Abstract
Introduction
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is an emerging viral pathogen that causes the novel coronavirus disease of 2019 (COVID-19) and may result in hypoxemic respiratory failure necessitating invasive mechanical ventilation in the most severe cases.
Objective
This narrative review provides evidence-based recommendations for the treatment of COVID-19 related respiratory failure requiring invasive mechanical ventilation.
Discussion
In severe cases, COVID-19 leads to hypoxemic respiratory failure that may meet criteria for acute respiratory distress syndrome (ARDS). The mainstay of treatment for ARDS includes a lung protective ventilation strategy with low tidal volumes (4–8 mL/kg predicted body weight), adequate positive end-expiratory pressure (PEEP), and maintaining a plateau pressure of < 30 cm H2O. While further COVID-19 specific studies are needed, current management should focus on supportive care, preventing further lung injury from mechanical ventilation, and treating the underlying cause.
Conclusions
This review provides evidence-based recommendations for the treatment of COVID-19 related respiratory failure requiring invasive mechanical ventilation.
Keywords: SARS-CoV-2, COVID-19, Acute respiratory distress syndrome, Respiratory failure, Lung protective strategy, Mechanical ventilation
1. Introduction
The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is an emerging viral pathogen that causes mild illness in some while others progress to respiratory failure requiring invasive mechanical ventilation [1,2]. The disease caused by SARS-CoV-2 has been termed the novel coronavirus disease of 2019 (COVID-19) [3]. Though incidence data are limited, a large case series of 1300 patients with COVID-19 from Italy found that 88% of critically ill patients required mechanical ventilation [4]. The mortality of those placed on mechanical ventilation is 24.5% to 28% in case series and may be even higher as many patients still remain in the hospital [4,5]. One study conducted prior to COVID-19 suggests a lung protective strategy started in the emergency department (ED) is associated with a reduction in hospital mortality, pulmonary complications, and days requiring mechanical ventilation [6]. Therefore, it is important for emergency medicine clinicians to be aware of the management of mechanically ventilated patients, particularly as these patients may be boarding in the ED for an extended period of time.
In the severest form, the characteristics of COVID-19 related respiratory failure may meet the definition of acute respiratory distress syndrome (ARDS) [[7], [8], [9], [10]]. ARDS is defined by the Berlin criteria (Table 1 ) [11] and exists on a spectrum as a heterogeneous syndrome caused by multiple etiologies [12,13]. Practice guidelines from the Surviving Sepsis Campaign and National Institutes of Health (NIH) on the management of COVID-19 recommend a lung protective strategy with a high positive end-expiratory pressure (PEEP) strategy and low tidal volumes (4–8 mL/kg predicted body weight) [[14], [15], [16]]. This review will discuss the physiology underlying COVID-19 related ARDS, lung protective ventilation strategies, individualized approaches to mechanical ventilation, additional therapies, and a recommended approach to mechanical ventilation for the emergency clinician.
Table 1.
The Berlin definition of the acute respiratory distress syndrome [11].
| Clinical Feature | Definition |
|---|---|
| Timing | Develops within one week of clinical insult |
| Chest Imaging | Bilateral opacities not otherwise explained by pleural effusions, lobar collapse, or nodules |
| Origin of Edema | Non-cardiogenic edema; edema not suspected to be from an elevated left atrial pressure causing hydrostatic edema; an echocardiogram may be needed in unclear cases |
| Oxygenation | Mild: PaO2/FiO2 of >200 mm Hg to < 300 mm Hg with PEEP or CPAP ≥5 cmH2O Moderate: PaO2/FiO2 of >100 mm Hg to < 200 mm Hg with PEEP ≥5 cmH2O Severe: PaO2/FiO2< 100 mm Hg with PEEP ≥5 cmH2O |
Abbreviations: FiO2, fraction of inspired oxygen; PaO2, partial pressure of arterial oxygen; PEEP, positive end-expiratory pressure; CPAP, continuous positive airway pressure.
2. Methods
This is a narrative review of invasive mechanical ventilation strategies for COVID-19 related respiratory failure. Authors conducted a literature review of PubMed and Google Scholar using keywords of “ARDS” OR “Acute Respiratory Distress Syndrome” OR “COVID-19” OR “SARS-CoV-2” OR “2019-nCoV” OR “hypoxemic respiratory failure” OR “mechanical ventilation” OR “driving pressure” OR “ventilator-induced lung injury” from 1994 to May 2020. Respiratory physiology studies from 1969 to 1994 were also included. Authors evaluated case reports and series, retrospective and prospective studies, systematic reviews and meta-analyses, and other narrative reviews. Authors also reviewed guidelines and supporting citations of included articles. Authors decided which studies to include for the review by consensus. When available, systematic reviews and meta-analyses were preferentially selected. These were followed sequentially by randomized controlled trials, prospective studies, retrospective studies, case reports, and other narrative reviews when alternate data were not available. Case reports, case controls, cohort studies, randomized clinical trials, meta-analyses and systematic reviews, and narrative reviews were included. Authors decided on the inclusion of 119 studies and included a total of 20 systematic reviews and meta-analyses, 30 randomized controlled trials, 8 prospective studies, 12 retrospective studies, 3 basic science experimental studies, 11 case reports or case series, 22 narrative reviews and 13 expert consensus documents and guidelines.
3. Discussion
3.1. Pathophysiology of acute respiratory distress syndrome and COVID-19
Acute respiratory distress syndrome is a complex and heterogeneous syndrome [11,17]. Causes of ARDS include non-infectious etiologies (e.g., trauma, pancreatitis), pulmonary infections, and non-pulmonary infections [12,17]. However, the common theme is an inflammatory response causing lung and systemic organ injury. A severe, hyperinflammatory, cytokine-mediated lung injury has also been implicated in COVID-19 [9,18]. Pro-inflammatory cytokines may interfere with the normal adaptive response of hypoxic vasoconstriction [19]. Damage to alveolar epithelium and endothelium leads to leakage of protein-rich fluid and non-cardiogenic pulmonary edema [17]. Consequently, the injured lung becomes at greater risk of atelectasis with impairment of surfactant, alveolar edema and hemorrhage, reduced lung compliance, increased ventilation-perfusion mismatching, and right-to-left shunting [17,20]. All of these factors contribute to hypoxemia. The histological characteristic of ARDS is diffuse alveolar damage, though interestingly this finding is not observed in all patients meeting the Berlin definition of ARDS [17]. This pattern of diffuse alveolar damage has also been noted in an autopsy case series of COVID-19 patients [19,21].
ARDS is a clinical diagnosis that relies on the 2012 Berlin definition (Table 1) [11]. Notably, ARDS is defined as an acute process with bilateral lung opacities on imaging not from cardiogenic edema and a partial pressure of arterial oxygen to the fraction of inspired oxygen (PaO2/FiO2) ratio of <300 mm Hg on a positive end-expiratory pressure (PEEP) of at least 5 cm H2O. ARDS is further categorized into mild, moderate, or severe, depending on the degree of impairment. Patients with COVID-19 requiring mechanical ventilation frequently meet the definition of ARDS [7,8].
Lung compliance in COVID-19 related respiratory failure is variable [7,22]. It is important to note that respiratory system compliance of < 40 mL/cm H2O was also originally considered in the Berlin definition of ARDS but was excluded after further evaluation [11]. Studies of ARDS in non-COVID-19 patients have also demonstrated a range of lung compliances and underlying causes [12,13], that are similar to currently published COVID-19 related ARDS patients [10]. This suggests that the treatments of ARDS developed over several decades remain applicable to the range of lung pathology observed in COVID-19 related respiratory failure [10,23]. Moreover, deviation from a lung protective ventilation strategy with a high VT and low PEEP has historically been shown to cause lung injury in animal models [[24], [25], [26], [27]].
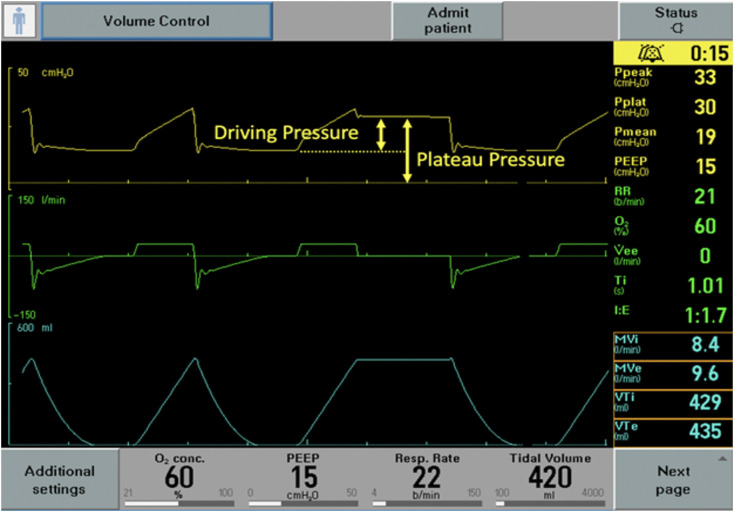
Once ARDS is diagnosed, the treatment focuses on addressing the underlying cause while preventing ventilator induced lung injury (VILI) [24]. There are multiple sources of lung injury (Table 2 ). Volutrauma occurs from excess volume or pressure leading to overdistension (i.e. stretching) of at-risk alveoli [17,24]. Lung injury caused by overdistension may be noted grossly by barotrauma (e.g. pneumothorax, pneumomediastinum) or occur silently on the alveolar level [24]. In ARDS, the functional lung volume is reduced due to alveolar injury, edema, and atelectasis [17]. The reduction in aerated lung space is the underpinning for the low VT of lung protective ventilation (LPV). The delivered VT generates a pressure within the lung. The generated pressure will vary depending on the size of the VT and an individual’s respiratory system compliance [28]. The plateau pressure (PPlat) estimates alveolar pressure, with a high PPlat suggesting alveolar overdistension [24,28]. Though a specific safe pressure threshold is unknown, it is recommended to maintain a PPlat < 30 cmH2O and a driving pressure (PPlat – PEEP) < 15 cm H2O [6,12,29]. The PPlat is measured with a 0.5-second pause at the end of inspiration in a passive patient when inspiratory flow reaches zero (Fig. 1 ) [12]. It is important the patient is passive (i.e. without any spontaneous respiratory effort) during the inspiratory hold, as any patient effort (e.g. expiratory or inspiratory effort) will distort the PPlat measurement. Additional injury may come in the form of atelectrauma—repetitive opening and closing of alveoli leading to alveolar injury and denaturing of surfactant [17,24]. Atelectrauma has been shown to increase inflammatory markers in animal models [12,17,[25], [26], [27]]. Atelectrauma is minimized by using appropriate PEEP to maintain alveolar aeration (i.e. recruitment) throughout the respiratory cycle [24].
Table 2.
Types of ventilator induced lung injury (VILI) [24].
| Injury | Mechanism | Minimization Strategy |
|---|---|---|
| Atelectrauma (Recruitment/derecruitment injury) | Lung injury caused by cyclic opening and collapse of atelectatic, but recruitable lung units. | Ensure appropriate PEEP and tidal volumes. |
| Barotrauma | Lung injury (e.g. pneumothorax, pneumomediastinum, etc.) caused by high transpulmonary pressure disrupting the alveolar structures. | Minimize excessive airway pressure and tidal volumes. |
| Biotrauma | Mechanical lung injury causes up-regulation and release of cytokines with a subsequent pulmonary and systemic inflammatory response causing multi-organ dysfunction. | Lung protective strategy while treating the underlying cause. Consider immunomodulating therapies (e.g. corticosteroids). |
| Oxygen toxicity | Injury caused by the inability of cells to overcome oxygen free radicals, and absorption atelectasis. | Turn down FiO2 as soon as possible to target an oxygen saturation of 92–96%. |
| Patient self-inflicted lung injury (P-SILI) | Intense inspiratory force by the patient causing high transpulmonary pressure swings. | Increase sedation with or without neuromuscular blockade if persistent, excessive, spontaneous respiratory effort is present. |
| Shearing injury | High shear forces at the junction of the collapsed and open lung units causing lung injury. | Use appropriate PEEP to maintain recruitment and low tidal volumes. Modes like airway pressure release ventilation (APRV) may reduce shear stress. |
| Volutrauma | Non-homogenous lung injury caused by alveolar overdistension. | Ensure a low tidal volume of 4–8 mL/kg PBW. |
Fig. 1.
An example of a plateau pressure, checked after an end inspiratory pause when inspiratory flow has reached zero. The plateau pressure is 30 cm H2O, in a volume control mode with a set 420 mL (6 mL/kg PBW) tidal volume. The driving pressure is 15 cm H2O (plateau pressure of 30 cm H2O - PEEP of 15 cm H2O). The driving pressure is related to the static compliance of the respiratory system (CRS) by CRS = Tidal Volume/Driving Pressure. In this patient the CRS is low at 28 mL/cm H2O.
3.2. A lung protective strategy
In COVID-19 related respiratory failure, care is focused on maintaining oxygenation while preventing VILI with lung protective ventilation (LPV). The mainstays of LPV are low VT, sufficient PEEP to maintain lung recruitment, and low airway pressures [17,[30], [31], [32], [33], [34]]. Though ARDS is a heterogeneous syndrome, the following is recommended for all ARDS patients: (1) VT of 4 to 8 mL/kg predicted body weight (PBW) and (2) targeting a PPlat <30 cm H2O (Table 3, Table 4 ) [15,34,35]. Emerging data from several American tertiary care centers support this approach in patients with COVID-19 requiring invasive mechanical ventilation [10].
Table 3.
The conventional lung protective ventilation strategy [12].
| Variable | Setting |
|---|---|
| Tidal Volume | 6 mL/kg PBW (Range: 4–8 mL/kg PBW) |
| Plateau pressure | Less than 30 cm H2O |
| Respiratory rate | Up to 35 breaths per minute, goal of pH 7.30–7.45 but may allow permissive hypercapnia with a pH >7.15 |
| Positive End Expiratory Pressure | Initiate at ≥5 cm H2O Titrate according to ARDSnet lower PEEP/higher FiO2 table |
| Oxygenation target | Titrate FiO2 to: |
| PaO2 | 55–80 mmHg |
| SpO2 | 88–95% |
Abbreviations: PBW (predicted body weight), PEEP (positive end-expiratory pressure).
Table 4.
PEEP/FiO2 titration strategies [12].
| Lower PEEP/FiO2 Combination | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| FiO2 | 0.3 | 0.4 | 0.4 | 0.5 | 0.5 | 0.6 | 0.7 | 0.7 | 0.7 | 0.8 | 0.9 | 0.9 | 0.9 | 1.0 |
| PEEP (cm H2O) | 5 | 5 | 8 | 8 | 10 | 10 | 10 | 12 | 14 | 14 | 14 | 16 | 18 | 18–24 |
| Higher PEEP/FiO2 Combination | ||||||||||||||
| FiO2 | 0.3 | 0.3 | 0.4 | 0.4 | 0.5 | 0.5 | 0.5 | 0.6 | 0.7 | 0.8 | 0.8 | 0.9 | 1.0 | |
| PEEP (cm H2O) | 5–12 | 14 | 14 | 16 | 16 | 18 | 20 | 20 | 20 | 20 | 22 | 22 | 22–24 | |
Abbreviations: PEEP (positive end-expiratory pressure), FiO2 (fraction of inspired oxygen).
The ARDSnet ARMA trial showed a mortality benefit in patients with ARDS using a low (6 mL/kg PBW), as compared to a high (12 mL/kg PBW), VT ventilation strategy, PEEP set by the lower PEEP/FiO2 table (Table 4) and a goal PPlat less than 30 cm H2O (Table 3) [12]. Though an initial improvement in oxygenation with a larger, 12 mL/kg VT may be seen, this trial showed the initial improvement in oxygenation with a larger VT is at the expense of a later increase in mortality [12]. Early initiation of LPV initiated in the ED is associated with improved mortality and patient outcomes [6]. The traditional LPV approach uses a PEEP/FiO2 table to determine the set PEEP based on the degree of hypoxemia and FiO2 requirement (Table 4) [12]. Currently, the Surviving Sepsis Campaign and NIH recommend a higher PEEP strategy over lower PEEP strategy for patients with ARDS due to COVID-19; however, this is a weak recommendation based on low-quality evidence [[14], [15], [16],36]. This prescriptive setting of PEEP has been criticized in COVID-19 because it lacks sufficient individualization to the variable response to PEEP [37].
3.3. An individualized approach
3.3.1. Description of PEEP and recruitment maneuvers in ARDS
The tenants of ARDS management and LPV are low Vt and cautious use of PEEP [17]. ARDS is a heterogeneous disease with a variety of potentially recruitable, PEEP-responsive lung units [38]. This heterogeneity means a single prescribed VT and PEEP impacts each lung unit differently depending on the local level of functional impairment (e.g. edema, atelectasis). An inherent tradeoff with any PEEP application is the need to balance improving oxygenation with potentially inducing VILI [39,40]. PEEP can be beneficial by recruiting and aerating collapsed lung units, improving gas exchange, and minimizing atelectrauma. However, it can also be harmful by over-distending the lung, increasing pulmonary vascular resistance, worsening ventilation and perfusion matching, and inducing hemodynamic instability by decreasing cardiovascular preload. Finding the optimal PEEP may not rest on one variable [22]. The elusive goal to find the optimal PEEP is not new [41] and strives to maximize the beneficial effects of PEEP while minimizing any potential harmful effects.
One method of PEEP adjustment was described in the ARDSnet trial in the form of a high and low PEEP/FiO2 table based on levels of hypoxemia [12] (Table 4). A mortality benefit has not been detected with the high PEEP/FiO2 compared to the low PEEP/FiO2 table [[42], [43], [44], [45]]. Despite lack of proven superiority, current recommendations support a high PEEP strategy for moderate to severe ARDS (PaO2/FiO2 < 200 mm Hg) which is consistent with current COVID-19 recommendations [[14], [15], [16],34,36,46]. The PEEP tables are unable to take into account the extent of individual lung injury and recruitability. For instance, a low PEEP in potentially recruitable lungs does not allow for the beneficial effects of PEEP while a high PEEP strategy with low lung recruitability can lead to over-distention and increased lung injury [47]. Marini and Gattinoni [37] suggest that different COVID-19 ARDS phenotypes require varied management strategies. These two “H” and “L” phenotypes are based on lung CT imaging, compliance, and response to PEEP [22,37,48,49]. However, these phenotypes for COVID-19 have not been validated in other studies and should not form the foundation of therapy. ARDS phenotypes have been previously described [50] and may provide insight into a personalized approach. Personalized ventilator strategies compared to a uniform approach may be helpful; however, caution must be used with personalization because if incorrectly assessed, mortality may increase [51].
There remains controversy surrounding the use of recruitment maneuvers. A recruitment maneuver is a prolonged inspiratory hold on higher levels of CPAP, such as 35–40 cm H2O for 30–40 s [16,52]. Evidence from a systematic review and meta-analysis showed improved oxygenation without an increased risk of barotrauma [52]. However, other studies show recruitment maneuvers with PEEP titration increase mortality compared to a standard PEEP/FIO2 table [53]. In COVID-19, if hypoxemia exists despite optimization of ventilator settings, recruitment maneuvers may be considered while monitoring for harmful effects such as oxygen desaturation, hypotension, or barotrauma [15,16].
3.3.2. Driving pressure
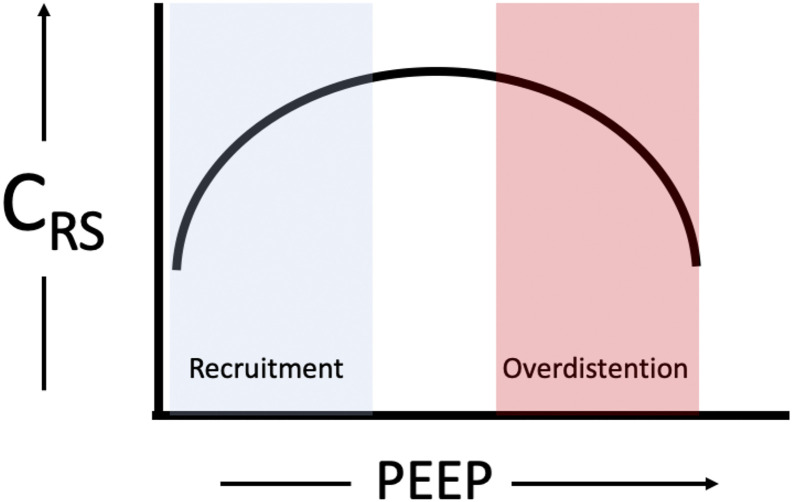
Driving pressure can be calculated easily at the bedside by obtaining the PPlat during an end-inspiratory pause in a passive patient in a volume-targeted mode of ventilation. It is calculated by PPlat - PEEP. The driving pressure reflects the static compliance of the respiratory system (CRS) by the equation of CRS= VT /(PPlat - PEEP). As the equation suggests, a change in VT or a change in pressure will affect the compliance of the respiratory system. It is possible that a change in PEEP may decrease the pressure associated with a VT (i.e. improve the CRS) if it is able to recruit previously non-aerated lung (Fig. 2 ) [29,54]. Similarly, it is also possible that an increase in PEEP could worsen CRS if an increase in pressure does not improve recruitment and instead causes overdistension (Fig. 2), leading to lung injury and worsening dead space or causing hemodynamic compromise [55]. The driving pressure may offer a better quantification of functional lung size as compared to PBW for a set VT because PBW is proportional to total lung size and not the reduced, functional lung size in ARDS [51,55].
Fig. 2.
A representation of the relationship between compliance of the respiratory system (CRS) and PEEP. If increasing PEEP improves recruitment, by aeration of previously non-aerated lung, then compliance will improve until the lungs are overdistended and compliance worsens.
Amato et. al. performed a retrospective review [29] of the ARDSnet trial data and showed that driving pressure was the ventilation variable that best stratified mortality risk in ARDS. Higher mortality was noted with a higher PPlat only when higher driving pressures were present. Similarly, protective effects of PEEP were noted only when associated with decreased driving pressures [29]. The association of higher driving pressures and higher mortality rate for mechanically ventilated patients with ARDS was also identified in a subsequent meta-analysis. Despite the strong association with mortality in retrospective studies [29,56], the routine use of driving pressure in ARDS has not demonstrated a mortality benefit in prospective randomized controlled trials [53,56]. One study using recruitment maneuvers followed by PEEP titration to the best CRS demonstrated an increase in mortality when compared to PEEP set by the low PEEP/FiO2 table [53]. This study suggests the combination of recruitment maneuvers and PEEP titration to best CRS should not be used together to set PEEP. Therefore, the driving pressure should be used as a complement—not a replacement—to the evidence-based VT, PPlat, and PEEP recommendations.
When ventilating an ED patient in a volume targeted mode, clinicians should re-evaluate the ventilator settings if the driving pressure is above 15 cm H20 [29]. The first option to decrease the driving pressure is to decrease the VT. Once a VT of ≤4–6 mL/kg PBW has been achieved, the next step is to adjust the PEEP and follow the change in driving pressure. At a fixed VT, the PEEP that leads to the smallest driving pressure is the ideal PEEP for improving respiratory system compliance (Fig. 2). This is accomplished by incrementally adjusting PEEP, to the range of the targeted PEEP from the PEEP/FiO2 table (Table 4), while monitoring the driving pressure. The goal of assessing driving pressure is to detect the individual heterogeneity of PEEP responsiveness seen in COVID-19 related ARDS. Driving pressure may aid in identifying those who may benefit, or be harmed, from a higher or lower PEEP than prescribed in the PEEP/FIO2 tables.
3.3.3. Transpulmonary pressure
Regional lung overdistention is a key factor in VILI, but it can be difficult to measure directly [24]. Overdistention occurs because of the high-pressure differences across the lung tissue, referred to as the transpulmonary pressure. The PPlat is the average alveolar pressure and often serves as a surrogate for inflation pressure and overdistention. In some instances, a high alveolar pressure (i.e. PPlat >30 cm H2O) may not reflect an injurious high transpulmonary pressure because of the counter-pressure (i.e. pleural pressure) provided by the chest wall and abdominal contents. As an example, a trumpet player with a very high alveolar pressure does not encounter elevated transpulmonary pressures because of the elevated pleural pressures generated to play the instrument [24,57]. Similarly, a stiff chest wall or rigid abdominal compartment may cause a high PPlat despite a safe, normal transpulmonary pressure [24]. One method of estimating pleural pressure is by using a balloon manometer to measure the esophageal pressures as a surrogate for pleural pressure. However, this complex strategy is not recommended in the emergency department as it is cumbersome and has not been shown to be beneficial when compared to empiric PEEP set by the PEEP/FiO2 tables (Table 4) [58,59].
3.4. Additional therapies in ARDS
3.4.1. Prone positioning
Prone positioning has been utilized for many years to improve oxygenation and outcomes in ARDS [60,61]. This position, commonly referred to as “proning,” utilizes gravitational effects to conform the shape of the lung to the chest cavity and ultimately reduce the pleural pressure gradient from non-dependent to dependent regions [62]. In addition to a more favorable and equitable distribution of aeration, proning increases end-expiratory lung volume, improves ventilation-perfusion matching, increases secretion clearance, and alters chest wall mechanics, leading to regional changes and improvements in overall lung ventilation [61,[63], [64], [65]]. Prone positioning has been shown in several studies to protect against VILI [62,[66], [67], [68], [69]] and also has a mortality benefit [60,70,71]. Proning should be considered in patients with ARDS and a PaO2/FiO2 ratio < 150 despite optimized ventilator settings [60]. This is consistent with the current COVID-19 recommendations where patients with moderate to severe ARDS may be proned for 12 to 16 h at a time [16]. Proning on mechanical ventilation is beneficial when started early, after 12–24 h of stabilization on the ventilator [60]. Unless there is a significant delay in the transfer, proning in the mechanically ventilated patient can generally wait for admission to the intensive care unit (ICU). If proning is started in the ED, neck and shoulder mobility should be assessed to ensure the patient can tolerate a prone position. Additionally, the team must wear appropriate PPE [15] and must be trained to monitor for pressure points and avoid accidental extubation, which can lead to loss of recruitment and potential exposure to the team. Due to the inherent risks and challenges, a collaborative approach with a practiced team is recommended prior to attempting prone positioning in the emergency department for the mechanically ventilated patient.
3.4.2. Inhaled pulmonary vasodilators
Inhaled pulmonary vasodilators predominantly distribute to ventilated alveoli causing localized vasodilation, thereby improving ventilation-perfusion matching [[72], [73], [74]]. While no mortality benefit has been demonstrated [[75], [76], [77]], inhaled nitric oxide and inhaled prostacyclins (e.g. epoprostenol) are used as rescue agents to reduce hypoxia-mediated vasoconstriction and improve oxygenation in severe ARDS [72,75,78,79]. The ease of delivery, inexpensive cost, and infrequent adverse events have made prostacyclins a more favorable choice [73], as inhaled nitric oxide [80] has been associated with increased renal impairment [81]. These decisions must be made within the local practice environment accounting for availability and ventilator circuit type. Also, there may be an increased risk of aerosolization with inhaled pulmonary vasodilators that should be taken into consideration. The Surviving Sepsis Campaign guidelines for COVID-19 recommend against the use of routine use of inhaled nitric oxide but suggest that a trial of inhaled pulmonary vasodilators may be used as a rescue therapy while monitoring for rapid improvement in oxygenation [16].
3.4.3. Neuromuscular blocking agents (NMBA)
Long-acting neuromuscular blocking agents (e.g. vecuronium and cisatracurium) used in moderate-to-severe ARDS have been shown to minimize patient-ventilator dyssynchrony, decrease work of breathing [82], improve oxygenation [83], reduce inflammatory biomarkers [84], and potentially increase the number of ventilator-free days and days outside the ICU [85]. The routine use of neuromuscular blockade in ARDS has been called into question after a 2019 multicenter randomized control trial evaluating the use of early paralytics and high PEEP in patients with moderate-to-severe ARDS found no difference in 90-day mortality when compared to usual therapy [86]. The evidence on neuromuscular use in COVID-19 induced ARDS is limited, and the long-term outcomes are unclear. In mechanically ventilated COVID-19 patients with moderate-to-severe ARDS, the Surviving Sepsis Campaign guidelines suggest using intermittent NMBA boluses instead of a continuous infusion to better facilitate lung protective ventilation [16]. The use of continuous NMBA infusions for up to 48 h should be reserved for patients with persistently high PPlat, poor oxygenation, and ventilator dyssynchrony [16].
3.4.4. Medications
The role of corticosteroids in the early and late stages of ARDS is controversial and widely debated. Two meta-analyses demonstrated reduced mortality, increased ventilator-free days, and accelerated resolution of disease when steroids were started several days after the onset of ARDS [87,88]. A more recent trial examining dexamethasone in patients with ARDS and a PaO2/FiO2 ratio of < 200 mmHg despite optimal ventilator settings suggested an improvement in outcomes [89]. However, a different meta-analysis did not support their use in either the acute or later fibroproliferative phases of the disease [90]. The current evidence of steroids use in COVID-19 induced ARDS is still emerging. A non-peer reviewed study of 26 patients with severe COVID-19 reported a decreased requirement for supplemental oxygenation and improvement on radiological chest imaging among patients who received corticosteroids [91]. There is a randomized control trial in Chongqing, China actively enrolling patients looking at the effect of glucocorticoids in COVID-19 patients with severe disease, which may provide further data [92]. Two more recent retrospective studies, albeit with small sample sizes and poor data controllability, reported that low-dose corticosteroid therapy may not delay viral clearance in COVID-19 patients [93,94]. Based on several Cochrane reviews on the use of steroids in viral pneumonia and a retrospective cohort study of patients with COVID-19 pneumonia [95], the Surviving Sepsis Campaign guidelines suggest the use of corticosteroids in critically ill patients with COVID-19 induced ARDS [16], while the NIH guidelines [15] make no recommendations based on limited evidence. Given the small improvements in mortality and faster resolution of septic shock observed from recent systematic reviews [96,97], they advise against the use of systemic corticosteroids of COVID-19 patients without ARDS unless the patient is in septic shock [16]. In patients with COVID-19, meeting criteria for ARDS, steroids should be considered in consultation with the admitting critical care team.
3.4.5. Venovenous extracorporeal membrane oxygenation
Venovenous extracorporeal membrane oxygenation (vvECMO) is a form of pulmonary bypass that uses an external membrane to allow for oxygen diffusion into the blood and the diffusion of carbon dioxide out of the blood. Over the last decade, several trials have shown increased use and potential benefits of vvECMO in severe ARDS when conventional ARDS management failed [98,99]. In COVID-19 patients, the Extracorporeal Life Support Organization (ELSO) guidelines state that indications for vvECMO should not differ from their usual recommendations or other existing guidelines [100,101]. The Surviving Sepsis Campaign guidelines for COVID-19 and ELSO recommend transfer to an ECMO experienced center for patients with severe ARDS and refractory hypoxemia despite maximal ARDS therapies [16].
3.5. An initial approach to COVID-19 hypoxemic respiratory failure
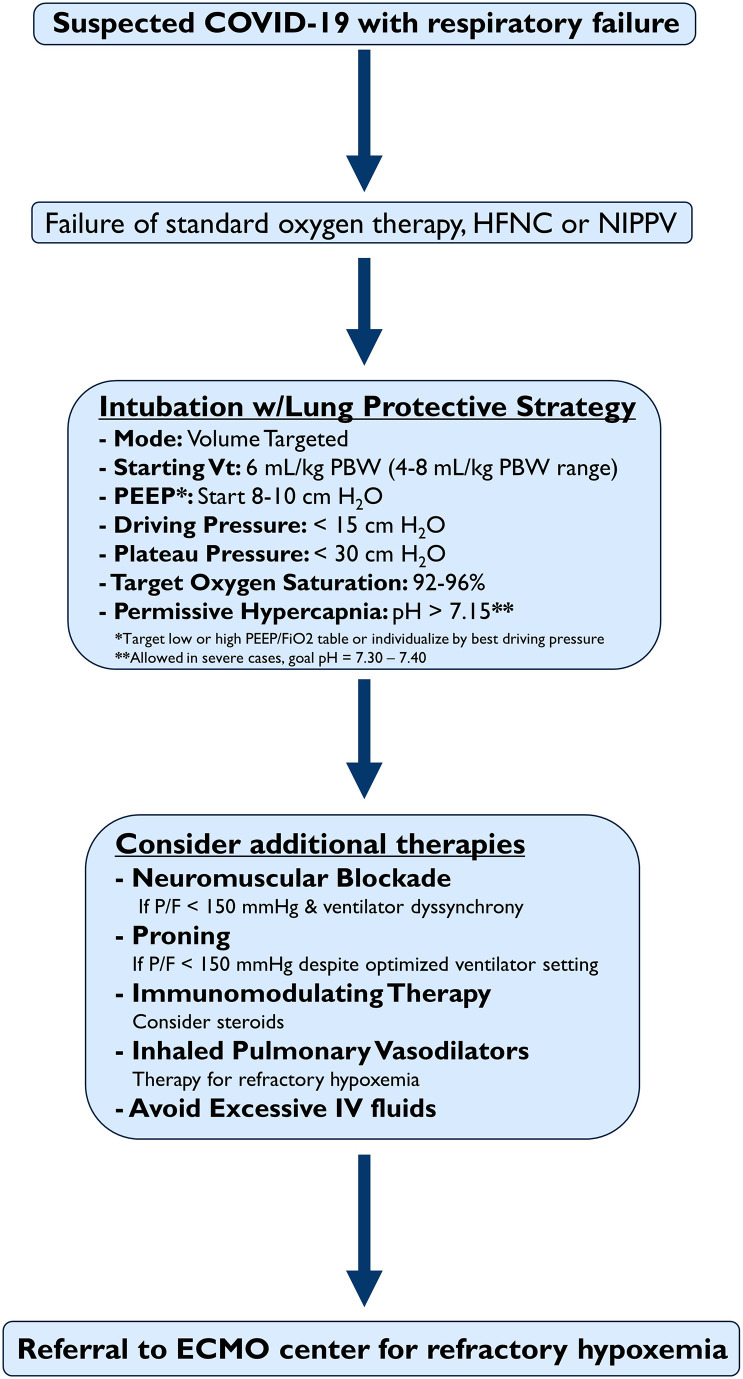
It is difficult to identify the optimal management of COVID-19 hypoxemic respiratory failure. Herein, we provide a rational approach based on the currently available evidence and lessons learned over the last few decades (Fig. 3 ). An initial approach to manage hypoxemia includes a trial of simple oxygen devices, high-flow nasal cannula (HFNC), or non-invasive positive pressure ventilation (NIPPV) if HFNC is not available [16]. A trial of HFNC or non-invasive positive pressure (NIPPV) is reasonable if intubation is not immediately indicated, if conventional oxygen devices fail to maintain an oxygen saturation of >90%, or if increased respiratory effort persists. HFNC has shown particular success in prior studies of hypoxemic respiratory failure and should be considered as a first-line treatment when simple oxygen devices fail to correct the hypoxemia [[14], [15], [16],102]. Non-invasive positive pressure (NIPPV) delivering PEEP in the form of continuous positive airway pressure (CPAP) may also have advantages, particularly with a helmet interface [103]. Bilevel positive pressure settings, delivering an additional inspiratory pressure, risk delivering injurious VT [102] and should be avoided unless otherwise indicated (e.g. an exacerbation of obstructive lung disease) [104]. The addition of awake, self-prone positioning or repositioning may improve oxygenation in patients with COVID-19 [105], but long-term effects of this practice are unclear. Personal protective equipment and airborne precautions must be utilized when using devices that can cause aerosolization [15]. The risks of aerosolization may not be increased with HFNC as compared to low flow oxygen devices [106]; if HFNC is used, a surgical mask to cover the device on the patient’s face is recommended.
Fig. 3.
A recommended initial approach to COVID-19 related hypoxemic respiratory failure in the Emergency Department. Abbreviations: HFNC (high flow nasal cannula), NIPPV (non-invasive positive pressure ventilation), PBW (predicted body weight), VT (tidal volume), P/F (PaO2/FiO2 ratio), IV (intravenous)
When HFNC or NIPPV is started, patients must be reassessed frequently—waiting until failure is associated with worse outcomes [102,107,108]. Patients on HFNC who remain tachypneic, have significant work of breathing, have rapidly escalating oxygen requirements, or remain hypoxic despite maximal flow (i.e. 60 L/min) and FiO2 should be intubated. The severity of hypoxemia, underlying illness, and clinical trajectory of these patients are important components of the decision to intubate. NIPPV failure rates for acute hypoxemic respiratory failure are higher in patients with pneumonia, sepsis, severe hypoxemia (PaO2/FiO2 < 150–200 mm Hg) [109,110], and persistently large tidal volumes (>9.5 mL/kg PBW) [111]. In those with persistently high respiratory effort, one must consider the possibility of self-inflicted lung injury. It is theorized that patients with a high respiratory effort are generating very high VT and high transpulmonary pressure, which could potentially lead to a self-inflicted lung injury [37,112]. Patients with persistently high respiratory efforts despite non-invasive support measures may benefit from early intubation, sedation, and control of VT and airway pressure.
Once invasive mechanical ventilation has started, LPV with low VT and appropriate PEEP should be started (Fig. 3 and Table 4). This strategy has been successfully used in a case series of COVID-19 patients [10] and is recommended by The Surviving Sepsis Campaign and NIH guidelines [[14], [15], [16]]. Ventilator modes vary between institution and device. A simple and effective strategy for the emergency department is to choose a volume targeted mode of ventilation (e.g. volume control). This allows for a safe, prescribed VT to be delivered. A pressure targeted mode (e.g. pressure control) is also an option but the delivered VT is less strict and will vary depending on the patient’s lung compliance, resistance, and the set inspiratory time. There is no outcome evidence to support the superiority of a pressure-controlled or volume-controlled mode of ventilation in ARDS [113]. Airway pressure release ventilation (APRV) has been used in ARDS [114] but the mode is complex, not available on every ventilator, and can lead to significant adverse events if used improperly [115]. For these reasons, APRV is not the initial preferred strategy in the ED, but this may vary by institution or local practice. Choosing a volume-controlled mode that allows a set VT is recommended. When the volume is set, the PPlat should be monitored by an end-inspiratory pause to ensure it remains < 30 cm H2O. Similarly, the driving pressure should be targeted < 15 cm H2O. If the PPlat or driving pressure are above these targets, decrease the VT by 1 mL/kg to a minimum of 4 mL/kg. The respiratory rate may need to be increased up to 35/min to maintain an appropriate minute ventilation. A pH target of 7.30–7.45 is recommended, but if necessary, permissive hypercapnia may be instituted, tolerating a pH > 7.15 [12].
Ideally, sedation should be kept as light as possible with a Richmond Agitation Sedation Scale of 0 (alert and calm) to −1 (drowsy but awakens to voice for >10 s); a lighter sedation strategy started in the ED is associated with improved outcomes [116]. Ventilator dyssynchrony is common [117] and places patients at risk for lung injury. A potential treatment for ventilator dyssynchrony is liberating the VT up to 8 mL/kg, if the PPlat and driving pressure are at safe levels, or adjusting the inspiratory flow pattern. If the PPlat or driving pressure is elevated or dyssynchrony persists, deep sedation with or without NMBAs may be needed.
Adjust the FiO2 to target an oxygen saturation of 92–96% [16]. The change from the original ARDS trial target of 88–95% [12] reflects a 2020 multicenter, randomized trial demonstrating a lack of benefit and potential of harm with a conservative (88–92%) vs. liberal (>96%) oxygenation strategy in ARDS [118]. The response to PEEP will be variable for all patients [38]. The PEEP/FiO2 tables (Table 4) provide guidance and have been used successfully in ARDS [12]. After intubation, start with a PEEP of 8–10 cm H20 and choose the range of PEEP by the high or low PEEP/FiO2 table (Table 4) [6]. Adjust the PEEP every 15 min by 2–3 cm H2O, while monitoring the PPlat, driving pressure, blood pressure, and pulse oximetry after each adjustment. A PPlat <30 cmH20 and driving pressure <15 cmH20 should be targeted. If pressures are above these levels, patients are at risk for VILI.
A conservative fluid resuscitation strategy should be used in patients with ARDS, as this may improve lung function [14,16,119]. Adjunctive therapies such as pulmonary vasodilators, corticosteroids, and proning should be considered on a case by case basis with input from the admitting critical care team. If the patient is unable to be oxygenated or ventilated despite optimized ventilator settings, consultation with an ECMO center should be considered.
4. Conclusion
In severe cases, COVID-19 leads to hypoxemic respiratory failure that may meet criteria for ARDS. While further COVID-19 specific studies are needed, the mainstay of treatment for COVID-19 related ARDS remains the early implementation, in the ED, of a lung protective ventilation strategy with low tidal volumes, adequate PEEP, and maintaining a plateau pressure of < 30 cm H2O. Adjunctive therapies such as corticosteroids, proning, NMBAs, pulmonary vasodilators, and ECMO in refractory cases should be considered on a case by case basis with input from the admitting critical care team.
Declaration of Competing Interest
None.
Acknowledgements
BL, TM, MR, MG, and DH conceived the idea for this manuscript and contributed substantially to the writing and editing of the review. This manuscript did not utilize any grants, and it has not been presented in abstract form. This clinical review has not been published, it is not under consideration for publication elsewhere, its publication is approved by all authors and tacitly or explicitly by the responsible authorities where the work was carried out, and that, if accepted, it will not be published elsewhere in the same form, in English or in any other language, including electronically without the written consent of the copyright-holder. This review does not reflect the views or opinions of the U.S. government, Department of Defense, U.S. Army, U.S. Air Force, or SAUSHEC EM Residency Program.
References
- 1.Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in china: summary of a report of 72 314 cases from the chinese center for disease control and prevention. JAMA. 2020 doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 2.Guan W.-J., Ni Z.-Y., Hu Y., Liang W.-H., Ou C.-Q., He J.-X. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Epidemiology Working Group for NCIP Epidemic Response, Chinese Center for Disease Control and Prevention The epidemiological characteristics of an outbreak of 2019 novel coronavirus diseases (COVID-19) in China. Zhonghua Liu Xing Bing Xue Za Zhi. 2020;41:145–151. doi: 10.3760/cma.j.issn.0254-6450.2020.02.003. [DOI] [PubMed] [Google Scholar]
- 4.Grasselli G., Zangrillo A., Zanella A., Antonelli M., Cabrini L., Castelli A. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy Region, Italy. JAMA. 2020;323:1574. doi: 10.1001/jama.2020.5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Richardson S., Hirsch J.S., Narasimhan M., Crawford J.M., McGinn T., Davidson K.W. Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City Area. JAMA. 2020 doi: 10.1001/jama.2020.6775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Fuller B.M., Ferguson I.T., Mohr N.M., Drewry A.M., Palmer C., Wessman B.T. Lung-protective ventilation initiated in the emergency department (LOV-ED): a quasi-experimental, before-after trial. Ann Emerg Med. 2017;70:406–418. doi: 10.1016/j.annemergmed.2017.01.013. [e4] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bhatraju P.K., Ghassemieh B.J., Nichols M., Kim R., Jerome K.R., Nalla A.K. Covid-19 in critically ill patients in the Seattle Region — case series. N Engl J Med. 2020 doi: 10.1056/NEJMoa2004500. [NEJMoa2004500] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Arentz M., Yim E., Klaff L., Lokhandwala S., Riedo F.X., Chong M. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State. JAMA. 2020;323:1612. doi: 10.1001/jama.2020.4326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Xiong Y., Liu Y., Cao L., Wang D., Guo M., Jiang A. Transcriptomic characteristics of bronchoalveolar lavage fluid and peripheral blood mononuclear cells in COVID-19 patients. Emerg Microbes Infect. 2020;9:761–770. doi: 10.1080/22221751.2020.1747363. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ziehr D.R., Alladina J., Petri C.R., Maley J.H., Moskowitz A., Medoff B.D. Respiratory pathophysiology of mechanically ventilated patients with COVID-19: a cohort study. Am J Respir Crit Care Med. 2020 doi: 10.1164/rccm.202004-1163LE. [rccm.202004-1163LE] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.ARDS Definition Task Force, Ranieri V.M., Rubenfeld G.D., Thompson B.T., Ferguson N.D., Caldwell E. Acute respiratory distress syndrome: the Berlin Definition. JAMA. 2012;307:2526–2533. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 12.Acute Respiratory Distress Syndrome Network, Brower R.G., Matthay M.A., Morris A., Schoenfeld D., Thompson B.T. Ventilation with lower tidal volumes as compared with traditional tidal volumes for acute lung injury and the acute respiratory distress syndrome. N Engl J Med. 2000;342:1301–1308. doi: 10.1056/NEJM200005043421801. [DOI] [PubMed] [Google Scholar]
- 13.Hager D.N., Krishnan J.A., Hayden D.L., Brower R.G. ARDS Clinical Trials Network. Tidal volume reduction in patients with acute lung injury when plateau pressures are not high. Am J Respir Crit Care Med. 2005;172:1241–1245. doi: 10.1164/rccm.200501-048CP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Poston J.T., Patel B.K., Davis A.M. Management of critically ill adults with COVID-19. JAMA. 2020 doi: 10.1001/jama.2020.4914. [DOI] [PubMed] [Google Scholar]
- 15.COVID-19 Treatment Guidelines Panel . National Institutes of Health; 2020. Coronavirus Disease 2019 (COVID-19) Treatment Guidelines.https://www.covid19treatmentguidelines.nih.gov/ Available at. (Accessed June 15, 2020) [PubMed] [Google Scholar]
- 16.Alhazzani W., Møller M.H., Arabi Y.M., Loeb M., Gong M.N., Fan E. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19) Intensive Care Med. 2020 doi: 10.1007/s00134-020-06022-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thompson B.T., Chambers R.C., Liu K.D. Acute respiratory distress syndrome. N Engl J Med. 2017;377:562–572. doi: 10.1056/NEJMra1608077. [DOI] [PubMed] [Google Scholar]
- 18.Zhou F., Yu T., Du R., Fan G., Liu Y., Liu Z. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet. 2020;395:1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fox S.E., Akmatbekov A., Harbert J.L., Li G., Brown J.Q., Vander Heide R.S. Pulmonary and cardiac pathology in Covid-19: the first autopsy series from New Orleans. Pathology. 2020 doi: 10.1101/2020.04.06.20050575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Luks A.M., Swenson E.R. COVID-19 lung injury and high altitude pulmonary edema: a false equation with dangerous implications. Ann ATS. 2020 doi: 10.1513/AnnalsATS.202004-327FR. [AnnalsATS.202004-327FR] [DOI] [PubMed] [Google Scholar]
- 21.Barton L.M., Duval E.J., Stroberg E., Ghosh S., Mukhopadhyay S. COVID-19 autopsies, Oklahoma, USA. Am J Clin Pathol. 2020 doi: 10.1093/ajcp/aqaa062. [aqaa062] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Gattinoni L., Coppola S., Cressoni M., Busana M., Rossi S., Chiumello D. Covid-19 does not lead to a “typical” acute respiratory distress syndrome. Am J Respir Crit Care Med. 2020 doi: 10.1164/rccm.202003-0817LE. [rccm.202003-0817LE] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Rice T.W., Janz D.R. In defense of evidence-based medicine for the treatment of COVID-19 ARDS. Ann ATS. 2020 doi: 10.1513/AnnalsATS.202004-325IP. [AnnalsATS.202004-325IP] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Slutsky A.S., Ranieri V.M. Ventilator-induced lung injury. N Engl J Med. 2013;369:2126–2136. doi: 10.1056/NEJMra1208707. [DOI] [PubMed] [Google Scholar]
- 25.Muscedere J.G., Mullen J.B., Gan K., Slutsky A.S. Tidal ventilation at low airway pressures can augment lung injury. Am J Respir Crit Care Med. 1994;149:1327–1334. doi: 10.1164/ajrccm.149.5.8173774. [DOI] [PubMed] [Google Scholar]
- 26.Tremblay L., Valenza F., Ribeiro S.P., Li J., Slutsky A.S. Injurious ventilatory strategies increase cytokines and c-fos m-RNA expression in an isolated rat lung model. J Clin Invest. 1997;99:944–952. doi: 10.1172/JCI119259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chu E.K., Whitehead T., Slutsky A.S. Effects of cyclic opening and closing at low- and high-volume ventilation on bronchoalveolar lavage cytokines. Crit Care Med. 2004;32:168–174. doi: 10.1097/01.CCM.0000104203.20830.AE. [DOI] [PubMed] [Google Scholar]
- 28.Hess D.R. Respiratory mechanics in mechanically ventilated patients. Respir Care. 2014;59:1773–1794. doi: 10.4187/respcare.03410. [DOI] [PubMed] [Google Scholar]
- 29.Amato M.B.P., Meade M.O., Slutsky A.S., Brochard L., Costa E.L.V., Schoenfeld D.A. Driving pressure and survival in the acute respiratory distress syndrome. N Engl J Med. 2015;372:747–755. doi: 10.1056/NEJMsa1410639. [DOI] [PubMed] [Google Scholar]
- 30.Rhodes A., Evans L.E., Alhazzani W., Levy M.M., Antonelli M., Ferrer R. Surviving sepsis campaign: international guidelines for management of sepsis and septic shock. Crit Care Med. 2017;45:486–552. doi: 10.1097/CCM.0000000000002255. [DOI] [PubMed] [Google Scholar]
- 31.Cannon J.W., Gutsche J.T., Brodie D. Optimal strategies for severe acute respiratory distress syndrome. Crit Care Clin. 2017;33:259–275. doi: 10.1016/j.ccc.2016.12.010. [DOI] [PubMed] [Google Scholar]
- 32.Griffiths M.J.D., McAuley D.F., Perkins G.D., Barrett N., Blackwood B., Boyle A. Guidelines on the management of acute respiratory distress syndrome. BMJ Open Respir Res. 2019;6 doi: 10.1136/bmjresp-2019-000420. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Howell M.D., Davis A.M. Management of ARDS in adults. JAMA. 2018;319:711. doi: 10.1001/jama.2018.0307. [DOI] [PubMed] [Google Scholar]
- 34.Fan E., Del Sorbo L., Goligher E.C., Hodgson C.L., Munshi L., Walkey A.J. An Official American Thoracic Society/European Society of Intensive Care Medicine/Society of Critical Care Medicine Clinical Practice Guideline: mechanical ventilation in adult patients with acute respiratory distress syndrome. Am J Respir Crit Care Med. 2017;195:1253–1263. doi: 10.1164/rccm.201703-0548ST. [DOI] [PubMed] [Google Scholar]
- 35.Kaku S., Nguyen C.D., Htet N.N., Tutera D., Barr J., Paintal H.S. Acute respiratory distress syndrome: etiology, pathogenesis, and summary on management. J Intensive Care Med. 2019 doi: 10.1177/0885066619855021. [088506661985502] [DOI] [PubMed] [Google Scholar]
- 36.Briel M., Meade M., Mercat A., Brower R.G., Talmor D., Walter S.D. Higher vs lower positive end-expiratory pressure in patients with acute lung injury and acute respiratory distress syndrome: systematic review and meta-analysis. JAMA. 2010;303:865–873. doi: 10.1001/jama.2010.218. [DOI] [PubMed] [Google Scholar]
- 37.Marini J.J., Gattinoni L. Management of COVID-19 respiratory distress. JAMA. 2020 doi: 10.1001/jama.2020.6825. [DOI] [PubMed] [Google Scholar]
- 38.Gattinoni L., Caironi P., Cressoni M., Chiumello D., Ranieri V.M., Quintel M. Lung recruitment in patients with the acute respiratory distress syndrome. N Engl J Med. 2006;354:1775–1786. doi: 10.1056/NEJMoa052052. [DOI] [PubMed] [Google Scholar]
- 39.Sahetya S.K., Goligher E.C., Brower R.G. Fifty years of research in ARDS. Setting positive end-expiratory pressure in acute respiratory distress syndrome. Am J Respir Crit Care Med. 2017;195:1429–1438. doi: 10.1164/rccm.201610-2035CI. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Del Sorbo L., Tonetti T., Ranieri V.M. Alveolar recruitment in acute respiratory distress syndrome: should we open the lung (no matter what) or may accept (part of) the lung closed? Intensive Care Med. 2019;45:1436–1439. doi: 10.1007/s00134-019-05734-7. [DOI] [PubMed] [Google Scholar]
- 41.Suter P.M., Fairley B., Isenberg M.D. Optimum end-expiratory airway pressure in patients with acute pulmonary failure. N Engl J Med. 1975;292:284–289. doi: 10.1056/NEJM197502062920604. [DOI] [PubMed] [Google Scholar]
- 42.Santa Cruz R., Rojas J.I., Nervi R., Heredia R., Ciapponi A. High versus low positive end-expiratory pressure (PEEP) levels for mechanically ventilated adult patients with acute lung injury and acute respiratory distress syndrome. Cochrane Database Syst Rev. 2013 doi: 10.1002/14651858.CD009098.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Brower R.G., Lanken P.N., MacIntyre N., Matthay M.A., Morris A., Ancukiewicz M. Higher versus lower positive end-expiratory pressures in patients with the acute respiratory distress syndrome. N Engl J Med. 2004;351:327–336. doi: 10.1056/NEJMoa032193. [DOI] [PubMed] [Google Scholar]
- 44.Meade M.O., Cook D.J., Guyatt G.H., Slutsky A.S., Arabi Y.M., Cooper D.J. Ventilation strategy using low tidal volumes, recruitment maneuvers, and high positive end-expiratory pressure for acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2008;299:637–645. doi: 10.1001/jama.299.6.637. [DOI] [PubMed] [Google Scholar]
- 45.Mercat A., J-CM Richard, Vielle B., Jaber S., Osman D., Diehl J.-L. Positive end-expiratory pressure setting in adults with acute lung injury and acute respiratory distress syndrome: a randomized controlled trial. JAMA. 2008;299:646–655. doi: 10.1001/jama.299.6.646. [DOI] [PubMed] [Google Scholar]
- 46.Walkey A.J., Del Sorbo L., Hodgson C.L., Adhikari N.K.J., Wunsch H., Meade M.O. Higher PEEP versus lower PEEP strategies for patients with acute respiratory distress syndrome. A systematic review and meta-analysis. Ann Am Thorac Soc. 2017;14:S297–S303. doi: 10.1513/AnnalsATS.201704-338OT. [DOI] [PubMed] [Google Scholar]
- 47.Chen L., Del Sorbo L., Grieco D.L., Junhasavasdikul D., Rittayamai N., Soliman I. Potential for lung recruitment estimated by the recruitment-to-inflation ratio in acute respiratory distress syndrome. A clinical trial. Am J Respir Crit Care Med. 2020;201:178–187. doi: 10.1164/rccm.201902-0334OC. [DOI] [PubMed] [Google Scholar]
- 48.Gattinoni L., Chiumello D., Rossi S. COVID-19 pneumonia: ARDS or not? Crit Care. 2020;24:154. doi: 10.1186/s13054-020-02880-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Gattinoni L., Chiumello D., Caironi P., Busana M., Romitti F., Brazzi L. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med. 2020 doi: 10.1007/s00134-020-06033-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Calfee C.S., Delucchi K., Parsons P.E., Thompson B.T., Ware L.B., Matthay M.A. Subphenotypes in acute respiratory distress syndrome: latent class analysis of data from two randomised controlled trials. Lancet Respir Med. 2014;2:611–620. doi: 10.1016/S2213-2600(14)70097-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Constantin J.-M., Jabaudon M., Lefrant J.-Y., Jaber S., Quenot J.-P., Langeron O. Personalised mechanical ventilation tailored to lung morphology versus low positive end-expiratory pressure for patients with acute respiratory distress syndrome in France (the LIVE study): a multicentre, single-blind, randomised controlled trial. Lancet Respir Med. 2019;7:870–880. doi: 10.1016/S2213-2600(19)30138-9. [DOI] [PubMed] [Google Scholar]
- 52.Goligher E.C., Hodgson C.L., Adhikari N.K.J., Meade M.O., Wunsch H., Uleryk E. Lung recruitment maneuvers for adult patients with acute respiratory distress syndrome. A systematic review and meta-analysis. Ann Am Thorac Soc. 2017;14:S304–S311. doi: 10.1513/AnnalsATS.201704-340OT. [DOI] [PubMed] [Google Scholar]
- 53.Writing Group for the Alveolar Recruitment for Acute Respiratory Distress Syndrome Trial (ART) Investigators, Cavalcanti A.B., Suzumura É.A., Laranjeira L.N., de Paisani D.M., Damiani L.P. Effect of lung recruitment and titrated positive end-expiratory pressure (PEEP) vs low PEEP on mortality in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2017;318:1335–1345. doi: 10.1001/jama.2017.14171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Aoyama H., Yamada Y., Fan E. The future of driving pressure: a primary goal for mechanical ventilation? J Intensive Care. 2018;6(64) doi: 10.1186/s40560-018-0334-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Spinelli E., Grieco D.L., Mauri T. A personalized approach to the acute respiratory distress syndrome: recent advances and future challenges. J Thorac Dis. 2019;11:5619–5625. doi: 10.21037/jtd.2019.11.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Aoyama H., Pettenuzzo T., Aoyama K., Pinto R., Englesakis M., Fan E. Association of driving pressure with mortality among ventilated patients with acute respiratory distress syndrome: a systematic review and meta-analysis. Crit Care Med. 2018;46:300–306. doi: 10.1097/CCM.0000000000002838. [DOI] [PubMed] [Google Scholar]
- 57.Bouhuys A. Physiology and musical instruments. Nature. 1969;221:1199–1204. doi: 10.1038/2211199a0. [DOI] [PubMed] [Google Scholar]
- 58.Beitler J.R., Sarge T., Banner-Goodspeed V.M., Gong M.N., Cook D., Novack V. Effect of titrating positive end-expiratory pressure (PEEP) with an esophageal pressure-guided strategy vs an empirical high PEEP-Fio2 strategy on death and days free from mechanical ventilation among patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2019;321:846–857. doi: 10.1001/jama.2019.0555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Talmor D., Sarge T., Malhotra A., O’Donnell C.R., Ritz R., Lisbon A. Mechanical ventilation guided by esophageal pressure in acute lung injury. N Engl J Med. 2008;359:2095–2104. doi: 10.1056/NEJMoa0708638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Guérin C., Reignier J., Richard J.-C., Beuret P., Gacouin A., Boulain T. Prone positioning in severe acute respiratory distress syndrome. N Engl J Med. 2013;368:2159–2168. doi: 10.1056/NEJMoa1214103. [DOI] [PubMed] [Google Scholar]
- 61.Gattinoni L., Tognoni G., Pesenti A., Taccone P., Mascheroni D., Labarta V. Effect of prone positioning on the survival of patients with acute respiratory failure. N Engl J Med. 2001;345:568–573. doi: 10.1056/NEJMoa010043. [DOI] [PubMed] [Google Scholar]
- 62.Scholten E.L., Beitler J.R., Prisk G.K., Malhotra A. Treatment of ARDS with prone positioning. Chest. 2017;151:215–224. doi: 10.1016/j.chest.2016.06.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Douglas W.W., Rehder K., Beynen F.M., Sessler A.D., Marsh H.M. Improved oxygenation in patients with acute respiratory failure: the prone position. Am Rev Respir Dis. 1977;115:559–566. doi: 10.1164/arrd.1977.115.4.559. [DOI] [PubMed] [Google Scholar]
- 64.Pappert D., Rossaint R., Slama K., Grüning T., Falke K.J. Influence of positioning on ventilation-perfusion relationships in severe adult respiratory distress syndrome. Chest. 1994;106:1511–1516. doi: 10.1378/chest.106.5.1511. [DOI] [PubMed] [Google Scholar]
- 65.Albert R.K., Leasa D., Sanderson M., Robertson H.T., Hlastala M.P. The prone position improves arterial oxygenation and reduces shunt in oleic-acid-induced acute lung injury. Am Rev Respir Dis. 1987;135:628–633. doi: 10.1164/arrd.1987.135.3.628. [DOI] [PubMed] [Google Scholar]
- 66.Broccard A., Shapiro R.S., Schmitz L.L., Adams A.B., Nahum A., Marini J.J. Prone positioning attenuates and redistributes ventilator-induced lung injury in dogs. Crit Care Med. 2000;28:295–303. doi: 10.1097/00003246-200002000-00001. [DOI] [PubMed] [Google Scholar]
- 67.Mentzelopoulos S.D. Prone position reduces lung stress and strain in severe acute respiratory distress syndrome. Eur Respir J. 2005;25:534–544. doi: 10.1183/09031936.05.00105804. [DOI] [PubMed] [Google Scholar]
- 68.Galiatsou E., Kostanti E., Svarna E., Kitsakos A., Koulouras V., Efremidis S.C. Prone position augments recruitment and prevents alveolar overinflation in acute lung injury. Am J Respir Crit Care Med. 2006;174:187–197. doi: 10.1164/rccm.200506-899OC. [DOI] [PubMed] [Google Scholar]
- 69.Papazian L., Gainnier M., Marin V., Donati S., Arnal J.-M., Demory D. Comparison of prone positioning and high-frequency oscillatory ventilation in patients with acute respiratory distress syndrome*. Crit Care Med. 2005;33:2162–2171. doi: 10.1097/01.CCM.0000181298.05474.2B. [DOI] [PubMed] [Google Scholar]
- 70.Sud S., Friedrich J.O., Taccone P., Polli F., Adhikari N.K.J., Latini R. Prone ventilation reduces mortality in patients with acute respiratory failure and severe hypoxemia: systematic review and meta-analysis. Intensive Care Med. 2010;36:585–599. doi: 10.1007/s00134-009-1748-1. [DOI] [PubMed] [Google Scholar]
- 71.Gattinoni L., Carlesso E., Taccone P., Polli F., Guérin C., Mancebo J. Prone positioning improves survival in severe ARDS: a pathophysiologic review and individual patient meta-analysis. Miner Anest. 2010;76:448–454. [PubMed] [Google Scholar]
- 72.Gebistorf F., Karam O., Wetterslev J., Afshari A. Inhaled nitric oxide for acute respiratory distress syndrome (ARDS) in children and adults. Cochrane Database Syst Rev. 2016 doi: 10.1002/14651858.CD002787.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Cherian S.V., Kumar A., Akasapu K., Ashton R.W., Aparnath M., Malhotra A. Salvage therapies for refractory hypoxemia in ARDS. Respir Med. 2018;141:150–158. doi: 10.1016/j.rmed.2018.06.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Kallet R.H.A. Comprehensive review of prone position in ARDS. Respir Care. 2015;60:1660–1687. doi: 10.4187/respcare.04271. [DOI] [PubMed] [Google Scholar]
- 75.Fuller B.M., Mohr N.M., Skrupky L., Fowler S., Kollef M.H., Carpenter C.R. The use of inhaled prostaglandins in patients with ARDS. Chest. 2015;147:1510–1522. doi: 10.1378/chest.14-3161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Dellinger R.P., Zimmerman J.L., Taylor R.W., Straube R.C., Hauser D.L., Criner G.J. Effects of inhaled nitric oxide in patients with acute respiratory distress syndrome: results of a randomized phase II trial. Crit Care Med. 1998;26:15–23. doi: 10.1097/00003246-199801000-00011. [DOI] [PubMed] [Google Scholar]
- 77.Adhikari N.K.J., Dellinger R.P., Lundin S., Payen D., Vallet B., Gerlach H. Inhaled nitric oxide does not reduce mortality in patients with acute respiratory distress syndrome regardless of severity: systematic review and meta-analysis*. Crit Care Med. 2014;42:404–412. doi: 10.1097/CCM.0b013e3182a27909. [DOI] [PubMed] [Google Scholar]
- 78.Pipeling M.R., Fan E. Therapies for refractory hypoxemia in acute respiratory distress syndrome. JAMA. 2010;304:2521. doi: 10.1001/jama.2010.1752. [DOI] [PubMed] [Google Scholar]
- 79.Kallet R.H. Adjunct therapies during mechanical ventilation: airway clearance techniques, therapeutic aerosols, and gases. Respir Care. 2013;58:1053–1073. doi: 10.4187/respcare.02217. [DOI] [PubMed] [Google Scholar]
- 80.Torbic H., Szumita P.M., Anger K.E., Nuccio P., LaGambina S., Weinhouse G. Inhaled epoprostenol vs inhaled nitric oxide for refractory hypoxemia in critically ill patients. J Crit Care. 2013;28:844–848. doi: 10.1016/j.jcrc.2013.03.006. [DOI] [PubMed] [Google Scholar]
- 81.Afshari A., Brok J., Møller A.M., Wetterslev J. The Cochrane Collaboration, Cochrane Database of Systematic Reviews. John Wiley & Sons, Ltd; Chichester, UK: 2010. Inhaled nitric oxide for acute respiratory distress syndrome (ARDS) and acute lung injury in children and adults. [p. CD002787.pub2] [DOI] [PubMed] [Google Scholar]
- 82.Alhazzani W., Alshahrani M., Jaeschke R., Forel J., Papazian L., Sevransky J. Neuromuscular blocking agents in acute respiratory distress syndrome: a systematic review and meta-analysis of randomized controlled trials. Crit Care. 2013;17 doi: 10.1186/cc12557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Gainnier M., Roch A., Forel J.-M., Thirion X., Arnal J.-M., Donati S. Effect of neuromuscular blocking agents on gas exchange in patients presenting with acute respiratory distress syndrome*. Crit Care Med. 2004;32:113–119. doi: 10.1097/01.CCM.0000104114.72614.BC. [DOI] [PubMed] [Google Scholar]
- 84.Forel J.-M., Roch A., Marin V., Michelet P., Demory D., Blache J.-L. Neuromuscular blocking agents decrease inflammatory response in patients presenting with acute respiratory distress syndrome*. Crit. Care Med. 2006;34:2749–2757. doi: 10.1097/01.CCM.0000239435.87433.0D. [DOI] [PubMed] [Google Scholar]
- 85.Papazian L., Forel J.-M., Gacouin A., Penot-Ragon C., Perrin G., Loundou A. Neuromuscular blockers in early acute respiratory distress syndrome. N Engl J Med. 2010;363:1107–1116. doi: 10.1056/NEJMoa1005372. [DOI] [PubMed] [Google Scholar]
- 86.The National Heart, Lung, and Blood Institute PETAL Clinical Trials Network Early neuromuscular blockade in the acute respiratory distress syndrome. N Engl J Med. 2019;380:1997–2008. doi: 10.1056/NEJMoa1901686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Meduri G.U., Bridges L., Shih M.-C., Marik P.E., Siemieniuk R.A.C., Kocak M. Prolonged glucocorticoid treatment is associated with improved ARDS outcomes: analysis of individual patients’ data from four randomized trials and trial-level meta-analysis of the updated literature. Intensive Care Med. 2016;42:829–840. doi: 10.1007/s00134-015-4095-4. [DOI] [PubMed] [Google Scholar]
- 88.Peter J.V., John P., Graham P.L., Moran J.L., George I.A., Bersten A. Corticosteroids in the prevention and treatment of acute respiratory distress syndrome (ARDS) in adults: meta-analysis. BMJ. 2008;336:1006–1009. doi: 10.1136/bmj.39537.939039.BE. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Villar J., Ferrando C., Martínez D., Ambrós A., Muñoz T., Soler J.A. Dexamethasone treatment for the acute respiratory distress syndrome: a multicentre, randomised controlled trial. Lancet Respir Med. 2020;8:267–276. doi: 10.1016/S2213-2600(19)30417-5. [DOI] [PubMed] [Google Scholar]
- 90.Agarwal R., Nath A., Aggarwal A.N., Gupta D. Do glucocorticoids decrease mortality in acute respiratory distress syndrome? A meta-analysis. Respirology. 2007;12:585–590. doi: 10.1111/j.1440-1843.2007.01060.x. [DOI] [PubMed] [Google Scholar]
- 91.Wang Y., Jiang W., He Q., Wang C., Wang B., Zhou P. Early, low-dose and short-term application of corticosteroid treatment in patients with severe COVID-19 pneumonia: single-center experience from Wuhan, China. Infect Dis. 2020 doi: 10.1101/2020.03.06.20032342. [DOI] [Google Scholar]
- 92.Qin Y.-Y., Zhou Y.-H., Lu Y.-Q., Sun F., Yang S., Harypursat V. Effectiveness of glucocorticoid therapy in patients with severe coronavirus disease 2019: protocol of a randomized controlled trial. Chin Med J. 2020;133:1080–1086. doi: 10.1097/CM9.0000000000000791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Fang X., Mei Q., Yang T., Li L., Wang Y., Tong F. Low-dose corticosteroid therapy does not delay viral clearance in patients with COVID-19. J Infect. 2020 doi: 10.1016/j.jinf.2020.03.039. [S0163445320301687] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Qin N., Cheng D., Yongtao L., Hong Z., Jun L., Xuan Z. Retrospective study of low-to-moderate dose glucocorticoids on viral clearance in patients with novel coronavirus pneumonia. Chin J Clin Infect Dis. 2020;13 doi: 10.3760/cma.j.cn115673-20200225-00072. [E009–E009] [DOI] [Google Scholar]
- 95.Wu C., Chen X., Cai Y., Xia J., Zhou X., Xu S. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020 doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Rygård S.L., Butler E., Granholm A., Møller M.H., Cohen J., Finfer S. Low-dose corticosteroids for adult patients with septic shock: a systematic review with meta-analysis and trial sequential analysis. Intensive Care Med. 2018;44:1003–1016. doi: 10.1007/s00134-018-5197-6. [DOI] [PubMed] [Google Scholar]
- 97.Rochwerg B., Oczkowski S.J., Siemieniuk R.A.C., Agoritsas T., Belley-Cote E., D’Aragon F. Corticosteroids in sepsis: an updated systematic review and meta-analysis. Crit Care Med. 2018;46:1411–1420. doi: 10.1097/CCM.0000000000003262. [DOI] [PubMed] [Google Scholar]
- 98.Combes A., Hajage D., Capellier G., Demoule A., Lavoué S., Guervilly C. Extracorporeal membrane oxygenation for severe acute respiratory distress syndrome. N Engl J Med. 2018;378:1965–1975. doi: 10.1056/NEJMoa1800385. [DOI] [PubMed] [Google Scholar]
- 99.Peek G.J., Mugford M., Tiruvoipati R., Wilson A., Allen E., Thalanany M.M. Efficacy and economic assessment of conventional ventilatory support versus extracorporeal membrane oxygenation for severe adult respiratory failure (CESAR): a multicentre randomised controlled trial. Lancet. 2009;374:1351–1363. doi: 10.1016/S0140-6736(09)61069-2. [DOI] [PubMed] [Google Scholar]
- 100.Combes A., Brodie D., Bartlett R., Brochard L., Brower R., Conrad S. Position paper for the organization of extracorporeal membrane oxygenation programs for acute respiratory failure in adult patients. Am J Respir Crit Care Med. 2014;190:488–496. doi: 10.1164/rccm.201404-0630CP. [DOI] [PubMed] [Google Scholar]
- 101.Bartlett R.H., Ogino M.T., Brodie D., McMullan D.M., Lorusso R., MacLaren G. Initial ELSO guidance document: ECMO for COVID-19 patients with severe cardiopulmonary failure. ASAIO J. 2020;66:472–474. doi: 10.1097/MAT.0000000000001173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Frat J.-P., Thille A.W., Mercat A., Girault C., Ragot S., Perbet S. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med. 2015;372:2185–2196. doi: 10.1056/NEJMoa1503326. [DOI] [PubMed] [Google Scholar]
- 103.Patel B.K., Wolfe K.S., Pohlman A.S., Hall J.B., Kress J.P. Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2016;315:2435–2441. doi: 10.1001/jama.2016.6338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Osadnik C.R., Tee V.S., Carson-Chahhoud K.V., Picot J., Wedzicha J.A., Smith B.J. Non-invasive ventilation for the management of acute hypercapnic respiratory failure due to exacerbation of chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2017;7 doi: 10.1002/14651858.CD004104.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Caputo N.D., Strayer R.J., Levitan R. Early self-proning in awake, non-intubated patients in the emergency department: a single ED’s experience during the COVID-19 pandemic. Acad Emerg Med. 2020;27:375–378. doi: 10.1111/acem.13994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Li J., Fink J.B., Ehrmann S. High-flow nasal cannula for COVID-19 patients: low risk of bio-aerosol dispersion. Eur Respir J. 2020 doi: 10.1183/13993003.00892-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Carrillo A., Gonzalez-Diaz G., Ferrer M., Martinez-Quintana M.E., Lopez-Martinez A., Llamas N. Non-invasive ventilation in community-acquired pneumonia and severe acute respiratory failure. Intensive Care Med. 2012;38:458–466. doi: 10.1007/s00134-012-2475-6. [DOI] [PubMed] [Google Scholar]
- 108.Kang B.J., Koh Y., Lim C.-M., Huh J.W., Baek S., Han M. Failure of high-flow nasal cannula therapy may delay intubation and increase mortality. Intensive Care Med. 2015;41:623–632. doi: 10.1007/s00134-015-3693-5. [DOI] [PubMed] [Google Scholar]
- 109.Ferreyro B.L., Angriman F., Munshi L., Sorbo L.D., Ferguson N.D., Rochwerg B. Association of noninvasive oxygenation strategies with all-cause mortality in adults with acute hypoxemic respiratory failure: a systematic review and meta-analysis. JAMA. 2020 doi: 10.1001/jama.2020.9524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Taha A., Larumbe-Zabala E., Abugroun A., Mohammedzein A., Naguib M.T., Patel M. Outcomes of noninvasive positive pressure ventilation in acute respiratory distress syndrome and their predictors: a national cohort. Crit Care Res Pract. 2019;2019:8106145. doi: 10.1155/2019/8106145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Carteaux G., Millán-Guilarte T., De Prost N., Razazi K., Abid S., Thille A.W. Failure of noninvasive ventilation for de novo acute hypoxemic respiratory failure: role of tidal volume*. Crit Care Med. 2016;44:282–290. doi: 10.1097/CCM.0000000000001379. [DOI] [PubMed] [Google Scholar]
- 112.Brochard L., Slutsky A., Pesenti A. Mechanical ventilation to minimize progression of lung injury in acute respiratory failure. Am J Respir Crit Care Med. 2016;195:438–442. doi: 10.1164/rccm.201605-1081CP. [DOI] [PubMed] [Google Scholar]
- 113.Chacko B., Peter J.V., Tharyan P., John G., Jeyaseelan L. Pressure-controlled versus volume-controlled ventilation for acute respiratory failure due to acute lung injury (ALI) or acute respiratory distress syndrome (ARDS) Cochrane Database Syst Rev. 2015;1 doi: 10.1002/14651858.CD008807.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Zhou Y., Jin X., Lv Y., Wang P., Yang Y., Liang G. Early application of airway pressure release ventilation may reduce the duration of mechanical ventilation in acute respiratory distress syndrome. Intensive Care Med. 2017;43:1648–1659. doi: 10.1007/s00134-017-4912-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Mireles-Cabodevila E., Kacmarek R.M. Should airway pressure release ventilation be the primary mode in ARDS? Respir Care. 2016;61:761–773. doi: 10.4187/respcare.04653. [DOI] [PubMed] [Google Scholar]
- 116.Fuller B.M., Roberts B.W., Mohr N.M., Knight W.A., Adeoye O., Pappal R.D. The ED-SED Study: a multicenter, prospective cohort study of practice patterns and clinical outcomes associated with emergency department SEDation for mechanically ventilated patients. Crit Care Med. 2019;47:1539–1548. doi: 10.1097/CCM.0000000000003928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Thille A.W., Rodriguez P., Cabello B., Lellouche F., Brochard L. Patient-ventilator asynchrony during assisted mechanical ventilation. Intensive Care Med. 2006;32:1515–1522. doi: 10.1007/s00134-006-0301-8. [DOI] [PubMed] [Google Scholar]
- 118.Barrot L., Asfar P., Mauny F., Winiszewski H., Montini F., Badie J. Liberal or conservative oxygen therapy for acute respiratory distress syndrome. N Engl J Med. 2020;382:999–1008. doi: 10.1056/NEJMoa1916431. [DOI] [PubMed] [Google Scholar]
- 119.National Heart, Lung, and Blood Institute Acute Respiratory Distress Syndrome (ARDS) Clinical Trials Network, Wiedemann H.P., Wheeler A.P., Bernard G.R., Thompson B.T., Hayden D. Comparison of two fluid-management strategies in acute lung injury. N Engl J Med. 2006;354:2564–2575. doi: 10.1056/NEJMoa062200. [DOI] [PubMed] [Google Scholar]