Abstract
Impairments in early-stage visual processing are observed in chronic psychosis. However, their presence, localization within the brain, and contribution to cognitive symptoms remain less well established early in disease course. The present study utilized magnetoencephalography (MEG) to examine sensory responses within primary visual cortex (V1). MEG was recorded from 38 individuals diagnosed with a schizophrenia spectrum illness at first psychotic episode (FESz) and 38 matched healthy controls (HC) during visual search tasks. The inverse solution for cortical activity contributing to the M100 visual evoked field was derived. Task performance and V1 activation were compared between groups. FESz exhibited a reduced V1 response relative to HC. This group deficit, however, was selective for the left hemisphere (LH). A similar interaction was observed for response time with FESz exhibiting slower responses to right visual field targets, a difference not observed among HC. Among FESz, larger LH V1 activity was associated with larger hallucination subscale scores on the Scale for the Assessment of Positive Symptoms. Early-stage visual processing deficits localized to V1 are present at disease onset in the schizophrenia spectrum. This impairment appears to be restricted to the LH, consistent with previous reports detailing a predominantly LH disease process in early psychosis, and activity within this region was associated with an increased experience of hallucinations. These findings detail the cortical responses contributing to visual processing impairments and their relationship with symptoms at disease onset, advancing our understanding of their developmental trajectory over the course of psychotic illness.
Keywords: schizophrenia spectrum, first-break, sensory impairment, lateralization
Introduction
Visual processing deficits have been well documented in schizophrenia using both behavioral paradigms such as backwards masking1,2 and neurophysiological techniques. In the latter case, deficits in visual evoked potentials (VEPs) recorded using electroencephalography (EEG) including the P13–6 and, to a lesser extent, C17,8 have been observed. Both VEPs are considered early-latency cortical components of the brain’s response to visual stimuli reflective of initial sensory-perceptual processes.9 In addition, there has been a growing appreciation for the contribution of these early-stage visual impairments to the more well-established deficits in selective attention and working memory.10,11 These findings emphasize the importance of dysfunctional sensory processes in psychosis and their role as potential mediators of its associated cognitive deficits, symptoms highly predictive of long-term functional outcomes.12–14 As a result, significant work has been dedicated to determining their utility as potential biomarkers of disease15,16 as well as targets for therapeutic interventions.17,18
Despite the association between impaired early-stage visual processing deficits and community functioning19,20 as well as the established impact of early interventions on these outcome measures,21,22 visual processing deficits early in disease course remain relatively understudied. Although most of the research in this area has focused on individuals in chronic stages of their illness, there has been one study that identified P1 deficits in adolescent cases of schizophrenia23 and another reporting a similar impairment at first psychotic break.24 In the study of adolescent onset psychosis, reductions in P1 were associated with deficits in working memory encoding, a similar relationship to that observed in chronic stages of the illness.10 Further investigation into the development of these sensory-perceptual visual processing deficits and their contribution to cognitive dysfunctions during early stages of a schizophrenia spectrum illness may provide novel information about primary disease processes and new targets for sensory remediation as therapeutic interventions.
Characterization of the neural mechanisms underlying these sensory deficits is equally critical to identifying potential treatment targets. Studies of visual processing conducted among chronic disease populations have applied inverse modeling to VEP deficits recorded at scalp EEG electrodes, localizing these impairments to early visual cortices as well as various regions along the dorsal visual stream.4,6 In an attempt to obtain a more refined localization of cortical deficits, studies have also combined EEG with fMRI. Using this approach, reductions in BOLD signals, a correlate of neural activity, have been observed within primary (V1) and secondary (V2) visual cortices.25 However, in contrast to EEG that suffers from tissue boundary- and scalp-induced distortions of electrical fields and the delayed hemodynamic response used to generate fMRI BOLD signals, magnetoencephalography (MEG) affords high spatial and temporal resolution combined in a single modality unaffected by different tissue conductivities.26 This precision becomes particularly important when interrogating sensory and perceptual processes operating on a millisecond time scale under the control of a complex network of cortical structures. Within the MEG visual processing literature, the M100 visual evoked field (VEF) is considered analogous to the P1 VEP. Leveraging the enhanced precision of this imaging modality, cortical source localization of the M100 VEF has identified V1 as its primary generator.27,28
The present investigation was designed to determine whether early-stage visual processing deficits were present among individuals with a schizophrenia spectrum illness following their first psychotic episode (FESz). To interrogate disruptions in the initial stages of visual processing, MEG was used to identify a reliable marker of early sensory-perceptual processing (ie, the M100) and distributed source modeling was applied to localize the cortical activity contributing to it within V1. Data were recorded during 2 visual search tasks that differentially emphasized either stimulus-driven, parallel search strategies (ie, a pop-out) or a controlled serial search of stimuli (ie, a serial search). We predicted impaired processing of target stimuli within V1 and associations between this deficit and performance on the visual search task. An effect of the attentional manipulation in the visual search task was not expected at this early stage of stimulus processing.
Methods
Participants
Participants included FESz and healthy controls (HC). Eighty-four participants (38 HC; 46 FESz) completed the study. Two FESz were excluded due to poor-quality MEG data. To ensure groups were matched on age, gender, Wechsler Abbreviated Scale of Intelligence IQ, and parental socioeconomic status (PSES), 6 additional FESz were excluded from analysis resulting in a final sample of 38 HC and 38 FESz. These 6 FESz individuals were excluded due to low PSES scores. Data from a subset of these participants (32 HC; 32 FESz) were included in a separate manuscript exploring neurophysiological markers of selective attention during visual search (Sklar et al, under review).
FESz were recruited from UPMC Western Psychiatric Hospital inpatient and outpatient services. The Structured Clinical Interview for DSM-IV (SCID-P) was used to establish diagnoses. Twenty-three FESz were diagnosed with schizophrenia, 6 with schizoaffective disorder (4 depressive and 2 bipolar types), and 9 with psychotic disorder not otherwise specified. FESz participated within 2 months of their first clinical contact and had less than 2 months of lifetime treatment with an antipsychotic medication. Thirteen (34%) were unmedicated at the time of testing. All patients were clinically stable at the time of testing.
All participants were screened for colorblindness using pseudoisochromatic plates and had at least 9 years of schooling. None had a history of head injury with sequelae, alcohol or drug addiction, or neurological comorbidity. All participants completed the MATRICS Cognitive Consensus Battery and FESz were assessed using the Scale for the Assessment of Negative Symptoms (SANS) and Scale for the Assessment of Positive Symptoms (SAPS).
All procedures were approved by the University of Pittsburgh IRB and participants provided informed consent and were paid for participation.
Stimuli and Procedures
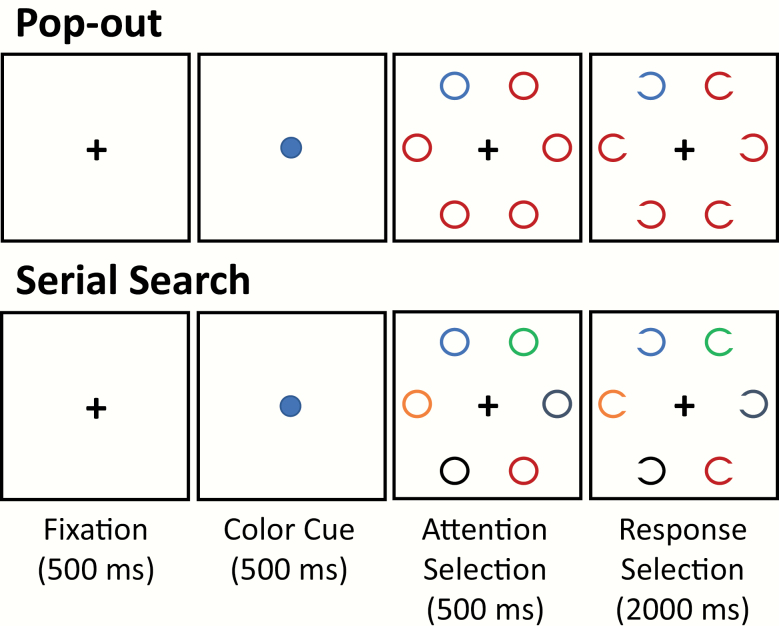
During the testing session, participants completed 2 visual search tasks depicted in figure 1. Trials from each task began with a centrally presented fixation cross (1°, 500 ms) followed by a central cue (0.65°, 500 ms) depicting the color of the to-be-presented target. A stimulus array (500 ms) comprised of 6 eccentrically arranged annuli, each subtending 0.65° and positioned 2° from fixation, was then presented. One annulus, presented in either the right (RVF) or left (LVF) visual field, was identified as the target based on its color. Finally, a small gap then appeared on either the right or left side of each annulus in the array. Participants were required to indicate the side of the gap on the target annulus via button-press within a 2000-ms time window. If the participant responded correctly on 0 or 1 of the previous 3 trials, the gap was 0.65°, 0.33° if 2 of 3 correct responses and 0.22° if the previous 3 responses were correct.
Fig. 1.
Pop-out and serial search tasks. Participants were instructed to identify the target annulus based on the color cue and then indicate via button-press the side on which the gap appears on the target.
During the pop-out (PO) task, all distractor annuli had a uniform color in contrast to the serial search (SS) task in which all colors of distractor annuli were unique. PO and SS tasks were run as separate blocks and their order was counterbalanced across participants in each group. Each block consisted of 144 trials, 72 with RVF targets and 72 with LVF targets.
MEG Recordings and Pre-processing
MEG data were recorded in a magnetically shielded room using a 306-channel whole-head system (Elekta Neuromag). Channels were arranged in 128 triplets, each comprising 1 magnetometer and 2 planar gradiometers. Data were recorded using a sampling rate of 1000 Hz (online bandpass filter = 0.1–330 Hz). Bipolar leads placed above and below the left eye (VEOG) and lateral to the outer canthi of both eyes (HEOG) recorded blinks and eye movements and bipolar ECG leads recorded cardiac activity. A 3D-digitizer (ISOTRAK; Polhemus, Inc., Colchester, VT) was used to record the location of 4 head position indicator coils placed on the scalp of each participant relative to their nasion and preauricular points. Head position was monitored continuously throughout the experiment. Neuromag MaxFilter software (http://imaging.mrc-cbu.cam.ac.uk/meg/Maxfilter_V2.2) was used to correct for head motion during the scan.
Electromagnetic noise originating from outside the MEG helmet was separated from brain signal using the temporal extension of the Signal Space Separation method.29 The MATLAB-based EEGLAB Toolbox30 was used to remove channels (interpolated following completion of pre-processing) and segments of data corrupted by excessive noise. There was no difference between HC (M = 2.2; SD = 1.9) and FESz (M = 2.9; SD = 2.2) in the number of channels interpolated (t74 = −1.5; P = .14). A high-pass filter (0.5 Hz; 12 dB/oct) was applied to the data, and an adaptive mixture independent component analysis was performed to remove eye-blink and ECG components.
Structural MRI scans, necessary for accurate cortical localization of MEG activity, were obtained for each participant. T1-weighted sagittal images were obtained using a Siemens TIM Trio 3 Tesla MRI system with a multi-echo 3D MPRAGE sequence (TR/TE/TI = 2530/1.74, 3.6, 5.46, 7.32/1260 ms, flip angle = 7°, field of view = 220 × 220 mm, 1-mm isotropic voxel size, 176 slices, GRAPPA acceleration factor = 2).
MEG Sensor Processing
Offline processing VEFs was conducted using BrainVision Analyzer2 (Brain Products GMBH). A low-pass (100 Hz; 24 dB/oct) filter was applied, followed by segmentation of the data into 600-ms epochs, including a 100-ms baseline window, constructed around the presentation of the stimulus array during each trial. Segments with data exceeding ± 5pT were excluded, and the remaining trial segments were averaged.
MEG Source Analysis
MRIlab (Elekta-Neuromag Oy, Helsinki, Finland) was used to register MEG sensor data to each participant’s structural MRI. Possible dipole sources were constrained to the gray/white matter boundary which was segmented from MRI data using Freesurfer (http://www.surfer.nmr.mgh.harvard.edu) and tessellated into an icosohedron with 5-mm spacing between vertices. Cortical activity contributing to the sensor-level data was estimated from the 204 gradiometers using MNE, a distributed inverse source solution. The forward solution, modeled as a single sphere, and the noise covariance matrix calculated from the baseline window were used to create a linear inverse operator using an orientation constraint of 0.4 with depth weighting applied.31,32 The dSPM statistic, an estimate of the signal-to-noise value, was calculated for each vertex normalizing the current estimate to prestimulus baseline variance.
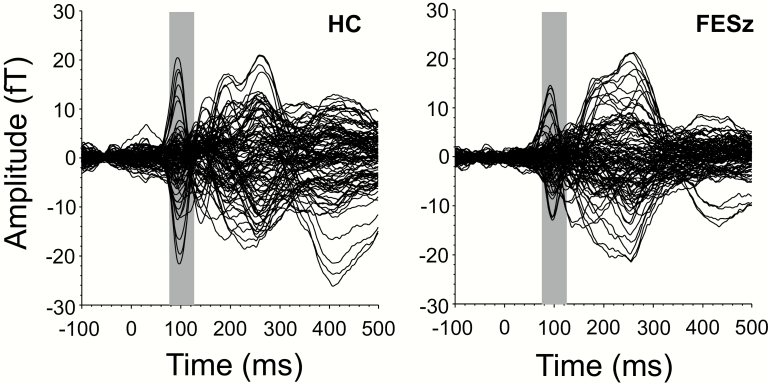
Given potential hemispheric difference in responses to the lateralized targets in the stimulus array, separate segments for LVF and RVF targets were derived. V1, defined using Brodmann Area Maps and Hinds V1 atlas available in Freesurfer, was selected as our a priori region of interest (ROI) given its role as the primary generator of the M100.26,27 Activity within this ROI in each hemisphere was measured over the 75- to 125-ms post-target time window corresponding to the M100 VEF, which is the earliest, clearly observable component depicted in the waveforms of figure 2.
Fig. 2.
Butterfly plots of visual evoked field (VEFs) in response to stimulus array collapsed across tasks. The gray box indicates the 75- to 100-ms time window of interest. Plots depict data recorded from one set of gradiometers.
Data Analysis
Demographic variables (table 1) were compared between groups using chi-square and t-tests. Task accuracy (ACC) and response time (RT) were subjected to a 2 (task: PO/SS) × 2 (group: HC/FESz) × 2 (visual field: LVF/RVF targets) analysis of variance (ANOVA). Given violations of normality, ACC and RT data were subject to log transformation prior to analysis. MEG sensor-level data were not subjected to statistical analyses, but rather used to visualize VEFs and establish a time window for comparison of their associated cortical source activity. A 2 (task: PO/SS) × 2 (group: HC/FESz) × 2 (hemisphere: LH/RH) ANOVA was applied to V1 cortical activity. Independent t-tests were used to follow-up significant effects between groups, and dependent t-tests were used to follow-up significant effects involving task condition and target location observed across groups. An analysis exploring potential effects of diagnostic classification was conducted to ensure that our 3 groups (ie, Schizophrenia, Schizoaffective Disorder, and Psychosis NOS) within the schizophrenia spectrum did not differ regarding outcome measures of interest. There was no main effect of diagnostic group or interaction involving diagnosis for measures of ACC (p’s > .1), RT (p’s > .1), or V1 activity (p’s > .2).
Table 1.
Demographics and Clinical Assessment Data (Mean ± SD) by Group
| HC (n = 38) | FESz (n = 38) | t/χ 2 | P | |
|---|---|---|---|---|
| Age | 22.8 ± 5.0 | 21.8 ± 4.4 | 0.91 | .37 |
| Female/male | 13/25 | 14/24 | 0.06 | 1.0 |
| Education (y) | 14.1 ± 3.2 | 12.4 ± 2.4 | 2.58 | .01 |
| WASI | 108.2 ± 10.6 | 107.3 ± 14.2 | 0.31 | .76 |
| MATRICS-Total | 48.2 ± 7.6 | 38.2 ± 14.3 | 3.79 | .00 |
| SES | 34.2 ± 14.6 | 29.4 ± 13.2 | 1.46 | .15 |
| PSES | 53.0 ± 13.7 | 47.6 ± 15.0 | 1.62 | .11 |
| SANS | — | 31.2 ± 8.0 | ||
| SAPS | — | 19.2 ± 11.6 | ||
| Unmedicated/medicated | — | 13/25 | ||
| Medication (CPZ mg/d) | — | 193.3 ± 165.5 |
Note: HC, healthy controls and FESz, first psychotic episode; CPZ, chlorpromazine equivalent dose; PSES, parental socioeconomic status; SANS, Scale for the Assessment of Negative Symptoms; SAPS, Scale for the Assessment of Positive Symptoms; SES, socioeconomic status; WASI, Wechsler Abbreviated Scale of Intelligence. Bolded values represent significant differences between groups.
Results
Task Performance
ACC and RT data are reported in table 2. Overall, FESz responded less accurately than HC during the visual search task (t7 4 = 2.64, P = .01, d = 0.61). ACC was also lower during SS compared with PO across groups (t75 = 3.24, P < .01, d = 0.37). Neither a main effect of visual field (P = .06) nor the interaction between group and target location (P = .08) reached statistical significance. No interactions involving task were observed (P’s > .2).
Table 2.
Results From the Visual Search Tasks (Mean ± SD) by Group and Target Location
| HC | FESz | |||
|---|---|---|---|---|
| LVF Targets | RVF Targets | LVF Targets | RVF Targets | |
| ACC (%) pop-out | 98.0 ± 3.1 | 98.3 ± 2.9 | 96.7 ± 5.4 | 95.2 ± 6.7 |
| ACC (%) serial search | 97.2 ± 2.4 | 96.9 ± 3.2 | 94.7 ± 5.0 | 94.0 ± 6.7 |
| RT (ms) pop-out | 532.9 ± 105 | 534.1 ± 116 | 599.3 ± 147 | 626.0 ± 159 |
| RT (ms) serial search | 530.3 ± 83 | 538.8 ± 85 | 591.0 ± 130 | 609.9 ± 134 |
| Contralateral V1 activity (dSPM) pop-out | 3.87 ± 1.5 | 4.27 ± 1.9 | 3.59 ± 1.5 | 3.40 ± 1.5 |
| Contralateral V1 activity (dSPM) serial search | 4.14 ± 2.0 | 4.51 ± 2.1 | 3.61 ± 1.6 | 3.33 ± 1.1 |
Note: FESz, first psychotic episode and HC, healthy controls; LVF, left visual field and RVF, right visual field; ACC, accuracy and RT, response time.
In addition to responding less accurately, FESz responded more slowly than HC (t74 = 2.66, P = .01, d = 0.61). There was also a main effect of visual field with slower responses to trials with RVF compared with LVF targets across groups (t75 = 3.23, P < .01, d = 0.37). An interaction was present between group and target location (F1,74 = 6.01, P = .02). Although the observed group difference was present for targets presented in both visual fields (LVF: t74 = −2.32, P = .02, d = 0.53; RVF: t74 = −2.69, P < .01, d = 0.62), FESz exhibited slower responses to RVF compared with LVF targets (t37 = 3.03, P < .01, d = 0.49) whereas HC did not (P = .13). There was no effect of task or any interactions involving it on RT (P’s > .05).
Sensor-Level Data
Butterfly plots of averaged waveforms recorded from one set of gradiometers for HC and FESz during both visual search tasks are depicted in figure 2. Although not subjected to statistical analysis, this data were used to identify the 75- to 125-ms time window of the M100 VEF, a homologue of the P1 recorded using EEG.
V1 Cortical Activity
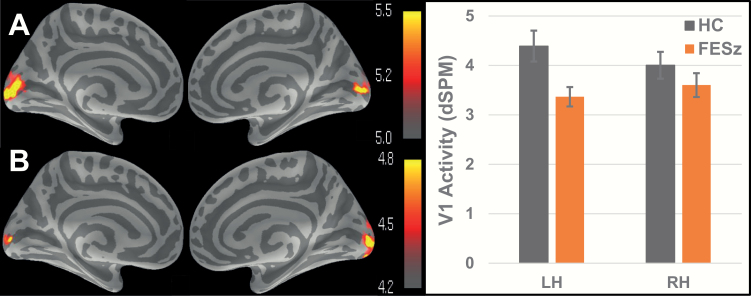
Cortical activity and its quantification averaged across the V1 ROI are depicted in figure 3, and means and SD are reported in table 2. Overall, FESz exhibited significantly reduced V1 activity compared with HC (t74 = −2.06, P = .04, d = 0.47) over the 75- to 125-ms time window. The observed group effect, however, differed based on hemisphere (F1,74 = 7.03, P = .01). FESz showed reduced LH (t74 = 2.78, P < .01, d = 0.64), but not RH (P = .27) V1 activity compared with HC. In addition, HC exhibited larger V1 activity in the LH compared with RH (t37 = 2.09, P = .04, d = 0.34), whereas no hemispheric effect was present among FESz (P = .11). As expected, there were no main effects of task (P = .26) and no interactions involving task (P’s > .1). No main effect of hemisphere was present (P = .52).
Fig. 3.
Cortical activity averaged over 75- to 125-ms post-stimulus array collapsed across tasks for HC (A) and first psychotic episode (FESz) (B). Bar graph depicts activity averaged across the V1 ROI. Of note, the scale has been set differently for healthy controls (HC) and FESz to allow for proper visualization of cortical activity. Error bars represent SEM.
Correlations Between Neurophysiology, Performance, and Clinical Measures
Among healthy controls, larger LH V1 activity was associated with more rapid responses to RVF targets (r = −.45, P < .01) and larger RH V1 activity was associated with more rapid responses to LVF targets (r = −.38, P = .02). There were no significant correlations between any measure of ACC and cortical activity (P’s > .1). In FESz, a similar inverse correlation between LH V1 activity and RVF target RT (r = −.34, P = .04), but not between RH V1 activity and LVF RT (P = .23), was present. There were no significant correlations between ACC measures and V1 activity in FESz (P’s > .2).
Among FESz, activity within LH V1 was positively correlated with total SAPS scores (r = .38; P = .02). This relationship was not present for the RH (P = .1). The association between a larger LH V1 response and higher SAPS scores was present for global ratings on the hallucination subscale (r = .37; P = .03), but not the delusions, bizarre behaviors, or thought disorder subscales (P’s > .1). Among the hallucinatory phenomena, LH V1 activity was associated with auditory (r = .38; P = .02), but not visual (r = .13; P = .43) hallucinations. No significant correlations were observed between V1 activity in either hemisphere and SANS or MATRICS overall and subscale scores (P’s > .05).
Medication Effects
There was no effect of medication status or interactions involving it on V1 activity (P’s > .1) or behavioral (P’s > .1) measure.
Discussion
Although deficits in visual processing have been extensively reported within the schizophrenia spectrum, their presence and the dysfunctional neural mechanisms contributing to them remain understudied at disease onset. The present study extends the existing literature documenting early-stage visual processing impairment in psychosis, identifying its presence at first break and coupling it to dysfunction within the left hemisphere (LH) primary visual cortex at this stage of the illness. In addition, activation within this region in response to visual stimuli, while reduced compared with HC, was associated with reports of increased hallucinatory phenomena. These findings detail impairments in sensory-perceptual processing and their relationship to symptoms afflicting individuals at disease onset, informing potential developmental trajectories of visual processing deficits in schizophrenia spectrum disorders.
The diminished response within V1 to stimuli during visual search may reflect histological changes within V1 in individuals diagnosed with schizophrenia including a reduction in neuronal population and overall volume of this region.33 This impairment is also consistent with anatomical changes such as cortical thinning34 and disrupted white matter integrity35,36 in medial occipital lobe detected using structural imaging techniques. In addition to these cortical abnormalities, impairments in lower-level structures involved in the relay of visual information to the cortex37 might have also contributed to the observed V1 deficit. Unfortunately, these investigations were mostly conducted in individuals during chronic stages of their illness and cannot speak directly to the impaired response observed in our sample. A preliminary analysis examining structural brain difference using the MRIs obtained in the current study revealed no significant differences in gray matter volume between FESz and HC within V1 (unpublished data). However, although structural MRI data were acquired to perform cortical source reconstruction of MEG sensor data, high-resolution T1 and T2 scans necessary for precise volumetric analyses were not obtained. Future studies utilizing scans better optimized for this purpose may reveal group differences not observed in the present data set.
Beyond a simple group effect, the relative impairment among FESz in V1 processing differed by hemisphere. This statistical interaction was in part due to a more robust LH response within HC, a finding previously observed among healthy individuals38 and possibly related to morphologic asymmetries in V1 including increased neuronal size39 and overall cellular density40 of the LH. The absence of a similar lateralization among FESz and their blunted LH V1 response compared with HC suggests an LH lateralized pathology in this patient population. This conclusion is supported by findings of a similar hemispheric interaction in task performance where FESz responds more slowly to targets located in the RVF, a pattern previously reported in patients during chronic stages of psychotic illness.41,42 The lateralized V1 dysfunction is also consistent with the broader schizophrenia spectrum literature identifying both structural43–45 and functional46,47 pathological processes preferentially affecting the LH. However, caution is warranted when drawing conclusions regarding the lateralization of the observed V1 deficit as the proximity of midline cortical structures begins to approach the limit at which MEG can isolate distinct regions of activity with a high degree of confidence.
The biological mechanisms underlying the asymmetry in V1 pathology remain uncertain, though it may reflect dysfunction within the magnocellular visual pathway present in individuals with schizophrenia.48 For example, stimuli presented with high temporal frequency, a characteristic biasing processing toward the magnocellular pathway, are detected more rapidly and with a higher degree of accuracy when presented in the right visual field in healthy individuals.49 However, to date, there has been no study documenting anatomical asymmetry within the magnocellular pathway that would suggest a LH dominance. Another possibility for the lateralization of V1 deficits in FESz relates to asymmetric top-down modulation of this cortical region. Although the V1 deficit observed 75–125 ms following stimulus presentations is suggestive of a purely sensory deficit, this response may represent a relatively late stage of information processing, a point at which cognitive control networks have been shown to exert influence over activity within visual cortices.50,51 In fact, even the earliest cortical responses to visual stimuli recorded within V1 (~50 ms) are susceptible to modulation by top-down processes.52,53 Given the presence of LH lateralized deficits within higher-order frontal and temporal networks in individuals diagnosed with a schizophrenia spectrum illness, it is possible that their modulation of activity within V1 is asymmetric as well.
Although the impaired V1 response among FESz was not associated with MATRICS scores, it was correlated with poorer performance during the visual search task indicating a relationship between deficits in early-stage visual processing and attention. Furthermore, despite reduced LH V1 activity relative to HC, larger stimulus-evoked activity within this region was associated with reports of more significant hallucinatory experiences among FESz. This finding is consistent with previously observed relationships between reports of hallucinations and stimulus-evoked gamma-band oscillations within sensory cortices.54–56 Based on these findings, it has been postulated that baseline hyperexcitability within these sensory regions contributes to both hallucinatory experiences and an impaired stimulus-evoked response. However, in contrast to these previous studies, the current sample exhibited predominantly cross-modal associations between V1 activity and auditory hallucinations. The absence of an intramodal correlation in the current sample is likely the result of the relative lack of visual hallucinations reported (mean SAPS item score of 0.9 with 14 nonhallucinators). Regarding the cross-modal association, the existence of structural connections between auditory and visual cortices57 and the presence of functional connectivity between sensory cortices in individuals with specific sensory impairments58,59 provide plausible explanations a link between pathology in the visual cortex and auditory symptoms. However, the mechanisms underlying this association remain largely speculative at this point and merit further consideration.
Conclusions
The present investigation used MEG recordings to examine early cortical responses evoked by target stimuli during a visual search task and their relationship to down-stream selective attention processes in individuals following their first psychotic episode. Localization of the cortical activity associated with the M100 VEF inculpates dysfunction within the primary visual cortex in sensory-level visual processing deficits at this early stage of the illness. Furthermore, the impaired V1 cortical response appears to be lateralized to the LH, adding to the conceptualization of an asymmetric disease process with pathology spanning across sensory modalities and cognitive domains in the LH. The contribution of higher-order, cognitive control processes to the observed V1 dysfunction at disease onset warrants further examination, as does potential lateralization of previously reported deficits in lower-level structures involved in the relay of visual information to the cortex. Connectivity analyses, both structural (eg, white-matter tractography) and functional (eg, frequency band synchronization and coupling), would also provide a more comprehensive understanding of the complex networks guiding our processing of the visual world and how it fails even at early stages of psychosis.
Acknowledgments
We thank the faculty and staff of the WPH Psychosis Recruitment and Assessment Core and the University of Pittsburgh Clinical Translational Science Institute (UL1 RR024153, Steven E. Reis, MD) for their assistance. None of the authors reported any biomedical financial interests or potential conflicts of interest.
Funding
Funded by National Institute of Mental Health (P50 MH103204) (David Lewis, MD, Director, DFS Project Co-PI).
References
- 1. Saccuzzo DP, Braff DL. Information-processing abnormalities: trait- and state-dependent components. Schizophr Bull. 1986;12(3):447–459. [DOI] [PubMed] [Google Scholar]
- 2. Schechter I, Butler PD, Silipo G, Zemon V, Javitt DC. Magnocellular and parvocellular contributions to backward masking dysfunction in schizophrenia. Schizophr Res. 2003;64(2–3):91–101. [DOI] [PubMed] [Google Scholar]
- 3. Basińska A. Altered electrophysiological pattern of target detection in schizophrenia in the continuous attention test. Acta Neurobiol Exp (Wars). 1998;58(3):207–220. [DOI] [PubMed] [Google Scholar]
- 4. Doniger GM, Foxe JJ, Murray MM, Higgins BA, Javitt DC. Impaired visual object recognition and dorsal/ventral stream interaction in schizophrenia. Arch Gen Psychiatry. 2002;59(11):1011–1020. [DOI] [PubMed] [Google Scholar]
- 5. Foxe JJ, Doniger GM, Javitt DC. Early visual processing deficits in schizophrenia: impaired P1 generation revealed by high-density electrical mapping. Neuroreport. 2001;12(17):3815–3820. [DOI] [PubMed] [Google Scholar]
- 6. Foxe JJ, Murray MM, Javitt DC. Filling-in in schizophrenia: a high-density electrical mapping and source-analysis investigation of illusory contour processing. Cereb Cortex. 2005;15(12):1914–1927. [DOI] [PubMed] [Google Scholar]
- 7. Butler PD, Martinez A, Foxe JJ, et al. Subcortical visual dysfunction in schizophrenia drives secondary cortical impairments. Brain. 2007;130(Pt 2):417–430. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Schechter I, Butler PD, Zemon VM, et al. Impairments in generation of early-stage transient visual evoked potentials to magno- and parvocellular-selective stimuli in schizophrenia. Clin Neurophysiol. 2005;116(9):2204–2215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Luck SJ, Heinze HJ, Mangun GR, Hillyard SA. Visual event-related potentials index focused attention within bilateral stimulus arrays. II. Functional dissociation of P1 and N1 components. Electroencephalogr Clin Neurophysiol. 1990;75(6):528–542. [DOI] [PubMed] [Google Scholar]
- 10. Dias EC, Butler PD, Hoptman MJ, Javitt DC. Early sensory contributions to contextual encoding deficits in schizophrenia. Arch Gen Psychiatry. 2011;68(7):654–664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Martínez A, Hillyard SA, Bickel S, Dias EC, Butler PD, Javitt DC. Consequences of magnocellular dysfunction on processing attended information in schizophrenia. Cereb Cortex. 2012;22(6):1282–1293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. González-Blanch C, Perez-Iglesias R, Pardo-García G, et al. Prognostic value of cognitive functioning for global functional recovery in first-episode schizophrenia. Psychol Med. 2010;40(6):935–944. [DOI] [PubMed] [Google Scholar]
- 13. Green MF, Kern RS, Braff DL, Mintz J. Neurocognitive deficits and functional outcome in schizophrenia: are we measuring the “right stuff”? Schizophr Bull. 2000;26(1):119–136. [DOI] [PubMed] [Google Scholar]
- 14. Vesterager L, Christensen TØ, Olsen BB, et al. Cognitive and clinical predictors of functional capacity in patients with first episode schizophrenia. Schizophr Res. 2012;141(2–3):251–256. [DOI] [PubMed] [Google Scholar]
- 15. Carter CS, Barch DM; CNTRICS Executive Committee Imaging biomarkers for treatment development for impaired cognition: report of the sixth CNTRICS meeting: Biomarkers recommended for further development. Schizophr Bull. 2012;38(1):26–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Javitt DC, Spencer KM, Thaker GK, Winterer G, Hajós M. Neurophysiological biomarkers for drug development in schizophrenia. Nat Rev Drug Discov. 2008;7(1):68–83. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Demmin DL, Fradkin SI, Silverstein SM. Remediation of visual processing impairments in schizophrenia: where we are and where we need to be. Current Behav Neurosci Rep. 2019;6(2):13–20. [Google Scholar]
- 18. Butler PD, Thompson JL, Seitz AR, Deveau J, Silverstein SM. Visual perceptual remediation for individuals with schizophrenia: rationale, method, and three case studies. Psychiatr Rehabil J. 2017;40(1):43–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Butler PD, Zemon V, Schechter I, et al. Early-stage visual processing and cortical amplification deficits in schizophrenia. Arch Gen Psychiatry. 2005;62(5):495–504. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Rassovsky Y, Horan WP, Lee J, Sergi MJ, Green MF. Pathways between early visual processing and functional outcome in schizophrenia. Psychol Med. 2011;41(3):487–497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Eack SM, Greenwald DP, Hogarty SS, Keshavan MS. One-year durability of the effects of cognitive enhancement therapy on functional outcome in early schizophrenia. Schizophr Res. 2010;120(1–3):210–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. ten Velden Hegelstad W, Haahr U, Larsen TK, et al. Early detection, early symptom progression and symptomatic remission after ten years in a first episode of psychosis study. Schizophr Res. 2013;143(2–3):337–343. [DOI] [PubMed] [Google Scholar]
- 23. Haenschel C, Bittner RA, Haertling F, et al. Contribution of impaired early-stage visual processing to working memory dysfunction in adolescents with schizophrenia: a study with event-related potentials and functional magnetic resonance imaging. Arch Gen Psychiatry. 2007;64(11):1229–1240. [DOI] [PubMed] [Google Scholar]
- 24. Yeap S, Kelly SP, Thakore JH, Foxe JJ. Visual sensory processing deficits in first-episode patients with Schizophrenia. Schizophr Res. 2008;102(1–3):340–343. [DOI] [PubMed] [Google Scholar]
- 25. Martínez A, Hillyard SA, Dias EC, et al. Magnocellular pathway impairment in schizophrenia: evidence from functional magnetic resonance imaging. J Neurosci. 2008;28(30):7492–7500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Tarkiainen A, Liljeström M, Seppä M, Salmelin R. The 3D topography of MEG source localization accuracy: effects of conductor model and noise. Clin Neurophysiol. 2003;114(10):1977–1992. [DOI] [PubMed] [Google Scholar]
- 27. Portin K, Vanni S, Virsu V, Hari R. Stronger occipital cortical activation to lower than upper visual field stimuli. Neuromagnetic recordings. Exp Brain Res. 1999;124(3):287–294. [DOI] [PubMed] [Google Scholar]
- 28. Suzuki M, Nagae M, Nagata Y, Kumagai N, Inui K, Kakigi R. Effects of refractive errors on visual evoked magnetic fields. BMC Ophthalmol. 2015;15:162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Uusitalo MA, Ilmoniemi RJ. Signal-space projection method for separating MEG or EEG into components. Med Biol Eng Comput. 1997;35(2):135–140. [DOI] [PubMed] [Google Scholar]
- 30. Delorme A, Makeig S. EEGLAB: an open source toolbox for analysis of single-trial EEG dynamics including independent component analysis. J Neurosci Methods. 2004;134(1):9–21. [DOI] [PubMed] [Google Scholar]
- 31. Hämäläinen M, Hari R. Magnetoencephalographic (MEG) characterization of dynamic brain activation. In: Toga A, Mazziotta J, eds. Brain Mapping: The Methods, 2nd ed.Amsterdam, The Netherlands: Academic Press; 2002:227–254. [Google Scholar]
- 32. Lin FH, Witzel T, Hämäläinen MS, Dale AM, Belliveau JW, Stufflebeam SM. Spectral spatiotemporal imaging of cortical oscillations and interactions in the human brain. Neuroimage. 2004;23(2):582–595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Dorph‐Petersen K-A, Pierri JN, Wu Q, Sampson AR, Lewis DA. Primary visual cortex volume and total neuron number are reduced in schizophrenia. J Comp Neurol. 2007;501(2):290–301. [DOI] [PubMed] [Google Scholar]
- 34. Narr KL, Toga AW, Szeszko P, et al. Cortical thinning in cingulate and occipital cortices in first episode schizophrenia. Biol Psychiatry. 2005;58(1):32–40. [DOI] [PubMed] [Google Scholar]
- 35. Ardekani BA, Nierenberg J, Hoptman MJ, Javitt DC, Lim KO. MRI study of white matter diffusion anisotropy in schizophrenia. Neuroreport. 2003;14(16):2025–2029. [DOI] [PubMed] [Google Scholar]
- 36. Palaniyappan L, Al-Radaideh A, Mougin O, Gowland P, Liddle PF. Combined white matter imaging suggests myelination defects in visual processing regions in schizophrenia. Neuropsychopharmacology. 2013;38(9):1808–1815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Gagné AM, Hébert M, Maziade M. Revisiting visual dysfunctions in schizophrenia from the retina to the cortical cells: a manifestation of defective neurodevelopment. Prog Neuropsychopharmacol Biol Psychiatry. 2015;62:29–34. [DOI] [PubMed] [Google Scholar]
- 38. Fu S, Greenwood PM, Parasuraman R. Brain mechanisms of involuntary visuospatial attention: an event-related potential study. Hum Brain Mapp. 2005;25(4):378–390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Jenner AR, Rosen GD, Galaburda AM. Neuronal asymmetries in primary visual cortex of dyslexic and nondyslexic brains. Ann Neurol. 1999;46(2):189–196. [DOI] [PubMed] [Google Scholar]
- 40. Amunts K, Armstrong E, Malikovic A, et al. Gender-specific left-right asymmetries in human visual cortex. J Neurosci. 2007;27(6):1356–1364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Carter CS, Robertson LC, Nordahl TE, Chaderjian M, Oshora-Celaya L. Perceptual and attentional asymmetries in schizophrenia: further evidence for a left hemisphere deficit. Psychiatry Res. 1996;62(2):111–119. [DOI] [PubMed] [Google Scholar]
- 42. Posner MI, Early TS , Reiman E, Pardo PJ, Dhawan M. Asymmetries in hemispheric control of attention in schizophrenia. Arch Gen Psychiatry. 1988;45:814–821. [DOI] [PubMed] [Google Scholar]
- 43. Burns J, Job D, Bastin ME, et al. Structural disconnectivity in schizophrenia: a diffusion tensor magnetic resonance imaging study. Br J Psychiatry. 2003;182:439–443. [PubMed] [Google Scholar]
- 44. Kasai T, Morotomi T, Katayama J, Kumada T. Attending to a location in three-dimensional space modulates early ERPs. Brain Res Cogn Brain Res. 2003;17(2):273–285. [DOI] [PubMed] [Google Scholar]
- 45. Hirayasu Y, Tanaka S, Shenton ME, et al. Prefrontal gray matter volume reduction in first episode schizophrenia. Cereb Cortex. 2001;11(4):374–381. [DOI] [PubMed] [Google Scholar]
- 46. Gur RE. Left hemisphere dysfunction and left hemisphere overactivation in schizophrenia. J Abnorm Psychol. 1978;87(2):226–238. [DOI] [PubMed] [Google Scholar]
- 47. Sommer IE, Ramsey NF, Kahn RS. Language lateralization in schizophrenia, an fMRI study. Schizophr Res. 2001;52(1–2):57–67. [DOI] [PubMed] [Google Scholar]
- 48. Butler PD. Early-stage visual processing deficits in schizophrenia. In: Lajtha A, Javitt D, Kantrowitz J, eds. Handbook of Neurochemistry and Molecular Neurobiology: Schizophrenia. Boston, MA: Springer US; 2009:331–352. [Google Scholar]
- 49. Okubo M, Nicholls ME. Hemispheric asymmetry in temporal resolution: contribution of the magnocellular pathway. Psychon Bull Rev. 2005;12(4):755–759. [DOI] [PubMed] [Google Scholar]
- 50. Di Russo F, Martínez A, Hillyard SA. Source analysis of event-related cortical activity during visuo-spatial attention. Cereb Cortex. 2003;13(5):486–499. [DOI] [PubMed] [Google Scholar]
- 51. Noesselt T, Hillyard SA, Woldorff MG, et al. Delayed striate cortical activation during spatial attention. Neuron. 2002;35(3):575–587. [DOI] [PubMed] [Google Scholar]
- 52. Kelly SP, Gomez-Ramirez M, Foxe JJ. Spatial attention modulates initial afferent activity in human primary visual cortex. Cereb Cortex. 2008;18(11):2629–2636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Poghosyan V, Ioannides AA. Attention modulates earliest responses in the primary auditory and visual cortices. Neuron. 2008;58(5):802–813. [DOI] [PubMed] [Google Scholar]
- 54. Spencer KM, Niznikiewicz MA, Nestor PG, Shenton ME, McCarley RW. Left auditory cortex gamma synchronization and auditory hallucination symptoms in schizophrenia. BMC Neurosci. 2009;10:85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Spencer KM, Nestor PG, Perlmutter R, et al. Neural synchrony indexes disordered perception and cognition in schizophrenia. Proc Natl Acad Sci USA. 2004;101(49):17288–17293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Shergill SS, Cameron LA, Brammer MJ, Williams SC, Murray RM, McGuire PK. Modality specific neural correlates of auditory and somatic hallucinations. J Neurol Neurosurg Psychiatry. 2001;71(5):688–690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Beer AL, Plank T, Greenlee MW. Diffusion tensor imaging shows white matter tracts between human auditory and visual cortex. Exp Brain Res. 2011;213(2–3):299–308. [DOI] [PubMed] [Google Scholar]
- 58. Finney EM, Fine I, Dobkins KR. Visual stimuli activate auditory cortex in the deaf. Nat Neurosci. 2001;4(12): 1171–1173. [DOI] [PubMed] [Google Scholar]
- 59. Wittenberg GF, Werhahn KJ, Wassermann EM, Herscovitch P, Cohen LG. Functional connectivity between somatosensory and visual cortex in early blind humans. Eur J Neurosci. 2004;20(7):1923–1927. [DOI] [PubMed] [Google Scholar]