Abstract
Terpenes and terpenoids are the largest groups of plant secondary metabolites. However, unlike polyphenols, they are rarely associated with geroprotective properties. Here we evaluated the conformity of the biological effects of terpenoids with the criteria of geroprotectors, including primary criteria (lifespan-extending effects in model organisms, improvement of aging biomarkers, low toxicity, minimal adverse effects, improvement of the quality of life) and secondary criteria (evolutionarily conserved mechanisms of action, reproducibility of the effects on different models, prevention of age-associated diseases, increasing of stress-resistance). The number of substances that demonstrate the greatest compliance with both primary and secondary criteria of geroprotectors were found among different classes of terpenoids. Thus, terpenoids are an underestimated source of potential geroprotectors that can effectively influence the mechanisms of aging and age-related diseases.
Keywords: terpenoids, carotenoids, antioxidants, aging, lifespan, geroprotectors
1. Introduction
Terpenoids are the largest group of plant secondary metabolites [1]. There are tens of thousands of naturally occurring hydrocarbons and they are one of the most structurally diverse classes of natural compounds. Terpenoids consist of C = 5, 10, 15, 20…, n > 40 carbon units and are classified as hemiterpenes (C5), monoterpenes (C10), sesquiterpenes (C15), diterpenes (C20), triterpenes (C30), tetraterpenes or carotenoids (C40), and polyterpenes (Cn, n > 40) [1]. Extensive biological investigations revealed a wide range of pharmacological and physiological activities of terpenoids and their derivatives [2,3,4]. However, terpenoids are rarely associated with anti-aging properties and may be underestimated as potential geroprotectors.
Geroprotectors are the pharmacological agents that decrease the rate of aging and extend lifespan. Despite the fact that terpenoids are the broad class of compounds, only a few of its representatives are well-known geroprotectors [5]. However, they are attracting increasing interest and such a systematic review of geroprotectors of various classes of terpenoids is necessary.
We proposed a set of primary and secondary selection criteria for potential geroprotector [6]. Primary criteria that should be met:
1. The life extension in experiments with wild type animal models. The geroprotector should prolong the life of the model beyond the intact maximum lifespan, protecting it from one or more mechanisms of aging.
2. Improvement of molecular, cellular, and physiological biomarkers to a younger state or slow down the progression of age-related changes in humans.
3. Most potential geroprotectors are preventive only when applied at relatively high concentrations. The lifespan-extending dose should be several orders of magnitude less than the toxic dose.
4. Minimal side effects at the therapeutic dosage at chronic application.
5. The potential benefit of taking a geroprotector may come after a long period. Potential geroprotectors should initially improve some parameters of health-related quality of life: physical, mental, emotional, or social functioning of the person.
Secondary selection criteria for potential geroprotector:
6. The target or mechanism of action of the geroprotector that extends the lifespan of the model should be evolutionarily conserved.
7. Reproducibility of geroprotective effects on different model organisms increases the possibility that effects will also be discovered in humans, even in the absence of a known conserved target.
8. Candidate geroprotectors should be able to delay the progress of one or several age-associated diseases in humans.
9. Potential geroprotectors should increase organism resistance to unfavorable environmental factors.
This review discusses terpenoid compounds belonging to different classes of this large group of substances, in terms of meeting the criteria for potential geroprotector and their potential for clinical use in relation to age-dependent diseases.
2. Extraction and Analysis of Terpenoids
Detection and structural identification play an important role in elucidating potential activities and developing therapeutic approaches to natural geroprotectors. Currently, advancement has been made in the determination of structure and studying of the chemical features of terpenes and terpenoids, as well as methods for their extraction. These methods are constantly improving, and new approaches are being developed [7].
All terpenes and terpenoids contain a hydrocarbon skeleton, which is formed from five-carbon precursors (isopentenyl diphosphate and dimethylallyl diphosphate) and polymerizes with the formation of prenyl diphosphates of various lengths. As a result of the removal of the diphosphate group, the intermediate allyl cations can be subjected to a series of chemical cascades with the formation of various compounds with linear and/or cyclized hydrocarbon chains, which are then further modified by the addition of various functional groups and adducts [2,3,4]. This feature of terpenes and terpenoids creates their remarkable chemical diversity and requires an appropriate approach for extraction, detection, and purification [8,9,10].
The preparation and analysis of natural compounds include four stages: (1) the release of biologically active compounds; (2) extraction; (3) purification of a target substance from an extract; (4) identification of the chemical structure of a target compound. The use of specific methods is determined depending on the size and complexity of a molecule, its physical properties (polarity, volatility), chemical properties, and some other parameters [7]. The polarity of a molecule is the most important feature that should be considered when determining the method of extraction, analysis, and purification of a substance [8,9,11].
Most primary terpenes without chemical modifications are non-polar. Some of them with the smallest molar mass (especially mono- and sesquiterpenes) can be volatile [12]. Non-volatile non-polar terpenes are extracted with hexane or other non-polar solvents [9]. In addition, to extract this fraction, silica can be used as a stationary phase. For the analysis of the obtained samples of non-polar terpenes, purification of the target molecules, and their structural identification, gas chromatography is used [9]. Additionally, thin layer chromatography (for more accurate identification of specific fractions with the target molecule) and high-performance liquid chromatography (for thorough purification) are applicable [7].
Isolation and purification of volatile non-polar terpenes have limitations associated with the need for their capture and the difficulties of separating substances from each other [7,8]. There is a molecular capture technique and novel approaches, such as solid-phase microextraction [13,14] or microwave-assisted extraction [15]. For structural identification, gas chromatography is also used [12].
The process of extraction and analysis of compounds with modifications requires other methods because they are polar molecules with greater variability of chemical properties. The degree of polarity depends on the type of modifications and their quantity. Modifications by the addition of methyl or hydroxyl groups provide a relatively low polarity of the compounds. For extraction, a suitable approach is the use of hexane (or polar methanol) as a solvent, but for analysis, liquid chromatography. However, it is possible to use gas chromatography, supplemented by derivatization, as well as thin-layer chromatography [7].
Modifications such as acylation, aroylation, glycosylation, and the addition of other functional groups increase the size and polarity of triterpenoids. For the extraction of such fractions, it is recommended to use polar solvents, in particular, methanol (or alternative methods, such as extraction with ionic liquid), and for analysis, high-performance liquid chromatography/electrospray tandem mass spectrometry [16,17]. Additional methods may be required to prepare the samples, depending on the chemical structure of specific terpenes and terpenoids [7,11].
3. Terpenoids as Potential Geroprotectors
3.1. Monoterpenes
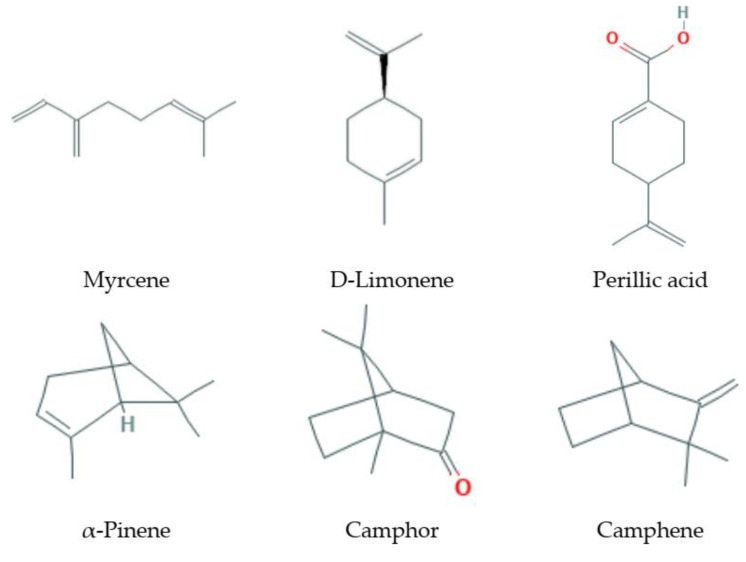
Monoterpenes (Figure 1) are isoprene dimers; they have the composition C10H16. These are easily volatile compounds with a pleasant smell that form the basis of essential oils of plants. According to the structure, monoterpenes are divided into two large groups: acyclic, with an open carbon chain (for example, myrcene, ocimene) and cyclic, which can contain both one cycle (limonene) and several (fennenes, pinenes); some bicyclic monoterpenes contain cyclopropane cycles (sabinene).
Figure 1.
Some monoterpenes that increase lifespan and delay age-related diseases.
3.1.1. Natural Sources
These substances are the main components of essential oils of fruits and the volatile turpentine fraction of conifer oleoresins. Camphene is found in the juniper oil, pettigrein—in the pine oil; dipentene—in oils of bergamot, coriander, sweet dill, lemon; limonene—in oils of bergamot, cumin, carrot, sweet dill, lemon, neroli, orange; pinene—in oils of coriander, cypress, eucalyptus, sweet dill, pine, rosemary; sylvestren—in oils of cypress, pine, and many other tree oils. Monoterpenes myrcene and ocimene are contained in essential oils of hops and laurel. Monoterpene alcohols, such as geraniol, are the main components of essential oils of rose, geranium, and other flower essences. Aldehydes (geranial) have a citrus smell and are contained in lemon essential oils. Camphor, a bicyclic monoterpene, is one of the major constituents of essential oils from rosemary, lavender, and sage [18]. In different parts of pine, the following were found: γ-terpinene and β-pinene—in the needles; limonene—in the bark; α-pinene and limonene—in the pollen [19]. Monoterpenes limonene is contained in the lemon oil and turpentine, and is a part of the oil of cumin. The highest content of limonene was found in representatives of the genus Citrus (Rutaceae) [20,21].
3.1.2. Lifespan Extension on Different Models
The hormetic-like effect of limonene was found in the Mediterranean fruit fly (Ceratitis capitata) model: despite the frank toxicity of limonene in high doses (LD90—39.74 nL per male and 75.51 nL per female), low doses (LD20—3.47 nL per male and 12.26 nL per female) increased the lifespan and female fertility in the case of protein-free diet [22]. Small concentrations of limonene (0.011 and 0.046 mM) also significantly increase both average and maximum lifespan of Drosophila melanogaster [23]. Exposure of the olive fruit flies (Bactrocera oleae) by the aroma of α-pinene, which is present in both olive fruit and leaves, increased the lifespan in males and fecundity in females under dietary restricted conditions [24]. However, studies conducted on the Mediterranean fruit fly did not confirm the pro-longevity effect [22].
3.1.3. Effects on Stress-Resistance
Low doses of camphor could be beneficial, inducing neurohormesis [25] or anti-tumor activity [26]. Camphor as a component of cosmetics can delay skin aging, decreasing the activity of elastase, elevating collagen expressions, activating proliferation of human primary dermal fibroblasts, and attenuating cellular senescence [18]. It has anti-mutagenic effects in small doses [27]. α-Pinene attenuated UV-induced photoaging by inhibiting the expression of matrix metalloproteinases in mouse skin [28]. Antioxidant properties were found for menthol [29] and γ-terpinene [30], these compounds prevent peroxidation of lipids and fatty acids. In Caenorhabditis elegans, the antioxidant activity of the mint essential oil was comparable to ascorbic acid [29]. Perillic acid showed protective properties in radiation-induced oxidative stress [31]. A mixture of geraniol and camphene prevented mitochondrial dysfunction, oxidative stress, and the release of apoptotic proteins in the liver during the nimesulide poisoning in rats [32]. The methanol extract of fennel seeds containing L-limonene softened oxidative stress and protected mouse cells from the damage caused by active forms of oxygen [33].
3.1.4. Toxicity and Side Effects
High doses of camphor have pronounced toxicity. In the Ames test, monoterpenoids (camphor, 1,8-cineol, citral, citronellol, menthol, except for terpineol) showed no mutagenic properties [34]. Low doses of monoterpenes, such as camphor, eucalyptol, and thujone, have a cytoprotective and antimutagenic effect, however, in large doses, their effects are opposite [27]. The products of the interaction of limonene with oxygen (oxygen hydroperoxides) could be aromatic allergens [35].
3.1.5. Life Quality Effects
Monoterpenes are often partially responsible for the aroma or odor of plants and are major odoriferous compounds of many flowers and fruits. Esters of terpene alcohols (citronellol, geraniol, nerol, farnesol, linalool, perillyl alcohol, menthol, borneol, carveol) are described as highly potent, reversible, and low toxic skin penetration enhancers [36]. D-limonene reduces overall stress levels and improves markers of inflammation [37,38,39]. D-limonene in Wistar rats caused the intense and persistent bradycardia associated with hypotension. In the in vivo model of arrhythmia, D-limonene (10 mg/kg) reduced the heart rate and arrhythmia [40]. In experiments with Oreochromis niloticus, Citrus bergamia peel oil containing limonene and linalool was added to the fish diet. Highest levels of total protein and lowest levels of serum cholesterol and triglycerides were observed in fish treated with 0.5 g per 100 g of bergamot oil, and fish growth rates were significantly increased [41].
3.1.6. Suppression of Pro-Aging or Activation of Anti-Aging Molecular Targets or Pathways
Hormesis pathways activated by phytochemicals include NRF2 and FOXO transcription factors that stimulate the production of antioxidant enzymes, protein chaperones, and neurotrophic factors [25]. Camphor induced the proliferation of primary human skin fibroblasts via PI3K/AKT and ERK signaling pathways. It attenuated an increase of the β-galactosidase (SA-β-gal) activity associated with aging, induced the expression of collagen (IA, IIIA, IVA types) and elastin in primary human dermal fibroblasts [18]. Myrcene ameliorates human skin extrinsic aging via decreasing the production of ROS, MMP-1, MMP-3, and IL-6, and increasing of TGF-1 and type I procollagen secretions. Myrcene treatment reduces the induction of mitogen-activated protein kinase (MAPK)-related signaling molecules such as p-ERK, p-p38, and p-JNK, and AP-1 [42]. Abisil, a substance of terpenes of Abies sibirica enchances the activity of a cellular energy sensor—AMPK—in mice [43].
3.1.7. Effects on Age-Related Diseases
Citronellol decreased hyperglycemia in streptozotocin-induced diabetic rats. The addition of citronellol to the STZ diet of rats positively influenced the maintenance of normal histological manifestation of liver cells and insulin-positive β-cells [44]. In a study of effects of limonene and perillic acid in C57BL/6 mice, a significant (65% and 67%) inhibition of the metastatic tumor formation was revealed [45]. Immunomodulatory activity (increase in total leukocyte count) was detected in Balb/c mice after the consumption of limonene and perillic acid [46]. D-limonene has chemopreventive activity against mammary, skin, liver, lung, and forestomach cancer in rodents [47]. D-limonene and its derivatives have chemotherapeutic and chemoprophylactic efficacy in cancer in various preclinical model systems [48]. On the cellular model of osteoarthritis, it was shown that myrcene has significant anti-inflammatory and anti-catabolic effects on human chondrocytes and is able to slow down the destruction of cartilage and the development of osteoarthritis. Myrcene and limonene prevent the increased expression of non-cartilage specific collagen I induced by IL-1β [49]. α-Terpineol has antitumor activity and acts by suppressing the transmission of NF-κB signals [50]. The protective effect of α-terpineol against disruption of synaptic plasticity of the hippocampus and spatial memory after transient cerebral ischemia in rats was revealed by facilitating long-term potentiation and suppressing lipid peroxidation in the hippocampus [51]. γ-Terpineol inhibited cell growth and caused apoptosis in human Bel-7402 cancer cells. A possible anti-cancer mechanism of γ-terpineol on human hepatoma cells is the induction of cellular apoptosis suppressing the growth of tumor cells [52]. Monoterpenes inhibit cell growth, cell cycle progression, and expression of the cyclin D1 gene in human breast cancer cell lines, and cause dose-dependent inhibition of cell proliferation [53]. Camphene reduces plasma cholesterol and triglycerides in rats with hyperlipidemia [54]. Terpenes of Abies sibirica affect molecular pathways associated with cancer and aging in human cells [5], induce apoptosis and inhibit proliferation in tumor cells in vitro, suppress tumor growth and angiogenesis in vivo [43].
3.1.8. Additional Activities
The antibacterial activity of the essential oil of Citrus hystrix with a concentration of 2% (by weight) showed a strong inhibitory effect against Bacillus subtilis and Escherichia coli [21]. Trichophyton rubrum is a fungus that causes chronic dermatophytosis in humans. Geraniol and citronellol exhibit antimicrobial properties damaging cell wall and cell membrane of T. rubrum by inhibiting ergosterol biosynthesis [55]. The essential oil of Santolina impressa, which includes β-pinene, 1,8-cineole, limonene, camphor, has a fungicidal effect on Cryptococcus neoformans, Epidermophyton floccosum, and Trichophytum rubrum [56]. The essential oil of leaves of Psidium guajava inhibited pathogenic human bacteria Curvularia lunata [57]. Essential oils from various aerial parts of Pinus eldarica show antibacterial properties against E. coli (essential oil from pollen). Essential oil from the cortex inhibited the growth of Candida albicans and Staphylococcus aureus, as well as a decreased growth of S. aureus, under the influence of the essential oil from the needles [19].
3.2. Sesquiterpenes
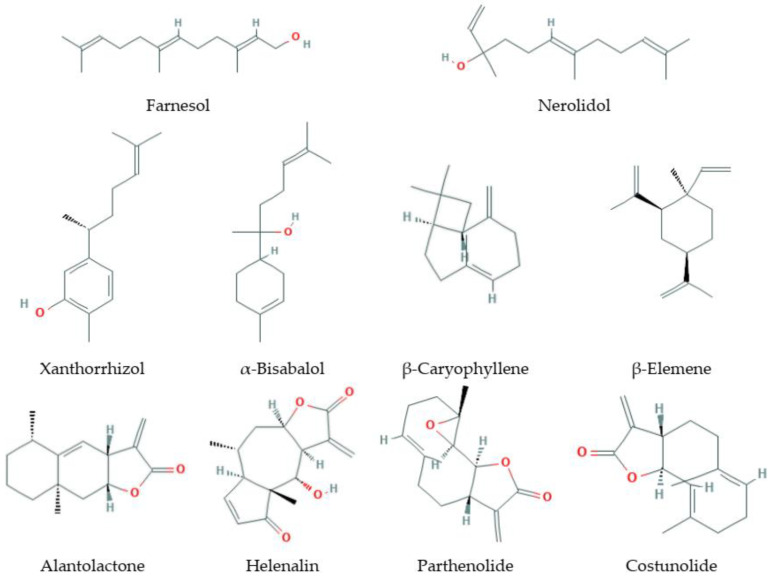
Sesquiterpenes (Figure 2) are C15-terpenoids built from three isoprene units. They are found particularly in higher plants and in many other living systems such as marine organisms and fungi. Usually sesquiterpenes are hydrocarbons or have oxygenated forms including lactones, alcohols, acids, aldehydes, and ketones.
Figure 2.
Some sesquiterpenes that increase lifespan and delay age-related diseases.
3.2.1. Natural Sources
Sesquiterpenes are a group of secondary metabolites in plants comprising a large group of over 5000 known compounds, being most common in families such as Apiaceae, Araceae, Araliaceae, Asteraceae, Cactaceae, Euphorbiaceae, Lamiaceae, Rutaceae, Solanaceae.
3.2.2. Lifespan Extension on Different Models
Lifespan extension experiments were made only for β-caryophyllene. This compound possesses a wide range of biological activities including antioxidant, anti-inflammatory, anti-cancerous, and local anesthetic actions. Pant et al. (2014) used C. elegans as a model system to elucidate the stress modulatory and lifespan prolonging action of β-caryophyllene. It was found that a 50 μM dose of this sesquiterpene increased the lifespan of C. elegans by over 22% and significantly reduced intracellular free radical levels, maintaining cellular redox homeostasis [58].
3.2.3. Effects on Stress-Resistance
It was shown that farnesol quells oxidative stress, reactive gliosis, and inflammation during acrylamide-induced neurotoxicity [59]. Nerolidol exhibits a protective effect against pentylenetetrazol-induced kindling, oxidative stress, and associated behavioral comorbidities in mice [60]. This compound has an effect against neuroinflammation, and oxidative stress induced by rotenone [61]. α-Bisabolol also has a protective effect on rotenone-induced toxicity in D. melanogaster [62], on altered hemodynamics, lipid peroxidation, and nonenzymatic antioxidants in isoproterenol-induced myocardial infarction [63]. It exhibits nephroprotective effects against ischemic-reperfusion acute kidney injury [64]. β-Elemene has a beneficial effect in traumatic brain injury by inflammatory pathway [65]. Xanthorrhizol prevents amyloid-β-induced oxidative modification and inactivation of neprilysin [66]. β-Caryophyllene has an antioxidant effect and plays a protective role for rat liver from CCl4-induced fibrosis by inhibiting hepatic stellate cell activation [67]. Furthermore, β-caryophyllene oxide has effects on xenobiotic-metabolizing enzymes in mice in vivo [68]. Costunolide protects mice against D-galactosamine and lipopolysaccharide-induced acute liver injury [69]. It exhibits antioxidant activity [70], for example, in experiment with H2O2-induced injury in PC12 cells [71]. In addition, this lactone protects against an ethanol-induced gastric ulcer in mice [72]. Parthenolide has a protective effect on LPS-induced acute lung injury [73]. Helenalin attenuates alcohol-induced hepatic fibrosis by enhancing ethanol metabolism, inhibiting oxidative stress, and suppressing HSC activation [74]. Alantolactone plays neuroprotective roles in traumatic brain injury in rats via anti-inflammatory, anti-oxidative, and anti-apoptosis pathways [75]. It prevents amyloid β25-35-induced toxicity in mouse cortical neurons and scopolamine-induced cognitive impairment in mice [76].
3.2.4. Effects on Aging Biomarkers
β-Elemene decreased levels of plasma endotoxin, serum TNF-α, and hepatic CD14 expression in rats with liver fibrosis [77]. Xanthorrhizol has hypolipidemic activities [78]. β-Caryophyllene demonstrated a hypocholesterolemic effect in rats fed cholesterol and fat-enriched diet [79]. It reduced the atherogenic index and coronary risk index in hypercholesterolemic rats [80] and protected in vitro neurovascular unit against oxygen-glucose deprivation and reoxygenation-induced injury [81], also it made a significant reduction in intestinal lipofuscin levels [58].
3.2.5. Toxicity and Side Effects
Nerolidol downregulates mitochondrial and cellular energetics [82]. It was also tested in in vivo genotoxicity assessment of nerolidol [83], demonstrating weak levels of dose-related DNA damage, and enhancing the average number of micronucleated cells. α-Bisabolol promotes cell death by inducing pores in mitochondria and lysosomes [84]. β-Caryophyllene showed the absence of adverse effects in female Swiss mice [85], and in a subchronic toxicity study in rats [86]. Costunolide induces micronuclei formation, chromosomal aberrations, cell cycle arrest, and mitochondrial-mediated apoptosis in Chinese hamster ovary cells [87]. Helenalin suppresses essential immune functions of activated CD4+ T-cells [88], Leydig, and adrenocortical cell steroidogenesis by inhibiting expression of the steroidogenic acute regulatory protein [89].
3.2.6. Life Quality Effects
Farnesol exerts anti-inflammatory and anti-allergic effects on ovalbumin-sensitized and challenged asthmatic mice [90], ameliorates serum allergic antibody titers and lipid profiles [91], exerts an antinociceptive effect as confirmed by histopathological analysis of the striatum and hippocampus in mice [92]. β-Caryophyllene modulates feeding behavior [58] and has a therapeutic potential from several pharmacological points [93].
3.2.7. Suppression of Pro-Aging or Activation of Anti-Aging Molecular Targets or Pathways
Farnesol and nerolidol can induce apoptosis of cancer cells [94,95]. β-Caryophyllene oxide potentiates TNF-α-induced apoptosis, and it inhibits invasion through down-modulation of NF-κB-regulated gene products [96]. β-Elemene enhances GAP-43 expression and neurite outgrowth by inhibiting RhoA kinase activation in rats with spinal cord injury [97]. It inhibits the proliferation of primary human airway granulation fibroblasts by downregulating the canonical Wnt/β-catenin pathway [98], attenuates macrophage activation and proinflammatory factor production via crosstalk with Wnt/β-catenin signaling pathway [99], induces the apoptosis of non-small-cell lung carcinoma cells through PERK/IRE1α/ATF6 pathway [100]. β-Elemene acts as an antitumor factor and downregulates the expression of survivin, Bcl-xL, and Mta-1 [101]. Xanthorrhizol induces apoptosis through ROS-mediated MAPK activation in human oral squamous cell carcinoma cells and inhibits DMBA-induced oral carcinogenesis in hamsters [102,103]. It was shown that xanthorrhizol regulates p53-, Bcl-2-, and caspase-dependent signaling pathway and induces apoptosis in HepG2 hepatoma cells [104]. β-Caryophyllene interacts with the main anti-aging genes SIR-2.1, SKN-1, and DAF-16 in C. elegans [58]. It attenuates focal cerebral ischemia-reperfusion injury via NRF2/HO-1 pathway in rats [105] and alleviates D-galactosamine and lipopolysaccharide-induced hepatic injury through suppression of the TLR4 and RAGE signaling pathways [106]. It exhibits antigenotoxic capacity in mice and has antioxidant and GST induction activities [107]. In addition, this compound ameliorates the Alzheimer-like phenotype in APP/PS1 mice through CB2 receptor activation and the PPARγ pathway [108].
Costunolide ameliorates lipoteichoic acid-induced acute lung injury via attenuating MAPK signaling pathway [109]. Activation of p53 by costunolide blocks glutaminolysis and inhibits proliferation in human colorectal cancer cells [110]. This compound protects lipopolysaccharide/D-galactosamine-induced acute liver injury in mice by inhibiting the NF-κB signaling pathway [111]. It inhibits proinflammatory cytokines and iNOS in activated murine BV2 microglia [112] and reveals inhibitory effects on the telomerase activity in human breast carcinoma cells [113]. Parthenolide is a direct inhibitor of the inflammasome [114]. It inhibits STAT3 signaling by covalently targeting Janus kinases [115], Wnt/β-catenin signaling by blocking synthesis of the transcriptional regulators TCF4/LEF1 [116] and FAK-mediated cell invasion [117]. Hsp72 is another intracellular target of this lactone [118]. Helenalin has differential effects on the proteome, metabolome, and the oxidative stress response in several immune cell types [119]. NF-κB p65 repression by helenalin contributes to the induction of autophagy cell death [120,121]. It produces inhibitory effects on 5-lipoxygenase and leukotriene C(4) synthase in human blood cells [122] and telomerase activity, attributed to the alkylation of the CYS445 residue [123]. Alantolactone induces apoptosis and suppresses migration in MCF-7 human breast cancer cells via the p38 MAPK, NF-κB, and NRF2 signaling pathways [124]. This lactone produces NRF2-mediated induction of detoxifying enzymes [125] and activation of apoptosis in human hepatoma cells [126]. Alantolactone exerts anti-inflammatory effects by inhibiting chemokine production and STAT1 phosphorylation in TNF-α and IFN-γ-induced in HaCaT cells [127] and sensitizes human pancreatic cancer cells to EGFR inhibitors through the inhibition of STAT3 signaling [128].
3.2.8. Effects on Age-Related Diseases
Farnesol has an anti-obesity effect in high fat diet-induced obese mice and induces the development of beige adipocytes in human adipose tissue-derived mesenchymal stem cells [129]. This compound has potential anti-inflammatory and anti-cancer properties [130]. Cardioprotection by farnesol includes the role of the mevalonate pathway [131]. Nerolidol has a different pharmacological application in treating neurodegenerative diseases [132]. Nerolidol demonstrates anticholinesterase, antioxidant, anti-nociceptive, anti-inflammatory, and anxiolytic activities, thus it is considered as a promising phytochemical for the development of therapeutic drugs [133]. This compound has a neuroprotective effect against neuroinflammation, and oxidative stress induced by rotenone [61]. It exhibits anti-nociceptive and anti-inflammatory activity with the involvement of the GABAergic system and proinflammatory cytokines [134]. α-Bisabolol prevents neuronal damage and memory deficits through the reduction of proinflammatory markers induced by permanent focal cerebral ischemia in mice [135]. It reduces pro-inflammatory cytokine production and ameliorates skin inflammation [136]. This compound exhibits anti-nociceptive and anti-inflammatory activities in rodents [137,138]. Xanthorrhizol possesses antioxidant and anti-inflammatory activities in hippocampal neurons and primary cultured microglia [139]. It was shown anti-hyperglycemic and anti-inflammatory effects of xanthorrhizol in high-fat diet-induced obese mice [140]. This sesquiterpene demonstrates diverse pharmacological activities and anticancer properties [141]. It could be used as a pharmaceutical agent in disease management including cancer, infectious diseases, inflammatory process, metabolic syndrome, and platelet disorder. β-Elemene attenuates atherosclerosis in apolipoprotein E-deficient mice via restoring NO levels and alleviating oxidative stress [142]. It reduces the progression of atherosclerosis in rabbits [143]. β-Elemene has diverse mechanisms of influence on cancer cell interaction [144,145]. β-Caryophyllene has many effects on diseases of the nervous system. It exhibits a neuroprotective effect on cerebral ischemia-reperfusion injury via regulation of necroptotic neuronal death and inflammation [146]. β-Caryophyllene attenuates oxidative stress, neuroinflammation, glial activation, and salvages dopaminergic neurons in a rat model of Parkinson’s disease [147]. It showed a neuroprotective effect against dopaminergic neuron injury in a murine model of Parkinson’s disease induced by MPTP [148] and against cerebral ischemic injury [149]. β-Caryophyllene has antioxidant, anti-inflammatory, anticancer, cardioprotective, hepatoprotective, gastroprotective, nephroprotective, antimicrobial, and immune-modulatory activity [150]. Costunolide suppresses inflammatory angiogenic response in a subcutaneous murine sponge model [151] and ameliorates the inflammatory process associated with experimental pleurisy in mice [152]. Parthenolide has effects on neurological diseases, cancer, metabolism regulation and inflammation, inhibits the initiation of experimental autoimmune neuritis [153], relieves pain and promotes M2 microglia/macrophage polarization in a rat model of neuropathy [154], shows a hepatoprotective effect in a rat model of nonalcoholic fatty liver disease [155]. It acts as an NF-κB inhibitor, that ameliorates diabetes-induced behavioral deficit, neurotransmitter imbalance, and neuroinflammation in the type 2 diabetes rat model [156]. Parthenolide shows anti-inflammatory effects [157], inhibiting pro-inflammatory cytokine production and exhibiting protection of collagen-induced arthritis in a rat [158].
3.2.9. Additional Activities
Farnesol interacts with thyroid hormone receptor (THR) β 1 and inhibits THR-mediated signaling in MCF-7 human breast cancer cells [159]. Local delivery of β-elemene improves locomotor functional recovery by alleviating endoplasmic reticulum stress and reducing neuronal apoptosis in rats with spinal cord injury [160]. β-Elemene influences α-tubulin polymerization in human hepatoma HepG2 cells [161] and supplies protection of endothelial cells, inhibition of neointimal hyperplasia in an injured artery [162]. β-Caryophyllene protects against alcoholic steatohepatitis by attenuating inflammation and metabolic dysregulation in mice [163]. This compound promotes osteoblastic mineralization and suppresses osteoclastogenesis and adipogenesis in mouse bone marrow cultures in vitro [164]. It modulates carbohydrate metabolism in streptozotocin-induced diabetic rats [165] and supplies gastric cytoprotection [166]. Xanthorrhizol demonstrates estrogenic activity [167] and has antibacterial activity against foodborne pathogens [168].
3.3. Diterpenes
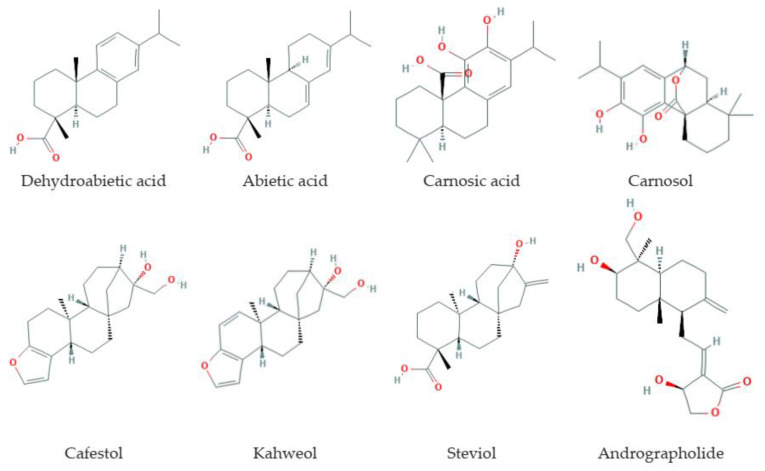
Diterpenes (Figure 3) are C20-terpenoids constructed from four isoprene links, with the general molecular formula (C10H16)2. Diterpenes can have a linear, bi-, tri-, tetra-, and penta- or macrocyclic structure, depending on their skeletal core. In nature, diterpenes are usually found in the polyoxygenated form with keto- and hydroxyl groups. Diterpenes are the basis of biologically important compounds such as retinol, retinal, and phytol.
Figure 3.
Some diterpenes that increase lifespan and delay age-related diseases.
3.3.1. Natural Sources
Diterpenes are widely distributed in plants of families such as Lamiaceae, Verbenaceae, Celastraceae, Euphorbiaceae, Thymelaeaceae, as well as in some genera such as Rhododendron and Taxus. Diterpenes and their derivatives are found in several food products (coffee, spices). Coffee diterpene alcohols, cafestol and kahweol, were isolated from green coffee beans (Coffea arabica L.). Carnosic acid (salvin) and carnosol are phenolic diterpenes present in spicy plants of the Lamiaceae family, such as rosemary (Rosmarinus officinalis), sage (Salvia officinalis, Salvia pachyphylla), oregano (Origanum vulgare) [169,170,171], basil (Ocimum basilicum), and thyme (Thymus vulgaris). Abietic and dehydroabietic acids are isolated from coniferous plants such as Pinus densiflora, P. sylvestris, Abies grandis [172], but also produced by representatives of several genera of cyanobacteria [173]. Andrographolide—bicyclic labdanum diterpenoid isolated from the stem and leaves of the medicinal plant Andrographis paniculata Nees. (Acanthaceae). Steviol is found in the leaves of Stevia rebaudiana Bertoni. (Asteraceae) [174]. Afidicoline is a tetracyclic diterpene antibiotic isolated from the fungus Cephalosporum aphidicola, also synthesized by Nigrospora oryzae [175].
3.3.2. Lifespan Extension on Different Models
Effects of increased lifespan were observed for dehydroabietic acid and carnosol in experiments on C. elegans. Dehydroabietic acid has been shown to increase the lifespan in C. elegans, as well as prevent the accumulation of lipofuscin and the process of fibrosis [172]. Nematodes treated with carnosol and carnosic acid were characterized by an increase in average and maximum lifespan [176,177].
3.3.3. Effects on Stress-Resistance
Diterpenes and diterpenoids have antioxidant activity. Carnosol and carnosic acid are inhibitors of lipid peroxidation, they prevent the oxidation of fatty acids, triglycerides, low-density lipoproteins in human aortic endothelial cells [178,179]. Under oxidative stress, nematodes treated with carnosol had a 21% increase in lifespan compared to controls, and under heat stress increased worm survival was higher by 9% [176]. The combined action of carnosic acid and carnosol against ROS and lipid radicals makes this diterpenoid tandem an effective antioxidant defense. In the Ames test, carnosol was found to have significant antioxidant and anti-mutagenic activity comparable to ascorbic acid [180]. In a micronucleus test, it was found that carnosol is even more effective than ascorbic acid in protection against gamma radiation [181]. Carnosol protects cells from eco-toxicants [182].
3.3.4. Effects on Aging Biomarkers
Diterpenes affect the molecular, metabolic, and functional biomarkers of aging. The anti-aging effects of dehydroabietic acid are mediated by the activation of SIRT1 [172]. Diterpenes isolated from the leaf extract of Croton tonkinensis showed inhibitory activity on SIRT1 [183]. The effect of the cafestol is due to the inhibition of secretion of ICAM-1, MCP-1, and IL-8 and inhibition of phosphorylation of ERK and p38. The mechanism of action of cafestol is associated with the activation of HO-1 and SIRT1 [184].
3.3.5. Toxicity and Side Effects
Diterpenes exhibit low toxicity. When using carnosic acid and carnosol, no visible damage to the body was observed, except for liver obesity in mice subjected to repeated administration of rosemary extract [185]. In animal and cell cultures, it has been shown that cafestol and kahweol show a wide range of biochemical effects, leading to a decrease in the genotoxicity of several carcinogens [186]. Steviol glycosides do not cause acute and subacute toxicity, allergic reactions, and they are not teratogenic, mutagenic, and carcinogenic substances. Their safety has been confirmed in numerous toxicological studies [187,188]. A few examples of side effects of some diterpenes are described. Therefore, high doses of taxol cause hair loss, bone marrow suppression, anemia, allergic reactions, muscle pain and diarrhea, heart problems, increased risk of infection, pneumonia, and neuropathy. The use of taxol during pregnancy is likely to cause birth defects in a fetus [189]. Abietic acid and dehydroabietic acid have pro-tumorigenesis cell transformation activity [190]. Dehydroabietic acid contributes to growth alterations and reproductive disturbances and affects liver energy metabolism in fishes [191,192]. In addition, diterpene abietane acids from pine needles and tips are abortifacient and toxic that was found in a study on cattle [193].
3.3.6. Life Quality Effects
The effect of rosemary extract and its main components, rosmarinic and carnosic acids, on SOD1-G93A transgenic mice, which are models of amyotrophic lateral sclerosis, was studied. Rosemary diterpenes significantly delayed motor dysfunction, weakening the degeneration of motor neurons and increasing the lifespan of mice, improved clinical assessment, and reduced body weight loss [194]. Carnosic acid reduced the accumulation of epididymal fat in mice [195]. Carnosol slowed down the processes associated with aging, including age-related pigmentation and neurodegenerative diseases, while it did not affect fertility and fat deposition [176]. Kahweol and cafestol influenced the formation of bone tissue, inhibiting the differentiation of osteoclasts. Cafestol had an inhibitory effect on osteoclastogenesis and contributed to the differentiation of osteoblasts [196].
3.3.7. Suppression of Pro-Aging or Activation of Anti-Aging Molecular Targets or Pathways
Diterpenes and diterpenoids affect several signaling pathways associated with aging, such as mTOR/AKT/PI3K, SIRT1, MAPK, NRF2, NF-κB, HSF1/HSP. Dehydroabietic acid shows itself to be an anti-aging agent that provides direct activation of SIRT1 [172]. Carnosic acid and carnosol induce the expression of heme oxygenase-1 (HO-1) gene and neuronal growth factors by inducing NRF2 and thus provide neuroprotective action. Carnosic acid has anti-inflammatory activity [197] and promotes neuronal differentiation [198]. Carnosol has anti-cancer, anti-inflammatory, and antioxidant effects mediated by modulating signaling cascades, including effects on molecules that regulate apoptosis (Bax/Bcl2), cell survival and proliferation (AKT/mTOR, MAPK), transcription factors, NF-κB, STAT3-6, and steroid androgen and estrogen receptors [199]. Kahweol enhanced the expression of HO-1, which provides neuroprotection from oxidative damage caused by 6-OHDA, and induced activation of PI3K and p38 [200]. Cafestol targets in endothelial cells are mitogen-activated protein kinases (MAPK), NRF2/HO-1 signaling pathway, and SIRT1 [184]. Cafestol weakened the action of intercellular adhesion molecules-1 (ICAM-1), monocyte chemoattractant protein (MSR-1), and the secretion of interleukin IL-8. Stevioside attenuated inflammation by reducing the expression of cytokine genes IL-6, TNF-α, and IL-1β in the mammary glands of mice infected with S. aureus, and inhibited the expression of cytokine genes by inactivating the MAPK, TLR2, and NF-κB pathways [201], suppressed proinflammatory cytokines [202,203,204]. Steviol and stevioside reduced the expression of IL-6, TNF-α, and IL-1β by inactivating NF-κB and activating Iκba in human colon carcinoma and THP-1 in vitro. These substances inhibit proinflammatory cytokines by increasing the level of IκBa [205,206]. Stevia leaf extract can effectively modulate the immune response and inhibit immunological disorders.
3.3.8. Effects on Age-Related Diseases
Diterpenes and diterpenoids can be used in the prevention and complex treatment of cancer, neurodegenerative, cardiovascular, and metabolic disorders. Anticancer activity is indicated for taxol, abietic acid, andrographolide, kahweol and cafestol, steviol, and carnosol. Carnosic acid and carnosol exhibit antioxidant, anti-inflammatory, anticarcinogenic, and neuroprotective activity [199]. Dehydroabietic acid protects against ulcers and positively affects the state of the cardiovascular system [207,208]. Rosemary diterpenes—carnosic acid and carnosol—improve the redox status of the mammalian brain and modulate neuroinflammation, acting as neuroprotectors [209]. The inhibitory effect of carnosic acid on neurodegeneration in the CA1 region of the hippocampus in an experimental model of Alzheimer’s disease in rats was noted. Carnosic acid prevents obesity and glucose intolerance in mice, activates AKT and AMPKα signaling, enhances glucose uptake by skeletal muscle cells, reduces body weight and epididymal fat accumulation [195]. Kahweol has anti-inflammatory and anti-angiogenic effects. Cafestol helps reduce the risk of type II diabetes by stimulating insulin secretion and increasing glucose uptake in muscle cells [210,211]. Cafestol reduces the overall expression of inflammatory molecules in endothelial cells, inhibits the proliferation of vascular endothelial cells [184]. Cafestol palate and kahweol act against angiogenesis-dependent disorders [212]. Coffee extract with caffeine and cafestol are promising agents for controlling age-related neurodegenerative diseases due to their high bioavailability and low toxicity [213]. Coffee diterpenoids have a positive effect on model animals with symptoms of neurodegenerative diseases. Neuroprotective effects are shown in Drosophila models of Alzheimer’s disease and polyglutamine disease [214].
Stevia rebaudiana leaf extract has an antidiabetic effect by lowering blood glucose levels in patients with type 2 diabetes [215]. Isosteviol exhibits anti-inflammatory, antihypertensive activity, regulates blood lipids, is an immunomodulator, inhibits DNA polymerase and DNA topoisomerase, having anti-tumor, antioxidant, and anti-tuberculosis effects [187]. Stevioside and steviol affect β-cells and stimulate insulin secretion in mice and rats [216,217,218]. Steviol glycosides can lower blood pressure by modulating calcium and potassium channels, and repeated administration of stevioside in both normal and hypertensive mice led to an increase in glomerular filtration and renal blood flow [187,219]. Stevioside has an antihyperglycemic and hypotensive effect [217]. Clerodane derivatives have an NGF-potentiating effect and significantly increase NGF-mediated neurite growth in PC12 cells and show antiulcer activity [220]. Andrographolide demonstrates anti-inflammatory [221] and antibacterial activity [222], exhibits anti-allergic [223], antioxidant [224], and anti-cancer effects [225,226].
3.3.9. Additional Activities
Diterpenes are diterpenoids and have antiviral, antibacterial, antiparasitic, and antifungal, antiprotozoal action. Some terpenoids are toxic to microorganisms and insects and play an important role in plant protection [220,227]. Extract of Stevia and its glycosides (for example, steviol), in addition to their value as sweeteners, have a therapeutic effect against cystic fibrosis. Carnosic acid and carnosol, isosteviol, andrographolide, dehydroabietic acid have several important protective properties, including anti-tuberculosis, antiseptic, and can be used in the treatment of colds, showing antibacterial and antiviral activity [171,187].
3.4. Triterpenes
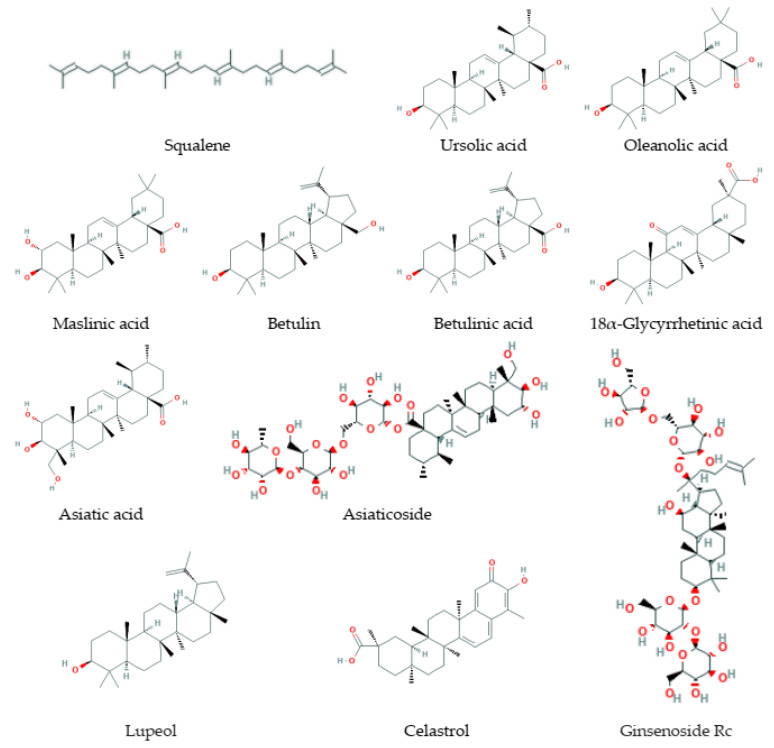
Triterpenes (Figure 4) are derived from the C30 precursor, squalene, that consists of six C5 isoprene units with following cyclization and generation of downstream triterpenoid structures such as steroids, sterols, saponins (glycosides), and others [228,229]. Triterpenoids comprise more than 20,000 recognized molecules [230]. Triterpenes and triterpenoids play an important physiological role in a cell and an organism. These compounds are essential for cellular membrane formation and function and are a basis for the formation of signaling molecules such as steroid hormones and cognate receptors. Squalene is cyclized to lanosterol (a primary cholesterol and ergosterol precursor) and to cycloartenol (a precursor of β-sitosterol) [228]. Triterpenes and their metabolites demonstrate a wide range of biological activities against aging, inflammation, cancerogenesis, neurodegenerative, cardiovascular, metabolic diseases, viral, bacterial, and fungal infections [229,231,232,233,234,235].
Figure 4.
Some triterpenes that increase lifespan and delay age-related diseases.
3.4.1. Natural Sources
Triterpenes and triterpenoids are biosynthesized by all known forms of life including bacteria, plants, fungus, and animals. Squalene is found both in many plants and animals including humans, it is an intermediate in the biosynthesis of phytosterol or cholesterol [236,237,238,239]. Triterpenes and triterpenoids with potential anti-aging activity are abundant in many plants. For example, oleanolic acid, ursolic acid, and betulinic acid are pentacyclic triterpene compounds contained in leaves, roots, and fruits of many plant species [240,241]. Some groups of triterpenoids are specific for plant species or families, particularly, ginsenosides (for Panax ginseng) [242] and limonoids (for citrus) [229]. Several triterpenes and triterpenoids were found in fungi, such as in fomitopsis (Fomitopsis pinicola), Poria (Wolfiporia extensa), and Reishi fungi (Ganoderma lucidum) [243,244,245]. Biological activity was also shown for synthetic triterpenoids [231,246,247,248], as well as there are methods for stimulation of triterpenoid production in medical plants [249].
3.4.2. Lifespan Extension on Different Models
Ursolic and oleanolic acids were described as phytochemicals with significant pro-longevity action. The treatment with 25–50 µM ursolic acid and plant extracts reached with this compound increased the mean and maximum lifespan of C. elegans and D. melanogaster up to 30% [250,251,252,253]. Oleanolic acid at the 100–600 μM concentration extends the mean lifespan of nematodes by 10–20% [254]. Stachys lavandulifolia extracts that have betulin, betulinic acid, oleanolic acid, and ursolic acid among constituents sufficiently extend the Drosophila life [253].
Other potential geroprotectors are cucurbitane triterpenoids. Sea cucumber (Holothuria scabra) extracts containing triterpene glycosides increased the nematode’s mean lifespan by 5–8% [255]. Cucurbitane triterpenoids from Momordica charantia fruits extended the yeast replicative lifespan as well [256]. In addition, ginsenoside Rc increased the longevity of nematodes grown on media with different cholesterol content [257].
18α-glycyrrhetinic acid in wild-type nematodes and in a model of Alzheimer’s disease [258]. Some other triterpenoids (betulin, azadiradione, celastrol) demonstrated a pro-longevity action in model animals with symptoms of neurodegenerative diseases as well (Parkinson’s disease, Huntington’s disease, amyotrophic lateral sclerosis) [259,260,261].
3.4.3. Effects on Stress-Resistance
Pro-longevity action of triterpenes and triterpenoids is associated with increased resistance to environmental stressors. Particularly, oleanolic acid [254], ursolic acid [250,251], as well as sea cucumber extract contained triterpene glycosides [255] improved the survival of nematode C. elegans in conditions of paraquat treatment and high temperatures. The cucurbitane glycoside increases yeast survival under oxidative stress and decreases ROS level [256]. This positive action can be mediated by the ROS scavenging activity and activation of antioxidant defense [251,254,262,263].
3.4.4. Effects on Aging Biomarkers
Triterpenoids attenuated molecular, metabolomic, and functional aging biomarkers.
Studies carried on cultures of mammalian and human cells revealed that triterpenoids (for example, ginsenoside Rg1 and lupeol) inhibit cellular senescence markers, p53, p16, p21, Rb, SA-β-galactosidase, upregulated by senescence-induced treatments such as D-galactose or UV light [262,264]. Ursolic acid was shown to increase molecular anti-aging biomarkers, SIRT1, SIRT6, PGC-1β, and α-Klotho, in mouse hypothalamus [265].
Squalene improved the mitochondrial energy status in the liver of aged mice. This triterpene minimized alterations in the activity of tricarboxylic acid cycle enzymes and respiratory marker enzymes [266]. In rats, red ginseng extract with triterpene saponins restored nine biomarkers related to energy and lipid metabolism, that demonstrated the prevention of age-associated impairment of kidney function and amino acid metabolism disorders [267]. Additionally, the application of ginsenoside Rg1 in mice with D-galactose-induced damage improved oxidation-associated biomarkers, pro-inflammatory cytokine secretion, expression of senescence-associated proteins, and prevented the premature ovarian failure that indicated it possible activity against loss of reproductive functions due to aging-related pathologies [268].
The combination of Spirulina and glycyrrhizin, a saponin from licorice root, prevents cognitive dysfunction in aged rats with obesity. This effect was accompanied by a decrease of glucose, cholesterol, leptin levels in the serum, as well as a reduction in acetylcholinesterase activity in the hippocampus [269]. Maslinic acid supplementation provides maintaining muscular functions in elderly persons [270].
3.4.5. Toxicity and Side Effects
Most triterpenes and triterpenoids that can be used as dietary components have low toxicity. For example, the subchronic toxicity and genotoxicity study of cycloastragenol did not reveal treatment-related mortality, sufficient side effects on different physiological parameters, or mutagenic action [271].
Undesirable effects of triterpenoids can be associated with its impact on reproductive functions. For example, consumption of ursolic acid by rats in an amount of 5 mg per kg of body weight suppresses spermatogenesis [272]. Additionally, triterpenes and triterpenoids can lead to gastrointestinal upsets [273].
3.4.6. Life Quality Effects
Triterpenoids, for example, ursolic acid, and echinocystic acid, are described to improve learning ability and memory in mice [274,275]. Ginsenoside Rg3 and Rh2 from red ginseng roots have a calming effect [276]. Another positive action of triterpenoids is the stimulation and maintaining of physical activity. In rodents, treatment with ursolic acid or celastrol improves weight loss, condition of muscle tissue and muscle mass elevation, increases stamina, helps to maintain high motor performance for a longer time [260,277,278,279,280]. In Drosophila, ursolic acid increases in climbing activity. Furthermore, this triterpenoid led to flies’ microbiota changes that contribute to life- and healthspan extension [281]. Maslinic acid sufficiently improved mobility in the elderly. Particularly, its supplementation in the combination with moderate resistance training increased upper muscle mass and reduced knee pain, preventing disability [270].
3.4.7. Suppression of Pro-Aging or Activation of Anti-Aging Molecular Targets or Pathways
Triterpenes and triterpenoids influence a set of pro- and anti-aging signaling pathways, particularly mTOR/AKT/PI3K, AMPK, SIRT1, MAPK, FOXO, NRF2, NF-κB, HSF1/HSPs pathways.
A set of triterpenoids demonstrate the ability to reduce phosphorylation levels of the mechanistic target of rapamycin (mTOR) and mTOR/AKT/PI3K pathway that lead to delay in aging and aging-related disorders (including cancer and neurodegeneration) [262,282,283,284,285,286,287,288]. Inhibition of mTOR mediates AMPK activation and autophagy induction, which play pro-longevity and anti-tumor role [283,284,285,286,287,288]. At the same time, ursolic acid in combination with leucine can stimulate myoblast differentiation and muscle mass increase through the induction of the mTOR pathway [278]. In addition, triterpenoid compounds exert anti-inflammatory action inhibiting NF-κB, COX-2, iNOS, TNF-α [260,289,290,291,292,293,294,295].
Triterpenoids and triterpenoid-containing extracts stimulate expression and enhance enzymatic activity of pro-longevity proteins SIRT1, SIRT6, PGC-1α, and JNK [251,265,275,281,296,297,298]. Furthermore, in silico, in vitro, and in vivo experiments demonstrated that ursolic acid directly binds to the outer surface of mammalian SIRT1 [297], as well as links with nematode JNK-1 ATP-binding site [250]. Oleanolic acid and sea cucumber extract modulated nuclear localization of FOXO/DAF-16 transcription factor and stimulated the activity of its downstream target genes sod-3, hsp-16.2, and ctl-1 [254,255], as well as ursolic acid-activated SKN-1/NRF2 target genes gcs-1 and daf-9 [251] in C. elegans. Triterpenes and triterpenoids restored the normal expression of antioxidant enzymes (particularly, superoxide dismutase, catalase, glutathione peroxidase, glutathione reductase, glutathione-S-transferase—downstream members of FOXO and NRF2 signaling pathways) and the level of antioxidant molecules including glutathione, improved markers of oxidative stress in different tissues of rodent models [262,266,299,300,301]. At the same time, lifespan studies carried on C. elegans showed that pro-longevity effects of treatment with triterpenoids were abolished or decreased by mutations in sir-2.1, jkk-1, jnk-1, sek-1, osr-1, daf-16, age-1, eat-2, skn-1 genes [250,251,254,255]. Additionally, triterpenoids were found to activate heat shock response (including HSF1 and HSP70 upregulation) and unfolded protein response [244,259,260].
3.4.8. Effects on Age-Related Diseases
Triterpenes and triterpenoids can be used in the prevention and complex therapy of cancers, neurodegenerative, cardiovascular, and metabolic disorders.
At the present time, there are a number of pieces of evidence that demonstrated anti-cancer properties of the pentacyclic triterpenoids of oleanane-, ursane, lupane, and friedelane types (including oleanolic, ursolic, betulinic, 18α-glycyrrhetinic, asiatic acids, celastrol, lupeol, among others) [231,302]. These compounds suppress tumor growth, reduce survival, and induce apoptosis of different types of cancer cells, including skin, breast, colon, prostate tumor cells, and others [290,303,304]. However, treatment with pentacyclic triterpenoids at effective anti-cancer concentrations had a toxic effect on normal cells in some cases [304]. A promising anti-cancer potential has triterpenoids extracted from formopsis, poria, and Reishi fungi. In vitro studies demonstrated their effects in murine Sarcoma cancer cell line and human leukemia, liver cancer, esophageal cancer, pancreatic cancer, prostate cancer cell lines [243,244,305]. In vivo study also revealed anti-tumor effects and demonstrated the survival improvement of tumor-bearing mice [243,244]. Furthermore, fungus triterpenoids had a little toxic impact on normal cells and tissues.
In addition, a number of semisynthetic derivatives of pentacyclic triterpenoids have been synthesized. Some of them were shown to have improved therapeutic activity, pharmacokinetic properties, and less toxicity for normal cells and tissues compared with parent compounds [231,247,248]. For the enhancing of bioavailability and therapeutic efficiency, delivery nanosystems are developed for triterpenoids such as ursolic, oleanolic, and betulinic acids [306,307]. Squalene can be used as an adjuvant in cancer chemotherapy and protect normal tissues against the toxic influence of some anti-cancer agents [308,309,310]. Squalene-based nanoparticles with cisplatin, doxorubicin, or paclitaxel were developed as prodrugs for targeted chemotherapy [311,312,313]. This approach can be also used for the treatment of other disorders. Particularly, squalene-adenosine nanoparticles have a high potential for the neuroprotection in stroke and spinal cord injury [314].
Triterpenes and triterpenoids can be used for the treatment of neurodegenerative disorders such as Alzheimer’s disease and Parkinson’s disease, and for the prevention of aging-dependent cognitive impairment. These compounds improve cognitive functions, learning and memory abilities, prevent synaptic plasticity dysfunction, β-amyloid peptide (Aβ) deposition and toxicity, suppress senescence and death of neural stem and neuronal cells, decrease inflammation and oxidative stress, correct metabolic and hormonal imbalance in models of accelerated senescence and neurodegeneration in vivo and in vitro [258,262,301,315,316,317,318,319].
Squalene and a number of triterpenoids can be used as cardioprotector agents due to their ability to reduce levels of low-density lipoprotein cholesterol (with a rise in the level of high-density lipoprotein cholesterol) and triglycerides, antioxidant and anti-inflammatory properties [289,320,321,322,323,324,325]. Triterpene compounds prevent structural changes in the myocardium, development of cardiovascular pathologies, and support normal cardiac function. In studies on rats, the protection action of squalene and triterpenoids against myocardial infarction, blood pressure increase, ischemia-reperfusion injury, chronic heart failure was found [320,321,322,323].
A range of pentacyclic triterpenoids contribute to metabolic syndrome through the regulation of proteins and signaling pathways involved in adipogenesis, lipolysis, fatty acid oxidation, insulin resistance, mitochondria biogenesis, gluconeogenesis, oxidative stress, and inflammation [233]. Squalene and triterpenoids can reverse hyperglycemia status, which is useful for the treatment of such metabolic diseases as diabetes mellitus and obesity. These compounds decrease levels of glucose in blood and triglycerides in the liver, stimulate insulin production, induce enzymatic and non-enzymatic antioxidant activities in model animals with diabetic symptoms, and diet with high fat or sucrose [326,327]. Triterpenoids can be applied to mitigate obesity and hyperlipidemia. These compounds lead to the destruction of lipids in adipocytes, inhibition of preadipocyte differentiation, and reduction of body fat content [251,321,328,329].
In addition, oleanolic acid has bone anti-resorption activity in aged female rats and can be applied in osteoporosis prevention [330]. Due to anti-inflammatory activity, triterpenoids, such as maslinic acid, can prevent related diseases, particularly arthritis [294,295]. Squalene and ginsenosides can be used for the protection against skin photoaging [242,273]. Their capacity was demonstrated in clinical trials and in vitro studies. Lupeol improved the selenite-induced cataract in rats and decreased the oxidative stress in eye tissues [300].
3.4.9. Additional Activities
Triterpenes and triterpenoids have antiviral, antibacterial, antifungal, antiparasitic action [230,331,332,333,334,335,336,337]. Furthermore, triterpenoids can be used as immunomodulator agents [246].
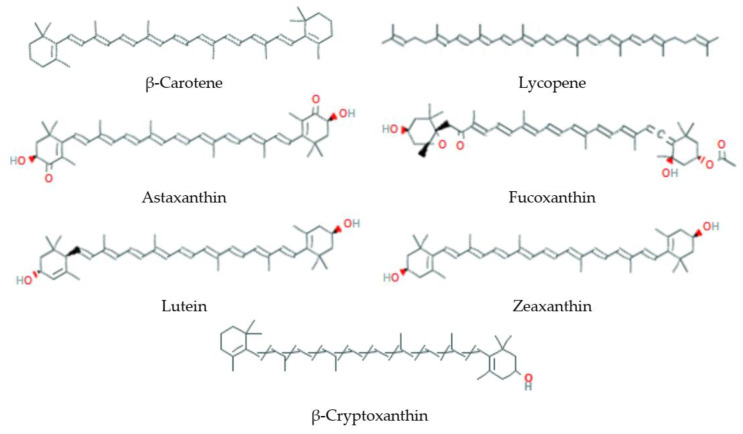
3.5. Tetraterpenes or Carotenoids
Carotenoids (Figure 5) are pigments that contain in their structure the C40 hydrocarbon backbone [338]. Depending on the presence of oxygen in the structure, carotenoids are divided into (i) unoxygenated carotenoids (β-carotene, α-carotene, lycopene) and (ii) oxygenated xanthophylls (astaxanthin, fucoxanthin, lutein). Furthermore, they can be divided into two groups depending on their ability to possess provitamin A activity. In nature, carotenoids play two important roles in the photosynthesis process by harvesting light and protecting an organism from excessive light exposure, which leads to ROS formation [339].
Figure 5.
Some carotenoids that increase lifespan and delay age-related diseases.
3.5.1. Natural Sources
Around 600 different carotenoids are known, 40 of which people consume with food [340]. In European countries, the most abundant carotenoids in food are lutein and β-carotene [341]. Lutein can be found in different green leafy vegetables like kale, spinach, asparagus, parsley, broccoli, zucchini, and lettuce [342]. β-Carotene, a precursor of Vitamin A, is the most abundant carotenoid in nature and contains in a variety of fruits and vegetables like spinach, pepper, lettuce, kale, and apricot [342].
3.5.2. Lifespan Extension on Different Models
Even though the influence of different carotenoid-containing extracts on lifespan parameters of model organisms is excessively studied [343,344,345], the data with pure carotenoids are limited. It was shown that β-carotene increased the median lifespan of D. melanogaster females up to 37% and the age of 90% mortality up to 11% [346]. In D. melanogaster males the effects of β-carotene were not stable. In another study, synthetic all-trans-carotene (100 μM) had no effects on the lifespan parameters of both Drosophila males and females [344]. This compound did not have statistically significant effects on the lifespan parameter of C. elegans at the concentrartions of 0.3–10 μM [346]. The statistically significant effects were also absent in experiments with mice [347].
Carotene lycopene had no effects on C. elegans lifespan parameters [348]. At the same time, pure synthesized lycopene at a dose of 7.5 ppm increased the mean lifespan of both D. melanogaster males and females by 4.8–8.1% as well as the maximum lifespan of females by 4.8% [345]. The positive effects were sex-dependent and more pronounced in females. They were accompanied by an increased SOD level.
Xanthophyll fucoxanthin in the concentration of 5 μM enhanced the median and maximum lifespan parameters in wild-type C. elegans species by 14% and 24%, respectively [346]. The addition of fucoxanthin also had positive effects on the longevity of D. melanogaster. In one study, it was shown that the compound increased the median lifespan of D. melanogaster females up to 49% and the age of 90% mortality up to 27% [346]. In D. melanogaster males the effects of fucoxanthin were not stable. In another study, the positive effects of 1 μM xanthophyll on D. melanogaster lifespan parameters were observed in both sexes [349].
Another xanthophyll astaxanthin (30 μM) increased the chronological lifespan of wild-type S. cerevisiae strains [350]. The positive effects were also observed on antioxidant (sod1Δ, sod2Δ, tsa1Δ, cta1Δ) and anti-apoptotic (pep4Δ, fis1Δ) deficient S. cerevisiae strains. Astaxanthin in concentrations 0.1–1 mM also increased the mean lifespan of wild-type C. elegans up to 31% [351]. It was shown that the positive effects on C. elegans lifespan parameters were observed only if the compound was fed to animals throughout their entire life or only at adult stage [348]. No effects were found, when astaxanthin has been fed to worms only from the larval L1 stage until adulthood. Astaxanthin increased longevity of the mealworm beetle Tenebrio molitor [352].
The addition of lutein in amounts of 0.03 and 0.1 mg to ml diet increased the mean and maximum lifespan of D. melanogaster males up to 11.4% and 16%, respectively [353]. The positive effects were accompanied by the upregulation of expression levels of a few antioxidant enzyme genes (Sod1, Sod2, Cat). The effects on flies’ females were not studied. Zeaxanthin had no effects on C. elegans lifespan parameters [348].
3.5.3. Effects on Stress-Resistance
β-Carotene increased resistance of Drosophila females to oxidative stress but had negative effects on the resistance of Drosophila males to heat shock [346]. The effects of fucoxanthin on different stress conditions were also controversial [346,349].
Lutein (0.1 mg/mL diet) enhanced resistance of D. melanogaster males to oxidative stress generated by the addition of paraquat and H2O2 [353].
A 2-h pretreatment with astaxanthin (30 μM) decreased sensitivity of S. cerevisiae antioxidant deficient strains (sod1Δ, sod2Δ, tsa1Δ, cta1Δ, ctt1Δ) to oxidative stress induced by H2O2 [350]. The positive effect was accompanied by the decreased levels of ROS and lipid peroxidation as well as an increased level of glutathione (GSH) in all studied antioxidant deficient strains. In strains tsa1Δ, cta1Δ, and ctt1Δ increased SOD enzyme activity was observed. Furthermore, astaxanthin decreased apoptotic cell death under acetic acid and H2O2 treatments in mutant strains, which exhibit increased apoptosis due to the lack of Fis1p or Pep4p proteins. Astaxanthin reduced growth defects in both antioxidant and anti-apoptotic deficient strains induced by H2O2 or acetic acid treatment. Astaxanthin decreased resistance of the mealworm beetle to a bacterial infection induced by using its two known entomopathogenic bacterial pathogens Bacillus cereus and Bacillus thuringiensis [352].
3.5.4. Effects on Aging Biomarkers
Fucoxanthin exerts protective effects in human fibroblasts cellular senescence [354]. The addition of fucoxanthin to D. melanogaster decreased the proportion of the male flies with increased intestinal permeability (Smurfs), a known-biomarker of aging in a number of species [355], and had no effects on this parameter in females [349].
Lutein (0.03–0.1 mg/mL) reduced the level of malondialdehyde, an end-product of lipid peroxidation, and a potential biomarker of aging [356], in D. melanogaster males [353]. The same effects were observed after the addition of pure synthesized lycopene [345].
One of the aging-related behavioral changes observed in organisms is a breakdown of sleep:wake cycles [357,358]. Fucoxanthin decreased sleep disturbance in old Drosophila females at night but decreased this parameter in younger flies [349]. In contrast to the previously mentioned positive effects of carotenoids on aging biomarkers, astaxanthin caused an immune depressive effect in the mealworm beetle [352].
3.5.5. Toxicity and Side Effects
Carotenoids are well-known antioxidants, however, under certain conditions, they may display pro-oxidant properties too. The key factors in explaining the dual role of carotenoids are oxygen pressure and carotenoids’ concentration [359]. Experimental data show that β-carotene can increase the possibility of cancer development in smoking men [360]. The analysis of randomized trials showed that β-carotene could also enhance mortality both in healthy people and people with various diseases [361]. Excessive consumption of β-carotene can lead to carotenosis, e.g., to skin orange discoloration [362].
However, in general, carotenoids are regarded as non-toxic compounds. They are commonly used as food colorants [362,363]. Nevertheless, the questions regarding which compounds can be used in which food, the compound’s levels, their sources, and purity are subject to legislation regulation in different countries [363].
3.5.6. Life Quality Effects
Both β-carotene (0.3–1 μM) and fucoxanthin (0.3–1 μM) increased locomotor activity in males but had no effects on this parameter in females [346,349]. Synthetic all-trans-carotene (100 μM) improved mobility of eight weeks of age females in a negative geotaxis test [344].
In D. melanogaster females, no statistically significant effects of β-carotene (0.3–1 μM) on the fecundity parameters like egg-laying and pupae development from eggs were observed [346]. Pure synthesized lycopene increased the reproductive activity of D. melanogaster females by enhancing the amount of F1 generation and the sexual capacity (mating rate and mating duration time) [345]. The effects of fucoxanthin in tested 0.3–1 μM concentrations on D. melanogaster females’ egg-laying were controversial. The compound stimulated egg-laying by 30–48% in young female flies on the first experimental week, but in most cases decreased this parameter by 2–77% on other weeks. Fucoxanthin enhanced the number of pupae development from eggs in the concentration of 1 μM [346,349]. Supplementation with astaxanthin increased larval development time of the mealworm beetle Tenebrio molitor [352].
3.5.7. Suppression of Pro-Aging or Activation of Anti-Aging Molecular Targets or Pathways
Carotenoids possess direct scavenging properties due to the presence of conjugated double bonds in their structure [364]. The compounds are also able to modify activities of different anti-aging molecular targets or pathways. For example, it was found that carotenoids can induce antioxidant defense mechanisms in the cell by activating the transcription factor NRF2 [365]. Lycopene and fucoxanthin were reported to increase the translocation of NRF2 to the nuclei and induce expression of phase II enzymes via activation of the ARE transcription system [366,367]. The effects of other tested carotenoids β-carotene, astaxanthin, and phytoene were less expressed [366].
Carotenoids inactivate NF-κB, which triggers the transcription of inflammatory cytokines [365]. For example, β-carotene suppressed activation of NF-κB pathway in LPS-pretreated RAW264.7 cells and peritoneal macrophages by decreasing translocation of NF-κB p65 subunit to the nuclei and phosphorylation of an NF-κB inhibitor protein IκB, which led to a suppression of IκB degradation. The β-carotene treatment also reduced the expression of inflammatory molecules [368,369].
Carotenoids modulate the MAPK activity. For example, β-carotene may activate or inactivate JNK and p38 depending on the concentration used [370]. Fucoxanthin activated JNK in cancer cells, which resulted in cell cycle arrest on G1-phase [371,372].
The fucoxanthin induced cell cycle arrest was also associated with increased GADD45 expression [371,372]. GADD45 overexpression led to increased D. melanogaster lifespan probably due to the resulting increase in the efficiency of detection and repair of spontaneous DNA damage [373].
The effects of carotenoids on the insulin/IGF-1 signaling pathway, a well-known longevity regulator, are controversial. For example, Yazaki et al. proposed that the insulin/IGF-1 signaling pathway might be one of the mechanisms of astaxanthin positive action on C. elegans lifespan [351]. They observed that in the age-1 mutant the positive effects of astaxanthin were less expressed than in wild-type worms. In daf-16 null mutants, no significant results on lifespan were found. At the same time, astaxanthin increased nuclear localization of the DAF-16 transcription factor as well as increased expression levels of DAF-16 target genes. The effects of astaxanthin were also associated with decreased ROS production in mitochondria. However, in another study, it was shown that the effects of astaxanthin on C. elegans were independent of the insulin/IGF-1 signaling pathway as the positive effects of the compound were also present in experiments with daf-2(e1370) and daf-16(mu86) strains [348]. Experiments with eat-2 (ad1116) mutants revealed that they were also independent of the dietary restriction mechanism. The main mechanism of astaxanthin proposed by the authors is that the compound affects biogenesis and activity of the mitochondrial respiratory chain complex III, which possibly results in decreased mtROS production due to the decreased electron leakage [348].
3.5.8. Effects on Age-Related Diseases
The possible role of carotenoids in inhibiting the development of various types of cancer is being actively discussed [374,375]. The anticancer activity was noted for several carotenoids (β-carotene, α-carotene, lycopene, lutein, zeaxanthin, β-cryptoxanthin, fucoxanthin, canthaxanthin, and astaxanthin). However, the negative effects were also reported [376].
There is evidence of neuroprotective effects of carotenoids. For example, it has been shown that carotenoid fucoxanthin decreased the pro-inflammatory response of microglia, reducing the production of neurotoxic mediators such as nitric oxide (NO) and pro-inflammatory cytokines (TNF-α, IL-6, IL-1β) [377]. The effects were associated with inhibition of the activation of mitogen-activated protein kinases (ERK, JNK, p38). In addition, fucoxanthin modulates the response of microglia to oxidative stress, activating the expression of antioxidant defense genes and reducing the level of free radicals in the cell.
The anti-obesity effects were reported for β-carotene, astaxanthin, β-cryptoxanthin, fucoxanthin, zeaxanthin, and lycopene in experiments using mice [378]. Carotenoids were able to inhibit adipogenesis and activate adipocyte browning and lipid catabolism. The direct influence of carotenoids on the brain function as a possible mechanism of carotenoids’ anti-obesity properties is being discussed. The available studies on humans, even though mostly dealing with mixtures of different carotenoids, confirm the beneficial effects of carotenoids on obesity.
Carotenoids prevent age-related cognitive decline. A few studies showed that the higher serum levels of lycopene, lutein, zeaxanthin, and β-carotene are associated with better cognitive performance in old people [379,380,381,382]. There is also evidence of beneficial effects of β-carotene in people with Alzheimer’s and Parkinson’s disease [383,384].
Carotenoids have potentially positive effects on cardiovascular health. Epidemiological studies show that there is a correlation between risk of cardiovascular diseases and atherosclerosis and concentration of carotenoids in dietary intake, plasma or serum, adipose tissue [385]. In clinical trials, the positive effects are also observed. For example, the supplementation with lycopene in form of cooked tomatoes, tomato extracts, or tomato juice lowered blood pressure, decreased concentration of lipid peroxidation products, reduced levels of total and LDL cholesterol, increased levels of antioxidant enzymes and resistance of LDL cholesterol to oxidation [385,386]. It also caused a decrease in levels of C-reactive protein and adhesion molecules (VCAM-1 and ICAM).
The positive correlation between carotenoids’ dietary intake or serum concentrations of carotenoids like β-carotene, α-carotene, β-cryptoxanthin and risk of type 2 diabetes was revealed in a number of studies [387,388,389,390,391]. However, contradictory data are also available [387,388].
Lutein and zeaxanthin are the most studied carotenoids in relation to eye health as they both are abundant in the macula, where they function as filters of blue light and provide protection against light-induced damage [392]. The potential beneficial effects of lutein and zeaxanthin on several eye diseases including age-related macular degeneration, cataracts, retinitis pigmentosa, retinopathy of prematurity, and diabetic retinopathy are being discussed [393,394,395]. The carotenoids supplementation had been reported to improve visual acuity, contrast sensitivity, and macular pigment optical density levels. It is reported that carotenoids like β-cryptoxanthin and lycopene have beneficial effects on bone health by activating osteoclasts and/or depressing the work of osteoblasts [396,397].
3.6. Polyterpenes
Polyterpenes are composed by many isoprenyl groups in the side chain ((C5H8)n, where n > 8). Some hardwoods produce polyterpenes—rubber and gutta (gutta-percha). Natural rubber consists of polyisoprene, in which double bonds are in the cis-conformation. Some plants produce polyisoprene, in which the double bond is in trans-conformation, this is gutta-percha. This class of terpenoids does not have geroprotectors properties, but these compounds are widely used materials because they have low toxicity [398,399,400,401].
3.7. Norisoprenoids
Norisoprenoids have 13 carbon atoms and are found in Vitis vinifera grape leaves. These include 3-oxo-α-ionol presented in the Muscat of Alexandria variety and derivatives of 7,8-dihydroionones, such as megastigman-3,9-diol and 3-oxo-7,8-dihydro-α-ionol, found in the Shiraz variety, also β-damascenone, 3-hydroxy-β-damascenone, 1,1,6-trimethyl-1,2-dihydronaphthalene in the Merlot grape [402,403,404,405,406]. This class of terpenoids does not have a geroprotectors property as well. Norisoprenoids add flavor to wine. Nowadays, clinical uses are being developed. For example, a method of sunburn prevention by β-damascenone in Skh-1 mice was studied [405].
3.8. Sesterterpenes
These terpenes, having 25 carbon atoms and five units of isoprene, are rarely found, and insufficiently explored. An example of sesterterpenes is geranylpharnesol. Geranylpharnesol has been shown to induce the differentiation of mouse myeloid leukemia M1 cells into macrophage-like cells. It was also found that geranylpharnesol can inhibit DNA and RNA synthesis by specifically inhibiting rRNA synthesis [407]. Studies on the tumor specificity of geranylpharnesol have shown that it has cytotoxicity against certain cultured human tumor cells [408].
3.9. Sesquarterpenes
Sesquarterpenes are composed of seven isoprene units and have the molecular formula C35H56. They are usually synthesized only in microorganisms. Sesquiterpenes are insufficiently explored. Examples of sesquarterpenoids are ferrugicadiol and tetraprenylcurcumen. It has been shown that ferrugicadiol from Calocedrus macrolepis var. Formosana is cytotoxic to human epidermoid carcinoma cells (KB cells) [409].
4. Possible Prospect of Terpene using as Anti-Aging Drugs
Due to their numbers and diversity, terpenoids offer much potential in an array of industrial and medicinal applications among all the secondary metabolites of plants [410]. Although most of the terpenes have not yet been fully investigated, they are known to have a wide range of medicinal applications among which are antiplasmodial, antiviral, anticancer, antidiabetic, and antidepressant activities [3,12,411]. Here we showed that most classes of terpenoids have representatives with the explicit geroprotective properties as antioxidants and inducers of the expression of cytoprotective mechanisms. The greatest compliance with the primary and secondary criteria of geroprotectors we found for the following terpenoids: limonene (monoterpene), β-caryophyllene (sesquiterpene), dehydroabietic acid, carnosol, carnosic acid (diterpenes), squalene, ursolic acid, oleanolic acid, maslinic acid, 18α-glycyrrhetinic acid, asiaticoside, ginsenosides (triterpenes), β-carotene, lycopene, fucoxanthin, astaxanthin, lutein, zeaxanthin, β-cryptoxanthin (tetraterpenes/carotenoids) (Table 1). In addition, terpenoids with known medicinal applications potentially could be repurposed to combat aging and prevent age-related conditions in humans [412].
Table 1.
Geroprotective potential of some terpenes and terpenoids.
| Compound (Molecular Formula) | Compliance with Criteria of Geroprotectors * | References | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Primary Criteria | Secondary Criteria | |||||||||
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | ||
| Monoterpenes (C10H16) | ||||||||||
| Limonene | + | + | - | - | + | + | + | + | + | [22,23,28,35,37,38,39,40,41,44,46,47,48,413,414,415,416,417] |
| Sesquiterpenes (C15H24) | ||||||||||
| β-Caryophyllene | + | + | + | + | + | + | 0 | + | + | [58,67,79,85,86,93,96,105,147,150,164,418,419,420,421,422,423,424] |
| Diterpenes ((C10H16)2) | ||||||||||
| Dehydroabietic acid | + | + | - | - | - | + | 0 | + | 0 | [172,190,191,192,193,207,208,425,426,427] |
| Carnosol | + | + | + | + | + | + | 0 | + | + | [176,181,185,186,199,209,428,429,430,431,432] |
| Carnosic acid | + | + | + | + | + | + | 0 | + | + | [177,185,194,195,197,209,429,433,434,435,436,437] |
| Triterpenes ((C10H16)3) | ||||||||||
| Squalene | 0 | + | 0 | + | 0 | + | 0 | + | 0 | [236,266,273,308,309,310,314,320,321,324,326] |
| –Ursolic acid | + | + | + | - | + | + | + | + | + | [231,250,251,265,281,297,298,302,438,439,440,441,442,443] |
| Oleanolic acid | + | 0 | + | - | + | + | 0 | + | + | [231,254,302,322,323,330,444,445,446,447,448] |
| Maslinic acid | 0 | + | + | + | + | + | 0 | + | 0 | [231,270,294,295,325,449] |
| 18α-Glycyrrhetinic acid | + | 0 | - | - | + | + | 0 | + | 0 | [231,258,439,450,451,452,453,454,455,456] |
| Asiaticoside | 0 | + | + | 0 | + | + | 0 | + | 0 | [231,301,302,457,458,459,460,461,462,463,464,465] |
| Ginsenosides | + | + | + | - | + | + | 0 | + | + | [257,262,268,466,467,468,469,470,471,472,473,474,475,476,477,478] |
| Tetraterpenes/carotenoids ((C10H16)4) | ||||||||||
| β-Carotene | - | + | + | - | + | + | - | + | - | [344,346,347,360,361,362,365,370,376,378,383,384,479,480,481,482,483,484] |
| Lycopene | + | + | + | + | + | + | - | + | 0 | [345,348,362,365,366,376,378,479,484,485,486,487,488,489] |
| Fucoxanthin | + | + | + | + | + | + | + | + | - | [346,349,362,365,367,371,372,377,378,490] |
| Astaxanthin | + | + | + | - | + | + | + | + | + | [326,350,351,352,362,365,376,378,491,492,493,494,495] |
| Lutein | + | + | + | + | + | + | 0 | + | + | [353,362,365,376,393,394,395,481,484,496,497,498,499,500] |
| Zeaxanthin | - | + | + | + | + | + | 0 | + | 0 | [348,362,365,376,378,393,394,395,481,484,498,499,500,501] |
| β-Cryptoxanthin | 0 | + | + | 0 | + | + | 0 | + | 0 | [362,376,378,479,483,484,499,502,503,504,505,506] |
* Primary criteria: 1. The life extension in wild-type animal models, 2. Improvement of aging biomarkers, 3. Low toxicity, 4. Minimal side effects, 5. Improvement of the quality of life; Secondary criteria: 6. Evolutionarily conserved mechanisms, 7. Reproducibility on different models, 8. Prevention of age-associated diseases, 9. Increased stress-resistance. + Compliance with criteria; - Not compliance with criteria; 0 Not investigated.
Along with their structural diversity and a broad spectrum of physiological activities, terpenes also allow for flexibility in the route of administration [411]. Possible routes of administration for terpenoids may include, but are not limited to, cutaneous (administration to the skin as anti-aging cream components [507]), oral (consumption as pharmacological substances, nutritional supplements, and bioactive food components [411]), and respiratory (inhaling the essential oils of aromatic plants or natural forest atmosphere [3,12,508]).
The experimental validation of the anti-aging activity of terpenes in humans is one of the major challenges to revealing new geroprotectors. According to a recent review, three major approaches are applicable to test anti-aging interventions in humans and accelerate their widespread use to improve human aging [509]. These approaches include testing of longevity interventions in the context of age-related disease or process indications; investigating the preventing effect of anti-aging interventions on multiple chronic diseases simultaneously; using non-invasive or minimally invasive strategies to measure biological age [509]. All these promising approaches can be applied to prove the activity of terpenoids as potential geroprotectors.
5. Conclusions
The unprecedented increase in the average human lifespan is one of the greatest accomplishments of the past century [510,511]. Life expectancy will continue to increase for the foreseeable future, which in combination with a rapid decline in human fertility will lead to population aging [510,512]. At the same time, healthspan (healthy, disease-free lifespan) has not increased as much as lifespan [511,513]. Since aging is the predominant risk factor for most chronic diseases, fragility, and disability in the elderly, population aging has become one of the main global challenges [511,514,515]. The development and implementation of effective geroprotective interventions can contribute to healthspan increasing and prevention or amelioration of age-related pathologies [6,509].
In this review, we showed that most classes of terpenoids have representatives with the potential geroprotective properties (Table 1). Thus, terpenoids are underestimated in their potential activities in terms of criteria of geroprotectors. We suggest that these compounds have a great prospect to become a new class of anti-aging drugs.
Author Contributions
Conceptualization, A.M., E.P. (Ekaterina Proshkina) and S.P.; writing—original draft preparation, E.P. (Ekaterina Proshkina); S.P.; T.B.; E.L.; L.K., E.P. (Elena Platonova); M.S. and A.M.; writing—review and editing, A.M., F.M. and M.S.; supervision, A.M. and F.M. All authors have read and agreed to the published version of the manuscript.
Funding
This research received funding within the framework of the state task on the theme “Development of geroprotective and radioprotective agents”, state registration № AAAA-A19-119021590022-2.
Conflicts of Interest
The authors declare that they have no conflict of interest except Fanya Maganova, who is the CEO of pharmacological company Initium-Pharm (Moscow, Russia). The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the result.
References
- 1.Yazaki K., Arimura G.I., Ohnishi T. ‘Hidden’ Terpenoids in Plants: Their Biosynthesis, Localization and Ecological Roles. Plant Cell Physiol. 2017;58:1615–1621. doi: 10.1093/pcp/pcx123. [DOI] [PubMed] [Google Scholar]
- 2.Martin-Smith M., Khatoon T. Biological Activity of the Terpenoids and Their Derivatives. In: Gillette J.R., Heinzelman R.V., Szmuszkovicz J., Leemann H.G., Stich K., Thomas M., Martin-Smith M., Khatoon T., Kunz W., Jucker E., editors. Progress in Drug Research/Fortschritte der Arzneimittelforschung/Progrès des recherches pharmaceutiques. Birkhäuser Basel; Basel, Switzerland: 1963. pp. 279–346. [DOI] [PubMed] [Google Scholar]
- 3.Brahmkshatriya P.P., Brahmkshatriya P.S. Terpenes: Chemistry, Biological Role, and Therapeutic Applications. In: Ramawat K.G., Mérillon J.-M., editors. Natural Products: Phytochemistry, Botany and Metabolism of Alkaloids, Phenolics and Terpenes. Springer; Berlin/Heidelberg, Germany: 2013. pp. 2665–2691. [DOI] [Google Scholar]
- 4.Jaeger R., Cuny E. Terpenoids with Special Pharmacological Significance: A Review. Nat. Prod. Commun. 2016;11:1934578X1601100946. doi: 10.1177/1934578X1601100946. [DOI] [PubMed] [Google Scholar]
- 5.Kudryavtseva A., Krasnov G., Lipatova A., Alekseev B., Maganova F., Shaposhnikov M., Fedorova M., Snezhkina A., Moskalev A. Effects of Abies sibirica terpenes on cancer- and aging-associated pathways in human cells. Oncotarget. 2016;7:83744–83754. doi: 10.18632/oncotarget.13467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Moskalev A., Chernyagina E., Tsvetkov V., Fedintsev A., Shaposhnikov M., Krut’ko V., Zhavoronkov A., Kennedy B.K. Developing criteria for evaluation of geroprotectors as a key stage toward translation to the clinic. Aging Cell. 2016;15:407–415. doi: 10.1111/acel.12463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jiang Z., Kempinski C., Chappell J. Extraction and Analysis of Terpenes/Terpenoids. Curr. Protoc. Plant Biol. 2016;1:345–358. doi: 10.1002/cppb.20024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Piñeiro Z., Palma M., Barroso C.G. Determination of terpenoids in wines by solid phase extraction and gas chromatography. Anal. Chim. Acta. 2004;513:209–214. doi: 10.1016/j.aca.2003.12.044. [DOI] [Google Scholar]
- 9.Harman-Ware A.E., Sykes R., Peter G.F., Davis M. Determination of Terpenoid Content in Pine by Organic Solvent Extraction and Fast-GC Analysis. Front. Energy Res. 2016:4. doi: 10.3389/fenrg.2016.00002. [DOI] [Google Scholar]
- 10.Nguyen T.D., Riordan-Short S., Dang T.T., O’Brien R., Noestheden M. Quantitation of Select Terpenes/Terpenoids and Nicotine Using Gas Chromatography-Mass Spectrometry with High-Temperature Headspace Sampling. ACS Omega. 2020;5:5565–5573. doi: 10.1021/acsomega.0c00384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Azmir J., Zaidul I.S.M., Rahman M.M., Sharif K.M., Mohamed A., Sahena F., Jahurul M.H.A., Ghafoor K., Norulaini N.A.N., Omar A.K.M. Techniques for extraction of bioactive compounds from plant materials: A review. J. Food Eng. 2013;117:426–436. doi: 10.1016/j.jfoodeng.2013.01.014. [DOI] [Google Scholar]
- 12.Marriott P.J., Shellie R., Cornwell C. Gas chromatographic technologies for the analysis of essential oils. J. Chromatogr. 2001;936:1–22. doi: 10.1016/S0021-9673(01)01314-0. [DOI] [PubMed] [Google Scholar]
- 13.Pawliszyn J. 1–Solid-Phase Microextraction in Perspective. In: Pawliszyn J., editor. Handbook of Solid Phase Microextraction. Elsevier; Oxford, AZ, USA: 2012. pp. 1–12. [DOI] [Google Scholar]
- 14.Bajer T., Ligor M., Ligor T., Buszewski B. Design of the extraction process for terpenes and other volatiles from allspice by solid-phase microextraction and hydrodistillation. J. Sep. Sci. 2016;39:769–775. doi: 10.1002/jssc.201500605. [DOI] [PubMed] [Google Scholar]
- 15.Chemat F., Abert-Vian M., Fernandez X. Microwave-Assisted Extraction of Essential Oils and Aromas. In: Chemat F., Cravotto G., editors. Microwave-Assisted Extraction for Bioactive Compounds: Theory and Practice. Springer; Boston, MA, USA: 2013. pp. 53–68. [DOI] [Google Scholar]
- 16.Martins M.A.R., Domańska U., Schröder B., Coutinho J.A.P., Pinho S.P. Selection of Ionic Liquids to be Used as Separation Agents for Terpenes and Terpenoids. ACS Sustain. Chem. Eng. 2015;4:548–556. doi: 10.1021/acssuschemeng.5b01357. [DOI] [Google Scholar]
- 17.Sun Y., Li W., Fitzloff J.F., van Breemen R.B. Liquid chromatography/electrospray tandem mass spectrometry of terpenoid lactones in Ginkgo biloba. J. Mass Spectrom. 2005;40:373–379. doi: 10.1002/jms.795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tran T.A., Ho M.T., Song Y.W., Cho M., Cho S.K. Camphor Induces Proliferative and Anti-senescence Activities in Human Primary Dermal Fibroblasts and Inhibits UV-Induced Wrinkle Formation in Mouse Skin. Phytother. Res. 2015;29:1917–1925. doi: 10.1002/ptr.5484. [DOI] [PubMed] [Google Scholar]
- 19.Ghaffari T., Kafil H.S., Asnaashari S., Farajnia S., Delazar A., Baek S.C., Hamishehkar H., Kim K.H. Chemical Composition and Antimicrobial Activity of Essential Oils from the Aerial Parts of Pinus eldarica Grown in Northwestern Iran. Molecules. 2019;24:3203. doi: 10.3390/molecules24173203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lin L.Y., Chuang C.H., Chen H.C., Yang K.M. Lime (Citrus aurantifolia (Christm.) Swingle) Essential Oils: Volatile Compounds, Antioxidant Capacity, and Hypolipidemic Effect. Foods. 2019;8:398. doi: 10.3390/foods8090398. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Md Othman S.N.A., Hassan M.A., Nahar L., Basar N., Jamil S., Sarker S.D. Essential Oils from the Malaysian Citrus (Rutaceae) Medicinal Plants. Medicines. 2016;3:13. doi: 10.3390/medicines3020013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Papanastasiou S.A., Bali E.-M.D., Ioannou C.S., Papachristos D.P., Zarpas K.D., Papadopoulos N.T. Toxic and hormetic-like effects of three components of citrus essential oils on adult Mediterranean fruit flies (Ceratitis capitata) PLoS ONE. 2017;12:e0177837. doi: 10.1371/journal.pone.0177837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fernández-Bedmar Z., Anter J., de La Cruz-Ares S., Muñoz-Serrano A., Alonso-Moraga A., Pérez-Guisado J. Role of citrus juices and distinctive components in the modulation of degenerative processes: Genotoxicity, antigenotoxicity, cytotoxicity, and longevity in Drosophila. J. Toxicol. Environ. Health Part A. 2011;74:1052–1066. doi: 10.1080/15287394.2011.582306. [DOI] [PubMed] [Google Scholar]
- 24.Gerofotis C.D., Ioannou C.S., Nakas C.T., Papadopoulos N.T. The odor of a plant metabolite affects life history traits in dietary restricted adult olive flies. Sci. Rep. 2016;6:28540. doi: 10.1038/srep28540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mattson M.P., Son T.G., Camandola S. Viewpoint: Mechanisms of action and therapeutic potential of neurohormetic phytochemicals. Dose Response. 2007;5:174–186. doi: 10.2203/dose-response.07-004.Mattson. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Moayedi Y., Greenberg S.A., Jenkins B.A., Marshall K.L., Dimitrov L.V., Nelson A.M., Owens D.M., Lumpkin E.A. Camphor white oil induces tumor regression through cytotoxic T cell-dependent mechanisms. Mol. Carcinog. 2019;58:722–734. doi: 10.1002/mc.22965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nikolić B., Vasilijević B., Mitić-Ćulafić D., Vuković-Gačić B., Knežević-Vukćević J. Comparative study of genotoxic, antigenotoxic and cytotoxic activities of monoterpenes camphor, eucalyptol and thujone in bacteria and mammalian cells. Chem. Biol. Interact. 2015;242:263–271. doi: 10.1016/j.cbi.2015.10.012. [DOI] [PubMed] [Google Scholar]
- 28.Karthikeyan R., Kanimozhi G., Madahavan N.R., Agilan B., Ganesan M., Prasad N.R., Rathinaraj P. Alpha-pinene attenuates UVA-induced photoaging through inhibition of matrix metalloproteinases expression in mouse skin. Life Sci. 2019;217:110–118. doi: 10.1016/j.lfs.2018.12.003. [DOI] [PubMed] [Google Scholar]
- 29.Wu Z., Tan B., Liu Y., Dunn J., Martorell Guerola P., Tortajada M., Cao Z., Ji P. Chemical Composition and Antioxidant Properties of Essential Oils from Peppermint, Native Spearmint and Scotch Spearmint. Molecules. 2019;24:2825. doi: 10.3390/molecules24152825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Foti M.C., Ingold K.U. Mechanism of inhibition of lipid peroxidation by gamma-terpinene, an unusual and potentially useful hydrocarbon antioxidant. J. Agric. Food Chem. 2003;51:2758–2765. doi: 10.1021/jf020993f. [DOI] [PubMed] [Google Scholar]
- 31.Pratheeshkumar P., Raphael T.J., Kuttan G. Protective role of perillic acid against radiation-induced oxidative stress, cytokine profile, DNA damage, and intestinal toxicity in mice. J. Environ. Pathol. Toxicol. Oncol. 2010;29:199–212. doi: 10.1615/JEnvironPatholToxicolOncol.v29.i3.40. [DOI] [PubMed] [Google Scholar]
- 32.Singh B.K., Tripathi M., Chaudhari B.P., Pandey P.K., Kakkar P. Natural terpenes prevent mitochondrial dysfunction, oxidative stress and release of apoptotic proteins during nimesulide-hepatotoxicity in rats. PLoS ONE. 2012;7:e34200. doi: 10.1371/journal.pone.0034200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mohamad R.H., El-Bastawesy A.M., Abdel-Monem M.G., Noor A.M., Al-Mehdar H.A., Sharawy S.M., El-Merzabani M.M. Antioxidant and anticarcinogenic effects of methanolic extract and volatile oil of fennel seeds (Foeniculum vulgare) J. Med. Food. 2011;14:986–1001. doi: 10.1089/jmf.2008.0255. [DOI] [PubMed] [Google Scholar]
- 34.Gomes-Carneiro M.R., Felzenszwalb I., Paumgartten F.J. Mutagenicity testing (+/-)-camphor, 1,8-cineole, citral, citronellal, (-)-menthol and terpineol with the Salmonella/microsome assay. Mutat. Res. 1998;416:129–136. doi: 10.1016/S1383-5718(98)00077-1. [DOI] [PubMed] [Google Scholar]
- 35.De Groot A. Limonene Hydroperoxides. Dermatitis. 2019;30:331–335. doi: 10.1097/DER.0000000000000465. [DOI] [PubMed] [Google Scholar]
- 36.Kopečná M., Macháček M., Nováčková A., Paraskevopoulos G., Roh J., Vávrová K. Esters of terpene alcohols as highly potent, reversible, and low toxic skin penetration enhancers. Sci. Rep. 2019;9:14617. doi: 10.1038/s41598-019-51226-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Bisson J.F., Menut C., d’Alessio P. Anti-inflammatory senescence actives 5203-L molecule to promote healthy aging and prolongation of lifespan. Rejuvenation Res. 2008;11:399–407. doi: 10.1089/rej.2008.0667. [DOI] [PubMed] [Google Scholar]
- 38.D’Alessio P.A., Bisson J.F., Bene M.C. Anti-stress effects of d-limonene and its metabolite perillyl alcohol. Rejuvenation Res. 2014;17:145–149. doi: 10.1089/rej.2013.1515. [DOI] [PubMed] [Google Scholar]
- 39.Ostan R., Bene M.C., Spazzafumo L., Pinto A., Donini L.M., Pryen F., Charrouf Z., Valentini L., Lochs H., Bourdel-Marchasson I., et al. Impact of diet and nutraceutical supplementation on inflammation in elderly people. Results from the RISTOMED study, an open-label randomized control trial. Clin. Nutr. 2016;35:812–818. doi: 10.1016/j.clnu.2015.06.010. [DOI] [PubMed] [Google Scholar]
- 40.Nascimento G.A.d., Souza D.S.d., Lima B.S., Vasconcelos C.M.L.d., Araújo A.A.d.S., Durço A.O., Quintans-Junior L.J., Almeida J.R.G.d.S., Oliveira A.P., Santana-Filho V.J.d., et al. Bradycardic and Antiarrhythmic Effects of the D-Limonene in Rats. Arq. Bras. De Cardiol. 2019;113:925–932. doi: 10.5935/abc.20190173. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Kesbiç O.S., Acar Ü., Yilmaz S., Aydin Ö.D. Effects of bergamot (Citrus bergamia) peel oil-supplemented diets on growth performance, haematology and serum biochemical parameters of Nile tilapia (Oreochromis niloticus) Fish. Physiol. Biochem. 2020;46:103–110. doi: 10.1007/s10695-019-00700-y. [DOI] [PubMed] [Google Scholar]
- 42.Hwang E., Ngo H.T.T., Park B., Seo S.A., Yang J.E., Yi T.H. Myrcene, an Aromatic Volatile Compound, Ameliorates Human Skin Extrinsic Aging via Regulation of MMPs Production. Am. J. Chin. Med. 2017;45:1113–1124. doi: 10.1142/S0192415X17500604. [DOI] [PubMed] [Google Scholar]
- 43.Nevzorova Y.A., Grossmann J., Trautwein C. Anti-tumorigenic and anti-angiogenic effects of natural conifer Abies sibirica terpenoids in vivo and in vitro. Biomed. Pharm. 2017;89:386–395. doi: 10.1016/j.biopha.2017.02.035. [DOI] [PubMed] [Google Scholar]
- 44.Srinivasan S., Muruganathan U. Antidiabetic efficacy of citronellol, a citrus monoterpene by ameliorating the hepatic key enzymes of carbohydrate metabolism in streptozotocin-induced diabetic rats. Chem. Biol. Interact. 2016;250:38–46. doi: 10.1016/j.cbi.2016.02.020. [DOI] [PubMed] [Google Scholar]
- 45.Raphael T.J., Kuttan G. Effect of naturally occurring monoterpenes carvone, limonene and perillic acid in the inhibition of experimental lung metastasis induced by B16F-10 melanoma cells. J. Exp. Clin. Cancer Res. 2003;22:419–424. [PubMed] [Google Scholar]
- 46.Raphael T.J., Kuttan G. Immunomodulatory activity of naturally occurring monoterpenes carvone, limonene, and perillic acid. Immunopharmacol. Immunotoxicol. 2003;25:285–294. doi: 10.1081/IPH-120020476. [DOI] [PubMed] [Google Scholar]
- 47.El-Minshawy A.M., Abdelgaleil S.A.M., Gadelhak G.G., Al-Eryan M.A., Rabab R.A. Effects of monoterpenes on mortality, growth, fecundity, and ovarian development of Bactrocera zonata (Saunders) (Diptera: Tephritidae) Environ. Sci. Pollut. Res. Int. 2018;25:15671–15679. doi: 10.1007/s11356-018-1780-1. [DOI] [PubMed] [Google Scholar]
- 48.Chow H.H., Salazar D., Hakim I.A. Pharmacokinetics of perillic acid in humans after a single dose administration of a citrus preparation rich in d-limonene content. Cancer Epidemiol. Biomark. Prev. 2002;11:1472–1476. [PubMed] [Google Scholar]
- 49.Rufino A.T., Ribeiro M., Sousa C., Judas F., Salgueiro L., Cavaleiro C., Mendes A.F. Evaluation of the anti-inflammatory, anti-catabolic and pro-anabolic effects of E-caryophyllene, myrcene and limonene in a cell model of osteoarthritis. Eur. J. Pharm. 2015;750:141–150. doi: 10.1016/j.ejphar.2015.01.018. [DOI] [PubMed] [Google Scholar]
- 50.Hassan S.B., Gali-Muhtasib H., Goransson H., Larsson R. Alpha terpineol: A potential anticancer agent which acts through suppressing NF-kappaB signalling. Anticancer Res. 2010;30:1911–1919. [PubMed] [Google Scholar]
- 51.Moghimi M., Parvardeh S., Zanjani T.M., Ghafghazi S. Protective effect of alpha-terpineol against impairment of hippocampal synaptic plasticity and spatial memory following transient cerebral ischemia in rats. Iran J. Basic Med. Sci. 2016;19:960–969. [PMC free article] [PubMed] [Google Scholar]
- 52.Wu Z.L., Yin Z.Q., Du Y.H., Feng R.Z., Ye K.C., Wei Q., Hu Y., He L., Liao L., Wang Y. gamma-terpineol inhibits cell growth and induces apoptosis in human liver cancer BEL-7402 cells in vitro. Int. J. Clin. Exp. Pathol. 2014;7:6524–6533. [PMC free article] [PubMed] [Google Scholar]
- 53.Bardon S., Picard K., Martel P. Monoterpenes inhibit cell growth, cell cycle progression, and cyclin D1 gene expression in human breast cancer cell lines. Nutr. Cancer. 1998;32:1–7. doi: 10.1080/01635589809514708. [DOI] [PubMed] [Google Scholar]
- 54.Vallianou I., Peroulis N., Pantazis P., Hadzopoulou-Cladaras M. Camphene, a plant-derived monoterpene, reduces plasma cholesterol and triglycerides in hyperlipidemic rats independently of HMG-CoA reductase activity. PLoS ONE. 2011;6:e20516. doi: 10.1371/journal.pone.0020516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Pereira Fde O., Mendes J.M., Lima I.O., Mota K.S., Oliveira W.A., Lima Ede O. Antifungal activity of geraniol and citronellol, two monoterpenes alcohols, against Trichophyton rubrum involves inhibition of ergosterol biosynthesis. Pharm. Biol. 2015;53:228–234. doi: 10.3109/13880209.2014.913299. [DOI] [PubMed] [Google Scholar]
- 56.Alves-Silva J.M., Zuzarte M., Goncalves M.J., Cruz M.T., Cavaleiro C., Salgueiro L. Unveiling the bioactive potential of the essential oil of a Portuguese endemism, St. Impressa. J. Ethnopharmacol. 2019;244:112120. doi: 10.1016/j.jep.2019.112120. [DOI] [PubMed] [Google Scholar]
- 57.Chaturvedi T., Singh S., Nishad I., Kumar A., Tiwari N., Tandon S., Saikia D., Verma R.S. Chemical composition and antimicrobial activity of the essential oil of senescent leaves of guava (Psidium guajava L.) Nat. Prod. Res. 2019:1–5. doi: 10.1080/14786419.2019.1648462. [DOI] [PubMed] [Google Scholar]
- 58.Pant A., Mishra V., Saikia S.K., Shukla V., Asthana J., Akhoon B.A., Pandey R. Beta-caryophyllene modulates expression of stress response genes and mediates longevity in Caenorhabditis elegans. Exp. Gerontol. 2014;57:81–95. doi: 10.1016/j.exger.2014.05.007. [DOI] [PubMed] [Google Scholar]
- 59.Santhanasabapathy R., Vasudevan S., Anupriya K., Pabitha R., Sudhandiran G. Farnesol quells oxidative stress, reactive gliosis and inflammation during acrylamide-induced neurotoxicity: Behavioral and biochemical evidence. Neuroscience. 2015;308:212–227. doi: 10.1016/j.neuroscience.2015.08.067. [DOI] [PubMed] [Google Scholar]
- 60.Kaur D., Pahwa P., Goel R.K. Protective Effect of Nerolidol Against Pentylenetetrazol-Induced Kindling, Oxidative Stress and Associated Behavioral Comorbidities in Mice. Neurochem. Res. 2016;41:2859–2867. doi: 10.1007/s11064-016-2001-2. [DOI] [PubMed] [Google Scholar]
- 61.Javed H., Azimullah S., Abul Khair S.B., Ojha S., Haque M.E. Neuroprotective effect of nerolidol against neuroinflammation and oxidative stress induced by rotenone. BMC Neurosci. 2016;17:58. doi: 10.1186/s12868-016-0293-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Leite G.O., Ecker A., Seeger R.L., Krum B.N., Lugokenski T.H., Fachinetto R., Sudati J.H., Barbosa N.V., Wagner C. Protective effect of (-)-alpha-bisabolol on rotenone-induced toxicity in Drosophila melanogaster. Can. J. Physiol. Pharm. 2018;96:359–365. doi: 10.1139/cjpp-2017-0207. [DOI] [PubMed] [Google Scholar]
- 63.Meeran M.F.N., Laham F., Al-Taee H., Azimullah S., Ojha S. Protective effects of alpha-bisabolol on altered hemodynamics, lipid peroxidation, and nonenzymatic antioxidants in isoproterenol-induced myocardial infarction: In vivo and in vitro evidences. J. Biochem. Mol. Toxicol. 2018;32:e22200. doi: 10.1002/jbt.22200. [DOI] [PubMed] [Google Scholar]
- 64.Sampaio T.L., Menezes R.R., da Costa M.F., Meneses G.C., Arrieta M.C., Chaves Filho A.J., de Morais G.B., Liborio A.B., Alves R.S., Evangelista J.S., et al. Nephroprotective effects of (-)-alpha-bisabolol against ischemic-reperfusion acute kidney injury. Phytomedicine. 2016;23:1843–1852. doi: 10.1016/j.phymed.2016.11.008. [DOI] [PubMed] [Google Scholar]
- 65.Meng X., Li N., Zhang Y., Fan D., Yang C., Li H., Guo D., Pan S. Beneficial Effect of beta-Elemene Alone and in Combination with Hyperbaric Oxygen in Traumatic Brain Injury by Inflammatory Pathway. Transl. Neurosci. 2018;9:33–37. doi: 10.1515/tnsci-2018-0007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Lim C.S., Han J.S. The antioxidant xanthorrhizol prevents amyloid-beta-induced oxidative modification and inactivation of neprilysin. Biosci. Rep. 2018;38 doi: 10.1042/BSR20171611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Calleja M.A., Vieites J.M., Montero-Melendez T., Torres M.I., Faus M.J., Gil A., Suarez A. The antioxidant effect of beta-caryophyllene protects rat liver from carbon tetrachloride-induced fibrosis by inhibiting hepatic stellate cell activation. Br. J. Nutr. 2013;109:394–401. doi: 10.1017/S0007114512001298. [DOI] [PubMed] [Google Scholar]
- 68.Lnenickova K., Svobodova H., Skalova L., Ambroz M., Novak F., Matouskova P. The impact of sesquiterpenes beta-caryophyllene oxide and trans-nerolidol on xenobiotic-metabolizing enzymes in mice in vivo. Xenobiotica. 2018;48:1089–1097. doi: 10.1080/00498254.2017.1398359. [DOI] [PubMed] [Google Scholar]
- 69.Mao J., Yi M., Wang R., Huang Y., Chen M. Protective Effects of Costunolide Against D-Galactosamine and Lipopolysaccharide-Induced Acute Liver Injury in Mice. Front. Pharm. 2018;9:1469. doi: 10.3389/fphar.2018.01469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Eliza J., Daisy P., Ignacimuthu S. Antioxidant activity of costunolide and eremanthin isolated from Costus speciosus (Koen ex. Retz) Sm. Chem. Biol. Interact. 2010;188:467–472. doi: 10.1016/j.cbi.2010.08.002. [DOI] [PubMed] [Google Scholar]
- 71.Cheong C.U., Yeh C.S., Hsieh Y.W., Lee Y.R., Lin M.Y., Chen C.Y., Lee C.H. Protective Effects of Costunolide against Hydrogen Peroxide-Induced Injury in PC12 Cells. Molecules. 2016;21:898. doi: 10.3390/molecules21070898. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Zheng H., Chen Y., Zhang J., Wang L., Jin Z., Huang H., Man S., Gao W. Evaluation of protective effects of costunolide and dehydrocostuslactone on ethanol-induced gastric ulcer in mice based on multi-pathway regulation. Chem. Biol. Interact. 2016;250:68–77. doi: 10.1016/j.cbi.2016.03.003. [DOI] [PubMed] [Google Scholar]
- 73.Jang Y.J., Back M.J., Fu Z., Lee J.H., Won J.H., Ha H.C., Lee H.K., Jang J.M., Choi J.M., Kim D.K. Protective effect of sesquiterpene lactone parthenolide on LPS-induced acute lung injury. Arch. Pharm. Res. 2016;39:1716–1725. doi: 10.1007/s12272-016-0716-x. [DOI] [PubMed] [Google Scholar]
- 74.Lin X., Zhang S., Huang R., Wei L., Tan S., Liang S., Tian Y., Wu X., Lu Z., Huang Q. Helenalin attenuates alcohol-induced hepatic fibrosis by enhancing ethanol metabolism, inhibiting oxidative stress and suppressing HSC activation. Fitoterapia. 2014;95:203–213. doi: 10.1016/j.fitote.2014.03.020. [DOI] [PubMed] [Google Scholar]
- 75.Wang X., Lan Y.L., Xing J.S., Lan X.Q., Wang L.T., Zhang B. Alantolactone plays neuroprotective roles in traumatic brain injury in rats via anti-inflammatory, anti-oxidative and anti-apoptosis pathways. Am. J. Transl. Res. 2018;10:368–380. [PMC free article] [PubMed] [Google Scholar]
- 76.Seo J.Y., Lim S.S., Kim J., Lee K.W., Kim J.S. Alantolactone and Isoalantolactone Prevent Amyloid β25-35-induced Toxicity in Mouse Cortical Neurons and Scopolamine-induced Cognitive Impairment in Mice. Phytother. Res. 2017;31:801–811. doi: 10.1002/ptr.5804. [DOI] [PubMed] [Google Scholar]
- 77.Liu J., Zhang Z., Gao J., Xie J., Yang L., Hu S. Downregulation effects of beta-elemene on the levels of plasma endotoxin, serum TNF-alpha, and hepatic CD14 expression in rats with liver fibrosis. Front. Med. 2011;5:101–105. doi: 10.1007/s11684-011-0111-4. [DOI] [PubMed] [Google Scholar]
- 78.Oon S.F., Nallappan M., Kassim N.K., Shohaimi S., Sa’ariwijaya M.S., Tee T.T., Cheah Y.H. Hypolipidemic activities of xanthorrhizol purified from centrifugal TLC. Biochem. Biophys. Res. Commun. 2016;478:1403–1408. doi: 10.1016/j.bbrc.2016.08.136. [DOI] [PubMed] [Google Scholar]
- 79.Harb A.A., Bustanji Y.K., Abdalla S.S. Hypocholesterolemic effect of beta-caryophyllene in rats fed cholesterol and fat enriched diet. J. Clin. Biochem. Nutr. 2018;62:230–237. doi: 10.3164/jcbn.17-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Baldissera M.D., Souza C.F., Grando T.H., Stefani L.M., Monteiro S.G. β-caryophyllene reduces atherogenic index and coronary risk index in hypercholesterolemic rats: The involvement of cardiac oxidative damage. Chem. Biol. Interact. 2017;270:9–14. doi: 10.1016/j.cbi.2017.04.008. [DOI] [PubMed] [Google Scholar]
- 81.Tian X., Peng J., Zhong J., Yang M., Pang J., Lou J., Li M., An R., Zhang Q., Xu L., et al. β-Caryophyllene protects in vitro neurovascular unit against oxygen-glucose deprivation and re-oxygenation-induced injury. J. Neurochem. 2016;139:757–768. doi: 10.1111/jnc.13833. [DOI] [PubMed] [Google Scholar]
- 82.Ferreira F.M., Palmeira C.M., Oliveira M.M., Santos D., Simoes A.M., Rocha S.M., Coimbra M.A., Peixoto F. Nerolidol effects on mitochondrial and cellular energetics. Toxicol. In Vitro. 2012;26:189–196. doi: 10.1016/j.tiv.2011.11.009. [DOI] [PubMed] [Google Scholar]
- 83.Piculo F., Guiraldeli Macedo C., de Andrade S.F., Luis Maistro E. In vivo genotoxicity assessment of nerolidol. J. Appl. Toxicol. 2011;31:633–639. doi: 10.1002/jat.1607. [DOI] [PubMed] [Google Scholar]
- 84.Rigo A., Vinante F. The antineoplastic agent alpha-bisabolol promotes cell death by inducing pores in mitochondria and lysosomes. Apoptosis. 2016;21:917–927. doi: 10.1007/s10495-016-1257-y. [DOI] [PubMed] [Google Scholar]
- 85.Oliveira G., Machado K.C., Machado K.C., da Silva A., Feitosa C.M., de Castro Almeida F.R. Non-clinical toxicity of beta-caryophyllene, a dietary cannabinoid: Absence of adverse effects in female Swiss mice. Regul. Toxicol. Pharm. 2018;92:338–346. doi: 10.1016/j.yrtph.2017.12.013. [DOI] [PubMed] [Google Scholar]
- 86.Schmitt D., Levy R., Carroll B. Toxicological Evaluation of beta-Caryophyllene Oil: Subchronic Toxicity in Rats. Int. J. Toxicol. 2016;35:558–567. doi: 10.1177/1091581816655303. [DOI] [PubMed] [Google Scholar]
- 87.Singireesu S., Misra S., Mondal S.K., Yerramsetty S., Sahu N., K S.B. Costunolide induces micronuclei formation, chromosomal aberrations, cytostasis, and mitochondrial-mediated apoptosis in Chinese hamster ovary cells. Cell Biol. Toxicol. 2018;34:125–142. doi: 10.1007/s10565-017-9411-z. [DOI] [PubMed] [Google Scholar]
- 88.Berges C., Fuchs D., Opelz G., Daniel V., Naujokat C. Helenalin suppresses essential immune functions of activated CD4+ T cells by multiple mechanisms. Mol. Immunol. 2009;46:2892–2901. doi: 10.1016/j.molimm.2009.07.004. [DOI] [PubMed] [Google Scholar]
- 89.Supornsilchai V., Soder O., Svechnikov K. Sesquiterpene lactone helenalin suppresses Leydig and adrenocortical cell steroidogenesis by inhibiting expression of the steroidogenic acute regulatory protein. Reprod. Toxicol. 2006;22:631–635. doi: 10.1016/j.reprotox.2006.04.011. [DOI] [PubMed] [Google Scholar]
- 90.Ku C.M., Lin J.Y. Farnesol, a sesquiterpene alcohol in herbal plants, exerts anti-inflammatory and antiallergic effects on ovalbumin-sensitized and -challenged asthmatic mice. Evid. Based Complement. Altern. Med. 2015;2015:387357. doi: 10.1155/2015/387357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Ku C.M., Lin J.Y. Farnesol, a sesquiterpene alcohol in essential oils, ameliorates serum allergic antibody titres and lipid profiles in ovalbumin-challenged mice. Allergol. Immunopathol. (Madr.) 2016;44:149–159. doi: 10.1016/j.aller.2015.05.009. [DOI] [PubMed] [Google Scholar]
- 92.De Oliveira Junior W.M., Benedito R.B., Pereira W.B., de Arruda Torres P., Ramos C.A., Costa J.P., da Rocha Tome A., de Sousa D.P., de Freitas R.M., de Fatima Formiga Melo Diniz M., et al. Farnesol: Antinociceptive effect and histopathological analysis of the striatum and hippocampus of mice. Fundam. Clin. Pharm. 2013;27:419–426. doi: 10.1111/j.1472-8206.2012.01030.x. [DOI] [PubMed] [Google Scholar]
- 93.Sharma C., Al Kaabi J.M., Nurulain S.M., Goyal S.N., Kamal M.A., Ojha S. Polypharmacological Properties and Therapeutic Potential of beta-Caryophyllene: A Dietary Phytocannabinoid of Pharmaceutical Promise. Curr. Pharm. Des. 2016;22:3237–3264. doi: 10.2174/1381612822666160311115226. [DOI] [PubMed] [Google Scholar]
- 94.Joo J.H., Jetten A.M. Molecular mechanisms involved in farnesol-induced apoptosis. Cancer Lett. 2010;287:123–135. doi: 10.1016/j.canlet.2009.05.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Biazi B.I., Zanetti T.A., Baranoski A., Corveloni A.C., Mantovani M.S. Cis-Nerolidol Induces Endoplasmic Reticulum Stress and Cell Death in Human Hepatocellular Carcinoma Cells through Extensive CYP2C19 and CYP1A2 Oxidation. Basic Clin. Pharm. Toxicol. 2017;121:334–341. doi: 10.1111/bcpt.12772. [DOI] [PubMed] [Google Scholar]
- 96.Kim C., Cho S.K., Kim K.D., Nam D., Chung W.S., Jang H.J., Lee S.G., Shim B.S., Sethi G., Ahn K.S. β-Caryophyllene oxide potentiates TNFα-induced apoptosis and inhibits invasion through down-modulation of NF-κB-regulated gene products. Apoptosis. 2014;19:708–718. doi: 10.1007/s10495-013-0957-9. [DOI] [PubMed] [Google Scholar]
- 97.Wang J., Li H., Yao Y., Ren Y., Lin J., Hu J., Zheng M., Song X., Zhao T., Chen Y.Y., et al. beta-Elemene Enhances GAP-43 Expression and Neurite Outgrowth by Inhibiting RhoA Kinase Activation in Rats with Spinal Cord Injury. Neuroscience. 2018;383:12–21. doi: 10.1016/j.neuroscience.2018.04.045. [DOI] [PubMed] [Google Scholar]
- 98.Xue C., Hong L.L., Lin J.S., Yao X.Y., Wu D.H., Lin X.P., Zhang J.M., Zhang X.B., Zeng Y.M. β-Elemene inhibits the proliferation of primary human airway granulation fibroblasts by down-regulating canonical Wnt/beta-catenin pathway. Biosci. Rep. 2018;38 doi: 10.1042/BSR20171386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Fang Y., Kang Y., Zou H., Cheng X., Xie T., Shi L., Zhang H. β-Elemene attenuates macrophage activation and proinflammatory factor production via crosstalk with Wnt/β-catenin signaling pathway. Fitoterapia. 2018;124:92–102. doi: 10.1016/j.fitote.2017.10.015. [DOI] [PubMed] [Google Scholar]
- 100.Liu Y., Jiang Z.Y., Zhou Y.L., Qiu H.H., Wang G., Luo Y., Liu J.B., Liu X.W., Bu W.Q., Song J., et al. β-Elemene regulates endoplasmic reticulum stress to induce the apoptosis of NSCLC cells through PERK/IRE1α/ATF6 pathway. Biomed. Pharm. 2017;93:490–497. doi: 10.1016/j.biopha.2017.06.073. [DOI] [PubMed] [Google Scholar]
- 101.Chen X., Wang Y., Luo H., Luo Z., Zhang T., Yang N., Long X., Xie H., Qiu W., Zhang B., et al. β-Elemene acts as an antitumor factor and downregulates the expression of survivin, Bcl-xL and Mta-1. Mol. Med. Rep. 2012;6:989–995. doi: 10.3892/mmr.2012.1018. [DOI] [PubMed] [Google Scholar]
- 102.Kim J.Y., An J.M., Chung W.Y., Park K.K., Hwang J.K., Kim du S., Seo S.R., Seo J.T. Xanthorrhizol induces apoptosis through ROS-mediated MAPK activation in human oral squamous cell carcinoma cells and inhibits DMBA-induced oral carcinogenesis in hamsters. Phytother. Res. 2013;27:493–498. doi: 10.1002/ptr.4746. [DOI] [PubMed] [Google Scholar]
- 103.Kang Y.J., Park K.K., Chung W.Y., Hwang J.K., Lee S.K. Xanthorrhizol, a natural sesquiterpenoid, induces apoptosis and growth arrest in HCT116 human colon cancer cells. J. Pharm. Sci. 2009;111:276–284. doi: 10.1254/jphs.09141FP. [DOI] [PubMed] [Google Scholar]
- 104.Handayani T., Sakinah S., Nallappan M., Pihie A.H. Regulation of p53-, Bcl-2- and caspase-dependent signaling pathway in xanthorrhizol-induced apoptosis of HepG2 hepatoma cells. Anticancer Res. 2007;27:965–971. [PubMed] [Google Scholar]
- 105.Lou J., Cao G., Li R., Liu J., Dong Z., Xu L. β-Caryophyllene Attenuates Focal Cerebral Ischemia-Reperfusion Injury by Nrf2/HO-1 Pathway in Rats. Neurochem. Res. 2016;41:1291–1304. doi: 10.1007/s11064-016-1826-z. [DOI] [PubMed] [Google Scholar]
- 106.Cho H.I., Hong J.M., Choi J.W., Choi H.S., Kwak J.H., Lee D.U., Kook Lee S., Lee S.M. β-Caryophyllene alleviates D-galactosamine and lipopolysaccharide-induced hepatic injury through suppression of the TLR4 and RAGE signaling pathways. Eur. J. Pharm. 2015;764:613–621. doi: 10.1016/j.ejphar.2015.08.001. [DOI] [PubMed] [Google Scholar]
- 107.Alvarez-Gonzalez I., Madrigal-Bujaidar E., Castro-Garcia S. Antigenotoxic capacity of beta-caryophyllene in mouse, and evaluation of its antioxidant and GST induction activities. J. Toxicol. Sci. 2014;39:849–859. doi: 10.2131/jts.39.849. [DOI] [PubMed] [Google Scholar]
- 108.Cheng Y., Dong Z., Liu S. β-Caryophyllene ameliorates the Alzheimer-like phenotype in APP/PS1 Mice through CB2 receptor activation and the PPARγ pathway. Pharmacology. 2014;94:1–12. doi: 10.1159/000362689. [DOI] [PubMed] [Google Scholar]
- 109.Chen Z., Zhang D., Li M., Wang B. Costunolide ameliorates lipoteichoic acid-induced acute lung injury via attenuating MAPK signaling pathway. Int. Immunopharmacol. 2018;61:283–289. doi: 10.1016/j.intimp.2018.06.017. [DOI] [PubMed] [Google Scholar]
- 110.Hu M., Liu L., Yao W. Activation of p53 by costunolide blocks glutaminolysis and inhibits proliferation in human colorectal cancer cells. Gene. 2018;678:261–269. doi: 10.1016/j.gene.2018.08.048. [DOI] [PubMed] [Google Scholar]
- 111.Wang Y., Zhang X., Zhao L., Shi M., Wei Z., Yang Z., Guo C., Fu Y. Costunolide protects lipopolysaccharide/d-galactosamine-induced acute liver injury in mice by inhibiting NF-kappaB signaling pathway. J. Surg. Res. 2017;220:40–45. doi: 10.1016/j.jss.2017.06.083. [DOI] [PubMed] [Google Scholar]
- 112.Rayan N.A., Baby N., Pitchai D., Indraswari F., Ling E.A., Lu J., Dheen T. Costunolide inhibits proinflammatory cytokines and iNOS in activated murine BV2 microglia. Front. Biosci. (Elite Ed.) 2011;3:1079–1091. doi: 10.2741/312. [DOI] [PubMed] [Google Scholar]
- 113.Choi S.H., Im E., Kang H.K., Lee J.H., Kwak H.S., Bae Y.T., Park H.J., Kim N.D. Inhibitory effects of costunolide on the telomerase activity in human breast carcinoma cells. Cancer Lett. 2005;227:153–162. doi: 10.1016/j.canlet.2005.01.011. [DOI] [PubMed] [Google Scholar]
- 114.Juliana C., Fernandes-Alnemri T., Wu J., Datta P., Solorzano L., Yu J.W., Meng R., Quong A.A., Latz E., Scott C.P., et al. Anti-inflammatory compounds parthenolide and Bay 11-7082 are direct inhibitors of the inflammasome. J. Biol. Chem. 2010;285:9792–9802. doi: 10.1074/jbc.M109.082305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Liu M., Xiao C., Sun M., Tan M., Hu L., Yu Q. Parthenolide Inhibits STAT3 Signaling by Covalently Targeting Janus Kinases. Molecules. 2018;23:1478. doi: 10.3390/molecules23061478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Zhu X., Yuan C., Tian C., Li C., Nie F., Song X., Zeng R., Wu D., Hao X., Li L. The plant sesquiterpene lactone parthenolide inhibits Wnt/beta-catenin signaling by blocking synthesis of the transcriptional regulators TCF4/LEF1. J. Biol. Chem. 2018;293:5335–5344. doi: 10.1074/jbc.M117.819300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Kwak S.W., Park E.S., Lee C.S. Parthenolide induces apoptosis by activating the mitochondrial and death receptor pathways and inhibits FAK-mediated cell invasion. Mol. Cell. Biochem. 2014;385:133–144. doi: 10.1007/s11010-013-1822-4. [DOI] [PubMed] [Google Scholar]
- 118.Shin M., McGowan A., DiNatale G.J., Chiramanewong T., Cai T., Connor R.E. Hsp72 Is an Intracellular Target of the alpha, beta-Unsaturated Sesquiterpene Lactone, Parthenolide. ACS Omega. 2017;2:7267–7274. doi: 10.1021/acsomega.7b00954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Zwicker P., Schultze N., Niehs S., Albrecht D., Methling K., Wurster M., Wachlin G., Lalk M., Lindequist U., Haertel B. Differential effects of Helenalin, an anti-inflammatory sesquiterpene lactone, on the proteome, metabolome and the oxidative stress response in several immune cell types. Toxicol. In Vitro. 2017;40:45–54. doi: 10.1016/j.tiv.2016.12.010. [DOI] [PubMed] [Google Scholar]
- 120.Lim C.B., Fu P.Y., Ky N., Zhu H.S., Feng X., Li J., Srinivasan K.G., Hamza M.S., Zhao Y. NF-kappaB p65 repression by the sesquiterpene lactone, Helenalin, contributes to the induction of autophagy cell death. BMC Complement. Altern. Med. 2012;12:93. doi: 10.1186/1472-6882-12-93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Lyss G., Knorre A., Schmidt T.J., Pahl H.L., Merfort I. The anti-inflammatory sesquiterpene lactone helenalin inhibits the transcription factor NF-kappaB by directly targeting p65. J. Biol. Chem. 1998;273:33508–33516. doi: 10.1074/jbc.273.50.33508. [DOI] [PubMed] [Google Scholar]
- 122.Tornhamre S., Schmidt T.J., Nasman-Glaser B., Ericsson I., Lindgren J.A. Inhibitory effects of helenalin and related compounds on 5-lipoxygenase and leukotriene C(4) synthase in human blood cells. Biochem. Pharm. 2001;62:903–911. doi: 10.1016/S0006-2952(01)00729-8. [DOI] [PubMed] [Google Scholar]
- 123.Zhang Z., Xu L., Cheung H.Y. The inhibitory effect of helenalin on telomerase activity is attributed to the alkylation of the CYS445 residue: Evidence from QM/MM simulations. J. Mol. Graph. Model. 2014;51:97–103. doi: 10.1016/j.jmgm.2014.04.012. [DOI] [PubMed] [Google Scholar]
- 124.Liu J., Liu M., Wang S., He Y., Huo Y., Yang Z., Cao X. Alantolactone induces apoptosis and suppresses migration in MCF7 human breast cancer cells via the p38 MAPK, NFkappaB and Nrf2 signaling pathways. Int. J. Mol. Med. 2018;42:1847–1856. doi: 10.3892/ijmm.2018.3751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Seo J.Y., Lim S.S., Kim J.R., Lim J.S., Ha Y.R., Lee I.A., Kim E.J., Park J.H., Kim J.S. Nrf2-mediated induction of detoxifying enzymes by alantolactone present in Inula helenium. Phytother. Res. 2008;22:1500–1505. doi: 10.1002/ptr.2521. [DOI] [PubMed] [Google Scholar]
- 126.Lei J.C., Yu J.Q., Yin Y., Liu Y.W., Zou G.L. Alantolactone induces activation of apoptosis in human hepatoma cells. Food Chem. Toxicol. 2012;50:3313–3319. doi: 10.1016/j.fct.2012.06.014. [DOI] [PubMed] [Google Scholar]
- 127.Lim H.S., Jin S.E., Kim O.S., Shin H.K., Jeong S.J. Alantolactone from Saussurea lappa Exerts Antiinflammatory Effects by Inhibiting Chemokine Production and STAT1 Phosphorylation in TNF-alpha and IFN-gamma-induced in HaCaT cells. Phytother. Res. 2015;29:1088–1096. doi: 10.1002/ptr.5354. [DOI] [PubMed] [Google Scholar]
- 128.Zheng H., Yang L., Kang Y., Chen M., Lin S., Xiang Y., Li C., Dai X., Huang X., Liang G., et al. Alantolactone sensitizes human pancreatic cancer cells to EGFR inhibitors through the inhibition of STAT3 signaling. Mol. Carcinog. 2019;58:565–576. doi: 10.1002/mc.22951. [DOI] [PubMed] [Google Scholar]
- 129.Kim H.L., Jung Y., Park J., Youn D.H., Kang J., Lim S., Lee B.S., Jeong M.Y., Choe S.K., Park R., et al. Farnesol Has an Anti-obesity Effect in High-Fat Diet-Induced Obese Mice and Induces the Development of Beige Adipocytes in Human Adipose Tissue Derived-Mesenchymal Stem Cells. Front. Pharm. 2017;8:654. doi: 10.3389/fphar.2017.00654. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Jung Y.Y., Hwang S.T., Sethi G., Fan L., Arfuso F., Ahn K.S. Potential Anti-Inflammatory and Anti-Cancer Properties of Farnesol. Molecules. 2018;23:2827. doi: 10.3390/molecules23112827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Szucs G., Murlasits Z., Torok S., Kocsis G.F., Paloczi J., Gorbe A., Csont T., Csonka C., Ferdinandy P. Cardioprotection by farnesol: Role of the mevalonate pathway. Cardiovasc. Drugs. 2013;27:269–277. doi: 10.1007/s10557-013-6460-2. [DOI] [PubMed] [Google Scholar]
- 132.De Carvalho R.B.F., De Almeida A.A.C., Campelo N.B., Lellis D., Nunes L.C.C. Nerolidol and its Pharmacological Application in Treating Neurodegenerative Diseases: A Review. Recent Pat. Biotechnol. 2018;12:158–168. doi: 10.2174/1872208312666171206123805. [DOI] [PubMed] [Google Scholar]
- 133.Chan W.K., Tan L.T., Chan K.G., Lee L.H., Goh B.H. Nerolidol: A Sesquiterpene Alcohol with Multi-Faceted Pharmacological and Biological Activities. Molecules. 2016;21:529. doi: 10.3390/molecules21050529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Fonseca D.V., Salgado P.R., de Carvalho F.L., Salvadori M.G., Penha A.R., Leite F.C., Borges C.J., Piuvezam M.R., Pordeus L.C., Sousa D.P., et al. Nerolidol exhibits antinociceptive and anti-inflammatory activity: Involvement of the GABAergic system and proinflammatory cytokines. Fundam Clin. Pharm. 2016;30:14–22. doi: 10.1111/fcp.12166. [DOI] [PubMed] [Google Scholar]
- 135.Fernandes M.Y.D., Carmo M., Fonteles A.A., Neves J.C.S., Silva A., Pereira J.F., Ferreira E.O., Lima N.M.R., Neves K.R.T., Andrade G.M. (-)-alpha-bisabolol prevents neuronal damage and memory deficits through reduction of proinflammatory markers induced by permanent focal cerebral ischemia in mice. Eur. J. Pharm. 2019;842:270–280. doi: 10.1016/j.ejphar.2018.09.036. [DOI] [PubMed] [Google Scholar]
- 136.Maurya A.K., Singh M., Dubey V., Srivastava S., Luqman S., Bawankule D.U. alpha-(-)-bisabolol reduces pro-inflammatory cytokine production and ameliorates skin inflammation. Curr. Pharm. Biotechnol. 2014;15:173–181. doi: 10.2174/1389201015666140528152946. [DOI] [PubMed] [Google Scholar]
- 137.Rocha N.F., Rios E.R., Carvalho A.M., Cerqueira G.S., Lopes Ade A., Leal L.K., Dias M.L., de Sousa D.P., de Sousa F.C. Anti-nociceptive and anti-inflammatory activities of (-)-alpha-bisabolol in rodents. Naunyn Schmiedebergs Arch. Pharm. 2011;384:525–533. doi: 10.1007/s00210-011-0679-x. [DOI] [PubMed] [Google Scholar]
- 138.Leite Gde O., Leite L.H., Sampaio Rde S., Araruna M.K., de Menezes I.R., da Costa J.G., Campos A.R. (-)-alpha-Bisabolol attenuates visceral nociception and inflammation in mice. Fitoterapia. 2011;82:208–211. doi: 10.1016/j.fitote.2010.09.012. [DOI] [PubMed] [Google Scholar]
- 139.Lim C.S., Jin D.Q., Mok H., Oh S.J., Lee J.U., Hwang J.K., Ha I., Han J.S. Antioxidant and antiinflammatory activities of xanthorrhizol in hippocampal neurons and primary cultured microglia. J. Neurosci. Res. 2005;82:831–838. doi: 10.1002/jnr.20692. [DOI] [PubMed] [Google Scholar]
- 140.Kim M.B., Kim C., Song Y., Hwang J.K. Antihyperglycemic and Anti-Inflammatory Effects of Standardized Curcuma xanthorrhiza Roxb. Extract and Its Active Compound Xanthorrhizol in High-Fat Diet-Induced Obese Mice. Evid. Based Complement. Altern. Med. 2014;2014:205915. doi: 10.1155/2014/205915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Oon S.F., Nallappan M., Tee T.T., Shohaimi S., Kassim N.K., Sa’ariwijaya M.S., Cheah Y.H. Xanthorrhizol: A review of its pharmacological activities and anticancer properties. Cancer Cell Int. 2015;15:100. doi: 10.1186/s12935-015-0255-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Liu M., Chen X., Ma J., Hassan W., Wu H., Ling J., Shang J. beta-Elemene attenuates atherosclerosis in apolipoprotein E-deficient mice via restoring NO levels and alleviating oxidative stress. Biomed. Pharm. 2017;95:1789–1798. doi: 10.1016/j.biopha.2017.08.092. [DOI] [PubMed] [Google Scholar]
- 143.Zhong Y., Liu J., Huo W.M., Duan W.L., Wang X., Shang J. beta-Elemene reduces the progression of atherosclerosis in rabbits. Chin. J. Nat. Med. 2015;13:415–420. doi: 10.1016/S1875-5364(15)30034-0. [DOI] [PubMed] [Google Scholar]
- 144.Jiang Z., Jacob J.A., Loganathachetti D.S., Nainangu P., Chen B. beta-Elemene: Mechanistic Studies on Cancer Cell Interaction and Its Chemosensitization Effect. Front. Pharm. 2017;8:105. doi: 10.3389/fphar.2017.00105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Jiang S., Ling C., Li W., Jiang H., Zhi Q., Jiang M. Molecular Mechanisms of Anti-cancer Activities of β-elemene: Targeting Hallmarks of Cancer. Anticancer Agents Med. Chem. 2016;16:1426–1434. doi: 10.2174/1871520616666160211123424. [DOI] [PubMed] [Google Scholar]
- 146.Yang M., Lv Y., Tian X., Lou J., An R., Zhang Q., Li M., Xu L., Dong Z. Neuroprotective Effect of beta-Caryophyllene on Cerebral Ischemia-Reperfusion Injury via Regulation of Necroptotic Neuronal Death and Inflammation: In Vivo and In Vitro. Front. Neurosci. 2017;11:583. doi: 10.3389/fnins.2017.00583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Ojha S., Javed H., Azimullah S., Haque M.E. beta-Caryophyllene, a phytocannabinoid attenuates oxidative stress, neuroinflammation, glial activation, and salvages dopaminergic neurons in a rat model of Parkinson disease. Mol. Cell. Biochem. 2016;418:59–70. doi: 10.1007/s11010-016-2733-y. [DOI] [PubMed] [Google Scholar]
- 148.Viveros-Paredes J.M., Gonzalez-Castaneda R.E., Gertsch J., Chaparro-Huerta V., Lopez-Roa R.I., Vazquez-Valls E., Beas-Zarate C., Camins-Espuny A., Flores-Soto M.E. Neuroprotective Effects of beta-Caryophyllene against Dopaminergic Neuron Injury in a Murine Model of Parkinson’s Disease Induced by MPTP. Pharmaceuticals. 2017;10:60. doi: 10.3390/ph10030060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Chang H.J., Kim J.M., Lee J.C., Kim W.K., Chun H.S. Protective effect of beta-caryophyllene, a natural bicyclic sesquiterpene, against cerebral ischemic injury. J. Med. Food. 2013;16:471–480. doi: 10.1089/jmf.2012.2283. [DOI] [PubMed] [Google Scholar]
- 150.Machado K.D.C., Islam M.T., Ali E.S., Rouf R., Uddin S.J., Dev S., Shilpi J.A., Shill M.C., Reza H.M., Das A.K., et al. A systematic review on the neuroprotective perspectives of beta-caryophyllene. Phytother. Res. 2018;32:2376–2388. doi: 10.1002/ptr.6199. [DOI] [PubMed] [Google Scholar]
- 151.Saraswati S., Alhaider A.A., Abdelgadir A.M. Costunolide suppresses an inflammatory angiogenic response in a subcutaneous murine sponge model. APMIS. 2018;126:257–266. doi: 10.1111/apm.12808. [DOI] [PubMed] [Google Scholar]
- 152.Butturini E., Di Paola R., Suzuki H., Paterniti I., Ahmad A., Mariotto S., Cuzzocrea S. Costunolide and Dehydrocostuslactone, two natural sesquiterpene lactones, ameliorate the inflammatory process associated to experimental pleurisy in mice. Eur. J. Pharm. 2014;730:107–115. doi: 10.1016/j.ejphar.2014.02.031. [DOI] [PubMed] [Google Scholar]
- 153.Zhang M., Liu R.T., Zhang P., Zhang N., Yang C.L., Yue L.T., Li X.L., Liu Y., Li H., Du J., et al. Parthenolide inhibits the initiation of experimental autoimmune neuritis. J. Neuroimmunol. 2017;305:154–161. doi: 10.1016/j.jneuroim.2017.02.003. [DOI] [PubMed] [Google Scholar]
- 154.Popiolek-Barczyk K., Kolosowska N., Piotrowska A., Makuch W., Rojewska E., Jurga A.M., Pilat D., Mika J. Parthenolide Relieves Pain and Promotes M2 Microglia/Macrophage Polarization in Rat Model of Neuropathy. Neural Plast. 2015;2015:676473. doi: 10.1155/2015/676473. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Bahabadi M., Mohammadalipour A., Karimi J., Sheikh N., Solgi G., Goudarzi F., Hashemnia M., Khodadadi I. Hepatoprotective effect of parthenolide in rat model of nonalcoholic fatty liver disease. Immunopharmacol. Immunotoxicol. 2017;39:233–242. doi: 10.1080/08923973.2017.1327965. [DOI] [PubMed] [Google Scholar]
- 156.Khare P., Datusalia A.K., Sharma S.S. Parthenolide, an NF-kappaB Inhibitor Ameliorates Diabetes-Induced Behavioural Deficit, Neurotransmitter Imbalance and Neuroinflammation in Type 2 Diabetes Rat Model. Neuromol. Med. 2017;19:101–112. doi: 10.1007/s12017-016-8434-6. [DOI] [PubMed] [Google Scholar]
- 157.Wang M., Li Q. Parthenolide could become a promising and stable drug with anti-inflammatory effects. Nat. Prod. Res. 2015;29:1092–1101. doi: 10.1080/14786419.2014.981541. [DOI] [PubMed] [Google Scholar]
- 158.Liu Q., Zhao J., Tan R., Zhou H., Lin Z., Zheng M., Romas E., Xu J., Sims N.A. Parthenolide inhibits pro-inflammatory cytokine production and exhibits protective effects on progression of collagen-induced arthritis in a rat model. Scand. J. Rheumatol. 2015;44:182–191. doi: 10.3109/03009742.2014.938113. [DOI] [PubMed] [Google Scholar]
- 159.Duncan R.E., Archer M.C. Farnesol induces thyroid hormone receptor (THR) beta1 but inhibits THR-mediated signaling in MCF-7 human breast cancer cells. Biochem. Biophys. Res. Commun. 2006;343:239–243. doi: 10.1016/j.bbrc.2006.02.145. [DOI] [PubMed] [Google Scholar]
- 160.Wang J., Li H., Ren Y., Yao Y., Hu J., Zheng M., Ding Y., Chen Y.Y., Shen Y., Wang L.L., et al. Local Delivery of beta-Elemene Improves Locomotor Functional Recovery by Alleviating Endoplasmic Reticulum Stress and Reducing Neuronal Apoptosis in Rats with Spinal Cord Injury. Cell Physiol. Biochem. 2018;49:595–609. doi: 10.1159/000492996. [DOI] [PubMed] [Google Scholar]
- 161.Mao Y., Zhang J., Hou L., Cui X. The effect of beta-elemene on alpha-tubulin polymerization in human hepatoma HepG2 cells. Chin. J. Cancer Res. 2013;25:770–776. doi: 10.3978/j.issn.1000-9604.2013.12.12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Wu L., Wang G., Tang S., Long G., Yin T. Protection of endothelial cells, inhibition of neointimal hyperplasia by beta-elemene in an injured artery. Cardiovasc. Drugs. 2011;25:233–242. doi: 10.1007/s10557-011-6305-9. [DOI] [PubMed] [Google Scholar]
- 163.Varga Z.V., Matyas C., Erdelyi K., Cinar R., Nieri D., Chicca A., Nemeth B.T., Paloczi J., Lajtos T., Corey L., et al. beta-Caryophyllene protects against alcoholic steatohepatitis by attenuating inflammation and metabolic dysregulation in mice. Br. J. Pharm. 2018;175:320–334. doi: 10.1111/bph.13722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Yamaguchi M., Levy R.M. beta-Caryophyllene promotes osteoblastic mineralization, and suppresses osteoclastogenesis and adipogenesis in mouse bone marrow cultures in vitro. Exp. Med. 2016;12:3602–3606. doi: 10.3892/etm.2016.3818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Basha R.H., Sankaranarayanan C. beta-Caryophyllene, a natural sesquiterpene, modulates carbohydrate metabolism in streptozotocin-induced diabetic rats. Acta Histochem. 2014;116:1469–1479. doi: 10.1016/j.acthis.2014.10.001. [DOI] [PubMed] [Google Scholar]
- 166.Tambe Y., Tsujiuchi H., Honda G., Ikeshiro Y., Tanaka S. Gastric cytoprotection of the non-steroidal anti-inflammatory sesquiterpene, beta-caryophyllene. Planta Med. 1996;62:469–470. doi: 10.1055/s-2006-957942. [DOI] [PubMed] [Google Scholar]
- 167.Anggakusuma, Yanti, Lee M., Hwang J.K. Estrogenic activity of xanthorrhizol isolated from curcuma xanthorrhiza ROXB. Biol. Pharm. Bull. 2009;32:1892–1897. doi: 10.1248/bpb.32.1892. [DOI] [PubMed] [Google Scholar]
- 168.Lee L.Y., Shim J.S., Rukayadi Y., Hwang J.K. Antibacterial activity of xanthorrhizol isolated from Curcuma xanthorrhiza Roxb. against foodborne pathogens. J. Food Prot. 2008;71:1926–1930. doi: 10.4315/0362-028X-71.9.1926. [DOI] [PubMed] [Google Scholar]
- 169.Wellwood C.R., Cole R.A. Relevance of carnosic acid concentrations to the selection of rosemary, Rosmarinus officinalis (L.), accessions for optimization of antioxidant yield. J. Agric. Food Chem. 2004;52:6101–6107. doi: 10.1021/jf035335p. [DOI] [PubMed] [Google Scholar]
- 170.Hossain M.B., Rai D.K., Brunton N.P., Martin-Diana A.B., Barry-Ryan C. Characterization of phenolic composition in Lamiaceae spices by LC-ESI-MS/MS. J. Agric. Food Chem. 2010;58:10576–10581. doi: 10.1021/jf102042g. [DOI] [PubMed] [Google Scholar]
- 171.Birtic S., Dussort P., Pierre F.X., Bily A.C., Roller M. Carnosic acid. Phytochemistry. 2015;115:9–19. doi: 10.1016/j.phytochem.2014.12.026. [DOI] [PubMed] [Google Scholar]
- 172.Kim J., Kang Y.-G., Lee J.-Y., Choi D.-H., Cho Y.-U., Shin J.-M., Park J.S., Lee J.H., Kim W.G., Seo D.B., et al. The natural phytochemical dehydroabietic acid is an anti-aging reagent that mediates the direct activation of SIRT1. Mol. Cell Endocrinol. 2015;412:216–225. doi: 10.1016/j.mce.2015.05.006. [DOI] [PubMed] [Google Scholar]
- 173.Park J.Y., Lee Y.K., Lee D.S., Yoo J.E., Shin M.S., Yamabe N., Kim S.N., Lee S., Kim K.H., Lee H.J., et al. Abietic acid isolated from pine resin (Resina Pini) enhances angiogenesis in HUVECs and accelerates cutaneous wound healing in mice. J. Ethnopharmacol. 2017;203:279–287. doi: 10.1016/j.jep.2017.03.055. [DOI] [PubMed] [Google Scholar]
- 174.Momtazi-Borojeni A.A., Esmaeili S.A., Abdollahi E., Sahebkar A. A Review on the Pharmacology and Toxicology of Steviol Glycosides Extracted from Stevia rebaudiana. Curr. Pharm. Des. 2017;23:1616–1622. doi: 10.2174/1381612822666161021142835. [DOI] [PubMed] [Google Scholar]
- 175.Bucknall R.A., Moores H., Simms R., Hesp B. Antiviral effects of aphidicolin, a new antibiotic produced by Cephalosporium aphidicola. Antimicrob. Agents Chemother. 1973;4:294–298. doi: 10.1128/AAC.4.3.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Lin C., Zhang X., Su Z., Xiao J., Lv M., Cao Y., Chen Y. Carnosol Improved Lifespan and Healthspan by Promoting Antioxidant Capacity in Caenorhabditis elegans. Oxid. Med. Cell. Longev. 2019;2019:5958043. doi: 10.1155/2019/5958043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Lin C., Zhang X., Xiao J., Zhong Q., Kuang Y., Cao Y., Chen Y. Effects on longevity extension and mechanism of action of carnosic acid in Caenorhabditis elegans. Food Funct. 2019;10:1398–1410. doi: 10.1039/C8FO02371A. [DOI] [PubMed] [Google Scholar]
- 178.Aruoma O.I., Halliwell B., Aeschbach R., Loligers J. Antioxidant and pro-oxidant properties of active rosemary constituents: Carnosol and carnosic acid. Xenobiotica. 1992;22:257–268. doi: 10.3109/00498259209046624. [DOI] [PubMed] [Google Scholar]
- 179.Loussouarn M., Krieger-Liszkay A., Svilar L., Bily A., Birtic S., Havaux M. Carnosic Acid and Carnosol, Two Major Antioxidants of Rosemary, Act through Different Mechanisms. Plant. Physiol. 2017;175:1381–1394. doi: 10.1104/pp.17.01183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Minnunni M., Wolleb U., Mueller O., Pfeifer A., Aeschbacher H.U. Natural antioxidants as inhibitors of oxygen species induced mutagenicity. Mutat. Res. 1992;269:193–200. doi: 10.1016/0027-5107(92)90200-L. [DOI] [PubMed] [Google Scholar]
- 181.Del Bano M.J., Castillo J., Benavente-Garcia O., Lorente J., Martin-Gil R., Acevedo C., Alcaraz M. Radioprotective-antimutagenic effects of rosemary phenolics against chromosomal damage induced in human lymphocytes by gamma-rays. J. Agric. Food Chem. 2006;54:2064–2068. doi: 10.1021/jf0581574. [DOI] [PubMed] [Google Scholar]
- 182.Fahim F.A., Esmat A.Y., Fadel H.M., Hassan K.F. Allied studies on the effect of Rosmarinus officinalis L. on experimental hepatotoxicity and mutagenesis. Int. J. Food Sci. Nutr. 1999;50:413–427. doi: 10.1080/096374899100987. [DOI] [PubMed] [Google Scholar]
- 183.Dao T.T., Le T.V., Nguyen P.H., Thuong P.T., Minh P.T., Woo E.R., Lee K.Y., Oh W.K. SIRT1 inhibitory diterpenoids from the Vietnamese medicinal plant Croton tonkinensis. Planta Med. 2010;76:1011–1014. doi: 10.1055/s-0029-1240863. [DOI] [PubMed] [Google Scholar]
- 184.Hao W.R., Sung L.C., Chen C.C., Chen P.Y., Cheng T.H., Chao H.H., Liu J.C., Chen J.J. Cafestol Inhibits Cyclic-Strain-Induced Interleukin-8, Intercellular Adhesion Molecule-1, and Monocyte Chemoattractant Protein-1 Production in Vascular Endothelial Cells. Oxid. Med. Cell. Longev. 2018;2018:7861518. doi: 10.1155/2018/7861518. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 185.Romo Vaquero M., Garcia Villalba R., Larrosa M., Yanez-Gascon M.J., Fromentin E., Flanagan J., Roller M., Tomas-Barberan F.A., Espin J.C., Garcia-Conesa M.T. Bioavailability of the major bioactive diterpenoids in a rosemary extract: Metabolic profile in the intestine, liver, plasma, and brain of Zucker rats. Mol. Nutr. Food Res. 2013;57:1834–1846. doi: 10.1002/mnfr.201300052. [DOI] [PubMed] [Google Scholar]
- 186.Cavin C., Holzhaeuser D., Scharf G., Constable A., Huber W.W., Schilter B. Cafestol and kahweol, two coffee specific diterpenes with anticarcinogenic activity. Food Chem. Toxicol. 2002;40:1155–1163. doi: 10.1016/S0278-6915(02)00029-7. [DOI] [PubMed] [Google Scholar]
- 187.Ullah A., Munir S., Mabkhot Y., Badshah S.L. Bioactivity Profile of the Diterpene Isosteviol and its Derivatives. Molecules. 2019;24:678. doi: 10.3390/molecules24040678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Curry L.L., Roberts A., Brown N. Rebaudioside A: Two-generation reproductive toxicity study in rats. Food Chem. Toxicol. 2008;46:S21–S30. doi: 10.1016/j.fct.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 189.Williams C., Bryant A. Short versus long duration infusions of paclitaxel for any advanced adenocarcinoma. Cochrane Database Syst. Rev. 2011 doi: 10.1002/14651858.CD003911.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Ohmori K., Kawamura Y. Cell transformation activities of abietic acid and dehydroabietic acid: Safety assessment of possible contaminants in paper and paperboard for food contact use. Food Addit. Contam. Part. A Chem. Anal. Control Expo. Risk Assess. 2009;26:568–573. doi: 10.1080/02652030802471221. [DOI] [PubMed] [Google Scholar]
- 191.Christianson-Heiska I.L., Haavisto T., Paranko J., Bergelin E., Isomaa B. Effects of the wood extractives dehydroabietic acid and betulinol on reproductive physiology of zebrafish (Danio rerio)-a two-generation study. Aquat. Toxicol. 2008;86:388–396. doi: 10.1016/j.aquatox.2007.12.001. [DOI] [PubMed] [Google Scholar]
- 192.Pandelides Z., Guchardi J., Holdway D. Dehydroabietic acid (DHAA) alters metabolic enzyme activity and the effects of 17beta-estradiol in rainbow trout (Oncorhynchus mykiss) Ecotoxicol. Environ. Saf. 2014;101:168–176. doi: 10.1016/j.ecoenv.2013.11.027. [DOI] [PubMed] [Google Scholar]
- 193.Stegelmeier B.L., Gardner D.R., James L.F., Panter K.E., Molyneux R.J. The toxic and abortifacient effects of ponderosa pine. Vet. Pathol. 1996;33:22–28. doi: 10.1177/030098589603300103. [DOI] [PubMed] [Google Scholar]
- 194.Shimojo Y., Kosaka K., Noda Y., Shimizu T., Shirasawa T. Effect of rosmarinic acid in motor dysfunction and life span in a mouse model of familial amyotrophic lateral sclerosis. J. Neurosci. Res. 2010;88:896–904. doi: 10.1002/jnr.22242. [DOI] [PubMed] [Google Scholar]
- 195.Ninomiya K., Matsuda H., Shimoda H., Nishida N., Kasajima N., Yoshino T., Morikawa T., Yoshikawa M. Carnosic acid, a new class of lipid absorption inhibitor from sage. Bioorg. Med. Chem. Lett. 2004;14:1943–1946. doi: 10.1016/j.bmcl.2004.01.091. [DOI] [PubMed] [Google Scholar]
- 196.Fukuma Y., Sakai E., Nishishita K., Okamoto K., Tsukuba T. Cafestol has a weaker inhibitory effect on osteoclastogenesis than kahweol and promotes osteoblast differentiation. BioFactors. 2015;41:222–231. doi: 10.1002/biof.1218. [DOI] [PubMed] [Google Scholar]
- 197.Poeckel D., Greiner C., Verhoff M., Rau O., Tausch L., Hornig C., Steinhilber D., Schubert-Zsilavecz M., Werz O. Carnosic acid and carnosol potently inhibit human 5-lipoxygenase and suppress pro-inflammatory responses of stimulated human polymorphonuclear leukocytes. Biochem. Pharm. 2008;76:91–97. doi: 10.1016/j.bcp.2008.04.013. [DOI] [PubMed] [Google Scholar]
- 198.Kosaka K., Mimura J., Itoh K., Satoh T., Shimojo Y., Kitajima C., Maruyama A., Yamamoto M., Shirasawa T. Role of Nrf2 and p62/ZIP in the neurite outgrowth by carnosic acid in PC12h cells. J. Biochem. 2010;147:73–81. doi: 10.1093/jb/mvp149. [DOI] [PubMed] [Google Scholar]
- 199.Johnson J.J. Carnosol: A promising anti-cancer and anti-inflammatory agent. Cancer Lett. 2011;305:1–7. doi: 10.1016/j.canlet.2011.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Hwang Y.P., Jeong H.G. The coffee diterpene kahweol induces heme oxygenase-1 via the PI3K and p38/Nrf2 pathway to protect human dopaminergic neurons from 6-hydroxydopamine-derived oxidative stress. FEBS Lett. 2008;582:2655–2662. doi: 10.1016/j.febslet.2008.06.045. [DOI] [PubMed] [Google Scholar]
- 201.Wang T., Guo M., Song X., Zhang Z., Jiang H., Wang W., Fu Y., Cao Y., Zhu L., Zhang N. Stevioside plays an anti-inflammatory role by regulating the NF-kappaB and MAPK pathways in S. aureus-infected mouse mammary glands. Inflammation. 2014;37:1837–1846. doi: 10.1007/s10753-014-9915-0. [DOI] [PubMed] [Google Scholar]
- 202.Boonkaewwan C., Ao M., Toskulkao C., Rao M.C. Specific immunomodulatory and secretory activities of stevioside and steviol in intestinal cells. J. Agric. Food Chem. 2008;56:3777–3784. doi: 10.1021/jf072681o. [DOI] [PubMed] [Google Scholar]
- 203.Yingkun N., Zhenyu W., Jing L., Xiuyun L., Huimin Y. Stevioside protects LPS-induced acute lung injury in mice. Inflammation. 2013;36:242–250. doi: 10.1007/s10753-012-9540-8. [DOI] [PubMed] [Google Scholar]
- 204.Zhao Y.T., Guo J.H., Wu Z.L., Xiong Y., Zhou W.L. Innate immune responses of epididymal epithelial cells to Staphylococcus aureus infection. Immunol. Lett. 2008;119:84–90. doi: 10.1016/j.imlet.2008.05.002. [DOI] [PubMed] [Google Scholar]
- 205.Boonkaewwan C., Burodom A. Anti-inflammatory and immunomodulatory activities of stevioside and steviol on colonic epithelial cells. J. Sci. Food Agric. 2013;93:3820–3825. doi: 10.1002/jsfa.6287. [DOI] [PubMed] [Google Scholar]
- 206.Boonkaewwan C., Toskulkao C., Vongsakul M. Anti-Inflammatory and Immunomodulatory Activities of Stevioside and Its Metabolite Steviol on THP-1 Cells. J. Agric. Food Chem. 2006;54:785–789. doi: 10.1021/jf0523465. [DOI] [PubMed] [Google Scholar]
- 207.Yarimizu T., Mitamura T., Suzuki S., Sakamoto S. Protective effects of an antiulcer agent, ecabet sodium on colorectal carcinogenesis in rodents. Oncol. Rep. 1998;5:1103–1107. doi: 10.3892/or.5.5.1103. [DOI] [PubMed] [Google Scholar]
- 208.Kang M.S., Hirai S., Goto T., Kuroyanagi K., Lee J.Y., Uemura T., Ezaki Y., Takahashi N., Kawada T. Dehydroabietic acid, a phytochemical, acts as ligand for PPARs in macrophages and adipocytes to regulate inflammation. Biochem. Biophys. Res. Commun. 2008;369:333–338. doi: 10.1016/j.bbrc.2008.02.002. [DOI] [PubMed] [Google Scholar]
- 209.De Oliveira M.R. The Dietary Components Carnosic Acid and Carnosol as Neuroprotective Agents: A Mechanistic View. Mol. Neurobiol. 2016;53:6155–6168. doi: 10.1007/s12035-015-9519-1. [DOI] [PubMed] [Google Scholar]
- 210.Mellbye F.B., Jeppesen P.B., Hermansen K., Gregersen S. Cafestol, a Bioactive Substance in Coffee, Stimulates Insulin Secretion and Increases Glucose Uptake in Muscle Cells: Studies in Vitro. J. Nat. Prod. 2015;78:2447–2451. doi: 10.1021/acs.jnatprod.5b00481. [DOI] [PubMed] [Google Scholar]
- 211.Mellbye F.B., Jeppesen P.B., Shokouh P., Laustsen C., Hermansen K., Gregersen S. Cafestol, a Bioactive Substance in Coffee, Has Antidiabetic Properties in KKAy Mice. J. Nat. Prod. 2017;80:2353–2359. doi: 10.1021/acs.jnatprod.7b00395. [DOI] [PubMed] [Google Scholar]
- 212.Moeenfard M., Cortez A., Machado V., Costa R., Luis C., Coelho P., Soares R., Alves A., Borges N., Santos A. Anti-Angiogenic Properties of Cafestol and Kahweol Palmitate Diterpene Esters. J. Cell. Biochem. 2016;117:2748–2756. doi: 10.1002/jcb.25573. [DOI] [PubMed] [Google Scholar]
- 213.McCall A.L., Millington W.R., Wurtman R.J. Blood-brain barrier transport of caffeine: Dose-related restriction of adenine transport. Life Sci. 1982;31:2709–2715. doi: 10.1016/0024-3205(82)90715-9. [DOI] [PubMed] [Google Scholar]
- 214.Trinh K., Andrews L., Krause J., Hanak T., Lee D., Gelb M., Pallanck L. Decaffeinated coffee and nicotine-free tobacco provide neuroprotection in Drosophila models of Parkinson’s disease through an NRF2-dependent mechanism. J. Neurosci. 2010;30:5525–5532. doi: 10.1523/JNEUROSCI.4777-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Gregersen S., Jeppesen P.B., Holst J.J., Hermansen K. Antihyperglycemic effects of stevioside in type 2 diabetic subjects. Metabolism. 2004;53:73–76. doi: 10.1016/j.metabol.2003.07.013. [DOI] [PubMed] [Google Scholar]
- 216.Jeppesen P.B., Gregersen S., Poulsen C.R., Hermansen K. Stevioside acts directly on pancreatic beta cells to secrete insulin: Actions independent of cyclic adenosine monophosphate and adenosine triphosphate-sensitive K+-channel activity. Metabolism. 2000;49:208–214. doi: 10.1016/S0026-0495(00)91325-8. [DOI] [PubMed] [Google Scholar]
- 217.Jeppesen P.B., Gregersen S., Rolfsen S.E., Jepsen M., Colombo M., Agger A., Xiao J., Kruhoffer M., Orntoft T., Hermansen K. Antihyperglycemic and blood pressure-reducing effects of stevioside in the diabetic Goto-Kakizaki rat. Metabolism. 2003;52:372–378. doi: 10.1053/meta.2003.50058. [DOI] [PubMed] [Google Scholar]
- 218.Chen J., Hou K., Qin P., Liu H., Yi B., Yang W., Wu W. RNA-Seq for gene identification and transcript profiling of three Stevia rebaudiana genotypes. BMC Genom. 2014;15:571. doi: 10.1186/1471-2164-15-571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Melis M.S. Stevioside effect on renal function of normal and hypertensive rats. J. Ethnopharmacol. 1992;36:213–217. doi: 10.1016/0378-8741(92)90046-T. [DOI] [PubMed] [Google Scholar]
- 220.Li R., Morris-Natschke S.L., Lee K.H. Clerodane diterpenes: Sources, structures, and biological activities. Nat. Prod. Rep. 2016;33:1166–1226. doi: 10.1039/C5NP00137D. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Shen Y.C., Chen C.F., Chiou W.F. Andrographolide prevents oxygen radical production by human neutrophils: Possible mechanism(s) involved in its anti-inflammatory effect. Br. J. Pharm. 2002;135:399–406. doi: 10.1038/sj.bjp.0704493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Kishore V., Yarla N.S., Bishayee A., Putta S., Malla R., Neelapu N.R., Challa S., Das S., Shiralgi Y., Hegde G., et al. Multi-targeting Andrographolide and its Natural Analogs as Potential Therapeutic Agents. Curr. Top. Med. Chem. 2017;17:845–857. doi: 10.2174/1568026616666160927150452. [DOI] [PubMed] [Google Scholar]
- 223.Xia Y.F., Ye B.Q., Li Y.D., Wang J.G., He X.J., Lin X., Yao X., Ma D., Slungaard A., Hebbel R.P., et al. Andrographolide attenuates inflammation by inhibition of NF-kappa B activation through covalent modification of reduced cysteine 62 of p50. J. Immunol. 2004;173:4207–4217. doi: 10.4049/jimmunol.173.6.4207. [DOI] [PubMed] [Google Scholar]
- 224.Batkhuu J., Hattori K., Takano F., Fushiya S., Oshiman K., Fujimiya Y. Suppression of NO production in activated macrophages in vitro and ex vivo by neoandrographolide isolated from Andrographis paniculata. Biol. Pharm. Bull. 2002;25:1169–1174. doi: 10.1248/bpb.25.1169. [DOI] [PubMed] [Google Scholar]
- 225.Dai L., Wang G., Pan W. Andrographolide Inhibits Proliferation and Metastasis of SGC7901 Gastric Cancer Cells. Biomed. Res. Int. 2017;2017:6242103. doi: 10.1155/2017/6242103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Banerjee M., Chattopadhyay S., Choudhuri T., Bera R., Kumar S., Chakraborty B., Mukherjee S.K. Cytotoxicity and cell cycle arrest induced by andrographolide lead to programmed cell death of MDA-MB-231 breast cancer cell line. J. Biomed. Sci. 2016;23:40. doi: 10.1186/s12929-016-0257-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Kannaste A., Laanisto L., Pazouki L., Copolovici L., Suhorutsenko M., Azeem M., Toom L., Borg-Karlson A.K., Niinemets U. Diterpenoid fingerprints in pine foliage across an environmental and chemotypic matrix: Isoabienol content is a key trait differentiating chemotypes. Phytochemistry. 2018;147:80–88. doi: 10.1016/j.phytochem.2017.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 228.Hillier S.G., Lathe R. Terpenes, hormones and life: Isoprene rule revisited. J. Endocrinol. 2019;242:R9–R22. doi: 10.1530/JOE-19-0084. [DOI] [PubMed] [Google Scholar]
- 229.Kushiro T., Ebizuka Y. 1.18–Triterpenes. In: Liu H.-W., Mander L., editors. Comprehensive Natural Products II’. Elsevier; Oxford, ME, USA: 2010. pp. 673–708. [DOI] [Google Scholar]
- 230.Jesus J.A., Lago J.H., Laurenti M.D., Yamamoto E.S., Passero L.F. Antimicrobial activity of oleanolic and ursolic acids: An update. Evid. Based Complement. Altern. Med. 2015;2015:620472. doi: 10.1155/2015/620472. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 231.Salvador J.A.R., Leal A.S., Valdeira A.S., Goncalves B.M.F., Alho D.P.S., Figueiredo S.A.C., Silvestre S.M., Mendes V.I.S. Oleanane-, ursane-, and quinone methide friedelane-type triterpenoid derivatives: Recent advances in cancer treatment. Eur. J. Med. Chem. 2017;142:95–130. doi: 10.1016/j.ejmech.2017.07.013. [DOI] [PubMed] [Google Scholar]
- 232.Ammon H.P. Boswellic Acids and Their Role in Chronic Inflammatory Diseases. Adv. Exp. Med. Biol. 2016;928:291–327. doi: 10.1007/978-3-319-41334-1_13. [DOI] [PubMed] [Google Scholar]
- 233.Sharma H., Kumar P., Deshmukh R.R., Bishayee A., Kumar S. Pentacyclic triterpenes: New tools to fight metabolic syndrome. Phytomedicine. 2018;50:166–177. doi: 10.1016/j.phymed.2018.09.011. [DOI] [PubMed] [Google Scholar]
- 234.Siddique H.R., Saleem M. Beneficial health effects of lupeol triterpene: A review of preclinical studies. Life Sci. 2011;88:285–293. doi: 10.1016/j.lfs.2010.11.020. [DOI] [PubMed] [Google Scholar]
- 235.Seo D.Y., Lee S.R., Heo J.W., No M.H., Rhee B.D., Ko K.S., Kwak H.B., Han J. Ursolic acid in health and disease. Korean J. Physiol. Pharm. 2018;22:235–248. doi: 10.4196/kjpp.2018.22.3.235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 236.Lou-Bonafonte J.M., Martinez-Beamonte R., Sanclemente T., Surra J.C., Herrera-Marcos L.V., Sanchez-Marco J., Arnal C., Osada J. Current Insights into the Biological Action of Squalene. Mol. Nutr. Food Res. 2018:e1800136. doi: 10.1002/mnfr.201800136. [DOI] [PubMed] [Google Scholar]
- 237.Kim S.K., Karadeniz F. Biological importance and applications of squalene and squalane. Adv. Food Nutr. Res. 2012;65:223–233. doi: 10.1016/B978-0-12-416003-3.00014-7. [DOI] [PubMed] [Google Scholar]
- 238.Popa O., Babeanu N.E., Popa I., Nita S., Dinu-Parvu C.E. Methods for obtaining and determination of squalene from natural sources. Biomed. Res. Int. 2015;2015:367202. doi: 10.1155/2015/367202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Bhilwade H.N., Tatewaki N., Nishida H., Konishi T. Squalene as novel food factor. Curr. Pharm. Biotechnol. 2010;11:875–880. doi: 10.2174/138920110793262088. [DOI] [PubMed] [Google Scholar]
- 240.Pérez-Camino M.C., Cert A. Quantitative determination of hydroxy pentacyclic triterpene acids in vegetable oils. J. Agric. Food Chem. 1999;47:1558–1562. doi: 10.1021/jf980881h. [DOI] [PubMed] [Google Scholar]
- 241.Jager S., Trojan H., Kopp T., Laszczyk M.N., Scheffler A. Pentacyclic triterpene distribution in various plants–rich sources for a new group of multi-potent plant extracts. Molecules. 2009;14:2016–2031. doi: 10.3390/molecules14062016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242.Lim T.G., Lee C.C., Dong Z., Lee K.W. Ginsenosides and their metabolites: A review of their pharmacological activities in the skin. Arch. Derm. Res. 2015;307:397–403. doi: 10.1007/s00403-015-1569-8. [DOI] [PubMed] [Google Scholar]
- 243.Gao Y., Wang P., Wang Y., Wu L., Wang X., Zhang K., Liu Q. In Vitro and In Vivo Activity of Fomitopsis Pinicola (Sw. Ex Fr.) Karst Chloroform (Fpkc) Extract Against S180 Tumor Cells. Cell Physiol. Biochem. 2017;44:2042–2056. doi: 10.1159/000485944. [DOI] [PubMed] [Google Scholar]
- 244.Cheng S., Swanson K., Eliaz I., McClintick J.N., Sandusky G.E., Sliva D. Pachymic acid inhibits growth and induces apoptosis of pancreatic cancer in vitro and in vivo by targeting ER stress. PLoS ONE. 2015;10:e0122270. doi: 10.1371/journal.pone.0122270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245.Wang J., Cao B., Zhao H., Feng J. Emerging Roles of Ganoderma Lucidum in Anti-Aging. Aging Dis. 2017;8:691–707. doi: 10.14336/AD.2017.0410. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Crowley V.M., Ayi K., Lu Z., Liby K.T., Sporn M., Kain K.C. Synthetic oleanane triterpenoids enhance blood brain barrier integrity and improve survival in experimental cerebral malaria. Malar J. 2017;16:463. doi: 10.1186/s12936-017-2109-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247.Ortega-Munoz M., Rodriguez-Serrano F., De Los Reyes-Berbel E., Mut-Salud N., Hernandez-Mateo F., Rodriguez-Lopez A., Garrido J.M., Lopez-Jaramillo F.J., Santoyo-Gonzalez F. Biological Evaluation and Docking Studies of Synthetic Oleanane-type Triterpenoids. ACS Omega. 2018;3:11455–11468. doi: 10.1021/acsomega.8b01034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 248.Liu X., Li B., Zhang Z., Wei Y., Xu Z., Qin S., Liu N., Zhao R., Peng J., Yang G., et al. Synthesis and Discovery Novel Anti-Cancer Stem Cells Compounds Derived from the Natural Triterpenoic Acids. J. Med. Chem. 2018;61:10814–10833. doi: 10.1021/acs.jmedchem.8b01445. [DOI] [PubMed] [Google Scholar]
- 249.Satheesan J., Narayanan A.K., Sakunthala M. Induction of root colonization by Piriformospora indica leads to enhanced asiaticoside production in Centella asiatica. Mycorrhiza. 2012;22:195–202. doi: 10.1007/s00572-011-0394-y. [DOI] [PubMed] [Google Scholar]
- 250.Negi H., Shukla A., Khan F., Pandey R. 3beta-Hydroxy-urs-12-en-28-oic acid prolongs lifespan in C. elegans by modulating JNK-1. Biochem. Biophys. Res. Commun. 2016;480:539–543. doi: 10.1016/j.bbrc.2016.10.073. [DOI] [PubMed] [Google Scholar]
- 251.Negi H., Saikia S.K., Pandey R. 3beta-Hydroxy-urs-12-en-28-oic Acid Modulates Dietary Restriction Mediated Longevity and Ameliorates Toxic Protein Aggregation in C. Elegans. J. Gerontol. A Biol. Sci. Med. Sci. 2017;72:1614–1619. doi: 10.1093/gerona/glx118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 252.Vayndorf E.M., Lee S.S., Liu R.H. Whole apple extracts increase lifespan, healthspan and resistance to stress in Caenorhabditis elegans. J. Funct. Foods. 2013;5:1236–1243. doi: 10.1016/j.jff.2013.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253.Altun D., Ayar A., Uysal H., Kara A.A., Unal E.L. Extended longevity of Drosophila melanogaster by water and ethanol extracts of Stachys lavandulifolia. Pharm. Biol. 2010;48:1291–1296. doi: 10.3109/13880201003789424. [DOI] [PubMed] [Google Scholar]
- 254.Zhang J., Lu L., Zhou L. Oleanolic acid activates daf-16 to increase lifespan in Caenorhabditis elegans. Biochem. Biophys. Res. Commun. 2015;468:843–849. doi: 10.1016/j.bbrc.2015.11.042. [DOI] [PubMed] [Google Scholar]
- 255.Jattujan P., Chalorak P., Siangcham T., Sangpairoj K., Nobsathian S., Poomtong T., Sobhon P., Meemon K. Holothuria scabra extracts possess anti-oxidant activity and promote stress resistance and lifespan extension in Caenorhabditis elegans. Exp. Gerontol. 2018;110:158–171. doi: 10.1016/j.exger.2018.06.006. [DOI] [PubMed] [Google Scholar]
- 256.Cao X., Sun Y., Lin Y., Pan Y., Farooq U., Xiang L., Qi J. Antiaging of Cucurbitane Glycosides from Fruits of Momordica charantia L. Oxid. Med. Cell. Longev. 2018;2018:1538632. doi: 10.1155/2018/1538632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 257.Lee J.H., Choi S.H., Kwon O.S., Shin T.J., Lee J.H., Lee B.H., Yoon I.S., Pyo M.K., Rhim H., Lim Y.H., et al. Effects of ginsenosides, active ingredients of Panax ginseng, on development, growth, and life span of Caenorhabditis elegans. Biol. Pharm. Bull. 2007;30:2126–2134. doi: 10.1248/bpb.30.2126. [DOI] [PubMed] [Google Scholar]
- 258.Papaevgeniou N., Sakellari M., Jha S., Tavernarakis N., Holmberg C.I., Gonos E.S., Chondrogianni N. 18alpha-Glycyrrhetinic Acid Proteasome Activator Decelerates Aging and Alzheimer’s Disease Progression in Caenorhabditis elegans and Neuronal Cultures. Antioxid. Redox. Signal. 2016;25:855–869. doi: 10.1089/ars.2015.6494. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 259.Singh B.K., Vatsa N., Nelson V.K., Kumar V., Kumar S.S., Mandal S.C., Pal M., Jana N.R. Azadiradione Restores Protein Quality Control and Ameliorates the Disease Pathogenesis in a Mouse Model of Huntington’s Disease. Mol. Neurobiol. 2018;55:6337–6346. doi: 10.1007/s12035-017-0853-3. [DOI] [PubMed] [Google Scholar]
- 260.Kiaei M., Kipiani K., Petri S., Chen J., Calingasan N.Y., Beal M.F. Celastrol blocks neuronal cell death and extends life in transgenic mouse model of amyotrophic lateral sclerosis. Neurodegener Dis. 2005;2:246–254. doi: 10.1159/000090364. [DOI] [PubMed] [Google Scholar]
- 261.Tsai C.W., Tsai R.T., Liu S.P., Chen C.S., Tsai M.C., Chien S.H., Hung H.S., Lin S.Z., Shyu W.C., Fu R.H. Neuroprotective Effects of Betulin in Pharmacological and Transgenic Caenorhabditis elegans Models of Parkinson’s Disease. Cell Transpl. 2017;26:1903–1918. doi: 10.1177/0963689717738785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262.Chen L., Yao H., Chen X., Wang Z., Xiang Y., Xia J., Liu Y., Wang Y. Ginsenoside Rg1 Decreases Oxidative Stress and Down-Regulates Akt/mTOR Signalling to Attenuate Cognitive Impairment in Mice and Senescence of Neural Stem Cells Induced by D-Galactose. Neurochem. Res. 2018;43:430–440. doi: 10.1007/s11064-017-2438-y. [DOI] [PubMed] [Google Scholar]
- 263.Smina T.P., Joseph J., Janardhanan K.K. Ganoderma lucidum total triterpenes prevent gamma-radiation induced oxidative stress in Swiss albino mice In Vivo. Redox Rep. 2016;21:254–261. doi: 10.1080/13510002.2015.1126098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264.Park Y.M., Park S.N. Inhibitory Effect of Lupeol on MMPs Expression using Aged Fibroblast through Repeated UVA Irradiation. Photochem. Photobiol. 2019;95:587–594. doi: 10.1111/php.13022. [DOI] [PubMed] [Google Scholar]
- 265.Bahrami S.A., Bakhtiari N. Ursolic acid regulates aging process through enhancing of metabolic sensor proteins level. Biomed. Pharm. 2016;82:8–14. doi: 10.1016/j.biopha.2016.04.047. [DOI] [PubMed] [Google Scholar]
- 266.Buddhan S., Sivakumar R., Dhandapani N., Ganesan B., Anandan R. Protective effect of dietary squalene supplementation on mitochondrial function in liver of aged rats. Prostaglandins Leukot Essent Fat. Acids. 2007;76:349–355. doi: 10.1016/j.plefa.2007.05.001. [DOI] [PubMed] [Google Scholar]
- 267.Sun J., Jiao C., Ma Y., Chen J., Wu W., Liu S. Anti-ageing effect of red ginseng revealed by urinary metabonomics using RRLC-Q-TOF-MS. Phytochem. Anal. 2018;29:387–397. doi: 10.1002/pca.2758. [DOI] [PubMed] [Google Scholar]
- 268.He L., Ling L., Wei T., Wang Y., Xiong Z. Ginsenoside Rg1 improves fertility and reduces ovarian pathological damages in premature ovarian failure model of mice. Exp. Biol. Med. (Maywood) 2017;242:683–691. doi: 10.1177/1535370217693323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 269.Madhavadas S., Subramanian S. Combination of Spirulina with glycyrrhizin prevents cognitive dysfunction in aged obese rats. Indian J. Pharm. 2015;47:39–44. doi: 10.4103/0253-7613.150327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270.Nagai N., Yagyu S., Hata A., Nirengi S., Kotani K., Moritani T., Sakane N. Maslinic acid derived from olive fruit in combination with resistance training improves muscle mass and mobility functions in the elderly. J. Clin. Biochem. Nutr. 2019;64:224–230. doi: 10.3164/jcbn.18-104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 271.Szabo N.J. Dietary safety of cycloastragenol from Astragalus spp.: Subchronic toxicity and genotoxicity studies. Food Chem. Toxicol. 2014;64:322–334. doi: 10.1016/j.fct.2013.11.041. [DOI] [PubMed] [Google Scholar]
- 272.Akbarsha M.A., Palanisamy M., Murugaian P., Lakshmi Latha P.N. Ursolic acid generates symplasts in rat spermatogenic clones. Phytother. Res. 1998;12:32–36. doi: 10.1002/(SICI)1099-1573(19980201)12:1<32::AID-PTR185>3.0.CO;2-4. [DOI] [Google Scholar]
- 273.Cho S., Choi C.W., Lee D.H., Won C.H., Kim S.M., Lee S., Lee M.J., Chung J.H. High-dose squalene ingestion increases type I procollagen and decreases ultraviolet-induced DNA damage in human skin in vivo but is associated with transient adverse effects. Clin. Exp. Derm. 2009;34:500–508. doi: 10.1111/j.1365-2230.2008.03133.x. [DOI] [PubMed] [Google Scholar]
- 274.Lu J., Zheng Y.L., Wu D.M., Luo L., Sun D.X., Shan Q. Ursolic acid ameliorates cognition deficits and attenuates oxidative damage in the brain of senescent mice induced by D-galactose. Biochem. Pharm. 2007;74:1078–1090. doi: 10.1016/j.bcp.2007.07.007. [DOI] [PubMed] [Google Scholar]
- 275.Park H.J., Kwon H., Lee S., Jung J.W., Ryu J.H., Jang D.S., Lee Y.C., Kim D.H. Echinocystic Acid Facilitates Neurite Outgrowth in Neuroblastoma Neuro2a Cells and Enhances Spatial Memory in Aged Mice. Biol. Pharm. Bull. 2017;40:1724–1729. doi: 10.1248/bpb.b17-00324. [DOI] [PubMed] [Google Scholar]
- 276.Kim T.W., Choi H.J., Kim N.J., Kim D.H. Anxiolytic-like effects of ginsenosides Rg3 and Rh2 from red ginseng in the elevated plus-maze model. Planta Med. 2009;75:836–839. doi: 10.1055/s-0029-1185402. [DOI] [PubMed] [Google Scholar]
- 277.Kim J.C., Kang Y.S., Noh E.B., Seo B.W., Seo D.Y., Park G.D., Kim S.H. Concurrent treatment with ursolic acid and low-intensity treadmill exercise improves muscle atrophy and related outcomes in rats. Korean J. Physiol. Pharm. 2018;22:427–436. doi: 10.4196/kjpp.2018.22.4.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 278.Kim M., Sung B., Kang Y.J., Kim D.H., Lee Y., Hwang S.Y., Yoon J.H., Yoo M.A., Kim C.M., Chung H.Y., et al. The combination of ursolic acid and leucine potentiates the differentiation of C2C12 murine myoblasts through the mTOR signaling pathway. Int. J. Mol. Med. 2015;35:755–762. doi: 10.3892/ijmm.2014.2046. [DOI] [PubMed] [Google Scholar]
- 279.Ebert S.M., Dyle M.C., Bullard S.A., Dierdorff J.M., Murry D.J., Fox D.K., Bongers K.S., Lira V.A., Meyerholz D.K., Talley J.J., et al. Identification and Small Molecule Inhibition of an Activating Transcription Factor 4 (ATF4)-dependent Pathway to Age-related Skeletal Muscle Weakness and Atrophy. J. Biol. Chem. 2015;290:25497–25511. doi: 10.1074/jbc.M115.681445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280.Kunkel S.D., Suneja M., Ebert S.M., Bongers K.S., Fox D.K., Malmberg S.E., Alipour F., Shields R.K., Adams C.M. mRNA expression signatures of human skeletal muscle atrophy identify a natural compound that increases muscle mass. Cell Metab. 2011;13:627–638. doi: 10.1016/j.cmet.2011.03.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281.Staats S., Wagner A.E., Luersen K., Kunstner A., Meyer T., Kahns A.K., Derer S., Graspeuntner S., Rupp J., Busch H., et al. Dietary ursolic acid improves health span and life span in male Drosophila melanogaster. BioFactors. 2019;45:169–186. doi: 10.1002/biof.1467. [DOI] [PubMed] [Google Scholar]
- 282.Gill B.S., Kumar S., Navgeet Triterpenes in cancer: Significance and their influence. Mol. Biol. Rep. 2016;43:881–896. doi: 10.1007/s11033-016-4032-9. [DOI] [PubMed] [Google Scholar]
- 283.Lee S.G., Kim M.M. Pachymic acid promotes induction of autophagy related to IGF-1 signaling pathway in WI-38 cells. Phytomedicine. 2017;36:82–87. doi: 10.1016/j.phymed.2017.09.020. [DOI] [PubMed] [Google Scholar]
- 284.Li B., Wu G.L., Dai W., Wang G., Su H.Y., Shen X.P., Zhan R., Xie J.M., Wang Z., Qin Z.H., et al. Aescin-induced reactive oxygen species play a pro-survival role in human cancer cells via ATM/AMPK/ULK1-mediated autophagy. Acta Pharm. Sin. 2018;39:1874–1884. doi: 10.1038/s41401-018-0047-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285.Sohretoglu D., Zhang C., Luo J., Huang S. ReishiMax inhibits mTORC1/2 by activating AMPK and inhibiting IGFR/PI3K/Rheb in tumor cells. Signal. Transduct. Target. 2019;4:21. doi: 10.1038/s41392-019-0056-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286.Wei Q., Zhang B., Li P., Wen X., Yang J. Maslinic Acid Inhibits Colon Tumorigenesis by the AMPK-mTOR Signaling Pathway. J. Agric. Food Chem. 2019;67:4259–4272. doi: 10.1021/acs.jafc.9b00170. [DOI] [PubMed] [Google Scholar]
- 287.Nie H., Wang Y., Qin Y., Gong X.G. Oleanolic acid induces autophagic death in human gastric cancer cells In Vitro and In Vivo. Cell Biol. Int. 2016;40:770–778. doi: 10.1002/cbin.10612. [DOI] [PubMed] [Google Scholar]
- 288.Zhang R., Zhang N., Zhang H., Liu C., Dong X., Wang X., Zhu Y., Xu C., Liu L., Yang S., et al. Celastrol prevents cadmium-induced neuronal cell death by blocking reactive oxygen species-mediated mammalian target of rapamycin pathway. Br. J. Pharm. 2017;174:82–100. doi: 10.1111/bph.13655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289.Zhao P., Wang Y., Zeng S., Lu J., Jiang T.M., Li Y.M. Protective effect of astragaloside IV on lipopolysaccharide-induced cardiac dysfunction via downregulation of inflammatory signaling in mice. Immunopharmacol. Immunotoxicol. 2015;37:428–433. doi: 10.3109/08923973.2015.1080266. [DOI] [PubMed] [Google Scholar]
- 290.Dai Y., Desano J., Tang W., Meng X., Meng Y., Burstein E., Lawrence T.S., Xu L. Natural proteasome inhibitor celastrol suppresses androgen-independent prostate cancer progression by modulating apoptotic proteins and NF-kappaB. PLoS ONE. 2010;5:e14153. doi: 10.1371/journal.pone.0014153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 291.Yoshikawa K., Inoue M., Matsumoto Y., Sakakibara C., Miyataka H., Matsumoto H., Arihara S. Lanostane triterpenoids and triterpene glycosides from the fruit body of Fomitopsis pinicola and their inhibitory activity against COX-1 and COX-2. J. Nat. Prod. 2005;68:69–73. doi: 10.1021/np040130b. [DOI] [PubMed] [Google Scholar]
- 292.Shishodia S., Majumdar S., Banerjee S., Aggarwal B.B. Ursolic acid inhibits nuclear factor-kappaB activation induced by carcinogenic agents through suppression of IkappaBalpha kinase and p65 phosphorylation: Correlation with down-regulation of cyclooxygenase 2, matrix metalloproteinase 9, and cyclin D1. Cancer Res. 2003;63:4375–4383. [PubMed] [Google Scholar]
- 293.Gao J., Tang X., Dou H., Fan Y., Zhao X., Xu Q. Hepatoprotective activity of Terminalia catappa L. leaves and its two triterpenoids. J. Pharm. Pharm. 2004;56:1449–1455. doi: 10.1211/0022357044733. [DOI] [PubMed] [Google Scholar]
- 294.Lee W., Kim J., Park E.K., Bae J.S. Maslinic Acid Ameliorates Inflammation via the Downregulation of NF-kappaB and STAT-1. Antioxidants. 2020;9:106. doi: 10.3390/antiox9020106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 295.Fukumitsu S., Villareal M.O., Fujitsuka T., Aida K., Isoda H. Anti-inflammatory and anti-arthritic effects of pentacyclic triterpenoids maslinic acid through NF-kappaB inactivation. Mol. Nutr. Food Res. 2016;60:399–409. doi: 10.1002/mnfr.201500465. [DOI] [PubMed] [Google Scholar]
- 296.Yang X.W., Ma L.Y., Zhou Q.L., Xu W., Zhang Y.B. SIRT1 activator isolated from artificial gastric juice incubate of total saponins in stems and leaves of Panax ginseng. Bioorg. Med. Chem. Lett. 2018;28:240–243. doi: 10.1016/j.bmcl.2017.12.067. [DOI] [PubMed] [Google Scholar]
- 297.Bakhtiari N., Mirzaie S., Hemmati R., Moslemee-Jalalvand E., Noori A.R., Kazemi J. Mounting evidence validates Ursolic Acid directly activates SIRT1: A powerful STAC which mimic endogenous activator of SIRT1. Arch. Biochem. Biophys. 2018;650:39–48. doi: 10.1016/j.abb.2018.05.012. [DOI] [PubMed] [Google Scholar]
- 298.Bakhtiari N., Hosseinkhani S., Tashakor A., Hemmati R. Ursolic acid ameliorates aging-metabolic phenotype through promoting of skeletal muscle rejuvenation. Med. Hypotheses. 2015;85:1–6. doi: 10.1016/j.mehy.2015.02.014. [DOI] [PubMed] [Google Scholar]
- 299.Sudhahar V., Kumar S.A., Varalakshmi P., Sundarapandiyan R. Mitigating role of lupeol and lupeol linoleate on hepatic lipemic-oxidative injury and lipoprotein peroxidation in experimental hypercholesterolemia. Mol. Cell. Biochem. 2007;295:189–198. doi: 10.1007/s11010-006-9288-2. [DOI] [PubMed] [Google Scholar]
- 300.Asha R., Gayathri Devi V., Abraham A. Lupeol, a pentacyclic triterpenoid isolated from Vernonia cinerea attenuate selenite induced cataract formation in Sprague Dawley rat pups. Chem. Biol. Interact. 2016;245:20–29. doi: 10.1016/j.cbi.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 301.Lin X., Huang R., Zhang S., Wei L., Zhuo L., Wu X., Tang A., Huang Q. Beneficial effects of asiaticoside on cognitive deficits in senescence-accelerated mice. Fitoterapia. 2013;87:69–77. doi: 10.1016/j.fitote.2013.03.023. [DOI] [PubMed] [Google Scholar]
- 302.Laszczyk M.N. Pentacyclic triterpenes of the lupane, oleanane and ursane group as tools in cancer therapy. Planta Med. 2009;75:1549–1560. doi: 10.1055/s-0029-1186102. [DOI] [PubMed] [Google Scholar]
- 303.Prasad S., Madan E., Nigam N., Roy P., George J., Shukla Y. Induction of apoptosis by lupeol in human epidermoid carcinoma A431 cells through regulation of mitochondrial, Akt/PKB and NFkappaB signaling pathways. Cancer Biol. 2009;8:1632–1639. doi: 10.4161/cbt.8.17.9204. [DOI] [PubMed] [Google Scholar]
- 304.De Los Reyes M.M., Oyong G.G., VA S.N., Shen C.C., Ragasa C.Y. Cytotoxic Compounds from Wrightia pubescens (R.Br.) Pharmacogn. Res. 2018;10:9–15. doi: 10.4103/pr.pr_45_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 305.Qu L., Li S., Zhuo Y., Chen J., Qin X., Guo G. Anticancer effect of triterpenes from Ganoderma lucidum in human prostate cancer cells. Oncol. Lett. 2017;14:7467–7472. doi: 10.3892/ol.2017.7153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 306.Valdes K., Morales J., Rodriguez L., Gunther G. Potential use of nanocarriers with pentacyclic triterpenes in cancer treatments. Nanomedicine. 2016;11:3139–3156. doi: 10.2217/nnm-2016-0251. [DOI] [PubMed] [Google Scholar]
- 307.Silva A.M., Alvarado H.L., Abrego G., Martins-Gomes C., Garduno-Ramirez M.L., Garcia M.L., Calpena A.C., Souto E.B. In Vitro Cytotoxicity of Oleanolic/Ursolic Acids-Loaded in PLGA Nanoparticles in Different Cell Lines. Pharmaceutics. 2019;11:362. doi: 10.3390/pharmaceutics11080362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 308.Senthilkumar S., Yogeeta S.K., Subashini R., Devaki T. Attenuation of cyclophosphamide induced toxicity by squalene in experimental rats. Chem. Biol. Interact. 2006;160:252–260. doi: 10.1016/j.cbi.2006.02.004. [DOI] [PubMed] [Google Scholar]
- 309.Senthilkumar S., Devaki T., Manohar B.M., Babu M.S. Effect of squalene on cyclophosphamide-induced toxicity. Clin. Chim. Acta. 2006;364:335–342. doi: 10.1016/j.cca.2005.07.032. [DOI] [PubMed] [Google Scholar]
- 310.Narayan Bhilwade H., Tatewaki N., Konishi T., Nishida M., Eitsuka T., Yasui H., Inanami O., Handa O., Naito Y., Ikekawa N., et al. The Adjuvant Effect of Squalene, an Active Ingredient of Functional Foods, on Doxorubicin-Treated Allograft Mice. Nutr. Cancer. 2019;71:1153–1164. doi: 10.1080/01635581.2019.1597900. [DOI] [PubMed] [Google Scholar]
- 311.Arias J.L., Reddy L.H., Othman M., Gillet B., Desmaele D., Zouhiri F., Dosio F., Gref R., Couvreur P. Squalene based nanocomposites: A new platform for the design of multifunctional pharmaceutical theragnostics. ACS Nano. 2011;5:1513–1521. doi: 10.1021/nn1034197. [DOI] [PubMed] [Google Scholar]
- 312.Bui D.T., Nicolas J., Maksimenko A., Desmaele D., Couvreur P. Multifunctional squalene-based prodrug nanoparticles for targeted cancer therapy. Chem. Commun. (Camb.) 2014;50:5336–5338. doi: 10.1039/C3CC47427E. [DOI] [PubMed] [Google Scholar]
- 313.Kotelevets L., Chastre E., Caron J., Mougin J., Bastian G., Pineau A., Walker F., Lehy T., Desmaele D., Couvreur P. A Squalene-Based Nanomedicine for Oral Treatment of Colon Cancer. Cancer Res. 2017;77:2964–2975. doi: 10.1158/0008-5472.CAN-16-1741. [DOI] [PubMed] [Google Scholar]
- 314.Gaudin A., Yemisci M., Eroglu H., Lepetre-Mouelhi S., Turkoglu O.F., Donmez-Demir B., Caban S., Sargon M.F., Garcia-Argote S., Pieters G., et al. Squalenoyl adenosine nanoparticles provide neuroprotection after stroke and spinal cord injury. Nat. Nanotechnol. 2014;9:1054–1062. doi: 10.1038/nnano.2014.274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 315.Yang L., Zhang J., Zheng K., Shen H., Chen X. Long-term ginsenoside Rg1 supplementation improves age-related cognitive decline by promoting synaptic plasticity associated protein expression in C57BL/6J mice. J. Gerontol. A Biol. Sci. Med. Sci. 2014;69:282–294. doi: 10.1093/gerona/glt091. [DOI] [PubMed] [Google Scholar]
- 316.Zhao H.F., Li Q., Li Y. Long-term ginsenoside administration prevents memory loss in aged female C57BL/6J mice by modulating the redox status and up-regulating the plasticity-related proteins in hippocampus. Neuroscience. 2011;183:189–202. doi: 10.1016/j.neuroscience.2011.03.048. [DOI] [PubMed] [Google Scholar]
- 317.Li N., Zhou L., Li W., Liu Y., Wang J., He P. Protective effects of ginsenosides Rg1 and Rb1 on an Alzheimer’s disease mouse model: A metabolomics study. J. Chromatogr. B Anal. Technol. Biomed. Life Sci. 2015;985:54–61. doi: 10.1016/j.jchromb.2015.01.016. [DOI] [PubMed] [Google Scholar]
- 318.Kaundal M., Akhtar M., Deshmukh R. Lupeol Isolated from Betula alnoides Ameliorates Amyloid Beta Induced Neuronal Damage via Targeting Various Pathological Events and Alteration in Neurotransmitter Levels in Rat’s Brain. J. Neurol. Neurosci. 2017;8:195. doi: 10.21767/2171-6625.1000195. [DOI] [Google Scholar]
- 319.Wei C.C., Chang C.H., Liao V.H. Anti-Parkinsonian effects of beta-amyrin are regulated via LGG-1 involved autophagy pathway in Caenorhabditis elegans. Phytomedicine. 2017;36:118–125. doi: 10.1016/j.phymed.2017.09.002. [DOI] [PubMed] [Google Scholar]
- 320.Farvin K.H., Anandan R., Kumar S.H., Shiny K.S., Mathew S., Sankar T.V., Nair P.G. Cardioprotective effect of squalene on lipid profile in isoprenaline-induced myocardial infarction in rats. J. Med. Food. 2006;9:531–536. doi: 10.1089/jmf.2006.9.531. [DOI] [PubMed] [Google Scholar]
- 321.Liu Y., Xu X., Bi D., Wang X., Zhang X., Dai H., Chen S., Zhang W. Influence of squalene feeding on plasma leptin, testosterone & blood pressure in rats. Indian J. Med. Res. 2009;129:150–153. [PubMed] [Google Scholar]
- 322.Du Y., Ko K.M. Effects of pharmacological preconditioning by emodin/oleanolic acid treatment and/or ischemic preconditioning on mitochondrial antioxidant components as well as the susceptibility to ischemia-reperfusion injury in rat hearts. Mol. Cell. Biochem. 2006;288:135–142. doi: 10.1007/s11010-006-9129-3. [DOI] [PubMed] [Google Scholar]
- 323.Du Y., Ko K.M. Oleanolic acid protects against myocardial ischemia-reperfusion injury by enhancing mitochondrial antioxidant mechanism mediated by glutathione and alpha-tocopherol in rats. Planta Med. 2006;72:222–227. doi: 10.1055/s-2005-916197. [DOI] [PubMed] [Google Scholar]
- 324.Ibrahim N., Fairus S., Zulfarina M.S., Naina Mohamed I. The Efficacy of Squalene in Cardiovascular Disease Risk-A Systematic Review. Nutrients. 2020;12:414. doi: 10.3390/nu12020414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 325.Guan T., Qian Y., Tang X., Huang M., Huang L., Li Y., Sun H. Maslinic acid, a natural inhibitor of glycogen phosphorylase, reduces cerebral ischemic injury in hyperglycemic rats by GLT-1 up-regulation. J. Neurosci. Res. 2011;89:1829–1839. doi: 10.1002/jnr.22671. [DOI] [PubMed] [Google Scholar]
- 326.Ravi Kumar S., Narayan B., Sawada Y., Hosokawa M., Miyashita K. Combined effect of astaxanthin and squalene on oxidative stress in vivo. Mol. Cell. Biochem. 2016;417:57–65. doi: 10.1007/s11010-016-2713-2. [DOI] [PubMed] [Google Scholar]
- 327.Ramu R., Shirahatti P.S., Nanjunda Swamy S., Zameer F., Dhananjaya B.L., Prasad N. Assessment of In Vivo Antidiabetic Properties of Umbelliferone and Lupeol Constituents of Banana (Musa sp. var. Nanjangud Rasa Bale) Flower in Hyperglycaemic Rodent Model. PLoS ONE. 2016;11:e0151135. doi: 10.1371/journal.pone.0151135. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 328.Zarse K., Bossecker A., Muller-Kuhrt L., Siems K., Hernandez M.A., Berendsohn W.G., Birringer M., Ristow M. The phytochemical glaucarubinone promotes mitochondrial metabolism, reduces body fat, and extends lifespan of Caenorhabditis elegans. Horm. Metab. Res. 2011;43:241–243. doi: 10.1055/s-0030-1270524. [DOI] [PubMed] [Google Scholar]
- 329.Wang S., Zhai C., Liu Q., Wang X., Ren Z., Zhang Y., Zhang Y., Wu Q., Sun S., Li S., et al. Cycloastragenol, a triterpene aglycone derived from Radix astragali, suppresses the accumulation of cytoplasmic lipid droplet in 3T3-L1 adipocytes. Biochem. Biophys. Res. Commun. 2014;450:306–311. doi: 10.1016/j.bbrc.2014.05.117. [DOI] [PubMed] [Google Scholar]
- 330.Cao S., Dong X.L., Ho M.X., Yu W.X., Wong K.C., Yao X.S., Wong M.S. Oleanolic Acid Exerts Osteoprotective Effects and Modulates Vitamin D Metabolism. Nutrients. 2018;10:247. doi: 10.3390/nu10020247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 331.Pokorny J., Borkova L., Urban M. Click Reactions in Chemistry of Triterpenes–Advances Towards Development of Potential Therapeutics. Curr. Med. Chem. 2018;25:636–658. doi: 10.2174/0929867324666171009122612. [DOI] [PubMed] [Google Scholar]
- 332.Zhao J., Chen J., Liu T., Fang J., Wan J., Zhao J., Li W., Liu J., Zhao X., Chen S. Anti-viral effects of urosolic acid on guinea pig cytomegalovirus in vitro. J. Huazhong Univ. Sci. Technol. Med. Sci. 2012;32:883–887. doi: 10.1007/s11596-012-1052-0. [DOI] [PubMed] [Google Scholar]
- 333.Kazakova O.B., Giniyatullina G.V., Yamansarov E.Y., Tolstikov G.A. Betulin and ursolic acid synthetic derivatives as inhibitors of Papilloma virus. Bioorg. Med. Chem. Lett. 2010;20:4088–4090. doi: 10.1016/j.bmcl.2010.05.083. [DOI] [PubMed] [Google Scholar]
- 334.Chang C.D., Lin P.Y., Hsu J.L., Shih W.L. Ursolic Acid Suppresses Hepatitis B Virus X Protein-mediated Autophagy and Chemotherapeutic Drug Resistance. Anticancer Res. 2016;36:5097–5107. doi: 10.21873/anticanres.11079. [DOI] [PubMed] [Google Scholar]
- 335.Kong L., Li S., Liao Q., Zhang Y., Sun R., Zhu X., Zhang Q., Wang J., Wu X., Fang X., et al. Oleanolic acid and ursolic acid: Novel hepatitis C virus antivirals that inhibit NS5B activity. Antivir. Res. 2013;98:44–53. doi: 10.1016/j.antiviral.2013.02.003. [DOI] [PubMed] [Google Scholar]
- 336.Jimenez-Arellanes A., Luna-Herrera J., Cornejo-Garrido J., Lopez-Garcia S., Castro-Mussot M.E., Meckes-Fischer M., Mata-Espinosa D., Marquina B., Torres J., Hernandez-Pando R. Ursolic and oleanolic acids as antimicrobial and immunomodulatory compounds for tuberculosis treatment. BMC Complement. Altern. Med. 2013;13:258. doi: 10.1186/1472-6882-13-258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 337.Chang W., Li Y., Zhang L., Cheng A., Liu Y., Lou H. Retigeric acid B enhances the efficacy of azoles combating the virulence and biofilm formation of Candida albicans. Biol. Pharm. Bull. 2012;35:1794–1801. doi: 10.1248/bpb.b12-00511. [DOI] [PubMed] [Google Scholar]
- 338.Mezzomo N., Ferreira S.R.S. Carotenoids Functionality, Sources, and Processing by Supercritical Technology: A Review. J. Chem. 2016;2016:1–16. doi: 10.1155/2016/3164312. [DOI] [Google Scholar]
- 339.Alcaino J., Baeza M., Cifuentes V. Carotenoid Distribution in Nature. Subcell Biochem. 2016;79:3–33. doi: 10.1007/978-3-319-39126-7_1. [DOI] [PubMed] [Google Scholar]
- 340.Rao A.V., Rao L.G. Carotenoids and human health. Pharm. Res. 2007;55:207–216. doi: 10.1016/j.phrs.2007.01.012. [DOI] [PubMed] [Google Scholar]
- 341.O’Neill M.E., Carroll Y., Corridan B., Olmedilla B., Granado F., Blanco I., Van den Berg H., Hininger I., Rousell A.M., Chopra M., et al. A European carotenoid database to assess carotenoid intakes and its use in a five-country comparative study. Br. J. Nutr. 2001;85:499–507. doi: 10.1079/BJN2000284. [DOI] [PubMed] [Google Scholar]
- 342.Perry A., Rasmussen H., Johnson E.J. Xanthophyll (lutein, zeaxanthin) content in fruits, vegetables and corn and egg products. J. Food Compos. Anal. 2009;22:9–15. doi: 10.1016/j.jfca.2008.07.006. [DOI] [Google Scholar]
- 343.Lashmanova E.A., Kuzivanova O.A., Dymova O.V., Moskalev A.A. The Effects of Cloudberry Fruit Extract on Drosophila melanogaster Lifespan and Stress Resistance. Adv. Gerontol. 2019;9:254–260. doi: 10.1134/S2079057019020127. [DOI] [PubMed] [Google Scholar]
- 344.Weinrich T., Xu Y., Wosu C., Harvey P.J., Jeffery G. Mitochondrial Function, Mobility and Lifespan Are Improved in Drosophila melanogaster by Extracts of 9-cis-beta-Carotene from Dunaliella salina. Mar. Drugs. 2019;17:279. doi: 10.3390/md17050279. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 345.Hu W., Dai D., Li W. Extension of Life Span and Improvement of Vitality in Drosophila melanogaster by Supplementation with Lycopene Mycelium Powder from Blakeslea Trispora. Food Sci. Technol. Res. 2013;19:513–518. doi: 10.3136/fstr.19.513. [DOI] [Google Scholar]
- 346.Lashmanova E., Proshkina E., Zhikrivetskaya S., Shevchenko O., Marusich E., Leonov S., Melerzanov A., Zhavoronkov A., Moskalev A. Fucoxanthin increases lifespan of Drosophila melanogaster and Caenorhabditis Elegans. Pharm. Res. 2015;100:228–241. doi: 10.1016/j.phrs.2015.08.009. [DOI] [PubMed] [Google Scholar]
- 347.Massie H.R., Ferreira J.R., Jr., DeWolfe L.K. Effect of dietary beta-carotene on the survival of young and old mice. Gerontology. 1986;32:189–195. doi: 10.1159/000212788. [DOI] [PubMed] [Google Scholar]
- 348.Hoffman R., Sultan L.D., Saada A., Hirschberg J., Osterzetser-Biran O., Gruenbaum Y. Astaxanthin extends lifespan via altered biogenesis of the mitochondrial respiratory chain complex III. Biorxiv. 2019 doi: 10.1101/698001. [DOI] [Google Scholar]
- 349.Nicoll A., Locarnini S. Review: Present and future directions in the treatment of chronic hepatitis B infection. J. Gastroenterol. Hepatol. 1997;12:843–854. doi: 10.1111/j.1440-1746.1997.tb00382.x. [DOI] [PubMed] [Google Scholar]
- 350.SJ S., Veerabhadrappa B., Subramaniyan S., Dyavaiah M. Astaxanthin enhances the longevity of Saccharomyces cerevisiae by decreasing oxidative stress and apoptosis. FEMS Yeast Res. 2019:19. doi: 10.1093/femsyr/foy113. [DOI] [PubMed] [Google Scholar]
- 351.Yazaki K., Yoshikoshi C., Oshiro S., Yanase S. Supplemental cellular protection by a carotenoid extends lifespan via Ins/IGF-1 signaling in Caenorhabditis elegans. Oxid. Med. Cell. Longev. 2011;2011:596240. doi: 10.1155/2011/596240. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 352.Dhinaut J., Balourdet A., Teixeira M., Chogne M., Moret Y. A dietary carotenoid reduces immunopathology and enhances longevity through an immune depressive effect in an insect model. Sci. Rep. 2017;7:12429. doi: 10.1038/s41598-017-12769-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 353.Zhang Z., Han S., Wang H., Wang T. Lutein extends the lifespan of Drosophila melanogaster. Arch. Gerontol. Geriatr. 2014;58:153–159. doi: 10.1016/j.archger.2013.07.007. [DOI] [PubMed] [Google Scholar]
- 354.Guvatova Z., Dalina A., Marusich E., Pudova E., Kobelyatskaya A., Krasnov G., Kudryavtseva A., Leonov S., Moskalev A. Protective effects of carotenoid fucoxanthin in fibroblasts cellular senescence. Mech. Ageing Dev. 2020:111260. doi: 10.1016/j.mad.2020.111260. [DOI] [PubMed] [Google Scholar]
- 355.Dambroise E., Monnier L., Ruisheng L., Aguilaniu H., Joly J.S., Tricoire H., Rera M. Two phases of aging separated by the Smurf transition as a public path to death. Sci. Rep. 2016;6:23523. doi: 10.1038/srep23523. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 356.Gil P., Farinas F., Casado A., Lopez-Fernandez E. Malondialdehyde: A possible marker of ageing. Gerontology. 2002;48:209–214. doi: 10.1159/000058352. [DOI] [PubMed] [Google Scholar]
- 357.Koh K., Evans J.M., Hendricks J.C., Sehgal A. A Drosophila model for age-associated changes in sleep:wake cycles. Proc. Natl. Acad. Sci. USA. 2006;103:13843–13847. doi: 10.1073/pnas.0605903103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 358.Mander B.A., Winer J.R., Walker M.P. Sleep and Human Aging. Neuron. 2017;94:19–36. doi: 10.1016/j.neuron.2017.02.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 359.Burton G.W., Ingold K.U. beta-Carotene: An unusual type of lipid antioxidant. Science. 1984;224:569–573. doi: 10.1126/science.6710156. [DOI] [PubMed] [Google Scholar]
- 360.Albanes D., Heinonen O.P., Huttunen J.K., Taylor P.R., Virtamo J., Edwards B.K., Haapakoski J., Rautalahti M., Hartman A.M., Palmgren J. Effects of alpha-tocopherol and beta-carotene supplements on cancer incidence in the Alpha-Tocopherol Beta-Carotene Cancer Prevention Study. Am. J. Clin. Nutr. 1995;62:1427S–1430S. doi: 10.1093/ajcn/62.6.1427S. [DOI] [PubMed] [Google Scholar]
- 361.Bjelakovic G., Nikolova D., Gluud L.L., Simonetti R.G., Gluud C. Antioxidant supplements for prevention of mortality in healthy participants and patients with various diseases. Cochrane Database Syst. Rev. 2012 doi: 10.1002/14651858.CD007176.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 362.Torregrosa-Crespo J., Montero Z., Fuentes J.L., Reig Garcia-Galbis M., Garbayo I., Vilchez C., Martinez-Espinosa R.M. Exploring the Valuable Carotenoids for the Large-Scale Production by Marine Microorganisms. Mar. Drugs. 2018;16:203. doi: 10.3390/md16060203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 363.Mortensen A. Carotenoids and other pigments as natural colorants. Pure Appl. Chem. 2006;78:1477–1491. doi: 10.1351/pac200678081477. [DOI] [Google Scholar]
- 364.Yeum K.-J., Aldini G., Russell R.M., Krinsky N.I. Antioxidant/Pro-oxidant Actions of Carotenoids. In: Britton G., Pfander H., Liaaen-Jensen S., editors. Carotenoids: Volume 5: Nutrition and Health. Birkhäuser Basel; Basel, Switzerland: 2009. pp. 235–268. [DOI] [Google Scholar]
- 365.Kaulmann A., Bohn T. Carotenoids, inflammation, and oxidative stress—Implications of cellular signaling pathways and relation to chronic disease prevention. Nutr. Res. 2014;34:907–929. doi: 10.1016/j.nutres.2014.07.010. [DOI] [PubMed] [Google Scholar]
- 366.Ben-Dor A., Steiner M., Gheber L., Danilenko M., Dubi N., Linnewiel K., Zick A., Sharoni Y., Levy J. Carotenoids activate the antioxidant response element transcription system. Mol. Cancer. 2005;4:177–186. [PubMed] [Google Scholar]
- 367.Liu C.L., Chiu Y.T., Hu M.L. Fucoxanthin enhances HO-1 and NQO1 expression in murine hepatic BNL CL.2 cells through activation of the Nrf2/ARE system partially by its pro-oxidant activity. J. Agric. Food Chem. 2011;59:11344–11351. doi: 10.1021/jf2029785. [DOI] [PubMed] [Google Scholar]
- 368.Bai S.K., Lee S.J., Na H.J., Ha K.S., Han J.A., Lee H., Kwon Y.G., Chung C.K., Kim Y.M. beta-Carotene inhibits inflammatory gene expression in lipopolysaccharide-stimulated macrophages by suppressing redox-based NF-kappaB activation. Exp. Mol. Med. 2005;37:323–334. doi: 10.1038/emm.2005.42. [DOI] [PubMed] [Google Scholar]
- 369.Li R., Hong P., Zheng X. beta-carotene attenuates lipopolysaccharide-induced inflammation via inhibition of the NF-kappaB, JAK2/STAT3 and JNK/p38 MAPK signaling pathways in macrophages. Anim. Sci. J. 2019;90:140–148. doi: 10.1111/asj.13108. [DOI] [PubMed] [Google Scholar]
- 370.Liu C., Russell R.M., Wang X.D. Low dose beta-carotene supplementation of ferrets attenuates smoke-induced lung phosphorylation of JNK, p38 MAPK, and p53 proteins. J. Nutr. 2004;134:2705–2710. doi: 10.1093/jn/134.10.2705. [DOI] [PubMed] [Google Scholar]
- 371.Satomi Y., Nishino H. Implication of mitogen-activated protein kinase in the induction of G1 cell cycle arrest and gadd45 expression by the carotenoid fucoxanthin in human cancer cells. Biochim. Biophys. Acta. 2009;1790:260–266. doi: 10.1016/j.bbagen.2009.01.003. [DOI] [PubMed] [Google Scholar]
- 372.Satomi Y. Fucoxanthin induces GADD45A expression and G1 arrest with SAPK/JNK activation in LNCap human prostate cancer cells. Anticancer Res. 2012;32:807–813. [PubMed] [Google Scholar]
- 373.Plyusnina E.N., Shaposhnikov M.V., Moskalev A.A. Increase of Drosophila melanogaster lifespan due to D-GADD45 overexpression in the nervous system. Biogerontology. 2011;12:211–226. doi: 10.1007/s10522-010-9311-6. [DOI] [PubMed] [Google Scholar]
- 374.Fiedor J., Burda K. Potential role of carotenoids as antioxidants in human health and disease. Nutrients. 2014;6:466–488. doi: 10.3390/nu6020466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 375.Kumar S.R., Hosokawa M., Miyashita K. Fucoxanthin: A marine carotenoid exerting anti-cancer effects by affecting multiple mechanisms. Mar. Drugs. 2013;11:5130–5147. doi: 10.3390/md11125130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 376.Tanaka T., Shnimizu M., Moriwaki H. Cancer chemoprevention by carotenoids. Molecules. 2012;17:3202–3242. doi: 10.3390/molecules17033202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 377.Pangestuti R., Vo T.S., Ngo D.H., Kim S.K. Fucoxanthin ameliorates inflammation and oxidative reponses in microglia. J. Agric. Food Chem. 2013;61:3876–3883. doi: 10.1021/jf400015k. [DOI] [PubMed] [Google Scholar]
- 378.Mounien L., Tourniaire F., Landrier J.F. Anti-Obesity Effect of Carotenoids: Direct Impact on Adipose Tissue and Adipose Tissue-Driven Indirect Effects. Nutrients. 2019;11:1562. doi: 10.3390/nu11071562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 379.Akbaraly N.T., Faure H., Gourlet V., Favier A., Berr C. Plasma carotenoid levels and cognitive performance in an elderly population: Results of the EVA Study. J. Gerontol. A Biol. Sci. Med. Sci. 2007;62:308–316. doi: 10.1093/gerona/62.3.308. [DOI] [PubMed] [Google Scholar]
- 380.Johnson E.J., Vishwanathan R., Johnson M.A., Hausman D.B., Davey A., Scott T.M., Green R.C., Miller L.S., Gearing M., Woodard J., et al. Relationship between Serum and Brain Carotenoids, alpha-Tocopherol, and Retinol Concentrations and Cognitive Performance in the Oldest Old from the Georgia Centenarian Study. J. Aging Res. 2013;2013:951786. doi: 10.1155/2013/951786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 381.Johnson E.J., McDonald K., Caldarella S.M., Chung H.Y., Troen A.M., Snodderly D.M. Cognitive findings of an exploratory trial of docosahexaenoic acid and lutein supplementation in older women. Nutr. Neurosci. 2008;11:75–83. doi: 10.1179/147683008X301450. [DOI] [PubMed] [Google Scholar]
- 382.Perrig W.J., Perrig P., Stahelin H.B. The relation between antioxidants and memory performance in the old and very old. J. Am. Geriatr. Soc. 1997;45:718–724. doi: 10.1111/j.1532-5415.1997.tb01476.x. [DOI] [PubMed] [Google Scholar]
- 383.Takeda A., Nyssen O.P., Syed A., Jansen E., Bueno-de-Mesquita B., Gallo V. Vitamin A and carotenoids and the risk of Parkinson’s disease: A systematic review and meta-analysis. Neuroepidemiology. 2014;42:25–38. doi: 10.1159/000355849. [DOI] [PubMed] [Google Scholar]
- 384.Li F.J., Shen L., Ji H.F. Dietary intakes of vitamin E, vitamin C, and beta-carotene and risk of Alzheimer’s disease: A meta-analysis. J. Alzheimers Dis. 2012;31:253–258. doi: 10.3233/JAD-2012-120349. [DOI] [PubMed] [Google Scholar]
- 385.Voutilainen S., Nurmi T., Mursu J., Rissanen T.H. Carotenoids and cardiovascular health. Am. J. Clin. Nutr. 2006;83:1265–1271. doi: 10.1093/ajcn/83.6.1265. [DOI] [PubMed] [Google Scholar]
- 386.Kulczyński B., Gramza-Michałowska A., Kobus-Cisowska J., Kmiecik D. The role of carotenoids in the prevention and treatment of cardiovascular disease—Current state of knowledge. J. Funct. Foods. 2017;38:45–65. doi: 10.1016/j.jff.2017.09.001. [DOI] [Google Scholar]
- 387.Sluijs I., Cadier E., Beulens J.W., van der A.D., Spijkerman A.M., van der Schouw Y.T. Dietary intake of carotenoids and risk of type 2 diabetes. Nutr. Metab. Cardiovasc. Dis. 2015;25:376–381. doi: 10.1016/j.numecd.2014.12.008. [DOI] [PubMed] [Google Scholar]
- 388.Montonen J., Knekt P., Jarvinen R., Reunanen A. Dietary antioxidant intake and risk of type 2 diabetes. Diabetes Care. 2004;27:362–366. doi: 10.2337/diacare.27.2.362. [DOI] [PubMed] [Google Scholar]
- 389.Arnlov J., Zethelius B., Riserus U., Basu S., Berne C., Vessby B., Alfthan G., Helmersson J. Serum and dietary beta-carotene and alpha-tocopherol and incidence of type 2 diabetes mellitus in a community-based study of Swedish men: Report from the Uppsala Longitudinal Study of Adult Men (ULSAM) study. Diabetologia. 2009;52:97–105. doi: 10.1007/s00125-008-1189-3. [DOI] [PubMed] [Google Scholar]
- 390.Coyne T., Ibiebele T.I., Baade P.D., Dobson A., McClintock C., Dunn S., Leonard D., Shaw J. Diabetes mellitus and serum carotenoids: Findings of a population-based study in Queensland, Australia. Am. J. Clin. Nutr. 2005;82:685–693. doi: 10.1093/ajcn/82.3.685. [DOI] [PubMed] [Google Scholar]
- 391.Sugiura M., Nakamura M., Ogawa K., Ikoma Y., Yano M. High-serum carotenoids associated with lower risk for developing type 2 diabetes among Japanese subjects: Mikkabi cohort study. BMJ Open Diabetes Res. Care. 2015;3:e000147. doi: 10.1136/bmjdrc-2015-000147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 392.Arteni A.A., Fradot M., Galzerano D., Mendes-Pinto M.M., Sahel J.A., Picaud S., Robert B., Pascal A.A. Structure and Conformation of the Carotenoids in Human Retinal Macular Pigment. PLoS ONE. 2015;10:e0135779. doi: 10.1371/journal.pone.0135779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 393.Buscemi S., Corleo D., Di Pace F., Petroni M.L., Satriano A., Marchesini G. The Effect of Lutein on Eye and Extra-Eye Health. Nutrients. 2018;10:1321. doi: 10.3390/nu10091321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 394.Hu B.J., Hu Y.N., Lin S., Ma W.J., Li X.R. Application of Lutein and Zeaxanthin in nonproliferative diabetic retinopathy. Int. J. Ophthalmol. 2011;4:303–306. doi: 10.3980/j.issn.2222-3959.2011.03.19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 395.Moschos M.M., Dettoraki M., Tsatsos M., Kitsos G., Kalogeropoulos C. Effect of carotenoids dietary supplementation on macular function in diabetic patients. Eye Vis. (Lond.) 2017;4:23. doi: 10.1186/s40662-017-0088-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 396.Yamaguchi M. Role of carotenoid beta-cryptoxanthin in bone homeostasis. J. Biomed. Sci. 2012;19:36. doi: 10.1186/1423-0127-19-36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 397.Kim L., Rao A.V., Rao L.G. Lycopene II—Effect on osteoblasts: The carotenoid lycopene stimulates cell proliferation and alkaline phosphatase activity of SaOS-2 cells. J. Med. Food. 2003;6:79–86. doi: 10.1089/109662003322233468. [DOI] [PubMed] [Google Scholar]
- 398.Virdee S.S., Thomas M.B. A practitioner’s guide to gutta-percha removal during endodontic retreatment. Br. Dent. J. 2017;222:251–257. doi: 10.1038/sj.bdj.2017.166. [DOI] [PubMed] [Google Scholar]
- 399.Belladonna F.G., Calasans-Maia M.D., Novellino Alves A.T., de Brito Resende R.F., Souza E.M., Silva E.J., Fidel S.R., De-Deus G. Biocompatibility of a self-adhesive gutta-percha-based material in subcutaneous tissue of mice. J. Endod. 2014;40:1869–1873. doi: 10.1016/j.joen.2014.07.013. [DOI] [PubMed] [Google Scholar]
- 400.Marending M., Bubenhofer S.B., Sener B., De-Deus G. Primary assessment of a self-adhesive gutta-percha material. Int. Endod. J. 2013;46:317–322. doi: 10.1111/j.1365-2591.2012.02117.x. [DOI] [PubMed] [Google Scholar]
- 401.Shanahan D.J., Duncan H.F. Root canal filling using Resilon: A review. Br. Dent. J. 2011;211:81–88. doi: 10.1038/sj.bdj.2011.573. [DOI] [PubMed] [Google Scholar]
- 402.Yuan F., Qian M.C. Development of C13-norisoprenoids, carotenoids and other volatile compounds in Vitis vinifera L. Cv. Pinot noir grapes. Food Chem. 2016;192:633–641. doi: 10.1016/j.foodchem.2015.07.050. [DOI] [PubMed] [Google Scholar]
- 403.Crupi P., Coletta A., Antonacci D. Analysis of carotenoids in grapes to predict norisoprenoid varietal aroma of wines from Apulia. J. Agric. Food Chem. 2010;58:9647–9656. doi: 10.1021/jf100564v. [DOI] [PubMed] [Google Scholar]
- 404.Wirth J., Guo W., Baumes R., Gunata Z. Volatile compounds released by enzymatic hydrolysis of glycoconjugates of leaves and grape berries from Vitis vinifera Muscat of Alexandria and Shiraz cultivars. J. Agric. Food Chem. 2001;49:2917–2923. doi: 10.1021/jf001398l. [DOI] [PubMed] [Google Scholar]
- 405.Song J., Shellie K.C., Wang H., Qian M.C. Influence of deficit irrigation and kaolin particle film on grape composition and volatile compounds in Merlot grape (Vitis vinifera L.) Food Chem. 2012;134:841–850. doi: 10.1016/j.foodchem.2012.02.193. [DOI] [PubMed] [Google Scholar]
- 406.Mathieu S., Terrier N., Procureur J., Bigey F., Gunata Z. A carotenoid cleavage dioxygenase from Vitis vinifera L.: Functional characterization and expression during grape berry development in relation to C13-norisoprenoid accumulation. J. Exp. Bot. 2005;56:2721–2731. doi: 10.1093/jxb/eri265. [DOI] [PubMed] [Google Scholar]
- 407.Ishikura H., Mochizuki T., Izumi Y., Usui T., Sawada H., Uchino H. Differentiation of mouse leukemic M1 cells induced by polyprenoids. Leuk Res. 1984;8:843–852. doi: 10.1016/0145-2126(84)90105-X. [DOI] [PubMed] [Google Scholar]
- 408.Sakagami H., Hashimoto K., Suzuki F., Ishihara M., Kikuchi H., Katayama T., Satoh K. Tumor-specificity and type of cell death induced by vitamin K2 derivatives and prenylalcohols. Anticancer Res. 2008;28:151–158. [PubMed] [Google Scholar]
- 409.Hsieh C.L., Tseng M.H., Shao Y.Y., Chang J.Y., Kuo C.C., Chang C.Y., Kuo Y.H. C35 terpenoids from the bark of Calocedrus macrolepis var. formosana with activity against human cancer cell lines. J. Nat. Prod. 2006;69:1611–1613. doi: 10.1021/np0600550. [DOI] [PubMed] [Google Scholar]
- 410.Zwenger S., Basu C. Plant terpenoids: Applications and future potentials. Biotechnol. Mol. Biol. Rev. 2008;3:1. [Google Scholar]
- 411.Cox-Georgian D., Ramadoss N., Dona C., Basu C. Therapeutic and Medicinal Uses of Terpenes. Med. Plants. 2019 doi: 10.1007/978-3-030-31269-5_15. [DOI] [Google Scholar]
- 412.Dönertaş H.M., Fuentealba M., Partridge L., Thornton J.M. Identifying Potential Ageing-Modulating Drugs In Silico. Trends Endocrinol. Metab. 2019;30:118–131. doi: 10.1016/j.tem.2018.11.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 413.Cruz G.S., Wanderley-Teixeira V., Oliveira J.V., CG D.A., Cunha F.M., Teixeira A.A.C., Guedes C.A., Dutra K.A., Barbosa D.R.S., Breda M.O. Effect of trans-anethole, limonene and your combination in nutritional components and their reflection on reproductive parameters and testicular apoptosis in Spodoptera frugiperda (Lepidoptera: Noctuidae) Chem. Biol. Interact. 2017;263:74–80. doi: 10.1016/j.cbi.2016.12.013. [DOI] [PubMed] [Google Scholar]
- 414.Gray C.A., Runyon J.B., Jenkins M.J., Giunta A.D. Mountain Pine Beetles Use Volatile Cues to Locate Host Limber Pine and Avoid Non-Host Great Basin Bristlecone Pine. PLoS ONE. 2015;10:e0135752. doi: 10.1371/journal.pone.0135752. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 415.Tang X.P., Guo X.H., Geng D., Weng L.J. d-Limonene protects PC12 cells against corticosterone-induced neurotoxicity by activating the AMPK pathway. Environ. Toxicol. Pharm. 2019;70:103192. doi: 10.1016/j.etap.2019.05.001. [DOI] [PubMed] [Google Scholar]
- 416.Miller J.A., Lang J.E., Ley M., Nagle R., Hsu C.H., Thompson P.A., Cordova C., Waer A., Chow H.H. Human breast tissue disposition and bioactivity of limonene in women with early-stage breast cancer. Cancer Prev. Res. 2013;6:577–584. doi: 10.1158/1940-6207.CAPR-12-0452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 417.Rehman M.U., Tahir M., Khan A.Q., Khan R., Oday O.H., Lateef A., Hassan S.K., Rashid S., Ali N., Zeeshan M., et al. D-limonene suppresses doxorubicin-induced oxidative stress and inflammation via repression of COX-2, iNOS, and NFkappaB in kidneys of Wistar rats. Exp. Biol. Med. (Maywood) 2014;239:465–476. doi: 10.1177/1535370213520112. [DOI] [PubMed] [Google Scholar]
- 418.Lindsey L.P., Daphney C.M., Oppong-Damoah A., Uchakin P.N., Abney S.E., Uchakina O.N., Khusial R.D., Akil A., Murnane K.S. The cannabinoid receptor 2 agonist, beta-caryophyllene, improves working memory and reduces circulating levels of specific proinflammatory cytokines in aged male mice. Behav. Brain Res. 2019;372:112012. doi: 10.1016/j.bbr.2019.112012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 419.Chavez-Hurtado P., Gonzalez-Castaneda R.E., Beas-Zarate C., Flores-Soto M.E., Viveros-Paredes J.M. beta-Caryophyllene Reduces DNA Oxidation and the Overexpression of Glial Fibrillary Acidic Protein in the Prefrontal Cortex and Hippocampus of d-Galactose-Induced Aged BALB/c Mice. J. Med. Food. 2020;23:515–522. doi: 10.1089/jmf.2019.0111. [DOI] [PubMed] [Google Scholar]
- 420.Srivastava S., Pant A., Trivedi S., Pandey R. Curcumin and beta-caryophellene attenuate cadmium quantum dots induced oxidative stress and lethality in Caenorhabditis elegans model system. Environ. Toxicol. Pharm. 2016;42:55–62. doi: 10.1016/j.etap.2016.01.001. [DOI] [PubMed] [Google Scholar]
- 421.Li H., Wang D., Chen Y., Yang M. beta-Caryophyllene inhibits high glucose-induced oxidative stress, inflammation and extracellular matrix accumulation in mesangial cells. Int. Immunopharmacol. 2020;84:106556. doi: 10.1016/j.intimp.2020.106556. [DOI] [PubMed] [Google Scholar]
- 422.Younis N.S., Mohamed M.E. beta-Caryophyllene as a Potential Protective Agent Against Myocardial Injury: The Role of Toll-Like Receptors. Molecules. 2019;24:1929. doi: 10.3390/molecules24101929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 423.Youssef D.A., El-Fayoumi H.M., Mahmoud M.F. Beta-caryophyllene alleviates diet-induced neurobehavioral changes in rats: The role of CB2 and PPAR-gamma receptors. Biomed. Pharm. 2019;110:145–154. doi: 10.1016/j.biopha.2018.11.039. [DOI] [PubMed] [Google Scholar]
- 424.Fidyt K., Fiedorowicz A., Strzadala L., Szumny A. beta-caryophyllene and beta-caryophyllene oxide-natural compounds of anticancer and analgesic properties. Cancer Med. 2016;5:3007–3017. doi: 10.1002/cam4.816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 425.Park N.H., Kang Y.G., Kim S.H., Bae I.H., Lee S.H., Kim D.Y., Hwang J.S., Kim Y.J., Lee T.R., Lee E.S. Dehydroabietic Acid Induces Regeneration of Collagen Fibers in Ultraviolet B-Irradiated Human Dermal Fibroblasts and Skin Equivalents. Ski. Pharmacol. Physiol. 2019;32:109–116. doi: 10.1159/000497103. [DOI] [PubMed] [Google Scholar]
- 426.Kamaya Y., Tokita N., Suzuki K. Effects of dehydroabietic acid and abietic acid on survival, reproduction, and growth of the crustacean Daphnia magna. Ecotoxicol. Environ. Saf. 2005;61:83–88. doi: 10.1016/j.ecoenv.2004.07.007. [DOI] [PubMed] [Google Scholar]
- 427.Xing L., Gutierrez-Villagomez J.M., Da Fonte D.F., Venables M.J., Trudeau V.L. Dehydroabietic acid cytotoxicity in goldfish radial glial cells In Vitro. Aquat. Toxicol. 2016;180:78–83. doi: 10.1016/j.aquatox.2016.09.009. [DOI] [PubMed] [Google Scholar]
- 428.Martin D., Rojo A.I., Salinas M., Diaz R., Gallardo G., Alam J., De Galarreta C.M., Cuadrado A. Regulation of heme oxygenase-1 expression through the phosphatidylinositol 3-kinase/Akt pathway and the Nrf2 transcription factor in response to the antioxidant phytochemical carnosol. J. Biol. Chem. 2004;279:8919–8929. doi: 10.1074/jbc.M309660200. [DOI] [PubMed] [Google Scholar]
- 429.Satoh T., McKercher S.R., Lipton S.A. Nrf2/ARE-mediated antioxidant actions of pro-electrophilic drugs. Free Radic. Biol. Med. 2013;65:645–657. doi: 10.1016/j.freeradbiomed.2013.07.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 430.Shi W., Xu G., Zhan X., Gao Y., Wang Z., Fu S., Qin N., Hou X., Ai Y., Wang C., et al. Carnosol inhibits inflammasome activation by directly targeting HSP90 to treat inflammasome-mediated diseases. Cell Death Dis. 2020;11:252. doi: 10.1038/s41419-020-2460-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 431.Samarghandian S., Azimi-Nezhad M., Borji A., Samini M., Farkhondeh T. Protective effects of carnosol against oxidative stress induced brain damage by chronic stress in rats. BMC Complement. Altern. Med. 2017;17:249. doi: 10.1186/s12906-017-1753-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 432.Lian K.C., Chuang J.J., Hsieh C.W., Wung B.S., Huang G.D., Jian T.Y., Sun Y.W. Dual mechanisms of NF-kappaB inhibition in carnosol-treated endothelial cells. Toxicol. Appl. Pharm. 2010;245:21–35. doi: 10.1016/j.taap.2010.01.003. [DOI] [PubMed] [Google Scholar]
- 433.Satoh T., Lipton S. Recent advances in understanding NRF2 as a druggable target: Development of pro-electrophilic and non-covalent NRF2 activators to overcome systemic side effects of electrophilic drugs like dimethyl fumarate. F1000Research. 2017;6:2138. doi: 10.12688/f1000research.12111.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 434.Lipton S.A., Rezaie T., Nutter A., Lopez K.M., Parker J., Kosaka K., Satoh T., McKercher S.R., Masliah E., Nakanishi N. Therapeutic advantage of pro-electrophilic drugs to activate the Nrf2/ARE pathway in Alzheimer’s disease models. Cell Death Dis. 2016;7:e2499. doi: 10.1038/cddis.2016.389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 435.Farr S.A., Niehoff M.L., Ceddia M.A., Herrlinger K.A., Lewis B.J., Feng S., Welleford A., Butterfield D.A., Morley J.E. Effect of botanical extracts containing carnosic acid or rosmarinic acid on learning and memory in SAMP8 mice. Physiol. Behav. 2016;165:328–338. doi: 10.1016/j.physbeh.2016.08.013. [DOI] [PubMed] [Google Scholar]
- 436.Taram F., Ignowski E., Duval N., Linseman D.A. Neuroprotection Comparison of Rosmarinic Acid and Carnosic Acid in Primary Cultures of Cerebellar Granule Neurons. Molecules. 2018;23:2956. doi: 10.3390/molecules23112956. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 437.Ishitobi H., Sanada Y., Kato Y., Ikuta Y., Shibata S., Yamasaki S., Lotz M.K., Matsubara K., Miyaki S., Adachi N. Carnosic acid attenuates cartilage degeneration through induction of heme oxygenase-1 in human articular chondrocytes. Eur. J. Pharm. 2018;830:1–8. doi: 10.1016/j.ejphar.2018.04.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 438.Ramos-Hryb A.B., Pazini F.L., Kaster M.P., Rodrigues A.L.S. Therapeutic Potential of Ursolic Acid to Manage Neurodegenerative and Psychiatric Diseases. CNS Drugs. 2017;31:1029–1041. doi: 10.1007/s40263-017-0474-4. [DOI] [PubMed] [Google Scholar]
- 439.Zhou J.X., Wink M. Evidence for Anti-Inflammatory Activity of Isoliquiritigenin, 18beta Glycyrrhetinic Acid, Ursolic Acid, and the Traditional Chinese Medicine Plants Glycyrrhiza glabra and Eriobotrya japonica, at the Molecular Level. Medicine. 2019;6:55. doi: 10.3390/medicines6020055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 440.Gharibi S., Bakhtiari N., Elham Moslemee J., Bakhtiari F. Ursolic Acid Mediates Hepatic Protection through Enhancing of anti-aging Biomarkers. Curr. Aging Sci. 2018;11:16–23. doi: 10.2174/1874609810666170531103140. [DOI] [PubMed] [Google Scholar]
- 441.Geerlofs L., He Z., Xiao S., Xiao Z.C. Repeated dose (90 days) oral toxicity study of ursolic acid in Han-Wistar rats. Toxicol. Rep. 2020;7:610–623. doi: 10.1016/j.toxrep.2020.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 442.Mlala S., Oyedeji A.O., Gondwe M., Oyedeji O.O. Ursolic Acid and Its Derivatives as Bioactive Agents. Molecules. 2019;24:2751. doi: 10.3390/molecules24152751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 443.Deng S., Shanmugam M.K., Kumar A.P., Yap C.T., Sethi G., Bishayee A. Targeting autophagy using natural compounds for cancer prevention and therapy. Cancer. 2019;125:1228–1246. doi: 10.1002/cncr.31978. [DOI] [PubMed] [Google Scholar]
- 444.Lu Y.F., Wan X.L., Xu Y., Liu J. Repeated oral administration of oleanolic acid produces cholestatic liver injury in mice. Molecules. 2013;18:3060–3071. doi: 10.3390/molecules18033060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 445.Zhao H., Liu J., Song L., Liu Z., Han G., Yuan D., Wang T., Dun Y., Zhou Z., Liu Z., et al. Oleanolic acid rejuvenates testicular function through attenuating germ cell DNA damage and apoptosis via deactivation of NF-kappaB, p53 and p38 signalling pathways. J. Pharm. Pharm. 2017;69:295–304. doi: 10.1111/jphp.12668. [DOI] [PubMed] [Google Scholar]
- 446.Rodriguez-Rodriguez R. Oleanolic acid and related triterpenoids from olives on vascular function: Molecular mechanisms and therapeutic perspectives. Curr. Med. Chem. 2015;22:1414–1425. doi: 10.2174/0929867322666141212122921. [DOI] [PubMed] [Google Scholar]
- 447.Fernandez-Aparicio A., Schmidt-RioValle J., Perona J.S., Correa-Rodriguez M., Castellano J.M., Gonzalez-Jimenez E. Potential Protective Effect of Oleanolic Acid on the Components of Metabolic Syndrome: A Systematic Review. J. Clin. Med. 2019;8:1294. doi: 10.3390/jcm8091294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 448.Ayeleso T.B., Matumba M.G., Mukwevho E. Oleanolic Acid and Its Derivatives: Biological Activities and Therapeutic Potential in Chronic Diseases. Molecules. 2017;22:1915. doi: 10.3390/molecules22111915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 449.Sanchez-Gonzalez M., Lozano-Mena G., Juan M.E., Garcia-Granados A., Planas J.M. Assessment of the safety of maslinic acid, a bioactive compound from Olea europaea L. Mol. Nutr. Food Res. 2013;57:339–346. doi: 10.1002/mnfr.201200481. [DOI] [PubMed] [Google Scholar]
- 450.Cheng X., Qiu L., Wang F. 18alpha-Glycyrrhetinic acid (GA) ameliorates fructose-induced nephropathy in mice by suppressing oxidative stress, dyslipidemia and inflammation. Biomed. Pharm. 2020;125:109702. doi: 10.1016/j.biopha.2019.109702. [DOI] [PubMed] [Google Scholar]
- 451.Lefaki M., Papaevgeniou N., Tur J.A., Vorgias C.E., Sykiotis G.P., Chondrogianni N. The dietary triterpenoid 18alpha-Glycyrrhetinic acid protects from MMC-induced genotoxicity through the ERK/Nrf2 pathway. Redox Biol. 2020;28:101317. doi: 10.1016/j.redox.2019.101317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 452.Ikarashi Y., Mizoguchi K. Neuropharmacological efficacy of the traditional Japanese Kampo medicine yokukansan and its active ingredients. Pharmacol. Ther. 2016;166:84–95. doi: 10.1016/j.pharmthera.2016.06.018. [DOI] [PubMed] [Google Scholar]
- 453.Wu S.Y., Wang W.J., Dou J.H., Gong L.K. Research progress on the protective effects of licorice-derived 18beta-glycyrrhetinic acid against liver injury. Acta Pharm. Sin. 2020 doi: 10.1038/s41401-020-0383-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 454.Kowalska A., Kalinowska-Lis U. 18beta-Glycyrrhetinic acid: Its core biological properties and dermatological applications. Int. J. Cosmet. Sci. 2019;41:325–331. doi: 10.1111/ics.12548. [DOI] [PubMed] [Google Scholar]
- 455.Kong S.Z., Chen H.M., Yu X.T., Zhang X., Feng X.X., Kang X.H., Li W.J., Huang N., Luo H., Su Z.R. The protective effect of 18beta-Glycyrrhetinic acid against UV irradiation induced photoaging in mice. Exp. Gerontol. 2015;61:147–155. doi: 10.1016/j.exger.2014.12.008. [DOI] [PubMed] [Google Scholar]
- 456.Li X., Sun R., Liu R. Natural products in licorice for the therapy of liver diseases: Progress and future opportunities. Pharm. Res. 2019;144:210–226. doi: 10.1016/j.phrs.2019.04.025. [DOI] [PubMed] [Google Scholar]
- 457.Al-Saeedi F.J. Study of the cytotoxicity of asiaticoside on rats and tumour cells. BMC Cancer. 2014;14:220. doi: 10.1186/1471-2407-14-220. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 458.Liang X., Yan Ni H., Si Wei C., Wen Juan W., Xu N., Cui S., Liu X.H., Zhang H., Yue Nan L., Liu S., et al. Antidepressant-like effect of asiaticoside in mice. Pharm. Biochem. Behav. 2008;89:444–449. doi: 10.1016/j.pbb.2008.01.020. [DOI] [PubMed] [Google Scholar]
- 459.Chen S.W., Wang W.J., Li W.J., Wang R., Li Y.L., Huang Y.N., Liang X. Anxiolytic-like effect of asiaticoside in mice. Pharm. Biochem. Behav. 2006;85:339–344. doi: 10.1016/j.pbb.2006.08.017. [DOI] [PubMed] [Google Scholar]
- 460.Namviriyachote N., Lipipun V., Akkhawattanangkul Y., Charoonrut P., Ritthidej G.C. Development of polyurethane foam dressing containing silver and asiaticoside for healing of dermal wound. Asian J. Pharm. Sci. 2019;14:63–77. doi: 10.1016/j.ajps.2018.09.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 461.Qiu J., Yu L., Zhang X., Wu Q., Wang D., Wang X., Xia C., Feng H. Asiaticoside attenuates lipopolysaccharide-induced acute lung injury via down-regulation of NF-kappaB signaling pathway. Int. Immunopharmacol. 2015;26:181–187. doi: 10.1016/j.intimp.2015.03.022. [DOI] [PubMed] [Google Scholar]
- 462.Zhang C., Chen S., Zhang Z., Xu H., Zhang W., Xu D., Lin B., Mei Y. Asiaticoside Alleviates Cerebral Ischemia-Reperfusion Injury via NOD2/Mitogen-Activated Protein Kinase (MAPK)/Nuclear Factor kappa B (NF-kappaB) Signaling Pathway. Med. Sci. Monit. 2020;26:e920325. doi: 10.12659/MSM.920325. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 463.Luo Y., Fu C., Wang Z., Zhang Z., Wang H., Liu Y. Asiaticoside attenuates the effects of spinal cord injury through antioxidant and antiinflammatory effects, and inhibition of the p38MAPK mechanism. Mol. Med. Rep. 2015;12:8294–8300. doi: 10.3892/mmr.2015.4425. [DOI] [PubMed] [Google Scholar]
- 464.Yin Z., Yu H., Chen S., Ma C., Ma X., Xu L., Ma Z., Qu R., Ma S. Asiaticoside attenuates diabetes-induced cognition deficits by regulating PI3K/Akt/NF-kappaB pathway. Behav. Brain Res. 2015;292:288–299. doi: 10.1016/j.bbr.2015.06.024. [DOI] [PubMed] [Google Scholar]
- 465.Razali N.N.M., Ng C.T., Fong L.Y. Cardiovascular Protective Effects of Centella asiatica and Its Triterpenes: A Review. Planta Med. 2019;85:1203–1215. doi: 10.1055/a-1008-6138. [DOI] [PubMed] [Google Scholar]
- 466.Yu S., Xia H., Guo Y., Qian X., Zou X., Yang H., Yin M., Liu H. Ginsenoside Rb1 retards aging process by regulating cell cycle, apoptotic pathway and metabolism of aging mice. J. Ethnopharmacol. 2020;255:112746. doi: 10.1016/j.jep.2020.112746. [DOI] [PubMed] [Google Scholar]
- 467.Qi R., Jiang R., Xiao H., Wang Z., He S., Wang L., Wang Y. Ginsenoside Rg1 protects against d-galactose induced fatty liver disease in a mouse model via FOXO1 transcriptional factor. Life Sci. 2020;254:117776. doi: 10.1016/j.lfs.2020.117776. [DOI] [PubMed] [Google Scholar]
- 468.Sun G.Z., Meng F.J., Cai H.Q., Diao X.B., Zhang B., Bai X.P. Ginsenoside Rg3 protects heart against isoproterenol-induced myocardial infarction by activating AMPK mediated autophagy. Cardiovasc. Diagn. 2020;10:153–160. doi: 10.21037/cdt.2020.01.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 469.Huang Q., Lou T., Wang M., Xue L., Lu J., Zhang H., Zhang Z., Wang H., Jing C., Zhao D., et al. Compound K inhibits autophagy-mediated apoptosis induced by oxygen and glucose deprivation/reperfusion via regulating AMPK-mTOR pathway in neurons. Life Sci. 2020;254:117793. doi: 10.1016/j.lfs.2020.117793. [DOI] [PubMed] [Google Scholar]
- 470.Wu C.Y., Hua K.F., Hsu W.H., Suzuki Y., Chu L.J., Lee Y.C., Takahata A., Lee S.L., Wu C.C., Nikolic-Paterson D.J., et al. IgA Nephropathy Benefits from Compound K Treatment by Inhibiting NF-kappaB/NLRP3 Inflammasome and Enhancing Autophagy and SIRT1. J. Immunol. 2020 doi: 10.4049/jimmunol.1900284. [DOI] [PubMed] [Google Scholar]
- 471.Wang X.M., She C., Li Q., Zhang D., Xu J.X., Li M.H., Li P., Xu H.B. Ginsenoside Rh3 activates Nrf2 signaling and protects endometrial cells from oxygen and glucose deprivation-reoxygenation. Aging (Albany Ny) 2020;12:6109–6119. doi: 10.18632/aging.103009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 472.Go G.Y., Jo A., Seo D.W., Kim W.Y., Kim Y.K., So E.Y., Chen Q., Kang J.S., Bae G.U., Lee S.J. Ginsenoside Rb1 and Rb2 upregulate Akt/mTOR signaling-mediated muscular hypertrophy and myoblast differentiation. J. Ginseng Res. 2020;44:435–441. doi: 10.1016/j.jgr.2019.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 473.Lu J.M., Yao Q., Chen C. Ginseng compounds: An update on their molecular mechanisms and medical applications. Curr. Vasc. Pharm. 2009;7:293–302. doi: 10.2174/157016109788340767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 474.Lee H., Hong Y., Tran Q., Cho H., Kim M., Kim C., Kwon S.H., Park S., Park J., Park J. A new role for the ginsenoside RG3 in antiaging via mitochondria function in ultraviolet-irradiated human dermal fibroblasts. J. Ginseng Res. 2019;43:431–441. doi: 10.1016/j.jgr.2018.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 475.Kim D.H., Park C.H., Park D., Choi Y.J., Park M.H., Chung K.W., Kim S.R., Lee J.S., Chung H.Y. Ginsenoside Rc modulates Akt/FoxO1 pathways and suppresses oxidative stress. Arch. Pharm Res. 2014;37:813–820. doi: 10.1007/s12272-013-0223-2. [DOI] [PubMed] [Google Scholar]
- 476.Sandner G., Mueller A.S., Zhou X., Stadlbauer V., Schwarzinger B., Schwarzinger C., Wenzel U., Maenner K., van der Klis J.D., Hirtenlehner S., et al. Ginseng Extract Ameliorates the Negative Physiological Effects of Heat Stress by Supporting Heat Shock Response and Improving Intestinal Barrier Integrity: Evidence from Studies with Heat-Stressed Caco-2 Cells, C. elegans and Growing Broilers. Molecules. 2020;25:835. doi: 10.3390/molecules25040835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 477.Kim C.Y., Kim Y.G., Sin S.J., Koo H., Cheon K., Kim D. Preventive Effect of Mebicar and Ginsenoside Rg1 on Neurobehavioral and Immunological Disruptions Caused by Intermittent Unpredictable Stress in Mice. Neuroimmunomodulation. 2018;25:49–58. doi: 10.1159/000489634. [DOI] [PubMed] [Google Scholar]
- 478.Davis M.P., Behm B. Ginseng: A Qualitative Review of Benefits for Palliative Clinicians. Am. J. Hosp. Palliat. Care. 2019;36:630–659. doi: 10.1177/1049909118822704. [DOI] [PubMed] [Google Scholar]
- 479.Nomura S.J., Robien K., Zota A.R. Serum Folate, Vitamin B-12, Vitamin A, gamma-Tocopherol, alpha-Tocopherol, and Carotenoids Do Not Modify Associations between Cadmium Exposure and Leukocyte Telomere Length in the General US Adult Population. J. Nutr. 2017;147:538–548. doi: 10.3945/jn.116.243162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 480.Boccardi V., Arosio B., Cari L., Bastiani P., Scamosci M., Casati M., Ferri E., Bertagnoli L., Ciccone S., Rossi P.D., et al. Beta-carotene, telomerase activity and Alzheimer’s disease in old age subjects. Eur J. Nutr. 2020;59:119–126. doi: 10.1007/s00394-019-01892-y. [DOI] [PubMed] [Google Scholar]
- 481.Cao Y., Wittert G., Taylor A.W., Adams R., Appleton S., Shi Z. Nutrient patterns and chronic inflammation in a cohort of community dwelling middle-aged men. Clin. Nutr. 2017;36:1040–1047. doi: 10.1016/j.clnu.2016.06.018. [DOI] [PubMed] [Google Scholar]
- 482.Dolara P., Bigagli E., Collins A. Antioxidant vitamins and mineral supplementation, life span expansion and cancer incidence: A critical commentary. Eur. J. Nutr. 2012;51:769–781. doi: 10.1007/s00394-012-0389-2. [DOI] [PubMed] [Google Scholar]
- 483.Min K.B., Min J.Y. Association between leukocyte telomere length and serum carotenoid in US adults. Eur. J. Nutr. 2017;56:1045–1052. doi: 10.1007/s00394-016-1152-x. [DOI] [PubMed] [Google Scholar]
- 484.Yuan C., Fondell E., Ascherio A., Okereke O.I., Grodstein F., Hofman A., Willett W.C. Long-Term Intake of Dietary Carotenoids Is Positively Associated with Late-Life Subjective Cognitive Function in a Prospective Study in US Women. J. Nutr. 2020 doi: 10.1093/jn/nxaa087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 485.Mazidi M., Katsiki N., George E.S., Banach M. Tomato and Lycopene Consumption Is Inversely Associated with Total and Cause-Specific Mortality: A Population-based Cohort Study, on behalf of the International Lipid Expert Panel (ILEP) Br. J. Nutr. 2019 doi: 10.1017/S0007114519002150. [DOI] [PubMed] [Google Scholar]
- 486.Nouri M., Nasr-Esfahani M.H., Tarrahi M.J., Amani R. The Effect of Lycopene Supplementation on Mood Status and Quality of Life in Infertile Men: A Randomized, Double-Blind, Placebo-Controlled Clinical Trial. Int. J. Fertil Steril. 2020;14:17–22. doi: 10.22074/ijfs.2020.5888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 487.Zhai L.L., Tang Z.G. Lycopene improves sperm quality: A promising nutrient for the treatment of male infertility. Phytother. Res. 2020 doi: 10.1002/ptr.6608. [DOI] [PubMed] [Google Scholar]
- 488.Chen D., Huang C., Chen Z. A review for the pharmacological effect of lycopene in central nervous system disorders. Biomed. Pharm. 2019;111:791–801. doi: 10.1016/j.biopha.2018.12.151. [DOI] [PubMed] [Google Scholar]
- 489.Zhang X., Yang Y., Wang Q. Lycopene can reduce prostate-specific antigen velocity in a phase II clinical study in Chinese population. Chin. Med. J. (Engl.) 2014;127:2143–2146. [PubMed] [Google Scholar]
- 490.Jiang X., Wang G., Lin Q., Tang Z., Yan Q., Yu X. Fucoxanthin prevents lipopolysaccharide-induced depressive-like behavior in mice via AMPK- NF-kappaB pathway. Metab. Brain Dis. 2019;34:431–442. doi: 10.1007/s11011-018-0368-2. [DOI] [PubMed] [Google Scholar]
- 491.Park J.S., Mathison B.D., Hayek M.G., Zhang J., Reinhart G.A., Chew B.P. Astaxanthin modulates age-associated mitochondrial dysfunction in healthy dogs. J. Anim. Sci. 2013;91:268–275. doi: 10.2527/jas.2012-5341. [DOI] [PubMed] [Google Scholar]
- 492.Hayashi M., Kawamura M., Kawashima Y., Uemura T., Maoka T. Effect of astaxanthin-rich extract derived from Paracoccus carotinifaciens on the status of stress and sleep in adults. J. Clin. Biochem. Nutr. 2020;66:92–102. doi: 10.3164/jcbn.19-113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 493.Jiang X., Yan Q., Liu F., Jing C., Ding L., Zhang L., Pang C. Chronic trans-astaxanthin treatment exerts antihyperalgesic effect and corrects co-morbid depressive like behaviors in mice with chronic pain. Neurosci. Lett. 2018;662:36–43. doi: 10.1016/j.neulet.2017.09.064. [DOI] [PubMed] [Google Scholar]
- 494.Comhaire F.H., El Garem Y., Mahmoud A., Eertmans F., Schoonjans F. Combined conventional/antioxidant “Astaxanthin” treatment for male infertility: A double blind, randomized trial. Asian J. 2005;7:257–262. doi: 10.1111/j.1745-7262.2005.00047.x. [DOI] [PubMed] [Google Scholar]
- 495.Nishida Y., Nawaz A., Kado T., Takikawa A., Igarashi Y., Onogi Y., Wada T., Sasaoka T., Yamamoto S., Sasahara M., et al. Astaxanthin stimulates mitochondrial biogenesis in insulin resistant muscle via activation of AMPK pathway. J. Cachexia Sarcopenia Muscle. 2020;11:241–258. doi: 10.1002/jcsm.12530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 496.Zeni A.L.B., Camargo A., Dalmagro A.P. Lutein prevents corticosterone-induced depressive-like behavior in mice with the involvement of antioxidant and neuroprotective activities. Pharm. Biochem. Behav. 2019;179:63–72. doi: 10.1016/j.pbb.2019.02.004. [DOI] [PubMed] [Google Scholar]
- 497.Cooke M.C., Coates A.M., Buckley E.S., Buckley J.D. Lutein Intake and Blood Lutein Concentration Are Positively Associated with Physical Activity in Adults: A Systematic Review. Nutrients. 2018;10:1186. doi: 10.3390/nu10091186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 498.Lindbergh C.A., Lv J., Zhao Y., Mewborn C.M., Puente A.N., Terry D.P., Renzi-Hammond L.M., Hammond B.R., Liu T., Miller L.S. The effects of lutein and zeaxanthin on resting state functional connectivity in older Caucasian adults: A randomized controlled trial. Brain Imaging Behav. 2020;14:668–681. doi: 10.1007/s11682-018-00034-y. [DOI] [PubMed] [Google Scholar]
- 499.Gerger P., Pai R.K., Stuckenschneider T., Falkenreck J., Weigert H., Stahl W., Weber B., Nelles G., Spazzafumo L., Schneider S., et al. Associations of Lipophilic Micronutrients with Physical and Cognitive Fitness in Persons with Mild Cognitive Impairment. Nutrients. 2019;11:902. doi: 10.3390/nu11040902. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 500.Jia Y.P., Sun L., Yu H.S., Liang L.P., Li W., Ding H., Song X.B., Zhang L.J. The Pharmacological Effects of Lutein and Zeaxanthin on Visual Disorders and Cognition Diseases. Molecules. 2017;22:610. doi: 10.3390/molecules22040610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 501.Edwards J.A. Zeaxanthin: Review of Toxicological Data and Acceptable Daily Intake. J. Ophthalmol. 2016;2016:3690140. doi: 10.1155/2016/3690140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 502.Unno K., Sugiura M., Ogawa K., Takabayashi F., Toda M., Sakuma M., Maeda K., Fujitani K., Miyazaki H., Yamamoto H., et al. Beta-cryptoxanthin, plentiful in Japanese mandarin orange, prevents age-related cognitive dysfunction and oxidative damage in senescence-accelerated mouse brain. Biol. Pharm. Bull. 2011;34:311–317. doi: 10.1248/bpb.34.311. [DOI] [PubMed] [Google Scholar]
- 503.Pearce K., Tremellen K. Influence of nutrition on the decline of ovarian reserve and subsequent onset of natural menopause. Hum. Fertil (Camb.) 2016;19:173–179. doi: 10.1080/14647273.2016.1205759. [DOI] [PubMed] [Google Scholar]
- 504.Pearce K., Tremellen K. Influence of Betaeta-Cryptoxanthin Supplementation on Ovarian Reserve and Fertility Status in Aged Wistar Rats. J. Diet. Suppl. 2020;17:273–285. doi: 10.1080/19390211.2018.1521489. [DOI] [PubMed] [Google Scholar]
- 505.Sahin K., Orhan C., Akdemir F., Tuzcu M., Sahin N., Yilmaz I., Juturu V. beta-Cryptoxanthin ameliorates metabolic risk factors by regulating NF-kappaB and Nrf2 pathways in insulin resistance induced by high-fat diet in rodents. Food Chem. Toxicol. 2017;107:270–279. doi: 10.1016/j.fct.2017.07.008. [DOI] [PubMed] [Google Scholar]
- 506.Zhang F., Shi D., Wang X., Zhang Y., Duan W., Li Y. beta-cryptoxanthin alleviates myocardial ischaemia/reperfusion injury by inhibiting NF-kappaB-mediated inflammatory signalling in rats. Arch. Physiol. Biochem. 2020:1–8. doi: 10.1080/13813455.2020.1760302. [DOI] [PubMed] [Google Scholar]
- 507.Mukherjee S., Date A., Patravale V., Korting H.C., Roeder A., Weindl G. Retinoids in the treatment of skin aging: An overview of clinical efficacy and safety. Clin. Interv. Aging. 2006;1:327–348. doi: 10.2147/ciia.2006.1.4.327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 508.Cho K.S., Lim Y.R., Lee K., Lee J., Lee J.H., Lee I.S. Terpenes from Forests and Human Health. Toxicol. Res. 2017;33:97–106. doi: 10.5487/TR.2017.33.2.097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 509.Partridge L., Fuentealba M., Kennedy B.K. The quest to slow ageing through drug discovery. Nat. Rev. Drug Discov. 2020 doi: 10.1038/s41573-020-0067-7. [DOI] [PubMed] [Google Scholar]
- 510.Sander M., Oxlund B., Jespersen A., Krasnik A., Mortensen E.L., Westendorp R.G.J., Rasmussen L.J. The challenges of human population ageing. Age Ageing. 2015;44:185–187. doi: 10.1093/ageing/afu189. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 511.Partridge L., Deelen J., Slagboom P.E. Facing up to the global challenges of ageing. Nature. 2018;561:45–56. doi: 10.1038/s41586-018-0457-8. [DOI] [PubMed] [Google Scholar]
- 512.Kontis V., Bennett J.E., Mathers C.D., Li G., Foreman K., Ezzati M. Future life expectancy in 35 industrialised countries: Projections with a Bayesian model ensemble. Lancet. 2017;389:1323–1335. doi: 10.1016/S0140-6736(16)32381-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 513.Crimmins E.M. Lifespan and Healthspan: Past, Present, and Promise. Gerontologist. 2015;55:901–911. doi: 10.1093/geront/gnv130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 514.Ogura S., Jakovljevic M.M. Editorial: Global Population Aging–Health Care, Social and Economic Consequences. Front. Public Health. 2018;6:335. doi: 10.3389/fpubh.2018.00335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 515.Christensen K., Doblhammer G., Rau R., Vaupel J.W. Ageing populations: The challenges ahead. Lancet. 2009;374:1196–1208. doi: 10.1016/S0140-6736(09)61460-4. [DOI] [PMC free article] [PubMed] [Google Scholar]