Abstract
Objective
To assess trends in the average costs and effectiveness of the French ultrasound screening programme for birth defects.
Design
A population-based study.
Setting
National Public Health Insurance claim database.
Participants
All pregnant women in the ‘Echantillon Généraliste des Bénéficiaires’, a permanent representative sample of 1/97 of the individuals covered by the French Health Insurance System.
Main outcomes measures
Trends in the costs and in the average cost-effectiveness ratio (ACER) of the screening programme (in € per case detected antenatally), per year, between 2006 and 2014. incremental cost-effectiveness ratio (ICER) from 1 year to another were also estimated. We assessed costs related to the ultrasound screening programme of birth defects excluding the specific screening of Down’s syndrome. The outcome for effectiveness was the prenatal detection rate of birth defects, assessed in a previous study. Linear and logistic regressions were used to analyse time trends.
Results
During the study period, there was a slight decrease in prenatal detection rates (from 58.2% in 2006 to 55.2% in 2014; p=0.015). The cost of ultrasound screening increased from €168 in 2006 to €258 per pregnancy in 2014 (p=0.001). We found a 61% increase in the ACER for ultrasound screening during the study period. ACERs increased from €9050 per case detected in 2006 to €14 580 per case detected in 2014 (p=0.001). ICERs had an erratic pattern, with a strong tendency to show that any increment in the cost of screening was highly cost ineffective.
Conclusion
Even if the increase in costs may be partly justified, we observed a diminishing returns for costs associated with the prenatal ultrasound screening of birth defects, in France, between 2006 and 2014.
Keywords: ultrasound, prenatal diagnosis, health economics
Strengths and limitations of this study.
We observed the chronological trend of the average cost-effectiveness ratio of the French ultrasound birth defect screening programme.
We used two large data sources: national registries of birth defect and the national claims database.
Almost all costs related to the screening were included.
We used a unique effectiveness criterion: the prenatal detection rate of birth defects.
Costs and prenatal detection rates had to be extrapolated from different samples because nationwide data are not available in France.
Introduction
Screening and prevention are not necessarily cost saving or more cost effective than treatment for existing conditions. A review of the cost-effectiveness literature found that ‘distributions of cost-effectiveness ratios for preventive measures and treatments are very similar—in other words, opportunities for efficient investment in healthcare programmes are roughly equal for prevention and treatment’.1
Screening and prevention can reduce morbidity and morbidity by allowing earlier diagnosis; however, this would not necessarily reduce costs if only a small part of the population targeted for prevention is at risk to be affected by a disease or in the absence of treatment. Screening a large population requires important resources, which may be better used to support other healthcare actions.2
All industrialised countries organise programmes for prenatal birth defects screening.3 Indeed, congenital abnormalities represent one of the leading cause of infant mortality, morbidity and long-term adverse developmental outcomes.4–6
So far, medico economic studies of prenatal screening for congenital anomalies have mostly focused on Down’s syndrome screening. Studies of prenatal screening for structural fetal anomalies are rare. Several reviews7 8 have highlighted the limits of the current literature on the subject, whether in comparisons of different modalities of screening9 10 or screening versus no screening.11 12 Moreover, while screening programmes for birth defects are organised in all industrialised countries, the cost-effectiveness (using global costs and prenatal detection rates) of such programmes was never evaluated at a national level. The available data in France suggest that detection rates of birth defects remain stable since the early 2000.13 14
Using national cost claims data and registry-based prenatal detection rates, our objective was to assess trends in the costs and effectiveness (cost per birth defect detected) of the French ultrasound screening programme for birth defects, by estimating both annual average cost-effectiveness ratio (ACER) and incremental cost-effectiveness ratio (ICER) from 1 year to another.
Data and methods
We assessed time trends for costs, efficacy and ACER of the French ultrasound screening programme for structural birth defects, between 2006 and 2014. In France, three ultrasound screening examinations are recommended for singleton pregnancies at 11–14, 20–25 and 30–35 weeks of gestation.15 A screening for birth defects is performed during these examinations, in addition to other analysis (placenta localisation, fetal growth estimation). The scans are performed by certified practitioners and examinations’ reports must include images of standard views following the French guidelines.15 Both public and private centres must comply with these guidelines. When a fetal anomaly is suspected, further examinations are performed by specialists and the parents are referred to prenatal referral centres.16 There are 48 referral centres for prenatal diagnosis in France and its overseas territories, providing full diagnostic workup, parents counselling and fetal treatments. When a severe fetal anomaly is confirmed, termination of pregnancy for fetal abnormality (TOPFA) can be authorised, regardless of the gestational age, according to the French regulations.17 TOFPA may be considered at the request of the parents, if the continuation of the pregnancy seriously endangers the mother’s health, or if there is a high probability for the future born infant to be affected by a severe and incurable disease. In addition to ultrasound screening examinations, a first trimester combined test is offered to all pregnant women for the evaluation of the risk of Down’s syndrome. This combined testing is based on maternal age, maternal biochemical markers and nuchal translucency measurement.18
The outcome for effectiveness was the prenatal detection rate of fetuses with birth defects. Details of the methodology for data sources, definition of cases, exclusion criteria, in particular for cases detected by other modalities than ultrasound and calculation methods have been reported elsewhere.13 The prenatal detection rate was defined as the ratio of the number of cases detected prenatally (positive screening) on the total number of cases per year. A case was defined as a fetus with at least one abnormality whatever the pregnancy outcome was (live birth or fetal loss after 20 WG or following a TOFPA). The screening was considered positive if the fetal anomaly was suspected by ultrasound during the pregnancy regardless the precise diagnosis after birth. For multiple abnormalities, the screening was considered positive if at least one had been detected prenatally. Fetal anomalies where the ones listed by European surveillance of congenital anomalies; EUROCAT (which excludes some minor abnormalities with very low medical or aesthetic impact).19
Cases detected following the first combined test were excluded from the analysis for both costs and efficacy as our aim was to focus on the ultrasound screening programme. However, increased nuchal translucency cases (>99th percentile) were included as these cases are related to the ultrasound screening (high risk for Down’s syndrome and other fetal anomalies, whatever the results of maternal biochemical markers). We used data from three French regional registries of birth defects. These public organisations identify cases with congenital anomalies over a predefined area (usually an administrative region). We included three registries: Auvergne, Paris and La Reunion, all members of EUROCAT, the European network for registries of birth defects20 and using the standards recommended for this purpose. We included the population of women who gave birth (live birth or fetal loss after 20 WG) or following a TOFPA in the areas covered by these three registries, during the study period (2006–2014). We excluded women not resident in those areas. Cost calculations included all procedures performed as part of the screening programme (number of ultrasound screening examinations), further examinations performed following the suspicion of a fetal abnormality and the operating costs of the referral centres. TOPFA costs were not included. We used the public healthcare system perspective and the time horizon of the pregnancy.
Data sources for calculation of costs were as follows:
Costs of procedures
The annual number of procedures for the screening and diagnosis of structural fetal anomalies and their costs were extracted from the national claims database, using the Echantillon Généraliste des Bénéficiaires (EGB), a permanent representative sample of 1/97 of the individuals covered by the French Health Insurance System for employees. This public insurance covered 85% of the total population.21 This claims database is exhaustive and covers the entire French population. The database contains information about procedures’ fees (including extra billing (or balance billing)), either performed in free-standing clinics or in hospital facilities, and cost sharing: costs for the healthcare system, both compulsory and complementary health insurance and out-of-pocket costs for patients. After the third month of pregnancy, all healthcare costs (including transportation) are covered by the statutory health insurance, the only out of pocket costs for patients are the extra billing which private practitioners may charge. Each episode of care is identified by a code. We identified pregnant women using delivery codes. Multiple codes are used for ultrasound examinations performed during the pregnancy, depending on the indication. We included procedures related to the detection of fetal anomalies (systematic first, second and third trimester ultrasound examinations, ultrasound examinations for the monitoring of a known fetal anomaly, fetal echocardiography, and amniocentesis, chorionic villus sampling). Procedures unrelated to the prenatal detection of fetal anomalies (dating ultrasound examinations before 11 WG, ultrasound examinations for fetal growth monitoring including Doppler, fetal well-being evaluation) were excluded. We excluded other imaging procedures (MRI or tomodensitometric examination), due to their very low occurrence in the population.
Infrastructure costs
In addition to the income resulting from their activity (ultrasound examinations, invasive tests and so on), referral centres in charge of prenatal diagnosis and genetic counselling are funded through a specific governmental grant. The annual amount of this grant was retrieved in official governmental data.22
Costs related to the specific screening for Down’s syndrome were excluded from the total costs of the programme. Cost of blood tests using maternal biochemical markers were not included, as well as invasive tests performed after a positive first trimester combined test. Costs of the first-trimester ultrasound scan were included in our cost-effectiveness analysis, even if the screening for Down’s syndrome was a part of these procedures.
We extrapolated the annual costs of procedures computed from the database to the entire population using the ratio of the total number of annual deliveries (data from the official governmental office for statistics, INSEE23) with the annual number of deliveries in the EGB sample. By adding the infrastructure costs, we estimated the total annual costs of the screening programme.
The ACER was estimated in € per case detected antenatally, for each year, as follow:
ACERt=Ct/(Nt * Rt * p)
Where:
t was the year.
C were costs of the ultrasound screening programme for birth defects.
N was the number of deliveries.
R was the prenatal detection rate for birth defects.
p was the prevalence of birth defects.
N was extracted from the official governmental office for statistics, INSEE.23 R and p were estimated from the dataset of the birth defects registers and previously published.13 p was the average prevalence of birth defects for the study period in the areas covered by the birth defects registers.
In addition to the ACERs, we estimated ICERs by comparing the costs and detection rates from 1 year to the next: ICERt = (Ct−Ct−1)/((Nt* Rt* p)−(Nt−1* Rt−1* p).
We used a simple linear regression to assess time trends in the costs of ultrasound screening and in the cost-effectiveness ratios. We used logistic regression for looking at trends in the odds of prenatal diagnosis. All analyses were conducted using Stata V.14.0 software (StataCorp, College Station, Texas, USA).
Results
In the 1/97 sample of the national claims database, we included a total number of 57 104 pregnant women for the 2006–2014 period, ranging from 5888 to 6882 per year (0.7% to 0.8% of the total number of deliveries in France). The mean maternal age in our sample was similar to that of the national population of pregnant women with the same trend of increase age during the study period (from 29.5 to 29.8 years old).24 In our sample, the proportion of multiple pregnancies was 1.6% during the study period.
The number of ultrasound screening examinations increased by approximately 20%, from 2.47 per pregnancy in 2006 to 2.98 in 2014 (p=0.005). We observed an increase of all types of ultrasound examinations, especially for the surveillance of fetal anomalies, which increased by three folds during this period (from 0.07 to 0.26 per pregnancy during the study period; p=0.003). The number of fetal echocardiographies increased from 0.01 to 0.03 per pregnancy (p<0.001). The average rate of invasive prenatal diagnosis procedures related to ultrasound screening was 0.016, and this rate did not change significantly over time (p=0.38). This absence of change resulted from two opposite trends: the total number of invasive procedures decreased during the study period (from 0.08 to 0.04 per pregnancy, p<0.001) but the estimated proportion of invasive procedures related to the ultrasound screening progressively increased over time (from 22% to 38% of the total number of invasive tests).
Ultrasound procedures were performed either in the private sector (by self-employed sonographers or in private for profit hospitals) or in public hospitals in 82% and 18%, respectively. Invasive procedures were mostly performed in public hospitals (65% of the total number).
The official fee for screening ultrasound was revised between 2007 and 2008, leading to a 19%, 15% and 27% increase in costs for the first, second and third trimester ultrasound scans, respectively. In addition, the proportion of the first, second and third trimester ultrasound procedures with extra billing increased during the study period from 40.5% to 46.5%, 40.9% to 47.1% and 42.8% to 44.6%, respectively. The average value of extra billing also increased for all ultrasound procedures during the study period. Consequently, the overall ultrasound screening procedures cost progressively increased during the study period from €139 685 270 in 2006 to €195 310 309 in 2014 (table 1). Detailed costs for first, second and third trimester screening ultrasound scans are available as online supplementary data.
Table 1.
Estimated total cost (and its repartition between procedures cost and infrastructure cost) and cost per pregnancy of the French ultrasound screening programme for birth defect between 2006 and 2014
| Total cost (€) | Including | Cost per pregnancy (€) | ||
| Procedures cost (€) | Infrastructure cost(€) | |||
| 2006 | 139 685 270 | 139 685 270 | 0 | 168 |
| 2007 | 147 545 154 | 137 655 154 | 9 890 000 | 180 |
| 2008 | 194 943 053 | 185 053 053 | 9 890 000 | 235 |
| 2009 | 194 453 720 | 184 563 720 | 9 890 000 | 236 |
| 2010 | 196 517 392 | 186 627 392 | 9 890 000 | 236 |
| 2011 | 195 435 422 | 185 545 422 | 9 890 000 | 237 |
| 2012 | 198 731 955 | 186 161 955 | 12 570 000 | 242 |
| 2013 | 203 869 909 | 188 959 909 | 14 910 000 | 251 |
| 2014 | 210 790 309 | 195 310 309 | 15 480 000 | 258 |
bmjopen-2019-036566supp001.pdf (178KB, pdf)
The specific governmental grant to referral centres was created in 2007, accounting for 6.7% of the total cost of the screening programme. Due to an increase since 2012, this grant accounted for 7.3% of the total costs in 2014 (table 1).
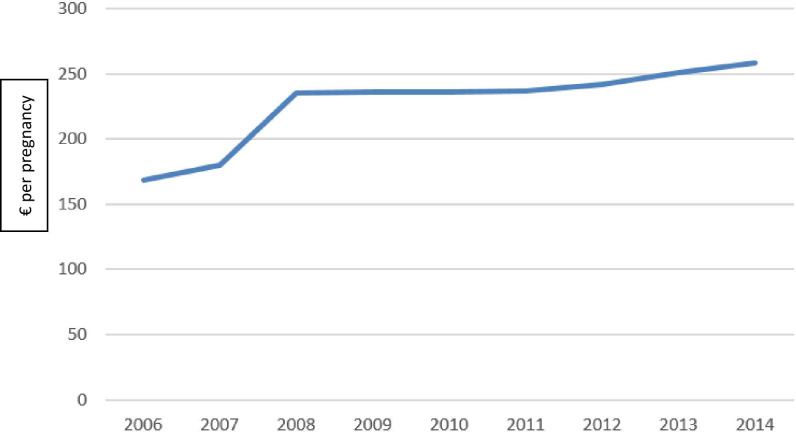
The overall cost of the screening programme in France was estimated to be €210 790 000, in 2014. Ultrasound examinations accounted for 99.5% of the total procedural costs (0.5% for invasive procedures). Seventy seven per cent of the total cost was covered by the public health insurance and 23% by patients (out-of-pocket or complementary insurance). Costs per pregnancy, total costs and their distribution are detailed in table 1, for the 2006–2014 period. The cost of screening increased from €168 in 2006 to €258 per pregnancy in 2014 (p=0.001) (figure 1).
Figure 1.
Chronological evolution of the cost per pregnancy (€) of the French ultrasound screening programme for birth defects.
We identified 15 989 cases of fetal anomalies (average: 1777 per year (range: 1661–1869)) from the registries between 2006 and 2014. The prevalence of birth defects during the study period was 3.2% and was fairly stable. The overall prenatal detection rate of birth defects following ultrasound screening during the study period was 57.0%, ranging from 53.9% (in 2012) to 58.7% (in 2008). The logistic regression found a small declining trend for the overall prenatal detection rate during the study period (OR=0.985; 95% CI 0.972 to 0.997; p=0.015).
ACER’s and ICER’s values for the 2006–2014 period are presented in table 2. Prevalence for birth defects was considered as stable during the study period (3.3%). We found a 61% increase in the ACER for ultrasound screening during the study period. ACER increased from €9050 per case detected in 2006 to €14 580 per case detected in 2014 (p=0.001). ICERs had an erratic pattern, with a strong tendency to show that any increment in the cost of screening was highly cost ineffective.
Table 2.
Evolution of the average cost-effectiveness ratio (ACER) and incremental cost-effectiveness ratio (ICER) of the French ultrasound screening programme for birth defect between 2006 and 2014, and parameters used for its calculation
| Total costs (€) | Detection rate (%) | Number of deliveries | ACER (€/detected case) | Difference in costs (€) | Difference in detected cases (n) | ICER (€/detected case) |
|
| 2006 | 139 685 270 | 58.2 | 829 352 | 9050 | |||
| 2007 | 147 545 154 | 57.9 | 818 705 | 9723 | 7 859 884 | −259 | Dominated |
| 2008 | 194 943 053 | 58.7 | 828 404 | 12 518 | 47 397 899 | 398 | 119 111 |
| 2009 | 194 453 720 | 57.2 | 824 641 | 12 886 | −489 333 | −482 | 1016 |
| 2010 | 196 517 392 | 57.2 | 832 799 | 12 901 | 2 063 672 | 142 | 14 549 |
| 2011 | 195 435 422 | 57.8 | 823 394 | 12 843 | −1 081 970 | −16 | 66 599 |
| 2012 | 198 731 955 | 53.9 | 821 047 | 14 026 | 3 296 533 | −1048 | Dominated |
| 2013 | 203 869 909 | 57.3 | 811 510 | 13 713 | 5 137 954 | 699 | 7355 |
| 2014 | 210 790 309 | 55.2 | 818 565 | 14 580 | 6 920 400 | −409 | Dominated |
ACERs calculation using the formula: ACERt=Ct/(Nt*Rt*p). Where: t was the year, C were costs of the ultrasound screening programme for birth defects (total costs), N was the number of deliveries, R was the prenatal detection rate for birth defects (detection rate), p was the total prevalence of birth defects (p=3.3%, for all the study period). ICERs calculation using the formula: ICERt=(Ct−Ct-1)/((Nt* Rt*p)−(Nt−1* Rt−1* p).
Discussion
Our study represents this first analysis of the French prenatal screening programme for structural birth defects (excluding Down’s syndrome). We found a trend of decreasing cost-effectiveness from €9050 in 2006 to €14 580 in 2014 (+62%) per case detected. This decrease in efficiency was essentially explained by the increase in costs even if we also found a small decrease in prenatal detection rates. Screening costs per pregnancy increased from €168 in 2006 to €258 in 2014, which was due to an increase in both the number and in the unit costs of ultrasound scans performed for prenatal detection of birth defects. Both the frequency and amounts of extra billing (or balance billing) also increased during the study period. To our knowledge, this is the first study to report on the efficiency of a national ultrasound screening programme for birth defects.
We chose to use both ACERs and ICERs. Indeed, our objective was not to compare the current screening policy to alternatives in order to maximise health benefits but to provide information on how much money is expected to be spent on average per identification of congenital anomaly. The use of ACER allowed simple comparisons from 1 year to another.
We have previously reported the prenatal detection rate of birth defects in France, between 2006 and 2014, using data from regional birth defects registers,13 all members of EUROCAT, the European network for registries of birth defects.20 This detection rate was 57.0%, and is covered the cases prenatally detected by ultrasound scans only (other screening modalities such as first trimester combined test with NT<99th were systematically excluded). The logistic regression even found a decreasing trend for the overall prenatal detection rate during the study period (OR=0.985; 95% CI 0.972 to 0.997; p=0.015). In parallel, the costs of the ultrasound screening programme for birth defects sharply increased from €168 in 2006 to €258 in 2014. Several factor could account for the increase in the costs of the ultrasound screening programme. The creation of a governmental grant for the referral centres of prenatal diagnosis in 2007 (and its revaluation since 2012) accounted for 21% of this increase. The official fee for screening ultrasounds was re-evaluated in 2008, which comprised 60% of the increase in costs. The remaining 19% were mostly due to the progressive increase in the number of ultrasound examinations per pregnancy, as well as the increase in extra billing practices. We observed a 51% increase of the total costs of the screening programme, comparatively, the healthcare spending increased by 24% between 2006 and 2014.25
As for all screening programmes, prenatal screening for fetal anomalies raises two issues, that is, its relevance and its cost effectiveness. The relevance of ultrasound screening programmes for birth defects is no longer a matter of debate. For certain birth defects, a prenatal diagnosis allows for optimal surveillance of pregnancies and perinatal management, in particular a delivery in an adequate maternity unit to improve postnatal management,26–30 and for some birth defects, administration of prenatal treatments (eg, fetal surgery).31 32 Moreover, in cases of severe fetal anomalies, parents may opt for termination of pregnancy, depending on their country’s legislation.33 Our goal was not to present results which could be directly extrapolated to other countries, with different screening programmes or even to countries with a three-scan strategy, because the uptake and performance of screening would differ. We suggest, however, that the cost-effectiveness of such programmes must be investigated. The number and timing of recommended screening ultrasound scans during pregnancy is the main difference between screening programmes across countries. In Europe, up to three screening ultrasound scans are usually reimbursed to low-risk pregnant women, but some countries recommend one (eg, Denmark, Finland and Sweden), two (eg, England, Belgium and Switzerland) and others three (eg, France, Italy and Spain) scans.3 In our study, the ultrasounds accounted for the majority of the screening programme costs. Hence differences in the number and fees of ultrasounds can result in substantial differences in the costs of one screening programme from another. In addition, several parameters may impact the number of ultrasound and overall programmes cost, including population coverage, false positive rates and care pathways for positive screenings. Detection rates may also vary from one programme to another. Indeed, several studies have shown significant variations among countries in prenatal detection rates for specific birth defects.34–37 To our knowledge, only one cost-effectiveness study attempted to compare different programmes of routine antenatal ultrasound screening, by modelling the number and the time of ultrasound screening examinations offered during pregnancy.10 However, the authors acknowledged that, due to uncertainty about both costs and effectiveness of ultrasound examinations, their model had important limitations.
There are several limitations to our study that should be addressed. First, the methodological choice of ACER is not recommended in economic evaluations. We justified our choice by the absence of a decisional policy context. We complemented the ACERs by an estimate of ICERS, which are not exactly a decision tool since no decision was planned, but rather an observation of how unplanned decisions by professionals resulted in highly ineffective allocation of resources. Second, costs and prenatal detection rates had to be extrapolated from different samples because nationwide data are not available in France. However, we do not believe that this difference between our study populations could have biased our results. Thus, prenatal diagnosis practices and policies are decided at a national level. Moreover, referral centres for prenatal diagnosis are equitably distributed across the country. Third, due to a modification in the calculation of the hospital’s funding between 2006 and 2009, the number of ultrasound examinations performed during this period in public hospitals was not recorded and had to be estimated. However, when we applied our estimation method on the 2010–2014 period and compared the estimated total costs to the real total cost (computed with the database), we noticed only small differences (between −2.7% and +0.8% depending on the year). In addition, public hospitals accounted for less than 20% of all ultrasound examinations performed, thus limiting a potential bias resulting from the estimation. Fourth, some inputs related to the screening were not included in the costs estimation. Some procedures indicated after a positive screening (fetal MRI and fetal treatment) do not have specific codes and could therefore not be identified. Consequently, our global cost estimation is slightly underestimated. Nevertheless, according to the French Agency of Biomedecine reports, such procedures are performed infrequently and the related cost should be insignificant.38 Fifth, the ultrasound prenatal screening not only focuses on the detection of fetal anomalies. Thus, other complications such as multiple gestation, placenta praevia or fetal growth anomalies might be found and contribute to the overall benefit of the screening. Some of these anomalies are more severe than others and their detection will radically change the management of the pregnancy. Our data does not allow identification these cases and therefore a precise quantification of the potential benefits in relation to costs. Sixth, we did not include the costs of subsequent decisions following the detection of birth defects, as we did not want to assume that the objective of the screening programme was to increase the number of terminations or to reduce the costs associated with postnatal care. And finally, although not specifically monitored, the increase in ultrasound scans is likely to increase non healthcare costs to women, with additional travel cost, time costs and missed days of work.
Conclusion
In France, between 2006 and 2014, the increase in costs of the ultrasound screening for birth defects was not associated to an increase in its efficacy. The observational design of our study limits the interpretation of the observed trends, especially for the increase in costs, which may be partly justified. Official fees for screening ultrasounds need to be revaluated to obtain fair pricing. However, we observed a clear diminishing returns for costs associated with prenatal ultrasound screening of birth defects, in France. An international comparison of ultrasound screening programmes for birth defects could be conducted if similar studies were performed in other countries.
Supplementary Material
Acknowledgments
Authors express their gratitude to the team of the birth defect registers which participate to the study and to the reviewers for their very constructive and insightful comments.
Footnotes
Contributors: CF, BK, J-MJ and ID-Z conceptualised the study and designed the analysis. FD was certified for the EGB database access. CF, FD, BK, HR and IP collected the data. All authors contributed to perform the analysis. CF, FD, BK, J-MJ and ID-Z wrote the final manuscript. J-MJ and ID-Z were the guarantor of the study.
Funding: The authors have not declared a specific grant for this research from any funding agency in the public, commercial or not-for-profit sectors.
Competing interests: None declared.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting or dissemination plans of this research.
Patient consent for publication: Not required.
Ethics approval: It was a time-trend observation on aggregated data, provided by an analysis on anonymised individual data, and didn’t require ethical approval. The EGB analysis, part of the SNIIRAM and property of the CNAMTS, was performed after INSERM approval and is covered by the Commission Nationale de l’Informatique et des Libertés (CNIL) (accords CNIL AT/CPZ/SVT/JB/DP/CR05222O du 14/06/2005 et DP/CR071761 du 28/08/2007).
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: Data are available upon reasonable request. The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
- 1.Cohen JT, Neumann PJ, Weinstein MC. Does preventive care save money? health economics and the presidential candidates. N Engl J Med 2008;358:661–3. 10.1056/NEJMp0708558 [DOI] [PubMed] [Google Scholar]
- 2.Russell LB. The role of prevention in health reform. N Engl J Med 1993;329:352–4. 10.1056/NEJM199307293290511 [DOI] [PubMed] [Google Scholar]
- 3.EUROCAT Central Registry EUROCAT special report: prenatal screening policies in Europe 2010. University of Ulster, 2010. [Google Scholar]
- 4.Lee K, Khoshnood B, Chen L, et al. Infant mortality from congenital malformations in the United States, 1970-1997. Obstet Gynecol 2001;98:620–7. 10.1097/00006250-200110000-00017 [DOI] [PubMed] [Google Scholar]
- 5.Hatton F, Bouvier-Colle MH, Blondel B, et al. [Trends in infant mortality in France: frequency and causes from 1950 to 1997]. Arch Pediatr 2000;7:489–500. 10.1016/s0929-693x(00)89004-1 [DOI] [PubMed] [Google Scholar]
- 6.Matthews TJ, MacDorman MF, Thoma ME. Infant mortality statistics from the 2013 period linked Birth/Infant death data set. Natl Vital Stat Rep 2015;64:1–30. [PubMed] [Google Scholar]
- 7.Ferrier C, Dhombres F, Guilbaud L, et al. [Ultrasound screening for birth defects: A medico-economic review]. Gynecol Obstet Fertil Senol 2017;45:408–15. 10.1016/j.gofs.2017.06.007 [DOI] [PubMed] [Google Scholar]
- 8.Roberts T, Henderson J, Mugford M, et al. Antenatal ultrasound screening for fetal abnormalities: a systematic review of studies of cost and cost effectiveness. BJOG 2002;109:44–56. 10.1111/j.1471-0528.2002.00223.x [DOI] [PubMed] [Google Scholar]
- 9.Vanara F, Bergeretti F, Gaglioti P, et al. Economic evaluation of ultrasound screening options for structural fetal malformations. Ultrasound Obstet Gynecol 2004;24:633–9. 10.1002/uog.1762 [DOI] [PubMed] [Google Scholar]
- 10.Roberts T, Mugford M, Piercy J. Choosing options for ultrasound screening in pregnancy and comparing cost effectiveness: a decision analysis approach. Br J Obstet Gynaecol 1998;105:960–70. 10.1111/j.1471-0528.1998.tb10258.x [DOI] [PubMed] [Google Scholar]
- 11.Long G, Sprigg A. A comparative study of routine versus selective fetal anomaly ultrasound scanning. J Med Screen 1998;5:6–10. 10.1136/jms.5.1.6 [DOI] [PubMed] [Google Scholar]
- 12.Leivo T, Tuominen R, Saari-Kemppainen A, et al. Cost-Effectiveness of one-stage ultrasound screening in pregnancy: a report from the Helsinki ultrasound trial. Ultrasound Obstet Gynecol 1996;7:309–14. 10.1046/j.1469-0705.1996.07050309.x [DOI] [PubMed] [Google Scholar]
- 13.Ferrier C, Dhombres F, Khoshnood B, et al. Trends in resource use and effectiveness of ultrasound detection of fetal structural anomalies in France: a multiple registry-based study. BMJ Open 2019;9:e025482. 10.1136/bmjopen-2018-025482 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Khoshnood B, Lelong N, Lecourbe A, et al. Registre des malformations congénitales de Paris. Surveillance épidémiologique et diagnostic prénatal des malformations : évolution sur 35 ans (1981-2014), 2014. [Google Scholar]
- 15.Sureau C, Henrion R. Rapport du comité national technique de l’échographie de dépistage prénatal, 2005. Available: https://www.cfef.org/archives/lettres/DocusCTE/rapportCTE.pdf [DOI] [PubMed]
- 16.Viossat P, Ville Y, Bessis R, et al. [Report of the French Comité national technique de l'échographie de dépistage prénatal (CNTEDP)--Recommendations for second line prenatal ultrasound]. Gynecol Obstet Fertil 2014;42:51–60. 10.1016/j.gyobfe.2013.11.004 [DOI] [PubMed] [Google Scholar]
- 17.LOI no 2001-588 du 4 juillet 2001 relative l’interruption volontaire de grossesse et la contraception. Available: http://admi.net/jo/20010707/MESX0000140L.html [Accessed 18 Apr 2019].
- 18.Arrêté Du 23 juin 2009 fixant les règles de bonnes pratiques en matière de dépistage et de diagnostic prénatals avec utilisation des marqueurs sériques maternels de la trisomie 21. Available: https://www.legifrance.gouv.fr/affichTexte.do?cidTexte=JORFTEXT000020814373 [Accessed 18 Jun 2019].
- 19.EUROCAT EUROCAT subgroups of congenital anomalies, version 2014, 2014. [Google Scholar]
- 20.Boyd PA, Haeusler M, Barisic I, et al. Paper 1: The EUROCAT network--organization and processes. Birth Defects Res A Clin Mol Teratol 2011;91 Suppl 1:S2–15. 10.1002/bdra.20780 [DOI] [PubMed] [Google Scholar]
- 21.Bezin J, Duong M, Lassalle R, et al. The National healthcare system claims databases in France, SNIIRAM and EGb: powerful tools for pharmacoepidemiology. Pharmacoepidemiol Drug Saf 2017;26:954–62. 10.1002/pds.4233 [DOI] [PubMed] [Google Scholar]
- 22.Agency for hospitals information (ATIH). Available: https://www.atih.sante.fr/dotations-annuelles-migac-et-fir
- 23.French national Institue of statistics and economic studies (INSEE). Available: https://www.insee.fr/fr/statistiques/2381380
- 24.French National Institue of Statistics and Economic Studies (INSEE) Average age of mothers at birth. Available: https://www.insee.fr/fr/statistiques/2381390#tableau-Donnes [Accessed 19 Jun 2019].
- 25.L’évolution de la CSBM depuis 1950. Available: https://drees.solidarites-sante.gouv.fr/IMG/pdf/fiche2-7.pdf [Accessed 25 May 2019].
- 26.Bonnet D, Coltri A, Butera G, et al. Detection of transposition of the great arteries in fetuses reduces neonatal morbidity and mortality. Circulation 1999;99:916–8. 10.1161/01.CIR.99.7.916 [DOI] [PubMed] [Google Scholar]
- 27.Tworetzky W, McElhinney DB, Reddy VM, et al. Improved surgical outcome after fetal diagnosis of hypoplastic left heart syndrome. Circulation 2001;103:1269–73. 10.1161/01.CIR.103.9.1269 [DOI] [PubMed] [Google Scholar]
- 28.Franklin O, Burch M, Manning N, et al. Prenatal diagnosis of coarctation of the aorta improves survival and reduces morbidity. Heart 2002;87:67–9. 10.1136/heart.87.1.67 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Wilson RD, Sogc Genetics Committee Special Contributor ,. Prenatal screening, diagnosis, and pregnancy management of fetal neural tube defects. J Obstet Gynaecol Can 2014;36:927–39. 10.1016/S1701-2163(15)30444-8 [DOI] [PubMed] [Google Scholar]
- 30.Yamamura Y, Swartout JP, Anderson EA, et al. Management of mild fetal pyelectasis: a comparative analysis. J Ultrasound Med 2007;26:1539–43. 10.7863/jum.2007.26.11.1539 [DOI] [PubMed] [Google Scholar]
- 31.Morris RK, Malin GL, Quinlan-Jones E, et al. The percutaneous shunting in lower urinary tract obstruction (PLUTO) study and randomised controlled trial: evaluation of the effectiveness, cost-effectiveness and acceptability of percutaneous vesicoamniotic shunting for lower urinary tract obstruction. Health Technol Assess 2013;17:1–232. 10.3310/hta17590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Adzick NS, Thom EA, Spong CY, et al. A randomized trial of prenatal versus postnatal repair of myelomeningocele. N Engl J Med 2011;364:993–1004. 10.1056/NEJMoa1014379 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Gissler M, Fronteira I, Jahn A, et al. Terminations of pregnancy in the European Union. BJOG 2012;119:324–32. 10.1111/j.1471-0528.2011.03189.x [DOI] [PubMed] [Google Scholar]
- 34.Garne E, Loane M, Wellesley D, et al. Congenital hydronephrosis: prenatal diagnosis and epidemiology in Europe. J Pediatr Urol 2009;5:47–52. 10.1016/j.jpurol.2008.08.010 [DOI] [PubMed] [Google Scholar]
- 35.Pedersen RN, Calzolari E, Husby S, et al. Oesophageal atresia: prevalence, prenatal diagnosis and associated anomalies in 23 European regions. Arch Dis Child 2012;97:227–32. 10.1136/archdischild-2011-300597 [DOI] [PubMed] [Google Scholar]
- 36.Garne E, Eurocat Working Group . Prenatal diagnosis of six major cardiac malformations in Europe--a population based study. Acta Obstet Gynecol Scand 2001;80:224–8. [PubMed] [Google Scholar]
- 37.Boyd PA, Loane M, Garne E, et al. Sex chromosome trisomies in Europe: prevalence, prenatal detection and outcome of pregnancy. Eur J Hum Genet 2011;19:231–4. 10.1038/ejhg.2010.148 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Agence biomédecine Diagnostic prénatal 2014, 2015. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
bmjopen-2019-036566supp001.pdf (178KB, pdf)