Abstract
Background
During the peak of COVID-19 pandemic, we have noted an increase in positive lower extremity CT angiogram (CTA) exams in patients presenting with leg ischemia. The goal of this study was to determine whether lower extremity arterial thrombosis was associated with COVID-19 and whether it was characterized by greater severity in these patients.
Methods
In this IRB approved retrospective propensity score–matched study, 16 SARS-CoV-2 positive patients who underwent CTA of the lower extremities and 32 SARS-CoV-2 negative patients observed from January to April in 2018-2020 were compared using three scoring system: two systems including all vessels with weighting given in one system to more proximal vessel and in the other to more distal vessels, and a third system where only the common iliac through popliteal arteries were considered. Correlation with presenting symptoms and outcomes was computed. Fisher exact tests were used to compare COVID-19 positive to negative patients regarding presence of clots and presenting symptoms. A Mantel-Haenszel test was used to associate outcome of death/amputation with COVID-19 adjusted by the history of peripheral vascular disease (PVD).
Results
Sixteen patients with confirmed COVID-19 (70 +/- 14 years, 7 women) underwent CTA and 32 propensity-score matched control patients (71 +/- 15 years, 16 women) were included. All COVID-19 patients (100%, 95%CI: 79-100%) had at least one thrombus while only 69% (95%CI: 50-84%) of controls had thrombi (p=0.02). 94% (95%CI: 70-99.8%) of COVID-19 patients had proximal thrombi compared to 47% (95%CI: 29-65%) of controls (p<0.001). Mean thrombus score using any of the three scoring systems yielded greater scores in the COVID-19 patients (p<0.001). Adjusted for history of PVD, death or limb amputation was more common in COVID-19 patients (OR 25, 95%CI 4.3-147, p<0.001). COVID-19 patients presenting with symptoms of leg ischemia only were more likely to avoid amputation or death than patients presenting also with pulmonary or systemic symptoms (p=0.001).
Conclusion
COVID-19 is associated with lower extremity arterial thrombosis characterized by greater clot burden and a more dire prognosis.
Summary
Patients with COVID-19 had lower extremity arterial thrombosis characterized by higher clot burden, involvement of proximal lower extremity arteries, and worse prognosis compared to control patients.
Key Results
■ All patients with COVID-19 infection undergoing lower extremity CTA had at least one lower extremity clot (100%) while only 69% of controls had clots (p=0.02).
■ Mean clot score for lower extremity vessels in patients with COVID-19 was greater than that of propensity score matched controls (p<0.001).
■ Adjusted for history of peripheral vascular disease, death or limb amputation was more common in patients with COVID-19 infection (odds ratio 25, p<0.001).
Introduction
COVID-19 caused by the SARS-CoV-2 virus is characterized by the respiratory symptoms implicit in the name SARS (severe acute respiratory syndrome). Still, with the increasing number of cases, it has also been linked to non-pulmonary targets including cardiac (1), gastrointestinal (2), skin (3), renal (4) and neurological (5) manifestations (4). Venous thromboembolism has also been shown to be an important cause of morbidity and mortality in COVID-19 patients both in the general inpatient and in the ICU setting, and even in patients receiving therapeutic anticoagulation (6,7,8). For example, in one autopsy series, unsuspected deep vein thrombosis was found in the majority of COVID-19 patients, and pulmonary embolism was the cause of death in one fourth of these patients (9). Arterial thrombosis has also been seen in COVID-19 patients reported within coronary arteries (6,10) and within the brain (6,11). There have also been case reports regarding mesenteric (12,13,14) and aortoiliac (6,12) thrombosis.
In our practice, in a COVID-19 hotspot, where at its peak (April 12, 2020) our hospital system had 1,194 inpatients diagnosed with COVID-19, we have observed an increased number of patients presenting with lower extremity ischemia and extensive arterial thromboses during the current pandemic. These patients typically presented to the emergency department with new symptoms of leg pain, coldness, discoloration and ulceration, and underwent lower extremity CTA. Diagnostic workup in many of these patients have shown them to be positive for the SARS-CoV-2 virus. With growing evidence of coagulopathy or vasculopathy (15) in COVID-19 patients, we investigated whether these cases were also due to COVID-19-related thrombosis. The goal of our study was to determine whether lower extremity arterial thrombosis is associated with SARS-CoV-2 infection and if there are differences in severity of lower extremity clot burden in patients who test positive for the virus.
Materials and Methods
Our institutional review board approved this propensity score–matched retrospective cohort study at an urban tertiary care medical center located in an epicenter of the COVID-19 pandemic. The requirement for written informed consent was waived.
Patients
Included in the study are all emergency department and inpatient cases of CTA of the abdominal aorta with lower extremity runoff performed during March and April of 2020, identified on the Picture Archiving and Communications System (PACS). This study covers the peak 2020 months of the pandemic in the New York City metro region (16). Thirty-eight patients underwent lower extremity CTA. Of these, 10 were not tested for the SARS-CoV-2 virus, 11 tested negative and one had a non-diagnostic CTA exam (Figure 1). The diagnosis in the 16 patients was based on RT-PCR testing as follows: Nine patients using the Cepheid Xpert Xpress SARS-CoV-2 assay (Cepheid, Sunnyvale, CA), two patients using the Hologic Panther Fusion real-time RT-PCR SARS-COV-2 assay (Hologic Inc., San Diego, CA), two patients using the Abbott RealTime SARS-COV-2 assay (Abbott Molecular, Des Plaines, IL) and three patients were diagnosed outside our hospital system (one at an urgent care clinic, one at an outside hospital and one at a nursing home) and details of the test used were not available.
Figure 1:
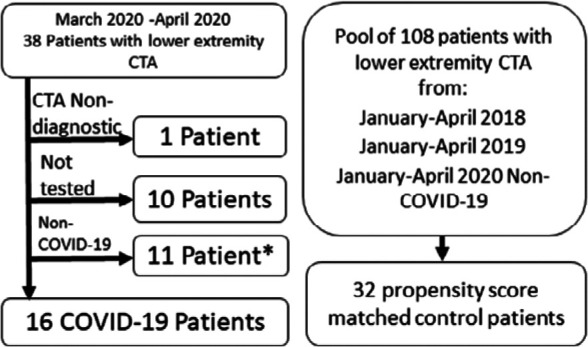
Patient flow for the study. Non-COVID-19 means that the patient tested negative for SARS CoV-2. *Two of these patients which tested negative were used as controls as described in the methods section.
For controls, a pool of all lower extremity CTAs performed on ED patients and inpatients during January through April 2018, January through April 2019 and on those who tested SARS-CoV-2 negative during January through April 2020 consisting of 108 cases was assembled. Our institution is not a level 1 trauma center and all CTA exams were performed for ischemia. We chose control patients from these time periods in consecutive years to avoid seasonal variation, which has been reported for cardiovascular disease (17,18). To reduce the potential confounding from demographic characteristics that differ between COVID-19 patients and controls, we used a method similar to propensity matching. We selected a total of 32 controls from a pool of 108 patients aged 40-95 by 2:1 matching on demographic data. The matching for 15 COVID-cases were done by propensity scores using the nearest neighbor method (19). The propensity scores were created by fitting a logistic regression model on the disease status with sex, age, BMI as the covariates. For the only COVID-19 case for which BMI was not available, matching was performed using ethnicity, sex and age. Among the selected 32 controls only two patients, who presented in March 2020, were tested for SARS-CoV-2 virus; one tested negative once with Hologic Panther Fusion real-time RT-PCR SARS-COV-2 assay and the other tested negative twice with the Cepheid Xpert Xpress SARS-CoV-2 assay.
All CTA exams were performed using GE LightSpeed VCT 64-slice MDCT scanners (GE Healthcare, Milwaukee, WI) following injection of 120 cc of Iopamidol 370 at 4-5 mL/sec. When multiple exams were performed on the same patient, only the first diagnostic exam was considered for this analysis. Blinded to COVID-19 status, images from each case were reviewed in consensus by two radiology attendings (one fellowship trained in body imaging with 14 years of post-training experience, and the other with fellowship training in emergency radiology and 9 years of post-training experience). Only initial CTA images were used for scoring, delayed images were available for only one case and were not used.
Case scoring
To the best of our knowledge there is no validated scoring system for calculating arterial clot burden in the lower extremities; therefore, we used three different systems to perform this task. Vessels were categorized into three zones: aortic (thoracic aorta and abdominal aorta), proximal (common iliac arteries, external iliac arteries, common femoral arteries, superficial femoral arteries, and popliteal arteries) and distal (anterior tibial, peroneal and posterior tibial arteries). Arteries of the foot were not assessed. Complete thrombosis of a single cross-section within the vessel was assigned the full score for that vessel whereas incomplete thrombosis of a vessel was assigned half the value. In the first system (“proximal weighted”), each aortic vessel was given a value of 3, each proximal vessel was given a value of 2 and each distal vessel was given a value of 1. In the second system (“distal weighted”) each aortic vessel was given a value of 1, each proximal vessel was given a value of 2 and each distal vessel was given a value of 3. In the third system (“unweighted proximal only”), only proximal vessels were considered, and each vessel was given a value of 2.
Imaging evidence of a chronic thrombosis included calcification within the walls and lumen of the vessel, decreased arterial caliber and presence of collaterals. Prior studies, if available, and clinical notes were used to help make the determination whether the thrombus was acute or chronic. Prior exam availability was similar between the groups; 2 of 16 (13%) COVID-19 patients and 5 of 32 (16%) control patients had prior studies available (p=1.0). Acute graft thrombosis was scored equivalent to the size of the vessel which it was replacing (e.g. an acutely thrombosed femoral-popliteal graft was scored as 4 as it was spanning 2 vessels, each with a value of 2), while a native chronically occluded vessel was scored as 0 (as it was chronic). If no prior studies were available for comparison, occlusion was deemed chronic if the non-enhancing vessel was diminutive in appearance, compared to the contralateral side, and peripherally calcified.
Chart review
Medical records were reviewed for COVID-19 status (based on RT-PCR testing of nasopharyngeal samples), demographics (age and sex), socioeconomic status expressed as standard deviations from the New York State mean income based on census tract data (20), lab tests, history of PVD, presenting symptoms, ICU stay and outcomes of death or leg amputation. Diagnostic test used to detect SARS-CoV-2 infection was recorded. SARS-CoV-2 IgM or IgG antibody testing was not in use during the time of our study. History of PVD was defined as prior vascular surgical consultation, testing or procedure due to symptoms of lower extremity arterial insufficiency. Presenting symptoms were classified as those pertaining to leg ischemia only versus a combination of leg symptoms and additional respiratory or systemic symptoms such as fever, cough, hypoxia, tachypnea, tachycardia, respiratory distress and/or altered mental status. An ICU stay was considered present if the patient spent any time during the admission in the ICU. Outcome of death or amputation was considered present only if it occurred during the index hospital admission.
Statistical analysis
We treated the COVID-19 patients and the controls as independent observations. Differences in patient characteristics between the two groups were assessed using Fisher’s exact test for dichotomous variables and Welch’s t-test for continuous variables. Fisher exact tests were used to compare presence and absence of clots in the two groups. Clot scores between SARS-Cov-2 positive and negative groups were compared using a linear regression model with history of PVD as a covariate. Association of serious outcomes (death or amputation) with COVID-19 status was tested using the Mantel-Haenszel test conditional on history of PVD. Association of presenting symptoms of patients with COVID-19 and their outcome was assessed using a Fisher's exact test. All statistical analyses were performed using the statistical software R, version 3.6.
Results
Patient characteristics and presentation
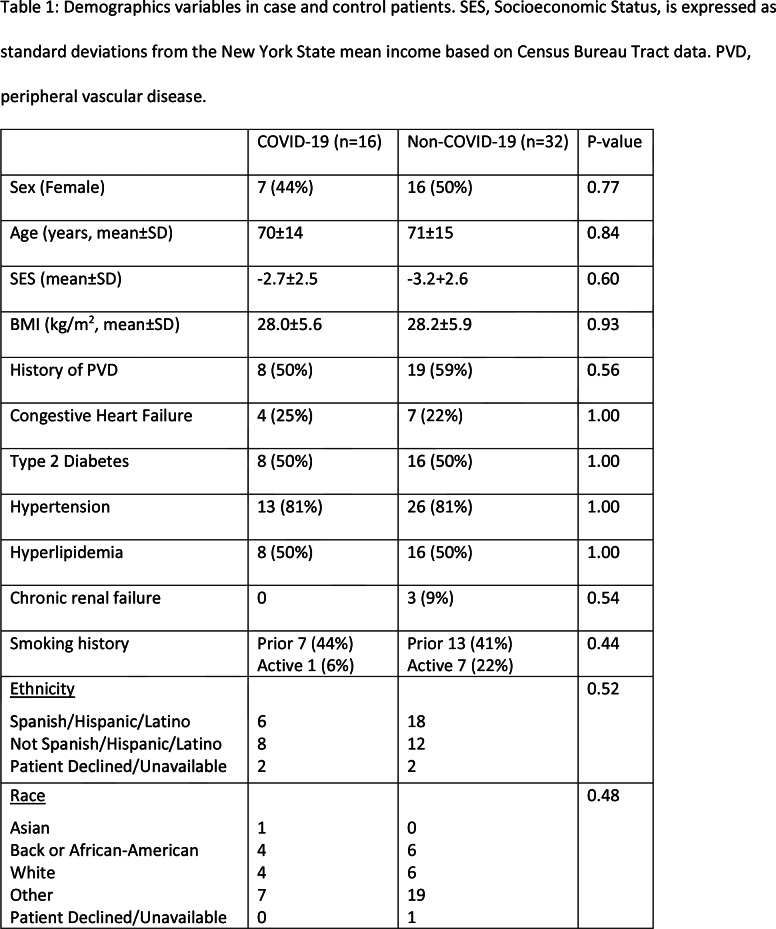
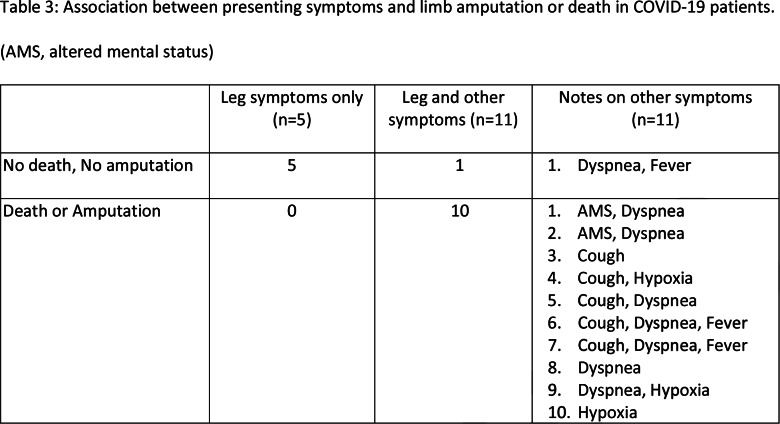
Tables 1 and 2 show the demographic and clinical characteristics of the two groups. Out of the 16 COVID-19 patients, 5 presented only with complaints of leg pain or discoloration, while the other 11 presented with additional systemic or respiratory symptoms (Table 3).
Table 1:
Demographics variables in case and control patients. SES, Socioeconomic Status, is expressed as standard deviations from the New York State mean income based on Census Bureau Tract data. PVD, peripheral vascular disease.
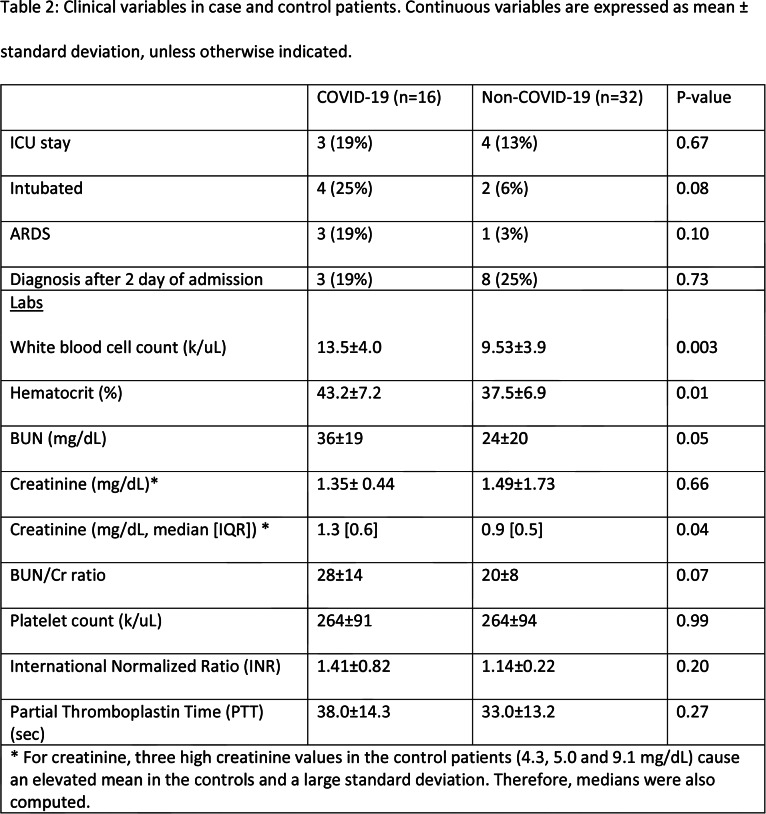
Table 2:
Clinical variables in case and control patients. Continuous variables are expressed as mean ± standard deviation, unless otherwise indicated.
Table 3:
Association between presenting symptoms and limb amputation or death in COVID-19 patients. (AMS, altered mental status)
Presence of clots and clot scores
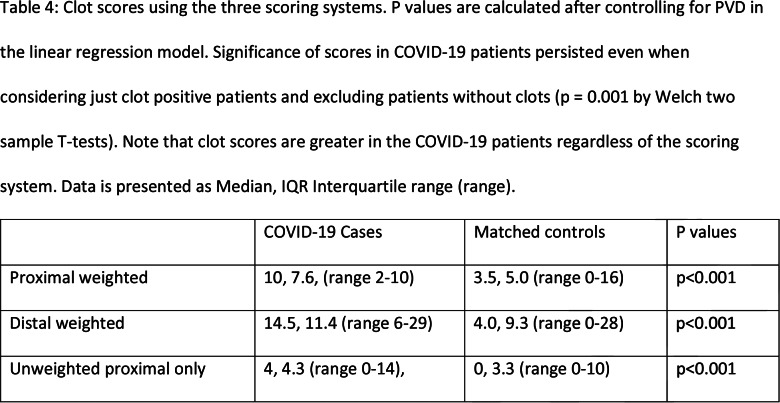
All sixteen COVID-19 patients (100%, 95%CI: 79-100%) had at least one clot (Figure 2) while only 22/32 patients (69%, 95%CI: 50-84%) had at least one clot among the controls (p= 0.02). When looking only at clots within the popliteal artery and more proximally, the COVID-19 patients had clots in 15/16 patients (94%, 95%CI: 70-99.8%) while 15/32 controls (47%, 95%CI: 29-65%) had a clot (p=0.002). Clot scores using the three systems are tabulated in Table 4. Echocardiography was performed in 7/16 (44%) COVID-19 patients and in 10/22 (45%) control patients (where the denominator 22 represents the number of control patients with leg clots). There was no statistically significant difference in echo performance rate (p=1.0). No exam detected cardiac clots.
Figure 2a:
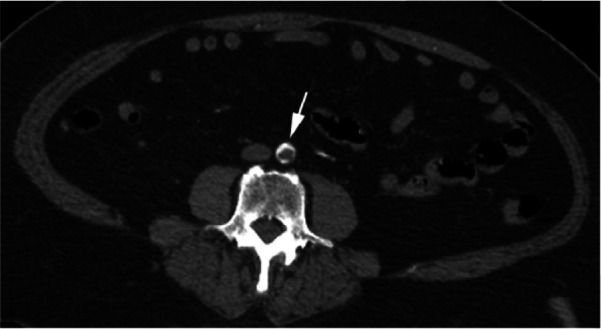
CTA images from a 63-year-old woman with COVID-19. Images through the (a) distal abdominal aorta, (b) proximal common iliac arteries, (c) external iliac arteries, (d) popliteal arteries, and (e) anterior tibial, posterior tibial, and peroneal arteries demonstrate lack of contrast opacification on the left (arrows). Hyperdensity along the periphery of the vessels in a and b corresponds to contrast rather than calcium. Note that the arrows in e point to the expected location of the vessels. (f) Oblique coronal reformatted CT image demonstrates clot at the aortic bifurcation (arrow).
Table 4:
Clot scores using the three scoring systems. P values are calculated after controlling for PVD in the linear regression model. Significance of scores in COVID-19 patients persisted even when considering just clot positive patients and excluding patients without clots (p = 0.001 by Welch two sample T-tests). Note that clot scores are greater in the COVID-19 patients regardless of the scoring system. Data is presented as Median, IQR Interquartile range (range).
Figure 2b:
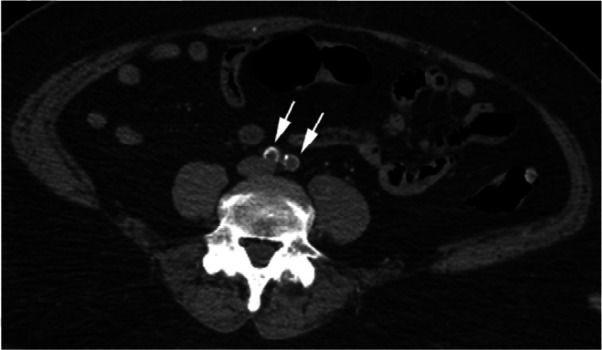
CTA images from a 63-year-old woman with COVID-19. Images through the (a) distal abdominal aorta, (b) proximal common iliac arteries, (c) external iliac arteries, (d) popliteal arteries, and (e) anterior tibial, posterior tibial, and peroneal arteries demonstrate lack of contrast opacification on the left (arrows). Hyperdensity along the periphery of the vessels in a and b corresponds to contrast rather than calcium. Note that the arrows in e point to the expected location of the vessels. (f) Oblique coronal reformatted CT image demonstrates clot at the aortic bifurcation (arrow).
Figure 2c:
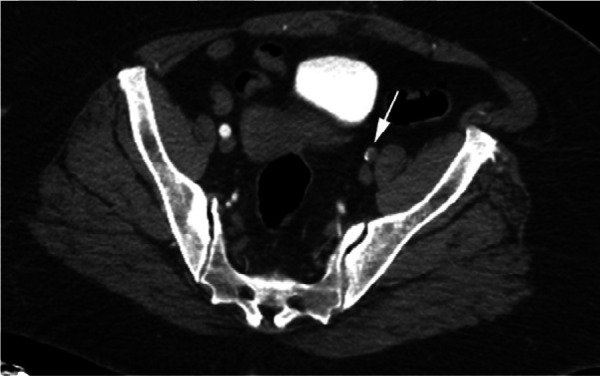
CTA images from a 63-year-old woman with COVID-19. Images through the (a) distal abdominal aorta, (b) proximal common iliac arteries, (c) external iliac arteries, (d) popliteal arteries, and (e) anterior tibial, posterior tibial, and peroneal arteries demonstrate lack of contrast opacification on the left (arrows). Hyperdensity along the periphery of the vessels in a and b corresponds to contrast rather than calcium. Note that the arrows in e point to the expected location of the vessels. (f) Oblique coronal reformatted CT image demonstrates clot at the aortic bifurcation (arrow).
Figure 2d:
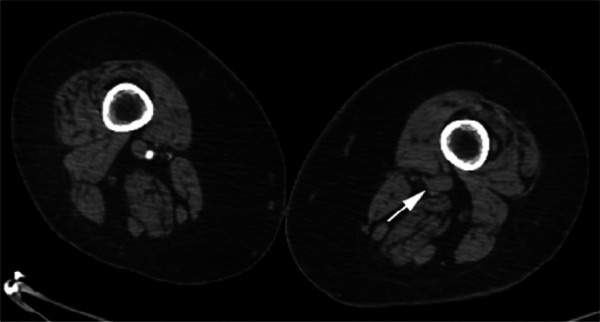
CTA images from a 63-year-old woman with COVID-19. Images through the (a) distal abdominal aorta, (b) proximal common iliac arteries, (c) external iliac arteries, (d) popliteal arteries, and (e) anterior tibial, posterior tibial, and peroneal arteries demonstrate lack of contrast opacification on the left (arrows). Hyperdensity along the periphery of the vessels in a and b corresponds to contrast rather than calcium. Note that the arrows in e point to the expected location of the vessels. (f) Oblique coronal reformatted CT image demonstrates clot at the aortic bifurcation (arrow).
Figure 2e:
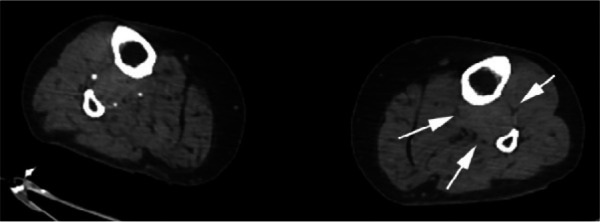
CTA images from a 63-year-old woman with COVID-19. Images through the (a) distal abdominal aorta, (b) proximal common iliac arteries, (c) external iliac arteries, (d) popliteal arteries, and (e) anterior tibial, posterior tibial, and peroneal arteries demonstrate lack of contrast opacification on the left (arrows). Hyperdensity along the periphery of the vessels in a and b corresponds to contrast rather than calcium. Note that the arrows in e point to the expected location of the vessels. (f) Oblique coronal reformatted CT image demonstrates clot at the aortic bifurcation (arrow).
Figure 2f:
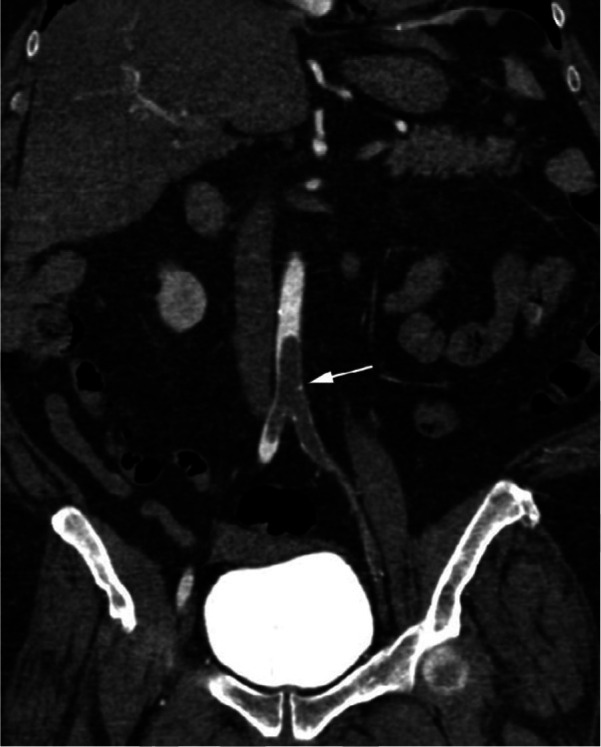
CTA images from a 63-year-old woman with COVID-19. Images through the (a) distal abdominal aorta, (b) proximal common iliac arteries, (c) external iliac arteries, (d) popliteal arteries, and (e) anterior tibial, posterior tibial, and peroneal arteries demonstrate lack of contrast opacification on the left (arrows). Hyperdensity along the periphery of the vessels in a and b corresponds to contrast rather than calcium. Note that the arrows in e point to the expected location of the vessels. (f) Oblique coronal reformatted CT image demonstrates clot at the aortic bifurcation (arrow).
Treatment and Outcomes
Patients either underwent surgery, anticoagulation (heparin or oral agents) or no treatment (one patient could not receive heparin due to concomitant stroke; two patients received palliative treatment only). 7 of 16 (44%) COVID-19 patients underwent surgery (three undergoing thrombectomy, three others undergoing thrombectomy followed by amputation, and one requiring amputation). Eleven of 32 (34%) control patients underwent surgery (6 undergoing thrombectomy, 2 undergoing thrombectomy and bypass, 2 undergoing angioplasty and 1 undergoing bypass). There was no difference in surgical intervention between groups (p=0.54).
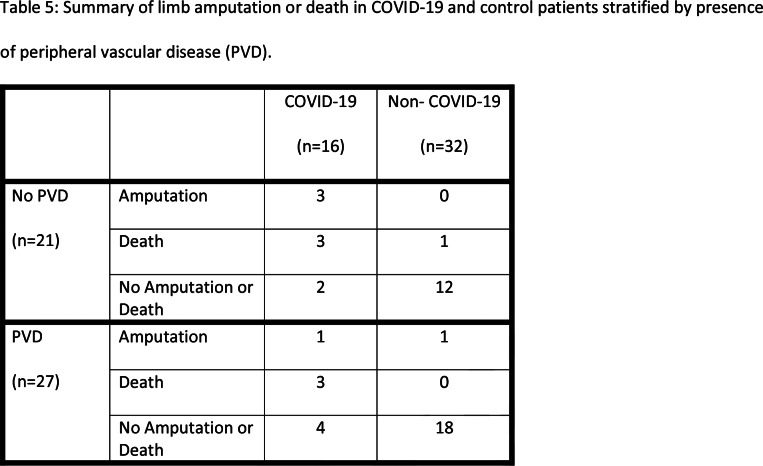
Table 5 summarizes presence of death or amputation in COVID-19 and non-COVID-19 patients. After adjusting for history of PVD using the Mantel-Haenszel method, death or limb amputation was significantly more common in COVID-19 patients (p<0.001). As it is possible that the association between the combined endpoint of death and amputation was due to overwhelming contribution from the death endpoint, we excluded these patients and looked at the association between COVID-19 and amputation alone. We found that even when patients who died were excluded, limb amputation was significantly more common in COVID-19 patients (p=0.02). COVID-19 patients who presented with symptoms of limb ischemia only were significantly more likely to avoid amputation or death than patients who presented with limb ischemia along with respiratory or systemic symptoms (Table 3, p=0.001).
Table 5:
Summary of limb amputation or death in COVID-19 and control patients stratified by presence of peripheral vascular disease (PVD).
Discussion
Results of this study are concordant with the growing body of evidence demonstrating increase in thromboembolic events in patients with COVID-19 disease (6,7,8,9,21,22). While venous thromboembolic events in COVID-19 patients are well recognized, this study demonstrates an association between lower extremity arterial thromboses and SARS-CoV-2 infection. Prominent features of this association are not only a high incidence of arterial thrombosis in COVID-19 patients presenting with ischemic leg symptoms (100% of cases in our cohort), but also a large thrombus burden (Table 4) and high frequency of thromboses involving proximal vessels.
In order to test our hypothesis of larger thrombus burden in COVID-19 patients, we had to design a system to quantify the severity of arterial thrombosis in the lower extremities as, to the best of our knowledge, no such grading system exists. We used three systems to approach this problem. First, by analogy with venous thrombosis grading (23), we created a system (“proximal weighted”) where larger capacity vessels (aorta) were given a higher score than calf vessels which have lower capacity. Second, as it can be argued that thrombosis of the more distal vessels leads to critical ischemia ultimately threatening limb loss, and therefore more distal vessels need to be assigned higher values, we created a second system (“distal weighted”) where calf vessels were assigned a higher score than the aorta. However, an inherent limitation of this system, which assigns a higher weighting to distal vessels, is the inability to always accurately differentiate in the lower leg between acute thrombosis or embolism and chronic occlusion due to atherosclerotic plaques and calcifications. In addition, issues with contrast opacification of the distal vessels may be created by peripheral vascular disease, low cardiac output or technical issues related to bolus timing, rather than actual occlusion. To mitigate this factor, we used a third system (“unweighted proximal only”) where only non-aorta proximal vessels were included, as diagnosis of acute thrombosis usually does not pose difficulty in vessels of larger caliber. All three grading methods yielded concordant statistically significant results verifying our findings (Table 4).
In this study, a significantly increased rate of leg amputation (25%) and death (38%) was found in COVID-19 patients compared to the control group (3% for both) irrespective of the time elapsed between presentation to the hospital and diagnosis by imaging. A similar high mortality rate (40%) was also shown by Bellosta et al. in their cohort of patients presenting with acute lower extremity ischemia and COVID-19 pneumonia (24). Our case and control groups were well matched for age, sex, BMI, socioeconomic status, race, ethnicity and history of smoking. There was also no significant difference in prevalence of preexisting conditions such as PVD, congestive heart failure (CHF), diabetes mellitus (DM), hypertension, chronic renal failure, hyperlipidemia or rate of ICU stay between the two groups. The latter parameter, however, is of uncertain relevance, since during the peak of the pandemic some patients may have been denied ICU entry due to shortage of beds or may not have received ICU level care due to stresses on staffing (including from intensivists, nurses and respiratory therapists). There was a greater percentage of COVID-19 patients who were intubated and were documented as having ARDS in comparison to the control group; however, this difference did not reach statistical significance.
Among the assessed laboratory tests, statistically significant differences between the two groups were observed in WBC count, hematocrit and creatinine (median value) which were mildly elevated in the COVID-19 patients. It is uncertain whether the mildly elevated mean WBC value of 13.5 k/uL was a symptom of the lower extremity ischemia or an indication of concurrent infection, which would portend a worse outcome. Mild elevation in BUN and creatinine (median value) among the COVID-19 patients seen in conjunction with elevation in hematocrit, may be due to dehydration and volume depletion in these patients, with associated prerenal azotemia. Dehydration has been proposed as a contributor to venous thromboembolic disease (25); however, correlation between dehydration and arterial thromboses in adult patients has not been established.
While the more severe outcomes in COVID-19 patients may reflect greater arterial thrombus burden, which may be a marker of increased mortality by itself (26), it can also be attributed to the aggressive nature of severe COVID-19 disease, which has been associated with cytokine storm, fulminant myocarditis (27) and atypical hypercoagulability causing development of thromboses despite patients being on therapeutic doses of anticoagulation (8). Additionally, it is possible, that unprecedented stressors on the healthcare system, particularly in the epicenter of the pandemic at its peak, may have indirectly contributed to these outcomes, for example by delaying the patient’s presentation following the onset of symptoms (28).
This study also suggests that pulmonary or systemic symptoms accompanying the lower extremity symptoms for which CTA was indicated, portents a worse prognosis regarding death and limb amputation. Conversely, a potentially important observation of this study was that if the presenting symptoms related to the leg only, then amputation or death could be avoided. It is unclear based on our data within what time period from the initial SARS-CoV-2 infection thromboses may set in, but awareness that leg ischemia may be associated with COVID-19 should prompt evaluation and treatment for limb ischemia.
One potential limitation of our study was a relatively small cohort of COVID-19 patients as for our analysis we chose COVID-19 patients who underwent CTA of the lower extremities during March and April 2020. However, this choice was dictated by the timing of the peak of disease in our area, for which the leading edge of patients was not tested due to limited testing availability. Indeed, this necessitated exclusion of 10 patients presenting for CTA of lower extremities in early March 2020 from our analysis as they were never tested for SARS-CoV-2 infection. Still, we would expect that this cohort would be one of the larger ones available, as our hospital system has seen approximately 6000 COVID-19 patients at the time of this writing.
Conclusion
This study demonstrates an association between COVID-19 and lower extremity arterial thrombosis with greater thrombus burden characterized by predilection for proximal arteries. Additionally, the incidence of death and amputation is significantly more common in COVID-19 patients, especially if there are also systemic or respiratory symptoms on presentation. Recognition of lower extremity ischemia as a symptom or complication of COVID-19 disease may allow for prompt diagnosis and treatment of this condition.
Footnotes
There was no funding support for this work.
References
- 1.Aghagoli G, Gallo Marin B, Soliman LB, Sellke FW. Cardiac involvement in Covid-19 patients: Risk factors, predictors, and complications: A review (published online ahead of print, 2020 Apr 19). J Card Surg. 2020;10.1111/jocs.14538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Bhayana R, Som A, Li MD, et al. Abdominal Imaging Findings in COVID-19: Preliminary Observations [published online ahead of print, 2020 May 11]. Radiology. 2020;201908. [DOI] [PMC free article] [PubMed]
- 3.Tang K, Wang Y, Zhang H, Zheng Q, Fang R, Sun Q. Cutaneous manifestations of the Coronavirus Disease 2019 (COVID-19): a brief review (published online ahead of print, 2020 May 7). Dermatol Ther. 2020;10.1111/dth.13528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Puelles VG, Lütgehetmann M, Lindenmeyer MT, et al. Multiorgan and Renal Tropism of SARS-CoV-2 (published online ahead of print, 2020 May 13). N Engl J Med. 2020;10.1056/NEJMc2011400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Needham EJ, Chou SH, Coles AJ, Menon DK. Neurological Implications of Covid-19 Infections (published online ahead of print, 2020 Apr 28). Neurocrit Care. 2020;1-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Lodigiani C, Iapichino G, Carenzo L, et al. Venous and arterial thromboembolic complications in Covid-19 patients admitted to an academic hospital in Milan, Italy (published online ahead of print, 2020 Apr 23). Thromb Res. 2020;191:9-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Middeldorp S, Coppens M, van Haaps TF, et al. Incidence of venous thromboembolism in hospitalized patients with COVID-19 (published online ahead of print, 2020 May 5). J Thromb Haemost. 2020;10.1111/jth.14888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Llitjos JF, Leclerc M, Chochois C, et al. High incidence of venous thromboembolic events in anticoagulated severe COVID-19 patients (published online ahead of print, 2020 Apr 22). J Thromb Haemost. 2020;10.1111/jth.14869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wichmann D, Sperhake JP, Lütgehetmann M, et al. Autopsy Findings and Venous Thromboembolism in Patients With COVID-19: A Prospective Cohort Study (published online ahead of print, 2020 May 6). Ann Intern Med. 2020;10.7326/M20-2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Dominguez-Erquicia P, Dobarro D, Raposeiras-Roubín S, Bastos-Fernandez G, Iñiguez-Romo A. Multivessel coronary thrombosis in a patient with COVID-19 pneumonia (published online ahead of print, 2020 May 6). Eur Heart J. 2020;ehaa393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hess DC, Eldahshan W, Rutkowski E. COVID-19-Related Stroke (published online ahead of print, 2020 May 7). Transl Stroke Res. 2020;1-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Vulliamy P, Jacob S, Davenport RA. Acute aorto-iliac and mesenteric arterial thromboses as presenting features of COVID-19 (published online ahead of print, 2020 Apr 30). Br J Haematol. 2020;10.1111/bjh.16760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.de Barry O, Mekki A, Diffre C, Seror M, Hajjam ME, Carlier RY. Arterial and venous abdominal thrombosis in a 79-year-old woman with COVID-19 pneumonia (published online ahead of print, 2020 Apr 29). Radiol Case Rep. 2020;10.1016/j.radcr.2020.04.055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Beccara L A, Pacioni C, Ponton S, Francavilla S, Cuzzoli A. Arterial Mesenteric Thrombosis as a Complication of SARS-CoV-2 Infection. Eur J Case Rep Intern Med. 2020;7(5):001690. Published 2020 Apr 30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Varga Z, Flammer AJ, Steiger P, et al. Endothelial cell infection and endotheliitis in COVID-19. Lancet. 2020;395(10234):1417-1418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.COVID-19: Data Summary - NYC Health. https://www1.nyc.gov/site/doh/covid/covid-19-data.page. Accessed 6/2/20. [Google Scholar]
- 17.Laohapensang K, Rerkasem K, Kattipattanapong V. Seasonal variation of Buerger's disease in Northern part of Thailand. Eur J Vasc Endovasc Surg. 2004;28(4):418-420. [DOI] [PubMed] [Google Scholar]
- 18.Mehta RH, Manfredini R, Hassan F, et al. Chronobiological patterns of acute aortic dissection. Circulation. 2002;106(9):1110-1115. [DOI] [PubMed] [Google Scholar]
- 19.Rosenbaum PR, Rubin DB, The central role of the propensity score in observational studies for causal effects, Biometrika. 1983;70(1):41–55. [Google Scholar]
- 20.Golestaneh L, Farzami A, Madu C, Johns T, Melamed ML, Norris KC. The association of neighborhood racial mix and ED visit count in a cohort of patients on hemodialysis. BMC Nephrol. 2019;20(1):343. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Klok F, Kruip M, van der Meer N, et al. Incidence of Thrombotic Complications in Critically Ill ICU Patients With Covid-19. Thromb Res 2020 Apr 10; S0049-3848(20)30120-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Llitjos J, Leclerc M, Chocois C. et al. High Incidence of Venous Thromboembolic Events in Anticoagulated Severe Covid-19 Patients. J Thromb Haemost 2020 Apr 22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ouriel K, Greenberg RK, Green RM, Massullo JM, Goines DR. A volumetric index for the quantification of deep venous thrombosis. J Vasc Surg. 1999;30(6):1060-1066. [DOI] [PubMed] [Google Scholar]
- 24.Bellosta R, Luzzani L, Natalini G, et al. Acute limb ischemia in patients with COVID-19 pneumonia [published online ahead of print, 2020 Apr 29]. J Vasc Surg. 2020;S0741-5214(20)31080-6. [DOI] [PMC free article] [PubMed]
- 25.Elias S, Hoffman R, Saharov G, Brenner B, Nadir Y. Dehydration as a Possible Cause of Monthly Variation in the Incidence of Venous Thromboembolism. Clin Appl Thromb Hemost. 2016;22(6):569-574. [DOI] [PubMed] [Google Scholar]
- 26.Grilz E. et al. Frequency, Risk Factors, and Impact on Mortality of Arterial Thromboembolism in Patients with Cancer . Haemotologica Sept 2018 103:1549-1556. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Clerkin KJ, Fried JA, Raikhelkar J, et al. COVID-19 and Cardiovascular Disease. Circulation. 2020;141(20):1648-1655. [DOI] [PubMed] [Google Scholar]
- 28.Hartnett KP, Kite-Powell A, DeVies J, et al. Impact of the COVID-19 Pandemic on Emergency Department Visits — United States, January1, 2019–May 30, 2020. MMWR Morb Mortal Wkly Rep. ePub: 3 June 2020. [DOI] [PMC free article] [PubMed]