Graphical abstract
Keywords: SARS-COV-2, Calorie restriction, Autophagy, Cytokine storm, Immune responses, COVID-19
Abstract
Severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) is the causative pathogen of deadly Coronavirus disease-19 (COVID-19) pandemic, which emerged as a major threat to public health across the world. Although there is no clear gender or socioeconomic discrimination in the incidence of COVID-19, individuals who are older adults and/or with comorbidities and compromised immunity have a relatively higher risk of contracting this disease. Since no specific drug has yet been discovered, strengthening immunity along with maintaining a healthy living is the best way to survive this disease. As a healthy practice, calorie restriction in the form of intermittent fasting (IF) in several clinical settings has been reported to promote several health benefits, including priming of the immune response. This dietary restriction also activates autophagy, a cell surveillance system that boosts up immunity. With these prevailing significance in priming host defense, IF could be a potential strategy amid this outbreak to fighting off SARS-CoV-2 infection. Currently, no review so far available proposing IF as an encouraging strategy in the prevention of COVID-19. A comprehensive review has therefore been planned to highlight the beneficial role of fasting in immunity and autophagy, that underlie the possible defense against SARS-CoV-2 infection. The COVID-19 pathogenesis and its impact on host immune response have also been briefly outlined. This review aimed at revisiting the immunomodulatory potential of IF that may constitute a promising preventive approach against COVID-19.
1. Introduction
COVID-19, which was first reported in Wuhan, China now emerged as a global pandemic. As of 23 June 2020, there is a total of 9,210,002 confirmed cases of COVID-19, including 474,799 deaths worldwide [1]. Individuals with pre-existing conditions (diabetes, hypertension, chronic bronchitis, cancer, etc.) [2] and compromised immune systems [3] are particularly vulnerable to this disease. Although the case fatality rate of the current outbreak (∼2 %) is lower than that of the previous two similar outbreaks, SARS (∼10 %) in 2002–2004 and MERS (∼34 %) in 2015 [4], the current one already exceeded the previous two in terms of the rate at which people are infected [1]. The matter of concern is that although the previous two outbreaks have been successfully contained, no suitable way has been found yet that can control the current outbreak. As newly emerged, no suitable therapy against COVID-19 has yet been discovered, nor even a clear concept about the pathogenesis of this disease. However, as patients with COVID-19 experience similar symptoms (such as sore throat, persistent high fever, and severe respiratory distress) like the previously emerged outbreaks, including SARS and MERS [5], the pathogenesis of this disease is more likely to be similar to that of those coronavirus diseases that involved massive cytokine storm [6,7]. Moreover, the consensus is that the disease can be fatal if the immune system is already compromised.
The immune system plays a critical role in fighting off SARS-CoV-2 infection, however, deregulated immune response may result in immunopathology and impaired pulmonary function [7,8]. Autophagy is a potential cell surveillance system that plays a pivotal role in the regulation of both innate [9] and adaptive immunity [10]. Induction of autophagy can potentially promote the immune system [11,12]. Targeting the immune system as well as the cellular processes (here, autophagy) that regulate immunity could offer a strategic tool against SARS-CoV-2 infection.
Fasting, a willful abstaining from eating for a certain period of time, is practiced as a religious ritual that has known to have a myriad of health benefits, including boosting up immunity, resistance to stress, slowing down aging process, and increasing longevity without noticeable side effects [[13], [14], [15]]. Fasting also has shown to activate autophagy [16,17], which in turn promotes immunity [18]. As the COVID-19 lacks a specific therapy, preventive measures that can prime host defense could help contain this disease. Considering the regulatory roles of fasting on autophagy and immunity, we anticipate that fasting may become a possible preventive strategy against COVID-19. In this review, we revisit the current knowledge of fasting as a possible important mediator that is involved in the diverse pathophysiological phenomena, including host immune response, autophagy, and the pathogenesis of SARS-CoV-2 infection. A better understanding of the physiological impacts of fasting is crucial to propagate a further investigation on this dietary practice as a novel preventive approach against SARS-CoV-2 infection.
2. SARS-CoV-2-associated immunopathogenesis, host immune response and immune evasion
SARS-CoV-2 infection shares common pathophysiology with other pathogenic coronaviruses, including SARS-CoV and MERS-CoV [19]. SARS-CoV-2 infects host cells through binding with angiotensin converting enzyme 2 (ACE2) receptor which is predominantly expressed in pulmonary alveolar epithelial cells [20,21]. Once inside the cell, the virus multiplies by taking over the host cell machinery and causes damage to the infected cells. SARS-CoV-2 infection and the damaged pulmonary cells induce a local immune response, that recruits macrophages and monocytes to respond to the infection [22].
In most cases, the immune response that follows viral infection readily subsides, and patients ultimately recover. However, in severe cases, patients may experience deadly consequences, including pneumonia which are associated with dysfunctional immune response, i.e., massive inflammatory cell infiltration and elevated and persistent levels of pro-inflammatory cytokines and chemokines (IL-1β, IL-2, IL-6, IL-7, IL-10, GM-CSF, IP-10, MCP-1, and TNF-α) in response of the innate immunity to viral infection [7,23]. These massive cytokine surges develop a severe immunopathological condition, termed as “cytokine storm” which, in turn, may lead to multiple pathological consequences, including extensive pulmonary edema, acute respiratory distress syndrome (ARDS), and multi-organ failure [7,24].
Along with innate immunity, the host body that encounters viral infection also develops the adaptive immune responses recruiting virus-specific T lymphocytes and B lymphocytes, respectively, to stimulate cell-mediated and humoral immune responses. These immune responses either potentiate inflammation or neutralize invading viruses. The antigen-presenting cells (APC) such as macrophages and dendritic cells present the viral antigen to T cells through human leukocyte antigen (HLA) [3]. Once activated, T cells are transformed into multiple forms, activating both cell-mediated and humoral immune response [3]. CD8 + T cells directly destroy virus-infected cells [25], whereas CD4 + T cells are crucial to prime both CD8 + T cells and B cells. Of the two subsets of CD4+, Th1 cells either activate natural killer cells or CD8 + T cells or may remain as memory T cells [3]. Whereas, CD4 + Th2 cells stimulate B cells to be converted into plasma B cells which then generate SARS-CoV-2-specific antibodies (mainly IgM and IgG) [3]. These antibodies, in turn, bind and neutralize SARS-CoV-2. Some of the B cells may form immune memory.
Like many other pathogenic microorganisms, SARS-CoV-2 also evolves mechanisms that help evade the host immune system. One such strategy is the persistent activation of NLRP3 (NACHT, LRR, and PYD domains-containing protein 3) inflammasome, a component of the innate immune system that induced caspase-1 activity and pro-inflammatory cytokines such as interleukin (IL)-1β and IL-18 secretion in macrophages [26]. Although the activation of NLRP3 inflammasome and the subsequent inflammation play crucial roles in the host antiviral immune responses, the aberrant NLRP3 inflammasome activation or chronic inflammation may also result in the severe pathological outcomes as was evident in an influenza A virus infection model in which the experimental animals experienced severe lung injury with an increased level of type I interferons and persistent NLRP3 inflammasome activation [27]. SARS-CoV infection also involves persistent activation of NLRP3 inflammasome by open reading frame 3a (ORF3a) [26,28]. Targeting NLRP3 inflammasome could, therefore, be a promising strategy for restraining viral infection [29].
3. Autophagy and immune responses
Autophagy is a lysosome dependent evolutionarily conserved process that breakdowns and recycles dysfunctional, lethal and mutant biomolecules, organelles, and invading pathogens to retain cellular homeostasis [[30], [31], [32]]. In autophagy, autophagosomes, a double membrane vesicles, engulf and fuse cytoplasmic elements that degraded and recycled the cargo [31,33] to produce sugars, nucleosides/nucleotides, amino acids, and fatty acids. These vital components can be channeled to the other metabolic pathways for cellular utilization [34].
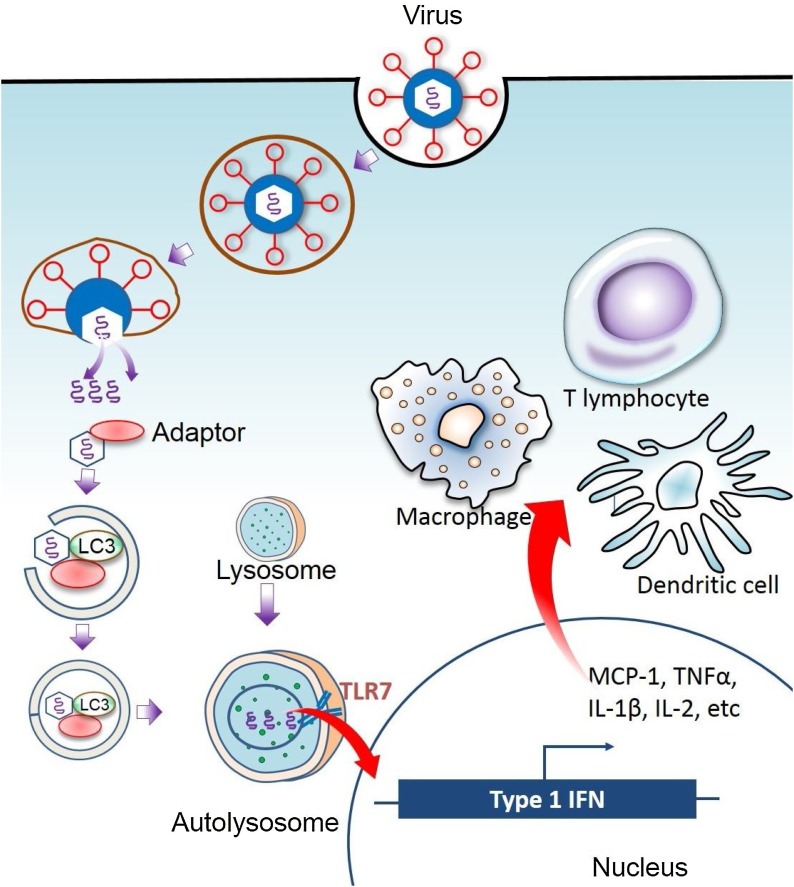
In addition, autophagy is associated with various pathophysiological processes, such as cell survival, cell death, aging, and immunity [35,36]. Autophagy is involved in the antigenic presentation of pathogen (for example, virus) components to the immune system [37,38]. Autophagy modulates the constituents of immune system, including T and B lymphocytes, dendritic cells, macrophages, and natural killer (NK) cells [39]. In innate as well as adaptive immune reactions, autophagy stimulates to maintain survival, homeostasis, proliferation, activation, as well as differentiation [11]. Besides, autophagy also encourages immune-mediated cells to release antibodies and cytokines [40]. During innate immunity, autophagy acts as a pattern of downstream receptors recognition through stimulation of the receptors of innate immunity containing nod-like receptors and toll-like receptors (TLR7), which triggers effector responses such as cytokine production, activation of NK T cell, and phagocytosis [41] (Fig. 1 ).
Fig. 1.
Autophagy-dependent innate immune response. Autophagy may induce innate immunity by delivering viral nucleic acids to endosomes containing Toll-like receptor 7 (TLR7), which stimulates the production of type 1 interferons (IFN) that, in turn, attract immune cells to the site of infection.
During adaptive immunity, autophagy plays an important role in major histocompatibility complex (MHC)-antigen presentation, lymphocyte development, thymic selection, inflammatory signaling, and cytokine regulation [10]. The adaptive immune reaction is regulated by CD4+ as well as CD8+ T cells [42]. T cell receptors act together with antigen-presenting cells to promote maturation of antibodies [43]. Autophagy can be enhanced by antigen presentation, and autophagy activation recruits ATG8/LC3 (autophagy-related 8/light chain 3) to phagosome membranes enclosed by the receptors of pathogen-associated molecular pattern that improve phagosomal fusion with lysosomes along with the transformation of phagosomal content [44]. These events contribute to increasing in antigen presentation and adaptive immunity.
Autophagy in APC plays a significant role in the presentation of endogenous antigens via MHC II, which is recognized by CD4+ T cells [45]. A fusion of viral antigens to the ATG8 (autophagy-related-gene 8) family protein LC3-II, which localizes to autophagosomal membranes, increases presentation to CD4+ T cells. Calorie restriction (CR), an autophagy-inducing factor, leads to an increase in the antigen presentation [46]. Autophagy also functions in the presentation of MHC class I-restricted antigens, which stimulate CD8+ T cells. Autophagy in APC can, therefore, greatly influence the responses of T cell subsets via both MHC I and II-dependent antigenic presentations [47].
Autophagy also regulates the survival and proliferation of T cells. The differentiation of each T cell subset is driven by specific cytokines, for example, IL-12 for TH1 cells, IL-4 for TH2 cells and TGFβ for regulatory CD4+ T cells [48] while combinations of cytokines for other T cell subsets such as THF-β, IL-1, and IL-6 for TH17 cells [48]. Both effector and regulatory CD4+ T cells play an essential role in host immune response, and a defect in these immune pathways are associated with numerous inflammatory diseases. Autophagy is known to have an inhibitory role in the differentiation of TH2 cells [49] while activating the differentiation and function of TH1 cells [49]. Therefore, cytokine-induced autophagy activation has a differential role in the regulation of differentiation and function of each CD4+ T cell subset [50].
Autophagy also plays a significant role in B cell development and survival. B cells with impaired autophagy fail to produce antibodies and cytokines [51]. Autophagy drives plasma cell differentiation and specific antibody production by enhancing antigen presentation [51]. Moreover, plasma cells require autophagy for the sustainable production of antibodies [52].
4. Fasting and autophagy
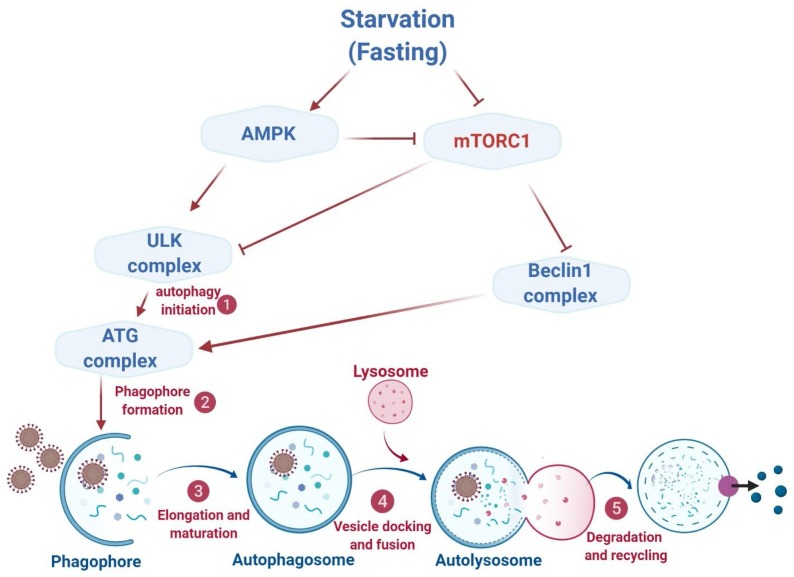
Autophagy is exclusively important during periods of stress and starvation because of its role in furnishing cells with nutrients and energy by recycling fuel-rich macromolecules [53]. Autophagy initiates with the triggering of Unc-51-like kinase (ULK) complex [54] which is regulated by the mechanistic target of rapamycin (mTOR) that can sense nutrient levels in the environment [55]. Under nutrient-rich conditions, mTOR phosphorylates ULK1/2 leading to the inhibition of autophagy. On the contrary, mTOR detaches from the ULK complex during periods of fasting or starvation leading to the activation of autophagy [54]. In addition, AMP-activated protein kinase negatively regulates mTOR, and also directly activates ULK1 complex, thereby acting as a positive regulator of autophagy in response to nutrient depletion. Fasting also upregulates several other autophagy-related proteins such as Atg6, Atg7, Atg8, LC3-II, Beclin1, p62, Sirt1, LAMP2, and ATG101 and thus potentially modulates autophagy [17].
Autophagy inhibition positively influences viral replication or virulence [[56], [57], [58]]. Many viruses inhibit autophagy by blocking autophagy-inducing pathways, AKT1/BECN1, for example, to promote virus replication [58,59]. A recent study has validated that SARS-CoV-2 infection also suppressed autophagy [60]. This study also demonstrated that the pharmacological intervention aimed at autophagy induction showed potentiality against this infection [60]. Similarly, intermittent fasting (IF) that causes nutrient depletion, the most potent known physiological autophagy-stimulator, can induce autophagy [17,61]. One study found that in rats that were starved for 24−46 h, most of the cells in almost every vital tissue had an increased number of autophagosomes [62]. Autophagy inhibition abrogated the anti-aging effects of fasting, indicating that fasting mediates autophagy induction [63]. Another study demonstrated that nutrient deprivation promoted longevity through the Sirtuin-1-dependent induction of autophagy [64]. The beneficial roles of fasting-mediated autophagy promotion have also been reported in functional homeostasis of many organs and tissues [17]. In addition to priming the host immune system, fasting-induced autophagy can improve cellular resistance to stress by increasing the metabolic buffering capacity of cells and thus preparing the human body to deal with various stresses (Fig. 2 ).
Fig. 2.
Fasting mediates autophagy. Autophagy receives fasting signals through two metabolic sensors such as mTOR and AMPK. Under the condition of nutrient depletion, mTOR detaches from the ULK1 complex leading to the activation of autophagy. Whereas, AMPK negatively regulates mTOR, and also directly activates ULK1 complex, thereby acting as a positive regulator of autophagy in response to nutrient depletion. Beclin1 complex is another autophagy activator that is negatively regulated by mTOR. Once autophagy is initiated, cytoplasmic elements (cargo) to be recycled are engulfed into double-membrane vesicles, termed as autophagosomes, which fuse with lysosomes forming autolysosomes, where cargos are degraded. Autophagy is a multistep process that includes (1) initiation, (2) membrane nucleation and phagophore formation, (3) phagophore elongation, (4) docking and fusion with the lysosome, and (5) degradation, which are regulated by autophagy-related proteins (ATGs). mTOR, mechanistic target of rapamycin; AMPK, AMP-activated protein kinase.
5. Fasting and immune responses
IF reduces inflammation and thus could offer some promising health benefits in certain disease conditions such as obesity, asthma, and rheumatoid arthritis, to which inflammatory response is crucially implicated [65]. Fasting enhanced insulin sensitivity and promoted cellular stress resistance [66], and thus help evolve resilience in immune response. IF improved clinical outcomes and caused a reduction of the biomarkers of inflammation (serum TNF-α) and oxidative stress (8-isoprostane, nitrotyrosine, and protein carbonyls) in asthma patients [67]. IF, an age-old obligatory practice by Muslims during the Holy month of Ramadan (over 14 h daily for 30 consecutive days from dawn to sunset), caused upregulation of key regulatory proteins of metabolism, DNA repair, and immune system and resulted in a serum proteome protective against inflammation and associated lifestyle diseases [68]. The potential molecular mechanism of fasting involves the triggering of adaptive cellular stress responses that prime host defense to confront with upcoming severe stress and counteract pathogenesis [65].
Reduction in fat mass correlates with a decline in serum pro-inflammatory cytokines, which indicates that approaches designed to promote fat loss could have beneficial outcomes, in particular, overcome the pro-inflammatory conditions associated with obesity [69]. One such approach could be the IF that helps normalize the systemic inflammatory status of the body by suppressing proinflammatory cytokines (IL-1β, IL-6, and TNF-α) and decreasing fat mass and circulating levels of leukocytes [70]. Supporting these findings, another study showed that intermittent CR positively modulates pro-inflammatory cytokine pathways by reducing the serum cytokine (IL-6 and TNF-α) and adipokine (leptin and IGF-I) levels in wild type female C57BL6 mice [71]. CR induces lipolysis resulting in the reduction of adipocyte size, increase adiponectin secretion, and reduce leptin, IL-1β, IL-6, VEGF-α, MCP-1, and CD-68 expression in white adipose tissue [72]. CR also enhances functional beige fat in mice [73,74]. CR reduces the numbers of circulating monocytes, as well as reduces monocyte metabolic and inflammatory activity in healthy humans and mice [75]. In addition, fasting upregulates gene expression of type 2 cytokines (Il-4, Il-5 and Il-13) that are important for the polarization of M2 macrophage (anti-inflammatory) [76].
Moreover, the potential immune-evading mechanism of SARS-CoV-2 that involves viral ORF3a-mediated persistent activation of NLRP3 can also be modulated by IF. During IF, conventional energy metabolism switches preferably towards fat catabolism with the production of ketones bodies as instant energy sources [77]. The β-hydroxybutyrate (BHB), a major ketone body that fuels many vital organs during fasting/starvation [78], may also help mitigate inflammation by blocking NLRP3 inflammasome overactivation. As evident in experimental models, BHB reduced the production of IL-1β and IL-18 mediated by NLRP3 inflammasome in human monocytes and suppressed caspase-1 activation and IL-1β production in the mouse [79]. These findings suggest that the anti-inflammatory effects of CR may be mechanistically linked to BHB-mediated inhibition of the NLRP3 inflammasome, and point to the potential use of interventions, IF as an example, that elevate circulating BHB against NLRP3-mediated proinflammatory diseases [79].
6. Prospects of fasting against COVID-19 and future directions
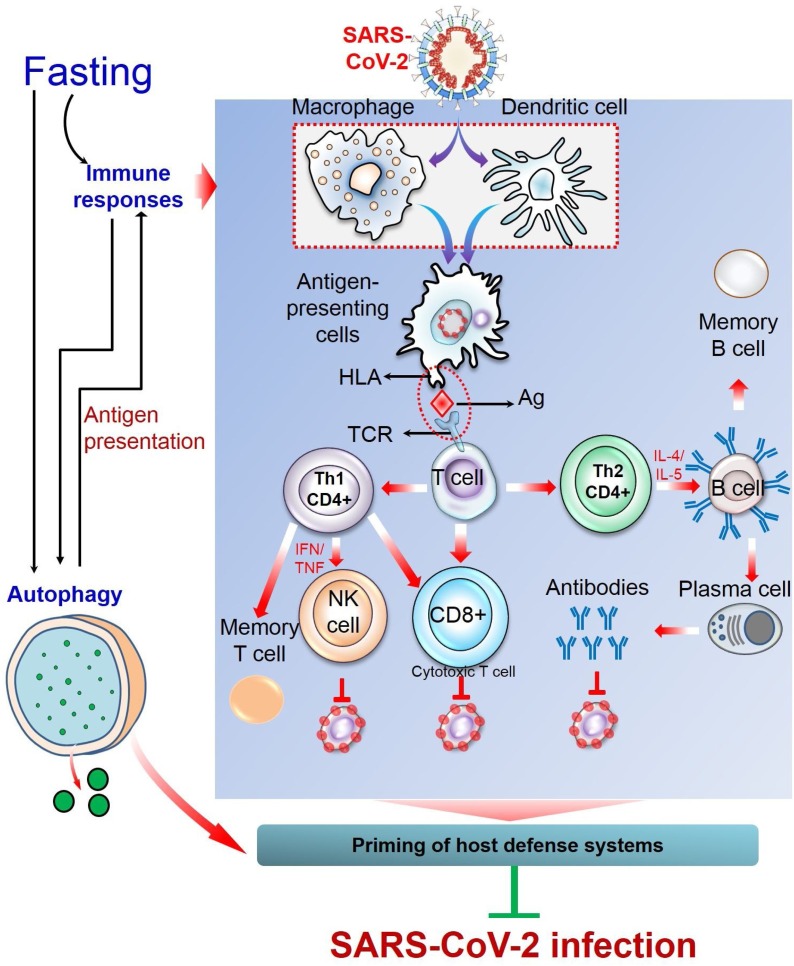
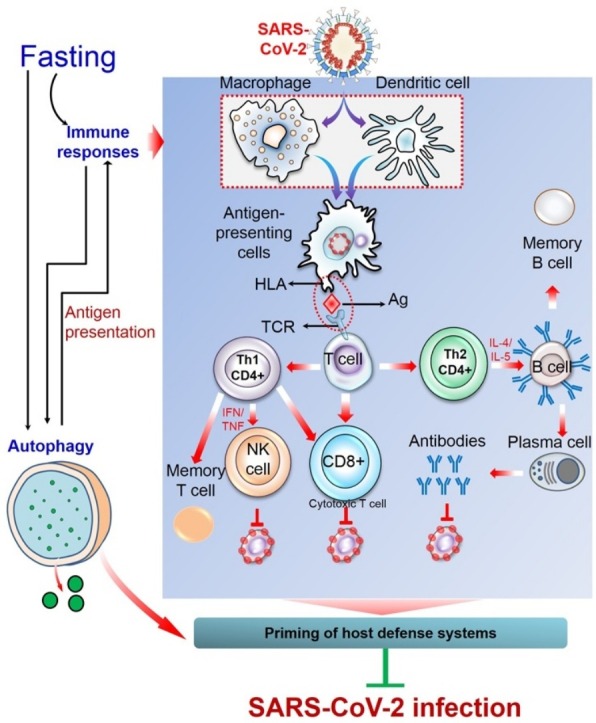
Since the symptoms of COVID-19 are more severe in individuals with pre-existing conditions and deficient in immunocompetence, the possible preventive measures are to control prevailing diseases and to boost up immune system. As already proposed here, IF could be an effective approach that may help prevent SARS-CoV-2 infection. This strategy of dietary restriction can directly (by activating immune response [80]) or indirectly (by inducing autophagy [16,17]) stimulate body surveillance system and boost up immunity, and thus prime host defense to cope with the confronting stresses. However, there is currently no experimental evidence that described the impacts of fasting against SARS-CoV-2 infection. Even no review proposed fasting as a preventive strategy against this disease. With addressing some salient physiological impacts of fasting on the host defense system, this review presents an insight into the potential benefits against SARS-CoV-2 infection that could be attained through observing IF (Fig. 3 ). However, individuals with pre-existing conditions should be aware of the possible complications of IF as CR may worsen their disease conditions. Moreover, even among seemingly healthy individuals, unplanned fasting can sometimes lead to unexpected consequences. COVID-19 patients are strongly advised not to fast during the course of infection as these dietary restrictions may put them at risk of nutritional deficiencies essential for their immune system. Although the health-promoting potentials of fasting are supported by several experimental evidences, a detailed investigation is warranted with an appropriate experimental model to exploit the full advantages of fasting in the prevention of SARS-CoV-2 infection.
Fig. 3.
Fasting as an intervention tool against SARS-CoV-2 infection. Fasting can prime the host defense system through activating multiple physiological processes, including immune responses and autophagy. In case of immune responses, the pulmonary alveolar epithelial cells that are infected with SARS-CoV-2 release damage-associated molecular patterns (DAMPs) such as nucleic acids, which are recognized by adjacent epithelial cells and resident macrophages, triggering the release of pro-inflammatory cytokines and chemokines (IL-6, IP-10, MIP1α, and MCP1). These mediators attract inflammatory cells, including macrophages, monocytes, and T cells to the site of infection, promoting further inflammation. In the dysfunctional immune response, there is a massive infiltration of inflammatory cells and further accumulation of pro-inflammatory mediators (IL-1β, IL-2, IL-6, IL-7, IL-10, G-CSF, IP-10, MCP-1, and TNF-α), leading to an immunopathological condition, referred to as ‘cytokine storm’ that causes multi-organ failure. On the contrary, in protective immune response, the antigen-presenting cells (macrophages and dendritic cells) present viral antigens to T cells which stimulate both cell-mediated and humoral immunity. CD8 + T cells kill virus-infected cells. Of the two subsets of CD4+, Th1 cells either activate natural killer cells or CD8 + T cells or may remain as memory T cells. Whereas, upon stimulation from CD4 + Th2 cells, B cells are converted into plasma B cells which generate SARS-CoV-2-specific antibodies that neutralize viruses. Another fasting-mediated cellular process is autophagy that either degrades viral particles (xenophagy) or activates innate and adaptive immunity. MIP1α, macrophage inflammatory protein 1α; MCP-1, monocyte chemoattractant protein 1; IP-10, interferon-γ-inducible protein 10; G-CSF, Granulocyte-macrophage colony-stimulating factor.
While IF is in practice in various religions and some of them have been proven to have potential health benefits, an appropriate fasting plan can also be adjusted on an individual basis. Along with observing IF, other health-benefiting practices such as exercise and meditation that help improve immunity are also highly recommended. Besides, a healthy diet enriched with functional ingredients that possess strong antioxidant, anti-inflammatory, and immunomodulatory properties should always be incorporated in the dietary chart. During fasting, care should be taken to ensure an adequate amount of essential micronutrients such as vitamin C, vitamin D, and zinc that help boost up the immunity and anti-stress mechanisms.
Author contributions
This work was a collaboration among all the authors. MAH and MJU designed outlines and drafted the manuscript. MAR, MSR, AAMS, RD, KSH, MF, and MJU wrote the initial draft of the manuscript. MJU and MAH reviewed the scientific contents described in the manuscript. All authors read and approved the final submitted version of the manuscript.
Declaration of Competing Interest
No conflict of interest from authors regarding the publication of this manuscript.
Acknowledgements
This work acknowledges the RP-Grant 2020 of Ewha Womans University, Republic of Korea. MAH, MSR, and MAR are grateful to the National Research Foundation of Korea (NRF) for Korea Research Fellowship (KRF) (#2018H1D3A1A01074712, #2017H1D3A1A02013844 and #2016H1D3A1908615, respectively) funded by the Ministry of Science and ICT, Republic of Korea.
References
- 1.2020. Worldometer, Coronavirus.https://www.worldometers.info/coronavirus/ [Google Scholar]
- 2.Guan W.-J., Liang W.-H., Zhao Y., Liang H.-R., Chen Z.-S., Li Y.-M., Liu X.-Q., Chen R.-C., Tang C.-L., Wang T., Ou C.-Q., Li L., Chen P.-Y., Sang L., Wang W., Li J.-F., Li C.-C., Ou L.-M., Cheng B., Xiong S., Ni Z.-Y., Xiang J., Hu Y., Liu L., Shan H., Lei C.-L., Peng Y.-X., Wei L., Liu Y., Hu Y.-H., Peng P., Wang J.-M., Liu J.-Y., Chen Z., Li G., Zheng Z.-J., Qiu S.-Q., Luo J., Ye C.-J., Zhu S.-Y., Cheng L.-L., Ye F., Li S.-Y., Zheng J.-P., Zhang N.-F., Zhong N.-S., He J.-X., C. China Medical Treatment Expert Group for Comorbidity and its impact on 1590 patients with Covid-19 in China: a nationwide analysis. Eur. Respir. J. 2020 doi: 10.1183/13993003.00547-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nikolich-Zugich J., Knox K.S., Rios C.T., Natt B., Bhattacharya D., Fain M.J. SARS-CoV-2 and COVID-19 in older adults: what we may expect regarding pathogenesis, immune responses, and outcomes. GeroScience. 2020:1–10. doi: 10.1007/s11357-020-00186-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Mahase E. Coronavirus: covid-19 has killed more people than SARS and MERS combined, despite lower case fatality rate. BMJ. 2020;368:m641. doi: 10.1136/bmj.m641. [DOI] [PubMed] [Google Scholar]
- 5.Petrosillo N., Viceconte G., Ergonul O., Ippolito G., Petersen E. COVID-19, SARS and MERS: are they closely related? Clin. Microbiol. Infect. 2020 doi: 10.1016/j.cmi.2020.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Duan K., Liu B., Li C., Zhang H., Yu T., Qu J., Zhou M., Chen L., Meng S., Hu Y., Peng C., Yuan M., Huang J., Wang Z., Yu J., Gao X., Wang D., Yu X., Li L., Zhang J., Wu X., Li B., Xu Y., Chen W., Peng Y., Hu Y., Lin L., Liu X., Huang S., Zhou Z., Zhang L., Wang Y., Zhang Z., Deng K., Xia Z., Gong Q., Zhang W., Zheng X., Liu Y., Yang H., Zhou D., Yu D., Hou J., Shi Z., Chen S., Chen Z., Zhang X., Yang X. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. PNAS. 2020;117(17):9490–9496. doi: 10.1073/pnas.2004168117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Channappanavar R., Perlman S. Pathogenic human coronavirus infections: causes and consequences of cytokine storm and immunopathology. Semin. Immunopathol. 2017;39(5):529–539. doi: 10.1007/s00281-017-0629-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Diao B., Wang C., Tan Y., Chen X., Liu Y., Ning L., Chen L., Li M., Liu Y., Wang G., Yuan Z., Feng Z., Wu Y., Chen Y. 2020. Reduction and Functional Exhaustion of T Cells in Patients With Coronavirus Disease 2019 (COVID-19) 2020.02.18.20024364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pan H.M., Chen L.X., Xu Y.H., Han W.D., Lou F., Fei W.Q., Liu S.P., Jing Z., Sui X.B. Autophagy-associated immune responses and cancer immunotherapy. Oncotarget. 2016;7(16):21235–21246. doi: 10.18632/oncotarget.6908. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Crotzer V.L., Blum J.S. Autophagy and adaptive immunity. Immunology. 2010;131(1):9–17. doi: 10.1111/j.1365-2567.2010.03321.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Cui B., Lin H., Yu J., Yu J., Hu Z. Autophagy and the immune response. Adv. Exp. Med. Biol. 2019;1206:595–634. doi: 10.1007/978-981-15-0602-4_27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kuballa P., Nolte W.M., Castoreno A.B., Xavier R.J. Autophagy and the immune system. Annu. Rev. Immunol. 2012;30:611–646. doi: 10.1146/annurev-immunol-020711-074948. [DOI] [PubMed] [Google Scholar]
- 13.Aris J.P., Alvers A.L., Ferraiuolo R.A., Fishwick L.K., Hanvivatpong A., Hu D., Kirlew C., Leonard M.T., Losin K.J., Marraffini M., Seo A.Y., Swanberg V., Westcott J.L., Wood M.S., Leeuwenburgh C., Dunn W.A., Jr. Autophagy and leucine promote chronological longevity and respiration proficiency during calorie restriction in yeast. Exp. Gerontol. 2013;48(10):1107–1119. doi: 10.1016/j.exger.2013.01.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Rickenbacher A., Jang J.H., Limani P., Ungethum U., Lehmann K., Oberkofler C.E., Weber A., Graf R., Humar B., Clavien P.A. Fasting protects liver from ischemic injury through Sirt1-mediated downregulation of circulating HMGB1 in mice. J. Hepatol. 2014;61(2):301–308. doi: 10.1016/j.jhep.2014.04.010. [DOI] [PubMed] [Google Scholar]
- 15.Golbidi S., Daiber A., Korac B., Li H., Essop M.F., Laher I. Health benefits of fasting and caloric restriction. Curr. Diab. Rep. 2017;17(12):123. doi: 10.1007/s11892-017-0951-7. [DOI] [PubMed] [Google Scholar]
- 16.Alirezaei M., Kemball C.C., Flynn C.T., Wood M.R., Whitton J.L., Kiosses W.B. Short-term fasting induces profound neuronal autophagy. Autophagy. 2010;6(6):702–710. doi: 10.4161/auto.6.6.12376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Bagherniya M., Butler A.E., Barreto G.E., Sahebkar A. The effect of fasting or calorie restriction on autophagy induction: a review of the literature. Ageing Res. Rev. 2018;47:183–197. doi: 10.1016/j.arr.2018.08.004. [DOI] [PubMed] [Google Scholar]
- 18.Brandhorst S., Choi I.Y., Wei M., Cheng C.W., Sedrakyan S., Navarrete G., Dubeau L., Yap L.P., Park R., Vinciguerra M., Di Biase S., Mirzaei H., Mirisola M.G., Childress P., Ji L., Groshen S., Penna F., Odetti P., Perin L., Conti P.S., Ikeno Y., Kennedy B.K., Cohen P., Morgan T.E., Dorff T.B., Longo V.D. A periodic diet that mimics fasting promotes multi-system regeneration, enhanced cognitive performance, and healthspan. Cell Metab. 2015;22(1):86–99. doi: 10.1016/j.cmet.2015.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Fu Y., Cheng Y., Wu Y. Understanding SARS-CoV-2-mediated inflammatory responses: from mechanisms to potential therapeutic tools. Virol. Sin. 2020 doi: 10.1007/s12250-020-00207-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Li W., Moore M.J., Vasilieva N., Sui J., Wong S.K., Berne M.A., Somasundaran M., Sullivan J.L., Luzuriaga K., Greenough T.C., Choe H., Farzan M. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426(6965):450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhou P., Yang X.L., Wang X.G., Hu B., Zhang L., Zhang W., Si H.R., Zhu Y., Li B., Huang C.L., Chen H.D., Chen J., Luo Y., Guo H., Jiang R.D., Liu M.Q., Chen Y., Shen X.R., Wang X., Zheng X.S., Zhao K., Chen Q.J., Deng F., Liu L.L., Yan B., Zhan F.X., Wang Y.Y., Xiao G.F., Shi Z.L. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579(7798):270–273. doi: 10.1038/s41586-020-2012-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tay M.Z., Poh C.M., Rénia L., MacAry P.A., Ng L.F.P. The trinity of COVID-19: immunity, inflammation and intervention. Nat. Rev. Immunol. 2020 doi: 10.1038/s41577-020-0311-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Huang C., Wang Y., Li X., Ren L., Zhao J., Hu Y., Zhang L., Fan G., Xu J., Gu X., Cheng Z., Yu T., Xia J., Wei Y., Wu W., Xie X., Yin W., Li H., Liu M., Xiao Y., Gao H., Guo L., Xie J., Wang G., Jiang R., Gao Z., Jin Q., Wang J., Cao B. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London, England) 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Qin C., Zhou L., Hu Z., Zhang S., Yang S., Tao Y., Xie C., Ma K., Shang K., Wang W., Tian D.-S. Dysregulation of immune response in patients with coronavirus 2019 (COVID-19) in Wuhan, China. Clin. Infect. Dis. 2020 doi: 10.1093/cid/ciaa248. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Yi Y., Lagniton P.N.P., Ye S., Li E., Xu R.H. COVID-19: what has been learned and to be learned about the novel coronavirus disease. Int. J. Biol. Sci. 2020;16(10):1753–1766. doi: 10.7150/ijbs.45134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Siu K.L., Yuen K.S., Castaño-Rodriguez C., Ye Z.W., Yeung M.L., Fung S.Y., Yuan S., Chan C.P., Yuen K.Y., Enjuanes L., Jin D.Y. Severe acute respiratory syndrome coronavirus ORF3a protein activates the NLRP3 inflammasome by promoting TRAF3-dependent ubiquitination of ASC. FASEB J. 2019;33(8):8865–8877. doi: 10.1096/fj.201802418R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Coates B.M., Staricha K.L., Koch C.M., Cheng Y., Shumaker D.K., Budinger G.R.S., Perlman H., Misharin A.V., Ridge K.M. J. Immunol. (Baltimore, Md. : 1950) 2018;200(7):2391–2404. doi: 10.4049/jimmunol.1701543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Nieto-Torres J.L., Verdiá-Báguena C., Jimenez-Guardeño J.M., Regla-Nava J.A., Castaño-Rodriguez C., Fernandez-Delgado R., Torres J., Aguilella V.M., Enjuanes L. Severe acute respiratory syndrome coronavirus E protein transports calcium ions and activates the NLRP3 inflammasome. Virology. 2015;485:330–339. doi: 10.1016/j.virol.2015.08.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhao C., Zhao W. NLRP3 Inflammasome-A key player in antiviral responses. Front. Immunol. 2020;11:211. doi: 10.3389/fimmu.2020.00211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Levine B., Klionsky D.J. Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev. Cell. 2004;6(4):463–477. doi: 10.1016/s1534-5807(04)00099-1. [DOI] [PubMed] [Google Scholar]
- 31.Galluzzi L., Baehrecke E.H., Ballabio A., Boya P., Bravo-San Pedro J.M., Cecconi F., Choi A.M., Chu C.T., Codogno P., Colombo M.I., Cuervo A.M., Debnath J., Deretic V., Dikic I., Eskelinen E.L., Fimia G.M., Fulda S., Gewirtz D.A., Green D.R., Hansen M., Harper J.W., Jäättelä M., Johansen T., Juhasz G., Kimmelman A.C., Kraft C., Ktistakis N.T., Kumar S., Levine B., Lopez-Otin C., Madeo F., Martens S., Martinez J., Melendez A., Mizushima N., Münz C., Murphy L.O., Penninger J.M., Piacentini M., Reggiori F., Rubinsztein D.C., Ryan K.M., Santambrogio L., Scorrano L., Simon A.K., Simon H.U., Simonsen A., Tavernarakis N., Tooze S.A., Yoshimori T., Yuan J., Yue Z., Zhong Q., Kroemer G. Molecular definitions of autophagy and related processes. EMBO J. 2017;36(13):1811–1836. doi: 10.15252/embj.201796697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rahman M.A., Rahman M.S., Uddin M.J., Uddin M.S., Mg P., Rhim H., Cho S. Molecular insights into therapeutic potential of autophagy modulation by natural products for cancer stem cells. Front. Cell Dev. Biol. 2020;8(283) doi: 10.3389/fcell.2020.00283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Mizushima N., Komatsu M. Autophagy: renovation of cells and tissues. Cell. 2011;147(4):728–741. doi: 10.1016/j.cell.2011.10.026. [DOI] [PubMed] [Google Scholar]
- 34.Kimmelman A.C., White E. Autophagy and tumor metabolism. Cell Metab. 2017;25(5):1037–1043. doi: 10.1016/j.cmet.2017.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Choi A.M., Ryter S.W., Levine B. Autophagy in human health and disease. N. Engl. J. Med. 2013;368(7):651–662. doi: 10.1056/NEJMra1205406. [DOI] [PubMed] [Google Scholar]
- 36.Mizushima N., Levine B. Autophagy in mammalian development and differentiation. Nat. Cell Biol. 2010;12(9):823–830. doi: 10.1038/ncb0910-823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Gannage M., Munz C. MHC presentation via autophagy and how viruses escape from it. Semin. Immunopathol. 2010;32(4):373–381. doi: 10.1007/s00281-010-0227-7. [DOI] [PubMed] [Google Scholar]
- 38.Crotzer V.L., Blum J.S. Autophagy and its role in MHC-mediated antigen presentation. J. Immunol. 2009;182(6):3335–3341. doi: 10.4049/jimmunol.0803458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Jiang G.M., Tan Y., Wang H., Peng L., Chen H.T., Meng X.J., Li L.L., Liu Y., Li W.F., Shan H. The relationship between autophagy and the immune system and its applications for tumor immunotherapy. Mol. Cancer. 2019;18 doi: 10.1186/s12943-019-0944-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Wu T.T., Li W.M., Yao Y.M. Interactions between autophagy and inhibitory cytokines. Int. J. Biol. Sci. 2016;12(7):884–897. doi: 10.7150/ijbs.15194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liu C.H., Liu H.Y., Ge B.X. Innate immunity in tuberculosis: host defense vs pathogen evasion. Cell. Mol. Immunol. 2017;14(12):963–975. doi: 10.1038/cmi.2017.88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wieczorek M., Abualrous E.T., Sticht J., Alvaro-Benito M., Stolzenberg S., Noe F., Freund C. Major histocompatibility complex (MHC) class I and MHC class II proteins: conformational plasticity in antigen presentation. Front. Immunol. 2017;8 doi: 10.3389/fimmu.2017.00292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Dhodapkar M.V., Dhodapkar K.M., Palucka A.K. Interactions of tumor cells with dendritic cells: balancing immunity and tolerance. Cell Death Differ. 2008;15(1):39–50. doi: 10.1038/sj.cdd.4402247. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Randow F., Munz C. Autophagy in the regulation of pathogen replication and adaptive immunity. Trends Immunol. 2012;33(10):475–487. doi: 10.1016/j.it.2012.06.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Münz C. Autophagy proteins in antigen processing for presentation on MHC molecules. Immunol. Rev. 2016;272(1):17–27. doi: 10.1111/imr.12422. [DOI] [PubMed] [Google Scholar]
- 46.Dengjel J., Schoor O., Fischer R., Reich M., Kraus M., Müller M., Kreymborg K., Altenberend F., Brandenburg J., Kalbacher H., Brock R., Driessen C., Rammensee H.G., Stevanovic S. Autophagy promotes MHC class II presentation of peptides from intracellular source proteins. Proc. Natl. Acad. Sci. U. S. A. 2005;102(22):7922–7927. doi: 10.1073/pnas.0501190102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Merkley S.D., Chock C.J., Yang X.O., Harris J., Castillo E.F. Modulating t cell responses via autophagy: the intrinsic influence controlling the function of both antigen-presenting cells and t cells. Front. Immunol. 2018;9(2914) doi: 10.3389/fimmu.2018.02914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Hirahara K., Nakayama T. CD4+ T-cell subsets in inflammatory diseases: beyond the Th1/Th2 paradigm. Int. Immunol. 2016;28(4):163–171. doi: 10.1093/intimm/dxw006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Kabat A.M., Harrison O.J., Riffelmacher T., Moghaddam A.E., Pearson C.F., Laing A., Abeler-Dörner L., Forman S.P., Grencis R.K., Sattentau Q., Simon A.K., Pott J., Maloy K.J. The autophagy gene Atg16l1 differentially regulates Treg and TH2 cells to control intestinal inflammation. eLife. 2016;5 doi: 10.7554/eLife.12444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Botbol Y., Patel B., Macian F. Common γ-chain cytokine signaling is required for macroautophagy induction during CD4+ T-cell activation. Autophagy. 2015;11(10):1864–1877. doi: 10.1080/15548627.2015.1089374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Jiang G.-M., Tan Y., Wang H., Peng L., Chen H.-T., Meng X.-J., Li L.-L., Liu Y., Li W.-F., Shan H. The relationship between autophagy and the immune system and its applications for tumor immunotherapy. Mol. Cancer. 2019;18(1):17. doi: 10.1186/s12943-019-0944-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Pengo N., Scolari M., Oliva L., Milan E., Mainoldi F., Raimondi A., Fagioli C., Merlini A., Mariani E., Pasqualetto E., Orfanelli U., Ponzoni M., Sitia R., Casola S., Cenci S. Plasma cells require autophagy for sustainable immunoglobulin production. Nat. Immunol. 2013;14(3):298–305. doi: 10.1038/ni.2524. [DOI] [PubMed] [Google Scholar]
- 53.Bento C.F., Renna M., Ghislat G., Puri C., Ashkenazi A., Vicinanza M., Menzies F.M., Rubinsztein D.C. Mammalian autophagy: how does it work? Annu. Rev. Biochem. 2016;85:685–713. doi: 10.1146/annurev-biochem-060815-014556. [DOI] [PubMed] [Google Scholar]
- 54.Hansen M., Rubinsztein D.C., Walker D.W. Autophagy as a promoter of longevity: insights from model organisms. Nat. Rev. Mol. Cell Biol. 2018;19(9):579–593. doi: 10.1038/s41580-018-0033-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Sohn M., Kim K., Uddin M.J., Lee G., Hwang I., Kang H., Kim H., Lee J.H., Ha H. Delayed treatment with fenofibrate protects against high-fat diet-induced kidney injury in mice: the possible role of AMPK autophagy. Am. J. Physiol. Renal Physiol. 2017;312(2):F323–F334. doi: 10.1152/ajprenal.00596.2015. [DOI] [PubMed] [Google Scholar]
- 56.Cherry S. VSV infection is sensed by Drosophila, attenuates nutrient signaling, and thereby activates antiviral autophagy. Autophagy. 2009;5(7):1062–1063. doi: 10.4161/auto.5.7.9730. [DOI] [PubMed] [Google Scholar]
- 57.Orvedahl A., MacPherson S., Sumpter R., Jr., Talloczy Z., Zou Z., Levine B. Autophagy protects against Sindbis virus infection of the central nervous system. Cell Host Microbe. 2010;7(2):115–127. doi: 10.1016/j.chom.2010.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Orvedahl A., Alexander D., Talloczy Z., Sun Q., Wei Y., Zhang W., Burns D., Leib D.A., Levine B. HSV-1 ICP34.5 confers neurovirulence by targeting the Beclin 1 autophagy protein. Cell Host Microbe. 2007;1(1):23–35. doi: 10.1016/j.chom.2006.12.001. [DOI] [PubMed] [Google Scholar]
- 59.Ku B., Woo J.S., Liang C., Lee K.H., Hong H.S., X. E, Kim K.S., Jung J.U., Oh B.H. Structural and biochemical bases for the inhibition of autophagy and apoptosis by viral BCL-2 of murine gamma-herpesvirus 68. PLoS Pathog. 2008;4(2):e25. doi: 10.1371/journal.ppat.0040025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Gassen J.P.Nils C., Bajaj Thomas, Dethloff Frederik, Emanuel Jackson, Weckmann Katja, Heinz Daniel E., Heinemann Nicolas, Lennarz Martina, Richter Anja, Niemeyer Daniela, Corman Victor M., Giavalisco Patrick, Drosten Christian, Müller Marcel A. Analysis of SARS-CoV-2-controlled autophagy reveals spermidine, MK-2206, and niclosamide as putative antiviral therapeutics. bioRxiv. 2020 [Google Scholar]
- 61.Kroemer G., Mariño G., Levine B. Autophagy and the integrated stress response. Mol. Cell. 2010;40(2):280–293. doi: 10.1016/j.molcel.2010.09.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Mizushima N. The role of the Atg1/ULK1 complex in autophagy regulation. Curr. Opin. Cell Biol. 2010;22(2):132–139. doi: 10.1016/j.ceb.2009.12.004. [DOI] [PubMed] [Google Scholar]
- 63.Martinez-Lopez N., Athonvarangkul D., Singh R. Autophagy and aging. Adv. Exp. Med. Biol. 2015;847:73–87. doi: 10.1007/978-1-4939-2404-2_3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Morselli E., Maiuri M.C., Markaki M., Megalou E., Pasparaki A., Palikaras K., Criollo A., Galluzzi L., Malik S.A., Vitale I., Michaud M., Madeo F., Tavernarakis N., Kroemer G. Caloric restriction and resveratrol promote longevity through the Sirtuin-1-dependent induction of autophagy. Cell Death Dis. 2010;1(1) doi: 10.1038/cddis.2009.8. e10-e10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Longo V.D., Mattson M.P. Fasting: molecular mechanisms and clinical applications. Cell Metab. 2014;19(2):181–192. doi: 10.1016/j.cmet.2013.12.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Mattson M.P., Longo V.D., Harvie M. Impact of intermittent fasting on health and disease processes. Ageing Res. Rev. 2017;39:46–58. doi: 10.1016/j.arr.2016.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Johnson J.B., Summer W., Cutler R.G., Martin B., Hyun D.H., Dixit V.D., Pearson M., Nassar M., Telljohann R., Maudsley S., Carlson O., John S., Laub D.R., Mattson M.P. Alternate day calorie restriction improves clinical findings and reduces markers of oxidative stress and inflammation in overweight adults with moderate asthma. Free Radic. Biol. Med. 2007;42(5):665–674. doi: 10.1016/j.freeradbiomed.2006.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Mindikoglu A.L., Abdulsada M.M., Jain A., Choi J.M., Jalal P.K., Devaraj S., Mezzari M.P., Petrosino J.F., Opekun A.R., Jung S.Y. Intermittent fasting from dawn to sunset for 30 consecutive days is associated with anticancer proteomic signature and upregulates key regulatory proteins of glucose and lipid metabolism, circadian clock, DNA repair, cytoskeleton remodeling, immune system and cognitive function in healthy subjects. J. Proteomics. 2020;217 doi: 10.1016/j.jprot.2020.103645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Harvie M.N., Pegington M., Mattson M.P., Frystyk J., Dillon B., Evans G., Cuzick J., Jebb S.A., Martin B., Cutler R.G., Son T.G., Maudsley S., Carlson O.D., Egan J.M., Flyvbjerg A., Howell A. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: a randomized trial in young overweight women. Int. J. Obes. 2011;35(5):714–727. doi: 10.1038/ijo.2010.171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Faris M.A., Kacimi S., Al-Kurd R.A., Fararjeh M.A., Bustanji Y.K., Mohammad M.K., Salem M.L. Intermittent fasting during Ramadan attenuates proinflammatory cytokines and immune cells in healthy subjects. Nutr. Res. 2012;32(12):947–955. doi: 10.1016/j.nutres.2012.06.021. [DOI] [PubMed] [Google Scholar]
- 71.Dogan S., Ray A., Cleary M.P. The influence of different calorie restriction protocols on serum pro-inflammatory cytokines, adipokines and IGF-I levels in female C57BL6 mice: short term and long term diet effects. Meta Gene. 2017;12:22–32. doi: 10.1016/j.mgene.2016.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Romero Mdel M., Fernández-López J.A., Esteve M., Alemany M. Different modulation by dietary restriction of adipokine expression in white adipose tissue sites in the rat. Cardiovasc. Diabetol. 2009;8:42. doi: 10.1186/1475-2840-8-42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Fabbiano S., Suarez-Zamorano N., Rigo D., Veyrat-Durebex C., Stevanovic Dokic A., Colin D.J., Trajkovski M. Caloric restriction leads to Browning of white adipose tissue through type 2 immune signaling. Cell Metab. 2016;24(3):434–446. doi: 10.1016/j.cmet.2016.07.023. [DOI] [PubMed] [Google Scholar]
- 74.Villarroya F., Cereijo R., Villarroya J., Gavaldà-Navarro A., Giralt M. Toward an understanding of how immune cells control Brown and beige adipobiology. Cell Metab. 2018;27(5):954–961. doi: 10.1016/j.cmet.2018.04.006. [DOI] [PubMed] [Google Scholar]
- 75.Jordan S., Tung N., Casanova-Acebes M., Chang C., Cantoni C., Zhang D., Wirtz T.H., Naik S., Rose S.A., Brocker C.N., Gainullina A., Hornburg D., Horng S., Maier B.B., Cravedi P., LeRoith D., Gonzalez F.J., Meissner F., Ochando J., Rahman A., Chipuk J.E., Artyomov M.N., Frenette P.S., Piccio L., Berres M.L., Gallagher E.J., Merad M. Dietary intake regulates the circulating inflammatory monocyte pool. Cell. 2019;178(5) doi: 10.1016/j.cell.2019.07.050. 1102–1114 e17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Kim K.-H., Kim Y.H., Son J.E., Lee J.H., Kim S., Choe M.S., Moon J.H., Zhong J., Fu K., Lenglin F., Yoo J.-A., Bilan P.J., Klip A., Nagy A., Kim J.-R., Park J.G., Hussein S.M.I., Doh K.-O., Hui C.-c., Sung H.-K. Intermittent fasting promotes adipose thermogenesis and metabolic homeostasis via VEGF-mediated alternative activation of macrophage. Cell Res. 2017;27(11):1309–1326. doi: 10.1038/cr.2017.126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.McGettrick A.F., O’Neill L.A. How metabolism generates signals during innate immunity and inflammation. J. Biol. Chem. 2013;288(32):22893–22898. doi: 10.1074/jbc.R113.486464. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Newman J.C., Verdin E. Ketone bodies as signaling metabolites. Trends Endocrinol. Metab. 2014;25(1):42–52. doi: 10.1016/j.tem.2013.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Youm Y.H., Nguyen K.Y., Grant R.W., Goldberg E.L., Bodogai M., Kim D., D’Agostino D., Planavsky N., Lupfer C., Kanneganti T.D., Kang S., Horvath T.L., Fahmy T.M., Crawford P.A., Biragyn A., Alnemri E., Dixit V.D. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome-mediated inflammatory disease. Nat. Med. 2015;21(3):263–269. doi: 10.1038/nm.3804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Cheng C.W., Adams G.B., Perin L., Wei M., Zhou X., Lam B.S., Da Sacco S., Mirisola M., Quinn D.I., Dorff T.B., Kopchick J.J., Longo V.D. Prolonged fasting reduces IGF-1/PKA to promote hematopoietic-stem-cell-based regeneration and reverse immunosuppression. Cell Stem Cell. 2014;14(6):810–823. doi: 10.1016/j.stem.2014.04.014. [DOI] [PMC free article] [PubMed] [Google Scholar]