To the Editor: The epidemic caused by 2019 novel coronavirus (2019-nCoV) has drawn public attention.[1] Huge infected population and enormous economic loss make it the urgent public health event to deal with. Confirmatory test targeting virus RNA was established at the early stage of outbreak and then used for 2019-nCoV infection diagnosis.[2] However, high risk of laboratory infection, high-qualified personnel, and strict operation condition hampered its application into primary hospitals and community clinics.[3] In this study, the serum immunoglobulin (Ig) M and IgG antibodies were detected in 2019-nCoV confirmed cases of different stages. Furthermore, three different immunological assays, chemiluminescent immunoassay (CLIA), gold immunochromatographic assay (GICA), and enzyme-linked immunosorbent assay (ELISA) were used for IgM and IgG detection.
A total of 22 confirmed patients were admitted to the Fifth Hospital of Shijiazhuang, the designated hospital for 2019-nCoV infection, from January 21st to February 24th, 2020. All patients were confirmed by reverse-transcription polymerase-chain-reaction assay (2019-nCoV RNA Test Kit, Daan Gene Company, China) for nasal and pharyngeal swab specimens, and were treated according to the national diagnosis and treatment plan.[4] This study was approved by the Ethics Commission of the Fifth Hospital of Shijiazhuang.The authors certify that they have obtained all appropriate patient consent forms. The clinical features and laboratory results were collected from electronic medical records. Typical ground-glass opacity in lung was observed in computed tomography scan results of all patients. Of the 22 patients, eight were females and 14 were males. Eleven patients had recent history of travel to epidemic areas, and the remaining ten had close contacts with their family members, who were confirmed to be infected by 2019-nCoV. The patients aged from 4 to 72 years, with the median age of 40 years. Most of the patients received oxygen therapy and anti-virus medication. Currently, all patients had recovered and discharged from hospital.
Due to the late delivery of the testing kits, some serum samples were not available. A total of 37 serum samples from 22 patients were obtained, of which ten came from the early stage (1–7 days after the onset of infection), 13 from the middle stage (8–14 days after the onset of infection), and 14 from the late stage (14–24 days after the onset of infection). The level of serum IgM and IgG antibodies were detected using three different commercial testing kits (CLIA, GICA, and ELISA) developed by the Beier Bioengineering Company (Beijing, China), which targeted the spike (S) and nucleocapsid (N) proteins of 2019-nCoV. For CLIA, samples with an concentration ≥8 arbitrary unit/mL were considered positive. For GICA, positive results showed the appearance of both control line and testing line. For ELISA, the absorbance at 450 nm (A450 nm) of each well was determined within 10 min using a microplate reader, and the cut-off value was 0.10+Anegative control. A value higher than the cut-off value was considered a positive result. All tests were conducted in the Laboratory of the Fifth Hospital of Shijiazhuang, according to the manufacturer's instructions and the technical guidelines for laboratory testing of new coronavirus infected pneumonia (second edition).[5]
As shown in Table 1, at the early, middle and late stages of infection, the serum IgM antibody was detected in 6/10, 7/13, and 11/14 of cases, respectively (at least one immunoassay was positive). Meanwhile, at the early and middle stages of infection, 5/10 and 10/13 cases demonstrated positive serum IgG results, whereas the serum IgG was detected in all cases (14/14) when it came to the late stage of infection.
Table 1.
The serum IgM and IgG levels in 2019-nCoV infected patients detected by different methods.
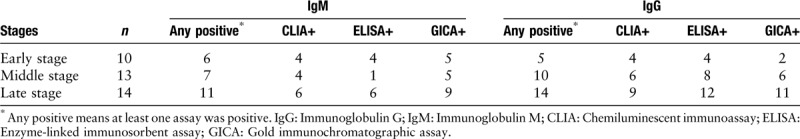
CLIA, GICA, and ELISA are important detection methods in clinical immunological diagnosis. Nevertheless, the positive rates of the three methods differed in detection of 2019-nCoV infection. GICA possessed a relatively higher positive rate in serum IgM detection (5/10, 5/13, and 9/14 in the early, middle, and late stages, respectively) and ELISA exhibited a comparatively higher positive rate in serum IgG testing (4/10, 8/13, and 12/14 in the early, middle, and late stages, respectively).
Detection of serum specific antibodies, IgM and IgG, is another crucial evidence for the diagnosis of infection. Easy operation, low work environmental requirement, and accessibility of blood samples make serological antibody test another choice for rapid screening and diagnosis of 2019-nCoV infection. The combination of GICA for serum IgM and ELISA for serum IgG might be an effective way for early screening and diagnosis for 2019-nCoV infection.
Acknowledgements
The authors thank the staff of the Fifth Hospital of Shijiazhuang for their contribution to this study. The authors appreciate the support provided by the Center for Disease Control and Prevention in Hebei Province.
Conflicts of interest
None.
Footnotes
How to cite this article: Gao HX, Li YN, Xu ZG, Wang YL, Wang HB, Cao JF, Yuan DQ, Li L, Xu Y, Zhang Z, Huang Y, Lu JH, Liu YZ, Dai EH. Detection of serum immunoglobulin M and immunoglobulin G antibodies in 2019 novel coronavirus infected patients from different stages. Chin Med J 2020;133:1479–1480. doi: 10.1097/CM9.0000000000000820
Hui-Xia Gao and Ya-Nan Li contributed equally to this work.
References
- 1.Stop the Wuhan virus. Nature 2020; 577:450.doi: 10.1038/d41586-020-00153-x. [DOI] [PubMed] [Google Scholar]
- 2.Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, et al. A novel coronavirus from patients with pneumonia in China. N Engl J Med 2020; 382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Yu F, Du L, Ojcius D, Pan C, Jiang S. Measures for diagnosing and treating infections by a novel coronavirus responsible for a pneumonia outbreak originating in Wuhan, China. Microbes Infect 2020; 22:74–79. doi: 10.1016/j.micinf.2020.01.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. COVID-19 Diagnosis and Treatment Plan (Trial Version 6) (In Chinese). Beijing: National Health Commission of the People's Republic of China; 2020. Available from: http://www.nhc.gov.cn/yzygj/s7653p/202002/8334a8326dd94d329df351d7da8aefc2/files/b218cfeb1bc54639af227f922bf6b817.pdf. [Accessed March 8, 2020] [Google Scholar]
- 5.Xu WZ, Li J, He XY, Zhang CQ, Mei SQ, Li CR, et al. The diagnostic value of joint detection of serum IgM and IgG antibodies to 2019-nCoV in 2019-nCoV infection (in Chinese). Chin J Lab Med 2020; [Epub ahead of print]. doi: 10.3760/cma.j.cn114452-20200223-00109. [Google Scholar]


