Abstract
Background-subtracted fast-scan cyclic voltammetry (FSCV) provides a method for detecting molecular fluctuations with high spatiotemporal resolution in the brain of awake and behaving animals. The rapid scan rates generate large background currents that are subtracted to reveal changes in analyte concentration. Although these background currents are relatively stable, small changes do occur over time. These changes, referred to as electrochemical drift, result in background-subtraction artifacts that constrain the utility of FSCV, particularly when quantifying chemical changes that gradually occur over long measurement times (minutes). The voltammetric features of electrochemical drift are varied and can span the entire potential window, potentially obscuring the signal from any targeted analyte. We present a straightforward method for extending the duration of a single FSCV recording window. First, we have implemented voltammetric waveforms in pairs that consist of a smaller triangular sweep followed by a conventional voltammetric scan. The initial, abbreviated waveform is used to capture drift information that can serve as a predictor for the contribution of electrochemical drift to the subsequent full voltammetric scan using partial-least-squares regression (PLSR). This double-waveform partial-least-squares regression (DW-PLSR) paradigm permits reliable subtraction of the drift component to the voltammetric data. Here, DW-PLSR is used to improve quantification of adenosine, dopamine, and hydrogen peroxide fluctuations occurring >10 min from the initial background position, both in vitro and in vivo. The results demonstrate that DW-PLSR is a powerful tool for evaluating and interpreting both rapid (seconds) and gradual (minutes) chemical changes captured in FSCV recordings over extended durations.
Keywords: voltammetry, striatum, multivariate, capacitance, carbon fiber, microelectrode, dopamine
Graphical Abstract
Introduction
Fast-scan cyclic voltammetry (FSCV) at carbon-fiber microelectrodes (CFME) is a powerful method for assessing rapid neurochemical dynamics in biological preparations ranging from single cells to freely moving animals.1–3 When combined with pharmacological or behavioral paradigms, this approach has provided remarkable insight into the molecular mechanisms underlying goal-directed behavior.4–7 Scan rates in the hundreds of volts per sec range have allowed sensitive, real-time measurements with molecular specificity for a wide variety of both electroactive and non-electroactive (through enzyme-modified electrodes) neurochemicals.8–11 However, the rapid scan rates generate charging currents that dwarf the faradaic currents generated by target analytes. This necessitates the use of background subtraction to reveal faradaic currents generated by neurochemicals present at the electrode surface. As a result, the duration of the measurement window (from a single reference point) is limited, generally to about 90 sec. After this point, background instability, or electrochemical drift, leads to subtraction artifacts. These complicate the evaluation of FSCV recordings, especially in intact animals.12–13
Drift is observed in practically all electrochemical systems ranging from pH probes to the electrospray emitters used in mass spectrometry.14–15 In FSCV, drift signals are diverse and stem from dynamic surface phenomena that predominately impact the double layer at the carbon electrode surface, shifting impedance. As a result, when sampling over an extended recording period (minutes – hours), a new background position must be selected for subtraction approximately every 90 sec. This limits the duration of a stable measurement window, and masks the detection of more gradual changes in neurotransmitter tone. It is important that the subtracted background signal is selected appropriately, because many neurochemical fluctuations are seemingly random and can be difficult to identify in the presence of drift or other interferents. As such, the use of traditional FSCV has been largely limited to studying phasic neurochemical events associated with some prominent external stimulus. This is generally accomplished over short measurement windows, precluding the study of gradual changes in analyte concentration.
Various strategies have been investigated to account for this problem. Early work by Millar and colleagues aimed to remove differential capacitance effects by characterizing non-faradiac and faradaic responses using paired voltammetric waveforms.16 Frequency domain analyses have also proved useful, as non-faradaic contributions can be isolated into the fundamental waveform frequency.17 More recently, an approach employing charge balanced waveforms was utilized to extend the measurement time window and evaluate changes in “tonic” extracellular dopamine; however, this required a significant reduction in temporal resolution.18 Fast-scan controlled adsorption voltammetry (FSCAV) is another approach that has been used to monitor resting levels of an analyte.19 It employs an alternating series of high and low frequency voltammetric pulses to manipulate adsorption at the electrode surface; however, this also requires a substantial reduction in temporal resolution. Wightman and colleagues developed a convolution-based approach that incorporates a small-amplitude potential step to sample the impedance state of the electrode before each sweep of the triangular voltammetric waveform.20 Finally, attempts have been made to extend the FSCV measurement window by using principal component regression (PCR) to quantify electrochemical drift for its removal from the overall signal, but this approach has had difficulty accounting for the wide variety of drift signals.12, 21–22
Previously, our lab developed double-waveform partial-least-squares regression (DW-PLSR) to remove shifts in pH (ΔpH) and dopamine (DA) signals from in vivo recordings that targeted detection of hydrogen peroxide (H2O2).23 In that work, voltammetric waveforms were applied in pairs. A smaller triangular sweep that excluded the redox window for the analyte of interest was scanned immediately prior to each conventional voltammetric sweep. Data collected in the smaller scan described the interferents, and also served as a PLSR predictor for the interfering signals collected with the full waveform immediately thereafter. This approach enabled successful removal of interfering signals (pH and DA), improving quantification of H2O2. Herein, a DW-PLSR strategy is used to predict and subtract electrochemical drift from extended FSCV recordings containing H2O2, DA, and adenosine fluctuations. Furthermore, we have demonstrated in vitro that drift signals from a set of electrodes can be used to create a model for drift removal that is not specific to a given electrode. The DW-PLSR model discriminates electrochemical drift from targeted analytes both in vitro and in vivo, and serves as a reliable means to simultaneously evaluate multiple neurochemical fluctuations at the same sensor, from a single reference point. This permits evaluation of concentration changes occurring on both the second and minute time scales, alleviating critical experimental and analytical constraints associated with the practice of FSCV.
Materials and Methods
Data Acquisition:
The double triangular waveform consisted of a small and large cyclic waveform ranging from −0.4 V to +0.3–0.8 V and −0.4 V to +1.4 V, respectively. This pair of triangular sweeps was applied using a scan rate of 400 V s−1 and separated by a 12 ms hold at −0.4 V. This waveform was applied with a frequency of 10 Hz, sampling at ∼37–50 kHz, using either a custom-built instrument for potential application and current transduction (Universal Electrochemistry Instrument, University of North Carolina at Chapel Hill, Department of Chemistry, Electronics Facility) or a WaveNeuro potentiostat (Pine Instruments, Durham, NC). High-Definition Cyclic Voltammetry software (HDCV, University of North Carolina at Chapel Hill) was used for waveform output, signal processing, and data analysis, in conjunction with data acquisition cards (National Instruments, Austin TX) used for measuring current and synchronizing the electrochemical cell with the flow-injection system. Analog filtering of the applied waveform was accomplished with a 2-pole Sallen-Key, low-pass filter at 2 kHz.
Animal Experiments:
Animal care and use was in accordance with North Carolina State University Institutional Animal Care and Use Committee (IACUC) guidelines. Male Sprague−Dawley rats (n=2 animals; 250−300 g; Charles River Laboratories, Raleigh, NC) were individually housed on a 12:12 h light/dark cycle with free access to food and water and allowed to acclimate to the facility for several days. For acute electrode preparations, the animal was urethane anesthetized (4 g kg−1 intraperitoneal), placed in a stereotaxic frame (David Kopf Instruments, Tujunga, CA), and holes for electrodes were drilled in the skull using stereotaxic coordinates, as previously described.24–25 The working electrode was placed in the dorsal striatum (+1.2 mm AP, +2.0 mm ML, −5.0 mm DV relative to bregma) for H2O2 measurements, and in the ventral striatum (+1.7 mm AP, +0.8 mm ML, −6.0 mm DV relative to bregma) for dopamine measurements; the Ag/AgCl reference electrode was placed in the contralateral forebrain. The animal’s body temperature was maintained at 37°C by a heating pad. All working electrodes were conditioned by application of the waveform at 30 Hz for at least 30 min prior to data collection.
Data Processing and Analysis:
All data were examined manually in HDCV before exporting to MATLAB R2018a (Mathworks, Natick, MA), where data were digitally filtered (first-order, low pass Bessel filter with 2 kHz cutoff) prior to further analyses. PLSR was accomplished using the “plsregress” function along with the SIMPLS algorithm to center the data, to reduce data dimensionality, and to construct vector components describing the variance. Mean-squared error was determined using a 5-fold cross validation, and was used to select the number of vector components retained in each PLSR model.26 Pearson’s linear correlation coefficient was used to compare drift-subtracted CVs to those obtained using the conventional background subtraction approach. All statistical analyses and graphical depiction of data were carried out using GraphPad Prism 6 (GraphPad Software, Inc., La Jolla, CA) or MATLAB R2018a.
Results and Discussion
Electrochemical Drift in FSCV Recordings
The term ‘electrochemical drift’ is used herein to describe a variety of voltammetric signals recorded at carbon-based sensors during FSCV experiments lasting minutes or longer. Some of these stem from physical changes. For example, etching of the carbon surface is known to occur over the lifetime of the sensor.27–28 This contributes to drift by altering the surface area (affecting capacitance) and the composition of chemical functionalities on the sensor surface.27, 29–30 Electrode fouling can occur via electropolymerization of redox-active species,31–32 tissue encapsulation, or adsorption of macromolecules to alter the active sensing surface (and its impedance properties).33–35 Additionally, ionic and molecular fluctuations are common in vivo, and can transiently alter the double-layer structure, shifting capacitance.20, 36–37 For example, the ΔpH signal contains current contributions across the entire potential window that are attributed to ion displacement by either hydroxide or hydronium ions, as well as shifts in the proton-coupled electron-transfer equilibria of surface-oxide functionalities.38–39 Furthermore, it has been shown that divalent cations (Ca2+ and Mg2+) interact with surface quinones in a potential-dependent manner.40 Due to the broad scope of sources that can potentially impact the carbon/solution interface, the drift signals observed in voltammetry data are highly diverse, can vary from one recording session to the next, and can contain multiple features across the potential-window.
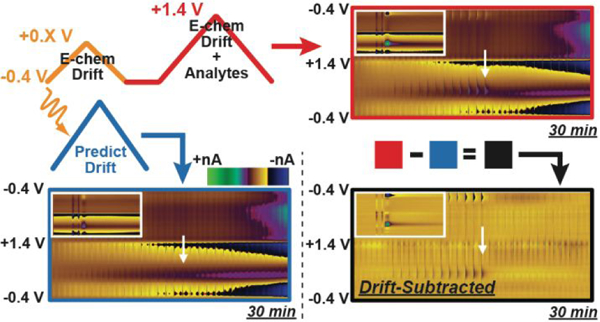
The Double-Waveform Partial-Least-Squares Regression Model
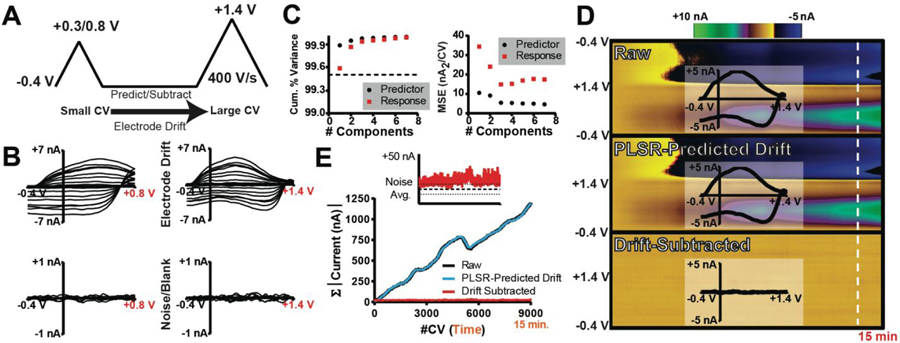
Figure 1 displays an outline of the DW-PLSR model, as it applies to an investigation of electrochemical drift. To reliably identify, predict, and subtract drift from voltammetric data, a double-triangular waveform is employed (Figure 1A). The small waveform (sWF) scans from a −0.4 V holding potential to +0.3/+0.8 V at 400 V s−1, and the large waveform (lWF) extends to +1.4 V at 400 V s−1. The switching potential of the sWF is chosen to exclude the oxidation potential of any analytes of interest, and to capture information specific to interferents; in this case electrochemical drift. A sWF switching potential of +0.3 V is insufficient to substantially oxidize DA, which prevents the model from treating it as an interferent.
Figure 1:
Introduction to the double-waveform partial-least-squares regression (DW-PLSR) paradigm. (A) Double waveform comprised of a smaller triangular sweep (sWF) paired with a larger sweep (lWF). (B) Training set CVs describing drift (top; 8 CVs) and noise (bottom; 3 CVs) recorded on the sWF (left) and the lWF (right). (C) 5-fold cross validation average mean-square error (left; nA2/CV) and cumulative percent variance (right) for both the predictor (black) and response (red). (D) Color plots displaying raw data collected with the lWF (top), PLSR-predicted drift (middle; three components), and drift-subtracted (bottom) data for a 15 min in vitro recording. Time and potential are plotted as the abscissa and ordinate, respectively. Current is depicted in false color, as indicated by the colorbar. Inset CVs were extracted from the data at the time point indicated by the white line. (E) Absolute current inherent to the CVs collected in the recordings displayed in (D), summed. Inset is a comparison of the total current in the drift-subtracted data to that in the training set describing the noise (bold black line is the mean; thinner lines represent the SD).
PLSR is a method for multivariate statistical analysis that is similar to principal component regression (PCR), which is commonly used to isolate individual chemical signals from complex FSCV data.21, 41 Notably, the two approaches differ in the construction of the principal components (PCs). PCR is an unsupervised dimensionality reduction method,42 as a set of predictors (CVs) are used to construct PCs without reference to the response (concentration). By contrast, PLSR is a supervised dimensionality reduction method that projects both predictor and response variables to a new vector space to determine the PCs that maximize the covariance of projected structures.26, 42 As such, PLSR generally describes training data more efficiently with fewer PCs (than PCR), and output prediction is often more robust.43–44
The DW-PLSR model is trained using data collected with the sWF and the lWF as the predictor and response, respectively. Figure 1B displays a representative PLSR training set comprised of sWF and lWF CVs for electrochemical drift and noise. Five-fold cross validation (Figure 1C) was used to determine principal component vectors that describe the training data, and cumulative percent variance (Figure 1C) was used to assess the noise in the data.44–45 Three principal components were retained in this model, as no improvement in the predictive power (mean-square error) was observed by retaining more components. Figure 1D displays a lWF color plot for an eventless 15 min in vitro recording, composed of CVs recorded over time. The inset CV describes electrochemical drift with current contributions that span the entire potential window. The PLSR-predicted drift for this recording is also shown, and is strikingly similar to the raw recording. Subtracting the PLSR-predicted drift from the raw recording results in the drift-subtracted color plot that is comprised solely of noise. To quantitatively assess the performance of DW-PLSR for drift subtraction, the absolute value of the current collected in each of these background-subtracted CVs was summed (Figure 1E). The CVs that comprise both the raw recording and the PLSR-predicted drift continually increase in current over the duration of the recording, indicative of electrochemical drift. Following drift subtraction, the CVs contain little current, indicating removal of drift. In fact, the total CV current following drift subtraction is comparable to the level of the noise (Figure 1E; inset). Thus, DW-PLSR can be used to account for electrochemical drift, and should serve as a useful tool in extending the time window over which chemical fluctuations can be reliably monitored from a single reference point.
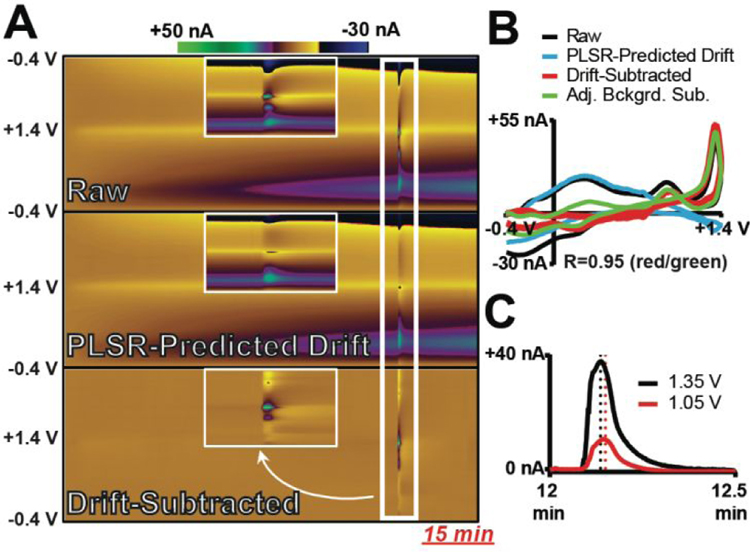
To investigate the ability of DW-PLSR to remove electrochemical drift that occurs concurrent with chemical fluctuations of redox-active species, a 15 min in vitro recording that contained a 2-sec bolus injection of 40-μM adenosine was analyzed via DW-PLSR. Adenosine is a purine nucleoside that plays an important biological role in energy transfer, a neuromodulator believed to play a role in regulation of blood flow, and is the subject of a number of studies incorporating FSCV.46–48 Adenosine undergoes two detectable, consecutive oxidations at a standard CFME. The first occurs at ~+1.3 V and, following sufficient production of the first oxidation product, the second oxidation at ~+1 V can also be monitored. Therefore, a +0.8 V upper potential limit was used for the sWF in this experiment. Figure 2A displays raw data collected with the lWF, PLSR-predicted drift, and drift-subtracted color plots. Inset are expanded color plots at the time point of the adenosine injection. The drift is readily identified by PLSR, and drift subtraction clearly reveals the short adenosine fluctuation. A comparison of CVs from each color plot is displayed in Figure 2B. Importantly, the ability to use the conventional approach to background subtraction is still possible with this approach – the data inherent to the initial, smaller waveform can simply be ignored. Figure 2B also contains a CV generated by subtracting a background CV that was collected immediately prior to the injection (conventional method). The currents reflected in the drift-subtracted CV only describe the two steps of adenosine oxidation, and the CV is consistent in shape and intensity with the CV generated by subtracting an adjacent background (R = 0.95). These data demonstrate that the DW-PLSR model can effectively remove drift to reveal a rapid adenosine fluctuation occurring 12 min after the initial background position. Furthermore, examination of the current versus time traces extracted at both oxidation potentials from the drift-subtracted data demonstrates that DW-PLSR is effective in preserving the time-delayed detection of the second oxidation step (Figure 2C). These data demonstrate the ability of DW-PLSR to evaluate an analyte whose signal subtly changes over time; a feature that PCR can struggle to handle.49–50
Figure 2:
DW-PLSR effectively removes drift from a 15 min recording containing a rapid fluctuation of adenosine (40 μM). (A) Color plots containing raw data collected with the lWF, PLSR-predicted drift (three components), and drift-subtracted data. Insets provide an expanded view of the adenosine fluctuation (white box). (B) CVs extracted at the time point of the adenosine injection. The CV generated using DW-PLSR for drift subtraction (red) correlates well with that generated using the conventional background-subtraction approach (green, R=0.95). (C) Current versus time traces extracted at the potential corresponding to the primary (+1.35 V; black) and secondary (+1.05 V; red) oxidation of adenosine.
Subtraction of Drift from Extended Dopamine Recordings
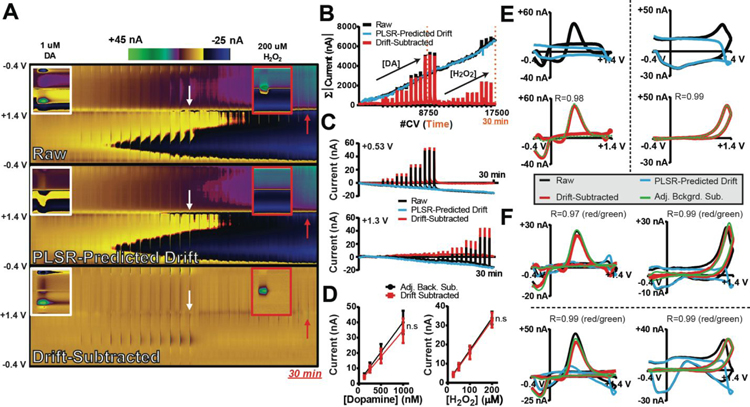
To be a broadly applicable, the DW-PLSR model must effectively remove drift from recordings that target a variety of analytes of interest. DA is one of the most widely studied neurotransmitters, and is the species most commonly monitored by FSCV. H2O2 is a reactive oxygen species that is produced as a byproduct of normal mitochondrial function. It can readily diffuse across the cell membrane, and can be monitored via FSCV.51 H2O2 can also modulate striatal DA release.52 Furthermore, it is generated by oxidase enzymes permitting indirect detection of non-electroactive molecules such as glucose, lactate, and acetyl(choline).8–11, 53 Thus, DW-PLSR was used to remove drift from a 30-min in vitro recording containing both DA and H2O2 fluctuations. In this experiment, a limit of +0.3 V was used in the sWF in order to prevent the oxidation of DA in the PLSR predictor. If significant DA signal was generated with the sWF, it would be treated as an interferent and would be removed with electrochemical drift.23
Figure 3A displays a 30 min flow-cell recording in which calibrations of both DA and H2O2 were performed, as well as color plots for PLSR-predicted drift, and resulting drift-subtracted data. Inset is an expanded view of the final 1 μM DA (~17 min) and 200 μM H2O2 (~29 min) injections. The drift training set and the model validation data for this electrode are shown in Supplemental Figure S1. Drift subtraction clearly reveals the analyte injections that were masked by drift in the raw data. Figure 3B displays the sum of the absolute value of the current in each CV collected over time. The summed raw current continually increases over the duration of the recording, indicative of electrochemical drift. As a result, current fluctuations due to the redox activity of the analytes are increasingly masked. The total (summed) current for voltammograms of PLSR-predicted drift collected over time overlaps with that of the raw recording, except it does not spike during analyte injection. Following drift subtraction, the total current contained in the CVs reflects two sets of spikes with increasing intensity – these are the calibration data for each analyte, clearly revealed. Figure 3C displays the current versus time traces extracted from each of the color plots at the oxidation potential for DA and H2O2. In this case, the contribution of drift would lead to an underestimation of both DA and H2O2 concentrations. Following drift subtraction, the data are baseline corrected. Figure 3D displays DA and H2O2 CVs extracted from the raw data, PLSR-predicted drift and drift-subtracted color plots at the time points indicated by the arrows in Figure 3A. CVs generated by adjacent background subtraction are overlaid with the drift-subtracted CVs, for comparison. This experiment was repeated on two additional electrodes, and corresponding CVs from those analyses are displayed in Figure 3F. The training sets, model validation data, and color plots for the two additional electrodes are provided in Supplemental Figure S1. It is important to note that although each drift signal exhibits unique features, the drift-subtracted CVs are consistently well correlated with those produced using adjacent background subtraction (R=0.97–0.99). Furthermore, calibrations produced via adjacent background subtraction (black) for both DA (39.2 ± 0.1 nA/μM) and H2O2 (0.168 ± 0.002 nA/μM) were consistent with those produced when drift was subtracted using the DW-PLSR model for both DA (34.2 ± 0.1 nA/μM) and H2O2 (0.163 ± 0.003 nA/μM). Calibration data were fit using linear regression (Figure 3E). One-way ANCOVA revealed no significant differences in slope or intercept for DA (slope: F(1,20)=0.303, p>0.05; intercept: F(1,21)=0.901, p>0.05) and H2O2 (slope: F(1,20)=0.042, p>0.05; intercept: F(1,21)=0.224, p>0.05) calibration lines. These data confirm that drift subtraction using the DW-PLSR model does not skew quantification of DA and H2O2 signals, even at time-points >15 min from the initial background position. Thus, DW-PLSR should provide a powerful tool for removing electrochemical drift from in vivo recordings.
Figure 3:
DW-PLSR can be used to effectively remove drift from 30 min recordings containing DA (125, 250, 500, and 1000 nM) and H2O2 (25, 50, 100, and 200 μM) bolus injections in triplicate. (A) Color plots of raw data collected using the lWF, as well as PLSR-predicted drift (three components), and drift-subtracted data collected at a single electrode. An expanded view of the color plots for the last DA (1 μM, white arrow and box) and H2O2 (200 μM, red arrow and box) injections are shown as insets. (B) The sum of the currents inherent to each CV comprising the color plots in (A), over time. Tick marks on abscissa indicate the time points of DA and H2O2 injections in the first and last half of the recording, respectively. (C) Current extracted at the oxidation potential for DA (top; +0.5 V) and H2O2 (bottom; +1.3 V), plotted as a function of time. (D) Calibration plots for DA (left) and H2O2 (right) created from the drift-subtracted data (initial background at 1 s), as well as by subtracting an adjacent background signal (black; conventional method). Data are displayed as the mean ± SD (n=3 electrodes). (E) CVs for 1 μM DA (left) and 200 μM H2O2 (right) extracted from the data at the time points indicated by the arrows in (A) for the raw (black), PLSR-predicted drift (blue), and drift-subtracted (red) data. A CV generated adjacent background subtraction (green) is shown for comparison. (F) DA and H2O2 CVs extracted at the same time points as in (D) for the other two electrodes used in this study.
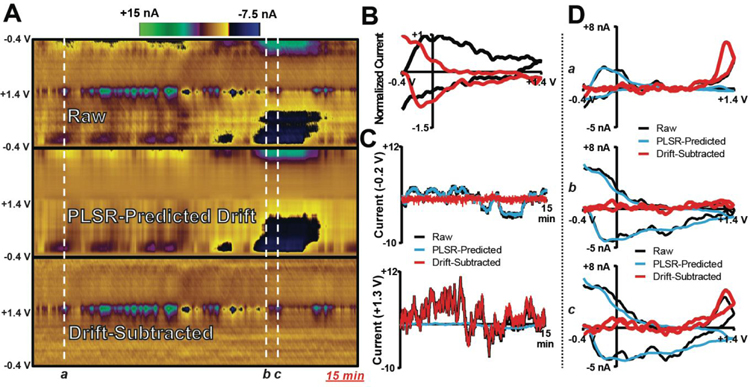
Drift Subtraction from Extended In Vivo Recordings
To evaluate effectiveness for in vivo recordings, the DW-PLSR model was used to analyze natural H2O2 fluctuations evident in a 15-min recording from the dorsal striatum of an anesthetized rat. A switching potential of +0.8 V was used in the sWF. Figure 4A displays the raw data, PLSR-predicted drift, and drift-subtracted color plots. The data contain noticeable H2O2 dynamics, indicated by the blue/green colors at ~+1.3 V, along with a relatively small contribution from drift. CVs for the observed drift signals are displayed in Figure 4B. Training set CVs as well as cross-validation and cumulative percent variance are displayed in Supplemental Figure S2. Figure 4C displays current versus time traces taken at −0.2 V, where drift is evident, as well as at +1.3 V, where H2O2 oxidation occurs. At −0.2 V, the raw and PLSR-predicted currents are consistent, resulting in baseline correction with subtraction of drift. At +1.3 V, the raw and drift-subtracted traces correlate, indicating a minimal contribution from electrochemical drift at ~+1.3 V in this recording. Figure 4D compares CVs selected from various time points in the recording, demonstrating the effectiveness of the model. Overall, drift subtraction using the DW-PLSR model provides for easy visualization and quantification of striatal H2O2 dynamics across this 15-min window.
Figure 4:
The DW-PLSR model removes drift from data collected in the dorsal striatum of an anesthetized rat, improving visualization of endogenous H2O2 transients. (A) Raw data, PLSR-predicted drift (4 components), and drift-subtracted color plots collected in a 15-min recording. (B) lWF drift signals indicative of training set data (normalized to the max current). (C) Current versus time traces extracted from the color plots at −0.2 V (top) and +1.3 V (bottom). (D) CVs extracted from the color plots at the time points indicated by the white lines and lowercase letters.
Next, the DW-PLSR model was employed for recordings in the ventral striatum of an anesthetized rat, a brain region containing significant dopaminergic input.54 Here, a +0.3 V switching potential was used in the sWF. The most striking data collected in this experiment are shown in Figure 5. These CVs were collected over 10 min at the very end of the experiment as the animal stopped breathing, due to extended anesthesia. This recording is a ‘worst-case scenario’, but it clearly demonstrates the utility of the model in even the most extreme case of ischemia. Ischemic events and spreading depression lead to the mass depolarization of neurons, dysregulation of oxygen and pH, and millimolar-scale changes in the concentrations of common extracellular ions (e.g. K+, Na+, Ca2+).55–58 These changes substantially affect the quality of the voltammetric data.
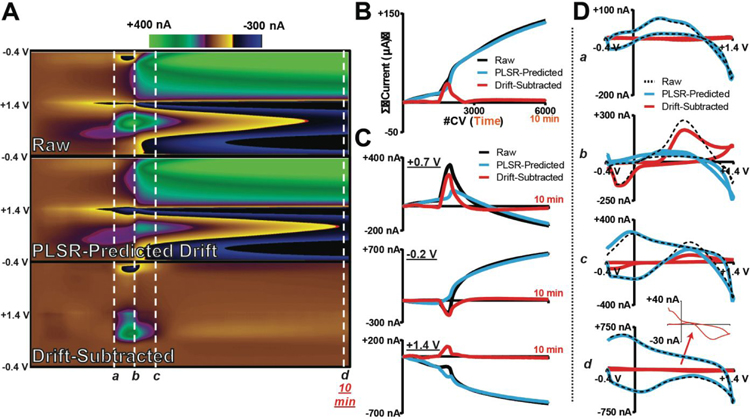
Figure 5:
The DW-PLSR model removes drift from voltammetric data collected in the ventral striatum of an anesthetized rat during a 10-min ischemic event. (A) Raw data, PLSR-predicted drift (3 components), and drift-subtracted color plots. (B) Total summed current of each CV collected over time in (A). (C) Current versus time traces extracted at +0.7 V (top), −0.2 V (middle), and +1.4 V (bottom) from each color plot. (D) CVs extracted from the color plots at the time points indicated by the white lines and lowercase letters (a–d). Inset is the re-scaled drift-subtracted CV (d).
The data in Figure 5A show a large DA signal (white line; b) collected during the ischemic event. It was recorded along with a significant amount of interfering drift. The drift was likely generated by the effects of ionic shifts on electrode impedance; variable impedance changes background size and shape.59 Training set CVs as well as cross-validation and cumulative percent variance data are displayed in Supplemental Figure S2. Figure 5B plots the summed current contained in the CVs, over time. Following drift subtraction, the total current is baseline corrected, and rises only at the time point of the DA fluctuation. Similarly, only features attributed to the DA redox couple are observed in the drift-subtracted color plot. The current versus times traces shown in Figure 5C were extracted at +0.7 V (DA oxidation), −0.2 V (dopamine-o-quinone reduction), and at +1.4 V. In this case, the origin of the current observed at +1.4 V is not clear. Figure 5D displays CVs extracted at various time points in the recording: prior to (a), during (b), and following (c) the large DA event, as well as at the end of the 10-min recording. Electrochemical drift was a significant contributor to each these signals, and in all cases the DW-PLSR model was effective for drift subtraction.
Interestingly, drift subtraction revealed a gradual decrease in DA ‘tone’ in the second half of the recording that had been masked by the much larger drift signal. This is evident as an undershoot in DA concentration in Figure 5C (top), and as an inverted DA CV in Figure 5D (bottom). The decrease is likely due to the diffusion and breakdown of extracellular DA, concurrent with a lack of replenished vesicular stores. However, it cannot be ruled out that some portion of the DA signal was captured within the much larger drift signal used in the training set, and erroneously subtracted from the data. Indeed, the maximum current of the largest drift CV in the training set is ~700 nA (Supplemental Figure S2), and the revealed decrease in DA is represented by only ~−30 nA. This presents a potential complication in monitoring slower, more gradual concentration changes. In order to reliably investigate minute-to-minute chemical dynamics occurring far from the initial background position, it is necessary that the training CVs be purely representative of drift.
Integrating Drift Signals from Multiple Electrodes for Drift Subtraction
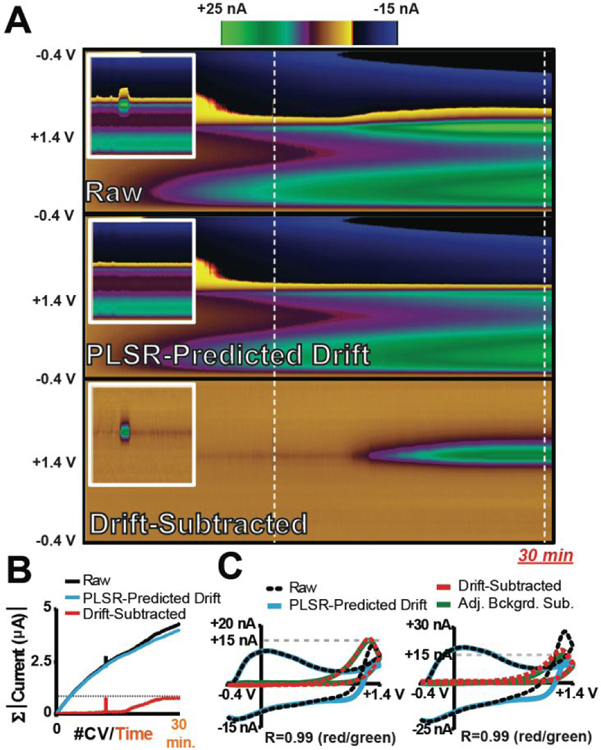
All models shown up to this point have been constructed using electrode-specific training sets. However, many studies employing FSCV incorporate permanently implanted electrodes that are used for extended recording sessions over the course of weeks to months.60–61 Electrochemical drift can vary from recording session to session, presenting a critical constraint; a drift training set may need to be made for every recording session.
Figure 6A displays a color plot of raw voltammetric data collected in a 30-min flow-cell recording. A bolus injection of H2O2 was introduced at the time point indicated by the first white dashed line. In the second half of the recording, a multi-pump system was used to slowly increase (15–30 min) the concentration of H2O2 in the flowing buffer to a final concentration equal to that of the bolus. The bolus injection of H2O2 can be deciphered at ~1.3 V in the raw voltammetric data, even in the presence of drift. However, it is far less straightforward to distinguish H2O2 from drift during the slow increase in H2O2 concentration. To evaluate if an electrode-specific training set is required for the effective removal of drift from these data, CVs describing drift were collected with multiple electrodes (n=10) and used to construct a DW-PLSR model. Supplemental Figure S3 displays normalized CVs for drift collected with the sWF and lWF at each electrode that are representative of those used to build the PLSR training set, as well as cross-validation mean-squared error and cumulative percent variance. Seven components were retained and used to predict and subtract drift from voltammetric data. It is visually evident that the color plot describing the PLSR-predicted drift for the recording describes the observed electrochemical drift, as the overall signal across the entirety of this color plot is similar to that evident in the raw data. Following drift subtraction, the color plot contains only two features at +1.3 V - the bolus injection and the slow increase in H2O2 concentration are clearly evident. A comparison of the total summed current of each CV over time for the raw, PLSR-predicted drift, and drift-subtracted data (Figure 6B) demonstrates that the model was effective for evaluating the slow increase in H2O2 concentration, as the total current gradually increases to the same magnitude as that of the bolus of H2O2 (same concentration). Figure 6C compares CVs extracted from the injection and near the end of the slow H2O2 increase. In both cases, the raw signal contains a significant drift contribution, which the PLSR prediction effectively captures. Following drift subtraction, the voltammetric signature for H2O2 is revealed, and CVs from both time points are of the same shape and intensity (R=0.99). Furthermore, a CV obtained by subtracting a background signal immediately prior to the bolus injection (green trace) is essentially indistinguishable from those obtained using DW-PLSR for drift subtraction (R=0.99). Supplemental Figure S3 displays CVs for bolus injections of different concentrations of H2O2 collected using three other electrodes that exhibited substantially different drift characteristics. These data reflect the ability of this DW-PLSR model to predict a variety of drift signals, and to generate drift-subtracted CVs that are consistent with those from adjacent background subtraction (R2=0.95–0.99). Thus, it is clearly possible to develop effective models for subtraction of drift from fast voltammetric data that are not specific to any given electrode, to reveal both rapid and gradual changes in analyte concentration over time.
Figure 6:
A DW-PLSR model constructed using drift signals from a collection of electrodes reveals both transient fluctuations and gradual increases in H2O2 concentration. (A) Representative raw data, PLSR-predicted drift (seven components), and drift-subtracted color plots for a 30-min recording in which both a bolus injection and a gradual increase in H2O2 occurred (same concentrations; 160 μM shown). Insets show an enhanced view of the injection. (B) The summed current of each CV as a function of time for the color plots in (A). (C) CVs extracted (white lines in A) from the injection (a; top) and near the end of the gradual increase in H2O2 concentration (b; bottom). In both plots, a CV generated by subtracting a background signal collected immediately prior to the time of the injection is included for comparison (green).
Conclusions
Electrochemical drift can be observed in any application of electrochemistry. In FSCV, drift occurs when changes at the carbon/solution interface impact the background signal. Drift not only limits the duration over which FSCV can accurately monitor rapid chemical fluctuations (typically the background is stable for 30–90 seconds), but it also negatively impacts selectivity and complicates interpretation of the data. Herein, we demonstrate that PLSR can be combined with a double voltammetric waveform to provide an effective means of determining and subtracting drift contributions from in vivo recordings. By using data collected with a sWF that does not encompass the redox activity of the analyte(s) as a predictor variable, the drift inherent to data collected with the subsequent lWF can be predicted and non-subjectively subtracted. A major advantage of this technique is that it simplifies training set construction for in vivo analyses – researchers are no longer required to search for analyte-pure CVs for all components of the chemical signal; rather, only interferent CVs are necessary. The DW-PLSR model can be used to subtract drift signals and reveal chemical fluctuations reflected by currents that span multiple orders of magnitude. In fact, it was demonstrated in vitro that drift information can be collected at a set of many electrodes, pooled, and used for the effective subtraction of drift as long as sufficient variety is included in the training set. However, it is important to note that potential sources of electrochemical drift are greater in vivo, and effective implementation of a training set that is not electrode-specific to the analysis of in vivo data will require more investigation. Importantly, drift subtraction did not alter the signal of any of the target analytes investigated. The DW-PLSR paradigm for drift removal promises to simplify data analysis and reduce user bias, as there is no longer a need to subtract backgrounds at multiple points over the course of a long (minutes) recording. As a result, it is expected that this approach will broaden the applicability of FSCV for monitoring chemical fluctuations occurring on multiple timescales.
Supplementary Material
Acknowledgements
The authors would like to thank Karen Butler for her assistance with data collection for data displayed in Figure 4. This work was supported by the U.S. National Institutes of Health (R01-NS076772 to L.A.S). C.J.M. is supported by an NSF Graduate Research Fellowship (DGE-1252376).
Footnotes
Supporting Information
The Supporting Information is available free of charge on the ACS Publications website at DOI:
Supporting Information. Displays additional data with respect to the PLSR analyses performed in this study, and a description of electrode fabrication and the flow-cell apparatus used for in vitro investigations
References
- 1.Michael AC; Borland L, Electrochemical methods for neuroscience. CRC Press: 2006. [PubMed] [Google Scholar]
- 2.Phillips PEM; Robinson DL; Stuber GD; Carelli RM; Wightman RM, Real-Time Measurements of Phasic Changes in Extracellular Dopamine Concentration in Freely Moving Rats by Fast-Scan Cyclic Voltammetry In Drugs of Abuse: Neurological Reviews and Protocols, Wang JQ, Ed. Humana Press: Totowa, NJ, 2003; pp 443–464. [DOI] [PubMed] [Google Scholar]
- 3.Pihel K; Schroeder TJ; Wightman RM, Rapid and Selective Cyclic Voltammetric Measurements of Epinephrine and Norepinephrine as a Method to Measure Secretion from Single Bovine Adrenal-Medullary Cells. Analytical Chemistry 1994, 66 (24), 4532–4537. [Google Scholar]
- 4.Cheer JF; Aragona BJ; Heien ML; Seipel AT; Carelli RM; Wightman RM, Coordinated accumbal dopamine release and neural activity drive goal-directed behavior. Neuron 2007, 54 (2), 237–44. [DOI] [PubMed] [Google Scholar]
- 5.Cheer JF; Wassum KM; Heien ML; Phillips PE; Wightman RM, Cannabinoids enhance subsecond dopamine release in the nucleus accumbens of awake rats. J Neurosci 2004, 24 (18), 4393–400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Phillips PE; Stuber GD; Heien ML; Wightman RM; Carelli RM, Subsecond dopamine release promotes cocaine seeking. Nature 2003, 422 (6932), 614–8. [DOI] [PubMed] [Google Scholar]
- 7.Roitman MF; Stuber GD; Phillips PE; Wightman RM; Carelli RM, Dopamine operates as a subsecond modulator of food seeking. J Neurosci 2004, 24 (6), 1265–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wassum KM; Tolosa VM; Wang J; Walker E; Monbouquette HG; Maidment NT, Silicon Wafer-Based Platinum Microelectrode Array Biosensor for Near Real-Time Measurement of Glutamate in Vivo. Sensors (Basel) 2008, 8 (8), 5023–5036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Smith SK; Lee CA; Dausch ME; Horman BM; Patisaul HB; McCarty GS; Sombers LA, Simultaneous Voltammetric Measurements of Glucose and Dopamine Demonstrate the Coupling of Glucose Availability with Increased Metabolic Demand in the Rat Striatum. Acs Chem Neurosci 2017, 8 (2), 272–280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Smith SK; Gosrani SP; Lee CA; McCarty GS; Sombers LA, Carbon-Fiber Microbiosensor for Monitoring Rapid Lactate Fluctuations in Brain Tissue Using Fast-Scan Cyclic Voltammetry. Analytical Chemistry 2018, 90 (21), 12994–12999. [DOI] [PubMed] [Google Scholar]
- 11.Parikh V; Kozak R; Martinez V; Sarter M, Prefrontal acetylcholine release controls cue detection on multiple timescales. Neuron 2007, 56 (1), 141–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hermans A; Keithley RB; Kita JM; Sombers LA; Wightman RM, Dopamine detection with fast-scan cyclic voltammetry used with analog background subtraction. Anal Chem 2008, 80 (11), 4040–8. [DOI] [PubMed] [Google Scholar]
- 13.Howell JO; Kuhr WG; Ensman RE; Wightman RM, Background Subtraction for Rapid Scan Voltammetry. Journal of Electroanalytical Chemistry 1986, 209 (1), 77–90. [Google Scholar]
- 14.Hunt SM; Sheil MM; Belov M; Derrick PJ, Probing the effects of cone potential in the electrospray ion source: consequences for the determination of molecular weight distributions of synthetic polymers. Anal Chem 1998, 70 (9), 1812–22. [DOI] [PubMed] [Google Scholar]
- 15.McLauchlan G; Rawlings JM; Lucas ML; McCloy RF; Crean GP; McColl KE, Electrodes for 24 hours pH monitoring--a comparative study. Gut 1987, 28 (8), 935–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Millar J; Williams GV, Fast Differential Ramp Voltammetry - a New Voltammetric Technique Designed Specifically for Use in Neuronal Tissue. Journal of Electroanalytical Chemistry 1990, 282 (1–2), 33–49. [Google Scholar]
- 17.Brazill SA; Bender SE; Hebert NE; Cullison JK; Kristensen EW; Kuhr WG, Sinusoidal voltammetry: a frequency based electrochemical detection technique. Journal of Electroanalytical Chemistry 2002, 531 (2), 119–132. [Google Scholar]
- 18.Oh Y; Park C; Kim DH; Shin H; Kang YM; DeWaele M; Lee J; Min HK; Blaha CD; Bennet KE; Kim IY; Lee KH; Jang DP, Monitoring In Vivo Changes in Tonic Extracellular Dopamine Level by Charge-Balancing Multiple Waveform Fast-Scan Cyclic Voltammetry. Anal Chem 2016, 88 (22), 10962–10970. [DOI] [PubMed] [Google Scholar]
- 19.Atcherley CW; Laude ND; Parent KL; Heien ML, Fast-scan controlled-adsorption voltammetry for the quantification of absolute concentrations and adsorption dynamics. Langmuir 2013, 29 (48), 14885–92. [DOI] [PubMed] [Google Scholar]
- 20.Johnson JA; Hobbs CN; Wightman RM, Removal of Differential Capacitive Interferences in Fast-Scan Cyclic Voltammetry. Anal Chem 2017, 89 (11), 6166–6174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Keithley RB; Wightman RM, Assessing principal component regression prediction of neurochemicals detected with fast-scan cyclic voltammetry. Acs Chem Neurosci 2011, 2 (9), 514–525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Keithley RB; Heien ML; Wightman RM, Multivariate concentration determination using principal component regression with residual analysis. Trends Analyt Chem 2009, 28 (9), 1127–1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Meunier CJ; Mitchell EC; Roberts JG; Toups JV; McCarty GS; Sombers LA, Electrochemical Selectivity Achieved Using a Double Voltammetric Waveform and Partial Least Squares Regression: Differentiating Endogenous Hydrogen Peroxide Fluctuations from Shifts in pH. Anal Chem 2018, 90 (3), 1767–1776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Roberts J; Lugo-Morales L; Loziuk P; Sombers L, Leading Methods in Dopamine Research. Humana Press; New York: 2013. [Google Scholar]
- 25.Paxinos G; Franklin KB, The mouse brain in stereotaxic coordinates. Gulf professional publishing: 2004. [Google Scholar]
- 26.Mehmood T; Liland KH; Snipen L; Saebo S, A review of variable selection methods in Partial Least Squares Regression. Chemometrics and Intelligent Laboratory Systems 2012, 118, 62–69. [Google Scholar]
- 27.Takmakov P; Zachek MK; Keithley RB; Walsh PL; Donley C; McCarty GS; Wightman RM, Carbon microelectrodes with a renewable surface. Anal Chem 2010, 82 (5), 2020–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.McCreery RL, Advanced carbon electrode materials for molecular electrochemistry. Chem Rev 2008, 108 (7), 2646–87. [DOI] [PubMed] [Google Scholar]
- 29.Roberts JG; Moody BP; McCarty GS; Sombers LA, Specific oxygen-containing functional groups on the carbon surface underlie an enhanced sensitivity to dopamine at electrochemically pretreated carbon fiber microelectrodes. Langmuir 2010, 26 (11), 9116–22. [DOI] [PubMed] [Google Scholar]
- 30.Mitchell EC; Dunaway LE; McCarty GS; Sombers LA, Spectroelectrochemical Characterization of the Dynamic Carbon-Fiber Surface in Response to Electrochemical Conditioning. Langmuir 2017, 33 (32), 7838–7846. [DOI] [PubMed] [Google Scholar]
- 31.Harreither W; Trouillon R; Poulin P; Neri W; Ewing AG; Safina G, Carbon nanotube fiber microelectrodes show a higher resistance to dopamine fouling. Anal Chem 2013, 85 (15), 7447–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hashemi P; Dankoski EC; Petrovic J; Keithley RB; Wightman RM, Voltammetric detection of 5-hydroxytryptamine release in the rat brain. Anal Chem 2009, 81 (22), 9462–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Scott FL; Svjetlana M; Matthew DJ; Jerrold LV; Cameron CM, In vivo impedance spectroscopy of deep brain stimulation electrodes. Journal of Neural Engineering 2009, 6 (4), 046001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Mercanzini A; Colin P; Bensadoun JC; Bertsch A; Renaud P, In vivo electrical impedance spectroscopy of tissue reaction to microelectrode arrays. IEEE Trans Biomed Eng 2009, 56 (7), 1909–18. [DOI] [PubMed] [Google Scholar]
- 35.Justin CW; Joseph AH; John D; William S; Daryl RK, Complex impedance spectroscopy for monitoring tissue responses to inserted neural implants. Journal of Neural Engineering 2007, 4 (4), 410. [DOI] [PubMed] [Google Scholar]
- 36.Nicholson C, Measurement of Extracellular Ions in the Brain. Trends in Neurosciences 1980, 3 (9), 216–218. [Google Scholar]
- 37.Rogers ML; Feuerstein D; Leong CL; Takagaki M; Niu X; Graf R; Boutelle MG, Continuous online microdialysis using microfluidic sensors: dynamic neurometabolic changes during spreading depolarization. Acs Chem Neurosci 2013, 4 (5), 799–807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Takmakov P; Zachek MK; Keithley RB; Bucher ES; McCarty GS; Wightman RM, Characterization of local pH changes in brain using fast-scan cyclic voltammetry with carbon microelectrodes. Anal Chem 2010, 82 (23), 9892–900. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Deakin MR; Kovach PM; Stutts KJ; Wightman RM, Heterogeneous Mechanisms of the Oxidation of Catechols and Ascorbic-Acid at Carbon Electrodes. Analytical Chemistry 1986, 58 (7), 1474–1480. [DOI] [PubMed] [Google Scholar]
- 40.Kim YJ; Wu W; Chun SE; Whitacre JF; Bettinger CJ, Catechol-mediated reversible binding of multivalent cations in eumelanin half-cells. Adv Mater 2014, 26 (38), 6572–9. [DOI] [PubMed] [Google Scholar]
- 41.Rodeberg NT; Sandberg SG; Johnson JA; Phillips PE; Wightman RM, Hitchhiker’s Guide to Voltammetry: Acute and Chronic Electrodes for in Vivo Fast-Scan Cyclic Voltammetry. Acs Chem Neurosci 2017, 8 (2), 221–234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Härdle W; Simar L, Applied multivariate statistical analysis. Springer: 2007; Vol. 22007. [Google Scholar]
- 43.Wentzell PD; Montoto LV, Comparison of principal components regression and partial least squares regression through generic simulations of complex mixtures. Chemometrics and Intelligent Laboratory Systems 2003, 65 (2), 257–279. [Google Scholar]
- 44.Geladi P; Kowalski BR, Partial Least-Squares Regression - a Tutorial. Analytica Chimica Acta 1986, 185, 1–17. [Google Scholar]
- 45.Lavine B; Workman J, Chemometrics. Anal Chem 2008, 80 (12), 4519–31. [DOI] [PubMed] [Google Scholar]
- 46.Dunwiddie TV; Masino SA, The role and regulation of adenosine in the central nervous system. Annu Rev Neurosci 2001, 24 (1), 31–55. [DOI] [PubMed] [Google Scholar]
- 47.Lee ST; Venton BJ, Regional Variations of Spontaneous, Transient Adenosine Release in Brain Slices. Acs Chem Neurosci 2018, 9 (3), 505–513. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ross AE; Venton BJ, Adenosine transiently modulates stimulated dopamine release in the caudate-putamen via A1 receptors. J Neurochem 2015, 132 (1), 51–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Borman RP; Wang Y; Nguyen MD; Ganesana M; Lee ST; Venton BJ, Automated Algorithm for Detection of Transient Adenosine Release. Acs Chem Neurosci 2017, 8 (2), 386–393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Ross AE; Venton BJ, Sawhorse waveform voltammetry for selective detection of adenosine, ATP, and hydrogen peroxide. Anal Chem 2014, 86 (15), 7486–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Spanos M; Gras-Najjar J; Letchworth JM; Sanford AL; Toups JV; Sombers LA, Quantitation of hydrogen peroxide fluctuations and their modulation of dopamine dynamics in the rat dorsal striatum using fast-scan cyclic voltammetry. Acs Chem Neurosci 2013, 4 (5), 782–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Bao L; Avshalumov MV; Patel JC; Lee CR; Miller EW; Chang CJ; Rice ME, Mitochondria are the source of hydrogen peroxide for dynamic brain-cell signaling. J Neurosci 2009, 29 (28), 9002–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Burmeister JJ; Gerhardt GA, Self-referencing ceramic-based multisite microelectrodes for the detection and elimination of interferences from the measurement of L-glutamate and other analytes. Anal Chem 2001, 73 (5), 1037–42. [DOI] [PubMed] [Google Scholar]
- 54.Robbins TW; Everitt BJ, Functions of dopamine in the dorsal and ventral striatum. Seminars in Neuroscience 1992, 4 (2), 119–127. [Google Scholar]
- 55.Borjigin J; Lee U; Liu T; Pal D; Huff S; Klarr D; Sloboda J; Hernandez J; Wang MM; Mashour GA, Surge of neurophysiological coherence and connectivity in the dying brain. Proc Natl Acad Sci U S A 2013, 110 (35), 14432–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hansen AJ, Effect of anoxia on ion distribution in the brain. Physiol Rev 1985, 65 (1), 101–48. [DOI] [PubMed] [Google Scholar]
- 57.Siemkowicz E; Hansen AJ, Brain extracellular ion composition and EEG activity following 10 minutes ischemia in normo- and hyperglycemic rats. Stroke 1981, 12 (2), 236–40. [DOI] [PubMed] [Google Scholar]
- 58.Hobbs CN; Holzberg G; Min AS; Wightman RM, Comparison of Spreading Depolarizations in the Motor Cortex and Nucleus Accumbens: Similar Patterns of Oxygen Responses and the Role of Dopamine. Acs Chem Neurosci 2017, 8 (11), 2512–2521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Meunier CJ; Roberts JG; McCarty GS; Sombers LA, Background Signal as an in Situ Predictor of Dopamine Oxidation Potential: Improving Interpretation of Fast-Scan Cyclic Voltammetry Data. Acs Chem Neurosci 2017, 8 (2), 411–419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Kishida KT; Saez I; Lohrenz T; Witcher MR; Laxton AW; Tatter SB; White JP; Ellis TL; Phillips PE; Montague PR, Subsecond dopamine fluctuations in human striatum encode superposed error signals about actual and counterfactual reward. Proc Natl Acad Sci U S A 2016, 113 (1), 200–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Schwerdt HN; Shimazu H; Amemori KI; Amemori S; Tierney PL; Gibson DJ; Hong S; Yoshida T; Langer R; Cima MJ; Graybiel AM, Long-term dopamine neurochemical monitoring in primates. Proc Natl Acad Sci U S A 2017, 114 (50), 13260–13265. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.