Abstract
Introduction
An individualised thromboprophylaxis was implemented in critically ill patients suffering from coronavirus disease 2019 (COVID-19) pneumonia to reduce mortality and improve clinical outcome. The aim of this study was to evaluate the effect of this intervention on clinical outcome.
Methods
In this mono-centric, controlled, before-after study, all consecutive adult patients with confirmed COVID-19 pneumonia admitted to ICU from March 13th to April 20th 2020 were included. A thromboprophylaxis protocol, including augmented LMWH dosing, individually tailored with anti-Xa measurements and twice-weekly ultrasonography screening for DVT, was implemented on March 31th 2020. Primary endpoint is one-month mortality. Secondary outcomes include two-week and three-week mortality, the incidence of VTE, acute kidney injury and continuous renal replacement therapy (CRRT). Multiple regression modelling was used to correct for differences between the two groups.
Results
46 patients were included in the before group, 26 patients in the after group. One month mortality decreased from 39.13% to 3.85% (p < 0.001). After correction for confounding variables, one-month mortality was significantly higher in the before group (p = 0.02, OR 8.86 (1.46, 53.75)). The cumulative incidence of VTE and CRRT was respectively 41% and 30.4% in the before group and dropped to 15% (p = 0.03) and 3.8% (p = 0.01), respectively. After correction for confounding variables, risk of VTE (p = 0.03, 6.01 (1.13, 32.12)) and CRRT (p = 0.02, OR 19.21 (1.44, 255.86)) remained significantly higher in the before group.
Conclusion
Mortality, cumulative risk of VTE and need for CRRT may be significantly reduced in COVID-19 patients by implementation of a more aggressive thromboprophylaxis protocol. Future research should focus on confirmation of these results in a randomized design and on uncovering the mechanisms underlying these observations.
Registration number
Keywords: COVID-19, Thromboprophylaxis, Venous thromboembolism, Mortality, Continuous renal replacement therapy
Highlights
-
•
An individualised thromboprophylaxis was implemented in critically ill COVID-19 patients.
-
•
One-month mortality was reduced after implementation of this protocol
-
•
Cumulative incidence of venous thromboembolism was lower after implementation.
-
•
Less need for continuous renal replacement therapy in the after group.
1. Introduction
The ongoing pandemic of coronavirus disease 2019 (COVID-19) in humans is caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2). This coronavirus was first detected in China in December 2019. This coronavirus was first detected in China in December 2019. The spectrum of disease severity of this viral infection is wide: from mild upper respiratory tract infection (RTI) in approximately 80%, to severe lower RTI (e.g. with dyspnoea, PaO2/FiO2 of <300 mm Hg, or bilateral lung involvement on imaging) in approximately 15% of cases and critical (e.g. with respiratory failure, shock, and/or multiorgan dysfunction) in 5% [1,2]. Hypoxemic respiratory failure due to acute respiratory distress syndrome (ARDS) has been reported as the dominant finding during intensive care unit (ICU) admission and as a major cause of death [1,4]. These clinical features may suggest that SARS-CoV-2 predominantly targets the respiratory tract. Indeed, the majority of the patients with severe COVID-19 initially present with single organ failure (i.e. respiratory insufficiency) [5]. Consequently, guidelines on the management of critically ill COVID-19 patients cover predominantly strong recommendations for ventilatory support [6]. Therefore, initial supportive therapy at our ICU, JESSA hospital Hasselt, Belgium, focused mainly at respiratory support with (non)-invasive mechanical ventilation with a higher positive end-expiratory pressure (PEEP) strategy. Hasselt is situated in the epicentre of the Belgian outbreak with the highest incidence across the country [7]. From the first admission to ICU on March 13th, admissions of critically ill COVID-19 patients to ICU grew exponentially. All patients were treated with routine low dose pharmacological venous thromboembolism (VTE) prophylaxis with LMWH according to the current guidelines in critically ill patients [8,9]. Nevertheless, deep vein thrombosis (DVT) screening using duplex ultrasound of all intubated and mechanically ventilated COVID-19 patients treated at one ward on March 29th, revealed a prevalence of DVT of more than 65% [10]. These findings suggested that thrombo-embolic phenomenona may be another, at that time less-well known, major cause of death of severe COVID-19 infection. This was further substantiated by the observation that elevated D-dimer levels, a marker of endogenous fibrinolysis and detectable in patients with DVT [11], are a risk factor for in-hospital mortality in patients with COVID-19 [[12], [13], [14]]. Therefore, we implemented an individualised thromboprophylaxis protocol with augmented doses of LMWH on March 31th in order to reduce mortality and improve clinical outcome.
The aim of this study was to evaluate the effect of this intervention on clinical outcome. The main hypothesis was that implementation of this intensified thromboprophylaxis protocol would have reduced mortality and improved clinical outcome in critically ill COVID-19 patients admitted to ICU.
2. Materials and methods
2.1. Methods
This mono-centric, investigator-initiated, longitudinal, controlled, before-after study is performed at the ICU department of the Jessa Hospital, Hasselt, Belgium. This study is approved by the ethical committee of JESSA Hospital Hasselt, Belgium on May 4th 2020 and registered on clinicaltials.gov (NCT04394000). Written informed consent was waived in light of the urgent need to collect data in the ongoing pandemic.
All adult patients diagnosed with COVID-19 pneumonia and admitted to ICU before April 20th 2020 were included in the study. Laboratory confirmation of COVID-19 was defined as a positive result on polymerase chain reaction (PCR) assays of nasopharyngeal swab samples, in accordance with the World Health Organization (WHO) protocol [15]. Only laboratory-confirmed patients were included in the analysis. From March 13th to May 3th 2020, data from 78 consecutive patients admitted to ICU were prospectively entered into a customized database that included demographic data, medical history, clinical symptoms or signs, laboratory results and clinical outcomes. This database was retrospectively reviewed. APACHE II score was assessed at admission to ICU [16]. The Sequential Organ Failure Assessment (SOFA) score [17] and PaO2/FiO2 (P/F ratio) were evaluated daily.
All COVID-19 patients were treated according to the COVID-protocol of the JESSA hospital (Additional File 1), based on the latest insights on COVID-19 [6].
2.1.1. Standard procedure
All patients admitted to ICU from March 13th 2020 until March 30th 2020 received routine low dose pharmacological VTE prophylaxis, i.e. once-daily subcutaneous injection of nadroparin calcium 2850 IU, according to the current guidelines in critically ill patients [8,9].
2.1.2. Intervention
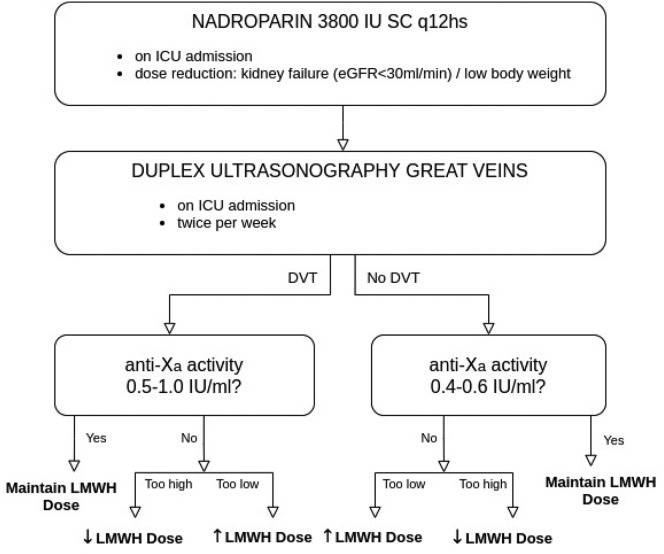
Based on the observation of Pellens et al. [10], we implemented an individualised, more aggressive thromboprophylaxis protocol on March 31th (Fig. 1 ). This individualised protocol contains three cornerstones: an increase in dosage of prophylactic LMWH close to therapeutic doses, introduction of routine venous ultrasonography and daily measurements of plasma anti-factor Xa activity. First, we empirically augmented the prophylactic dose of nadroparin calcium to 3800 IU every 12 h subcutaneously. A dose reduction based on clinical judgment was made for patients with severe kidney failure (eGFR < 30 ml/min) and cachectic patients (total body weight < 40 kg). Second, all patients were screened two times per week for the presence of DVT in the large veins (i.e. the inferior caval vein, iliac, femoral, popliteal, jugular, subclavian and brachial veins) with duplex ultrasonography, regardless of clinical suspicion. This ultrasound technique uses a combination of a venous compression ultrasound and a venous Doppler ultrasound. By applying pressure, one can evaluate the collapsibility of the deep veins. When a vein is not fully collapsible, it indicates the presence of a blood clot. The collapsibility is tested every few cm. Pulsed wave Doppler is used to evaluate the presence and velocity of blood flows within the vein. This examination was consequently performed bedside by a radiologist rather than forming part of a general point-of-care ultrasound examination. Third, we in introduced daily measurement of anti-factor Xa (anti-Xa) activity in all patients. The anti-Xa test is a functional assay, measuring the direct inhibition of factor Xa by any LMWH. Anti-Xa testing was performed with the ACL top 550 (Werfen®). Blood samples were taken 4 to 6 h after subcutaneous administration of LMWH. High-intermediate prophylactic anti-Xa activity of 0,4–0,6 IU/ml was targeted in patients without objectified need for therapeutic anticoagulation (i.e. medical history or presence of DVT on ultrasonography). In patients with need for therapeutic anticoagulation, a therapeutic anti-Xa concentration of 0,5–1,0 IU/ml was targeted (Fig. 1).
Fig. 1.
Overview of the individualised and intensified thromboprophylaxis protocol implemented on March 30th 2020. DVT: deep venous thrombosis, LMWH: low molecular weight heparin.
2.1.3. Outcome parameters
The primary endpoint is one-month mortality. Secondary outcomes include two-week and three-week mortality, the incidence of VTE, the incidence of acute kidney injury and continuous renal replacement therapy (CRRT), the lowest P/F-ratio and highest SOFA-score during ICU stay, length of stay (LOS) in the ICU and hospital LOS, and highest bilirubin, AST and ALT during ICU stay. All patients were followed for at least 30 days after submission to ICU. Data set was closed at June 2th 2020.
2.1.4. Definitions
Acute kidney failure was diagnosed according to the KDIGO clinical practice guidelines [18]. ARDS was diagnosed according to the Berlin Definition [19]. Sepsis and septic shock were defined according to the 2016 Third International Consensus Definition for Sepsis and Septic Shock.
2.1.5. Statistical analysis
Patients admitted to ICU from March 13th until March 30th 2020 treated with low-dose thromboprophylaxis and patients admitted to ICU March 31th until April 20th 2020 treated with an intensified thromboprophylaxis protocol were compared in terms of demographic data, medical history and clinical outcomes. Categorical variables are presented as frequency (%) and groups were compared using Fisher's exact test. Continuous variables are presented as median with interquartile range and groups were compared using Wilcoxon rank sum test.
To compare the two groups for one-month mortality, incidence of acute kidney failure, CRRT and VTE, a logistic regression model, with a Firth correction, was implemented. Worst (lowest) P/F ratio and worst (highest) SOFA score, highest bilirubin, highest AST and highest ALT during ICU stay were compared by means of linear regression model. A naturally logarithmic transformation of these endpoints was used, to obtain normally distributed residuals.
Age, gender, BMI, hypertension and diabetes together with the SOFA and Apache II score at admission to ICU and D-dimer level and white blood cell count at admission to ICU [12,13,20] were incorporated as explanatory (confounding) variables in the logistic and linear regression models to correct for possible differences between the two groups (e.g. the intensified thromboprophylaxis group is younger). The following stepwise strategy was adopted. First, two groups were compared without a correction for patient characteristics and risk factors. Next, models adjusting the comparison were fitted by incorporating one explanatory/confounding variable at a time. Only variables with a p-value ≤ 0.1 were included in the final multiple regression model.
Based on the final multiple regression model, odds ratios (95% confidence interval) for before versus after group were calculated. An odds ratio larger than 1 indicates an increased risk in the before group. For the continuous variables the ratio (95% confidence interval) of the geometric means were obtained, based on the final multiple regression models. A ratio larger than 1 indicates that the geometric mean of the outcome in the before group is higher than in the after group.
A p-value < 0.05 is considered statistically significant and a p-value < 0.1 is considered a statistical tendency. All analyses were conducted with SAS version 9.4.
3. Results
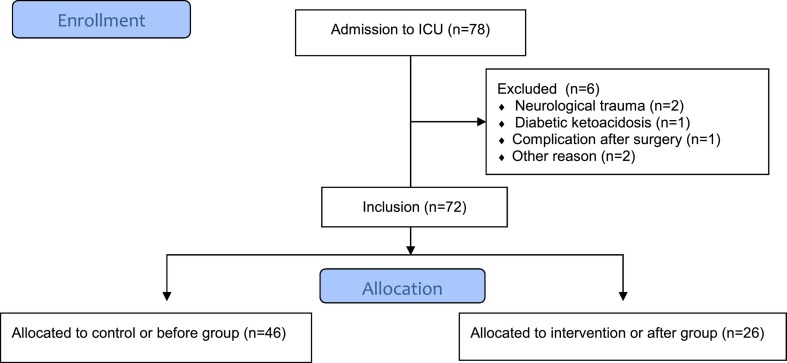
In total, 78 COVID-19 patients were admitted to the COVID-19 ICU from March 13th until April 20th 2020. Six COVID-19 patients, admitted to the COVID ICU for other reasons (i.e. neurological trauma, diabetic ketoacidosis, complication after surgery) than COVID-19 pneumonia, were excluded from analysis. All 46 patients admitted to ICU from March 13th until March 30th were included in the control or ‘before’ group and all 26 patients admitted to ICU from March 31th until April 20th were included in the intervention or ‘after’ group (Additional File 2).
Median age in the before group was 69.50 (62.00, 76.00) vs 62.00 (56.00, 73.00) in the after group (p = 0.03) with 73.91% males in the before group vs 57.69% in the after group (p = 0.19). No differences in BMI (before group: 26.21(24.22, 29.38) vs after group: 27.00 (25.00, 30.72), p = 0.73), Apache II scores (before group: 13.00 (10.00, 16.00) vs after group: 11.00 (9.00, 14.00), p = 0.09) and SOFA scores (before group: 3.50 (2.00, 9.00) vs after group: 4.00 (3.00, 5.00), p = 0.69) were seen between both groups. Baseline characteristics are presented in Table 1 .
Table 1.
Demographics and clinical characteristics at admission to ICU.
| Before |
After |
p-Value | |
|---|---|---|---|
| n = 46 | n = 26 | ||
| Age (years) | 69.50 (62.00, 76.00) | 62.00 (56.00, 73.00) | 0.03 |
| Gender (male) | 34 (73,91%) | 15 (57,69%) | 0.19 |
| BMI (n = 68) | 26.21(24.22, 29.38) | 27.00 (25.00, 30.72) | 0.73 |
| Smoking (%) | 5 (10.87%) | 0 (0.00%) | 0.15 |
| Apache II | 13.00 (10.00, 16.00) | 11.00 (9.00, 14.00) | 0.09 |
| SOFA score | 3.50 (2.00, 9.00) | 4.00 (3.00, 5.00) | 0.69 |
| Comorbidity | |||
| Hypertension | 29 (63.04%) | 11 (42.31%) | 0.14 |
| Diabetes | 14 (30.43%) | 6 (23.08%) | 0.59 |
| Chronic kidney disease | 6 (13.04%) | 5 (19.23%) | 0.51 |
| Chronic liver disease | 1 (2.17%) | 1 (3.85%) | 1.00 |
| Clinical parameters (n = 71) | |||
| Fever | 41 (89.13%) | 17 (68.00%) | 0.05 |
| Cough | 43 (93.48%) | 20 (80.00%) | 0.12 |
| Dyspnoea | 38 (82.61%) | 18 (72.00%) | 0.37 |
| Sputum production | 5 (10.87%) | 4 (16.00%) | 0.71 |
| Myalgia | 25 (54.35%) | 15 (60.00%) | 0.80 |
| Headache | 5 (10.87%) | 4 (16.00%) | 0.71 |
| Diarrhea | 7 (15.22%) | 8 (32.00%) | 0.13 |
| Rhinorrhoea | 4 (8.70%) | 1 (4.00%) | 0.65 |
| Sore throat | 2 (4.35%) | 0 (0.00%) | 0.54 |
| Nausea | 8 (17.40%) | 8 (32.00%) | 0.23 |
| Chest pain | 7 (15.22%) | 2 (8.00%) | 0.48 |
| Dizziness | 4 (8.70%) | 7 (28.00%) | 0.04 |
| Breathing rate (n = 62) | 25.00 (20.00, 30.00) | 23.00 (18.00, 25.00) | 0.06 |
| Heart rate | 88.00 (78.00, 100.00) | 90.00 (78.00, 100.00) | 0.74 |
| Systolic blood pressure | 142.50 (125.00, 163.00) | 143.50 (122.00, 168.00) | 0.87 |
| Glasgow Coma Scale | 15.00 (14.00, 15.00) | 15.00 (14.00, 15.00) | 0.92 |
| Laboratory findings | |||
| White blood cells | 7.46 (6.10, 9.38) | 8.90 (7.12, 10.15) | 0.08 |
| D-dimers (n = 57) | 0.97 (0.64, 1.67) | 2.18 (0.67, 5.20) | 0.12 |
| Troponin (n = 65) | 18.10 (13.40, 25.45) | 15.90 (10.70, 19.20) | 0.17 |
| Lactate dehydrogenase (n = 64) | 440.00 (390.00, 530.00) | 410.00 (360.00, 490.00) | 0.18 |
Data are presented as n (%) or as median (25th, 75th percentile). A p-value < 0.05 is considered statistically significant. Fisher's exact is used for binary variables and Wilcoxon rank sum test for continuous variables.
The primary outcome, i.e. 1 month mortality was 39.13% (18/46) in the before group and 3.85% (1/26) in the after group (p < 0.001).
One-week mortality was 19.57% (9/46) in before group and 3.85% (1/26) in the after group (p = 0.08), two-week mortality was 30.43% (14/46) in the before group and 3.85% (1/26) in the after group (p = 0.01) and three-week mortality was 36.96% (17/46) in the before group and 3.85% (1/16) in the after group (p = 0.01) (Fig. 2 ).
Fig. 2.
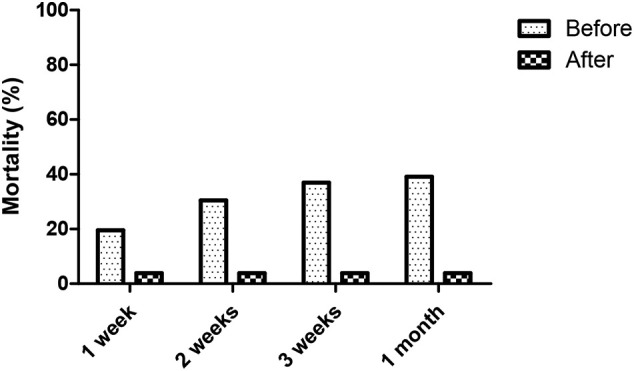
Mortality at 1 week, 2 weeks, 3 weeks and 1 month. All patients in the before group and after group had one month follow-up.
The incidence of VTE, the incidence of acute kidney injury and CRRT, the lowest P/F-ratio and highest SOFA-score during ICU stay, LOS ICU and hospital LOS, and highest bilirubin, AST and ALT during ICU stay, stratified per group are shown in Table 2 .
Table 2.
Outcome parameters.
| Before |
After |
p-Value | |
|---|---|---|---|
| n = 46 | n = 26 | ||
| ARDS (n = 65) | 44 (97.78%) | 16 (80.00%) | 0.03 |
| Acute kidney injury | 30 (65.22%) | 11 (42.31%) | 0.08 |
| Acute heart failure | 8 (17.39%) | 2 (7.69%) | 0.31 |
| Sepsis (n = 69) | 35 (77.78%) | 7 (29.17%) | <0.01 |
| CRRT | 14 (30.43%) | 1 (3.85%) | 0.01 |
| VTE | 19 (41.30%) | 4 (15.38%) | 0.03 |
| LOS (ICU) (days) | 13.00 (7.00, 32.00) | 11.00 (4.00, 20.00) | 0.03 |
| LOS (Hospital) (days) | 21.00 (12.00, 34.00) | 18.50 (13.00, 23.00) | 0.18 |
| Worst P/F ratio (n = 65) | 68.75 (60.00, 85.00) | 79.86 (55.29, 107.42) | 0.55 |
| Worst SOFA score | 11.00 (6.00, 15.00) | 6.00 (4.00, 9.00) | <0.01 |
| Bilirubin | 1.58 (0.78, 3.28) | 0.72 (0.44, 1.16) | <0.01 |
| AST | 123.00 (61.00, 192.00) | 64.50 (45.00, 101.00) | 0.01 |
| ALT | 78.00 (40.00, 169.00) | 62.00 (33.00, 77.00) | 0.05 |
Data are presented as n (%) or as median (25th, 75th percentile). A p-value < 0.05 is considered statistically significant. Fisher's exact is used for binary variables and Wilcoxon rank sum test for continuous variables. Length of stay (LOS) was measured until 3th May 2020 or in case of deceased patients until death.
One patient in the intervention group suffered major haemorrhage after removal of a pleural drain, requiring surgical exploration and transfusion of packed red cells. This patient however was discharged from the ICU one week after the event. Furthermore, no major adverse events were noted.
In the before group, 1 patient received zovirax (2.1%), in the after group 1 patient received remdesivir (3.84%). Almost all patients in the before group (97.8%) received antibiotic treatment while this was 80.8% in the after group. 13 patients in the before group (28.2%) and 6 patients in the after group (23.1%) received corticosteroids during their stay at ICU.
The results of the final 9 multiple regression models are presented in Table 3 . For 1 month mortality, gender, BMI, hypertension, diabetes, white blood cells, D-dimers, SOFA and Apache II score were not statistically significant after univariate logistic regression analysis. Therefore, only age was included in the multiple regression analysis. The final logistic regression model for acute kidney failure incorporated gender, SOFA score at admission and hypertension; the model for CRRT included SOFA score at admission, obesity and D-dimers. The model for VTE included D-dimers. The comparison for lowest P/F ratio was corrected for age; the model for highest SOFA score and bilirubin included gender and SOFA score at admission. The final model for AST included the SOFA score at admission as explanatory variable, ALT was corrected for age, gender and diabetes.
Table 3.
Ratio of geometric means of the before group versus the after group, corrected for explanatory variables (multiple logistic regression models).
| OR (95% CI) | p-Value | |
|---|---|---|
| 1 month mortality | 8.86 (1.46, 53.75) | 0.02 |
| Acute kidney failure | 1.51 (0.45, 5.03) | 0.50 |
| CRRT | 19.21 (1.44, 255.86) | 0.02 |
| VTE | 6.01 (1.13, 32.12) | 0.03 |
| Lowest P/F ratio | 0.89 (0.69, 1.15) | 0.36 |
| Highest SOFA score | 1.58 (1.20, 2.07) | <0.01 |
| Bilirubin | 1.88 (1.17, 3.02) | <0.01 |
| AST | 1.74 (1.11, 2.71) | 0.01 |
| ALT | 1.48 (0.95, 2.32) | 0.08 |
Data are presented as OR (95% CI). A p-value < 0.05 is considered statistically significant. OR: Odds ratio, CI confidence interval.
4. Discussion
In this mono-centric before-after study involving consecutive critically ill COVID patients admitted to the ICU, one-month mortality significantly decreased from 39.13% to 3.85% by applying more aggressive thromboprophylaxis. After correction for group differences, reduction of one-month mortality was statistically significant. The cumulative incidence of VTE was up to 41% in the before group, despite routine thromboprophylaxis with low-dose LMWH. Cumulative incidence of VTE is probably underestimated in the before group since several patients had already died without being screened for VTE.
After implementation of the individualised, more aggressive thromboprophylaxis protocol, incidence of VTE dropped to 15%. After correction for group differences, reduction of cumulative VTE incidence remained statistically significant. After correction for group differences, also the risk of need for CRRT, higher SOFA score and risk of more severe hepatic laboratory abnormalities, were all in favor of the intervention group. No adverse events affecting survival were noted.
These results are in line with the recent finding that high D-dimers on admission can predict in-hospital mortality in patients with COVID-19 [[12], [13], [14]]. The presence of D-dimers suggests significant thrombus formation and breakdown in the body. Thus, D-dimers in fact are a symptom of development of COVID-induced hypercoagulopathy, i.e. the formation of arterial and venous thromboembolism [5]. Therefore, the association between high D-dimers and mortality in COVID-19 pneumonia may be attributed to massive pulmonary embolism (PE) and resulting obstructive shock [21]. This hypothesis is substantiated by recently published clinical reports on the incidence of thromboembolic complications in COVID-19 patients [[22], [23], [24], [25]]. Klok et al. found a 49% cumulative incidence of thrombotic complications in a multicenter cohort of 184 critically ill COVID-19 patients admitted to ICU [22]. All patients received at least standard doses of LMWH [22]. Helms et al. demonstrated a 43% prevalence of thrombotic complications in a French multicenter ICU study of 150 COVID-19 patients despite prophylactic or therapeutic anticoagulation [24]. Moreover, they confirmed that COVID-19 ARDS patients developed significantly more thrombotic complications compared to non-COVID ARDS patients [24]. Poissy et al. reported a 20.6% incidence of PE in a single-center cohort of 107 consecutive COVID-19 patients admitted to ICU, within a median time from ICU admission of 6 days (range 1 to 18 days) [23]. They confirmed that high D-dimers on ICU admission were associated with a higher PE risk [23].
ICU mortality in the before group of this study is comparable to ICU mortality reported in literature.
ICU mortality in a multicenter cohort of 1591 COVID-19 patients admitted to ICU's in Lombardy, Italy, was 26% [26]. Mortality in a small case series of 24 COVID-19 patients admitted to ICU in the Seattle region was even 50% [27].
The high difference in CRRT was in favor of the intervention group and was only attributable to the higher incidence of severe acute kidney injury in the before group. During the whole study period, sufficient CRRT machines were available. The relationship between VTE and AKI is still undetermined. However, massive pulmonary embolism is found to be a strong predictor of AKI [28]. The underlying mechanism is not well understood but may be explained by occurrence of pulmonary hypertension and right heart failure with subsequent hemodynamic instability and renal hypoperfusion [28].
Several pathways of action of LMWH have been described that may explain the observed beneficial effects of increased LMWH dosage in this cohort [29]. The anticoagulant effect of LMWH alone may already explain the reduced risk of VTE and associated death, observed in this study [5,30]. However, LMWH also have potent anti-inflammatory properties as they can decrease the level of inflammatory biomarkers [29,31]. These properties may be very important in light of the growing evidence that an exuberant immune response, i.e. a cytokine storm, may be responsible for a more severe disease course after COVID-19 rather than viral virulence [32]. Last but not least, LMWH may even decrease viral infectivity of SARS-CoV2 by blocking cellular entrance of this virus [33] since Factor Xa has been shown to facilitate SARS-CoV entry into the host cells [34].
On April 11th, the Italian Medicines Agency (AIFA) published a document on heparin use in COVID-19 patients, suggesting doses close to therapeutic ones in severe cases, despite the lack of scientific evidence [35]. We believe that this study provides scientific evidence for this thesis.
Guidelines of the American College of Chest Physicians give a grade 2C recommendation of using LMWH over no prophylaxis but doesn't suggest any LMWH dosage in critically ill patients [8]. After observation of a high prevalence of DVT in our critically ill COVID-19 patients, we decided to increase the prophylactic dose of nadroparin to 3800 IU q12h in all patients and to individually tailor LMWH doses close to therapeutic ranges, based on monitoring plasma anti-Xa activity. Up to date there is no clear-cut evidence that anti-Xa measurements reduce mortality neither morbidity [36]. Therefore, recommendations for the management of LMWH therapy state that routine measurements of anti-Xa activity is not necessary [37]. In contrast, Hirsch already in 2004 suggested that screening of anti-Xa activity might be useful in obese patients, as well as in patients with kidney failure [36]. In light of the high incidence of obesity, kidney failure [12] and VTE in critically ill COVID-19 patients, routine measurements of anti-Xa activity should be considered in this patient group.
Our protocol also included routine duplex ultrasonography of the great veins on admission and twice weekly by a radiologist. The current guidelines however state: ‘in critically ill patients, we suggest against routine ultrasound screening for DVT’ [8]. Ultrasonography performed by radiologist however has an excellent sensitivity (91–95%) and specificity (98%) for detection of DVT [38].
This study has several limitations. The quasi experimental study design is associated with a risk of unidentified confounders. This was the only solution since randomization was not possible because of a need for quick intervention. Due to the mono-centric design of this study, the generalizability of these results may be limited. In the course of the pandemic, we also adapted therapy at other domains. For example, national guidelines for admission to ICU became more stringent in order to secure sufficient ICU capacity. This may explain differences between the two groups, such as age. The relatively small size of this study and the sparsity of the data (especially for mortality and CRRT) results in high odds ratios and wide confidence intervals. The possibility to correct for multiple explanatory variables at the same time is also limited for this reason. Finally, the individualised thromboprophylaxis protocol was implemented on March 31th in all COVID-19 patients admitted to ICU at that time. Therefore, all patients included in the before group who were not discharged from ICU at that time, also received this treatment. This treatment bias however, may have negatively affected differences in outcome.
5. Conclusion
In conclusion, our results suggest that mortality, cumulative risk of VTE and need for CRRT may be reduced in critically ill COVID-19 patients by implementing a more aggressive thromboprophylaxis protocol containing close to therapeutic LMWH dosing, individually tailored with routine anti-Xa measurements combined with systematically ultrasonography screening for DVT. Therefore, we advocate the adoption of this protocol in an attempt to improve clinical outcome in critically ill COVID-19 patients. Future research should focus on confirmation of these results in a randomized design and on uncovering the mechanisms underlying these observations.
List of abbreviations
- ARDS
acute respiratory distress syndrome
- DVT
deep vein thrombosis
- VTE
venous thromboembolism
- ICU
intensive care unit
- LMWH
low-molecular-weight-heparine
- NMBA
neuromuscular blocking agents
- PE
pulmonary embolism
- PEEP
positive end expiratory pressure
- P/F ratio
ratio of arterial oxygen partial pressure to fractional inspired oxygen concentration
- SARS-CoV-2
severe acute respiratory syndrome coronavirus 2
- SD
standard deviation
Ethical approval and consent to participate
This study is approved by the ethical committee of JESSA Hospital Hasselt, Belgium on May 4th 2020. Written informed consent was waived in light of the urgent need to collect data in the ongoing pandemic.
Financial support and sponsorship
The study was be funded solely by departmental funding.
Declaration of competing interest
The authors declare that they have no conflicting interests. The authors affirm that this study has not received any funding/assistance from a commercial organization, and we do not keep any commercial relationships that may lead to a conflict of interests.
Acknowledgments
Acknowledgements
This study is part of the Limburg Clinical Research Program (LCRP) UHasselt-ZOL-Jessa, supported by the foundation Limburg Sterk Merk, province of Limburg, Flemish government, Hasselt University, Ziekenhuis Oost-Limburg and Jessa Hospital.
Authors' contributions
B.S. and J.D. conceived of the study.
B.S., J.D., J.V and M.V. initiated the study design and D.R, L.H,K.V., P.M, helped with implementation.
L.B. was responsible for the statistical analyses.
C.V., L.G, B.P. and I.C. were responsible for the data collection.
B. S, J.V., M.V. and J.O. were responsible for the data interpretation.
All authors were responsible for the writing of the paper and approved the final manuscript.
Presentations
None.
Consent for publication
All authors give consent for publication of the manuscript.
Availability of supporting data
Not applicable.
Trial sponsor
JESSA Hospital dirk.ramaekers@jessazh.be
D. Ramaekers, Medical Director JESSA Hospital, Jessa Hospital – Hasselt, Virga-Jesse Campus, Stadsomvaart 11, 3500 Hasselt, Belgium.
The sponsor had no role in the design of this study and will not have any role during its execution, analyses, interpretation of the data, or decision to submit results.
Appendix A
Additional File 1. COVID protocol in the Jessa Hospital, Hasselt, Belgium
According to this protocol, all patients admitted to our ICU received an IV-infusion with glucose 5% at 60 ml/h as maintenance fluid and stress ulcer prophylaxis with Pantoprazole 40 mg intravenously daily. Prophylactic antibiotic therapy was initiated for 5 days, using Amoxicillin-clavulanic acid 1 g intravenously 4 times a day or Moxifloxacin 400 mg intravenously once daily in case of known allergy to penicillin. The protocol also comprised the administration of hydroxychloroquine orally starting with a loading dose of 400 mg twice on the first day, followed by 200 mg twice a day, for 4 days, with daily monitoring of the QTC-interval by ECG. This therapy was interrupted if the QTC-interval exceeded 500 ms since QTC-interval prolongation is one of the well-known important side-effects of hydroxychloroquine potentially causing severe cardiac arrhythmias.
Ventilatory support was initiated with a high-flow nasal cannula or non-invasive mechanical ventilation as long as the patient was cooperative to this treatment. In case of respiratory fatigue, patients were sedated and intubated and invasive mechanical ventilation was started according to the ARDS-network guidelines that included a protocol based on low-oxygen and high-PEEP. This was based on the first reports that the viral pneumonia caused by SARS-CoV-2 mimicked an ARDS-like pattern [6]. Sedation was performed by a combination of propofol, midazolam and piritramide aiming for the lowest level of sedation by witch the patient would tolerate the mechanical ventilation. Adjustments were made guided by spO2-levels, which are continuously monitored and arterial blood gasses taken every 4 h. In case of hypotension due to vasoplegia, norepinephrine was used as first choice vasopressor.
Additional File 2. Flowchart of the study
References
- 1.Wang D., Hu B., Hu C. Clinical characteristics of 138 hospitalized patients with 2019 novel Coronavirus–infected pneumonia in Wuhan, China. Jama. 2020;323(11) doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Chan JFW, Yuan SY, Kok KH, et al. A familial cluster of pneumonia associated with the 2019 novel coronavirus indicating person-to-person transmission: a study of a family cluster. The Lances. 395(10223):514-523. [DOI] [PMC free article] [PubMed]
- 4.Wu C., Chen X., Cai Y. Risk factors associated with acute respiratory distress syndrome and death in patients with coronavirus disease 2019 pneumonia in Wuhan, China. JAMA Intern Med. 2020;180(7):934–943. doi: 10.1001/jamainternmed.2020.0994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Thachil J., Tang N., Gando S. ISTH interim guidance on recognition and management of coagulopathy in COVID-19. Journal of thrombosis and haemostasis : JTH. May 2020;18(5):1023–1026. doi: 10.1111/jth.14810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Alhazzani W., Møller M.H., Arabi Y.M. Surviving Sepsis Campaign: guidelines on the management of critically ill adults with Coronavirus Disease 2019 (COVID-19) Intensive care medicine. 2020;46:854–887. doi: 10.1007/s00134-020-06022-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.van Halem K., Cox J., Messiaen P. Care for adult non-ICU Covid-19 patients: early experiences from a Belgian tertiary care centre. The Netherlands journal of medicine. Apr 2020;78(3):111–115. [PubMed] [Google Scholar]
- 8.Kahn S.R., Lim W., Dunn A.S. Prevention of VTE in nonsurgical patients: antithrombotic therapy and prevention of thrombosis, 9th ed: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines. Chest. Feb 2012;141(2 Suppl):e195S–e226S. doi: 10.1378/chest.11-2296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schunemann H.J., Cushman M., Burnett A.E. American Society of Hematology 2018 guidelines for management of venous thromboembolism: prophylaxis for hospitalized and nonhospitalized medical patients. Blood advances. Nov 27 2018;2(22):3198–3225. doi: 10.1182/bloodadvances.2018022954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Pellens B.R.M., Van Tornout M., De Mey N., Dubois J., De Pauw I., Ramaekers D., Stessel B. Prevalence of deep venous thrombosis in ventilated COVID-19 patients: a mono-center cross-sectional study. JECCM. 2020 (Article in review) [Google Scholar]
- 11.Wells P.S., Anderson D.R., Rodger M. Evaluation of D-dimer in the diagnosis of suspected deep-vein thrombosis. The New England journal of medicine. Sep 25 2003;349(13):1227–1235. doi: 10.1056/NEJMoa023153. [DOI] [PubMed] [Google Scholar]
- 12.Zhou F., Yu T., Du R. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. Lancet (London, England) Mar 28 2020;395(10229):1054–1062. doi: 10.1016/S0140-6736(20)30566-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Zhang L., Yan X., Fan Q. D-dimer levels on admission to predict in-hospital mortality in patients with Covid-19. Journal of thrombosis and haemostasis: JTH. 2020;18:1324–1329. doi: 10.1111/jth.14859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tang N., Li D., Wang X., Sun Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. Journal of thrombosis and haemostasis : JTH. Apr 2020;18(4):844–847. doi: 10.1111/jth.14768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.WHO. Laboratory Testing for Coronavirus Disease 2019 (COVID-19) in Suspected Human Cases. Interim Guidance. 1-7.
- 16.Knaus W.A., Draper E.A., Wagner D.P., Zimmerman J.E. APACHE II: a severity of disease classification system. Critical care medicine. Oct 1985;13(10):818–829. [PubMed] [Google Scholar]
- 17.Vincent J.L., Moreno R., Takala J. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive care medicine. Jul 1996;22(7):707–710. doi: 10.1007/BF01709751. [DOI] [PubMed] [Google Scholar]
- 18.Khwaja A. KDIGO clinical practice guidelines for acute kidney injury. Nephron. Clinical practice. 2012;120(4):c179–c184. doi: 10.1159/000339789. [DOI] [PubMed] [Google Scholar]
- 19.Force A.D.T., Ranieri V.M., Rubenfeld G.D. Acute respiratory distress syndrome: the Berlin Definition. Jama. Jun 20 2012;307(23):2526–2533. doi: 10.1001/jama.2012.5669. [DOI] [PubMed] [Google Scholar]
- 20.Du R.H., Liang L.R., Yang C.Q. Predictors of mortality for patients with COVID-19 pneumonia caused by SARS-CoV-2: a prospective cohort study. The European respiratory journal. May 2020;55(5) doi: 10.1183/13993003.00524-2020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Creel-Bulos C., Hockstein M., Amin N., Melhem S., Truong A., Sharifpour M. Acute Cor pulmonale in critically Ill patients with Covid-19. N. Engl. J. Med. 2020;382(21):e70. doi: 10.1056/NEJMc2010459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Klok F.A., Kruip M., van der Meer N.J.M. Confirmation of the high cumulative incidence of thrombotic complications in critically ill ICU patients with COVID-19: an updated analysis. Thromb. Res. 2020;191:148–150. doi: 10.1016/j.thromres.2020.04.041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Poissy J., Goutay J., Caplan M. Pulmonary embolism in COVID-19 patients: awareness of an increased prevalence. Circulation. 2020;142:184–186. doi: 10.1161/CIRCULATIONAHA.120.047430. [DOI] [PubMed] [Google Scholar]
- 24.Helms J., Tacquard C., Severac F. High risk of thrombosis in patients with severe SARS-CoV-2 infection: a multicenter prospective cohort study. Intensive Care Med. 2020;46:1089–1098. doi: 10.1007/s00134-020-06062-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Lodigiani C., Iapichino G., Carenzo L. Venous and arterial thromboembolic complications in COVID-19 patients admitted to an academic hospital in Milan, Italy. Thrombosis research. Apr 23 2020;191:9–14. doi: 10.1016/j.thromres.2020.04.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Grasselli G., Zangrillo A., Zanella A. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 Admitted to ICUs of the Lombardy Region, Italy. Jama. 2020;323(16):1574–1581. doi: 10.1001/jama.2020.5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Bhatraju P.K., Ghassemieh B.J., Nichols M. Covid-19 in critically Ill patients in the Seattle Region - case series. N. Engl. J. Med. 2020;382(21):2012–2022. doi: 10.1056/NEJMoa2004500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Chang C.H., Fu C.M., Fan P.C. Acute kidney injury in patients with pulmonary embolism: a population-based cohort study. Medicine. Mar 2017;96(9):e5822. doi: 10.1097/MD.0000000000005822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Thachil J. The versatile heparin in COVID-19. Journal of thrombosis and haemostasis : JTH. May 2020;18(5):1020–1022. doi: 10.1111/jth.14821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Tang N., Bai H., Chen X., Gong J., Li D., Sun Z. Anticoagulant treatment is associated with decreased mortality in severe coronavirus disease 2019 patients with coagulopathy. Journal of Thrombosis and Haemostasis. 2020;18(5):1094–1099. doi: 10.1111/jth.14817. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Mousavi S., Moradi M., Khorshidahmad T., Motamedi M. Anti-inflammatory effects of heparin and its derivatives: a systematic review. Advances in pharmacological sciences. 2015;2015:507151. doi: 10.1155/2015/507151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jamilloux Y., Henry T., Belot A. Should we stimulate or suppress immune responses in COVID-19? Cytokine and anti-cytokine interventions. Autoimmunity reviews. May 3 2020:102567. doi: 10.1016/j.autrev.2020.102567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Belen-Apak F.B., Sarialioglu F. The old but new: can unfractioned heparin and low molecular weight heparins inhibit proteolytic activation and cellular internalization of SARS-CoV2 by inhibition of host cell proteases? Medical hypotheses. Apr 20 2020;142:109743. doi: 10.1016/j.mehy.2020.109743. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Du L., Kao R.Y., Zhou Y. Cleavage of spike protein of SARS coronavirus by protease factor Xa is associated with viral infectivity. Biochemical and biophysical research communications. Jul 20 2007;359(1):174–179. doi: 10.1016/j.bbrc.2007.05.092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ciavarella A., Peyvandi F., Martinelli I. Where do we stand with antithrombotic prophylaxis in patients with COVID-19? Thrombosis research. Apr 20 2020;191:29. doi: 10.1016/j.thromres.2020.04.023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Hirsh J., Raschke R. Heparin and low-molecular-weight heparin: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. Sep 2004;126(3 Suppl):188S–203S. doi: 10.1378/chest.126.3_suppl.188S. [DOI] [PubMed] [Google Scholar]
- 37.N. F. Vol 992. Humana Press; Totowa, NJ: 2013. Anti-factor Xa (Anti-Xa) Assay. [Google Scholar]
- 38.West J.R., Shannon A.W., Chilstrom M.L. What is the accuracy of emergency physician-performed ultrasonography for deep venous thrombosis? Ann Emerg Med. Jun 2015;65(6):699–701. doi: 10.1016/j.annemergmed.2014.06.025. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.