Graphical abstract
Keywords: Type I Interferons, COVID-19, SARS-CoV-2, Coronavirus, Mucosal treatments, Immunomodulation, Antiviral therapy, Antiviral Immune Response, Beta Interferon
Highlights
-
•
IFN-I, in particular IFN-β, are promising drugs for SARS-CoV2 infection.
-
•
Early infection in elderly patients is the best setting to exploit IFN-I immunomodulatory activity.
-
•
Caution should be given in using continuous IFN-I treatments at high doses.
-
•
Mucosal IFN-I delivery is promising but deserves further clinical investigation.
-
•
Attention should be paid to IFN-I treatment in patients with severe COVID-19.
Abstract
Coronavirus disease 2019 (COVID-19) first emerged in late 2019 in China. At the time of writing, its causative agent SARS-CoV-2 has spread worldwide infecting over 9 million individuals and causing more than 460,000 deaths. In the absence of vaccines, we are facing the dramatic challenge of controlling COVID-19 pandemic. Among currently available drugs, type I Interferons (IFN-I) – mainly IFN-α and β –represent ideal candidates given their direct and immune-mediated antiviral effects and the long record of clinical use. However, the best modalities of using these cytokines in SARS-CoV-2 infected patients is a matter of debate. Here, we discuss how we can exploit the current knowledge on IFN-I system to tailor the most promising dosing, timing and route of administration of IFN-I to the disease stage, with the final aim of making these cytokines a valuable therapeutic strategy in today's fight against COVID-19 pandemic.
1. Introduction
The rapid and devastating outbreak of Coronavirus disease 2019 (COVID-19) pandemic and the lack of approved treatments for any human coronavirus (CoV) infection highlight the urgent need to develop strategies to prevent infection, control virus spreading and disease exacerbation. Several options can be envisaged, ranging from prophylactic vaccine to targeted antiviral drugs. However, new interventions are likely to require months to years to be readily-available, thus priority is being given to the repurposing of existing antiviral agents [1].
Since COVID-19 outbreak, more than 2000 clinical trials have been authorized to identify the drugs or drug combinations capable of attenuating the virulence of the disease [2]. Some of these trials include the use of Interferons (IFN) alone or in combination with other compounds. IFN are a cytokine family linking innate and adaptive immune responses, with an important role in the inhibition of viral replication, through different receptor molecules and effector proteins [3]. There are three types of IFN, Type I (IFN-α/β), Type II (IFN-γ), and Type III (IFN-λ). Although all three are likely involved in protection against CoV infection, Type I IFN (IFN-I) and IFN-λ are the most studied in this area [4]. Interestingly, although IFN-α and β share the same receptor and exhibit similar biological activities [5], their diverse receptor affinity may be responsible for some differential effects [5]. Some studies point to IFN-β1b and IFN-β1a as the most potent subtypes for SARS-CoV inhibition and likely even more for SARS-CoV-2 [[6], [7], [8]].
IFN-I were first discovered more than 60 years ago as antiviral substances produced by influenza virus-infected cells, capable of markedly inhibiting viral replication in target cells [9]. These cytokines were the firsts to be cloned and have been extensively used in patients with some viral diseases [10] and cancer (IFN-α) [11], and in the treatment of relapsing-remitting multiple sclerosis (MS) (IFN-β, [12]). Indeed, IFN-I are pleiotropic factors endowed with multiple activities, including both a broad-spectrum antiviral activity [9,10] and a remarkable antiproliferative and immunoregulatory function [13].
The antiviral activity of IFN-I has been extensively exploited for the treatment of viral chronic infections [10]. Nevertheless, as highlighted by the long clinical records of IFN-I use, the complex nature of the IFN-I system and the dysregulated IFN-I response in SARS-CoV-2 infection point to the need of a fine-tuning of route, timing and dose of administration to balance clinical efficacy and side effects. In this Perspective article, we briefly review the role of IFN-I signaling in COVID-19 pathogenesis and discuss some critical issues that need to be taken into account for optimally exploiting IFN-I in today’s fight against COVID-19 pandemic.
2. SARS-CoV-2: one virus, multiple battlefronts
Differently from the 2002 severe acute respiratory syndrome (SARS)-CoV, SARS-CoV-2 has a lower virulence with a large number of asymptomatic infections and mild upper respiratory tract illness [14,15]. This is a determinant factor for its increased transmissibility, considering that asymptomatic or presymptomatic carriers might contribute to viral transmission, especially in high-risk populations [16]. Even though upper respiratory infections are often spontaneously resolved, some of these eventually progress in more severe viral pneumonia. Severe COVID-19 patients are characterized by a strong immune activation (reviewed in [17]) resulting in lung infiltration by proinflammatory macrophages and granulocytes [18], development of acute respiratory distress syndrome, respiratory failure and subsequent fatality (Fig. 1 ). Age, sex and comorbidities are main factors affecting the risk of developing severe COVID-19 [19,20]. Ageing is indeed characterized by the gradual development of a chronic subclinical systemic inflammation and by acquired immune impairment, particularly in the male population [21]. In particular, recent studies have shown that ageing affects B lymphocyte-driven acquired immunity [22], attenuates the upregulation of co-stimulatory molecules for T cell priming and reduces IFN-I production by alveolar phagocytes in response to viral infection [19].
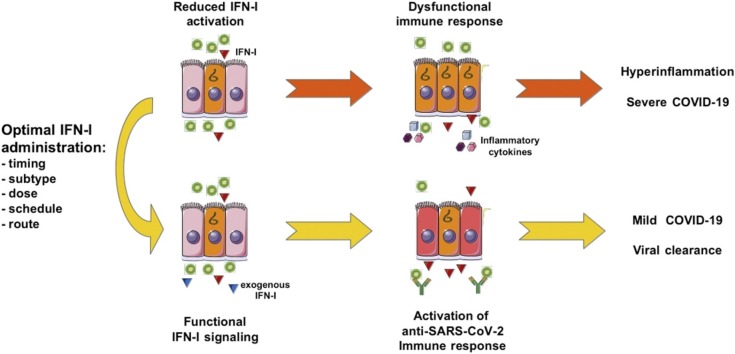
Fig. 1.
COVID-19 and IFN-I treatment.
In the majority of patients (left panels) early phase infection results in a physiological IFN-I activation that favors the development of anti-SARS-CoV2 antibodies and virus-specific T cells in addition to the paracrine direct antiviral effect. This immune activation leads to a resolution of the infection without any need of treatment. Instead, approximately 15–20 % of infected patients (middle panels), because of reduced early IFN-I activation show a dysfunctional immune response with higher viral spread. This situation is then followed by an excessive granulocyte and macrophage infiltration, secreting high amounts of inflammatory cytokines that lead to severe forms of COVID-19. This group of patients would strongly benefit of an IFN-I treatment at the early phase of infection (right panels) as it would re-establish a functional immune response that would then lead to resolution of the infection. At bottom, diagrams summarizing the variation over time of infection levels and immune response for each scenario. This figure was created using Servier Medical Art templates.
Thus, due to the different pathogenic mechanisms intervening in early stage versus late stage of SARS-CoV-2 infection (reviewed in [17]) and to the role of comorbidities in disease exacerbation, interventions should be carefully tailored to the stage of the disease and to the target population with the aim of avoiding the progression to severe clinical manifestations.
3. CoV and the hijack of IFN-I
Although our knowledge on SARS-CoV-2 biology is still limited, the overall genomic similarity of SARS-CoV-2 with SARS- and MERS-CoV, as well as the immunologic changes observed during infection [23] point to a dysregulated IFN-I response in COVID-19 pathogenesis and disease outcome [24]. Diminished levels of IFN-I were already detected during the course of SARS and MERS infections. In particular, SARS patients revealed no expression of IFN-I or IFN-stimulated genes (ISG) in peripheral blood mononuclear cells, suggesting a viral-induced inhibition of their expression [18,25]. Furthermore, a downregulated expression of IFN-α and of some ISG (RIG-I, MDA5 and IRF3/7) was associated with the severity of disease and fatal outcomes in MERS patients [26]. Likewise, SARS-CoV-2 infection (either in cell lines, primary cell cultures, ferrets, and COVID-19 patients) drives a lower antiviral transcriptional response that is marked by low IFN-I and IFN-III levels and elevated chemokine expression, which could explain the proinflammatory disease state associated with COVID-19. In patients with severe COVID-19, an impaired IFN-I signalling associated with persistent blood viral load and an exacerbated inflammatory response was recently reported [27]. Along these lines, IFN immune profiling in critically ill COVID-19 patients revealed a peak in IFN α2 production 8–10 days after symptoms onset. A small group of patients had suppressed IFN-I production, in particular, patients with no IFN-α production presented poorer outcome and longer intensive care unit stay [28].
Other studies suggest that rather than a complete absence, the IFN response may be delayed. Compelling evidence indicated that human bronchial epithelial cells are capable of promoting active, but delayed IFN-related antiviral responses to SARS-CoV and MERS-CoV infection [29,30]. These results are in line with data on mouse model of SARS-CoV infection, where a robust viral replication associated with delayed IFN-I signaling was reported to orchestrate the inflammatory responses, leading to accumulation of pathogenic monocytes-macrophages promoting lung immunopathology, vascular leakage and suboptimal T cell response with decreased survival [31]. Notably, while early exogenous administration of IFN-β completely protected mice from lethal MERS-CoV infection by inhibiting virus replication and inflammatory cytokine production, delayed IFN-β therapy caused instead a striking increase in IFN-I, ISG, and inflammatory cytokine levels, resulting in fatal disease [32]. In conclusion, in comparison to other respiratory viruses, SARS-CoV-2 infection drives a lower antiviral transcriptional response that is marked by low IFN-I and IFN-III levels and elevated chemokine expression, which could explain the proinflammatory disease state associated with COVID-19 [33]. Overall, these observations outline the critical role of IFN-I in both protective and pathogenic events during CoV infections, thus strengthening the need of fine tuning the IFN-I signaling with respect to the kinetics of CoV replication for an optimal protective response.
4. Multifaceted aspects of IFN-I effects: not only antiviral cytokines
The IFN response represents the major first line of defense against viruses. Under steady state conditions, IFN-I are produced at low levels. However, upon RNA viral recognition through the endosomal (i.e. Toll-like receptors) or cytosolic (RIG-I-like receptors) pattern recognition receptors, the activation of downstream signaling pathways leads to the induction of IFN-I as well as IFN-λ, which, in turn, stimulates the transcription of a variety of ISG, ultimately leading to a broad-spectrum antimicrobial response [34]. The antiviral activity of IFN-I is, however, intimately linked to its impressive capacity to regulate activation and function of various immune cell populations. Among these activities, the activation of antigen-presenting dendritic cells (DC) and natural killer (NK) cells, and the localization, expansion or differentiation of virus-specific T lymphocytes and of antibody-producing B lymphocytes represent key aspects of the antiviral defence. In the early phases of infection, IFN-I contribute to limit viral spread by activating NK cells [35] and enhancing their cytotoxic activity against infected cells [9]. Concurrently, IFN-I variously inhibit or promote the differentiation of precursors into DC (reviewed in [13]). DC exposed to IFN-I show a peculiar ability to cross-present viral antigens to naïve CD8+ T cells [36], promote Th1 cell differentiation, T cell memory turnover [37] and favor the recruitment of NK, T and B cells at the site of infection [38]. Notably, the interplay IFN-I/DC is crucial for the generation of protective antibodies against reference and viral antigens [[39], [40], [41], [42]]. In fact, the stimulation of IFN signaling by exogenous IFN-I or by IFN inducers, such as poly(I:C), strongly enhance the primary antibody response (especially IgG2a) to reference antigens [39] or to a standard influenza vaccine [42] in mice. Interestingly, even in the absence of a direct antiviral effect, the continuous production of IFN-α by a recombinant γ-herpesvirus elicits a significant antiviral immune response attenuating the severity of the infection and protecting against subsequent viral challenge [43]. This evidence supports the concept that IFN-I, rapidly produced in response to viral infection, stimulate the generation of a protective humoral immunity. Based on these premises, IFN-I were used in pilot studies as a vaccine adjuvant in infective diseases [44]. IFN-α local injection in close proximity of the HBV vaccine showed evidence of clinical efficacy in the absence of any toxicity [44].
Overall, although our knowledge of the interplay between SARS-CoV-2 and the host immune system is still limited, we believe that IFN-I can represent not only a direct antiviral treatment restricting early viral spread, but also a valuable tool to enhance humoral and cellular immunity, thus halting viral infection from progressing towards a more severe form of disease.
5. IFN-I and CoV infection: a matter of timing?
The past experience on IFN-I in cancer and chronic viral infection was mainly based on repeated intravenous (i.v.) or subcutaneous (s.c.) injections of high doses (10–20 millions) of IFN-α, often resulting in relevant side effects and refractoriness phenomena that hampered clinical success [45]. The peculiar scenario offered by SARS-CoV-2, characterized by the possible rapid progression towards severe COVID-19 pneumonia, represents a new challenge, and requires a rethinking of the timing and modality of administration of these cytokines. The therapeutic interventions for SARS-CoV-2 need to be tailored to virus pathogenesis and subsequent disease: in early phases, they should counteract virus replication and spreading to favor the development of an effective antiviral immune response; at later time points, treatments should, in principle, reduce the deleterious effects of virus-induced cytokine storm on lung tissues [46].
Although IFN-α and -β demonstrated robust in vitro efficacy against CoV, disappointing results were achieved when transposed to human diseases [[47], [48], [49], [50]]. Upon emergence of previous SARS- and MERS-CoV epidemics, patients were administered off-label with recombinant IFN-α2a, IFN-α2b and IFN-β1, either as monotherapy or in combination, but the rate of success was limited [51]. In some cases, in line with data emerging from animal models [32], the disappointing results obtained with IFN-I were ascribed to the late timing of administration of the cytokine [51], thus suggesting the need for an early therapeutic intervention to fully exploit the properties of these cytokines. Since the beginning of COVID-19 pandemic, IFN-α and -β administration at early time points was included in several clinical studies reported by WHO [2], with the common intent to achieve a general antiviral effect. In fact, IFN-I was often administered together with other compounds (Lopinavir, Ritonavir, Remdesivir, Chloroquine), even though no evidence-based or mechanism-driven rationale clearly supported these combinations. We believe that the early administration of IFN-β, by counteracting SARS-CoV-2 immune evasion mechanisms and linking innate and adaptive immune response, will result not only in an effective limitation of virus replication, but will also prompt the elicitation of humoral and cellular immune response. The beneficial effect of early IFN-β supplementation might be potentially higher in patients likely experiencing an age-related impairment of IFN-I response (Fig. 1).
Besides being less effective if given when viral replication is reaching its peak, attention should be paid to the possible adverse effects of IFN-I administration when SARS-CoV-2 cytokine release syndrome is already ongoing. It is clear that cytokine accumulation observed in the plasma of severe COVID-19 patients is not the result of systemic immune activation, but it is rather caused by virus-mediated stimulation of alveolar macrophages and lung epithelial cells [52]. Nevertheless, we cannot exclude that the immunomodulation phenomena occurring upon IFN-α or β systemic treatment may concur in the stimulation of pro-inflammatory cytokines in the lung [53], thus causing an additive effect that can eventually exacerbate SARS-CoV-2 immunopathology. For this reason, we believe that the most appropriate therapeutic window for a safe and effective use of IFN-α or β administration is limited to the first 10 days since the SARS-CoV-2 diagnosis. In light of the evidence available so far, the use of IFN-I for the treatment of severe COVID-19 patients should be discouraged.
6. Mucosal delivery of IFN-I: is this strategy ready for efficiently fighting COVID-19 pandemic?
Since the respiratory mucosa is the primary portal of entry for SARS-CoV-2, there is increasing interest in developing mucosal treatments for either prevention or treatment of COVID-19. The first attempts to deliver IFN-I to the nasal mucosa to counteract respiratory infections date back in the 1970s [54]. Since then, intranasal administration of IFN-I has been repeatedly explored as prophylactic measure against respiratory infections with variable results that mostly depended on the dose and on the type of patients [55]. The intranasal route has also been explored for the delivery of IFN-I [56] or IFN-I-inducers [57] as adjuvants for influenza vaccines in animal models. In all these studies, intranasal IFN-I was well tolerated and devoid of any evident toxicity. However, concerns exist towards the possibility that the intranasal route may unwantedly deliver compounds to the central nervous system, thus causing toxicity [58]. Among mucosal routes, sublingual administration is gaining interest due to the rapid bioavailability of the applied compound and the lack of hepatic or neural toxicity. In healthy individuals an oral low dosage IFN-α preparation proved safe and effective as prophylaxis against respiratory viral infections [59]. The data available thus far suggest that the beneficial effects of sublingual IFN-I may reside in the local interactions between the cytokine and specific cell populations, including the intraepithelial γδ T cells, which enter the circulation to mediate the antiviral effects of IFN-I [60]. In contrast to the parenteral administration, mucosal administration of IFN-I may have the advantage of being less toxic (if any) and easy to perform, while retaining the capability of inducing both local and systemic effects potentially protective in the very early phases of SARS-CoV-2 infection. Several clinical trials using inhaled IFN-α either as monotherapy or in combination with other treatments have been authorized so far worldwide [2]. In a study recently published on MedRxiv, intranasal administration of IFN-α to medical staff at risk of SARS-CoV-2 infection results in complete protection [61]. Since the beneficial effects of mucosal IFN-I are critically dependent on the timing of administration with regard to the stage of the immune or inflammatory stimulus, further studies are needed to confirm IFN-I efficacy against SARS-CoV-2 infection and to better characterize the biological activities, the mechanisms of action and the optimal modalities of mucosal administration.
7. Closing remarks and perspectives
The COVID-19 outbreak has emphasized the urgent need to identify antiviral agents immediately available to combat SARS-CoV-2 infection. As shown in Table 1 , among the many trials authorized all over the world to test the possible effectiveness of different drug candidates, almost 50 trials, to the best of our knowledge, imply the use of either IFN-α, or IFN-β at different dosages and by different delivery routes, alone as well as in combination with other drugs [2]. While such a complex scenario testifies the current interest in the potential use of IFN-I against COVID-19 pandemic [62], it also gives evidence of the lack of consensus on the best formulation, dose and delivery route for these cytokines.
Table 1.
Current clinical trials assessing IFN-I activity on confirmed SARS-CoV-2 patients§.
| Trial ID | Status | IFN subtype | Alone or in Combination | Age | Delivery route |
|---|---|---|---|---|---|
| ChiCTR2000029638 | Recruiting | Supercompound | Alone | 18−75 | mucosal |
| ChiCTR2000030117 | Recruiting | Alpha | Combination (antivirals) | 18−70 | |
| ChiCTR2000030013 | Not Recruiting | Alpha | Alone | n.a.‡ | |
| NCT04254874 | Recruiting | Peg-Alpha-2b | Combination (antivirals) | >18 | |
| NCT04349410 | Recruiting | Alpha-2b | Alone | all | |
| ChiCTR2000029600 | Recruiting | Alpha | Alone & in combination | 16−75 | |
| ChiCTR2000029756 | Recruiting | Alpha | Alone | 18−60 | |
| ChiCTR2000031196 | Recruiting | Alpha | Combination (antivirals) | 16−85 | |
| NCT04293887 | Not recruiting | Alpha-1b | Combination (standard care) | >18 | |
| NCT04291729 | Not recruiting | n.a. | Combination (antivirals) | 18−75 | |
| NCT04320238 | Recruiting | Alpha-1b | Alone & in combination | 18−65 | |
| ChiCTR2000030535 | Recruiting | Alpha | Combination (antivirals) | n.a. | |
| NCT04251871 | Recruiting | Alpha | Combination (antivirals + traditional chinese medicine) | 14−80 | |
| ChiCTR2000030480 | Recruiting | Alpha-1b | Alone | 18−110 | |
| NCT04275388 | Not recruiting | Alpha | Combination (antivirals) | <100 | |
| ChiCTR2000029989 | Not Recruiting | Alpha-1b | Alone | >60 | |
| NCT04273763 | Recruiting | Alpha | Combination (antivirals) | 18−80 | |
| ChiCTR2000030166 | Not Recruiting | Alpha | Combination (antivirals) | n.a. | systemic |
| NCT04379518 | Not recruiting | Alpha-2b | Combination (Rintatolimod) | >18 | |
| RPCEC00000307 | Recruiting | Alpha-2b | Combination (antivirals + antibiotics + hydroxychloroquine) | >19 | |
| IRCT20161206031256N3 | Recruiting | Alpha-2a, Beta-1a | Combination (standard care) | >18 | |
| ChiCTR2000030922 | Recruiting | Long acting Alpha-2a | Combination (antivirals) | 18−65 | n.a. |
| ChiCTR2000029387 | Recruiting | Alpha | Combination (antivirals) | 18−65 | n.a. |
| IRCT20200511047396N1 | Recruiting | Beta-1a | Alone | >18 | mucosal |
| IRCT20080901001165N53 | Recruiting | Beta-1a | Combination (standard of care) | >18 | |
| NCT04385095 | Recruiting | Beta-1a | Alone | >18 | |
| NCT04315948 | Recruiting | Beta-1a | Combination (antivirals) | >18 | |
| EUCTR2020−001023-14-GB | Authorised | Beta | Combination (antivirals) | n.a. | |
| ISRCTN83971151 | Recruiting | Beta | Combination (antivirals) | n.a. | systemic |
| IRCT20151227025726N12 | Recruiting | Beta | Combination (antivirals) | >18 | |
| IRCT20100228003449N28 | Recruiting | Beta | Combination (antivirals) | 18−75 | |
| NCT04276688 | Recruiting | Beta | Combination (antivirals) | >18 | |
| NCT04350281 | Recruiting | Beta-1b | Combination (hydroxychloroquine) | >18 | |
| IRCT20200412047042N1 | Not Recruiting | Beta | Combination (antivirals + hydroxychloroquine) | n.a. | |
| CTRI/2020/04/024,773 | Recruiting | Beta-1a | Combination (antivirals) | 18−99 | |
| NCT04324463 | Recruiting | Beta | Alone & in combination | >18 | |
| IRCT20100228003449N27 | Recruiting | Beta-1b | Combination (antivirals) | 18−65 | |
| EUCTR2020−001366-11-ES/IE/IT/LT/PT/RO/LV; IRCT20200405046953N1; PER-010−20 | Authorised | Beta-1a | Combination (antivirals) | >18 | |
| NCT04350671 | Recruiting | Beta-1a | Combination (antivirals + hydroxychloroquine) | >50 | |
| IRCT20190804044429N1 | Recruiting | Beta-1b | Combination (antivirals + hydroxychloroquine) | >18 | |
| IRCT20200516047468N1 | Not Recruiting | Beta | Combination (antibiotics + Vit.D) | >18 | |
| EUCTR2020−001262-11-ES | Authorised | Beta-1b | Combination (Standard of care) | >18 | |
| NCT04350684 | Recruiting | Beta-1a | Combination (antivirals + hydroxychloroquine) | >18 | n.a. |
| IRCT20120225009124N4 | Recruiting | Beta | Combination (antivirals + hydroxychloroquine + dexamethasone) | 18−70 | n.a. |
| NCT04343768 | Not recruiting | Beta-1a, Beta-1b | Combination (antivirals + hydroxychloroquine) | >18 | n.a. |
Data updated to June 18, 2020; ‡n.a.: not available.
In some cases, and in most of the Chinese studies, IFN-α and IFN-β administration by either inhalation or intranasal instillation is proposed, assuming that a mucosal delivery of IFN-I, besides being targeted at the infection site, could be more compliant and less toxic than the parental administration, especially for prophylaxis in subjects at high risk of infection or in early phases of household management of infected patients. However, the mechanisms of action of intranasal IFN-I are still poorly identified. Further studies are needed to characterize the pharmacodynamics and pharmacokinetics of mucosal IFN-I preparation, thus defining the risks of central nervous system toxicity [especially when IFN-β is used [63] and achieving a consensus on this strategy. By contrast, much more is known on the pharmacodynamics and pharmacokinetics of IFN-α and IFN-β administered by both s.c. and i.v routes, their mechanisms of action and toxicity limitations. A continuous systemic treatment, somehow inspired by the conventional use of these cytokines in patients with chronic hepatitis C infection and cancer, has been suggested in some of the emerging COVID-19 clinical studies. However, taking into account some critical aspects related to toxicity and refractoriness recently underlined [11,45] and discussed above, we believe this treatment schedule should be discouraged in favor of a discontinuous treatment schedule.
In the majority of the proposed studies, IFN-α and -β are being combined with other drugs, mainly antivirals, although in some cases the rationale for combination is poorly defined. In a recent randomized, phase 2 trial examining the combination of IFN-β1b, lopinavir/ritonavir and ribavirin compared with lopinavir/ritonavir alone, the investigators observed significant symptoms alleviation, shortening of the duration of viral shedding and of hospital stay only in IFN-treated patients with mild to moderate COVID-19 [64]. Notably, due to the absence of single treatment arms in this clinical study, it cannot be discerned whether the specific contribution of IFN-β1a to clinical outcome improvement was a direct antiviral or immune-mediated effect. Thus, further studies to investigate the clinical impact of IFNβ-1b alone in COVID-19 patients are warranted [65].
While some of the above-mentioned issues will be clarified once the results of more preclinical and early phase clinical studies are available, we believe that the knowledge of IFN-I biology and the past experience of the clinical use of these cytokines should be carefully considered in the design of new clinical studies against COVID-19. In the light of the current information on SARS-CoV-2 pathogenesis, we speculate that IFN-I system integrity may be, at least in part, responsible for the spontaneous resolution of SARS-CoV-2 infection in the majority of infected patients (estimated around 80 %). We assume that in the remaining 20 % of people a defective IFN-I system may favor SARS-CoV-2 spread, eventually causing the development of severe forms of COVID-19 and dismal prognosis. We believe that this fraction of the population, in which age, sex, comorbidities and genetic predisposition, may represent determinants of IFN impairment and important cofactors in disease outcome [[19], [20], [21]], could greatly benefit from a time-limited, intermittent, systemic (s.c. or i.m.) IFN-I administration (2–4 million per injection) at the earliest time of SARS-CoV-2 diagnosis (Fig. 1).
Further research efforts should be dedicated to understand the role of the endogenous IFN-I signaling in the susceptibility to SARS-CoV-2 infection and in the progression to the severe forms of COVID-19, in order to identify markers of resistance to the disease and tailor IFN treatments to the patients with the most severe IFN-I system biological impairments.
In conclusion, we believe that to optimally exploit its multiple biological activities against COVID-19 pandemic, low-dose, intermittent IFN-I treatment schedules should be used in the early stages of infection to compensate for virus-induced IFN-impairment or IFN-deficiency in the frail population, thus accelerating virus clearance and avoiding the progression to the severe forms of COVID-19.
Authors contribution
FB conceptualized the manuscript; EA and LB led the review process, made substantial contributions to discussions of the content and drafted the initial manuscript together with FB; SG and LC intellectually contributed to discussions of the content and to the development, writing and revision of the manuscript. All authors reviewed, edited and approved the manuscript before submission.
Declaration of Competing Interest
The authors declare no competing interests.
Biographies

Eleonora Aricò graduated in Biological Sciences in 1998 at the University “La Sapienza” in Rome, where she also received the specialization degree in “Applied Genetics” in 2002. Since then, she works as Researcher at the Istituto Superiore di Sanità, in Rome, where she carries out her research activity in the field of cancer immunology and immunotherapy, in both animal models and clinical studies, with a particular focus on type I Interferons. She worked as visiting fellow at the University of Edinburgh, Scotland, UK, in the context of an European Grant on type I IFN as immune adjuvant (2000). She also spent two years at the Department of Transfusion Medicine of the National Institute of Health (Bethesda, US), working for a joint Italy-US program on clinical studies with IFN-I (2004–2006). In 2017, she joined FaBioCell, the cell factory of the Istituto Superiore di Sanità, where she takes part in the development and clinical application of cell-based immunotherapy strategies for cancer and regenerative diseases.

Laura Bracci is Researcher at the Department of Oncology and Molecular Medicine of the Istituto Superiore di Sanità. She completed her PhD in Medical Microbiology and Immunology in 2005 and the same year she joined the Unit of Onco-immunology at the Department of Research at the University Hospital of Basel (Switzerland) where she carried out studies on DC biology in the context of TLR stimulation. She performed and coordinated studies aimed at disclosing the mechanisms underlying the immunoadjuvant activities of systemic and mucosal type I Interferons in both influenza infection as well as tumor models. The research activity is documented by thirty-six publications in international peer-reviewed journals (H-index: 21) in the fields of interferon research, infectious diseases, oncology and immunology.

Luciano Castiello obtained his PhD in Biology Applied to Human Health, University of Rome “Roma Tre” (Rome, Italy). He is currently the Qualified Person of the GMP facility of the Istituto Superiore di Sanità. Previously, from 2009 to 2013 he was visiting fellow at the Cell Processing Section of the Clinical Center of the National Institutes of Health (Bethesda, MD, USA). Winner of the Marie Curie Sklodovska Fellowship in 2013 at the Istituto Superiore di Sanità, he has also worked at the Pasteur Institute of Rome. He has published more than twenty scientific papers mainly in oncology, cellular therapies, and immunology.

Sandra Gessani is Senior Researcher at the Center for Gender-specific medicine of the Istituto Superiore di Sanità. She has a long lasting experience on the mechanisms regulating IFN expression, both in the antiviral and the antitumor response. An important achievement was breaking the dogma that type II IFN expression was a prerogative of lymphoid cells by demonstrating its constitutive expression in macrophages and providing the first example for its autocrine regulation in myeloid cells (Di Marzio et al., J. Exp. Med. 1994). Dr. Gessani’s group also pioneered studies on the functional interaction between human DC and γδ T lymphocytes (Conti et al., J. Immunol. 2005). Her recent work focused on the relationship among diet, inflammation and cancer (Donninelli et al., Frontiers Immunol. 2017; Del Cornò et al. Frontiers Immunol. 2019).

Filippo Belardelli received his degree in Biological Sciences in 1975 at the University “La Sapienza” in Rome, where he obtained the specialization degree in Microbiology in 1979. In 1980, he became staff investigator of the Laboratory of Virology at the Istituto Superiore di Sanità (ISS), where was appointed as Section Director in 1983. He was the Director of the Department of Cell Biology and Neurosciences (2006–2011) and of the Department of Hematology, Oncology and Molecular Medicine (2011–2016) at the ISS. Dr. Belardelli carried out important biomedical research activities at the ISS as well as in several prestigious laboratories, including the Laboratory of Viral Oncology of the “Institut de Recherches Scientifiques sur le Cancer” in Villejuif and the Department of Immunology of the Scripps Research Institute in La Jolla. The research activity is documented by approximately 290 publications in international journals (H-index: 59) regarding the fields of interferon and cytokine research, infectious diseases, oncology and immunology. In 2017, he became Senior Research Associate of the Institute of Translational Pharmacology of the Italian National Research Council.
Footnotes
Supplementary material related to this article can be found, in the online version, at doi:https://doi.org/10.1016/j.cytogfr.2020.07.010.
Appendix A. Supplementary data
The following is Supplementary data to this article:
References
- 1.Hung I.F.-N., Lung K.-C., Tso E.Y.-K., Liu R., Chung T.W.-H., Chu M.-Y., Ng Y.-Y., Lo J., Chan J., Tam A.R., Shum H.-P., Chan V., Wu A.K.-L., Sin K.-M., Leung W.-S., Law W.-L., Lung D.C., Sin S., Yeung P., Yip C.C.-Y., Zhang R.R., Fung A.Y.-F., Yan E.Y.-W., Leung K.-H., Ip J.D., Chu A.W.-H., Chan W.-M., Ng A.C.-K., Lee R., Fung K., Yeung A., Wu T.-C., Chan J.W.-M., Yan W.-W., Chan W.-M., Chan J.F.-W., Lie A.K.-W., Tsang O.T.-Y., Cheng V.C.-C., Que T.-L., Lau C.-S., Chan K.-H., To K.K.-W., Yuen K.-Y. Triple combination of interferon beta-1b, lopinavir–ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet. 2020;395:1695–1704. doi: 10.1016/S0140-6736(20)31042-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.2020. International Clinical Trials Registry Platform (ICTRP)https://www.who.int/ictrp/data/en/ [Google Scholar]
- 3.Negishi H., Taniguchi T., Yanai H. The interferon (IFN) class of cytokines and the IFN regulatory factor (IRF) transcription factor family. Cold Spring Harb. Perspect. Biol. 2018;10 doi: 10.1101/cshperspect.a028423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Park A., Iwasaki A., Type I., Type I. Interferons – induction, signaling, evasion, and application to combat COVID-19. Cell Host Microbe. 2020;27:870–878. doi: 10.1016/j.chom.2020.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Schreiber G., Piehler J. The molecular basis for functional plasticity in type I interferon signaling. Trends Immunol. 2015;36:139–149. doi: 10.1016/j.it.2015.01.002. [DOI] [PubMed] [Google Scholar]
- 6.Lokugamage K.G., Hage A., Schindewolf C., Rajsbaum R., Menachery V.D. Cold Spring Harbor Laboratory; 2020. SARS-CoV-2 is Sensitive To Type I Interferon Pretreatment. [DOI] [Google Scholar]
- 7.Hensley L.E., Fritz E.A., Jahrling P.B., Karp C.L., Huggins J.W., Geisbert T.W. Interferon-β 1a and SARS coronavirus replication. Emerg. Infect. Dis. 2004;10:317–319. doi: 10.3201/eid1002.030482. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Scagnolari C., Vicenzi E., Bellomi F., Stillitano M.G., Pinna D., Poli G., Clementi M., Dianzani F., Antonelli G. Increased sensitivity of SARS-coronavirus to a combination of human type I and type II interferons. Antivir. Ther. (Lond.) 2004;9:1003–1011. [PubMed] [Google Scholar]
- 9.Vilcek J. Fifty years of interferon research: aiming at a moving target. Immunity. 2006;25:343–348. doi: 10.1016/j.immuni.2006.08.008. [DOI] [PubMed] [Google Scholar]
- 10.Lin F.-C., Young H.A. Interferons: Success in anti-viral immunotherapy. Cytokine Growth Factor Rev. 2014;25:369–376. doi: 10.1016/j.cytogfr.2014.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Aricò E., Castiello L., Capone I., Gabriele L., Belardelli F. Type i interferons and cancer: an evolving story demanding novel clinical applications. Cancers (Basel). 2019;11 doi: 10.3390/cancers11121943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dumitrescu L., Constantinescu C.S., Tanasescu R. Recent developments in interferon-based therapies for multiple sclerosis. Expert Opin. Biol. Ther. 2018;18:665–680. doi: 10.1080/14712598.2018.1462793. [DOI] [PubMed] [Google Scholar]
- 13.Rizza P., Moretti F., Capone I., Belardelli F. Role of type I interferon in inducing a protective immune response: perspectives for clinical applications. Cytokine Growth Factor Rev. 2015;26:195–201. doi: 10.1016/j.cytogfr.2014.10.002. [DOI] [PubMed] [Google Scholar]
- 14.Fung S.-Y., Yuen K.-S., Ye Z.-W., Chan C.-P., Jin D.-Y. A tug-of-war between severe acute respiratory syndrome coronavirus 2 and host antiviral defence: lessons from other pathogenic viruses. Emerg. Microbes Infect. 2020;9:558–570. doi: 10.1080/22221751.2020.1736644. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Zou L., Ruan F., Huang M., Liang L., Huang H., Hong Z., Yu J., Kang M., Song Y., Xia J., Guo Q., Song T., He J., Yen H.-L., Peiris M., Wu J. SARS-CoV-2 viral load in upper respiratory specimens of infected patients. N. Engl. J. Med. 2020;382:1177–1179. doi: 10.1056/NEJMc2001737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Arons M.M., Hatfield K.M., Reddy S.C., Kimball A., James A., Jacobs J.R., Taylor J., Spicer K., Bardossy A.C., Oakley L.P., Tanwar S., Dyal J.W., Harney J., Chisty Z., Bell J.M., Methner M., Paul P., Carlson C.M., McLaughlin H.P., Thornburg N., Tong S., Tamin A., Tao Y., Uehara A., Harcourt J., Clark S., Brostrom-Smith C., Page L.C., Kay M., Lewis J., Montgomery P., Stone N.D., Clark T.A., Honein M.A., Duchin J.S., Jernigan J.A. Presymptomatic SARS-CoV-2 infections and transmission in a skilled nursing facility. N. Engl. J. Med. 2020;382:2081–2090. doi: 10.1056/NEJMoa2008457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Vabret N., Britton G.J., Gruber C., Hegde S., Kim J., Kuksin M., Levantovsky R., Malle L., Moreira A., Park M.D., Pia L., Risson E., Saffern M., Salomé B., Esai Selvan M., Spindler M.P., Tan J., van der Heide V., Gregory J.K., Alexandropoulos K., Bhardwaj N., Brown B.D., Greenbaum B., Gümüş Z.H., Homann D., Horowitz A., Kamphorst A.O., Curotto de Lafaille M.A., Mehandru S., Merad M., Samstein R.M., Agrawal M., Aleynick M., Belabed M., Brown M., Casanova-Acebes M., Catalan J., Centa M., Charap A., Chan A., Chen S.T., Chung J., Bozkus C.C., Cody E., Cossarini F., Dalla E., Fernandez N., Grout J., Ruan D.F., Hamon P., Humblin E., Jha D., Kodysh J., Leader A., Lin M., Lindblad K., Lozano-Ojalvo D., Lubitz G., Magen A., Mahmood Z., Martinez-Delgado G., Mateus-Tique J., Meritt E., Moon C., Noel J., O’Donnell T., Ota M., Plitt T., Pothula V., Redes J., Reyes Torres I., Roberto M., Sanchez-Paulete A.R., Shang J., Schanoski A.S., Suprun M., Tran M., Vaninov N., Wilk C.M., Aguirre-Ghiso J., Bogunovic D., Cho J., Faith J., Grasset E., Heeger P., Kenigsberg E., Krammer F., Laserson U. Immunology of COVID-19: current state of the science. Immunity. 2020 doi: 10.1016/j.immuni.2020.05.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yu S.-Y. Gene expression profiles in peripheral blood mononuclear cells of SARS patients. World J. Gastroenterol. 2005;11:5037. doi: 10.3748/wjg.v11.i32.5037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z., Xiang J., Wang Y., Song B., Gu X., Guan L., Wei Y., Li H., Wu X., Xu J., Tu S., Zhang Y., Chen H., Cao B. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/s0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Conti P., Younes A. Coronavirus COV-19/SARS-CoV-2 affects women less than men: clinical response to viral infection. J. Biol. Regul. Homeost. Agents. 2020;34 doi: 10.23812/Editorial-Conti-3. [DOI] [PubMed] [Google Scholar]
- 21.Bonafè M., Prattichizzo F., Giuliani A., Storci G., Sabbatinelli J., Olivieri F. 2020. Inflamm-Aging: Why Older Men Are the Most Susceptible to SARS-Cov-2 Complicated Outcomes; pp. 1–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Márquez E.J., han Chung C., Marches R., Rossi R.J., Nehar-Belaid D., Eroglu A., Mellert D.J., Kuchel G.A., Banchereau J., Ucar D. Sexual-dimorphism in human immune system aging. Nat. Commun. 2020;11:1–17. doi: 10.1038/s41467-020-14396-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y., Xie C., Ma K., Shang K., Wang W., Tian D.-S. Dysregulation of immune response in patients with COVID-19 in Wuhan, China. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Acharya D., Liu G., Gack M.U. Dysregulation of type I interferon responses in COVID-19. Nat. Rev. Immunol. 2020:1–2. doi: 10.1038/s41577-020-0346-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Reghunathan R., Jayapal M., Hsu L.Y., Chng H.H., Tai D., Leung B.P., Melendez A.J. Expression profile of immune response genes in patients with severe acute respiratory syndrome. BMC Immunol. 2005;6:2. doi: 10.1186/1471-2172-6-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Faure E., Poissy J., Goffard A., Fournier C., Kipnis E., Titecat M., Bortolotti P., Martinez L., Dubucquoi S., Dessein R., Gosset P., Mathieu D., Guery B. Distinct immune response in two MERS-CoV-infected patients: Can we go from bench to bedside? PLoS One. 2014;9 doi: 10.1371/journal.pone.0088716. e88716. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hadjadj J., Yatim N., Barnabei L., Corneau A., Boussier J., Pere H., Charbit B., Bondet V., Chenevier-Gobeaux C., Breillat P., Carlier N., Gauzit R., Morbieu C., Pene F., Marin N., Roche N., Szwebel T.-A., Smith N., Merkling S., Treluyer J.-M., Veyer D., Mouthon L., Blanc C., Tharaux P.-L., Rozenberg F., Fischer A., Duffy D., Rieux-Laucat F., Kerneis S., Terrier B. Impaired type I interferon activity and exacerbated inflammatory responses in severe Covid-19 patients. MedRxiv. 2020 doi: 10.1101/2020.04.19.20068015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Trouillet-Assant S., Viel S., Gaymard A., Pons S., Richard J.-C., Perret M., Villard M., Brengel-Pesce K., Lina B., Mezidi M., Bitker L., Belot A., Mouton W., Oriol G., Compagnon C., Generenaz L., Cheynet V., Ader F., Becker A., Benech N., Chauvelot P., Chidiac C., Conrad A., Ferry T., Miailhes P., Perpoint T., Perry M., Pouderoux C., Roux S., Triffault-Fillit C., Valour F., Hodane Y., Chauvelot L., Chabert P., Provoost J., David G., Folliet L., Lecam P., Billaud G., Bouscambert M., Escuret V., Frobert E., Bal A., Destras G., Josset L., Morfin F., Munier C., Valette M., Venet F., Garnier L., Pescarmona R., Lombard C., Walzer T. Type I IFN immunoprofiling in COVID-19 patients. J. Allergy Clin. Immunol. 2020;0 doi: 10.1016/j.jaci.2020.04.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Yoshikawa T., Hill T.E., Yoshikawa N., Popov V.L., Galindo C.L., Garner H.R., Peters C.J., Te Tseng C. Dynamic innate immune responses of human bronchial epithelial cells to severe acute respiratory syndrome-associated coronavirus infection. PLoS One. 2010;5 doi: 10.1371/journal.pone.0008729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Menachery V.D., Yount B.L., Josset L., Gralinski L.E., Scobey T., Agnihothram S., Katze M.G., Baric R.S. Attenuation and Restoration of Severe Acute Respiratory Syndrome Coronavirus Mutant Lacking 2’-O-Methyltransferase Activity. J. Virol. 2014;88:4251–4264. doi: 10.1128/jvi.03571-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Channappanavar R., Fehr A.R., Vijay R., Mack M., Zhao J., Meyerholz D.K., Perlman S. Dysregulated type I interferon and inflammatory monocyte-macrophage responses cause lethal pneumonia in SARS-CoV-Infected mice. Cell Host Microbe. 2016;19:181–193. doi: 10.1016/j.chom.2016.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Channappanavar R., Fehr A.R., Zheng J., Wohlford-Lenane C., Abrahante J.E., Mack M., Sompallae R., McCray P.B., Meyerholz D.K., Perlman S. IFN-I response timing relative to virus replication determines MERS coronavirus infection outcomes. J. Clin. Invest. 2019;129:3625–3639. doi: 10.1172/JCI126363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Blanco-Melo D., Nilsson-Payant B.E., Liu W.C., Uhl S., Hoagland D., Møller R., Jordan T.X., Oishi K., Panis M., Sachs D., Wang T.T., Schwartz R.E., Lim J.K., Albrecht R.A. B.R. tenOever, imbalanced host response to SARS-CoV-2 drives development of COVID-19. Cell. 2020 doi: 10.1016/j.cell.2020.04.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Akira S., Uematsu S., Takeuchi O. Pathogen recognition and innate immunity. Cell. 2006;124:783–801. doi: 10.1016/j.cell.2006.02.015. [DOI] [PubMed] [Google Scholar]
- 35.Müller L., Aigner P., Stoiber D. Type I interferons and natural killer cell regulation in cancer. Front. Immunol. 2017;8:304. doi: 10.3389/fimmu.2017.00304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Le Bon A., Etchart N., Rossmann C., Ashton M., Hou S., Gewert D., Borrow P., Tough D.F. Cross-priming of CD8+ T cells stimulated by virus-induced type I interferon. Nat. Immunol. 2003;4:1009–1015. doi: 10.1038/ni978. [DOI] [PubMed] [Google Scholar]
- 37.Tough D.F., Borrow P., Sprent J. Induction of bystander T cell proliferation by viruses and type I interferon in vivo. Science (80-.) 1996;272:1947–1950. doi: 10.1126/science.272.5270.1947. [DOI] [PubMed] [Google Scholar]
- 38.McNab F., Mayer-Barber K., Sher A., Wack A., O’Garra A. Type I interferons in infectious disease. Nature Publishing Group. 2015 doi: 10.1038/nri3787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Le Bon A., Schiavoni G., D’Agostino G., Gresser I., Belardelli F., Tough D.F. Type i interferons potently enhance humoral immunity and can promote isotype switching by stimulating dendritic cells in vivo. Immunity. 2001;14:461–470. doi: 10.1016/s1074-7613(01)00126-1. [DOI] [PubMed] [Google Scholar]
- 40.Santini S.M., Lapenta C., Logozzi M., Parlato S., Spada M., Di Pucchio T., Belardelli F. Type I interferon as a powerful adjuvant for monocyte-derived dendritic cell development and activity in vitro and in Hu-PBL-SCID mice. J. Exp. Med. 2000;191:1777–1788. doi: 10.1084/jem.191.10.1777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Lapenta C., Santini S.M., Logozzi M., Spada M., Andreotti M., Di Pucchio T., Parlato S., Belardelli F. Potent immune response against HIV-1 and protection from virus challenge in hu-PBL-SCID mice immunized with inactivated virus-pulsed dendritic cells generated in the presence of IFN-α. J. Exp. Med. 2003;198:361–367. doi: 10.1084/jem.20021924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Proietti E., Bracci L., Puzelli S., Di Pucchio T., Sestili P., De Vincenzi E., Venditti M., Capone I., Seif I., De Maeyer E., Tough D., Donatelli I., Belardelli F. Type I IFN as a natural adjuvant for a protective immune response: lessons from the influenza vaccine model. J. Immunol. 2002;169:375–383. doi: 10.4049/jimmunol.169.1.375. [DOI] [PubMed] [Google Scholar]
- 43.Aricò E., Monque D.M., D’Agostino G., Moschella F., Venditti M., Kalinke U., Allen D.J., Nash A.A., Belardelli F., Ferrantini M. MHV-68 producing mIFNα1 is severely attenuated in vivo and effectively protects mice against challenge with wt MHV-68. Vaccine. 2011;29:3935–3944. doi: 10.1016/j.vaccine.2011.03.092. [DOI] [PubMed] [Google Scholar]
- 44.Miquilena-Colina M.E., Lozano-Rodríguez T., García-Pozo L., Sáez A., Rizza P., Capone I., Rapicetta M., Chionne P., Capobianchi M., Selleri M., Castilletti C., Belardelli F., Lo Iacono O., García-Monzón C. Recombinant interferon-alpha2b improves immune response to hepatitis B vaccination in haemodialysis patients: results of a randomised clinical trial. Vaccine. 2009;27:5654–5660. doi: 10.1016/j.vaccine.2009.07.014. [DOI] [PubMed] [Google Scholar]
- 45.Antonelli G., Scagnolari C., Moschella F., Proietti E. Twenty-five years of type I interferon-based treatment: a critical analysis of its therapeutic use. Cytokine Growth Factor Rev. 2015;26:121–131. doi: 10.1016/j.cytogfr.2014.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Matricardi P.M., Negro R.W.D., Nisini R., First The. Comprehensive immunological model of COVID-19: implications for prevention, diagnosis, and public health measures. Preprint). 2020:1–38. doi: 10.1111/pai.13271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Stockman L.J., Bellamy R., Garner P. SARS: Systematic Review of Treatment Effects. PLoS Med. 2006;3:e343. doi: 10.1371/journal.pmed.0030343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Sheahan T.P., Sims A.C., Leist S.R., Schäfer A., Won J., Brown A.J., Montgomery S.A., Hogg A., Babusis D., Clarke M.O., Spahn J.E., Bauer L., Sellers S., Porter D., Feng J.Y., Cihlar T., Jordan R., Denison M.R., Baric R.S. Comparative therapeutic efficacy of remdesivir and combination lopinavir, ritonavir, and interferon beta against MERS-CoV. Nat. Commun. 2020;11 doi: 10.1038/s41467-019-13940-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Arabi Y.M., Asiri A.Y., Assiri A.M., Aziz Jokhdar H.A., Alothman A., Balkhy H.H., AlJohani S., Al Harbi S., Kojan S., Al Jeraisy M., Deeb A.M., Memish Z.A., Ghazal S., Al Faraj S., Al-Hameed F., AlSaedi A., Mandourah Y., Al Mekhlafi G.A., Sherbeeni N.M., Elzein F.E., Almotairi A., Al Bshabshe A., Kharaba A., Jose J., Al Harthy A., Al Sulaiman M., Mady A., Fowler R.A., Hayden F.G., Al-Dawood A., Abdelzaher M., Bajhmom W., Hussein M.A. Treatment of Middle East respiratory syndrome with a combination of lopinavir/ritonavir and interferon-β1b (MIRACLE trial): statistical analysis plan for a recursive two-stage group sequential randomized controlled trial. Trials. 2020;21:8. doi: 10.1186/s13063-019-3846-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Omrani A.S., Saad M.M., Baig K., Bahloul A., Abdul-Matin M., Alaidaroos A.Y., Almakhlafi G.A., Albarrak M.M., Memish Z.A., Albarrak A.M. Ribavirin and interferon alfa-2a for severe Middle East respiratory syndrome coronavirus infection: a retrospective cohort study. Lancet Infect. Dis. 2014;14:1090–1095. doi: 10.1016/S1473-3099(14)70920-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Zumla A., Chan J.F.W., Azhar E.I., Hui D.S.C., Yuen K.Y. Coronaviruses-drug discovery and therapeutic options. Nat. Rev. Drug Discov. 2016;15:327–347. doi: 10.1038/nrd.2015.37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Coperchini F., Chiovato L., Croce L., Magri F., Rotondi M. The cytokine storm in COVID-19: an overview of the involvement of the chemokine/chemokine-receptor system. Cytokine Growth Factor Rev. 2020;53:25–32. doi: 10.1016/j.cytogfr.2020.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Makris S., Paulsen M., Johansson C. Type I interferons as regulators of lung inflammation. Front. Immunol. 2017;8:259. doi: 10.3389/fimmu.2017.00259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Merigan T.C., Hall T.S., Reed S.E., Tyrrell D.A.J.J., Unit M.R.C.C.C., Hospital H. Inhibition of respiratory virus infection by locally applied Interferon. Lancet. 1973;301:563–567. doi: 10.1016/S0140-6736(73)90714-9. [DOI] [PubMed] [Google Scholar]
- 55.Velkov T., Abdul Rahim N., Zhou Q.T., Chan H.K., Li J. Inhaled anti-infective chemotherapy for respiratory tract infections: successes, challenges and the road ahead. Adv. Drug Deliv. Rev. 2015;85:65–82. doi: 10.1016/j.addr.2014.11.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bracci L., Canini I., Puzelli S., Sestili P., Venditti M., Spada M., Donatelli I., Belardelli F., Proietti E. Type I IFN is a powerful mucosal adjuvant for a selective intranasal vaccination against influenza virus in mice and affects antigen capture at mucosal level. Vaccine. 2005;23:2994–3004. doi: 10.1016/j.vaccine.2004.12.006. [DOI] [PubMed] [Google Scholar]
- 57.Dhakal S., Ghimire S., Renu S., Ross K.A., Lakshmanappa Y.S., Hogshead B.T., Bernardo P., Lee C.W., Wannemuehler M.J., Narasimhan B., Renukaradhya G.J. Evaluation of CpG-ODN-adjuvanted polyanhydride-based intranasal influenza nanovaccine in pigs. Vet. Microbiol. 2019;237:108401. doi: 10.1016/j.vetmic.2019.108401. [DOI] [PubMed] [Google Scholar]
- 58.Thorne R.G., Hanson L.R., Ross T.M., Tung D., Frey W.H. Delivery of interferon-β to the monkey nervous system following intranasal administration. Neuroscience. 2008;152:785–797. doi: 10.1016/j.neuroscience.2008.01.013. [DOI] [PubMed] [Google Scholar]
- 59.Bennett A.L., Smith D.W., Cummins M.J., Jacoby P.A., Cummins J.M., Beilharz M.W. Low-dose oral interferon alpha as prophylaxis against viral respiratory illness: a double-blind, parallel controlled trial during an influenza pandemic year, Influenza Other Respi. Viruses. 2013;7:854–862. doi: 10.1111/irv.12094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Bocci V. Absorption of Cytokines via Oropharyngeal-Associated Lymphoid Tissues: Does an Unorthodox Route Improve the Therapeutic Index of Interferon? Clin. Pharmacokinet. 1991;21:411–417. doi: 10.2165/00003088-199121060-00002. [DOI] [PubMed] [Google Scholar]
- 61.Meng Z., Wang T., Li C., Chen X., Li L., Qin X., Li H., Luo J. An experimental trial of recombinant human interferon alpha nasal drops to prevent coronavirus disease 2019 in medical staff in an epidemic area. MedRxiv. 2020 doi: 10.1101/2020.04.11.20061473. 2020.04.11.20061473. [DOI] [Google Scholar]
- 62.Sallard E., Lescure F.-X., Yazdanpanah Y., Mentre F., Peiffer-Smadja N. Type 1 interferons as a potential treatment against COVID-19. Antiviral Res. 2020;178:104791. doi: 10.1016/j.antiviral.2020.104791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Ross T.M., Martinez P.M., Renner J.C., Thorne R.G., Hanson L.R., Frey W.H. Intranasal administration of interferon beta bypasses the blood-brain barrier to target the central nervous system and cervical lymph nodes: a non-invasive treatment strategy for multiple sclerosis. J. Neuroimmunol. 2004;151:66–77. doi: 10.1016/j.jneuroim.2004.02.011. [DOI] [PubMed] [Google Scholar]
- 64.Fan-Ngai Hung I., Lung K.-C., Yuk-Keung Tso E., Liu R., Wai-Hin Chung T., Chu M.-Y., Ng Y.-Y., Lo J., Chan J.W., Raymond Tam A., Shum H.-P., Chan V., Ka-Lun Wu A., Sin K.-M., Leung W.-S., Law W.-L., Christopher Lung D., Sin S., Yeung P., Chik-Yan Yip C., Ruiqi Zhang R., Yim-Fong Fung A., Yuen-Wing Yan E., Leung K.-H., Daniel Ip J., Wing-Ho Chu A., Chan W.-M.W.-M., Chin-Ki Ng A., Lee R., Fung K., Yeung A., Wu T.-C., Wai-Man Chan J., Yan W.-W., Chan W.-M.W.-M., Fuk-Woo Chan J., Kwok-Wai Lie A., Tak-Yin Tsang O., Chi-Chung Cheng V., Que T.-L., Lau C.-S., Chan K.-H., Kai-Wang To K., Yuen K.-Y., Hung I.F.-N., Lung K.-C., Tso E.Y.-K., Liu R., Chung T.W.-H., Chu M.-Y., Ng Y.-Y., Lo J., Chan J.W., Tam A.R., Shum H.-P., Chan V., Wu A.K.-L., Sin K.-M., Leung W.-S., Law W.-L., Lung D.C., Sin S., Yeung P., Yip C.C.-Y., Zhang R.R., Fung A.Y.-F., Yan E.Y.-W., Leung K.-H., Ip J.D., Chu A.W.-H., Chan W.-M.W.-M., Ng A.C.-K., Lee R., Fung K., Yeung A., Wu T.-C., Chan J.W., Yan W.-W., Chan W.-M.W.-M., Chan J.W., Lie A.K.-W., Tsang O.T.-Y., Cheng V.C.-C., Que T.-L., Lau C.-S., Chan K.-H., To K.K.-W., Yuen K.-Y. Triple combination of interferon beta-1b, lopinavir–ritonavir, and ribavirin in the treatment of patients admitted to hospital with COVID-19: an open-label, randomised, phase 2 trial. Lancet. 2020;395:1695–1704. doi: 10.1016/s0140-6736(20)31042-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Shalhoub S. Interferon beta-1b for COVID-19. Lancet. 2020;395:1670–1671. doi: 10.1016/s0140-6736(20)31101-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.