Abstract
Introduction
Emerging data have described poor clinical outcomes from infection with the novel severe acute respiratory syndrome coronavirus 2 (SARS-CoV 2) among African American patients and those from underserved socioeconomic groups. We sought to describe the clinical characteristics and outcomes of acute kidney injury (AKI) in this special population.
Methods
This is a retrospective study conducted in an underserved area with a predominance of African American patients with coronavirus disease 2019 (COVID-19). Descriptive statistics were used to characterize the sample population. The onset of AKI and relation to clinical outcomes were determined. Multivariate logistic regression was used to determine factors associated with AKI.
Results
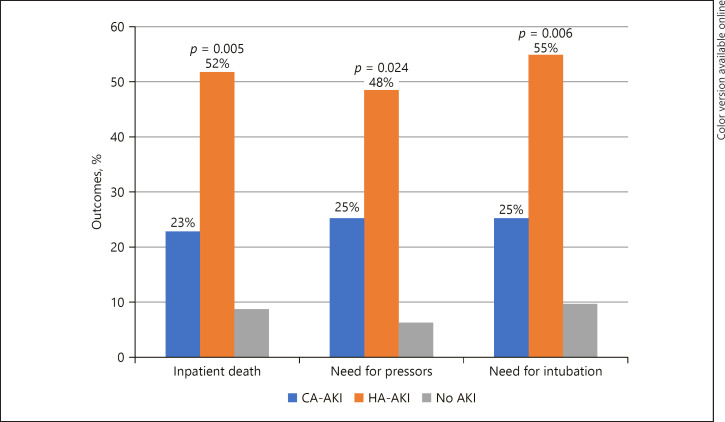
Nearly half (49.3%) of the patients with COVID-19 had AKI. Patients with AKI had a significantly lower baseline estimated glomerular filtration rate (eGFR) and higher FiO<sub>2</sub> requirement and D-dimer levels on admission. More subnephrotic proteinuria and microhematuria was seen in these patients, and the majority had a pre-renal urine electrolyte profile. Patients with hospital-acquired AKI (HA-AKI) as opposed to those with community-acquired AKI (CA-AKI) had higher rates of in-hospital death (52 vs. 23%, p = 0.005), need for vasopressors (42 vs. 25%, p = 0.024), and need for intubation (55 vs. 25%, p = 0.006). A history of heart failure was significantly associated with AKI after adjusting for baseline eGFR (OR 3.382, 95% CI 1.121–13.231, p = 0.032).
Conclusion
We report a high burden of AKI among underserved COVID-19 patients with multiple comorbidities. Those who had HA-AKI had worse clinical outcomes compared to those who with CA-AKI. A history of heart failure is an independent predictor of AKI in patients with COVID-19.
Keywords: COVID-19, Novel coronavirus, Acute kidney injury, Heart failure
Introduction
In December 2019, several pneumonia cases in Wuhan, China were shown to be caused by a novel coronavirus causing coronavirus disease 2019 (COVID-19) [1]. COVID-19 typically presents as an acute respiratory illness that can quickly progress to respiratory failure. Multiorgan involvement is also common, including damage to the kidneys, heart, and gastrointestinal tract, mirroring the presence of the angiotensin-converting enzyme 2 (ACE2) receptors in these organs, which serves as a portal of entry for the virus [2]. In particular, acute kidney injury (AKI) as one major complication of COVID-19 occurred in 17–29% of in-hospital patients based on reports from China, the USA, and some European countries [3, 4, 5, 6]. The potential mechanisms of AKI in COVID-19 patients include direct viral cytopathic effects, cytokine damage, microthrombi in the renal circulation, and hypoperfusive injury. Furthermore, AKI was found to be a significant poor prognostic marker among patients with COVID-19 [3]. The described burden of AKI in patients with COVID-19 has predominantly been reported in patients of non-African American origin. There are emerging data on worse outcomes of COVID-19 in African American patients [7] and in patients from underserved socioeconomic groups who also have higher rates of preexisting cardiorenal metabolic disease risk factors, such as hypertension, diabetes mellitus, and obesity. These comorbidities have in turn been shown to portend an overall worse prognosis [8, 9, 10]. The primary objective of this study is to describe the onset, nature, characteristics, and predictive factors of AKI in this high-risk patient population with COVID-19.
Materials and Methods
This is a single-center retrospective study. We included all patients >18 years of age who were admitted to a tertiary inner-city hospital with a confirmed diagnosis of COVID-19 via polymerase chain reaction (PCR). We excluded patients who were still hospitalized as of the time of chart review and analysis, as the final disposition related to outcomes could not be ascertained at the time of writing this report. We also excluded patients with incomplete data on clinical outcomes, those on maintenance hemodialysis, and patients on remdesivir or convalescent plasma therapy as both these were introduced in our institution protocol at the time of this data analysis. The definition used for AKI in this study was an increase in serum creatinine by at least 0.3 mg/dL or 1.5-fold from the baseline based on Kidney Disease Improving Global Outcome (KDIGO) criteria [11]. Patients admitted to the hospital with AKI based on the first serum creatinine measured were denoted as having community-acquired AKI (CA-AKI). In contrast, patients were identified as having hospital-acquired AKI (HA-AKI) when AKI was not apparent upon hospital admission but developed during hospitalization. The baseline serum creatinine level was defined as the lowest measured serum creatinine within the preceding 3 months. Baseline estimated glomerular filtration rate (eGFR) was computed using the CKD-API equation using the latest serum creatinine value available in that same period of time. All data were extracted from electronic medical records. Demographic variables were presented using descriptive statistics and frequencies. Categorical variables were analyzed with χ2 testing. Demographic and clinical variables were tabulated. The χ2 test was used to analyze the relationship between demographic or comorbidity variables and AKI, need for intubation, and vasopressor use. Multivariate logistic regression was used to look at the factors associated with AKI. We utilized 95% confidence intervals, which are presented when appropriate. Our study was approved by the institutional review board (IRB No. 2020-435).
Results
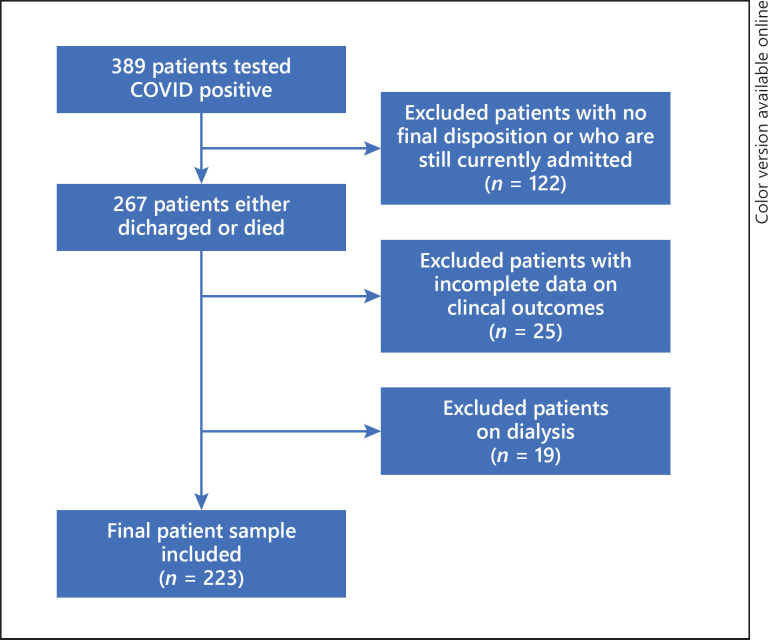
Of the 389 patients who tested positive for COVID-19 via PCR, 122 were excluded due to being currently hospitalized, as well as 19 patients who were on dialysis and 25 patients with incomplete clinical data, leaving a final sample of 223 patients (Fig. 1). In that final sample, the mean age was 65.91 ± 14.95 years, 48% were female, and 68% were African American. Of the total patients (n = 223), 72% had hypertension, 47% had diabetes, and 49% had documented AKI. Of the patients who had AKI, 72% were diagnosed on admission (CA-AKI), while 28% developed AKI during the hospital stay (HA-AKI). There were no differences in rates of AKI based on gender (p = 0.52) or race (p = 0.50). However, there were significantly more cases of AKI among older patients (>60 years old) compared to younger subjects (84 vs. 16%, p < 0.0001). Among patients with AKI, there was a significantly higher incidence of heart failure (19 vs. 4%, p = 0.002), hypertension (80 vs. 64%, p = 0.003), and baseline chronic kidney disease regardless of stage (27 vs. 9%, p = 0.001). Of the patients with AKI, 8% needed continuous renal replacement therapy (CRRT) or intermittent hemodialysis (HD). Among those who required CRRT/HD, 89% were intubated and 78% died. Baseline eGFR was significantly lower among the patients who had AKI compared to those without (79.61 vs. 99.16 mL/min/1.73 m2, p = 0.002). On admission, those with AKI had higher FiO2 requirements (44 vs. 34%, p = 0.007) and D-dimer levels (5,468 vs. 2,248 ng/mL, p = 0.04) than those without. On urinalysis, patients who had AKI had significantly more proteinuria (74 vs. 48%, p = 0.003), which was predominantly subnephrotic (87%) via the urine protein to creatinine ratio, and microscopic hematuria (65 vs. 42%, p = 0.014). The majority (69%) of patients who had AKI had a prerenal urine electrolyte profile, while 31% had an elevated fraction excretion of sodium. This pattern did not show significant differences by age, gender, ethnicity, or human immunodeficiency virus (HIV) status. There were also no significant differences in urinary electrolyte profile between patients with CA-AKI and HA-AKI (prerenal profile in CA-AKI vs. HA-AKI = 64 vs. 81%, p = 0.436) as well as those intubated and non-intubated (39 vs. 31%, p = 0.140). The development of AKI was associated with poor clinical outcomes, including death, need for intubation, and need for vasopressors (Table 1). Moreover, patients who had HA-AKI did worse with these clinical outcomes when compared to patients who had CA-AKI (Fig. 2, 3). Multivariate logistic regression found that age (OR 1.029, 95% CI 1.001–1.057, p = 0.042) and history of heart failure (OR 3.851, 95% CI 1.121–13.231, p = 0.032) were shown to be significantly associated with AKI after adjusting for baseline eGFR (Table 2).
Fig. 1.
Flow diagram for the study.
Table 1.
Characteristics and outcomes of patients with COVID-19
| With AKI (n = 110) | Without AKI (n = 113) | p value | |||
|---|---|---|---|---|---|
| Age | 70.33±12.18 | 61.60±16.15 | <0.0001 | ||
| Female gender | 50 (46) | 58 (51) | 0.422 | ||
| Ethnicity/race | |||||
| African American | 73 (66) | 78 (69) | 0.388 | ||
| Caucasian | 6 (6) | 8 (7) | |||
| Hispanic | 11 (10) | 15 (13) | |||
| Other | 20 (18) | 12 (11) | |||
| Comorbidities | |||||
| BMI | 28.97±8.31 | 30.69±9.97 | 0.172 | ||
| COPD | 12 (11) | 15 (13) | 0.683 | ||
| Asthma | 6 (6) | 12 (11) | 0.219 | ||
| Heart failure | 19 (17) | 5 (4) | 0.002 | ||
| Atrial fibrillation | 15 (14) | 8 (7) | 0.126 | ||
| Diabetes | 58 (53) | 46 (41) | 0.082 | ||
| Coronary artery disease | 20 (18) | 15 (13) | 0.360 | ||
| Hypertension | 104 (80) | 76 (64) | 0.003 | ||
| ACE/ARB | 39 (36) | 39 (35) | 0.889 | ||
| CKD (any stage) | 29 (27) | 10 (9) | 0.001 | ||
| CKD stage 1 | 44 (40) | 59 (52) | 0.014 | ||
| CKD stage 2 | 38 (35) | 42 (37) | |||
| CKD stage 3A | 11 (10) | 10 (9) | |||
| CKD stage 3B | 9 (8) | 0 (0) | |||
| CKD stage 4 | 8 (7) | 2 (2) | |||
| CA-AKI | 79 (32) | N/A | |||
| HA-AKI | History of kidney transplant | 31 (28) | 3 (3) | 2 (2) | 0.680 |
| History of HIV | 4 (4) | 2 (2) | 0.442 | ||
| Clinical and lab parameters | |||||
| FiO2% requirement on admission | 44±32 | 34±23 | 0.007 | ||
| Serum ferritin on admission, ng/mL | 1,942±2,977 | 1,385±2,065 | 0.201 | ||
| D-Dimer on admission, ng/mL | 5,468±7,990 | 2,248±3,125 | 0.004 | ||
| CRP on admission, mg/L | 160±129 | 120±89 | 0.097 | ||
| Procalcitonin on admission, ng/mL | 2.53±6.06 | 1.91±8.52 | 0.645 | ||
| Baseline eGFR | 79.61±36.86 | 99.16±49.52 | 0.002 | ||
| eGFR on admission | 47.73±29.91 | 90.55±47.09 | <0.0001 | ||
| Lowest eGFR in 1st week | 34.99±23.70 | 84.53±38.66 | <0.0001 | ||
| Hydroxychloroquine | 67 (61) | 64 (57) | 0.587 | ||
| Steroids | 30 (27) | 19 (17) | 0.075 | ||
| Tocilizumab | 10 (9) | 9 (8) | 0.814 | ||
| Clinical outcomes | |||||
| Inpatient death | 34 (31) | 10 (9) | <0.0001 | ||
| Need for vasopressors | 35 (32) | 7 (6) | <0.0001 | ||
| Need for intubation | 37 (34) | 11 (10) | <0.0001 | ||
Data are presented as the mean ± SD or n (%). CKD, chronic kidney disease; CA-AKI, community-acquired AKI; HA-AKI, hospital-acquired AKI; N/A, not available.
Fig. 2.
Clinical outcomes of COVID-19 patients with CA-AKI, HA-AKI, and without AKI.
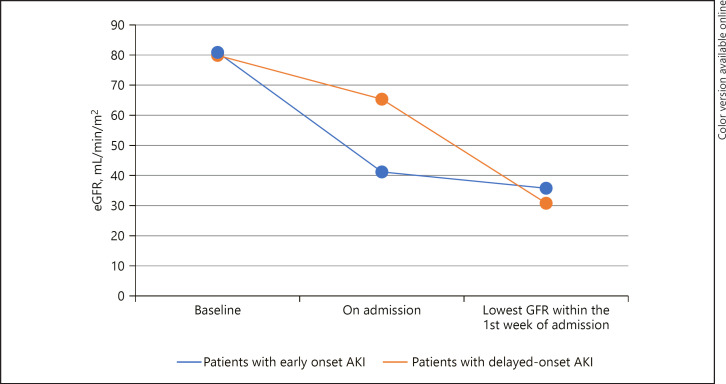
Fig. 3.
GFR trend of COVID-19 patients with CA-AKI versus HA-AKI.
Table 2.
Multivariate logistic regression on factors associated with AKI among patients admitted with COVID-19
| OR | 95% lower limit | 95% upper limit | p value | |
|---|---|---|---|---|
| Age | 1.029 | 1.001 | 1.057 | 0.042 |
| BMI | 1.002 | 0.964 | 1.042 | 0.907 |
| Male | ref. | |||
| Female | 0.705 | 0.373 | 1.330 | 0.280 |
| African American | ref. | |||
| Caucasian | 1.874 | 0.451 | 7.785 | 0.387 |
| Hispanic | 0.897 | 0.305 | 2.635 | 0.843 |
| Other | 1.597 | 0.589 | 4.334 | 0.358 |
| COPD | 0.801 | 0.287 | 2.234 | 0.671 |
| DM | 1.435 | 0.747 | 2.754 | 0.278 |
| HTN | 1.750 | 0.791 | 3.872 | 0.167 |
| HF | 3.851 | 10.121 | 13.231 | 0.032 |
| CAD | 0.543 | 0.215 | 1.373 | 0.197 |
| Baseline eGFR | 0.991 | 0.982 | 1.000 | 0.055 |
| ACEi/ARB use | 0.663 | 0.335 | 1.313 | 0.239 |
OR, odds ratio; COPD, chronic obstructive pulmonary disease; DM, diabetes mellitus; HTN, hypertension; HF, heart failure; CAD, coronary artery disease; eGFR, estimated glomerular filtration rate; ACEi/ARB, angiotensin-converting enzyme inhibitors/angiotensin receptor blockers.
Discussion
In this retrospective study conducted in a tertiary hospital serving predominantly African American patients as well as patients from lower socioeconomic strata, we observed that almost half of those hospitalized with confirmed COVID-19 had CA-AKI. This reported incidence of AKI among COVID-19 patients is higher compared to those reported from other regions, including China, the USA, and Europe, since December 2019 [3, 4, 5, 6].
African American patients have a low renin phenotype leading to a suppressed renin-angiotensin-aldosterone system. This theoretically confers a benefit in terms of developing acute respiratory distress syndrome, which is associated with angiotensin-1 (AT1) receptor activation [12]. However, given their high prevalence of comorbidities, including heart failure, chronic kidney disease, diabetes, and hypertension, they are also at very high risk of developing COVID-19-related complications, including AKI [8, 9, 10, 13, 14]. However, African American race was not a predictor of AKI in this analysis, which may be the result of a small sample size.
Interestingly, heart failure was consistently associated with AKI in this report, even after adjusting for baseline eGFR in the multivariate logistic regression, suggesting a possible cardiorenal component to the development of worsening kidney function. In COVID-19 patients with underlying heart failure, the development of cardiorenal syndrome (CRS) could be a type 1 CRS phenotype as a sequela of cardiomyopathy from cytokine storm/myocarditis, a type 3 CRS phenotype from the development of AKI leading to cardiac myocyte injury, or a type 5 CRS presentation with dual cardiac and kidney injury from inflammation, increased vascular permeability, and microthrombi [15, 16]. Also, heart-kidney crosstalk could be contributory to this observation, as cardiomyopathy can lead to renal congestion, hypotension, and renal hypoperfusion leading to a reduction in GFR [16]. In relation to this, one retrospective study reported a significant association of right ventricular dilation to mortality among COVID-19 patients [17]. The myocardial dysfunction can possibly be attributed to hypoxia, thrombotic events, direct viral damage, and cytokine storm in critically ill patients with severe COVID-19 [17]. This then can also potentially induce renal dysfunction via the cardiorenal mechanism. It is notable that in our study there was a significantly greater number of patients who needed intubation in the group with AKI compared to those without. High requirements of positive pressure ventilation in patients with COVID-19 can lead to potential deleterious hemodynamic consequences of reduced cardiac output contributing to renal hypoperfusion, which may play a role in the mechanism of AKI in these patients [18]. In our study, more patients who were intubated had a higher rate of having a pre-renal profile compared to those who were not intubated. However, this was not statistically significant, which is probably related to the relatively low absolute number of intubations.
This finding is also clinically relevant in the context of reported data on clinical outcomes with the use of angiotensin-converting enzyme inhibitors/angiotensin receptor blockers (ACEi/ARB) in patients with COVID-19 [8, 19, 20]. Similarly, the premature discontinuation of ACE inhibitors or ARBs in heart failure patients in the absence of hemodynamic deterioration or severe hyperkalemia is associated with harm [21]. In our study, ACE/ARB use was not associated either AKI or inpatient death. Thus, it may be prudent to consider maintenance of ACEI/ARB in patients with COVID-19, particularly in those with a history of heart failure unless contraindicated by hemodynamic compromise or refractory hyperkalemia. Future controlled studies looking at the effect of the renin angiotensin aldosterone blockade on cardiovascular and kidney outcomes are necessary to better define the risk/benefit profile with these agents in patients with COVID-19.
In terms of other laboratory and clinical findings among our COVID-19 patients, a few observations were made. Patients with AKI have significantly higher D-dimer levels as well as FiO2 requirement on admission than those without AKI. Given these findings, it is possible that this particular group of patients had more severe COVID-19 disease on presentation. Although D-dimer has low specificity, several studies have suggested that it can be elevated in patients with AKI with activated coagulation being associated with systemic inflammatory syndrome as well in severe COVID-19 infection, along with increased interleukin-6 levels, as a consequence of cytokine release syndrome [22, 23]. Furthermore, increased FiO2 requirement indicative of alveolar damage and impending respiratory failure can also contribute to renal medullary hypoxia compounding the renal hypoperfusion from combined effects of endothelial damage, third-space fluid loss, and hypotension from COVID-19-induced cytokine release [16]. Urine sediments of these patients showed subnephrotic proteinuria and microhematuria, which are consistent with autopsy findings of kidney tissues of COVID-19 patients showing severe acute tubular necrosis with macrophage infiltration of the tubulointerstitium and diffuse proximal tubule injury with erythrocyte aggregates obstructing the peritubular capillaries [24]. Lastly, patients with AKI had significantly lower GFR both at baseline and during admission compared to those without AKI, which is congruous with the well-established observation that a low eGFR at baseline is considered to be a strong and consistent risk factor for AKI, especially in the critical care setting [25].
The findings from our study are in consonance with those from prior studies suggesting that AKI is a significant risk factor of clinical deterioration and in-hospital death among COVID-19 patients [3, 4, 6, 26]. Patients who developed HA-AKI had poorer outcomes than those with CA-AKI. Perhaps those with CA-AKI may have had easily reversible etiologies, including poor oral intake and volume depletion, while HA-AKI may signal multiorgan deterioration in the setting of severe COVID-19 or even the influence of decompensated cardiorenal mechanisms in our population. This assumption is supported by the fact that a low urine fractional excretion of sodium (FENa) or urea (FEUrea) profile was seen in the majority of the AKI patients regardless of age, gender, ethnicity, HIV status, and onset of AKI. Arguably, low FENa can also be seen in clear-cut causes of intrinsic kidney disease, such as contrast-induced nephropathy, acute interstitial nephritis, rhabdomyolysis, and early tubular obstruction, which could be present in our patients as well [27]. However, based on the comparison between urine electrolytes in CA-AKI and HA-AKI, a prerenal urine index per se does not help prognosticate AKI in these patients given the lack of serial profiling and documenting any change in this pattern, especially in those with HA-AKI.
Limitations
Our study has some limitations. First, the definition of AKI did not include the urine volume-based criterion of <0.5 mL/kg/h for 6 h [11], which may have led to underestimation of AKI incidence. Also, given the retrospective nature of the study, we were not able to determine if the change in kidney function at baseline was within the 48-h time period component as per the criteria of CA-AKI. Second, the effects of COVID-19 on long-term outcomes could not be assessed. The data for this study came from a single-center retrospective analysis with a predominance of African American patients with high rates of medical comorbidities; thus, the generalizability of the study findings may be limited. Third, the presence of hematuria was only ascertained using routine urinalysis by the laboratory; thus, the diagnostic significance of this finding is limited. Lastly, the majority of patients had a pre-renal urine electrolyte profile in both CA-AKI versus HA-AKI, but we were not able to determine whether patients with HA-AKI might have subsequently progressed to a different urine electrolyte profile due to the lack of serial urine electrolyte profiling.
Conclusion
The burden of AKI among underserved patients with COVID-19 who are predominantly African Americans with multiple comorbidities was high. HA-AKI is associated with worse clinical outcomes compared to CA-AKI. A history of heart failure is an independent predictor of AKI with COVID-19. Other pathophysiologic mechanisms for AKI development, such as cardiorenal syndrome, have to be considered among COVID-19 patients with multiple comorbidities.
Statement of Ethics
This study was approved by the institutional review board (IRB No. 2020-435).
Disclosure Statement
None of the authors have any conflicts of interest to disclose.
Funding Sources
There was no monetary or material support for this research investigation.
Author Contributions
Dr. Pelayo, Dr. Lo, and Dr. Rangaswami designed the study, analyzed/interpreted its results, and drafted and critically revised the paper. Dr. Gopalakrishnan conceived the original idea and supervised the study. All the other authors (Dr. Bhargav, Dr. Gul, Dr. Peterson, Dr. DeJoy III, Dr. Salacup, Dr. Albano, Dr. Azmaiparashvili, and Dr. Aponte) helped in collecting the data, contributed to the analysis and discussion of the results, and approved the final version of the study.
References
- 1.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020 Feb;395((10223)):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Zhou X, Chen K, Zou J. Single-cell RNA-seq data analysis on the receptor ACE2 expression reveals the potential risk of different human organs vulnerable to 2019-nCoV infection. Front Med. 2020;14((2)):185–92. doi: 10.1007/s11684-020-0754-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cheng Y, Luo R, Wang K, Zhang M, Wang Z, Dong L, et al. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020 May;97((5)):829–38. doi: 10.1016/j.kint.2020.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Guan WJ, Ni ZY, Hu Y, Liang WH, Ou CQ, He JX, et al. China Medical Treatment Expert Group for Covid-19 Clinical Characteristics of Coronavirus Disease 2019 in China [published online ahead of print February 28, 2020] N Engl J Med. 2020 Apr;382((18)):1708–20. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Arentz M, Yim E, Klaff L, Lokhandwala S, Riedo FX, Chong M, et al. Characteristics and outcomes of 21 critically ill patients with COVID-19 in Washington State [published online ahead of print March 19, 2020] JAMA. 2020 Mar;323((16)):1612. doi: 10.1001/jama.2020.4326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.ICNARC − Reports. https://www.icnarc.org/Our-Audit/Audits/Cmp/Reports. Accessed April 23, 2020.
- 7.Yancy CW. COVID-19 and African Americans [published online ahead of print April 15, 2020] JAMA. 2020 Apr;323((19)):1891. doi: 10.1001/jama.2020.6548. [DOI] [PubMed] [Google Scholar]
- 8.Mehra MR, Desai SS, Kuy S, Henry TD, Patel AN. Cardiovascular disease, drug therapy, and mortality in Covid-19 [published online ahead of print May 1, 2020] N Engl J Med. 2020 May;:NEJMoa2007621. doi: 10.1056/NEJMoa2007621. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 9.Du RH, Liang LR, Yang CQ, Wang W, Cao TZ, Li M, et al. Predictors of mortality for patients with COVID-19 pneumonia caused by SARS-CoV-2: a prospective cohort study [published online ahead of print April 8, 2020] Eur Respir J. 2020 May;55((5)):2000524. doi: 10.1183/13993003.00524-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zheng Z, Peng F, Xu B, Zhao J, Liu H, Peng J, et al. Risk factors of critical & mortal COVID-19 cases: A systematic literature review and meta-analysis [published online ahead of print April 23, 2020] J Infect. 2020 Apr;S0163-4453((20)):30234–6. doi: 10.1016/j.jinf.2020.04.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kidney International. KDIGO clinical practice guidelines for acute kidney injury. Kidney Int Suppl. 2012;2:8–12. [Google Scholar]
- 12.Zilbermint M, Hannah-Shmouni F, Stratakis CA. Genetics of Hypertension in African Americans and Others of African Descent. Int J Mol Sci. 2019 Mar;20((5)):1081. doi: 10.3390/ijms20051081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Park YW, Zhu S, Palaniappan L, Heshka S, Carnethon MR, Heymsfield SB. The metabolic syndrome: prevalence and associated risk factor findings in the US population from the Third National Health and Nutrition Examination Survey, 1988-1994. Arch Intern Med. 2003 Feb;163((4)):427–36. doi: 10.1001/archinte.163.4.427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Lea JP, Greene EL, Nicholas SB, et al. Cardiorenal metabolic syndrome in the african diaspora: rationale for including CKD in the metabolic syndrome definition. Ethn Dis. 2009;19((suppl 2)):11–4. [PubMed] [Google Scholar]
- 15.Rangaswami J, Bhalla V, Blair JE, Chang TI, Costa S, Lentine KL, et al. American Heart Association Council on the Kidney in Cardiovascular Disease and Council on Clinical Cardiology Cardiorenal Syndrome: Classification, Pathophysiology, Diagnosis, and Treatment Strategies: A Scientific Statement From the American Heart Association. Circulation. 2019 Apr;139((16)):e840–78. doi: 10.1161/CIR.0000000000000664. [DOI] [PubMed] [Google Scholar]
- 16.Ronco C, Reis T. Kidney involvement in COVID-19 and rationale for extracorporeal therapies [published online ahead of print April 9, 2020] Nat Rev Nephrol. 2020 Jun;16((6)):308–10. doi: 10.1038/s41581-020-0284-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Arguilan E, Sud K, Vogel B, et al. Right ventricular dilatation in hospitalized patients with COVID-19 infection [published online ahead of print May 7, 2020] JACC Cardiovasc Imaging. doi: 10.1016/j.jcmg.2020.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Koyner JL, Murray PT. Mechanical ventilation and the kidney. Blood Purif. 2010;29((1)):52–68. doi: 10.1159/000259585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Mancia G, Rea F, Ludergnani M, Apolone G, Corrao G. Renin–angiotensin–aldosterone system blockers and the risk of Covid-19 [published online ahead of print May 1 2020] N Engl J Med. 2020 May;:NEJMoa2006923. doi: 10.1056/NEJMoa2006923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Reynolds HR, Adhikari S, Pulgarin C, Troxel AB, Iturrate E, Johnson SB, et al. Renin-Angiotensin-Aldosterone System Inhibitors and Risk of Covid-19 [published online ahead of print May 1 2020] N Engl J Med. 2020 May;:NEJMoa2008975. doi: 10.1056/NEJMoa2008975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Oliveros E, Oni ET, Shahzad A, Kluger AY, Lo KB, Rangaswami J, et al. Benefits and Risks of Continuing Angiotensin-Converting Enzyme Inhibitors, Angiotensin II Receptor Antagonists, and Mineralocorticoid Receptor Antagonists during Hospitalizations for Acute Heart Failure [published online ahead of print February 14, 2020] Cardiorenal Med. 2020;10((2)):69–84. doi: 10.1159/000504167. [DOI] [PubMed] [Google Scholar]
- 22.Gordge MP, Faint RW, Rylance PB, Ireland H, Lane DA, Neild GH. Plasma D dimer: a useful marker of fibrin breakdown in renal failure. Thromb Haemost. 1989 Jun;61((3)):522–5. [PubMed] [Google Scholar]
- 23.Simmons EM, Himmelfarb J, Sezer MT, Chertow GM, Mehta RL, Paganini EP, et al. PICARD Study Group Plasma cytokine levels predict mortality in patients with acute renal failure. Kidney Int. 2004 Apr;65((4)):1357–65. doi: 10.1111/j.1523-1755.2004.00512.x. [DOI] [PubMed] [Google Scholar]
- 24.Su H, Yang M, Wan C, et al. Renal histopathological analysis of 26 postmortem findings of patients with COVID-19 in China. Kidney Int. 2020 doi: 10.1016/j.kint.2020.04.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Grams ME, Sang Y, Ballew SH, Gansevoort RT, Kimm H, Kovesdy CP, et al. CKD Prognosis Consortium A Meta-analysis of the Association of Estimated GFR, Albuminuria, Age, Race, and Sex With Acute Kidney Injury. Am J Kidney Dis. 2015 Oct;66((4)):591–601. doi: 10.1053/j.ajkd.2015.02.337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Xiao G, Hu H, Wu F, et al. Acute kidney injury in patients hospitalized with COVID-19 in Wuhan, China: a singlecenter retrospective observational study. medRxiv. 2020. https://doi.org/10.1101/2020.04.06.20055194 https://www.medrxiv.org/content/10.1101/2020.04.06.20055194v1. [DOI] [PMC free article] [PubMed]
- 27.Carvounis CP, Nisar S, Guro-Razuman S. Significance of the fractional excretion of urea in the differential diagnosis of acute renal failure. Kidney Int. 2002 Dec;62((6)):2223–9. doi: 10.1046/j.1523-1755.2002.00683.x. [DOI] [PubMed] [Google Scholar]