Abstract
Purpose of Review
To provide information on characteristics and use of various ceramics in spine fusion and future directions.
Recent Findings
In most recent years, focus has been shifted to the use of ceramics in minimally invasive surgeries or implementation of nanostructured surface modification features to promote osteoinductive properties. In addition, effort has been placed on the development of bioactive synthetics. Core characteristic of bioactive synthetics is that they undergo change to simulate a beneficial response within the bone. This change is based on chemical reaction and various chemical elements present in the bioactive ceramics. Recently, a synthetic 15-amino acid polypeptide bound to an anorganic bone material which mimics the cell-binding domain of type-I collagen opened a possibility for osteogenic and osteoinductive roles of this hybrid graft material.
Summary
Ceramics have been present in the spine fusion arena for several decades; however, their use has been limited. The major obstacle in published literature is small sample size resulting in low evidence and a potential for bias. In addition, different physical and chemical properties of various ceramics further contribute to the limited evidence. Although ceramics have several disadvantages, they still hold a great promise as a value-based graft material with being easily available, relatively inexpensive, and non-immunogenic.
Keywords: Ceramics, Synthetics, Hydroxyapatite, Beta-tricalcium phosphate, Bioactive, Spine fusion
Introduction
The incidence of spinal fusion has been steadily increasing over the past couple of decades [1, 2]. Over the last 18 years, the annual mean for spinal fusion procedures performed in the USA was 317,534 [2]. It has been well established in the spine literature that the gold standard for achieving arthrodesis is iliac crest bone graft (ICBG). However, potential complications including donor site morbidity, increased operative time and blood loss, increased length of stay, and limited supply have been reported [3]. As a result, there has been a growing trend in the use of bone graft substitutes and bone graft extenders in an effort to mitigate these risks. Although there are various graft substitutes, none of them has all three characteristics of ICBG: being osteoconductive, osteogenic, and osteoinductive.
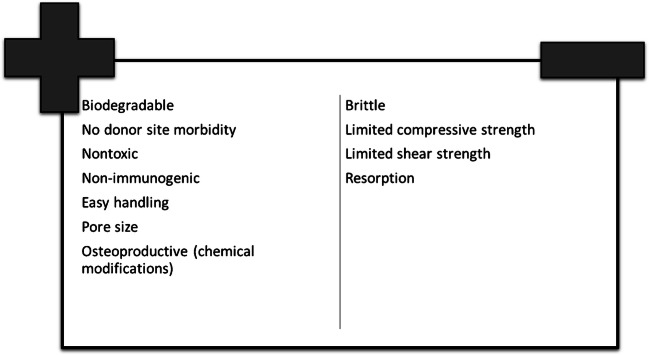
Ceramics are a class of bone graft substitutes that provide osteoconductive properties without any osteogenic or osteoinductive potential [4, 5]. Examples of the most commonly used ceramics include hydroxyapatite (HA), beta-tricalcium phosphate (β-TCP), calcium phosphate (CaP), calcium sulfate, and bioactive synthetics such as silicate-substituted calcium phosphate (Si-CaP) and bioglass (BAG). In newer generation of ceramics, bioactive synthetics go beyond osteoconductive role, and they have osteoproductive characteristics which promote cell stimulation towards osteogenesis and growth factor production. Ceramics are an attractive alternative given that they are an inert substance that is non-toxic, non-immunogenic, and without risk of infection. They are fully customizable, easily stored, and available in virtually unlimited supply. Lastly, when compared with other graft materials, they are also a less expensive option [6]. The main disadvantage is the inherent lack of mechanical strength (Fig. 1). Ceramics are brittle with low fracture resistance and tensile strength [7]. Therefore, they are commonly protected until bone ingrowth has occurred.
Fig. 1.
Pros and cons of ceramics—first to third generations
The purpose of this review was to describe evolution of various ceramics and their use in spine fusion.
Hydroxyapatite
Hydroxyapatite (HA) is one of the most commonly used ceramic in orthopedics. HA is a crystalline structure composed of calcium phosphate and is a major component of the inorganic bone matrix. HA functions as an osteoconductive matrix without any osteogenic cells or osteoinductive potential. HA shares many of the advantages as other types of ceramics, such as being an inert substance that is easy to sterilize and readily available as an off the shelf product. However, one of the main disadvantages is its inherent lack of strength. Therefore, HA is rarely used as a stand-alone bone substitute and must be augmented with instrumented fusion to allow for its incorporation within the surrounding bone. Multiple studies have demonstrated its clinical effectiveness in both the cervical and lumbar spine.
In a prospective randomized controlled trial (RCT) by Korovessis et al., 60 patients with degenerative lumbar stenosis undergoing posterior lumbar fusion (PLF) were divided into three groups: autologous iliac crest bone graft (ICBG) bilaterally, ICBG on one side and HA + local bone (LB) + bone marrow (BM) on the contralateral side, or HA + LB + BM bilaterally [8•]. All three groups, regardless of graft used, progressed to fusion based on radiographs and CT scans performed at 12 months postoperatively. Authors noted incomplete fusion between transverse processes, which they attributed to the progressive resorption of HA that began at the 6-month time point. This, however, had no effect on the overall fusion rate or functional outcomes.
Similarly, HA has also been used to achieve high fusion rates in the cervical spine. Yoshi et al. performed a prospective study evaluating patents who underwent anterior cervical discectomy and fusion (ACDF) with either HA + percutaneously harvested ICBG vs tricortical ICBG alone [9]. They found that at 2-year follow-up, there were no significant differences in recovery rates, fusion rates, or sagittal alignment.
In addition to augmenting fusion rates, HA has also been used as a bone graft extender. Multiple studies have demonstrated that HA can be used a bone graft extender to address some of the drawbacks with ICBG harvest, while at the same time achieving comparable fusion rates to the gold standard [10–12]. Yoo et al. evaluated 88 patients undergoing minimally invasive transformational lumbar interbody fusion (MI-TLIF) [13]. Patients were divided into three groups: group I autograft, group II HA + autograft (> 50%), and group II HA + autograft (< 50%). Fusion rates at 2 years postoperatively increased as the amount of autograft increased (90.9%, 87.8%, and 85.7% for groups I–III respectively); however, there was no statistical significant difference between the groups showing the value of ceramic graft extenders.
Beta-Tricalcium Phosphate
Another commonly used ceramic is β-TCP. β-TCP is a synthetic bone graft substitute similar to normal bone [14]. It is composed of 39% calcium and 20% phosphate [5]. β-TCP shares the same osteoconductive properties of other ceramics, enabling it to function as an effective carrier for both cells and growth factors. When compared with other ceramics such as hydroxyapatite (HA), β-TCP has a higher porosity and therefore resorbs at a much faster rate, typically within 6 weeks [15]. Despite this limitation, multiple studies have used it successfully as a bone graft substitute as well as a bone graft extender.
Dai et al. conducted a prospective randomized control trial (RCT) evaluating fusion rates in patients undergoing instrumented PLF for degenerative lumbar spinal stenosis treated with either β-TCP combined with local autograft or ICBG [16]. Out of the 62 patients included in this study, all 32 patients in β-TCP group and 30 patients in ICBG group demonstrated evidence of radiographic fusion within the 3-year follow-up period. Authors found no significant difference in fusion rates or functional outcome scores between groups at the time of final follow-up. Gan et al. used β-TCP combined with autologous enriched mesenchymal stem cells (MSCs) in 41 patients undergoing posterior spinal fusion (PSF) [17]. At 34.5 months, 95.1% of patients had a successful fusion based on radiographic and clinic assessment. Authors attributed the success of this composite graft to the osteogenic and osteoinductive properties of MSCs that when combined with an osteoconductive scaffold, such as β-TCP, allows for the ingrowth of cells and blood vessels. A systematic review by Nickoli et al. found 30 studies that used ceramics as a bone graft extender in patients undergoing PLF [11]. They found that the fusion rate for β-TCP was 92.5% (319 patients out of 349). This was significantly higher than the fusion rate for all ceramic-based bone grafts, which was 86.4%.
More recently, there have been number of studies that have evaluated the use of β-TCP with newer minimally invasive surgery (MIS) techniques. A retrospective review by Abassi et al. used β-TCP with autologous bone marrow aspirate (BMA) in 24 patients undergoing oblique lateral lumbar interbody fusion (OLLIF) [18]. After discectomy was performed, authors packed all available disc space with β-TCP and BMA. Fusion was confirmed radiographically by two independent observers in all 24 patients at the 1-year follow-up visit.
Efforts have been made to create composite ceramic grafts, often a combination of β-TCP and HA, in an attempt to maximize fusion rates. Multiple studies have used them as standalone bone substitutes as well as bone graft extenders. Parker et al. comparted the use of AttraX (95% β-TCP + 5% HA) with rhBMP-2 in 135 patients undergoing extreme lateral interbody fusion (XLIF). Fusion rate at the 2-year postoperative visit was 80% for β-TCP group compared with 96% for rhBMP-2 group [19•]. However, this difference was only significant when β-TCP was used as a standalone graft. No difference in fusion rate was seen when both groups were supplemented by instrumented fusion. Conversely, Malham et al. used Mastergraft (85% β-TCP + 15% HA) as a bone graft extender by combining it with rhBMP-2 in patients undergoing XLIF [20]. Supplemental fixation was used to avoid subsidence, maintain stability, or correct any deformities. Authors reported a fusion rate of 95.2% for standalone group vs 80% for supplemental fixation group at 1-year follow-up.
Calcium Sulfate
Calcium sulfate has long been used as a bone substitute, particularly in orthopedic trauma, and has been praised for its availability, low-cost, and osteoconductive properties [21]. The crystalline structure of calcium sulfate is similar to that of cancellous bone, providing architecture for the introduction of capillaries and mesenchymal stem cells [22]. The lack of osteoinductive or osteogenic properties, as well as quick resorption time (1–3 months), hinders the use of calcium sulfate as a standalone graft and mandates the use of additional substances such as demineralized bone matrix or local allograft for maximized effect [21, 23].
Owing to the traditional use of calcium sulfate in trauma, a handful of recent studies have shown that pedicle screw fixation is improved with calcium sulfate augmentation in unstable thoracolumbar burst fractures [24–26]. Calcium sulfate injections have also been shown in sheep models to reduce fracture risk in osteoporotic vertebral bodies [27].
Previous reviews have demonstrated similar results between calcium sulfate and autologous graft for lumbar fusion across multiple studies [23•]. However, more recent clinical investigations of calcium sulfate for spinal fusion are limited. In a prospective study of 68 patients with cervical degenerative disc disease being treated with one- or two-level discectomy, Xie et al. showed comparable results between polyether-ether-ketone (PEEK) interbody cages with calcium sulfate/demineralized bone matrix (CS/DBM) and those with autogenous iliac cancellous bone [28]. At 12-month follow-up, the CS/DBM group showed 94.3% fusion compared with 100% in the ICBG group. Both groups had 100% fusion at final follow-up (24 months). No significant difference was found in follow-up clinical symptom score or lordotic angle between the two groups. The complication rate for the ICBG group (18.2%) was significantly higher than the CS/DBM group (8.6%) [28].
PMMA
Polymethylmethacrylate (PMMA) has been a staple of orthopedic surgery for years. Traditionally utilized as a cement in total joint replacement, PMMA was subsequently expanded into uses such as vertebroplasty and kyphoplasty. The methylmethacrylate monomer is capable of polymerizing at room temperature, is low-cost, and has good biocompatibility, although it is bioinert and thus has minimal bone remodeling capacity [29, 30].
Studies comparing PMMA against other materials for use in cages for anterior cervical fusion have yielded variable results. A review by Noordhoek et al. compared PEEK cages, titanium cages, cage-screw-combinations (CSC), and PMMA cages. Of the seventy-one studies included, only 6 looked at PMMA, but a significantly higher incidence of subsidence occurred in PMMA vs PEEK or CSC (30.2% vs 23.5% and 15.1%) [31]. A recent single-blind randomized controlled study by Farrokhi et al. comparing PEEK cage with a novel PMMA cage enrolled 64 patients that were randomly assigned to undergo ACDF using either the acrylic cage (n = 32) or PEEK cage (n = 32). At 12-month follow-up, there was a statistically significant difference in the disc space height between acrylic and PEEK (5 ± 0.9 mm and 4.25 ± 1 mm, respectively), as well as intervertebral angle correction (3.38 ± 1.9 and 3.16 ± degrees, respectively) [32].
Recently, PMMA has been utilized for pedicle screw augmentation to increase pull-out strength and resistance to failure in osteoporotic patients [33]. Low-quality bone has increased risk for implant failure, and while a number of techniques have been assessed to improve fixation in these patients, PMMA-augmented screws have demonstrated to be most successful [34]. Four retrospective studies (ranging from 28 to 313 osteoporotic patients) examined the safety and efficacy of lumbar fusion using cement injectable cannulated pedicle screws (CICPS) [34–37]. Three studies utilized Visual Analog Scale and Oswestry Disability Index to assess efficacy and showed statistically significant decreases in low back and lower leg pain [35–37]. They also examined complications and found leakage rates between 6.46 and 10.3%, with all cases of leakage being clinically insignificant. The fourth retrospective study, by Martín-Fernández et al., was the largest (313 patients) and showed a leakage rate of 62.3%, none of which showed major clinical complications [34]. The same study found a revision rate of 17.9%, mostly in patients with > 4 level reconstruction.
A prospective study by Cao et al. prospectively compared unilateral transforaminal lumbar interbody fusion using traditional pedicle screws and PMMA-augmented screws in osteoporotic patients [38]. The fusion rate with PMMA was significantly higher (91.3% vs 79.2%), and at 2-year follow-up, there was a significant difference in disc height in the PMMA group vs non-PMMA (10.7 ± 1.6 mm vs 8.7 ± 2.3 mm).
Bioactive Synthetics: Si-CaP and Bioactive Glass
Silicate-substituted calcium phosphate (Si-CaP) is a new generation of ceramics formed by a partial substitution of phosphate with silicate (usually around 0.8% by weight) [39]. Silicate has been shown to have a role in bone metabolism and can upregulate osteoblast proliferation and differentiation, promote osteoinductive gene expression, and increase type I collagen synthesis [40, 41]. Adding silicate to the ceramic increases negative surface charge leading to increased osteoblast attraction to the surface of the material and additionally induces porosity, making it an osteoinductive material [42]. Wheeler et al. compared Si-CaP to iliac crest autograft and found fusion mass to be higher with Si-CaP, while bony bridging was equivalent when compared with autograft treatment [40]. A study by Jenis et al. showed a fusion rate of 76.5% at 24 months of follow-up following posterolateral lumbar fusion [43]. In two separate studies, Alimi and et al. found fusion rates of 90% and 82.9% following spinal fusion (cervical, thoracic, and lumbar procedures included) [39, 44]. Licina et al. compared Si-CaP with rhBMP-2, and fusion was observed in 9/9 and 8/9 patients, respectively, following posterolateral lumbar fusion with similar alleviation of pain and improvement in quality of life [45]. A prospective study by Pimenta et al. compared Si-CaP with rhBMP-2 bone graft [46]. Pimenta et al. reported complete fusion with both graft materials with rhBMP-2 resulting in more rapid early postoperative fusion following XLIF. More recently, Mokawem et al. found a combined fusion rate of 98.9% following TLIF and LLIF using SiCaP-packed 3D-printed lamellar titanium cages, while Bolger et al. found the use of Si-CaP to have a fusion rate of 86.3% at 12 months following PLF surgery [47, 48]. Additionally, Lerner et al. found Si-CaP augmented with BMA to be an effective and safe bone graft extender in scoliosis surgery [49].
Bioactive glass is an osteoconductive bone substitute that is composed of 46.1 mol% SiO2, 24.4 mol% Na2O, 26.9 mol% CaO and 2.6 mol% P2O5 (45S5 bioactive glass) [50]. Bioactive glass bonds strongly to bone due to the formation of an HCA layer which interacts with collagen fibrils of damaged bone [50]. Once this HCA layer is formed, dissolution of calcium and silica ions stimulates attachment of osteoblasts, cell division, and upregulation of osteogenic genes that is dose dependent to ion release [51, 52]. Early animal model studies found bioactive glass to induce either comparable rates of fusion or higher volumes of fusion mass than autograft [53–55]. Ilharreborde et al. conducted a comparative study of bioglass vs iliac crest autograft for spinal fusion in adolescent idiopathic scoliosis and found complete fusion with both graft materials in 88 patients [56]. A study by Frantzen et al. found a higher fusion rates with bioglass with a higher silica content than 45S5 BAG (53.9 vs 46.1 mol% for 45S5) in patients undergoing L4/5 and L5/S1 spinal fusion for spondylolisthesis [57]. A recent study by Barrey et al. assessed 30 patients with a wide range of degenerative and traumatic conditions of the cervical or lumbar spine who underwent spinal fusion with bioactive glass and found an overall fusion rate of 93% at 1-year postop [58].
P-15
Recently, P-15, a synthetic 15-amino acid polypeptide that mimics the cell-binding domain of type-I collagen, has emerged as a bone graft alternative in the setting of spinal fusion. Serving as an attachment site for osteogenic cells to type I collagen, P-15 also stimulates osteoblastogenesis by promoting the differentiation of bone marrow stromal cells to osteoblasts. Furthermore, it enhances the cellular production and subsequent secretion of various growth factors, cytokines, and bone morphogenetic proteins that signal for further osteoblastogenesis [59, 60]. i-Factor Bone Graft (Cerapedics, Inc., Westminster, CO) is a bone graft substitute which consists of P-15 bound to an anorganic bone material (ABM); it therefore contains both osteoconductive and osteoinductive properties. To date, favorable fusion rates with use of P-15 have been demonstrated in a limited number of clinical studies in the setting of cervical fusion [61, 62].
In a single-blinded randomized controlled IDE trial, Arnold et al. demonstrated non-inferiority of i-Factor when compared with local autograft bone in the setting of single-level ACDF utilizing a cortical ring allograft. At 12-month follow-up, fusion rates with use of i-Factor were comparable to that of autograft (88.97% compared with 85.82%, respectively; P = 0.0004) with no significant differences in adverse events or patient reported clinical outcomes between the two groups [61]. In the 2-year follow-up study, overall fusion rates had improved with no significant differences between groups (94.87% vs 93.79% in autograft group; P = 0.2513). However, use of i-Factor was associated with overall improved clinical outcomes when utilizing study defined endpoints of success (fusion, Neck Disability Index > 15, neurological success, and absence of reoperations; 69.83% in the i-Factor group vs 56.35% in the autograft group; P = 0.0302) [62].
In the setting of lumbar fusion surgery, preliminary animal studies have additionally shown P-15 to be a useful fusion adjunct [59, 63]. While high-quality clinical data is limited, in their series of 110 patients who underwent either single or multi-level ALIF utilizing ABM/P-15, Mobb et al. demonstrated high fusion rates (97.5%, 81%, and 100% in single-level, two-level, three-level fusions, respectively) and favorable patient-reported outcome measures at 2-year follow-up [64].
While P-15 may prove to be a useful adjunct in the setting of spinal fusions, additional high-quality and independently conducted clinical studies are warranted to further delineate its clinical utility.
Conclusions
Ceramics have been present in the spine fusion arena for several decades; however, their use has been limited. The major obstacles in published literature are small sample size and different physical and chemical properties resulting in low evidence and a potential risk for bias. Although non-inferiority with the use of ceramics as a bone graft extender when compared with ICBG has been demonstrated, many published studies were not adequately powered to show a significant difference. Even with limitations ceramics hold a promise as a value-based graft material being easily available, relatively inexpensive, and non-immunogenic. Chemical modifications and addition of various elements have a potential to not only enhance cell differentiation and growth factor production but also stimulate vascularization and help prevention of surgical site infection. In patients with risk factors such as osteoporosis, bioactive synthetics can play an important role with addition of strontium which has a potential of inhibiting osteoclast activity. Finally, cost is one of the key variables when it comes to spine conditions and their treatments. Studies have shown that ceramics are less expensive than other graft materials [65]. Well-designed prospective studies on new ceramics and hybrids are warranted to solidify the clinical impact and utility in various spine fusion procedures.
Compliance with Ethical Standards
Conflict of Interest
No conflicts of interest for the current study. Disclosures outside of submitted work: Brandon Ortega, Carson Gardner, Sidney Roberts, and Andrew Chung have no disclosures. Jeffrey Wang: royalties—Biomet, Seaspine, Amedica, DePuy Synthes; investments/options—Bone Biologics, Pearldiver, Electrocore, Surgitech; board of directors—North American Spine Society, AO Foundation (20,000 honorariums for board position, plus travel for board meetings), Cervical Spine Research Society; editorial boards—Spine, the Spine Journal, Clinical Spine Surgery, Global Spine Journal; fellowship funding (paid directly to institution)—AO Foundation. Zorica Buser: consultancy—Cerapedics, The Scripps Research Institute, Xenco Medical (past), AO Spine (past); research support—SeaSpine (past, paid to the institution), Next Science (paid directly to institution), Motion Metrics (paid directly to institution); North American Spine Society—committee member; Lumbar Spine Society—co-chair research committee; AOSpine Knowledge Forum Degenerative—associate member; AOSNA Research Committee—committee member.
Footnotes
This article is part of the Topical Collection on Updates in Spine Surgery—Techniques, Biologics, and Non-operative Management
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Brandon Ortega, Email: Brandon.Ortega@med.usc.edu.
Carson Gardner, Email: chgardne@usc.edu.
Sidney Roberts, Email: sirobert@usc.edu.
Andrew Chung, Email: andrewchung84@gmail.com.
Jeffrey C. Wang, Email: jeffrey.wang@med.usc.edu
Zorica Buser, Email: zbuser@usc.edu.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
- 1.Buser Z, Ortega B, D’Oro A, Pannell W, Cohen JR, Wang J, et al. Spine degenerative conditions and their treatments: national trends in the United States of America. Glob Spine J. 2018;8(1):57–67. doi: 10.1177/2192568217696688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.John J, Mirahmadizadeh A, Seifi A. Association of insurance status and spinal fusion usage in the United States during two decades. J Clin Neurosci. 2018;51:80–84. doi: 10.1016/j.jocn.2018.02.013. [DOI] [PubMed] [Google Scholar]
- 3.Gruskay JA, Basques BA, Bohl DD, Webb ML, Grauer JN. Short-term adverse events, length of stay, and readmission after iliac crest bone graft for spinal fusion. Spine. 2014;39(20):1718–1724. doi: 10.1097/BRS.0000000000000476. [DOI] [PubMed] [Google Scholar]
- 4.Tay BK, Patel VV, Bradford DS. Calcium sulfate and calcium phosphate-based bone substitutes. Mimicry of the mineral phase of bone. Orthop Clin North Am. 1999;30:615–623. doi: 10.1016/S0030-5898(05)70114-0. [DOI] [PubMed] [Google Scholar]
- 5.Vaz K, Verma K, Protopsaltis T, Schwab F, Lonner B, Errico T. Bone grafting options for lumbar spine surgery: a review examining clinical efficacy and complications. SAS J. 2010;4(3):75–86. doi: 10.1016/j.esas.2010.01.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Khan SN, Fraser JF, Sandhu HS, Cammisa FP, Jr, Girardi FP, Lane JM. Use of osteopromotive growth factors, demineralized bone matrix, and ceramics to enhance spinal fusion. J Am Acad Orthop Surg. 2005;13(2):129–137. doi: 10.5435/00124635-200503000-00006. [DOI] [PubMed] [Google Scholar]
- 7.Cook RW, Hsu WK. Ceramics: clinical evidence for ceramics in spine fusion. Semin Spine Surgery. 2016;28(4):217–225. doi: 10.1053/j.semss.2016.08.004. [DOI] [Google Scholar]
- 8.Korovessis P, Koureas G, Zacharatos S, Papazisis Z, Lambiris E. Correlative radiological, self-assessment and clinical analysis of evolution in instrumented dorsal and lateral fusion for degenerative lumbar spine disease. Autograft versus coralline hydroxyapatite. Eur Spine J. 2005;14:630–638. doi: 10.1007/s00586-004-0855-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yoshii T, Yuasa M, Sotome S, Yamada T, Sakaki K, Hirai T, Taniyama T, Inose H, Kato T, Arai Y, Kawabata S, Tomizawa S, Enomoto M, Shinomiya K, Okawa A. Porous/dense composite hydroxyapatite for anterior cervical discectomy and fusion. Spine. 2013;38(10):833–840. doi: 10.1097/BRS.0b013e3182801390. [DOI] [PubMed] [Google Scholar]
- 10.Lee JH, Hwang CJ, Song BW, Koo KH, Chang BS, Lee CK. A prospective consecutive study of instrumented posterolateral lumbar fusion using synthetic hydroxyapatite (Bongros-HA) as a bone graft extender. J Biomed Mater Res A. 2009;90(3):804–810. doi: 10.1002/jbm.a.32113. [DOI] [PubMed] [Google Scholar]
- 11.Nickoli MS, Hsu WK. Ceramic-based bone grafts as a bone grafts extender for lumbar spine arthrodesis: a systematic review. Glob Spine J. 2014;4(3):211–216. doi: 10.1055/s-0034-1378141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Garin C, Boutrand S. Natural hydroxyapatite as a bone graft extender for posterolateral spine arthrodesis. Int Orthop. 2016;19. [DOI] [PubMed]
- 13.Yoo JS, Min SH, Yoon SH. Fusion rate according to mixture ratio and volumes of bone graft in minimally invasive transforaminal lumbar interbody fusion: minimum 2-year follow-up. Eur J Orthop Surg Traumatol. 2015;25:183–189. doi: 10.1007/s00590-014-1529-6. [DOI] [PubMed] [Google Scholar]
- 14.Brandoff JF, Silber JS, Vaccaro AR. Contemporary alternatives to synthetic bone grafts for spine surgery. Am J Orthop. 2008;37:410–414. [PubMed] [Google Scholar]
- 15.Jarcho M. Calcium phosphate ceramics as hard tissue prosthetics. Clin Orthop. 1981;157:259–278. [PubMed] [Google Scholar]
- 16.Dai LY, Jiang LS. Single-level instrumented posterolateral fusion of lumbar spine with beta-tricalcium phosphate versus autograft: a prospective, randomized study with 3-year follow-up. Spine. 2008;33(12):1299–1304. doi: 10.1097/BRS.0b013e3181732a8e. [DOI] [PubMed] [Google Scholar]
- 17.Gan Y, Dai K, Zhang P, Tang T, Zhu Z, Lu J. The clinical use of enriched bone marrow stem cells combined with porous beta-tricalcium phosphate in posterior spinal fusion. Biomaterials. 2008;29:3973–3982. doi: 10.1016/j.biomaterials.2008.06.026. [DOI] [PubMed] [Google Scholar]
- 18.Abbasi H, Miller L, Abbasi A, Orandi V, Khaghany K. Minimally invasive scoliosis surgery with oblique lateral lumbar interbody fusion: single surgeon feasibility study. Cureus. 2017;9(6):e1389. doi: 10.7759/cureus.1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Parker RM, Malham GM. Comparison of a calcium phosphate bone substitute with recombinant human bone morphogenetic protein-2: a prospective study of fusion rates, clinical outcomes and complications with 24-month follow-up. Eur Spine J. 2017;26(3):754–763. doi: 10.1007/s00586-016-4927-0. [DOI] [PubMed] [Google Scholar]
- 20.Malham GM, Ellis NJ, Parker RM, Blecher CM, White R, Goss B, Seex KA. Maintenance of segmental lordosis and disk height in stand-alone and instrumented extreme lateral interbody fusion (XLIF) Clin Spine Surg. 2017;30(2):E90–E98. doi: 10.1097/BSD.0b013e3182aa4c94. [DOI] [PubMed] [Google Scholar]
- 21.Campana V, Milano G, Pagano E, Barba M, Cicione C, Salonna G, et al. Bone substitutes in orthopaedic surgery: from basic science to clinical practice. J Mater Sci Mater Med. 2014;25:2445–2461. doi: 10.1007/s10856-014-5240-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Roberts TT, Rosenbaum AJ. Bone grafts, bone substitutes and orthobiologics. Organogenesis. 2012;8(4):114–124. doi: 10.4161/org.23306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Buser Z, Brodke DS, Youssef JA, Meisel HJ, Myhre SL, Hashimoto R, et al. Synthetic bone graft versus autograft or allograft for spinal fusion: a systematic review. J Neurosurg. 2016;25(4):509–516. doi: 10.3171/2016.1.SPINE151005. [DOI] [PubMed] [Google Scholar]
- 24.Kao FC, Hsieh MK, Yu CW, Tsai TT, Lai PL, Niu CC, Chen LH, Chen WJ. Additional vertebral augmentation with posterior instrumentation for unstable thoracolumbar burst fractures. Injury. 2017;48(8):1806–1812. doi: 10.1016/j.injury.2017.06.015. [DOI] [PubMed] [Google Scholar]
- 25.Liao J, Chen W, Wang H. Treatment of thoracolumbar burst fractures by short-segment pedicle screw fixation using a combination of two additional pedicle screws and vertebroplasty at the level of the fracture: a finite element analysis. BMC Musculoskelet Disord. 2017;18:262(18). doi: 10.1186/s12891-017-1623-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Liao J, Fan K. Posterior short-segment fixation in thoracolumbar unstable burst fractures – transpedicular grafting or six-screw construct? Clin Neurol Neurosurg. 2017;153:56–63. doi: 10.1016/j.clineuro.2016.12.011. [DOI] [PubMed] [Google Scholar]
- 27.Liu D, Wu ZX, Zhang Y, Wang CR, Xie QY, Gong K, Zhang B, Quan Y, Pan XM. Local treatment of osteoporotic sheep vertebral body with calcium sulfate for decreasing the potential fracture risk: microstructural and biomechanical evaluations. Clin Spine Surg. 2016;29(7):E358–E364. doi: 10.1097/BSD.0b013e3182a22a96. [DOI] [PubMed] [Google Scholar]
- 28.Xie Y, Li H, Yuan J, Fu L, Yang J, Zhang P. A prospective randomized comparison of PEEK cage containing calcium sulphate or demineralized bone matrix with autograft in anterior cervical interbody fusion. Int Orthop. 2015;39:1129–1136. doi: 10.1007/s00264-014-2610-. [DOI] [PubMed] [Google Scholar]
- 29.Hoppe S, Keel MJB. Pedicle screw augmentation in osteoporotic spine: indications, limitations and technical aspects. Eur J Trauma Emerg Surg. 2017;43:3–8. doi: 10.1007/s00068-016-0750-x. [DOI] [PubMed] [Google Scholar]
- 30.Webb JCJ, Spencer RF. The role of polymethylmethacrylate bone cement in modern orthopaedic surgery. J Bone Joint Surg Br. 2007;89-B:851–857. doi: 10.1302/0301-620X.89B7.19148. [DOI] [PubMed] [Google Scholar]
- 31.Noordhoek I, Koning MT, Jacobs WCH, Vleggeert-Lankamp CLA. Incidence and clinical relevance of cage subsidence in anterior cervical discectomy and fusion: a systematic review. Acta Neurochir. 2018;160:873–880. doi: 10.1007/s00701-018-3490-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Farrokhi M, Nikoo Z, Gholami M, Hosseini K. Comparison between acrylic cage and polyetheretherketone (PEEK) cage in single-level anterior cervical discectomy and fusion: randomized clinical trial. Clin Spine Surg. 2017;30:38–46. doi: 10.1097/BSD.0000000000000251. [DOI] [PubMed] [Google Scholar]
- 33.Elder BD, Lo SF, Holmes C, Goodwin CR, Kosztowski TA, Lina IA, et al. The biomechanics of pedicle screw augmentation with cement. Spine J. 2015;15(6):1432–1445. doi: 10.1016/j.spinee.2015.03.016. [DOI] [PubMed] [Google Scholar]
- 34.Martín-Fernández M, López-Herradón A, Piñera AR, Tomé-Bermejo F, Duart JM, Vlad MD, Rodríguez-Arguisjuela MG, Alvarez-Galovich L. Potential risks of using cement-augmented screws for spinal fusion in patients with low bone quality. Spine J. 2017;17:1192–1199. doi: 10.1016/j.spinee.2017.04.029. [DOI] [PubMed] [Google Scholar]
- 35.Wang Z, Liu Y, Rong Z, Wang C, Liu X, Zhang F, Zhang Z, Xu J, Dai F. Clinical evaluation of a bone cement-injectable cannulated pedicle screw augmented with polymethylmethacrylate: 128 osteoporotic patients with 42 months of follow up. Clinics. 2019;74:1–10. doi: 10.6061/clinics/2019/e346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rong Z, Zhang F, Xiao J, Wang Z, Luo F, Zhang Z, Xu J, Dai F. Application of cement-injectable cannulated pedicle screw in treatment of osteoporotic thoracolumbar vertebral compression fracture (AO type A): a retrospective study of 28 cases. World Neurosurg. 2018;120:e247–e258. doi: 10.1016/j.wneu.2018.08.045. [DOI] [PubMed] [Google Scholar]
- 37.Dai F, Liu Y, Zhang F, Sun D, Luo F, Zhang Z, Xu J. Surgical treatment of the osteoporotic spine with bone cement-injectable cannulated pedicle screw fixation: technical description and preliminary application in 43 patients. Clinics. 2015;70:114–119. doi: 10.6061/clinics/2015(02)08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cao Y, Liang Y, Wan S, Jiang C, Jiang X, Chen Z. Pedicle screw with cement augmentation in unilateral transforaminal lumbar interbody fusion: a 2-year follow-up study. World Neurosurg. 2018;118:e288–e295. doi: 10.1016/j.wneu.2018.06.181. [DOI] [PubMed] [Google Scholar]
- 39.Nagineni VV, James AR, Alimi M, Hofstetter C, Shin BJ, Njoku I, Jr, Tsiouris AJ, Härtl R. Silicate-substituted calcium phosphate ceramic bone graft replacement for spinal fusion procedures. Spine (Phila Pa 1976) 2012;37:E1264–E1272. doi: 10.1097/BRS.0b013e318265e22e. [DOI] [PubMed] [Google Scholar]
- 40.Wheeler DL, Jenis LG, Kovach ME, Marini J, Turner AS. Efficacy of silicated calcium phosphate graft in posterolateral lumbar fusion in sheep. Spine J. 2007;7:308–317. doi: 10.1016/j.spinee.2006.01.005. [DOI] [PubMed] [Google Scholar]
- 41.Carlisle EM. Silicon: a possible factor in bone calcification. Science. 1970;167:279–280. doi: 10.1126/science.167.3916.279. [DOI] [PubMed] [Google Scholar]
- 42.Cameron K, Travers P, Chander C, Buckland T, Campion C, Noble B. Directed osteogenic differentiation of human mesenchymal stem/precursor cells on silicate substituted calcium phosphate. J Biomed Mater Res A. 2013;101:13–22. doi: 10.1002/jbm.a.34261. [DOI] [PubMed] [Google Scholar]
- 43.Jenis LG, Banco RJ. Efficacy of silicate-substituted calcium phosphate ceramic in posterolateral instrumented lumbar fusion. Spine (Phila Pa 1976) 2010;35:E1058–E1063. doi: 10.1097/BRS.0b013e3181df196f. [DOI] [PubMed] [Google Scholar]
- 44.Alimi M, Navarro-Ramirez R, Parikh K, Njoku I, Hofstetter CP, Tsiouris AJ, Härtl R. Radiographic and clinical outcome of silicate-substituted calcium phosphate (Si-CaP) ceramic bone graft in spinal fusion procedures. Clin Spine Surg. 2017;30:E845–EE52. doi: 10.1097/BSD.0000000000000432. [DOI] [PubMed] [Google Scholar]
- 45.Licina P, Coughlan M, Johnston E, Pearcy M. Comparison of silicate-substituted calcium phosphate (Actifuse) with recombinant human bone morphogenetic protein-2 (infuse) in posterolateral instrumented lumbar fusion. Glob Spine J. 2015;5:471–478. doi: 10.1055/s-0035-1566230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Pimenta L, Marchi L, Oliveira L, Coutinho E, Amaral R. A prospective, randomized, controlled trial comparing radiographic and clinical outcomes between stand-alone lateral interbody lumbar fusion with either silicate calcium phosphate or rh-BMP2. J Neurol Surg A Cent Eur Neurosurg. 2013;74:343–350. doi: 10.1055/s-0032-1333420. [DOI] [PubMed] [Google Scholar]
- 47.Mokawem M, Katzouraki G, Harman CL, Lee R. Lumbar interbody fusion rates with 3D-printed lamellar titanium cages using a silicate-substituted calcium phosphate bone graft. J Clin Neurosci. 2019;68:134–139. doi: 10.1016/j.jocn.2019.07.011. [DOI] [PubMed] [Google Scholar]
- 48.Bolger C, Jones D, Czop S. Evaluation of an increased strut porosity silicate-substituted calcium phosphate, SiCaP EP, as a synthetic bone graft substitute in spinal fusion surgery: a prospective, open-label study. Eur Spine J. 2019;28:1733–1742. doi: 10.1007/s00586-019-05926-1. [DOI] [PubMed] [Google Scholar]
- 49.Lerner T, Liljenqvist U. Silicate-substituted calcium phosphate as a bone graft substitute in surgery for adolescent idiopathic scoliosis. Eur Spine J. 2013;22(Suppl 2):S185–S194. doi: 10.1007/s00586-012-2485-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jones JR. Review of bioactive glass: from Hench to hybrids. Acta Biomater. 2013;9:4457–4486. doi: 10.1016/j.actbio.2012.08.023. [DOI] [PubMed] [Google Scholar]
- 51.Xynos ID, Edgar AJ, Buttery LD, Hench LL, Polak JM. Gene-expression profiling of human osteoblasts following treatment with the ionic products of Bioglass 45S5 dissolution. J Biomed Mater Res. 2001;55:151–157. doi: 10.1002/1097-4636(200105)55:2<151::AID-JBM1001>3.0.CO;2-D. [DOI] [PubMed] [Google Scholar]
- 52.Kaufmann EA, Ducheyne P, Shapiro IM. Effect of varying physical properties of porous, surface modified bioactive glass 45S5 on osteoblast proliferation and maturation. J Biomed Mater Res. 2000;52:783–796. doi: 10.1002/1097-4636(20001215)52:4<783::AID-JBM24>3.0.CO;2-J. [DOI] [PubMed] [Google Scholar]
- 53.Lindfors NC, Tallroth K, Aho AJ. Bioactive glass as bone-graft substitute for posterior spinal fusion in rabbit. J Biomed Mater Res. 2002;63:237–244. doi: 10.1002/jbm.10177. [DOI] [PubMed] [Google Scholar]
- 54.Bergman SALL. Bone in-fill of non-healing calvarial defects using particulate bioglass and autogenous bone. Bioceramics. 1995;8:17–21. [Google Scholar]
- 55.BW C. The use of Bioglass for spinal arthrodesis and iliac crest repair-an in vivo sheep model. In I O ed. Proceedings of the North American Spine Society, Annual Meeting, San Francisco, CA, October 28–31.: Elsevier Science, 1998:214–6.
- 56.Ilharreborde B, Morel E, Fitoussi F, Presedo A, Souchet P, Penneçot GF, Mazda K. Bioactive glass as a bone substitute for spinal fusion in adolescent idiopathic scoliosis: a comparative study with iliac crest autograft. J Pediatr Orthop. 2008;28:347–351. doi: 10.1097/BPO.0b013e318168d1d4. [DOI] [PubMed] [Google Scholar]
- 57.Frantzén J, Rantakokko J, Aro HT, Heinänen J, Kajander S, Gullichsen E, Kotilainen E, Lindfors NC. Instrumented spondylodesis in degenerative spondylolisthesis with bioactive glass and autologous bone: a prospective 11-year follow-up. J Spinal Disord Tech. 2011;24:455–461. doi: 10.1097/BSD.0b013e31822a20c6. [DOI] [PubMed] [Google Scholar]
- 58.Barrey C, Broussolle T. Clinical and radiographic evaluation of bioactive glass in posterior cervical and lumbar spinal fusion. Eur J Orthop Surg Traumatol. 2019;29:1623–1629. doi: 10.1007/s00590-019-02477-5. [DOI] [PubMed] [Google Scholar]
- 59.Sherman BP, Lindley EM, Turner AS, Seim HB, 3rd, Benedict J, Burger EL, et al. Evaluation of ABM/P-15 versus autogenous bone in an ovine lumbar interbody fusion model. Eur Spine J Off Publ Eur Spine Soc Eur Spinal Deform Soc Eur Sect Cerv Spine Res Soc. 2010;19(12):2156–2163. doi: 10.1007/s00586-010-1546-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yang XB, Bhatnagar RS, Li S, Oreffo ROC. Biomimetic collagen scaffolds for human bone cell growth and differentiation. Tissue Eng. 2004;10(7–8):1148–1159. doi: 10.1089/ten.2004.10.1148. [DOI] [PubMed] [Google Scholar]
- 61.Arnold PM, Sasso RC, Janssen ME, Fehlings MG, Smucker JD, Vaccaro AR, Heary RF, Patel AI, Goulet B, Kalfas IH, Kopjar B. Efficacy of i-factor bone graft versus autograft in anterior cervical discectomy and fusion: results of the prospective, randomized, single-blinded Food and Drug Administration investigational device exemption study. Spine. 2016;41(13):1075–1083. doi: 10.1097/BRS.0000000000001466. [DOI] [PubMed] [Google Scholar]
- 62.Arnold PM, Sasso RC, Janssen ME, Fehlings MG, Heary RF, Vaccaro AR, et al. i-FactorTM bone graft vs autograft in anterior cervical discectomy and fusion: 2-year follow-up of the randomized single-blinded Food and Drug Administration investigational device exemption study. Neurosurgery. 2018;83(3):377–384. doi: 10.1093/neuros/nyx432. [DOI] [PubMed] [Google Scholar]
- 63.Axelsen MG, Overgaard S, Jespersen SM, Ding M. Comparison of synthetic bone graft ABM/P-15 and allograft on uninstrumented posterior lumbar spine fusion in sheep. J Orthop Surg. 2019;14(1):2. doi: 10.1186/s13018-018-1042-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Mobbs RJ, Maharaj M, Rao PJ. Clinical outcomes and fusion rates following anterior lumbar interbody fusion with bone graft substitute i-FACTOR, an anorganic bone matrix/P-15 composite. J Neurosurg Spine. 2014;21(6):867–876. doi: 10.3171/2014.9.SPINE131151. [DOI] [PubMed] [Google Scholar]
- 65.Skovrlj B, Guzman JZ, Al Maaieh M, Cho SK, Iatridis JC, Qureshi SA. Cellular bone matrices: viable stem cell-containing bone graft substitutes. Spine J. 2014;14(11):2763–2772. doi: 10.1016/j.spinee.2014.05.024. [DOI] [PMC free article] [PubMed] [Google Scholar]