Abstract
Lung cancer stem cells (CSCs) play a pivotal role in tumor development, drug resistance, metastasis and recurrence of lung cancer. Thus, it is of great importance to study the mechanism by which CSCs are regulated. In this study, we demonstrate that the deubiquitinase USP4 is critically important in promoting lung cancer stemness. Silencing of USP4 leads to reduction of Oct4 and Sox2 expression, decreased CD133+ cell population and inhibition of tumorsphere formation. Conversely, ectopic expression of USP4 significantly enhances lung cancer cell stemness, which is effectively rescued by simultaneous silencing of Twist1. Mechanistically, we identified USP4 as a novel deubiquitinase of Twist1. USP4 binds to, deubiquitinates and stabilizes Twist1 protein. Furthermore, we show that USP4 expression is elevated in human lung cancer specimens and is positively correlated with Twist1 expression. High expression of USP4/Twist1 is associated with poor clinical outcomes of lung cancer patients. Together, this study highlights an important role for USP4 in lung cancer stemness and suggests USP4 as a potential target for lung cancer diagnosis and treatment.
Keywords: USP4, Twist1, lung cancer, CSCs, stemness
1. Introduction
Lung cancer is the most common cancer worldwide and a leading cause of cancer-related death [1]. Histopathologically, lung cancer is categorized into small cell lung cancer (SCLC) and non-small cell lung cancer (NSCLC), of which NSCLC accounts for 85% of all lung cancer cases. In recent years, development of NSCLC therapy, including chemotherapy, radiotherapy, targeted therapy and immunotherapy, greatly improves both survival and prognosis for patients [2]. However, a major challenge in NSCLC treatment is that most of NSCLC patients do not respond well to immunotherapy and that tumor recurrence and drug resistance after treatment remain [3]. Thus, it is of great importance to illustrate new mechanisms and new drug targets involved in NSCLC tumorigenesis and drug resistance.
It has been well documented that cancer stem cells (CSCs) play a critical role in cancer development, tumorigenesis, drug resistance, metastasis and recurrence [4,5,6,7]. CSCs are a small sub-population of tumor cells with the capacity for self-renewal, differentiation, tumor initiation and innate resistance to chemotherapy and radiation. These characters associated with CSCs are collectively called stemness. In NSCLC, CSCs can be identified and characterized by expression of CSCs markers, including CD133, CD44 and aldehyde dehydrogenase (ALDH) [8,9,10,11]. Several embryonic transcription factors, including Oct4, Sox2, Nanog, MYC and KLF4, are essential in the regulation of stemness of CSCs [8,12,13,14,15]. In addition, CSCs characteristically exhibit activation of several highly conserved pathways involved in cell renewal and tissue homeostasis, including Hedgehog, Notch, WNT/β-Catenin and TNF-α, to promote cell survival, self-renewal, and metastasis [16,17,18,19]. Targeting proteins critical for CSCs stemness has been a potential strategy for cancer treatment [20,21].
Twist1, a basic helix-loop-helix (bHLH) transcription factor, plays a critical role in promoting EMT, tumor metastasis, cancer stemness and drug resistance [5,6]. Twist1 can form homo-dimers or hetero-dimers with other bHLH proteins, which directly regulate the transcription of EMT or stemness-related genes [22,23]. Twist1 is a key transcription factor to upregulate expression of Oct4 and Sox2 [24,25]. Expression of Twist1 is elevated in various human cancers, including cancers of lung, breast, prostate and liver [26,27,28,29,30,31,32,33]. It has been reported that Twist1 is critical for oncogene-driven cell proliferation since silencing of Twist1 induces senescence in lung adenocarcinoma cells driven by various oncogenic drivers [26]. Activation of TGF-β signaling upregulates Twist1 expression, which in turn promotes lung cancer stemness [34]. Several E3 ubiquitin ligases, including FBXL14, β-TRCP, FBXO45 and Pirh2, have been shown to bind to and degrade Twist1 protein [35,36,37]. In addition, HR23A protein, a poly-ubiquitin chain carrier, targets and degrades Twist1 to inhibit lung cancer stemness [38].
Deubiquitinating enzymes (DUBs) are a group of proteases that catalyze the cleavage of protein–ubiquitin bonds. USPs (ubiquitin-specific proteases), the largest subfamily of DUB, play critical roles in development of various malignant tumors [39,40]. USP4 is the first deubiquitinating enzyme identified in mammalian cells [41] and is frequently overexpressed in various cancers, including lung adenocarcinoma and glioblastoma [42,43]. A body of evidence indicates that USP4 functions as a potent oncogene, through regulating p53 signaling, Wnt/β-catenin and TGF-β/SMAD signaling pathways, to promote tumorigenesis and cancer development [44,45,46,47]. However, whether USP4 directly regulates lung cancer stemness remains unclear.
In this study, we demonstrate that USP4 is critically important in maintaining lung cancer cell stemness. We show that USP4 is a novel deubiquitinating enzyme of Twist1 protein. These results highlight USP4 as a critical factor in promoting lung cancer stemness and as a potentially useful lung cancer prognosis marker.
2. Results
2.1. USP4 Promotes Lung Cancer Cell Stemness
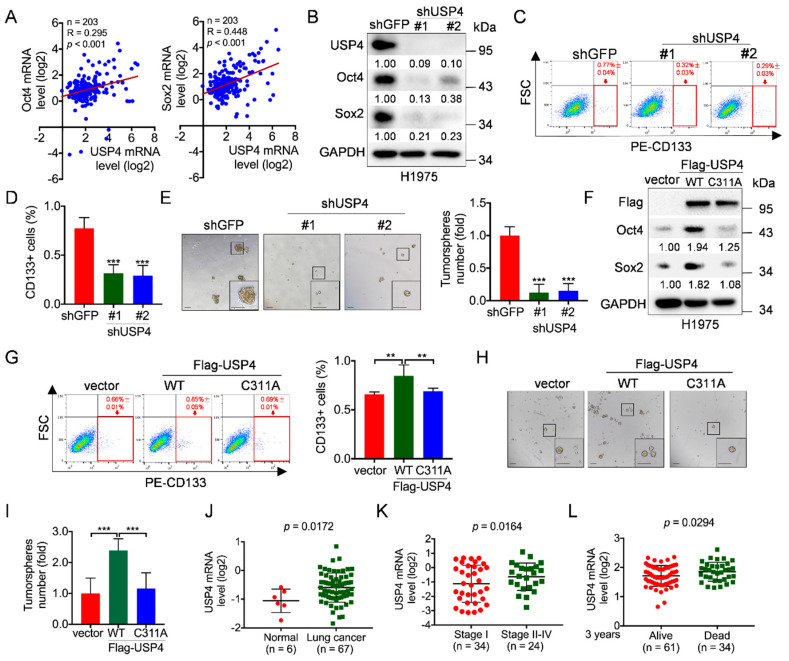
Cancer stem cells (CSCs) play a pivotal role in lung cancer development, metastasis and recurrence [4,5,6,7]. Thus, it is of great importance to understand the molecular bases by which CSCs are regulated. Given the critical roles of ubiquitin-specific proteases (USPs) in regulation of protein stability and cancer development, we aimed to identify novel USP(s) in the regulation of CSCs. Therefore, we first analyzed the correlation of expression between USPs and stemness transcript factors exemplified by Oct4/Sox2, utilizing clinical lung cancer samples from Oncomine database [48]. As shown in Figure 1A and Figure S1A–I, expression of USP1 was significantly correlated with expression of Oct4 and Sox2 in human lung cancers, in keeping with previous reports [49,50]. Notably, expression of USP4 was also significantly correlated with expression of Oct4 (R = 0.295; p < 0.001; n = 203) and Sox2 (R = 0.448; p < 0.001; n = 203), suggesting that USP4 is a potential positive regulator of lung cancer stemness.
Figure 1.
USP4 promotes lung cancer cell stemness and its high expression is correlated with human lung cancer patients. (A) The Oncomine dataset “Bhattacharjee Lung” was used to analyze Pearson correlation of USP4 and Oct4/Sox2 expression. (B–E) H1975 cells stably expressing shRNA against USP4 (shUSP4-#1 or shUSP4-#2) were subjected to (B) Western blot analyses, (C–D) FACS analyses for CD133-stained cells or (E) tumorsphere formation assay. Respective images and quantitation were shown. Data from three independent experiments in triplicates were presented as means ± SD. *** p < 0.001. Scale bar = 100 μm. (F–I) H1975 cells stably expressing Flag-USP4 or Flag-USP4C311A were subjected to (F) Western blot analyses, (G) FACS analyses for CD133-stained cells or (H–I) tumorsphere formation assay. Respective images and quantitation were shown. Data from three independent experiments in duplicates were presented as means ± SD. ** p < 0.01, *** p < 0.001. Scale bar = 100 μm. (J) The Oncomine dataset “Gaber lung” was used to analyze USP4 mRNA levels in normal human lung tissues and lung cancers. (K) The Oncomine dataset “Bild lung” was used to analyze USP4 mRNA levels in stage I or stage II-IV human lung cancers. (L) The Oncomine dataset “Raponi lung” was used to analyze USP4 mRNA levels in 3 year-alive or 3 year-dead human lung cancer patients.
Next, we aimed to confirm the relationship between USP4 and Oct4/Sox2. We used shRNAs specific for USP4 to facilitate the knockdown of USP4 in human non-small cell lung cancer (NSCLC) H1975 and A549 cells. As shown in Figure 1B and Figure S2A, silencing of USP4 significantly reduced Oct4 and Sox2 protein expression, suggesting that silencing of USP4 can inhibit lung cancer cell stemness. Indeed, silencing of USP4 significantly led to reduced population of CD133+ cells, a marker for CSCs in NSCLC, concomitant with reduced tumorsphere formation (Figure 1C–E and Figure S3A–C). Conversely, ectopic expression of wild-type USP4, but not USP4C311A mutant defective in deubiquitinating enzymatic activity, significantly upregulated Oct4 and Sox2 protein expression, concomitant with both increased population of CD133+ cells and increased capacity of tumorsphere formation (Figure 1F–I, Figure S2B and Figure S4A–C). Together, these data indicate that USP4 is a critical factor to promote lung cancer stemness, which is dependent on its deubiquitinating enzymatic activity.
These results prompted us to verify expression levels of USP4 in lung cancer. Clinical analyses of Oncomine “Gaber lung” dataset showed that USP4 mRNA levels were elevated in lung cancer specimens compared to normal tissues (Figure 1J), and analyses of Oncomine “Bild lung” dataset showed that stage II-IV lung cancer specimens had elevated USP4 mRNA levels compared to stage I lung cancer specimens (Figure 1K). Furthermore, higher levels of USP4 mRNA were significantly associated (p = 0.0294) with poor overall three-year survival of lung cancer patients (Figure 1L). Together, these findings indicate that USP4 is critically important in promoting lung cancer stemness and is associated with lung cancer clinical prognosis.
2.2. USP4 Promotes Lung Cancer Stemness Via Upregulation of Twist1 Protein Expression
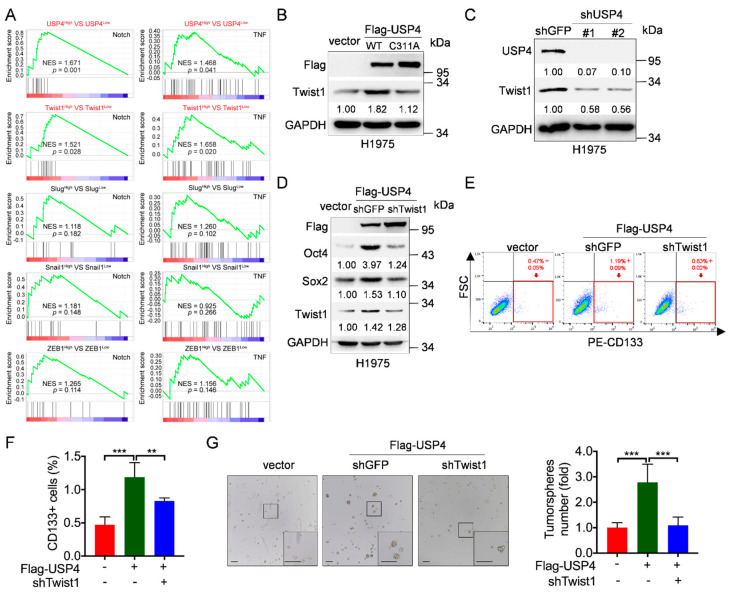
We then investigated the molecular bases by which USP4 promotes lung cancer stemness. It has been reported that EMT-associated transcriptional factors, including Twist1, Snail, Slug and ZEB1, play a critical role in lung cancer stemness [34,38]. We therefore performed Gene Set Enrichment Analyses (GSEA) on gene signatures of Notch and TNF signaling, both of which are important in maintaining lung cancer stemness [16,19]. As shown in Figure 2A, high expression of both USP4 and Twist1 was significantly associated with gene signatures of Notch and TNF signaling, suggesting that Twist1 may play a role in USP4-mediated lung cancer stemness. We then examined the effect of USP4 on Twist1 expression. As shown in Figure 2B and Figure S5A, ectopic expression of USP4, but not USP4C311A, led to a significant increase in Twist1 protein expression in H1975 cells. Conversely, silencing of USP4 dramatically reduced Twist1 protein expression in H1975 and A549 cells (Figure 2C and Figure S3A). Consistently, ectopic expression of USP4 also increased Twist1 protein expression in A549 cells (Figure S4A).
Figure 2.
USP4 promotes lung cancer stemness via upregulation of Twist1 protein expression. (A) Gene Set Enrichment Analyses (GSEA) analyzed the effects of USP4 or transcriptional factors associated with EMT (Twist1, Slug, Snail and ZEB1) on Notch signaling and TNF signaling. GSE19804 dataset was used to these analyses. NES: normalized enrichment score. (B) H1975 cells stably expressing Flag-USP4 or Flag-USP4C311A were subjected to Western blot analyses. (C) H1975 cells stably expressing shRNAs against USP4 (shUSP4-#1 or shUSP4-#2) were subjected to Western blot analyses. (D–G) H1975 cells stably expressing Flag-USP4 and either shTwist1 or shGFP were subjected to (D) Western blot analyses, (E–F) FACS analyses for CD133-stained cells or (G) tumorsphere formation assay. Respective images and quantitation were shown. Data from three independent experiments in duplicates were presented as means ± SD. ** p < 0.01, *** p < 0.001. Scale bar = 100 μm.
To investigate the causative role of Twist1 in USP4-mediated lung cancer stemness, we performed the rescuing experiments. As shown in Figure 2D, silencing of Twist1 completely rescued USP4-induced upregulation of Oct4 and Sox2 protein expression. Consistently, silencing of Twist1 significantly reversed USP4-induced increase of CD133+ cell population and tumorsphere formation (Figure 2E–G and Figure S5B). Together, these results demonstrate that Twist1 is a critical downstream effector in USP4-induced lung cancer stemness.
2.3. USP4 Is a Deubiquitinase of Twist1 Protein
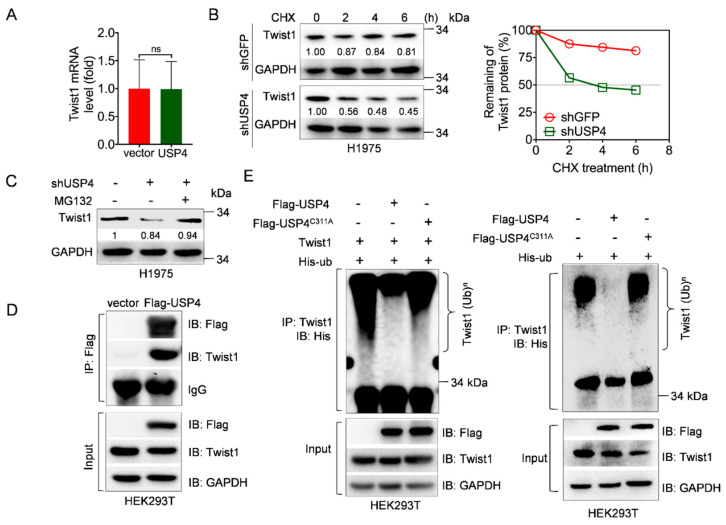
We next investigated the mechanism by which USP4 upregulates Twist1 expression. As shown in Figure 3A, ectopic expression of USP4 had little effects on the steady-state Twist1 mRNA levels. Instead, silencing of USP4 significantly shortened the half-life of Twist1 protein (Figure 3B). In addition, treatment with MG132, a well-known proteasome inhibitor, markedly rescued silencing of USP4-induced downregulation of Twist1 (Figure 3C). These results indicate that USP4 stabilizes Twist1 protein via proteasome pathway.
Figure 3.
USP4 is a deubiquitinase of Twist1 protein. (A) H1975 cells stably expressing USP4 or a vector control were subjected to Q-PCR analyses. ns stands for no significance. (B) H1975 cells stably expressing shUSP4 or shGFP were treated with 50 μg/mL cycloheximide (CHX) for an indicated time interval, and then subjected to Western blot analyses. Twist1 protein levels were quantified and the plot was presented. (C) H1975 cells stably expressing shRNAs against USP4 (shUSP4) were treated with or without 20 μM MG132 for 12 h prior to Western blot analyses. (D) HEK293T cells were transfected with Flag-USP4 expressing plasmids for 36 h. Cells were treated with 20 μM MG132 for 6 h prior to Co-IP experiments. (E) HEK293T cells were co-transfected with Twist1 and His-ubiquitin in the presence of either Flag-USP4 or Flag-USP4C311A expressing plasmids for 36 h (left panel). HEK293T cells were co-transfected with His-ubiquitin and either Flag-USP4 or Flag-USP4C311A expressing plasmids for 36 h (right panel). Cells were then treated with 20 μM MG132 for 6 h prior to immunoprecipitation (IP) and Western blot analyses.
Next, we investigated whether USP4 can function as deubiquitinating enzyme for Twist1. To this end, we first asked whether USP4 can form a stable protein complex with Twist1. Co-Immunoprecipitation assay showed that USP4 interacted with Twist1 (Figure 3D). Moreover, ectopic expression of USP4, but not USP4C311A, dramatically removed polyubiquitin of endogenous Twist1 in vivo (Figure 3E). These results indicate that USP4 is a novel deubiquitinating enzyme to protect Twist1 from proteasome-mediated degradation.
2.4. Clinical Validation of Correlation between USP4 and Twist1 Expression and Its Association with Overall Survival in Lung Cancer
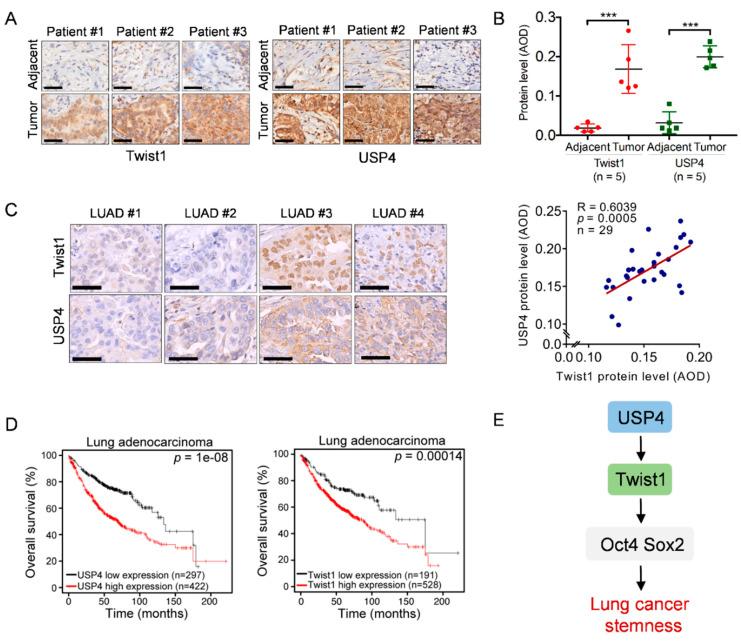
Our data indicate that USP4 stabilizes Twist1 to promote lung cancer cell stemness, which prompted us to verify the clinical relevance of USP4–Twist1 in human lung cancer. As shown in Figure 4A,B, compared to adjacent tissues, expression of both USP4 and Twist1 proteins were high in biopsy specimens of lung adenocarcinoma patients. We further examined the clinical correlation between USP4 and Twist1 using tissue microarrays of lung adenocarcinoma. A positive correlation was observed between USP4 and Twist1 expression (R = 0.6039, p = 0.0005) (Figure 4C). Clinical analysis by Kaplan–Meier dataset showed that lung cancer patients with high USP4 or Twist1 levels exhibited poor overall survival (OS) (Figure 4D). Together, these results indicate that the USP4–Twist1 axis plays a critical role in lung cancer stemness and clinical prognoses (Figure 4E).
Figure 4.
High expression of USP4 is positively correlated with Twist1 and associated with poor overall survival in human lung cancers. (A,B) Consecutive tissue slides derived from human lung adnocarcinoma (treatment-naïve) and adjacent normal tissues were subjected to immunohistochemistry (IHC) for expression of USP4 and Twist1 expression. Representative images of IHC staining were shown (A). Stainings were quantified by average optical density (AOD) (B). *** p < 0.001. Scale bar = 50 μm. (C) Consecutive tissue microarray slides derived from human lung adenocarcinoma (LUAD) were subjected to IHC analyses for Pearson correlation of USP4 and Twist1 expression. Representative images of IHC staining were shown. Stainings were quantified by average optical density (AOD). Scale bar = 50 μm. (D) Kaplan–Meier plots of overall survival of human lung cancer patients were stratified by the USP4 or Twist1 mRNA expression levels in the patient tumor samples. (E) A working model depicting that USP4 deubiquitinates and stabilizes Twist1 protein to promote lung cancer stemness.
3. Discussion
Lung cancer is a leading cause of cancer incidence and mortality worldwide [1]. Accumulating evidence indicates that CSCs are responsible for resistance to conventional therapies due to the stemness properties [51]. Several DUBs have been shown to be important in the regulation of lung cancer stemness. For instance, USP21 deubiquitinates and stabilizes Nanog protein stability [52]. USP17 and OTUD3 promote lung cancer stemness through mediating TRAF2/TRAF3 complex and stabilizing GRP78, respectively [53,54]. Herein, we show that USP4 is critically important in promoting lung cancer stemness via stabilizing Twist1 expression. This notion is supported by the evidence that ectopic expression of USP4, but not USP4C311A, significantly upregulates Oct4 and Sox2 expression, increases the population of CD133+ cells and promotes mammosphere formation, all of which can be rescued by simultaneous silencing of Twist1.
EMT transcriptional factors, Snail/Slug, Twist1 and ZEB1, are pivotal in regulation of tumor metastasis and stemness [38]. It has been reported that expression of these factors is tightly controlled by DUBs. Snail protein can be deubiquitinated and stabilized by USP26, USP11, USP10, USP37 or OTUB1 [55,56,57,58,59]. Slug can be stabilized by USP10 and DUB3 [55,60]. ZEB1 is stabilized by USP51 [61]. Twist1 protein, on the other hand, can be stabilized by deubquitinase DUB3 in response to IL-6 [35,36,37,60]. In this study, we uncover that USP4 is a novel deubquitinase of Twist1 and that silencing of Twist dramatically inhibits USP4-induced stemness of lung cancer cells, suggesting that the USP4–Twist1 axis plays a critical role in lung cancer stemness.
The biological function of USP4 in cancer development appears to be complex. It has been reported that USP4 can deubiquitinate and stabilize TRAF2/TRAF6 to inhibit TNFα-induced cancer cell migration [62]. In addition, USP4 expression could be upregulated by CircBMPR2-miR-553 to suppress breast cancer resistance to tamoxifen [63]. These results suggest a role for USP4 in growth suppression. However, USP4 is highly expressed in various cancers [42]. A wealth of evidence strongly suggests that USP4 functions as a tumor-promoting protein, as exemplified by the ability of USP4 to promote tumorigenesis via deubiquitinating and stabilizing ARF-BP1, β-catenin and TGF-βRI [47,64,65,66]. In addition, USP4 can also promote cell cycle procession and tumorigenesis by deubiquitinating SART3 and Ro52 [45,46]. Our results presented in this study provide another series of strong evidence supporting USP4 as an oncoprotein.
Importantly, our results showed clear clinical relevance, supported by the observations that expression of USP4 and Twist1 is positively correlated in lung cancers and that high expression of USP4/Twist1 is associated with poor overall survival in lung adenocarcinoma patients. Together, our findings demonstrate that USP4 deubiquitinates and stabilizes Twist1 protein to promote lung cancer stemness. Thus, targeting the USP4–Twist1 axis may represent a novel therapeutic approach for lung cancer treatment.
4. Materials and Methods
4.1. Cell Culture
Human non-small cell lung cancer H1975 cells were cultured in RPMI-1640 medium (Hyclone, Logan, UT, USA) supplemented with 10% fetal bovine serum (FBS; Hyclone, Logan, UT, USA), 100 units/mL penicillin (GIBCO, Rockville, MD, USA) and 100 μg/mL streptomycin (GIBCO, Rockville, MD, USA). Human non-small cell lung cancer A549 cells and human embryonic kidney HEK293T cells were cultured in DMEM medium supplemented with 10% fetal bovine serum (FBS; Hyclone, Logan, UT, USA), 100 units/mL penicillin (GIBCO, Rockville, MD, USA) and 100 μg/mL streptomycin (GIBCO, Rockville, MD, USA). Cells were maintained in a humidified 37 °C incubator under a 5% CO2 atmosphere.
4.2. Plasmids and Lentiviral Infection
Twist1 recombinant expressing vector (EX-U1219-Lv105) was purchased from GENECOPIEA. A pLVX-puro vector was used to generate recombinant lentiviruses expressing human USP4. The USP4C311A mutants were generated by KOD-Plus-Mutagenesis kit (SMK-101, Toyobo Osaka). Short hairpin RNA constructs targeting human USP4 or Twist1 were generated using pLKO.1-puro vector as described [67]. All plasmid constructs used in this study were confirmed by DNA sequencing. The amplification primer and specific shRNA sequences used in this study are listed below:
For Flag-USP4, Forward: 5′ GAGGATCTATTTCCGGTGAAGAGGAG
ATCTGCCGCCGCGA 3′; Reverse: 5′ TCTAGAACTAGTCTCGAGGTTA
AACCTTATCGTCGTCAT 3′. For Flag-USP4C311A, Forward: 5′ ACCGCCT
TCATGAACTCCGCTTTGC 3′; Reverse: 5′ GTTTCCCAGGTTTCCAAGTC
CACAG 3′
For anti-USP4, # 1: CCCAACTGTAAGAAGCATCAA; # 2 GCCCAGAAT
GTGCTAAGGTTT. For anti-Twist1: GCTGAGCAAGATTCAGACC
4.3. Plasmid Transfection, Lentiviral Infection and RNA Interference
Cells at 70–80% confluence were transfected with expressing plasmids by Lipofectamine 2000 transfection reagent (Invitrogen, Carlsbad, CA, USA). Recombinant lentiviruses were amplified by co-transfecting psPAX2 and pMD2.G packaging plasmids and lentiviral expressing plasmids using Lipofectamine 2000 in HEK293T. Viruses were collected at 60 h after transfection. Cells at 50% confluence were infected with recombinant lentiviruses in the presence of 10 µg/mL polybrene, followed by 12 h incubation.
4.4. Western Blot, Immunoprecipitation and Immunohistochemistry (IHC) Analyses
For Western blot analysis, cells were collected, washed with cold PBS, and resuspended in EBC250 lysis buffer (250 mM NaCl, 50 mM Tris pH 8.0, 0.5% Nonidet P-40, 50 mM NaF, 1 mM phenylmethylsulfonyl fluoride, 2 μg/mL aprotinin, and 2 μg/mL leupeptin). Equal amounts of total protein were loaded, separated by SDS-PAGE, transferred to PVDF membranes (Millipore, Darmstadt, Germany), and hybridized to an appropriate primary antibody and HRP-conjugated secondary antibody for subsequent detection by enhanced chemiluminescence. The immunoblots were quantitated by Image Lab 5.0 and normalized to the loading control GAPDH.
Immunoprecipitation and immunohistochemistry analyses were performed according to procedures described previously [68,69]. Human tumor tissue microarray slides (Cat# HLugA030PG02) were purchased from Outdo Biotech Co., (Shanghai, China). Slides were scanned by NanoZoomer (Hamamatsu, Japan) and images were quantified by integrated optical density (IOD) using Image-Pro Plus 6.0 software from Media Cybernetics Inc. (Rockville, MD, USA). Average optical density (AOD) was calculated using the formula: AOD = IOD/Area. Antibodies for USP4 (612819, 1:500 for WB, 1:100 for IHC) and Oct4 (618077, 1:1000 for WB) were purchased from zenbioscience (Chengdu, China). Antibody for Sox2 (AB5603, 1:500 for WB) was purchased from Merck (Darmstadt, Germany). Antibodies for GAPDH (AB0037, 1:1000 for WB) and Twist1 (CY2578, 1:1000 for WB, 1:200 for IHC) were purchased from Abways (Shanghai, China). Antibody for Flag (14793, 1:1000 for WB) was purchased from Cell Signaling Technology (Danvers, MA, USA).
4.5. Tumorspheres Formation Assay
20,000 cells were seeded on the 6-ultral-low-adherient plates and grown in DMEM/F12 medium containing 5 mg/mL insulin (Sigma, Taufkirchen, Germany), 2% B27 (Invitrogen, Carlsbad, CA, USA), 20 ng/mL epidermal growth factor (R&D systems), 100 units/mL penicillin (GIBCO, Rockville, MD, USA) and 100 μg/mL streptomycin (GIBCO, Rockville, MD, USA) for 7 days. Tumorspheres were defined to the diameter of spheres that were greater than 50 μm. The number of tumorspheres of each well were counted, normalized and presented to reflect tumorsphere formation ability.
4.6. Quantitative PCR
Total RNA was extracted from cells using RNeasy plus Mini Kit (cat# 74134, QIAGEN, Germantown, MD, USA) and reverse-transcribed according to the manufacturer’s instructions. Q-PCR was performed for Twist1 and GAPDH. The Q-PCR reactions were carried out in CFX-960 Real time PCR System (Bio-Rad, CA, USA) and using Bio-Rad SoFast Eva-Green Supermix (Bio-Rad, Hercules, CA, USA) according to the manufacturer’s instructions. Relative quantitation values were calculated using the ΔΔCt method. Q-PCR primer for Twist1, Forward: GTCCGCAGTCTTACGAGGAG; Reverse: GCTTGAGGGTC
TGAATCTTGCT. For GAPDH, Forward: TGGACTCCACGACGTACTCA; Reverse: AATCCCATCACCATCTTCCA.
4.7. Flow Cytometry Assay
To analyze CD133+ cell population, cells were harvested and washed twice with PBS, then were re-suspended in staining buffer (Bio-Rad), and cell numbers were counted. Cells were incubated with Phycoerythrin (PE) conjugated anti-CD133 antibody (12-1338-42, Invitrogen, Carlsbad, CA, USA) or PE conjugated IgG1 kappa isotype control antibody (12-4714-81, Invitrogen, Carlsbad, CA, USA) for 30 min at 4 °C. Cells were then washed twice with staining buffer and re-suspended by staining buffer, and analyzed by FACS machine (Calibur, Bio-Rad, Hercules, CA, USA). The data of flow cytometry assay were analyzed by FlowjoX software.
4.8. Clinical Relevance Analysis
Pearson correlation of USPs and Oct4/Sox2 expression was analyzed using Oncomine dataset “Bhattacharjee Lung”. The mRNA levels of USP4 in normal human lung tissues and lung cancers were analyzed using Oncomine dataset “Gaber lung”. The mRNA levels of USP4 in stage I or stage II–IV human lung cancers were analyzed using Oncomine dataset “Bild lung”. The mRNA levels of USP4 in 3 year-alive or 3 year-dead human lung cancer patients were analyzed using Oncomine dataset “Raponi lung”. Kaplan–Meier plots of overall survival of human lung cancer patients stratified by the USP4 or Twist1 mRNA expression levels were analyzed using the Kaplan–Meier survival datasets. The high and low groups were defined by the best cutoff.
Gene signature enrichment analyses (GSEA) were performed by GSEA software (v 4.0.3, UC San Diego and Broad Institute Inc., San Diego, CA, USA) based on GSE19804 dataset available on GEO database. The mean value of each gene expression involved in the assay was used to divided into “high” and “low” groups. Notch and TNF signaling related genes were described in Figure S6.
4.9. Statistical Analysis
GraphPad Prism 6.0 (GraphPad Software Incorporation, San Diego, CA, USA) was used for data recording, collection, processing, and calculation. All cell-based experiments were performed at least three times in duplicates. Data were presented as means ± SD. Quantitative data were analyzed statistically using Student’s t-test to assess significance.
5. Conclusions
Our results demonstrate that deubiquitinase USP4 promotes lung cancer cell stemness via upregulation of Twist1, Oct4 and Sox2 expression. Mechanistically, we have identified USP4 as a novel deubiquitinase of Twist1. In addition, USP4 expression is elevated in human lung cancer specimens and is positively correlated with Twist1 expression. High expression of USP4/Twist1 is associated with poor clinical outcomes of lung cancer patients. Thus, targeting USP4 may represent a novel therapeutic approach for lung cancer treatment.
Acknowledgments
The authors thank Lingqiang Zhang (Beijing Institute of Lifeomics, Beijing, China) for providing ubiquitin plasmid. The authors thank members of Z.-X.J.X. laboratory for stimulating discussions during the course of study.
Supplementary Materials
The following are available online at https://www.mdpi.com/2072-6694/12/6/1582/s1, Figure S1: Correlation of USPs and Oct4/Sox2 expression in lung adenocarcinoma; Figure S2: USP4 significantly regulates proteins expression of Oct4 and Sox2; Figure S3: Silencing of USP4 downregulates Twist1, Oct4 and Sox2 expression and inhibits stemness of lung cancer A549 cells; Figure S4: Ectopic expression of USP4 upregulates Twist1, Oct4 and Sox2 expression and promotes stemness of lung cancer A549 cells; Figure S5: Ectopic expression of USP4 increases Oct4 and Sox2 protein levels via upregulation of Twist1 protein expression; Figure S6: Genes related to TNF and Notch signaling used for Gene signature enrichment analysis.
Author Contributions
Z.-X.J.X., M.N. and F.L. conceived the project and performed the project planning; F.L., Q.H., T.H., J.X., Y.Y., S.X., L.D., M.F. and R.G. performed research; M.N., Z.-X.J.X. and F.L. analyzed data and wrote the manuscript. All authors have read and agreed to the published version of the manuscript.
Funding
This research was supported by National Natural Science Foundation of China, grant number 31701242, 81520108020, 81830108 and 81861148031, National Key R&D Program of China, grant number 2018YFC2000100, and the Fundamental Research Funds for the Central Universities, grant number SCU2019D013, SCU2017D006, SCU2018D006.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Siegel R.L., Miller K.D., Jemal A. Cancer statistics, 2019. CA Cancer J. Clin. 2019;69:7–34. doi: 10.3322/caac.21551. [DOI] [PubMed] [Google Scholar]
- 2.Melosky B. Current treatment algorithms for patients with metastatic non-small cell, non-squamous lung cancer. Front. Oncol. 2017;7:38. doi: 10.3389/fonc.2017.00038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nikolaou M., Pavlopoulou A., Georgakilas A.G., Kyrodimos E. The challenge of drug resistance in cancer treatment: A current overview. Clin. Exp. Metastasis. 2018;35:309–318. doi: 10.1007/s10585-018-9903-0. [DOI] [PubMed] [Google Scholar]
- 4.Eramo A., Lotti F., Sette G., Pilozzi E., Biffoni M., di Virgilio A., Conticello C., Ruco L., Peschle C., de Maria R. Identification and expansion of the tumorigenic lung cancer stem cell population. Cell Death Differ. 2008;15:504–514. doi: 10.1038/sj.cdd.4402283. [DOI] [PubMed] [Google Scholar]
- 5.Sullivan J.P., Minna J.D., Shay J.W. Evidence for self-renewing lung cancer stem cells and their implications in tumor initiation, progression, and targeted therapy. Cancer Metastasis Rev. 2010;29:61–72. doi: 10.1007/s10555-010-9216-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chiou S.H., Wang M.L., Chou Y.T., Chen C.J., Hong C.F., Hsieh W.J., Chang H.T., Chen Y.S., Lin T.W., Hsu H.S., et al. Coexpression of Oct4 and Nanog enhances malignancy in lung adenocarcinoma by inducing cancer stem cell-like properties and epithelial-mesenchymal transdifferentiation. Cancer Res. 2010;70:10433–10444. doi: 10.1158/0008-5472.CAN-10-2638. [DOI] [PubMed] [Google Scholar]
- 7.Singh A., Settleman J. EMT, cancer stem cells and drug resistance: An emerging axis of evil in the war on cancer. Oncogene. 2010;29:4741–4751. doi: 10.1038/onc.2010.215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chen Y.C., Hsu H.S., Chen Y.W., Tsai T.H., How C.K., Wang C.Y., Hung S.C., Chang Y.L., Tsai M.L., Lee Y.Y., et al. Oct-4 expression maintained cancer stem-like properties in lung cancer-derived CD133-positive cells. PLoS ONE. 2008;3:e2637. doi: 10.1371/journal.pone.0002637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Nishino M., Ozaki M., Hegab A.E., Hamamoto J., Kagawa S., Arai D., Yasuda H., Naoki K., Soejima K., Saya H., et al. Variant CD44 expression is enriching for a cell population with cancer stem cell-like characteristics in human lung adenocarcinoma. J. Cancer. 2017;8:1774–1785. doi: 10.7150/jca.19732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.De Aberasturi A.L., Redrado M., Villalba M., Larzabal L., Pajares M.J., Garcia J., Evans S.R., Garcia-Ros D., Bodegas M.E., Lopez L., et al. TMPRSS4 induces cancer stem cell-like properties in lung cancer cells and correlates with ALDH expression in NSCLC patients. Cancer Lett. 2016;370:165–176. doi: 10.1016/j.canlet.2015.10.012. [DOI] [PubMed] [Google Scholar]
- 11.Ho M.M., Ng A.V., Lam S., Hung J.Y. Side population in human lung cancer cell lines and tumors is enriched with stem-like cancer cells. Cancer Res. 2007;67:4827–4833. doi: 10.1158/0008-5472.CAN-06-3557. [DOI] [PubMed] [Google Scholar]
- 12.Bora-Singhal N., Perumal D., Nguyen J., Chellappan S. Gli1-mediated regulation of Sox2 facilitates self-renewal of stem-like cells and confers resistance to EGFR Inhibitors in non-small cell lung cancer. Neoplasia. 2015;17:538–551. doi: 10.1016/j.neo.2015.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Hu F., Li C., Zheng X., Zhang H., Shen Y., Zhou L., Yang X., Han B., Zhang X. Lung adenocarcinoma resistance to therapy with EGFRtyrosine kinase inhibitors is related to increased expression of cancer stem cell markers SOX2, OCT4 and Nanog. Oncol. Rep. 2020;43:727–735. doi: 10.3892/or.2019.7454. [DOI] [PubMed] [Google Scholar]
- 14.Liu K., Xu S.H., Chen Z., Zeng Q.X., Li Z.J., Chen Z.M. TRPM7 overexpression enhances the cancer stem cell-like and metastatic phenotypes of lung cancer through modulation of the Hsp90alpha/uPA/MMP2 signaling pathway. BMC Cancer. 2018;18:1167. doi: 10.1186/s12885-018-5050-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jen J., Liu C.Y., Chen Y.T., Wu L.T., Shieh Y.C., Lai W.W., Wang Y.C. Oncogenic zinc finger protein ZNF322A promotes stem cell-like properties in lung cancer through transcriptional suppression of c-Myc expression. Cell Death Differ. 2019;26:1283–1298. doi: 10.1038/s41418-018-0204-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Hassan K.A., Wang L., Korkaya H., Chen G., Maillard I., Beer D.G., Kalemkerian G.P., Wicha M.S. Notch pathway activity identifies cells with cancer stem cell-like properties and correlates with worse survival in lung adenocarcinoma. Clin. Cancer Res. 2013;19:1972–1980. doi: 10.1158/1078-0432.CCR-12-0370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Giroux-Leprieur E., Costantini A., Ding V.W., He B. Hedgehog signaling in lung cancer: From oncogenesis to cancer treatment resistance. Int. J. Mol. Sci. 2018;19:2835. doi: 10.3390/ijms19092835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liu T., Wu X., Chen T., Luo Z., Hu X. Downregulation of DNMT3A by miR-708-5p inhibits lung cancer stem cell-like phenotypes through repressing wnt/beta-catenin signaling. Clin. Cancer Res. 2018;24:1748–1760. doi: 10.1158/1078-0432.CCR-17-1169. [DOI] [PubMed] [Google Scholar]
- 19.Lu C.-H., Yeh D.-W., Lai C.-Y., Liu Y.-L., Huang L.-R., Lee A.Y.-L., Jin S.L.C., Chuang T.-H. USP17 mediates macrophage-promoted inflammation and stemness in lung cancer cells by regulating TRAF2/TRAF3 complex formation. Oncogene. 2018;37:6327–6340. doi: 10.1038/s41388-018-0411-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yang L., Shi P., Zhao G., Xu J., Peng W., Zhang J., Zhang G., Wang X., Dong Z., Chen F., et al. Targeting cancer stem cell pathways for cancer therapy. Signal Transduct Target Ther. 2020;5:8. doi: 10.1038/s41392-020-0110-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ranji P., Kesejini T.S., Saeedikhoo S., Alizadeh A.M. Targeting cancer stem cell-specific markers and/or associated signaling pathways for overcoming cancer drug resistance. Tumour Biol. 2016;37:13059–13075. doi: 10.1007/s13277-016-5294-5. [DOI] [PubMed] [Google Scholar]
- 22.Maia A.M., da Silva J.H., Mencalha A.L., Caffarena E.R., Abdelhay E. Computational modeling of the bHLH domain of the transcription factor TWIST1 and R118C, S144R and K145E mutants. BMC Bioinform. 2012;13:184. doi: 10.1186/1471-2105-13-184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Zhu Q.Q., Ma C., Wang Q., Song Y., Lv T. The role of TWIST1 in epithelial-mesenchymal transition and cancers. Tumour Biol. 2016;37:185–197. doi: 10.1007/s13277-015-4450-7. [DOI] [PubMed] [Google Scholar]
- 24.Hata T., Rajabi H., Yamamoto M., Jin C., Ahmad R., Zhang Y., Kui L., Li W., Yasumizu Y., Hong D., et al. Targeting MUC1-C inhibits TWIST1 signaling in triple-negative breast cancer. Mol. Cancer Ther. 2019;18:1744–1754. doi: 10.1158/1535-7163.MCT-19-0156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Izadpanah M.H., Abbaszadegan M.R., Fahim Y., Forghanifard M.M. Ectopic expression of TWIST1 upregulates the stemness marker OCT4 in the esophageal squamous cell carcinoma cell line KYSE30. Cell Mol. Biol. Lett. 2017;22:33. doi: 10.1186/s11658-017-0065-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Burns T.F., Dobromilskaya I., Murphy S.C., Gajula R.P., Thiyagarajan S., Chatley S.N., Aziz K., Cho Y.J., Tran P.T., Rudin C.M. Inhibition of TWIST1 leads to activation of oncogene-induced senescence in oncogene-driven non-small cell lung cancer. Mol. Cancer Res. 2013;11:329–338. doi: 10.1158/1541-7786.MCR-12-0456. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hung J.J., Yang M.H., Hsu H.S., Hsu W.H., Liu J.S., Wu K.J. Prognostic significance of hypoxia-inducible factor-1alpha, TWIST1 and snail expression in resectable non-small cell lung cancer. Thorax. 2009;64:1082–1089. doi: 10.1136/thx.2009.115691. [DOI] [PubMed] [Google Scholar]
- 28.Li Q.Q., Xu J.D., Wang W.J., Cao X.X., Chen Q., Tang F., Chen Z.Q., Liu X.P., Xu Z.D. Twist1-mediated adriamycin-induced epithelial-mesenchymal transition relates to multidrug resistance and invasive potential in breast cancer cells. Clin. Cancer Res. 2009;15:2657–2665. doi: 10.1158/1078-0432.CCR-08-2372. [DOI] [PubMed] [Google Scholar]
- 29.Ai L., Kim W.J., Alpay M., Tang M., Pardo C.E., Hatakeyama S., May W.S., Kladde M.P., Heldermon C.D., Siegel E.M., et al. TRIM29 suppresses TWIST1 and invasive breast cancer behavior. Cancer Res. 2014;74:4875–4887. doi: 10.1158/0008-5472.CAN-13-3579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Shiota M., Zardan A., Takeuchi A., Kumano M., Beraldi E., Naito S., Zoubeidi A., Gleave M.E. Clusterin mediates TGF-beta-induced epithelial-mesenchymal transition and metastasis via Twist1 in prostate cancer cells. Cancer Res. 2012;72:5261–5272. doi: 10.1158/0008-5472.CAN-12-0254. [DOI] [PubMed] [Google Scholar]
- 31.Cho K.H., Jeong K.J., Shin S.C., Kang J., Park C.G., Lee H.Y. STAT3 mediates TGF-beta1-induced TWIST1 expression and prostate cancer invasion. Cancer Lett. 2013;336:167–173. doi: 10.1016/j.canlet.2013.04.024. [DOI] [PubMed] [Google Scholar]
- 32.Sun T., Zhao N., Zhao X.L., Gu Q., Zhang S.W., Che N., Wang X.H., Du J., Liu Y.X., Sun B.C. Expression and functional significance of Twist1 in hepatocellular carcinoma: Its role in vasculogenic mimicry. Hepatology. 2010;51:545–556. doi: 10.1002/hep.23311. [DOI] [PubMed] [Google Scholar]
- 33.Wushou A., Hou J., Zhao Y.J., Shao Z.M. Twist-1 up-regulation in carcinoma correlates to poor survival. Int. J. Mol. Sci. 2014;15:21621–21630. doi: 10.3390/ijms151221621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Giuseppe P., Virginia T., Rosa C., Renato F., Aantonello L.R., Eleonora L., Nicola M., Francesca P., Nicola N., Gaetano R. Epithelial to mesenchymal transition by TGFβ-1 induction increases stemness characteristics in primary non small cell lung cancer cell line. PloS ONE. 2011;6:e21548. doi: 10.1371/journal.pone.0021548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Yang-Hartwich Y., Tedja R., Roberts C.M., Goodner-Bingham J., Cardenas C., Gurea M., Sumi N.J., Alvero A.B., Glackin C.A., Mor G. p53-Pirh2 Complex Promotes Twist1 Degradation and Inhibits EMT. Mol. Cancer Res. 2019;17:153–164. doi: 10.1158/1541-7786.MCR-18-0238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Xu M., Zhu C., Zhao X., Chen C., Zhang H., Yuan H., Deng R., Dou J., Wang Y., Huang J., et al. Atypical ubiquitin E3 ligase complex Skp1-Pam-Fbxo45 controls the core epithelial-to-mesenchymal transition-inducing transcription factors. Oncotarget. 2015;6:979–994. doi: 10.18632/oncotarget.2825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lander R., Nordin K., LaBonne C. The F-box protein Ppa is a common regulator of core EMT factors Twist, Snail, Slug, and Sip1. J. Cell Biol. 2011;194:17–25. doi: 10.1083/jcb.201012085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Yu C.Y., Liu B.H., Tang S.Y., Liang R.Y., Hsu K.H., Chuang S.M. HR23A-knockdown lung cancer cells exhibit epithelial-to-mesenchymal transition and gain stemness properties through increased Twist1 stability. Biochim. Biophys. Acta Mol. Cell Res. 2019;1866:118537. doi: 10.1016/j.bbamcr.2019.118537. [DOI] [PubMed] [Google Scholar]
- 39.Dikic I. Proteasomal and autophagic degradation systems. Annu. Rev. Biochem. 2017;86:193–224. doi: 10.1146/annurev-biochem-061516-044908. [DOI] [PubMed] [Google Scholar]
- 40.McClurg U.L., Robson C.N. Deubiquitinating enzymes as oncotargets. Oncotarget. 2015;6:9657–9668. doi: 10.18632/oncotarget.3922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wada K., Kamitani T. UnpEL/Usp4 is ubiquitinated by Ro52 and deubiquitinated by itself. Biochem. Biophys. Res. Commun. 2006;342:253–258. doi: 10.1016/j.bbrc.2006.01.144. [DOI] [PubMed] [Google Scholar]
- 42.Gray D.A., Inazawa J., Gupta K., Wong A., Ueda R., Takahashi T. Elevated expression of Unph, a proto-oncogene at 3p21.3, in human lung tumors. Oncogene. 1995;10:2179–2183. [PubMed] [Google Scholar]
- 43.Zhou Y., Liang P., Ji W., Yu Z., Chen H., Jiang L. Ubiquitin-specific protease 4 promotes glioblastoma multiforme via activating ERK pathway. Onco Targets Ther. 2019;12:1825–1839. doi: 10.2147/OTT.S176582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Cheng J., Guo J., North B.J., Wang B., Cui C.P., Li H., Tao K., Zhang L., Wei W. Functional analysis of deubiquitylating enzymes in tumorigenesis and development. Biochim. Biophys. Acta Rev. Cancer. 2019;1872:188312. doi: 10.1016/j.bbcan.2019.188312. [DOI] [PubMed] [Google Scholar]
- 45.Wada K., Tanji K., Kamitani T. Oncogenic protein UnpEL/Usp4 deubiquitinates Ro52 by its isopeptidase activity. Biochem. Biophys. Res. Commun. 2006;339:731–736. doi: 10.1016/j.bbrc.2005.11.076. [DOI] [PubMed] [Google Scholar]
- 46.Song E.J., Werner S.L., Neubauer J., Stegmeier F., Aspden J., Rio D., Harper J.W., Elledge S.J., Kirschner M.W., Rape M. The Prp19 complex and the Usp4Sart3 deubiquitinating enzyme control reversible ubiquitination at the spliceosome. Genes. Dev. 2010;24:1434–1447. doi: 10.1101/gad.1925010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhang L., Zhou F., Drabsch Y., Gao R., Snaar-Jagalska B.E., Mickanin C., Huang H., Sheppard K.A., Porter J.A., Lu C.X., et al. USP4 is regulated by AKT phosphorylation and directly deubiquitylates TGF-beta type I receptor. Nat. Cell Biol. 2012;14:717–726. doi: 10.1038/ncb2522. [DOI] [PubMed] [Google Scholar]
- 48.Bhattacharjee A., Richards W.G., Staunton J., Li C., Monti S., Vasa P., Ladd C., Beheshti J., Bueno R., Gillette M., et al. Classification of human lung carcinomas by mRNA expression profiling reveals distinct adenocarcinoma subclasses. Proc. Natl. Acad. Sci. USA. 2001;98:13790–13795. doi: 10.1073/pnas.191502998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Das D.S., Das A., Ray A., Song Y., Samur M.K., Munshi N.C., Chauhan D., Anderson K.C. Blockade of deubiquitylating enzyme USP1 inhibits DNA repair and triggers apoptosis in multiple myeloma cells. Clin. Cancer Res. 2017;23:4280–4289. doi: 10.1158/1078-0432.CCR-16-2692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Jeon H.M., Sohn Y.W., Oh S.Y., Kim S.H., Beck S., Kim S., Kim H. ID4 imparts chemoresistance and cancer stemness to glioma cells by derepressing miR-9*-mediated suppression of SOX2. Cancer Res. 2011;71:3410–3421. doi: 10.1158/0008-5472.CAN-10-3340. [DOI] [PubMed] [Google Scholar]
- 51.Shibue T., Weinberg R.A. EMT, CSCs, and drug resistance: The mechanistic link and clinical implications. Nat. Rev. Clin. Oncol. 2017;14:611–629. doi: 10.1038/nrclinonc.2017.44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Liu X., Yao Y., Ding H., Han C., Chen Y., Zhang Y., Wang C., Zhang X., Zhang Y., Zhai Y., et al. USP21 deubiquitylates Nanog to regulate protein stability and stem cell pluripotency. Signal Transduct. Target. Ther. 2016;1:16024. doi: 10.1038/sigtrans.2016.24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Lu C.H., Yeh D.W., Lai C.Y., Liu Y.L., Huang L.R., Lee A.Y., Jin S.C., Chuang T.H. Correction: USP17 mediates macrophage-promoted inflammation and stemness in lung cancer cells by regulating TRAF2/TRAF3 complex formation. Oncogene. 2019;38:5742–5743. doi: 10.1038/s41388-019-0831-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Du T., Li H., Fan Y., Yuan L., Guo X., Zhu Q., Yao Y., Li X., Liu C., Yu X., et al. The deubiquitylase OTUD3 stabilizes GRP78 and promotes lung tumorigenesis. Nat. Commun. 2019;10:2914. doi: 10.1038/s41467-019-10824-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Ouchida A.T., Kacal M., Zheng A., Ambroise G., Zhang B., Norberg E., Vakifahmetoglu-Norberg H. USP10 regulates the stability of the EMT-transcription factor Slug/SNAI2. Biochem. Biophys. Res. Commun. 2018;502:429–434. doi: 10.1016/j.bbrc.2018.05.156. [DOI] [PubMed] [Google Scholar]
- 56.Zhou H., Liu Y., Zhu R., Ding F., Cao X., Lin D., Liu Z. OTUB1 promotes esophageal squamous cell carcinoma metastasis through modulating Snail stability. Oncogene. 2018;37:3356–3368. doi: 10.1038/s41388-018-0224-1. [DOI] [PubMed] [Google Scholar]
- 57.Cai J., Li M., Wang X., Li L., Li Q., Hou Z., Jia H., Liu S. USP37 promotes lung cancer cell migration by stabilizing snail protein via deubiquitination. Front. Genet. 2019;10:1324. doi: 10.3389/fgene.2019.01324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Li L., Zhou H., Zhu R., Liu Z. USP26 promotes esophageal squamous cell carcinoma metastasis through stabilizing Snail. Cancer Lett. 2019;448:52–60. doi: 10.1016/j.canlet.2019.02.007. [DOI] [PubMed] [Google Scholar]
- 59.Wang W., Wang J., Yan H., Zhang K., Liu Y. Upregulation of USP11 promotes epithelialtomesenchymal transition by deubiquitinating Snail in ovarian cancer. Oncol. Rep. 2019;41:1739–1748. doi: 10.3892/or.2018.6924. [DOI] [PubMed] [Google Scholar]
- 60.Lin Y., Wang Y., Shi Q., Yu Q., Liu C., Feng J., Deng J., Evers B.M., Zhou B.P., Wu Y. Stabilization of the transcription factors slug and twist by the deubiquitinase dub3 is a key requirement for tumor metastasis. Oncotarget. 2017;8:75127–75140. doi: 10.18632/oncotarget.20561. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Zhou Z., Zhang P., Hu X., Kim J., Yao F., Xiao Z., Zeng L., Chang L., Sun Y., Ma L. USP51 promotes deubiquitination and stabilization of ZEB1. Am. J. Cancer Res. 2017;7:2020–2031. [PMC free article] [PubMed] [Google Scholar]
- 62.Xiao N., Li H., Luo J., Wang R., Chen H., Chen J., Wang P. Ubiquitin-specific protease 4 (USP4) targets TRAF2 and TRAF6 for deubiquitination and inhibits TNFalpha-induced cancer cell migration. Biochem. J. 2012;441:979–986. doi: 10.1042/BJ20111358. [DOI] [PubMed] [Google Scholar]
- 63.Liang Y., Song X., Li Y., Ma T., Su P., Guo R., Chen B., Zhang H., Sang Y., Liu Y., et al. Targeting the circBMPR2/miR-553/USP4 Axis as a potent therapeutic approach for breast cancer. Mol. Ther. Nucleic Acids. 2019;17:347–361. doi: 10.1016/j.omtn.2019.05.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Zhang X., Berger F.G., Yang J., Lu X. USP4 inhibits p53 through deubiquitinating and stabilizing ARF-BP1. EMBO J. 2011;30:2177–2189. doi: 10.1038/emboj.2011.125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Li Z., Hao Q., Luo J., Xiong J., Zhang S., Wang T., Bai L., Wang W., Chen M., Wang W., et al. USP4 inhibits p53 and NF-kappaB through deubiquitinating and stabilizing HDAC2. Oncogene. 2016;35:2902–2912. doi: 10.1038/onc.2015.349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Yun S.I., Kim H.H., Yoon J.H., Park W.S., Hahn M.J., Kim H.C., Chung C.H., Kim K.K. Ubiquitin specific protease 4 positively regulates the WNT/beta-catenin signaling in colorectal cancer. Mol. Oncol. 2015;9:1834–1851. doi: 10.1016/j.molonc.2015.06.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Bergholz J., Zhang Y., Wu J., Meng L., Walsh E.M., Rai A., Sherman M.Y., Xiao Z.X. DeltaNp63alpha regulates Erk signaling via MKP3 to inhibit cancer metastasis. Oncogene. 2014;33:212–224. doi: 10.1038/onc.2012.564. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Yi Y., Chen D., Ao J., Sun S., Wu M., Li X., Bergholz J., Zhang Y., Xiao Z.X. Metformin promotes AMP-activated protein kinase-independent suppression of DeltaNp63alpha protein expression and inhibits cancer cell viability. J. Biol. Chem. 2017;292:5253–5261. doi: 10.1074/jbc.M116.769141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Chen Y., Li Y., Peng Y., Zheng X., Fan S., Yi Y., Zeng P., Chen H., Kang H., Zhang Y., et al. DeltaNp63alpha down-regulates c-Myc modulator MM1 via E3 ligase HERC3 in the regulation of cell senescence. Cell Death Differ. 2018;25:2118–2129. doi: 10.1038/s41418-018-0132-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.