Abstract
CD137 (4-1BB) is a surface glycoprotein that belongs to the tumour necrosis factor receptor family (TNFRSF9). Its expression is induced on activation on a number of leucocyte types. Interestingly, for cancer immunotherapy, CD137 becomes expressed on primed T and natural killer (NK) cells, which on ligation provides powerful costimulatory signals. Perturbation of CD137 by CD137L or agonist monoclonal antibodies on activated CD8 T cells protects such antigen-specific cytotoxic T lymphocytes from apoptosis, enhances effector functionalities and favours persistence and memory differentiation. As a consequence, agonist antibodies exert potent antitumour effects in mouse models and the CD137 signalling domain is critical in chimeric antigen receptors (CAR) of CAR T cells approved to be used in the clinic. New formats of CD137 agonist moieties are being clinically developed, seeking potent costimulation targeted to the tumour microenvironment to avoid liver inflammation side effects, that have thus far limited and delayed clinical development.
Keywords: CD137, 4-1BB, cancer immunotherapy, mAb
Introduction
Cancer immunotherapy is already an important pillar in oncology treatments. T cells are the main mediators of efficacy. These immune cell subsets can specifically recognise tumour cells and kill them when properly activated. The understanding of the underlying biological processes that determine T-cell fate on activation in the tumour microenvironment (TME) represents one of the main cornerstones for the future of cancer immunotherapy. Enabling immune responses by targeting immune inhibitory signals (immune checkpoints) using PD-1/PD-L1 and/or CTLA-4 blockers has shown remarkable clinical efficacy against a large number of malignancies, but responses are still limited to a small fraction of patients.1 In parallel with T-cell inhibition blockade, immune cell stimulation by agonistic agents acting on costimulatory receptors such as CD137, OX40, TNFR2, CD27, CD40 or GITR, which are mainly expressed on T cells, constitute an area of very active preclinical and clinical research.
CD137 biological background
CD137/4-1BB (tnfrsf9) is a costimulatory receptor and a member of the tumour necrosis factor receptor superfamily (TNFRSF) that was initially discovered on activated T cells.2 CD137 is transiently expressed on activated T cells that have encountered cognate antigen, activated NK cells or mature dendritic cells (DCs). To date, a unique functional ligand has been identified for CD137, CD137L/4-1BBL (tnfsf9), expressed on the surface of professional antigen-presenting cells such as DCs, macrophages and B cells.3 Structurally, no enzymatic activity has been described for the intracellular domain of CD137 which is believed to function by recruiting TRAF1, TRAF2 and maybe TRAF3 adaptor proteins that mediate K63 polyubiquitination reactions in the CD137 signalosome.4 CD137L trimerisation leads to CD137 receptor clustering and TRAFs-mediated activation of NF-κB and MAPK intracellular signalling cascades, ultimately leading to cell activation, proliferation and survival.5 Bcl-XL, Bfl-1 induction and downregulation of BIM have been identified as antiapoptotic mechanisms, that seems to involve a role for interference with activation-induced cell death.6
On T cells, T cell receptor (TCR) stimulation and subsequent CD3 signalling induce transient expression of CD137 that on ligation with agonist antibodies or the natural ligand probably favours Th1 responses. Of note, CD28 costimulation results in markedly enhanced CD137 surface expression.7 Initial reports using CD137 monoclonal antibodies (mAbs) in mouse and human cells demonstrated that CD137 ligation causes strong costimulatory signals in T cells that in cooperation with TCR signalling gives rise to interleukin (IL)-2 and interferon ɣ (IFN-ɣ) production as well as T-cell proliferation.8 Further studies have shown that, in addition to inducing effector cytokine production, CD137 costimulation favours T cell memory and effector differentiation,5 protects T cells from apoptosis,6 changes the mitochondrial metabolism to increase T-cell respiratory capacities9 10 and induces overall DNA demethylation and chromatin reprogramming.11 In tumour models, in vitro and in vivo studies point towards a principal role of CD137 in CD8+ T cells rather than in CD4+ T cells, even though CD137 is inducible on both CD8+ and CD4+ T cells, including CD4+ FoxP3+ regulatory T cells (Tregs).12 Of note, the functional role of CD137 signalling on Th1 CD4+ and Tregs is still poorly understood and it may not be as physiologically relevant as for CD8+ T cells. On activated NK cells, CD137 triggering results in increased cell activation.13 Reports on enhancement of antibody-dependent cell cytotoxicity and cytotoxic functions have been retracted,14 15 but there is evidence that it contributes to NK activation.16
Because of (1) the strong costimulatory activity on cytotoxic T cells that boosts cell proliferation and survival, (2) the specific expression on antigen-primed tumour-specific T cells in the TME and draining lymph nodes17 18 and (3) the sustained signalling over time causing long-term changes in tumour-specific T cells,19 CD137 is considered an attractive target to enhance cancer immunotherapy. Furthermore, recent evidence on CD137 expression on CD4+ FoxP3+ Tregs indicated that CD137 can be used as a target to reduce/deplete Tregs which more brightly express CD137 and thereby offer a window for selective depletion.12
CD137-based cancer immunotherapy in preclinical models
The first in vivo evidence for the therapeutic potential of CD137 targeting in cancer was shown in transplantable tumour models of sarcoma and mastocytoma in which CD8+ T cell-dependent complete tumour eradication was attained using agonistic anti-CD137 mAbs.20 Ever since, accumulating preclinical data support the antitumour activity of CD137 agonism in several immunogenic and non-immunogenic tumour models. These same tumour models have been crucial for the understanding of the mechanistic principles behind CD137 costimulation in cancer. Among them, it has been shown that CD137 agonists could prevent cytotoxic T lymphocyte (CTL) anergy and thereby break T cell tolerance towards tumour antigens,21 restore T cell dysfunctionality in the TME22 and increase persistence of tumour-specific T cells.23
On their way to the preclinical development of CD137 agonists, initial efforts were made to force expression of CD137L on poorly immunogenic sarcoma cells7 or the prostate-specific membrane antigen (PSMA) recognising T cells,24 this strategy resulted in increased immunogenicity and enhanced antitumour activity. However, studies using agonistic mAbs showed stronger antitumour activity probably because of stronger agonistic actions and thus became the focus of translational research. Similar to other TNFRSF agonists such as CD40 mAbs, the road to the development of sufficiently agonistic CD137 has not been straightforward. In order to attain functional CD137 signalling and costimulation, agonist CD137 mAb must trimerise the receptor, a feature that requires effective crosslinking of the Fc to FcɣRs. In line with this, Buchan et al suggested a sequential mAb treatment scheme using a dual Fc engineering strategy to make the most of the CD137-specific expression pattern, first treating with an activating FcɣRs-binding mIgG2a mAb to deplete intratumour Tregs brightly expressing CD137, followed by an inhibitory FcɣRs-binding mIgG1 mAb to provide strong costimulation to CD8 CTLs.25
Along with the efficacy of anti-CD137 antibody as a monotherapy, a broad number of synergistic combinations have been reported. Checkpoint inhibitory receptors such as PD-1 or CTLA-4 are commonly found to be upregulated in dysfunctional T cells within the TME across multiple types of malignant diseases.1 Checkpoint receptors on binding their ligands repress T-cell activation intracellular signals that had frequently been induced by costimulatory receptors. To make the most of these facts, simultaneous blockade of checkpoint inhibitor receptors in conjunction with agonistic mAbs triggering costimulatory receptors makes sense26. This can be achieved also with bispecific antibodies encompassing both activities in a single moiety. In this regard, true synergistic combinations are a major goal in cancer immunotherapy.27 CD137-resistant ovarian and lung cancer models become amenable to eradication on combined treatment with anti-CD137 mAb with PD-1 blocking mAb, resulting in potent synergistic effects that correlate with increased T-cell survival and effector functions.28 Interestingly, CD137 and PD-1 coexpression is likely to be restricted to neoantigen-specific tumour-infiltrating CD8+ T cells,29 suggesting the rational of combining both pathways for immunotherapeutic purposes. Combination of CTLA-4 blocking mAb with anti-CD137 mAb also resulted in enhanced CD8+ T-cell mediated immune responses in mouse models of melanoma.30 In addition to checkpoint blockade, combination with immunostimulatory mAbs such as CD40,31 OX4032 or CD2033 has been reported to potentiate the antitumour effects.
Other therapeutic strategies including radiotherapy,34 gene therapy35 or adoptive cell therapy synergise with CD137 stimulation. Combination of CD137 mAb with adoptive T cell therapy in mouse models of B16OVA and OVA-specific CD8+ T cell (OT1) transfer resulted in long-lasting tumour control elicited by enhanced effector functions of the transferred and endogenous CD8+ T cells that when visualised by intravital microscopy behaved more aggressively against malignant cells.23
CD137 immunotherapy in clinical development
Two approaches encompassing CD137 have been evaluated in the clinic: (1) anti-CD137-targeting mAbs and bispecific Abs and (2) the cytoplasmatic CD137 domain forming part of chimeric antigen receptors (CAR). Today, CD19 targeting CAR-T cells are the only CD137-based approach approved by the FDA for the treatment of B-cell paediatric leukaemia and refractory B-cell lymphoma.36 In contrast to CD28 encompassing CARs, CD137 intracellular signalling domain-based CARs are those achieving best adoptive T-cell persistence.37 In addition, stronger metabolic fitness and beneficial epigenetic reprograming have been associated with CD137 containing CARs.38
Two agonist anti-CD137 mAb have been tested in the clinical setting; urelumab (BMS-663513), a fully human IgG4-based anti-CD137 mAb that does not block CD137L–CD137 interactions and utolimumab (PF-05082566), a ligand-blocking-humanised IgG1 mAb (table 1).
Table 1.
Summary of combinatorial approaches with agonistic CD137 monoclonal antibody (mAb) under clinical evaluation
| Agent | mAb | Main characteristics | Combination under clinical evaluation | Additional biological agent | Condition | Phase | NCT |
| Urelumab (BMS-663513) |
Fully human IgG4 | High agonist activity | Rituximab (anti-CD20) |
Chronic lymphocytic leukaemia | II (withdrawn) | NCT02420938 | |
| Non-ligand blocking | Liver inflammation with doses of ≥1 mg/kg | B-cell non-Hodgkin’s lymphoma | I (completed) | NCT01775631 | |||
| Nivolumab (anti-PD-1) |
Solid tumours (intratumour urelumab) | I and II (not recruiting) | NCT03792724 | ||||
| Solid tumours and B-cell non-Hodgkin’s lymphoma | I and II (completed) | NCT02253992 | |||||
| Muscle-invasive urothelial carcinoma of the bladder (neadjuvant nivolumab) | II (recruting) | NCT02845323 | |||||
| Advanced and/or metastatic malignant tumours | I (active, not recruiting) | NCT02534506 | |||||
| Multiple metastases in advanced solid tumours | I (recruiting) | NCT03431948 | |||||
| Recurrent globlastoma | I (active) | NCT02658981 | |||||
| GVAX (pancreas vaccine) | Surgically resectable pancreatic cancer | I and II (recruiting) | NCT02451982 | ||||
| Tumour- infiltrating lymphocytes | Metastatic melanoma | I (active, not recruiting) | NCT02652455 | ||||
| Cetuximab (anti-EGFR) |
Advanced/metastatic colorectal cancer | I (completed) | NCT02110082 | ||||
| Elotuzumab (anti-CS1) | Multiple myeloma | I (completed) | NCT02252263 | ||||
| Ipilumumab (anti-CTLA-4) |
Malignant melanoma | I (withdrawn) | NCT00803374 | ||||
| Utolimumab (PF-05082566) |
Humanised IgG1 | Weak agonist activity | Pembrolizumab (anti-PD-1) |
Advanced solid tumours | I (completed) | NCT02179918 | |
| Ligand blocking | No dose-limiting toxicities | Mogamulizumab (anti-CCR4) |
Advanced solid tumours | I (terminated) | NCT02444793 | ||
| Rituxumab (anti-CD20) |
Solid tumours or B-cell lymphomas | I (completed) | NCT01307267 | ||||
| Cetuximab (anti-EGFR) |
Advanced colorectal cancer | I (recruiting) | NCT03290937 | ||||
| Trastuzumab (anti-HER2) |
Advanced HER2-positive breast cancer | I (recruiting) | NCT03364348 | ||||
| Avelumab (anti-PD-L1) |
Advanced HER2 +breast cancer | II (recruting) | NCT03414658 | ||||
| Avelumab (anti-PD-L1) |
Triple negative breast cancer | II (recruiting) | NCT03971409 | ||||
| Locally advanced or metastatic solid tumours | II (recruiting) | NCT02554812 | |||||
| PF-8600 (OX-40 agonist) | Advanced solid tumours | I and II (recruiting) | NCT03217747 | ||||
| Rituximab (anti-CD20) |
Relapsed or refractory diffuse large B-cell lymphoma | I (completed) | NCT02951156 | ||||
| Rituximab (anti-CD20) |
Relapsed or refractory diffuse large B-cell lymphoma or mantle cell lymphoma | I (recruiting) | NCT03440567 | ||||
| PF-8600 (OX-40 agonist) | Advanced or metastatic carcinoma | I (active, not recruiting) | NCT02315066 | ||||
| ISA101b (HPV16 E6/E7 peptides vaccine) | HPV-16-positive oropharyngeal cancer | II (active, not recruiting) | NCT03258008 | ||||
| Primed CD8 +tumour antigen-specific T cells | Aldesleukin | Recurrent ovarian cancer | I (recruiting) | NCT03318900 |
Initial reports testing urelumab in phase I and II trials showed activity as a single agent in patients with advanced cancer, but trials had to be terminated because of treatment-related severe adverse effects in the form of liver inflammation.39 The hepatotoxicity observed in the patients had not been observed in non-human primates due to lower binding affinities to cynomolgus CD137. An integrated safety analysis of urelumab revealed life-threating liver inflammation associated with doses of ≥1 mg/kg that required a drastic dose reduction to 0.1 mg/kg to be safe.40 Such dose reduction maintained little pharmacodynamic activity and resulted in loss of efficacy as monotherapy except for a few B-cell non-Hodgkin’s lymphoma cases, where some objective responses were observed. Despite the dose reduction requirement, the obtained safety prolife supported evaluation of urelumab in combination with other immune-modulating agents. Low doses of urelumab in combination with the anti-PD1 mAb nivolumab showed acceptable tolerability and promising clinical responses have been reported in patients with metastatic melanoma irrespective of PD-L1 tumour expression.41 Urelumab is further being combined with cetuximab (NCT02110082), rituximab (NCT01775631) and elotuzumab (NCT02252263) (table 1).
On the contrary, utolimumab has no dose-limiting toxicities and shows preliminary antitumour activity in patients with advanced cancer. Probably because of being a weak CD137-agonist, utolimumab has shown little clinical effect as a monotherapy.42 Phase 1 combination strategies combining utolimumab with rituximab in patients with relapsed or refractory non-Hodgkin’s lymphoma43 and utilimumab with pembrolizumab in patients with advanced solid tumours44 have demonstrated tolerability, safety and preliminary clinical activity. Another combinatorial strategy being tested is the combination of utomilumab with intravenous infusion of CD8 +tumour antigen-specific T cells (NCT03318900; table 1).
CD137 mAb therapy aims to stimulating the activity of tumour-specific T cells already within the TME. Tumour susceptibility to the mAb treatment is likely to depend on a pre-existing tumour T-cell infiltration and on CD137 expression levels on such tumour-infiltrating T cells. For clinical purposes, well-defined prognostic and predictive markers for CD137 treatment are still missing. Thus, biomarker discovery and validation for CD137-based immunotherapies currently constitute a major research effort.
Lessons learnt: moving CD137 agonists towards safer tumour targeting
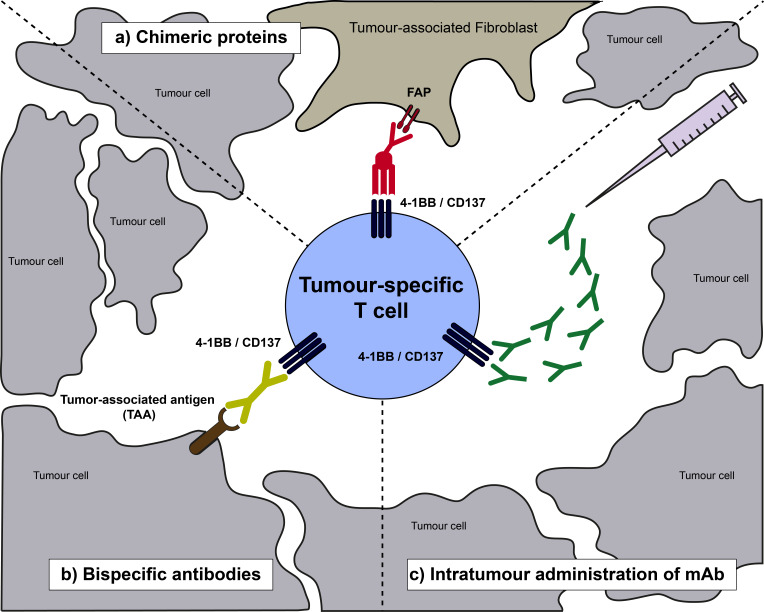
Preclinical studies trying to elucidate the hepatotoxicity related to CD137 agonists showed hepatic polyclonal CD8+ T cell activation and IFN-ɣ secretion as the main mediators of liver damage.45 Further studies link CD137 mAb binding to liver resident monocyte and Kupffer cell that secrete tumour necrosis factor α (TNF-α) and IL-27 and activate CD8+ T cells, ultimately leading to liver damage.46 Prompted by the preclinical mechanistic insight and based on the clinical experiences using CD137 mAb, that when sufficiently agonistic lead to severe liver inflammation, new efforts are in place to specifically bring CD137 agonism to the tumour tissue microenvironment (figure 1).
Figure 1.
Schematic representation of a tumour-infiltrating T lymphocyte expressing CD137 as a result of priming by tumour-antigen recognition. Then, novel strategies to make the most of CD137 costimulation are represented including bispecifics and direct intratumour injections. FAP, fibroblast activation protein; mAb, monoclonal antibody.
Different approaches are on the table, among them the most advanced ones under clinical development are the CD137-based bispecific constructs that are designed to bring CD137 costimulation specifically to the TME. A bispecific antibody targeting CD137 on T cells and the oncogenic antigen HER2 expressed on tumour cells (HER2/4-1BB Ab) has been the first CD137-based bispecific Ab tested in the clinic.47 Recently, results presented from the phase 1 single-agent dose escalation trial evaluating HER2/4-1BB Ab in patients with HER2+ solid tumours, indicated good tolerability of the construct and showed evidence of clinical activity.48 Another tumour targeting bispecific construct being explored in the clinic is the PD-L1 targeting PD-L1/4-1BB Ab (NCT03809624). Preclinically, bispecific Ab targeting the tumour antigen 5T4 and CD137 (5T4/4-1BB Ab) has shown promising activity in cell cultures.49 In addition, a chimeric protein consisting on a trimeric form of CD137L integrated into a fibroblast activation protein (FAP), a highly expressed protein on the tumour stroma, recognising bispecific construct (FAP/4-1BBL) has shown impressive anti-tumoural activities against tumour mouse models and tumour-targeting in spontaneous cancer bearing rhesus monkeys, with no signs of toxicity in macaques.50 In line with the bispecific designs and to avoid FcɣR binding mediated liver toxicity, Fc lacking trimeric CD137 constructs with sufficient agonistic activity and without liver toxicity have been successfully evaluated in mouse models.51
Another approach to specifically provide CD137 costimulation to the TME includes intratumour injections of low doses of the CD137 mAb.52 Following preclinical evidence demonstrating the feasibility and antitumour efficacy of the intratumour administration, a clinical trial injecting low doses of CD137 mAb directly into metastatic lesions and in combination with systemic nivolumab is being conducted in patients with solid tumours (NCT03792724).
Conclusion
The immunobiology of CD137 provides the basis for our understanding of the role of CD137 costimulation in cancer and arguably presents CD137 as one of the most potent costimulatory receptors for primed CD8+ T cells. Preclinical data convincingly show strong antitumour activity of CD137 agonists. Consequently, CD137 emerges as a robust candidate to be targeted for cancer treatment. The beginnings of CD137 agonist mAb in the clinic were overshadowed by hepatic toxicities in a not well-defined fraction of patients. In spite of such caveats, (1) safe combinatorial strategies, involving checkpoint inhibitors and CD137 agonists, designed to simultaneously normalise and stimulate the inhibited antitumour immune responses or (2) strategies to specifically target CD137 agonists to the TME are likely to broaden the therapeutic window. In the next years, we need to harness a promising foal to turn it into a powerful warhorse against cancer.
Footnotes
Contributors: IE and IM wrote the manuscript. All the authors reviewed and edited the manuscript.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: IM is a paid consultant for Bristol-Myers Squibb, Roche, AstraZeneca, Pharmamar, Alligator, Numab, F-star, Servier, and MSD, and reports receiving commercial research grants from Alligator, Bristol-Myers Squibb, Roche, and Pharmamar.
Patient consent for publication: Not required.
Provenance and peer review: Commissioned; externally peer reviewed.
References
- 1.Ribas A, Wolchok JD. Cancer immunotherapy using checkpoint blockade. Science 2018;359:1350–5. 10.1126/science.aar4060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kwon BS, Weissman SM. cDNA sequences of two inducible T-cell genes. Proc Natl Acad Sci U S A 1989;86:1963–7. 10.1073/pnas.86.6.1963 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Goodwin RG, Din WS, Davis-Smith T, et al. Molecular cloning of a ligand for the inducible T cell gene 4-1BB: a member of an emerging family of cytokines with homology to tumor necrosis factor. Eur J Immunol 1993;23:2631–41. 10.1002/eji.1830231037 [DOI] [PubMed] [Google Scholar]
- 4.Zapata JM, Perez-Chacon G, Carr-Baena P, et al. CD137 (4-1BB) signalosome: complexity is a matter of TRAFs. Front Immunol 2018;9:2618. 10.3389/fimmu.2018.02618 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cannons JL, Lau P, Ghumman B, et al. 4-1BB ligand induces cell division, sustains survival, and enhances effector function of CD4 and CD8 T cells with similar efficacy. J Immunol 2001;167:1313–24. 10.4049/jimmunol.167.3.1313 [DOI] [PubMed] [Google Scholar]
- 6.Lee H-W, Park S-J, Choi BK, et al. 4-1BB promotes the survival of CD8+ T lymphocytes by increasing expression of Bcl-xL and Bfl-1. J Immunol 2002;169:4882–8. 10.4049/jimmunol.169.9.4882 [DOI] [PubMed] [Google Scholar]
- 7.Melero I, Bach N, Hellström KE, et al. Amplification of tumor immunity by gene transfer of the co-stimulatory 4-1BB ligand: synergy with the CD28 co-stimulatory pathway. Eur J Immunol 1998;28:1116–21. [DOI] [PubMed] [Google Scholar]
- 8.Shuford WW, Klussman K, Tritchler DD, et al. 4-1BB costimulatory signals preferentially induce CD8+ T cell proliferation and lead to the amplification in vivo of cytotoxic T cell responses. J Exp Med 1997;186:47–55. 10.1084/jem.186.1.47 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Menk AV, Scharping NE, Rivadeneira DB, et al. 4-1BB costimulation induces T cell mitochondrial function and biogenesis enabling cancer immunotherapeutic responses. J Exp Med 2018;215:1091–100. 10.1084/jem.20171068 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Teijeira A, Labiano S, Garasa S, et al. Mitochondrial morphological and functional reprogramming following CD137 (4-1BB) costimulation. Cancer Immunol Res 2018;6:798–811. 10.1158/2326-6066.CIR-17-0767 [DOI] [PubMed] [Google Scholar]
- 11.Aznar MA, Labiano S, Diaz-Lagares A, et al. CD137 (4-1BB) costimulation modifies DNA methylation in CD8+ T cell–relevant genes. Cancer Immunol Res 2018;6:69–78. 10.1158/2326-6066.CIR-17-0159 [DOI] [PubMed] [Google Scholar]
- 12.Freeman ZT, Nirschl TR, Hovelson DH, et al. A conserved intratumoral regulatory T cell signature identifies 4-1BB as a pan-cancer target. J Clin Invest 2020;130:1405–16. 10.1172/JCI128672 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Choi BK, Kim YH, Kim CH, et al. Peripheral 4-1BB signaling negatively regulates NK cell development through IFN-gamma. J Immunol 2010;185:1404–11. 10.4049/jimmunol.1000850 [DOI] [PubMed] [Google Scholar]
- 14.Kohrt HE, Houot R, Goldstein MJ, et al. Cd137 stimulation enhances the antilymphoma activity of anti-CD20 antibodies. Blood 2011;117:2423–32. 10.1182/blood-2010-08-301945 [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 15.Kohrt HE, Colevas AD, Houot R, et al. Targeting CD137 enhances the efficacy of cetuximab. J Clin Invest 2019;129:2595. 10.1172/JCI129689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ochoa MC, Perez-Ruiz E, Minute L, et al. Daratumumab in combination with urelumab to potentiate anti-myeloma activity in lymphocyte-deficient mice reconstituted with human NK cells. Oncoimmunology 2019;8:1599636. 10.1080/2162402X.2019.1599636 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wolfl M, Kuball J, Ho WY, et al. Activation-Induced expression of CD137 permits detection, isolation, and expansion of the full repertoire of CD8+ T cells responding to antigen without requiring knowledge of epitope specificities. Blood 2007;110:201–10. 10.1182/blood-2006-11-056168 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ye Q, Song D-G, Poussin M, et al. CD137 accurately identifies and enriches for naturally occurring tumor-reactive T cells in tumor. Clin Cancer Res 2014;20:44–55. 10.1158/1078-0432.CCR-13-0945 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sanchez-Paulete AR, Labiano S, Rodriguez-Ruiz ME, et al. Deciphering CD137 (4-1BB) signaling in T-cell costimulation for translation into successful cancer immunotherapy. Eur J Immunol 2016;46:513–22. 10.1002/eji.201445388 [DOI] [PubMed] [Google Scholar]
- 20.Melero I, Shuford WW, Newby SA, et al. Monoclonal antibodies against the 4-1BB T-cell activation molecule eradicate established tumors. Nat Med 1997;3:682–5. 10.1038/nm0697-682 [DOI] [PubMed] [Google Scholar]
- 21.Wilcox RA, Flies DB, Zhu G, et al. Provision of antigen and CD137 signaling breaks immunological ignorance, promoting regression of poorly immunogenic tumors. J Clin Invest 2002;109:651–9. 10.1172/JCI0214184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Williams JB, Horton BL, Zheng Y, et al. The EGR2 targets LAG-3 and 4-1BB describe and regulate dysfunctional antigen-specific CD8+ T cells in the tumor microenvironment. J Exp Med 2017;214:381–400. 10.1084/jem.20160485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Weigelin B, Bolaños E, Teijeira A, et al. Focusing and sustaining the antitumor CTL effector killer response by agonist anti-CD137 mAb. Proc Natl Acad Sci U S A 2015;112:7551–6. 10.1073/pnas.1506357112 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Stephan MT, Ponomarev V, Brentjens RJ, et al. T cell-encoded CD80 and 4-1BBL induce auto- and transcostimulation, resulting in potent tumor rejection. Nat Med 2007;13:1440–9. 10.1038/nm1676 [DOI] [PubMed] [Google Scholar]
- 25.Buchan SL, Dou L, Remer M, et al. Antibodies to costimulatory receptor 4-1BB enhance anti-tumor immunity via T regulatory cell depletion and promotion of CD8 T cell effector function. Immunity 2018;49:958–70. 10.1016/j.immuni.2018.09.014 [DOI] [PubMed] [Google Scholar]
- 26.Azpilikueta A, Agorreta J, Labiano S, et al. Successful immunotherapy against a transplantable mouse squamous lung carcinoma with anti-PD-1 and Anti-CD137 monoclonal antibodies. J Thorac Oncol 2016;11:524–36. 10.1016/j.jtho.2016.01.013 [DOI] [PubMed] [Google Scholar]
- 27.Melero I, Berman DM, Aznar MA, et al. Evolving synergistic combinations of targeted immunotherapies to combat cancer. Nat Rev Cancer 2015;15:457–72. 10.1038/nrc3973 [DOI] [PubMed] [Google Scholar]
- 28.Wei H, Zhao L, Hellstrom I, et al. Dual targeting of CD137 co-stimulatory and PD-1 co-inhibitory molecules for ovarian cancer immunotherapy. Oncoimmunology 2014;3:e28248. 10.4161/onci.28248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gros A, Robbins PF, Yao X, et al. PD-1 identifies the patient-specific CD8⁺ tumor-reactive repertoire infiltrating human tumors. J Clin Invest 2014;124:2246–59. 10.1172/JCI73639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Curran MA, Kim M, Montalvo W, et al. Combination CTLA-4 blockade and 4-1BB activation enhances tumor rejection by increasing T-cell infiltration, proliferation, and cytokine production. PLoS One 2011;6:e19499. 10.1371/journal.pone.0019499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Westwood JA, Matthews GM, Shortt J, et al. Combination anti-CD137 and anti-CD40 antibody therapy in murine myc-driven hematological cancers. Leuk Res 2014;38:948–54. 10.1016/j.leukres.2014.05.010 [DOI] [PubMed] [Google Scholar]
- 32.Lee S-J, Myers L, Muralimohan G, et al. 4-1BB and OX40 dual costimulation synergistically stimulate primary specific CD8 T cells for robust effector function. J Immunol 2004;173:3002–12. 10.4049/jimmunol.173.5.3002 [DOI] [PubMed] [Google Scholar]
- 33.Souza-Fonseca-Guimaraes F, Blake SJ, Makkouk A, et al. Anti-CD137 enhances anti-CD20 therapy of systemic B-cell lymphoma with altered immune homeostasis but negligible toxicity. Oncoimmunology 2016;5:e1192740. 10.1080/2162402X.2016.1192740 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Rodriguez-Ruiz ME, Rodriguez I, Garasa S, et al. Abscopal effects of radiotherapy are enhanced by combined immunostimulatory mAbs and are dependent on CD8 T cells and Crosspriming. Cancer Res 2016;76:5994–6005. 10.1158/0008-5472.CAN-16-0549 [DOI] [PubMed] [Google Scholar]
- 35.Quetglas JI, Dubrot J, Bezunartea J, et al. Immunotherapeutic synergy between anti-CD137 mAb and intratumoral administration of a cytopathic Semliki Forest virus encoding IL-12. Mol Ther 2012;20:1664–75. 10.1038/mt.2012.56 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.June CH, O'Connor RS, Kawalekar OU, et al. CAR T cell immunotherapy for human cancer. Science 2018;359:1361–5. 10.1126/science.aar6711 [DOI] [PubMed] [Google Scholar]
- 37.Zhao Z, Condomines M, van der Stegen SJC, et al. Structural design of engineered costimulation determines tumor rejection kinetics and persistence of CAR T cells. Cancer Cell 2015;28:415–28. 10.1016/j.ccell.2015.09.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Kawalekar OU, O'Connor RS, Fraietta JA, et al. Distinct signaling of coreceptors regulates specific metabolism pathways and impacts memory development in car T cells. Immunity 2016;44:380–90. 10.1016/j.immuni.2016.01.021 [DOI] [PubMed] [Google Scholar]
- 39.Sznol M, Hodi FS, Margolin K, et al. Phase I study of BMS-663513, a fully human anti-CD137 agonist monoclonal antibody, in patients (PTS) with advanced cancer (CA). JCO 2008;26:3007 10.1200/jco.2008.26.15_suppl.3007 [DOI] [Google Scholar]
- 40.Segal NH, Logan TF, Hodi FS, et al. Results from an integrated safety analysis of Urelumab, an agonist Anti-CD137 monoclonal antibody. Clin Cancer Res 2017;23:1929–36. 10.1158/1078-0432.CCR-16-1272 [DOI] [PubMed] [Google Scholar]
- 41.Massarelli E. Clinical safety and efficacy as- sessment of the CD137 agonist urelumab alone and in combination with nivolumab in patients with hematologic and solid tumor malignancies In: Proceeding of the 31st annual meeting and associated programs of the Society for immunotherapy of cancer. 239, 2016. [Google Scholar]
- 42.Segal NH, Gopal AK, Bhatia S, et al. A phase 1 study of PF-05082566 (anti-4-1BB) in patients with advanced cancer. JCO 2014;32:3007 10.1200/jco.2014.32.15_suppl.3007 [DOI] [Google Scholar]
- 43.Gopal AK, Bartlett NL, Levy R, et al. A phase I study of PF-05082566 (anti-4-1BB) + rituximab in patients with CD20+ NHL. JCO 2015;33:3004 10.1200/jco.2015.33.15_suppl.3004 [DOI] [Google Scholar]
- 44.Tolcher AW, Sznol M, Hu-Lieskovan S, et al. Phase Ib study of Utomilumab (PF-05082566), a 4-1BB/CD137 agonist, in combination with pembrolizumab (MK-3475) in patients with advanced solid tumors. Clin Cancer Res 2017;23:5349–57. 10.1158/1078-0432.CCR-17-1243 [DOI] [PubMed] [Google Scholar]
- 45.Dubrot J, Milheiro F, Alfaro C, et al. Treatment with anti-CD137 mAbs causes intense accumulations of liver T cells without selective antitumor immunotherapeutic effects in this organ. Cancer Immunol Immunother 2010;59:1223–33. 10.1007/s00262-010-0846-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Bartkowiak T, Jaiswal AR, Ager CR, et al. Activation of 4-1BB on liver myeloid cells triggers hepatitis via an Interleukin-27-Dependent pathway. Clin Cancer Res 2018;24:1138–51. 10.1158/1078-0432.CCR-17-1847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Hinner MJ, Aiba RSB, Jaquin TJ, et al. Tumor-localized costimulatory T-cell engagement by the 4-1BB/HER2 bispecific antibody-anticalin fusion PRS-343. Clin Cancer Res 2019;25:5878–89. 10.1158/1078-0432.CCR-18-3654 [DOI] [PubMed] [Google Scholar]
- 48.Piha-Paul S, Bendell J, Tolcher A. Phase 1 dose escalation study of PRS-343, a HER2/4-1BB bispecific molecule, in patients with HER2+ malignancies. 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer, National Harbor, MD, 2019. [Google Scholar]
- 49.Nelson M, Miller R, Bader R. Potent tumor-directed T cell activation and tumor inhibition induced by ALG.APV-527, a 4-1BB x 5T4 ADAPTIRTM bispecific antibody. 34th Annual Meeting & Pre-Conference Programs of the Society for Immunotherapy of Cancer, National Harbor, MD, 2019. [Google Scholar]
- 50.Claus C, Ferrara C, Xu W, et al. Tumor-targeted 4-1BB agonists for combination with T cell bispecific antibodies as off-the-shelf therapy. Sci Transl Med 2019;11:eaav5989. 10.1126/scitranslmed.aav5989 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Compte M, Harwood SL, Muñoz IG, et al. A tumor-targeted trimeric 4-1BB-agonistic antibody induces potent anti-tumor immunity without systemic toxicity. Nat Commun 2018;9:4809. 10.1038/s41467-018-07195-w [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Palazón A, Martínez-Forero I, Teijeira A, et al. The HIF-1α hypoxia response in tumor-infiltrating T lymphocytes induces functional CD137 (4-1BB) for immunotherapy. Cancer Discov 2012;2:608–23. 10.1158/2159-8290.CD-11-0314 [DOI] [PubMed] [Google Scholar]