ABSTRACT
Objective:
The objectives of this study were to clarify whether prevertebral soft tissue (PVST) thickening increases the risk of dysphagia, to identify at which vertebral level determining PVST thickness is useful, and to determine cutoff values.
Methods:
A total of 80 patients with traumatic cervical spinal cord injury (TCSCI) treated over a 43-month period at a single regional institution specializing in cervical spinal cord injuries participated in the study. The exclusion criteria were having undergone anterior cervical spine surgery; a history of complicated traumatic brain injury, complicated vertebral artery injury, or brain injury; endotracheal intubation at day 30 after onset; the disappearance of neurological symptoms within 24 hours after onset; and the use of a halo vest. The associations between PVST thicknesses at C1, C2, C3, C6, and C7 measured by CT on the day of onset and the presence of dysphagia (Food Intake LEVEL Scale score <8) at 30 and 60 days after onset of TCSCI were analyzed using ROC curves to calculate the maximum area under the curve and the PVST cutoff values for these vertebrae. Associations between various risk factors, including PVST thickness, and dysphagia at days 30 and 60 after onset were examined using univariate and multivariate analyses.
Results:
Independent associations with dysphagia were found with the C3 PVST thickness (day 30: ≥8.3 mm, day 60: ≥9.4 mm) and tracheostomy.
Conclusion:
PVST thickness or injury seems to be an independent risk factor for dysphagia. By measuring PVST, it is possible to estimate the severity of dysphagia even in acute conditions.
Keywords: dysphagia, prevertebral soft tissue injury, traumatic spinal cord injury
INTRODUCTION
Dysphagia is a relatively common complication of traumatic cervical spinal cord injury (TCSCI)1,2,3,4,5,6,7,8,9,10,11) that can cause a variety of respiratory complications,1,2,3,4,5,6,7,8,9,10,11) negatively impact vital prognosis, and increase medical costs. Furthermore, the growing emphasis on achieving early oral feeding training, which is thought to shorten medical treatment times, means that the rapid diagnosis of dysphagia is essential.
Discovering risk factors for dysphagia after TCSCI would help to achieve early diagnosis and the determination of prognosis. A number of studies has investigated risk factors such as age, neurological level, full or partial paralysis, tracheostomy, use of a respirator, and cervical spine surgery.1,2,3,4,5,6,7,8,9,10,11) While most reports have found a correlation between dysphagia and tracheostomy,1,3,4,5,6,7,8,9,10) the results for other risk factors have been mixed.
Moreover, while tracheostomy has long been reported as having an effect on dysphagia,12,13,14,15,16,17,18) several recent studies have not observed any impact of tracheostomy on dysphagia or swallowing motion.19,20,21,22,23) Such findings raise doubt as to whether tracheostomy is indeed a risk factor for dysphagia. Therefore, there is a possibility that there exist more important risk factors than tracheostomy.
TCSCI is sometimes complicated by damage to the prevertebral soft tissue (PVST),24,25) which shows up on images as PVST thickening.25) The pharyngeal plexus and pharyngeal constrictor form part of the PVST,26) which, if damaged, may cause dysphagia.10,27) Indeed, some studies have observed a correlation between postoperative PVST swelling and dysphagia after anterior cervical spinal surgery (ACSS).28,29)
Very few studies have examined the correlation between PVST damage and dysphagia. We could find only one relevant report, by Ihalainen et al.,11) which examined PVST thickness at C3 and C6 as potential risk factors for dysphagia, but no correlations were found.11) However, Ihalainen et al. did not measure the PVST thickness immediately after TCSCI; moreover, their reasons for selecting C3 and C6 were unclear. Furthermore, their cutoff values for PVST thickness were based on the upper bounds from a report by Rojas et al.,25) although it is uncertain whether these values are appropriate for determining the risk factors for dysphagia.
The purpose of the present study was to clarify whether PVST thickening or injury after TCSCI increases the risk of dysphagia, to identify at which vertebral level determining PVST thickness is useful, and to determine cutoff values. Furthermore, we aimed to examine a variety of risk factors to determine which are independent factors.
METHODS
Subjects
A retrospective search was made for patients with TCSCI admitted to Yonemori Hospital, Kagoshima, from September 2014 to April 2018 using the hospital’s medical records database. Yonemori Hospital accepts patients with spinal cord injuries from all areas of Kagoshima Prefecture 24 hours a day. The exclusion criteria were a history of ACSS, complicated traumatic brain injury, complicated vertebral artery injury, cerebrovascular disorder, traumatic brain injury, or neurogenerative disease; endotracheal intubation at day 30 after onset; not having undergone cervical CT before endotracheal intubation on the day of onset; the disappearance of neurological symptoms within 24 hours; the use of a halo vest; and missing information on any of the previous items.
Data Collection
Oral Intake Ability (Dysphagia)
Food Intake LEVEL Scale (FILS)30) scores on day 30 and day 60 after onset were examined retrospectively from medical records. A FILS score <8 was considered to be dysphagia. A FILS score of 8 means “The patient eats three meals by excluding food that is particularly difficult to swallow.”30) As a patient’s oral intake becomes more restricted, the FILS score decreases, progressing to no oral intake at FILS scores of ≤3.
PVST Thickness
PVST thickness was measured using sagittal sections on cervical CT at C1, C2, C3, C6, and C7 on the day of onset before endotracheal intubation. Because the characteristics of the soft tissue anterior to C4 and C5 make it difficult to set the upper bounds for PVST thickness,25) measurements at these vertebrae were not performed.
Risk Factors
Age, gender, ASIA Impairment Scale (AIS) at onset, neurological level, the duration of dependence on mechanical ventilation, the presence or absence of tracheostomy, the presence or absence of interbody fusion (excluding patients who underwent ACSS), and the or absence of laminoplasty were examined retrospectively from medical records.
Associations between dysphagia at day 30, age, and the duration of mechanical ventilation were examined using ROC) curve analysis, and cutoff values were calculated using the Youden index. These were classified as ≥/<66 years old and ≥/<3 days, respectively. Patients were also classified as AIS A/B or C/D. The neurological level was classified as C4 and above or C5 and below.
Statistical Analysis
ROC curve analysis was used to examine associations between PVST thickness and dysphagia to calculate the area under the curve (AUC) and cutoff values for PVST at the C1, C2, C3, C6, and C7 levels. The associations between these cutoff values and dysphagia were examined with the chi-square test.
Next, using the chi-square test, we investigated the associations between dysphagia at 30 and 60 days and age, sex, AIS, neurological level, the duration of dependence on mechanical ventilation, the presence or absence of tracheostomy, the presence or absence of interbody fusion (excluding patients who underwent ACSS), and the presence or absence of laminoplasty.
Logistic regression analysis was performed on PVST thickness of not less than cut-off value at the vertebral level with the greatest AUC and risk factors for dysphagia that had significant associations in the chi-square test. Variable selection with a stepwise model (Bayesianinformation criterion reduction) was also used. The vertebral level at which the PVST thickness had the greatest AUC was determined using ROC curve analysis. EZR version 1.4031) was used for the statistical analyses and calculations.
Ethical Considerations
We conducted this study in accordance with the Declaration of Helsinki and the study was approved by the Ethical Review Board of Yonemori Hospital (approval number: 18002).
RESULTS
Sample Size
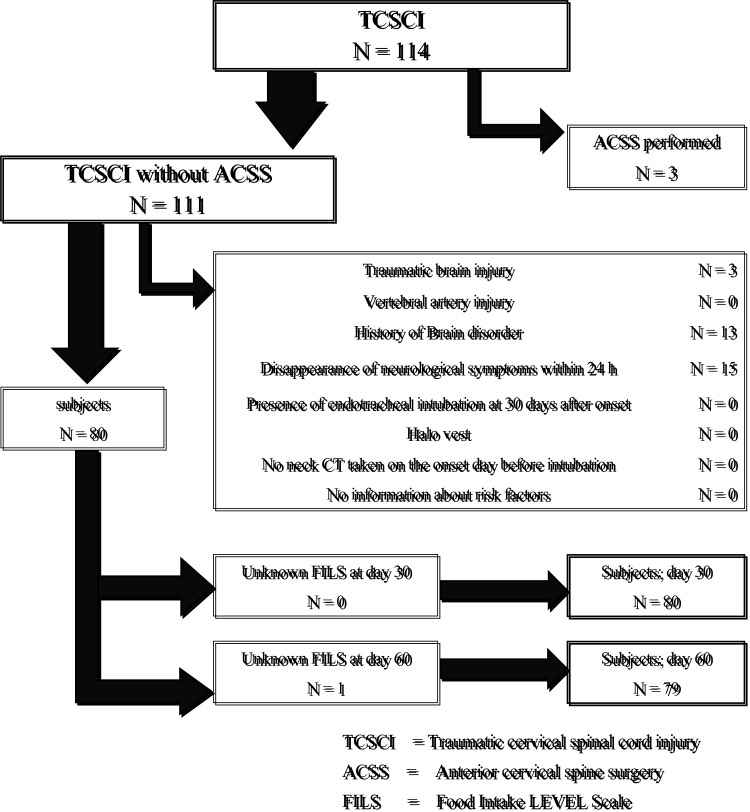
Figure 1 shows the flow diagram for patient selection. There were 80 and 79 patients at day 30 and 60 after onset, respectively. Figure 2 shows the patients’ age distribution and Table 1 shows the affected neurological levels and AIS categories.
Fig. 1.
Patient selection flow chart
Fig. 2.
Age distribution of study participants
Table 1. ASIA Impairment Scale (AIS) for each vertebral level.
| Neurological Level | Total | |||||||||
| C1 | C2 | C3 | C4 | C5 | C6 | C7 | C8 | |||
| AIS | A | 0 | 0 | 0 | 5 (4)* | 6 | 0 | 2 | 0 | 13 (12)* |
| B | 0 | 0 | 4 | 6 | 0 | 1 | 0 | 0 | 11 | |
| C | 0 | 0 | 1 | 13 | 5 | 1 | 1 | 0 | 21 | |
| D | 0 | 0 | 2 | 10 | 9 | 9 | 5 | 0 | 35 | |
| E | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| Total | 0 | 0 | 7 | 34 (33)* | 20 | 11 | 8 | 0 | 80 (79)* | |
* (): number of patients at day 60
Determining PVST Thickness Cutoff Values for Each Vertebral Level
Table 2 shows the ROC curve analyses for dysphagia and PVST thickness at each vertebral level and the results of chi-square testing of the cutoff values. Associations were observed for all vertebrae except C7 at day 30 and for all vertebrae at day 60. C3 had the largest AUC at both 30 and 60 days. Further analyses were carried out using the PVST thickness at C3 (C3:PVST). Using the Youden index, the C3:PVST cutoff values were 8.3 mm at day 30 and 9.4 mm at day 60.
Table 2. ROC curve analysis for PVST thickness and chi-square test using the cutoff values.
| Day 30 | ||||||||||
| ROC | Chi-square test | |||||||||
| AUC | 95% CI | Cutoff value | Sensitivity | Specificity | PVST thickness |
Dysphagia | P value | |||
| Yes | No | |||||||||
| n=17 | n=63 | |||||||||
| C1 | 0.673 | 0.508–0.837 | 4.9 | 0.529 | 0.889 | ≥4.9 | 9 | 7 | <0.001* | |
| C2 | 0.833 | 0.72–0.947 | 5.9 | 0.706 | 0.921 | ≥5.9 | 12 | 6 | <0.001* | |
| C3 | 0.877 | 0.787–0.968 | 8.3 | 0.765 | 0.921 | ≥8.3 | 13 | 6 | <0.001* | |
| C6 | 0.703 | 0.571–0.834 | 12.3 | 0.941 | 0.476 | ≥12.3 | 16 | 33 | 0.0043* | |
| C7 | 0.558 | 0.382–0.734 | 13.3 | 0.412 | 0.81 | ≥13.3 | 7 | 12 | 0.114 | |
| Day 60 | ||||||||||
| ROC | Chi-square test | |||||||||
| AUC | 95% CI | Cutoff value | Sensitivity | Specificity | PVST thickness | Dysphagia | P value | |||
| Yes | No | |||||||||
| n=9 | n=70 | |||||||||
| C1 | 0.662 | 0.452–0.872 | 4.9 | 0.556 | 0.843 | ≥4.9 | 5 | 11 | 0.0183* | |
| C2 | 0.799 | 0.652–0.945 | 5.9 | 0.778 | 0.843 | ≥5.9 | 7 | 11 | <0.001* | |
| C3 | 0.828 | 0.685–0.971 | 9.4 | 0.778 | 0.914 | ≥9.4 | 7 | 6 | <0.001* | |
| C6 | 0.795 | 0.669–0.921 | 15.1 | 0.667 | 0.786 | ≥15.1 | 6 | 15 | 0.0127* | |
| C7 | 0.764 | 0.6–0.929 | 13.3 | 0.667 | 0.829 | ≥13.3 | 6 | 12 | 0.00359* | |
C3 had the largest AUC at both 30 and 60 days. Further analyses used PVST thickness at C3.
The cutoff values were determined using the Youden index.
Association with the Tested Risk Factors
Table 3 shows the associations between dysphagia and various risk factors. No association was found with age, sex, AIS, or neurological level.
Table 3. Risk factors for dysphagia in traumatic cervical spinal cord injury.
| Day 30 n=80 | Day 60 n=79 | ||||||
| Dysphagia | Chi-square test | Dysphagia | Chi-square test | ||||
| Yes | No | P value | Yes | No | P value | ||
| n=17 | n=63 | n=9 | n=70 | ||||
| Age ≥66 years | 11 | 26 | 0.148 | 7 | 29 | 0.0881 | |
| Gender male | 14 | 48 | 0.832 | 8 | 53 | 0.642 | |
| AIS A and B | 12 | 12 | <0.001* | 5 | 18 | 0.143 | |
| Neurological level C4 and above | 12 | 29 | 0.127 | 6 | 34 | 0.504 | |
| Mechanical ventilator use ≥3 days | 9 | 3 | <0.001* | 5 | 6 | <0.001* | |
| Tracheostomy operation | 8 | 2 | <0.001* | 5 | 4 | <0.001* | |
| Interbody fusion (without ACSS) |
14 | 12 | <0.001* | 8 | 18 | <0.001* | |
| Laminoplasty (without interbody fusion) |
0 | 12 | 0.117 | 0 | 12 | 0.392 | |
| PVST thickness at C3 ≥8.3mm | 13 | 6 | <0.001* | – | – | – | |
| PVST thickness at C3 ≥9.4mm | – | – | – | 7 | 6 | <0.001* | |
Multivariate Analysis
Table 4 shows the results of logistic regression analyses using risk factors that showed significant associations in the chi-square tests. At day 30 after onset, a significant association was observed only for C3:PVST ≥8.3 mm [odds ratio (OR) 13.60, 95% confidence interval (95% CI) (2.38–78.0)]. At day 60, an association was observed for C3:PVST ≥9.4 mm and tracheostomy. Using a stepwise model for variable selection, C3:PVST and tracheostomy at both 30 and 60 days were selected, both of which showed significant associations.
Table 4. Logistic regression model summaries assessing risk factors for dysphagia.
| Day 30 | Univariate analysis | Multivariate analysis | Stepwise selection model | ||||||||
| Odds ratio | (95% CI) | P value | Odds ratio | (95% CI) | P value | Odds ratio | (95% CI) | P value | |||
| AIS A or B | 10.2 | (3.02–34.50)* | <0.001* | 2.8 | (0.45–17.40) | 0.27 | |||||
| PVST thickness (C3) ≥8.3 mm | 30.9 | (7.60–125.0)* | <0.001* | 13.6 | (2.38–78.0)* | 0.0033* | 23.4 | (4.98–110.0)* | <0.001* | ||
| Tracheostomy | 27.1 | (4.950–148.0)* | <0.001* | 9.59 | (0.52–176.0) | 0.13 | 17 | (2.090–139.0)* | 0.0081* | ||
| Mechanical ventilator Use >3 days | 22.5 | (5.020–101.00)* | <0.001* | 0.77 | (0.06–9.86) | 0.84 | |||||
| Interbody fusion | 19.8 | (4.910–80.20)* | <0.001* | 4.1 | (0.63–26.70) | 0.14 | |||||
| Day 60 | Univariate analysis | Multivariate analysis | Stepwise selection model | ||||||||
| Odds ratio | (95% CI) | P value | Odds ratio | (95% CI) | P value | Odds ratio | (95% CI) | P value | |||
| PVST Thickness (C3) ≥9.4 mm | 14.4 | (2.310–90.50)* | 0.00434* | 19.8 | (2.36–167.0)* | 0.006* | 25.2 | (3.69–172.0)* | <0.001* | ||
| Tracheostomy | 69 | (6.710–710.0)* | <0.001* | 36.2 | (1.09–1200.0)* | 0.044* | 11 | (1.35–89.60)* | 0.025* | ||
| Mechanical ventilator Use >3 days | 16.5 | (2.600–105.0)* | 0.00297* | 0.1 | (0.00–4.24) | 0.23 | |||||
| Interbody fusion | 4.73 | (0.80600–27.700) | 0.0853 | 7.52 | (0.49–115.00) | 0.15 | |||||
DISCUSSION
Although a variety of risk factors for dysphagia as a complication of TCSCI have been reported,1,2,3,4,5,6,7,8,9,10,11) the results of the present study indicate that only the PVST thickness at C3 and tracheostomy are independent risk factors. According to Rojas et al.,25) the normal mean C3:PVST is 4.2 mm (range 2–9.5 mm) with a cutoff value of 7 mm.25) The cutoff values in the present study were 8.3 mm at day 30 after onset and 9.4 mm at day 60 after onset, which are slightly higher than their upper bound but are within their range. This does not necessarily mean that risk increases when the cutoff value reported by Rojas et al.25) is exceeded; rather, it is surmised that dysphagia risk would increase in cases with more than a certain level of PVST thickening. Further, the C3:PVST cutoff value at post-onset day 60 was higher than that at day 30, which suggests that greater PVST thickening may prolong dysphagia.
PVST Injury
In this study, we demonstrated an association between increased C3:PVST and dysphagia. The oropharynx and hypopharynx are at the C3 vertebra level and contain the pharyngeal plexus and the pharyngeal constrictor muscles. Consequently, increased C3:PVST may suggest damage to the pharyngeal plexus and pharyngeal constrictor muscles.
A few studies have reported a correlation between post-ACSS dysphagia and PVST edema,26,28,29) which may be due to damage to the pharyngeal plexus or pharyngeal constrictor.26) However, PVST in TCSCI associated with hyperextension can cause pharyngeal dysphagia similar to that seen after ACSS. Pharyngeal phase problems have been reported in dysphagia caused by TCSCI, including delayed swallowing reflex and reduced pharyngeal contractions.6) These findings support the idea of damage to the pharyngeal plexus or pharyngeal constrictor.
Tracheostomy
Multiple studies have found a correlation between tracheostomy and TCSCI-associated dysphagia1,3,4,5,6,7,8,9,10); however, opinions are divided over whether tracheostomy itself is a cause of dysphagia.12,13,14,15,16,17,18,19,20,21,22,23)
Tracheostomy increases aspiration, regardless of the underlying disease,12,13,14,15) and this is thought to be caused by a variety of factors.13,16,17,18) However, the presence or absence of a tracheostomy tube has not been found to affect the movements of the hyoid bone and larynx.22) Moreover, laryngeal elevation, pharyngeal constriction, and the opening of the upper esophageal sphincter have not been found to change after tracheostomy tube removal,23) neither has increased aspiration been found after tracheostomy.19,20,21) Therefore, it cannot be concluded that performing a tracheostomy will have a negative impact on swallowing function in all cases.
Further, airway secretion-processing dysfunction caused by respiratory muscle paralysis in TCSCI increases airway secretions.11) An increase in airway secretions is difficult to distinguish from the accumulation of pharyngeal secretions caused by pharyngeal dysphagia, which could delay the decision to begin oral feeding.
In the present study, an independent association with the presence of a tracheostomy could not be confirmed until after day 30 after onset in a multivariate analysis unless a stepwise model was used, which indicates that the association is not as strong as that for PVST damage.
Other Risk Factors
In the present study, complete/incomplete tetraplegia, neurological level, and the duration of dependence on mechanical ventilation were not found to be independent risk factors. Previous results on these factors have not been consistent.1,2,3,4,5,6,7,8,9,10,11) Complete/incomplete quadriplegia, neurological level, and the dependence on mechanical ventilation may be associated with reduced airway clearance caused by ventilatory dysfunction, which could be compounding factors attributed to tracheostomy.
Correlations of dysphagia with age have been observed in some studies1,6,7,10) but not in others.2,3,5) In the present study, non-TCSCI causes of dysphagia were excluded, which may have eliminated cases of age-related dysphagia from the cohort.
Studies of cervical spinal surgery and dysphagia have shown correlations with ACSS.1,4) We believe the present study’s exclusion of patients with a variety of complicating conditions, such as a history of ACSS that could be associated with dysphagia, and the application of multivariate analysis eliminated the predicted confounding factors.
Limitations
This was a single-center study. The distributions of the age at onset, AIS, and injury level were similar to those in an epidemiological study of spinal cord injuries at Rosai Hospitals and related facilities throughout Japan.32) However, according to an epidemiological survey of spinal cord injuries in Japan, the annual incidence of cervical spinal cord injury was 30.15 per million nationwide (1990–1992),33) 25.41 per million in Fukuoka Prefecture (2005–2007),34) and 105.39–109.26 per million in Tokushima Prefecture (2011–2012).35) Based on these reports, the estimated number of cervical cord injuries during the study period in Kagoshima Prefecture, with a population of about 1.6 million (based on the 2015 Population Census of Japan) is 149–401. Therefore, the size of the current study cohort was small.
FILS scores were obtained from a retrospective examination of medical records and were used in this study to assess improvement in dysphagia. FILS evaluates oral feeding ability; consequently, it may not accurately reflect swallowing function. A videofluoroscopic swallowing study and/or fiberoptic endoscopic evaluation of swallowing should be used to obtain a more accurate assessment of swallowing function. Furthermore, pulmonary function, oral function, and willingness to eat should be evaluated.
CONCLUSION
PVST thickening and tracheostomy exhibited independent associations with dysphagia as a complication of TCSCI. C3:PVST ≥8.3 mm was associated with dysphagia at day 30 (OR 23.4) and C3:PVST ≥9.4 mm was associated with dysphagia at day 60 after onset (OR 25.2).
Two factors may coexist in dysphagia as a complication of TCSCI—pharyngeal dysphagia accompanying damage to the prevertebral soft tissue and problems with airway clearance resulting from respiratory muscle paralysis. The distinction between these two causes is important in the decision to start oral intake. Furthermore, by measuring PVST, it is possible to estimate dysphagia even in acute conditions.
Footnotes
CONFLICTS OF INTEREST: The authors have no conflicts of interest directly relevant to the content of this article.
REFERENCES
- 1.Kirshblum S,Johnston MV,Brown J,O’Connor KC,Jarosz P: Predictors of dysphagia after spinal cord injury. Arch Phys Med Rehabil 1999;80:1101–1105. 10.1016/S0003-9993(99)90068-0 [DOI] [PubMed] [Google Scholar]
- 2.Wolf C,Meiners TH: Dysphagia in patients with acute cervical spinal cord injury. Spinal Cord 2003;41:347–353. 10.1038/sj.sc.3101440 [DOI] [PubMed] [Google Scholar]
- 3.Abel R,Ruf S,Spahn B: Cervical spinal cord injury and deglutition disorders. Dysphagia 2004;19:87–94. 10.1007/s00455-003-0511-y [DOI] [PubMed] [Google Scholar]
- 4.Brady S,Miserendino R,Statkus D,Miserendino R,Statkus D,Springer T,Hakel M,Stambolis V: Predictors to dysphagia and recovery after cervical spinal cord injury during acute rehabilitation. J Appl Res 2004;4:1–11. [Google Scholar]
- 5.Seidl RO,Nusser-Müller-Busch R,Kurzweil M,Niedeggen A: Dysphagia in acute tetraplegics: a retrospective study. Spinal Cord 2010;48:197–201. 10.1038/sc.2009.102 [DOI] [PubMed] [Google Scholar]
- 6.Shin JC,Yoo JH,Lee YS,Goo HR,Kim DH: Dysphagia in cervical spinal cord injury. Spinal Cord 2011;49:1008–1013. 10.1038/sc.2011.34 [DOI] [PubMed] [Google Scholar]
- 7.Shem K,Castillo K,Wong S,Chang J: Dysphagia in individuals with tetraplegia: incidence and risk factors. J Spinal Cord Med 2011;34:85–92. 10.1179/107902610X12911165974981 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chaw E,Shem K,Castillo K,Wong S,Chang J: Dysphagia and associated respiratory considerations in cervical spinal cord injury. Top Spinal Cord Inj Rehabil 2012;18:291–299. 10.1310/sci1804-291 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shem KL,Castillo K,Wong SL,Chang J,Kao MC,Kolakowsky-Hayner SA: Diagnostic accuracy of bedside swallow evaluation versus videofluoroscopy to assess dysphagia in individuals with tetraplegia. PM R 2012;4:283–289. 10.1016/j.pmrj.2012.01.002 [DOI] [PubMed] [Google Scholar]
- 10.Hayashi T,Fujiwara Y,Sakai H,Maeda T,Ueta T,Shiba K: Risk factors for severe dysphagia in acute cervical spinal cord injury. Spinal Cord 2017;55:940–943. 10.1038/sc.2017.63 [DOI] [PubMed] [Google Scholar]
- 11.Ihalainen T,Rinta-Kiikka I,Luoto TM,Thesleff T,Helminen M,Korpijaakko-Huuhka AM,Ronkainen A: Risk factors for laryngeal penetration-aspiration in patients with acute traumatic cervical spinal cord injury. Spine J 2018;18:81–87. 10.1016/j.spinee.2017.06.033 [DOI] [PubMed] [Google Scholar]
- 12.DeVita MA,Spierer-Rundback L: Swallowing disorders in patients with prolonged orotracheal intubation or tracheostomy tubes. Crit Care Med 1990;18:1328–1330. 10.1097/00003246-199012000-00004 [DOI] [PubMed] [Google Scholar]
- 13.Bonanno PC: Swallowing dysfunction after tracheostomy. Ann Surg 1971;174:29–33. 10.1097/00000658-197107010-00005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Betts RH: Post-tracheostomy aspiration. N Engl J Med 1965;273:155. 10.1056/NEJM196507152730309 [DOI] [PubMed] [Google Scholar]
- 15.Nash M: Swallowing problems in the tracheotomized patient. Otolaryngol Clin North Am 1988;21:701–709. [PubMed] [Google Scholar]
- 16.Elpern EH,Scott MG,Petro L,Ries MH: Pulmonary aspiration in mechanically ventilated patients with tracheostomies. Chest 1994;105:563–566. 10.1378/chest.105.2.563 [DOI] [PubMed] [Google Scholar]
- 17.Shaker R,Milbrath M,Ren J,Campbell B,Toohill R,Hogan W: Deglutitive aspiration in patients with tracheostomy: effect of tracheostomy on the duration of vocal cord closure. Gastroenterology 1995;108:1357–1360. 10.1016/0016-5085(95)90682-7 [DOI] [PubMed] [Google Scholar]
- 18.Sasaki CT,Suzuki M,Horiuchi M,Kirchner JA: The effect of tracheostomy on the laryngeal closure reflex. Laryngoscope 1977;87:1428–1440. 10.1288/00005537-197709000-00003 [DOI] [PubMed] [Google Scholar]
- 19.Leder SB,Ross DA: Investigation of the causal relationship between tracheotomy and aspiration in the acute care setting. Laryngoscope 2000;110:641–644. 10.1097/00005537-200004000-00019 [DOI] [PubMed] [Google Scholar]
- 20.Leder SB,Joe JK,Ross DA,Coelho DH,Mendes J: Presence of a tracheotomy tube and aspiration status in early, postsurgical head and neck cancer patients. Head Neck 2005;27:757–761. 10.1002/hed.20239 [DOI] [PubMed] [Google Scholar]
- 21.Leder SB,Ross DA: Confirmation of no causal relationship between tracheotomy and aspiration status: a direct replication study. Dysphagia 2010;25:35–39. 10.1007/s00455-009-9226-z [DOI] [PubMed] [Google Scholar]
- 22.Terk AR,Leder SB,Burrell MI: Hyoid bone and laryngeal movement dependent upon presence of a tracheotomy tube. Dysphagia 2007;22:89–93. 10.1007/s00455-006-9057-0 [DOI] [PubMed] [Google Scholar]
- 23.Kang JY,Choi KH,Yun GJ,Kim MY,Ryu JS: Does removal of tracheostomy affect dysphagia? A kinematic analysis. Dysphagia 2012;27:498–503. 10.1007/s00455-012-9396-y [DOI] [PubMed] [Google Scholar]
- 24.Maeda T,Ueta T,Mori E,Yugue I,Kawano O,Takao T,Sakai H,Okada S,Shiba K: Soft-tissue damage and segmental instability in adult patients with cervical spinal cord injury without major bone injury. Spine 2012;37:E1560–E1566. 10.1097/BRS.0b013e318272f345 [DOI] [PubMed] [Google Scholar]
- 25.Rojas CA,Vermess D,Bertozzi JC,Whitlow J,Guidi C,Martinez CR: Normal thickness and appearance of the prevertebral soft tissues on multidetector CT. AJNR Am J Neuroradiol 2009;30:136–141. 10.3174/ajnr.A1307 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Anderson KK,Arnold PM: Oropharyngeal dysphagia after anterior cervical spine surgery: a review. Global Spine J 2013;3:273–285. 10.1055/s-0033-1354253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Iruthayarajah J,McIntyre A,Mirkowski M,Welch-West P,Loh E,Teasell R: Risk factors for dysphagia after a spinal cord injury: a systematic review and meta-analysis. Spinal Cord 2018;56:1116–1123. 10.1038/s41393-018-0170-3 [DOI] [PubMed] [Google Scholar]
- 28.Kang SH,Kim DK,Seo KM,Kim K,Kim Y: Multi-level spinal fusion and postoperative prevertebral thickness increase the risk of dysphagia after anterior cervical spine surgery. J Clin Neurosci 2011;18:1369–1373. 10.1016/j.jocn.2011.02.033 [DOI] [PubMed] [Google Scholar]
- 29.Lee MJ,Bazaz R,Furey CG,Yoo J: Risk factors for dysphagia after anterior cervical spine surgery: a two-year prospective cohort study. Spine J 2007;7:141–147. 10.1016/j.spinee.2006.02.024 [DOI] [PubMed] [Google Scholar]
- 30.Kunieda K,Ohno T,Fujishima I,Hojo K,Morita T: Reliability and validity of a tool to measure the severity of dysphagia: The Food Intake LEVEL Scale. J Pain Symptom Manage 2013;46:201–206. 10.1016/j.jpainsymman.2012.07.020 [DOI] [PubMed] [Google Scholar]
- 31.Kanda Y: Investigation of the freely available easy-to-use software ‘EZR’ for medical statistics. Bone Marrow Transplant 2013;48:452–458. 10.1038/bmt.2012.244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.National Spinal Cord Injury Database. Website [in Japanese]. [cited 2019 Jun 12]. Available from: http://www.kibirihah.johas.go.jp/003_reha/10_sekison_db.html.
- 33.Shingu H,Ohama M,Ikata T,Katoh S,Akatsu T: A nationwide epidemiological survey of spinal cord injuries in Japan from January 1990 to December 1992. Paraplegia 1995;33:183–188. [DOI] [PubMed] [Google Scholar]
- 34.Sakai H,Ueta T,Shiba K: Epidemiological survey of spinal cord injury in Fukuoka prefecture [in Japanese] Bone Jt Nerve 2011;1:475–480. [Google Scholar]
- 35.Katoh S,Enishi T,Sato N,Sairyo K: High incidence of acute traumatic spinal cord injury in a rural population in Japan in 2011 and 2012: an epidemiological study. Spinal Cord 2014;52:264–267. 10.1038/sc.2014.13 [DOI] [PubMed] [Google Scholar]