Abstract
Inflammation, a common feature of many diseases, is an essential immune response that enables survival and maintains tissue homeostasis. However, in some conditions, the inflammatory process becomes detrimental, contributing to the pathogenesis of a disease. Targeting inflammation by using nanomedicines (i.e. nanoparticles loaded with a therapeutic active principle), either through the recognition of molecules overexpressed onto the surface of activated macrophages or endothelial cells, or through enhanced vasculature permeability, or even through biomimicry, offers a promising solution for the treatment of inflammatory diseases. After providing a brief insight on the pathophysiology of inflammation and current therapeutic strategies, the review will discuss, at a pre-clinical stage, the main innovative nanomedicine approaches that have been proposed in the past five years for the resolution of inflammatory disorders, finally focusing on those currently in clinical trials.
Keywords: Nanoparticles, Drug delivery systems, Inflammation, Biomimetic nanoparticles, Pre-clinical assessment, Clinical application
Abbreviations: 5-ASA, 5-AminoSalicylic Acid (also called Mesalazine); APCs, antigen-presenting cells; AMPS, 2-Acrylamido-2-MethylPropane-Sulfonic acid; ATP, Adenosine Triphosphate; bFGF, basic fibroblast growth factor; COVID-19, coronavirus disease 2019; DC, dendritic cells; Dex, Dexamethasone; EAE, experimental autoimmune encephalomyelitis; ELVIS, Extravasation through Leaky Vasculature and subsequent Inflammatory cell-mediated Sequestration; EPR, Enhanced Permeability and Retention; FDA, Food and Drug Administration; HA, Hyaluronic Acid; HIF, Hypoxia-Inducible transcription Factor; IBD, Inflammatory Bowel Diseases; ICAM, Inter-Cellular Adhesion Molecule; IFN-γ, Interferon gamma; IL-10, Interleukin 10; LMWH, Low Molecular Weight Heparin; LPS, LipoPolySaccharide; LXR, Liver X Receptor; MAPK, Mitogen Activated Protein Kinase; MPO, Myeloperoxidase; MPSS, MethylPrednisolone Sodium Succinate; NF-κB, Nuclear Factor-kappa B; NIPAM, N-Isopropyl acrylamide; NPs, Nanoparticles; NSAID, Non-Steroidal Anti-inflammatory Drug; pDNA, plasmid DeoxyriboNucleic Acid; PEG, Polyethylene Glycol; PEI, Polyethyleneimine; PLGA, Poly(Lactic-co-Glycolic Acid); PRR, Pattern-Recognition Receptor; PSL, PhosphatidylSerine-containing Liposome; ROS, Radical Oxygen Species; RA, Rheumatoid arthritis; TCR, T cell receptors; TLR, Toll-Like Receptor; TNF-α, Tumor Necrosis Factor alpha; VCAM, Vascular Cell Adhesion Molecule; VEGF, Vascular Endothelial Growth Factor
Graphical Abstract
1. Introduction
Inflammation represents a mechanism of the body’s defence, originating from multiple causes such as infectious agents (viruses, bacteria), physical agents, radical oxygen species (ROS), metabolic stress (hypoxia), to cite only few examples [1]. In some situations, the inflammatory process may, however, become detrimental. The COVID-19 infection, associated in certain cases with a strong cytokine storm, a viral sepsis and an uncontrolled systemic inflammation, leading to acute respiratory distress represents an example of such adverse reaction [2,3].
Historically, the first to define the clinical symptoms of inflammation was Celsus in the first century AD, who described four cardinal signs of inflammation as follows: “rubor et tumor cum calore et dolore” (redness and swelling with heat and pain). In 1858, Virchow added a fifth cardinal sign: “functio laesa” (disturbance of function) [1].
Depending on the origin of the agent, inflammation can be classified by whether it is caused by an external agent or an endogenous abnormal response. Based on its duration, inflammation can either be acute or chronic. Acute inflammation has been considered a defence of innate immune response induced by infection or injury [4], while chronic inflammation can accompany some pathological states without any infection or injury, like in obesity [5].
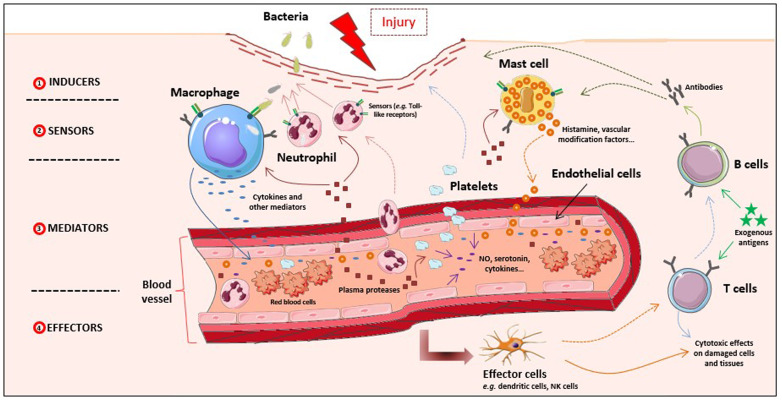
Recently, Antonelli and Kushner redefined inflammation as the innate immune response to potentially harmful stimuli such as pathogens, injury, and metabolic stress [6]. Indeed, proper inflammatory responses initiate a broad spectrum of innate immune protection and orchestrate long-term adaptive immunity toward specific pathogens. As such, the host immune system brings out layers of regulatory mechanisms that in return modulate the initiation, progression and resolution of inflammation [6,7]. Generally, the inflammatory response (Fig. 1 ) is perceived as a reaction of recognition of the infectious agent (inducers), a step which involves specialized (sensors) body cells as well as circulating proteins. After recognition, there is a production of mediators of inflammation which target the diseased tissue [6,8]. But both, innate and adaptive immune system, trigger the inflammatory process. Indeed, the components of the innate immunity include phagocytes (i.e., neutrophils, monocytes, macrophages), inflammation-related serum proteins (e.g., complement, C-reactive protein), cell receptors that signal a defensive response (e.g., Toll-like receptors) and cells that release cytokines and inflammatory mediators (i.e., macrophages, mast cells, natural-killer cells) [7]. The complement system not only acts as the first line of defense for the body, but also links innate and adaptive immunity and plays an important role in peripheral lymph nodes to enhance B and T cell responses. Activation of adaptative immunity occurs after innate immunity signaling and/or antigen presentation by specialized cells known as antigen-presenting cells (APCs), which include dendritic cells (DC) and macrophages. In contrast to the innate immune system, the adaptive immune system is highly specific against one or more antigens after their recognition by specialized receptors at the surface of B and T lymphocytes [9]. B cells are the main producers of antibodies which recognize and bind antigens. T cells are lymphocytes that express T cell receptors (TCR) on their surface and play essential roles in cell-mediated immunity. However, more and more data show that this distinction between innate immunity and adaptive immunity, does not correspond to reality since enormous interdependence between the two immune responses exists. For example, nitric oxide and reactive oxygen species (ROS) produced by macrophages, dendritic cells or other components of the innate immunity can modulate T cell function and survival [10].
Fig. 1.
Schematic representation of the inflammatory response.
Briefly, inducer signals (1) (e.g. wound, pathogens) trigger sensors (2) on inflammatory cells present in the damaged area, which start to induce the production and release of multiple mediators (3). Mediators, including plasma proteases, chemokines, cytokines, and vascular modification factors lead in turn to vascular modifications, recruitment of blood platelets and other inflammatory cells such as phagocytic leukocytes (e.g. neutrophils) and act on effector cells (4) and tissues to resolve inflammation. Adaptive immunity is also implicated in inflammation response, through lymphocytes B and T able to recognize and respond to antigens presented by antigen-presenting cells such as dendritic cells. Adaptive mechanisms may function either by direct cytotoxic effects or by secretion of antibodies interacting with elements of the innate inflammatory response (complement proteins, phagocytic cells...).
In most cases, the acute inflammatory response is controlled over time, and is normally finished once the insult is eliminated and the damaged tissue repaired. However, in some situations, when the inflammatory inducer persists, a chronic inflammatory response may establish. And when the inflammatory processes last for a long period of time, inflammation may become harmful and damaging for the tissues, which can in turn, contribute to the development and pathogenesis of chronic diseases of altered homeostasis. For example, lymphocyte T cells are thought to be the main triggers of autoimmune disease processes. In addition to innate immunity, adaptive mechanisms, through antigen recognition by the lymphocytes, production of antibodies, and their interaction with complement and phagocytic cells of the innate immunity such as macrophages, control both the nature and shape of the inflammatory responses. Thus, through this retro-activation of innate mechanisms, adaptive responses frequently provoke a persistent inflammation, which contributes to the process of chronic inflammation [7].
Inflammation is a very complex mechanism with, however, new advances in the understanding of its molecular mechanisms each year. Excellent reviews exist in this area describing the pathophysiological mechanisms [1,6,8]. It is now known that a large spectrum of diseases are accompanied by an uncontrolled inflammatory response, among these being sepsis, asthma, obesity and type 2 diabetes, neurodegenerative diseases, cardiovascular diseases (e.g. atherosclerosis, cardiac ischemia/ reperfusion), cancer, bowel disease, Crohn disease, rheumatoid polyarthritis, to cite only a few [11]. The understanding of the pathophysiology of the inflammation has allowed to identify proteins expressed by certain cells during inflammation (e.g. ICAM, VCAM, Selectins, ...). Additional phenomena such as the EPR (enhanced permeability and retention) effect have been identified. This effect, which is an interesting feature of inflamed tissues, leads to an increased vascular permeability coupled to a deregulated neovascularization [12]. EPR effect was first described in tumors were discontinuous endothelium in newly formed and immature vessels is formed. In inflammatory diseases, a similar effect was described with, however, some specificity. Thus, loss of endothelium integrity can result in abnormal neoangiogenesis observed in diseased tissues. The neovascularization mechanism is described to be induced by hypoxia and inflammation which are critical factors. Hypoxia promotes activation of hypoxia-inducible transcription factors (e.g. HIF-1, HIF-2) and subsequently expression of growth factor (e.g. endothelial growth factor (VEGF), basic fibroblast growth factor (bFGF)), finally leading to a loss of intercellular junctions between endothelial cells [12,13]. On the other hand, neutrophils, which are predominant in acute inflammation, promote vascular permeability by releasing neutrophil elastase, myeloperoxidase and MMP-9, which contribute to enhanced vascular permeability. Wang et al., referred this phenomenon as ELVIS effect (Extravasation through Leaky Vasculature and subsequent Inflammatory cell-mediated Sequestration) and use it to deliver polymer prodrugs [14,15]. Other studies have exploited the leaky vasculature during inflammation to deliver nanomedicines like PEGylated polycyanoacrylate [16] or liposomes [17] to the spinal cord of rats with experimental autoimmune encephalomyelitis (EAE).
Today, the therapeutic strategies include the use of steroidal or non-steroidal anti-inflammatory drugs. But two additional therapeutic approaches have recently emerged: the use of anti-leukotrienes that inhibit the recruitment of inflammatory cells and of pro-inflammatory cytokine inhibitors, like anti-TNF alpha or anti-IL-1 monoclonal antibodies. However, these treatments are sometimes not sufficient to obtain an optimal pharmacological activity. Drug limitations include non-specific biodistribution, low bioavailability and/or short half-life into the body. Moreover, high dosages are needed to be administered to patients, which cause off-target side effects with only modest success in controlling inflammatory disease symptoms [18].
To avoid some of these limitations, the use of nanoparticles (NPs), with a size from a few tenths to a few hundred of nanometers, has gained increasing interest. It is nowadays possible to obtain biocompatible nanomedicines (i.e. NPs loaded with active principle) with a highly controlled shape, size and surface charge [[19], [20], [21], [22]]. Moreover, a passive exploitation of this characteristic leaky vasculature has allowed an increased delivery and accumulation of nanomedicines through sub-endothelial space. Additionally, targeting moieties on nanoparticles’ surface may allow an active accumulation and a controlled drug release into the diseased cells and tissues, reducing toxicity and side-effects [[23], [24], [25]]. Therefore, many nanomedicines were developed with the aim to treat diseases with an inflammatory background, including cancer [26,27], cardiovascular pathologies [[28], [29], [30]], autoimmune diseases [31], metabolic syndrome [32], neurodegenerative diseases [22,33,34].
We have excluded in this review nanomedicines dedicated to cancer therapy where inflammatory processes occurs, because there are already excellent reviews on this subject [[35], [36], [37]]. Nevertheless, to the best of our knowledge, there is no recent review making the state of the art on the use of nanomedicines when inflammation in general becomes detrimental.
This review will focus on the new nanomedicine concepts that have emerged during the last five years concerning the management of inflammatory diseases at the pre-clinical stage. The last part deals with the very low number of nanomedicines in clinical trials.
2. Recent nanomedicines for the treatment of inflammation
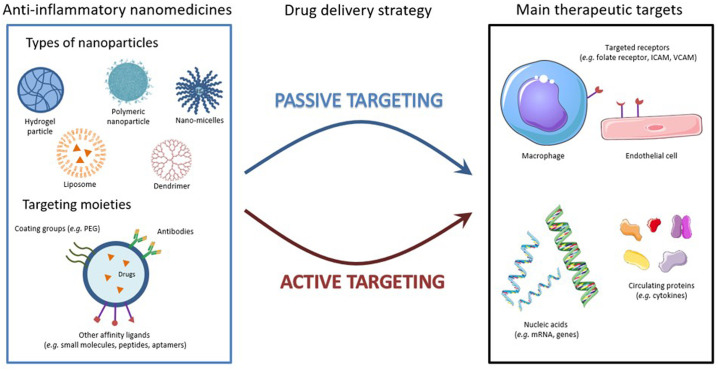
A better understanding of the molecular and cellular events underlying the inflammatory response has opened some new perspectives in the treatment of inflammation, particularly through the development of well-designed nanomedicines. Indeed, nanoparticles (NPs) can be specifically engineered to go preferentially to the target tissue from the site of administration, thus addressing issues of conventional therapies such as off-target organ side effects and systemic toxicity, exacerbated by frequent and long-term dosing. The formulation of nanoparticles regulating the expression of pro- and anti-inflammatory molecules and targeting inflammatory sensors or macrophages through phagocytosis, holds great promises for the treatment of inflammatory diseases (Fig. 2 , Table 1 ). In addition, using nanocarriers to specifically target effector cells, particularly antigen-presenting cells, could be of great value to promote cellular response or immune tolerance thanks to their modulability which allows them to passively (optimizing the size and/or surface charge of nanoparticles) or actively (decorating nanoparticles with specific antibodies) target these cells [38]. Considerable attention to develop more effective anti-inflammatory nanomedicines, in order to overcome the side effects observed with conventional therapy, has resulted in the development on anti-inflammatory nanomedicines.
Fig. 2.
Anti-inflammatory nanomedicines: strategies and targets.
Different types of nanoparticles were developed or are still in pre-clinical development for the management of inflammation. Among them liposomes, polymer nanoparticles, micelles, dendrimers, or hydrogel-based formulations. These nanoparticles could be naked or functionalized with targeting moieties, such as coating groups and antibodies or other affinity ligands. This allows them to target passively, through the leaky vasculature or actively the main actors of inflammation, including macrophages, endothelial cells, membrane receptors on inflammatory cells, anti-inflammatory genes and cytokines.
Table 1.
Some examples of new innovative nanomedicine systems for pre-clinical therapies against inflammatory disorders.
| Nanocarrier | Active principle | Inflammatory lesion | Animal model | Treatment time | Tested doses and route of administration | Main results | Ref |
|---|---|---|---|---|---|---|---|
| Eudragit S100 polymeric NPs | Prednisolone | Healthy | Wistar Albino rats | Every 30mins, from 1h to 6h | 5 mg/kg of NPs, oral administration |
|
[45] |
| Pegylated liposomes | Prednisolone | Renal ischemia and reperfusion injury | 8-week-old male LEW/HanHsd rats | 96h | 10 mg/kg of NPs, intravenous (i.v.) injection |
|
[47] |
| Hyaluronic acid-coated solid lipid NPs | Prednisolone | Collagen-antibody induced model of arthritis in Freud’s adjuvant | 6-week-old male DBA/1 mice | 4 injections, once every three days | 15 mg/kg of equivalent prednisolone each time, intra-articular injections |
|
[170] |
| Nano-sized elastic niosomes | Prednisolone | Clove oil-induced severe ocular inflammation | Albino rabbits | Every 4h, 3 times a day, for 6 days | Equivalent to 500 μg of prednisolone (A or P) each time, ocular delivery |
|
[50] |
| Polycaprolactone NPs dispersed in fibrin glue-based gel system | Methyl-prednisolone sodium succinate | Acute spinal cord injury | Male Wistar Albino rats | 24h | Doses = N/A, topical and intraperitoneal administration |
|
[51] |
| Hyaluronic acid hydrogel-based hydroxyl-terminated polyamidoamine dendrimers | Dexamethasone | Corneal alkali burn model | 8-week-old Lewis rats | 24h, 72h, 7 days or 14 days | Equivalent to 1.76 mg /kg of Dex, subconjunctival injection |
|
[52] |
| Poly(ethylene glycol)-poly( εcaprolactone) polymer micelles | Dexamethasone | Adjuvant arthritis model by mycobacteria inoculation in Freund’s adjuvant | 6-8 weeks Wistar rats | 4 injections of Dex, on days 14, 16, 18, 20 after arthritis induction | 0.8 mg/kg of Dex, intravenous injection |
|
[53] |
| Spherical polymeric nanoconstructs | Dexamethasone | Dextran sodium sulfate-induced model of arthritis | 8-week-old female C57Bl/6J mice | At day 2 after lesion: 6 consecutive treatments distributed until 16 days | 5 mg/kg each time, i.v. injections |
|
[54] |
| Solid lipid NPs | Dexamethasone and Butyrate | Dextran sodium sulfate-induced model of arthritis | 8-week-old male BALB/c mice | Daily treatment started from day 6 after lesion, for 3 days | 0.1 mg/kg of dexamethasone and 4 mg/kg of Butyrate, oral administration |
|
[55] |
| N-(2-hydroxypropyl)methacrylamide (HPMA) copolymer NPs | Dexamethasone | Adjuvant-induced arthritis | Male Lewis rats | One injection, at day 14, 15, 16 or 17 after arthritis induction | 10 mg/kg of dexamethasone, intravenous injection |
|
[56] |
| Dual pH- and time-dependant polymeric NPs composed of Eudragit FS30D and Eudragit RS100 | Budesonide | Dextran sodium sulfate-induced model of colitis | 7-week-old mice | 2h, 6h, or 10h | 0.5 mg/kg of drug (0.168 mg/kg of budesonide), oral administration |
|
[58] |
| PLGA NPs | Budesonide | Oxazolone-mediated experimental colitis | 8 to 12-week-old BALB/c mice | 18h | 42 μg of NPs, oral administration |
|
[60] |
| Phosphatidyl serine:phosphatidyl choline (3:7 mol/mol) liposomes | Recombinant mouse IL-10 | 12 weeks High Fat Diet-induced obesity and atherosclerosis | C57Bl/6 mice | One treatment after 12 weeks of diet | 1 μg/mouse of equivalent rIL-10, intraperitoneal injection |
|
[69] |
| Biodegradable polyester polymeric NPs made with core polymer I, II and PLGA-PEG-ColIV | Recombinant mouse IL-10 | Zymosan A (0.2 mg/mouse)-induced peritonitis (1) and 12 weeks of Western Diet-induced atherosclerosis (2) | 8 to 10-week-old female C57Bl/6J (1) and 8 to 10-week-old male Ldlr-/- (2) | 4h treatment (1) and 1/week for 4 weeks (2) | 100 or 500 ng/mouse of equivalent rIL-10 (1) and 5 μg of rIL-10/ i.v. injection (2) |
|
[70] |
| Poly(NIPAm-co-AMPS) NPs | Anti-inflammatory cell penetrating peptide KAFAK | Osteoarthritis model induced by removal of native aggrecan of cartilage explants and IL1β treatment | 3-month-old bovine knee joints explants | One treatment after 2 days of culture | 50 μg NPs, treatment on cartilage explants |
|
[79] |
| NGPEGSS NPs system incorporating NIPAm, AMPS, BAC, and PEG polymers | Anti-inflammatory cell penetrating peptide KAFAK | Osteoarthritis model by removal of native aggrecan cartilage explants and IL1β treatment | 3-month-old bovine knee joint explants | One treatment after 2 days of culture | 50 μg NPs, treatment on cartilage explants |
|
[81] |
| Chitosan/poly(γ-glutamic acid) NPs | Diclofenac | LPS-induced inflammation | In vitro human macrophages | 24h, 48h | 0.7 mg/mL, in vitro treatment |
|
[64] |
| Mannose-modified trimethyl chitosan-cysteine NPs | TNF-α siRNA | Acute hepatic injury induced by LPS and D-GaIN | Male Sprague-Dawley rats | 2h | 20 μg or 50 μg of equivalent TNF-α siRNA/kg, oral administration |
|
[82] |
| Nanoemulsion formulation of cationic lipid DOTAP | TNF-α siRNA | LPS intranigral injection-induced Parkinson’s disease model | Male Sprague-Dawley rats | 6h, 24h | 1.5 mg/kg of NPs, intranasal administration |
|
[115] |
| Poly(ethylene glycol)-poly(caprolactone) polymer micelles conjugated with Tat cell-penetrating peptide and siRNA targeting TNF-α | TNF-α siRNA | transient middle cerebral artery occlusion model of cerebral ischemia-reperfusion injury | 10-week-old Sprague-Dawley male rats | At 30 min after middle cerebral artery occlusion | Equivalent of 30 μg of siRNA, intranasal administration |
|
[84] |
| Dendrimers functionalized with cationic pyrrolidinium or morpholinium surface groups | TNF-α siRNA | LPS-induced acute lung inflammation model by intranasal administration of LPS | 6 to 8-week-old female CD-1 mice | 28h, 96h | Pre-treatment 24h before inflammation, with 2 mg/kg of eq. siRNA concentration, intranasal administration |
|
[85] |
| Modified chitosan nano-carrier deploying folic acid, diethylethylamine and PEG groups | TNF-α siRNA | Collagen-antibody induced model of arthritis | 8 to 12-week-old female DBA/1 mice | Treatment at day 1, 3, 5, 7 after induction of inflammation and sacrifice at day 10 | 4 intraperitoneal injections at 50 μg of siRNA each |
|
[86] |
| Tuftsin/alginate NPs | IL-10 pDNA | Adjuvant arthritis model by inoculation with Mycobacterium butyricum in Freund’s adjuvant | Male Lewis rats | From 1h to 24h | Intraperitoneal injection at day 19 post-lesion of 3 mg of NPs |
|
[90] |
| Hyaluronic acid/ polyetyleneimine NPs | IL-10 and IL-4 pDNAs | Intraperitoneal injection of Brewer-thioglycollate medium to recruit peritoneal macrophages | 6 to 8-week-old C57Bl/6 mice | 48h | 2h post inflammation, intraperitoneal injection of 100 μg of equivalent pDNA |
|
[91] |
| Hyaluronic acid/chitosan NPs | Cytokine response modifier A pDNA | Anterior cruciate ligament transection model of osteoarthritis | 6 to 8-week-old Male Sprague-Dawley rats | 3 injections, every 4 weeks starting 4 weeks after lesion | 4 μg of NPs/rat each time, i.v. injections |
|
[48] |
| Mannose-functionalized dendrimeric NPs using polyamidoamine dendrimer | Liver X receptor (LXR) ligand T0901317 | 12 weeks of Western Diet to induce atherosclerosis | Ldlr-/- mice | Once per week for 4 weeks after the 12 weeks of diet | 200 μg of NPs by i.v. injection |
|
[108] |
| Biodegradable diblock PLGA-b-PEG copolymer NPs | Liver X receptor (LXR) ligand GW3965 | Zymosan A-induced peritonitis | Ldlr-/- mice | 5h treatment (1) or over 2 weeks (2) | 10 mg/kg of equivalent agonist 1h prior to Zymosan injection (1) or 6 injections of 10 mg/kg (2), i.v. injections |
|
[109] |
| Phospholipid-reconstituted ApoA-I peptide-derived synthetic HDL | Liver X receptor (LXR) ligand T0901317 | Atherogenic diet for 14 weeks | 8-week-old male ApoE-deficient mice | 3 times a week for 6 weeks | 30 mg/kg of NPs, which is equal to 1.5 mg/kg of ligands, i.v. injections |
|
[110] |
| Raspberry-like core/satellite NPs with methyl violagen-functionalized polymeric NPs (core) and azobenzene (corona) | IL-1Ra antagonist protein | Healthy | 8 to 10-week-old male Sprague-Dawley rats | One treatment | 5 μg of cy7-labelled NPs, intra-articular injection |
|
[77] |
| Oleic acid-incorporated liquid crystalline monoolein-based NPs | Tacrolimus (FK506) | Daily topical dose (1.5 mg) of Imiquimod on shaved back for 5 days to induce a skin inflammation (psoriasis) | 8 to 11-week-old BALB/c mice | Once a day, for 7 days after inflammation induction | 30 μg of equivalent Tacrolimus each time, topical administration |
|
[111] |
| Positively charged PEP−PEG−PBG polymer micelles | FK506 | Dry eye disease animal model by subcutaneous injection of scopolamine | 6-week-old C57BL/6 mice | 4 times a day for 5 days | Topical application of 30 μg of the formulation each time |
|
[112] |
| PEGylated liposomes | FK506 | Experimental autoimmune myocarditis model induced by immunization with porcine myosin | 7-week-old male Lewis rats | Treatment on day 14 and 17, sacrificed on day 21 | 0.035 mg/kg, 0.17 mg/kg or 0.35 mg/kg of equivalent drug per treatment, i.v. injection |
|
[113] |
| PEGylated liposomes | Cyclosporin A | Transient middle cerebral artery occlusion by 90 min brain ischemia induction, followed by 48h reperfusion | Male Wistar rats | 5min after ischemia, for 48h | 2.5 mg/kg of equivalent cyclosporin A, i.v. injection |
|
[114] |
| Omega-3 fatty acid rich flaxseed oil-based nanoemulsion system | Cyclosporin A | LPS model of neuroinflammation | Sprague-Dawley rats | 9h | Pre-treatment 3h prior to inflammation, with 5 mg/kg of NPs, intranasal administration |
|
[115] |
| Silicon dioxide NPs | Mesalazine | 5% dextran sodium sulphate-induced ulcerative colitis model | 8 to 9-week-old male BALB/c mice | Once a day, for 7 days, sacrifice on day 8 |
100 mg/kg per day of NPs, oral administration |
|
[116] |
| Squalene-adenosine NPs | Tocopherol (VitE) | Endotoxemia model of inflammation induced by intraperitoneal injection of LPS | 8 to 12-week-old Male C57BL/6 and female BALB/c mice | One treatment, 30 min after LPS injection | 15 mg/kg SQAd NPs (5.5 mg/kg of equivalent adenosine) and 15 mg/kg VitE, intravenous injection |
|
[129] |
2.1. Steroid-based nanomedicines
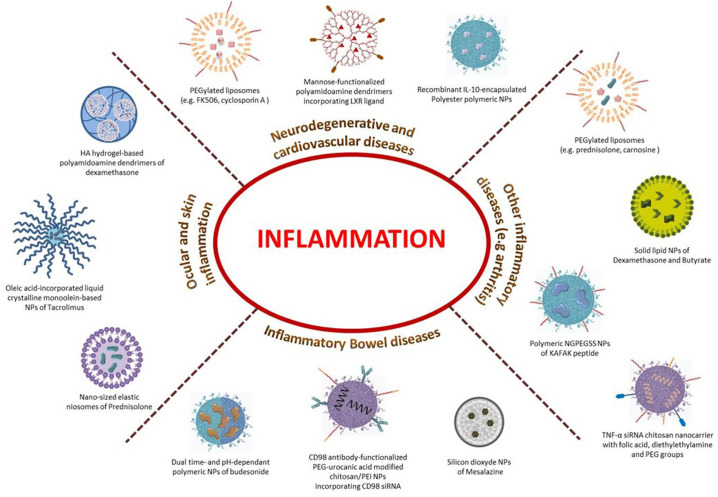
Corticosteroids are a class of steroid hormones involved in a wide range of physiological processes, including stress response, immune response and regulation of inflammation. Particularly, glucocorticoids are potent anti-inflammatory corticosteroids, regardless of the inflammation's cause, whose effect is mainly through a lipocortin-1 synthesis mechanism [39,40]. Some examples of the mostly used glucocorticoids for the treatment of inflammation are hydrocortisone and derivatives, prednisolone, dexamethasone or budesonide. Their therapeutic strength is extensively recognized, but they inexorably exhibit severe side effects after few weeks of treatment (such as weight gain, high blood pressure, high blood sugar, or even cataracts, bone loss, and muscle weakness) [41,42] limiting their clinical use. Thus, various nano-formulations of corticosteroids [43] were proposed to overcome those limitations (Fig. 3 , Table 1).
Fig. 3.
Examples of anti-inflammatory diseases and their innovative nanomedicines.
A large spectrum of materials and different strategies are developed for the treatment of inflammatory diseases.
Prednisolone is one of the most popular corticosteroids used for encapsulation into nanocarriers. For example, prednisolone nanocapsules, composed of a polymer wall surrounding an oily core, and coated with a pH-responsive enteric polymer (Eudragit S100), were designed and tested in healthy rats. This oral system allowed a high encapsulation efficiency of ca. 90% and a drug release triggered into the colon, as a consequence of neutral pH. This should increase the selectivity and efficacy of prednisolone for the treatment of inflammatory bowel diseases (IBD) or other colonic diseases [44].
Similarly, for oral administration, spherical polymer microsponges with a size of around 30μm, but with nano-porous microstructures, were also loaded with prednisolone and coated with Eudragit S100. The microsponges structure facilitated plastic deformation which was needed for the formation of mechanically strong tablets applicable for clinics. In addition, these microsponges displayed a high drug loading (around 86%), allowing dose and dose frequency reduction which should decrease prednisolone side effects and have some interest for the treatment of IBD [45]. However, the proof of concept was only validated in vitro, while in vivo studies are still lacking to validate this approach.
Another interesting strategy, based on orally or/and rectally administered lectin-decorated glucocorticoid loaded nanoparticles, was developed for an active targeting and selective adhesion to the inflamed tissue in vitro in experimental colitis. This strategy seems to be a promising tool for treatment of inflammatory bowel diseases [46]. Other recent pre-clinical studies have further demonstrated the therapeutic added value of prednisolone nano-formulations. For example, prednisolone loaded liposomes were assessed in a 8-week-old male LEWIS rat model of renal ischaemia-reperfusion injury, and liposomes were found to accumulate in the inflamed kidney after systemic delivery. The treatment increased the presence of anti-inflammatory macrophages and reduced monocyte chemoattractant protein-1 mRNA production, concomitant to a reduced pro-inflammatory profile in the kidney [47]. Although leading to very interesting data, it is important to note that the experiments were performed with young age animals and the final point of analysis remained quite short (i.e. 96 h). And young animals respond more efficiently than older ones, whereas, in the daily medical practice elderly people represent the population of patients the more affected by inflammatory diseases.
Rheumatoid arthritis (RA) is a chronic, systemic inflammatory disease treated with high doses of glucocorticoids. To avoid non-specific body biodistribution, solid-lipid nanoparticles were coated with hyaluronic acid in order to target hyaluronic receptor CD44 over-expressed on the surface of synovial lymphocytes, macrophages and fibroblasts in inflamed joints in RA. After intravenous injection into 6-week-old mice with collagen-induced arthritis, a significant improvement of the disease has been obtained, likely due to the specific accumulation of NPs into the inflamed tissue [48]. Folate-targeted double liposomes (i.e. small unilamellar vesicles encapsulated into a big liposome) containing two therapeutic agents (Prednisolone and Methotrexate) were also developed and assessed by systemic administration in collagen-induced arthritis in rats. These liposomes displayed high stability, enhanced loading capability and further enhanced the site-specific drug delivery to activated macrophages at inflamed joints in RA [49]. Gaafar et al. designed elastic niosome (“ethoniosomes”) nanoparticles loaded also with prednisolone for ocular delivery. The ethoniosomes composed of Span60 and cholesterol and using ethanol 20% v/v as a hydrating solution to the surfactant/lipid materials were tested in rabbits subjected to a clove oil-induced severe ocular inflammation. A reduction of half of the time for complete healing was observed, in addition to a reduced intraocular pressure, one of the main side effects observed with conventional anti-inflammatory treatments. Besides, this system demonstrated good physical stability for at least 2 months at the fridge temperature with good ocular tolerability and minimal ocular irritation, suggesting good potential for translation into the clinic [50].
Methylprednisolone, already used in clinics for spinal cord injury treatment, is unfortunately accompanied by dose-related side effects due to systemic injection. The loading of methylprednisolone sodium succinate (MPSS) into polycaprolactone NPs dispersed in a fibrin glue-based gel system was tested in a rat model of induced spinal cord injury either by topical or intraperitoneal administration. The localized delivery of MPSS onto the lesion site enabled a reduced damage on spinal cord [51]. This therapeutic effect was explained by a dramatic decrease of caspase-3 and a moderate decrease in pro-inflammatory cytokines.
Dexamethasone (Dex) is another glucocorticoid drug with anti-inflammatory activity. A recent study demonstrates that dendrimer-Dex gel attenuated corneal inflammation more efficiently than free-Dex, by decreasing macrophage infiltration and pro-inflammatory cytokines expression, in a rat corneal inflammation model. The designed system is a biocompatible hydrogel conjugated to Dex and based on hydroxyl-terminated polyamidoamine dendrimers and hyaluronic acid, cross-linked via thiolene click chemistry for subconjunctival injection [52]. Noteworthy, a single subconjunctival injection of this nano-formulation led to a prolonged efficacy for a period of 2 weeks with a reduced central corneal thickness and an improved corneal clarity, without elevation of the intraocular pressure. Moreover, the subconjunctival route is clinically accessible and can allow the administration of a relatively large volume of nanomedicine. Dexamethasone nanomedicines could also be very efficient for the treatment of rheumatoid arthritis or ulcerative colitis, bypassing the serious adverse effects associated with important doses of drug free. Thus, Wang et al. developed self -assembled poly(ethylene glycol) / poly(εcaprolactone) polymer micelles loaded with Dex for intravenous injection at low dose of Dex (0.8 mg/kg body weight) in a rat model of arthritis. Although displaying moderate adverse effects, these polymeric micelles demonstrated substantial anti-inflammatory potential, including suppression of paw swelling and erythema, down-regulation of pro-inflammatory cytokines expression and protection of articular cartilage and bone from degradation and erosion [53].
Spherical polymer nanoconstructs loaded with Dex were also intravenously injected to a mouse model of colitis. A strong systemic anti-inflammatory effect was observed through a reduced macrophage infiltration and the expression of pro-inflammatory cytokines. These cellular effects were accompanied by a better animal recovery manifested by a reduced weight loss and a decreased rectal bleeding. These benefits were less evident in animals treated with the drug free [54]. Furthermore, using the same model, Dianzani et al. assessed the efficacy of orally administered multi-drug nanoparticles co-loaded with Dexamethasone and Butyrate. Both in vitro and in vivo, the treatment induced a significant decrease of pro-inflammatory cytokine release, which was more effective at doses 10-fold lower than the dose required to achieve the similar pharmacological efficacy with the single free drugs treatments [55]. If delivering simultaneously multiple drugs may be interesting, further research is, however, necessary to better understand the exact mechanism underlying the enhanced anti-inflammatory effect of this multidrug nano-formulation. Very recently, by using an interesting approach, Wang’s team optimized a prodrug composed of N-(2-hydroxypropyl) methacrylamide (HPMA) copolymer linked to dexamethasone for the treatment of inflammatory arthritis. By intravenous injection in a rat model of arthritis, they observed very effective and safe therapeutic anti-inflammatory effects compared to other prodrug formulations. This attractive study promoted the importance of identifying the most potent therapeutic nanomedicines by evaluating the design and screening of activation mechanism [56].
Budesonide is a topical anti-inflammatory synthetic steroid, which was formulated for oral administration as first-line therapy in mild to moderate ileocecal Crohn’s disease patients [57]. In order to minimize early drug release and activity in the stomach and small intestine, Naeem et al. encapsulated Budesonide into dual pH- and time-dependent polymer nanoparticles composed of Eudragit FS30D, as a pH-sensitive polymer, and Eudragit RS100, as a controlled-release polymer. It was shown that in a mouse model of colitis, dual pH/time-dependent systems had greater therapeutic potential than single Eudragit FS30D or single Eudragit RS100 system. This beneficial effect was explained by the release of Budesonide at the right place (i.e. colon) during the right time (i.e. 24h) [58]. The use of Eudragit with various functionalities, either pH-sensitive or for controlled release, opens interesting perspective for clinical applications, particularly for colon-targeting treatments such as in IBD. Thus, nanotechnology has been used in oral dosage formulation design as strategies to further enhance uptake into diseased tissue within the colon [59]
Other nano-formulations to target and decrease inflammation are in development. For example, Budesonide loaded into Hyaluronic Acid (HA) or poly(lactic-co-glycolic acid) (PLGA) nanoparticles exhibited higher anti-inflammatory effects than the free drug in vitro on human colon carcinoma Caco-2 cell line and on inflamed intestinal mucosa in mice respectively [60,61].
Together these preclinical results underline the feasibility to improve the delivery of anti-inflammatory drugs in vivo. However, to be closer to the human diseases, the choice of the animal model, the duration of the treatment and the end points deserve to be better thoughtful, as will be discussed below (see part 4).
2.2. Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) nanomedicines
Non-Steroidal Anti-Inflammatory Drugs (NSAIDs) act through the inhibition of cyclooxygenase proteins that metabolize arachidonic acid into prostaglandins [62]. Due to short half-life and high percentage of protein binding, high doses of NSAIDs are needed to be efficient, which in turn causes undesirable side effects, such as an increased risk of gastrointestinal and cardiovascular complications [62,63]. Some NSAIDs NPs have recently been in development in order to counteract these side effects but, for the moment, they are only investigated at an in vitro pre-clinical stage.
Gonçalves et al. have loaded Diclofenac, a NSAID that inhibits prostaglandin synthesis by reducing cyclooxygenase-1 and cyclooxygenase-2 with relative equivalence, into chitosan-poly(γ-glutamic acid) NPs. This approach did not show any cytotoxicity for human macrophages. NPs were rapidly phagocyted and activated these cells, also inhibiting prostaglandin E2 and IL-6 production [64]. More recently, Shang et al. designed a novel intestinal targeted drug delivery system for oral administration; this system that combines PEG950 and methacrylic acid (MAA)) hydrogel and Ibuprofen-loaded PLGA NPs, could site-specifically deliver the drug into alkaline environment at a sustained-release manner. Such process improved the bioavailability and decreased the side effects of Ibuprofen [65].
Celecoxib is a COX-2 inhibitor indicated for long-term treatment of rheumatoid arthritis. It is administered orally, but unfortunately, this molecule shows adverse systemic complications in patients. In rats with experimental arthritis, the topical application of a gel containing this anti-inflammatory drug into solid lipid nanoparticles was found more efficient in inhibiting the inflammatory process, compared to untreated or conventional gel treated animals [66].
2.3. Nano-encapsulated anti-inflammatory mediators
Anti-inflammatory mediators represent another approach for the treatment of inflammatory diseases. For instance, cytokine IL-10, one of the main anti-inflammatory cytokines possesses multiple effects in immuno-regulation and inflammation [67]. It mainly downregulates the expression of other pro- or anti-inflammatory cytokines, enhances immune B cell survival, proliferation, as well as, antibody production. Besides, IL-10 is implied in blocking NF-κB activity and is involved in the regulation of the JAK-STAT signalling pathway. But its clinical use is complicated by important cytokine-related side effects [68]. To overcome these limitations, new IL-10 anti-inflammatory nano-formulations were developed (Table 1).
A typical example is the development of nano-sized phosphatidylserine-containing liposomes (PSL) conjugated to IL-10. Toita et al. showed in a mouse model of obesity that intraperitoneal injection of IL-10-conjugated PSL had a better macrophage targeting ability and an enhanced anti-inflammatory effect than PSL alone, due to the synergistic anti-inflammatory effects of IL-10 and PSL. It was suggested that this association could be used as a macrophage-targeted therapy for inflammation-related diseases [69].
Another example is the development of biodegradable polyester NPs incorporating IL-10 for the targeted delivery of atherosclerotic plaques. Intravenous injection of these NPs significantly reduced acute inflammation in an atherosclerotic murine model and were shown to be more potent than IL-10 free. In addition, it was observed that plaque rupture was prevented by increasing the fibrous cap thickness, simultaneously to the decrease of the necrotic cores [70]. More recently, Baganizi et al. have bioconjugated recombinant IL-10 to poly(vinylpyrolidone)-coated silver NPs. These nanoparticles showed an improvement of the cytokine stability and a better anti-inflammatory effectiveness in mouse macrophages as manifested by a decrease in IL-6 and TNFα production [71]. However, this study was limited by the fact that silver NPs could not be delivered in vivo for inflammation treatment. Indeed, at the cellular level, toxicities have been reported, including reactive oxygen species generation, DNA damages and cytokine induction during in vitro studies [72,73]. In vivo, studies also indicated adverse effects on many organs of the circulatory, respiratory, central nervous, hepatic and dermal systems [74,75].
Other studies focused on the nano-formulation of antagonists of the pro-inflammatory cytokine IL-1 receptor. Particularly, self-assembled IL-1 receptor antagonist (IL-1Ra)-presenting nanoparticles were developed. Two examples of this kind of NPs are: IL-1Ra-poly(2-hydroxyethyl methacrylate)-pyridine NPs and IL-1Ra core/satellite NPs, based on methyl viologen-functionalized polymeric NPs as the core, and a trans-isomer of azobenzene modified IL-1Ra as the corona. The pharmacological efficacy of these nanodevices were investigated after local injection in a rat model of inflamed joints [76,77] and both IL-1Ra-presenting nanoparticles demonstrated interesting anti-inflammatory effects, mainly inhibiting the IL-1β-stimulated pro-inflammatory production.
2.4. Anti-inflammatory peptides nanomedicines
Anti-inflammatory peptides, which are small bioactive molecules containing up to 50 amino acids, are raising increasing interest [78]. For example, the anti-inflammatory peptide KAFAK, an inhibitor of mitogen-activated protein kinase-2, was assessed in a cartilage explant inflammation model. To increase the peptide’s therapeutic lifetime, KAFAK was first loaded into co-polymerized 2-acrylamido-2-methylpropane sulfonic acid (AMPS) and N-isopropylacrylamide (NIPAM) monomers, to form pH-dependant poly(NIPAM-co-AMPS) nanoparticles [79,80]. These NPs demonstrated the ability to selectively diffuse through the degraded cartilage explants before to release the peptide, allowing a significant reduction of pro-inflammatory cytokine production. However, the authors noted that the high affinity of KAFAK towards the nanoparticles core and the lack of core degradation resulted in the release of less than half of the loaded peptides, thus limiting the therapeutic applications of this approach [80]. Then, the same authors developed a more complex delivery system, based on poly(NIPAM) nanoparticles with degradable disulfide crosslinks (abbreviated as NGPEGSS), loaded with KAFAK peptides. The system relies on the co-polymerization of NIPAM and AMPS monomers, coated with polyethylene glycol (PEG) and using the degradable crosslinker bis(acryloyl)cystamine (BAC) for more effective release into the intracellular compartment [80,81]. Ex vivo, a preferential accumulation of the NPs was observed throughout the inflamed aggrecan-depleted cartilage explants compared to their healthy counterparts and, simultaneously, a significant reduction of IL-6 pro-inflammatory cytokine was measured. Moreover, these NPs were stable and released less than 10% of the loaded KAFAK over 96h at pH=7.4 in a non-reducing environment. Thus, their ability to diffuse into the damaged cartilage, maintaining the majority of their payload prior to cell uptake by chondrocytes and macrophages, combined with a release of the peptide into these cells, represented an improvement comparatively to the treatment with the free peptide or with the previously described poly(NIPAM-co-AMPS) NPs [81].
2.5. Nanomedicines for gene therapy
Another strategy to control the inflammation is based on the manipulation of genetic material. In recent years, significant progresses have been made to design gene delivery systems to obtain enhanced therapeutic efficacy with less off-target side effects. It is expected that targeted gene therapy could reduce the above-mentioned limitation by localizing the targeted gene at the site of the inflammation. Small interfering RNAs (siRNAs) were found to be a promising approach for silencing specific genes involved in inflammation. However, siRNA delivery into the cytosol of immune cells remains a challenge.
Many different mediators were considered for the development of gene nano-therapies (Table 1). For example, siRNA silencing proinflammatory cytokine tumor necrosis factor (TNFα) expression was entrapped into mannose-modified trimethyl chitosan-cysteine nanoparticles. In vitro, the NPs were internalized by rat peritoneal exudate cells, through endocytosis and micropinocytosis pathways. But more interestingly, in a rat model of acute hepatic injury, TNFα siRNA-loaded NPs orally administered induced an effective TNFα knockdown at a very low dose (i.e. 50 μg/kg) [82]. Along the same line, cationic lipid nanoemulsions were also proposed for the delivery of anti-TNFα siRNA to the brain. It was shown that after intranasal administration in a rat model of LPS-induced neuroinflammation, the nanoemulsion concentrated preferentially into the brain, inducing a local down-regulation of TNFα [83]. Similarly, poly(ethylene glycol)-poly(caprolactone) polymeric micelles conjugated with anti- TNFα siRNA were administered intranasally in a rat model of cerebral ischemia-reperfusion injury associated with neuroinflammation. The rats subjected to the micelles treatment exerted a decrease of TNFα production and a significant improvement of their neurological score, as compared with the treatment using naked anti- TNFα siRNA [84].
Very recently, intranasal administration of cationic phosphorus dendriplexes loaded with anti-TNFα siRNA were shown very effective in a murine acute lung inflammatory model. Indeed, the authors observed a specific targeting toward macrophages, resulting in a major inhibition of TNFα, as well as, a better anti-inflammatory regulation of other cytokines [85].
Since in inflammation sites there is an overexpression of folic acid receptors by macrophages, a modified chitosan nanocarrier has been constructed, deploying folic acid, diethylethylamine and PEG groups to deliver TNFα siRNA. A significant decrease of inflammation in a murine arthritis model, close to human disease, was observed in animal receiving intraperitoneal injections of these NPs [86]. Nevertheless, the observed results should be discussed since the effects were observed only after multiple days of treatment, whereas TNFα levels usually tend to decrease rapidly (within a day) after siRNA treatment.
In order to downregulate TNFα and reduce RA progression, Aldayel et al. designed PEGylated solid-lipid nanoparticles composed of lecithin and cholesterol and loaded with lyophilized TNF-α-siRNA. When this formulation was intravenously injected in a mouse model of collagen antibody-induced arthritis refractory to methotrexate therapy, the results obtained showed a good treatment efficacy, opening the hope for patients refractory to methotrexate [87]. Similar results have been obtained with lipid-polymer hybrid nanoparticles loaded with siRNA directed against TNFα. Intra-articular injection, in an experimental arthritis mouse model, showed that this nanotherapy could efficiently suppress the inflammatory process, this effect being obtained with a low TNFα-siRNA dose (i.e 1 μg) [88].
Gene therapy was also evaluated on a different aspect of inflammation, relying on the control of macrophage polarization. Indeed, M1 macrophages are pro-inflammatory and have a central role in host defence against infection, while M2 macrophages are associated with responses to anti-inflammatory reactions and tissue remodelling [89]. Therefore, re-polarization of macrophages from the M1 phenotypes to M2 phenotypes by plasmid DNA (pDNA), siRNA or miRNA, represents an exciting prospect for the treatment of inflammatory diseases. In this context, IL-10 pDNA was encapsulated into alginate-based nanoparticles decorated with tuftsin, a tetrapeptide located in the heavy chain of immunoglobulin G which actively targets macrophages [90]. After intraperitoneal administration, these NPs were found to concentrate in inflamed paws of arthritic rats, due to a continuous recruitment of macrophages in the inflamed tissues, and a sustained local and systemic IL-10 expression was observed. Another nano-formulation of IL-10 pDNA was designed, using hyaluronic acid-poly(ethyleneimine) (HA-PEI) NPs. In this study, IL-10 pDNA and IL-4 pDNA, who are both supposed to play a key role in the polarization of macrophages, were encapsulated into these NPs. It was found that the in vitro transfection occurred in peritoneal macrophages over-expressing CD44 receptor and increased M2 phenotype surface markers due to the expression of IL-4 and IL-10. Besides, the intraperitoneal administration of these NPs to LPS-induced inflamed mice also significantly increased the M2 markers expression, showed elevated levels of serum IL-10 and reduced inflammation [91]. When microRNA-223, which is a miRNA that has been proved to alter macrophage polarization and activation, was encapsulated in the same nanocarrier [92], similar re-polarization of macrophages and anti-inflammatory effects were observed in vitro [93].
Other similar approaches are described in Table 1. But the crucial message here is that if gene nanotherapy could be effective in pre-clinical rodent models, it remains urgent to validate the proof-of-concept in bigger animal models, closer to human diseases.
2.6. Biomimetic nanoparticles
When injected intravenously, most of the nanomedicines are rapidly captured and eliminated by the immune system if they are not PEGylated. Biomimetic nanoparticles are hybrid nanostructures in which the uppermost layer is similar to a cell membrane (e.g. erythrocytes, immune cells, cancer cells and platelets) [94,95]. In this category are included cell membrane-coated nanoparticles and liposomes engineered with cell membrane proteins. This strategy brings the biological characteristics of the source cells and leads to an exhibited prolonged circulation of the nanoparticles and an active targeting capability; moreover, these biomimetic nanoparticles are less likely to be recognized by the immune system since the membrane coating has the inherent characteristics of the mother cells. Excellent reviews have been published in this field [[96], [97], [98], [99]] and we cover here only the very recent publications.
Current clinical treatment of rheumatoid arthritis primarily targets the inflammatory response by using anti-cytokine biologics such as those inhibiting tumour necrosis factor alpha (TNF-α) and interleukin (IL-1). However, the response rate still remains unsatisfactory [100]. Since neutrophils play an important role in resolving inflammation and repairing tissues damage [101], in a very interesting approach Zhang et al., fused neutrophil membrane onto polymeric cores and injected these hybrid biomimetic nanoparticles into the knee joint in a mouse model of collagen-induced arthritis and in a human transgenic mouse model of arthritis. The results showed a significant therapeutic efficacy by reducing joint damage and suppressing overall arthritis severity [102]. Of note, the authors used a biodegradable polymer which is FDA approved (i.e PLGA), to prepare the nanoparticles core. In another strategy, macrophage membrane-coated polymeric nanoparticles have shown to bind and neutralize endotoxins and to inhibit systemic inflammatory response in a mice lipopolysaccharide (LPS) model of inflammation after intravenous injection [103].
In another study, biomimetic nanoparticles were synthesized using membrane proteins purified from activated J774 macrophages and loaded with rapamycine. In a model of ApoE-/- mice with atherosclerotic plaques, it was observed that systemic injection of these hybrid NPs was able to suppress cell proliferation within the aorta [104].
Other strategies have been developed such as leukocyte-mimetic liposomes with the ability to translocate through inflamed endothelial cell layers [105]. Macrophage-derived microvesicles-coated PLGA nanoparticles were able to target rheumatoid arthritis (RA) after intravenous injection, displaying significant enhancement of the therapeutic efficacy in a collagen-induced arthritis mouse model. Similarly, macrophage-coated nanoparticles encapsulating tacrolimus significantly suppressed the progression of RA in mice [106].
Together these studies suggest that hybrid biomimetic nanomedicines represent a new and promising approach, since they possess unique biodistribution characteristics exhibited by different cell types used and, flexible designs derived from adaptable nanoparticle cores. However, some important concerns remain including possible immune reactions and complement activation after their administration. Besides, the scaling-up and the sterilisation of these biomimetic nanodevices also require specific developments.
2.7. Other applications of nanoparticles for immune system modulation
Strategies to activate or inhibit receptors implied in the inflammatory response are also potential ways to reduce inflammation (Table 1, Fig. 3). One example is the liver X receptors (LXRs), whose activation in macrophages is atheroprotective and suppresses inflammation [107]. Thus, some recent studies focused on the formulation of LXR agonists in macrophage-targeted nanoparticles, such as mannose functionalized dendrimeric NPs [108], biodegradable diblock PLGA-b-PEG copolymer NPs [109], or synthetic high density lipoprotein NPs [110]. Intravenous injection of all these different nano-formulations were found to increase the expression of LXR target genes, to exert anti-inflammatory effects and to inhibit the development and necrosis of plaques in LDLr-deficient and ApoE-deficient mouse models of atherosclerosis. Particularly, these nano-formulations allowed an enhanced cholesterol efflux [[108], [109], [110]], a reduction of CD68-positive macrophage content of plaques by 50%[109] and an up-regulation of expression of ATP-binding cassette transporters [110].
Nanomedicines with immunosuppressants are also in development for improving the pharmacological efficacy and reducing the adverse events of these molecules. Topical administration of tacrolimus (FK506) loaded onto monolein nanoparticles were tested in a psoriasis-like skin inflammation in mice, and appeared to be more efficient than tacrolimus free dissolved in propylene glycol [111]. Positively charged Tacrolimus-loaded nanomicelles were found to prolong eye surface retention, to enhance corneal permeability, to exert anti-apoptotic effects on the corneal epithelium and overall to suppress expression of inflammatory related factors in a dry eye disease rabbit model [112]. Besides, PEGylated liposomes loaded with the immunosuppressive drug FK506, were found to inhibit the expression of cytokines, such as interferon-γ and tumor necrosis factor-α, and reduced inflammation and fibrosis of the myocardium in a rat experimental autoimmune myocarditis after systemic administration [113]. Liposomal nano-formulations were also used to encapsulate cyclosporine A, another immunosuppressive drug. For example, intravenous administration of liposomal-cyclosporine A could improve cerebral ischemia and reperfusion injuries like neuroinflammation in a rat model, by inhibiting the inflammatory responses including MPO activity and TNFα level [114]. A further study on neuroinflammation in rats also proved the anti-inflammatory efficacy of cyclosporine-A using a cationic oil-in-water nanoemulsion upon intranasal administration [115].
Studies on mesalazine (5-ASA), an aminosalicylate anti-inflammatory drug mainly used in inflammatory bowel disease, focused in ameliorating the contact between the drug and the inflamed tissues by increasing adhesion and retention time. Practically, 5-ASA loaded onto silicon dioxide nanoparticles were tested in a dextran sodium sulfate-induced mouse model of ulcerative colitis. A significant decrease of IL-6, TNF-α and MPO was noted after oral administration of these NPs [116]. Along the same line, a very recent study designed an inclusion complex of 5-ASA in hydroxypropyl-β-cyclodextrin loaded onto chitosan NPs. This delivery system exhibited a sustained-release profile, and the NPs were more effective than the free drug in inhibiting the secretion of pro-inflammatory cytokine-stimulated colon cancer cells [117]. In another study, polymeric nanoparticles were rectally administered to protect low molecular weight heparin (LMWH) from intestinal degradation and to provide targeted delivery to inflamed tissue in experimental colitis mice. Such a combination shows a highly specific therapy by its protection against degradation in luminal environment and selective drug delivery inflamed intestinal tissue [118].
An unusual approach consisted in the design of prodrugs that will spontaneously self-assemble as nanoparticles in solution. This is what our group has developed through the construction of nanoparticles made of squalene, a natural and biocompatible lipid precursor of the cholesterol’s biosynthesis. Of note, this lipid is already used in clinic as vaccine adjuvant. Squalene has been employed as a building block for the bio-conjugation with different hydrophilic drug molecules, and demonstrated the ability to self-assemble in aqueous medium in the form of nanoparticles [119,120]. In general, these squalene-based nanoparticles were found to be non-cytotoxic, exhibited a high drug payload, an enhanced therapeutic response, and they were quite simple to prepare. Based on this concept, very recently, we discovered a new painkiller nanomedicine consisting in leu-enkephalin conjugated to squalene. And, the nanoparticles showed prolonged anti-pain effects on inflammatory pain in rats. Since they were found to target the inflamed area after intravenous administration and to act only on the peripheral opioid receptors, this nanomedicine opens the door to the treatment of intense pain without inducing tolerance and addiction, as observed with morphine and related synthetic opioids. The targeting of the inflamed area was found to occur by the EPR-like effect [121].
Squalene has also been conjugated to adenosine, a crucial regulatory autocrine and paracrine factor with anti-inflammatory properties [122,123]. Adenosine is physiologically presents at low levels in unstressed tissues, and is rapidly released from the intracellular space to the extracellular space in response to pathophysiological conditions, such as hypoxia, ischemia, inflammation or trauma, to generate various cellular responses that aim to restore tissue homeostasis [122,123]. Nevertheless, the use of adenosine in clinic is limited because of a very short plasma half-life and the need to administer high dosages which induce important side effects. Squalenoyl-adenosine nanoparticles may therefore represent a promising approach in the treatment of inflammatory disorders surpassing adenosine limitations. Intravenous administration of these nanoparticles already demonstrated a significant neuroprotective effect in experimental models of stoke in mice and spinal cord injury in a rats, where inflammation is part of the diseases [124].
Since inflammation is accompanied by the overproduction of radical oxygen species [125], molecules with antioxidant capacities have been formulated as nanoparticles in order to increase the targeting of inflamed tissues and improve drug bioavailability. For example lycopene, a carotenoid drug with antioxidant and anti-inflammatory activity [126] and glutathione [127] were both tested in animals with experimental rheumatoid arthritis. Wu et al. also developed andrographolide-loaded poly(ethylene glycol) and poly(propylene sulphide) micelles which showed to synchronically alleviate inflammation and oxidative stress in an ApoE-/- atherosclerotic mice model [128]. In addition, our group recently developed a novel antioxidant/anti-inflammatory multidrug squalene-based nanoparticle formulation, allowing simultaneous delivery of adenosine and tocopherol in tandem to targeted sites by exploiting dysfunctions of the endothelial barrier at the sites of acute inflammation. These nanoparticles demonstrated promising anti-inflammatory and protective effects in mice models of endotoxemia and lethal systemic shock, with potential to treat uncontrolled inflammation associated with many diseases, including severe COVID-19 [129].
Another interesting example is represented by metallic nanoparticles, the archetype being gold nanoparticles. The use of gold for therapeutic purposes dates back to Antiquity in China and Egypt, but only recently the scientific community developed a strong interest in gold nanoparticles for biomedical applications, especially because they can be easily functionalised and possess imaging and therapeutic capabilities [130]. The anti-inflammatory property of nanogold was recently explained by the down regulation of inflammatory mediators like TNF-α, IL-1β, COX-2 and transcription factor NF-kB (Nuclear factor-kB) [131]. Although systemic administration of these nanoparticles produced a significant anti-inflammatory activity in vivo, on different mouse models [132,133], it has to be noted that gold, even if well tolerated by the body, is not a biodegradable material and may remain for a long time into the body, as previously demonstrated in a mouse model [134].
Apart from being used as drug delivery platforms, NPs have also been engineered to modulate the immune system. The induction of antigen-specific adaptive immunity exclusively occurs in lymphoid tissues, where T and B cells meet antigen or antigen-loaded APC (dendritic cells, macrophages). Since dendritic cells can activate T cells, vaccination protocols targeting DCs are promising approaches to enhance immune responses. When injected into the blood, nanoparticles interact most frequently with antigen-presenting cells (APCs). Among the APCs, dendritic cells (DCs), which are the most specialized, are capturing and processing antigens before migration to lymphoid tissues, allowing T or B cells activation. By this way, nanoparticles may allow modulating the immune response. For example, De Koker et al. engineered hybrid nanoparticles composed of silica- poly(methacrylic acid) polymer. When injected in vivo, these NPs target and accumulate in lymph nodes, suggesting a possible application for the lymphatic delivery of antigens and immune-modulation [135]. And Muller et al. designed a versatile nanoparticle vaccine platform to promote an antigen-specific humoral response. These NPs were able to sustain a prolonged antigen presentation to APCs and to produce a strong immune response, as compared to the free antigen. Moreover, the size, charge, and surface characteristics of the NPs were found to be key parameters for improving the lymphatic trafficking of the NPs and their subsequent uptake by APCs [136].
To overcome resistance or production of antibodies which may occur after treatment with Rituximab (anti-CD20), another interesting approach was developed by Kopeček et al. who developed polymer prodrugs to target B cells. This approach, initially tested for B-cell malignancy treatment [[137], [138], [139], [140]], was recently evaluated in a pre-clinical model of inflammatory rheumatoid arthritis (RA) [141]. The treatment, tested in a collagen-induced rheumatoid arthritis in mice, was composed of two conjugates: (i) a bispecific engager, Fab′-MORF1 (obtained by linking Fab′ fragment of CD20 mAb to the 3′ end of a morpholino oligonucleotide (MORF1) via a thioether bond) and (ii) a receptor crosslinking mediated by HPMA-based P-(MORF2). Compared to untreated mice, a delayed progress of RA was observed. And this new approach could open the way for the treatment of other autoimmune diseases.
However, this research area based on the modulation of the immune system has mainly been developed for cancer immunotherapy (see reviews [[142], [143], [144]]), while the studies reporting the modulation of immune system in other inflammatory diseases are less. In this sense, Gao et al., designed an innovative antibacterial vaccine. The authors used Escherichia coli as a model pathogen membrane and coated them onto small gold nanoparticles (AuNPs) with a diameter of 30 nm. When injected subcutaneously, the NPs induced rapid activation and maturation of dendritic cells in the lymph nodes of the vaccinated mice, with in addition, the generation of durable antibody responses. These results demonstrated that using natural bacterial membranes to coat synthetic nanoparticles holds great promise for antibacterial vaccines [145]. Autoimmune diseases are caused by antigenically complex immune responses of the adaptive and innate immune system against specific cells, tissues or organs. Type 1 diabetes is a T cell-dependent autoimmune disease that is characterized by the destruction of insulin-producing β cells in the pancreas. Yeste et al., engineered nanoparticles to deliver a tolerogenic molecule, for inducing a tolerogenic phenotype in DCs. A significant delay of diabetes onset was observed in nonobese diabetic mice receiving this immunomodulatory nanoformulation [146]. For the future, a deeper understanding on the NP-immune cell crosstalk could guide the development of nano-immunotherapeutic approaches with an increased therapeutic efficacy.
3. Clinical trials and commercial markets
Many nanomedicines, mainly liposomes, have entered clinical practice, and even more are being investigated in clinical trials for a wide variety of therapeutic and imaging indications. To date, only few tenths of nanomedicines are FDA approved and few hundreds of clinical trials are investigating new nanomedicines [147], cancer therapy being the main application. Recently, three other nanomedicines have been approved: Patisiran/ONPATTRO, VYXEOS, AND NBTXR3/Hensify [148]. However, to the best of our knowledge, very few anti-inflammatory nanoparticles have succeeded to reach clinical trials (Table 2 ).
Table 2.
Some examples of nanomedicines under clinical trials.
| Nanocarrier | Active principle | Disease | Patients and clinical phase | Main Results | Ref. |
|---|---|---|---|---|---|
| PEGylated liposomes | Prednisolone | Illio-femoral atherosclerosis | 14 patients, mean age 70 years (± 7 years) |
|
[150] |
| PEGylated liposomes | Prednisolone | Moderate to severe active ulcerative colitis | 18 patients (iv administration) |
|
[155] |
| Liposome | Cyclosporine (lipogel or cream) | Chronic plaque psoriasis | 38 patients (receiving cyclosporine lipogel or conventional cyclosporine cream or placebo |
|
[156] |
| Glutatione-PEGylated liposomes | Methyl prednisolone | – | Assessment of safety, pharmacokinetics and pharmacodynamics |
|
[171] |
| PLGA | Pitavastatine | Healthy volunteers | 40 patients, Phase I, iv administration |
|
[158] |
| Nanoemulsion (oil in water emulsion) | Tretinoin | Acnee vulgaris | 10 patients with mild to moderate acnee vulgaris lesions |
|
[159] |
| PEGylated silica -gold nanoshells | Photothermolysis (ultrasound 10 sec to 10 W/cm2) | Acnee vulgaris | 37 patients |
|
[172] |
Anti-inflammatory liposomal formulations are one of the most promising nanomedicines for the treatment of acute and chronic inflammation. For example, after intravenous administration, long-circulating prednisolone-containing liposomes were found to accumulate into macrophages of patients with symptomatic iliofemoral atherosclerosis, as a result of prolonged circulation half-life. Nevertheless, in contrast to the pre-clinical efficacy data obtained in rabbits [149], short-term treatment did not reduce arterial wall permeability or arterial wall inflammation in humans [[150], [151], [152]]. This may have resulted from an insufficient dose of prednisolone reaching the plaque; and although increasing the dose will likely resolve this issue, it would also introduce the risk of adverse effects. Another possible explanation may be a timing issue since, based on pre-clinical results, the authors evaluated the clinical results only after 10 days of treatment. But most studies evaluating the impact of anti-inflammatory agents in human cardiovascular diseases showed therapeutic efficacy only after several weeks of treatment [150]. Despite the lack of anti-inflammatory activity, the study proved for the first time that targeted delivery of nanomedicines to atherosclerotic lesions is feasible in humans. This could provide guidance for the development of future nano-formulations for the imaging or the treatment of atherosclerosis.
Interestingly, such prednisolone-encapsulated liposome formulations were also tested in phase II clinical trials for the treatment of arteriovenous fistula [153], graves ophthalmopathy, and ulcerative colitis [154]. Recently, the clinical study for the treatment of ulcerative colitis exhibited strong positive results in 70% of the patients, with 4 patients out of 13 in remission, and 7 out of 13 showing a reduction of the endoscopy sub-score [155]. In addition, a phase III clinical trial for the treatment of rheumatoid arthritis has started but no results are available for the moment.
A liposomal formulation of cyclosporine A in a lipogel was also developed for a topical application to treat mild to moderate stable plaque psoriasis and tested in an early-phase clinical trial [156]. The clinical performance of this liposomal gel was superior to that of cyclosporine incorporated in a conventional dermatological formulation at equal drug concentration. Pharmacokinetic studies and other safety evaluation protocols indicated the good tolerance of the liposomal formulation which has induced uniform reduction of plaque elevation [156]. Other liposomal formulations are currently in development, such as liposomal corticosteroids or liposomal dexamethasone, whose clinical translation has started very recently in patients with progressive multiple myeloma [157].
Pitavastatin is a statin known to have vasodilating properties and to modulate inflammatory response. To evaluate the safety, tolerability, and pharmacokinetics for intravenous administration of Pitavastatin-loaded PLGA nanoparticles (NK-104-NP), a phase I clinical trial was recently conducted in healthy Japanese male subjects. The study proved that NK-104-NPs exhibited dose-dependent pharmacokinetics and was well tolerated with no significant adverse effects in healthy volunteers [158].
In a very recent pilot clinical study, an oil-in-water nanoemulsion containing all trans- retinoic acid (Tretinoin), showed a good efficiency against acne after topical application [159]. Similarly, when subjected to focal thermolysis of sebaceous gland by near infrared laser, silica-gold nanoshells (150 nm) coated with PEG, were effective to provide a significant reduction in occurrence and appearance of inflammatory lesions [160].
In conclusion, very few nanomedicines specifically designed for the treatment of inflammation are nowadays assessed in clinical trials. In order to operate translation from the bench to the bedside, several experimental challenges need to be addressed. Key issues related to the clinical development of anti-inflammatory nanoparticles include biological challenges such as understanding the biology of specific diseases and the biological interaction of nanoparticles in patients, commercialization hurdles related to large-scale manufacturing, to cost-effectiveness in comparison to current therapies and to regulatory standards, biocompatibility and safety [161]. Reducing complexity in nanoparticle design and synthesis should, therefore, be one of the main goals to generate clinically translatable nano-sized therapeutics.
4. General discussion
In recent years, cellular and molecular advances have improved the comprehension of the inflammatory response, leading to the discovery of multiple targets for biological therapies against inflammatory diseases. In addition to a variety of safe and quite effective anti-inflammatory agents already available, such as aspirin and other nonsteroidal anti-inflammatory drugs, many biological agents have also been developed including antibodies and nucleic acids for cytokine inhibition or to reduce the extravasation of immune cells into the inflamed tissues [162]. In spite of promising progresses made in developing such treatments, the widespread distribution of the inflammatory signaling pathways in multiple cell types and tissues leads to important off-target side effects and to systemic toxicities. Combined with a relatively low bioavailability or/and limited half-lives, necessitating repeated and expensive high dosages responsible for severe side effects, current therapies still remain with clinical hurdles to overcome.
This has prompted the academic research to design nanocarriers in order to improve the delivery of anti-inflammatory compounds. After administration, such nanomedicines follow a kinetic process of absorption, distribution, metabolization and elimination. In addition, they distribute in a different way in the body depending if their surface is naked, decorated with PEG, or loaded with a ligand for an active targeting. In this way, numerous nanoparticles loaded with various anti-inflammatory molecules were developed and have shown real therapeutic improvements in pre-clinical models (Fig. 3).
Other than being drug delivery carriers, nanoparticles have also been used as vaccine adjuvant and for modulation of the immune system. Different data suggest that the shape of the NPs can affect the interactions with innate immune system. Once injected into the body the NPs immediately encounter the innate immune system and generate specific responses as foreign substances. Apart from the coating with poly (ethylene glycol) that delays the protein corona formation at their surface, the physicochemical properties of NPs (size, charge, hydrophobicity and stiffness) will also determine their interaction with the innate system.
For example, it was shown that the uptake of nanorods by macrophages was more efficient than the uptake of nanospheres [163]. And a comparison of the complement activation using spherical, rods and disks polystyrene NPs evidenced that rods and disks generated more important complement activation than spheres at later time points [164].
But today it remains still unclear which physical properties or chemical composition of nanoparticles can drive to immune modulation.
There is also an urgent need to develop new in vitro and in vivo pre-clinical models, better mimicking the pathophysiology of the disease in humans. For instance, the EPR effect has become during decades a general condition to allow the targeting of nanoparticles or liposomes through leaky vasculatures towards tumor or inflamed areas, in most of the experimental pre-clinical rodent models. But in the clinical reality, the occurrence of the EPR effect is far to be systematic. Various other parameters need to be taken into consideration, like the distribution and diffusion deep into the inflamed tissue (which is bigger in humans than in mice), the extend of drug release, the blood perfusion of the inflamed area, the nature of the extracellular matrix, etc. Thus, concerning the inflammatory processes, are the pre-clinical models really reliable? And to what extent the observed vascular permeability in pre-clinical models is transposable to human? Answering those questions and developing clinically relevant animal models is a key to allow a better selection of disease-driven nano-formulations [165].
Different administration ways, oral, local, topical or intravenous have been tested with anti-inflammatory nanomedicines. While intravenous delivery allows a direct access to the whole body, the local delivery proved to be effective, allowing to directly concentrate the drug into the diseased tissue. If the choice of administration route depends evidently on the localization of the inflammation site, it should also customize the choice of the materials and the design of the nanomedicines (presence or not of PEG or other ligands).
The route of administration such as intravenous or the direct injection into the inflamed site can influence the induction of the immune response. Thus, when NPs are administered intravenously, the protein corona formed at the surface of the NPs and the complement eventually activated can alter biodistribution and therapeutic efficacy. But when the NPs are administered locally, the immune response is mainly dependent on the tissue residential cells.
Chemical and physical properties of the NPs, including size, surface charge, and surface chemistry, are other important factors in the context of systemic inflammation. For example, it was observed that LPS-induced inflammation resulted in splenic macrophage polarization and altered leukocyte uptake of polystyrene nanoparticles depending on their size [166]. Concerning local administration, poly(lactic acid) particles need a size of at least 3 μm to guarantee the retention in inflamed joints [167].
Of note, as detailed in this review, different types of nanocarriers have been employed for the delivery of anti-inflammatory drugs, including liposomes, polymer nanoparticles, micelles or prodrugs. To compare their delivery abilities is quite hazardous because the experimental conditions are diverse. Liposomes are evidently the most advanced in terms of clinical translation and some liposomal formulations are even FDA approved. However, they generally display a lower stability in biological fluids than polymer nanoparticles or prodrugs, sometimes leading to a fast drug leakage. And their drug loading capacities are often limited. On the other hand, in case of intravenous administration, polymer micelles may disassemble due to a high critical micellar concentration which doesn’t occur with polymer nanoparticles.
But as shown in this review, the clinical translation of the nanoparticles remains still limited. This is due, in part, to the complexity related to the pharmaceutical development of nanomedicines, including their biocompatibility and safety. We also note that chronic inflammatory diseases bring together cofounding factors such as age, overweight and obesity and sometimes genetic predisposition to cite only a few. However, most of the preclinical studies to assess anti-inflammatory nanomedicines are performed on young and healthy animals, with an induced accelerated inflammatory disease and subject to normal fed. These factors should therefore be carefully considered in order to improve the translation from preclinical models to the clinic. In addition, the difference between rodent and human immune systems may represent an important obstacle for translating studies performed in mice into human clinical trials. This is the reason why there is an urgent need to develop pre-clinical experimental models with pathophysiological similarities closer to human inflammatory disease.
Bio-inspired nanodevices represent a new generation of innovative nanomedicines with the aim to mimic natural circulatory cells. They are able to increase the blood circulation time and to improve the distribution of the loaded drug towards the inflammatory cells and tissues. However, their immunogenicity, scale-up and characterization remain important hurdles before starting clinical trials.
Besides, problems related to the scaling-up, the government regulations and the overall cost-effectiveness if compared to the current available anti-inflammatory therapies are other important limitations in the success of the anti-inflammatory nanomedicines in general. The often complex architectural design of many nanomedicines described in this review, likely results in difficulties for performing reproducible sample preparations, safe and in good quantities [168]. And this is particularly important since a slight modification of the size, the shape and/or the nanoparticle surface chemistry may dramatically influence the stability, the interaction with biological media, as well as, the biodistribution and clearance of the nanocarriers. Thus, reliable and standardized methodologies to obtain reproducible nanoparticles are absolutely required [169]. There is often a gap between the academic environment where innovative nanocarriers are designed and the industrial context, where reproducible preparation and manufacturing processes supported by analytical methodologies are needed.
5. Conclusion
As shown in this review, there is an increasing interest to improve the delivery of small drug molecules or biotherapeutics (i.e. nucleic acids, peptides, proteins) for the resolution of inflammatory diseases. This represents a unique opportunity for pharmaceutically developable nanoscaled drug delivery systems, especially to optimize drug safety and efficacy through improved biodistribution and pharmacokinetics and to resolve the issues related to the difficulty to administer fragile biomolecules.
Author contributions
The manuscript was written with the contribution of all the authors.
Declaration of Competing Interest
The authors declare no competing interest.
Acknowledgments
The authors gratefully acknowledge the financial support from the 7th EuroNanoMed-II call for proposals, project NanoHeart n°ANR-16-ENM2-0005-01.
References
- 1.Chovatiya R., Medzhitov R. Stress, inflammation, and defense of homeostasis. Mol. Cell. 2014;54:281–288. doi: 10.1016/j.molcel.2014.03.030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hossam W.F.E., Ashour M., Rahman M. Masudur, Hatem A. Elshabrawy insights into the recent 2019 novel coronavirus (SARS-CoV-2) in light of past human coronavirus outbreaks. Pathogens. 2020;9 doi: 10.3390/pathogens9030186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.SA A.A. Meo, Al-Khlaiwi T., Meo I.M., Halepoto D.M., Iqbal M., Usmani A.M., Hajjar W., Ahmed N. Novel coronavirus 2019-nCoV: prevalence, biological and clinical characteristics comparison with SARS-CoV and MERS-CoV. Eur. Rev. Med. Pharmacol. Sci. 2012-2019;24(2020) doi: 10.26355/eurrev_202002_20379. [DOI] [PubMed] [Google Scholar]
- 4.Xiao T.S. Innate immunity and inflammation. Cell. Mol. Immunol. 2017;14:1–3. doi: 10.1038/cmi.2016.45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hotamisligil G.S. Endoplasmic reticulum stress and the inflammatory basis of metabolic disease. Cell. 2010;140:900–917. doi: 10.1016/j.cell.2010.02.034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Antonelli M., Kushner I. It's time to redefine inflammation. FASEB J. 2017;31:1787–1791. doi: 10.1096/fj.201601326R. [DOI] [PubMed] [Google Scholar]
- 7.Cronkite D.A., Strutt T.M. The regulation of inflammation by innate and adaptive lymphocytes. J Immunol Res. 2018;2018:1467538. doi: 10.1155/2018/1467538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Medzhitov R. Inflammation 2010: new adventures of an old flame. Cell. 2010;140:771–776. doi: 10.1016/j.cell.2010.03.006. [DOI] [PubMed] [Google Scholar]
- 9.Chaplin D.D. Overview of the immune response. J. Allergy Clin. Immunol. 2010;125:S3–23. doi: 10.1016/j.jaci.2009.12.980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Harrison D.G., Guzik T.J., Lob H.E., Madhur M.S., Marvar P.J., Thabet S.R., Vinh A., Weyand C.M. Inflammation, immunity, and hypertension. Hypertension. 2011;57:132–140. doi: 10.1161/HYPERTENSIONAHA.110.163576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Molinaro R., Boada C., Del Rosal G.M., Hartman K.A., Corbo C., Andrews E.D., Toledano-Furman N.E., Cooke J.P., Tasciotti E. Vascular inflammation: a novel access route for nanomedicine. Methodist Debakey Cardiovasc. J. 2016;12:169–174. doi: 10.14797/mdcj-12-3-169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Durymanov M., Kamaletdinova T., Lehmann S.E., Reineke J. Exploiting passive nanomedicine accumulation at sites of enhanced vascular permeability for non-cancerous applications. J. Control. Release. 2017;261:10–22. doi: 10.1016/j.jconrel.2017.06.013. [DOI] [PubMed] [Google Scholar]
- 13.Azzopardi E.A., Ferguson E.L., Thomas D.W. The enhanced permeability retention effect: a new paradigm for drug targeting in infection. J. Antimicrob. Chemother. 2013;68:257–274. doi: 10.1093/jac/dks379. [DOI] [PubMed] [Google Scholar]
- 14.Wang D., Goldring S.R. The bone, the joints and the Balm of Gilead. Mol. Pharm. 2011;8:991–993. doi: 10.1021/mp200328t. [DOI] [PubMed] [Google Scholar]
- 15.Yuan F., Quan L.D., Cui L., Goldring S.R., Wang D. Development of macromolecular prodrug for rheumatoid arthritis. Adv. Drug Deliv. Rev. 2012;64:1205–1219. doi: 10.1016/j.addr.2012.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Calvo P., Gouritin B., Villarroya H., Eclancher F., Giannavola C., Klein C., Andreux J.P., Couvreur P. Quantification and localization of PEGylated polycyanoacrylate nanoparticles in brain and spinal cord during experimental allergic encephalomyelitis in the rat. Eur. J. Neurosci. 2002;15:1317–1326. doi: 10.1046/j.1460-9568.2002.01967.x. [DOI] [PubMed] [Google Scholar]
- 17.Cavaletti G., Cassetti A., Canta A., Galbiati S., Gilardini A., Oggioni N., Rodriguez-Menendez V., Fasano A., Liuzzi G.M., Fattler U., Ries S., Nieland J., Riccio P., Haas H. Cationic liposomes target sites of acute neuroinflammation in experimental autoimmune encephalomyelitis. Mol. Pharm. 2009;6:1363–1370. doi: 10.1021/mp8001478. [DOI] [PubMed] [Google Scholar]
- 18.Tabas I., Glass C.K. Anti-inflammatory therapy in chronic disease: challenges and opportunities. Science. 2013;339:166–172. doi: 10.1126/science.1230720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Banerjee A., Qi J., Gogoi R., Wong J., Mitragotri S. Role of nanoparticle size, shape and surface chemistry in oral drug delivery. J. Control. Release. 2016;238:176–185. doi: 10.1016/j.jconrel.2016.07.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jindal A.B. The effect of particle shape on cellular interaction and drug delivery applications of micro- and nanoparticles. Int. J. Pharm. 2017;532:450–465. doi: 10.1016/j.ijpharm.2017.09.028. [DOI] [PubMed] [Google Scholar]
- 21.Jo D.H., Kim J.H., Lee T.G. Size, surface charge, and shape determine therapeutic effects of nanoparticles on brain and retinal diseases. Nanomedicine. 2015;11:1603–1611. doi: 10.1016/j.nano.2015.04.015. [DOI] [PubMed] [Google Scholar]
- 22.Agrahari V., Burnouf P.A., Burnouf T. Nanoformulation properties, characterization, and behavior in complex biological matrices: Challenges and opportunities for brain-targeted drug delivery applications and enhanced translational potential. Adv. Drug Deliv. Rev. 2019;148:146–180. doi: 10.1016/j.addr.2019.02.008. [DOI] [PubMed] [Google Scholar]
- 23.Duncan G.A., Bevan M.A. Computational design of nanoparticle drug delivery systems for selective targeting. Nanoscale. 2015;7:15332–15340. doi: 10.1039/c5nr03691g. [DOI] [PubMed] [Google Scholar]
- 24.Li H., Zhang X., Wang K., Liu H., Wei Y. Facile preparation of biocompatible and robust fluorescent polymeric nanoparticles via PEGylation and cross-linking. ACS Appl. Mater. Interfaces. 2015;7:4241–4246. doi: 10.1021/am5085308. [DOI] [PubMed] [Google Scholar]
- 25.Santos-Martinez M.J., Rahme K., Corbalan J.J., Faulkner C., Holmes J.D., Tajber L., Medina C., Radomski M.W. Pegylation increases platelet biocompatibility of gold nanoparticles. J. Biomed. Nanotechnol. 2014;10:1004–1015. doi: 10.1166/jbn.2014.1813. [DOI] [PubMed] [Google Scholar]
- 26.Iqbal J., Abbasi B.A., Ahmad R., Mahmood T., Ali B., Khalil A.T., Kanwal S., Shah S.A., Alam M.M., Badshah H., Munir A. Nanomedicines for developing cancer nanotherapeutics: from benchtop to bedside and beyond. Appl. Microbiol. Biotechnol. 2018;102:9449–9470. doi: 10.1007/s00253-018-9352-3. [DOI] [PubMed] [Google Scholar]
- 27.Tran S., DeGiovanni P.J., Piel B., Rai P. Cancer nanomedicine: a review of recent success in drug delivery. Clin. Transl. Med. 2017;6:44. doi: 10.1186/s40169-017-0175-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Kim K.S., Khang G., Lee D. Application of nanomedicine in cardiovascular diseases and stroke. Curr. Pharm. Des. 2011;17:1825–1833. doi: 10.2174/138161211796390967. [DOI] [PubMed] [Google Scholar]
- 29.Martin Gimenez V.M., Kassuha D.E., Manucha W. Nanomedicine applied to cardiovascular diseases: latest developments. Ther. Adv. Cardiovasc. Dis. 2017;11:133–142. doi: 10.1177/1753944717692293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dormont M.V.F., Couvreur P. Nanoplumbers: biomaterials to fight cardiovascular diseases. Mater. Today. 2018;21:122–143. [Google Scholar]
- 31.Gharagozloo M., Majewski S., Foldvari M. Therapeutic applications of nanomedicine in autoimmune diseases: from immunosuppression to tolerance induction. Nanomedicine. 2015;11:1003–1018. doi: 10.1016/j.nano.2014.12.003. [DOI] [PubMed] [Google Scholar]
- 32.Veiseh O., Tang B.C., Whitehead K.A., Anderson D.G., Langer R. Managing diabetes with nanomedicine: challenges and opportunities. Nat. Rev. Drug Discov. 2015;14:45–57. doi: 10.1038/nrd4477. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Goldsmith M., Abramovitz L., Peer D. Precision nanomedicine in neurodegenerative diseases. ACS Nano. 2014;8:1958–1965. doi: 10.1021/nn501292z. [DOI] [PubMed] [Google Scholar]
- 34.Kabanov A.V., Gendelman H.E. Nanomedicine in the diagnosis and therapy of neurodegenerative disorders. Prog. Polym. Sci. 2007;32:1054–1082. doi: 10.1016/j.progpolymsci.2007.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Molinaro R., Corbo C., Livingston M., Evangelopoulos M., Parodi A., Boada C., Agostini M., Tasciotti E. Inflammation and cancer: in medio stat nano. Curr. Med. Chem. 2018;25:4208–4223. doi: 10.2174/0929867324666170920160030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Salvioni L., Rizzuto M.A., Bertolini J.A., Pandolfi L., Colombo M., Prosperi D. Thirty years of cancer nanomedicine: success, frustration, and hope. Cancers (Basel) 2019;11 doi: 10.3390/cancers11121855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Irvine D.J., Dane E.L. Enhancing cancer immunotherapy with nanomedicine. Nat. Rev. Immunol. 2020;20:321–334. doi: 10.1038/s41577-019-0269-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Dacoba T.G., Olivera A., Torres D., Crecente-Campo J., Alonso M.J. Modulating the immune system through nanotechnology. Semin. Immunol. 2017;34:78–102. doi: 10.1016/j.smim.2017.09.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Barnes P.J. Glucocorticoids. Chem. Immunol. Allergy. 2014;100:311–316. doi: 10.1159/000359984. [DOI] [PubMed] [Google Scholar]
- 40.Oppong E., Cato A.C. Effects of Glucocorticoids in the Immune System. Adv. Exp. Med. Biol. 2015;872:217–233. doi: 10.1007/978-1-4939-2895-8_9. [DOI] [PubMed] [Google Scholar]
- 41.Hussain Ali E.-M.C., Windbergs Maike, Lehr Claus-Michael. Nanomedicines for the treatment of inflammatory bowel diseases. Eur. J. Nanomed. 2013;5:23–38. [Google Scholar]
- 42.Oray M., Abu Samra K., Ebrahimiadib N., Meese H., Foster C.S. Long-term side effects of glucocorticoids. Expert Opin. Drug Saf. 2016;15:457–465. doi: 10.1517/14740338.2016.1140743. [DOI] [PubMed] [Google Scholar]
- 43.Youshia J., Lamprecht A. Size-dependent nanoparticulate drug delivery in inflammatory bowel diseases. Expert Opin. Drug Deliv. 2016;13:281–294. doi: 10.1517/17425247.2016.1114604. [DOI] [PubMed] [Google Scholar]
- 44.Kshirsagar S.J., Bhalekar M.R., Patel J.N., Mohapatra S.K., Shewale N.S. Preparation and characterization of nanocapsules for colon-targeted drug delivery system. Pharm. Dev. Technol. 2012;17:607–613. doi: 10.3109/10837450.2011.557732. [DOI] [PubMed] [Google Scholar]
- 45.Kumari A., Jain A., Hurkat P., Tiwari A., Jain S.K. Eudragit S100 coated microsponges for Colon targeting of prednisolone. Drug Dev. Ind. Pharm. 2018;44:902–913. doi: 10.1080/03639045.2017.1420079. [DOI] [PubMed] [Google Scholar]
- 46.Moulari B., Beduneau A., Pellequer Y., Lamprecht A. Lectin-decorated nanoparticles enhance binding to the inflamed tissue in experimental colitis. J. Control. Release. 2014;188:9–17. doi: 10.1016/j.jconrel.2014.05.046. [DOI] [PubMed] [Google Scholar]
- 47.van Alem C.M.A., Boonstra M., Prins J., Bezhaeva T., van Essen M.F., Ruben J.M., Vahrmeijer A.L., van der Veer E.P., de Fijter J.W., Reinders M.E., Meijer O., Metselaar J.M., van Kooten C., Rotmans J.I. Local delivery of liposomal prednisolone leads to an anti-inflammatory profile in renal ischaemia-reperfusion injury in the rat. Nephrol. Dial. Transplant. 2018;33:44–53. doi: 10.1093/ndt/gfx204. [DOI] [PubMed] [Google Scholar]
- 48.Zhou P.H., Qiu B., Deng R.H., Li H.J., Xu X.F., Shang X.F. Chondroprotective effects of hyaluronic acid-chitosan nanoparticles containing plasmid DNA encoding cytokine response modifier A in a rat knee osteoarthritis model. Cell. Physiol. Biochem. 2018;47:1207–1216. doi: 10.1159/000490217. [DOI] [PubMed] [Google Scholar]
- 49.Verma A., Jain A., Tiwari A., Saraf S., Panda P.K., Agrawal G.P., Jain S.K. Folate conjugated double liposomes bearing prednisolone and methotrexate for targeting rheumatoid arthritis. Pharm. Res. 2019;36:123. doi: 10.1007/s11095-019-2653-0. [DOI] [PubMed] [Google Scholar]
- 50.Gaafar P.M., Abdallah O.Y., Farid R.M., Abdelkader H. Preparation, characterization and evaluation of novel elastic nano-sized niosomes (ethoniosomes) for ocular delivery of prednisolone. J. Liposome Res. 2014;24:204–215. doi: 10.3109/08982104.2014.881850. [DOI] [PubMed] [Google Scholar]
- 51.Karabey-Akyurek Y., Gurcay A.G., Gurcan O., Turkoglu O.F., Yabanoglu-Ciftci S., Eroglu H., Sargon M.F., Bilensoy E., Oner L. Localized delivery of methylprednisolone sodium succinate with polymeric nanoparticles in experimental injured spinal cord model. Pharm. Dev. Technol. 2017;22:972–981. doi: 10.3109/10837450.2016.1143002. [DOI] [PubMed] [Google Scholar]
- 52.Soiberman U., Kambhampati S.P., Wu T., Mishra M.K., Oh Y., Sharma R., Wang J., Al Towerki A.E., Yiu S., Stark W.J., Kannan R.M. Subconjunctival injectable dendrimer-dexamethasone gel for the treatment of corneal inflammation. Biomaterials. 2017;125:38–53. doi: 10.1016/j.biomaterials.2017.02.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Wang Q., Jiang J., Chen W., Jiang H., Zhang Z., Sun X. Targeted delivery of low-dose dexamethasone using PCL-PEG micelles for effective treatment of rheumatoid arthritis. J. Control. Release. 2016;230:64–72. doi: 10.1016/j.jconrel.2016.03.035. [DOI] [PubMed] [Google Scholar]
- 54.Lee A., De Mei C., Fereira M., Marotta R., Yoon H.Y., Kim K., Kwon I.C., Decuzzi P. Dexamethasone-loaded polymeric nanoconstructs for monitoring and treating inflammatory bowel disease. Theranostics. 2017;7:3653–3666. doi: 10.7150/thno.18183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Dianzani C., Foglietta F., Ferrara B., Rosa A.C., Muntoni E., Gasco P., Della Pepa C., Canaparo R., Serpe L. Solid lipid nanoparticles delivering anti-inflammatory drugs to treat inflammatory bowel disease: effects in an in vivo model. World J. Gastroenterol. 2017;23:4200–4210. doi: 10.3748/wjg.v23.i23.4200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Jia Z., Zhao G., Wei X., Kong D., Sun Y., Zhou Y., Lele S.M., Fehringer E.V., Garvin K.L., Goldring S.R., Wang D. Structural optimization of HPMA copolymer-based dexamethasone prodrug for improved treatment of inflammatory arthritis. J. Control. Release. 2020;324:560–573. doi: 10.1016/j.jconrel.2020.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Greenberg G.R., Feagan B.G., Martin F., Sutherland L.R., Thomson A.B., Williams C.N., Nilsson L.G., Persson T. Oral budesonide for active Crohn's disease. Canadian inflammatory bowel disease study group. N. Engl. J. Med. 1994;331:836–841. doi: 10.1056/NEJM199409293311303. [DOI] [PubMed] [Google Scholar]
- 58.Naeem M., Choi M., Cao J., Lee Y., Ikram M., Yoon S., Lee J., Moon H.R., Kim M.S., Jung Y., Yoo J.W. Colon-targeted delivery of budesonide using dual pH- and time-dependent polymeric nanoparticles for colitis therapy. Drug Des. Dev. Ther. 2015;9:3789–3799. doi: 10.2147/DDDT.S88672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Hua S., Marks E., Schneider J.J., Keely S. Advances in oral nano-delivery systems for colon targeted drug delivery in inflammatory bowel disease: selective targeting to diseased versus healthy tissue. Nanomedicine. 2015;11:1117–1132. doi: 10.1016/j.nano.2015.02.018. [DOI] [PubMed] [Google Scholar]
- 60.Ali H., Weigmann B., Collnot E.M., Khan S.A., Windbergs M., Lehr C.M. Budesonide loaded PLGA nanoparticles for targeting the inflamed intestinal mucosa–pharmaceutical characterization and fluorescence imaging. Pharm. Res. 2016;33:1085–1092. doi: 10.1007/s11095-015-1852-6. [DOI] [PubMed] [Google Scholar]
- 61.Vafaei S.Y., Esmaeili M., Amini M., Atyabi F., Ostad S.N., Dinarvand R. Self assembled hyaluronic acid nanoparticles as a potential carrier for targeting the inflamed intestinal mucosa. Carbohydr. Polym. 2016;144:371–381. doi: 10.1016/j.carbpol.2016.01.026. [DOI] [PubMed] [Google Scholar]
- 62.Wongrakpanich S., Wongrakpanich A., Melhado K., Rangaswami J. A comprehensive review of non-steroidal anti-inflammatory drug use in the elderly. Aging Dis. 2018;9:143–150. doi: 10.14336/AD.2017.0306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Sostres C., Gargallo C.J., Arroyo M.T., Lanas A. Adverse effects of non-steroidal anti-inflammatory drugs (NSAIDs, aspirin and coxibs) on upper gastrointestinal tract. Best Pract. Res. Clin. Gastroenterol. 2010;24:121–132. doi: 10.1016/j.bpg.2009.11.005. [DOI] [PubMed] [Google Scholar]
- 64.Goncalves R.M., Pereira A.C., Pereira I.O., Oliveira M.J., Barbosa M.A. Macrophage response to chitosan/poly-(gamma-glutamic acid) nanoparticles carrying an anti-inflammatory drug. J. Mater. Sci. Mater. Med. 2015;26:167. doi: 10.1007/s10856-015-5496-1. [DOI] [PubMed] [Google Scholar]
- 65.Shang Q., Huang S., Zhang A., Feng J., Yang S. The binary complex of poly(PEGMA-co-MAA) hydrogel and PLGA nanoparticles as a novel oral drug delivery system for ibuprofen delivery. J. Biomater. Sci. Polym. Ed. 2017;28:1874–1887. doi: 10.1080/09205063.2017.1354677. [DOI] [PubMed] [Google Scholar]
- 66.Nirbhavane P., Sharma G., Singh B., Khuller G.K., Goni V.G., Patil A.B., Katare O.P. Preclinical explorative assessment of celecoxib-based biocompatible lipidic nanocarriers for the management of CFA-induced rheumatoid arthritis in Wistar rats. AAPS PharmSciTech. 2018;19:3187–3198. doi: 10.1208/s12249-018-1148-3. [DOI] [PubMed] [Google Scholar]
- 67.Ip W.K.E., Hoshi N., Shouval D.S., Snapper S., Medzhitov R. Anti-inflammatory effect of IL-10 mediated by metabolic reprogramming of macrophages. Science. 2017;356:513–519. doi: 10.1126/science.aal3535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Viscido A., Capannolo A., Latella G., Caprilli R., Frieri G. Nanotechnology in the treatment of inflammatory bowel diseases. J. Crohns Colitis. 2014;8:903–918. doi: 10.1016/j.crohns.2014.02.024. [DOI] [PubMed] [Google Scholar]
- 69.Toita R., Kawano T., Murata M., Kang J.H. Anti-obesity and anti-inflammatory effects of macrophage-targeted interleukin-10-conjugated liposomes in obese mice. Biomaterials. 2016;110:81–88. doi: 10.1016/j.biomaterials.2016.09.018. [DOI] [PubMed] [Google Scholar]
- 70.Kamaly N., Fredman G., Fojas J.J., Subramanian M., Choi W.I., Zepeda K., Vilos C., Yu M., Gadde S., Wu J., Milton J., Carvalho Leitao R., Rosa Fernandes L., Hasan M., Gao H., Nguyen V., Harris J., Tabas I., Farokhzad O.C. Targeted interleukin-10 nanotherapeutics developed with a microfluidic chip enhance resolution of inflammation in advanced atherosclerosis. ACS Nano. 2016;10:5280–5292. doi: 10.1021/acsnano.6b01114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Baganizi D.R., Nyairo E., Duncan S.A., Singh S.R., Dennis V.A. Interleukin-10 conjugation to carboxylated PVP-coated silver nanoparticles for improved stability and therapeutic efficacy. Nanomaterials (Basel) 2017;7 doi: 10.3390/nano7070165. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Stensberg M.C., Wei Q., McLamore E.S., Porterfield D.M., Wei A., Sepulveda M.S. Toxicological studies on silver nanoparticles: challenges and opportunities in assessment, monitoring and imaging. Nanomedicine (London) 2011;6:879–898. doi: 10.2217/nnm.11.78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Yildirimer L., Thanh N.T., Loizidou M., Seifalian A.M. Toxicology and clinical potential of nanoparticles. Nano Today. 2011;6:585–607. doi: 10.1016/j.nantod.2011.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Sung J.H., Ji J.H., Yoon J.U., Kim D.S., Song M.Y., Jeong J., Han B.S., Han J.H., Chung Y.H., Kim J., Kim T.S., Chang H.K., Lee E.J., Lee J.H., Yu I.J. Lung function changes in Sprague-Dawley rats after prolonged inhalation exposure to silver nanoparticles. Inhal. Toxicol. 2008;20:567–574. doi: 10.1080/08958370701874671. [DOI] [PubMed] [Google Scholar]
- 75.Bilberg K., Hovgaard M.B., Besenbacher F., Baatrup E. In vivo toxicity of silver nanoparticles and silver ions in zebrafish (Danio rerio) J. Toxicol. 2012;2012:293784. doi: 10.1155/2012/293784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Agarwal R., Volkmer T.M., Wang P., Lee L.A., Wang Q., Garcia A.J. Synthesis of self-assembled IL-1Ra-presenting nanoparticles for the treatment of osteoarthritis. J. Biomed. Mater. Res. A. 2016;104:595–599. doi: 10.1002/jbm.a.35601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Wang T., Tang Y., He X., Yan J., Wang C., Feng X. Self-assembled raspberry-like core/satellite nanoparticles for anti-inflammatory protein delivery. ACS Appl. Mater. Interfaces. 2017;9:6902–6907. doi: 10.1021/acsami.6b16277. [DOI] [PubMed] [Google Scholar]
- 78.Gupta S., Sharma A.K., Shastri V., Madhu M.K., Sharma V.K. Prediction of anti-inflammatory proteins/peptides: an insilico approach. J. Transl. Med. 2017;15:7. doi: 10.1186/s12967-016-1103-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Bartlett R.L., 2nd, Sharma S., Panitch A. Cell-penetrating peptides released from thermosensitive nanoparticles suppress pro-inflammatory cytokine response by specifically targeting inflamed cartilage explants. Nanomedicine. 2013;9:419–427. doi: 10.1016/j.nano.2012.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Poh S., Lin J.B., Panitch A. Release of anti-inflammatory peptides from thermosensitive nanoparticles with degradable cross-links suppresses pro-inflammatory cytokine production. Biomacromolecules. 2015;16:1191–1200. doi: 10.1021/bm501849p. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Lin J.B., Poh S., Panitch A. Controlled release of anti-inflammatory peptides from reducible thermosensitive nanoparticles suppresses cartilage inflammation. Nanomedicine. 2016;12:2095–2100. doi: 10.1016/j.nano.2016.05.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.He C., Yin L., Song Y., Tang C., Yin C. Optimization of multifunctional chitosan-siRNA nanoparticles for oral delivery applications, targeting TNF-alpha silencing in rats. Acta Biomater. 2015;17:98–106. doi: 10.1016/j.actbio.2015.01.041. [DOI] [PubMed] [Google Scholar]
- 83.Yadav S., Gandham S.K., Panicucci R., Amiji M.M. Intranasal brain delivery of cationic nanoemulsion-encapsulated TNFalpha siRNA in prevention of experimental neuroinflammation. Nanomedicine. 2016;12:987–1002. doi: 10.1016/j.nano.2015.12.374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Kanazawa T., Kurano T., Ibaraki H., Takashima Y., Suzuki T., Seta Y. Therapeutic effects in a transient middle cerebral artery occlusion rat model by nose-to-brain delivery of anti-TNF-alpha siRNA with cell-penetrating peptide-modified polymer micelles. Pharmaceutics. 2019;11 doi: 10.3390/pharmaceutics11090478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Bohr A., Tsapis N., Andreana I., Chamarat A., Foged C., Delomenie C., Noiray M., El Brahmi N., Majoral J.P., Mignani S., Fattal E. Anti-inflammatory effect of anti-TNF-alpha SiRNA cationic phosphorus dendrimer nanocomplexes administered intranasally in a murine acute lung injury model. Biomacromolecules. 2017;18:2379–2388. doi: 10.1021/acs.biomac.7b00572. [DOI] [PubMed] [Google Scholar]
- 86.Shi Q., Rondon-Cavanzo E.P., Dalla Picola I.P., Tiera M.J., Zhang X., Dai K., Benabdoune H.A., Benderdour M., Fernandes J.C. In vivo therapeutic efficacy of TNFalpha silencing by folate-PEG-chitosan-DEAE/siRNA nanoparticles in arthritic mice. Int. J. Nanomedicine. 2018;13:387–402. doi: 10.2147/IJN.S146942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Aldayel A.M., O'Mary H.L., Valdes S.A., Li X., Thakkar S.G., Mustafa B.E., Cui Z. Lipid nanoparticles with minimum burst release of TNF-alpha siRNA show strong activity against rheumatoid arthritis unresponsive to methotrexate. J. Control. Release. 2018;283:280–289. doi: 10.1016/j.jconrel.2018.05.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Jansen M.A.A., Klausen L.H., Thanki K., Lyngso J., Skov Pedersen J., Franzyk H., Nielsen H.M., van Eden W., Dong M., Broere F., Foged C., Zeng X. Lipidoid-polymer hybrid nanoparticles loaded with TNF siRNA suppress inflammation after intra-articular administration in a murine experimental arthritis model. Eur. J. Pharm. Biopharm. 2019;142:38–48. doi: 10.1016/j.ejpb.2019.06.009. [DOI] [PubMed] [Google Scholar]
- 89.Liu Y.C., Zou X.B., Chai Y.F., Yao Y.M. Macrophage polarization in inflammatory diseases. Int. J. Biol. Sci. 2014;10:520–529. doi: 10.7150/ijbs.8879. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Jain S., Tran T.H., Amiji M. Macrophage repolarization with targeted alginate nanoparticles containing IL-10 plasmid DNA for the treatment of experimental arthritis. Biomaterials. 2015;61:162–177. doi: 10.1016/j.biomaterials.2015.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Tran T.H., Rastogi R., Shelke J., Amiji M.M. Modulation of macrophage functional polarity towards anti-inflammatory phenotype with plasmid DNA delivery in CD44 targeting hyaluronic acid nanoparticles. Sci. Rep. 2015;5:16632. doi: 10.1038/srep16632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Haneklaus M., Gerlic M., O'Neill L.A., Masters S.L. miR-223: infection, inflammation and cancer. J. Intern. Med. 2013;274:215–226. doi: 10.1111/joim.12099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Tran T.H., Krishnan S., Amiji M.M. MicroRNA-223 induced repolarization of peritoneal macrophages using CD44 targeting hyaluronic acid nanoparticles for anti-inflammatory effects. PLoS One. 2016;11 doi: 10.1371/journal.pone.0152024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Carmona-Ribeiro A.M. Preparation and characterization of biomimetic nanoparticles for drug delivery. Methods Mol. Biol. 2012;906:283–294. doi: 10.1007/978-1-61779-953-2_22. [DOI] [PubMed] [Google Scholar]
- 95.Hu C.M., Fang R.H., Wang K.C., Luk B.T., Thamphiwatana S., Dehaini D., Nguyen P., Angsantikul P., Wen C.H., Kroll A.V., Carpenter C., Ramesh M., Qu V., Patel S.H., Zhu J., Shi W., Hofman F.M., Chen T.C., Gao W., Zhang K., Chien S., Zhang L. Nanoparticle biointerfacing by platelet membrane cloaking. Nature. 2015;526:118–121. doi: 10.1038/nature15373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Yan H., Shao D., Lao Y.H., Li M., Hu H., Leong K.W. Engineering cell membrane-based nanotherapeutics to target inflammation. Adv. Sci. (Weinh.) 2019;6:1900605. doi: 10.1002/advs.201900605. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Jin K., Luo Z., Zhang B., Pang Z. Biomimetic nanoparticles for inflammation targeting. Acta Pharm. Sin. B. 2018;8:23–33. doi: 10.1016/j.apsb.2017.12.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Vijayan V., Uthaman S., Park I.K. Cell membrane-camouflaged nanoparticles: a promising biomimetic strategy for cancer theragnostics. Polymers (Basel) 2018;10 doi: 10.3390/polym10090983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Wang H., Liu Y., He R., Xu D., Zang J., Weeranoppanant N., Dong H., Li Y. Cell membrane biomimetic nanoparticles for inflammation and cancer targeting in drug delivery. Biomater. Sci. 2020;8:552–568. doi: 10.1039/c9bm01392j. [DOI] [PubMed] [Google Scholar]
- 100.Firestein G.S., McInnes I.B. Immunopathogenesis of Rheumatoid Arthritis. Immunity. 2017;46:183–196. doi: 10.1016/j.immuni.2017.02.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Horckmans M., Ring L., Duchene J., Santovito D., Schloss M.J., Drechsler M., Weber C., Soehnlein O., Steffens S. Neutrophils orchestrate post-myocardial infarction healing by polarizing macrophages towards a reparative phenotype. Eur. Heart J. 2017;38:187–197. doi: 10.1093/eurheartj/ehw002. [DOI] [PubMed] [Google Scholar]
- 102.Zhang Q., Dehaini D., Zhang Y., Zhou J., Chen X., Zhang L., Fang R.H., Gao W. Neutrophil membrane-coated nanoparticles inhibit synovial inflammation and alleviate joint damage in inflammatory arthritis. Nat. Nanotechnol. 2018;13:1182–1190. doi: 10.1038/s41565-018-0254-4. [DOI] [PubMed] [Google Scholar]
- 103.Thamphiwatana S., Angsantikul P., Escajadillo T., Zhang Q., Olson J., Luk B.T., Zhang S., Fang R.H., Gao W., Nizet V., Zhang L. Macrophage-like nanoparticles concurrently absorbing endotoxins and proinflammatory cytokines for sepsis management. Proc. Natl. Acad. Sci. U. S. A. 2017;114:11488–11493. doi: 10.1073/pnas.1714267114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Boada C., Zinger A., Tsao C., Zhao P., Martinez J.O., Hartman K., Naoi T., Sukhoveshin R., Sushnitha M., Molinaro R., Trachtenberg B., Cooke J.P., Tasciotti E. Rapamycin-loaded biomimetic nanoparticles reverse vascular inflammation. Circ. Res. 2020;126:25–37. doi: 10.1161/CIRCRESAHA.119.315185. [DOI] [PubMed] [Google Scholar]
- 105.Fukuta T., Yoshimi S., Tanaka T., Kogure K. Leukocyte-mimetic liposomes possessing leukocyte membrane proteins pass through inflamed endothelial cell layer by regulating intercellular junctions. Int. J. Pharm. 2019;563:314–323. doi: 10.1016/j.ijpharm.2019.04.027. [DOI] [PubMed] [Google Scholar]
- 106.Li R., He Y., Zhu Y., Jiang L., Zhang S., Qin J., Wu Q., Dai W., Shen S., Pang Z., Wang J. Route to rheumatoid arthritis by macrophage-derived microvesicle-coated nanoparticles. Nano Lett. 2019;19:124–134. doi: 10.1021/acs.nanolett.8b03439. [DOI] [PubMed] [Google Scholar]
- 107.Zelcer N., Tontonoz P. Liver X receptors as integrators of metabolic and inflammatory signaling. J. Clin. Invest. 2006;116:607–614. doi: 10.1172/JCI27883. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.He H., Yuan Q., Bie J., Wallace R.L., Yannie P.J., Wang J., Lancina M.G., 3rd, Zolotarskaya O.Y., Korzun W., Yang H., Ghosh S. Development of mannose functionalized dendrimeric nanoparticles for targeted delivery to macrophages: use of this platform to modulate atherosclerosis. Transl. Res. 2018;193:13–30. doi: 10.1016/j.trsl.2017.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Zhang X.Q., Even-Or O., Xu X., van Rosmalen M., Lim L., Gadde S., Farokhzad O.C., Fisher E.A. Nanoparticles containing a liver X receptor agonist inhibit inflammation and atherosclerosis. Adv. Healthc. Mater. 2015;4:228–236. doi: 10.1002/adhm.201400337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Guo Y., Yuan W., Yu B., Kuai R., Hu W., Morin E.E., Garcia-Barrio M.T., Zhang J., Moon J.J., Schwendeman A., Eugene Chen Y. Synthetic high-density lipoprotein-mediated targeted delivery of liver X receptors agonist promotes atherosclerosis regression. EBioMedicine. 2018;28:225–233. doi: 10.1016/j.ebiom.2017.12.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Thapa R.K., Yoo B.K. Evaluation of the effect of tacrolimus-loaded liquid crystalline nanoparticles on psoriasis-like skin inflammation. J. Dermatolog. Treat. 2014;25:22–25. doi: 10.3109/09546634.2012.755250. [DOI] [PubMed] [Google Scholar]
- 112.Sen Lin C.G., Wang Doudou, Xie Qi, Wu Biao, Wang Jingjie, Nan Kaihui, Zheng Qinxiang, Chen Wei. Overcoming the anatomical and physiological barriers in topical eye surface medication using a peptide-decorated polymeric micelle. ACS Appl. Mater. Interfaces. 2019;11 doi: 10.1021/acsami.9b13851. [DOI] [PubMed] [Google Scholar]
- 113.Okuda K., Fu H.Y., Matsuzaki T., Araki R., Tsuchida S., Thanikachalam P.V., Fukuta T., Asai T., Yamato M., Sanada S., Asanuma H., Asano Y., Asakura M., Hanawa H., Hao H., Oku N., Takashima S., Kitakaze M., Sakata Y., Minamino T. Targeted therapy for acute autoimmune myocarditis with nano-sized liposomal FK506 in rats. PLoS One. 2016;11 doi: 10.1371/journal.pone.0160944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Partoazar A., Nasoohi S., Rezayat S.M., Gilani K., Mehr S.E., Amani A., Rahimi N., Dehpour A.R. Nanoliposome containing cyclosporine A reduced neuroinflammation responses and improved neurological activities in cerebral ischemia/reperfusion in rat. Fundam. Clin. Pharmacol. 2017;31:185–193. doi: 10.1111/fcp.12244. [DOI] [PubMed] [Google Scholar]
- 115.Yadav S., Pawar G., Kulkarni P., Ferris C., Amiji M. CNS delivery and anti-inflammatory effects of intranasally administered cyclosporine-A in cationic nanoformulations. J. Pharmacol. Exp. Ther. 2019;370:843–854. doi: 10.1124/jpet.118.254672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Tang H., Xiang D., Wang F., Mao J., Tan X., Wang Y. 5-ASA-loaded SiO2 nanoparticles-a novel drug delivery system targeting therapy on ulcerative colitis in mice. Mol. Med. Rep. 2017;15:1117–1122. doi: 10.3892/mmr.2017.6153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Tang P., Sun Q., Zhao L., Pu H., Yang H., Zhang S., Gan R., Gan N., Li H. Mesalazine/hydroxypropyl-beta-cyclodextrin/chitosan nanoparticles with sustained release and enhanced anti-inflammation activity. Carbohydr. Polym. 2018;198:418–425. doi: 10.1016/j.carbpol.2018.06.106. [DOI] [PubMed] [Google Scholar]
- 118.Yazeji T., Moulari B., Beduneau A., Stein V., Dietrich D., Pellequer Y., Lamprecht A. Nanoparticle-based delivery enhances anti-inflammatory effect of low molecular weight heparin in experimental ulcerative colitis. Drug Deliv. 2017;24:811–817. doi: 10.1080/10717544.2017.1324530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Couvreur P., Stella B., Reddy L.H., Hillaireau H., Dubernet C., Desmaele D., Lepetre-Mouelhi S., Rocco F., Dereuddre-Bosquet N., Clayette P., Rosilio V., Marsaud V., Renoir J.M., Cattel L. Squalenoyl nanomedicines as potential therapeutics. Nano Lett. 2006;6:2544–2548. doi: 10.1021/nl061942q. [DOI] [PubMed] [Google Scholar]
- 120.Desmaele D., Gref R., Couvreur P. Squalenoylation: a generic platform for nanoparticular drug delivery. J. Control. Release. 2012;161:609–618. doi: 10.1016/j.jconrel.2011.07.038. [DOI] [PubMed] [Google Scholar]
- 121.Feng J., Lepetre-Mouelhi S., Gautier A., Mura S., Cailleau C., Coudore F., Hamon M., Couvreur P. A new painkiller nanomedicine to bypass the blood-brain barrier and the use of morphine. Sci. Adv. 2019;5 doi: 10.1126/sciadv.aau5148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Antonioli L., Blandizzi C., Pacher P., Hasko G. Immunity, inflammation and cancer: a leading role for adenosine. Nat. Rev. Cancer. 2013;13:842–857. doi: 10.1038/nrc3613. [DOI] [PubMed] [Google Scholar]
- 123.Hasko G., Linden J., Cronstein B., Pacher P. Adenosine receptors: therapeutic aspects for inflammatory and immune diseases. Nat. Rev. Drug Discov. 2008;7:759–770. doi: 10.1038/nrd2638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Gaudin A., Yemisci M., Eroglu H., Lepetre-Mouelhi S., Turkoglu O.F., Donmez-Demir B., Caban S., Sargon M.F., Garcia-Argote S., Pieters G., Loreau O., Rousseau B., Tagit O., Hildebrandt N., Le Dantec Y., Mougin J., Valetti S., Chacun H., Nicolas V., Desmaele D., Andrieux K., Capan Y., Dalkara T., Couvreur P. Squalenoyl adenosine nanoparticles provide neuroprotection after stroke and spinal cord injury. Nat. Nanotechnol. 2014;9:1054–1062. doi: 10.1038/nnano.2014.274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Egea J., Fabregat I., Frapart Y.M., Ghezzi P., Gorlach A., Kietzmann T., Kubaichuk K., Knaus U.G., Lopez M.G., Olaso-Gonzalez G., Petry A., Schulz R., Vina J., Winyard P., Abbas K., Ademowo O.S., Afonso C.B., Andreadou I., Antelmann H., Antunes F., Aslan M., Bachschmid M.M., Barbosa R.M., Belousov V., Berndt C., Bernlohr D., Bertran E., Bindoli A., Bottari S.P., Brito P.M., Carrara G., Casas A.I., Chatzi A., Chondrogianni N., Conrad M., Cooke M.S., Costa J.G., Cuadrado A., Dang P. My-Chan, De Smet B., Debelec-Butuner B., Dias I.H.K., Dunn J.D., Edson A.J., El Assar M., El-Benna J., Ferdinandy P., Fernandes A.S., Fladmark K.E., Forstermann U., Giniatullin R., Giricz Z., Gorbe A., Griffiths H., Hampl V., Hanf A., Herget J., Hernansanz-Agustin P., Hillion M., Huang J., Ilikay S., Jansen-Durr P., Jaquet V., Joles J.A., Kalyanaraman B., Kaminskyy D., Karbaschi M., Kleanthous M., Klotz L.O., Korac B., Korkmaz K.S., Koziel R., Kracun D., Krause K.H., Kren V., Krieg T., Laranjinha J., Lazou A., Li H., Martinez-Ruiz A., Matsui R., McBean G.J., Meredith S.P., Messens J., Miguel V., Mikhed Y., Milisav I., Milkovic L., Miranda-Vizuete A., Mojovic M., Monsalve M., Mouthuy P.A., Mulvey J., Munzel T., Muzykantov V., Nguyen I.T.N., Oelze M., Oliveira N.G., Palmeira C.M., Papaevgeniou N., Pavicevic A., Pedre B., Peyrot F., Phylactides M., Pircalabioru G.G., Pitt A.R., Poulsen H.E., Prieto I., Rigobello M.P., Robledinos-Anton N., Rodriguez-Manas L., Rolo A.P., Rousset F., Ruskovska T., Saraiva N., Sasson S., Schroder K., Semen K., Seredenina T., Shakirzyanova A., Smith G.L., Soldati T., Sousa B.C., Spickett C.M., Stancic A., Stasia M.J., Steinbrenner H., Stepanic V., Steven S., Tokatlidis K., Tuncay E., Turan B., Ursini F., Vacek J., Vajnerova O., Valentova K., Van Breusegem F., Varisli L., Veal E.A., Yalcin A.S., Yelisyeyeva O., Zarkovic N., Zatloukalova M., Zielonka J., Touyz R.M., Papapetropoulos A., Grune T., Lamas S., Schmidt H., Di Lisa F., Daiber A. European contribution to the study of ROS: A summary of the findings and prospects for the future from the COST action BM1203 (EU-ROS) Redox Biol. 2017;13:94–162. doi: 10.1016/j.redox.2017.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Moia V.M., Leal Portilho F., Almeida Padua T., Barbosa Correa L., Ricci-Junior E., Cruz Rosas E., Alencar L. Magalhaes Rebelo, Sinfronio F. Savio Mendes, Sampson A., Iram S. Hussain, Alexis F., de OliveiraHenriques M.D., Santos-Oliveira R. Lycopene used as anti-inflammatory nanodrug for the treatment of rheumathoid arthritis: animal assay, pharmacokinetics, ABC transporter and tissue deposition. Colloids Surf. B: Biointerfaces. 2020;188 doi: 10.1016/j.colsurfb.2020.110814. [DOI] [PubMed] [Google Scholar]
- 127.Kadry M.O. Liposomal glutathione as a promising candidate for immunological rheumatoid arthritis therapy. Heliyon. 2019;5 doi: 10.1016/j.heliyon.2019.e02162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Wu T., Chen X., Wang Y., Xiao H., Peng Y., Lin L., Xia W., Long M., Tao J., Shuai X. Aortic plaque-targeted andrographolide delivery with oxidation-sensitive micelle effectively treats atherosclerosis via simultaneous ROS capture and anti-inflammation. Nanomedicine. 2018;14:2215–2226. doi: 10.1016/j.nano.2018.06.010. [DOI] [PubMed] [Google Scholar]
- 129.Flavio Dormont R.B., Cailleau Catherine, Reynaud Franceline, Peramo Arnaud, Gendron Amandine, Mougin Julie, Gaudin Françoise, Varna Mariana, Couvreur Patrick. Squalene-based multidrug nanoparticles for improved mitigation of uncontrolled inflammation. Sci. Adv. 05 Jun 2020;6(23) doi: 10.1126/sciadv.aaz5466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Varna M., Xuan H.V., Fort E. Gold nanoparticles in cardiovascular imaging. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2018;10 doi: 10.1002/wnan.1470. [DOI] [PubMed] [Google Scholar]
- 131.Khan M.A., Khan M.J. Nano-gold displayed anti-inflammatory property via NF-kB pathways by suppressing COX-2 activity. Artif. Cells Nanomed. Biotechnol. 2018;46:1149–1158. doi: 10.1080/21691401.2018.1446968. [DOI] [PubMed] [Google Scholar]
- 132.Zhao Y., He Z., Wang R., Cai P., Zhang X., Yuan Q., Zhang J., Gao F., Gao X. Comparison of the therapeutic effects of gold nanoclusters and gold nanoparticles on rheumatoid arthritis. J. Biomed. Nanotechnol. 2019;15:2281–2290. doi: 10.1166/jbn.2019.2848. [DOI] [PubMed] [Google Scholar]
- 133.Gao F., Yuan Q., Cai P., Gao L., Zhao L., Liu M., Yao Y., Chai Z., Gao X. Au clusters treat rheumatoid arthritis with uniquely reversing cartilage/bone destruction. Adv. Sci. (Weinh.) 2019;6:1801671. doi: 10.1002/advs.201801671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Pannerec-Varna R.P., Bousquet G., Ferreira I., Leboeuf C., Boisgard R., Gapihan G., Verine J., Palpant B., Bossy E. In vivo uptake andcellular distribution of gold nanoshells in a preclinicalmodel of xenografted human renal cancer. Gold Bull. 2013;46:257–265. [Google Scholar]
- 135.De Koker S., Cui J., Vanparijs N., Albertazzi L., Grooten J., Caruso F., De Geest B.G. Engineering polymer hydrogel nanoparticles for lymph node-targeted delivery. Angew. Chem. Int. Ed. Eng. 2016;55:1334–1339. doi: 10.1002/anie.201508626. [DOI] [PubMed] [Google Scholar]
- 136.Mueller S.N., Tian S., DeSimone J.M. Rapid and persistent delivery of antigen by lymph node targeting PRINT nanoparticle vaccine carrier to promote humoral immunity. Mol. Pharm. 2015;12:1356–1365. doi: 10.1021/mp500589c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Chu T.W., Yang J., Zhang R., Sima M., Kopecek J. Cell surface self-assembly of hybrid nanoconjugates via oligonucleotide hybridization induces apoptosis. ACS Nano. 2014;8:719–730. doi: 10.1021/nn4053827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Chu T.W., Zhang R., Yang J., Chao M.P., Shami P.J., Kopecek J. A two-step pretargeted nanotherapy for CD20 crosslinking may achieve superior anti-lymphoma efficacy to rituximab. Theranostics. 2015;5:834–846. doi: 10.7150/thno.12040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Li L., Yang J., Wang J., Kopecek J. Amplification of CD20 cross-linking in rituximab-resistant B-lymphoma cells enhances apoptosis induction by drug-free macromolecular therapeutics. ACS Nano. 2018;12:3658–3670. doi: 10.1021/acsnano.8b00797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Wang J., Li L., Yang J., Clair P.M., Glenn M.J., Stephens D.M., Radford D.C., Kosak K.M., Deininger M.W., Shami P.J., Kopecek J. Drug-free macromolecular therapeutics induce apoptosis in cells isolated from patients with B cell malignancies with enhanced apoptosis induction by pretreatment with gemcitabine. Nanomedicine. 2019;16:217–225. doi: 10.1016/j.nano.2018.12.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Wang J., Li Y., Li L., Yang J., Kopecek J. Exploration and evaluation of therapeutic efficacy of drug-free macromolecular therapeutics in collagen-induced rheumatoid arthritis mouse model. Macromol. Biosci. 2020;20 doi: 10.1002/mabi.201900445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Du J., Zhang Y.S., Hobson D., Hydbring P. Nanoparticles for immune system targeting. Drug Discov. Today. 2017;22:1295–1301. doi: 10.1016/j.drudis.2017.03.013. [DOI] [PubMed] [Google Scholar]
- 143.Goldberg M.S. Improving cancer immunotherapy through nanotechnology. Nat. Rev. Cancer. 2019;19:587–602. doi: 10.1038/s41568-019-0186-9. [DOI] [PubMed] [Google Scholar]
- 144.Zupancic E., Curato C., Paisana M., Rodrigues C., Porat Z., Viana A.S., Afonso C.A.M., Pinto J., Gaspar R., Moreira J.N., Satchi-Fainaro R., Jung S., Florindo H.F. Rational design of nanoparticles towards targeting antigen-presenting cells and improved T cell priming. J. Control. Release. 2017;258:182–195. doi: 10.1016/j.jconrel.2017.05.014. [DOI] [PubMed] [Google Scholar]
- 145.Gao W., Fang R.H., Thamphiwatana S., Luk B.T., Li J., Angsantikul P., Zhang Q., Hu C.M., Zhang L. Modulating antibacterial immunity via bacterial membrane-coated nanoparticles. Nano Lett. 2015;15:1403–1409. doi: 10.1021/nl504798g. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Yeste A., Takenaka M.C., Mascanfroni I.D., Nadeau M., Kenison J.E., Patel B., Tukpah A.M., Babon J.A., DeNicola M., Kent S.C., Pozo D., Quintana F.J. Tolerogenic nanoparticles inhibit T cell-mediated autoimmunity through SOCS2. Sci. Signal. 2016;9 doi: 10.1126/scisignal.aad0612. [DOI] [PubMed] [Google Scholar]
- 147.Anselmo A.C., Mitragotri S. Nanoparticles in the clinic. Bioeng. Transl. Med. 2016;1:10–29. doi: 10.1002/btm2.10003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Anselmo A.C., Mitragotri S. Nanoparticles in the clinic: an update. Bioeng. Transl. Med. 2019;4 doi: 10.1002/btm2.10143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Lobatto M.E., Fayad Z.A., Silvera S., Vucic E., Calcagno C., Mani V., Dickson S.D., Nicolay K., Banciu M., Schiffelers R.M., Metselaar J.M., van Bloois L., Wu H.S., Fallon J.T., Rudd J.H., Fuster V., Fisher E.A., Storm G., Mulder W.J. Multimodal clinical imaging to longitudinally assess a nanomedical anti-inflammatory treatment in experimental atherosclerosis. Mol. Pharm. 2010;7:2020–2029. doi: 10.1021/mp100309y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.van der Valk F.M., van Wijk D.F., Lobatto M.E., Verberne H.J., Storm G., Willems M.C., Legemate D.A., Nederveen A.J., Calcagno C., Mani V., Ramachandran S., Paridaans M.P., Otten M.J., Dallinga-Thie G.M., Fayad Z.A., Nieuwdorp M., Schulte D.M., Metselaar J.M., Mulder W.J., Stroes E.S. Prednisolone-containing liposomes accumulate in human atherosclerotic macrophages upon intravenous administration. Nanomedicine. 2015;11:1039–1046. doi: 10.1016/j.nano.2015.02.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.A Proof of Concept Study to Determine the Local Delivery and Efficacy of Nanocort - Full Text View. 2012. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01647685
- 152.Silencing Inflammatory Activity by Injecting Nanocort in Patients at Risk for Atherosclerotic Disease - Full Text View. 2012. ClinicalTrials.govhttps://clinicaltrials.gov/ct2/show/NCT01601106
- 153.Voorzaat B.M., van Schaik J., van der Bogt K.E., Vogt L., Huisman L., Gabreels B.A., van der Meer I.M., van Eps R.G., Eefting D., Weijmer M.C., Groeneveld H.O., van Nieuwenhuizen R.C., Boom H., Verburgh C.A., van der Putten K., Rotmans J.I. Improvement of radiocephalic fistula maturation: rationale and design of the Liposomal Prednisolone to Improve Hemodialysis Fistula Maturation (LIPMAT) study - a randomized controlled trial. J. Vasc. Access. 2017;18:114–117. doi: 10.5301/jva.5000673. [DOI] [PubMed] [Google Scholar]
- 154.Liposome Encapsulated Prednisolone - AdisInsight. 2019. https://adisinsight.springer.com/drugs/800028576 (n.d)
- 155.n.d.h.e.n. Enceladus Pharmaceuticals BV Announces Positive Results From a Phase 2a study in Patients With Active Ulcerative Colitis With Nanocort® | Enceladus Pharmaceuticals. 2016. [Google Scholar]
- 156.Kumar R., Dogra S., Amarji B., Singh B., Kumar S., Sharma K. Vinay, Mahajan R., Katare O.P. Efficacy of novel topical liposomal formulation of cyclosporine in mild to moderate stable plaque psoriasis: a randomized clinical trial. JAMA Dermatol. 2016;152:807–815. doi: 10.1001/jamadermatol.2016.0859. [DOI] [PubMed] [Google Scholar]
- 157.n.d.h. A Evaluation of the Safety of Oncocort IV Pegylated Liposomal Dexamethasone Phosphate in Patients With Progressive Multiple Myeloma - Full Text View. 2012. ClinicalTrials.gov
- 158.Nakano K., Matoba T., Koga J.I., Kashihara Y., Fukae M., Ieiri I., Shiramoto M., Irie S., Kishimoto J., Todaka K., Egashira K. Safety, tolerability, and pharmacokinetics of NK-104-NP. Int. Heart J. 2018;59:1015–1025. doi: 10.1536/ihj.17-555. [DOI] [PubMed] [Google Scholar]
- 159.Sabouri M., Samadi A., Ahmad Nasrollahi S., Farboud E.S., Mirrahimi B., Hassanzadeh H., Nassiri Kashani M., Dinarvand R., Firooz A. Tretinoin loaded nanoemulsion for acne vulgaris: fabrication, physicochemical and clinical efficacy assessments. Skin Pharmacol. Physiol. 2018;31:316–323. doi: 10.1159/000488993. [DOI] [PubMed] [Google Scholar]
- 160.S.A.P.R.o.T.I.C.S.i.A. S. Inc. 2019. https://www.prnewswire.com/news-releases/sebacia-announces-positive-results-of-two-independent-clinical-studies-in-acne-300026759.html (n.d.)
- 161.Hua S., de Matos M.B.C., Metselaar J.M., Storm G. Current trends and challenges in the clinical translation of nanoparticulate nanomedicines: pathways for translational development and commercialization. Front. Pharmacol. 2018;9:790. doi: 10.3389/fphar.2018.00790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Dinarello C.A. Anti-inflammatory agents: present and future. Cell. 2010;140:935–950. doi: 10.1016/j.cell.2010.02.043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Doshi N., Mitragotri S. Macrophages recognize size and shape of their targets. PLoS One. 2010;5 doi: 10.1371/journal.pone.0010051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Wibroe P.P., Anselmo A.C., Nilsson P.H., Sarode A., Gupta V., Urbanics R., Szebeni J., Hunter A.C., Mitragotri S., Mollnes T.E., Moghimi S.M. Bypassing adverse injection reactions to nanoparticles through shape modification and attachment to erythrocytes. Nat. Nanotechnol. 2017;12:589–594. doi: 10.1038/nnano.2017.47. [DOI] [PubMed] [Google Scholar]
- 165.Leong H.S., Butler K.S., Brinker C.J., Azzawi M., Conlan S., Dufes C., Owen A., Rannard S., Scott C., Chen C., Dobrovolskaia M.A., Kozlov S.V., Prina-Mello A., Schmid R., Wick P., Caputo F., Boisseau P., Crist R.M., McNeil S.E., Fadeel B., Tran L., Hansen S.F., Hartmann N.B., Clausen L.P.W., Skjolding L.M., Baun A., Agerstrand M., Gu Z., Lamprou D.A., Hoskins C., Huang L., Song W., Cao H., Liu X., Jandt K.D., Jiang W., Kim B.Y.S., Wheeler K.E., Chetwynd A.J., Lynch I., Moghimi S.M., Nel A., Xia T., Weiss P.S., Sarmento B., das Neves J., Santos H.A., Santos L., Mitragotri S., Little S., Peer D., Amiji M.M., Alonso M.J., Petri-Fink A., Balog S., Lee A., Drasler B., Rothen-Rutishauser B., Wilhelm S., Acar H., Harrison R.G., Mao C., Mukherjee P., Ramesh R., McNally L.R., Busatto S., Wolfram J., Bergese P., Ferrari M., Fang R.H., Zhang L., Zheng J., Peng C., Du B., Yu M., Charron D.M., Zheng G., Pastore C. On the issue of transparency and reproducibility in nanomedicine. Nat. Nanotechnol. 2019;14:629–635. doi: 10.1038/s41565-019-0496-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Chen K.H., Lundy D.J., Toh E.K., Chen C.H., Shih C., Chen P., Chang H.C., Lai J.J., Stayton P.S., Hoffman A.S., Hsieh P.C. Nanoparticle distribution during systemic inflammation is size-dependent and organ-specific. Nanoscale. 2015;7:15863–15872. doi: 10.1039/c5nr03626g. [DOI] [PubMed] [Google Scholar]
- 167.Pradal J., Maudens P., Gabay C., Seemayer C.A., Jordan O., Allemann E. Effect of particle size on the biodistribution of nano- and microparticles following intra-articular injection in mice. Int. J. Pharm. 2016;498:119–129. doi: 10.1016/j.ijpharm.2015.12.015. [DOI] [PubMed] [Google Scholar]
- 168.Dormont F., Rouquette M., Mahatsekake C., Gobeaux F., Peramo A., Brusini R., Calet S., Testard F., Lepetre-Mouelhi S., Desmaele D., Varna M., Couvreur P. Translation of nanomedicines from lab to industrial scale synthesis: The case of squalene-adenosine nanoparticles. J. Control. Release. 2019;307:302–314. doi: 10.1016/j.jconrel.2019.06.040. [DOI] [PubMed] [Google Scholar]
- 169.Lamprecht A. Nanomedicines in gastroenterology and hepatology. Nat. Rev. Gastroenterol. Hepatol. 2015;12:195–204. doi: 10.1038/nrgastro.2015.37. [DOI] [PubMed] [Google Scholar]
- 170.Zhou M., Hou J., Zhong Z., Hao N., Lin Y., Li C. Targeted delivery of hyaluronic acid-coated solid lipid nanoparticles for rheumatoid arthritis therapy. Drug Deliv. 2018;25:716–722. doi: 10.1080/10717544.2018.1447050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Kanhai S., Stavrakaki I., Gladdines W., Gaillard P.J., Klaassen E.S., Groeneveld G.J. Glutathione-PEGylated liposomal methylprednisolone in comparison to free methylprednisolone: slow release characteristics and prolonged lymphocyte depression in a first-in-human study. Br. J. Clin. Pharmacol. 2018;84:1020–1028. doi: 10.1111/bcp.13525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Paithankar D., Munavalli Girish, Kauvar Arielle, Lloyd Jenifer, Blomgren Richard, Faupel Linda, Meyer Todd, Mitragotri Samir. Ultrasonic delivery of silica–gold nanoshells for photothermolysis of sebaceous glands in humans: Nanotechnology from the bench to clinic. J. Control. Release. 2015;206:30–36. doi: 10.1016/j.jconrel.2015.03.004. [DOI] [PubMed] [Google Scholar]