Highlights
-
•
Vascular development depends on the timely differentiation of endothelial and smooth muscle cells.
-
•
Vascular aging and vascular disease are influenced by endothelial and vascular smooth muscle cell compartments.
-
•
A survey of the literature on the role of PDE5 in vascular development, aging and disease is reported.
-
•
The role of PDE5 on vascular development, aging and disease needs to be further investigated by its genetic ablation.
Keywords: PDE5, VSMCs, ECs, cGMP, Nitric oxide
Abstract
Vascular tree development depends on the timely differentiation of endothelial and vascular smooth muscle cells. These latter are key players in the formation of the vascular scaffold that offers resistance to the blood flow. This review aims at providing an overview on the role of PDE5, the cGMP-specific phosphodiesterase that historically attracted much attention for its involvement in male impotence, in the regulation of vascular smooth muscle cell function. The overall goal is to underscore the importance of PDE5 expression and activity in this cell type in the context of the organs where its function has been extensively studied.
1. Introduction
The ramification of the mammalian vascular network occurs very early during embryogenesis in a complex and multifactorial process known as vasculogenesis (Yancopoulos et al., 2000). In humans, it starts approximately at day 18 after fertilization while in the mouse at embryonic day 7 from the extraembryonic mesoderm as formation of blood islands (yolk sac, connecting stalk and chorion) (Larsen, 1998) and then proceeds within the splanchnic mesoderm by angioblast conversion into endothelial cells (ECs) (Goldie et al., 2008).
From pre-existing vessels, the sprouting of capillaries leads to the formation of new microvasculature in a process known as angiogenesis that distributes to avascularized tissues bringing oxygen and nutrients (Carmeliet, 2000; Risau, 1997). Recruitment of peri-endothelial vascular smooth muscle cells (VSMCs) from the ECs lining blood vessels consolidates the vasculogenetic process and provides a contractile protection layer to neo-formed vessels (Carmeliet, 2000). The origin of EC and VSMC lineages has been shown to be highly heterogeneous and strictly depends on the organ in which the vascular web is developing.
2. The role of endothelium
Angioblasts are the first endothelial progenitor cells (EPCs) within the blood islands of the yolk sac that reach the embryo to form the primitive vascular tree (Coffin et al., 1991, Hatzopoulos et al., 1998, Risau, 1997). Initially, homogeneous EPCs differentiate into specific phenotypes that are needed to support the diverse functions involved in sustaining blood flow, cell permeability and transport that each organ requires during development. Accordingly, ECs express specific molecular markers that identify and differentiate arterious, venous, and lymphatic endothelia, by the expression of specific gene signatures. ECs can be also recruited from bone marrow (BM) by action of vascular endothelial growth factor (VEGF) in order to induce neovascularization in physiological and pathological conditions (Asahara et al., 1999; Bertolini et al., 2006). BM-ECs also play a role in tumor neoangiogenesis, although their contribution to cancer vessels can be variable, depending on cancer type or grade (Duda et al., 2006; Ruzinova et al., 2003).
3. The role of VSMCs
If ECs originate from different regional sources and show high level of complexity, VSMCs are no exception. VSMC heterogeneity has been extensively described according to their proliferative or quiescent status that involve phenotypic switching and differences in cell morphology at confluence, production of growth factors or differential expression of cyclic nucleotide hydrolyzing enzymes (Dolci et al., 2006; Owens et al., 2004). An additional level of heterogeneity comes from lineage tracing studies that identified distinct sources of VSMC progenitors colonizing different vessels, or even different areas of the same vessel (Le Lievre and Le Douarin, 1975).
VSMCs can be recruited to small vessels to form pericytes that regulate tissue microcirculation and vascular leakage (Armulik et al., 2010; Daneman et al., 2010). Pericyte origin can be the same as that of the arterial VSMCs, as demonstrated for the cephalic neural crest cells that colonize brain arteries and microvasculature (Etchevers et al., 2001), or for endocardial cells that colonize part of the coronary bed (Chen et al., 2016), however more studies are needed to understand where they originate from in other locations during development and in the adulthood. In the adult, arterial walls contain both proliferative and differentiated, contractile VSMCs within the medial layer and progenitor cells, mostly in the tunica adventitia, that contribute to form a stem cell reservoir (Majesky et al., 2011). Several studies have shown that arterial VSMCs possess a different gene expression signature compared to the venous counterpart (reviewed in (Aird, 2007). VSMCs of arteries synthesize and secrete the majority of extracellular matrix (ECM) proteins and proteoglycans that confer elasticity and compliance to large arterious vessels and alteration of cell-ECM interaction leads to arterial stiffness during aging and disease [for a review see (Lacolley et al., 2017)].
4. NO/cGMP/PDE5 pathway activity in EC and VSMC physiology
Nitric oxide (NO) is a free radical gas, known to activate autocrine/paracrine pathways. Endothelial nitric oxide is a potent stimulus for angiogenesis (van der Zee et al., 1997) and its synthesis is controlled by VEGF that induces the specific endothelial NO synthase (eNOS) (van der Zee et al., 1997). As almost every cell, ECs are responsive to NO that activates soluble guanylyl cyclase (sGC) to produce cyclic guanosine monophosphate (cGMP), which in turn activates protein kinase G (PKG). NO produced by eNOS in endothelial cells acts paracrinally on neighbouring smooth muscle cells targeting sGC (Farah et al., 2018).
PKG acts on several downstream targets in VSMCs among which vasodilator-stimulated phosphoprotein (VASP), F-actin, extracellular signal-regulated kinase (ERK) 1/2 and p38 mitogen-activated protein kinase (MAPK), each contributing to NO angiogenic potential (Chen et al., 2008; Zhang et al., 2003).
Type 5 phosphodiesterase (PDE5) belongs to a family of phosphodiesterases (PDEs) that hydrolyze cyclic nucleotides cAMP and/or cGMP second messengers (Bender and Beavo, 2006). Among the family members, PDE5 is the predominant phosphodiesterase in vascular and trabecular smooth muscle cells that hydrolyzes specifically cGMP under basal condition (low calcium level), whereas PDE1, activated by Ca2+ / calmodulin, plays a pivotal role in high calcium conditions as, for example, during muscle contraction (Laursen et al., 2017). We previously reported that PDE5 localizes in cytoplasmic vesicles and discrete cytoplasmic foci corresponding to centrosomes of several cell types (Dolci et al., 2006). We also showed that PDE5 levels are inversely related to the mitotic activity of SMCs. Treatment of VSMCs with platelet-derived grow factor (PDGF), a potent mitogen for this cell type, downregulates PDE5 expression, an effect that is similar to that of human chorionic gonadotropin (hCG) on human myometrial cells (Carosa et al., 2014; Dolci et al., 2006).
5. PDE5 inhibitors
Serendipitously discovered more than 20 years ago with the aim to protect the damaged cardiac tissue by inhibiting the PDE5, sildenafil is the first and one of the most powerful PDE5 inhibitors (PDE5i) identified that showed penile erections as a common side effect [see (Ghofrani et al., 2006) and references therein]. The cardioprotection was found relatively weak, but thanks to the specific high expression of PDE5 in the VSMCs of the penis, the drug was classified as the first oral treatment of erectile dysfunction (Goldstein et al., 1998). To date, four marketed PDE5i are available with identical pharmacodynamic properties, targeting the same enzyme, but with different specificities and pharmacokinetic characteristics (Jannini et al., 2012). Avanafil is the PDE5i with the highest specificity for PDE5 (Wang et al., 2012). The lowest cross-reaction with the other PDEs account for its excellent tolerability profile (Corona et al., 2016). Vardenafil has a pharmacokinetic profile similar to sildenafil, but with a lower IC50, indicating how much of a drug is needed to inhibit PDE5 activity by 50 % (Porst et al., 2001). Tadalafil, which, at low daily dose, is also approved for the treatment of urinary lower tract symptoms (Gonzalez and Kaplan, 2006), has the worst safety profile for the cross reaction with type 11 muscular PDE (Bischoff, 2004) producing severe back pain and myalgia in 2–3 % of patients treated. However, this drug shows the longest half-life (around 17 h, vs the average 4–5 of the others), a merit in absence of side effects, but a risk when adverse events are present.
The vascular activity of each PDE5i has been demonstrated to be peculiar. This might be linked to the stereochemical characteristics of the binding to the enzymatic substrate or possible cross-reaction with other substrates. In fact, 10 mg vardenafil daily did not alter endothelial function or arterial stiffness (Melehan et al., 2018) and the same drug at a 20 mg regimen, as well 60 mg of tadalafil (a dose trice of the maximal for the on demand use and 12 times of that prescribed for the daily use) did not impact on arterial oxygenation in patients with pulmonary arterial hypertension (PAH), a severe vascular disease leading to a progressive increase in pulmonary vascular resistance (Galie et al., 2005). On the contrary, this specific effect, comparable to that of NO, was only seen with 50 mg of sildenafil (Kuschner, 2006). For this reason, sildenafil is approved, as for NO and tadalafil (at 40 mg/day, twice the maximal dose for ED), for the use in PAH. The specific ability in targeting the vascular performance was further demonstrated with penile dynamic Color Doppler Ultrasound (CDU), which found that only sildenafil, and not tadalafil nor vardenafil, was able to improve the hemodynamics of the penis of impotent patients (Jannini et al., 2009). Sildenafil cross specificity to other PDEs may potentially account for the differential effects on vasodilation of this drug compared to vardenafil or tadalafil. While sildenafil shows a lower, albeit high, selectivity ratio for PDE1 with respect to vardenafil (40 vs 140, respectively), tadalafil is highly selective with respect to PDE1, while selectivity ratio with other PDEs is more than 1000 (Saenz de Tejada et al., 2001).
6. Role of PDE5 in VSMC and EC aging and disease: lessons from PDE5i
Vascular aging is a degenerative process that affects the arterial walls representing the condition that precedes the onset of vascular disease, starting with the chronic inflammation of the intima-media unit, when dysmetabolic cues alter the local homeostasis. Modifications of the vascular tunica involve phenotypic changes of VSMCs, including cell death, calcification and mechano-sensing ability, contractile or proliferative switch, that in turn affect EC fitness leading to thickening and stiffening of the vascular wall (Lacolley et al., 2018).
Despite VSMC heterogeneity during development, PDE5 is highly expressed in VSMCs from almost every organ and is one of the most abundant cGMP-PDEs present in arterial SMCs together with PDE1A, 1B, and 1C, PDE3A and 3B (Liu et al., 2008). Three PDE5 isoforms have been identified in humans, including PDE5A1, PDE5A2, and PDE5A3 that differ in their respective 5′ terminus mRNAs originating from three alternative first exons in the pre-mRNA (Campolo et al., 2018; Kotera et al., 2000; Lin et al., 2000). The three isoforms, that show differential amino acid lengths, are all expressed in aortic VSMCs (Cesarini et al., 2019), possess similar cGMP catalytic activity and are sensitive to PDE5 specific inhibitors (see below) (Lin et al., 2000).
VSMC contractility is tightly regulated by NO/cGMP/PDE5 signalling, and dysregulation of this pathway can modify the vascular biology determining hypertension and/or age-related modification of vascular system (Mergia and Stegbauer, 2016) (Durik et al., 2012). In particular, age-related reduction of NO bioavailability is responsible for reduction in endothelium-dependent dilation, enhanced vasoconstriction, and dysregulation of tissue perfusion [(Ungvari et al., 2018) and references therein]. In agreement, it has been shown that PDE5 and PDE1 mRNA levels and activity are elevated in senescent human VSMCs (Bautista Nino et al., 2015) and that such alterations can modify the vascular tone and contribute to the development of hypertension (Stegbauer et al., 2013) implying that PDE5i might indeed improve VSMC tone regulation.
The majority of PDE5 studies on vascular aging and disease have been based on acute or chronic PDE5i administration to block PDE5 activity, assuming that this condition might equal PDE5 absence, since, to date, no animal models for PDE5 deletion have been reported. Thus, other non-enzymatic actions or cross-reactions with additional substrates can not be excluded.
In this section, we reported PDE5 expression and/or enzymatic activity inhibition data studied in the different anatomical regions involving effects on VSMC and EC vascular compartments.
6.1. Aorta
PDE5 is the most expressed cGMP-PDE in the human aorta (Cesarini et al., 2019). Its expression is activated in the muscular layer early during embryogenesis. In 7 weeks old human embryo, as well as in E12 mouse embryos, PDE5 expression is initially found in the ventral mesenchymal area of the thoraco-abdominal segment that corresponds to the localization site of migratory VSMCs from somites in the chick dorsal aorta (Hungerford et al., 1996). We recently found that PDE5 isoforms A1 and A3 are both up-regulated by Notch3 in VSMCs in vitro, while all the three isoforms are strongly downregulated in thoracic aortic aneurysms (TAAs) (Cesarini et al., 2019). In line with our reports, three independent case reports of patients experiencing aortic aneurysm dissection following the abuse of sildenafil have been described (Famularo et al., 2001; Nachtnebel et al., 2006; Tiryakioglu et al., 2009).
6.2. Heart
PDE5 expression in the heart is quite controversial. While positivity to anti-PDE5 antibodies has been reported in coronary VSMCs (Corbin et al., 2005; Shan et al., 2012), healthy myocardium does not seem to express high levels of the enzyme (Degen et al., 2015). A consistent upregulation of PDE5 has been, however, detected in congestive heart failure and right ventricular hypertrophy (Lu et al., 2010; Pokreisz et al., 2009; Shan et al., 2012). In these conditions, angiotensin II stimulation of VSMCs determines the induction of PDE5 that, in turn, reduces cGMP/PKG signalling increasing the angiotensin II response (Kass et al., 2007). This loop can exacerbate hypertension and vascular proliferation in diseases involving renin/angiotensin stimulations. Sildenafil treatment prevents ventricular hypertrophy induced by transverse aortic ligation (Takimoto et al., 2005) and protects against ischemia-reperfusion injury. Indeed, Ockaili et al. demonstrated that infusion of sildenafil decreases the infarct size in a rabbit model (Ockaili et al., 2002). A recent study on a large Swedish cohort demonstrated that treatment for erectile dysfunction (ED) after a first myocardial infarct was associated with a reduced mortality and heart failure hospitalisation. Only patients treated with PDE5i had a reduced risk, which appeared to be dose-dependent, strongly favouring for a role of PDE5i in the relaxation of coronary SMC tone (Andersson et al., 2017). Chronic use of sildenafil improves diabetic cardiomyopathy via an anti-remodelling effect, resulting in improved cardiac kinetics and circulating markers (Giannetta et al., 2012). PDE5i-dependent heart protection operated by tadalafil has been shown to be achieved through PGK-dependent hydrogen sulfide (H2S) intracellular signaling, which contributes to the opening of mitochondrial ATP-sensitive potassium channels and cell homeostasis maintenance (Salloum et al., 2009). Moreover, inhibition of PDE5 has been shown to enhance ischemia-induced angiogenesis by mobilizing EPCs through a PKG-dependent HIF-1/VEGF pathway (Sahara et al., 2010). In this scenario, it is conceivable to hypothesize future cardiac indications, albeit still debated (Corinaldesi et al., 2016). All these studies and the large bibliography on the beneficial effects of PDE5i on heart vascular function definitively prove that PDE5i do not show cardiovascular life-threatening adverse effects even if their synergistic interaction with NO-donors, such as nitrates frequently used for angina, should be avoided due to low blood pressure or syncope as side effects.
6.3. Dermis
Vasomotor dysfunction of the cutaneous surface is a wide-spread disorder in older populations. It has been shown that PDE5 plays an important role in the regulation of capillary blood flow of extremities as demonstrated by successful PDE5i treatment of patients with therapy-resistant Raynaud’s phenomenon (Fries et al., 2005;). This condition is caused by episodic vasospasm and ischaemia of the extremities in response to cold temperatures or to emotional stress and can affect individuals at any age. In elderly patients, Raynaud’s phenomenon is often a secondary event that masks a secondary disease, such as occlusive arterial, rheumatologic or haematologic diseases (Goundry et al., 2012). Velocity of capillary blood flow, severely impaired in these patients, was able to increase up to >400 % in sildenafil groups compared to placebo treated patients (Fries et al., 2005). PDE5 has also been shown to be expressed in the dermal compartment of mouse hair follicles, and in vitro treatment of dermal papillae with sildenafil induces VEGF and platelet derived growth factor A (PDGFA) secretion, increased dermal proliferation and neoangiogenesis (Choi et al., 2018), although studies in humans have not proved conclusive evidences (Sarifakioglu et al., 2006).
6.4. Kidney
PDE5 plays a pivotal role in the vascular dysfunction that characterizes the renovascular hypertension; indeed, sildenafil, the most frequently used treatment for impotence in renal failure (Vecchio et al., 2010), is able to restore the depressed baroreflex control in a rat model of renovascular hypertension (Cavalcanti et al., 2016). Although within the kidney parenchyma PDE5 shows a vascular expression only, studies have also demonstrated the beneficial effects of sildenafil treatment on renal cell apoptosis, oxidative stress and mesangial matrix deposition in chronic kidney disease and in diabetic mouse models as a result of renal microcirculation improvement (Jeong et al., 2009; Pofi et al., 2017; Rodriguez-Iturbe et al., 2005).
6.5. Female genital apparatus and placental annexes
As mentioned above, PDE5 is expressed in myometrium and in myometrial arteries and its levels are down-regulated by hCG in vitro (Belmonte et al., 2005). Its inhibition by sildenafil improves embryo implantation and pregnancy rates in a cohort of in vitro fertilization (IVF) low success patients (Sher and Fisch, 2000), suggesting that PDE5 inhibition by hCG supports embryo implantation and early pregnancy. PDE5 immunoreactivity was also detected in VSMCs of the pseudocavernous tissue of the vaginal wall and in clitoral corpora cavernosa (D'Amati et al., 2002; Park et al., 1998), however it is not clear if it might play a role in female sexual dysfunction (FSD) and clinical trials on the use of PDE5i to treat FSD need further studies.
PDE5 is also highly expressed in umbilical arteries and counteracts fetal umbilical artery relaxation (Santos-Silva et al., 2008), suggesting a role in pregnancy-induced hypertension and preeclampsia. In agreement with this observation, a recent metanalysis quoted a large amount of animal and human studies that highlighted the beneficial effects of sildenafil on fetal growth restriction (FGR) due to placental insufficiency (Paauw et al., 2017). More recent results obtained from the clinical trial STRIDER and from a randomised controlled trial (Sharp et al., 2018), however, showed no such effect and no indications of efficacy.
6.6. Male genital apparatus
PDE5 is abundantly expressed in the male genital apparatus; however, it is its localization in VSCMs of penile corpora cavernosa that made PDE5i discovery one of the most important revolutions of the last century. Such localization is conserved along the phylogenetic tree in low mammals at least down to snakes (Capel et al., 2011). Interestingly, PDE5 deregulation causes priapism in eNOS1-3 KO mice (Champion et al., 2005) indicating that PDE5 is a key player of erection control in different species that evolved with or without the os penis, or baculum (Cellerino and Jannini, 2005).
Indeed, PDE5 expression is ontogenetically regulated and androgen-dependent in rats, with the highest levels of mRNA and protein expression in adult corpus cavernosum-derived SMCs (CC-SMCs) (Carosa et al., 2009; Morelli et al., 2004). PDGF induces proliferation of CC-SMCs and down-regulates PDE5 expression suggesting, as demonstrated for myometrial cells, that PDE5 marks the contractile phenotype of SMCs. Hyperoxia conditions positively control PDE5 gene expression while, as expected, hypoxia down-regulates it. In agreement with these results we found that Hif1α overexpression in vitro down-regulated PDE5 protein levels and identified in silico two canonical Hif Responsive Elements (HREs) within the first and the second PDE5 promoter (unpublished results).
A beneficial effect on steroidogenesis and age-related regressive changes in the rat testis has been reported when elderly animals were chronically treated with PDE5i, although the target cells of PDE5 inhibition have not been identified (Sokanovic et al., 2018).
6.7. Lung
While PDE3, PDE4 and PDE5 are the most expressed PDEs in bronchial/lung parenchyma, the most expressed PDE in fetal and postnatal lung arteries is PDE5 (Hanson et al., 1998). Its expression, as well as its enzymatic activity, is strongly up-regulated by hyperoxia in in vitro cultured pulmonary fetal VSMCs and in vivo (Farrow et al., 2008), playing a pivotal role in PAH. Indeed, persistent PAH of the newborn, a life-threatening condition, has been shown to be linked to PDE5 overexpression and overactivation (Farrow et al., 2008), and several studies have shown that sildenafil mediated inhibition of PDE5 reduces pulmonary vascular resistance and vessels remodelling in PAH (Prasad et al., 2000; Wharton et al., 2005). The reduction of pulmonary vascular resistance and pressure after selective PDE5 inhibition are also associated with blood decrease of Endothelin-1 (ET-1) and attenuation of lung vessel hypoxia-induced inflammatory cytokine expression (Rossi et al., 2008) (Tsai et al., 2006). Recent clinical trials have succeeded in demonstrating the efficacy of sildenafil alone or in combination with ET-1 receptor antagonists to improve physical performances of patients affected by pulmonary hypertension alone or complicated by collagenopathies (SUPER-1 and COMPASS-2, respectively (Kario et al., 2019; Krishnan and Horn, 2016)). Covid-19 viral lung infection, the pandemia that is currently causing thousands of victims for respiratory insufficiency, has been hypothesized to affect not only the alveolar compartment but also the pulmonary microvasculature. Given the efficacy of NO on reducing PAH, a clinical trial has now opened in USA to test the effects of NO inhalation on Covid-19 dependent acute respiratory distress syndrome (ARDS). Theoretically, by reducing the pulmonary arterial pressure NO will improve oxygen saturation and reduce the burden on the right ventricle. If NO inhalation will prove to be helpful, also PDE5i administration will be definitely beneficial to treat ARDS.
6.8. Blood
PDE5 expression in blood cells is confined to megakaryocytes and platelets. We and others have demonstrated that PDE5 is expressed both in fetal and postnatal platelet precursors and in terminally differentiated elements (Cesarini et al., 2019; Mullershausen et al., 2001). Physiologically, PDE5 activity blunts the NO/cGMP/PKG activated pathway within a short period of time contributing to the control of platelet secretion and aggregation. Two other PDE isoenzymes, PDE2 and PDE3, with activity for both cAMP and cGMP are found in platelets (Hidaka and Asano, 1976), explaining why PDE5i alone do not show anti-coagulant side effects (Fig. 1 ).
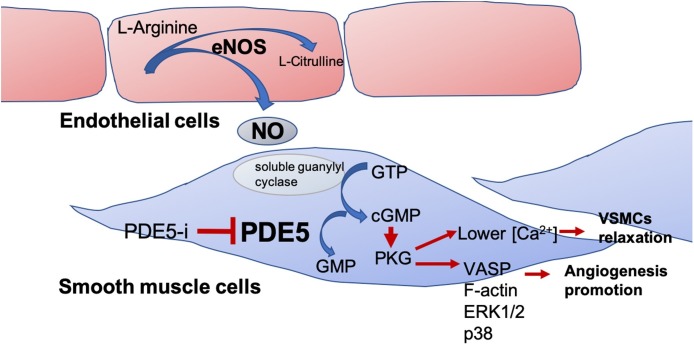
Fig. 1.
Schematic representation of NO/PDE5/PKG pathway in smooth muscle cells.
NO released from endothelial cells activates soluble guanylate cyclase that produces cGMP from GTP. cGMP activates PKG that phosphorylates several substrates thus inhibiting smooth muscle contraction and promoting angiogenesis. PDE5 reverses this condition by hydrolizing cGMP to GMP while PDE5i (sildenafil, vardenafil, tadalafil, avanafil) all promote smooth muscle relaxation by inhibiting cGMP accumulation.
6.9. Brain
A role of PDE5 together with PDE1 has been highlighted in age-dependent cerebral vasculature alterations. Indeed, in a mouse model of age-dependent vascular dysfunctions, the inhibition of both PDE5 and 1 restored the vascular functions (Bautista Nino et al., 2015; Olmestig et al., 2017; Pauls et al., 2018). Moreover, in a rat model of cerebral ischemia, sildenafil treatment improved angiogenesis and synaptogenesis around the ischemic boundary regions (Zhang et al., 2005).
Since PDE5 was detected in cerebral and meningeal arteries both in humans and rodents (Pauls et al., 2018), its involvement was analysed also in cerebral small vessel disease (SVD), a pathology that affects up to 70 % of older adults and that represents the most common cause of vascular cognitive impairment. Hypoperfusion due to SVD causes severe changes in subcortical areas that represent the first symptoms in Alzheimer’s disease. By improving cerebral blood flow, hemodynamic function and oxygen metabolism, sildenafil was shown to impact on Alzheimer’s disease (Sheng et al., 2017). A phase 2 trial (named PASTIS) examining whether tadalafil can increase blood flow to the deep vessels within the brain, is near to closing stage and will clarify this important issue (Pauls et al., 2017).
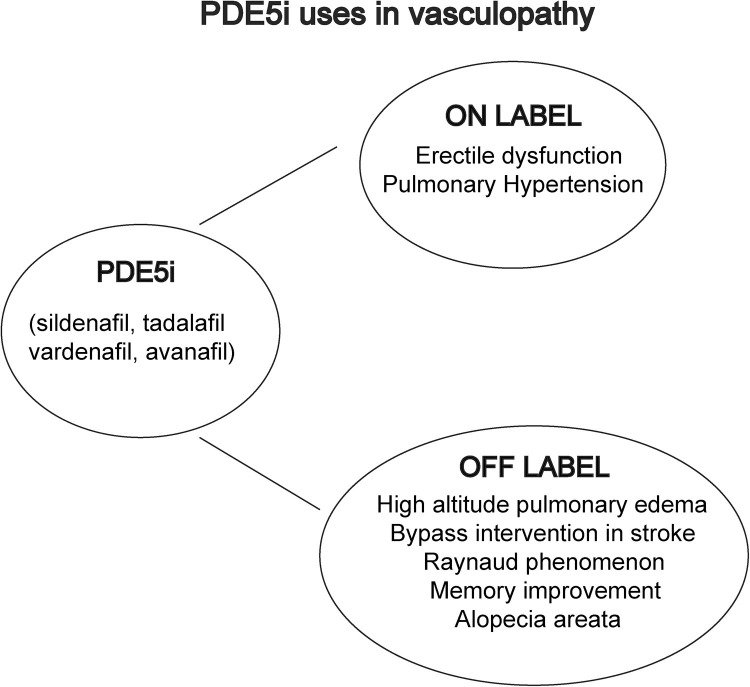
A summary of PDE5i on label and off label uses in vasculopathy is reported in Fig. 2 .
Fig. 2.
PDE5i use in pathologies linked to vascular defects. On label refers to approved uses while off label refers to unapproved uses of PDE5i for the indications reported.
7. PDE5 and ECs
PDE5 is expressed in VSMCs but not or at least not at immunodetectable levels in ECs, however its inhibition exerts important effects on endothelium. When querying for PDE5/PDE5i and endothelial cells in Medline databases, an amount of reports identifies endothelium-dependent vasodilation as the beneficial effect of PDE5 inhibition on ECs. Although NO and PDE5i-mediated cGMP increase induces relaxation of VSMCs, an event that enhances O2 tension at endothelial level, few data are present in literature that identify molecular targets specific of ECs that are influenced by VSMC relaxation. In particular, it has been shown that PDE5 inhibition promotes the recruitment of EPCs, which consistently contribute to neoangiogenesis and vascular repair (Diller et al., 2008) (Foresta et al., 2009), the decrease of ET-1 blood levels and of hypoxia-induced inflammatory cytokines such as (Intercellular Adhesion Molecule 1) ICAM-1, (Vascular Cell Adhesion Protein 1) VCAM-1, C-reactive protein (CRP) and Interleukin (IL)-6 (Vlachopoulos et al., 2009). In line with this observation, we previously reported that blood level of CXCL10 and CXCL8 chemokines released by inflammation-activated human cardiomyocytes and endothelial cells, respectively, significantly decreased after sildenafil treatment in diabetic patients at the onset of cardiomyopathy (Di Luigi et al., 2016; Giannattasio et al., 2019) (Morano et al., 2007).
A summary of PDE5 expression in VSMC and EC vascular compartments is reported in Table 1 .
Table 1.
PDE5 localization and effects of its activity or inhibition.
| PDE5 Localization | Effect | Ref. |
|---|---|---|
| Human myometrial cells, C2C12 myoblasts and rat corpora cavernosa cells | PDE5 expression is associated to quiescent, contractile SMCs and its levels are down regulated when VSMCs resume proliferation, suggesting that PDE5 is a marker of the contractile phenotype | Dolci et al., 2006 |
| Arterial SMCs | Developmental increase in PDE5 may enable the ductus to constrict and remodel itself more efficiently after birth | Liu et al., 2008 |
| Aortic VSMCs | PDE5 isoforms A1 and A3 are both up-regulated by Notch3 in VSMCs in vitro, while all the three isoforms are strongly downregulated in thoracic aortic aneurysms | Cesarini et al., 2019 |
| Senescent human VSMCs | High levels of PDE5 cause vasoconstriction response that alters the vascular tone and contributes to the development of hypertension | Bautista Nino et al., 2015; Bautista Nino et al., 2015; Stegbauer et al., 2013 |
| Coronary VSMCs | Upregulated myocardial PDE5 expression may contribute to the progression of heart failure | Corbin et al., 2005; Shan et al., 2012 |
| Myometrium and myometrial arteries | PDE5 marks the contractile phenotype of VSMCs | Belmonte et al., 2005 |
| VSMCs of pseudocavernous tissue of vaginal wall and clitoral corpora cavernosa | PDE5 might play a possible role in female sexual dysfunction | D’Amati et al., 2002; Park et al., 1998 |
| Umbilical arteries | PDE5 is highly expressed in umbilical arteries and counteracts fetal umbilical artery relaxation | Santos-Silva et al., 2008 |
| VSMCs of penile corpora cavernosa | PDE5 marks the contractile phenotype of SMCs | Carosa et al., 2009; Morelli et al., 2004 |
| Pulmonary arteries | PDE5 inhibition reduces pulmonary vascular resistance and vessel remodelling | Hanson et al., 1998 |
| Cerebral and meningeal arteries | PDE5 inhibition improves cerebral blood flow, hemodynamic function and oxygen metabolism | Pauls et al., 2018 |
8. Concluding remarks
This review has focused on the role of PDE5 on vascular development, aging and disease reporting on data obtained using specific PDE5 inhibitors. From all the results reviewed it emerges that PDE5 expression marks the contractile phenotype of VSMCs, that its “correct expression” correlates to the VSMC fitness and that its overactivity/overexpression is deleterious for the vascular health of almost all the organs of an individual. Although the huge amount of functional data present in the literature are based on results obtained by inhibition of PDE5, to date no results have been reported on the impact of PDE5 deletion on these processes, since no PDE5 knockout mouse models have been described yet. In particular it is not known if PDE5 absence might be compatible with correct embryonic or postnatal development and if other functions, apart from the enzymatic one, are attributable to this specific phosphodiesterase. Thus, further work is needed to clarify how PDE5 correct expression and function impact on this important issue.
Declaration of Competing Interest
EAJ is or has been consultant and/or paid speaker for Bayer, Ibsa, Lundbeck, Menarini, Otsuka, Pfizer, and Shionogi. All the other authors declare no conflict of interests
Acknowledgements
ADB, CC and SD were supported by PRIN grant #2017ATZ2YK_002
EAJ was supported by the PRIN grant #2017S9KTNE_002
References
- Aird W.C. Phenotypic heterogeneity of the endothelium: II. Representative vascular beds. Circ. Res. 2007;100:174–190. doi: 10.1161/01.RES.0000255690.03436.ae. [DOI] [PubMed] [Google Scholar]
- Andersson D.P., Trolle Lagerros Y., Grotta A., Bellocco R., Lehtihet M., Holzmann M.J. Association between treatment for erectile dysfunction and death or cardiovascular outcomes after myocardial infarction. Heart. 2017;103:1264–1270. doi: 10.1136/heartjnl-2016-310746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Armulik A., Genove G., Mae M., Nisancioglu M.H., Wallgard E., Niaudet C., He L., Norlin J., Lindblom P., Strittmatter K., Johansson B.R., Betsholtz C. Pericytes regulate the blood-brain barrier. Nature. 2010;468:557–561. doi: 10.1038/nature09522. [DOI] [PubMed] [Google Scholar]
- Asahara T., Takahashi T., Masuda H., Kalka C., Chen D., Iwaguro H., Inai Y., Silver M., Isner J.M. VEGF contributes to postnatal neovascularization by mobilizing bone marrow-derived endothelial progenitor cells. EMBO J. 1999;18:3964–3972. doi: 10.1093/emboj/18.14.3964. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bautista Nino P.K., Durik M., Danser A.H., de Vries R., Musterd-Bhaggoe U.M., Meima M.E., Kavousi M., Ghanbari M., Hoeijmakers J.H., O’Donnell C.J., Franceschini N., Janssen G.M., De Mey J.G., Liu Y., Shanahan C.M., Franco O.H., Dehghan A., Roks A.J. Phosphodiesterase 1 regulation is a key mechanism in vascular aging. Clin. Sci. 2015;129:1061–1075. doi: 10.1042/CS20140753. [DOI] [PubMed] [Google Scholar]
- Belmonte A., Ticconi C., Dolci S., Giorgi M., Zicari A., Lenzi A., Jannini E.A., Piccione E. Regulation of phosphodiesterase 5 expression and activity in human pregnant and non-pregnant myometrial cells by human chorionic gonadotropin. J. Soc. Gynecol. Investig. 2005;12:570–577. doi: 10.1016/j.jsgi.2005.09.002. [DOI] [PubMed] [Google Scholar]
- Bender A.T., Beavo J.A. Cyclic nucleotide phosphodiesterases: molecular regulation to clinical use. Pharmacol. Rev. 2006;58:488–520. doi: 10.1124/pr.58.3.5. [DOI] [PubMed] [Google Scholar]
- Bertolini F., Shaked Y., Mancuso P., Kerbel R.S. The multifaceted circulating endothelial cell in cancer: towards marker and target identification. Nat. Rev. Cancer. 2006;6:835–845. doi: 10.1038/nrc1971. [DOI] [PubMed] [Google Scholar]
- Bischoff E. Potency, selectivity, and consequences of nonselectivity of PDE inhibition. Int. J. Impot. Res. 2004;16(Suppl 1):S11–14. doi: 10.1038/sj.ijir.3901208. [DOI] [PubMed] [Google Scholar]
- Campolo F., Zevini A., Cardarelli S., Monaco L., Barbagallo F., Pellegrini M., Cornacchione M., Di Grazia A., De Arcangelis V., Gianfrilli D., Giorgi M., Lenzi A., Isidori A.M., Naro F. Identification of murine phosphodiesterase 5A isoforms and their functional characterization in HL-1 cardiac cell line. J. Cell. Physiol. 2018;233:325–337. doi: 10.1002/jcp.25880. [DOI] [PubMed] [Google Scholar]
- Capel R.O., Monica F.Z., Porto M., Barillas S., Muscara M.N., Teixeira S.A., Arruda A.M., Pissinatti L., Pissinatti A., Schenka A.A., Antunes E., Nahoum C., Cogo J.C., de Oliveira M.A., De Nucci G. Role of a novel tetrodotoxin-resistant sodium channel in the nitrergic relaxation of corpus cavernosum from the South American rattlesnake Crotalus durissus terrificus. J. Sex. Med. 2011;8:1616–1625. doi: 10.1111/j.1743-6109.2011.02246.x. [DOI] [PubMed] [Google Scholar]
- Carmeliet P. Mechanisms of angiogenesis and arteriogenesis. Nat. Med. 2000;6:389–395. doi: 10.1038/74651. [DOI] [PubMed] [Google Scholar]
- Carosa E., Rossi S., Giansante N., Gravina G.L., Castri A., Dolci S., Botti F., Morelli A., Di Luigi L., Pepe M., Lenzi A., Jannini E.A. The ontogenetic expression pattern of type 5 phosphodiesterase correlates with androgen receptor expression in rat corpora cavernosa. J. Sex. Med. 2009;6:388–396. doi: 10.1111/j.1743-6109.2008.01091.x. [DOI] [PubMed] [Google Scholar]
- Carosa E., Castri A., Forcella C., Sebastiani G., Di Sante S., Gravina G.L., Ronchi P., Cesarini V., Dolci S., Di Stasi S., Lenzi A., Jannini E.A. Platelet-derived growth factor regulation of type-5 phosphodiesterase in human and rat penile smooth muscle cells. J. Sex. Med. 2014;11:1675–1684. doi: 10.1111/jsm.12568. [DOI] [PubMed] [Google Scholar]
- Cavalcanti C.O., Alves R.R., de Oliveira A.L., Cruz J.C., de Franca-Silva M.S., Braga V.A., Balarini C.M. Inhibition of PDE5 restores depressed baroreflex sensitivity in renovascular hypertensive rats. Front. Physiol. 2016;7:15. doi: 10.3389/fphys.2016.00015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cellerino A., Jannini E.A. Why humans need type 5 phosphodiesterase inhibitors. Int. J. Androl. 2005;28(Suppl 2):14–17. doi: 10.1111/j.1365-2605.2005.00582.x. [DOI] [PubMed] [Google Scholar]
- Cesarini V., Pisano C., Rossi G., Balistreri C.R., Botti F., Antonelli G., Ruvolo G., Jannini E.A., Dolci S. Regulation of PDE5 expression in human aorta and thoracic aortic aneurysms. Sci. Rep. 2019;9:12206. doi: 10.1038/s41598-019-48432-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Champion H.C., Bivalacqua T.J., Takimoto E., Kass D.A., Burnett A.L. Phosphodiesterase-5A dysregulation in penile erectile tissue is a mechanism of priapism. Proc. Natl. Acad. Sci. U. S. A. 2005;102:1661–1666. doi: 10.1073/pnas.0407183102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen H., Levine Y.C., Golan D.E., Michel T., Lin A.J. Atrial natriuretic peptide-initiated cGMP pathways regulate vasodilator-stimulated phosphoprotein phosphorylation and angiogenesis in vascular endothelium. J. Biol. Chem. 2008;283:4439–4447. doi: 10.1074/jbc.M709439200. [DOI] [PubMed] [Google Scholar]
- Chen Q., Zhang H., Liu Y., Adams S., Eilken H., Stehling M., Corada M., Dejana E., Zhou B., Adams R.H. Endothelial cells are progenitors of cardiac pericytes and vascular smooth muscle cells. Nat. Commun. 2016;7:12422. doi: 10.1038/ncomms12422. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi H.I., Kang B.M., Jang J., Hwang S.T., Kwon O. Novel effect of sildenafil on hair growth. Biochem. Biophys. Res. Commun. 2018;505:685–691. doi: 10.1016/j.bbrc.2018.09.164. [DOI] [PubMed] [Google Scholar]
- Corbin J.D., Beasley A., Blount M.A., Francis S.H. High lung PDE5: a strong basis for treating pulmonary hypertension with PDE5 inhibitors. Biochem. Biophys. Res. Commun. 2005;334:930–938. doi: 10.1016/j.bbrc.2005.06.183. [DOI] [PubMed] [Google Scholar]
- Corinaldesi C., Di Luigi L., Lenzi A., Crescioli C. Phosphodiesterase type 5 inhibitors: back and forward from cardiac indications. J. Endocrinol. Invest. 2016;39:143–151. doi: 10.1007/s40618-015-0340-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Corona G., Rastrelli G., Burri A., Jannini E.A., Maggi M. The safety and efficacy of Avanafil, a new 2(nd) generation PDE5i: comprehensive review and meta-analysis. Expert Opin. Drug Saf. 2016;15:237–247. doi: 10.1517/14740338.2016.1130126. [DOI] [PubMed] [Google Scholar]
- D’Amati G., di Gioia C.R., Bologna M., Giordano D., Giorgi M., Dolci S., Jannini E.A. Type 5 phosphodiesterase expression in the human vagina. Urology. 2002;60:191–195. doi: 10.1016/s0090-4295(02)01663-1. [DOI] [PubMed] [Google Scholar]
- Daneman R., Zhou L., Kebede A.A., Barres B.A. Pericytes are required for blood-brain barrier integrity during embryogenesis. Nature. 2010;468:562–566. doi: 10.1038/nature09513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Degen C.V., Bishu K., Zakeri R., Ogut O., Redfield M.M., Brozovich F.V. The emperor’s new clothes: PDE5 and the heart. PLoS One. 2015;10:e0118664. doi: 10.1371/journal.pone.0118664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Luigi L., Corinaldesi C., Colletti M., Scolletta S., Antinozzi C., Vannelli G.B., Giannetta E., Gianfrilli D., Isidori A.M., Migliaccio S., Poerio N., Fraziano M., Lenzi A., Crescioli C. Phosphodiesterase type 5 inhibitor sildenafil decreases the proinflammatory chemokine CXCL10 in human cardiomyocytes and in subjects with diabetic cardiomyopathy. Inflammation. 2016;39:1238–1252. doi: 10.1007/s10753-016-0359-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diller G.P., van Eijl S., Okonko D.O., Howard L.S., Ali O., Thum T., Wort S.J., Bedard E., Gibbs J.S., Bauersachs J., Hobbs A.J., Wilkins M.R., Gatzoulis M.A., Wharton J. Circulating endothelial progenitor cells in patients with Eisenmenger syndrome and idiopathic pulmonary arterial hypertension. Circulation. 2008;117:3020–3030. doi: 10.1161/CIRCULATIONAHA.108.769646. [DOI] [PubMed] [Google Scholar]
- Dolci S., Belmonte A., Santone R., Giorgi M., Pellegrini M., Carosa E., Piccione E., Lenzi A., Jannini E.A. Subcellular localization and regulation of type-1C and type-5 phosphodiesterases. Biochem. Biophys. Res. Commun. 2006;341:837–846. doi: 10.1016/j.bbrc.2006.01.035. [DOI] [PubMed] [Google Scholar]
- Duda D.G., Cohen K.S., Kozin S.V., Perentes J.Y., Fukumura D., Scadden D.T., Jain R.K. Evidence for incorporation of bone marrow-derived endothelial cells into perfused blood vessels in tumors. Blood. 2006;107:2774–2776. doi: 10.1182/blood-2005-08-3210. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Durik M., Kavousi M., van der Pluijm I., Isaacs A., Cheng C., Verdonk K., Loot A.E., Oeseburg H., Bhaggoe U.M., Leijten F., van Veghel R., de Vries R., Rudez G., Brandt R., Ridwan Y.R., van Deel E.D., de Boer M., Tempel D., Fleming I., Mitchell G.F., Verwoert G.C., Tarasov K.V., Uitterlinden A.G., Hofman A., Duckers H.J., van Duijn C.M., Oostra B.A., Witteman J.C., Duncker D.J., Danser A.H., Hoeijmakers J.H., Roks A.J. Nucleotide excision DNA repair is associated with age-related vascular dysfunction. Circulation. 2012;126:468–478. doi: 10.1161/CIRCULATIONAHA.112.104380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Etchevers H.C., Vincent C., Le Douarin N.M., Couly G.F. The cephalic neural crest provides pericytes and smooth muscle cells to all blood vessels of the face and forebrain. Development. 2001;128:1059–1068. doi: 10.1242/dev.128.7.1059. [DOI] [PubMed] [Google Scholar]
- Famularo G., Polchi S., Di Bona G., Manzara C. Acute aortic dissection after cocaine and sildenafil abuse. J. Emerg. Med. 2001;21:78–79. doi: 10.1016/s0736-4679(01)00345-6. [DOI] [PubMed] [Google Scholar]
- Farah C., Michel L.Y.M., Balligand J.L. Nitric oxide signalling in cardiovascular health and disease. Nat. Rev. Cardiol. 2018;15:292–316. doi: 10.1038/nrcardio.2017.224. [DOI] [PubMed] [Google Scholar]
- Farrow K.N., Groh B.S., Schumacker P.T., Lakshminrusimha S., Czech L., Gugino S.F., Russell J.A., Steinhorn R.H. Hyperoxia increases phosphodiesterase 5 expression and activity in ovine fetal pulmonary artery smooth muscle cells. Circ. Res. 2008;102:226–233. doi: 10.1161/CIRCRESAHA.107.161463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Foresta C., De Toni L., Di Mambro A., Garolla A., Ferlin A., Zuccarello D. The PDE5 inhibitor sildenafil increases circulating endothelial progenitor cells and CXCR4 expression. J. Sex. Med. 2009;6:369–372. doi: 10.1111/j.1743-6109.2008.01014.x. [DOI] [PubMed] [Google Scholar]
- Fries R., Shariat K., von Wilmowsky H., Bohm M. Sildenafil in the treatment of Raynaud’s phenomenon resistant to vasodilatory therapy. Circulation. 2005;112:2980–2985. doi: 10.1161/CIRCULATIONAHA.104.523324. [DOI] [PubMed] [Google Scholar]
- Galie N., Ghofrani H.A., Torbicki A., Barst R.J., Rubin L.J., Badesch D., Fleming T., Parpia T., Burgess G., Branzi A., Grimminger F., Kurzyna M., Simonneau G., Sildenafil Use in Pulmonary Arterial Hypertension Study, G Sildenafil citrate therapy for pulmonary arterial hypertension. N. Engl. J. Med. 2005;353:2148–2157. doi: 10.1056/NEJMoa050010. [DOI] [PubMed] [Google Scholar]
- Ghofrani H.A., Osterloh I.H., Grimminger F. Sildenafil: from angina to erectile dysfunction to pulmonary hypertension and beyond. Nature reviews. Drug Discov. 2006;5:689–702. doi: 10.1038/nrd2030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giannattasio S., Corinaldesi C., Colletti M., Di Luigi L., Antinozzi C., Filardi T., Scolletta S., Basili S., Lenzi A., Morano S., Crescioli C. The phosphodiesterase 5 inhibitor sildenafil decreases the proinflammatory chemokine IL-8 in diabetic cardiomyopathy: in vivo and in vitro evidence. J. Endocrinol. Invest. 2019;42:715–725. doi: 10.1007/s40618-018-0977-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giannetta E., Isidori A.M., Galea N., Carbone I., Mandosi E., Vizza C.D., Naro F., Morano S., Fedele F., Lenzi A. Chronic Inhibition of cGMP phosphodiesterase 5A improves diabetic cardiomyopathy: a randomized, controlled clinical trial using magnetic resonance imaging with myocardial tagging. Circulation. 2012;125:2323–2333. doi: 10.1161/CIRCULATIONAHA.111.063412. [DOI] [PubMed] [Google Scholar]
- Goldie L.C., Nix M.K., Hirschi K.K. Embryonic vasculogenesis and hematopoietic specification. Organogenesis. 2008;4:257–263. doi: 10.4161/org.4.4.7416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goldstein I., Lue T.F., Padma-Nathan H., Rosen R.C., Steers W.D., Wicker P.A. Oral sildenafil in the treatment of erectile dysfunction. Sildenafil Study Group. New Engl. J. Med. 1998;338:1397–1404. doi: 10.1056/NEJM199805143382001. [DOI] [PubMed] [Google Scholar]
- Gonzalez R.R., Kaplan S.A. Tadalafil for the treatment of lower urinary tract symptoms in men with benign prostatic hyperplasia. Expert Opin. Drug Metab. Toxicol. 2006;2:609–617. doi: 10.1517/17425255.2.4.609. [DOI] [PubMed] [Google Scholar]
- Goundry B., Bell L., Langtree M., Moorthy A. Diagnosis and management of Raynaud’s phenomenon. BMJ. 2012;344:e289. doi: 10.1136/bmj.e289. [DOI] [PubMed] [Google Scholar]
- Hanson K.A., Burns F., Rybalkin S.D., Miller J.W., Beavo J., Clarke W.R. Developmental changes in lung cGMP phosphodiesterase-5 activity, protein, and message. Am. J. Respir. Crit. Care Med. 1998;158:279–288. doi: 10.1164/ajrccm.158.1.9711042. [DOI] [PubMed] [Google Scholar]
- Hidaka H., Asano T. Human blood platelet 3’: 5’-cyclic nucleotide phosphodiesterase. Isolation of low-Km and high-Km phosphodiesterase. Biochim. Biophys. Acta. 1976;429:485–497. doi: 10.1016/0005-2744(76)90296-5. [DOI] [PubMed] [Google Scholar]
- Hungerford J.E., Owens G.K., Argraves W.S., Little C.D. Development of the aortic vessel wall as defined by vascular smooth muscle and extracellular matrix markers. Dev. Biol. 1996;178:375–392. doi: 10.1006/dbio.1996.0225. [DOI] [PubMed] [Google Scholar]
- Jannini E.A., Isidori A.M., Gravina G.L., Aversa A., Balercia G., Bocchio M., Boscaro M., Carani C., Corona G., Fabbri A., Foresta C., Forti G., Francavilla S., Granata A.R., Maggi M., Mansani R., Palego P., Spera G., Vetri M., Lenzi A., Endotrial Study, G The ENDOTRIAL study: a spontaneous, open-label, randomized, multicenter, crossover study on the efficacy of sildenafil, tadalafil, and vardenafil in the treatment of erectile dysfunction. J. Sex. Med. 2009;6:2547–2560. doi: 10.1111/j.1743-6109.2009.01375.x. [DOI] [PubMed] [Google Scholar]
- Jannini E.A., DeRogatis L.R., Chung E., Brock G.B. How to evaluate the efficacy of the phosphodiesterase type 5 inhibitors. J. Sex. Med. 2012;9:26–33. doi: 10.1111/j.1743-6109.2011.02611.x. [DOI] [PubMed] [Google Scholar]
- Jeong K.H., Lee T.W., Ihm C.G., Lee S.H., Moon J.Y., Lim S.J. Effects of sildenafil on oxidative and inflammatory injuries of the kidney in streptozotocin-induced diabetic rats. Am. J. Nephrol. 2009;29:274–282. doi: 10.1159/000158635. [DOI] [PubMed] [Google Scholar]
- Kario K., Yamasaki K., Yagi K., Tsukamoto M., Yamazaki S., Okawara Y., Tomitani N., Kanegae H. Effect of suvorexant on nighttime blood pressure in hypertensive patients with insomnia: the SUPER-1 study. J. Clin. Hypertens. 2019;21:896–903. doi: 10.1111/jch.13505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kass D.A., Champion H.C., Beavo J.A. Phosphodiesterase type 5: expanding roles in cardiovascular regulation. Circ. Res. 2007;101:1084–1095. doi: 10.1161/CIRCRESAHA.107.162511. [DOI] [PubMed] [Google Scholar]
- Kotera J., Fujishige K., Omori K. Immunohistochemical localization of cGMP-binding cGMP-specific phosphodiesterase (PDE5) in rat tissues. J. Histochem. Cytochem. 2000;48:685–693. doi: 10.1177/002215540004800512. [DOI] [PubMed] [Google Scholar]
- Krishnan U., Horn E.M. A contemporary approach to pulmonary arterial hypertension. Curr. Atheroscler. Rep. 2016;18:58. doi: 10.1007/s11883-016-0608-z. [DOI] [PubMed] [Google Scholar]
- Kuschner W.G. Sildenafil citrate therapy for pulmonary arterial hypertension. N. Engl. J. Med. 2006;354:1091–1093. author reply 1091-1093. [PubMed] [Google Scholar]
- Lacolley P., Regnault V., Segers P., Laurent S. Vascular smooth muscle cells and arterial stiffening: relevance in development, aging, and disease. Physiol. Rev. 2017;97:1555–1617. doi: 10.1152/physrev.00003.2017. [DOI] [PubMed] [Google Scholar]
- Lacolley P., Regnault V., Avolio A.P. Smooth muscle cell and arterial aging: basic and clinical aspects. Cardiovasc. Res. 2018;114:513–528. doi: 10.1093/cvr/cvy009. [DOI] [PubMed] [Google Scholar]
- Larsen W.J. 2nd ed. Churchill Livingstone; New York: 1998. Essentials of Human Embriology; pp. 191–193. [Google Scholar]
- Laursen M., Beck L., Kehler J., Christoffersen C.T., Bundgaard C., Mogensen S., Mow T.J., Pinilla E., Knudsen J.S., Hedegaard E.R., Grunnet M., Simonsen U. Novel selective PDE type 1 inhibitors cause vasodilatation and lower blood pressure in rats. Br. J. Pharmacol. 2017;174:2563–2575. doi: 10.1111/bph.13868. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le Lievre C.S., Le Douarin N.M. Mesenchymal derivatives of the neural crest: analysis of chimaeric quail and chick embryos. J. Embryol. Exp. Morphol. 1975;34:125–154. [PubMed] [Google Scholar]
- Lin C.S., Lau A., Tu R., Lue T.F. Expression of three isoforms of cGMP-binding cGMP-specific phosphodiesterase (PDE5) in human penile cavernosum. Biochem. Biophys. Res. Commun. 2000;268:628–635. doi: 10.1006/bbrc.2000.2187. [DOI] [PubMed] [Google Scholar]
- Liu H., Manganiello V., Waleh N., Clyman R.I. Expression, activity, and function of phosphodiesterases in the mature and immature ductus arteriosus. Pediatr. Res. 2008;64:477–481. doi: 10.1203/PDR.0b013e3181827c2c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Z., Xu X., Hu X., Lee S., Traverse J.H., Zhu G., Fassett J., Tao Y., Zhang P., dos Remedios C., Pritzker M., Hall J.L., Garry D.J., Chen Y. Oxidative stress regulates left ventricular PDE5 expression in the failing heart. Circulation. 2010;121:1474–1483. doi: 10.1161/CIRCULATIONAHA.109.906818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majesky M.W., Dong X.R., Regan J.N., Hoglund V.J. Vascular smooth muscle progenitor cells: building and repairing blood vessels. Circ. Res. 2011;108:365–377. doi: 10.1161/CIRCRESAHA.110.223800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melehan K.L., Hoyos C.M., Hamilton G.S., Wong K.K., Yee B.J., McLachlan R.I., O’Meagher S., Celermajer D., Ng M.K., Grunstein R.R., Liu P.Y. Randomized trial of CPAP and vardenafil on erectile and arterial function in men with obstructive sleep apnea and erectile dysfunction. J. Clin. Endocrinol. Metab. 2018;103:1601–1611. doi: 10.1210/jc.2017-02389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mergia E., Stegbauer J. Role of phosphodiesterase 5 and cyclic GMP in hypertension. Curr. Hypertens. Rep. 2016;18:39. doi: 10.1007/s11906-016-0646-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morano S., Mandosi E., Fallarino M., Gatti A., Tiberti C., Sensi M., Gandini L., Buchetti B., Lenti L., Jannini E.A., Lenzi A. Antioxidant treatment associated with sildenafil reduces monocyte activation and markers of endothelial damage in patients with diabetic erectile dysfunction: a double-blind, placebo-controlled study. Eur. Urol. 2007;52:1768–1774. doi: 10.1016/j.eururo.2007.04.042. [DOI] [PubMed] [Google Scholar]
- Morelli A., Filippi S., Mancina R., Luconi M., Vignozzi L., Marini M., Orlando C., Vannelli G.B., Aversa A., Natali A., Forti G., Giorgi M., Jannini E.A., Ledda F., Maggi M. Androgens regulate phosphodiesterase type 5 expression and functional activity in corpora cavernosa. Endocrinology. 2004;145:2253–2263. doi: 10.1210/en.2003-1699. [DOI] [PubMed] [Google Scholar]
- Mullershausen F., Russwurm M., Thompson W.J., Liu L., Koesling D., Friebe A. Rapid nitric oxide-induced desensitization of the cGMP response is caused by increased activity of phosphodiesterase type 5 paralleled by phosphorylation of the enzyme. J. Cell Biol. 2001;155:271–278. doi: 10.1083/jcb.200107001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nachtnebel A., Stollberger C., Ehrlich M., Finsterer J. Aortic dissection after sildenafil-induced erection. South Med. J. 2006;99:1151–1152. doi: 10.1097/01.smj.0000240732.65859.aa. [DOI] [PubMed] [Google Scholar]
- Ockaili R., Salloum F., Hawkins J., Kukreja R.C. Sildenafil (Viagra) induces powerful cardioprotective effect via opening of mitochondrial K(ATP) channels in rabbits. Am. J. Physiol. Heart Circ. Physiol. 2002;283:H1263–1269. doi: 10.1152/ajpheart.00324.2002. [DOI] [PubMed] [Google Scholar]
- Olmestig J.N.E., Marlet I.R., Hainsworth A.H., Kruuse C. Phosphodiesterase 5 inhibition as a therapeutic target for ischemic stroke: a systematic review of preclinical studies. Cell. Signal. 2017;38:39–48. doi: 10.1016/j.cellsig.2017.06.015. [DOI] [PubMed] [Google Scholar]
- Owens G.K., Kumar M.S., Wamhoff B.R. Molecular regulation of vascular smooth muscle cell differentiation in development and disease. Physiol. Rev. 2004;84:767–801. doi: 10.1152/physrev.00041.2003. [DOI] [PubMed] [Google Scholar]
- Paauw N.D., Terstappen F., Ganzevoort W., Joles J.A., Gremmels H., Lely A.T. Sildenafil during pregnancy: a preclinical meta-analysis on fetal growth and maternal blood pressure. Hypertension. 2017;70:998–1006. doi: 10.1161/HYPERTENSIONAHA.117.09690. [DOI] [PubMed] [Google Scholar]
- Park K., Moreland R.B., Goldstein I., Atala A., Traish A. Sildenafil inhibits phosphodiesterase type 5 in human clitoral corpus cavernosum smooth muscle. Biochem. Biophys. Res. Commun. 1998;249:612–617. doi: 10.1006/bbrc.1998.9206. [DOI] [PubMed] [Google Scholar]
- Pauls M.M.H., Clarke N., Trippier S., Betteridge S., Howe F.A., Khan U., Kruuse C., Madigan J.B., Moynihan B., Pereira A.C., Rolfe D., Rostrup E., Haig C.E., Barrick T.R., Isaacs J.D., Hainsworth A.H. Perfusion by Arterial Spin labelling following Single dose Tadalafil in Small vessel disease (PASTIS): study protocol for a randomised controlled trial. Trials. 2017;18:229. doi: 10.1186/s13063-017-1973-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pauls M.M., Moynihan B., Barrick T.R., Kruuse C., Madigan J.B., Hainsworth A.H., Isaacs J.D. The effect of phosphodiesterase-5 inhibitors on cerebral blood flow in humans: a systematic review. J. Cereb. Blood Flow Metab. 2018;38:189–203. doi: 10.1177/0271678X17747177. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pofi R., Fiore D., De Gaetano R., Panio G., Gianfrilli D., Pozza C., Barbagallo F., Xiang Y.K., Giannakakis K., Morano S., Lenzi A., Naro F., Isidori A.M., Venneri M.A. Phosphodiesterase-5 inhibition preserves renal hemodynamics and function in mice with diabetic kidney disease by modulating miR-22 and BMP7. Sci. Rep. 2017;7:44584. doi: 10.1038/srep44584. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pokreisz P., Vandenwijngaert S., Bito V., Van den Bergh A., Lenaerts I., Busch C., Marsboom G., Gheysens O., Vermeersch P., Biesmans L., Liu X., Gillijns H., Pellens M., Van Lommel A., Buys E., Schoonjans L., Vanhaecke J., Verbeken E., Sipido K., Herijgers P., Bloch K.D., Janssens S.P. Ventricular phosphodiesterase-5 expression is increased in patients with advanced heart failure and contributes to adverse ventricular remodeling after myocardial infarction in mice. Circulation. 2009;119:408–416. doi: 10.1161/CIRCULATIONAHA.108.822072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Porst H., Rosen R., Padma-Nathan H., Goldstein I., Giuliano F., Ulbrich E., Bandel T. The efficacy and tolerability of vardenafil, a new, oral, selective phosphodiesterase type 5 inhibitor, in patients with erectile dysfunction: the first at-home clinical trial. Int. J. Impot. Res. 2001;13:192–199. doi: 10.1038/sj.ijir.3900713. [DOI] [PubMed] [Google Scholar]
- Prasad S., Wilkinson J., Gatzoulis M.A. Sildenafil in primary pulmonary hypertension. N. Engl. J. Med. 2000;343:1342. doi: 10.1056/NEJM200011023431814. [DOI] [PubMed] [Google Scholar]
- Risau W. Mechanisms of angiogenesis. Nature. 1997;386:671–674. doi: 10.1038/386671a0. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Iturbe B., Ferrebuz A., Vanegas V., Quiroz Y., Espinoza F., Pons H., Vaziri N.D. Early treatment with cGMP phosphodiesterase inhibitor ameliorates progression of renal damage. Kidney Int. 2005;68:2131–2142. doi: 10.1111/j.1523-1755.2005.00669.x. [DOI] [PubMed] [Google Scholar]
- Rossi R., Nuzzo A., Lattanzi A., Coppi F., Modena M.G. Sildenafil improves endothelial function in patients with pulmonary hypertension. Pulm. Pharmacol. Ther. 2008;21:172–177. doi: 10.1016/j.pupt.2007.01.008. [DOI] [PubMed] [Google Scholar]
- Ruzinova M.B., Schoer R.A., Gerald W., Egan J.E., Pandolfi P.P., Rafii S., Manova K., Mittal V., Benezra R. Effect of angiogenesis inhibition by Id loss and the contribution of bone-marrow-derived endothelial cells in spontaneous murine tumors. Cancer Cell. 2003;4:277–289. doi: 10.1016/s1535-6108(03)00240-x. [DOI] [PubMed] [Google Scholar]
- Saenz de Tejada I., Angulo J., Cuevas P., Fernandez A., Moncada I., Allona A., Lledo E., Korschen H.G., Niewohner U., Haning H., Pages E., Bischoff E. The phosphodiesterase inhibitory selectivity and the in vitro and in vivo potency of the new PDE5 inhibitor vardenafil. Int. J. Impot. Res. 2001;13:282–290. doi: 10.1038/sj.ijir.3900726. [DOI] [PubMed] [Google Scholar]
- Sahara M., Sata M., Morita T., Nakajima T., Hirata Y., Nagai R. A phosphodiesterase-5 inhibitor vardenafil enhances angiogenesis through a protein kinase G-dependent hypoxia-inducible factor-1/vascular endothelial growth factor pathway. Arterioscler. Thromb. Vasc. Biol. 2010;30:1315–1324. doi: 10.1161/ATVBAHA.109.201327. [DOI] [PubMed] [Google Scholar]
- Salloum F.N., Chau V.Q., Hoke N.N., Abbate A., Varma A., Ockaili R.A., Toldo S., Kukreja R.C. Phosphodiesterase-5 inhibitor, tadalafil, protects against myocardial ischemia/reperfusion through protein-kinase g-dependent generation of hydrogen sulfide. Circulation. 2009;120:S31–36. doi: 10.1161/CIRCULATIONAHA.108.843979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos-Silva A.J., Cairrao E., Morgado M., Alvarez E., Verde I. PDE4 and PDE5 regulate cyclic nucleotides relaxing effects in human umbilical arteries. Eur. J. Pharmacol. 2008;582:102–109. doi: 10.1016/j.ejphar.2007.12.017. [DOI] [PubMed] [Google Scholar]
- Sarifakioglu E., Degim I.T., Gorpelioglu C. Determination of the sildenafil effect on alopecia areata in childhood: an open-pilot comparison study. J. Dermatolog. Treat. 2006;17:235–237. doi: 10.1080/09546630600895003. [DOI] [PubMed] [Google Scholar]
- Shan X., Quaile M.P., Monk J.K., French B., Cappola T.P., Margulies K.B. Differential expression of PDE5 in failing and nonfailing human myocardium. Circ. Heart Fail. 2012;5:79–86. doi: 10.1161/CIRCHEARTFAILURE.111.961706. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharp A., Cornforth C., Jackson R., Harrold J., Turner M.A., Kenny L.C., Baker P.N., Johnstone E.D., Khalil A., von Dadelszen P., Papageorghiou A.T., Alfirevic Z. Maternal sildenafil for severe fetal growth restriction (STRIDER): a multicentre, randomised, placebo-controlled, double-blind trial. Lancet Child Adolesc Health. 2018;2:93–102. doi: 10.1016/S2352-4642(17)30173-6. [DOI] [PubMed] [Google Scholar]
- Sheng M., Lu H., Liu P., Li Y., Ravi H., Peng S.L., Diaz-Arrastia R., Devous M.D., Womack K.B. Sildenafil improves vascular and metabolic function in patients with Alzheimer’s disease. J. Alzheimers Dis. 2017;60:1351–1364. doi: 10.3233/JAD-161006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sher G., Fisch J.D. Vaginal sildenafil (Viagra): a preliminary report of a novel method to improve uterine artery blood flow and endometrial development in patients undergoing IVF. Hum. Reprod. 2000;15:806–809. doi: 10.1093/humrep/15.4.806. [DOI] [PubMed] [Google Scholar]
- Sokanovic S.J., Capo I., Medar M.M., Andric S.A., Kostic T.S. Long-term inhibition of PDE5 ameliorates aging-induced changes in rat testis. Exp. Gerontol. 2018;108:139–148. doi: 10.1016/j.exger.2018.04.007. [DOI] [PubMed] [Google Scholar]
- Stegbauer J., Friedrich S., Potthoff S.A., Broekmans K., Cortese-Krott M.M., Quack I., Rump L.C., Koesling D., Mergia E. Phosphodiesterase 5 attenuates the vasodilatory response in renovascular hypertension. PLoS One. 2013;8 doi: 10.1371/journal.pone.0080674. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Takimoto E., Champion H.C., Li M., Belardi D., Ren S., Rodriguez E.R., Bedja D., Gabrielson K.L., Wang Y., Kass D.A. Chronic inhibition of cyclic GMP phosphodiesterase 5A prevents and reverses cardiac hypertrophy. Nat. Med. 2005;11:214–222. doi: 10.1038/nm1175. [DOI] [PubMed] [Google Scholar]
- Tiryakioglu S.K., Tiryakioglu O., Turan T., Kumbay E. Aortic dissection due to sildenafil abuse. Interact. Cardiovasc. Thorac. Surg. 2009;9:141–143. doi: 10.1510/icvts.2009.205849. [DOI] [PubMed] [Google Scholar]
- Tsai B.M., Turrentine M.W., Sheridan B.C., Wang M., Fiore A.C., Brown J.W., Meldrum D.R. Differential effects of phosphodiesterase-5 inhibitors on hypoxic pulmonary vasoconstriction and pulmonary artery cytokine expression. Ann. Thorac. Surg. 2006;81:272–278. doi: 10.1016/j.athoracsur.2005.06.040. [DOI] [PubMed] [Google Scholar]
- Ungvari Z., Tarantini S., Donato A.J., Galvan V., Csiszar A. Mechanisms of vascular aging. Circ. Res. 2018;123:849–867. doi: 10.1161/CIRCRESAHA.118.311378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van der Zee R., Murohara T., Luo Z., Zollmann F., Passeri J., Lekutat C., Isner J.M. Vascular endothelial growth factor/vascular permeability factor augments nitric oxide release from quiescent rabbit and human vascular endothelium. Circulation. 1997;95:1030–1037. doi: 10.1161/01.cir.95.4.1030. [DOI] [PubMed] [Google Scholar]
- Vecchio M., Navaneethan S.D., Johnson D.W., Lucisano G., Graziano G., Saglimbene V., Ruospo M., Querques M., Jannini E.A., Strippoli G.F. Interventions for treating sexual dysfunction in patients with chronic kidney disease. Cochrane Database Syst. Rev. 2010 doi: 10.1002/14651858.CD007747.pub2. [DOI] [PubMed] [Google Scholar]
- Vlachopoulos C., Ioakeimidis N., Rokkas K., Stefanadis C. Cardiovascular effects of phosphodiesterase type 5 inhibitors. J. Sex. Med. 2009;6:658–674. doi: 10.1111/j.1743-6109.2008.01107.x. [DOI] [PubMed] [Google Scholar]
- Wang R., Burnett A.L., Heller W.H., Omori K., Kotera J., Kikkawa K., Yee S., Day W.W., DiDonato K., Peterson C.A. Selectivity of avanafil, a PDE5 inhibitor for the treatment of erectile dysfunction: implications for clinical safety and improved tolerability. J. Sex. Med. 2012;9:2122–2129. doi: 10.1111/j.1743-6109.2012.02822.x. [DOI] [PubMed] [Google Scholar]
- Wharton J., Strange J.W., Moller G.M., Growcott E.J., Ren X., Franklyn A.P., Phillips S.C., Wilkins M.R. Antiproliferative effects of phosphodiesterase type 5 inhibition in human pulmonary artery cells. Am. J. Respir. Crit. Care Med. 2005;172:105–113. doi: 10.1164/rccm.200411-1587OC. [DOI] [PubMed] [Google Scholar]
- Yancopoulos G.D., Davis S., Gale N.W., Rudge J.S., Wiegand S.J., Holash J. Vascular-specific growth factors and blood vessel formation. Nature. 2000;407:242–248. doi: 10.1038/35025215. [DOI] [PubMed] [Google Scholar]
- Zhang R., Wang L., Zhang L., Chen J., Zhu Z., Zhang Z., Chopp M. Nitric oxide enhances angiogenesis via the synthesis of vascular endothelial growth factor and cGMP after stroke in the rat. Circ. Res. 2003;92:308–313. doi: 10.1161/01.res.0000056757.93432.8c. [DOI] [PubMed] [Google Scholar]
- Zhang L., Zhang R.L., Wang Y., Zhang C., Zhang Z.G., Meng H., Chopp M. Functional recovery in aged and young rats after embolic stroke: treatment with a phosphodiesterase type 5 inhibitor. Stroke. 2005;36:847–852. doi: 10.1161/01.STR.0000158923.19956.73. [DOI] [PubMed] [Google Scholar]