Introduction
Cutaneous T-cell lymphoma (CTCL) is a common cutaneous malignancy. Although early-stage disease is treatable with local therapies, including topical corticosteroids, retinoids, and alkylating agents, more advanced disease poses a therapeutic challenge. CTCL is highly radiation sensitive, but recurrence is common. Advanced disease is often lethal, with death caused by progression or bacterial sepsis from cutaneous infection.1 Many systemic treatments have been attempted for advanced disease, but complete remission is uncommon. More severe disease and poor outcomes are noted in African-Americans, making CTCL an example of racial disparities.
In this case report, we described remission in a patient with rapidly progressive CTCL with the use of concurrent radiation therapy and the inexpensive NADPH oxidase inhibitor gentian violet, and mogamulizumab. Gentian violet has been used in humans for more than a century, but recent understanding of its antitumor properties has led to novel uses in neoplastic diseases.2 Gentian violet has properties that are valuable in the patient with CTCL, namely antibacterial, antipruritic, and antiangiogenic properties. We demonstrate the induction of remission of a patient with end-stage CTCL who previously was referred to hospice but was then treated with a regimen of gentian violet, radiation therapy, and mogamulizumab. We describe this case to explore if this regimen deserves further study among heavily pretreated patients with advanced CTCL.
Case report
The patient is a 50-year-old African-American woman who presented to her dermatologist for evaluation in 2013. She reported a 5-year history of pruritic, erythematous skin eruptions that transformed into hypopigmented plaques. She received steroid injections that helped transiently, and she applied topical corticosteroids for possible eczema to no avail. Over 2 years, she had an 8-cm tumor on the left wrist, as well as additional tumors on the face and bilateral upper and lower extremities (Fig 1). In May 2013, biopsy of one of the tumors found CTCL. Polymerase chain reaction analysis was positive for T-cell receptor γ clonal rearrangement. Flow cytometric analysis of peripheral blood analysis did not show abnormal cell populations to indicate systemic involvement. CD3, CD4, and CD5 were positive by immunoperoxidase. Positron emission tomography (PET)/Computed tomography (CT) scan showed a borderline left axillary lymph node with a standardized uptake value maximum of 3.2 thought to be reactive, but there was no evidence of additional disease. She was referred to the radiation oncology department and received total skin electron irradiation (TSEI) to a total dose of 36 Gy followed by an additional boost to the underdosed areas.
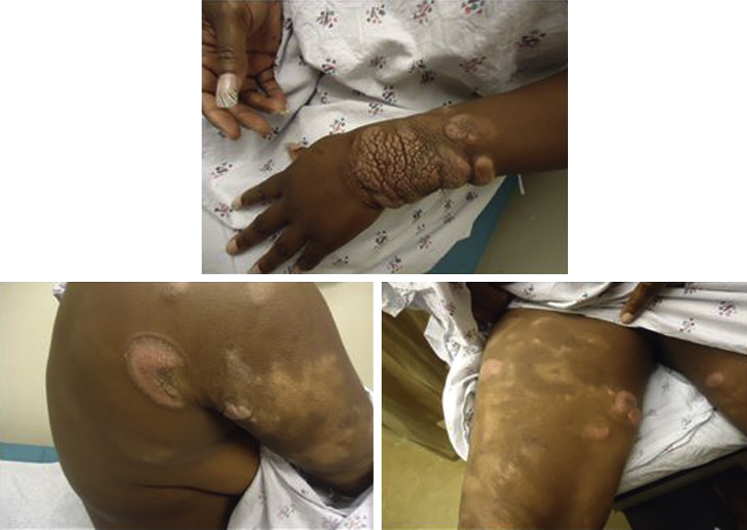
Fig 1.
The patient presented in 2013 with patches and plaques over the body. Notably, she had an 8-cm tumor on the left wrist.
The patient completed treatment in January 2014 and achieved a near-complete response. Subsequently, she received interferon, bexarotene, and photochemotherapy, but they were stopped because of poor tolerance, so she received interferon and vorinostat. In January 2015, she required local radiation treatment (4 Gy) to recurrences on the right wrist and left wrist. In March 2015, she presented with sloughing of the palms and soles, as well as blistering of the inframammary folds affecting her ability to perform activities of daily living. Culture of a sole was positive for a Pseudomonas and Staphylococcus aureus infection. Biopsy was positive for CTCL. She required weeks of intravenous antibiotics for treatment. Repeat PET/CT scan found new lymphadenopathy in the bilateral axillary, epitrochlear, inguinal, pelvic, and left external iliac lymph nodes suggestive of disease progression, so liposomal doxorubicin was initiated. She had a complete response and received repeat TSEI to a lower total dose (12 Gy) in September 2015 as conditioning for planned allogenic stem cell transplant. The patient ultimately did not receive a transplant for social reasons. Therefore, she started a treatment regimen of bexarotene with topical mechlorethamine, clobetasol, and triamcinolone.
She again had sloughing of the hands, arms, and breasts that was painful. She was restarted on liposomal doxorubicin and received 5 additional cycles (12 total). In December 2016, the lesions on the breasts and arms worsened, so she was switched to gemcitabine. She elected to stop treatment May 2017 to pursue holistic treatments. Afterwards, Pseudonomas bacteremia developed secondary to infected skin of the hands requiring hospitalization and intravenous antibiotics. She was hospitalized for additional infections resulting in bacteremia necessitating intravenous antibiotics. She still wished to take garlic; vitamin D, B12, B17; and turmeric instead of additional chemotherapy. In March 2018, lesions on the back, buttocks, and thighs opened. Repeat skin biopsy on March 9, 2018 found large cell transformation of the skin that was CD30 positive. These lesions were addressed with local radiation (4 Gy) on March 22, 2018 in combination with topical gentian violet to the open areas. She was amenable to restarting conventional therapy and brentuximab, an antibody to CD 30, was initiated on March 29, 2018.
However, she developed DRESS syndrome (drug rash with eosinophilia and systemic symptoms) after the fourth cycle, so it was stopped. She was hospitalized on multiple occasions for infections from open lesions throughout the year. On October 18, 2018, she received local radiation (4 Gy) to lesions on the bilateral eyes, forehead, hands, and feet followed by local radiation to the breast on November 15, 2018. She continued to apply topical gentian violet to the open areas. To our knowledge, the concurrent use of gentian violet and radiation has not been attempted previously. Although gentian violet is effective in controlling local infection, it was uncertain whether concurrent use would diminish or potentiate the effects of radiation. Mogamulizumab, an antibody of the chemokine receptor type 4 (CCR4), was initiated on November 15, 2018. The irradiated areas had a nice response with the gentian violet and closed. In January 2019, however, tumors developed involving the lip, left posterior shoulder, and neck that were treated locally with radiation (4 Gy) (Fig 2). She remained on mogamulizumab and continued to apply gentian violet to any open areas. She had not had a recurrence of any of the treated lesions.
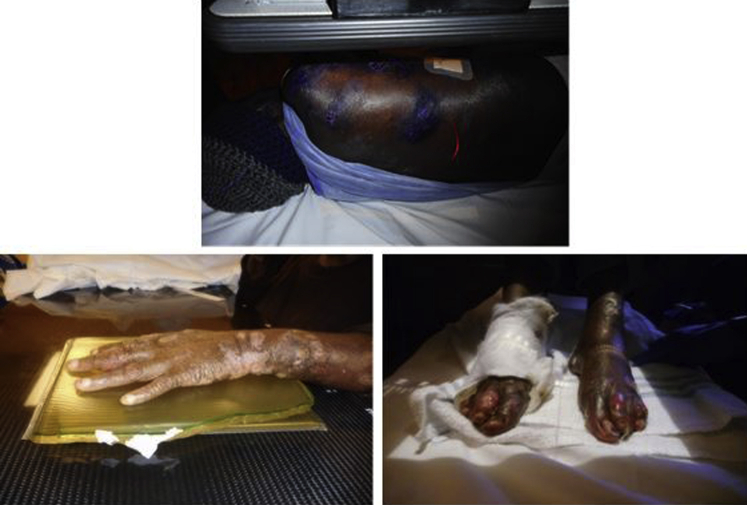
Fig 2.
In 2018, she had painful lesions on the arms, feet, around the eyes, and on the trunk. Focal radiation therapy and gentian violet were used locally.
In February 2020, PET/CT scan did find a hypermetabolic 14-cm necrotic left inguinal lymph node. Symptomatically, she experienced left leg edema and shooting pain causing difficulty with ambulation. On February 13, 2020, she received local radiation to the left inguinal mass, which was presumed to be tumor (4 Gy). The left inguinal mass had a significant response to the treatment. At this time, she remains on mogamulizumab and has no open lesions. She reports an excellent quality of life and has not been hospitalized for an infection in more than a year (Fig 3). Before, her open skin lesions caused her significant pain, which exacerbated her depression. She was even on home hospice for some time. With the combination of local treatment with radiation and gentian violet with mogamulizumab, her end-stage CTLC is in remission.
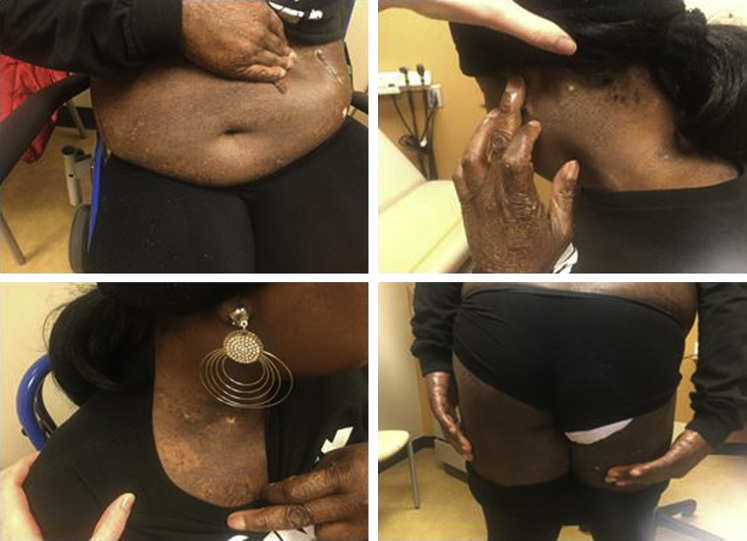
Fig 3.
The patient presented in 2020 with no open areas. Previous lesions on the thighs, trunk, hands, and neck resolved.
Discussion
Advanced CTCL remains an unmet therapeutic need. Multiple systemic therapies have been attempted including systemic retinoids, histone deacetylase (HDAC) inhibitors (vorinostat), lenalidomide, extracorporeal photopheresis, and targeted antibodies including alemtuzumab (anti-CD52), brentuximab (anti-CD30), and mogamulizumab (anti-CCR4).1, 2, 3 The results of these therapies are often partial response, with decreased burden of disease, but not complete remission. TSEI and focal radiation therapy are often used for symptom management. Historically, long courses of treatment were recommended. Emerging evidence shows that single-fraction, low-dose radiation can induce a good response. Low-dose radiation is advantageous as it has minimal side effects and can be repeated in cases of recurrence.4, 5, 6 In this case report, we show a complete remission of a patient with a regimen of topical gentian violet, radiation, and mogamulizumab.
Bacterial infection and superinfection are well known phenomena in patients with advanced CTCL.7 In addition to being a source of sepsis through impaired barrier function, there is increasing data suggesting that Staphylococcal species contribute to disease progression. This may occur through several mechanisms. First, chronic antigenic stimulation and bacterial superantigens may lead to the development of clonal T proliferation.8, 9, 10, 11 Staphylococcal superantigens have been implicated in the development of lymphomatoid papulosis, another clonal lymphoproliferative disorder, and an atopic T helper cell 2 profile was noted with a female predominance.8 Staphylococcal α toxin kills normal T lymphocytes but spares malignant T lymphocytes because of a downregulation of its receptor, ADAM10.10 Finally, antibacterial therapies have recently shown some symptomatic benefit in patients with advanced CTCL.12
Gentian violet is a small molecule that has been used in humans for more than 100 years. No evidence of its carcinogenicity has been found in human use.13 Our laboratory discovered that it is an NADPH oxidase inhibitor and, as such, found that it has antitumor properties in patients with melanoma and lymphoma in vivo.14, 15, 16, 17 Based on our work, gentian violet was found to have efficacy against early plaque-stated CTCL. In this case report, we demonstrate remission of advanced CTCL using a regimen containing gentian violet, radiation, and mogamulizumab. Given that NADPH oxidase plays a role in maintaining a T helper cell 2 immune profile,18 gentian violet may act in part to induce an immune response by a shift to a T helper cell 1 profile. Indeed, the same types of bacteria that promote atopic dermatitis (Staphylococcus and Streptococcus species) also promote CTCL proliferation. The patient received prophylactic antibiotic therapy, but this was insufficient to prevent multiple admissions for bacterial infection.
We present a patient with end-stage CTCL who experienced a complete remission after the combination of gentian violet, low-dose radiation therapy, and mogamulizumab. We believe it is unlikely to be a spontaneous resolution because the patient had progressive disease for at least 7 years. This is a first-in-human case of the concurrent use of gentian violet, mogamulizumab, and radiation. Addition of gentian violet could have mechanisms of action including enhancement of immunotherapy, induction of an abscopal effect, and changing the microbiome of the patient. The precise role of gentian violet, radiation, and mogamulizumab cannot be extrapolated from a single case. However, this patient's response, improvement in quality of life, and duration of remission, is quite unexpected. Because complete remissions of advanced CTCL are rare, this case suggests possibilities that should be tested in larger clinical trials.
Footnotes
Funding sources: None.
Conflicts of interest: None disclosed.
References
- 1.Quaglino P., Maule M., Prince H.M. Global patterns of care in advanced stage mycosis fungoides/Sezary syndrome: a multicenter retrospective follow-up study from the Cutaneous Lymphoma International Consortium. Ann Oncol. 2017;28(10):2517–2525. doi: 10.1093/annonc/mdx352. [DOI] [PubMed] [Google Scholar]
- 2.Maley A.M., Arbiser J.L. Gentian violet: a 19th century drug re-emerges in the 21st century. Exp Dermatol. 2013;22(12):775–780. doi: 10.1111/exd.12257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Querfeld C., Rosen S.T., Guitart J. Results of an open-label multicenter phase 2 trial of lenalidomide monotherapy in refractory mycosis fungoides and Sezary syndrome. Blood. 2014;123(8):1159–1166. doi: 10.1182/blood-2013-09-525915. [DOI] [PubMed] [Google Scholar]
- 4.Thomas T.O., Agrawal P., Guitart J. Outcome of patients treated with a single-fraction dose of palliative radiation for cutaneous T-cell lymphoma. Int J Radiat Oncol Biol Phys. 2013;85(3):747–753. doi: 10.1016/j.ijrobp.2012.05.034. [DOI] [PubMed] [Google Scholar]
- 5.Neelis K.J., Schimmel E.C., Vermeer M.H. Low-dose palliative radiotherapy for cutaneous B- and T-cell lymphomas. Int J Radiat Oncol Biol Phys. 2009;74(1):154–158. doi: 10.1016/j.ijrobp.2008.06.1918. [DOI] [PubMed] [Google Scholar]
- 6.Jawed S.I., Myskowski P.L., Horwitz S., Moskowitz A., Querfeld C. Primary cutaneous T-cell lymphoma (mycosis fungoides and Sezary syndrome): part I. Diagnosis: clinical and histopathologic features and new molecular and biologic markers. J Am Acad Dermatol. 2014;70(2):205.e1–205.e16. doi: 10.1016/j.jaad.2013.07.049. quiz 221-202. [DOI] [PubMed] [Google Scholar]
- 7.Mirvish J.J., Pomerantz R.G., Falo L.D., Jr., Geskin L.J. Role of infectious agents in cutaneous T-cell lymphoma: facts and controversies. Clin Dermatol. 2013;31(4):423–431. doi: 10.1016/j.clindermatol.2013.01.009. [DOI] [PubMed] [Google Scholar]
- 8.Kadin M.E., Hamilton R.G., Vonderheid E.C. Evidence linking atopy and staphylococcal superantigens to the pathogenesis of lymphomatoid papulosis, a recurrent CD30+ cutaneous lymphoproliferative disorder. PLoS One. 2020;15(2):e0228751. doi: 10.1371/journal.pone.0228751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Boursi B., Haynes K., Mamtani R., Yang Y.X. An association between newly diagnosed cutaneous T cell lymphoma and prior impetigo: a nested case-control study. Arch Dermatol Res. 2016;308(9):661–664. doi: 10.1007/s00403-016-1684-1. [DOI] [PubMed] [Google Scholar]
- 10.Blumel E., Willerslev-Olsen A., Gluud M. Staphylococcal alpha-toxin tilts the balance between malignant and non-malignant CD4(+) T cells in cutaneous T-cell lymphoma. Oncoimmunology. 2019;8(11):e1641387. doi: 10.1080/2162402X.2019.1641387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Willerslev-Olsen A., Krejsgaard T., Lindahl L.M. Bacterial toxins fuel disease progression in cutaneous T-cell lymphoma. Toxins (Basel) 2013;5(8):1402–1421. doi: 10.3390/toxins5081402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lindahl L.M., Willerslev-Olsen A., Gjerdrum L.M.R. Antibiotics inhibit tumor and disease activity in cutaneous T-cell lymphoma. Blood. 2019;134(13):1072–1083. doi: 10.1182/blood.2018888107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Arbiser J.L. Gentian violet is safe. J Am Acad Dermatol. 2009;61(2):359. doi: 10.1016/j.jaad.2009.03.029. [DOI] [PubMed] [Google Scholar]
- 14.Arbiser J.L., Bips M., Seidler A., Bonner M.Y., Kovach C. Combination therapy of imiquimod and gentian violet for cutaneous melanoma metastases. J Am Acad Dermatol. 2012;67(2):e81–e83. doi: 10.1016/j.jaad.2011.10.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wu J., Wood G.S. Analysis of the effect of gentian violet on apoptosis and proliferation in cutaneous T-cell lymphoma in an in vitro study. JAMA Dermatol. 2018;154(10):1191–1198. doi: 10.1001/jamadermatol.2018.2756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Cowan N., Coman G., Duffy K., Wada D.A. Treatment of recalcitrant mycosis fungoides with topical gentian violet. JAAD Case Rep. 2019;5(5):413–415. doi: 10.1016/j.jdcr.2019.03.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Rao S., Morris R., Rice Z.P., Arbiser J.L. Regression of diffuse B-cell lymphoma of the leg with intralesional gentian violet. Exp Dermatol. 2018;27(1):93–95. doi: 10.1111/exd.13418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Lacy P., Abdel-Latif D., Steward M., Musat-Marcu S., Man S.F., Moqbel R. Divergence of mechanisms regulating respiratory burst in blood and sputum eosinophils and neutrophils from atopic subjects. J Immunol. 2003;170(5):2670–2679. doi: 10.4049/jimmunol.170.5.2670. [DOI] [PubMed] [Google Scholar]