Abstract
Coronaviruses (CoVs) are large positive stranded enveloped RNA viruses that generally cause enteric and respiratory diseases in humans and in animals. Most human CoVs have recently attracted global attention to their lethal potential and great infectious capacity. A highly pathogenic CoV, called COVID-19 or SARS‐CoV-2, dramatically emerged in December 2019 in Wuhan, China. This new CoV has caused severe pneumonia in China and rapidly spreads around the world, the COVID-19 pandemic. Growing evidence pieces show that viruses, such as CoVs, can enter the central nervous system from different pathways and inducing neurotoxicity. Therefore, it is urgent to make clear whether SARS-CoV-2 has access to the central nervous system and can cause direct neuronal effects. Moreover, a brain–lung–brain axis is been proposed from the scientific community where severe neurological dysfunction and injury are associated with lung injury, and vice versa. In this axis, virus-induced inflammation and oxidative stress could be the common mechanisms responsible for CoV neurological symptoms. Therefore, is important to make clear whether SARS-CoV-2 lung damage can cause direct or indirect neuronal effects.
Keywords: Coronaviruses, COVID-19, SARS-CoV-2, Central nervous system, Brain–lung–brain axis
1. Introduction
COVID-19 is much more than a health emergency, it has the potential to create devastating social and economic crises that will leave deep scars. Nations are rushing to slow the diffusion of the virus by treating patients, limiting travel, quarantining citizens, canceling large matches, concerts, and close schools. People with COVID-19 generally develop respiratory symptoms but the increasing evidence shows that some patients with a severe infection also develop neurological ailments like confusion, stroke, seizure, or loss of smell and taste. Severe acute respiratory syndrome (SARS) is caused by a coronavirus (CoV), SARS-CoV-2 is a novel coronavirus identified as the cause of coronavirus disease 2019 (COVID-19), which is enveloped non-segmented positive-sense RNA virus (subgenus sarbecovirus, Orthocoronavirinae subfamily) (Zhu et al., 2020). Based on their morphology as spherical virions with a core shell and surface projections resembling a solar corona, they are termed coronaviruses. RNA viruses, such as CoV, infect both humans and animals (Cui et al., 2019). Genetic analysis shows that SARS-CoV-2 has a highly homologous sequence with SARS-CoV (79.6 %). Furthermore, SARS-CoV-2 is 96 % identical at the whole-genome level to a bat coronavirus (Zhou et al., 2020). Moreover, the entry of COVID-19 into human host cells has been identified to use the same receptor as SARS‐CoV (Yu et al., 2020). The recent COVID-19 epidemic has been started in Wuhan, China; and this highly contagious disease has spread throughout China and other part of the world (Table 1 ) (Huang et al., 2020; Song et al., 2019; Lu et al., 2020). The symptoms of COVID-19 infection usually appear after an incubation period of about five days.
Table 1.
Coronavirus disease 2019 situation report. Data as received by WHO from national authorities by 10:00 CEST, 3 May 2020.
| Globally | RISK ASSESSMENT |
| 3 349 786 cases (82 763) | Global Level Very High |
| 238 628 deaths (8657) | |
| European Region | Region of the Americas |
| 1 518 895 cases (27 032) | 1 384 641 cases (44 050) |
| 142 667 deaths (2081) | 78 409 deaths (6213) |
| Western Pacific Region | Eastern Mediterranean Region |
| 151 444 cases (1041) | 200 609 cases (5618) |
| 6229 deaths (63) | 7871 deaths (130) |
| African Region | South-East Asia Region |
| 29 438 cases (1465) | 64 047 cases (3557) |
| 1064 deaths (51) | 2375 deaths (119) |
The adult populations, called sensitive, are at significantly increased risk of mortality (Wang et al., 2020). This effect is due to changes in aging; age-related comorbidity conditions such as heart and lung disease, diabetes, and dementia. The presence of multiple diseases in older patients may be considered as a mark of frailty, which increases the person's vulnerability to stress and impairs the multisystemic compensatory effort to restore homeostasis. The immune system of older adults presents numerous age-related changes, indicated as immune senescence (Nikolich-Zugich, 2018). These changes concern both the innate (Solana et al., 2006) and adaptive (Goronzy et al., 2012; Kogut et al., 2012) immune systems, as well as the immuno-response in time and space (Nikolich-Zugich, 2018) which works effectively in young but deteriorates with age. Neurotropic and neuroinvasive capabilities of coronaviruses have been described in humans. Upon infection, coronavirus enters the CNS, causing inflammation and demyelination (Bohmwald et al., 2018). Recent studies discussed the neuroinvasive potential of SARS-CoV-2; in fact, some infected subjects did show neurological effects. A recent multicenter study has identified frequent (i.e. more than 80 % of the cohort) gustatory and olfactory impairments in included patients. Moreover, studies have shown that lung damage can be associated with brain injury and neurocognitive dysfunction and vice versa, indicating the existence of a brain-lung-brain axis (Stevens and Puybasset, 2011). In this brief review, we will discuss the evidence on the occurrence of central nervous system (CNS) involvement and neurological manifestations in patients with COVID-19. Moreover, we have reported evidence indicating whether COVID-19 might cause direct and/or indirect effect on the neuronal system or whether the eventual virus-induced neuronal damage may cause a respiratory syndrome.
2. Direct effects of COVID-19 on brain
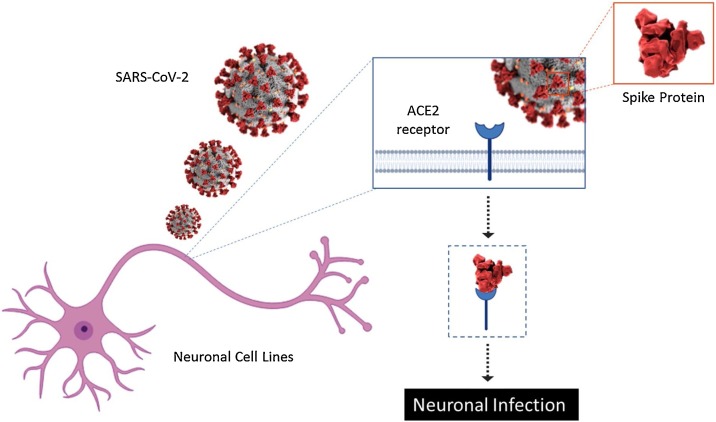
Increasing evidence showed that coronaviruses are not always limited to the respiratory system and that they may invade the CNS. In fact, detection of some RNA of human-coronavirus in human brain samples clearly demonstrates that these respiratory pathogens are naturally neuroinvasive in humans and suggests that they establish a persistent infection in human CNS (Arbour et al., 2000). Different evidence, in vitro, demonstrated the susceptibility of various human neural cell lines to infection by human CoVs. Primary cultures of human astrocytes and microglia showed susceptibility to infection by the OC43 strain of human CoV (Bonavia et al., 1997). Moreover, Arbour and collaborators reported an acute and persistent Infection in Human Neural Cell Lines by Human CoV. They characterized the susceptibility of various human neural cell lines (astrocytoma cell lines U-87 MG, U-373 MG, and GL-15, as well as neuroblastoma SK-N-SH, neuroglioma H4, oligodendrocytic MO3.13, and the CHME-5 immortalized fetal microglial cell lines) to an acute infection by Human CoV (Arbour et al., 1999). Spike proteins (S) are essential for viral entry into host cells. S protein of SARS-CoV-2 bound to the cell receptor ACE2 (angiotensin-converting enzyme 2), which is present on non-immune cells, such as respiratory and intestinal epithelial cells, endothelial cells, kidney cells (renal tubules) and cerebral neurons and immune cells, such as alveolar monocytes/macrophages causing his entry (Fig. 1 ) (Magrone et al., 2020).
Fig. 1.
Schematic representation of the mechanism to infection of the SARS-CoV-2 in neuronal cell lines.
Therefore, the neuroinvasive capacity has been demonstrated as a common characteristic of CoVs, and on the base of similarity between SARS‐CoV and SARS-CoV-2, it is quite likely that SARS-CoV-2 also possesses a like potential (Li et al., 2020). Although the etiology of COVID-19-induced neurodegeneration remains unknown or poorly understood, it has been suggested that these human respiratory pathogens could be associated with the initiation or exacerbation of neurological diseases (Desforges et al., 2014). Chen and collaborators have reported that, in Wuhan city, some subjects infected with COVID-19 did show neurological effects such as headache (about 8%), nausea and vomiting (1%) (Chen et al., 2020). Moreover, in one recent paper, Chinese scientists noted that on 214 COVID‐19 patients up to 36.4 % reported neurologic symptoms such as acute cerebrovascular diseases, consciousness impairment, and skeletal muscle symptoms (Mao et al., 2020). Moriguchi and collaborators report the first case of meningitis associated with COVID-19. The RNA of SARS-CoV-2 was not identified in the nasopharyngeal swab but was detected in cerebrospinal fluid by gene sequencing. A brain Magnetic Resonance Imaging (MRI) showed hyperintensity along the wall of right lateral ventricle and hyperintense signal changes in the right mesial temporal lobe and hippocampus, suggesting the possibility of COVID-19 meningitis (Moriguchi et al., 2020).
Viruses may enter the CNS by different routes involving the vasculature, the olfactory and trigeminal nerves, the cerebrospinal fluid, and the lymphatic system (Desforges et al., 2020). However, the exact route by which SARS‐CoV enters the CNS has still not been identified. The hematogenous or lymphatic route seems improbably, especially in the early stage of infection, since almost no virus particle was detected in the non-neuronal cells in the infected brain areas [Ding et al., 2004; Gu et al., 2005; Xu et al., 2005). Different evidence shows that CoVs as first step invade peripheral nerve terminals, and successively their obtained access to the CNS by synapse‐connected route (trans‐synaptic transfer) (Li et al., 2013). This route has been well documented for the avian bronchitis virus (Matsuda et al., 2004; Chasey and Alexander, 1976). Recently Butowt and Bilinska proposed that the olfactory epithelium from the nasal cavity could be a more appropriate tissue for detection of SARS-CoV-2 virus at the earliest stages, with respect to commonly used sputum or nasopharyngeal swabs (Butowt and Bilinska, 2020). They point out that the different types of non-neuronal cells present in the olfactory epithelium, which express the ACE2 receptor, could facilitate the binding, replication and accumulation of SARS-CoV-2 (Butowt and Bilinska, 2020). This may be the underlying mechanism for the recently reported cases of smell dysfunction in patients with COVID-19. The authors proposed the possibility that viral brain infection starts from the olfactory neurons (Butowt and Bilinska, 2020). Nose-to-brain route is use in biomedicine to brain drug delivery, avoiding the difficulties due to the BBB and the problems connected with systemic administration (Picone et al., 2018). The circulation of the virus in the bloodstream its interaction with the capillary endothelium and the subsequent formation of the viral particles could determine the destruction of the brain capillary endothelium allowing the access of the virus to the brain. Destruction of the endothelium of the brain capillaries and hemorrhage in the brain tissue can have lethal effects long before neuronal damage occurs (Mannan Baig et al., 2020).
Therefore, viral infection in brain may be accompanied by vascular endothelium dysfunction and brain neuroinflammation, especially in frail or older patients (Toth et al., 2017). These changes may be much worse under the hypoxic conditions of acute respiratory distress syndrome caused by COVID-19. Some authors suggest that the neuroinvasive potential of SARS-CoV-2 could play a role in the respiratory failure of COVID‐19 patients since the central respiratory failure progresses rapidly after the SARS-CoV-2 entry into the CNS and damages the brainstem where the pneumotaxic center is located (Li et al., 2020). In contrast, Turtle says that respiratory failure caused by pneumonia is clinically distinct from that caused by brain failure and the possibility of CNS entry by SARS-CoV-2 remains plausible, but if so, this is a very rare event (Turtle, 2020). However, another relevant factor linking the virus to neurological disease is that in multiple sclerosis (MS) both viruses and environmental factors are considered important for the etiopathology of disease. For this aim since 1993 viruses, have been the subject of extensive research and discussion (Kurtzke, 1993). Evidence for the presence of coronaviral genes in human brain tissue of MS subject has been found (Murray et al., 1992; Dessau et al., 2001).
3. Potential indirect effects of COVID-19 on brain
The development of a systemic inflammatory response syndrome is closely linked to severe viral infections. Virus-induced oxidative stress could be mediated by an early phase of liberation of pro-inflammatory cytokines. In fact, the acute inflammation there is an imbalance between increased production of radicals and the availability of antioxidant molecules and may result in increased systemic oxidative stress (Biswas, 2016). The brain is especially vulnerable to reactive oxygen species (ROS) because this tissue is a major metabolizer of oxygen and yet has relatively feeble protective antioxidant mechanisms. The death of neurons is an important point of the pathophysiological process of nervous system diseases. Therefore, inflammation leads to increased levels of ROS, which can induce oxidative stress at the site of inflammation (Zuo et al., 2019). On the other hand, a number of reactive oxygen/nitrogen species can initiate an intracellular signaling cascade that enhances pro-inflammatory gene expression (Lorenzen et al., 2017). Thus, inflammation and oxidative stress are closely related pathophysiological events (Lugrin et al., 2014). The brain–lung–brain axis has been proposed from scientific community where severe neurological dysfunction and injury are associated to lung injury (Stevens and Puybasset, 2011). Chronic obstructive pulmonary disease (COPD) is a progressive condition characterized by airflow limitation associated with an abnormal inflammatory response. In this pathology, several systemic biomarkers of oxidative stress are available, including ROS themselves (Zinellu et al., 2016).
The examination of lung specimens from mild COVID-19 patients showed edema, proteinaceous exudate with globules, patchy inflammatory cellular infiltration and moderate formation of hyaline membranes (Tian et al., 2020). In a recent review, Channappanavar and Perlman have examined that in the SARS-CoV infected animal model, marked inflammatory and immune responses may activate a “cytokine storm”, and apoptosis responses and even death (Channappanavar and Perlman, 2017). Specifically, in the blood of patients infected with SARS-CoV-2, there is a marked increase in interleukin 1β (IL-1β), interferon γ (IFN-γ), interferon-inducible protein 10 (IP-10), and monocyte chemoattractant protein 1(MCP-1), as well as IL-4 and IL-10 when compared to that of SARS patients (Huang et al., 2020). Collectively, the finding indicates that inflammation is a major feature in COVID-19 patients. Thus, excessive inflammation, depressed immune system, and an activate cytokine storm substantially contribute to the negative consequences of SARS-CoV-2. An excessive immune system response could induced organs failure, (cardiac, hepatic and renal systems) before than neurodegeneration (Zirui Tay et al., 2020).
In a cellular model, respiratory syncytial virus infection, induces ROS production and consequently oxidative stress. In fact, the findings suggest an imbalance between ROS production and antioxidant cellular defenses through the negative modulation of superoxide dismutase (SOD) 1, 2, and 3, catalase, glutathione peroxidase (GPx), and glutathione S-transferase (GST) (Hosakote et al., 2009). Taken together, these data suggest that lung inflammation could determine systemic oxidative stress. In particular, the high inflammatory capacity of SARS-CoV-2 could generate a high level of ROS able to damage the brain. In fact, systemic oxidative stress has an important role in neurodegenerative diseases (ND) etiology (Cervellati et al., 2020). Moreover, inflammatory damage to the Blood-Brain Barrier (BBB) surface has been linked to various neurological disorders and CNS infections. The neuroinflammatory signaling has been strongly linked to elevated levels of pro-inflammatory cytokines such as tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6) (Rochfort and Cummins, 2015) The brain is susceptible to minimal imbalances of the redox state due to its high energy and metabolic request, imbalances of the redox state favor tissue injury and neuroinflammatory mechanisms activation paving the way for neurodegeneration (Martínez Leo and Segura Campos, 2019; Nuzzo et al., 2014; Cevenini et al., 2010). Therefore, inflammation and oxidative stress systemic, induced by SARS-CoV-2 lung injury, could has effect in CNS causing neuronal dysfunction.
Another particular aspect of indirect effect of COVID-19 infection is the regulation of circulating cytotoxic lymphocytes such as natural killer (NK) cells, in fact these cells are necessary for the control of general viral infection (Di Bona et al., 2014). In particular, Zheng and Collaborators showed that the total number of NK and CD8 + T cells was decreased markedly in patients with SARS-CoV-2 infection. The data highlight the importance of improving the immune response of NK cells and avoiding exhaustion of cytotoxic lymphocytes at the early stage of SARS-CoV-2 infection (Zheng et al., 2020). When a virus proliferates in tissue cells of lung this leads to alveolar gas exchange disorders causing hypoxia in the CNS, increasing anaerobic metabolism in the mitochondria of brain cells (Di Carlo et al., 2012; Abdennour et al., 2012). Hypoxia can cause cerebral vasodilation, swelling of brain cells, interstitial edema, cerebral blood flow obstruction, ischemia, and congestion (Guo et al., 2020). In this condition the brain function gradually deteriorates, drowsiness, and bulbar conjunctival edema (Guo et al., 2020). Patients with COVID-19 often suffer from severe hypoxia, hypoxia injury may cause subsequent nervous system damage [55]. In addition, for patients at particular risk of developing cerebrovascular disease, hypoxia may also induce the occurrence of acute cerebrovascular disease.
4. Conclusions
Although the SARS-CoV-2 brain infection and the related effects is not been demonstrated with certainty, this possibility remains plausible (direct brain effect). Moreover, on the other side, lung injuries could have effect on CNS causing neuronal dysfunction (indirect brain effect). For these considerations, we stress the need to begin the research on COVID-19 actions in the CNS (Fig. 2 ). Moreover, is very important to monitor both the early and long-term neurocognitive effects of COVID-19. Therefore, one possibility of counteracting the COVID-19 effects is the use of antiviral therapies combined with neuroprotective drugs. For example, using antiviral drugs that can cross the blood-brain barrier combined with agents that specifically target both inflammation and oxidative stress could be considered.
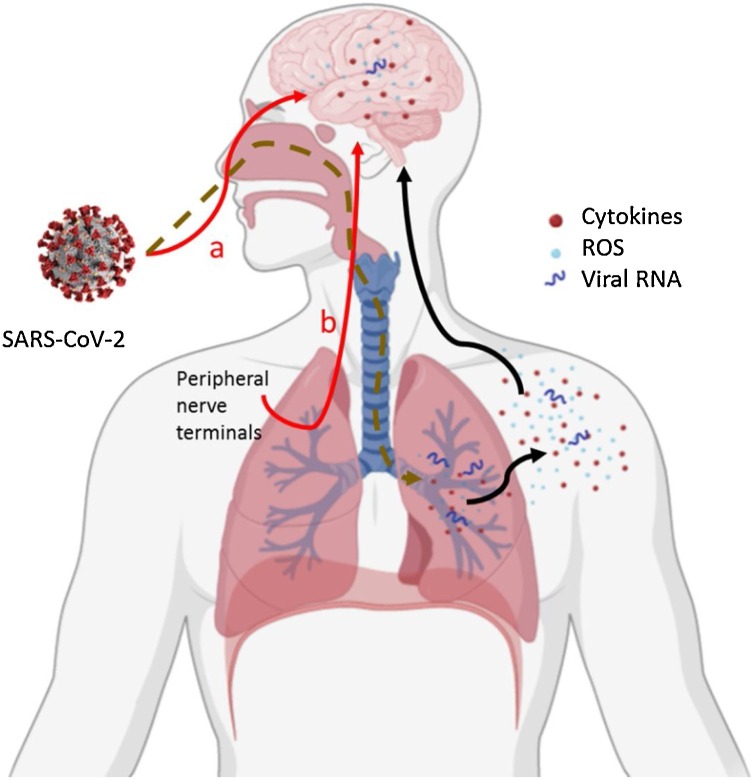
Fig. 2.
The possible routes through which SARS-CoV-2 to reach the brain. Red line: via olfactory epithelium (a) and/or after infection peripheral nerve terminals by trans‐synaptic transfer (b) inducing possible direct damage by neuro-inflammation and oxidative stress. Black line: After lung infection, the systemic inflammation and oxidative stress get to the CNS inducing possible indirect damage.
Funding
This research received no external funding.
Declaration of Competing Interest
The authors declare no conflict of interest.1
Footnotes
The image was generated by BioRender web software.
References
- Abdennour L., Zeghal C., Dème M., Puybasset L. Interaction brain-lungs. Ann. Fr. Anesth. Reanim. 2012;31:101–107. doi: 10.1016/j.annfar.2012.04.013. [DOI] [PubMed] [Google Scholar]
- Arbour N., Côté G., Lachance C., Tardieu M., Cashman N.R., Talbot P.J. Acute and persistent infection of human neural cell lines by human coronavirus OC43. J. Virol. 1999;73:3338–3350. doi: 10.1128/jvi.73.4.3338-3350.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arbour N., Day R., Newcombe J., Talbot P.J. Neuroinvasion by human respiratory coronaviruses. Virology. 2000;74:8913–8921. doi: 10.1128/jvi.74.19.8913-8921.2000. https://doi.org/110.1128/JVI.74.19.8913-8921.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Biswas S.K. Does the interdependence between oxidative stress and inflammation explain the antioxidant paradox? Oxid. Med. Cell. Longev. 2016:5698931. doi: 10.1155/2016/5698931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohmwald K., Galvez N., Ríos M., Kalergis A.M. Neurologic alterations due to respiratory virus infections. Front. Cell. Neurosci. 2018;12:386. doi: 10.3389/fncel.2018.00386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bonavia A., Arbour N., Yong V.W., Talbot P.J. Infection of primary cultures of human neural cells by human coronaviruses 229E and OC43. J. Virol. 1997;71:800–806. doi: 10.1128/jvi.71.1.800-806.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butowt R., Bilinska K. SARS-CoV-2: olfaction, brain infection, and the urgent need for clinical samples allowing earlier virus detection. ACS Chem. Neurosci. 2020;13 doi: 10.1021/acschemneuro.0c00172. [DOI] [PubMed] [Google Scholar]
- Cervellati C., Trentini A., Pecorelli A., Valacchi G. Inflammation in neurological disorders: the thin boundary between brain and periphery. Antioxid. Redox Signal. 2020:9. doi: 10.1089/ars.2020.8076. [DOI] [PubMed] [Google Scholar]
- Cevenini E., Caruso C., Candore G., Capri M., Nuzzo D., Duro G., Rizzo C., Colonna-Romano G., Lio D., Di Carlo D., Palmas M.G., Scurti M., Pini E., Franceschi C., Vasto S. Age-related inflammation: the contribution of different organs, tissues and systems. How to face it for therapeutic approaches. Curr. Pharm. Des. 2010;16:609–618. doi: 10.2174/138161210790883840. [DOI] [PubMed] [Google Scholar]
- Channappanavar R., Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017;39:529–539. doi: 10.1007/s00281-017-0629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chasey D., Alexander D.J. Morphogenesis of avian infectious bronchitis virus in primary chick kidney cells. Arch. Virol. 1976;52:101–111. doi: 10.1007/bf01317869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen N., Zhou M., Dong X., Qu J., Gong F., Han Y., Qiu Y., Wang J., Liu Y., Wei Y., et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;(395):507–513. doi: 10.1016/S0140-6736(20)30211-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cui J., Li F., Shi Z.L. Origin and evolution of pathogenic coronaviruses. Nat. Rev. Microbiol. 2019;17:181–192. doi: 10.1038/s41579-018-0118-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desforges M., Le Coupanec A., Brison E., Meessen-Pinard M., Talbot P.J. Neuroinvasive and neurotropic human respiratory coronaviruses: potential neurovirulent agents in humans. Adv. Exp. Med. Biol. 2014;807:75–96. doi: 10.1007/978-81-322-1777-0_6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Desforges M., Le Coupanec A., Dubeau P., Bourgouin A., Lajoie L., Dubé M., Talbot P.J. Human coronaviruses and other respiratory viruses: underestimated opportunistic pathogens of the central nervous system? Viruses. 2020;20 doi: 10.3390/v12010014. pii: E14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dessau R.B., Lisby G., Frederiksen J.L. Coronaviruses in brain tissue from patients with multiple sclerosis. Acta Neuropathol. 2001;101:601–604. doi: 10.1007/s004010000331. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Di Bona D., Scafidi V., Plaia A., Colomba C., Nuzzo D., Occhino C., Tuttolomondo A., Giammanco G., De Grazia S., Montalto G., Duro G., Cippitelli M., Caruso C. HLA and killer cell immunoglobulin-like receptors influence the natural course of CMV infection. J. Infect. Dis. 2014;210:1083–1089. doi: 10.1093/infdis/jiu226. [DOI] [PubMed] [Google Scholar]
- Di Carlo M., Giacomazza D., Picone P., Nuzzo D., San Biagio P.L. Are oxidative stress and mitochondrial dysfunction the key players in the neurodegenerative diseases? Free Radic. Res. 2012;46:1327–1338. doi: 10.3109/10715762.2012.714466. [DOI] [PubMed] [Google Scholar]
- Ding Y., He L., Zhang Q., Huang Z., Che X., Hou J., Wang H., Shen H., Qiu L., Li Z., Geng J., Cai J., Han H., Li X., Kang W., Weng D., Liang P., Jiang S. Organ distribution of severe acute respiratory syndrome (SARS) associated coronavirus (SARS‐CoV) in SARS patients: implications for pathogenesis and virus transmission pathways. J. Pathol. 2004;203:622–630. doi: 10.1002/path.1560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goronzy J.J., Li G., Yu M., Weyand C.M. Signaling pathways in aged T cells - a reflection of T cell differentiation, cell senescence and host environment. Semin. Immunol. 2012;24:365–372. doi: 10.1016/j.smim.2012.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gu J., Gong E., Zhang B., Zheng J., Gao Z., Zhong Y., Zou W., Zhan J., Wang S., Xie Z., Zhuang H., Wu B., Zhong H., Shao H., Fang W., Gao D., Pei F., Li X., He Z., Xu D., Shi X., Anderson V.M., Leong A.S. Multiple organ infection and the pathogenesis of SARS. J. Exp. Med. 2005;202:415–424. doi: 10.1084/jem.20050828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guo Y.R., Cao Q.D., Hong Z.S., Tan Y.Y., Chen S.D., Jin H.J., Tan K.S., Wang D.Y., Yan Y. The origin, transmission and clinical therapies on coronavirus disease 2019 (COVID-19) outbreak - an update on the status. Mil. Med. Res. 2020;13:11. doi: 10.1186/s40779-020-00240-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hosakote Y.M., Liu T., Castro S.M., Garofalo R.P., Casola A. Respiratory syncytial virus induces oxidative stress by modulating antioxidant enzymes. Am. J. Respir. Cell Mol. Biol. 2009;41:348–357. doi: 10.1165/rcmb.2008-0330OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London, England) 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kogut I., Scholz J.L., Cancro M.P., Cambier J.C. B cell maintenance and function in aging. Semin. Immunol. 2012;24:342–349. doi: 10.1016/j.smim.2012.04.004. [DOI] [PubMed] [Google Scholar]
- Kurtzke J.F. Epidemiologic evidence for multiple sclerosis as an infection. Clin. Microbiol. Rev. 1993;6:382–427. doi: 10.1128/cmr.6.4.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y.C., Bai W.Z., Hirano N., Hayashida T., Taniguchi T., Sugita Y., Tohyama K., Hashikawa T. Neurotropic virus tracing suggests a membranous‐coating‐mediated mechanism for transsynaptic communication. J. Comp. Neurol. 2013;521:203–212. doi: 10.1002/cne.23171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y.C., Bai W.Z., Hashikawa T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med. Virol. 2020;27 doi: 10.1002/jmv.25728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lorenzen I., Mullen L., Bekeschus S., Hanschmann E.M. Redox regulation of inflammatory processes is enzymatically controlled. Oxid. Med. Cell. Longev. 2017:8459402. doi: 10.1155/2017/8459402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu R., Zhao X., Li J., Niu P., Yang B., Wu H., Wang W., Song H., Huang B., Zhu N., Bi Y., Ma X., Zhan F., Wang L., Hu T., Zhou H., Hu Z., Zhou W., Zhao L., Chen J., Meng Y., Wang J., Lin Y., Yuan J., Xie Z., Ma J., Liu W.J., Wang D., Xu W., Holmes E.C., Gao G.F., Wu G., Chen W., Shi W., Tan W. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;(22):565–574. doi: 10.1016/S0140-6736(20)30251-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lugrin J., Rosenblatt-Velin N., Parapanov R., Liaudet L. The role of oxidative stress during inflammatory processes. Biol. Chem. 2014;395:203–230. doi: 10.1515/hsz-2013-0241. [DOI] [PubMed] [Google Scholar]
- Magrone T., Magrone M., Jirillo E. Focus on Receptors for Coronaviruses with Special Reference to Angiotensin-converting Enzyme 2 as a Potential Drug Target - A Perspective. Endocr. Metab. Immune Disord. Drug Targets. 2020;27 doi: 10.2174/1871530320666200427112902. [DOI] [PubMed] [Google Scholar]
- Mannan Baig A., Khaleeq A., Ali U., Syeda H. Evidence of the COVID-19 virus targeting the CNS: tissue distribution, host–virus interaction, and proposed neurotropic mechanisms. ACS Chem. Neurosci. 2020;11:995–998. doi: 10.1021/acschemneuro.0c00122. [DOI] [PubMed] [Google Scholar]
- Mao L., Wang M., Chen S., He Q., Chang J., Hong C., Zhou Y., Wang D., Li Y., Jin H., Hu B. Neurological Manifestations of hospitalized Patients with COVID-19 in Wuhan, China: a retrospective case series study. medRxiv preprint. 2020 doi: 10.1101/2020.02.22.20026500. Posted February. [DOI] [Google Scholar]
- Martínez Leo E.E., Segura Campos M.R. Systemic oxidative stress: a key point in neurodegeneration - a review. J. Nutr. Health Aging. 2019;23:694–699. doi: 10.1007/s12603-019-1240-8. [DOI] [PubMed] [Google Scholar]
- Matsuda K., Park C.H., Sunden Y., Kimura T., Ochiai K., Kida H., Umemura T. The vagus nerve is one route of transneural invasion for intranasally inoculated influenza a virus in mice. Vet. Pathol. 2004;41:101–107. doi: 10.1354/vp.41-2-101. [DOI] [PubMed] [Google Scholar]
- Moriguchi T., Harii N., Goto J., Harada D., Sugawara H., Takamino J., Ueno M., Sakata H., Kondo K., Myose N., Nakao A., Takeda M., Haro H., Inoue O., Suzuki-Inoue K., Kubokawa K., Ogihara S., Sasaki T., Kinouchi H., Kojin H., Ito M., Onishi H., Shimizu T., Sasaki Y., Enomoto N., Ishihara H., Furuya S., Yamamoto T., Shimada S. A first Case of Meningitis/Encephalitis associated with SARS-Coronavirus-2. Int. J. Infect. Dis. 2020;20:30195–30198. doi: 10.1016/j.ijid.2020.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Murray R.S., Brown B., Brian D., Cabirac G.F. Detection of coronavirus RNA and antigen in multiple sclerosis brain. Ann. Neurol. 1992;31:525–533. doi: 10.1002/ana.410310511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nikolich-Zugich J. The twilight of immunity: emerging concepts in aging of the immune system. Nat. Immunol. 2018;19:10–19. doi: 10.1038/s41590-017-0006-x. [DOI] [PubMed] [Google Scholar]
- Nuzzo D., Picone P., Caruana L., Vasto S., Barera A., Caruso C., Di Carlo M. Inflammatory mediators as biomarkers in brain disorders. Inflammation. 2014;37:639–648. doi: 10.1007/s10753-013-9780-2. [DOI] [PubMed] [Google Scholar]
- Picone P., Sabatino M.A., Ditta L.A., Amato A., San Biagio P.L., Mulè F., Giacomazza D., Dispenza C., Di Carlo M. Nose-to-brain delivery of insulin enhanced by a nanogel carrier. J. Control. Release. 2018;28(270):23–36. doi: 10.1016/j.jconrel.2017.11.040. [DOI] [PubMed] [Google Scholar]
- Rochfort K.D., Cummins P.M. The blood-brain barrier endothelium: a target for pro-inflammatory cytokines. Biochem. Soc. Trans. 2015;43:702–706. doi: 10.1042/BST20140319. [DOI] [PubMed] [Google Scholar]
- Solana R., Pawelec G., Tarazona R. Aging and innate immunity. Immunity. 2006;24:491–494. doi: 10.1016/j.immuni.2006.05.003. [DOI] [PubMed] [Google Scholar]
- Song Z., Xu Y., Bao L., Zhang L., Yu P., Qu Y., Zhu H., Zhao W., Han Y., Qin C. From SARS to MERS, thrusting coronaviruses into the spotlight. Viruses. 2019;11(1) doi: 10.3390/v11010059. pii: E59. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stevens R.D., Puybasset L. The brain–lung–brain axis. Intensive Care Med. 2011;37:1054–1056. doi: 10.1007/s00134-011-2233-1. [DOI] [PubMed] [Google Scholar]
- Tian S., Hu W., Niu L., Liu H., Xu H., Xiao S. Pulmonary Pathology of Early Phase SARSCoV-2 Pneumonia. Preprints. 2020 doi: 10.20944/preprints202002.0220.v1. www.Preprints.Org) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toth P., Tarantini S., Csiszar A., Ungvari Z. Functional vascular contributions to cognitive impairment and dementia: mechanisms and consequences of cerebral autoregulatory dysfunction, endothelial impairment, and neurovascular uncoupling in aging. Am J Physiol-Heart Circulatory Physiol. 2017;312:H1–H20. doi: 10.1152/ajpheart.00581.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turtle L. Respiratory failure alone does not suggest central nervous system invasion by SARS-CoV-2. J. Med. Virol. 2020:4. doi: 10.1002/jmv.25828. [DOI] [PubMed] [Google Scholar]
- Wang D., Hu B., Hu C., Zhu F., Liu X., Zhang J., Wang B., Xiang H., Cheng Z., Xiong Y., Zhao Y., Li Y., Wang X., Peng Z. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus-infected pneumonia in Wuhan, China. JAMA. 2020 doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J., Zhong S., Liu J., Li L., Li Y., Wu X., Li Z., Deng P., Zhang J., Zhong N., Ding Y., Jiang Y. Detection of severe acute respiratory syndrome coronavirus in the brain: potential role of the chemokine mig in pathogenesis. Clin. Infect. Dis. 2005;41:1089–1096. doi: 10.1086/444461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu F., Du L., Ojcius D.M., Pan C., Jiang S. Measures for diagnosing and treating infections by a novel coronavirus responsible for a pneumonia outbreak originating in Wuhan, China. Microbes Infect. 2020;22:74–79. doi: 10.1016/j.micinf.2020.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zheng M., Gao Y., Wang G., Song G., Liu S., Sun D., Xu Y., Tian Z. Functional exhaustion of antiviral lymphocytes in COVID-19 patients. Cell. Mol. Immunol. 2020;17:533–535. doi: 10.1038/s41423-020-0402-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhou P., Yang X.L., Wang X.G., Hu B., Zhang L., Zhang W., Si H.R., Zhu Y., Li B., Huang C.L., Chen H.D., Chen J., Luo Y., Guo H., Jiang R.D., Liu M.Q., Chen Y., Shen X.R., Wang X., Zheng X.S., Zhao K., Chen Q.J., Deng F., Liu L.L., Yan B., Zhan F.X., Wang Y.Y., Xiao G.F., Shi Z.L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu N., Zhang D., Wang W., Li X., Yang B., Song J., Zhao X., Huang B., Shi W., Lu R., Niu P., Zhan F., Ma X., Wang D., Xu W., Wu G., Gao G.F., Tan W. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med. 2020;20:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zinellu E., Zinellu A., Fois A.G., Carru C., Pirina P. Circulating biomarkers of oxidative stress in chronic obstructive pulmonary disease: a systematic review. Respir. Res. 2016;14:150. doi: 10.1186/s12931-016-0471-z. https://doi.org/110.1186/s12931-016-0471-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zirui Tay M., Meng Poh C., Rénia L., MacAry P.A., Ng L.F.P. The trinity of COVID-19: immunity, inflammation and intervention. Nat. Rev. Immunol. 2020;6:363–374. doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zuo L., Prather E.R., Stetskiv M., Garrison D.E., Meade J.R., Peace T.I., Zhou T. Inflammaging and oxidative stress in human diseases: from molecular mechanisms to novel treatments. Int. J. Mol. Sci. 2019;10:20. doi: 10.3390/ijms20184472. pii: E4472. [DOI] [PMC free article] [PubMed] [Google Scholar]