of epitopes and stoichiometry of antibody neutralization is similar to authentic SARS-CoV-2. A direct comparison with a clinical isolate of SARS-CoV-2 is necessary to establish the utility of VSV-SARS-CoV-2-SΔ21 for assays of viral entry and antibody neutralization.
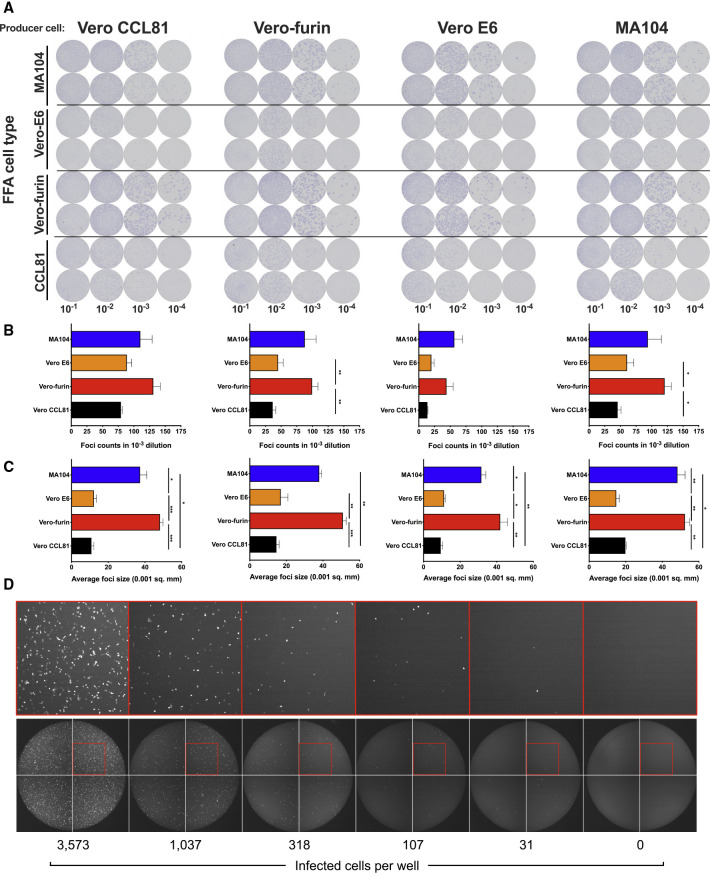
We designed a high-throughput assay for titrating SARS-CoV-2 that could be applied to multiple cell substrates. Instead of using a plaque assay—which relies on the capacity for a virus to cause cell death, which can vary across cell types—we developed a focus-forming assay (FFA) and viral antigen detection as a measure of infectivity. We propagated SARS-CoV-2 in four different producer cell types (Vero CCL81, Vero E6, Vero-furin, and MA104 cells) and then measured the number and size of foci after staining recipient cells with an anti-S mAb. With SARS-CoV-2 stocks generated from each producer cell type, we observed distinct foci across recipient cell substrates at approximately 30 h post-inoculation (Figure 2 A). We consistently observed the highest viral titers and largest foci sizes with Vero-furin and MA104 cells (Figures 2B and 2C). However, the larger foci were more difficult to enumerate on an automated Immunospot reader and required additional manual quality control analysis. Because of this, we used Vero E6 cells for our rapid focus-reduction neutralization tests (FRNT) in subsequent experiments.
Figure 2.
Development of a SARS-CoV-2 Focus-Forming Assay and a VSV-SARS-CoV-2-SΔ21 eGFP-Reduction Assay
(A–C) Representative focus forming assay images (A) of viral stocks generated from each producer cell type (top) were developed on the indicated cell substrates (indicated on the left side). Data are representative of two independent experiments. Foci obtained in (A) were counted (B) and the size was determined (C) using an ImmunoSpot plate reader (∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001 by one-way ANOVA with Tukey’s multiple comparisons test; error bars indicate standard error of the mean).
(D) Representative serial dilution series of VSV-SARS-CoV-2-SΔ21 on Vero E6 cells. The total number of infected cells per well was quantified using an automated microscope. Insets of enhanced magnification are shown in red. Data are representative of two independent experiments.
A High-Throughput, eGFP-Based Neutralization Assay for VSV-SARS-CoV-2-SΔ21
In parallel, we developed a high-throughput method to measure neutralization of VSV-SARS-CoV-2-SΔ21. As VSV-SARS-CoV-2-SΔ21 encodes an eGFP reporter and viral gene expression is robust, eGFP-positive cells can be quantified 7.5 h post-infection using a fluorescence microscope with automated counting analysis software. This approach enabled the development of an eGFP-reduction neutralization test (GRNT) (Figure 2D).
Neutralization of VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 by Human Antibodies
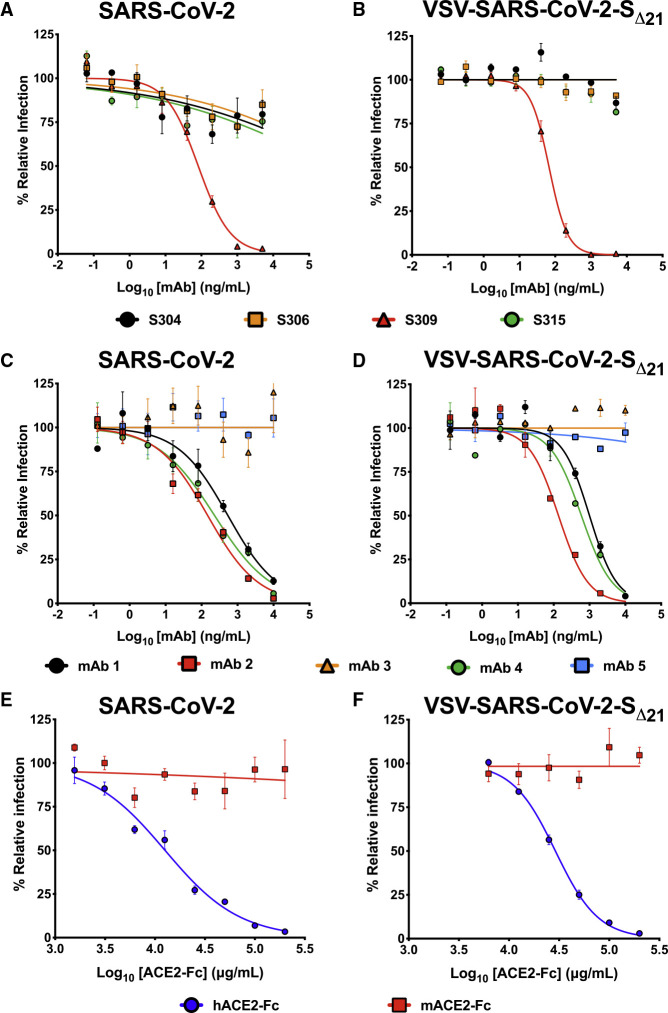
Members of our group recently identified human mAbs from memory B cells of a SARS-CoV survivor that bind to SARS-CoV-2 S (Pinto et al., 2020). We tested a subset of these (mAbs 304, 306, 309, and 315) for their ability to inhibit VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 infections on Vero E6 cells. While three of these mAbs showed poor inhibitory activity, mAb 309 potently neutralized both SARS-CoV-2 and VSV-SARS-CoV-2-SΔ21 (Figures 3A and 3B) with similar EC50 values between the two assays (81 and 67 ng/mL for SARS-CoV-2 and VSV-SARS-CoV-2-SΔ21, respectively). To broaden the test panel, we evaluated the activity of a panel of mAbs generated as part of a phage display library (Miersch et al., 2020) by both FRNT and GRNT. Many of these mAbs exhibited moderate neutralization activities in the EC50 range of 100 to 500 ng/mL (Figures 3C and 3D). Nonetheless, we observed the same neutralization trend between VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 with highly correlated EC50 values (<2-fold differences).
Figure 3.
Neutralization of VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 by Human Monoclonal Antibodies and hACE2 Decoy Receptors
(A and B) Cross-reactive mAbs isolated from a SARS-CoV survivor were tested for neutralizing activity against SARS-CoV-2 (A) or VSV-SARS-CoV-2-SΔ21 (B) (n = 2 and 3, respectively).
(C and D) SARS-CoV-2 RBD-specific antibodies obtained from a phage library were tested for their capacity to neutralize SARS-CoV-2 (C) or VSV-SARS-CoV-2-SΔ21 (D) (n = 2 and 2, respectively).
(E and F) hACE2-Fc or mACE2-Fc were tested for their neutralization activity against SARS-CoV-2 (E) or VSV-SARS-CoV-2-SΔ21 (F) (n = 2 and 3, respectively).
Error bars in (A)–(F) represent the standard error of the mean. See also Figure S3.
Neutralization by Human ACE2-Fc Receptor Decoy Proteins
Human ACE2 (hACE2) is an entry receptor for both SARS-CoV and SARS-CoV-2 (Letko et al., 2020; Li et al., 2003, 2005; Wrapp et al., 2020). As a soluble hACE2-Fc decoy protein has been proposed as a therapeutic for SARS-CoV-2 (Kruse, 2020), in part based on its ability to inhibit SARS-CoV infection in cell culture (Moore et al., 2004), we tested whether hACE2-Fc could inhibit infection of VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 using our FRNT and GRNT assays. When pre-mixed with VSV-SARS-CoV-2-SΔ21 or SARS-CoV-2, hACE2-Fc, but not murine ACE2-Fc (mACE2-Fc), it dose dependently and equivalently inhibited infection of recipient Vero E6 cells (Figures 3E and 3F). As expected, hACE2-Fc did not inhibit infection of wild-type VSV, confirming that neutralization was specific to the SARS-CoV-2 S protein (Figure S3). We noted that a relatively high concentration of hACE2-Fc was required for inhibition, with EC50 values of 29 and 12.6 μg/mL for VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2, respectively. Thus, although soluble hACE2-Fc decoy proteins similarly inhibit infection of VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2, the potency is less than anticipated, which suggests that the receptor-binding domain (RBD) on the S protein on the surface of both viruses may not be fully accessible in solution.
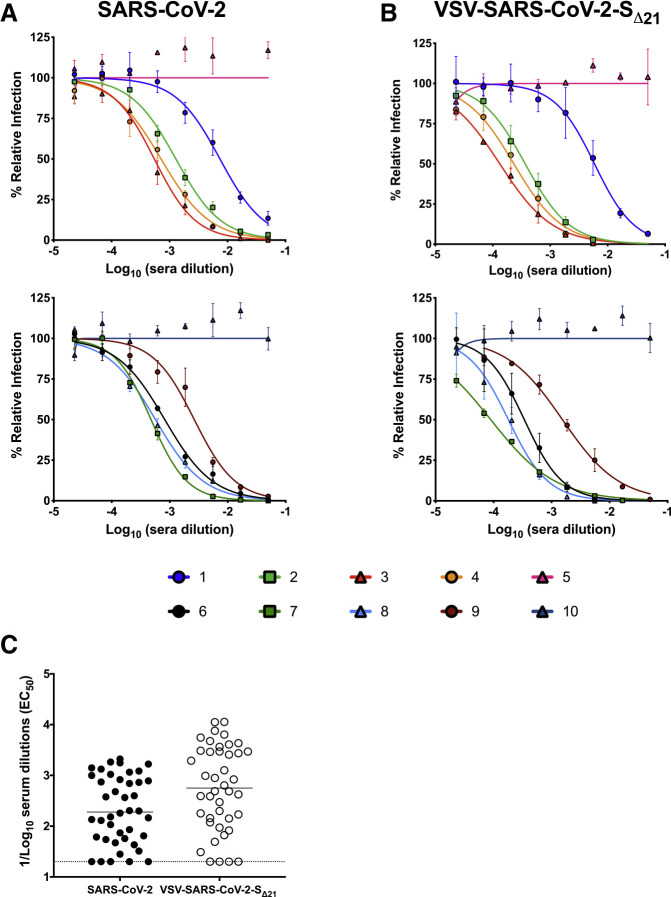
Neutralization of VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 by Human Immune Serum
As part of studies to evaluate immune convalescent plasma as a possible therapy for SARS-CoV-2-infected patients (Bloch et al., 2020), we obtained 42 serum samples from 20 individuals at different time points after the onset of COVID-19 symptoms (Table 1 ). These samples were pre-screened using a commercially available IgG ELISA. We tested each sample for neutralization of VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 on Vero E6 cells. We observed that sera with ELISA-negative or indeterminate results generally showed low inhibitory titers (EC50 < 1/100), whereas ELISA-positive sera generated a broad range of neutralizing antibody activity (EC50 > 1/200 to > 1/1,900) (Figures 4 A and S4). Remarkably, neutralization of VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 was similar across the entire panel of samples (Figures 4B, 4C, and S4).
Table 1.
Human Serum ELISA IgG
| Serum | Days Post Symptom Onset | Euroimmun IgG |
Epitope IgG |
||
|---|---|---|---|---|---|
| Index | Reactive | Index | Reactive | ||
| 1 | 14 | 1.3 | + | 2.6 | + |
| 2 | 12 | 6.0 | + | 3.5 | + |
| 3 | 17 | 10.2 | + | 3.9 | + |
| 4 | 16 | 14.9 | + | 4.5 | + |
| 5 | 5 | 0.2 | − | 1.2 | + |
| 6 | 19 | 8.6 | + | 4.4 | + |
| 7 | 17 | 7.1 | + | 4.1 | + |
| 8 | 10 | 5.1 | + | 2.0 | + |
| 9 | 14 | 6.8 | + | 3.1 | + |
| 10 | 6 | 0.2 | − | 1.1 | + |
| 11 | - | 5.6 | + | 0.9 | +/− |
| 12 | - | <0.8 | − | 1.3 | + |
| 13 | 9 | 0.7 | − | 1.1 | + |
| 14 | 20 | 3.6 | + | 2.9 | + |
| 15 | 13 | 0.4 | − | 2.0 | + |
| 16 | 13 | 0.5 | − | 0.9 | − |
| 17 | 11 | 0.3 | − | 0.8 | − |
| 18 | 10 | 0.2 | − | 0.7 | − |
| 19 | 14 | 0.9 | +/− | 1.2 | + |
| 20 | 10 | 0.4 | − | 1.5 | + |
| 21 | 11 | 0.5 | − | 2.0 | + |
| 22 | 10 | 0.3 | − | 0.6 | − |
| 23 | 17 | 7.6 | + | 4.2 | + |
| 24 | 14 | 3.5 | + | 3.3 | + |
| 25 | 13 | 1.5 | + | 2.9 | + |
| 26 | 17 | 14.2 | + | 4.4 | + |
| 27 | 13 | 0.5 | − | 1.9 | + |
| 28 | 14 | 9.2 | + | 4.6 | + |
| 29 | 13 | 3.9 | + | 2.8 | + |
| 30 | 16 | 3.4 | + | 4.6 | + |
| 31 | 15 | 10.7 | + | 3.6 | + |
| 32 | 6 | 0.5 | − | 1.1 | + |
| 33 | 11 | 0.4 | − | 1.8 | + |
| 34 | 12 | 0.6 | − | 2.5 | + |
| 35 | 14 | 3.7 | + | 4.1 | + |
| 36 | 20 | 11.6 | + | 4.2 | + |
| 37 | 7 | 0.7 | − | 1.2 | + |
| 38 | 8 | 1.7 | + | 1.9 | + |
| 39 | 7 | 0.3 | − | 0.7 | − |
| 40 | 9 | 3.4 | + | 2.2 | + |
| 41 | 18 | 10.3 | + | 4.1 | + |
| 42 | 17 | 10.3 | + | 4.6 | + |
Serum samples from 20 individuals were collected at different time points post onset of COVID-19 symptoms and screened using two ELISA assays (Euroimmun or Epitope). The serum numbers correspond to those of Figures 4 and S3. IgG index values were calculated by dividing the O.D. of the serum sample by a reference O.D. control, and ratios were interpreted using the following criteria as recommended by the manufacturer: Negative (−) < 0.8, Indeterminate (+/−) 0.8–1.1, Positive (+) ≥ 1.1.
Figure 4.
Human Immune Serum Neutralization of SARS-CoV-2 and VSV-SARS-CoV-2-SΔ21
(A and B) Representative neutralization curves of serum from SARS-CoV-2-infected donors with low, medium, and high inhibitory activity against SARS-CoV-2 (A) or VSV-SARS-CoV-2-SΔ21 (B) (n = 2 and 2, respectively). Error bars in (A) and (B) represent the standard error of the mean.
(C) EC50 values of all human serum tested for neutralization of SARS-CoV-2 and VSV-SARS-CoV-2-SΔ21. Differences in the geometric mean or median titers were 3.0-fold between FRNT and GRNT assays. See also Figure S4.
VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 Neutralization Assays Are Highly Correlative
We determined the extent to which the VSV-SARS-CoV-2-SΔ21 and SARS-CoV-2 neutralization tests correlated with each other. We compared the GRNT and FRNT EC50 values obtained in assays with mAbs, polyclonal sera, and soluble ACE2 protein (Figure 5 ). For the samples with neutralizing activity, we observed a remarkably strong correlation between the two assays (r = 0.9285; p < 0.001). Moreover, all 11 of the samples that were deemed non-neutralizing in one assay had the same result in the second assay. Together, these results establish the utility of using VSV-SARS-CoV-2-SΔ21 as a surrogate for authentic SARS-CoV-2 infection in entry inhibition and neutralization studies.
Figure 5.
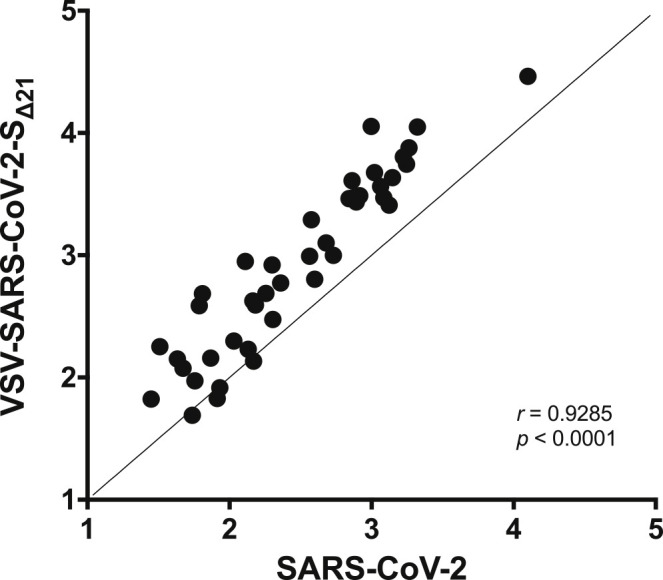
Correlation Analysis of Neutralization of SARS-CoV-2 and VSV-SARS-CoV-2-SΔ21
EC50 values determined in Figures 3A–3D and 4A–4B were used to determine correlation between neutralization assays. Spearman’s correlation r and p values are indicated.
Discussion
Emerging viral pathogens have caused numerous epidemics and several pandemics over the last century. The most recent example, SARS-CoV-2, has spread to nearly every country in the world in just a few months, causing millions of infections and hundreds of thousands of deaths (https://www.worldometers.info/coronavirus/). Rapid responses to viral outbreaks and generation of countermeasures require readily accessible tools to facilitate study and evaluate antiviral activity. Here, we generated a high-titer, replication-competent chimeric VSV expressing the SARS-CoV-2 S protein that performs similarly to a SARS-CoV-2 clinical isolate across multiple neutralization tests. As access to BSL3 facilities is limited, the finding that VSV-SARS-CoV-2-SΔ21 is neutralized similarly by decoy receptors, mAbs, and polyclonal antibodies in comparison to authentic SARS-CoV-2 is important. This tool will enable academic, government, and industry investigators to rapidly perform assays that interrogate SARS-CoV-2 entry, neutralization, and inhibition at a BSL2 level, which should simplify and expedite the discovery of therapeutic interventions and analysis of functional humoral immune responses.
Upon recovery of VSV-SARS-CoV-2-SAA, we selected for a mutant, which contained a 21-amino acid deletion in the cytoplasmic tail. As truncation of the cytoplasmic tail eliminates the modified KxHxx ER retention signal, we suggest that this mutation facilitates more efficient incorporation of the SARS-CoV-2 S protein into the VSV particles. Although truncation of the cytoplasmic tail of HIV envelope protein resulted in conformational alterations in the ectodomain of the protein (Chen et al., 2015), based on the extensive neutralization data presented here, including correlation to neutralization of a clinical isolate of SARS CoV-2, a 21-amino acid truncation does not appear to substantively alter the structure of the S protein ectodomain. It remains to be determined whether fully wild-type S protein can incorporate efficiently into VSV. Indeed, similar mutations were generated in the SARS-CoV S protein cytoplasmic tail to boost incorporation into retroviruses and VSV pseudotypes (Fukushi et al., 2005; Giroglou et al., 2004; Moore et al., 2004).
The value of a chimeric virus depends on its capacity to present viral surface antigens in a similar way to its authentic counterpart (Garbutt et al., 2004). Indeed, the morphology of the bullet-shaped rhabdovirus and the spherical coronavirus and the density and geometry of S protein display could differentially impact antibody engagement and neutralization. Despite this concern, our extensive testing of VSV-SARS-CoV-2-SΔ21 with antibodies and soluble ACE2-Fc proteins showed similar neutralization profiles compared to authentic, fully infectious SARS-CoV-2. Thus, VSV-SARS-CoV-2-SΔ21, despite the structural differences of the virion, provides a useful tool for screening antibodies, entry-based antiviral agents, and vaccine responses against SARS-CoV-2. Indeed, convalescent plasma is under investigation as a potential COVID-19 therapeutic (Chen et al., 2020). Our studies suggest that, in addition to testing for anti-S or anti-RBD antibodies (Shen et al., 2020), neutralization assays with VSV-SARS-CoV-2-SΔ21 may be a convenient and rapid method to obtain functional information about immune plasma preparations to enable prioritization prior to passive transfer to COVID-19 patients.
Coronaviruses possess a roughly 30 kb RNA genome, which requires that they encode a proofreading enzyme (ExoN in nsp14) (Denison et al., 2011) to counteract the error rate of the viral-RNA-dependent RNA polymerase. The lack of such proofreading enzymes in the genomes of rhabdoviruses suggests that selection of escape mutants to inhibitors of the coronavirus S protein will be faster in VSV-SARS-CoV-2, which further increases the utility of this chimeric virus. Our FRNT and GRNT assays can be used to establish evidence of prior SARS-CoV-2 infection or vaccination, as well as determine waning of functional responses over time, as the likelihood of cross-neutralizing responses with other cosmopolitan coronaviruses (e.g., HCoV-229E and HCoV-OC43) is exceedingly low. Overall, VSV-SARS-CoV-2-SΔ21 and our FRNT and GRNT assays can facilitate the development and evaluation of antibody- or entry-based countermeasures against SARS-CoV-2 infection. A similar VSV-SARS-CoV-2 chimera as a reporter of antibody mediated neutralization is described in a companion paper (Dieterle et al., 2020).
Limitations of Study
Our experiments do not directly test the safety of the recombinant VSV-eGFP-SARS-CoV-2. VSV-Indiana and chimeric VSV in which the native envelope protein is replaced with that of the envelope proteins of Ebola (Garbutt et al., 2004; Jones et al., 2005; Takada et al., 2003), Lassa (Geisbert et al., 2005), Andes (Brown et al., 2011), highly pathogenic avian influenza (Furuyama et al., 2020), and many other viruses are handled at BSL2. VSV-Ebola is a licensed human vaccine, ERVEBO, distributed by Merck. We are actively evaluating VSV-SARS-CoV-2 as a vaccine candidate, and mice inoculated with this virus do not develop disease (J.B.C., P.W.R., S.P.J.W., and M.S.D., unpublished data). Notwithstanding these data, a chimeric VSV containing both the F and G genes of Nipah virus remains pathogenic in mice (van den Pol et al., 2017), suggesting that appropriate caution should be used in handling VSV-SARS-CoV-2 at BSL2.
STAR★Methods
Key Resources Table
| REAGENT or RESOURCE | SOURCE | IDENTIFIER |
|---|---|---|
| Antibodies | ||
| Anti-human IgG peroxidase | Sigma-Aldrich | Cat# A6029-1ML; RRID: AB_258272 |
| CR3022 | ter Meulen et al., 2006; Yuan et al., 2020 | N/A |
| Goat anti-human IgG - H&L (HRP) | Abcam | Cat# ab6858-1MG; RRID: AB_955433 |
| Phage display antibody set | Miersch et al., Bioraxiv https://doi.org/10.1101/2020.06.05.137349 | N/A |
| VIR antibody set | Pinto et al., 2020 | S309 PDB: 6WS6 |
| Bacterial and Virus Strains | ||
| 2019 n-CoV/USA_WA1/2020 | CDC (gift from Natalie Thornburg) | N/A |
| Vaccinia virus vTF7-3 | Fuerst et al., 1986 | N/A |
| VSV-eGFP | Chandran et al., 2005 | N/A |
| Biological Samples | ||
| Patient serum set | This study | N/A |
| Chemicals, Peptides, and Recombinant Proteins | ||
| Blasticidin S HCl | GIBCO/Thermo Fisher | Cat# A1113903 |
| Cytosine arabinoside | Sigma-Aldrich | Cat# C1768 |
| Formaldehyde Solution | Millipore Sigma | Cat# FX0410-5 |
| HEPES, free acid | Millipore Sigma | Cat #5310-OP |
| Hoechst 33342 | Invitrogen/Thermo Fisher | Cat# H3570 |
| Human ACE2-Fc | This study | GenBank: BAB40370.1 & AAC82527.1 |
| L-[35S]-cysteine | Perkin Elmer | Cat# NEG022T |
| L-[35S]-methionine | Perkin Elmer | Cat# NEG009T |
| Lipofectamine 2000 Transfection Reagent | Invitrogen/Thermo Fisher | Cat# 11668019 |
| Mouse ACE2-Fc | This study | NCBI Reference Sequence: NP_001123985.1 |
| Phosphotungstic acid hydrate | Sigma-Aldrich | Cat# 79690-25G |
| Pierce ECL Western Blotting Substrate | Thermo Scientific/Thermo Fisher | Cat# 32106 |
| Protein A Agarose Resin | GoldBio | Cat# P-400-100 |
| TRIzol Reagent | Invitrogen/Thermo Fisher | Cat# 15596018 |
| TrueBlue peroxidase substrate | KPL/SeraCare | Cat# 5510-0050 |
| Critical Commercial Assays | ||
| Anti-SARS-CoV-2 ELISA (IgG) serology test | Euroimmun US, Inc., 1 Bloomfield Ave, Mountain Lakes, NJ 07046 | N/A |
| Deposited Data | ||
| Nucleotide sequence of VSV-eGFP-SARS-CoV-2-SΔ21 | This study | BioProject: PRJNA635934; SRA: SRR11878607 |
| Experimental Models: Cell Lines | ||
| A549 | Gift from Jan Carette | N/A |
| BHK-21 | ATCC | Cat# CCL-10 |
| BSRT7/5 | Buchholz et al., 1999 | N/A |
| Caco-2 | ATCC | Cat# HTB-37 |
| Caco-2 BBe1 | ATCC | Cat# CRL-2102 |
| Calu-3 | Gift from Stanley Perlman | N/A |
| Expi293F | Thermo Fisher | Cat# A14527 |
| ExpiCHO | GIBCO/Thermo Fisher | Cat# A29127 |
| HEK293 | ATCC | Cat# CRL-1573 |
| HEK293T | ATCC | Cat# CRL-3216 |
| H1Hela | Gift from Jan Carette | N/A |
| HepG2 | ATCC | Cat# HB-8065 |
| HT-29 | ATCC | Cat# HTB-38 |
| Huh7.5.1 | Gift from Harry Greenberg | |
| MA104 | Gift from Harry Greenberg | N/A |
| Vero CCL81 | ATCC | Cat# CCL-81 |
| Vero E6 | ATCC | Cat# CRL-1586 |
| Vero E6-TMPRSS2 | This paper | N/A |
| Vero-furin | Mukherjee et al., 2016 | N/A |
| Experimental Models: Organisms/Strains | ||
| N/A | N/A | N/A |
| Oligonucleotides | ||
| TruSeq RNA Single Indexes Set A | Illumina | Cat# 20020492 |
| TruSeq RNA Single Indexes Set B | Illumina | Cat# 20020493 |
| Recombinant DNA | ||
| pCAGGS-VSV-G | Stanifer et al., 2011 | N/A |
| pFM1.2-hACE2-Fc | This paper | GenBank: AB046569.1 |
| pFM1.2-mACE2-Fc | This paper | NCBI Reference Sequence: NM_001130513.1 |
| pGEM3-VSV L | Whelan et al., 1995 | N/A |
| pGEM3-VSV G | Stanifer et al., 2011 | N/A |
| pGEM3-VSV N | Whelan et al., 1995 | N/A |
| pGEM3-VSV P | Whelan et al., 1995 | N/A |
| pLX304-TMPRSS2 | Zang et al., 2020 | N/A |
| psPAX2 | Gift from Didier Trono (unpublished data) | Addgene Cat# 12260 |
| pVSV-eGFP | Chandran et al., 2005 | N/A |
| pVSV-eGFP-SARS-CoV-2-SAA | This study | Spike mutated from MN908947.3 |
| pVSV-eGFP-SARS-CoV-2-S MERS SAA cytoplasmic tail switch | This paper | Spike mutated from a codon-optimized version of AHI48572.1 |
| pVSV-eGFP-SARS-CoV-2-S VSV G #1 cytoplasmic tail switch | This paper | VSV Indiana G (strain Orsay) |
| pVSV-eGFP-SARS-CoV-2-S VSV G #2 cytoplasmic tail switch | This paper | VSV Indiana G (strain Orsay) |
| pVSV-eGFP-SARS-CoV-2-S VSV G Ecto/TM/tail switch | This paper | VSV Indiana G (strain Orsay) |
| pVSV-eGFP-SARS-CoV-2-S VSV G TM/tail switch | This paper | VSV Indiana G (strain Orsay) |
| Software and Algorithms | ||
| BBMap 38.79 | Bushnell et al., 2017 | https://jgi.doe.gov/data-and-tools/bbtools/ |
| Fastp 0.20.0 | Chen et al., 2018 | https://github.com/OpenGene/fastp |
| Image Analyses: GE InCell Analyzer 1000 Workstation | GE Life Sciences (now Cytiva) | N/A |
| NUCmer 3.1 | Delcher et al., 2002 | http://mummer.sourceforge.net/ |
| SAMtools 1.9 | Li et al., 2009 | http://www.htslib.org/ |
| SPAdes 3.13.0 | Bankevich et al., 2012 | http://cab.spbu.ru/software/spades/ |
| Statistics: Prism 8.0 | GraphPad | N/A |
| Other | ||
| TruSeq Stranded Total RNA Library Prep Kit with Ribo-Zero Human/Mouse/Rat | Illumina | Cat #20020596 |
Resource Availability
Lead Contact
Further information and requests for resources and reagents should be directed to and will be fulfilled by the Lead Contact author Sean P.J. Whelan (spjwhelan@wustl.edu).
Materials Availability
All plasmids, antibodies, cells, viruses, and cell lines developed are available under Material Transfer Agreements from Washington University School of Medicine.
Data and Code Availability
The authors declare that all data supporting the findings of this study are available within the paper. The sequence of VSV-eGFP-SARS-CoV-2-SΔ21 (passage 12) generated during this study is available through NCBI (SRA: SRR11878607, https://www.ncbi.nlm.nih.gov/sra/?term=SRR11878607; BioProject: PRJNA635934).
Experimental Model and Subject Details
Cells
Cells were maintained in humidified incubators at 34 or 37°C and 5% CO2 in the indicated media. BSRT7/5, Vero CCL81, Vero E6, Vero E6-TMPRSS2, A549, Caco-2, Caco-2 BBe1, Calu-3, Huh7.5.1, HepG2, H1Hela, BHK-21, HEK293, and HEK293T were maintained in DMEM (Corning or VWR) supplemented with glucose, L-glutamine, sodium pyruvate, and 10% fetal bovine serum (FBS). Vero-furin cells (Mukherjee et al., 2016) also were supplemented with 5 μg/mL of Blasticidin S HCl (GIBCO). MA104 cells were maintained in Medium 199 (GIBCO) containing 10% FBS. HT-29 cells were cultured in complete DMEM/F12 (Thermo Fisher) supplemented with sodium pyruvate, non-essential amino acids, and HEPES (Millipore Sigma). Vero E6-TMPRSS2 cells were generated using a lentivirus vector. Briefly, HEK293T producer cells were transfected with pLX304-TMPRSS2, pCAGGS-VSV-G, and psPAX2, and cell culture supernatants were collected at 48 h and clarified by centrifugation at 1,000 x g for 5 min. The resulting lentivirus was used to infect Vero E6 cells for 24 h, and cells were selected with 40 μg/mL Blasticidin S HCl for 7 days.
Recombinant VSV
Recovery of recombinant VSV was performed as described (Whelan et al., 1995). Briefly, BSRT7/5 cells (Buchholz et al., 1999) were inoculated with vaccinia virus vTF7-3 (Fuerst et al., 1986) and subsequently transfected with T7-expression plasmids encoding VSV N, P, L, and G, and an antigenomic copy of the viral genome. Cell culture supernatants were collected at 56-72 h, clarified by centrifugation (5 min at 1,000 x g), and filtered through a 0.22 μm filter. Virus was plaque-purified on Vero CCL81 cells in the presence of 25 μg/mL of cytosine arabinoside (Sigma-Aldrich), and plaques in agarose plugs were amplified on Vero CCL81 cells. Viral stocks were amplified on MA104 cells at an MOI of 0.01 in Medium 199 containing 2% FBS and 20 mM HEPES pH 7.7 at 34°C. Viral supernatants were harvested upon extensive cytopathic effect and clarified of cell debris by centrifugation at 1,000 x g for 5 min. Aliquots were maintained at −80°C. Construction and use of VSV-SARS-CoV-2 was approved by the Washington University School of Medicine Institutional Biosafety Committee at Biosafety level 2.
SARS-CoV-2
SARS-CoV-2 strain 2019 n-CoV/USA_WA1/2020 was obtained from the Centers for Disease Control and Prevention (gift of Natalie Thornburg). Virus was passaged in the indicated producer cells (Figures 2A–2C). Work with SARS-CoV-2 was approved by the Washington University School of Medicine Institutional Biosafety Committee at Biosafety level 3 with positive pressure respirators.
Method Details
Plasmids
The S gene of SARS-CoV-2 isolate Wuhan-Hu-1 (GenBank MN908947.3) was synthesized in two fragments (Integrated DNA Technologies) and inserted into an infectious molecular clone of VSV (Chandran et al., 2005; Whelan et al., 1995) as previously (Carette et al., 2011; Jae et al., 2014). Modifications to the cytoplasmic tail were assembled identically, and accession numbers or references to the amino acid sequences used for the MERS and VSV G tail mutants can be found in the Key Resources Table. Other plasmids were previously described: VSV N, P, L and G expression plasmids (Stanifer et al., 2011; Whelan et al., 1995), psPAX2 (Addgene), and pLX304-TMPRSS2 (Zang et al., 2020).
Next Generation Sequencing
Total RNA was extracted from Vero CCL81 cells infected with VSV-SARS-CoV-2-SΔ21 using TRIzol (Invitrogen) according to the manufacturer’s protocol. RNA was used to generate next generation sequencing libraries using TruSeq Stranded Total RNA library kit with Ribo Zero ribosomal subtraction (Illumina). The libraries were quantified using a bioanalyzer (Agilent) and pooled at an equal molar concentration and used to generate paired end 250 bp reads on a MiSeq (Illumina). Raw sequencing data was processed using fastp v0.20.0 (Chen et al., 2018) to cut adaptor sequences and filter out sequences with a Phred quality score < 30. Processed reads were aligned to the reference plasmid sequence using BBMap v38.79 (Bushnell et al., 2017). Reads that mapped to the reference were extracted using SAMtools 1.9 (Li et al., 2009). The extracted mapped reads were used as input for de novo assembly with SPAdes v3.13.0 (Bankevich et al., 2012). Assembled contigs produced a 14.2 kb consensus sequence, which was subsequently aligned to the reference plasmid using NUCmer v3.1 (Delcher et al., 2002). Consensus sequences for each RNA sample were generated by aligning contigs to the reference plasmid sequence pVSV(1+)-eGFP-SARS-CoV-2-S with SnapGene v5.0.
Western Blotting
Purified VSV virions were incubated in non-reducing denaturation buffer (55 mM Tris-HCl pH 6.8, 1.67% (w/v) SDS, 7.5% (w/v) glycerol) at 100°C for 5 min. Viral proteins were separated on a 8% acrylamide gel, transferred onto a nitrocellulose membrane, and incubated with human anti-SARS antibody CR3022 diluted in Tris-buffered saline containing 1% Tween-20 (TBS-T) and 5% milk, followed by incubation with HRP-conjugated goat anti-human antibody (Abcam) diluted in TBS-T containing 1% milk. HRP activity was visualized using the Pierce ECL western blotting kit (Thermo Scientific) and imaged with a ChemiDocTM MP Imager (Bio-Rad).
Metabolic Radiolabeling of Virions
To generate high titer stocks of VSV-SARS-CoV-2, BSRT7/5 cells were transfected with pCAGGS-VSV-G in Opti-MEM (GIBCO) using Lipofectamine 2000 (Invitrogen) and infected 7 h later with VSV-SARS-CoV-2-SΔ21 at an MOI of 0.1 in DMEM containing 2% FBS and 20 mM HEPES pH 7.7. Viral stocks were collected at 48 hpi, and used to infect fresh cells (MOI of 10) for labeling of viral proteins. At 4 hpi, cells were starved in serum free, methionine/cysteine free DMEM (Corning), and exposed to 15 μCi/mL [35S]-methionine and [35S]-cysteine (Perkin Elmer) from 5-24 h in the presence of actinomycin D. Cell culture supernatants were collected, clarified by centrifugation (1,500 x g, 5 min), and analyzed by SDS-PAGE and detected by phosphoimage analysis (Li et al., 2006).
Transmission Electron Microscopy
Purified viruses were adhered to glow-discharged, carbon-coated copper grids. Samples were stained with 2% (w/v) phosphotungstic acid (Sigma-Aldrich), pH 7.1, in H2O and viewed on a JEOL 1200 EX transmission electron microscope (JEOL USA Inc.) equipped with an AMT 8-megapixel digital camera and AMT Image Capture Engine V602 software (Advanced Microscopy Techniques).
Monoclonal Antibodies
Phage-displayed Fab libraries were panned against immobilized SARS-CoV-2 spike RBD in multiple rounds using established methods (Persson et al., 2013). Following four rounds of selection, phage ELISAs were used to screen 384 clones to identify those that bound specifically to RBD. The complementarity determining regions of Fab-phage clones were decoded by sequencing the variable regions and cloning them into mammalian expression vectors for expression and purification of human IgG1 proteins, as described (Tao et al., 2019). A subset of the panel of mAbs was tested for neutralization as a part of this study.
Another set of mAbs (S304, S306, S309, S310 and S315) were isolated from EBV-immortalized memory B cells from a SARS-CoV survivor (Traggiai et al., 2004) and are cross-reactive to SARS-CoV-2 (Pinto et al., 2020). Recombinant antibodies were expressed in ExpiCHO cells transiently co-transfected with plasmids expressing the heavy and light chain as previously described (Stettler et al., 2016).
Human Sera
Human samples were collected from PCR-confirmed COVID-19 patients. Serum samples were obtained by routine phlebotomy at different days post symptom onset (range: day 5 - 20). Samples were prescreened by the Euroimmun anti-SARS-CoV-2 IgG ELISA (Lubeck, Germany), a qualitative assay with the Food and Drug Administration Emergency Use Authorization that detects antibodies to the SARS-CoV-2 S protein. This study was approved by the Mayo Clinic Institutional Review Board.
Protein Expression and Purification
DNA fragments encoding human ACE2 (hACE2 residues 1-615) and mouse ACE2 (mACE2, residues 1-615) were synthesized and cloned into pFM1.2 with a C-terminal HRV-3C protease cleavage site (LEVLFQGP) and a human IgG1 Fc region as previously described (Raj et al., 2013). We transiently transfected plasmids into Expi293F cells and harvested cell supernatants 4 days post transfection. Secreted hACE2-Fc and mACE2-Fc proteins were purified by protein A chromatography (GoldBio).
GFP-Reduction Neutralization Test
Patient samples were heat-inactivated at 56°C for 30 min. Indicated dilutions of samples were incubated with 102 PFU of VSV-SARS-CoV-2-SΔ21 for 1 h at 37°C. Antibody-virus complexes were added to Vero E6 cells in 96-well plates and incubated at 37°C for 7.5 h. Cells subsequently were fixed in 2% formaldehyde (Millipore Sigma) containing 10 μg/mL Hoechst 33342 nuclear stain (Invitrogen) for 45 min at room temperature, when fixative was replaced with PBS. Images were acquired with the InCell 2000 Analyzer (GE Healthcare) automated microscope in both the DAPI and FITC channels to visualize nuclei and infected cells (i.e., eGFP-positive cells), respectively (4X objective, 4 fields per well, covering the entire well). Images were analyzed using the Multi Target Analysis Module of the InCell Analyzer 1000 Workstation Software (GE Healthcare). GFP-positive cells were identified in the FITC channel using the top-hat segmentation method and subsequently counted within the InCell Workstation software. The sensitivity and accuracy of GFP-positive cell number determinations were validated using a serial dilution of virus. A background number of GFP+ cells was subtracted from each well using an average value determined from at least 4 uninfected wells. Data were processed using Prism software (GraphPad Prism 8.0). ACE2 neutralization assays using VSV-SARS-CoV-2-SΔ21 were conducted similarly but using an MOI of 1. Infection and imaging of Calu-3 cells was performed using similar methods, though cells were infected at an MA104-calculated MOI of 20. Cells were imaged using a 10X objective, with 9 fields per well. FITC and DAPI fields were overlaid using ImageJ and contrast-adjusted identically.
Focus-Reduction Neutralization Test
Indicated dilutions of mAbs, sera, or protein were incubated with 102 FFU of SARS-CoV-2 for 1 h at 37°C. Antibody-virus complexes were added to indicated cell monolayers in 96-well plates and incubated at 37°C for 1 h. Subsequently, cells were overlaid with 1% (w/v) methylcellulose in MEM supplemented with 2% FBS. Plates were harvested 30 h later by removing overlays and fixed with 4% paraformaldehyde in PBS for 20 min at room temperature. Plates were washed and sequentially incubated with 1 μg/mL of CR3022 anti-S antibody (ter Meulen et al., 2006; Yuan et al., 2020) and HRP-conjugated goat anti-human IgG in PBS supplemented with 0.1% saponin and 0.1% BSA. SARS-CoV-2-infected cell foci were visualized using TrueBlue peroxidase substrate (KPL) and quantitated on an ImmunoSpot microanalyzer (Cellular Technologies). Data were processed using Prism software (GraphPad Prism 8.0).
Quantification and Statistical Analysis
All statistical tests were performed using GraphPad Prism 8.0 software as described in the indicated figure legends. Statistical significance was assigned when P values were < 0.05. The number of independent experiments and the statistical tests used are indicated in the relevant Figure legends. Error bars indicate standard error of the mean unless stated otherwise.
Acknowledgments
This study was supported by NIH contracts and grants (75N93019C00062, HHSN272201700060C, R01 AI127828, R37 AI059371, and U01 AI151810), the Defense Advanced Research Project Agency (HR001117S0019), and gifts from Washington University in Saint Louis. J.B.C. is supported by a Helen Hay Whitney Foundation postdoctoral fellowship. We thank Natalie Thornburg for providing the clinical isolate of SARS-CoV-2, James Rini for providing RBD used to detect phage display mAbs, Jan Carette for providing A549 and H1Hela cells, Harry Greenberg for providing Huh7.5.1 and MA104 cells, and Stanley Perlman for providing Calu-3 cells. We also thank Wandy Beatty and the Molecular Microbiology Imaging Facility at the Washington University School of Medicine for taking the electron microscopy pictures. Some of the figures were created using BioRender.com.
Author Contributions
J.B.C. performed SARS-CoV-2 neutralization experiments. P.W.R. generated and characterized VSV-SARS-CoV-2-SΔ21 and performed neutralization experiments. R.E.C., Z.L., L.M.B., M.A.T., S.D., and Q.Z. provided experimental assistance. S.T., L.D., and D.W. prepared RNA-seq libraries and assembled the VSV-SARS-CoV-2-SΔ21 sequence. H.Z. and D.H.F. generated and provided purified proteins; D.C. and H.W.V. provided the recombinant mAbs; S.M., M.U., S.S., and G.A. provided other recombinant mAbs; and E.S.T. and J.P.H. identified and provided the human immune serum. M.X.G.I. helped with automated microscope use and analysis. J.B.C., P.W.R., S.P.J.W., and M.S.D. wrote the initial draft, with the other authors providing editing comments.
Declaration of Interests
M.S.D. is a consultant for Inbios, Eli Lilly, Vir Biotechnology, and NGM Biopharmaceuticals and is on the Scientific Advisory Board of Moderna. The Diamond laboratory has received unrelated funding under sponsored research agreements from Moderna and Emergent BioSolutions. D.C. and H.W.V. are employees of Vir Biotechnology Inc. and may hold shares in Vir Biotechnology Inc. S.P.J.W. and P.W.R. have filed a disclosure with Washington University for the recombinant VSV.
Published: July 3, 2020
Footnotes
Supplemental Information can be found online at https://doi.org/10.1016/j.chom.2020.06.021.
Supplemental Information
References
- Bai Y., Yao L., Wei T., Tian F., Jin D.Y., Chen L., Wang M. Presumed Asymptomatic Carrier Transmission of COVID-19. JAMA. 2020 doi: 10.1001/jama.2020.2565. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bankevich A., Nurk S., Antipov D., Gurevich A.A., Dvorkin M., Kulikov A.S., Lesin V.M., Nikolenko S.I., Pham S., Prjibelski A.D. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comp. Biol. 2012;19:455–477. doi: 10.1089/cmb.2012.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bloch E.M., Shoham S., Casadevall A., Sachais B.S., Shaz B., Winters J.L., van Buskirk C., Grossman B.J., Joyner M., Henderson J.P. Deployment of convalescent plasma for the prevention and treatment of COVID-19. J Clin Invest. 2020;130:2757–2765. doi: 10.1172/JCI138745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown K.S., Safronetz D., Marzi A., Ebihara H., Feldmann H. Vesicular stomatitis virus-based vaccine protects hamsters against lethal challenge with Andes virus. J. Virol. 2011;85:12781–12791. doi: 10.1128/JVI.00794-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchholz U.J., Finke S., Conzelmann K.K. Generation of bovine respiratory syncytial virus (BRSV) from cDNA: BRSV NS2 is not essential for virus replication in tissue culture, and the human RSV leader region acts as a functional BRSV genome promoter. J. Virol. 1999;73:251–259. doi: 10.1128/jvi.73.1.251-259.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bushnell B., Rood J., Singer E. BBMerge - Accurate paired shotgun read merging via overlap. PLoS ONE. 2017;12:e0185056. doi: 10.1371/journal.pone.0185056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carette J.E., Raaben M., Wong A.C., Herbert A.S., Obernosterer G., Mulherkar N., Kuehne A.I., Kranzusch P.J., Griffin A.M., Ruthel G. Ebola virus entry requires the cholesterol transporter Niemann-Pick C1. Nature. 2011;477:340–343. doi: 10.1038/nature10348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chandran K., Sullivan N.J., Felbor U., Whelan S.P., Cunningham J.M. Endosomal proteolysis of the Ebola virus glycoprotein is necessary for infection. Science. 2005;308:1643–1645. doi: 10.1126/science.1110656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen J., Kovacs J.M., Peng H., Rits-Volloch S., Lu J., Park D., Zablowsky E., Seaman M.S., Chen B. HIV-1 ENVELOPE. Effect of the cytoplasmic domain on antigenic characteristics of HIV-1 envelope glycoprotein. Science. 2015;349:191–195. doi: 10.1126/science.aaa9804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen S., Zhou Y., Chen Y., Gu J. fastp: an ultra-fast all-in-one FASTQ preprocessor. Bioinformatics. 2018;34:i884–i890. doi: 10.1093/bioinformatics/bty560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen L., Xiong J., Bao L., Shi Y. Convalescent plasma as a potential therapy for COVID-19. Lancet Infect. Dis. 2020;20:398–400. doi: 10.1016/S1473-3099(20)30141-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Delcher A.L., Phillippy A., Carlton J., Salzberg S.L. Fast algorithms for large-scale genome alignment and comparison. Nucleic Acids Res. 2002;30:2478–2483. doi: 10.1093/nar/30.11.2478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Denison M.R., Graham R.L., Donaldson E.F., Eckerle L.D., Baric R.S. Coronaviruses: an RNA proofreading machine regulates replication fidelity and diversity. RNA Biol. 2011;8:270–279. doi: 10.4161/rna.8.2.15013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dieterle M.E., Haslwanter D., Bortz R.H., Wirchnianski A.S., Lasso G., Vergnolle O., Abbasi S.A., Fels J.M., Laudermilch E., Florez C. A replication-competent vesicular stomatitis virus for studies of SARS-CoV-2 spike-mediated cell entry and its inhibitionCell. Host & Microbe. 2020;28 doi: 10.1016/j.chom.2020.06.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuerst T.R., Niles E.G., Studier F.W., Moss B. Eukaryotic transient-expression system based on recombinant vaccinia virus that synthesizes bacteriophage T7 RNA polymerase. Proc. Natl. Acad. Sci. USA. 1986;83:8122–8126. doi: 10.1073/pnas.83.21.8122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fukushi S., Mizutani T., Saijo M., Matsuyama S., Miyajima N., Taguchi F., Itamura S., Kurane I., Morikawa S. Vesicular stomatitis virus pseudotyped with severe acute respiratory syndrome coronavirus spike protein. J. Gen. Virol. 2005;86:2269–2274. doi: 10.1099/vir.0.80955-0. [DOI] [PubMed] [Google Scholar]
- Fukushi S., Mizutani T., Saijo M., Kurane I., Taguchi F., Tashiro M., Morikawa S. Evaluation of a novel vesicular stomatitis virus pseudotype-based assay for detection of neutralizing antibody responses to SARS-CoV. J. Med. Virol. 2006;78:1509–1512. doi: 10.1002/jmv.20732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Furuyama W., Reynolds P., Haddock E., Meade-White K., Quynh Le M., Kawaoka Y., Feldmann H., Marzi A. A single dose of a vesicular stomatitis virus-based influenza vaccine confers rapid protection against H5 viruses from different clades. NPJ Vaccines. 2020;5:4. doi: 10.1038/s41541-019-0155-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garbutt M., Liebscher R., Wahl-Jensen V., Jones S., Möller P., Wagner R., Volchkov V., Klenk H.D., Feldmann H., Ströher U. Properties of replication-competent vesicular stomatitis virus vectors expressing glycoproteins of filoviruses and arenaviruses. J. Virol. 2004;78:5458–5465. doi: 10.1128/JVI.78.10.5458-5465.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisbert T.W., Jones S., Fritz E.A., Shurtleff A.C., Geisbert J.B., Liebscher R., Grolla A., Ströher U., Fernando L., Daddario K.M. Development of a new vaccine for the prevention of Lassa fever. PLoS Med. 2005;2:e183. doi: 10.1371/journal.pmed.0020183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giroglou T., Cinatl J., Jr., Rabenau H., Drosten C., Schwalbe H., Doerr H.W., von Laer D. Retroviral vectors pseudotyped with severe acute respiratory syndrome coronavirus S protein. J. Virol. 2004;78:9007–9015. doi: 10.1128/JVI.78.17.9007-9015.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guan W.J., Ni Z.Y., Hu Y., Liang W.H., Ou C.Q., He J.X., Liu L., Shan H., Lei C.L., Hui D.S.C., China Medical Treatment Expert Group for Covid-19 Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hoffmann M., Kleine-Weber H., Schroeder S., Krüger N., Herrler T., Erichsen S., Schiergens T.S., Herrler G., Wu N.H., Nitsche A. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell. 2020;181:271–280.e8. doi: 10.1016/j.cell.2020.02.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jae L.T., Raaben M., Herbert A.S., Kuehne A.I., Wirchnianski A.S., Soh T.K., Stubbs S.H., Janssen H., Damme M., Saftig P. Virus entry. Lassa virus entry requires a trigger-induced receptor switch. Science. 2014;344:1506–1510. doi: 10.1126/science.1252480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones S.M., Feldmann H., Ströher U., Geisbert J.B., Fernando L., Grolla A., Klenk H.D., Sullivan N.J., Volchkov V.E., Fritz E.A. Live attenuated recombinant vaccine protects nonhuman primates against Ebola and Marburg viruses. Nat. Med. 2005;11:786–790. doi: 10.1038/nm1258. [DOI] [PubMed] [Google Scholar]
- Kobinger G.P., Limberis M.P., Somanathan S., Schumer G., Bell P., Wilson J.M. Human immunodeficiency viral vector pseudotyped with the spike envelope of severe acute respiratory syndrome coronavirus transduces human airway epithelial cells and dendritic cells. Hum. Gene Ther. 2007;18:413–422. doi: 10.1089/hum.2006.194. [DOI] [PubMed] [Google Scholar]
- Kruse R.L. Therapeutic strategies in an outbreak scenario to treat the novel coronavirus originating in Wuhan, China. F1000Res. 2020;9:72. doi: 10.12688/f1000research.22211.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei C., Qian K., Li T., Zhang S., Fu W., Ding M., Hu S. Neutralization of SARS-CoV-2 spike pseudotyped virus by recombinant ACE2-Ig. Nat. Commun. 2020;11:2070. doi: 10.1038/s41467-020-16048-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Letko M., Marzi A., Munster V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat. Microbiol. 2020;5:562–569. doi: 10.1038/s41564-020-0688-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W., Moore M.J., Vasilieva N., Sui J., Wong S.K., Berne M.A., Somasundaran M., Sullivan J.L., Luzuriaga K., Greenough T.C. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li F., Li W., Farzan M., Harrison S.C. Structure of SARS coronavirus spike receptor-binding domain complexed with receptor. Science. 2005;309:1864–1868. doi: 10.1126/science.1116480. [DOI] [PubMed] [Google Scholar]
- Li J., Wang J.T., Whelan S.P. A unique strategy for mRNA cap methylation used by vesicular stomatitis virus. Proc. Natl. Acad. Sci. USA. 2006;103:8493–8498. doi: 10.1073/pnas.0509821103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li H., Handsaker B., Wysoker A., Fennell T., Ruan J., Homer N., Marth G., Abecasis G., Durbin R., 1000 Genome Project Data Processing Subgroup The Sequence Alignment/Map format and SAMtools. Bioinformatics. 2009;25:2078–2079. doi: 10.1093/bioinformatics/btp352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lontok E., Corse E., Machamer C.E. Intracellular targeting signals contribute to localization of coronavirus spike proteins near the virus assembly site. J. Virol. 2004;78:5913–5922. doi: 10.1128/JVI.78.11.5913-5922.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McBride C.E., Li J., Machamer C.E. The cytoplasmic tail of the severe acute respiratory syndrome coronavirus spike protein contains a novel endoplasmic reticulum retrieval signal that binds COPI and promotes interaction with membrane protein. J. Virol. 2007;81:2418–2428. doi: 10.1128/JVI.02146-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miersch S., Ustav M.J., Li Z., Case J.B., Ganaie S., Matusali G., Colavita F., Lapa D., Capobianchi M.R., Novelli G. Synthetic antibodies neutralize SARS-CoV-2 infection of mammalian cells. bioRxiv. 2020 doi: 10.1101/2020.06.05.137349. [DOI] [Google Scholar]
- Moore M.J., Dorfman T., Li W., Wong S.K., Li Y., Kuhn J.H., Coderre J., Vasilieva N., Han Z., Greenough T.C. Retroviruses pseudotyped with the severe acute respiratory syndrome coronavirus spike protein efficiently infect cells expressing angiotensin-converting enzyme 2. J. Virol. 2004;78:10628–10635. doi: 10.1128/JVI.78.19.10628-10635.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mukherjee S., Sirohi D., Dowd K.A., Chen Z., Diamond M.S., Kuhn R.J., Pierson T.C. Enhancing dengue virus maturation using a stable furin over-expressing cell line. Virology. 2016;497:33–40. doi: 10.1016/j.virol.2016.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nie J., Li Q., Wu J., Zhao C., Hao H., Liu H., Zhang L., Nie L., Qin H., Wang M. Establishment and validation of a pseudovirus neutralization assay for SARS-CoV-2. Emerg. Microbes Infect. 2020;9:680–686. doi: 10.1080/22221751.2020.1743767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ou X., Liu Y., Lei X., Li P., Mi D., Ren L., Guo L., Guo R., Chen T., Hu J. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020;11:1620. doi: 10.1038/s41467-020-15562-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Persson H., Ye W., Wernimont A., Adams J.J., Koide A., Koide S., Lam R., Sidhu S.S. CDR-H3 diversity is not required for antigen recognition by synthetic antibodies. J. Mol. Biol. 2013;425:803–811. doi: 10.1016/j.jmb.2012.11.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pinto D., Park Y.J., Beltramello M., Walls A.C., Tortorici M.A., Bianchi S., Jaconi S., Culap K., Zatta F., De Marco A. Cross-neutralization of SARS-CoV-2 by a human monoclonal SARS-CoV antibody. Nature. 2020;583:290–295. doi: 10.1038/s41586-020-2349-y. [DOI] [PubMed] [Google Scholar]
- Raj V.S., Mou H., Smits S.L., Dekkers D.H., Müller M.A., Dijkman R., Muth D., Demmers J.A., Zaki A., Fouchier R.A. Dipeptidyl peptidase 4 is a functional receptor for the emerging human coronavirus-EMC. Nature. 2013;495:251–254. doi: 10.1038/nature12005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ruch T.R., Machamer C.E. The coronavirus E protein: assembly and beyond. Viruses. 2012;4:363–382. doi: 10.3390/v4030363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shen C., Wang Z., Zhao F., Yang Y., Li J., Yuan J., Wang F., Li D., Yang M., Xing L. Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma. JAMA. 2020;323:1582–1589. doi: 10.1001/jama.2020.4783. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stanifer M.L., Cureton D.K., Whelan S.P. A recombinant vesicular stomatitis virus bearing a lethal mutation in the glycoprotein gene uncovers a second site suppressor that restores fusion. J. Virol. 2011;85:8105–8115. doi: 10.1128/JVI.00735-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stettler K., Beltramello M., Espinosa D.A., Graham V., Cassotta A., Bianchi S., Vanzetta F., Minola A., Jaconi S., Mele F. Specificity, cross-reactivity, and function of antibodies elicited by Zika virus infection. Science. 2016;353:823–826. doi: 10.1126/science.aaf8505. [DOI] [PubMed] [Google Scholar]
- Takada A., Feldmann H., Stroeher U., Bray M., Watanabe S., Ito H., McGregor M., Kawaoka Y. Identification of protective epitopes on ebola virus glycoprotein at the single amino acid level by using recombinant vesicular stomatitis viruses. J. Virol. 2003;77:1069–1074. doi: 10.1128/JVI.77.2.1069-1074.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tao Y., Mis M., Blazer L., Ustav M., Jnr, Steinhart Z., Chidiac R., Kubarakos E., O’Brien S., Wang X., Jarvik N. Tailored tetravalent antibodies potently and specifically activate Wnt/Frizzled pathways in cells, organoids and mice. eLife. 2019;8:e46134. doi: 10.7554/eLife.46134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- ter Meulen J., van den Brink E.N., Poon L.L., Marissen W.E., Leung C.S., Cox F., Cheung C.Y., Bakker A.Q., Bogaards J.A., van Deventer E. Human monoclonal antibody combination against SARS coronavirus: synergy and coverage of escape mutants. PLoS Med. 2006;3:e237. doi: 10.1371/journal.pmed.0030237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tortorici M.A., Veesler D. Structural insights into coronavirus entry. Adv. Virus Res. 2019;105:93–116. doi: 10.1016/bs.aivir.2019.08.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Traggiai E., Becker S., Subbarao K., Kolesnikova L., Uematsu Y., Gismondo M.R., Murphy B.R., Rappuoli R., Lanzavecchia A. An efficient method to make human monoclonal antibodies from memory B cells: potent neutralization of SARS coronavirus. Nat. Med. 2004;10:871–875. doi: 10.1038/nm1080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van den Pol A.N., Mao G., Chattopadhyay A., Rose J.K., Davis J.N. Chikungunya, Influenza, Nipah, and Semliki Forest Chimeric Viruses with Vesicular Stomatitis Virus: Actions in the Brain. J. Virol. 2017;91 doi: 10.1128/JVI.02154-16. e02154-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walls A.C., Park Y.J., Tortorici M.A., Wall A., McGuire A.T., Veesler D. Structure, Function, and Antigenicity of the SARS-CoV-2 Spike Glycoprotein. Cell. 2020;181:281–292.e6. doi: 10.1016/j.cell.2020.02.058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Watanabe Y., Allen J.D., Wrapp D., McLellan J.S., Crispin M. Site-specific glycan analysis of the SARS-CoV-2 spike. Science. 2020:eabb9983. doi: 10.1126/science.abb9983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whelan S.P., Ball L.A., Barr J.N., Wertz G.T. Efficient recovery of infectious vesicular stomatitis virus entirely from cDNA clones. Proc. Natl. Acad. Sci. USA. 1995;92:8388–8392. doi: 10.1073/pnas.92.18.8388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wrapp D., Wang N., Corbett K.S., Goldsmith J.A., Hsieh C.L., Abiona O., Graham B.S., McLellan J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science. 2020 doi: 10.1126/science.abb2507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yuan M., Wu N.C., Zhu X., Lee C.D., So R.T.Y., Lv H., Mok C.K.P., Wilson I.A. A highly conserved cryptic epitope in the receptor binding domains of SARS-CoV-2 and SARS-CoV. Science. 2020;368:630–633. doi: 10.1126/science.abb7269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zang R., Gomez Castro M.F., McCune B.T., Zeng Q., Rothlauf P.W., Sonnek N.M., Liu Z., Brulois K.F., Wang X., Greenberg H.B. TMPRSS2 and TMPRSS4 promote SARS-CoV-2 infection of human small intestinal enterocytes. Sci. Immunol. 2020;5:eabc3582. doi: 10.1126/sciimmunol.abc3582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang T., Wu Q., Zhang Z. Probable Pangolin Origin of SARS-CoV-2 Associated with the COVID-19 Outbreak. Curr Biol. 2020;30:1346–1351.e2. doi: 10.1016/j.cub.2020.03.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou P., Yang X.L., Wang X.G., Hu B., Zhang L., Zhang W., Si H.R., Zhu Y., Li B., Huang C.L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The authors declare that all data supporting the findings of this study are available within the paper. The sequence of VSV-eGFP-SARS-CoV-2-SΔ21 (passage 12) generated during this study is available through NCBI (SRA: SRR11878607, https://www.ncbi.nlm.nih.gov/sra/?term=SRR11878607; BioProject: PRJNA635934).