Summary
The novel coronavirus disease 2019 (COVID‐19) is rapidly expanding and causing many deaths all over the world with the World Health Organization (WHO) declaring a pandemic in March 2020. Current therapeutic options are limited and there is no registered and/or definite treatment or vaccine for this disease or the causative infection, severe acute respiratory coronavirus 2 syndrome (SARS‐CoV‐2). Angiotensin‐converting enzyme 2 (ACE2), a part of the renin‐angiotensin system (RAS), serves as the major entry point into cells for SARS‐CoV‐2 which attaches to human ACE2, thereby reducing the expression of ACE2 and causing lung injury and pneumonia. Vitamin D, a fat‐soluble‐vitamin, is a negative endocrine RAS modulator and inhibits renin expression and generation. It can induce ACE2/Ang‐(1‐7)/MasR axis activity and inhibits renin and the ACE/Ang II/AT1R axis, thereby increasing expression and concentration of ACE2, MasR and Ang‐(1‐7) and having a potential protective role against acute lung injury (ALI)/acute respiratory distress syndrome (ARDS). Therefore, targeting the unbalanced RAS and ACE2 down‐regulation with vitamin D in SARS‐CoV‐2 infection is a potential therapeutic approach to combat COVID‐19 and induced ARDS.
Keywords: angiotensin‐converting enzyme 2, COVID‐19, vitamin D
Abbreviations
- 1,25(OH)2D3
1,25‐dihydroxyvitamin D
- 25(OH)D
25‐hydroxyvitamin D
- ACE2
angiotensin‐converting enzyme 2
- AGTR1A
type 1a Ang II receptor
- ALI
acute lung injury
- Ang II
angiotensin II
- APC
antigen presentation cells
- ARDS
acute respiratory distress syndrome
- AT1
Ang II receptor type 1
- BKB1R
bradykinin B1 receptor
- COVID‐19
coronavirus disease 2019
- CVD
cardiovascular disease
- DABK
desArg9‐bradykinin
- IFN‐γ
interferon γ
- KKS
Kinin‐Kallikrein system
- LPS
lipopolysaccharide
- NF‐κB
nuclear factor‐κB
- NOS
nitric oxide synthase
- RAS
renin‐angiotensin system
- SARS‐CoV‐2
severe acute respiratory coronavirus 2 syndrome
- TGF‐β
transforming growth factor‐β
- TNF‐α
tumor necrosis factor α
- VDR
vitamin D receptor
- WHO
World Health Organization
1. INTRODUCTION
The novel coronavirus disease 2019 (COVID‐19) is a kind of pneumonia caused by a new member of the Coronoviridae family called severe acute respiratory coronavirus 2 syndrome (SARS‐CoV‐2) which started from Wuhan Province in China in December 2019. 1 The clinical features of this disease include fever, dry cough, dyspnea, myalgia and fatigue. 2 A small number of patients also experience expectoration, headaches, hemoptysis and diarrhea. 2 Common laboratory findings are leukopenia and lymphopenia. 2 The COVID‐19 can be complicated by interstitial pneumonia together with alveolar destruction resulting in severe acute respiratory distress syndrome (ARDS) and ultimately death. 3 Because the COVID‐19 epidemic is rapidly expanding and causes many deaths on a daily basis all over the world, the World Health Organization (WHO) declared a pandemic on 11th March 2020. 4 This quick and uncontrolled spread of the disease can cause major concerns in patients with underlying diseases such as diabetes mellitus, cardiovascular diseases (CVDs), hypertension, pulmonary and rheumatic diseases. 2 This newly emerging disease imposes a lot of negative impacts on the economy and healthcare structures in affected countries. Currently, therapeutic options are limited and there is no registered and/or definite treatment or vaccine for this problem. Alternative solutions to prevent and control this virus are therefore desirable.
Vitamin D is a fat‐soluble micronutrient and also a hormone produced in body from sunlight. Besides its function in preserving bone integrity, it has a role in maturation of different cells such as immune cells. 5 Vitamin D deficiency is identified as a global public health matter and numerous investigations have reported that poor vitamin D status can affect a variety of diseases. 6 A majority of healthy individuals have low vitamin D concentration, mainly at the end of the winter. 5 Furthermore, individuals who stay at home or institution as well as night workers and elderly subjects, who have low exposure to sunlight, may suffer from vitamin D deficiency. 6 Since COVID‐19 was recognized in winter 2019 and mainly influenced middle‐aged and elderly subjects, it has been supposed that virus‐infected subjects may not have adequate vitamin D levels. Even though the mechanisms responsible for vitamin D function in the host immune system have been explained, 7 , 8 the interplay between viral infections and vitamin D remains a confusing matter, and plausible interactions between viral infections and vitamin D do not appear to be simple.
Angiotensin‐converting enzyme 2 (ACE2), a part of the renin‐angiotensin system (RAS), serves as the major entry point into cells for some coronaviruses such as HCoV‐NL63, SARS‐CoV and SARS‐CoV‐2 that cause SARS and COVID‐19, respectively. 9 , 10 , 11 , 12 It has also been shown that SARS‐CoV‐2 attaches to human ACE2 through its spike glycoprotein, thereby reducing the expression of ACE2 and causing lung injury and pneumonia. 12 , 13 , 14 , 15 Therefore, considering the importance of vitamin D in RAS and regulating ACE and ACE2, 16 we supposed that vitamin D could be considered as a potential therapeutic option for this novel coronavirus.
This narrative mini‐review has been conducted using an online search on PubMed and Google scholar in order to discuss the potential pathways connecting vitamin D, COVID‐19 and its therapy, with a focus on the RAS and ACE2.
2. ACE2: POTENTIAL TARGET IN COVID‐19 PATHOPHYSIOLOGY
Coronaviruses, the viruses with the largest single stranded‐RNA genome, belong to the family of Coronaviridae. 17 Coronaviruses cause enzootic diseases in birds and mammals, including humans. 18 Furthermore, the host immune response can be essential for the pathogenesis of clinical features. 19 When the virus binds to the host cells via its target receptor and enters the cells, its antigen will be presented to the antigen presentation cells (APC), which is a central part of the body's anti‐viral immunity. Antigen presentation then stimulates the body's humoral and cellular immunity, which are mediated by virus‐specific B and T cells. The antibody profile against acute viral infections including SARS‐CoV and SARS‐CoV‐2 has a typical pattern of IgM and IgG production. 20 Moreover, the number of CD4+ and CD8+ T cells decreased significantly in the peripheral blood of infected patients. 20 In addition, the inflammatory response and secretion of cytokines and chemokines, which occurs during acute viral infections like SARS‐CoV and SARS‐CoV‐2 attracts immune cells, particularly monocytes and T lymphocytes from the blood into the infected site. 21 In most subjects, recruited cells clear the infection in the lung, the immune response recedes and patients recover. 21 However, in some patients, a dysfunctional immune response occurs, which triggers a cytokine storm that mediates extensive lung inflammation. 21
ACE2 is a part of the RAS identified in 2000. 22 It is the human homologue of ACE but is different from ACE in substrate specificity. 22 ACE2 is a membrane protein found in nearly all body tissues with relatively higher expression in respiratory epithelial cells, alveolar cells type I and II, heart, kidney, blood vessels and the gastrointestinal tract. 9 , 23 , 24 ACE2 catalyzes the cleavage of angiotensin II (Ang II) (a vasoconstrictor peptide) into angiotensin 1‐7 [Ang‐(1‐7)] (a vasodilator), thereby reducing blood pressure. 25 Ang‐(1‐7) stimulates nitric oxide synthase (NOS) and further antagonizes Ang II activity through its AT1 receptor. 26 It seems that ACE/ACE2 ratio has a considerable impact on various diseases like diabetes, hypertension, and nephropathy. 27
ACE2is also the receptor on host cell surface and the major entry point into cells for some coronaviruses such as HCoV‐NL63, SARS‐CoV which causes SARS and SARS‐CoV‐2 which causes COVID‐19. 9 , 10 , 11 , 12 SARS‐CoV‐2 binds to human ACE2 through its spike glycoprotein and reduces ACE2 expression, thereby causing excessive generation of AngII via the enzyme ACE. Ang II has been demonstrated to stimulate cell growth and proliferation of lung fibroblasts 28 , 29 and up‐regulates the expression of transforming growth factor‐β (TGF‐β), 30 a central player in fibrogenesis that is associated with the differentiation of fibroblast to myofibroblasts, which dramatically increases extracellular matrix protein deposition. 31 Ang II is involved in lung inflammation through the generation of reactive oxygen species and the release of proinflammatory cytokines. 32 , 33 Ang II has been shown to induce alveolar epithelial cell apoptosis, 34 which is believed to initiate the fibrotic process and is also involved in promoting procoagulatory effects through activating plasminogen activator inhibitor‐1. 35 Induction of type 1a Ang II receptor (AGTR1A) enhances pulmonary vascular permeability, thereby causing lung injury and pneumonia; this possibly explains the lung damage following decreasing ACE2 expression. 12 , 13 , 14 , 15 In an animal study of ARDS, ACE2 knockout mice exhibited intense lung disease, characterized by increases in vascular permeability and lung edema. 36 Also, ACE2 overexpression decreased lipopolysaccharide (LPS)‐induced ARDS through the Ang‐(1‐7)/Mas pathway by suppressing extracellular signal‐regulated kinase/nuclear factor‐κB (NF‐κB) activation. 37 Increase in the ACE/ACE2 ratio that occurs during COVID‐19 presumably affects kidney damage initiation. 26 Meanwhile, ACE2 affects biologic peptides outside the RAS such as the Kinin‐Kallikrein system, Apelin‐13 and dynorphin A peptide. These peptides can be elevated and impose unfavorable effects by attenuatingACE2 expression. 38 Furthermore, ACE2 hydrolyzes the active bradykinin metabolite DABK (desArg9‐bradykinin). 39 A decreased activity of ACE2 impairs the inactivation of DABK and therefore, enhances its signaling through bradykinin B1 receptor (BKB1R). As a consequence, fluid extravasation and leukocyte recruitment to the lung occur. 40
3. INTERPLAY BETWEEN VITAMIN D AND ACE2: PROPOSED POTENTIAL THERAPY FOR COVID‐19
Vitamin D is a fat‐soluble vitamin and a hormone produced in the body from 7‐dehydrocholesterol with the help of sunlight or ultraviolet irradiation. In the liver, vitamin D converts to 25‐hydroxyvitamin D [25(OH)D], which is the principal circulating vitamin D metabolite and is commonly used as a vitamin D status index. The active form of vitamin D is named 1,25‐dihydroxyvitamin D [1,25(OH)2D3], which is synthesized in the kidney. 41 Immune cells are not only targets for active vitamin D, but are also local producers of vitamin D and are able to activate this hormone in a local fashion, thereby arguing for an autocrine or paracrine function for this vitamin within the immune system. 42 From this view, inadequate vitamin D levels can be linked to the immune anomalies, such as higher infection rates and autoimmunity. 42 According to the recent review, vitamin D is capable in lowering the risk of COVID‐19 infections and deaths through different mechanisms including maintenance of cell junctions, strengthening cellular immunity by reducing the cytokine storm with impacts on tumor necrosis factor α (TNF‐α) and interferon γ (IFN‐γ), 43 and modulating adaptive immunity by suppressing T helper cell type 1 responses and promoting T regulatory cells induction. 44 Vitamin D deficiency can therefore be indicated in ARDS, and heart failure as well as in sepsis, 42 and these can all be features of critically ill COVID‐19 patients. Based on a report by Tian et al, 45 vitamin D deficiency may be linked with a high risk of severity in COVID‐19. Vitamin D deficiency enhances the cytokine storm, 46 thereby, it is a risk factor for and/or a driver of the excessive and persistent inflammation, which is a main characteristic of ARDS and may be considerably lethal in subjects with SARS‐CoV‐2 infection. 47 , 48 , 49 In addition to the immune‐modulatory effects that include downregulation of pro‐inflammatory cytokines, vitamin D has a plausible protective role against acute lung injury (ALI)/ARDS. The expression of vitamin D receptor (VDR) is high in the lung, and according to Kong et al, 50 VDR‐knockout mice demonstrated ALI with higher severity and mortality in comparison with wild type mice following LPS treatment. Vitamin D deficiency was also noted to cause (ALI)/ARDS directly. 47 , 51 , 52 Moreover, Shi et al 53 reported that VDR‐knockout mice exhibited severe ALI induced by LPS; whereas a vitamin D analog, paricalcitol, improved LPS‐induced ALI and maintained alveolar barrier activity. In addition, according to Xu et al, 54 vitamin D attenuated LPS‐induced ALI by modulating the RAS. It has been reported that 1,25(OH)2D3 is a negative endocrine RAS modulator and inhibits renin expression and generation. 55 , 56 Compared to subjects with sufficient 25(OH)D levels (≥30 ng/mL), those with insufficiency (15‐29.9 ng/mL) and deficiency (<15 ng/mL) had higher circulating Ang II concentrations. Furthermore, those with vitamin D deficiency had significantly blunted renal plasma flow responses to infused Ang II. 57 Since there is no clear definition regarding the optimal concentration of vitamin D to use in different clinical settings including patients with COVID‐19, so this fact may impact its interaction with ACE2 in COVID‐19. Persistent deficiency in vitamin D level may activate the RAS, thereby leading to chronic CVD and decreased lung function as well as inducing RAS activation lung fibrosis. 58 Patients with these comorbidities account for a higher proportion of critically ill cases in COVID‐19. 58 The COVID‐19 infection becomes more severe especially in elderly, hypertensive, diabetic and obese patients and smokers. 59 Particularly in the presence of diabetes mellitus and accompanying comorbid conditions such as hypertension, obesity, old age and smoking, cytosolic pH is low, thus the virus easily may enter the cell by attaching to ACE2. 60 , 61 Especially in elderly patients with diabetes, COVID‐19 infection will be more severe since cytosolic pH is lower. 61 Recently, it has been reported that CVD or RAS blockade medications may increase ACE2 levels, augmenting available substrate for SARS‐CoV‐2 infection. 62 Furthermore, circulating ACE2, shed from endothelial cells is a biomarker of hypertension and heart failure 63 as well as diabetes, 64 reflecting increased ACE2 activity. SARS‐CoV‐2 infection down‐regulates ACE2 activity, resulting in toxic Ang II over‐accumulation which in turn may cause ARDS or fulminant myocarditis. 62 In addition, some medications such as antibiotics, anti‐inflammatory agents, antihypertensives and endocrine drugs decrease serum 25(OH)D levels via activating the pregnane‐X receptor. 65 Medication use usually increases with age. 65 Therefore, normalization of vitamin D concentrations can lower RAS activity by transcriptional suppression of renin expression. 66 Xu et al 54 also showed that vitamin D suppressed renin, ACE and AngII expression and increased ACE2 concentration in LPS‐induced ALI. Thus, vitamin D may decrease LPS‐induced ALI by inducing ACE2/Ang‐(1‐7) axis and suppressing renin and the ACE/Ang II/AT1R axis. 54 In another study by Yang et al, 67 1,25(OH)2D3 enhanced mRNA expression of VDR and ACE2 as well as VDR and ACE2 levels in rat models of LPS‐induced ALI compared to controls, indicating that increased expression of ACE2 mRNA and VDR mRNA had a role in protection against ALI development. Furthermore, clinical features and pathological changes of lung tissues in the calcitriol‐treated LPS rats were remarkably milder than controls. 67 Lin et al 68 observed that calcitriol decreased ACE concentration and ACE/ACE2 ratio and enhanced ACE2 concentration in diabetic rats. In addition, calcitriol treatment remarkably suppressed p38 mitogen‐activated protein kinase and extracellular signal‐regulated kinase (ERK) phosphorylation, but not c‐Jun N‐terminal kinases phosphorylation, and effectively weakened ACE up‐regulation and ACE2 down‐regulation. 68 Accordingly, administration of the synthetic vitamin D analog, paricalcitol, led to increased levels of ACE2 in tubular cells and decreased levels of ACE2 within the circulation in an animal model of type I diabetes, thereby slowing the development of diabetic nephropathy. 69 Cui et al 70 also reported that calcitriol suppressed Ang II receptor type 1 (AT1) and ACE and reduced Ang II formation in the spontaneously hypertensive rats. In addition, calcitriol had noticeable influence on ACE2/Ang‐(1‐7)/MasR axis with elevated expression of ACE2, MasR and Ang‐(1‐7) production. 70 The proposed potential therapeutic effect of vitamin D for COVID‐19 and induced ARDS is summarized in Figure 1.
FIGURE 1.
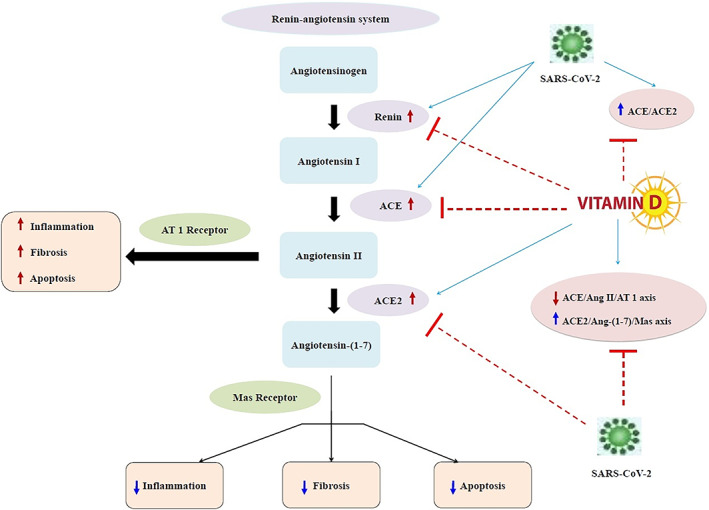
The proposed potential therapeutic effect of vitamin D for COVID‐19 and induced ARDS. ACE, angiotensin‐converting enzyme; ACE2: angiotensin‐converting enzyme 2
While the beneficial effects of vitamin D on COVID‐19 infection are mainly from observational studies of disease incidence or prevalence with respect to serum 25(OH)D concentrations, 43 most vitamin D randomized clinical trials have not declared that vitamin D supplementation reduced the risk of non‐skeletal disorders. 71 , 72 Although it is more likely that any protective effect of vitamin D against COVID‐19 is related to suppression of cytokine response and reduced severity/risk for ARDS, there is also evidence from a meta‐analysis that regular oral vitamin D2/D3 intake (in doses up to 2000 IU/day) is safe and protective against acute respiratory tract infection, especially in subjects with vitamin D deficiency. 73 Thus, it seems possible that vitamin D prophylaxis (without over‐dosing) may decrease the severity of illness caused by SARS‐CoV‐2, especially in settings where hypovitaminosis D is common. 74 Rhodes et al 75 also indicated that it would be reasonable to consider vitamin D supplementation to protect musculoskeletal health in those at risk of deficiency due to being housebound. Moreover, Marik et al 76 suggested that vitamin D deficiency may partly explain the geographic variations in the reported case fatality rate of COVID‐19, indicating that supplementation with vitamin D may reduce the mortality from this pandemic. In addition, significant relationship was observed between vitamin D levels and the number COVID‐19 cases and especially the mortality caused by this infection. 77 The most vulnerable group of population for COVID‐19 is also the one that has the most deficit in vitamin D. 77 To reduce the risk of infection, it is recommended that individuals at risk of COVID‐19 take 10 000 IU/day of vitamin D3 for a few weeks to rapidly increase 25(OH)D levels above 40‐60 ng/mL, followed by 5000 IU/day. 43 For treatment of subjects who become infected with COVID‐19, higher vitamin D3 doses might be useful. 43
4. CONCLUSION
Taking into account the above‐mentioned evidence, it seems that targeting the unbalanced RAS and ACE2 down‐regulation with vitamin D in SARS‐CoV‐2 infection may be a potential therapeutic approach to combat the expanding of COVID‐19 and induced ARDS. This mini‐review opens a new window for such a protective action of vitamin D in COVID‐19 and induced ARDS. Further controlled clinical studies of vitamin D as a potential COVID‐19 therapeutic option as well as determining the appropriate dosage and the presence of any safety concerns are warranted.
CONFLICT OF INTEREST
The authors have no competing interest.
Malek Mahdavi A. A brief review of interplay between vitamin D and angiotensin‐converting enzyme 2: Implications for a potential treatment for COVID‐19. Rev Med Virol. 2020;30:e2119. 10.1002/rmv.2119
REFERENCES
- 1. Zhou P, Yang XL, Wang XG, Hu B, et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020;579:270‐273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Huang C, Wang Y, Li X, Ren L, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan. China Lancet Lond Engl. 2020;395:497‐506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Yang X, Yu Y, Xu J, et al. Clinical course and outcomes of critically ill patients with SARS‐CoV‐2 pneumonia in Wuhan, China: a single‐centered, retrospective, observational study. Lancet Respir Med. 2020;8:475‐481. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. https://www.who.int/emergencies/diseases/novel-coronavirus-2019/events-as-they-happen.
- 5. Tangpricha V, Pearce EN, Chen TC, Holick MF. Vitamin D insufficiency among free‐living healthy young adults. Am J Med. 2002;112:659‐662. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Holick MF. Sunlight and vitamin D for bone health and prevention of autoimmune diseases, cancers, and cardiovascular disease. Am J Clin Nutr. 2004;80:1678S‐1688S. [DOI] [PubMed] [Google Scholar]
- 7. Sassi F, Tamone C, D'Amelio P. Vitamin D: nutrient, hormone, and immunomodulator. Nutrients. 2018;10:1656‐1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Chun RF, Liu PT, Modlin RL, Adams JS, Hewison M. Impact of vitamin D on immune function: lessons learned from genome‐wide analysis. Front Physiol. 2014;5:151‐165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Xu X, Chen P, Wang J, et al. Evolution of the novel coronavirus from the ongoing Wuhan outbreak and modeling of its spike protein for risk of human transmission. Sci China Life Sci. 2020;63:457‐460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Fehr AR, Perlman S. Coronaviruses: an overview of their replication and pathogenesis. Methods Mol Biol. 2015;1282:1‐23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Li F. Receptor recognition and cross‐species infections of SARS coronavirus. Antiviral Res. 2013;100:246‐254. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Kuba K, Imai Y, Rao S, et al. A crucial role of angiotensin converting enzyme 2 (ACE2) in SARS coronavirus‐induced lung injury. NatMed. 2005;11:875‐879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Li W, Moore MJ, Vasilieva N, et al. Angiotensin‐converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450‐454. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Chen Y, Guo Y, Pan Y, Zhao ZJ. Structure analysis of the receptor binding of 2019‐nCoV. Biochem Biophys Res Commun. 2020;525:135‐140. pii: S0006‐291X(20)30339–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Lu R, Zhao X, Li J, Niu P, et al. Genomic characterization and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565‐574. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Li YC. Molecular mechanism of vitamin D in the cardiovascular system. J Investig Med. 2011;59:868‐871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Banerjee A, Kulcsar K, Misra V, Frieman M, Mossman K. Bats and coronaviruses. Viruses. 2019;11:41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Schoeman D, Fielding BC. Coronavirus envelope protein: current knowledge. Virol J. 2019;16:69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Hoffmann M, Kleine‐Weber H, Schroeder S, et al. SARS‐CoV‐2 cell entry depends on ACE2 and TMPRSS2 and is blocked by a clinically proven protease inhibitor. Cell. 2020;181:271‐280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Li X, Geng M, Peng Y, Meng L, Lu S. Molecular immune pathogenesis and diagnosis of COVID‐19. J Pharm Anal. 2020;10:102‐108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Tay MZ, Poh CM, Rénia L, MacAry PA, Ng LFP. The trinity of COVID‐19: immunity, inflammation and intervention. Nat Rev Immunol. 2020;1‐12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Donoghue M, Hsieh F, Baronas E, et al. A novel angiotensin converting enzyme‐related carboxypeptidase (ACE2) converts angiotensin I to angiotensin 1‐9. Circ Res. 2000;87:E1‐E9. [DOI] [PubMed] [Google Scholar]
- 23. Harmer D, Gilbert M, Borman R, Clark KL. Quantitative mRNA expression profiling of ACE 2, a novel homologue of angiotensin converting enzyme. FEBS Lett. 2002;532:107‐110. [DOI] [PubMed] [Google Scholar]
- 24. Xu H, Zhong L, Deng J, et al. High expression of ACE2 receptor of 2019‐nCoV on the epithelial cells of oral mucosa. Int J Oral Sci. 2020;12:1‐5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Keidar S, Kaplan M, Gamliel‐Lazarovich A. ACE2 of the heart: from angiotensin I to angiotensin (1‐7). Cardiovasc Res. 2007;73:463‐469. [DOI] [PubMed] [Google Scholar]
- 26. Mizuiri S, Ohashi Y. ACE and ACE2 in kidney disease. World J Nephrol. 2015;4:74‐82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Imai Y, Kuba K, Penninger JM. The discovery of angiotensin‐converting enzyme 2 and its role in acute lung injury in mice. Exp Physiol. 2008;93:543‐548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Marshall RP, McAnulty RJ, Laurent GJ. Angiotensin II is mitogenic for human lung fibroblasts via activation of the type 1 receptor. Am J Respir Crit Care Med. 2000;161:1999‐2004. [DOI] [PubMed] [Google Scholar]
- 29. Marshall RP, Gohlke P, Chambers RC, et al. Angiotensin II and the fibroproliferative response to acute lung injury. Am J Physiol Lung Cell Mol Physiol. 2004;286:L156‐L164. [DOI] [PubMed] [Google Scholar]
- 30. Uhal BD, Kim JK, Li X, Molina‐Molina M. Angiotensin‐TGF‐beta 1 crosstalk in human idiopathic pulmonary fibrosis: autocrine mechanisms in myofibroblasts and macrophages. Curr Pharm des. 2007;13:1247‐1256. [DOI] [PubMed] [Google Scholar]
- 31. Guo W, Shan B, Klingsberg RC, Qin X, Lasky JA. Abrogation of TGF‐beta1‐induced fibroblast‐myofibroblast differentiation by histone deacetylase inhibition. Am J Physiol Lung Cell Mol Physiol. 2009;297:L864‐L870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Mastruzzo C, Crimi N, Vancheri C. Role of oxidative stress in pulmonary fibrosis. Monaldi Arch Chest Dis. 2002;57:173‐176. [PubMed] [Google Scholar]
- 33. Okada M, Suzuki K, Matsumoto M, et al. Effects of angiotensin on the expression of fibrosis‐associated cytokines, growth factors, and matrix proteins in human lung fibroblasts. J Clin Pharm Ther. 2009;34:288‐299. [DOI] [PubMed] [Google Scholar]
- 34. Wang R, Zagariya A, Ibarra‐Sunga O, et al. Angiotensin II induces apoptosis in human and rat alveolar epithelial cells. Am J Physiol. 1999;276:L885‐L889. [DOI] [PubMed] [Google Scholar]
- 35. Liu RM. Oxidative stress, plasminogen activator inhibitor 1, and lung fibrosis. Antioxid Redox Signal. 2008;10:303‐319. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Imai Y, Kuba K, Rao S, et al. Angiotensin‐converting enzyme 2 protects from severe acute lung failure. Nature. 2005;436:112‐116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Li Y, Zeng Z, Cao Y, et al. Angiotensin‐converting enzyme 2 prevents lipopolysaccharide‐induced rat acute lung injury via suppressing the ERK1/2 and NF‐κB signaling pathways. Sci Rep. 2016;6:1‐14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Tang Z, Wang Z, Hu Z, Zhang M, Li L, Li B. The role of bradykinin in lung ischemia‐reperfusion injury in a rat lung transplantation model. Acta Cir Bras. 2016;31:807‐812. [DOI] [PubMed] [Google Scholar]
- 39. Vickers C, Hales P, Kaushik V, et al. Hydrolysis of biological peptides by human angiotensin‐converting enzyme‐related carboxypeptidase. J Biol Chem. 2002;277:14838‐14843. [DOI] [PubMed] [Google Scholar]
- 40. Sodhi CP, Wohlford‐Lenane C, Yamaguchi Y, et al. Attenuation of pulmonary ACE2 activity impairs inactivation of des‐Arg(9) bradykinin/BKB1R axis and facilitates LPS‐induced neutrophil infiltration. Am J Physiol Lung Cell Mol Physiol. 2018;314:L17‐L31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Holick MF, Vitamin D. deficiency. N Engl J Med. 2007;357:266‐281. [DOI] [PubMed] [Google Scholar]
- 42. Baeke F, Takiishi T, Korf H, Gysemans C, Mathieu C. Vitamin D: modulator of the immune system. Curr Opin Pharmacol. 2010;10:482‐496. [DOI] [PubMed] [Google Scholar]
- 43. Grant WB, Lahore H, McDonnell SL, et al. Evidence that Vitamin D supplementation could reduce risk of influenza and COVID‐19 infections and deaths. Nutrients. 2020;12:988‐1015. pii: E988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Cantorna MT, Snyder L, Lin YD, Yang L. Vitamin D and 1,25(OH)2D regulation of T cells. Nutrients. 2015;7:3011‐3021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45. Tian Y, Rong L. Letter: COVID‐19, and vitamin D. Authors’ Reply Aliment PharmacolTher. 2020;51:995‐996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Khare D, Godbole NM, Pawar SD, et al. Calcitriol (1, 25[OH]2 D3) pre‐ and post‐treatment suppresses inflammatory response to influenza a (H1N1) infection in human lung A549 epithelial cells. Eur J Nutr. 2013;52:1405‐1415. [DOI] [PubMed] [Google Scholar]
- 47. Dancer RC, Parekh D, Lax S, et al. Vitamin D deficiency contributes directly to the acute respiratory distress syndrome (ARDS). Thorax. 2015;70:617‐624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Parekh D, Thickett DR, Turner AM. Vitamin D deficiency and acute lung injury. Inflamm Allergy Drug Targets. 2013;12:253‐261. [DOI] [PubMed] [Google Scholar]
- 49. Brown R, Sarkar A. Vitamin D deficiency: a factor in COVID‐19, progression, severity and mortality?‐an urgent call for research. Mito Fit Preprint Arch. 2020;1‐29. [Google Scholar]
- 50. Kong J, Zhu X, Shi Y, et al. VDR attenuates acute lung injury by blocking Ang‐2‐Tie‐2 pathway and renin‐angiotensin system. Mol Endocrinol. 2013;27:2116‐2125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51. Park S, Lee MG, Hong SB, Lim CM, Koh Y, Huh JW. Effect of vitamin D deficiency in Korean patients with acute respiratory distress syndrome. Korean J Intern Med. 2019;34:685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Onwuneme C, Martin F, McCarthy R, et al. The association of vitamin D status with acute respiratory morbidity in preterm infants. J Pediatr. 2015;166:1175‐1180. [DOI] [PubMed] [Google Scholar]
- 53. Shi YY, Liu TJ, Fu JH, et al. Vitamin D/VDR signaling attenuates lipopolysaccharide‐induced acute lung injury by maintaining the integrity of the pulmonary epithelial barrier. Mol Med Rep. 2016;13:1186‐1194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54. Xu J, Yang J, Chen J, Luo Q, Zhang Q, Zhang H. Vitamin D alleviates lipopolysaccharide‐induced acute lung injury via regulation of the renin‐angiotensin system. Mol Med Rep. 2017;16:7432‐7438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55. Li YC, Qiao G, Uskokovic M, Xiang W, Zheng W, Kong J. Vitamin D: a negative endocrine regulator of the renin‐angiotensin system and blood pressure. J Steroid Biochem Mol Biol. 2004;89‐90:387‐392. [DOI] [PubMed] [Google Scholar]
- 56. Li YC, Kong J, Wei M, Chen ZF, Liu SQ, Cao LP. 1,25‐Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin‐angiotensin system. J Clin Invest. 2002;110:229‐238. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Tomaschitz A, Pilz S, Ritz E, et al. Independent association between 1,25‐dihydroxyvitamin D, 25‐hydroxyvitamin D and the renin‐angiotensin system: the Ludwigshafen risk and cardiovascular health (LURIC) study. Clin Chim Acta. 2010;411:1354‐1360. [DOI] [PubMed] [Google Scholar]
- 58. Shi Y, Liu T, Yao LI, et al. Chronic vitamin D deficiency induces lung fibrosis through activation of the renin‐angiotensin system. Sci Rep. 2017;7:3312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Cure E, Cumhur CM. Comment on organ‐protective effect of angiotensin‐converting enzyme 2 and its effect on the prognosis of COVID‐19. J Med Virol. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Resnick LM, Gupta RK, Sosa RE, Corbett ML, Laragh JH. Intracellular pH in human and experimental hypertension. Proc Natl Acad Sci. 1987;84:7663‐7667. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Cure E, Cumhur CM. Angiotensin‐converting enzyme inhibitors and angiotensin receptor blockers may be harmful in patients with diabetes during COVID‐19 pandemic. Diabetes Metab Syndr. 2020;14:349‐350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Hanff TC, Harhay MO, Brown TS, Cohen JB, Mohareb AM. Is there an association between COVID‐19 mortality and the renin‐angiotensin system – a call for epidemiologic investigations. Clin Infect Dis. 2020; pii: ciaa329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Uri K, Fagyas M, ManyineSiket I, et al. New perspectives in the renin‐angiotensinaldosterone system (RAAS) IV: circulating ACE2 as a biomarker of systolic dysfunction in human hypertension and heart failure. PLoS One. 2014;9:e87845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Soro‐Paavonen A, Gordin D, Forsblom C, et al. Circulating ACE2 activity is increased in patients with type 1 diabetes and vascular complications. J Hypertens. 2012;30:375‐383. [DOI] [PubMed] [Google Scholar]
- 65. Grober U, Kisters K. Influence of drugs on vitamin D and calcium metabolism. Dermatoendocrinol. 2012;4:158‐166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Yuan W, Pan W, Kong J, et al. 1,25‐dihydroxyvitamin D3 suppresses renin gene transcription by blocking the activity of the cyclic AMP response element in the renin gene promoter. J Biol Chem. 2007;282:29821‐29830. [DOI] [PubMed] [Google Scholar]
- 67. Yang J, Zhang H, Xu Z. Effect of Vitamin D on ACE2 and Vitamin D Receptor Expression in Rats with LPS‐Induced Acute Lung Injury. 2016. [Google Scholar]
- 68. Lin M, Gao P, Zhao T, et al. Calcitriol regulates angiotensin‐converting enzyme and angiotensin converting‐enzyme 2 in diabetic kidney disease. Mol Biol Rep. 2016;43:397‐406. [DOI] [PubMed] [Google Scholar]
- 69. Riera M, Anguiano L, Clotet S, et al. Paricalcitol modulates ACE2 shedding and renal ADAM17 in NOD mice beyond proteinuria. Am J Physiol Renal Physiol. 2016;310:F534‐F546. [DOI] [PubMed] [Google Scholar]
- 70. Cui C, Xu P, Li G, et al. Vitamin D receptor activation regulates microglia polarization and oxidative stress in spontaneously hypertensive rats and angiotensin II‐exposed microglial cells: role of renin‐angiotensin system. Redox Biol. 2019;26:101295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Autier P, Mullie P, Macacu A, et al. Effect of vitamin D supplementation on non‐skeletal disorders: a systematic review of meta‐analyses and randomised trials. Lancet Diabetes Endocrinol. 2017;5:986‐1004. [DOI] [PubMed] [Google Scholar]
- 72. Rejnmark L, Bislev LS, Cashman KD, et al. Non‐skeletal health effects of vitamin D supplementation: a systematic review on findings from meta‐analyses summarizing trial data. PLoS One. 2017;12:e0180512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Martineau AR, Jolliffe DA, Hooper RL, et al. Vitamin D supplementation to prevent acute respiratory tract infections: systematic review and meta‐analysis of individual participant data. BMJ. 2017;356:i6583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Panarese A, Shahini E. Letter: COVID‐19, and vitamin D. Aliment Pharmacol Ther. 2020;51:993‐995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75. Rhodes JM, Subramanian S, Laird E, Kenny RA. Editorial: low population mortality from COVID‐19 in countries south of latitude 35 degrees north supports vitamin D as a factor determining severity. Aliment Pharmacol Ther. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Marik PE, Kory P, Varon J. Does vitamin D status impact mortality from SARS‐CoV‐2 infection?. Med Drug Discov. 2020;6:1‐2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77. Ilie PC, Stefanescu S, Smith L. The role of vitamin D in the prevention of coronavirus disease 2019 infection and mortality. Aging Clin Exp Res. 2020. [DOI] [PMC free article] [PubMed] [Google Scholar]


