Abstract
The global pandemic of COVID-19 has been associated with infections and deaths among health-care workers. This Viewpoint of infectious aerosols is intended to inform appropriate infection control measures to protect health-care workers. Studies of cough aerosols and of exhaled breath from patients with various respiratory infections have shown striking similarities in aerosol size distributions, with a predominance of pathogens in small particles (<5 μm). These are immediately respirable, suggesting the need for personal respiratory protection (respirators) for individuals in close proximity to patients with potentially virulent pathogens. There is no evidence that some pathogens are carried only in large droplets. Surgical masks might offer some respiratory protection from inhalation of infectious aerosols, but not as much as respirators. However, surgical masks worn by patients reduce exposures to infectious aerosols to health-care workers and other individuals. The variability of infectious aerosol production, with some so-called super-emitters producing much higher amounts of infectious aerosol than most, might help to explain the epidemiology of super-spreading. Airborne infection control measures are indicated for potentially lethal respiratory pathogens such as severe acute respiratory syndrome coronavirus 2.
Introduction
The global pandemic of COVID-19, caused by severe acute respiratory syndrome coronavirus 2 [SARS-CoV-2]) has been associated with infections and deaths among health-care workers.1 There have been conflicting recommendations from health authorities on the use of masks or respirators to protect health-care workers.2, 3, 4 When I first reviewed personal respiratory protection against tuberculosis for health-care workers more than 20 years ago,5 there was very little information on infectious aerosols. Since then, colleagues in various disciplines have provided a wealth of data. The purpose of this Viewpoint is to review the scientific literature on the aerosols generated by individuals with respiratory infections, and to discuss how these data inform the optimal use of masks, respirators, and other infection-control measures to protect health-care workers from those aerosols. This is not a review of the literature on the use of surgical masks or respirators, as several have been done already.6, 7, 8, 9, 10, 11
Traditional view of infectious aerosols
Current infection control policies are based on the premise that most respiratory infections are transmitted by large respiratory droplets—ie, larger than 5 μm—produced by coughing and sneezing, then deposited onto exposed fomite or mucosal surfaces.12 Proximity has often been considered a proxy for respiratory droplets,13, 14 reflected by statements such as “Proximity to the index case was associated with transmission which is consistent with droplet spread.”15 Airborne transmission has often been attributed to infectious droplet nuclei produced by the desiccation of suspended droplets, and defined as 5 μm or smaller in size. This has been thought to occur only for tuberculosis and a few other pathogens. Thus, surgical masks have been recommended for use against most respiratory infections.
Particles and plumes
Infectious aerosols are suspensions of pathogens in particles in the air, subject to both physical and biological laws. Particle size is the most important determinant of aerosol behaviour. Particles that are 5 μm or smaller in size can remain airborne indefinitely under most indoor conditions16 unless there is removal due to air currents or dilution ventilation. This same size range of particles (ie, <5 μm) deposits in the lower respiratory tract in humans12, 17 as well as in guinea pigs, mice, and monkeys.18 Particles sized 6–12 μm deposit in the upper airways of the head and neck.18
Sophisticated imaging studies have shown that plumes of aerosols are generated by sneezing or coughing (appendix p 1).19, 20 The aerosol plume contains the highest concentration of particles, which then dissipate in the air over time and distance. That distance is now much farther than previously appreciated, travelling up to 7–8 m.19 A re-analysis21 of the size of particles emitted by an average person that would fall to the ground within 2 m is 60–100 μm, and these can be carried more than 6 m away by sneezing. Obviously, health-care workers doing procedures close to a patient's mouth, such as intubations, bronchoscopies, or dental work can easily be exposed to such aerosol plumes. There is a wide range of particle sizes within the plumes.22 However, the most important questions are whether pathogens are in those plumes and whether their size is consistent with transmission. Studies of cough aerosols and exhaled breath offer answers to those questions.
Cough aerosol studies
Pathogens have been isolated in the aerosols generated by coughing from patients with various respiratory infections. Studies of those that included methods to measure particle sizes have consistently found pathogens in small particles (ie, <5 μm; table 1 ). Other studies without particle size data focused on other outcomes,16, 33, 34 or used methods that could not provide sizing data.35, 36, 37, 38, 39, 40
Table 1.
Summary of studies of infectious aerosols collected from coughs with particle size data
| Pathogen, n/N (%) patients | Containment method and sampling time | Aerosol sampling method | Small particle size range (μm; % of total aerosol) | Median CFU (range) | Comment | |
|---|---|---|---|---|---|---|
| Denver, CO, USA (Fennelly et al, 2004)23 | Mycobacterium tuberculosis, 4/16 (25%) | Plexiglass box, 2 × 5 min | Two Andersen cascade impactors | Most <4·7 | NR (3–633) | Development study: all MDR-TB; no HIV |
| Kampala, Uganda (Fennelly et al, 2012)24 | M tuberculosis, 28/101 (28%) | Stainless steel cylinder: 30 L, 2 × 5 min | Two Andersen cascade impactors | <4·7 (96%) | 16 (1–710) | Feasibility study: 8 (8%) MDR-TB; 49/84 (58%) HIV-positive |
| Cape Town, South Africa (Patterson et al, 2018)25 | M tuberculosis, 15/35 (43%) by culture; 25/27 (93% by PCR) | Custom chamber 1400 L | Andersen cascade impactor and polycarbonate filter | <4·7 (59%) | 2·5 (1–14) | .. |
| Cape Town, South Africa (Theron et al, 2020)26 | M tuberculosis, 142/452 (31%) | 10 L polypropylene chamber; 5 min | One Andersen cascade impactor | <4·7 (60%) | 2–4 (1–310) | .. |
| Brisbane, QLD, Australia (Wainwright et al, 2009)27 | Pseudomonas aeruginosa, 25/28 (89%) | Stainless steel cylinder: 30 L, 2 × 5 min | Andersen cascade impactor | <4·7 (72%) | NR (0–13 485) | .. |
| Brisbane, QLD, Australia (Knibbs et al, 2014)28 | P aeruginosa, 17/18 (94%) at 4 m | Stainless steel distance rig | One Andersen cascade impactor | <4·7 (58%) at 4 m | Mean 14·3 (95% CI 10·9–18·7) for small fraction | .. |
| Morgantown, WV, USA (Lindsley et al, 2010)29 | Influenza A, 32/38 (84%) | Mechanical spirometer (10 L) | NIOSH sampler | <4 (65%) | .. | .. |
| Morgantown, WV, USA (Lindsley et al, 2012)30 | Influenza, N=9 | Mechanical spirometer with HEPA filtered air | Laser particle spectrometer | Average count median diameter 0·63 (SD 0·05) | Average particles per cough 75 400 (SD 97 300) | No viable sampling |
| Winston-Salem, NC, USA (Bischoff et al, 2013)31 | Influenza A and B, 26/61 (43%) | Inpatient rooms and emergency department | Andersen cascade impactor | <4·7 (more than 75% at 1 ft); almost 100% at 6 ft | 5 (19%) emitted 32 times more than others | |
| Sydney, NSW, Australia (Gralton et al, 2013)32 | 23/28 (80%) mixed viruses | Custom unit | Andersen cascade impactor | <4·7 | Not measured | HRV, RSV, influenza A, and parainfluenza |
CFU=colony-forming units. NR=not recorded. MDR-TB=multidrug-resistant tuberculosis. HEPA=high-efficiency particulate air. NIOSH=US National Institute of Occupational Safety and Health. HEPA=high efficiency particulate air. HRV=human rhinovirus. RSV=respiratory syncytial virus.
Key messages.
-
•
Infectious aerosols are particles with potentially pathogenic viruses, bacteria, and fungi suspended in the air, which are subject to the same physical laws as other airborne particulate matter. The biology of the pathogens predicts their airborne survival, infectivity, virulence, and other characteristics.
-
•
Particle size is the most important determinant of aerosol behaviour.
-
•
Small aerosol particles smaller than 5 μm in aerodynamic size are most likely to remain airborne for indefinite periods (unless there is removal due to air currents or dilution ventilation), and to be deposited in the lower respiratory tract.
-
•
Infection control guidelines have stated that most respiratory infections are transmitted by respiratory droplets—ie, particles larger than 5–10 μm in size. Airborne transmission has been attributed to only a few pathogens, notably Mycobacterium tuberculosis, via infectious droplet nuclei that are particles sized 5 μm or smaller. The use of airborne infection isolation rooms and respirator masks has been recommended only to protect against airborne transmission.
-
•
These recommendations have been based on old data and inferences. Over the past two decades, investigators have collected and directly measured the particle sizes of infectious aerosols emitted from individuals with respiratory infections from aerosols generated by cough and from exhaled breath.
-
•
The studies reviewed in this paper consistently show that humans produce infectious aerosols in a wide range of particle sizes, but pathogens predominate in small particles (<5 μm that are immediately respirable by exposed individuals.
-
•
Data are accumulating that severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes COVID-19, is transmitted by both small and large particle aerosols.
-
•
These data suggest that health-care workers should be protected from these potentially infectious aerosols when working in close proximity to patients.
-
•
Some surgical masks might offer respiratory protection compared with not wearing a mask. Filtering facepiece respirators offer more respiratory protection than surgical masks, and powered air purifying respirator (PAPRs) hoods offer the best protection for most health-care settings.
-
•
Face shields can help decrease exposures to and contamination from large particle aerosols, but they do not offer inhalation protection against small particle aerosols.
-
•
PAPRs have built-in eye protection. Surgical masks and other respirators require a face shield or goggles to protect the eyes to prevent infection.
-
•
Masking of patients can help to partly reduce infectious aerosol exposures to health-care workers, but are not a substitute for physical distancing and other infection control measures.
-
•
Aerosolisation of respiratory pathogens is highly variable, at least partly due to the log-normal distribution of infectious aerosols, consistent with so-called super-spreading.
-
•
Airborne infection isolation rooms and other infection control measures against airborne infection are indicated for virulent respiratory pathogens such as SARS-CoV-2.
Tuberculosis
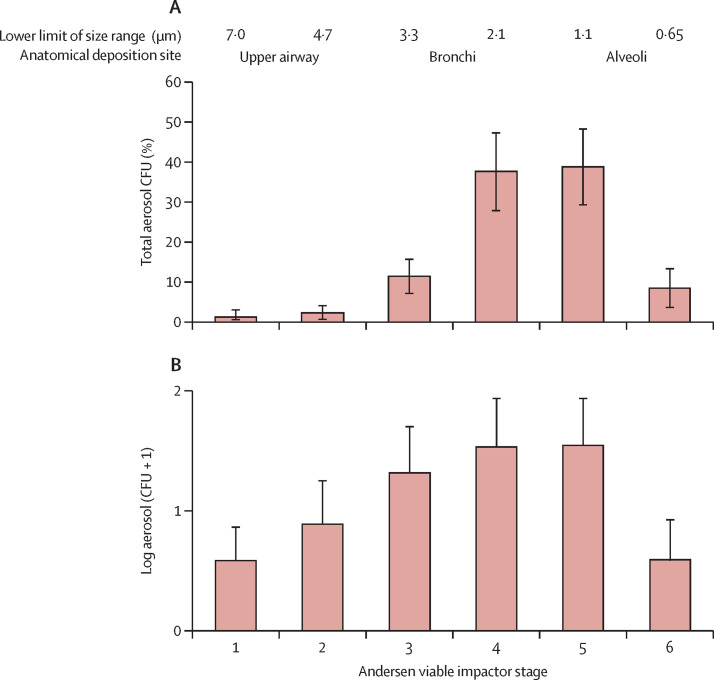
When culturable cough aerosols produced by patients with tuberculosis were directly measured, most (96%) of the culturable Mycobacterium tuberculosis were in particles smaller than 4·7 μm (figure 1 ).24 There were few M tuberculosis in large particles (ie, >7·0 μm) and on settle plates (11% with any colony-forming units [CFU]).24 Culturable cough aerosols from index cases of tuberculosis were found to be the best predictor of new infections of tuberculosis in their household contacts.34 A consistent finding in tuberculosis aerosol studies is the variability of infectious aerosol production from patients with pulmonary tuberculosis.33 These data suggest that a few patients with tuberculosis are infectious via cough aerosols, and some are very infectious,41 coherent with the epidemiological observation of super-spreading.42, 43
Figure 1.
Particle size distributions of cough aerosols from (A) patients with tuberculosis24 and (B) patients with cystic fibrosis infected with Pseudomonas aeruginosa27
(A) Reproduced from Fennelly et al,24 by permission of the American Thoracic Society. (B) Reproduced from Wainwright et al.27 Error bars represent 95% CIs. CFU=colony forming units.
M tuberculosis has also been detected in a 1·4 m3 chamber, using both molecular and culture-based methods.25 Most (59%) of the particles were smaller than 3·3 μm. In the largest study26 of cough aerosols in tuberculosis, almost half of the patients with drug-resistant-tuberculosis generated cough aerosols, and the highest counts of viable bacilli were in the 2·1–4·7 μm size range, consistent with previous studies.24, 25
Cystic fibrosis
Pseudomonas aeruginosa has been collected from cough aerosols in patients with cystic fibrosis.27 These patients generated a particle size distribution that was only slightly larger than that noted in patients with tuberculosis (figure 1). There were relatively few large particles containing bacteria on the settle plates (median 6 CFU) or in a wash of the connecting tubing (1120 CFU, 95% CI 200–6060).27 In a follow-up study, the investigators found that viable P aeruginosa from cough aerosols could travel 4 m and remain culturable for up to 45 min.28
Influenza and other viruses
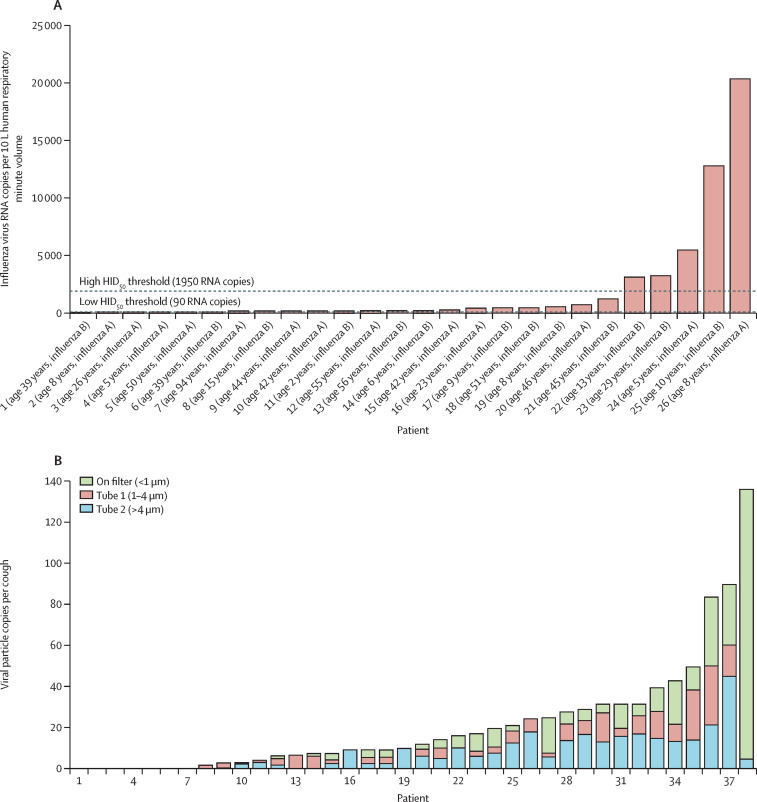
To study the effect of distance, cough aerosols were collected at distances of 1 ft, 3 ft, and 6 ft from 61 patients with influenza (influenza A or influenza B).31 Particles smaller than 4·7 μm were collected at all three sampling sites. At 6 ft (1·83 m), hardly any large particles (ie, ≥4·7 μm) were detected. The magnitude of the influenza aerosol output was log-normally distributed, again coherent with super-spreading (figure 2 ). In a separate study using a different bioaerosol sampler,29 viral RNA was detected in cough aerosols in 38 (81%) of 47 patients with influenza. 35% of the viral RNA was in particles larger than 4 μm, and 65% was in particles sized 4 μm or smaller (figure 2).
Figure 2.
Log-normal distributions of the magnitudes of cough aerosols from patients with influenza using (A) an Andersen cascade impactor31 and (B) a NIOSH two-stage aerosol sampler,29 coherent with super-spreading
(A) Reproduced from Bischoff et al,31 by permission of Oxford University Press. (B) Reproduced from Lindsley et al.29 NIOSH=US National Institute of Occupational Safety and Health. HID50=50% human infectious dose.
In children and adults with upper respiratory infections, PCR assays have detected various viruses.32 During coughing, 82% of participants produced small particles (<4·7 μm) containing virus, versus 57% who produced larger particles.
Exhaled breath aerosol studies
In studies of exhaled breath aerosols with particle size measurements, pathogens were consistently found in small particles (<5 μm; table 2 ). Other studies assayed exhaled breath condensates or filters,38, 48 or used other methods that cannot provide particle size distributions such as direct impaction onto a Petri dish40 or into liquid media.36 However, most particles in exhaled breath are smaller than 4 μm, with a median between 0·7 and 1·0 μm.49
Table 2.
Summary of studies of infectious aerosols collected from exhaled breath with particle size data
| Pathogen, n/N (%) patients | Containment method and sampling time | Aerosol sampling method | Particle size range (μm; % of total aerosol) | Median CFU or viral copies (range) | Comment | |
|---|---|---|---|---|---|---|
| Sydney, NSW, Australia (Gralton et al, 2013)32 | Mixed viruses 31/52 (60%) | Custom unit 10 min | Andersen cascade impactor | <4·7; 25/31 (81%) | Not measured | HRV, RSV, influenza A, and parainfluenza |
| Hong Kong, China (Fabian et al, 2008)44 | Influenza A, 3/5 (60%); Influenza B, 1/7 (14%) | Oronasal face mask 20 min | Teflon filters and optical particle counter | <1; >87% | (<3·2 to 20 viral particles) | |
| Lowell, MA, USA (Milton et al, 2013)45 | 34/37 (92%); 20 influenza A; 17 influenza B | Head inside cone-shaped collector 30 min | Gesundheit-II: sit impactor for coarse fraction; water condenser plus slit impactor for fine fraction | ≤5 (fine fraction): 34/37 (92%) >5 (coarse fraction): 16/37 (43%) |
Maximum viral copies: Fine: 1·3 × 105 Coarse: 2·9 × 104 |
Fine particles contained 8·8 times more virus than coarse particles |
| College Park, MD, USA (Yan et al, 2018)46 | 52/134 (39%) culture positive in fine aerosols; coarse aerosols not cultured | Head inside cone-shaped collector 30 min | Gesundheit-II: slit impactor for coarse fraction; water condenser plus slit impactor for fine fraction | ≤5 (fine fraction): 166/218 (76%) PCR-positive >5 (coarse fraction): 88/218 (40%) PCR-positive |
≤5 (fine fraction): 3·8 × 104 geometric mean RNA copies >5 (coarse fraction): 1·2 × 104 geometric mean RNA copies |
|
| Hong Kong (Leung et al, 2020)47 | Mixed viruses 49/132 (37%) | Head inside cone-shaped collector 30 min | Gesundheit-II: slit impactor for coarse fraction; water condenser plus slit impactor for fine fraction | ≤5 (fine fraction): 4/10 (40%) coronavirus, 19/34 (56%) rhinovirus >5 (coarse fraction): 3/10 (30%) coronavirus, 6/23 (26%) influenza, 9/32 (28%) rhinovirus |
Median log10 copies; ≤5 (fine fraction): coronavirus 0·3, influenza 0·3, rhinovirus 1·8 >5 (coarse fraction): coronavirus 0·3, influenza 0·3, rhinovirus 0·3 |
CFU=colony-forming units. HRV=human rhinovirus. RSV=respiratory syncytial virus.
Several virus types have been detected in exhaled breath condensates using PCR, such as influenza,50, 51, 52 human rhinovirus,50, 52 respiratory syncytial virus,50, 52 cytomegalovirus,53, 54 Epstein-Barr virus,53 human papillomavirus,55 and Torque teno virus.56 Bacteria have also been detected by PCR in exhaled breath condensates, especially Haemophilus influenzae, and also P aeruginosa, Escherichia coli, Stenotrophomonas maltophilia, meticillin-sensitive Staphylococcus aureus, and meticillin-resistant S aureus.57 Viral and bacterial pathogens were isolated from exhaled breath condensates in the same patients, including influenza A, respiratory syncytial virus, S aureus, H influenzae, Legionella pneumophila, and Mycoplasma pneumoniae.58 Cladosporium, Alternaria, and Penicillium species have been detected in the exhaled breath condensates of patients with asthma.59 In a study of exposures to patients with Pneumocystis jirovecii colonies, the exhaled breath was positive by PCR in two (50%) of four critically ill patients and in two (22%) of nine exposed health-care workers with colonies.60
Once direct measurement of particles containing viruses in exhaled breath was technically feasible, most particles (87%) with influenza viral RNA were found to be smaller than 1 μm.44 Exhaled influenza viral generation rates were estimated to be from fewer than 3·2 to 20 virus particles per min. Further developments enabled detection of so-called fine versus coarse particles (ie, ≤5 μm vs >5 μm).45 Influenza viral RNA was detected in the exhaled breath of 34 (92%) of 37 adults.45 The fine particles contained 8·8-times (95% CI 4·1–19·0) more viral copies than did the coarse ones. Respiratory viruses have been found in both coughs (82% of participants) and exhaled breath (81% of participants).32 Similarly, influenza virus was found in similar amounts in coughs (53% of participants) and breath (42% of participants).36 Human rhinovirus was collected more frequently in exhaled breath than in cough aerosols using a filter method.38 Findings from two studies with comparable particle size data showed that influenza virus in exhaled breath is contained in smaller particles than influenza virus in cough (figure 3 ).29, 44
Figure 3.
Proportions of influenza aerosol particles sizes in cough29 and exhaled breath44 sample collections
Data extracted from primary references29, 44 for comparison. Influenza virus in exhaled breath is emitted in smaller particles than influenza virus in cough aerosols.
Three studies did not detect M tuberculosis in exhaled breath condensates.61, 62, 63 In a study of 16 patients with tuberculosis requiring mechanical ventilation, PCR assays of filters in the expired air were positive in 12 (75%).64 However, two studies65, 66 using face-mask sampling from patients with tuberculosis have detected M tuberculosis in exhaled breath. In the first,65 a N95 respirator with a sampling membrane was worn for 5 min. Patients with tuberculosis were instructed to cough, talk, and breathe normally. M tuberculosis-specific RNA, suggesting viability, was detected in all 15 participants. In a more detailed 24-h study of 78 patients with tuberculosis, M tuberculosis was detected more frequently in face-mask samples (86%) than in sputum (21%).66
The most probable mechanism to explain the presence of pathogens in exhaled breath is that the opening of collapsed bronchioles generates aerosols, but there are other theories such as vocal cord closure and vibration.49, 67 These mechanisms might explain transmission from asymptomatic individuals.68 However, there are no data supporting transmission from infectious aerosols in exhaled breath, as most of these studies were focused on diagnostics.
Room air and personal sampling studies
Infectious aerosols have also been collected from room air, suggesting the potential for exposures to health-care workers. Varicella-zoster virus, known to be one of the most contagious viruses, was detected by PCR in the room air of 64 (82%) of 78 patients with varicella and in the room air of nine (70%) of 13 patients with herpes zoster, suggesting airborne transmission.69 Measles is another very infectious virus. Aerosol sampling was done in the room of a young woman with measles at the head of her bed, and at 0·61 m and 0·91 m away from her head (0·91 m=foot of the bed). PCR assays were positive for measles RNA in the particles smaller than 4·7 μm collected at all locations; however, particles larger than 4·7 μm were only positive for virus at the head of the bed. None of the samples were positive by tissue culture.70
M tuberculosis has been detected in hospital air by PCR from settle plates71 and filters.64, 72, 73 In an outpatient clinic in South Africa, M tuberculosis was detected by PCR more frequently from personal air samplers worn by health-care workers (in nine [36%] of 25) than by stationary samplers (in two [8·3%] of 24).74 Influenza virus has been detected using PCR in personal samplers worn by health-care workers and in ambient air samples from an emergency department: 50% of the airborne virus particles were 4 μm or smaller.75 Influenza A was also detected by PCR in 19% of personal samplers and 17% of stationary samplers in an urgent care clinic.76 In the same clinic, respiratory syncytial virus RNA was detected in 38% of the personal samplers and 32% of the stationary samplers; 42% of the particles containing influenza and 9% of the particles containing respiratory syncytial virus were smaller than 4·1 μm. In a smaller study,77 influenza A viral RNA was detected in five (50%) of ten sample collections. Most (four of five) were from particles larger than 4 μm, and one was from particles sized 1–4 μm. In another study,78 six (37·5%) of 16 air samples near patients with influenza were positive by PCR in all particle size ranges tested—ie, smaller than 1 μm, 1–4 μm, and larger than 4 μm.
In a large study79 in a busy inner-city emergency department, influenza was detected in 53 (42%) of 125 personal samplers worn by 30 health-care workers, in 28 (43%) of 96 room air samples, in 23 (76%) of 30 surface samples, and on three (25%) of 12 respirators worn while exposed to a patient with confirmed influenza. In a separate study at a large hospital in China,80 influenza was detected in 15 (79%) of 19 air samples in all particle size ranges (<1 μm, 1–4 μm, and >4 μm). Total influenza virus ranged from 3715 to 119 371 copies per m3. Similar to the study in which influenza virus and respiratory syncytial virus were both detected by PCR,76 viable respiratory syncytial virus was collected from room air near 22 (92%) of 24 infected infants and young children on a general ward and near all ten patients in the intensive care unit; most of the virus was contained in particles smaller than 4·7 μm.81 Human rhinovirus RNA has been isolated from the air of office buildings, but no specific size range was specified.82
Adenovirus was detected by PCR from eight (29%) of 28 air samples in a paediatric ward in Singapore,83 and in 18%84 and 36%85 of air samples from two paediatric emergency departments in Taiwan. Adenovirus DNA was also detected in 21 (77%) of air samples and in 78 (72%) of surface samples in five toilets in the nephrology ward of an Italian hospital.86 M pneumoniae DNA was also detected in 46% of air samples from a paediatric outpatient department in Taiwan.84
P jirovecii DNA has been detected in the room air in multiple studies. The DNA was first isolated from 17 (57%) of 30 rooms of patients with Pneumocystis pneumonia, but was also detected in six (29%) of 21 other hospital rooms.87 A subsequent study found DNA from air samples taken at 1 m from the head of 15 (79·8%) of 19 patients, and in four (33·3%) of 12 of samples taken 8 m away.88 Nosocomial transmission of Pneumocystis has been supported by the finding of air samples positive for the DNA in four (29%) of 14 air samples and in two (22%) of nine health-care workers exposed during bronchoscopy.89 Similarly, air samples were positive for P jirovecii DNA in seven (47%) of 15 critical-care unit rooms, and nine (8·8%) of 102 health-care workers had colonies.60 This study was then extended to the detection of DNA from rooms of patients with Pneumocystis colonies (but without pneumonia).90, 91, 92
Few aerosol data were available from the SARS-CoV pandemic in 2003. In Toronto, air sampling with a slit sampler yielded two of ten samples that were positive for SARS-CoV by PCR but negative on viral culture. Both PCR and cultures were negative on 28 filter samples.93 Retrospective analyses strongly suggested that airborne transmission probably occurred in Hong Kong.94, 95, 96 The Middle East Respiratory Syndrome (MERS) coronavirus was isolated from seven room air samples from dedicated MERS units in two South Korean hospitals. All seven were positive by PCR and four of seven were positive on viral culture.97
Infectious aerosols of SARS CoV-2
Since the outbreak of COVID-19, there has been a question over airborne transmission of SARS-CoV-2. Similar to that seen with SARS-CoV, there was only a mild reduction in viability over a 3-h period in an experimental aerosol generated in a laboratory, consistent with a potential for airborne spread.98 To date, there are no published reports of cough aerosol or exhaled breath sampling from patients with COVID-19, but SARS-CoV-2 has been detected in the air of hospitals in China99 and the USA.100 The virus was detected in both surface and air samples in another hospital in Wuhan, China, with positive PCR tests on 14 (35%) of 40 air samples from the intensive care unit and two (12·5%) of 16 air samples from the general ward.101 It appears that SARS-CoV-2 has the potential to be spread by all modes of transmission: direct contact (ie, person-to-person) and indirect contact (eg, via contaminated objects and aerosol).100 It is not yet clear which mode occurs most frequently. Air sampling for SARS-CoV-2 was negative in three studies,102, 103, 104 but two included small numbers of patients in rooms with high rates of dilution ventilation,102, 103 and one study included a small number of air samples using inefficient impinger devices.104 The outbreaks of COVID-19 in nursing homes,105 choirs,106 and correctional facilities107 are reminiscent of tuberculosis outbreaks and suggestive of both traditional airborne transmission and so-called super-spreading epidemiology.42, 43, 108 Experiments using the golden hamster model have shown 100% efficient aerosol transmission among animals caged separately as well as by direct contact.109
A new paradigm of infectious aerosols
These data show that infectious aerosols from humans exist in a wide range of particle sizes that are strikingly consistent across studies, methods, and pathogens. There is no evidence to support the concept that most respiratory infections are associated with primarily large droplet transmission. In fact, small particle aerosols are the rule, rather than the exception, contrary to current guidelines.12 These small particles occur without a need for a prolonged time to allow for desiccation, and they are of a size that is immediately respirable. These data also add evidence that could update the current dichotomous infection control guidelines, as was proposed 9 years ago.110
The logic that transmission within close proximity defines respiratory droplet spread is fallacious, as small particle aerosols are in the highest concentration close to patients and dissipate with distance. There is epidemiological evidence of an increased risk of tuberculosis transmission within close proximity.111, 112, 113 Individuals sharing a bed with a source patient with tuberculosis are more likely to be infected than people sharing the same room; in turn, people sharing the same room as the source case have a higher risk than individuals in a different room.114, 115, 116 An outbreak associated with an aerosol-generating device used to clean a tuberculous abscess revealed a gradient of tuberculin reactivity, with higher rates among patients in rooms closest to the source case's room.117 Physical distancing decreases transmission potential from pathogens in small particles as well as in large particles, although small particles have a greater capacity to travel further.
The variability of transmission among respiratory pathogens appears to be less dependent on the physical particle size emitted by the diseased person, as current guidelines suggest, but more by biological factors such as the size of the emitted inoculum, the ability of the pathogen to survive desiccation and other stresses of aerosolisation and airborne transport, and environmental factors such as air movement, temperature and humidity, and host defences.
Implications of infectious aerosol data for infection control practice
Because of the large number of patients in health-care settings, health-care workers are likely to have frequent exposures to highly infectious cases. They might also have more cumulative inhaled doses and infections, although it is unknown if this is involved in the pathogenesis of COVID-19. Infection control measures might not only reduce the probability of infection, but might also reduce the size of the inhaled inoculum, which has been associated with disease severity in influenza118, 119 and other diseases.120 This might be especially important for small particle aerosols, as 1 μm aerosols of Bacillus anthracis caused higher mortality in animals than 12 μm aerosols in a seminal study.121
Masks versus respirators
Modelling studies122, 123, 124 and simulated workplace protection studies125, 126, 127 in the USA have shown benefits of various types of respirators and little to no protection from surgical masks. A study in the UK found that surgical masks could reduce inert aerosol exposure by two times, but filtering facepiece respirators reduced the exposure by a factor of 100 or higher.128 In a study of influenza aerosols, surgical masks reduced exposure by an average of six times, but there was a wide range of reduction from 1·1 to 55 times, depending on the design of the mask.129 Two randomised trials130, 131 did not show any benefit of N95 respirators over surgical masks in reducing respiratory illnesses, and two showed that the respirators were protective.132, 133 However, none of the trials used quantitative fit testing, and two had surprisingly low failure rates (1·1–2·6%)132, 133 compared with 60% found in a panel study for the same N95 respirators.134 The low failure rates suggest a problem with fit testing.
Filtering facepiece respirators are only as effective as their fit, as the weak point of these respirators is the face-mask leak.135, 136 Unfortunately, there has been little operational research on the process of fit-testing respirators for health-care workers. There is wide variability among filtering facepiece respirators, and “it may be of more benefit…to wear a respirator model with good-fitting characteristics without fit testing than to wear a respirator model with poor-fitting characteristics after passing a fit-test.”134 Similarly, there are some surgical masks that offer good protection, but as they are not certified or regulated as devices for respiratory protection, it is difficult to know which is the best to use. There is a pressing need for research in this area. Face shields can decrease inhalation exposures to wearers and surface contamination of filtering facepiece respirators by aerosol particles of a median diameter of 8·5 μm by 96% and 97%, respectively, but they only reduce inhalation exposures to smaller particle aerosols of 3·4 μm by 23%.137
Masks to prevent transmission from the wearer
Although surgical masks offer little protection from inhaled agents, they have a role in protecting health-care workers when worn by patients. Placing surgical masks on patients with multidrug-resistant tuberculosis decreased transmission to guinea pigs by 56%,138 and masking of patients with cystic fibrosis reduced P aeruginosa air contamination by 8%.139 Surgical masks reduced the quantity of influenza viral RNA by 2·8 times in small particles and by 25 times in large ones.45 More recently, surgical masks effectively reduced large droplets (>5 μm) of seasonal coronaviruses from three of ten patients to 0 of 11 (p=0·09) and small aerosols (<5 μm) from four of ten patients to 0 of 11 (p=0·04).47 Similarly, surgical masks reduced droplets of influenza from six of 23 to one of 27 (p=0·04). However, the reduction in influenza small aerosols (<5 μm) was not significant. There is mounting evidence suggesting that the wearing of masks can reduce transmission of SARS-CoV-2 in community and health-care settings.140
A major limitation to much of the data on infectious aerosols of viruses is the reliance on PCR findings; few studies have evaluated viability using cell cultures or other methods. Viability itself can be difficult to assess. Aerosolisation from the respiratory tract produces multiple stresses on microbes that can decrease their viability, usually defined by the ability to be cultured. Indoors, desiccation predominates, but temperature, radiation, oxygen, ozone and its reaction products, and other exposures can also damage viral lipids, proteins, and nucleic acids.141 Aerosol sampling itself can produce additional stresses, including mechanical trauma, additional desiccation, and injury in post-sampling processes and extraction.142 PCR assays are usually easier to do logistically than using cell cultures for viral sampling. For example, our group was able to directly sample influenza virus onto monolayers of cell cultures in the laboratory, but this proved impractical for transport to and from clinical sites because of the sensitivity of the cells to spillage and pH stresses.143 These multiple factors, as well as inherent physical inefficiencies of air samplers, suggest that most infectious aerosol data are probably underestimates of the exposures to health-care workers.
Obviously, infectious individuals breathe continuously 24 h per day, but there are no data on possible circadian rhythms or variability in output. By contrast, coughing can be very paroxysmal and sporadic. Although 24-h cough frequency can be measured, it has not been linked to aerosol production. There is only one study of the association between cough aerosol production by tuberculosis index cases and new infections in exposed contacts;34 however, no studies have documented transmission of any respiratory infections exclusively via large respiratory droplets or fomites. Although the data reviewed here indicate that there are small proportions of patients who are highly infectious and probably super-spreaders,42, 43 until a diagnostic test or other method is available to identify them, we must consider all patients with respiratory pathogens as potentially infectious.
Discussion
This Viewpoint suggests that infection control guidelines should be re-evaluated to account for the predominance of small particles within infectious aerosols. Protective devices available to health-care workers have a range of protection, increasing from surgical masks to filtering facepiece respirators to powered air-purifying respirators. Although these are indicated for close encounters, their limitations highlight the need for improved administrative controls, such as more rapid diagnosis and isolation, and the development of vaccines and treatments. These data support calls for the recognition of aerosol (ie, traditional airborne) transmission of SARS-CoV-2.144 This could facilitate the use of enhanced dilution and directional ventilation and other environmental control options—eg, air disinfection with ultraviolet germicidal irradiation,145 which might be especially helpful in congregate settings such as nursing homes. Implementation of improved infection control measures could prevent future morbidity and mortality among health-care workers.
Search strategy and selection criteria
References in the English language for this Viewpoint were identified through searches of PubMed for articles published from 1966 to June, 2020, with the terms “particles”, “health care workers”, “coronavirus”, “COVID-19”, “SARS Co-V-2”, “cough aerosols”, “exhaled breath”, “TB”, “air”, “influenza”, “respirators”, “personal respiratory protection. Some papers known to the author were located using Google Scholar if not found in PubMed.
Contributors
I am funded entirely by the Division of Intramural Research, Pulmonary Branch, National Heart, Lung and Blood Institute (NHLBI), National Institutes of Health (NIH), Bethesda, MD, USA. I conceived this paper, and did all the searches, manuscript drafts, and revisions. No medical writers were used. The views presented here are mine alone and do not represent official policy of the NIH, NHLBI, or any other organisation.
Declaration of interests
I declare no competing interests.
Supplementary Material
References
- 1.Chou R, Dana T, Buckley DI, Selph S, Fu R, Totten AM. Epidemiology of and risk factors for coronavirus infection in health care workers: a living rapid review. Ann Intern Med. 2020 doi: 10.7326/M20-1632. published online May 5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.WHO . World Health Organization; Geneva: March 29, 2020. Modes of transmission of virus causing COVID-19: implications for IPC precaution recommendations.https://www.who.int/publications-detail/modes-of-transmission-of-virus-causing-covid-19-implications-for-ipc-precaution-recommendations [Google Scholar]
- 3.Public Health England Guidance on infection prevention and control for COVID-19. Jan 10, 2020; update May 3, 2020. https://www.gov.uk/government/publications/wuhan-novel-coronavirus-infection-prevention-and-control
- 4.US Centers for Disease Control and Prevention Interim infection prevention and control recommendations for patients with suspected or confirmed coronavirus disease 2019 (COVID-19) in healthcare settings. 2020. https://www.cdc.gov/coronavirus/2019-ncov/hcp/infection-control-recommendations.html
- 5.Fennelly KP. Personal respiratory protection against Mycobacterium tuberculosis. Clin Chest Med. 1997;18:1–17. doi: 10.1016/s0272-5231(05)70352-x. [DOI] [PubMed] [Google Scholar]
- 6.Canadian Agency for Drugs and Technologies in Health; Ottawa: 2014. Respiratory precautions for protection from bioaerosols or infectious agents: a review of the clinical effectiveness and guidelines. CADTH Rapid Response Reports. [PubMed] [Google Scholar]
- 7.Bartoszko JJ, Farooqi MAM, Alhazzani W, Loeb M. Medical masks vs N95 respirators for preventing COVID-19 in health care workers a systematic review and meta-analysis of randomized trials. Influenza Other Respir Viruses. 2020;14:365–373. doi: 10.1111/irv.12745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bin-Reza F, Lopez Chavarrias V, Nicoll A, Chamberland ME. The use of masks and respirators to prevent transmission of influenza: a systematic review of the scientific evidence. Influenza Other Respir Viruses. 2012;6:257–267. doi: 10.1111/j.1750-2659.2011.00307.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Long Y, Hu T, Liu L, et al. Effectiveness of N95 respirators versus surgical masks against influenza: a systematic review and meta-analysis. J Evid Based Med. 2020;13:93–101. doi: 10.1111/jebm.12381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Smith JD, MacDougall CC, Johnstone J, Copes RA, Schwartz B, Garber GE. Effectiveness of N95 respirators versus surgical masks in protecting health care workers from acute respiratory infection: a systematic review and meta-analysis. CMAJ. 2016;188:567–574. doi: 10.1503/cmaj.150835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Verbeek JH, Rajamaki B, Ijaz S, et al. Personal protective equipment for preventing highly infectious diseases due to exposure to contaminated body fluids in healthcare staff. Cochrane Database Syst Rev. 2020;5 doi: 10.1002/14651858.CD011621.pub5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Siegel JD, Rhinehart E, Jackson M, Chiarello L, Health Care Infection Control Practices Advisory Committee 2007 Guideline for isolation precautions: preventing transmission of infectious agents in health care settings. Am J Infect Control. 2007;35:S65–S164. doi: 10.1016/j.ajic.2007.10.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Brankston G, Gitterman L, Hirji Z, Lemieux C, Gardam M. Transmission of influenza A in human beings. Lancet Infect Dis. 2007;7:257–265. doi: 10.1016/S1473-3099(07)70029-4. [DOI] [PubMed] [Google Scholar]
- 14.Tellier R. Transmission of influenza A in human beings. Lancet Infect Dis. 2007;7:759–760. doi: 10.1016/S1473-3099(07)70269-4. [DOI] [PubMed] [Google Scholar]
- 15.Wong TW, Lee CK, Tam W, et al. Cluster of SARS among medical students exposed to single patient, Hong Kong. Emerg Infect Dis. 2004;10:269–276. doi: 10.3201/eid1002.030452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Wells WF. Airborne contagion and air hygiene: an ecological study of droplet infections. Harvard University Press; Cambridge: 1955. Aerodynamics of droplet nuclei; pp. 13–19. [Google Scholar]
- 17.Brown JH, Cook KM, Ney FG, Hatch T. Influence of particle size upon the retention of particulate matter in the human lung. Am J Public Health Nations Health. 1950;40:450–480. doi: 10.2105/ajph.40.4.450. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Harper GJ, Morton JD. The respiratory retention of bacterial aerosols: experiments with radioactive spores. J Hyg. 1953;51:372–385. doi: 10.1017/s0022172400015801. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Bourouiba L. Turbulent gas clouds and respiratory pathogen emissions: potential implications for reducing transmission of COVID-19. JAMA. 2020 doi: 10.1001/jama.2020.4756. published online March 26. [DOI] [PubMed] [Google Scholar]
- 20.Tang JW, Settles GS. Images in clinical medicine. Coughing and aerosols. N Engl J Med. 2008;359:e19. doi: 10.1056/NEJMicm072576. [DOI] [PubMed] [Google Scholar]
- 21.Xie X, Li Y, Chwang AT, Ho PL, Seto WH. How far droplets can move in indoor environments–revisiting the Wells evaporation-falling curve. Indoor Air. 2007;17:211–225. doi: 10.1111/j.1600-0668.2007.00469.x. [DOI] [PubMed] [Google Scholar]
- 22.Bahl P, Doolan C, De Silva C, Chughtai AA, Bourouiba L, Macintyre CR. Airborne or droplet precautions for health workers treating COVID-19? J Infect Dis. 2020 doi: 10.1093/infdis/jiaa189. published online April 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fennelly KP, Martyny JW, Fulton KE, Orme IM, Cave DM, Heifets LB. Cough-generated aerosols of Mycobacterium tuberculosis: a new method to study infectiousness. Am J Respir Crit Care Med. 2004;169:604–609. doi: 10.1164/rccm.200308-1101OC. [DOI] [PubMed] [Google Scholar]
- 24.Fennelly KP, Jones-Lopez EC, Ayakaka I, et al. Variability of infectious aerosols produced during coughing by patients with pulmonary tuberculosis. Am J Respir Crit Care Med. 2012;186:450–457. doi: 10.1164/rccm.201203-0444OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Patterson B, Morrow C, Singh V, et al. Detection of Mycobacterium tuberculosis bacilli in bio-aerosols from untreated TB patients. Gates Open Research. 2018;1:11. doi: 10.12688/gatesopenres.12758.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Theron G, Limberis J, Venter R, et al. Bacterial and host determinants of cough aerosol culture positivity in patients with drug-resistant versus drug-susceptible tuberculosis. Nature Med. 2020 doi: 10.1038/s41591-020-0940-2. published online June 29. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Wainwright CE, France MW, O'Rourke P, et al. Cough-generated aerosols of Pseudomonas aeruginosa and other Gram-negative bacteria from patients with cystic fibrosis. Thorax. 2009;64:926–931. doi: 10.1136/thx.2008.112466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Knibbs LD, Johnson GR, Kidd TJ, et al. Viability of Pseudomonas aeruginosa in cough aerosols generated by persons with cystic fibrosis. Thorax. 2014;69:740–745. doi: 10.1136/thoraxjnl-2014-205213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Lindsley WG, Blachere FM, Thewlis RE, et al. Measurements of airborne influenza virus in aerosol particles from human coughs. PLoS One. 2010;5 doi: 10.1371/journal.pone.0015100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lindsley WG, Pearce TA, Hudnall JB, et al. Quantity and size distribution of cough-generated aerosol particles produced by influenza patients during and after illness. J Occup Environ Hyg. 2012;9:443–449. doi: 10.1080/15459624.2012.684582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bischoff WE, Swett K, Leng I, Peters TR. Exposure to influenza virus aerosols during routine patient care. J Infect Dis. 2013;207:1037–1046. doi: 10.1093/infdis/jis773. [DOI] [PubMed] [Google Scholar]
- 32.Gralton J, Tovey ER, McLaws ML, Rawlinson WD. Respiratory virus RNA is detectable in airborne and droplet particles. J Med Virol. 2013;85:2151–2159. doi: 10.1002/jmv.23698. [DOI] [PubMed] [Google Scholar]
- 33.Acuña-Villaorduña C, Schmidt-Castellani LG, Marques-Rodrigues P, et al. Cough-aerosol cultures of Mycobacterium tuberculosis in the prediction of outcomes after exposure. A household contact study in Brazil. PLoS One. 2018;13 doi: 10.1371/journal.pone.0206384. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Jones-Lopez EC, Namugga O, Mumbowa F, et al. Cough aerosols of Mycobacterium tuberculosis predict new infection: a household contact study. Am J Respir Crit Care Med. 2013;187:1007–1015. doi: 10.1164/rccm.201208-1422OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Huynh KN, Oliver BG, Stelzer S, Rawlinson WD, Tovey ER. A new method for sampling and detection of exhaled respiratory virus aerosols. Clin Infect Dis. 2008;46:93–95. doi: 10.1086/523000. [DOI] [PubMed] [Google Scholar]
- 36.Lindsley WG, Blachere FM, Beezhold DH, et al. Viable influenza A virus in airborne particles expelled during coughs versus exhalations. Influenza Other Respir Viruses. 2016;10:404–413. doi: 10.1111/irv.12390. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Lindsley WG, Noti JD, Blachere FM, et al. Viable Influenza A virus in airborne particles from human coughs. J Occup Environ Hyg. 2015;12:107–113. doi: 10.1080/15459624.2014.973113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Stelzer-Braid S, Oliver BG, Blazey AJ, et al. Exhalation of respiratory viruses by breathing, coughing, and talking. J Med Virol. 2009;81:1674–1679. doi: 10.1002/jmv.21556. [DOI] [PubMed] [Google Scholar]
- 39.Stelzer-Braid S, Tovey ER, Willenborg CM, et al. Absence of back to school peaks in human rhinovirus detections and respiratory symptoms in a cohort of children with asthma. J Med Virol. 2016;88:578–587. doi: 10.1002/jmv.24371. [DOI] [PubMed] [Google Scholar]
- 40.Johnson DF, Druce JD, Birch C, Grayson ML. A quantitative assessment of the efficacy of surgical and N95 masks to filter influenza virus in patients with acute influenza infection. Clin Infect Dis. 2009;49:275–277. doi: 10.1086/600041. [DOI] [PubMed] [Google Scholar]
- 41.Fennelly KP. Variability of airborne transmission of Mycobacterium tuberculosis: implications for control of tuberculosis in the HIV era. Clin Infect Dis. 2007;44:1358–1360. doi: 10.1086/516617. [DOI] [PubMed] [Google Scholar]
- 42.Galvani AP, May RM. Epidemiology: dimensions of superspreading. Nature. 2005;438:293–295. doi: 10.1038/438293a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Lloyd-Smith JO, Schreiber SJ, Kopp PE, Getz WM. Superspreading and the effect of individual variation on disease emergence. Nature. 2005;438:355–359. doi: 10.1038/nature04153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Fabian P, McDevitt JJ, DeHaan WH, et al. Influenza virus in human exhaled breath: an observational study. PLoS One. 2008;3 doi: 10.1371/journal.pone.0002691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Milton DK, Fabian MP, Cowling BJ, Grantham ML, McDevitt JJ. Influenza virus aerosols in human exhaled breath: particle size, culturability, and effect of surgical masks. PLoS Pathog. 2013;9 doi: 10.1371/journal.ppat.1003205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Yan J, Grantham M, Pantelic J, et al. Infectious virus in exhaled breath of symptomatic seasonal influenza cases from a college community. Proc Natl Acad Sci USA. 2018;115:1081–1086. doi: 10.1073/pnas.1716561115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Leung NHL, Chu DKW, Shiu EYC, et al. Respiratory virus shedding in exhaled breath and efficacy of face masks. Nat Med. 2020;26:676–680. doi: 10.1038/s41591-020-0843-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tovey ER, Stelzer-Braid S, Toelle BG, et al. Rhinoviruses significantly affect day-to-day respiratory symptoms of children with asthma. J Allergy Clin Immunol. 2015;135:663–669. doi: 10.1016/j.jaci.2014.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bake B, Larsson P, Ljungkvist G, Ljungström E, Olin AC. Exhaled particles and small airways. Respir Res. 2019;20:8. doi: 10.1186/s12931-019-0970-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Houspie L, De Coster S, Keyaerts E, et al. Exhaled breath condensate sampling is not a new method for detection of respiratory viruses. Virol J. 2011;8:98. doi: 10.1186/1743-422X-8-98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.St George K, Fuschino ME, Mokhiber K, Triner W, Spivack SD. Exhaled breath condensate appears to be an unsuitable specimen type for the detection of influenza viruses with nucleic acid-based methods. J Virol Methods. 2010;163:144–146. doi: 10.1016/j.jviromet.2009.08.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Turchiarelli V, Schinkel J, Molenkamp R, et al. Repeated virus identification in the airways of patients with mild and severe asthma during prospective follow-up. Allergy. 2011;66:1099–1106. doi: 10.1111/j.1398-9995.2011.02600.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Carpagnano GE, Lacedonia D, Natalicchio MI, et al. Viral colonization in exhaled breath condensate of lung cancer patients: possible role of EBV and CMV. Clin Respir J. 2018;12:418–424. doi: 10.1111/crj.12531. [DOI] [PubMed] [Google Scholar]
- 54.Costa C, Bucca C, Bergallo M, Solidoro P, Rolla G, Cavallo R. Unsuitability of exhaled breath condensate for the detection of herpesviruses DNA in the respiratory tract. J Virol Methods. 2011;173:384–386. doi: 10.1016/j.jviromet.2011.02.004. [DOI] [PubMed] [Google Scholar]
- 55.Carpagnano GE, Koutelou A, Natalicchio MI, et al. HPV in exhaled breath condensate of lung cancer patients. Br J Cancer. 2011;105:1183–1190. doi: 10.1038/bjc.2011.354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chikasue K, Kimura M, Ikeda K, et al. Detection of Torque teno virus DNA in exhaled breath by polymerase chain reaction. Acta Med Okayama. 2012;66:387–397. doi: 10.18926/AMO/48963. [DOI] [PubMed] [Google Scholar]
- 57.Zheng Y, Chen H, Yao M, Li X. Bacterial pathogens were detected from human exhaled breath using a novel protocol. J Aerosol Science. 2018;117:224–234. doi: 10.1016/j.jaerosci.2017.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Zakharkina T, Koczulla AR, Mardanova O, Hattesohl A, Bals R. Detection of microorganisms in exhaled breath condensate during acute exacerbations of COPD. Respirology. 2011;16:932–938. doi: 10.1111/j.1440-1843.2011.01977.x. [DOI] [PubMed] [Google Scholar]
- 59.Carpagnano GE, Malerba M, Lacedonia D, et al. Analysis of the fungal microbiome in exhaled breath condensate of patients with asthma. Allergy Asthma Proc. 2016;37:41–46. doi: 10.2500/aap.2016.37.3943. [DOI] [PubMed] [Google Scholar]
- 60.Valade S, Azoulay E, Damiani C, Derouin F, Totet A, Menotti J. Pneumocystis jirovecii airborne transmission between critically ill patients and health care workers. Intensive Care Med. 2015;41:1716–1718. doi: 10.1007/s00134-015-3835-9. [DOI] [PubMed] [Google Scholar]
- 61.Jain R, Schriever CA, Danziger LH, Cho SH, Rubinstein I. The IS6110 repetitive DNA element of Mycobacterium tuberculosis is not detected in exhaled breath condensate of patients with active pulmonary tuberculosis. Respiration. 2007;74:329–333. doi: 10.1159/000101786. [DOI] [PubMed] [Google Scholar]
- 62.Kawada H, Yamazato M, Shinozawa Y, et al. [Exhaled breath-generated aerosols of Mycobacterium tuberculosis in patients with active pulmonary tuberculosis during normal breathing] Kekkaku. 2008;83:387–391. [PubMed] [Google Scholar]
- 63.Schreiber J, Meyer C, Rusch-Gerdes S, et al. Mycobacterium tuberculosis gene-amplification in breath condensate of patients with lung tuberculosis. Eur J Med Res. 2002;7:290–291. [PubMed] [Google Scholar]
- 64.Wan GH, Lu SC, Tsai YH. Polymerase chain reaction used for the detection of airborne Mycobacterium tuberculosis in health care settings. Am J Infect Control. 2004;32:17–22. doi: 10.1016/s0196-6553(03)00090-7. [DOI] [PubMed] [Google Scholar]
- 65.Shaikh A, Sriraman K, Vaswani S, Oswal V, Mistry N. Detection of Mycobacterium tuberculosis RNA in bioaerosols from pulmonary tuberculosis patients. Int J Infect Dis. 2019;86:5–11. doi: 10.1016/j.ijid.2019.06.006. [DOI] [PubMed] [Google Scholar]
- 66.Williams CM, Abdulwhhab M, Birring SS, et al. Exhaled Mycobacterium tuberculosis output and detection of subclinical disease by face-mask sampling: prospective observational studies. Lancet Infect Dis. 2020;20:607–617. doi: 10.1016/S1473-3099(19)30707-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Patterson B, Wood R. Is cough really necessary for TB transmission? Tuberculosis. 2019;117:31–35. doi: 10.1016/j.tube.2019.05.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Rothe C, Schunk M, Sothmann P, et al. Transmission of 2019-nCoV infection from an asymptomatic contact in Germany. N Engl J Med. 2020;382:970–971. doi: 10.1056/NEJMc2001468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Sawyer MH, Chamberlin CJ, Wu YN, Aintablian N, Wallace MR. Detection of varicella-zoster virus DNA in air samples from hospital rooms. J Infect Dis. 1994;169:91–94. doi: 10.1093/infdis/169.1.91. [DOI] [PubMed] [Google Scholar]
- 70.Bischoff WE, McNall RJ, Blevins MW, et al. Detection of measles virus RNA in air and surface specimens in a hospital setting. J Infect Dis. 2016;213:600–603. doi: 10.1093/infdis/jiv465. [DOI] [PubMed] [Google Scholar]
- 71.Vadrot C, Bex V, Mouilleseaux A, Squinazi F, Darbord JC. Detection of Mycobacterium tuberculosis complex by PCR in hospital air samples. J Hosp Infect. 2004;58:262–267. doi: 10.1016/j.jhin.2004.04.026. [DOI] [PubMed] [Google Scholar]
- 72.Mastorides SM, Oechler RL, Greene JN, Sinnott JT, Sandin RL. Detection of airborne Mycobacterium tuberculosis by air filtration and polymerase chain reaction. Clin Infect Dis. 1997;25:756. doi: 10.1086/516953. [DOI] [PubMed] [Google Scholar]
- 73.Zhang B, Wang Z, Tong X. Methods for detecting mycobacterium tuberculosis in the air of wards for tuberculosis patients. Zhonghua Jie He He Hu Xi Za Zhi. 1997;20:101–103. (in Chinese). [PubMed] [Google Scholar]
- 74.Matuka O, Singh TS, Bryce E, et al. Pilot study to detect airborne Mycobacterium tuberculosis exposure in a South African public healthcare facility outpatient clinic. J Hosp Infect. 2015;89:192–196. doi: 10.1016/j.jhin.2014.11.013. [DOI] [PubMed] [Google Scholar]
- 75.Blachere FM, Lindsley WG, Pearce TA, et al. Measurement of airborne influenza virus in a hospital emergency department. Clin Infect Dis. 2009;48:438–440. doi: 10.1086/596478. [DOI] [PubMed] [Google Scholar]
- 76.Lindsley WG, Blachere FM, Davis KA, et al. Distribution of airborne influenza virus and respiratory syncytial virus in an urgent care medical clinic. Clin Infect Dis. 2010;50:693–698. doi: 10.1086/650457. [DOI] [PubMed] [Google Scholar]
- 77.Leung NHL, Zhou J, Chu DKW, et al. Quantification of influenza virus RNA in aerosols in patient rooms. PLoS One. 2016;11 doi: 10.1371/journal.pone.0148669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Yip L, Finn M, Granados A, et al. Influenza virus RNA recovered from droplets and droplet nuclei emitted by adults in an acute care setting. J Occup Environ Hyg. 2019;16:341–348. doi: 10.1080/15459624.2019.1591626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Rule AM, Apau O, Ahrenholz SH, et al. Healthcare personnel exposure in an emergency department during influenza season. PLoS One. 2018;13 doi: 10.1371/journal.pone.0203223. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Shiu EYC, Huang W, Ye D, et al. Frequent recovery of influenza A but not influenza B virus RNA in aerosols in pediatric patient rooms. Indoor Air. 2020 doi: 10.1111/ina.12669. https//:doi.org.10.1111/ina.12669 published online March 23. [DOI] [PubMed] [Google Scholar]
- 81.Kulkarni H, Smith CM, Lee DDH, Hirst RA, Easton AJ, O'Callaghan C. Evidence of respiratory syncytial virus spread by aerosol. Time to revisit infection control strategies? Am J Respir Crit Care Med. 2016;194:308–316. doi: 10.1164/rccm.201509-1833OC. [DOI] [PubMed] [Google Scholar]
- 82.Myatt TA, Johnston SL, Zuo Z, et al. Detection of airborne rhinovirus and its relation to outdoor air supply in office environments. Am J Respir Crit Care Med. 2004;169:1187–1190. doi: 10.1164/rccm.200306-760OC. [DOI] [PubMed] [Google Scholar]
- 83.Yadana S, Coleman KK, Nguyen TT, et al. Monitoring for airborne respiratory viruses in a general pediatric ward in Singapore. J Public Health Res. 2019;8 doi: 10.4081/jphr.2019.1407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Wan G-H, Huang C-G, Huang Y-C, et al. Surveillance of airborne adenovirus and Mycoplasma pneumoniae in a hospital pediatric department. PLoS One. 2012;7 doi: 10.1371/journal.pone.0033974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Tseng CC, Chang LY, Li CS. Detection of airborne viruses in a pediatrics department measured using real-time qPCR coupled to an air-sampling filter method. J Environ Health. 2010;73:22–28. [PubMed] [Google Scholar]
- 86.Verani M, Bigazzi R, Carducci A. Viral contamination of aerosol and surfaces through toilet use in health care and other settings. Am J Infect Control. 2014;42:758–762. doi: 10.1016/j.ajic.2014.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Bartlett MS, Vermund SH, Jacobs R, et al. Detection of Pneumocystis carinii DNA in air samples: likely environmental risk to susceptible persons. J Clin Microbiol. 1997;35:2511–2513. doi: 10.1128/jcm.35.10.2511-2513.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Choukri F, Menotti J, Sarfati C, et al. Quantification and spread of Pneumocystis jirovecii in the surrounding air of patients with Pneumocystis pneumonia. Clin Infect Dis. 2010;51:259–265. doi: 10.1086/653933. [DOI] [PubMed] [Google Scholar]
- 89.Morilla R, Martínez-Rísquez MT, De La Horra C, et al. Airborne acquisition of Pneumocystis in bronchoscopy units: a hidden danger to healthcare workers. Med Mycol. 2018 doi: 10.1093/mmy/myy093. https//:doi.org.10.1093/mmy/myy093 published online Oct 4. [DOI] [PubMed] [Google Scholar]
- 90.Fréalle E, Valade S, Guigue N, et al. Diffusion of Pneumocystis jirovecii in the surrounding air of patients with Pneumocystis colonization: frequency and putative risk factors. Med Mycol. 2017;55:568–572. doi: 10.1093/mmy/myw113. [DOI] [PubMed] [Google Scholar]
- 91.Le Gal S, Pougnet L, Damiani C, et al. Pneumocystis jirovecii in the air surrounding patients with Pneumocystis pulmonary colonization. Diagn Microbiol Infect Dis. 2015;82:137–142. doi: 10.1016/j.diagmicrobio.2015.01.004. [DOI] [PubMed] [Google Scholar]
- 92.Pougnet L, Grall A, Moal M-C, et al. Pneumocystis jirovecii exhalation in the course of Pneumocystis pneumonia treatment. Infect Control Hosp Epidemiol. 2018;39:627–630. doi: 10.1017/ice.2018.45. [DOI] [PubMed] [Google Scholar]
- 93.Booth TF, Kournikakis B, Bastien N, et al. Detection of airborne severe acute respiratory syndrome (SARS) coronavirus and environmental contamination in SARS outbreak units. J Infect Dis. 2005;191:1472–1477. doi: 10.1086/429634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Li Y, Huang X, Yu ITS, Wong TW, Qian H. Role of air distribution in SARS transmission during the largest nosocomial outbreak in Hong Kong. Indoor Air. 2005;15:83–95. doi: 10.1111/j.1600-0668.2004.00317.x. [DOI] [PubMed] [Google Scholar]
- 95.Yu IT, Li Y, Wong TW, et al. Evidence of airborne transmission of the severe acute respiratory syndrome virus. N Engl J Med. 2004;350:1731–1739. doi: 10.1056/NEJMoa032867. [DOI] [PubMed] [Google Scholar]
- 96.Yu ITS, Qiu H, Tse LA, Wong TW. Severe acute respiratory syndrome beyond Amoy Gardens: completing the incomplete legacy. Clin Infect Dis. 2014;58:683–686. doi: 10.1093/cid/cit797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Kim S-H, Chang SY, Sung M, et al. Extensive viable Middle East Respiratory Syndrome (MERS) coronavirus contamination in air and surrounding environment in MERS isolation wards. Clin Infect Dis. 2016;63:363–369. doi: 10.1093/cid/ciw239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and surface stability of SARS-CoV-2 as compared with SARS-CoV-1. N Engl J Med. 2020;382:1564–1567. doi: 10.1056/NEJMc2004973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Liu Y, Ning Z, Chen Y, et al. Aerodynamic analysis of SARS-CoV-2 in two Wuhan hospitals. Nature. 2020;582:557–560. doi: 10.1038/s41586-020-2271-3. [DOI] [PubMed] [Google Scholar]
- 100.Santarpia JL, Rivera DN, Herrera V, et al. Aerosol and surface transmission potential of SARS-CoV-2. medRxiv. 2020 https://doi.org/2020.03.23.20039446 published online June 3. (preprint). [Google Scholar]
- 101.Guo Z-D, Wang Z-Y, Zhang S-F, et al. Aerosol and surface distribution of severe acute respiratory syndrome coronavirus 2 in hospital wards, Wuhan, China, 2020. Emerg Infect Dis. 2020;26:1583–1591. doi: 10.3201/eid2607.200885. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Cheng VC-C, Wong S-C, Chan VW-M, et al. Air and environmental sampling for SARS-CoV-2 around hospitalized patients with coronavirus disease 2019 (COVID-19) Infect Control Hosp Epidemiol. 2020 doi: 10.1017/ice.2020.282. https//:doi.org.10.1017/ice.2020.282 published online June 8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Ong SWX, Tan YK, Chia PY, et al. Air, surface environmental, and personal protective equipment contamination by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) from a symptomatic patient. JAMA. 2020;323 doi: 10.1001/jama.2020.3227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Faridi S, Niazi S, Sadeghi K, et al. A field indoor air measurement of SARS-CoV-2 in the patient rooms of the largest hospital in Iran. Sci Total Environ. 2020;725 doi: 10.1016/j.scitotenv.2020.138401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Roxby AC, Greninger AL, Hatfield KM, et al. Detection of SARS-CoV-2 Among residents and staff members of an independent and assisted living community for older adults—Seattle, Washington, 2020. MMWR Morb Mortal Wkly Rep. 2020;69:416–418. doi: 10.15585/mmwr.mm6914e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Hamner L, Dubbel P, Capron I, et al. High SARS-CoV-2 attack rate following exposure at a choir practice—Skagit County, WA, March 2020. MMWR Morb Mortal Wkly Rep. 2020;69:606–610. doi: 10.15585/mmwr.mm6919e6. [DOI] [PubMed] [Google Scholar]
- 107.Wallace M, Hagan L, Curran KG, et al. COVID-19 in correctional and detention facilities—United States, February–April 2020. MMWR Morb Mortal Wkly Rep. 2020;69:587–590. doi: 10.15585/mmwr.mm6919e1. [DOI] [PubMed] [Google Scholar]
- 108.Riou J, Althaus CL. Pattern of early human-to-human transmission of Wuhan 2019 novel coronavirus (2019-nCoV), December 2019 to January 2020. Euro Surveill. 2020;25 doi: 10.2807/1560-7917.ES.2020.25.4.2000058. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Sia SF, Yan L-M, Chin AWH, et al. Pathogenesis and transmission of SARS-CoV-2 in golden hamsters. Nature. 2020 doi: 10.1038/s41586-020-2342-5. https//:doi.org.10.1038/s41586-020-2342-5 published online May 14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Gralton J, Tovey E, McLaws ML, Rawlinson WD. The role of particle size in aerosolised pathogen transmission: a review. J Infect. 2011;62:1–13. doi: 10.1016/j.jinf.2010.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kenyon TA, Valway SE, Ihle WW, Onorato IM, Castro KG. Transmission of multidrug-resistant Mycobacterium tuberculosis during a long airplane flight. N Engl J Med. 1996;334:933–938. doi: 10.1056/NEJM199604113341501. [DOI] [PubMed] [Google Scholar]
- 112.MacIntyre CR, Carnie J, Randall M. Risk of transmission of tuberculosis among inmates of an Australian prison. Epidemiol Infect. 1999;123:445–450. doi: 10.1017/s095026889900312x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Mandalakas AM, Kirchner HL, Lombard C, et al. Well-quantified tuberculosis exposure is a reliable surrogate measure of tuberculosis infection. Int J Tuberc Lung Dis. 2012;16:1033–1039. doi: 10.5588/ijtld.12.0027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Acuna-Villaorduna C, Jones-Lopez EC, Fregona G, et al. Intensity of exposure to pulmonary tuberculosis determines risk of tuberculosis infection and disease. Eur Respir J. 2018;51 doi: 10.1183/13993003.01578-2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.McIntosh AI, Doros G, Jones-Lopez EC, et al. Extensions to Bayesian generalized linear mixed effects models for household tuberculosis transmission. Stat Med. 2017;36:2522–2532. doi: 10.1002/sim.7303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Lutong L, Bei Z. Association of prevalence of tuberculin reactions with closeness of contact among household contacts of new smear-positive pulmonary tuberculosis patients. Int J Tuberc Lung Dis. 2000;4:275–277. [PubMed] [Google Scholar]
- 117.Hutton MD, Stead WW, Cauthen GM, Bloch AB, Ewing WM. Nosocomial transmission of tuberculosis associated with a draining abscess. J Infect Dis. 1990;161:286–295. doi: 10.1093/infdis/161.2.286. [DOI] [PubMed] [Google Scholar]
- 118.Alford RH, Kasel JA, Gerone PJ, Knight V. Human influenza resulting from aerosol inhalation. Proc Soc Exp Biol Med. 1966;122:800–804. doi: 10.3181/00379727-122-31255. [DOI] [PubMed] [Google Scholar]
- 119.Hemmink JD, Morgan SB, Aramouni M, et al. Distinct immune responses and virus shedding in pigs following aerosol, intra-nasal and contact infection with pandemic swine influenza A virus, A(H1N1)09. Vet Res. 2016;47:103. doi: 10.1186/s13567-016-0390-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Fennelly KP, Jones-Lopez EC. Quantity and quality of inhaled dose predicts immunopathology in tuberculosis. Front Immunol. 2015;6:1–13. doi: 10.3389/fimmu.2015.00313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Druett HA, Henderson DW, Packman L, Peacock S. Studies on respiratory infection. I. The influence of particle size on respiratory infection with anthrax spores. J Hyg (Lond) 1953;51:359–371. doi: 10.1017/s0022172400015795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Fennelly KP, Nardell EA. The relative efficacy of respirators and room ventilation in preventing occupational tuberculosis. Infect Control Hosp Epidemiol. 1998;19:754–759. doi: 10.1086/647719. [DOI] [PubMed] [Google Scholar]
- 123.Gammaitoni L, Nucci MC. Using a mathematical model to evaluate the efficacy of TB control measures. Emerg Infect Dis. 1997;3:335–342. doi: 10.3201/eid0303.970310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Nicas M. Respiratory protection and the risk of Mycobacterium tuberculosis infection. Am J Indust Med. 1995;27:317–333. doi: 10.1002/ajim.4700270302. [DOI] [PubMed] [Google Scholar]
- 125.Duling MG, Lawrence RB, Slaven JE, Coffey CC. Simulated workplace protection factors for half-facepiece respiratory protective devices. J Occup Environ Hyg. 2007;4:420–431. doi: 10.1080/15459620701346925. [DOI] [PubMed] [Google Scholar]
- 126.Elmashae RBY, Grinshpun SA, Reponen T, Yermakov M, Riddle R. Performance of two respiratory protective devices used by home-attending health care workers (a pilot study) J Occup Environ Hyg. 2017;14:D145–D149. doi: 10.1080/15459624.2017.1319571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Gao S, Koehler RH, Yermakov M, Grinshpun SA. Performance of facepiece respirators and surgical masks against surgical smoke: simulated workplace protection factor study. Ann Occup Hyg. 2016;60:608–618. doi: 10.1093/annhyg/mew006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Gawn JM, Clayton M, Makison C, Crook B. Health and Safety Executive; Norwich, UK: 2008. Evaluating the protection afforded by surgical masks against influenza bioaerosols. [Google Scholar]
- 129.Makison Booth C, Clayton M, Crook B, Gawn JM. Effectiveness of surgical masks against influenza bioaerosols. J Hosp Infect. 2013;84:22–26. doi: 10.1016/j.jhin.2013.02.007. [DOI] [PubMed] [Google Scholar]
- 130.Loeb M, Dafoe N, Mahony J, et al. Surgical mask vs N95 respirator for preventing influenza among health care workers. JAMA. 2009;302:1865–1871. doi: 10.1001/jama.2009.1466. [DOI] [PubMed] [Google Scholar]
- 131.Radonovich LJ, Simberkoff MS, Bessesen MT, et al. N95 respirators vs medical masks for preventing influenza among health care personnel. JAMA. 2019;322:824–833. doi: 10.1001/jama.2019.11645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.MacIntyre CR, Wang Q, Cauchemez S, et al. A cluster randomized clinical trial comparing fit-tested and non-fit-tested N95 respirators to medical masks to prevent respiratory virus infection in health care workers. Influenza Other Respir Viruses. 2011;5:170–179. doi: 10.1111/j.1750-2659.2011.00198.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Macintyre CR, Wang Q, Seale H, et al. A randomized clinical trial of three options for N95 respirators and medical masks in health workers. Am J Respir Crit Care Med. 2013;187:960–966. doi: 10.1164/rccm.201207-1164OC. [DOI] [PubMed] [Google Scholar]
- 134.Coffey CC, Lawrence RB, Campbell DL, Zhuang Z, Calvert CA, Jensen PA. Fitting characteristics of eighteen N95 filtering-facepiece respirators. J Occup Environ Hyg. 2004;1:262–271. doi: 10.1080/15459620490433799. [DOI] [PubMed] [Google Scholar]
- 135.Grinshpun SA, Haruta H, Eninger RM, Reponen T, McKay RT, Lee SA. Performance of an N95 filtering facepiece particulate respirator and a surgical mask during human breathing: two pathways for particle penetration. J Occup Environ Hyg. 2009;6:593–603. doi: 10.1080/15459620903120086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Qian Y, Willeke K, Grinshpun SA, Donnelly J, Coffey CC. Performance of N95 respirators: filtration efficiency for airborne microbial and inert particles. Am Ind Hyg Assoc J. 1998;59:128–132. doi: 10.1080/15428119891010389. [DOI] [PubMed] [Google Scholar]
- 137.Lindsley WG, Noti JD, Blachere FM, Szalajda JV, Beezhold DH. Efficacy of face shields against cough aerosol droplets from a cough simulator. J Occup Environ Hyg. 2014;11:509–518. doi: 10.1080/15459624.2013.877591. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Dharmadhikari AS, Mphahlele M, Stoltz A, et al. Surgical face masks worn by patients with multidrug-resistant tuberculosis: impact on infectivity of air on a hospital ward. Am J Respir Crit Care Med. 2012;185:1104–1109. doi: 10.1164/rccm.201107-1190OC. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Wood ME, Stockwell RE, Johnson GR, et al. Face masks and cough etiquette reduce the cough aerosol concentration of Pseudomonas aeruginosa in people with cystic fibrosis. Am J Respir Crit Care Med. 2018;197:348–355. doi: 10.1164/rccm.201707-1457OC. [DOI] [PubMed] [Google Scholar]
- 140.Prather KA, Wang CC, Schooley RT. Reducing transmission of SARS-CoV-2. Science. 2020;368:1422–1424. doi: 10.1126/science.abc6197. [DOI] [PubMed] [Google Scholar]
- 141.Cox CS. Airborne bacteria and viruses. Sci Prog. 1989;73:469–499. [PubMed] [Google Scholar]
- 142.Pan M, Lednicky JA, Wu CY. Collection, particle sizing and detection of airborne viruses. J Appl Microbiol. 2019;127:1596–1611. doi: 10.1111/jam.14278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Fennelly K, Tribby M, Wu C-Y, et al. Collection and measurement of aerosols of viable influenza virus in liquid media in an Andersen cascade impactor. Virus Adapt Treat. 2014;7:1–9. [Google Scholar]
- 144.Somsen GA, van Rijn C, Kooij S, Bem RA, Bonn D. Small droplet aerosols in poorly ventilated spaces and SARS-CoV-2 transmission. Lancet Respir Med. 2020;8:658–659. doi: 10.1016/S2213-2600(20)30245-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Morawska L, Tang JW, Bahnfleth W, et al. How can airborne transmission of COVID-19 indoors be minimised? Environ Int. 2020;142 doi: 10.1016/j.envint.2020.105832. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.