Abstract
Early childhood growth and development is conditioned by the consecutive events belonging to perinatal programming. This critical window of life will be very sensitive to any event altering programming of the main body functions. Programming of gut function, which is starting right after conception, relates to a very well-established series of cellular and molecular events associating all types of cells present in this organ, including neurons, endocrine and immune cells. At birth, this machinery continues to settle with the establishment of extra connection between enteric and other systemic systems and is partially under the control of gut microbiota activity, itself being under the densification and the diversification of microorganisms’ population. As thus, any environmental factor interfering on this pre-established program may have a strong incidence on body functions. For all these reasons, pregnant women, fetuses and infants will be particularly susceptible to environmental factors and especially food contaminants. In this review, we will summarize the actual understanding of the consequences of repeated low-level exposure to major food contaminants on gut homeostasis settlement and on brain/gut axis communication considering the pivotal role played by the gut microbiota during the fetal and postnatal stages and the presumed consequences of these food toxicants on the individuals especially in relation with the risks of developing later in life non-communicable chronic diseases.
Keywords: Perinatal repeated low-level exposure, Gut homeostasis, Brain gut microbiota axis, Food contaminants, Non-communicable chronic diseases, Epigenetics
Core tip: Increase of non-communicable chronic disease in the world is now clearly associated to environmental factors exposure during life and more especially during fetal and postnatal life. This review outlines the actual understanding of the consequences, on pregnant women, babies and infants, of repeated exposure to low level food contaminants on the settlement and maturation of the gut with specific focus on gut homeostasis, brain gut axis exchanges and the pivotal role played by the microbiota. However, their exact mechanisms of action on health, and particularly the molecular ones including epigenetic regulations still need to be clarified.
INTRODUCTION
Intrauterine and post-partum periods belong to the very sensitive time periods at the origin of the adaptation of the body to environmental stimuli also described as adaptative behavior thanks to developmental plasticity. This is phenomenon by which a single phenotype can give rise to a range of different physiologic or morphologic states in response to different environmental conditions during development. However, this adaptation ability is somehow restraint to a certain point. Indeed, since Barker et al[1] described the Developmental Origin of Health and Diseases (DOHaD), several original publications have pointed out that a poor fetal and postnatal settlement of gut microbiota will necessarily remodulate neuro-immuno-endocrine maturation of the gut. This will then be responsible for the modification of the establishment of the brain neuronal connections at the origin of a higher risk of developing later in life non communicable chronic diseases affecting the gut and the brain functions (for review see[2]).
Environmental factors are known to impact placental development at the origin of fetal growth retardation and postnatal major modification of the neuro-immuno-endocrine functions maturation and cross communication leading to chronic diseases. However, while the impact of food contaminants on the appearance of these diseases is not to be debated, investigation related to their impact on the brain gut microbiota axis development and maturation during perinatal life are needed to correlate all the events for the nutrition of pregnant women and infants.
This review is aimed at describing the actual understanding of the indirect and direct repeated low-level exposure of respectively the fetus and the infant to major categories of food contaminants and the consequences on development and maturation mainly of the brain gut microbiota axis through their impact on neuro-immuno-endocrine modulation.
SETTLEMENT AND MATURATION OF THE BRAIN-GUT-MICROBIOTA AXIS
Intestinal homeostasis settlement is defined as the establishment of an equilibrium between gut microbiota, immune system and epithelial cell integrity[3]. This event occurs during the 1000 first days of life, from fetal stage to 2 years old[4]. Because gut microbiota plays a central role in the maturation of gut mucosa and immune system, many studies focused their interest on where and when will start maternal microbiota transmission. Early 20th century, the idea that gut microbiota acquisition in infant starts at delivery, following contamination with vaginal and fecal bacteria from maternal flora, was accepted. However, new methods based on independent-cultured molecular technics suggest that bacterial transfer from the mother to their infant begins in utero[5] although the exact mechanisms of this event are not well understood. In 2005 and 2008, Jiménez et al[6,7] detected the presence of bacteria in amniotic fluid, umbilical cord and meconium or first stool of a newborn, by polymerase chain reaction. More recently studies were performed on the identification of microbiome in human placenta following termed and pretermed gestations. Firmicutes, Tenericutes, Proteobacteria, Bacteroidetes, Fusobacteria phyla and nonpathogenic commensal microbiota were found and associated with antenatal infection[8,9]. Nevertheless, even if bacteria patterns could be transmitted from mother to children in the womb, differences of newborn microbiota composition were observed depending on the mode of delivery, preterm birth and the use of antibiotics[10-13]. Currently, it has been acknowledged that Staphylococci, Streptococci, Enterococci, Lactobacillus and Bifidobacteria are the first bacteria which will colonize our gastrointestinal tract from vaginal and fecal flora and some of them come from lactation[14]. Before the third year, the interindividual variability is higher than in adults, but food diet diversification generates gut microbiota evolution by increasing their richness/diversity and will tend to reach a stability in time also called eubiosis state[15]. Gut microbiota contributes in health benefits such as metabolism of food, competition against pathogen, growth of intestinal cells and stimulation of the immune system among others[16,17]. During this period, child microbiota remains very sensitive to various influences that can impact its composition and cause long term effects on our physiology[18]. Nonetheless, a modification of gut microbiota inducing dysbiosis is usually associated with many diseases as inflammatory bowel disease, autism, type 2 diabetes, obesity, autoimmune diseases and allergy[19,20]. For example, clinical trials provide proofs concerning the relationship between allergy development and microbiota dysbiosis[21] in function of delivery mode in infants[22]. The behavior of children in the first 3 years of life clearly promotes significant exposure to microbes: Breastfeeding and direct contact with maternal skin, permanent introduction of objects, hands or feet into mouth, especially during crawling and early walking stages when hands are in contact onto floor surfaces. However, the actual excessive hygiene of our environment decreases the exposition to microbes in early life impacting immune system maturation. This has been mentioned in the rise of immune disorders (autoimmune diseases, allergies, etc.) for more than 40 years as “hygiene hypothesis”[23,24].
By contrast, as the intestinal microbiota of preterm infants at birth is less diverse than in full-term infants, it presents an immature intestine with underdeveloped peristalsis, barrier function and immunity. The intestine is therefore, a potential source of inflammation and infections by pathogenic bacteria such as Enterobacter, Enterococcus, and Staphylococcus[25,26].
Mucosal tissue homeostasis also results from the perinatal establishment of mucosal-induced immune tolerance, phenomenon regulated by a set of signals provided by innate immune cells that shape the adaptative immune responses. Through its barrier function, cell contact-mediated signals and production of cytokines, this regulatory immune network is controlled by the mucosal epithelium. Perinatal maturation of the mucosal immune system may also be modulated by direct or undirect effect of environmental factors whichever route of exposure considered, During pregnancy, maternal and fetal immune system communicate in a bi-directional manner. The development and maturation of the immune system starts early in fetal life and continues throughout the infancy and early childhood. These are windows of high susceptibility and vulnerability to environmental insults such as malnutrition, stress or environmental contaminants. The development of fetal immune system is characterized by the presence of several distinctive features that promote fetal active tolerance against both in utero maternal antigens and exogenous antigens, such as infectious agents. It is well established that endogenous and exogenous factors shape the development and maturation of the intestinal immune system. In rodents and human, the formation of secondary lymphoid structures, such as Peyer’s patches or mesenteric lymph nodes (MLNs), occurs before birth. However, their size and development of germinal centers depend on the postnatal microbial colonization[27,28]. Myeloid-derived cells, such as macrophages, dendritic cells (DCs) or brain microglial cells, begin to populate the tissues as early as gestational weeks 4 to 7 and continue to increase in number throughout the second trimester[29]. Then from week 8 to 18, the thymus will be populated by lymphoid cells through the process of lymphocyte maturation and positive and negative selection (central tolerance). The population will continue to expand within the thymus as gestation proceeds[29]. The first mature T cells are seen in peripheral tissues between weeks 10 and 12 of gestation and a significant number are circulating at the end of the second trimester[30]. In mice, innate cells such as lymphoid tissue induced cells, natural killer cells or T helper 2 like cells, migrate from fetal liver to the gut mucosa thanks to endogenous signals within the first 4 wk after birth[31]. Bacterial colonization will stimulate the recruitment of forkhead box protein 3 (FOXP3) and regulatory T cells (Treg) to the gut mucosa and secretion of the anti-inflammatory cytokine interleukin-10 (IL-10)[31,32]. By this mechanism, Treg cells keep pro-inflammatory Th cells under control to preserve the epithelial barrier. It has been observed a great abundance of Treg cells in human fetal MLNs[33] and their homing seems to be particularly important in infancy[34]. Bifidobacterium infantis, largely present in the gut lumen, strongly induces FOXP3+ Treg cells in human infants[35]. Reversely, with the development of the innate immune response most of microorganisms colonizing the small intestine and the colon will be anaerobes. The small intestinal mucosa of human newborns has a mature crypt-villus architecture, with continuous stem cell proliferation, and epithelial cell migration and differentiation. Changes in the composition of antimicrobial peptides and in antibacterial activity during early postnatal period have also been observed in the intestinal lumen of human neonates[36]. Intestinal immune homeostasis is also established thanks to a close cooperation between the intestinal epithelial cells (IECs) and the components of sub-epithelial gut associated lymphoid tissue. In the intestinal mucosa, molecules frequently associated with pathogens may be recognized by surface pattern recognition receptors and among them, Toll-like receptors (TLRs). Upon activation of TLR signaling, IECs will release transforming growth factor beta (TGFβ) by which tolerogenic phenotypes of DCs are elicited[37]. DCs then secrete IL-10 at the origin of an immune response dominated by Treg cells. In parallel, Th1 immunity may be induced and anti-inflammatory cytokines will be secreted[38,39]. Generation of FOXP3+ Treg and concomitant establishment of immune tolerance result from two steps. On the one hand, a founder pool of FOXP3+ Tregs is produced by the activation of antigen specific naive T cells in gut-draining MLNs. On the other hand, homing of activated Treg to intestinal lamina propria thanks to intestinal macrophages. Homing FOXP3+ Treg cells propagate, and immune tolerance is irreversibly installed. To provide passive immunity to the fetus or the neonate, maternal antibodies are transferred to the fetus by the placenta (starting as early as 6 wk of gestation with a rapid increase by week 24-26) and then to the infant through breast feeding. Peripheral activation of the immune system with production of cytokines can affect the central nervous system (CNS) directly via cytokines crossing the brain blood barrier or indirectly by activating the hypothalamic pituitary adrenal axis[40].
At last, the homeostasis of maternal and fetal endocrine milieu is also important to allow normal development and prevent adverse effects arising from early insults. Alteration of gestational endocrine environment by diseases will then perturb hormonal homeostasis and may be at the origin of fetal programming of altered endocrine profile in fetus. An important pathway by which gut microbes and their metabolites communicate with CNS involves the cells making up the endocrine system of the gut[41]. Enteroendocrine cells are intersped between gut epithelial cells throughout the length of the gut and are able to release different types of molecules that can enter the systemic circulation and have an incidence on the CNS regulatory pathways. 5-Hydroxytryptamin or serotonin (5-HT), the main regulating hormone of gastrointestinal motility and secretion, is produced by the enterochromaffin cells and stored in these cells and enteric neurons but also in CNS. Most of the molecules contributing to the neuroendocrine signaling within the brain-gut-microbiota axis originate from the gut microbiota metabolism of food as it is the case for tryptophan, a precursor to the neurotransmitter 5-HT[42].
While neuronal connections start to settle during the fetal period, their complete development won’t be achieved before adulthood. This is the reason why brain/periphery neuronal organization will remain very sensitive during a wide lifespan window. Thanks to epidemiological observation, the developing brain is known to be very sensitive to both external and internal environmental cues as early as prenatal life. During this period, maternal metabolic and immune activities will be dependent of the external environment. Their reactivity will interfere with the neurodevelopment in the womb and any alteration of maternal endocrine or immune response may have strong irreversible consequences on fetal development through epigenetic modifications. These studies linked neurodevelopmental disorders such as schizophrenia or autism with microbial infection during fetal life[43-45]. While placenta is considered to protect the fetus placing him in an almost sterile environment, the maternal environment will, by many ways, condition fetal brain development. Indeed, maternal microbiota metabolites may cross the placental barrier and affect the fetal brain construction programming which is dependent on placental neurohormones such as serotonin, essential for the forebrain development[46] or metabolic hormones such as adipokines or steroids controlling cell proliferation, differentiation, neurodevelopment[47,48]. Furthermore, maternal gut microbiome regulates serotonin production by enterochromaffin cells[49]. Its depletion results in uttered brain development[50]. Maternal dysbiosis will also influence blood brain barrier formation[51].
Brain development will continue postnatally, and metabolic programming of energy expenditure will be conditioned by a proper settlement of the Arcuate nucleus/hypothalamic connections that take place the first couple of days after birth. Synaptogenesis begins shortly after birth and reaches a maximal level around 2 years of age before synaptic refinement during mid-adolescence to get adult neuronal organization[52].
This period of neuronal development parallels the gut microbiota settlement and maturation a process known to be essential for the establishment of a proper immune function[56-60], the neuroendocrine system[61] and metabolic regulation[15,62]. Gut microbes communicate with the CNS through at least three parallel and interacting channels involving endocrine, nervous and immune signaling mechanisms. As thus, the settlement of this bidirectional communication is very crucial for preserving health according to a large body of literature. Alteration of this cross talk will be at the origin of the pathogenesis and pathophysiology of classic brain/gut disorders such as gastrointestinal disorders such as irritable bowel syndrome or inflammatory bowel diseases but also a growing list of psychiatric disorders or autism[63,64], Parkinson’s disease[65] or chronic pain[66]. One can suspect a mis settlement of this bidirectional communication to be at the origin of all of these chronic non-communicable diseases the alteration of gut microbiota settlement early in life.
During a sensitive window of development, environmental chemical exposure to chemicals, particularly through nutritional route, will act when tissues are forming, to affect the phenotype, shift the immune-metabolism-microbiota interactions, with consequences on the neuro-immuno-endocrine pathways at both the gut and the brain levels and this will then impact organ functions and chronic diseases development susceptibility later in life[67]. Furthermore, many mechanisms presented here are under genetic control including specific gene expression at each developmental and maturation stage. These mechanisms are allowed by particular transcription factors that interact with DNA to activate or inhibit gene expression. Epigenetic mechanisms are linked to accessibility of DNA to these transcription factors and are not transmitted with the classical mendelian genetics. Epigenetic is linked to biochemical modifications that affect DNA compaction and accessibility, essentially methylation of DNA on the specific nitrogen base cytosine and post translational modifications of Histone proteins. miRNA are another epigenetic mechanism where these small RNAs from 21 to 24 nucleotides are able to hybridise mainly in the 3’-UTR region of mRNA and block translation or cause mRNA degradation[68]. Alterations of epigenetic modifications are presented as key events of developmental programming[69,70]. Epigenetic mechanisms are inherently malleable and may accumulate over time which will, during the developmental programming of individuals, have profound effects on gene expression and predispose to the development of disease phenotypes profile[71].
The “Trialogue” among bacterial inhabitants, intestinal barriers and gut -associated neuro-immuno-endocrine components should normally ensure genesis and preservation of a stable symbiotic relationship consolidating the gut homeostasis. However, if the organism gets into contact with environmental toxicants, carried inside the lumen through food intake, these conditions could be at the origin of instability increasing the risk to develop chronic disorders, especially in critical windows of life such as perinatal stage.
ROLE OF FOOD CONTAMINANTS ON FETAL PROGRAMMING
Based on all this literature information, one can question whether prenatal or postnatal food contaminants exposure could somehow reprogram the fetus or infant overall development and more especially may interfere with the microbiota settlement, reconditioning the maturation of the gut mucosa not only at the immune but also at the neuroendocrine level. One can speculate that all of these modifications will naturally impact the developing connection between the gut and the brain. This has clearly been evidenced that exposition to certain chemical substances during postnatal life and early childhood will predispose to significant and irreversible damage in the developing brain. Pregnancy is a peculiar state of high vulnerability for the woman and the fetus because of the necessary adaptation to a putatively noxious environment which may modify the symbiotic interaction between mother and baby and could, in turn, influence the fetal programming process. This could result in some permanent, often subtle alterations in different organs among which gut or brain. Many factors during and after pregnancy may influence the future health status of the human being: Genetic status, inflammation during pregnancy, but also diet. Indeed, food not only provides nutrients but is also the most important source of environmental contaminants of diverse origins including heavy metals[72] or persistent organic pollutants (POPs) coming from industrial activities and found in food and particularly in fatty fish[73]. Some other, such as pesticides, may be detected in harvested crops[74] or may appear during storage which is the case for fungi or bacteria toxins[75] or during food processing and particularly heating with the appearance of Maillard reaction products (MRPs)[76]. Fetuses are exposed to contaminants from their mother’s diet. Since all of these substances are unintentionally found in food products, they can impair health under certain circumstances. Human fetus is not a small adult. This vulnerable subpopulation is probably at a greater risk from food contaminant because of a higher absorption rate, a poor detoxification elimination capacity, rapid cell proliferation, and immature repairing mechanisms. In this part, we will review the consequences of food contaminants exposure of the mother during pregnancy on the maturation of fetus and then on infant and child.
Fetal exposure to antibiotics
As mentioned above, maternal microbiota is transmitted to the fetus during pregnancy through mechanisms partly understood and, during natural parturition, microflora is ingested into the neonatal gut, establishing the initial microbial population provided that delivery was realized without caesarean, without disinfectants, intrapartum antibiotics, antiseptic creams, etc[77]. As thus, perturbations of vaginal maternal microflora (diet, body composition, infection, antibiotic treatment, stress, probiotics, etc.) will definitely have significant consequences on the offspring. During pregnancy, vaginal bacterial infections may alter early neurodevelopment and seems to be related with autism spectrum disorder[78] and following the delivery it may alter the microbial assembly of the neonatal gut. This will decrease diversity and stability of the microbiota populations that will influence fermentation, digestion and absorption of metabolites, increase of antibiotic resistant bacteria such as Klebsiella, Citrobacter, Enterobacter, E. coli[79-81]. Altogether, these effects will alter the development of the neonate and the infant and will be increasing its disease risk.
In rodents, exposure of dams to antibiotics during lactation is responsible for Lactobacilli abundance depletion, an increased fat mass and altered secretion of metabolic hormones in their offspring[82,83].
Fetal exposure to pesticides
Impacts of pesticides on the offspring after maternal exposure has been widely documented in the literature. However, few studies evidenced their deleterious effects at repeated low-level exposure. Many of these have long been described as endocrine disruptors. Organophosphates and carbamates such as chlorpyriphos (CPF), malathion, diazinon, carbaryl share cholinesterase inhibitory activities. Moreover, these organophosphate do have a strong incidence on microbiota balance[84]. It has been evidenced that exposing dams to diazinon results in behavioural disturbances in their offspring[85]. It is acknowledged that environmental chemicals, especially endocrine disrupting factors, will induce functional modification of gene expression and when these changes occur in fetal life this may be at the origin of increased risk of dysfunction and disease later in life although any phenotype change will be observed at birth[67].
Carbofuran, an anticholinesterase carbamate, is commonly used as an insecticide, nematicide, and acaricide in agricultural practice throughout the world. Carbofuran and/or its major metabolites can cross the placental barrier and produce serious effects on the maternal-placental-fetal unit. Carbofuran's toxicity can be potentiated by simultaneous exposure with other cholinesterase inhibitors[86].
Chlorpyrifos [O,O-diethyl-O-(3,5,6-trichloro-2-pyridinyl)phosphorothioate: CPF] is an organophosphate insecticide used worldwide to treat fruit and vegetable crops. The most widely studied organophosphate, CPF has been clearly identified as a neuron killer, neural cell migration and brain connection disruptor and its residues are often detected in food and drinking water[87]. Although the digestive tract is the first organ to come into contact with food contaminants, little is known about CPF’s impact on the epithelial barrier. Studies from Joly Condette et al[88], have shown that chronic CPF-exposure during critical pre- and postnatal periods of organ development and maturation alters epithelial barrier function, which in turn is associated with elevated permeability and bacterial translocation. Furthermore, the barrier dysfunction is associated with changes in tight junction’s protein expression. In the same rat model, rat pups exposed to 5 mg/kg/d CPF were both significantly smaller (body length) and lighter than controls. Exposure to CPF was associated with changes in the histological structures (shorter and thinner intestinal villi), an intestinal microbial dysbiosis and increased bacterial translocation in the spleen and liver. These significant microbial changes in the gut were associated with impaired epithelium protection (mucin-2) and microbial pattern recognition receptors (TLRs 2 and 4) genes expression[84]. In summary, pesticide residues in food may impact the digestive tract function and its ability to adapt to environmental changes. In rats, this effect appears to be even greater at the time of weaning (i.e., when food diversification occurs). Increasing evidence indicates that CPF is involved in metabolic disorders. Data from Reygner et al[89], indicate that developmental exposure to CPF interferes with metabolism with dose related effects evident at adulthood. Today, as developmental intellectual disorders affect one out of six children in industrialized countries, there is a growing interest in identifying impacts of pesticides on brain development and maturation and in the etiology of intellectual impairments. A study from the same French team assessed whether maternal ingestion of low CPF dose in rats could impair the cerebral function of their progeny[90]. According to the results, the progeny of CPF-treated dams showed slower negative geotaxis as neonates, lower novelty exploration as juveniles and faster startle reflex as adolescents and adults. This data suggests that developmental CPF relevant to human exposure may impair novelty-related activity and sensori-motor functions, thus adaptability to the environment. This data supports the hypothesis that CPF may contribute to behavioural disorders including acquisition retardation and consequences as an adult. Another organophosphate compound has also been studied for its potential health impacts. Acephate is a pesticide that targets insects which also belongs to the organochlorines. As for CPF, a Wistar rat animal model suggests that acephate exposure during pregnancy and lactation causes alterations in maternal glucose metabolism and programs the offspring to be susceptible to type 2 diabetes at adulthood[91].
Organochlorine pesticides (OCPs) are environmental contaminants that persist in the environment and bioaccumulate through the food chain in humans and animals. The results from Yamazaki et al[92] support the hypothesis that prenatal exposure to OCPs, especially cis-heptachlor epoxide, may have an adverse effect on the neurodevelopment of infants at specific ages, even at low levels. Chlordecone is a persistent OCP that was used in the French West Indies until the early 1990s for banana weevil borer control. In a prospective longitudinal study conducted in Guadeloupe (Timoun mother-child cohort study), exposure to chlordecone was measured at birth from an umbilical cord blood sample and from a breast milk sample collected at 3 mo postpartum. The results suggest that prenatal exposure to chlordecone is associated with specific impairments in fine motor function in boys, and add to the growing evidence that exposure to OCPs early in life impairs child development[93]. Similarly, in the same birth cohort Timoun in Guadeloupe, another objective of the study was to examine the association between prenatal exposure to chlordecone and fetal growth measured by birth weight[94]. They found a significant U-shaped association between birth weight and chlordecone exposure, within the upper quartiles of gestational weight gain or excessive gestational weight gain. Chlordecone exposure may affect fetal growth, particularly when excessive gestational weight gain is present. Other pesticides such as dichlorodiphenyl-trichloroethane and its highly toxic metabolite dichloro-diphenyl-dichloroethylene (DDE) are persistent OCPs which are lipophilic environmental pollutants that accumulate in the food chain. These chemicals have recently been under scrutiny for their possible health hazards such as cancer and reproductive outcomes including low birth weight. The study from Khanjani et al[95] investigated whether mothers with a higher contamination of pesticides were different from mothers with low contamination in relation to their offspring's birth outcomes such as birth weight, small for gestation age, prematurity, head circumference, sex ratio, and previous miscarriage or still birth. In this case, those pesticides were not found to be associated with adverse birth outcomes in contaminated mothers in the range of contamination of our population (< 7.5 mg/kg lipid in maternal milk), although there is weak evidence that sex ratio may be affected. Maternal exposure to Great Lakes sport-caught fish contaminated by DDE suggest that fetal DDE exposure (as indicated by maternal serum DDE concentration) may decrease birth weight[96].
Amitraz is an active plant protection product, which has an antiparasitic effect, and which belongs to the chemical family of formamidines. Kim et al[97] investigated the potential adverse effects of amitraz on the initiation and maintenance of pregnancy in Sprague-Dawley rats as well as its effects on embryo-fetal development after maternal exposure during the entire pregnancy period. Amitraz was administered to pregnant rats by gavage from days 1 to 19 of gestation at dose levels of 0, 3, 10, and 30 mg/kg/d. All dams underwent a caesarean section on day 20 of gestation and their fetuses were examined for any external, visceral, and skeletal abnormalities. At 30 mg/kg, maternal toxicity manifested as an increase in the incidence of abnormal clinical signs and a lower body weight gain and food intake. Developmental toxicity included an increase in the fetal death rate, a decrease in the litter size, and a reduction in the fetal body weight. In addition, there was an increase in the incidence of fetal external, visceral, and skeletal abnormalities. At a dose below 10 mg/kg, maternal toxicity observed included a decrease in the body weight gain and a decrease in food intake associated to a minimal developmental toxicity. This includes a decrease in the fetal body weight, an increase in the visceral and skeletal aberrations, and a delay in fetal ossification. These results show that amitraz administered during the entire pregnancy period in rats is embryotoxic and teratogenic at the maternally toxic dose (i.e., 30 mg/kg/d) and is minimally embryotoxic at a minimally maternally toxic dose (i.e., 10 mg/kg/d).
Diuron is widely used as a weed killer to kill unwanted grasses and other annual and evergreen broadleaf weeds, especially in viticulture. It is also used in gardens and for weeding roadsides or railways. A perinatal exposure to diuron [3-(3,4-dichlorophenyl)-1-1-dimethylurea] might exert adverse effects on rat lymphoid organs. Pregnant Sprague-Dawley rats were exposed to diuron at 500, 750 or 1250 ppm in the diet from gestational days 12 to 21 (GD 12-21) and during lactation. Flow cytometric analysis revealed a significant reduction in B lymphocytes (CD45RA+) in male pups but T lymphocytes (CD4+, CD8+ and CD4+/CD8+) were not markedly affected. Thus, data suggest that Diuron-induced maternal toxicity in dams exposed to high dose and perinatal exposure to this herbicide produced spleen toxicity as evidenced by a reduction in B lymphocyte number in male Sprague-Dawley pups[98].
Fetal exposure to mycotoxins
Because of the growing evidence of the deleterious effects of pesticides on health, more and more people are choosing to consume organic products. However, there is growing attention in western countries related to the increasing consumption of organic products since the actual means of crop storage conditions may not prevent from mycotoxins food contamination, especially when it relates to organic products. This concern is even more important when it comes to pregnant women and infants. Indeed, cereals intended for human consumption may be contaminated by fungi species of the genera Aspergillus, Penicillium, Fusarium and Alternaria, which are able to produce highly noxious secondary metabolites: Mycotoxins[75]. The most relevant worldwide mycotoxins in cereals are aflatoxins (AF), ochratoxin A (OTA), patulin (PAT), fumonisins, zearalenone (ZE) and trichothecenes including T-2 toxin and deoxynivalenol (DON)[99]. Based on current available evidence, it is probable that a placental transfer of mycotoxins occurs as early as gestational stage and accumulates in the fetal circulation towards the end of pregnancy. Mycotoxins from human maternal contaminated food consumption can cross placental barrier, be metabolize by the developing fetus and can affect fetal systems[100]. This vulnerable population is probably at a greater risk from carcinogenic environmental toxins because of high absorption rate, poor elimination capacity, rapid cell proliferation, and immature repairing mechanisms[101]. Few studies are carried out on the effect of mycotoxins on the fetus during gestation. Moreover, an occupational exposure to mycotoxins in grain induces labor at an early stage of pregnancy[102].
Only one human study investigated AF exposure and prematurity, but animal studies suggest AF exposure may increase risk for prematurity and pregnancy loss. The fetus could be affected by maternal AF exposure through direct toxicity as well as indirect toxicity, via maternal systemic inflammation, impaired placental growth, or elevation of placental cytokines. The cytotoxic and systemic effects of AF could plausibly mediate maternal anemia, intrauterine growth restriction, fetal loss, and preterm birth[103]. A study realised on a specific Human population submitted to AF exposition showed perturbation on epigenetic modifications. Precisely, it has been shown a correlation between AF rate in mother during gestation and a differential methylation pattern of many genes in child after birth, susceptible to affect gene expression[104]. Indeed, another study on cells from the digestive tract showed that exposition to another mycotoxin fumonisin B1 altered methylation of gene promotors, showing that this mechanism is possibly general[105].
Wangikar et al[106], 2014 studied the effect of OTA and aflatoxin B1 (AFB1) mycotoxins administered orally to pregnant rats, revealing that these mycotoxins caused visceral anomalies such as gastroschisis. Arora et al[107], reported that a single dose of AFB1 given in a pregnant female by the stomach tube caused protrusion of intestines after exposure on gestation day 8. Treatment of pregnant rats with AFB1 resulted in the formation of benign and malignant tumors in the liver, stomach, intestine, endocrine organs, and the central and peripheral nervous system in the offspring.
ZE, an estrogenic mycotoxin produced by Fusarium graminearum or F. roseum, is one of the most common contaminants of cereal grains world-wide. The objective of the study from Collins and colleagues was to determine the effects of ZE on in utero development of rats[108]. Pregnant female Charles River Sprague-Dawley rats were gavaged once daily with ZE (in corn oil) at doses of 0, 1, 2, 4, or 8 mg/kg body weight on GD 6-19. In summary, ZE was maternally toxic and fetotoxic but not teratogenic.
Fetal exposure to other organic pollutants
Bisphenol A (BPA) is an organic compound in the aromatic family, used primarily in the manufacture of plastics and resins. It is pointed out as a risk factor in development of food allergy and food intolerance, two adverse food reactions increasing worldwide. Menard et al[109] evaluated the consequences of perinatal (end of gravity to weaning) exposure to low doses of BPA on immune-specific response to the food antigen ovalbumin (OVA) at adulthood. When BPA-treated OVA-tolerized rats were orally challenged with OVA, colonic inflammation occurred, with neutrophil infiltration, increased IFNγ, and decreased TGFβ. They show that perinatal exposure to BPA altered oral tolerance and immunization to dietary antigens (OVA). In summary, the naive immune system of neonate is vulnerable to low doses of BPA that trigger food intolerance later in life.
POPs are highly resistant chemicals, including dioxins, furans, polychlorinated biphenyls and OCPs, created by industrial activities and found in food with the highest levels found in fatty fish[73]. These highly toxic compounds have been found to cross the placenta and to be excreted in breast milk[110]. Early life exposure to POPs is associated with developmental immunotoxicity and neurodevelopmental disorders like autism spectrum disorders (for review see Dietert et al[111]). This complex interaction between the immune system, environmental pollutants and neurodevelopment has been described by different authors although the exact mechanisms are still not well understood[112]. However, prenatal and postnatal exposure to POPs are clearly associated with brain damage[113,114]. At last, most of POPs and particularly tetrachlorodibenzofluranes, polychlorinated bisphenyls (PCBs) or chlorothalonil, another OCP, has been described to have strong incidence on gut microbiota composition[115] but this has still to be confirmed in infants.
Fetal exposure to heavy metals
For the general population, the most common source of exposure to toxic heavy metals are through the air and dietary intake. Due to their high vulnerability, pregnant women and infants are even more concerned than any other by exposure to them since their effect on health are major. Mercurials are global environmental pollutants deriving from natural processes and anthropogenic activities. Most human exposure to mercury occurs through the intake of fish, shellfish, and sea mammals contaminated with methylmercury (MeHg)[116]. The neurotoxic hazard posed by MeHg to humans and the unique susceptibility of the developing brain have been well documented following the mass poisonings occurring in Japan and Iraq. Why the foetus displays different neuropathological effects and a higher sensitivity to MeHg relative to the adult is still unknown. Depending on the degree of in utero exposure, MeHg may result in effects ranging from foetal death to subtle neurodevelopmental delays. On the basis of epidemiological studies performed in populations having moderate chronic MeHg exposure, no definitive consensus has been reached to date on the safety level of maternal exposure during pregnancy[117]. In the study from Cambier et al[118], pregnant rat mothers were contaminated with environmentally relevant doses of 36 and 76 ng MeHg/g of food using diets containing naturally mercury-containing fish. Newborns were protected against Hg exposure by the placental barrier since in newborns from mothers fed the diet containing 76 ng MeHg/g of food, the concentrations of Hg in brain, kidney, liver and skeletal muscles represented 12, 3, 21 and 18% of those of their mother's tissues, respectively. These results suggest the existence, at least in rats, of a threshold level in terms of MeHg exposure above which the placental barrier collapses. In behavioural tests performed at 5 and 6 wk of age, MeHg-exposed rats showed a significant deficit in motor coordination in the rotarod test and a learning disability in the passive avoidance response test, compared with controls. Histopathologically, focal cerebellar dysplasia, including the heterotopic location of Purkinje cells and granule cells, was observed. These abnormalities may be induced by the effect of highly accumulated MeHg in the brain during the gestation period[119]. As shown above, foetuses are particularly sensitive to MeHg exposure and adverse effects on infant development have been associated with levels of exposure that result in few, if any, signs of maternal clinical illness or toxicity. High levels of prenatal exposure in humans result in neurobehavioral effects such as cerebral palsy and severe mental retardation. Prenatal exposure to MeHg in communities with chronic low-level exposure is related to decreased birth weight and early sensorimotor dysfunction such as delayed onset of walking[120]. Moreover, late pregnancy exposure to mercury intensified the toxic effects of lead contamination on infant neurodevelopment at 6 mo of age[121]. Cadmium is known to have endocrine disrupting activities[122]. In utero cadmium exposure in human has been associated to modification in methylation of genes associated to lipid metabolism and bone mineralisation[123]. As a result, one can program increased systemic fat accumulation and increased incidence of bone fractures and osteoporosis later in life[123]. Arsenic is a potent toxicant and carcinogen and can be found in a large variety of food including fish and rice[124]. A clinical study, based on blood cord samples, revealed that exposure of pregnant women to arsenic was responsible for the increased expression of 12 miRNA predicted to be involved in regulation of signaling pathways commonly deregulated in cancer and diabetes mellitus[125]. This study thus confirmed the deregulation of miRNA expression following arsenic fetal exposure and partly explains how arsenic contributes to the development of these diseases.
Children from Faroe Island born from apparently healthy mothers who ate mercury contaminated fish during pregnancy showed altered neuronal connections evidenced by decreased scores on attentiveness and memory[126]. Lead exposure may cause increased risk of pregnancy hypertension and miscarriage[127]. Arsenic, lead and MeHg have been associated with reduced length of gestation or preterm birth[127-130]. A French group has reported results from cross studies of women before pregnancy and during the third trimester. They evidenced that exposure before pregnancy was higher than in the last trimester. The reason of this difference seems to relies partly on the increase of body weight that was not compensated by parallel increase in food intake[131]. This was one of the first study in France revealing the level of dietary exposure to food contaminants for the foetus and how it is affected by changes of food intake during pregnancy and seasonal variation. Dairy consumption is associated with lower mercury and lead levels in both pregnant women and children. Lead absorption is enhanced under calcium deficiency whereas lead can interfere with the cycle of calcium homeostasis and function[132,133]. This explains why high dairy intake, meaning high calcium intake, might reduce lead absorption and its related neurotoxic effects[134]. So far, very few studies evaluated joint exposure to heavy metals. To our knowledge, only one recent work illustrated that joint exposure to lead and mercury in late pregnancy negatively affected the neurodevelopment of infants aged 6 mo[121].
Fetal exposure to MRPs
Since the first human discovered the fire, our food habits evolved a lot and this evolution increased markedly this past century in parallel with deep modification of our way of life especially in northern modernized countries. Food cooking became necessary to improve food nutritional and organoleptic qualities, hygiene but also facilitates its storage on a longer period. However, this transformation process comes with the appearance of new compounds among which MRPs. This wide set of molecules appears through cooking and results from the reaction between the carbonyl group of a reduced sugar and a free amine group of amino acid, typically the ε-amino group of lysine residues in proteins. This initial reaction gives then the rise to a large bunch of molecules through multiple rearrangements at the origin of the taste of cooked food, the brownish to grilled color of food. However, advanced glycation end-products (AGEs), acrylamide or heterocyclic amine belonging to glycotoxins may also have negative effects on health[135]. Dietary AGEs get absorbed through the small intestine and their blood concentration increases lifelong and further exacerbates their endogenous production[136]. Exposure to high AGEs during fetal development could lead to inflammatory states at young age such as juvenile diabetes[137]. In rodents as well as human, consumption of AGEs rich diet (AGE-RD) negatively affects glucose homeostasis and favors the appearance of free radicals. Csongová et al[138], recently evidenced that AGE-RD consumption during pregnancy might accelerate the maturation of reflexes in their offspring. Furthermore, they showed that this diet would predispose the male progeny to weight gain and affect their glucose metabolism[138]. Unfortunately, the study did not indicate any impact on the digestive tract. Literature describes that endogenously produced AGEs are able to interact with the receptor for advanced glycation end products (RAGE) leading to prolonged activation of nuclear factor kappa B (NF-κB) which result in the transcription of pro-inflammatory cytokines, including TNFα[139,140]. Then, restricting AGE intake could be beneficial in reducing levels of some inflammatory markers, helping to limit the risks of developing later in life the progression of chronic conditions[141]. Two longitudinal studies have shown a relationship between maternal acrylamide exposure and lower birth weight or higher risk of having a small baby for gestational age[142,143]. Furthermore, increasing maternal exposure to dietary acrylamide during pregnancy was associated with higher prevalence of children being overweight/obese at 3, 5 and 8 years of age[144]. Food acrylamide, largely found in fried potatoes, breakfast cereals, biscuits and coffee is extensively absorbed from the gastrointestinal tract and after reaching the systemic circulation us rapidly distributed into the tissues. Acrylamide is recognized as a neurotoxicant[145] and can exert reproductive and developmental toxicity effects[146]. This is rather critical since it has been estimated that 10%-50% of acrylamide passes the placental barrier[147,148]. Hormonal and endocrine effect of acrylamide is equivocal. Acrylamide modifies thyroid activity since it has been reported that urinary acrylamide metabolites were negatively associated with free thyroxine[149] in adolescents and young adults. Fetal thyroid function might as well have an impact on fetal growth[150] since thyroid hormones are essential for optimal growth. This could partly explain growth retardation observed in acrylamide exposed children. Diamanti-Kandarakis et al[151] showed that exposure of dietary AGEs affects metabolic and hormonal functions of female rats. Six months exposure to highly heated rodents chow rich in AGE increased: (1) Deposition of AGEs in tissues; (2) Expression of RAGE; and (3) Elevated plasma testosterone levels in theca cells of ovaries. Furthermore, female Wistar rats fed high-AGE diet had declined levels of estradiol and progesterone. These female sex hormones are crucial for implanting the fertilized egg in the uterus, maintaining pregnancy and also to regulate the estrous and menstrual cycles[151]. Such effects may then have severe repercussions onto the developing fetus. It has been evidenced that peripheral blood mononuclear cells of rats isolated from high-AGE diet-fed female rats in had increased expression of RAGE, the AGEs receptor associated with the proinflammatory pathways[152,153]. This might be a mechanism also stimulated in the developing child which could contribute to alteration of the programming function.
ROLE OF FOOD CONTAMINANTS ON INFANT AND CHILD DEVELOPMENT AND MATURATION
While fetal exposure to food contaminants may strongly condition developmental programming, one cannot exclude the postnatal exposure to these contaminants as an important factor contributing to modification of this programming. Disruption of the developing gut and of the establishing cross communication between the brain and the gut by food contaminants may lead to a reduced capacity to settle local and systemic neuro-immuno-endocrine functions and an increased risk to develop neurological and inflammatory disorders. Indeed, strong or novel evolutionary challenges may be at the origin of disruption of normal signaling pathways explaining the appearance soon or late of these disorders. However, while more and more studies evidence the consequences of maternal exposure during pregnancy on the fetus and the infant/child, very few studies focused on the direct exposure of the baby and the infant to food contaminants. This will be important to figure out all these effects since infants and children are very vulnerable to food contaminants since they consume high amounts of food in comparison with their weight, have a higher metabolic rate and thus get a higher exposure to them by contrast to adults. In this part of the review, we will summarize the actual state of the art, considering that most of the below cited studies will often be related to gut microbiota settlement but there are very few works related to neurotoxicity of food contaminants on juvenile individuals.
Postnatal exposure to antibiotics
Pharmaceutical products among which laxatives[154] or antibiotics[155] have a major influence on the gut microbiota activity and may have repercussions on the settlement of local and systemic interactions with the neuro-immuno-endocrine functions. Disrupting the overall eubiosis results in shift of abundance of resident gut bacteria and predisposes the child to certain inflammatory or functional disorders[156]. Use of antibiotics during the postnatal period may severely alter this fragile settling ecosystem especially when they are given very early and for long periods of time[157]. This disrupted profile associated to a decreased microbial activity will be conditions favoring the settlement of enteric pathogens[158,159]. This has been associated with long term disruption with intestinal barrier function but will also impact the growth of dominant bacteria in the human gut[157], confirming that antibiotic exposure in early life car render the infant susceptible to numerous diseases later in life[160,161]. The perturbation of composition and biodiversity of the gut microbiota is often associated to an increase of Proteobacteria because of a higher content of antimicrobial resistance genes in this phylum[162] and the disruption of our microbiota caused by antibiotics favors the development of non-communicable chronic diseases (NCCDs) characteristic of dysbiosis. At last, the alteration of the microbiota composition observed in infants treated with antibiotics was close to the one of children from mothers treated with antibiotics before delivery, with consequences late in life[163]. Moreover, antibiotics treatment has the main disadvantages of enriching our microbiota with antibiotics resistant genes which might become later on a problem towards the gut homeostasis. Furthermore, disrupting microbiota settlement will also disrupt its overall metabolizing activity which concerns its ability to transform food contaminants and limit their impact on our health (for review see Flandroy et al[164]). On top of this, although the use of antibiotics might be very important to prevent sepsis in preterm or newborn infants, they might, under some circumstances, interfere with neurotransmitters receptors and particularly those of gamma-amino butyric acid[165] or of serotonin[166], compromising correct central and peripheral neuronal circuitry which may under prolonged exposure, have serious consequences[167]. These toxicological effects have been evidenced for amoxicillin in juvenile rats[168].
Rotavirus infection is a very common cause of gastroenteritis since it targets mainly the gastrointestinal tract. In animals, several observations are concordant: Rotavirus infection decreases intestinal enzymes activities in both piglets[169] and young mice[170,171]. In rabbits, it impairs epithelial homeostasis by altering the intestinal brush border membrane[172]. Rotavirus infection in children results in the occurrence of a flat mucosa with total virus atrophy[173]. Exposition during early life to environmental microorganisms strongly affects protein expression at the brush border since it clearly stimulates TLR5 and TLR9 gene expressions of exposed new born mice[174]. This mismatch at birth will fragilize the maturation of the intestinal epithelium.
Postnatal exposure to pesticides
There is growing evidence from animal studies suggesting that early life exposure to toxic stress may magnify the effects of environmental chemical exposure on neurodevelopment[175]. Air pollution and pesticides may affect growth during infancy or childhood, and they are associated with neurodevelopmental and behavioral problems[176].
Organophosphates pesticides are neurotoxic at high doses and target the nervous system of children, and are associated with changes in neurotransmitters including serotonin, norepinephrine, acetylcholine, and dopamine. The associations of organophosphate pesticide and child IQ at 7 years old was noted by Stein et al[177] in 2016 and was already observed[178] as more important in children that also experience certain social adversities. Those who have the highest exposure to environmental toxicants are often the same populations and individuals who experience the greatest adversity. Children with high prenatal exposure to organophosphates who also experience specific early and persistent toxic stress may be at a greater risk for adverse cognitive development[177]. However, postnatal organophosphates studies are not numerous and most of the results are inconsistent[179].
Prolonged breastfeeding was associated with increased of OCPs in Norwegian children blood samples[180]. In studies that measured the levels of DDE and PCBs, organochlorine compounds postnatally, authors reported a slower growth and a higher risk of endocrine function alteration in children[181].
Glyphosate is one of the agro-chemical which should also deserve a special attention in infants and children. In fact, this herbicide is inhibiting the shikimate pathway, which, when present in bacteria, controls the synthesis of aromatic compounds such as tyrosine or tryptophan, key intermediaries in systemic effects of our microbiota (see the first section of this review)[164]. From now, there seems to be no direct effect of neonatal exposure to glyphosate on gut brain axis maturation. However, since it may alter uterus development, one can’t exclude any alteration of the gut homeostasis[182], since it has been evidenced that this herbicide could temporarily modify commensal bacterial community composition[183].
Postnatal exposure to mycotoxins
Infants and young children are more vulnerable to mycotoxins than adults, due to their constantly growing, poorly developed nervous, immune, reproductive, and digestive systems, high rate of metabolism, and restricted diet. Childhood exposure to mycotoxins, induced by the consumption of contaminated foods : Breast milk[184], infant formula or complementary foods with cereals[185], vegetables or nuts[186,187]. This has been associated with poor child growth and development[188], increased susceptibility to infections and many others effects like immune disorders[189], tumors development[190], precocious puberty[191,192], etc. Assessment of health risks related to mycotoxins ingestion during childhood has been evaluated in the last years. The presence of mycotoxins in the intestinal fluid of a child could cause more significant damage to the intestinal enterocytes because of the reduced dimension of the intestinal epithelium compared to that of an adult. An outbreak in the children population of Kashmir Valley was associated with the consumption of naturally contaminated bread with a varying quantities of trichothecene mycotoxins[193]. Symptoms of gastrointestinal illness appeared 15 min to 1 h after consuming it. DON belonging to these trichothecenes, can act on the viability and the proliferation of immune cells. This event then inhibits proteins biosynthesis that prevent cytokine production[194]. The direct consequence of this event is a higher susceptibility to infection disease which is of peculiar importance on such vulnerable people that are babies and infants. Moreover, it was evidenced that DON was affecting the intestinal barrier function in chicken[195].
AF may be generated by Aspergillus Fungi. One of its metabolites, a 4-hydroxylated form, can be secreted in mammal milk after ingestion of the mycotoxin. As for DON, AF reduce anti-inflammatory cytokines IL-4 expression together with the increase of susceptibility to infection[196].
Another outbreak of diarrhea in children in Sweden was related to consumption of fruit contaminated with PAT[197]. Indeed, it was reported that consumption of high concentration of mycotoxins (DON and PAT) could cause gastroenteritis with vomiting in humans[198,199]. PAT is cytotoxic to enterocytes, increase permeability across intestinal Caco-2 monolayers and alter ion transport in intact intestinal mucosae[199,200]. Fusarium mycotoxins are quite frequently found in baby food in particular in the developing countries. Despite of efforts made to correct the heating process to limit or eliminate the mycotoxins from maize or soy-bean, it has been evidenced that this food transformation not only does not eliminate mycotoxins but rather help to generate many others thus presenting a higher risk for the consumer and particularly babies[184].
Other groups of metabolites produced by some Aspergillus or Penicillium are Ochratoxins. However, from the actual knowledge, it seems not to have any consequence neither on the gut nor on the grain but rather on kidneys.
At last, fumonisins, Fusarium mycotoxins, may also be found in baby and infant food. From the actual literature, it has not been so far determined that they are responsible for gut disease apart from a foodborne disease outbreak in India[193]. However, they seem to alter the settlement of the nervous system since they are associated to neural tube defects in the Mexico-Texan border[201].
Postnatal exposure to other organic pollutants
Several POPs have been associated with increased children's behavior problems[202]. Some studies have pointed out that exposition for 24 h to 5 pollutants [TCDD, deltamethrin, hexabromocyclododecane, Benzo(a)Pyrene and 2-amino-1-methyl-6-phenylimidazo(4,5-b)pyridine] and one mixture of pollutants polycyclic aromatic hydrocarbures (PAHs) significantly shifted the microbial volatile pattern, suggesting that the activity of the microbiota might have been affected[203]. Moreover, their direct or indirect consequences on neuronal communication and network development has already been described[204]. Taking all these informations into account, one can’t exclude the possibility of misshaping the gut microbiota settlement in newborns and the sensible consequences on his/her health. POPs, PCBs, PAHs have been described as significantly altering the microbiota metabolism at the origin of the induction of a proinflammatory status of the gut[203]. 2,3,7,8-tetrachlorodibenzofurane, a POP, may shift the firmicutes/Bacteroidetes ratio which is at the origin of alteration of bile acid metabolism[205]. Furthermore, PAHs can be transformed by the gut microbiota to estrogenic metabolites[206].
In the same line, phthalates, a category of food plasticizers, may strongly unbalance the settlement of gut brain microbiota axis. Indeed, exposure to di-ethylhexyl phthalate through lactation showed negative association with mental index[202]. Phthalate metabolite concentrations were higher in the 2- and 5-years old children, compared to 11 years old ones. Postnatal high exposure to phthalic acid ester is related to attention deficit hyperactivity disorder symptoms in children[207], and particularly diethylphthalate are inducing gut dysbiosis and consistent weight loss. One of the mechanisms proposed was through the aryl hydrocarbon receptor activation[208]. Moreover, child exposition to Phthalates has been correlated with the decrease of DNA methylation of specific genes among which TNFα. As this demethylation is linked to a decrease of TNFα protein level, this could explain the gene expression deregulation observed during the inflammatory response as it was suggested for asthma[209]. These recent observations play in favor for a correlation that can be pointed out between contaminants and molecular pathways involved in a given disease.
Duration of breastfeeding was the main determinant and was significantly associated with increased POP concentrations in the children. Prolonged breastfeeding was associated with increased concentrations of POPs such as PCBs, and polybrominated diphenyl ethers in children blood samples[180]. In their study, Caspersen et al[180], revealed that concentrations of POPs in 3 years old children were 1.4-fold higher than in an independent sample of pregnant women.
Postnatal exposure to heavy metals
There are a dozen of studies concerning prenatal and postnatal exposure to heavy metals such as cadmium, arsenic, mercury or lead. Authors have reported in all of them a slower postnatal growth in children but due to a limited number of studies per specific heavy metals authors could not conclude on any other side effects[210]. Exposure to mercury, cadmium and lead in early life can disrupt the development of the nervous system and such exposure can affect the child’s cognitive, motor and behavioral development[72]. The main route of exposure to cadmium are inhalation then ingestion due to consumption of cereals and vegetables grown on contaminated soils.
On 278 nine-year-old children, 21% had total toenail mercury concentrations (1.5 to 6 μg/g) higher than the United State Environmental Protection Agency recommended levels (1 μg/g Hg) for optimal health in children, associated with an aggressive behavior[211].
A decrease of intelligence quotient at 1.5 and 5 years was associated with childhood duration and amount of arsenic exposure[212]. From other studies, it was established in mice that gut microbiota could metabolize arsenic intro potent toxic metabolites[213] that will interfere with neuronal signaling, limit brain weight but also neurons and glia number[214].
Postnatal exposure to MRPs
Depending on length and temperature of food heating, the amount, type and diversity of MRPs may significantly change. No direct evidence of MRPs incidence on the brain gut microbiota axis development has been described so far although most of AGEs are absorbed through the gut[215]. However, there are a couple of evidences confirming the activation of an immune response in the presence of MRPs probably due to the fixation of these molecules on RAGE and its subsequent activation of the NF-κB pathway leading to the release of pro-inflammatory cytokines such as TNFα, IL-6 or IL-1β and the increase of tissue oxidative stress along with an inflammatory response[216]. We and others are currently working on the establishment of any correlation with perinatal events.
Babies exposed to acrylamide show growth retardation[217]. This has been partly explained by prenatal alteration of fetal programming of the thyroid. However, another window of susceptibility should be investigated, and child’s postnatal exposition should be considered in addition to prenatal exposition. Another possible mechanism linking acrylamide exposure and growth is through oxidative stress and inflammation. Indeed, acrylamide exposure can result in increased oxidative stress through increased expression of CYP2E1, resulting further in heightened perinatal inflammatory status[218,219]. Dietary AGEs are responsible for oxidative stress and inflammation in several tissues by interfering with vital hormones involving physiological reactions. Exposure to high AGEs during childhood is responsible for inflammatory status. This speculation is supported by the relationship between high maternal AGEs and infant plasma insulin levels[137]. New born and infants may be very concerned by AGEs especially those that need to receive hydrolyzed infant formulas that have very high levels of Nε-carboxy-methyllysine (CML) as compared to breast milk due to the heat treatment of milk proteins in presence of lactose[220]. In a recent review, we suggested that neoformed compounds from the Maillard Reaction and especially CML could be a new risk factor for allergy[221] since they may react with RAGE receptors expressed at the surface of numerous immune cells (mononuclear cells, DCs and T cells) present in areas where AGEs are often more abundant[222-224].
CONCLUSION
Consequences on risks of developing non-communicable chronic diseases
Nutrition is somehow conditioning the metabolic imprinting of each individual. Recent literature clearly established that qualitative early nutrition is a prerequisite to limit the risks of developing later in life chronic diseases (Figure 1). From the first studies describing the DOHaD, several original articles have pointed out that a poor fetal and postnatal settlement of gut microbiota will necessarily remodels neuro-immuno-endocrine maturation of the gut (Figure 2) that will, in turn, modify brain neuronal connections establishment leading to a significantly higher risk of developing later in life NCCD, either at the gut of the brain level see[2] for review. However, this poor establishment of an efficient gut barrier may have other deleterious effects that will result into other NCCD affecting the endocrine function: Diabetes[224], hypertension[1] but also general behavior. Indeed, it has long been evidenced that developmental exposure to testosterone produced by fetal testis programs male sexual behavior[225-227] while excessive exposure of female fetus to this hormone during critical windows of differentiation will masculinize the female brain[228,229]. Many other factors may also be responsible of fetal and postnatal major programming modifications which will increase the risk of developing NCCD. Mother undernutrition or malnutrition, poor placental development will be at the origin of growth retardation. Compensatory growth during early life is also a risk factor for the development of adult disease in the offspring. Moreover, maternal stress, conditioning corticotropin releasing hormone and cortisol release, has been evidenced to alter irreversibly perinatal programming as well as maternal disease state especially when associated with prescription drugs such as antibiotics. Mother lifestyle: Family size, living place, presence of pets in households etc. will also leave epigenetic marks at the origin of the remodeling of the programming of the offspring and the possibility of their transmission to the upcoming generation.
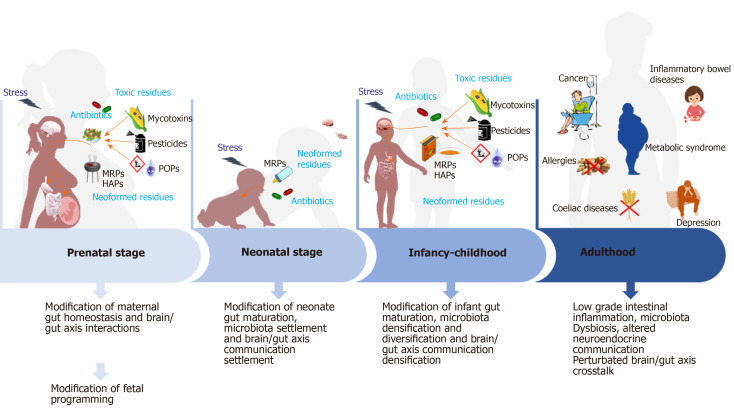
Figure 1.
Hypothesis on the impact of food contaminants on prenatal and postnatal gut homeostasis mis-programming and its consequences on the etiology of non-communicable chronic diseases. During pregnancy and infancy, repeated exposure of fetus and then infant to food contaminants is presumed to be at the origin of altered gut homeostasis due to mis settlement of neuro-immuno-endocrine cross talks and microbiota diversification and densification. Furthermore, also due to these contaminants, the brain/gut axis exchanges might not settle properly. All these modifications during the perinatal period may, later in life, predispose to the appearance of non-communicable chronic diseases among which celiac disease, allergies, inflammatory bowel disease, metabolic syndrome, depression, etc. At the bottom of the figure, are presented the main consequences on gut homeostasis observed at each stage of life. MRPs: Maillard reaction products; POPs: Persistent organic pollutants.
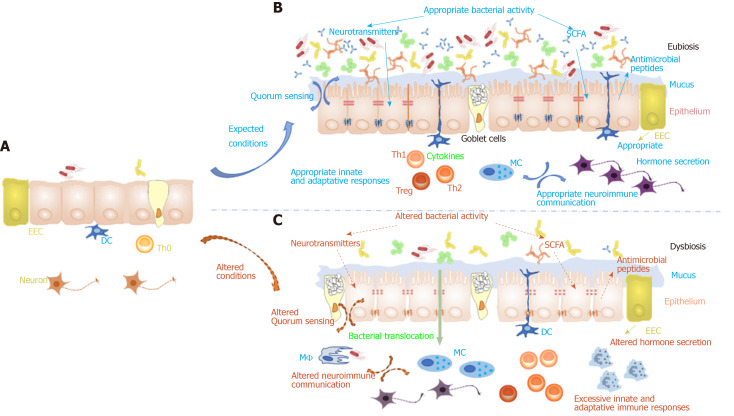
Figure 2.
Impact of food contaminants on gut homeostasis settlement and stability. A: At birth the intestinal epithelial environment is still immature with fairly no secretion, very few microorganisms and immature neuro-immuno-endocrine interactions; B: These interactions settle progressively during infancy and childhood to be fully functional at adulthood; C: However, when the conditions are not met and more especially if the child has been submitted during the fetal stage and infant stage to food contaminants, this might be at the origin of gut homeostasis misshaping observed at adulthood. This could then be responsible for gut permeability increase responsible for a mild “leaky gut”, predisposing to intestinal neuro-immuno-endocrine communications alterations. This will in turn be at the origin of systemic neuro-immuno-endocrine communication misshaping contributing to an alteration of crosstalk between the brain and the gut.
This past couple of years the qualification of some molecular epigenetics has clearly added strong insight into the effects of environmental stimuli during perinatal programming. It especially gave information on the importance of the timing of exposures to these stimuli on later human health. So far, investigation of the consequences of all these environmental factors is running on and gives interesting evidences of the mechanisms responsible for NCCD. This is the reason why we must explore the interaction of genetic and environmental components through lifetime with a special focus on early development. However, the main work to be undertaken will be to correlate all these events with each other to be able to improve recommendation to the population in terms of nutrition behaviors on the one side and lifestyle on the other side.
Footnotes
Conflict-of-interest statement: Authors declare no conflict of interests for this article.
Manuscript source: Invited manuscript
Peer-review started: January 31, 2020
First decision: May 1, 2020
Article in press: May 30, 2020
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: France
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Mortazavian AM S-Editor: Dou Y L-Editor: A E-Editor: Ma YJ
Contributor Information
Elodie Sarron, Transformations and Agroressources (EA 7519), Institut Polytechnique UniLaSalle, Université d'Artois, Beauvais 60026, France.
Maxime Pérot, Transformations and Agroressources (EA 7519), Institut Polytechnique UniLaSalle, Université d'Artois, Beauvais 60026, France.
Nicolas Barbezier, Transformations and Agroressources (EA 7519), Institut Polytechnique UniLaSalle, Université d'Artois, Beauvais 60026, France.
Carine Delayre-Orthez, Transformations and Agroressources (EA 7519), Institut Polytechnique UniLaSalle, Université d'Artois, Beauvais 60026, France.
Jérôme Gay-Quéheillard, Périnatalité et risques Toxiques, UMR-I-01, Université de Picardie Jules Verne, Amiens 80000, France.
Pauline M Anton, Transformations and Agroressources (EA 7519), Institut Polytechnique UniLaSalle, Université d'Artois, Beauvais 60026, France. pauline.anton@unilasalle.fr.
References
- 1.Barker DJ, Osmond C. Low birth weight and hypertension. BMJ. 1988;297:134–135. doi: 10.1136/bmj.297.6641.134-b. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cripps RL, Martin-Gronert MS, Ozanne SE. Fetal and perinatal programming of appetite. Clin Sci (Lond) 2005;109:1–11. doi: 10.1042/CS20040367. [DOI] [PubMed] [Google Scholar]
- 3.Sommer F, Bäckhed F. The gut microbiota--masters of host development and physiology. Nat Rev Microbiol. 2013;11:227–238. doi: 10.1038/nrmicro2974. [DOI] [PubMed] [Google Scholar]
- 4.Robertson RC, Manges AR, Finlay BB, Prendergast AJ. The Human Microbiome and Child Growth - First 1000 Days and Beyond. Trends Microbiol. 2019;27:131–147. doi: 10.1016/j.tim.2018.09.008. [DOI] [PubMed] [Google Scholar]
- 5.Romano-Keeler J, Weitkamp JH. Maternal influences on fetal microbial colonization and immune development. Pediatr Res. 2015;77:189–195. doi: 10.1038/pr.2014.163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Jiménez E, Fernández L, Marín ML, Martín R, Odriozola JM, Nueno-Palop C, Narbad A, Olivares M, Xaus J, Rodríguez JM. Isolation of commensal bacteria from umbilical cord blood of healthy neonates born by cesarean section. Curr Microbiol. 2005;51:270–274. doi: 10.1007/s00284-005-0020-3. [DOI] [PubMed] [Google Scholar]
- 7.Jiménez E, Marín ML, Martín R, Odriozola JM, Olivares M, Xaus J, Fernández L, Rodríguez JM. Is meconium from healthy newborns actually sterile? Res Microbiol. 2008;159:187–193. doi: 10.1016/j.resmic.2007.12.007. [DOI] [PubMed] [Google Scholar]
- 8.Aagaard K, Ma J, Antony KM, Ganu R, Petrosino J, Versalovic J. The placenta harbors a unique microbiome. Sci Transl Med. 2014;6:237ra65. doi: 10.1126/scitranslmed.3008599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Stout MJ, Conlon B, Landeau M, Lee I, Bower C, Zhao Q, Roehl KA, Nelson DM, Macones GA, Mysorekar IU. Identification of intracellular bacteria in the basal plate of the human placenta in term and preterm gestations. Am J Obstet Gynecol. 2013;208:226.e1–226.e7. doi: 10.1016/j.ajog.2013.01.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Grönlund MM, Lehtonen OP, Eerola E, Kero P. Fecal microflora in healthy infants born by different methods of delivery: permanent changes in intestinal flora after cesarean delivery. J Pediatr Gastroenterol Nutr. 1999;28:19–25. doi: 10.1097/00005176-199901000-00007. [DOI] [PubMed] [Google Scholar]
- 11.Biasucci G, Benenati B, Morelli L, Bessi E, Boehm G. Cesarean delivery may affect the early biodiversity of intestinal bacteria. J Nutr. 2008;138:1796S–1800S. doi: 10.1093/jn/138.9.1796S. [DOI] [PubMed] [Google Scholar]
- 12.Arboleya S, Sánchez B, Solís G, Fernández N, Suárez M, Hernández-Barranco AM, Milani C, Margolles A, de Los Reyes-Gavilán CG, Ventura M, Gueimonde M. Impact of Prematurity and Perinatal Antibiotics on the Developing Intestinal Microbiota: A Functional Inference Study. Int J Mol Sci. 2016:17. doi: 10.3390/ijms17050649. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Itani T, Ayoub Moubareck C, Melki I, Rousseau C, Mangin I, Butel MJ, Karam Sarkis D. Establishment and development of the intestinal microbiota of preterm infants in a Lebanese tertiary hospital. Anaerobe. 2017;43:4–14. doi: 10.1016/j.anaerobe.2016.11.001. [DOI] [PubMed] [Google Scholar]
- 14.Martin R, Nauta AJ, Ben Amor K, Knippels LM, Knol J, Garssen J. Early life: gut microbiota and immune development in infancy. Benef Microbes. 2010;1:367–382. doi: 10.3920/BM2010.0027. [DOI] [PubMed] [Google Scholar]
- 15.Arrieta MC, Stiemsma LT, Amenyogbe N, Brown EM, Finlay B. The intestinal microbiome in early life: health and disease. Front Immunol. 2014;5:427. doi: 10.3389/fimmu.2014.00427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sekirov I, Russell SL, Antunes LC, Finlay BB. Gut microbiota in health and disease. Physiol Rev. 2010;90:859–904. doi: 10.1152/physrev.00045.2009. [DOI] [PubMed] [Google Scholar]
- 17.Prakash S, Rodes L, Coussa-Charley M, Tomaro-Duchesneau C. Gut microbiota: next frontier in understanding human health and development of biotherapeutics. Biologics. 2011;5:71–86. doi: 10.2147/BTT.S19099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Dogra S, Sakwinska O, Soh SE, Ngom-Bru C, Brück WM, Berger B, Brüssow H, Karnani N, Lee YS, Yap F, Chong YS, Godfrey KM, Holbrook JD. Rate of establishing the gut microbiota in infancy has consequences for future health. Gut Microbes. 2015;6:321–325. doi: 10.1080/19490976.2015.1078051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Clemente JC, Ursell LK, Parfrey LW, Knight R. The impact of the gut microbiota on human health: an integrative view. Cell. 2012;148:1258–1270. doi: 10.1016/j.cell.2012.01.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Neu J, Rushing J. Cesarean versus vaginal delivery: long-term infant outcomes and the hygiene hypothesis. Clin Perinatol. 2011;38:321–331. doi: 10.1016/j.clp.2011.03.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ling Z, Li Z, Liu X, Cheng Y, Luo Y, Tong X, Yuan L, Wang Y, Sun J, Li L, Xiang C. Altered fecal microbiota composition associated with food allergy in infants. Appl Environ Microbiol. 2014;80:2546–2554. doi: 10.1128/AEM.00003-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Negele K, Heinrich J, Borte M, von Berg A, Schaaf B, Lehmann I, Wichmann HE, Bolte G LISA Study Group. Mode of delivery and development of atopic disease during the first 2 years of life. Pediatr Allergy Immunol. 2004;15:48–54. doi: 10.1046/j.0905-6157.2003.00101.x. [DOI] [PubMed] [Google Scholar]
- 23.Strachan DP. Hay fever, hygiene, and household size. BMJ. 1989;299:1259–1260. doi: 10.1136/bmj.299.6710.1259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Alexandre-Silva GM, Brito-Souza PA, Oliveira ACS, Cerni FA, Zottich U, Pucca MB. The hygiene hypothesis at a glance: Early exposures, immune mechanism and novel therapies. Acta Trop. 2018;188:16–26. doi: 10.1016/j.actatropica.2018.08.032. [DOI] [PubMed] [Google Scholar]
- 25.Grier A, Qiu X, Bandyopadhyay S, Holden-Wiltse J, Kessler HA, Gill AL, Hamilton B, Huyck H, Misra S, Mariani TJ, Ryan RM, Scholer L, Scheible KM, Lee YH, Caserta MT, Pryhuber GS, Gill SR. Impact of prematurity and nutrition on the developing gut microbiome and preterm infant growth. Microbiome. 2017;5:158. doi: 10.1186/s40168-017-0377-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Korpela K, Blakstad EW, Moltu SJ, Strømmen K, Nakstad B, Rønnestad AE, Brække K, Iversen PO, Drevon CA, de Vos W. Intestinal microbiota development and gestational age in preterm neonates. Sci Rep. 2018;8:2453. doi: 10.1038/s41598-018-20827-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.van de Pavert SA, Mebius RE. New insights into the development of lymphoid tissues. Nat Rev Immunol. 2010;10:664–674. doi: 10.1038/nri2832. [DOI] [PubMed] [Google Scholar]
- 28.Weitkamp JH, Rudzinski E, Koyama T, Correa H, Matta P, Alberty B, Polk DB. Ontogeny of FOXP3(+) regulatory T cells in the postnatal human small intestinal and large intestinal lamina propria. Pediatr Dev Pathol. 2009;12:443–449. doi: 10.2350/08-09-0533.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Leibnitz R. In: Holladay S, editor. Development of the human immune system. In: Holladay S. Developmental Immunotoxicology. Boca Raton, FL: CRC Press, 2005: 21-44. [Google Scholar]
- 30.Mold J, McCune J. Immunological tolerance during fetal development: from mouse to man. Adv Immunol. 2012;115:73–111. doi: 10.1016/B978-0-12-394299-9.00003-5. [DOI] [PubMed] [Google Scholar]
- 31.Sawa S, Cherrier M, Lochner M, Satoh-Takayama N, Fehling HJ, Langa F, Di Santo JP, Eberl G. Lineage relationship analysis of RORgammat+ innate lymphoid cells. Science. 2010;330:665–669. doi: 10.1126/science.1194597. [DOI] [PubMed] [Google Scholar]
- 32.Round JL, Lee SM, Li J, Tran G, Jabri B, Chatila TA, Mazmanian SK. The Toll-like receptor 2 pathway establishes colonization by a commensal of the human microbiota. Science. 2011;332:974–977. doi: 10.1126/science.1206095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Michaëlsson J, Mold JE, McCune JM, Nixon DF. Regulation of T cell responses in the developing human fetus. J Immunol. 2006;176:5741–5748. doi: 10.4049/jimmunol.176.10.5741. [DOI] [PubMed] [Google Scholar]
- 34.Grindebacke H, Stenstad H, Quiding-Järbrink M, Waldenström J, Adlerberth I, Wold AE, Rudin A. Dynamic development of homing receptor expression and memory cell differentiation of infant CD4+CD25high regulatory T cells. J Immunol. 2009;183:4360–4370. doi: 10.4049/jimmunol.0901091. [DOI] [PubMed] [Google Scholar]
- 35.O'Mahony C, Scully P, O'Mahony D, Murphy S, O'Brien F, Lyons A, Sherlock G, MacSharry J, Kiely B, Shanahan F, O'Mahony L. Commensal-induced regulatory T cells mediate protection against pathogen-stimulated NF-kappaB activation. PLoS Pathog. 2008;4:e1000112. doi: 10.1371/journal.ppat.1000112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hooper LV, Wong MH, Thelin A, Hansson L, Falk PG, Gordon JI. Molecular analysis of commensal host-microbial relationships in the intestine. Science. 2001;291:881–884. doi: 10.1126/science.291.5505.881. [DOI] [PubMed] [Google Scholar]
- 37.Zeuthen LH, Fink LN, Frokiaer H. Epithelial cells prime the immune response to an array of gut-derived commensals towards a tolerogenic phenotype through distinct actions of thymic stromal lymphopoietin and transforming growth factor-beta. Immunology. 2008;123:197–208. doi: 10.1111/j.1365-2567.2007.02687.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Granucci F, Zanoni I. Role of Toll like receptor-activated dendritic cells in the development of autoimmunity. Front Biosci. 2008;13:4817–4826. doi: 10.2741/3041. [DOI] [PubMed] [Google Scholar]
- 39.Rutella S, Locatelli F. Intestinal dendritic cells in the pathogenesis of inflammatory bowel disease. World J Gastroenterol. 2011;17:3761–3775. doi: 10.3748/wjg.v17.i33.3761. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Silverman MN, Pearce BD, Biron CA, Miller AH. Immune modulation of the hypothalamic-pituitary-adrenal (HPA) axis during viral infection. Viral Immunol. 2005;18:41–78. doi: 10.1089/vim.2005.18.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Furness JB, Rivera LR, Cho HJ, Bravo DM, Callaghan B. The gut as a sensory organ. Nat Rev Gastroenterol Hepatol. 2013;10:729–740. doi: 10.1038/nrgastro.2013.180. [DOI] [PubMed] [Google Scholar]
- 42.Ruddick JP, Evans AK, Nutt DJ, Lightman SL, Rook GA, Lowry CA. Tryptophan metabolism in the central nervous system: medical implications. Expert Rev Mol Med. 2006;8:1–27. doi: 10.1017/S1462399406000068. [DOI] [PubMed] [Google Scholar]
- 43.Bale TL, Baram TZ, Brown AS, Goldstein JM, Insel TR, McCarthy MM, Nemeroff CB, Reyes TM, Simerly RB, Susser ES, Nestler EJ. Early life programming and neurodevelopmental disorders. Biol Psychiatry. 2010;68:314–319. doi: 10.1016/j.biopsych.2010.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Finegold SM, Molitoris D, Song Y, Liu C, Vaisanen ML, Bolte E, McTeague M, Sandler R, Wexler H, Marlowe EM, Collins MD, Lawson PA, Summanen P, Baysallar M, Tomzynski TJ, Read E, Johnson E, Rolfe R, Nasir P, Shah H, Haake DA, Manning P, Kaul A. Gastrointestinal microflora studies in late-onset autism. Clin Infect Dis. 2002;35:S6–S16. doi: 10.1086/341914. [DOI] [PubMed] [Google Scholar]
- 45.Mittal VA, Ellman LM, Cannon TD. Gene-environment interaction and covariation in schizophrenia: the role of obstetric complications. Schizophr Bull. 2008;34:1083–1094. doi: 10.1093/schbul/sbn080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Goehler LE, Park SM, Opitz N, Lyte M, Gaykema RP. Campylobacter jejuni infection increases anxiety-like behavior in the holeboard: possible anatomical substrates for viscerosensory modulation of exploratory behavior. Brain Behav Immun. 2008;22:354–366. doi: 10.1016/j.bbi.2007.08.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Sullivan R, Wilson DA, Feldon J, Yee BK, Meyer U, Richter-Levin G, Avi A, Michael T, Gruss M, Bock J, Helmeke C, Braun K. The International Society for Developmental Psychobiology annual meeting symposium: Impact of early life experiences on brain and behavioral development. Dev Psychobiol. 2006;48:583–602. doi: 10.1002/dev.20170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Satokari R, Grönroos T, Laitinen K, Salminen S, Isolauri E. Bifidobacterium and Lactobacillus DNA in the human placenta. Lett Appl Microbiol. 2009;48:8–12. doi: 10.1111/j.1472-765X.2008.02475.x. [DOI] [PubMed] [Google Scholar]
- 49.Fu J, Bonder MJ, Cenit MC, Tigchelaar EF, Maatman A, Dekens JA, Brandsma E, Marczynska J, Imhann F, Weersma RK, Franke L, Poon TW, Xavier RJ, Gevers D, Hofker MH, Wijmenga C, Zhernakova A. The Gut Microbiome Contributes to a Substantial Proportion of the Variation in Blood Lipids. Circ Res. 2015;117:817–824. doi: 10.1161/CIRCRESAHA.115.306807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Mazer C, Muneyyirci J, Taheny K, Raio N, Borella A, Whitaker-Azmitia P. Serotonin depletion during synaptogenesis leads to decreased synaptic density and learning deficits in the adult rat: a possible model of neurodevelopmental disorders with cognitive deficits. Brain Res. 1997;760:68–73. doi: 10.1016/s0006-8993(97)00297-7. [DOI] [PubMed] [Google Scholar]
- 51.Côté F, Fligny C, Bayard E, Launay JM, Gershon MD, Mallet J, Vodjdani G. Maternal serotonin is crucial for murine embryonic development. Proc Natl Acad Sci USA. 2007;104:329–334. doi: 10.1073/pnas.0606722104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Herschkowitz N, Kagan J, Zilles K. Neurobiological bases of behavioral development in the first year. Neuropediatrics. 1997;28:296–306. doi: 10.1055/s-2007-973720. [DOI] [PubMed] [Google Scholar]
- 53.Manco M. Gut microbiota and developmental programming of the brain: from evidence in behavioral endophenotypes to novel perspective in obesity. Front Cell Infect Microbiol. 2012;2:109. doi: 10.3389/fcimb.2012.00109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Rothman SM, Griffioen KJ, Wan R, Mattson MP. Brain-derived neurotrophic factor as a regulator of systemic and brain energy metabolism and cardiovascular health. Ann N Y Acad Sci. 2012;1264:49–63. doi: 10.1111/j.1749-6632.2012.06525.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Zola SM, Squire LR, Teng E, Stefanacci L, Buffalo EA, Clark RE. Impaired recognition memory in monkeys after damage limited to the hippocampal region. J Neurosci. 2000;20:451–463. doi: 10.1523/JNEUROSCI.20-01-00451.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Karmarkar D, Rock KL. Microbiota signalling through MyD88 is necessary for a systemic neutrophilic inflammatory response. Immunology. 2013;140:483–492. doi: 10.1111/imm.12159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Abt MC, Osborne LC, Monticelli LA, Doering TA, Alenghat T, Sonnenberg GF, Paley MA, Antenus M, Williams KL, Erikson J, Wherry EJ, Artis D. Commensal bacteria calibrate the activation threshold of innate antiviral immunity. Immunity. 2012;37:158–170. doi: 10.1016/j.immuni.2012.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Arpaia N, Campbell C, Fan X, Dikiy S, van der Veeken J, deRoos P, Liu H, Cross JR, Pfeffer K, Coffer PJ, Rudensky AY. Metabolites produced by commensal bacteria promote peripheral regulatory T-cell generation. Nature. 2013;504:451–455. doi: 10.1038/nature12726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Artis D. Epithelial-cell recognition of commensal bacteria and maintenance of immune homeostasis in the gut. Nat Rev Immunol. 2008;8:411–420. doi: 10.1038/nri2316. [DOI] [PubMed] [Google Scholar]
- 60.Kaplan JL, Shi HN, Walker WA. The role of microbes in developmental immunologic programming. Pediatr Res. 2011;69:465–472. doi: 10.1203/PDR.0b013e318217638a. [DOI] [PubMed] [Google Scholar]
- 61.Clarke G, O'Mahony SM, Dinan TG, Cryan JF. Priming for health: gut microbiota acquired in early life regulates physiology, brain and behaviour. Acta Paediatr. 2014;103:812–819. doi: 10.1111/apa.12674. [DOI] [PubMed] [Google Scholar]
- 62.Goulet O. Potential role of the intestinal microbiota in programming health and disease. Nutr Rev. 2015;73 Suppl 1:32–40. doi: 10.1093/nutrit/nuv039. [DOI] [PubMed] [Google Scholar]
- 63.Vuong HE, Hsiao EY. Emerging Roles for the Gut Microbiome in Autism Spectrum Disorder. Biol Psychiatry. 2017;81:411–423. doi: 10.1016/j.biopsych.2016.08.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Cryan JF, Dinan TG. Mind-altering microorganisms: the impact of the gut microbiota on brain and behaviour. Nat Rev Neurosci. 2012;13:701–712. doi: 10.1038/nrn3346. [DOI] [PubMed] [Google Scholar]
- 65.Sampson TR, Debelius JW, Thron T, Janssen S, Shastri GG, Ilhan ZE, Challis C, Schretter CE, Rocha S, Gradinaru V, Chesselet MF, Keshavarzian A, Shannon KM, Krajmalnik-Brown R, Wittung-Stafshede P, Knight R, Mazmanian SK. Gut Microbiota Regulate Motor Deficits and Neuroinflammation in a Model of Parkinson's Disease. Cell. 2016;167:1469–1480.e12. doi: 10.1016/j.cell.2016.11.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Amaral FA, Sachs D, Costa VV, Fagundes CT, Cisalpino D, Cunha TM, Ferreira SH, Cunha FQ, Silva TA, Nicoli JR, Vieira LQ, Souza DG, Teixeira MM. Commensal microbiota is fundamental for the development of inflammatory pain. Proc Natl Acad Sci USA. 2008;105:2193–2197. doi: 10.1073/pnas.0711891105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Barouki R, Gluckman PD, Grandjean P, Hanson M, Heindel JJ. Developmental origins of non-communicable disease: implications for research and public health. Environ Health. 2012;11:42. doi: 10.1186/1476-069X-11-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Vickers MH. Early life nutrition, epigenetics and programming of later life disease. Nutrients. 2014;6:2165–2178. doi: 10.3390/nu6062165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Godfrey KM, Lillycrop KA, Burdge GC, Gluckman PD, Hanson MA. Epigenetic mechanisms and the mismatch concept of the developmental origins of health and disease. Pediatr Res. 2007;61:5R–10R. doi: 10.1203/pdr.0b013e318045bedb. [DOI] [PubMed] [Google Scholar]
- 70.Lane RH. Fetal programming, epigenetics, and adult onset disease. Clin Perinatol. 2014;41:815–831. doi: 10.1016/j.clp.2014.08.006. [DOI] [PubMed] [Google Scholar]
- 71.Jirtle RL, Skinner MK. Environmental epigenomics and disease susceptibility. Nat Rev Genet. 2007;8:253–262. doi: 10.1038/nrg2045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Papadopoulou E, Haug LS, Sakhi AK, Andrusaityte S, Basagaña X, Brantsaeter AL, Casas M, Fernández-Barrés S, Grazuleviciene R, Knutsen HK, Maitre L, Meltzer HM, McEachan RRC, Roumeliotaki T, Slama R, Vafeiadi M, Wright J, Vrijheid M, Thomsen C, Chatzi L. Diet as a Source of Exposure to Environmental Contaminants for Pregnant Women and Children from Six European Countries. Environ Health Perspect. 2019;127:107005. doi: 10.1289/EHP5324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Schecter A, Colacino J, Haffner D, Patel K, Opel M, Päpke O, Birnbaum L. Perfluorinated compounds, polychlorinated biphenyls, and organochlorine pesticide contamination in composite food samples from Dallas, Texas, USA. Environ Health Perspect. 2010;118:796–802. doi: 10.1289/ehp.0901347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Valeri L, Mazumdar MM, Bobb JF, Claus Henn B, Rodrigues E, Sharif OIA, Kile ML, Quamruzzaman Q, Afroz S, Golam M, Amarasiriwardena C, Bellinger DC, Christiani DC, Coull BA, Wright RO. The Joint Effect of Prenatal Exposure to Metal Mixtures on Neurodevelopmental Outcomes at 20-40 Months of Age: Evidence from Rural Bangladesh. Environ Health Perspect. 2017;125:067015. doi: 10.1289/EHP614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Liew WP, Mohd-Redzwan S. Mycotoxin: Its Impact on Gut Health and Microbiota. Front Cell Infect Microbiol. 2018;8:60. doi: 10.3389/fcimb.2018.00060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ravichandran G, Lakshmanan DK, Raju K, Elangovan A, Nambirajan G, Devanesan AA, Thilagar S. Food advanced glycation end products as potential endocrine disruptors: An emerging threat to contemporary and future generation. Environ Int. 2019;123:486–500. doi: 10.1016/j.envint.2018.12.032. [DOI] [PubMed] [Google Scholar]
- 77.Jašarević E, Rodgers AB, Bale TL. A novel role for maternal stress and microbial transmission in early life programming and neurodevelopment. Neurobiol Stress. 2015;1:81–88. doi: 10.1016/j.ynstr.2014.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Zerbo O, Qian Y, Yoshida C, Grether JK, Van de Water J, Croen LA. Maternal Infection During Pregnancy and Autism Spectrum Disorders. J Autism Dev Disord. 2015;45:4015–4025. doi: 10.1007/s10803-013-2016-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Tanaka M, Nakayama J. Development of the gut microbiota in infancy and its impact on health in later life. Allergol Int. 2017;66:515–522. doi: 10.1016/j.alit.2017.07.010. [DOI] [PubMed] [Google Scholar]
- 80.Keski-Nisula L, Kyynäräinen HR, Kärkkäinen U, Karhukorpi J, Heinonen S, Pekkanen J. Maternal intrapartum antibiotics and decreased vertical transmission of Lactobacillus to neonates during birth. Acta Paediatr. 2013;102:480–485. doi: 10.1111/apa.12186. [DOI] [PubMed] [Google Scholar]
- 81.Fallani M, Young D, Scott J, Norin E, Amarri S, Adam R, Aguilera M, Khanna S, Gil A, Edwards CA, Doré J Other Members of the INFABIO Team. Intestinal microbiota of 6-week-old infants across Europe: geographic influence beyond delivery mode, breast-feeding, and antibiotics. J Pediatr Gastroenterol Nutr. 2010;51:77–84. doi: 10.1097/MPG.0b013e3181d1b11e. [DOI] [PubMed] [Google Scholar]
- 82.Cox LM, Blaser MJ. Pathways in microbe-induced obesity. Cell Metab. 2013;17:883–894. doi: 10.1016/j.cmet.2013.05.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Cox LM, Yamanishi S, Sohn J, Alekseyenko AV, Leung JM, Cho I, Kim SG, Li H, Gao Z, Mahana D, Zárate Rodriguez JG, Rogers AB, Robine N, Loke P, Blaser MJ. Altering the intestinal microbiota during a critical developmental window has lasting metabolic consequences. Cell. 2014;158:705–721. doi: 10.1016/j.cell.2014.05.052. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Joly Condette C, Bach V, Mayeur C, Gay-Quéheillard J, Khorsi-Cauet H. Chlorpyrifos Exposure During Perinatal Period Affects Intestinal Microbiota Associated With Delay of Maturation of Digestive Tract in Rats. J Pediatr Gastroenterol Nutr. 2015;61:30–40. doi: 10.1097/MPG.0000000000000734. [DOI] [PubMed] [Google Scholar]
- 85.Spyker JM, Avery DL. Neurobehavioral effects of prenatal exposure to the organophosphate Diazinon in mice. J Toxicol Environ Health. 1977;3:989–1002. doi: 10.1080/15287397709529633. [DOI] [PubMed] [Google Scholar]
- 86.Gupta RC. Carbofuran toxicity. J Toxicol Environ Health. 1994;43:383–418. doi: 10.1080/15287399409531931. [DOI] [PubMed] [Google Scholar]
- 87.Bolles HG, Dixon-White HE, Peterson RK, Tomerlin JR, Day EW Jr, Oliver GR. U.S. market basket study to determine residues of the insecticide chlorpyrifos. J Agric Food Chem. 1999;47:1817–1822. doi: 10.1021/jf980962d. [DOI] [PubMed] [Google Scholar]
- 88.Joly Condette C, Khorsi-Cauet H, Morlière P, Zabijak L, Reygner J, Bach V, Gay-Quéheillard J. Increased gut permeability and bacterial translocation after chronic chlorpyrifos exposure in rats. PLoS One. 2014;9:e102217. doi: 10.1371/journal.pone.0102217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Reygner J, Lichtenberger L, Elmhiri G, Dou S, Bahi-Jaber N, Rhazi L, Depeint F, Bach V, Khorsi-Cauet H, Abdennebi-Najar L. Inulin Supplementation Lowered the Metabolic Defects of Prolonged Exposure to Chlorpyrifos from Gestation to Young Adult Stage in Offspring Rats. PLoS One. 2016;11:e0164614. doi: 10.1371/journal.pone.0164614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Laporte B, Gay-Quéheillard J, Bach V, Villégier AS. Developmental neurotoxicity in the progeny after maternal gavage with chlorpyrifos. Food Chem Toxicol. 2018;113:66–72. doi: 10.1016/j.fct.2018.01.026. [DOI] [PubMed] [Google Scholar]
- 91.Ribeiro TA, Prates KV, Pavanello A, Malta A, Tófolo LP, Martins IP, Oliveira JC, Miranda RA, Gomes RM, Vieira E, Franco CC, Barella LF, Francisco FA, Alves VS, Silveira SD, Moreira VM, Fabricio GS, Palma-Rigo K, Sloboda DM, Mathias PC. Acephate exposure during a perinatal life program to type 2 diabetes. Toxicology. 2016;372:12–21. doi: 10.1016/j.tox.2016.10.010. [DOI] [PubMed] [Google Scholar]
- 92.Yamazaki K, Araki A, Nakajima S, Miyashita C, Ikeno T, Itoh S, Minatoya M, Kobayashi S, Mizutani F, Chisaki Y, Kishi R. Association between prenatal exposure to organochlorine pesticides and the mental and psychomotor development of infants at ages 6 and 18 months: The Hokkaido Study on Environment and Children's Health. Neurotoxicology. 2018;69:201–208. doi: 10.1016/j.neuro.2017.11.011. [DOI] [PubMed] [Google Scholar]
- 93.Boucher O, Simard MN, Muckle G, Rouget F, Kadhel P, Bataille H, Chajès V, Dallaire R, Monfort C, Thomé JP, Multigner L, Cordier S. Exposure to an organochlorine pesticide (chlordecone) and development of 18-month-old infants. Neurotoxicology. 2013;35:162–168. doi: 10.1016/j.neuro.2013.01.007. [DOI] [PubMed] [Google Scholar]
- 94.Hervé D, Costet N, Kadhel P, Rouget F, Monfort C, Thomé JP, Multigner L, Cordier S. Prenatal exposure to chlordecone, gestational weight gain, and birth weight in a Guadeloupean birth cohort. Environ Res. 2016;151:436–444. doi: 10.1016/j.envres.2016.08.004. [DOI] [PubMed] [Google Scholar]
- 95.Khanjani N, Sim MR. Maternal contamination with dichlorodiphenyltrichloroethane and reproductive outcomes in an Australian population. Environ Res. 2006;101:373–379. doi: 10.1016/j.envres.2005.10.003. [DOI] [PubMed] [Google Scholar]
- 96.Weisskopf MG, Anderson HA, Hanrahan LP, Kanarek MS, Falk CM, Steenport DM, Draheim LA Great Lakes Consortium. Maternal exposure to Great Lakes sport-caught fish and dichlorodiphenyl dichloroethylene, but not polychlorinated biphenyls, is associated with reduced birth weight. Environ Res. 2005;97:149–162. doi: 10.1016/j.envres.2004.01.014. [DOI] [PubMed] [Google Scholar]
- 97.Kim JC, Shin JY, Yang YS, Shin DH, Moon CJ, Kim SH, Park SC, Kim YB, Kim HC, Chung MK. Evaluation of developmental toxicity of amitraz in Sprague-Dawley rats. Arch Environ Contam Toxicol. 2007;52:137–144. doi: 10.1007/s00244-006-0021-7. [DOI] [PubMed] [Google Scholar]
- 98.Domingues A, Grassi TF, Spinardi-Barbisan AL, Barbisan LF. Developmental exposure to diuron causes splenotoxicity in male Sprague-Dawley rat pups. J Environ Sci Health B. 2012;47:420–426. doi: 10.1080/03601234.2012.657054. [DOI] [PubMed] [Google Scholar]
- 99.Piacentini KC, Ferranti LS, Pinheiro M, Bertozzi BG, Rocha LO. Mycotoxin contamination in cereal-based baby foods. Curr Opin Food Sci. 2019;30:73–78. [Google Scholar]
- 100.Wild CP, Rasheed FN, Jawla MF, Hall AJ, Jansen LA, Montesano R. In-utero exposure to aflatoxin in west Africa. Lancet. 1991;337:1602. doi: 10.1016/0140-6736(91)93295-k. [DOI] [PubMed] [Google Scholar]
- 101.Woo CS, El-Nezami H. Maternal-Fetal Cancer Risk Assessment of Ochratoxin A during Pregnancy. Toxins (Basel) 2016;8:87. doi: 10.3390/toxins8040087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Kristensen P, Irgens LM, Andersen A, Bye AS, Sundheim L. Gestational age, birth weight, and perinatal death among births to Norwegian farmers, 1967-1991. Am J Epidemiol. 1997;146:329–338. doi: 10.1093/oxfordjournals.aje.a009274. [DOI] [PubMed] [Google Scholar]
- 103.Smith LE, Prendergast AJ, Turner PC, Humphrey JH, Stoltzfus RJ. Aflatoxin Exposure During Pregnancy, Maternal Anemia, and Adverse Birth Outcomes. Am J Trop Med Hyg. 2017;96:770–776. doi: 10.4269/ajtmh.16-0730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Hernandez-Vargas H, Castelino J, Silver MJ, Dominguez-Salas P, Cros MP, Durand G, Le Calvez-Kelm F, Prentice AM, Wild CP, Moore SE, Hennig BJ, Herceg Z, Gong YY, Routledge MN. Exposure to aflatoxin B1 in utero is associated with DNA methylation in white blood cells of infants in The Gambia. Int J Epidemiol. 2015;44:1238–1248. doi: 10.1093/ije/dyv027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Demirel G, Alpertunga B, Ozden S. Role of fumonisin B1 on DNA methylation changes in rat kidney and liver cells. Pharm Biol. 2015;53:1302–1310. doi: 10.3109/13880209.2014.976714. [DOI] [PubMed] [Google Scholar]
- 106.Wangikar PB, Dwivedi P, Sinha N. Effect in rats of simultaneous prenatal exposure to ochratoxin A and aflatoxin B1. I. Maternal toxicity and fetal malformations. Birth Defects Res B Dev Reprod Toxicol. 2004;71:343–351. doi: 10.1002/bdrb.20021. [DOI] [PubMed] [Google Scholar]
- 107.Arora RG, Appelgren LE, Bergman A. Distribution of [14C]-labelled aflatoxin B1 in mice. Acta Pharmacol Toxicol (Copenh) 1978;43:273–279. doi: 10.1111/j.1600-0773.1978.tb02265.x. [DOI] [PubMed] [Google Scholar]
- 108.Collins TF, Sprando RL, Black TN, Olejnik N, Eppley RM, Alam HZ, Rorie J, Ruggles DI. Effects of zearalenone on in utero development in rats. Food Chem Toxicol. 2006;44:1455–1465. doi: 10.1016/j.fct.2006.04.015. [DOI] [PubMed] [Google Scholar]
- 109.Menard S, Guzylack-Piriou L, Leveque M, Braniste V, Lencina C, Naturel M, Moussa L, Sekkal S, Harkat C, Gaultier E, Theodorou V, Houdeau E. Food intolerance at adulthood after perinatal exposure to the endocrine disruptor bisphenol A. FASEB J. 2014;28:4893–4900. doi: 10.1096/fj.14-255380. [DOI] [PubMed] [Google Scholar]
- 110.Fromme H, Mosch C, Morovitz M, Alba-Alejandre I, Boehmer S, Kiranoglu M, Faber F, Hannibal I, Genzel-Boroviczény O, Koletzko B, Völkel W. Pre- and postnatal exposure to perfluorinated compounds (PFCs) Environ Sci Technol. 2010;44:7123–7129. doi: 10.1021/es101184f. [DOI] [PubMed] [Google Scholar]
- 111.Dietert RR, Dietert JM. Potential for early-life immune insult including developmental immunotoxicity in autism and autism spectrum disorders: focus on critical windows of immune vulnerability. J Toxicol Environ Health B Crit Rev. 2008;11:660–680. doi: 10.1080/10937400802370923. [DOI] [PubMed] [Google Scholar]
- 112.Park HY, Hertz-Picciotto I, Sovcikova E, Kocan A, Drobna B, Trnovec T. Neurodevelopmental toxicity of prenatal polychlorinated biphenyls (PCBs) by chemical structure and activity: a birth cohort study. Environ Health. 2010;9:51. doi: 10.1186/1476-069X-9-51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Ribas-Fitó N, Sala M, Kogevinas M, Sunyer J. Polychlorinated biphenyls (PCBs) and neurological development in children: a systematic review. J Epidemiol Community Health. 2001;55:537–546. doi: 10.1136/jech.55.8.537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Walkowiak J, Wiener JA, Fastabend A, Heinzow B, Krämer U, Schmidt E, Steingrüber HJ, Wundram S, Winneke G. Environmental exposure to polychlorinated biphenyls and quality of the home environment: effects on psychodevelopment in early childhood. Lancet. 2001;358:1602–1607. doi: 10.1016/S0140-6736(01)06654-5. [DOI] [PubMed] [Google Scholar]
- 115.Claus SP, Guillou H, Ellero-Simatos S. The gut microbiota: a major player in the toxicity of environmental pollutants? NPJ Biofilms Microbiomes. 2016;2:16003. doi: 10.1038/npjbiofilms.2016.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Clemens S, Ma JF. Toxic Heavy Metal and Metalloid Accumulation in Crop Plants and Foods. Annu Rev Plant Biol. 2016;67:489–512. doi: 10.1146/annurev-arplant-043015-112301. [DOI] [PubMed] [Google Scholar]
- 117.Castoldi AF, Coccini T, Manzo L. Neurotoxic and molecular effects of methylmercury in humans. Rev Environ Health. 2003;18:19–31. doi: 10.1515/reveh.2003.18.1.19. [DOI] [PubMed] [Google Scholar]
- 118.Cambier S, Fujimura M, Bourdineaud JP. A likely placental barrier against methylmercury in pregnant rats exposed to fish-containing diets. Food Chem Toxicol. 2018;122:11–20. doi: 10.1016/j.fct.2018.09.066. [DOI] [PubMed] [Google Scholar]
- 119.Sakamoto M, Kakita A, Wakabayashi K, Takahashi H, Nakano A, Akagi H. Evaluation of changes in methylmercury accumulation in the developing rat brain and its effects: a study with consecutive and moderate dose exposure throughout gestation and lactation periods. Brain Res. 2002;949:51–59. doi: 10.1016/s0006-8993(02)02964-5. [DOI] [PubMed] [Google Scholar]
- 120.Gilbert SG, Grant-Webster KS. Neurobehavioral effects of developmental methylmercury exposure. Environ Health Perspect. 1995;103 Suppl 6:135–142. doi: 10.1289/ehp.95103s6135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Shah-Kulkarni S, Lee S, Jeong KS, Hong YC, Park H, Ha M, Kim Y, Ha EH. Prenatal exposure to mixtures of heavy metals and neurodevelopment in infants at 6 months. Environ Res. 2020;182:109122. doi: 10.1016/j.envres.2020.109122. [DOI] [PubMed] [Google Scholar]
- 122.Woods MM, Lanphear BP, Braun JM, McCandless LC. Gestational exposure to endocrine disrupting chemicals in relation to infant birth weight: a Bayesian analysis of the HOME Study. Environ Health. 2017;16:115. doi: 10.1186/s12940-017-0332-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Järup L, Akesson A. Current status of cadmium as an environmental health problem. Toxicol Appl Pharmacol. 2009;238:201–208. doi: 10.1016/j.taap.2009.04.020. [DOI] [PubMed] [Google Scholar]
- 124.Carlin DJ, Naujokas MF, Bradham KD, Cowden J, Heacock M, Henry HF, Lee JS, Thomas DJ, Thompson C, Tokar EJ, Waalkes MP, Birnbaum LS, Suk WA. Arsenic and Environmental Health: State of the Science and Future Research Opportunities. Environ Health Perspect. 2016;124:890–899. doi: 10.1289/ehp.1510209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Rager JE, Bailey KA, Smeester L, Miller SK, Parker JS, Laine JE, Drobná Z, Currier J, Douillet C, Olshan AF, Rubio-Andrade M, Stýblo M, García-Vargas G, Fry RC. Prenatal arsenic exposure and the epigenome: altered microRNAs associated with innate and adaptive immune signaling in newborn cord blood. Environ Mol Mutagen. 2014;55:196–208. doi: 10.1002/em.21842. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Grandjean P, Weihe P, White RF, Debes F, Araki S, Yokoyama K, Murata K, Sørensen N, Dahl R, Jørgensen PJ. Cognitive deficit in 7-year-old children with prenatal exposure to methylmercury. Neurotoxicol Teratol. 1997;19:417–428. doi: 10.1016/s0892-0362(97)00097-4. [DOI] [PubMed] [Google Scholar]
- 127.Yazbeck C, Thiebaugeorges O, Moreau T, Goua V, Debotte G, Sahuquillo J, Forhan A, Foliguet B, Magnin G, Slama R, Charles MA, Huel G. Maternal blood lead levels and the risk of pregnancy-induced hypertension: the EDEN cohort study. Environ Health Perspect. 2009;117:1526–1530. doi: 10.1289/ehp.0800488. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Miranda ML, Edwards S, Maxson PJ. Mercury levels in an urban pregnant population in Durham County, North Carolina. Int J Environ Res Public Health. 2011;8:698–712. doi: 10.3390/ijerph8030698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Rahman A, Vahter M, Ekström EC, Rahman M, Golam Mustafa AH, Wahed MA, Yunus M, Persson LA. Association of arsenic exposure during pregnancy with fetal loss and infant death: a cohort study in Bangladesh. Am J Epidemiol. 2007;165:1389–1396. doi: 10.1093/aje/kwm025. [DOI] [PubMed] [Google Scholar]
- 130.Vigeh M, Yokoyama K, Seyedaghamiri Z, Shinohara A, Matsukawa T, Chiba M, Yunesian M. Blood lead at currently acceptable levels may cause preterm labour. Occup Environ Med. 2011;68:231–234. doi: 10.1136/oem.2009.050419. [DOI] [PubMed] [Google Scholar]
- 131.Chan-Hon-Tong A, Charles MA, Forhan A, Heude B, Sirot V. Exposure to food contaminants during pregnancy. Sci Total Environ. 2013;458-460:27–35. doi: 10.1016/j.scitotenv.2013.03.100. [DOI] [PubMed] [Google Scholar]
- 132.Kwong WT, Friello P, Semba RD. Interactions between iron deficiency and lead poisoning: epidemiology and pathogenesis. Sci Total Environ. 2004;330:21–37. doi: 10.1016/j.scitotenv.2004.03.017. [DOI] [PubMed] [Google Scholar]
- 133.Talpur S, Afridi HI, Kazi TG, Talpur FN. Interaction of Lead with Calcium, Iron, and Zinc in the Biological Samples of Malnourished Children. Biol Trace Elem Res. 2018;183:209–217. doi: 10.1007/s12011-017-1141-9. [DOI] [PubMed] [Google Scholar]
- 134.Gomes WR, Devóz PP, Araújo ML, Batista BL, Barbosa F Jr, Barcelos GRM. Milk and Dairy Products Intake Is Associated with Low Levels of Lead (Pb) in Workers highly Exposed to the Metal. Biol Trace Elem Res. 2017;178:29–35. doi: 10.1007/s12011-016-0913-y. [DOI] [PubMed] [Google Scholar]
- 135.Ames JM. Dietary Maillard Reaction Products: Implications for Human Health and Disease. Czech J Food Sci. 2009;27:S66–S69. [Google Scholar]
- 136.Koschinsky T, He CJ, Mitsuhashi T, Bucala R, Liu C, Buenting C, Heitmann K, Vlassara H. Orally absorbed reactive glycation products (glycotoxins): an environmental risk factor in diabetic nephropathy. Proc Natl Acad Sci USA. 1997;94:6474–6479. doi: 10.1073/pnas.94.12.6474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Mericq V, Piccardo C, Cai W, Chen X, Zhu L, Striker GE, Vlassara H, Uribarri J. Maternally transmitted and food-derived glycotoxins: a factor preconditioning the young to diabetes? Diabetes Care. 2010;33:2232–2237. doi: 10.2337/dc10-1058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Csongová M, Gurecká R, Koborová I, Celec P, Domonkos E, Uličná O, Somoza V, Šebeková K. The effects of a maternal advanced glycation end product-rich diet on somatic features, reflex ontogeny and metabolic parameters of offspring mice. Food Funct. 2018;9:3432–3446. doi: 10.1039/c8fo00183a. [DOI] [PubMed] [Google Scholar]
- 139.Goldberg T, Cai W, Peppa M, Dardaine V, Baliga BS, Uribarri J, Vlassara H. Advanced glycoxidation end products in commonly consumed foods. J Am Diet Assoc. 2004;104:1287–1291. doi: 10.1016/j.jada.2004.05.214. [DOI] [PubMed] [Google Scholar]
- 140.Meguid El Nahas A, Bello AK. Chronic kidney disease: the global challenge. Lancet. 2005;365:331–340. doi: 10.1016/S0140-6736(05)17789-7. [DOI] [PubMed] [Google Scholar]
- 141.Moller DE. Potential role of TNF-alpha in the pathogenesis of insulin resistance and type 2 diabetes. Trends Endocrinol Metab. 2000;11:212–217. doi: 10.1016/s1043-2760(00)00272-1. [DOI] [PubMed] [Google Scholar]
- 142.Duarte-Salles T, von Stedingk H, Granum B, Gützkow KB, Rydberg P, Törnqvist M, Mendez MA, Brunborg G, Brantsæter AL, Meltzer HM, Alexander J, Haugen M. Dietary acrylamide intake during pregnancy and fetal growth-results from the Norwegian mother and child cohort study (MoBa) Environ Health Perspect. 2013;121:374–379. doi: 10.1289/ehp.1205396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Pedersen M, von Stedingk H, Botsivali M, Agramunt S, Alexander J, Brunborg G, Chatzi L, Fleming S, Fthenou E, Granum B, Gutzkow KB, Hardie LJ, Knudsen LE, Kyrtopoulos SA, Mendez MA, Merlo DF, Nielsen JK, Rydberg P, Segerbäck D, Sunyer J, Wright J, Törnqvist M, Kleinjans JC, Kogevinas M NewGeneris Consortium. Birth weight, head circumference, and prenatal exposure to acrylamide from maternal diet: the European prospective mother-child study (NewGeneris) Environ Health Perspect. 2012;120:1739–1745. doi: 10.1289/ehp.1205327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Kadawathagedara M, Tong ACH, Heude B, Forhan A, Charles MA, Sirot V, Botton J, The Eden Mother-Child Cohort Study Group. Dietary acrylamide intake during pregnancy and anthropometry at birth in the French EDEN mother-child cohort study. Environ Res. 2016;149:189–196. doi: 10.1016/j.envres.2016.05.019. [DOI] [PubMed] [Google Scholar]
- 145.Ferguson SA, Garey J, Smith ME, Twaddle NC, Doerge DR, Paule MG. Preweaning behaviors, developmental landmarks, and acrylamide and glycidamide levels after pre- and postnatal acrylamide treatment in rats. Neurotoxicol Teratol. 2010;32:373–382. doi: 10.1016/j.ntt.2010.01.010. [DOI] [PubMed] [Google Scholar]
- 146.Yilmaz BO, Yildizbayrak N, Aydin Y, Erkan M. Evidence of acrylamide- and glycidamide-induced oxidative stress and apoptosis in Leydig and Sertoli cells. Hum Exp Toxicol. 2017;36:1225–1235. doi: 10.1177/0960327116686818. [DOI] [PubMed] [Google Scholar]
- 147.Annola K, Karttunen V, Keski-Rahkonen P, Myllynen P, Segerbäck D, Heinonen S, Vähäkangas K. Transplacental transfer of acrylamide and glycidamide are comparable to that of antipyrine in perfused human placenta. Toxicol Lett. 2008;182:50–56. doi: 10.1016/j.toxlet.2008.08.006. [DOI] [PubMed] [Google Scholar]
- 148.Sörgel F, Weissenbacher R, Kinzig-Schippers M, Hofmann A, Illauer M, Skott A, Landersdorfer C. Acrylamide: increased concentrations in homemade food and first evidence of its variable absorption from food, variable metabolism and placental and breast milk transfer in humans. Chemotherapy. 2002;48:267–274. doi: 10.1159/000069715. [DOI] [PubMed] [Google Scholar]
- 149.Lin CY, Lin LY, Chen YC, Wen LL, Chien KL, Sung FC, Chen PC, Su TC. Association between measurements of thyroid function and the acrylamide metabolite N-Acetyl-S-(propionamide)-cysteine in adolescents and young adults. Environ Res. 2015;136:246–252. doi: 10.1016/j.envres.2014.08.043. [DOI] [PubMed] [Google Scholar]
- 150.Shields BM, Knight BA, Hill A, Hattersley AT, Vaidya B. Fetal thyroid hormone level at birth is associated with fetal growth. J Clin Endocrinol Metab. 2011;96:E934–E938. doi: 10.1210/jc.2010-2814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Diamanti-Kandarakis E, Piperi C, Korkolopoulou P, Kandaraki E, Levidou G, Papalois A, Patsouris E, Papavassiliou AG. Accumulation of dietary glycotoxins in the reproductive system of normal female rats. J Mol Med (Berl) 2007;85:1413–1420. doi: 10.1007/s00109-007-0246-6. [DOI] [PubMed] [Google Scholar]
- 152.Chatzigeorgiou A, Kandaraki E, Piperi C, Livadas S, Papavassiliou AG, Koutsilieris M, Papalois A, Diamanti-Kandarakis E. Dietary glycotoxins affect scavenger receptor expression and the hormonal profile of female rats. J Endocrinol. 2013;218:331–337. doi: 10.1530/JOE-13-0175. [DOI] [PubMed] [Google Scholar]
- 153.Clarke RE, Dordevic AL, Tan SM, Ryan L, Coughlan MT. Dietary Advanced Glycation End Products and Risk Factors for Chronic Disease: A Systematic Review of Randomised Controlled Trials. Nutrients. 2016;8:125. doi: 10.3390/nu8030125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Falony G, Joossens M, Vieira-Silva S, Wang J, Darzi Y, Faust K, Kurilshikov A, Bonder MJ, Valles-Colomer M, Vandeputte D, Tito RY, Chaffron S, Rymenans L, Verspecht C, De Sutter L, Lima-Mendez G, D'hoe K, Jonckheere K, Homola D, Garcia R, Tigchelaar EF, Eeckhaudt L, Fu J, Henckaerts L, Zhernakova A, Wijmenga C, Raes J. Population-level analysis of gut microbiome variation. Science. 2016;352:560–564. doi: 10.1126/science.aad3503. [DOI] [PubMed] [Google Scholar]
- 155.Jernberg C, Löfmark S, Edlund C, Jansson JK. Long-term impacts of antibiotic exposure on the human intestinal microbiota. Microbiology. 2010;156:3216–3223. doi: 10.1099/mic.0.040618-0. [DOI] [PubMed] [Google Scholar]
- 156.Mitsou EK, Kirtzalidou E, Pramateftaki P, Kyriacou A. Antibiotic resistance in faecal microbiota of Greek healthy infants. Benef Microbes. 2010;1:297–306. doi: 10.3920/BM2010.0007. [DOI] [PubMed] [Google Scholar]
- 157.Fouhy F, Guinane CM, Hussey S, Wall R, Ryan CA, Dempsey EM, Murphy B, Ross RP, Fitzgerald GF, Stanton C, Cotter PD. High-throughput sequencing reveals the incomplete, short-term recovery of infant gut microbiota following parenteral antibiotic treatment with ampicillin and gentamicin. Antimicrob Agents Chemother. 2012;56:5811–5820. doi: 10.1128/AAC.00789-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Dethlefsen L, Huse S, Sogin ML, Relman DA. The pervasive effects of an antibiotic on the human gut microbiota, as revealed by deep 16S rRNA sequencing. PLoS Biol. 2008;6:e280. doi: 10.1371/journal.pbio.0060280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Rousseau C, Levenez F, Fouqueray C, Doré J, Collignon A, Lepage P. Clostridium difficile colonization in early infancy is accompanied by changes in intestinal microbiota composition. J Clin Microbiol. 2011;49:858–865. doi: 10.1128/JCM.01507-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Penders J, Thijs C, Vink C, Stelma FF, Snijders B, Kummeling I, van den Brandt PA, Stobberingh EE. Factors influencing the composition of the intestinal microbiota in early infancy. Pediatrics. 2006;118:511–521. doi: 10.1542/peds.2005-2824. [DOI] [PubMed] [Google Scholar]
- 161.Cho I, Yamanishi S, Cox L, Methé BA, Zavadil J, Li K, Gao Z, Mahana D, Raju K, Teitler I, Li H, Alekseyenko AV, Blaser MJ. Antibiotics in early life alter the murine colonic microbiome and adiposity. Nature. 2012;488:621–626. doi: 10.1038/nature11400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Gensollen T, Iyer SS, Kasper DL, Blumberg RS. How colonization by microbiota in early life shapes the immune system. Science. 2016;352:539–544. doi: 10.1126/science.aad9378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Tanaka S, Kobayashi T, Songjinda P, Tateyama A, Tsubouchi M, Kiyohara C, Shirakawa T, Sonomoto K, Nakayama J. Influence of antibiotic exposure in the early postnatal period on the development of intestinal microbiota. FEMS Immunol Med Microbiol. 2009;56:80–87. doi: 10.1111/j.1574-695X.2009.00553.x. [DOI] [PubMed] [Google Scholar]
- 164.Flandroy L, Poutahidis T, Berg G, Clarke G, Dao MC, Decaestecker E, Furman E, Haahtela T, Massart S, Plovier H, Sanz Y, Rook G. The impact of human activities and lifestyles on the interlinked microbiota and health of humans and of ecosystems. Sci Total Environ. 2018;627:1018–1038. doi: 10.1016/j.scitotenv.2018.01.288. [DOI] [PubMed] [Google Scholar]
- 165.Norrby SR. Neurotoxicity of carbapenem antibiotics: consequences for their use in bacterial meningitis. J Antimicrob Chemother. 2000;45:5–7. [Google Scholar]
- 166.Ge X, Ding C, Zhao W, Xu L, Tian H, Gong J, Zhu M, Li J, Li N. Antibiotics-induced depletion of mice microbiota induces changes in host serotonin biosynthesis and intestinal motility. J Transl Med. 2017;15:13. doi: 10.1186/s12967-016-1105-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Bhattacharyya S, Darby R, Berkowitz AL. Antibiotic-induced neurotoxicity. Curr Infect Dis Rep. 2014;16:448. doi: 10.1007/s11908-014-0448-3. [DOI] [PubMed] [Google Scholar]
- 168.Atli O, Demir-Ozkay U, Ilgin S, Aydin TH, Akbulut EN, Sener E. Evidence for neurotoxicity associated with amoxicillin in juvenile rats. Hum Exp Toxicol. 2016;35:866–876. doi: 10.1177/0960327115607948. [DOI] [PubMed] [Google Scholar]
- 169.Shepherd RW, Butler DG, Cutz E, Gall DG, Hamilton JR. The mucosal lesion in viral enteritis. Extent and dynamics of the epithelial response to virus invasion in transmissible gastroenteritis of piglets. Gastroenterology. 1979;76:770–777. doi: 10.1016/S0016-5085(79)80177-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Collins J, Starkey WG, Wallis TS, Clarke GJ, Worton KJ, Spencer AJ, Haddon SJ, Osborne MP, Candy DC, Stephen J. Intestinal enzyme profiles in normal and rotavirus-infected mice. J Pediatr Gastroenterol Nutr. 1988;7:264–272. doi: 10.1097/00005176-198803000-00017. [DOI] [PubMed] [Google Scholar]
- 171.Katyal R, Rana SV, Vaiphei K, Ohja S, Singh K, Singh V. Effect of rotavirus infection on small gut pathophysiology in a mouse model. J Gastroenterol Hepatol. 1999;14:779–784. doi: 10.1046/j.1440-1746.1999.01948.x. [DOI] [PubMed] [Google Scholar]
- 172.Halaihel N, Liévin V, Alvarado F, Vasseur M. Rotavirus infection impairs intestinal brush-border membrane Na(+)-solute cotransport activities in young rabbits. Am J Physiol Gastrointest Liver Physiol. 2000;279:G587–G596. doi: 10.1152/ajpgi.2000.279.3.G587. [DOI] [PubMed] [Google Scholar]
- 173.Bishop RF, Davidson GP, Holmes IH, Ruck BJ. Virus particles in epithelial cells of duodenal mucosa from children with acute non-bacterial gastroenteritis. Lancet. 1973;2:1281–1283. doi: 10.1016/s0140-6736(73)92867-5. [DOI] [PubMed] [Google Scholar]
- 174.Weng M, Walker WA. The role of gut microbiota in programming the immune phenotype. J Dev Orig Health Dis. 2013;4:203–214. doi: 10.1017/S2040174412000712. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Bellinger DC. Lead neurotoxicity and socioeconomic status: conceptual and analytical issues. Neurotoxicology. 2008;29:828–832. doi: 10.1016/j.neuro.2008.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Tsai MS, Chen MH, Lin CC, Liu CY, Chen PC. Children's environmental health based on birth cohort studies of Asia (2) - air pollution, pesticides, and heavy metals. Environ Res. 2019;179:108754. doi: 10.1016/j.envres.2019.108754. [DOI] [PubMed] [Google Scholar]
- 177.Stein LJ, Gunier RB, Harley K, Kogut K, Bradman A, Eskenazi B. Early childhood adversity potentiates the adverse association between prenatal organophosphate pesticide exposure and child IQ: The CHAMACOS cohort. Neurotoxicology. 2016;56:180–187. doi: 10.1016/j.neuro.2016.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178.Bouchard MF, Chevrier J, Harley KG, Kogut K, Vedar M, Calderon N, Trujillo C, Johnson C, Bradman A, Barr DB, Eskenazi B. Prenatal exposure to organophosphate pesticides and IQ in 7-year-old children. Environ Health Perspect. 2011;119:1189–1195. doi: 10.1289/ehp.1003185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.González-Alzaga B, Lacasaña M, Aguilar-Garduño C, Rodríguez-Barranco M, Ballester F, Rebagliato M, Hernández AF. A systematic review of neurodevelopmental effects of prenatal and postnatal organophosphate pesticide exposure. Toxicol Lett. 2014;230:104–121. doi: 10.1016/j.toxlet.2013.11.019. [DOI] [PubMed] [Google Scholar]
- 180.Caspersen IH, Kvalem HE, Haugen M, Brantsæter AL, Meltzer HM, Alexander J, Thomsen C, Frøshaug M, Bremnes NM, Broadwell SL, Granum B, Kogevinas M, Knutsen HK. Determinants of plasma PCB, brominated flame retardants, and organochlorine pesticides in pregnant women and 3 year old children in The Norwegian Mother and Child Cohort Study. Environ Res. 2016;146:136–144. doi: 10.1016/j.envres.2015.12.020. [DOI] [PubMed] [Google Scholar]
- 181.Burns JS, Williams PL, Korrick SA, Hauser R, Sergeyev O, Revich B, Lam T, Lee MM. Association between chlorinated pesticides in the serum of prepubertal Russian boys and longitudinal biomarkers of metabolic function. Am J Epidemiol. 2014;180:909–919. doi: 10.1093/aje/kwu212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 182.Guerrero Schimpf M, Milesi MM, Ingaramo PI, Luque EH, Varayoud J. Neonatal exposure to a glyphosate based herbicide alters the development of the rat uterus. Toxicology. 2017;376:2–14. doi: 10.1016/j.tox.2016.06.004. [DOI] [PubMed] [Google Scholar]
- 183.Nielsen LN, Roager HM, Casas ME, Frandsen HL, Gosewinkel U, Bester K, Licht TR, Hendriksen NB, Bahl MI. Glyphosate has limited short-term effects on commensal bacterial community composition in the gut environment due to sufficient aromatic amino acid levels. Environ Pollut. 2018;233:364–376. doi: 10.1016/j.envpol.2017.10.016. [DOI] [PubMed] [Google Scholar]
- 184.Lombard MJ. Mycotoxin exposure and infant and young child growth in Africa: what do we know? Ann Nutr Metab. 2014;64 Suppl 2:42–52. doi: 10.1159/000365126. [DOI] [PubMed] [Google Scholar]
- 185.Raiola A, Tenore GC, Manyes L, Meca G, Ritieni A. Risk analysis of main mycotoxins occurring in food for children: An overview. Food Chem Toxicol. 2015;84:169–180. doi: 10.1016/j.fct.2015.08.023. [DOI] [PubMed] [Google Scholar]
- 186.Skaug MA. Analysis of Norwegian milk and infant formulas for ochratoxin A. Food Addit Contam. 1999;16:75–78. doi: 10.1080/026520399284235. [DOI] [PubMed] [Google Scholar]
- 187.Ojuri OT, Ezekiel CN, Eskola MK, Šarkanj B, Babalola AD, Sulyok M, Hajšlová J, Elliott CT, Krska R. Mycotoxin co-exposures in infants and young children consuming household- and industrially-processed complementary foods in Nigeria and risk management advice. Food Control. 2019;98:312–22. [Google Scholar]
- 188.Gong YY, Egal S, Hounsa A, Turner PC, Hall AJ, Cardwell KF, Wild CP. Determinants of aflatoxin exposure in young children from Benin and Togo, West Africa: the critical role of weaning. Int J Epidemiol. 2003;32:556–562. doi: 10.1093/ije/dyg109. [DOI] [PubMed] [Google Scholar]
- 189.Turner PC, Moore SE, Hall AJ, Prentice AM, Wild CP. Modification of immune function through exposure to dietary aflatoxin in Gambian children. Environ Health Perspect. 2003;111:217–220. doi: 10.1289/ehp.5753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Sherif SO, Salama EE, Abdel-Wahhab MA. Mycotoxins and child health: the need for health risk assessment. Int J Hyg Environ Health. 2009;212:347–368. doi: 10.1016/j.ijheh.2008.08.002. [DOI] [PubMed] [Google Scholar]
- 191.Hannon WH, Hill RH Jr, Bernert JT Jr, Haddock L, Lebron G, Cordero JF. Premature thelarche in Puerto Rico: a search for environmental estrogenic contamination. Arch Environ Contam Toxicol. 1987;16:255–262. doi: 10.1007/BF01054942. [DOI] [PubMed] [Google Scholar]
- 192.Sáenz de Rodríguez CA. Environmental hormone contamination in Puerto Rico. N Engl J Med. 1984;310:1741–1742. doi: 10.1056/NEJM198406283102612. [DOI] [PubMed] [Google Scholar]
- 193.Bhat RV, Beedu SR, Ramakrishna Y, Munshi KL. Outbreak of trichothecene mycotoxicosis associated with consumption of mould-damaged wheat production in Kashmir Valley, India. Lancet. 1989;1:35–37. doi: 10.1016/s0140-6736(89)91684-x. [DOI] [PubMed] [Google Scholar]
- 194.Desjardins A. Mechanisms of actions of trichothecenes. In: Fusarium mycotoxins: Genetic chemistry and biology. Saint Paul: Minnesota, 2006: 53-54. [Google Scholar]
- 195.Girgis GN, Barta JR, Brash M, Smith TK. Morphologic changes in the intestine of broiler breeder pullets fed diets naturally contaminated with Fusarium mycotoxins with or without coccidial challenge. Avian Dis. 2010;54:67–73. doi: 10.1637/8945-052809-Reg.1. [DOI] [PubMed] [Google Scholar]
- 196.Hendrickse RG. Of sick turkeys, kwashiorkor, malaria, perinatal mortality, heroin addicts and food poisoning: research on the influence of aflatoxins on child health in the tropics. Ann Trop Med Parasitol. 1997;91:787–793. doi: 10.1080/00034989760545. [DOI] [PubMed] [Google Scholar]
- 197.Berg T, Rasmussen G, Thorup I. Mycotoxins in Danish Foods. Danish, SØborg: National Food Agency of Health, 1995. [Google Scholar]
- 198.FAO. Safety evaluation of certain mycotoxins in food. In: (IPCS) IP on CS. Fifty-sixth meeting of the Joint FAO/WHO Expert Committee on Food Additives. Geneva, 2001: 712. [Google Scholar]
- 199.Mahfoud R, Maresca M, Garmy N, Fantini J. The mycotoxin patulin alters the barrier function of the intestinal epithelium: mechanism of action of the toxin and protective effects of glutathione. Toxicol Appl Pharmacol. 2002;181:209–218. doi: 10.1006/taap.2002.9417. [DOI] [PubMed] [Google Scholar]
- 200.Mohan HM, Collins D, Maher S, Walsh EG, Winter DC, O'Brien PJ, Brayden DJ, Baird AW. The mycotoxin patulin increases colonic epithelial permeability in vitro. Food Chem Toxicol. 2012;50:4097–4102. doi: 10.1016/j.fct.2012.07.036. [DOI] [PubMed] [Google Scholar]
- 201.Marin S, Ramos AJ, Cano-Sancho G, Sanchis V. Mycotoxins: occurrence, toxicology, and exposure assessment. Food Chem Toxicol. 2013;60:218–237. doi: 10.1016/j.fct.2013.07.047. [DOI] [PubMed] [Google Scholar]
- 202.Kim S, Eom S, Kim HJ, Lee JJ, Choi G, Choi S, Kim S, Kim SY, Cho G, Kim YD, Suh E, Kim SK, Kim S, Kim GH, Moon HB, Park J, Kim S, Choi K, Eun SH. Association between maternal exposure to major phthalates, heavy metals, and persistent organic pollutants, and the neurodevelopmental performances of their children at 1 to 2years of age- CHECK cohort study. Sci Total Environ. 2018;624:377–384. doi: 10.1016/j.scitotenv.2017.12.058. [DOI] [PubMed] [Google Scholar]
- 203.Defois C, Ratel J, Garrait G, Denis S, Le Goff O, Talvas J, Mosoni P, Engel E, Peyret P. Food Chemicals Disrupt Human Gut Microbiota Activity And Impact Intestinal Homeostasis As Revealed By In Vitro Systems. Sci Rep. 2018;8:11006. doi: 10.1038/s41598-018-29376-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Gascon M, Verner MA, Guxens M, Grimalt JO, Forns J, Ibarluzea J, Lertxundi N, Ballester F, Llop S, Haddad S, Sunyer J, Vrijheid M. Evaluating the neurotoxic effects of lactational exposure to persistent organic pollutants (POPs) in Spanish children. Neurotoxicology. 2013;34:9–15. doi: 10.1016/j.neuro.2012.10.006. [DOI] [PubMed] [Google Scholar]
- 205.Claus SP, Guillou H, Ellero-Simatos S. Erratum: The gut microbiota: a major player in the toxicity of environmental pollutants? NPJ Biofilms Microbiomes. 2017;3:17001. doi: 10.1038/npjbiofilms.2017.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Van de Wiele T, Vanhaecke L, Boeckaert C, Peru K, Headley J, Verstraete W, Siciliano S. Human colon microbiota transform polycyclic aromatic hydrocarbons to estrogenic metabolites. Environ Health Perspect. 2005;113:6–10. doi: 10.1289/ehp.7259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Ku HY, Tsai TL, Wang PL, Su PH, Sun CW, Wang CJ, Wang SL. Prenatal and childhood phthalate exposure and attention deficit hyperactivity disorder traits in child temperament: A 12-year follow-up birth cohort study. Sci Total Environ. 2020;699:134053. doi: 10.1016/j.scitotenv.2019.134053. [DOI] [PubMed] [Google Scholar]
- 208.Zhang L, Nichols RG, Correll J, Murray IA, Tanaka N, Smith PB, Hubbard TD, Sebastian A, Albert I, Hatzakis E, Gonzalez FJ, Perdew GH, Patterson AD. Persistent Organic Pollutants Modify Gut Microbiota-Host Metabolic Homeostasis in Mice Through Aryl Hydrocarbon Receptor Activation. Environ Health Perspect. 2015;123:679–688. doi: 10.1289/ehp.1409055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Wang IJ, Karmaus WJ, Chen SL, Holloway JW, Ewart S. Effects of phthalate exposure on asthma may be mediated through alterations in DNA methylation. Clin Epigenetics. 2015;7:27. doi: 10.1186/s13148-015-0060-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Vrijheid M, Casas M, Gascon M, Valvi D, Nieuwenhuijsen M. Environmental pollutants and child health-A review of recent concerns. Int J Hyg Environ Health. 2016;219:331–342. doi: 10.1016/j.ijheh.2016.05.001. [DOI] [PubMed] [Google Scholar]
- 211.Karatela S, Paterson J, Ward NI. Domain specific effects of postnatal toenail methylmercury exposure on child behaviour. J Trace Elem Med Biol. 2017;41:10–15. doi: 10.1016/j.jtemb.2017.01.003. [DOI] [PubMed] [Google Scholar]
- 212.Hamadani JD, Tofail F, Nermell B, Gardner R, Shiraji S, Bottai M, Arifeen SE, Huda SN, Vahter M. Critical windows of exposure for arsenic-associated impairment of cognitive function in pre-school girls and boys: a population-based cohort study. Int J Epidemiol. 2011;40:1593–1604. doi: 10.1093/ije/dyr176. [DOI] [PubMed] [Google Scholar]
- 213.Pinyayev TS, Kohan MJ, Herbin-Davis K, Creed JT, Thomas DJ. Preabsorptive metabolism of sodium arsenate by anaerobic microbiota of mouse cecum forms a variety of methylated and thiolated arsenicals. Chem Res Toxicol. 2011;24:475–477. doi: 10.1021/tx200040w. [DOI] [PubMed] [Google Scholar]
- 214.Tolins M, Ruchirawat M, Landrigan P. The developmental neurotoxicity of arsenic: cognitive and behavioral consequences of early life exposure. Ann Glob Health. 2014;80:303–314. doi: 10.1016/j.aogh.2014.09.005. [DOI] [PubMed] [Google Scholar]
- 215.Finot PA. Historical perspective of the Maillard reaction in food science. Ann N Y Acad Sci. 2005;1043:1–8. doi: 10.1196/annals.1333.001. [DOI] [PubMed] [Google Scholar]
- 216.Kierdorf K, Fritz G. RAGE regulation and signaling in inflammation and beyond. J Leukoc Biol. 2013;94:55–68. doi: 10.1189/jlb.1012519. [DOI] [PubMed] [Google Scholar]
- 217.Wang Z, Hall SD, Maya JF, Li L, Asghar A, Gorski JC. Diabetes mellitus increases the in vivo activity of cytochrome P450 2E1 in humans. Br J Clin Pharmacol. 2003;55:77–85. doi: 10.1046/j.1365-2125.2003.01731.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Nguyen MU, Wallace MJ, Pepe S, Menheniott TR, Moss TJ, Burgner D. Perinatal inflammation: a common factor in the early origins of cardiovascular disease? Clin Sci (Lond) 2015;129:769–784. doi: 10.1042/CS20150045. [DOI] [PubMed] [Google Scholar]
- 219.Dittrich R, Hoffmann I, Stahl P, Müller A, Beckmann MW, Pischetsrieder M. Concentrations of Nepsilon-carboxymethyllysine in human breast milk, infant formulas, and urine of infants. J Agric Food Chem. 2006;54:6924–6928. doi: 10.1021/jf060905h. [DOI] [PubMed] [Google Scholar]
- 220.Baskara I, Niquet-Leridon C, Anton PM, Delayre-Orthez C. Neoformed Compounds From the Maillard Reaction in Infant Formulas: a New Risk Factor for Allergy? EMJ Allergy Immunol. 2017;2:87–93. [Google Scholar]
- 221.Shimoike T, Inoguchi T, Umeda F, Nawata H, Kawano K, Ochi H. The meaning of serum levels of advanced glycosylation end products in diabetic nephropathy. Metabolism. 2000;49:1030–1035. doi: 10.1053/meta.2000.7738. [DOI] [PubMed] [Google Scholar]
- 222.Sharp PS, Rainbow S, Mukherjee S. Serum levels of low molecular weight advanced glycation end products in diabetic subjects. Diabet Med. 2003;20:575–579. doi: 10.1046/j.1464-5491.2003.00973.x. [DOI] [PubMed] [Google Scholar]
- 223.Ge J, Jia Q, Liang C, Luo Y, Huang D, Sun A, Wang K, Zou Y, Chen H. Advanced glycosylation end products might promote atherosclerosis through inducing the immune maturation of dendritic cells. Arterioscler Thromb Vasc Biol. 2005;25:2157–2163. doi: 10.1161/01.ATV.0000181744.58265.63. [DOI] [PubMed] [Google Scholar]
- 224.Hales CN, Barker DJ, Clark PM, Cox LJ, Fall C, Osmond C, Winter PD. Fetal and infant growth and impaired glucose tolerance at age 64. BMJ. 1991;303:1019–1022. doi: 10.1136/bmj.303.6809.1019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Forest MG. Role of androgens in fetal and pubertal development. Horm Res. 1983;18:69–83. doi: 10.1159/000179780. [DOI] [PubMed] [Google Scholar]
- 226.Gorski RA. Sexual dimorphisms of the brain. J Anim Sci. 1985;61 Suppl 3:38–61. doi: 10.1093/ansci/61.supplement_3.38. [DOI] [PubMed] [Google Scholar]
- 227.Jost A. Genetic and hormonal factors in sex differentiation of the brain. Psychoneuroendocrinology. 1983;8:183–193. doi: 10.1016/0306-4530(83)90055-0. [DOI] [PubMed] [Google Scholar]
- 228.Phoenix CH, Goy RW, Gerall AA, Young WC. Organizing action of prenatally administered testosterone propionate on the tissues mediating mating behavior in the female guinea pig. Endocrinology. 1959;65:369–382. doi: 10.1210/endo-65-3-369. [DOI] [PubMed] [Google Scholar]
- 229.Wood RI, Foster DL. Sexual differentiation of reproductive neuroendocrine function in sheep. Rev Reprod. 1998;3:130–140. doi: 10.1530/ror.0.0030130. [DOI] [PubMed] [Google Scholar]