Abstract
Background
Myocardial ischemia mediates the progression of multiple cardiovascular diseases and leads to serious damage to the morphology, function, and metabolism of cardiomyocytes. The serum level of the hormone Meteorin-like (METRNL) was lower in patients with coronary artery disease and was negatively correlated with inflammatory cytokines. The aim of the present study was to determine the relationship between METRNL and myocardial ischemia/reperfusion (MI/R) injury, and investigate the molecular mechanisms implicated the pathogenesis of myocardial ischemia.
Material/Methods
In the present study, H9C2 cells underwent oxygen-glucose deprivation and reperfusion (OGD/R) treatment to establish a MI/R cell model. Quantitative real-time polymerase chain reaction was performed to analyze the expression of target gene. Western blot was used to evaluate the protein expression. Cell Counting Kit-8 assay was employed to detect the cell viability. Enzyme-linked immunosorbent assay was carried out to determine the levels of inflammatory cytokines. Finally, flow cytometry and TUNEL staining were used to detect the apoptotic levels of cardiomyocytes.
Results
The results showed that the expression of METRNL was downregulated in H9C2 cells during OGD/R. Interestingly, METRNL overexpression inhibited the inflammation, apoptosis and endoplasmic reticulum stress in H9C2 cells during OGD/R, which were totally reversed by PAK2 silencing. In addition, METRNL overexpression induced activation of AMPK-PAK2 signaling cascade.
Conclusions
METRNL attenuates MI/R injury-induced cardiomyocytes apoptosis by alleviating endoplasmic reticulum stress via activation of AMPK-PAK2 signaling in H9C2 cells. Our findings support that METRNL might be a promising target for treatment of myocardial ischemia in the future.
MeSH Keywords: Apoptosis, Endoplasmic Reticulum, Myocardial Ischemia
Background
Myocardial ischemia is an acute and severe pathological condition, which leads to serious damage to the morphology, function, and metabolism of cardiomyocytes [1,2]. Emerging evidence has demonstrated that myocardial ischemia mediates the progression of multiple cardiovascular diseases, such as coronary artery disease, heart failure, and myocardial fibrosis, and contributes to high morbidity and mortality [3,4]. Moreover, ischemic heart disease has been identified as the leading cause of human death worldwide, and a threat to public health and imposed great economic burden on families and society [5]. Clinically, early restoration of blood reperfusion is recognized as the most effective way to attenuate poor outcomes of patients with myocardial infarction [6]. However, the process of reperfusion further aggravates damage of cardiomyocyte morphology and function, a condition defined as myocardial ischemia/reperfusion (MI/R) injury [7]. Recently, percutaneous coronary intervention and thrombolytic therapy have been widely regarded as the most effective therapeutic strategy for patients with ischemic heart disease [8]. However, the mortality rate of ischemic heart disease is still high, and general population remains at risk of MI/R injury, particularly seniors [9]. Unfortunately, the specific mechanism of MI/R injury has not been fully illuminated [10]. Thus, our study aimed to investigate the underlying mechanism implicated in pathogenesis of myocardial ischemia.
As the primary site of protein production and secretion, endoplasmic reticulum (ER) plays a crucial role in cell function [11]. Inflammation and oxidative stress are induced by multiple factors, including ischemia, hypoxia, and heat shock, all of which can disrupt the functions of ER and cause the accumulation of unfolded and misfolded proteins, which is referred to as ER stress. Accumulating evidence demonstrated that ER stress is closely related to the development of pathological cardiovascular diseases [12]. I/R injury induces the aberrant secretion of inflammatory cytokines and cardiomyocyte apoptosis, and eventually triggers severe ER stress [13]. A previous study reported that the blocking of ER stress-induced cardiomyocyte apoptosis is a credible strategy for myocardial ischemia therapy [14].
The hormone Meteorin-like (METRNL) is recognized as a novel adipokine that is produced by adipose tissue [15]. It was observed in a study that the serum METRNL concentration was lower in patients with coronary artery disease compared to the healthy individuals [16]. Zhengxia et al. also demonstrated that the serum level of METRNL was decreased and was negatively correlated with inflammatory markers including interleukin (IL)-1β and IL-11 [17]. However, the role of METRNL in myocardial infarction remains unclear. METRNL alleviated lipid-mediated inflammatory response and insulin resistance via activation of 5′ AMP-activated protein kinase (AMPK) or PPARδ signaling pathways in mice skeletal muscle [18]. Interestingly, the activation of AMPK signaling pathways was reported to be an effective strategy to attenuate the ER stress [19]. Thus, we presume that METRNL could be a potential target for ischemic heart disease treatment.
In the current study, the oxidative-glucose deprivation/re-oxygenation (OGD/R) injury model was conducted in H9C2 cells to determine the relationships between METRNL and MI/R injury. Our study aims at investigating the molecular mechanisms implicated the pathogenesis of myocardial ischemia. Our findings provided evidence to support that METRNL attenuates MI/R injury-induced cardiomyocytes apoptosis and may be an effective target for ischemic heart disease.
Material and Methods
Cell culture and oxygen-glucose deprivation and reperfusion (OGD/R)
The H9C2 cell line, derived from embryonic rat heart cells, was obtained from American Type Culture Collection. H9C2 cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) containing 10% fetal bovine serum (FBS; Gibco, USA), 100 U/mL penicillin, and 100 μg/mL streptomycin at 37°C in a humidified atmosphere containing 5% CO2 and 95% air atmosphere.
When cultured H9C2 cells reached confluency, ischemia was induced by deprivation of oxygen and glucose for 2 hours in serum/glucose-free DMEM medium in an anaerobic chamber (95% N2 and 5% CO2). Then, H9C2 cells were maintained in the glucose containing DMEM with 10% FBS under normal growth conditions (95% air and 5% CO2) for another 12 hours to reoxygenation. Control cells were incubated under normoxic conditions.
Quantitative real-time polymerase chain reaction (qRT-PCR)
After treatment, total RNA was extracted from H9C2 cells with TRIzol reagent (Takara, Japan). Reverse Transcription Kit (Thermo, USA) was employed to synthesize the cDNA. After authentication of RNA quality and quantity, TaqMan MicroRNA Reverse Transcription kit was used to synthesize complementary DNA according to manufacturer’s protocols. A SYBR green qPCR assay kit (TaKaRa, Japan) was employed to amplify the expression levels of METRNL and p21-activated kinase 2 (PAK2) with glyceraldehyde 3-phosphate dehydrogenase (GAPDH) as the internal references. The 2−ΔΔCt method was adopted to calculate the relative expression of target genes and house-keeping genes. All experiments were performed 3 independent times. The primer sequences for qPCR amplification were as follows:
-
METRNL forward, 5′-TGCATAATAAACCGAGGTT-3′ and
reverse, 5′-CTCCGTGGGCCGCTGCCCT-3′;
-
PAK2 forward, 5′-TCTTCCTCCCCCAGGGTTG-3′ and
reverse, 5′-AATCGAGCCCACTGTTCTGG-3′;
-
and GAPDH forward, 5′-CGGAGTCAACGGATTTGGTCGTAT-3′ and
reverse, 5′-AGCCTTCTCCATGGTGGTGAAGAC-3′.
Western blot
After treatment, H9C2 cells were lysed with radioimmunoprecipitation assay (RIPA) buffer containing protease inhibitors to extract total protein. Equal amounts of proteins were fractionated by sodium dodecyl sulphate-polyacrylamide gel electrophoresis (SDS-PAGE). Then, the proteins were transferred onto polyvinylidene difluoride (PVDF) membranes and incubated at 4°C overnight with primary antibodies, followed by incubation with corresponding horseradish peroxidase (HRP)-labeled secondary antibodies at room temperature for 2 hours. Primary antibodies, including anti-METRNL antibody (1: 1000), anti-Bcl2 antibody (1: 1000), anti-Bax antibody (1: 1000), anti-cleaved caspase-3 antibody (1: 500), anti-caspase-3 antibody (1: 1000), anti-caspase-12 antibody (1: 1000), anti-PARP antibody (1: 1000), anti-GRP78 antibody (1: 1000), anti-ATF4 antibody (1: 1000), anti-ATF6 antibody (1: 1000), anti-CHOP antibody (1: 1000), anti-PAK2 antibody (1: 1000), anti-p-AMPK antibody (1: 1000), and anti-AMPK antibody (1: 1000) were obtained from Cell Signaling Technology. The protein bands were visualized by Tanon-5200 Chemiluminescence Imager (Tanon, Shanghai, China) with electrochemiluminescence (ECL) assay and the intensity of the bands was normalized to GAPDH expression. All experiments were performed 3 independent times.
Cell viability assay
Cell Counting Kit-8 (CCK-8) assay was employed to detect the viability of the H9C2 cells. Cells were plated at a density of 5.0×103/well in 96-well plates. After 24 hours of incubation, OGD/R treatment was performed at different time points. Then CCK-8 reagent was added to each well and cultured at 37°C for 1 hour. The absorbance was determined at 450 nm using a Microplate Reader (Pulangxin Technology) at indicated time (0, 6, 12, and 24 hours). All experiments were repeated at least 3 independent times.
Enzyme-linked immunosorbent assay (ELISA)
After treatment, levels of inflammatory cytokines including tumor necrosis factor (TNF)-α, IL-1β, and IL-6 in supernatants of H9C2 cells were determined by specific enzyme-linked immunosorbent assay (ELISA) kit (Invitrogen, Carlsbad, CA, USA), following the manufacturer’s instructions. Absorbance was assayed at 450 nm by a microplate reader (Bio-Rad, Hercules, CA, USA). All experiments were performed 3 independent times.
Flow cytometry
Cell apoptosis was determined by Annexin V-fluorescein isothiocyanate (FITC)/propidium iodide (PI) apoptosis detection kit (Thermo Fisher Scientific). Briefly, H9C2 cells were seeded in 6-well plates (1×105 cells/well) and transfected with or without overexpression (Ov)-METRNL. Then, all cells underwent OGD/R treatment followed by washing and trypsinization. H9C2 cells were incubated with Annexin V-FITC for 20 minutes. Thereafter, PI staining solution was added and incubated for another 20 minutes under darkness. A flow cytometer (Becton Dickinson Biosciences, San Jose, CA, USA) was employed to analyze cell apoptosis rate. In the scatter diagram, the H9C2 cells in the LR (early apoptosis cells) and UR (late apoptosis cells) regions were identified as apoptotic cells.
TUNEL assay
To assess apoptosis rate of H9C2 cells transfected with or without Ov-METRNL and shRNA-PAK2, terminal deoxynucleotidyl transferase dUTP nick end labeling immunohistochemical (TUNEL) staining was carried out. The TUNEL Apoptosis Detection Kit I, POD (Boster, CA, USA) was used according to the manufacturer’s procedure. Then, cells were reacted with diaminobenzene (DAB, Boster, CA, USA) for 10 minutes. Thereafter, cells were counterstained with hematoxylin (Solarbio, Beijing, China) for 30 seconds. TUNEL positive cells were captured under an optical microscope at least 5 random separate fields. The percentage of apoptosis cells was calculated and analyzed.
Statistical analysis
All statistical analyses were performed with SPSS 18.0 software. Measured data with normal distribution were presented as mean±standard deviation (SD). Two independent samples were compared by t-test. For comparison among multiple groups, one-way ANOVA with the LSD t-test was applied. P<0.05 was considered statistically significant.
Results
The expression of METRNL was downregulated in H9C2 cells during OGD/R
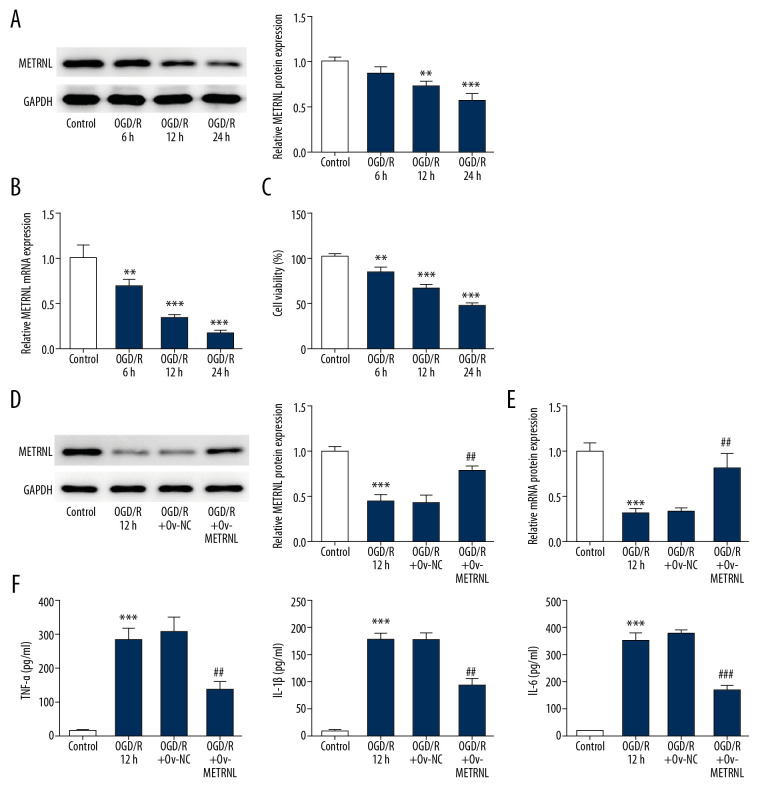
To detect the METRNL expression, western blot and qRT-PCR were performed. The results are shown in Figure 1A and 1B. The expression of METRNL was significantly decreased in H9C2 cells with the increase of OGD/R time, suggesting the METRNL expression was closely associated with ischemia progression. In addition, CCK-8 assay suggested that the survival rate of H9C2 cells was decreased by OGD/R treatment in a time-dependent manner (Figure 1C). Taking these factors together, OGD/R treatment for 12 hours was selected for further experiments.
Figure 1.
The expression of METRNL was downregulated in H9C2 cells during OGD/R, and METRNL overexpression inhibited the inflammatory cytokines secretion. (A, D) The expression of METRNL in H9C2 cells was determined by western blot. (B, E) Relative expression of METRNL in H9C2 cells was evaluated by qRT-PCR. (C) The viability of H9C2 cells was analyzed by CCK8 assay. (F) The levels of inflammatory cytokines including TNF-α, IL-1β, and IL-6 were measured by corresponding ELISA kits. Error bars represent the mean±SEM from 3 independent experiments. * P < 0.05, ** P<0.01, *** P<0.001 versus Control; ## P<0.01, ### P<0.001 versus OGD/R 12 hours. METRNL – Meteorin-like; OGD/R – oxygen-glucose deprivation and reperfusion; qRT-PCR – quantitative real-time polymerase chain reaction; CCK-8 – Cell Counting Kit-8; TNF – tumor necrosis factor; IL – interleukin; ELISA – enzyme-linked immunosorbent assay; SEM – standard error of the mean.
METRNL overexpression inhibited the inflammatory cytokines secretion in H9C2 cells during OGD/R
To regulate the METRNL expression, overexpression (Ov)-METRNL plasmids were conducted. Western blot and qRT-PCR were carried out to determine the efficiency of transfection. As shown in Figure 1D and 1E, the upregulation of METRNL was observed in the OGD/R+Ov-METRNL group. Moreover, the levels of inflammatory cytokines, including TNF-α, IL-1β, and IL-6, were evaluated as shown in the corresponding ELISA used to investigate the role of METRNL in MI/R injury. The results showed that OGD/R treatment dramatically increased release of inflammatory cytokines in H9C2 cells, while METRNL overexpression attenuated abnormal secretion of these inflammatory cytokines (Figure 1F). The data indicated that METRNL overexpression inhibited the inflammatory cytokines secretion in H9C2 cells during OGD/R.
METRNL overexpression suppressed the apoptosis of H9C2 cells induced by OGD/R
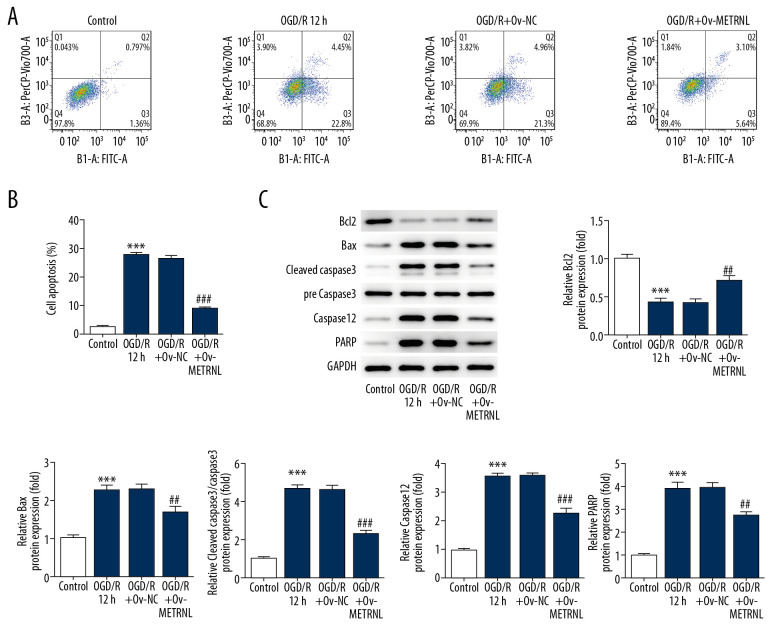
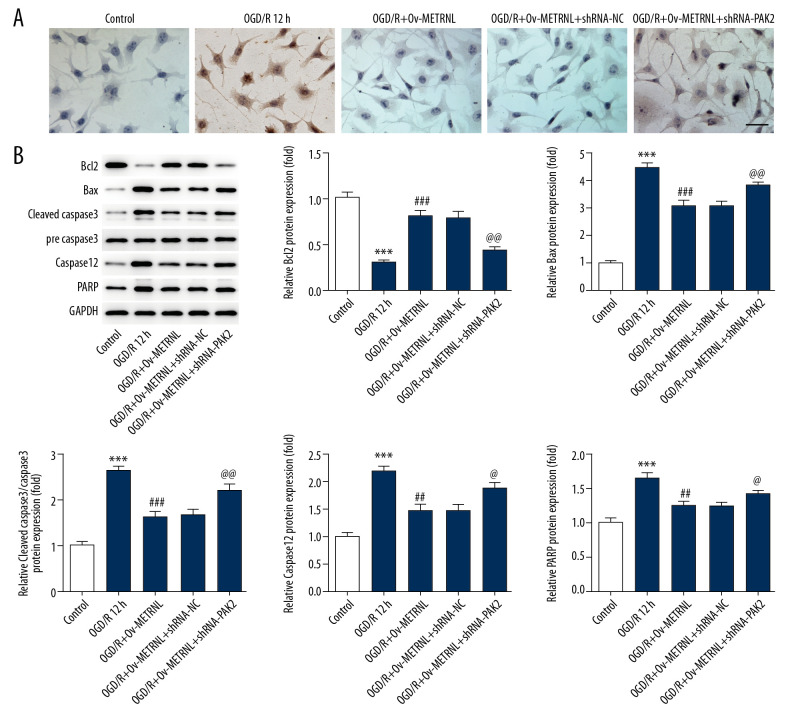
To further investigate the role of METRNL in MI/R injury, flow cytometry and western blot were performed to evaluate the apoptosis rate of H9C2 cells and expression of proteins related to apoptosis, respectively. As shown in Figure 2A and 2B, OGD/R treatment significantly elevated cardiomyocytes apoptosis rate compared with control group, which was reversed by METRNL overexpression. Furthermore, western blot results demonstrated that OGD/R treatment led to reduction of anti-apoptotic proteins (Bcl2) while upregulation of proapoptotic proteins (Bax), cleaved caspase-3, caspase-12, and poly ADP-ribose polymerase (PARP) wherever METRNL overexpression blocked these phenomenon (Figure 2C). The data suggested that METRNL overexpression suppressed the OGD/R-induced apoptosis in H9C2 cells.
Figure 2.
METRNL overexpression suppressed the apoptosis of H9C2 cells induced by OGD/R. (A, B) The apoptosis rate of H9C2 cells was determined by flow cytometry. (C) The expressions of Bcl2, Bax, cleaved caspase-3, pre caspase-3, caspase-12, and PARP were evaluated by western blot. Error bars represent the mean±SEM from 3 independent experiments. *** P<0.001 versus Control; ## P<0.01, ### P<0.001 versus OGD/R 12 hours. METRNL – Meteorin-like; OGD/R – oxygen-glucose deprivation and reperfusion; PARP – poly ADP-ribose polymerase; SEM – standard error of the mean.
METRNL overexpression inhibited ER stress and promoted the PAK2 expression
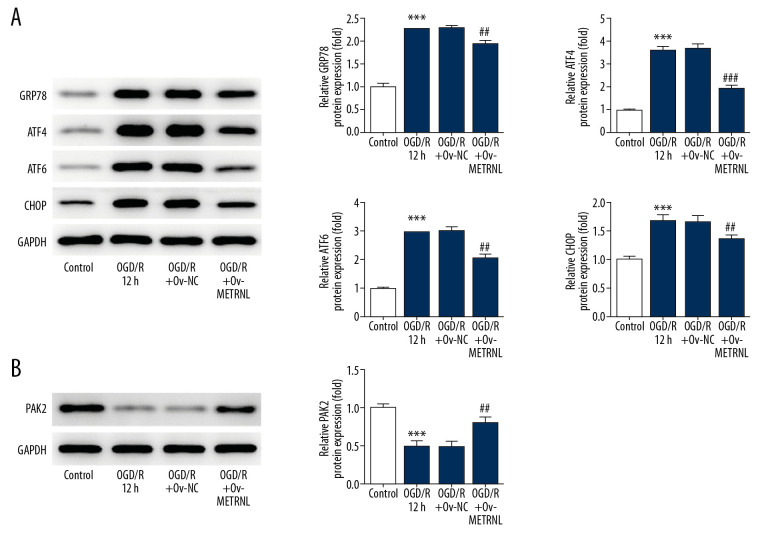
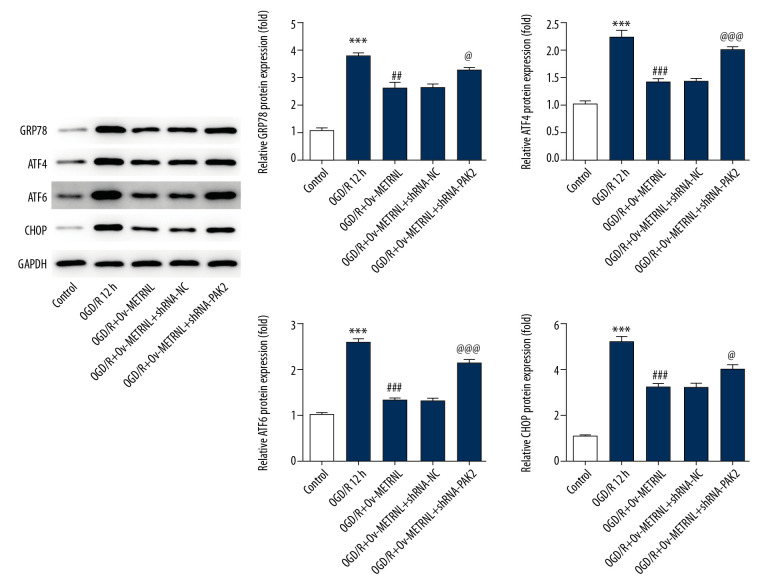
ER stress induces the unfolded protein response, mediated by ER chaperone (glucose-regulated protein 78, GRP78), ER stress transducers activating transcription factor 4 (ATF4), ATF6, and C/EBP-homologus protein (CHOP) [20]. To study the impact of METRNL on ER function, the expressions of proteins, including GRP78, ATF4, ATF6, and CHOP were evaluated by western blot. As shown in Figure 3A, the expressions of the aforementioned proteins were remarkably elevated by OGD/R treatment, suggesting OGD/R treatment induced activation of ER stress. Interestingly, METRNL overexpression played a suppressive role in the expression of aforementioned proteins. Additionally, a previous study reported that PAK2 was identified as a novel regulator of ER function [21]. Western blot results showed that OGD/R treatment caused decrease of PAK2 expression, which was abolished by METRNL overexpression (Figure 3B). The data implied that METRNL overexpression inhibited ER stress and promoted PAK2 expression.
Figure 3.
METRNL overexpression inhibited ER stress and promoted the PAK2 expression. (A, B) The expressions of GRP78, ATF4, ATF6, CHOP, and PAK2 were determined by western blot. Error bars represent the mean±SEM from 3 independent experiments. *** P<0.001 versus Control; ## P<0.01, ### P<0.001 versus OGD/R 12 hours. METRNL – Meteorin-like; ER – endoplasmic reticulum; PAK2 – p21-activated kinase 2; GRP78 – glucose-regulated protein 78; ATF – activating transcription factor; CHOP – C/EBP-homologus protein; SEM – standard error of the mean; OGD/R – oxygen-glucose deprivation and reperfusion.
PAK2 silencing reversed the inhibitive effect of METRNL overexpression on inflammatory response and cell apoptosis induced by OGD/R
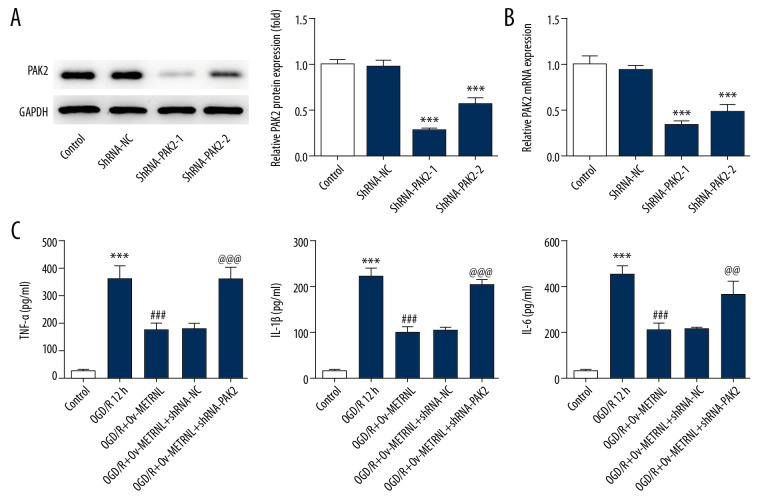
To investigate the underlying mechanism implicated in the role of METRNL in MI/R injury, PAK2 expression was downregulated by shRNA-PAK2 plasmids. Western blot and qRT-PCR were carried out to evaluate the efficiency of transfection. It was found that shRNA-PAK2-1 induced more significant reduction of PAK2 expression than shRNA-PAK2-2 (Figure 4A, 4B), and therefore, shRNA-PAK2-1 was selected in the next experiments.
Figure 4.
PAK2 silencing reversed the inhibitive effect of METRNL overexpression on inflammatory response. (A) The expression of PAK2 in H9C2 cells was determined by western blot. (B) Relative expression of PAK2 in H9C2 cells was evaluated by qRT-PCR. (C) The levels of inflammatory cytokines including TNF-α, IL-1β, and IL-6 were measured by corresponding ELISA kits. Error bars represent the mean±SEM from three independent experiments. *** P<0.001 versus Control; ### P<0.001 versus OGD/R 12 hours; @@ P<0.01, @@@ P<0.001 versus OGD/R+Ov-METRNL. PAK2 – p21-activated kinase 2; METRNL – Meteorin-like; qRT-PCR – quantitative real-time polymerase chain reaction; TNF – tumor necrosis factor; IL – interleukin; ELISA – enzyme-linked immunosorbent assay; SEM – standard error of the mean; OGD/R – oxygen-glucose deprivation and reperfusion.
As demonstrated by ELISA results, high levels of inflammatory cytokines were clearly observed with transfection of shRNA-PAK2 (Figure 4C). Moreover, PAK2 silencing blocked the suppressive effect of METRNL overexpression on cell apoptosis induced by OGD/R, indicated by TUNEL assay results (Figure 5A). In addition, PAK2 silencing abolished METRNL overexpression-induced Bcl2 upregulation and the downregulation of proteins including Bax, cleaved caspase-3, caspase-12. and PARP (Figure 5B). These findings supported that METRNL suppressed the inflammation and cell apoptosis induced by OGD/R via activation of PAK2.
Figure 5.
PAK2 silencing blocked the inhibitive effect of METRNL overexpression on cell apoptosis induced by OGD/R. (A) The apoptosis rate of H9C2 cells was analyzed by TUNEL assay (200x magnification). (B) The expressions of Bcl2, Bax, cleaved caspase-3, pre caspase-3, caspase-12, and PARP were evaluated by western blot. Error bars represent the mean±SEM from 3 independent experiments. *** P<0.001 versus Control; ## P<0.01, ### P<0.001 versus OGD/R 12 hours; @ P<0.05, @@ P<0.01 versus OGD/R+Ov-METRNL. PAK2 – p21-activated kinase 2; METRNL – Meteorin-like; OGD/R – oxygen-glucose deprivation and reperfusion; TUNEL – terminal deoxynucleotidyl transferase dUTP nick end labeling immunohistochemical; PARP – poly ADP-ribose polymerase; SEM – standard error of the mean; Ov-METRNL – overexpression-METRNL.
PAK2 silencing reversed the inhibitive effect of METRNL overexpression on ER stress induced by OGD/R
To confirm the effect of METRNL overexpression on ER stress, the expressions of GRP78, ATF4, ATF6, and CHOP were determined by western blot after transfection of shRNA-PAK2. The expression levels of above proteins were increased in H9C2 cells from the OGD/R+Ov-METRNL+shRNA-PAK2 group compared with the OGD/R+Ov-METRNL group (Figure 6), suggesting PAK2 silencing reversed the suppressive effect of METRNL overexpression on ER stress activation induced by OGD/R.
Figure 6.
PAK2 silencing reversed the inhibitive effect of METRNL overexpression on c ER stress induced by OGD/R. The expressions of GRP78, ATF4, ATF6, and CHOP were determined by western blot. Error bars represent the mean±SEM from 3 independent experiments. *** P<0.001 versus Control; ## P<0.01, ### P<0.001 versus OGD/R 12 hours; @ P<0.05, @@@ P<0.001 versus OGD/R+Ov-METRNL. PAK2 – p21-activated kinase 2; METRNL – Meteorin-like; ER – endoplasmic reticulum; OGD/R – oxygen-glucose deprivation and reperfusion; GRP78 – glucose-regulated protein 78; ATF – activating transcription factor; CHOP – C/EBP-homologus protein; SEM – standard error of the mean.
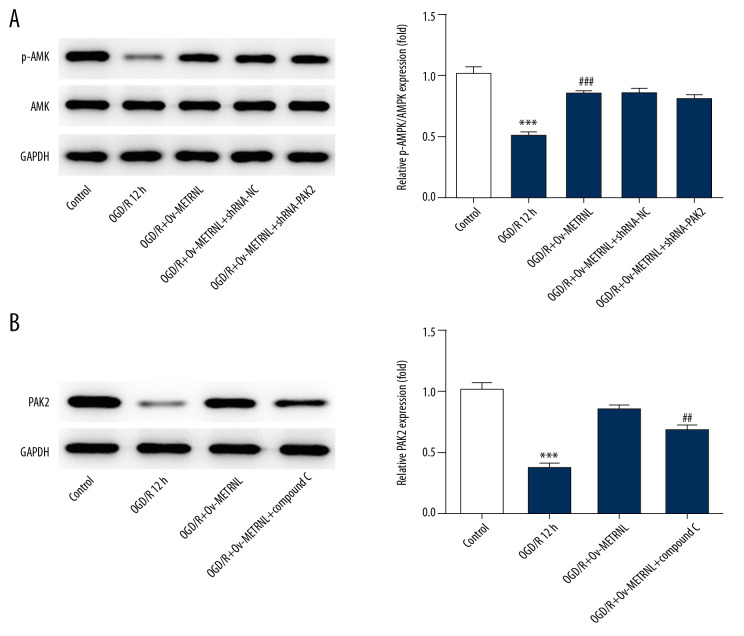
METRNL overexpression induced activation of AMPK-PAK2 signaling cascade
To further investigate the underlying mechanism implicated the role of METRNL in MI/R injury, the expressions of p-AMPK, AMPK, and PAK2 were analyzed by western blot in H9C2 cells transfected with or without Ov-METRNL and shRNA-PAK2. As shown in Figure 7A and 7B, OGD/R treatment induced downregulation of p-AMPK and PAK2, which was reversed by METRNL overexpression. Total AMPK expression had no significant difference. Interestingly, PAK2 silencing had no obvious effect on phosphorylation of AMPK, while AMPK inhibitor, compound C, blocked the positive effect of METRNL on PAK2 expression, suggesting PAK2 is the downstream signaling protein of AMPK. These data proposed that METRNL could play suppressive roles in cardiomyocytes apoptosis and ER stress via activation of AMPK-PAK2 signaling cascade.
Figure 7.
(A, B) METRNL overexpression induced activation of AMPK-PAK2 signaling cascade. The expressions of p-AMPK, AMPK and PAK2 were determined by western blot. Error bars represent the mean±SEM from three independent experiments. **** P<0.001 versus Control; ## P<0.01, ### P<0.001 versus OGD/R 12 hours. METRNL – Meteorin-like; AMPK – 5′ AMP-activated protein kinase; PAK2 – p21-activated kinase 2; SEM – standard error of the mean.
Discussion
Myocardial ischemia is involved in the progression of multiple cardiovascular diseases including coronary artery disease, myocardial fibrosis, and heart failure [22,23]. Recently, METRNL, which is a newly discovered adipokine, was proven to be significantly associated with the pathogenesis of coronary artery disease [16]. Although an earlier study demonstrated that serum METRNL concentration was decreased in patients with coronary artery disease and was negatively correlated with the severity of coronary artery disease [17], its role in myocardial infarction of cardiovascular disease has not been defined. Hence, the aim of the present study was to measure the level of METRNL and explore its underlying mechanism in MI/R injury cell model induced by OGD/R treatment.
In the present study, our findings indicated that the expression of METRNL was downregulated in H9C2 cells during OGD/R (Figure 1). To investigate the underlying mechanisms of METRNL in MI/R injury, further experiments were performed to confirm whether METRNL had a regulative effect on ER stress in H9C2 cells. Accumulating evidence has demonstrated that ER stress plays a crucial role in myocardial ischemia [24]. Excessive inflammatory response and oxidative stress impair the ER functions and cause accumulation of unfolded/misfolded proteins, triggering activation of ER stress [25]. Aberrant myocardial apoptosis induced by prolonged ER stress contributes to the subsequent occurrence of heart failure [26]. In our study, inflammation, apoptosis, and ER stress activation were induced by OGD/R treatment in H9C2 cells. Interestingly, the overexpression of METRNL suppressed the inflammatory response, apoptosis, and ER stress in H9C2 cells during OGD/R treatment (Figure 2). Kerget et al. reported that lower METRNL level was observed in patients with chronic obstructive pulmonary disease, and METRNL was negatively correlated with inflammatory factors [27], which is consistent with our results. Therefore, ER stress could be a promising strategy for myocardial ischemia.
Another interesting finding of our study was that METRNL overexpression enhanced the expression of PAK2 (Figure 3), a novel regulator of ER function. A recent study revealed that melatonin protects against cardiomyocytes reperfusion injury via suppression of ER stress through inactivation of PAK2 [28]. To confirm whether the protective effects of METRNL in MI/R injury were dependent on PAK2 upregulation, the expression of PAK2 was downregulated by shRNA-PAK2. PAK2 silencing blocked the beneficial actions exerted by METRNL overexpression. In our study, knockdown of PAK2 aggravated the inflammatory response (Figure 4), apoptosis (Figure 5), and ER stress (Figure 6), suggesting METRNL could attenuate cardiomyocytes apoptosis via inactivation of ER stress through upregulation of PAK2. More importantly, the upregulation of p-AMPK by METRNL was observed in this study, and our data hinted that compound C, AMPK inhibitor, led to a reduction in PAK2 expression, suggesting AMPK-PAK2 signaling mediates the role of METRNL in cardiomyocytes apoptosis induced by MI/R injury (Figure 7). Jung et al. reported that METRNL alleviated inflammation and insulin resistant via AMPK or PPARδ-dependent pathways [18], which is consistent with our study. Meanwhile, AMPK activation has inhibitive impact on ER stress [19]. Moreover, Xing et al. also demonstrated that AMPK-Pak2 axis was identified as a novel signaling cascade responsible for ER stress under hypoxia-reoxygenation injury in neuroblastoma cell [21]. Consequently, METRNL alleviated MI/R-induced ER stress via activation of AMPK-PAK2 signaling in cardiomyocytes, suggesting METRNL may be expected to represent as a promising target for treatment of myocardial ischemia in the future.
Conclusions
In summary, METRNL attenuates MI/R injury-induced cardiomyocytes apoptosis by alleviating ER stress via activation of AMPK-PAK2 signaling in H9C2 cells. Our findings support that METRNL might be a promising target for treatment of myocardial ischemia in the future.
Footnotes
Conflict of interest
None.
Source of support: This work was supported by Youth Research Project of Health and Family Planning Commission in Fujian Province, China (Grant number: 2016-1-67)
References
- 1.Xu W, Yu H, Ma R, et al. Apelin protects against myocardial ischemic injury by inhibiting dynamin-related protein 1. Oncotarget. 2017;8(59):100034–44. doi: 10.18632/oncotarget.21777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hou X, Fu M, Cheng B, et al. Galanthamine improves myocardial ischemia-reperfusion-induced cardiac dysfunction, endoplasmic reticulum stress-related apoptosis, and myocardial fibrosis by suppressing AMPK/Nrf2 pathway in rats. Ann Transl Med. 2019;7(22):634. doi: 10.21037/atm.2019.10.108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Zhao X, Jia Y, Chen H, et al. Plasma-derived exosomal miR-183 associates with protein kinase activity and may serve as a novel predictive biomarker of myocardial ischemic injury. Exp Ther Med. 2019;18(1):179–87. doi: 10.3892/etm.2019.7555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics-2016 update: A report from the American Heart Association. Circulation. 2016;133(4):e38–360. doi: 10.1161/CIR.0000000000000350. [DOI] [PubMed] [Google Scholar]
- 5.Wang R, Wang M, Zhou J, et al. Shuxuening injection protects against myocardial ischemia-reperfusion injury through reducing oxidative stress, inflammation and thrombosis. Ann Transl Med. 2019;7(20):562. doi: 10.21037/atm.2019.09.40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Li S, Cao C, Chen H, et al. Atheroprotective effects of statins in patients with unstable angina by regulating the blood-borne microRNA network. Mol Med Rep. 2017;16(1):817–27. doi: 10.3892/mmr.2017.6616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Heusch G, Musiolik J, Gedik N, Skyschally A. Mitochondrial STAT3 activation and cardioprotection by ischemic postconditioning in pigs with regional myocardial ischemia/reperfusion. Circ Res. 2011;109(11):1302–8. doi: 10.1161/CIRCRESAHA.111.255604. [DOI] [PubMed] [Google Scholar]
- 8.Giustino G, Dangas GD. Ischemia-reperfusion injury and ischemic post-conditioning in acute myocardial infarction: Lost in translation. Catheter Cardiovasc Interv. 2017;90(7):1068–69. doi: 10.1002/ccd.27436. [DOI] [PubMed] [Google Scholar]
- 9.Gao L, Liu Y, Guo S, et al. Circulating long noncoding RNA HOTAIR is an essential mediator of acute myocardial infarction. Cell Physiol Biochem. 2017;44(4):1497–508. doi: 10.1159/000485588. [DOI] [PubMed] [Google Scholar]
- 10.Zhu S, Xu T, Luo Y, et al. Luteolin enhances sarcoplasmic reticulum Ca2+-ATPase activity through p38 MAPK signaling thus improving rat cardiac function after ischemia/reperfusion. Cell Physiol Biochem. 2017;41(3):999–1010. doi: 10.1159/000460837. [DOI] [PubMed] [Google Scholar]
- 11.Benham AM. Protein secretion and the endoplasmic reticulum. Cold Spring Harb Perspect Biol. 2012;4(8):a012872. doi: 10.1101/cshperspect.a012872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Groenendyk J, Sreenivasaiah PK, Kim DH, et al. Biology of endoplasmic reticulum stress in the heart. Circ Res. 2010;107(10):1185–97. doi: 10.1161/CIRCRESAHA.110.227033. [DOI] [PubMed] [Google Scholar]
- 13.Gao J, Guo Y, Liu Y, et al. Protective effect of FBXL10 in myocardial ischemia reperfusion injury via inhibiting endoplasmic reticulum stress. Respir Med. 2020;161:105852. doi: 10.1016/j.rmed.2019.105852. [DOI] [PubMed] [Google Scholar]
- 14.Yao Y, Lu Q, Hu Z, et al. A non-canonical pathway regulates ER stress signaling and blocks ER stress-induced apoptosis and heart failure. Nat Commun. 2017;8(1):133. doi: 10.1038/s41467-017-00171-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Luo L, Liu M. Adipose tissue in control of metabolism. J Endocrinol. 2016;231(3):R77–99. doi: 10.1530/JOE-16-0211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Dadmanesh M, Aghajani H, Fadaei R, Ghorban K. Lower serum levels of Meteorin-like/Subfatin in patients with coronary artery disease and type 2 diabetes mellitus are negatively associated with insulin resistance and inflammatory cytokines. PLoS One. 2018;13(9):e0204180. doi: 10.1371/journal.pone.0204180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Liu ZX, Ji HH, Yao MP, et al. Serum METRNL is associated with the presence and severity of coronary artery disease. J Cell Mol Med. 2019;23(1):271–80. doi: 10.1111/jcmm.13915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Jung TW, Lee SH, Kim HC, et al. METRNL attenuates lipid-induced inflammation and insulin resistance via AMPK or PPARdelta-dependent pathways in skeletal muscle of mice. Exp Mol Med. 2018;50(9):122. doi: 10.1038/s12276-018-0147-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim SH, Park JW. Morin hydrate attenuates CSE-induced lipid accumulation, ER stress, and oxidative stress in RPE cells: Implications for age-related macular degeneration. Free Radic Res. 2019;53(8):865–74. doi: 10.1080/10715762.2019.1637862. [DOI] [PubMed] [Google Scholar]
- 20.Gao L, Chen H, Li C, et al. ER stress activation impairs the expression of circadian clock and clock-controlled genes in NIH3T3 cells via an ATF4-dependent mechanism. Cell Signal. 2019;57:89–101. doi: 10.1016/j.cellsig.2019.01.008. [DOI] [PubMed] [Google Scholar]
- 21.Xing J, Xu H, Liu C, et al. Melatonin ameliorates endoplasmic reticulum stress in N2a neuroblastoma cell hypoxia-reoxygenation injury by activating the AMPK-Pak2 pathway. Cell Stress Chaperones. 2019;24(3):621–33. doi: 10.1007/s12192-019-00994-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Beltran C, Pardo R, Bou-Teen D, et al. Enhancing glycolysis protects against ischemia-reperfusion injury by reducing ROS production. Metabolites. 2020;10(4) doi: 10.3390/metabo10040132. pii; E132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhou H, Hu B, Liu X. Thyroid hormone metabolite 3-iodothyronamine (T1AM) alleviates hypoxia/reoxygenation-induced cardiac myocyte apoptosis via Akt/FoxO1 pathway. Med Sci Monit. 2020;26:e923195. doi: 10.12659/MSM.923195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zhang Q, Shi J, Guo D, et al. Qishen granule alleviates endoplasmic reticulum stress-induced myocardial apoptosis through IRE-1-CRYAB pathway in myocardial ischemia. J Ethnopharmacol. 2020;252:112573. doi: 10.1016/j.jep.2020.112573. [DOI] [PubMed] [Google Scholar]
- 25.Tabas I, Ron D. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat Cell Biol. 2011;13(3):184–90. doi: 10.1038/ncb0311-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Omidkhoda N, Wallace Hayes A, Reiter RJ, Karimi G. The role of MicroRNAs on endoplasmic reticulum stress in myocardial ischemia and cardiac hypertrophy. Pharmacol Res. 2019;150:104516. doi: 10.1016/j.phrs.2019.104516. [DOI] [PubMed] [Google Scholar]
- 27.Kerget B, Afsin DE, Kerget F, et al. Is METRNL an adipokine involved in the anti-inflammatory response to acute exacerbations of COPD? Lung. 2020;198(2):307–14. doi: 10.1007/s00408-020-00327-4. [DOI] [PubMed] [Google Scholar]
- 28.Wang S, Bian W, Zhen J, et al. Melatonin-mediated Pak2 activation reduces cardiomyocyte death through suppressing hypoxia reoxygenation injury-induced endoplasmic reticulum stress. J Cardiovasc Pharmacol. 2019;74(1):20–29. doi: 10.1097/FJC.0000000000000678. [DOI] [PubMed] [Google Scholar]