Abstract
Background
SARS‐CoV‐2 coronavirus infection ranges from asymptomatic through to fatal COVID‐19 characterized by a ‘cytokine storm’ and lung failure. Vitamin D deficiency has been postulated as a determinant of severity.
Objectives
To review the evidence relevant to vitamin D and COVID‐19.
Methods
Narrative review.
Results
Regression modelling shows that more northerly countries in the Northern Hemisphere are currently (May 2020) showing relatively high COVID‐19 mortality, with an estimated 4.4% increase in mortality for each 1 degree latitude north of 28 degrees North (P = 0.031) after adjustment for age of population. This supports a role for ultraviolet B acting via vitamin D synthesis. Factors associated with worse COVID‐19 prognosis include old age, ethnicity, male sex, obesity, diabetes and hypertension and these also associate with deficiency of vitamin D or its response. Vitamin D deficiency is also linked to severity of childhood respiratory illness. Experimentally, vitamin D increases the ratio of angiotensin‐converting enzyme 2 (ACE2) to ACE, thus increasing angiotensin II hydrolysis and reducing subsequent inflammatory cytokine response to pathogens and lung injury.
Conclusions
Substantial evidence supports a link between vitamin D deficiency and COVID‐19 severity but it is all indirect. Community‐based placebo‐controlled trials of vitamin D supplementation may be difficult. Further evidence could come from study of COVID‐19 outcomes in large cohorts with information on prescribing data for vitamin D supplementation or assay of serum unbound 25(OH) vitamin D levels. Meanwhile, vitamin D supplementation should be strongly advised for people likely to be deficient.
Keywords: vitamin D, COVID‐19, cytokine

Introduction
The SARS‐CoV‐2 coronavirus is an enveloped RNA virus, infection by which provokes a remarkable range of responses from complete lack of symptoms through to cytokine storm and life‐threatening acute respiratory distress syndrome (ARDS) [1, 2]. The explanations for this extremely variable prognosis are unclear. Mortality from coronavirus infectious disease 2019 (COVID‐19) is higher amongst people who are older, male, obese, diabetic, hypertensive, or who are from black, Asian, or minority ethnic (BAME) demographics. All these factors are associated with increased prevalence of vitamin D deficiency or, as in male sex, with reduced impact of vitamin D on the immune response. Vitamin D is, like cortisone and sex hormones, a cholesterol‐derived steroid hormone, and it modulates expression of around 5% of human genes including many relevant to the immune response to pathogens. We have therefore examined the evidence that vitamin D deficiency might be a factor determining severity of COVID‐19.
Association between northerly latitude and increased mortality from COVID‐19
There is currently (May 2020) a significant association between northerly latitude and mortality from COVID‐19 expressed per million population across the 117 countries with more than 1 million population and more than 150 recorded cases at time of sampling (Fig. 1) [3]. Much of this association is due to the younger age of populations in some countries. Adjusting for per cent of population ≥ 65 years does however leave a significant relationship between latitude and COVID‐19 mortality (P = 0.031) with an estimated 4.4% increase in mortality for each 1 degree latitude north of 28 degrees North (Table 1). Addition of neither pollution (particles of matter < 2.5 µm diameter (PM2.5) micrograms per m3) nor population density per country added significant explanatory power to a model containing latitude and age. An association between northerly latitude and mortality has also been noted amongst African Americans across the United States [4].
Figure 1.
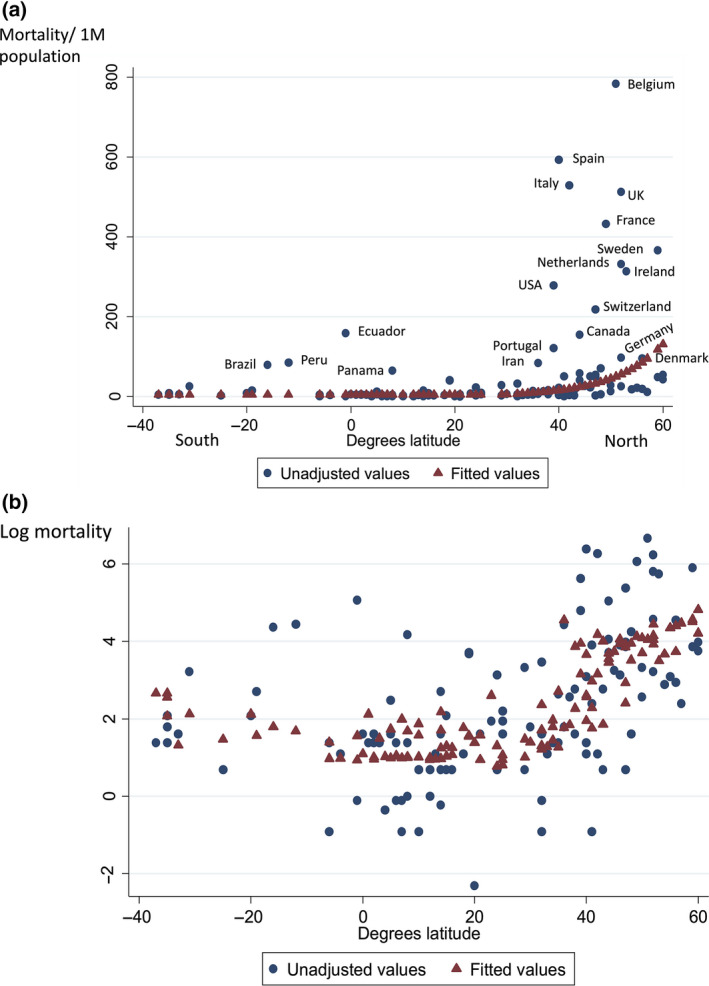
(a) COVID‐19 mortality per 1 million population by country compared with latitude of capital cities. Fitted values are derived from a piecewise linear model of the logarithm of mortality on latitude. This was based on a threshold of 28 degrees North that explained the greatest amount of variation. (b) Logarithm of COVID‐19 mortality per 1 million compared with latitude with and without adjustment for age (%≥65 years). Data accessed 18 May 2020. Reproduced from [3] with permission.
Table 1.
Associations between COVID‐19 mortality by country, latitude and % of population ≥ 65 years (from [3], data accessed 18 May 2020).
| Variable | Regression coefficient | Standard error | P‐value | % of variation explained | Effect size (95% CI) a |
|---|---|---|---|---|---|
| Univariate models | |||||
| Latitude | 0.1074 | 0.0142 | <0.0005 | 33.1 | 11.3% (8.3–14.5%) |
| % ≥65 | 0.1766 | 0.0199 | <0.0005 | 40.4 | 19.3% (14.8–24.1%) |
| Multivariate model | |||||
| Latitude | 0.0428 | 0.0196 | 0.031 | 43.0 | 4.4% (0.4–8.5%) |
| % ≥65 | 0.1281 | 0.0291 | <0.0005 | 13.7% (7.4–20.3%) | |
The effect size is, for latitude, the percentage increase in mortality from one location, situated at least 28°north, to another location one degree further north and, for % ≥ 65, the percentage increase in mortality for each one % increase in % ≥65
Associations between COVID‐19 mortality and latitude suggest a possible effect of ultraviolet light. A substantial source of vitamin D comes from synthesis in the skin from its precursor 7‐dehydrocholesterol as a consequence of ultraviolet light (UVB) exposure. People living far from the equator may therefore become vitamin D deficient in the winter and spring, with levels lowest from December to May [5]. It is estimated that at latitudes below 35 degrees, either side of the equator UVB radiation is sufficient for year‐round vitamin D synthesis, although this will also depend on diet, skin colour, clothing, time spent outdoors and use of sunscreen [6].
Interpretation of country‐to‐country variation is further complicated by variable approaches to supplementation and vitamin D food fortification initiatives. Thus, vitamin D levels are generally well maintained despite relative lack of UVB exposure in Nordic countries due to widespread use of supplements and food fortification, whereas deficiency is commoner in the United Kingdom and in southern European countries, [7, 8] and particularly amongst persons over 80 years and those in institutions. We have recently reported a significant correlation between COVID‐19 mortality and reported average serum vitamin D levels across European countries [9]. Vitamin D deficiency defined as <30 nmol L−1 is found in over 10% of Europeans [7], but it has been suggested that a level of at least 50 nmol L−1 may be optimal [6]. Wuhan itself, where the outbreak started, is at latitude 31 degrees north; however, air pollution is also a major factor limiting UVB radiation and has previously been very marked over this densely populated (11M) city [10]. There are currently no population‐based vitamin D data available from Wuhan. In sunnier Brazil, whose capital Brasilia is at −16 degrees latitude, there is now high mortality but meta‐analysis has shown 28% prevalence of vitamin D deficiency [11].
Alternative explanations for the north‐south gradient in COVID‐19 mortality are arguably less plausible. Although population density expressed per country does not currently associate with COVID‐19 mortality, it could be speculated that cities tend to be smaller and urban populations less densely crowded further south but there are many examples of high population cities below 28 degrees latitude north – Karachi 14.9M, Hong Kong 7.4M, Mexico City 8.9M, Nairobi 4.3M and Sidney 5.2M, for example. It can also be pointed out that there is a north‐south gradient for diagnosed cases, however, if true – for diagnosis rate is of course very dependent on testing frequency ‐ this may well reflect the longer period of infectivity that is associated with more severe COVID‐19 illness. It does not seem very likely that the virus has simply had less opportunity to spread south of the equator given that we are now 4 months into the pandemic.
Ultraviolet light, particularly UVB, has also been shown to have direct immunosuppressive effects on the skin, that include suppression of contact sensitivity at the UV‐irradiated site and induction of antigen‐specific tolerance mediated by regulatory T lymphocytes. Significant systemic immunosuppression has also been demonstrated in experimental models [12]. Various mediators are thought to be responsible for these effects. These include cis‐urocanic acid, generated by the isomerization of trans‐urocanic acid, which binds to the serotonin receptor on antigen‐presenting cells, keratinocytes and mast cells, and also cyclobutene pyrimidine dimers, generated by UV‐mediated nucleotide damage, and oxidation products of membrane lipids.
UV light could also reduce viability of free virus in the environment. Although UVC light (200–280 nm wavelength) that has a strong germicidal effect does not penetrate the earth’s atmosphere, UVB (280–320 nm) has a weaker but significant anti‐viral effect that may shorten the survival of the virus on surfaces and thus reduce infection rates [13]. Higher temperature and higher humidity can also decrease viral survival in the environment and have been shown to correlate with reduced COVID‐19 infection rates and mortality across 166 countries [14]. The evidence is contradictory though: a recent study in the United States has linked UVB, and higher temperature, but not rainfall with lower SARS‐CoV‐2 infection rates [15], whereas a study across Chinese cities has shown no association with either UVB or temperature and R 0 or infection rates [16].
Thus, although the association with latitude implies that COVID‐19 may prove seasonal, the mechanisms underlying this could include any or all of impacts of UVB on the immune system mediated by vitamin D synthesis, other consequences of the actions of UVB in the skin and direct effects on environmental survival of SARS‐CoV‐2 consequent to UVB, humidity or temperature. Of these, only an effect of UVB mediated by vitamin D synthesis would readily explain the associations between COVID‐19 mortality and ethnicity.
Vitamin D chemistry and biology
Vitamin D, uniquely amongst the vitamins, is a steroid hormone. It is fat‐soluble and exists in two forms, vitamin D2 (ergocalciferol) and vitamin D3 (cholecalciferol). Both are generated by the action of UVB, splitting a single (9,10) carbon–carbon bond in their respective precursors – ergosterol and cholesterol thus generating a secosteroid or ‘cut’ steroid. Ergosterol is the precursor in fungi and plankton and cholesterol the precursor in animals. The chemical structure of vitamin D therefore has close similarities with that of the other cholesterol‐derived hormones such as cortisol, aldosterone, testosterone and oestrogen (Fig. 2). Vitamin D3 has greater affinity for the circulating vitamin D binding protein and a substantially longer half‐life in the circulation than vitamin D2 [18]. This is probably only of major significance if taken by intermittent bolus dosing rather than daily supplementation [19]. After the generation of cholecalciferol (or ergocalciferol), further hydroxylation is required in the liver and then the kidney to generate the active 1,25 dihydroxycholecalciferol. It should be noted though that macrophages/dendritic cells also have the ability to convert 25(OH)D to 1,25(OH)2D via CYP27B1 and that clinical consequences of vitamin D deficiency correlate better with serum concentration of 25(OH)D rather than with the (1000‐fold lower) serum concentration of 1,25(OH)2D [20]. Although lymphocytes also express CYP27B1, this is at substantially lower level and the regulatory (anti‐inflammatory) effect of vitamin D on human lymphocytes in mixed cell culture requires the presence of antigen‐presenting dendritic cells [21]. The daily requirement of vitamin D is estimated at between 5 and 20 µg (200 to 800 IU) [6], and it is not easy to achieve this through diet alone. Oily fish is the only really substantial natural dietary source though farmed versus wild fish concentrations vary. Liver and eggs also contain vitamin D but a single egg only provides about 5% of the daily requirement. Mushrooms need to be subjected to UV irradiation and even then will only provide modest amounts of vitamin D2. For the majority of people, the main source of vitamin D is its generation by the action of UVB on cholesterol in the skin. This is evidenced by the fact that in the UK, blood levels of vitamin D are approximately 50% lower in February than in September [22] (Fig. 2). That seasonal differences are not even greater reflects the long half‐life of vitamin D in the body (2–3 months), predominantly in fat stores, in contrast to its relatively short half‐life in the blood (2–3 weeks) [23].
Figure 2.
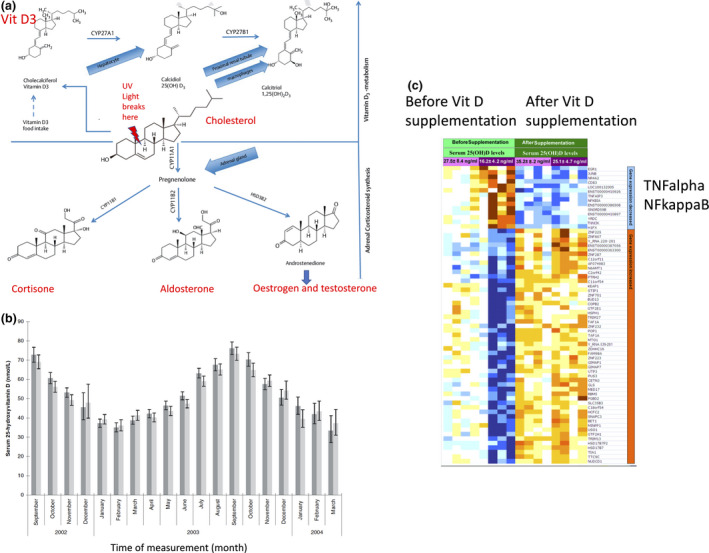
(a) Synthesis of vitamin D, adrenal and sex hormones from cholesterol. Adapted from Muscogiuri et al. [17] with permission. (b) Seasonal variation in serum vitamin D levels (mean [95% CI]) amongst 7437 white British (1958 British birth cohort) at age 45. Dark bar = male, pale bar = female. From Hypponen and Power [22] with permission. (c) Heatmaps of vitamin D‐responsive genes in mixed leucocytes from individuals before and after 2 months of vitamin D supplementation. Four individuals had prior vitamin D deficiency, and 4 individuals had prior normal levels. Blue = decreased expression. Brown = increased expression. It can be seen that vitamin D responsiveness equalized between the two sets of individuals after supplementation. From Hossain‐nezhad et al. [28] with permission.
Vitamin D is best known for its effects on calcium and phosphate absorption, osteoclast activation, and hence on bone calcification and muscle strength [24]. However, the vitamin D receptor is very widely expressed, including by all leucocyte classes [25].
In the blood, approximately 85% of vitamin D is bound to vitamin D binding protein (DBP), 15% to albumin and just 0.03% of 25(OH)D3 and 0.4% of total 1,25(OH)2D3 are free vitamin D [26]. It is thought that in most cells, only free vitamin D can enter the cell. Cellular entry by protein‐bound vitamin D is dependent on expression of the cell surface receptor proteins megalin and cubilin [20] and is largely restricted to the kidney, parathyroid and placenta. Free vitamin D diffuses through the plasma membrane and binds to the vitamin D receptor (VDR) in the cell nucleus where the vitamin D/VDR complex then interacts with vitamin D response elements in the genome. It is estimated that vitamin D affects the transcription of around 1000 genes, that is around 5% of the human genome [27]. In pooled leucocytes, it has been shown that at least 60 genes are vitamin D‐responsive [28] and nearly two hundred genes in monocyte/macrophage cells [29, 30, 31] (Fig. 2). Vitamin D therefore has substantial effects on the immune system that are highly relevant to the response to pathogens.
Impact of vitamin D on immunological response to pathogens – clinical studies
A protective effect of cod‐liver oil in tuberculosis was recognized in an early therapeutic trial conducted at London’s Brompton hospital in the 1840s [32] and the role of vitamin D in this effect was recognized in the 1940s, initially with the successful treatment of cutaneous tuberculosis and subsequently with many confirmatory studies [33]. Since then, knowledge of the impacts of vitamin D on the immune system has expanded greatly and there has been increasing recent focus into its role in determining response to viral infections, particularly respiratory viruses.
The evidence both from laboratory studies and from clinical studies is that vitamin D status has probably only a small impact on risk for viral infection but a much more important impact on inflammatory response and hence severity. Thus, a meta‐analysis looking at impact of supplementary vitamin D on risk for upper respiratory tract infection showed a statistically significant but very modest reduction, from 42.2% to 40.3%, in risk of one or more infections [34]. However, amongst those who were vitamin D deficient at baseline the reduction in infection rate was greater – from 55.0% down to 40.5%. A beneficial effect was only seen with regular daily dosing and not with intermittent bolus dosing. Probably more impressive is the association between vitamin D deficiency and severity of respiratory disease – for example the need for intensive care in 1016 infants hospitalized with bronchiolitis (22% if vitamin D < 20 ng mL−1 (50 nmol L−1), compared with 12% if vitamin D > 30 ng mL−1 (75 nmol L−1); P = 0.003) [35].
A study in Irish people >60 years old and healthy apart from hypertension showed a strong correlation between vitamin D deficiency and increase in both IL‐6 and C‐reactive protein [36]. A detailed investigation of the impacts of vitamin D, gender and seasonality on cytokines has subsequently been undertaken in 534 healthy subjects as part of the Human Functional Genomics Project [37]. This showed that monocyte inflammatory cytokine responses to lipopolysaccharide and Candida albicans are substantially greater in men. Several inflammatory cytokines, including TNF‐alpha, interleukin beta and interleukin 6, were shown to be higher in summer, mostly showing no relationship with vitamin D.
Interpreting serum vitamin D levels during illness – the negative acute phase effect
Studies of vitamin D levels in individuals who are already ill or have raised inflammatory markers have to be interpreted with caution. Controlled studies in calves infected with bovine diarrhoea virus (BVDV) showed that serum vitamin D levels fell by 57% during the acute phase response to illness [38] and similar falls have been documented in humans following orthopaedic surgery and acute pancreatitis [39]. Both serum vitamin D binding protein (DBP) and albumin concentrations fall in illness and total vitamin D levels will fall as a consequence. It is therefore almost inevitable that there will be a correlation between lower total serum vitamin D levels and increased COVID‐19 severity. There are several possible ways of getting around this problem:
Measurement of free vitamin D
Total 25(OH)D serum concentration in serum is generally acknowledged to be the de facto biomarker of vitamin D status. However, as already noted, the unbound (i.e. free) concentration of 25(OH)D is below 0.5% of the total concentration [26, 40]. Emerging evidence points to a critical role for free, rather than total 25(OH)D in mediating important cellular processes related to immunity. For instance, in vitro studies have demonstrated reduced immune functions of dendritic cells [21] and adherent monocytes [41] by increasing DBP in culture media which reduces the amount of free 25 (OH)D. This is analogous to free thyroid hormones or testosterone which are physiologically more relevant than their total concentration.
Measurement of free 25(OH)D has been challenging due to its very low serum concentrations (approximately 10‐fold less than free thyroid hormones) and has historically relied on cumbersome radioactive tracer‐based methods [42]. More recently, two further assays, including an ELISA [43] and a high‐throughput mass spectrometric method [44] for direct measurement of free 25(OH)D, have been introduced, but require further clinical and technical validation. Thus, computational methods which rely on concentrations of total ligands and DBP and their in vitro measured affinity constant are often used to calculate free 25(OH)D [45]. It is important to note, however, that the biological significance of DBP’s various allelic forms on DBP concentrations and affinity differences is yet to be fully established. The experimentally measured affinity differences for vitamin D metabolites for various genotypic forms of DBP with the exception of one study [46] appear to be small [47, 48]. On the other hand, genotype has been consistently shown to alter serum DBP concentrations [49, 50]. In summary, the main role of DBP in determining free 25(OH)D levels appears to be DBP concentration‐dependent rather than genotype‐dependent. Thus, measurement of serum DBP and albumin alongside total 25(OH)D should allow a robust computational approach for calculating free 25(OH)D and permit a better correlation between vitamin D status and COVID‐19 severity.
Associations with vitamin D receptor polymorphism
Vitamin D receptor polymorphisms impact on vitamin D response. Meta‐analysis has shown a highly significant relationship (P = 0.007 OR 1.52) between hospitalization for respiratory syncytial virus (RSV) bronchiolitis and possession of a minor allele for a vitamin D receptor polymorphism (Fok1‐f rs2228570) that lowers transcriptional activity of the vitamin D receptor [51]. This polymorphism has an allele frequency of 13 to 38% in healthy subjects so it could be very informative to know whether this allele is also seen with higher frequency in people with more severe COVID‐19.
Mendelian randomization
The difficulty in interpreting serum vitamin D levels during illness has led investigators to consider the application of Mendelian randomization. This uses gene polymorphisms that predict vitamin D status as a surrogate for vitamin D deficiency. One approach that has proved successful has used gene polymorphisms associated with risk of skin colour, tanning, or freckling. This identified a group of gene polymorphisms that together were predictive of vitamin D status [52]. However, this included genes such as HERC2 that are major determinants of blue eye colour [53] which in turn are strongly associated with reduced pigmentation and enhanced vitamin D response to UVB in white individuals [54]. This approach may therefore be less effective in a population that contains mixed ethnicities.
A broader statistical approach is to use a genome‐wide association study (GWAS) to identify, in a hypothesis‐free fashion, polymorphisms that associate with vitamin D deficiency. This has been done in a remarkable study across 79,366 European‐ancestry individuals [55]. Polymorphisms at six loci were informative with high significance. However, the overall estimate of heritability of serum vitamin D concentrations was found to be only 7.5% and with only 38% of that heritability accounted for by the identified polymorphisms. This approach will therefore only be useable with very large sample sizes. Moreover, a different GWAS would need to be performed for other ethnic groups.
Measurement of vitamin D in hair or other tissues
Study of vitamin D levels in hair samples has been proposed as a way of avoiding the negative acute phase response effect of severe illness on serum vitamin D levels [56]. This approach is proving reliable in measurement of other steroid hormones such as cortisol [57, 58] but would need further validation.
Impact of vitamin D on immunological response to pathogens – laboratory studies
In keeping with the clinical studies, experimental evidence shows that vitamin D has shown only inconsistent effects on viral replication in human respiratory epithelial cell culture but markedly down‐regulates production of pro‐inflammatory cytokines including TNF‐alpha and IL‐6 by various mechanisms including inhibition of viral‐induced NF‐kappaB activation [59].
Vitamin D receptors are expressed by most immune cells including activated T cells, B‐cells and dendritic cells, and macrophages. Vitamin D is important for killing of phagocytosed bacteria, including Mycobacterium tuberculosis [60, 61] and E. coli, [62] by macrophages. An important part of this bactericidal effect relates to the induction by vitamin D of cathelicidin, a cationic bactericidal peptide [63]. Cathelicidin (LL‐37) can be produced not only by macrophages but also by epithelial cells and has been shown to have anti‐viral activity, particularly against enveloped viruses [64]. Vitamin D has been shown to induce an anti‐viral effect against rhinovirus in cultured respiratory epithelial cells [65], an effect that can also be demonstrated by addition of exogenous cathelicidin [66]. An effect of cathelicidin against influenza has also been shown [67]. Currently though, the impacts of vitamin D on macrophage defence against viral pathogens have demonstrated a predominant impact on cytokine response rather than on viral killing [68]. Some of the work has focussed on Dengue fever, a viral infection that is well known for its very marked cytokine activation and risk of organ failure [69, 70], although vitamin D deficiency has paradoxically been shown to correlate with reduced risk of septic shock in Dengue fever [71]. Experiments have shown consistent suppression of inflammatory cytokine response to pathogens by vitamin D, in macrophages, and also in T cells and in various animal models of pneumonia and pneumonitis [72, 73, 74]. Cytokines suppressed include IL‐6 that has been incriminated in COVID‐19‐associated ARDS.
Given that vitamin D may regulate the response of nearly two hundred genes in the monocyte/macrophage, it is not surprising that its suppressive effect on macrophage cytokine responsiveness has been shown to be effected via more than one pathway. Vitamin D has been shown to regulate the production of inflammatory signalling mediated by both NF‐kappaB and STAT‐1 [75] with MAPkinase activation as an important precursor [72]. There has therefore been increasing speculation that vitamin D deficiency could contribute to a risk of more serious COVID‐19 disease with increased risk of cytokine storm and consequent acute respiratory distress syndrome (ARDS) [76, 77].
Vitamin D immune response and gender
The impact of vitamin D on suppressing the immune response has been shown to differ between men and women. Vitamin D induces reduction in pro‐inflammatory cytokines IL17 and interferon gamma and increase of interleukin 10 production by CD4 + T lymphocytes, effects that are much greater in T lymphocytes from women than from men. Similarly, anti‐CD3‐ and anti‐CD28‐stimulated peripheral blood mononuclear cells from men generated less than half the number of regulatory CD4 + CD25 + FoxP3 + T lymphocytes in response to vitamin D compared with cells from women, but this gender difference disappeared when oestradiol was added [78]. In keeping with this, a Norwegian study has looked at the effect of weekly supplementation with 20 000 IU vitamin D3 or placebo on the human transcriptome in prediabetic individuals with impaired glucose tolerance. Fifty‐eight genes were shown to be significantly affected by vitamin D in men compared with 185 in women (P < 0.05). In women, 51 genes showed a 2‐log difference in expression compared with only a single gene in men [79]. Genes affected included those related to the interleukin signalling pathway and B cell‐mediated immunity. The authors speculated that the gender difference might be related to oestrogen‐dependent effects on synthesis of the vitamin D binding protein.
Increased prevalence of vitamin D deficiency amongst people with risk factors for severe COVID‐19 including ethnicity, diabetes, hypertension, obesity and institutionalization
Vitamin D deficiency is commoner in obese individuals, people with type 2 diabetes, hypertension, and most strikingly amongst ethnic minorities in Europe and North America – where darker skin pigmentation reduces skin synthesis and eightfold increased prevalence of deficiency is reported (Table 2) [80, 81, 82, 83]. All of these are demographics that have been associated with increased risk of severe COVID‐19. Vitamin D deficiency is also substantially commoner amongst people who are institutionalized including prisoners and people in care homes [84, 85, 86, 87]. Since vitamin D is fat‐soluble, its deficiency is also more likely in people with chronic digestive disorders such as Crohn’s disease or chronic pancreatitis but, hopefully, most will be receiving supplements.
Table 2.
Associations between vitamin D status and demographic variables associated with COVID‐19 mortality
| Author/year | Demographic variable | Type of study/location | n | Findings | Conclusions |
|---|---|---|---|---|---|
| Kunutsor et al 2013 80 | Hypertension | Meta‐analysis | 283 537 | Relative risk for hypertension reduced by 0.88 (95% CI 0.81–0.97) per 10 ng mL−1 increment in vitamin D levels | Inverse correlation between vitamin D status and hypertension |
| Mauss et al 2015 81 | Diabetes | Cross‐sectional (Germany) | 1821 |
Vit D < 10 ng mL−1 associated with increasing HbA1c P ≤ 0.001 And type 2 diabetes OR 2.55 (95% CI 1.16–5.12) |
Strong inverse correlation between vitamin D status, fasting glucose, HbA1c and type 2 diabetes |
| Yao et al 2015 82 | Obesity | Meta‐analysis | 13 209 | Vitamin D deficiency (varying definitions) OR 3.43 (95% CI 2.33 to −5.06) for obesity | Strong inverse correlation between vitamin D status and obesity |
| Herrick et al 2019 83 | Ethnicity | Cohort study (USA) | 16 180 |
Prevalence of vitamin D deficiency (<30 nmol L−1) 17.5% (95% CI 15.2–20.0) in non‐Hispanic black; 2.1% (95% CI 1.5–2.7) in non‐Hispanic white |
Strong association between ethnicity and vitamin D deficiency. No gender difference |
Vitamin D, the renin–angiotensin system and COVID‐19
The receptor for SARS‐CoV‐2, as for SARS‐CoV, is angiotensin‐converting enzyme 2 (ACE2) so there is intense interest in factors that alter its expression or function. ACE2 has potentially contradictory roles. Given that it is the receptor for SARS‐CoV‐2, it would be reasonable to assume that greater expression of ACE2 would be bad for the human host. However, since the discovery of ACE2 twenty years ago various studies have shown that it has a crucial role in protecting against acute lung injury and ARDS in experimental models [88, 89, 90]. The balance between ACE2 and ACE seems crucial as ACE2 counteracts the effects of ACE by hydrolysing angiotensin II to angiotensin (1‐7). Since angiotensin II is central to the development of ARDS, this is a very important protective mechanism. So, more ACE2 is good – at least in respect of reducing risk of ARDS, and ACE2 also has a protective role against cardiovascular diseases [91, 92]. ACE2 is highly expressed on human lung alveolar cells but also on vascular endothelial cells, smooth muscle cells, renal tubular epithelium and small intestinal enterocytes. There is frustratingly little published information on its expression, or perhaps more importantly on the ratio of ACE2:ACE expression, in children, males, elderly, varying ethnicity etc other than very small human studies [93] or animal studies. The gene encoding ACE2 is carried on the X chromosome. Serum assays have shown no sex differences in ACE2 concentration overall but higher serum ACE2 in older women [94]. Studies in rats have however shown substantially reduced ACE2 expression with ageing and particularly in older males [95].
Vitamin D has been shown experimentally to increase ACE2, reduce ACE expression, reduce angiotensin II production and reduce damage in lipopolysaccharide (lps)‐induced lung injury in rats [96]. Similarly, vitamin D receptor gene knockout mice show much more severe acute lung injury and increased mortality in an lps‐sepsis model of ARDS with amelioration by antagonists of angiotensin II [73]. Vitamin D also suppresses expression of renin, the rate‐limiting enzyme in the renin‐angiotensin cascade [97]. These effects are clearly highly relevant to a potential role of vitamin D in protecting against ARDS in COVID‐19.
Vitamin D deficiency, lupus anticoagulant‐associated thrombosis and COVID‐19
Venous and arterial thrombo‐embolic events are common in severe COVID‐19, affecting 28% of cases admitted to intensive care, despite thromboprophylaxis, in an Italian case series of 388 patients [98]. There has been considerable interest in the effects of vitamin D on coagulation but large studies have failed to show an impact of vitamin D status or supplementation on the risks for cardiovascular disease or thromboembolism [99, 100].
A much stronger case can be made though for a protective effect of vitamin D against thrombotic complications of the anti‐phospholipid syndrome [101], and here there are intriguing parallels with the thrombotic tendency in COVID‐19 [102]. Studies have shown a prevalence of up to 70% for vitamin D deficiency amongst patients with anti‐phospholipid syndrome and meta‐analysis of 4 case–control studies including 325 cases and 507 controls showed an odds ratio of 3.06 (P < 0.001) for frequency of vitamin D deficiency in patients with anti‐phospholipid syndrome [103]. In keeping with an effect of vitamin D, marked seasonality has been shown for anti‐phospholipid antibody titres in healthy controls with lower levels in summer months [104].
A systematic investigation of 56 patients hospitalized for COVID‐19 found 25 (45%) positive for lupus anticoagulant on the basis of coagulation tests (dilute Russell’s viper venom time (DRVVT) and activated partial thromboplastin time (aPTT) although anticardiolipin or anti‐beta2‐glycoprotein antibodies were only detected in 10% [105]. Another study reported that 20% of 216 patients positive for SARS‐CoV‐2 were found to have a prolonged activated partial thromboplastin time (aPTT) and when most of these were further tested 91% were positive on lupus anticoagulant assay [106]. It is well recognized that lupus anticoagulant activity may develop transiently, typically for two to three months, in other viral infections [107].
Seasonal variation of vitamin D deficiency and implications for the COVID‐19 pandemic
In the absence of vitamin D supplementation, there is marked seasonal variation in vitamin D levels. In the UK, for example, sunlight does not contain sufficient UVB to allow skin vitamin D synthesis until April and in northern Europe blood levels in nonsupplemented individuals may not rise substantially until late May or June [6]. Similarly, because of its relatively long half‐life in fat stores of 2–3 months, levels in the Southern Hemisphere typically do not drop until June. Moreover, older people and people with dark skin have much lower dermal synthesis of vitamin D in response to UVB [108]. The seasonality of respiratory virus infections is of course very well documented for influenza, human coronavirus and respiratory syncytial virus (RSV) – the ‘winter viruses’ [109], although other factors such as temperature and humidity are likely also to underlie this. There is sound evidence linking vitamin D deficiency with risk for or severity of influenza [76, 110] and RSV [51, 75]. If COVID‐19 severity is strongly related to vitamin D status, this too may prove to be a winter virus, since more severe COVID‐19 illness probably results in a longer period of infectivity. Current lock‐down measures could of course blunt the normal summer rise in vitamin D.
Vitamin D in the COVID‐19 pandemic – current knowledge
There are very limited peer‐reviewed studies currently published, and the current data are ‘soft’. The simplest but possibly the most informative is a questionnaire‐based study in Italian patients with Parkinson’s disease (n = 1486) and their family members (‘controls’ n = 1207) [111]. One hundred and five (7.1%) of patients and 92 (7.6%) family members had confirmed or probable COVID‐19. Vitamin D supplements had been taken by 13/105 (12.4%) COVID‐19 cases compared with 316/1381 (22.9%) unaffected – after age adjustment OR 0.56 (95% CI 0.32–0.99; P = 0.048) for vitamin D supplements reducing odds of COVID‐19 infection.
Another study from Italy has reported serum vitamin D levels taken with 7 weeks of SARS‐CoV‐2 PCR testing – mostly with 3 days of test [112]. Amongst 107 patients with available data, the 27 SARS‐CoV‐2 positives had median 25(OH)D 11.1 ng mL−1 (IQR 8.2–21.0) compared with the 80 SARS‐CoV‐2 negatives who had median 25(OH)D 24.6 ng mL−1 (IQR 8.9–30.5; P = 0.004). Because of the proximity of vitamin D assay to PCR testing, it is possible that vitamin D levels could have been lowered as a consequence of a negative acute phase response.
It should be noted that both of these studies are looking at the possible impact of vitamin D on risk for infection. No peer‐reviewed studies have yet been published looking at possible impacts of vitamin D on COVID‐19 severity.
Several preprints that have not yet undergone peer review are available online, but some of these are problematic. One of the more complete studies reports a retrospective cohort from Chicago of 4314 patients tested for COVID‐19 all of whom had a vitamin D level in the year before testing [113]. In multivariate analysis that adjusted for age and ethnicity, being likely vitamin D deficient (previous deficient level and lack of increased treatment) increased risk of testing positive for COVID‐19 (RR 1.77, P < 0.02). It should again be noted that this addresses whether vitamin D impacts on risk for infection but does not inform about risk of COVID‐19 severity.
Three studies, two currently online as preprints and one peer‐reviewed, have used historical vitamin D levels, measured between 2006 and 2010 in individuals sampled for the UK Biobank. These studies have shown no association between historical vitamin D levels (season‐adjusted) and testing positive for COVID‐19 but have not yet assessed COVID‐19 severity [114, 115, 116].
Studies are urgently needed that examine COVID‐19 outcomes in relation to vitamin D status or supplementation. An additional problem that needs addressing is possible confounding by a ‘healthy user’ effect, that is people with higher vitamin D levels possibly leading a healthier lifestyle in other ways. Care should be taken to adjust for relevant confounders such as deprivation, smoking and exercise.
Implications for current guidance
Many countries have recommendations for use of vitamin D supplements. Current UK guidance at https://www.nhs.uk/news/food‐and‐diet/the‐new‐guidelines‐on‐vitamin‐d‐what‐you‐need‐to‐know/ is that ‘adults and children over the age of one should have 10 µg (400 International units [IU]) of vitamin D every day. This means that some people may want to consider taking a supplement’. Recommendations from other countries vary, and the World Health Organization recommends a daily intake of 5 µg (200 IU) for adults but rising to 15 µg (600 IU) over 65 years [6]. The proportion of the population taking supplements is largely unknown although in one Irish study only 4% of men and 15% of women over 50 regularly took supplements [87].
The relevant advisory bodies: European Food Standards Agency, UK Scientific Advisory Committee on Nutrition and US Institute of Medicine, all consider an upper intake limit of 4000 IU day−1 in adults [7]. The consensus recommendations for vitamin D intakes for older adults or those with little sun exposure (household or confined) are a daily intake of 10–20 µg (400–800 IU day−1). Due to an inadequate dietary intake and lack of mandatory fortification in Europe and the United Kingdom, a vitamin D supplement may be required to achieve this. However, lifestyle and demographic factors also need to be taken into account and the ‘one size fits all’ will not achieve sufficient blood 25(OH)D concentrations across all people in a similar timeframe. For instance, the response to vitamin D supplementation is blunted in those who are overweight or obese [117, 118, 119, 120]. Furthermore, men possibly have a lower response to supplementation than women [117] and people with intestinal inflammatory conditions such as Crohn’s disease have poorer absorption. Baseline vitamin D status is also critical, and older adults may have a lower response [121]. Consequently, those with very low vitamin D concentrations, obesity, chronic intestinal disease or other conditions that affect vitamin D metabolism require either a much longer run‐in time period of supplementation or a higher dose to reach a sufficient level in the same timeframe.
Identification of appropriate supplementation dose depends on the target serum 25(OH)D concentration. Although >25 or 30 nmol L−1 is generally accepted as adequate for musculoskeletal health, it is recommended by the US Institute of Medicine that a higher level, >50 nmol L−1, should be achieved [118]. Some authorities would recommend a still higher target level of >75 nmol L−1, but this is controversial. Extrapolation from a large number of cohort studies suggests that for adults, supplementation with 1000 IU day−1 should be adequate, even in obese individuals, for achieving >50 nmol L−1. However, to achieve >75 nmol L−1 would typically require supplementation of between 3000 IU day−1 and 4000 IU day−1 for an obese individual [118].
Finally, consideration also needs to be given to other dietary components that are required for vitamin D metabolism/function. For instance, the metabolism of vitamin D into the active form is a magnesium‐dependent process, whilst it also acts a co‐factor for vitamin D binding protein [122]. Dietary intakes of magnesium have been highlighted as low in both the US and UK populations [123, 124], and the recommended intakes are around 420 mg day−1 for men and 320 mg/day−1 for women [125].
Implications for research
Research should include urgent observational studies comparing blood levels of vitamin D in the population with subsequent outcomes in COVID‐19 illness, but the caveat that vitamin D levels may fall during the acute phase response of a pyrexial illness may make interpretation difficult. Assay of free vitamin D may largely get around this problem. Simple observational studies of associations between prior vitamin D supplementation and COVID‐19 outcomes could be the fastest route to useful evidence.
Randomized controlled trials are regarded as the ‘gold standard’ for evidence but are probably harder to conduct when the intervention under test is an established vitamin rather than the more usual trial of a novel and potentially riskier drug. There are currently eleven clinical trials of vitamin D in COVID‐19 registered on clintrials.gov (Table 3). Nine of these are in symptomatic patients. Supplementing vitamin D in people who are already ill might be too late to be effective, although certainly worthy of study. Two studies will address prophylaxis, one in people >60 who are institutionalized (Lille) and the other in healthcare workers and relatives of affected patients (Tehran). Results from some of these studies will hopefully become available over the next few months.
Table 3.
Trials of vitamin D in COVID‐19 registered on clintrials.gov (5 June 2020)
| Clinical trial number | Title | Location | Subjects | Intervention | Proposed sample size | Primary outcome measure | Estimated primary completion |
|---|---|---|---|---|---|---|---|
| NCT04411446 | Cholecalciferol to improve the outcomes of COVID‐19 patients (CARED) | Argentina | Nonsevere, symptomatic and hospitalized | Single oral dose of 500 000 IU oral vitD3 vs placebo | 1265 | Need for respiratory support and change in respiratory SOFA a score | Dec 2020 |
| NCT04407286 | Vitamin D testing and treatment for COVID‐19 | Arizona USA | Nonsevere symptomatic patients with low levels of vitamin D | Open‐label cholecalciferol 10 000 IU day−1 bd (age 18–69 years) or 15 000 IU day−1 tds (age 70+) for 2 weeks. Continue after 2 weeks at this dose if deficient. If insufficient after 2 weeks, 5000 IU day−1 | 100 | Normalization of vitamin D levels and change in severity of COVID‐19 symptoms from baseline to 2 weeks | Aug 2020 |
| NCT04395768 | International ALLIANCE study of therapies to prevent progression of COVID‐19 | Australia | Symptomatic COVID‐19 | Multiple treatments including hydroxychloroquine, azithromycin, zinc, vitamin D3 5000 IU daily for 14 days, vitamin B12 with or without vitamin C | 200 | Change in severity and duration of symptoms, length of hospital stay and need for mechanical ventilation or mortality within 15 days | May 2021 |
| NCT04386850 b | Oral 25‐hydroxyvitamin D3 and COVID‐19 | Tehran, Iran | Symptomatic COVID‐19 | Oral 25‐hydroxy vitamin D3 25 mcg od for 2 months | 1500 | Hospitalization, disease duration, death and need for oxygen support | Nov 2020 |
| NCT04386850 b | Oral 25‐hydroxyvitamin D3 and COVID‐19 | Tehran, Iran | Healthcare provider or a close patient relative with a negative COVID‐19 test living with COVID‐19‐positive patients | Oral 25‐hydroxy vitamin D3 25 mcg od for 2 months | 1500 | Diagnosis of COVID‐19 infection of any severity, hospitalization, disease duration, death and need for oxygen support | Nov 2020 |
| NCT04385940 | Vitamin D and COVID‐19 management | Alberta USA | Nonsevere. symptomatic patients | Daily low dose (1000 IU day−1) compared to weekly high dose (ergocalciferol 50 000 IU twice during first week and one dose over second and third weeks) | 64 | Symptom recovery (time from intervention to day 21) | Aug 2020 |
| NCT04366908 | Prevention and treatment with calcifediol of COVID‐19 coronavirus‐induced acute respiratory syndrome (SARS) | Cordoba, Spain |
18–90 COVID‐19 pcr diagnosis |
Best available therapy (BAT) plus calcifediol 266 µg ×2 on day 1, then ×1 on days 3,7,14,21,28 vs BAT | 1008 | Admission to ITU or death by day 28 | July 2020 |
| NCT04363840 | The LEAD COVID‐19 trial: Low risk, early aspirin and vitamin D to reduce COVID‐19 hospitalizations | New Orleans USA | COVID‐19 diagnosis < 24 h |
50 000 IU VitD3 oral Once weekly ×2 plus aspirin 81 mg od (both arms) |
1080 | Hospitalization within 2 weeks | Dec 2020 |
| NCT04351490 | Impact of zinc and vitamin D3 supplementation on the survival of institutionalized patients infected with COVID‐19 | Lille, France | >60 institutionalized |
Zinc gluconate 15 mgs ×2/day VitD 2000 IU day−1 for 2 months vs usual care |
3140 | Survival 2 months | July 2020 |
| NCT04344041 | COVID‐19 and vitamin D supplementation: a multicentre randomized trial of high‐dose versus standard‐dose vitamin D3 in high‐risk COVID‐19 patients (CoVitTrial) | Angers, France | High risk ≥ 70 year diagnosed within 48 h | Vit D 400 000 IU single dose versus Vit D 50 000 IU single dose | 260 | All‐cause mortality 14 day | July 2020 |
| NCT04334005 | Vitamin D on prevention and treatment of COVID‐19 | Granada Spain | Nonsevere symptomatic |
Single dose 25 000 IU oral vitD3 vs usual care |
200 | All‐cause mortality | June 2020 |
SOFA‐sequential organ failure assessment score.
NCT04386850 has two cohorts: a treatment study for COVID‐19‐positive patients and a prevention study for healthcare providers (HCP) or close patient relatives living with COVID‐19‐positive patients.
Conclusions
Urgent research is needed to assess whether vitamin D deficiency is associated with increased COVID‐19 severity and to determine the effects of vitamin D supplementation. Meanwhile, given the strong circumstantial and biological evidence, and the relative safety of vitamin D supplementation, it seems sensible to advocate its use more widely during this pandemic, particularly for all those people at risk of vitamin D deficiency. The potential gain if the hypothesis is correct would be massive.
These points are summarized in Table 4.
Table 4.
Summary
|
Vitamin D deficiency as a possible factor determining COVID‐19 severity
|
Conflict of interest statement
JMR with the University of Liverpool and Provexis UK holds a patent for use of a soluble fibre preparation as maintenance therapy for Crohn’s disease plus a patent for its use in antibiotic‐associated diarrhoea. Patent also held with the University of Liverpool and others in relation to use of modified heparins in cancer therapy. SS has received speaker fees from MSD, Actavis, AbbVie, Dr Falk pharmaceuticals, Shire and received educational grants from MSD, AbbVie, Actavis and is an advisory board member for AbbVie, Dr Falk pharmaceutics and Vifor pharmaceuticals. EL, GG and RAK have no conflicts to declare.
Funding
None.
Authors and contributions
All authors contributed to conception of the article. JMR and SS wrote the first draft, and all authors contributed to revision and approved the final version.
Author Contribution
Jonathan Rhodes: Conceptualization (equal); Formal analysis (lead); Writing‐original draft (lead); Writing‐review & editing (equal). Sreedhar Subramanian: Conceptualization (supporting); Writing‐original draft (supporting); Writing‐review & editing (equal). Eamon Laird: Conceptualization (supporting); Writing‐original draft (supporting); Writing‐review & editing (equal). George Griffin: Conceptualization (equal); Writing‐original draft (supporting); Writing‐review & editing (equal). Rose Anne Kenny: Conceptualization (equal); Writing‐original draft (supporting); Writing‐review & editing (equal).
Acknowledgements
We are very grateful to Prof Frank Dunstan, Emeritus Professor of Statistics, Cardiff University, for his very helpful comments.
Rhodes JM, Subramanian S, Laird E, Griffin G, Kenny RA (Institute of Translational Medicine, University of Liverpool, Liverpool, UK; Trinity College Dublin, Dublin, Ireland; St George’s, University of London, London, UK; Mercers Institute for Ageing, St James Hospital, Dublin 8, Ireland). Perspective: Vitamin D deficiency and COVID-19 severity – plausibly linked by latitude, ethnicity, impacts on cytokines, ACE2 and thrombosis. J Intern Med 2021; 289: 97–115. 10.1111/joim.13149
[Correction added on 21 September 2020 after original online publication: The copyright line for this article was changed]
[Correction added on 27 July 2020, after first online publication: “Graphical abstract” has been corrected in this current version.]
References
- 1. Pascarella G, Strumia A, Piliego C et al. COVID‐19 diagnosis and management: a comprehensive review. J Intern Med 2020; 288: 192–206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Docherty AB, Harrison EM, Green CA. et al. Features of 16,749 hospitalised UK patients with COVID‐19 using the ISARIC WHO Clinical Characterisation Protocol. medRxiv 2020.04.23.20076042; doi: 10.1101/2020.04.23.20076042 [DOI] [PMC free article] [PubMed]
- 3. Rhodes J, Dunstan F, Laird E, Subramanian S, Kenny RA. COVID‐19 mortality increases with northerly latitude after adjustment for age suggesting a link with ultraviolet and vitamin D. BMJ Nutrit., Prevent. Health 2020. 10.1136/bmjnph-2020-000110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Kohlmeier M. Avoidance of vitamin D deficiency to slow the COVID‐19 pandemic. BMJ Nutrit., Prevent. Health 2020. 10.1136/bmjnph-2020-000096 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Hansen L, Tjønneland A, Køster B et al. Vitamin D Status and seasonal variation among Danish children and adults: A descriptive study. Nutrients 2018; 10: 1801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Roth DE, Abrams SA, Aloia J et al. Global prevalence and disease burden of vitamin D deficiency: a roadmap for action in low‐ and middle‐income countries. Ann N Y Acad Sci 2018; 1430: 44–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Lips P, Cashman KD, Lamberg‐Allardt C et al. Current vitamin D status in European and Middle East countries and strategies to prevent vitamin D deficiency: a position statement of the European Calcified Tissue Society. Eur J Endocrinol 2019; 180: 23–54. [DOI] [PubMed] [Google Scholar]
- 8. Manios Y, Moschonis G, Lambrinou CP et al. A systematic review of vitamin D status in southern European countries. Eur J Nutr 2018; 57: 2001–2036. [DOI] [PubMed] [Google Scholar]
- 9. Laird E, Rhodes J, Kenny RA. Vitamin D and inflammation: potential implications for severity of COVID‐19. Irish Med J 2020; 113: P81. [PubMed] [Google Scholar]
- 10. https://earthobservatory.nasa.gov/images/146362/airborne‐nitrogen‐dioxide‐plummets‐over‐china Accessed April 27th 2020.
- 11. Pereira‐Santos M, Santos JYGD, Carvalho GQ, Santos DBD, Oliveira AM. Epidemiology of vitamin D insufficiency and deficiency in a population in a sunny country: Geospatial meta‐analysis in Brazil. Crit Rev Food Sci Nutr 2019; 59: 2102–2109. [DOI] [PubMed] [Google Scholar]
- 12. Bernard JJ, Gallo RL, Krutmann J. Photoimmunology: how ultraviolet radiation affects the immune system. Nat Rev Immunol 2019; 19: 688–701. [DOI] [PubMed] [Google Scholar]
- 13. Lytle CD, Sagripanti JL. Predicted inactivation of viruses of relevance to biodefense by solar radiation. J Virol 2005; 79: 14244–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Wu Y, Jing W, Liu J et al. Effects of temperature and humidity on the daily new cases and new deaths of COVID‐19 in 166 countries. Sci Total Environ 2020; 729: 139051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Sehra ST, Salciccioli JD, Wiebe DJ, Fundin S, Baker JF. Maximum daily temperature, precipitation, ultra‐violet light and rates of transmission of SARS‐Cov‐2 in the United States [published online ahead of print, 2020 May 30]. Clin Infect Dis 2020; ciaa681. 10.1093/cid/ciaa681 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Yao Y, Pan J, Liu Z et al. No association of COVID‐19 transmission with temperature or UV radiation in Chinese cities. Eur Respir J 2020; 55: 2000517. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Muscogiuri G, Altieri B, Penna‐Martinez M, Badenhoop K. Focus on vitamin D and the adrenal gland. Horm Metab Res 2015; 47: 239–46. [DOI] [PubMed] [Google Scholar]
- 18. Armas LA, Hollis BW, Heaney RP. Vitamin D2 is much less effective than vitamin D3 in humans. J Clin Endocrinol Metab 2004; 89: 5387–91. [DOI] [PubMed] [Google Scholar]
- 19. Tripkovic L, Lambert H, Hart K et al. Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25‐hydroxyvitamin D status: a systematic review and meta‐analysis. Am J Clin Nutr 2012; 95: 1357–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Chun RF, Shieh A, Gottlieb C et al. Vitamin D Binding Protein and the Biological Activity of Vitamin D. Front Endocrinol (Lausanne) 2019; 10: 718. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Jeffery LE, Wood AM, Qureshi OS et al. Availability of 25‐hydroxyvitamin D(3) to APCs controls the balance between regulatory and inflammatory T cell responses. J Immunol 2012; 189: 5155–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Hypponen E, Power C. Hypovitaminosis D in British adults at age 45y: nationwide cohort study of dietary and lifestyle predictors. Am J Clin Nutr 2007; 85: 860–8. [DOI] [PubMed] [Google Scholar]
- 23. Martinaityte I, Kamycheva E, Didriksen A, Jakobsen J, Jorde R. Vitamin D stored in fat tissue during a 5‐year intervention affects serum 25‐hydroxyvitamin D levels the following year. J Clin Endocrinol Metab 2017; 102: 3731–8. [DOI] [PubMed] [Google Scholar]
- 24. Laird E, Ward M, McSorley E, Strain JJ, Wallace J. Vitamin D and bone health: potential mechanisms. Nutrients 2010; 2: 693–724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Pike JW, Meyer MB, Lee SM, Onal M, Benkusky NA. The vitamin D receptor: contemporary genomic approaches reveal new basic and translational insights. J Clin Invest 2017; 127: 1146–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Bikle DD, Schwartz J. Vitamin D binding protein, total and free vitamin D levels in different physiological and pathophysiological conditions. Front Endocrinol (Lausanne) 2019; 10: 317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Carlberg C, Muñoz A. An update on vitamin D signaling and cancer [published online ahead of print, 2020 May 30]. Semin Cancer Biol 2020; S1044–579X(20)30114. 10.1016/j.semcancer.2020.05.018 [DOI] [PubMed] [Google Scholar]
- 28. Hossein‐nezhad A, Spira A, Holick MF. Influence of vitamin D status and vitamin D3 supplementation on genome wide expression of white blood cells: a randomized double‐blind clinical trial. PLoS One 2013; 8: e58725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Carlberg C, Seuter S, de Mello VDF et al. Primary vitamin D target genes allow a categorization of possible benefits of vitamin D3 supplementation. PLoS One 2013; 8: e71042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Nurminen V, Seuter S, Carlberg C. Primary vitamin D target genes of human monocytes. Front Physiol 2019; 10: 194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Ramagopalan SV, Heger A, Berlanga AJ et al. A ChIP‐seq defined genome‐wide map of vitamin D receptor binding: associations with disease and evolution. Genome Res 2010; 20: 1352–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Green M. Cod liver oil and tuberculosis. BMJ 2011; 343: d7505. [DOI] [PubMed] [Google Scholar]
- 33. Huang SJ, Wang XH, Liu ZD et al. Vitamin D deficiency and the risk of tuberculosis: a meta‐analysis. Drug Des Devel Ther 2016; 11: 91–102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Martineau AR, Jolliffe DA, Hooper RL et al. Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta‐analysis of individual participant data. Brit Med J 2017; 356: i6583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Vo P, Koppel C, Espinola JA et al. Vitamin D Status at the time of hospitalization for bronchiolitis and its association with disease severity. J Pediatr 2018; 203: 416–422. [DOI] [PubMed] [Google Scholar]
- 36. Laird E, McNulty H, Ward M et al. Vitamin D deficiency is associated with inflammation in older Irish adults. J Clin Endocrinol Metab 2014; 99: 1807–15. [DOI] [PubMed] [Google Scholar]
- 37. Ter Horst R, Jaeger M, Smeekens SP et al. Host and environmental factors influencing individual human cytokine responses. Cell 2016; 167: 1111–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Nonnecke BJ, McGill JL, Ridpath JF, Sacco RE, Lippolis JD, Reinhardt TA. Acute phase response elicited by experimental bovine diarrhea virus (BVDV) infection is associated with decreased vitamin D and E status of vitamin‐replete preruminant calves. J Dairy Sci 2014; 97: 5566–79. [DOI] [PubMed] [Google Scholar]
- 39. Silva MC, Furlanetto TW. Does serum 25‐hydroxyvitamin D decrease during acute‐phase response? A systematic review. Nutr Res 2015; 35: 91–96. [DOI] [PubMed] [Google Scholar]
- 40. Bikle DD, Malmstroem S, Schwartz J. Current controversies: are free vitamin metabolite levels a more accurate assessment of vitamin D status than total levels? Endocrinol Metab Clin North Am 2017; 46: 901–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Chun RF, Lauridsen AL, Suon L et al. Vitamin D‐binding protein directs monocyte responses to 25‐hydroxy‐ and 1,25‐dihydroxyvitamin D. J Clin Endocrinol Metab 2010; 95: 3368–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Bikle DD, Siiteri PK, Ryzen E, Haddad JG. Serum protein binding of 1,25‐dihydroxyvitamin D: a reevaluation by direct measurement of free metabolite levels. J Clin Endocrinol Metab 1985; 61: 969–75. [DOI] [PubMed] [Google Scholar]
- 43. Diasource Free 25OH Vitamin D ELISA Available at: https://www.diasource‐diagnostics.com/RUO‐Products/ImmunoAssays/Bone‐Metabolism/Vitamin‐D/Free‐25OH‐Vitamin‐D‐ELISA‐96‐tests
- 44. Berg AH, Bhan I, Powe C, Karumanchi SA, Xu D, Thadhani RI. Acute homeostatic changes following vitamin D2 supplementation. J Endocr Soc 2017; 1: 1135–49. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Chun RF, Peercy BE, Adams JS, Hewison M. Vitamin D binding protein and monocyte response to 25‐hydroxyvitamin D and 1,25‐dihydroxyvitamin D: analysis by mathematical modeling. PLoS One 2012; 7: e30773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Arnaud J, Constans J. Affinity differences for vitamin D metabolites associated with the genetic isoforms of the human serum carrier protein (DBP). Hum Genet 1993; 92: 183–8. [DOI] [PubMed] [Google Scholar]
- 47. Kawakami M, Imawari M, Goodman DS. Quantitative studies of the interaction of cholecalciferol (vitamin D3) and its metabolites with different genetic variants of the serum binding protein for these sterols. Biochem J 1979; 179: 413–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Boutin B, Galbraith RM, Arnaud P. Comparative affinity of the major genetic variants of human group‐specific component (vitamin D‐binding protein) for 25‐(OH) vitamin D. J Steroid Biochem 1989; 32: 59–63. [DOI] [PubMed] [Google Scholar]
- 49. Schwartz JB, Gallagher JC, Jorde R et al. Determination of Free 25(OH)D concentrations and their relationships to total 25(OH)D in multiple clinical populations. J Clin Endocrinol Metab 2018; 103: 3278–88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Lauridsen AL, Vestergaard P, Nexo E. Mean serum concentration of vitamin D‐binding protein (Gc globulin) is related to the Gc phenotype in women. Clin Chem 2001; 47: 753–6. [PubMed] [Google Scholar]
- 51. McNally JD, Sampson M, Matheson LA, Hutton B, Little J. Vitamin D receptor (VDR) polymorphisms and severe RSV bronchiolitis: a systematic review and meta‐analysis. Pediatr Pulmonol 2014; 49: 790–9. [DOI] [PubMed] [Google Scholar]
- 52. Bonilla C, Gilbert R, Kemp JP et al. Using genetic proxies for lifecourse sun exposure to assess the causal relationship of sun exposure with circulating vitamin d and prostate cancer risk. Cancer Epidemiol Biomarkers Prev 2013; 22: 597–606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Donnelly MP, Paschou P, Grigorenko E et al. A global view of the OCA2‐HERC2 region and pigmentation. Hum Genet 2012; 131: 683–696. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Kimlin M, Harrison S, Nowak M, Moore M, Brodie A, Lang C. Does a high UV environment ensure adequate vitamin D status? J Photochem Photobiol B 2007; 89: 139–147. [DOI] [PubMed] [Google Scholar]
- 55. Jiang X, O'Reilly PF, Aschard H et al. Genome‐wide association study in 79,366 European‐ancestry individuals informs the genetic architecture of 25‐hydroxyvitamin D levels. Nat Commun 2018; 9: 260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56. Zgaga L, Laird E, Healy M. 25‐Hydroxyvitamin D measurement in human hair: results from a proof‐of‐concept study. Nutrients 2019; 11: 423. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Iob E, Steptoe A. Cardiovascular disease and hair cortisol: a novel biomarker of chronic stress. Curr Cardiol Rep 2019; 21: 116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Feeney JC, O'Halloran AM, Kenny RA. The association between hair cortisol, hair cortisone, and cognitive function in a population‐based cohort of older adults: results from the Irish longitudinal study on ageing. J Gerontol A Biol Sci Med Sci 2020; 75: 257–265. [DOI] [PubMed] [Google Scholar]
- 59. Greiller CL, Martineau AR. Modulation of the immune response to respiratory viruses by vitamin D. Nutrients 2015; 7: 4240–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. McMurray DN, Bartow RA, Mintzer CL, Hernandez‐Frontera E. Micronutrient status and immune function in tuberculosis. Ann N Y Acad Sci 1990; 587: 59–69. [DOI] [PubMed] [Google Scholar]
- 61. Bloom BR, Modlin RL. Mechanisms of defense against intracellular pathogens mediated by human macrophages. Microbiol Spectr 2016; 4. 10.1128/microbiolspec.MCHD-0006-2015 [DOI] [PubMed] [Google Scholar]
- 62. Flanagan PK, Chiewchengchol D, Wright HL et al. Killing of Escherichia coli by Crohn's disease monocyte‐derived macrophages and its enhancement by hydroxychloroquine and vitamin D. Inflamm Bowel Dis 2015; 21: 1499–510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Hewison M. Antibacterial effects of vitamin D. [published correction appears in Nat Rev Endocrinol. 2011;7:436] Nat Rev Endocrinol 2011; 7: 337–45. [DOI] [PubMed] [Google Scholar]
- 64. Ahmed A, Siman‐Tov G, Hall G, Bhalla N, Narayanan A. Human antimicrobial peptides as therapeutics for viral infections. Viruses 2019; 11: 704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Telcian AG, Zdrenghea MT, Edwards MR et al. Vitamin D increases the antiviral activity of bronchial epithelial cells in vitro. Antiviral Res 2017; 137: 93–101. [DOI] [PubMed] [Google Scholar]
- 66. Schögler A, Muster RJ, Kieninger E et al. Vitamin D represses rhinovirus replication in cystic fibrosis cells by inducing LL‐37. Eur Respir J 2016; 47: 520–530. [DOI] [PubMed] [Google Scholar]
- 67. Barlow PG, Svoboda P, Mackellar A et al. Antiviral activity and increased host defense against influenza infection elicited by the human cathelicidin LL‐37. PLoS One 2011; 6: e25333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Zdrenghea MT, Makrinioti H, Bagacean C, Bush A, Johnston SL, Stanciu LA. Vitamin D modulation of innate immune responses to respiratory viral infections. Rev Med Virol 2017; 27: 10.1002/rmv.1909. [DOI] [PubMed] [Google Scholar]
- 69. Arboleda JF, Fernandez GJ, Urcuqui‐Inchima S. Vitamin D‐mediated attenuation of miR‐155 in human macrophages infected with dengue virus: Implications for the cytokine response. Infect Genet Evol 2019; 69: 12–21. [DOI] [PubMed] [Google Scholar]
- 70. Puerta‐Guardo H, Medina F, De la Cruz Hernández SI, Rosales VH, Ludert JE, del Angel RM. The 1α,25‐dihydroxy‐vitamin D3 reduces dengue virus infection in human myelomonocyte (U937) and hepatic (Huh‐7) cell lines and cytokine production in the infected monocytes. Antiviral Res 2012; 94: 57–61. Erratum. In: Antiviral Res 2012;94:297. Medina, Fernando [added]. [DOI] [PubMed] [Google Scholar]
- 71. Villamor E, Villar LA, Lozano A, Herrera VM, Herrán OF. Vitamin D serostatus and dengue fever progression to dengue hemorrhagic fever/dengue shock syndrome. Epidemiol Infect 2017; 145: 2961–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Zhang Y, Leung DY, Richers BN et al. Vitamin D inhibits monocyte/macrophage proinflammatory cytokine production by targeting MAPK phosphatase‐1. J Immunol 2012; 188: 2127–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Kong J, Zhu X, Shi Y et al. VDR attenuates acute lung injury by blocking Ang‐2‐Tie‐2 pathway and renin‐angiotensin system. Mol Endocrinol 2013; 27: 2116–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Tsujino I, Ushikoshi‐Nakayama R, Yamazaki T, Matsumoto N, Saito I. Pulmonary activation of vitamin D3 and preventive effect against interstitial pneumonia. J Clin Biochem Nutr 2019; 65: 245–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Stoppelenburg AJ, von Hegedus JH, Huis in't Veld R, Bont L, Boes M Defective control of vitamin D receptor‐mediated epithelial STAT1 signalling predisposes to severe respiratory syncytial virus bronchiolitis. J Pathol 2014; 232: 57–64. [DOI] [PubMed] [Google Scholar]
- 76. Grant WB, Lahore H, McDonnell SL et al. Evidence that vitamin D supplementation could reduce risk of Influenza and COVID‐19 infections and deaths. Nutrients 2020; 12: E988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Rhodes JM, Subramanian S, Laird E, Kenny RA. Editorial: low population mortality from COVID‐19 in countries south of latitude 35 degrees North supports vitamin D as a factor determining severity. Aliment Pharmacol Ther 2020; 51: 1434–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78. Correale J, Ysrraelit MC, Gaitán MI. Gender differences in 1,25 dihydroxyvitamin D3 immunomodulatory effects in multiple sclerosis patients and healthy subjects. J Immunol 2010; 185: 4948–58. [DOI] [PubMed] [Google Scholar]
- 79. Pasing Y, Fenton CG, Jorde R, Paulssen RH. Changes in the human transcriptome upon vitamin D supplementation. J Steroid Biochem Mol Biol 2017; 173: 93–9. [DOI] [PubMed] [Google Scholar]
- 80. Kunutsor SK, Apekey TA, Steur M. Vitamin D and risk of future hypertension: meta‐analysis of 283,537 participants. Eur J Epidemiol 2013; 28: 205–21. [DOI] [PubMed] [Google Scholar]
- 81. Mauss D, Jarczok MN, Hoffmann K, Thomas GN, Fischer JE. Association of vitamin D levels with type 2 diabetes in older working adults. Int J Med Sci 2015; 12: 362–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82. Yao Y, Zhu L, He L et al. A meta‐analysis of the relationship between vitamin D deficiency and obesity. Int J Clin Exp Med 2015; 8: 14977–84. [PMC free article] [PubMed] [Google Scholar]
- 83. Herrick KA, Storandt RJ, Afful J et al. Vitamin D status in the United States, 2011–2014. Am J Clin Nutr 2019; 110: 150–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84. Jacobs ET, Mullany CJ. Vitamin D deficiency and inadequacy in a correctional population. Nutrition 2015; 31: 659–63. [DOI] [PubMed] [Google Scholar]
- 85. Nwosu BU, Maranda L, Berry R et al. The vitamin D status of prison inmates. PLoS One 2014; 9: e90623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86. Arnljots R, Thorn J, Elm M, Moore M, Sundvall PD. Vitamin D deficiency was common among nursing home residents and associated with dementia: a cross sectional study of 545 Swedish nursing home residents. BMC Geriatr 2017; 17: 229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87. Laird E, Kenny RA.Vitamin D deficiency in Ireland: Implications for COVID‐19. Results from the Irish longitudinal study on ageing. April 4 2020. 10.38018/TildaRe.2020-05 [DOI]
- 88. Yan T, Xiao R, Lin G. Angiotensin‐converting enzyme 2 in severe acute respiratory syndrome coronavirus and SARS‐CoV‐2: A double‐edged sword? FASEB J 2020; 34: 6017–26. 10.1096/fj.202000782 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Imai Y, Kuba K, Rao S et al. Angiotensin‐converting enzyme 2 protects from severe acute lung failure. Nature 2005; 436: 112–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90. Kuba K, Imai Y, Rao S et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus‐induced lung injury. Nat Med 2005; 11: 875–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Kuba K, Imai Y, Penninger JM. Multiple functions of angiotensin‐converting enzyme 2 and its relevance in cardiovascular diseases. Circ J 2013; 77: 301–8. [DOI] [PubMed] [Google Scholar]
- 92. Zheng YY, Ma YT, Zhang JY, Xie X. COVID‐19 and the cardiovascular system. Nat Rev Cardiol 2020; 17: 259–260. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93. Zhao Y, Zhao Z, Wang Y et al. Single‐cell RNA expression profiling of ACE2, the receptor of SARS‐CoV‐2 bioRxiv doi: 10.1101/2020.01.26.919985 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Fernández‐Atucha A, Izagirre A, Fraile‐Bermúdez AB et al. Sex differences in the aging pattern of renin‐angiotensin system serum peptidases. Biol Sex Differ 2017; 8: 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Xie X, Chen J, Wang X, Zhang F, Liu Y. Age‐ and gender‐related difference of ACE2 expression in rat lung. [published correction appears in Life Sci. 2006;79:2499. Xudong, Xie [corrected to Xie, Xudong]; Junzhu, Chen [corrected to Chen, Junzhu]; Xingxiang, Wang [corrected to Wang, Xingxiang]; Furong, Zhang [corrected to Zhang, Furong]; Yanrong, Liu [corrected to Liu, Yanrong]] Life Sci 2006; 78: 2166–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Xu J, Yang J, Chen J, Luo Q, Zhang Q, Zhang H. Vitamin D alleviates lipopolysaccharide‐induced acute lung injury via regulation of the renin‐angiotensin system. Mol Med Rep 2017; 16: 7432–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Yuan W, Pan W, Kong J et al. 1,25‐dihydroxyvitamin D3 suppresses renin gene transcription by blocking the activity of the cyclic AMP response element in the renin gene promoter. J Biol Chem 2007; 282: 29821–30. [DOI] [PubMed] [Google Scholar]
- 98. Lodigiani C, Iapichino G, Carenzo L et al. Venous and arterial thromboembolic complications in COVID‐19 patients admitted to an academic hospital in Milan, Italy. Thrombosis Res 2020; 191: 9–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Scragg R, Stewart AW, Waayer D et al. Effect of Monthly High‐Dose Vitamin D Supplementation on cardiovascular disease in the Vitamin D assessment study: A randomized clinical trial. JAMA Cardiol 2017; 2: 608–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Vučković BA, van Rein N, Cannegieter SC, Rosendaal FR, Lijfering WM. Vitamin supplementation on the risk of venous thrombosis: results from the MEGA case‐control study. Am J Clin Nutr 2015; 101: 606–12. [DOI] [PubMed] [Google Scholar]
- 101. García‐Carrasco M, Jiménez‐Herrera EA, Gálvez‐Romero JL et al. The anti‐thrombotic effects of vitamin D and their possible relationship with antiphospholipid syndrome. Lupus 2018; 27: 2181–9. [DOI] [PubMed] [Google Scholar]
- 102. Svenungsson E, Antovic A. The antiphospholipid syndrome ‐ often overlooked cause of vascular occlusions? J Intern Med 2020; 287: 349–72. [DOI] [PubMed] [Google Scholar]
- 103. Riancho‐Zarrabeitia L, Cuberia M, Munoz P et al. Vitamin D and antiphospholipid syndrome: a retrospective cohort study and metaanalysis. Semin Arthritis Rheum 2018; 47: 877–82. [DOI] [PubMed] [Google Scholar]
- 104. Luong TH, Rand JH, Wu XX, Godbold JH, Gascon‐Lema M, Tuhrim S. Seasonal distribution of antiphospholipid antibodies. Stroke 2001; 32: 1707–11. [DOI] [PubMed] [Google Scholar]
- 105. Harzallah I, Debliquis A, Drénou B. Lupus anticoagulant is frequent in patients with Covid‐19 [published online ahead of print, 2020 Apr 23]. J Thromb Haemost 2020. 10.1111/jth.14867 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Bowles L, Platton S, Yartey N et al. Lupus anticoagulant and abnormal coagulation tests in patients with Covid‐19. N Engl J Med 2020. 10.1056/NEJMc2013656 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107. Uthman IW, Gharavi AE. Viral infections and antiphospholipid antibodies. Semin Arthritis Rheum 2002; 31: 256–63. [DOI] [PubMed] [Google Scholar]
- 108. Wacker M, Holick MF. Sunlight and Vitamin D: A global perspective for health. Dermatoendocrinol 2013; 5: 51–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Moriyama M, Hugentobler WJ, Iwasaki A. Seasonality of respiratory viral infections [published online ahead of print, 2020 Mar 20]. Annu Rev Virol 2020; 7: 1. 10.1146/annurev-virology-012420-022445 [DOI] [PubMed] [Google Scholar]
- 110. Urashima M, Segawa T, Okazaki M, Kurihara M, Wada Y, Ida H. Randomized trial of vitamin D supplementation to prevent seasonal influenza A in schoolchildren. Am J Clin Nutr 2010; 91: 1255–60. [DOI] [PubMed] [Google Scholar]
- 111. Fasano A, Cereda E, Barichella M et al. COVID‐19 in Parkinson's disease patients living in Lombardy, Italy. Mov Disord 2020. 10.1002/mds.28176 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112. D'Avolio A, Avataneo V, Manca A et al. 25‐Hydroxyvitamin D concentrations are lower in patients with positive PCR for SARS‐CoV‐2. Nutrients 2020; 12: E1359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113. Meltzer DO, Best TJ, Zhang H, Vokes T, Arora V, Solway J. Association of vitamin D deficiency and treatment with COVID‐19 incidence. Preprint. medRxiv. 2020. Published 2020 May 13. 10.1101/2020.05.08.20095893 [DOI] [Google Scholar]
- 114. Hastie CE, Mackay DF, Ho F et al. Vitamin D concentrations and COVID‐19 infection in UK Biobank. Diabetes Metab Syndr 2020; 14: 561–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115. Darling AL, Ahmadi KR, Kate A, Ward KA et al. Vitamin D status, body mass index, ethnicity and COVID‐19: Initial analysis of the first‐reported UK Biobank COVID‐19 positive cases (n 580) compared with negative controls (n 723) medRxiv 2020; 10.1101/2020.04.29.20084277 [DOI]
- 116. Raisi‐Estabragh Z, McCracken C, Bethell MS et al.Greater risk of severe COVID‐19 in non‐White ethnicities is not explained by cardiometabolic, socioeconomic, or behavioural factors, or by 25(OH)‐vitamin D status: study of 1,326 cases from the UK Biobank medRxiv 2020. 10.1101/2020.06.01.20118943 [DOI] [PMC free article] [PubMed]
- 117. Wu Z, Camargo CA Jr, Reid IR et al. What factors modify the effect of monthly bolus dose vitamin D supplementation on 25‐hydroxyvitamin D concentrations? J Steroid Biochem Mol Biol 2020; 201: 105687. [DOI] [PubMed] [Google Scholar]
- 118. Zittermann A, Ernst JB, Gummert JF, Börgermann J. Vitamin D supplementation, body weight and human serum 25‐hydroxyvitamin D response: a systematic review. Eur J Nutr 2014; 53: 367–374. [DOI] [PubMed] [Google Scholar]
- 119. Mazahery H, von Hurst PR. Factors affecting 25‐hydroxyvitamin D concentration in response to vitamin D supplementation. Nutrients 2015; 7: 5111–5142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. de Oliveira LF, de Azevedo LG, da Mota Santana J, de Sales LPC, Pereira‐Santos M. Obesity and overweight decreases the effect of vitamin D supplementation in adults: systematic review and meta‐analysis of randomized controlled trials. Rev Endocr Metab Disord 2020; 21: 67–76. [DOI] [PubMed] [Google Scholar]
- 121. Harris SS, Dawson‐Hughes B, Perrone GA. Plasma 25‐hydroxyvitamin D responses of younger and older men to three weeks of supplementation with 1800 IU/day of vitamin D. J Am Coll Nutr 1999; 18: 470–474. [DOI] [PubMed] [Google Scholar]
- 122. Rosanoff A, Dai Q, Shapses SA. Essential nutrient interactions: Does low or suboptimal magnesium status interact with vitamin D and/or calcium status? Adv Nutr 2016; 7: 25–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123. Ford ES, Mokdad AH. Dietary magnesium intake in a national sample of US adults. J Nutr 2003; 133: 2879–2882. [DOI] [PubMed] [Google Scholar]
- 124. Derbyshire E. Micronutrient intakes of British adults across mid‐life: a secondary analysis of the UK national diet and nutrition survey. Front Nutr 2018; 5: 55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125. Institute of Medicine (US) Standing Committee on the Scientific Evaluation of Dietary Reference Intakes . Dietary Reference Intakes for Calcium, Phosphorus, Magnesium, Vitamin D, and Fluoride. Washington (DC): National Academies Press (US); 1997. [PubMed] [Google Scholar]


