Abstract
Background
Hypertrophic scar is associated with excessive proliferation of fibroblasts, the accumulation of collagen fibers, and angiogenesis associated with chronic inflammation. Scar resection, combined with radiotherapy, is widely used in clinical practice, but timing remains controversial. This study aimed to investigate the association between the timing of postoperative radiotherapy and the effects on hypertrophic scar in a rabbit model.
Material/Methods
Forty New Zealand white rabbits, 8–12 months old, weighing 1.8–2.3 kg were used in the model of hypertrophic scar and underwent surgical resection with or without postoperative radiotherapy. The study groups included: Group 1, the non-resection group; Group 2, the resection and non-radiotherapy group; Group 3, the immediate postoperative radiotherapy group; Group 4, the 12-hour postoperative radiotherapy group; Group 5, the 24-hour postoperative radiotherapy group; Group 6, the 48-hour postoperative radiotherapy group; Group 7, the 72-hour postoperative radiotherapy group; and Group 8, the 120-hour postoperative radiotherapy group. The rabbit ear skin was observed after treatment, and the hypertrophic scar index (HI), fibroblast numerical area density (NA), and collagen fiber area density (AA) were determined.
Results
The HI, NA, and AA were significantly lower after 48 hours of postoperative radiotherapy (P<0.05), with the effects occurring mainly within 24 hours. There was no difference in HI, NA, and AA between the radiotherapy and non-radiotherapy groups within 24 hours after surgery.
Conclusions
In a rabbit model of hypertrophic scar, surgical resection combined with radiotherapy resulted in an optimal effect within 24 hours after surgery.
MeSH Keywords: Cicatrix, Hypertrophic; Radiotherapy, Adjuvant; Time-to-Treatment
Background
During normal wound healing, anabolism and catabolism of collagen are in equilibrium. However, increased proliferation of fibroblasts results in the pathological change of increased collagen deposition, resulting in hypertrophic scar [1,2]. Injury to the skin can result from trauma, burns, surgery, vaccination, skin piercing, folliculitis, acne, and herpes zoster infection, all of which can lead to hypertrophic scar [1,2],
Hypertrophic scar is a common clinical problem that is treated by surgical resection, topical application of drugs, pressure compression, radiotherapy, and laser treatment [3,4]. However, treatment may not always be curative, and current treatments may be associated with pain, skin atrophy, pigmentation, and also have a high recurrence rate [3,4]. For example, the recurrence rate of hypertrophic scar following surgical resection alone is between 45–100% [3,4], and the recurrence rate following radiotherapy alone is between 37–100% [5–7]. However, the recurrence rate after combined surgical excision and postoperative electron beam radiation is between 2.8–71% [8,9]. Therefore, surgical resection of hypertrophic scar combined with radiotherapy is widely used in clinical practice.
Hypertrophic scars result from increased proliferation of fibroblasts with deposition of increased extracellular matrix (ECM) and collagen, angiogenesis, and chronic inflammation [1,2]. Ionizing radiation can directly inhibit and destroy fibroblasts and accelerate the apoptosis of fibroblasts and vascular endothelial cells resulting in the reduction of ECM and collagen deposition [10–12].
Currently, there are no recommendations for the optimal timing and duration of the use of local irradiation following the surgical excision of hypertrophic scar. Previous studies have indicated that the total local radiation dose delivered is more effective than the choice of the radiation source, the schedule of the radiotherapy, and the size of the dose given [13]. The findings from other studies have shown that that radiotherapy dose does not have a dose-response effect and that radiation may be effective early after surgery [13–16]. Given these latest findings, the hypothesis that drove the present study was that more information is required regarding the effect of the time interval between surgical excision of hypertrophic scar and the beginning of radiotherapy. Previous authors have recommended a time interval of between 3 hours to 7 days [13]. However, previous studies have mainly been in vitro studies, and the results of in vivo research data are awaited.
A rabbit model of hypertrophic scar uses injury to the rabbit ear, which can produce lesions similar to human hypertrophic scars [17]. The ventral skin of the rabbit ear has a similar structure to human skin [17]. Therefore, this study aimed to investigate the association between the timing of postoperative radiotherapy and the effects on hypertrophic scar in a rabbit model.
Material and Methods
Animals and study groups
The China Medical University Review Board approved this study. Rabbits were purchased from the Animal Experiment Center of China Medical University. New Zealand white rabbits, 8–12 months old, weighing 1.8–2.3 kg, were used for the animal model of hypertrophic scar (Table 1).
Table 1.
The study groups in the rabbit model and the effects of timing of radiotherapy on hypertrophic scar following surgery.
| No. of groups | Groups | Timing of the first radiotherapy | Cases of infection | |
|---|---|---|---|---|
| Non-infected | Infected | |||
| Group 1 | Non-resection group | – | – | – |
| Group 2 | Resection and non-radiotherapy group | – | 30 | 0 |
| Group 3 | Immediate postoperative radiotherapy group | Immediately after surgery | 29 | 1 |
| Group 4 | 12-hour postoperative radiotherapy group | 12 hours after surgery | 30 | 0 |
| Group 5 | 24-hour postoperative radiotherapy group | 24 hours after surgery | 30 | 0 |
| Group 6 | 48-hour postoperative radiotherapy group | 48 hours after surgery | 27 | 3 |
| Group 7 | 72-hour postoperative radiotherapy group | 72 hours after surgery | 29 | 1 |
| Group 8 | 120-hour postoperative radiotherapy group | 120 hours after surgery | 28 | 2 |
Forty rabbits in the model of hypertrophic scar underwent surgical resection with or without postoperative radiotherapy. The study groups included: Group 1, the non-resection group; Group 2, the resection and non-radiotherapy group; Group 3, the immediate postoperative radiotherapy group; Group 4, the 12-hour postoperative radiotherapy group; Group 5, the 24-hour postoperative radiotherapy group; Group 6, the 48-hour postoperative radiotherapy group; Group 7, the 72-hour postoperative radiotherapy group; and Group 8, the 120-hour postoperative radiotherapy group (Table 1).
Establishment of the rabbit model of hypertrophic scar
Surgery on the rabbit ear followed standard procedures. Anesthesia was administered by a thigh intramuscular injection of ketamine/xylazine (0.2 ml/kg). The ventral area of the rabbit ear was shaved and cleaned. The site of the skin incision was marked with gentian violet in the middle and lower part of the ventral side of the rabbit ear. The incision was a circular shape with a diameter of 6 mm. The entire skin layer was excised between the skin and the ear cartilage using a sharp scalpel. Four wounds were made in each rabbit ear at a distance of 1 cm, resulting in 320 wounds in 40 rabbits (Table 1).
Hemostasis of the wound was achieved by compression using sterile gauze, which was fixed with tape. The rabbits were maintained in cages. The next day, the dressings were removed. Povidone-iodine was applied to the wounds, which were allowed to heal naturally.
Hypertrophic scar resection
After anesthesia, the skin was cleaned with 2% of iodophor-containing cotton wool, partially disinfected, and a sterile towel was placed over the rabbit ear. The hypertrophic scar tissue with a surrounding area of 0.2 cm of normal skin was resected.
Radiotherapy
A two-photon linear accelerator with a 5 mV electron beam was used (Siemens, Munich, Germany) with 2 mm skin application (MED-TEC, Orange City, IA, USA). The irradiation field included 0.5 cm around the incision, using a lead film to protect other areas. The total radiation dose was 1800 cGy. After the first radiotherapy session (Table 1), each radiotherapy interval was 24 hours, with 300 cGy each time, and continuous irradiation was performed six times.
Post-treatment monitoring of hypertrophic scars
After surgery and radiotherapy, wound healing of the rabbit ears was closely observed, including the healing time (time of eschar loss exfoliation), the size of the scar (the diameter), and the presence of infection (whether or not postoperative infection occurred).
Tissue sampling of hypertrophic scars
The rabbits were euthanized at 30 days after surgery. The specimens included normal skin with a diameter of 3 mm around the scar. After the rabbit ear skin specimen was harvested, it was sectioned and fixed in 4% formaldehyde buffer for 48 hours. After dehydration using graded ethanol, the specimens were embedded in paraffin wax.
Hematoxylin and eosin (H&E) staining
The embedded fixed rabbit ear skin tissues were sectioned onto glass slides, deparaffinized in CitriSolv (Thermofisher Scientific, Waltham, MA, USA), and then hydrated in graded ethanol to distilled water. The tissue sections were stained with hematoxylin solution followed by 1% hydrochloric acid, ethanol, and eosin. The tissue sections were then dehydrated in graded ethanol and cover-slipped.
van Gieson stain for collagen
The slides were deparaffinized in CitriSolv (Thermofisher Scientific, Waltham, MA, USA), hydrated in graded ethanol to distilled water. The tissue sections were stained with van Gieson stain for collagen. Incubation of the tissue section in van Gieson solution was followed by dehydration in 95% and 100% ethanol and cover-slipped.
Indices used for the evaluation of hypertrophic scar in the rabbit model
The rabbit ear skin was observed after treatment, and the hypertrophic scar index (HI), fibroblast numerical area density (NA), and collagen fiber area density (AA) were determined by low-power light microscopy on the routinely stained slides (H&E).
The following formula was used to determine the HI:
where, A is the vertical thickness of the highest point of the scar dome to the ear cartilage surface; B is the vertical thickness of the normal skin around the scar to the ear cartilage surface.
Ten fields of view of the routinely stained tissue sections were randomly selected from the superficial, deep, central, and lateral aspects of the hypertrophic scar. The number of fibroblasts per unit area in the routinely stained tissue sections was counted, and the average number represented NA. In the van Giesen-stained slides, collagen (stained red) was assessed in ten random fields of view selected from the superficial, deep, central, and lateral aspects of the hypertrophic scar. The AA of the collagen was calculated by image analysis using Image-Pro Plus version 6.0 software (Mediacy, Rockville, MD, USA), with the average AA calculated in each case.
Statistical analysis
Data were analyzed using SPSS version 17.0 software (IBM Corp., Armonk, NY, USA). The measurement data were expressed as the mean±standard deviation (SD). The difference between multiple groups was analyzed by one-way analysis of variance (ANOVA). The Levene test was used to determine variance consistency. The least significant difference (LSD) test was used to complete the comparison between groups. The count data in multiple groups was evaluated using the chi-squared (χ2) test. A p-value <0.05 was considered to be statistically significant.
Results
Early radiotherapy significantly inhibited hypertrophic scar formation in the rat model
The rat model of hypertrophic scar included the following study groups: Group 1, the non-resection group; Group 2, the resection and non-radiotherapy group; Group 3, the immediate postoperative radiotherapy group; Group 4, the 12-hour postoperative radiotherapy group; Group 5, the 24-hour postoperative radiotherapy group; Group 6, the 48-hour postoperative radiotherapy group; Group 7, the 72-hour postoperative radiotherapy group; and Group 8, the 120-hour postoperative radiotherapy group. After treatment, three main indices were measured: the hypertrophic scar index (HI); the fibroblast numerical area density (NA); and the collagen fiber area density (AA).
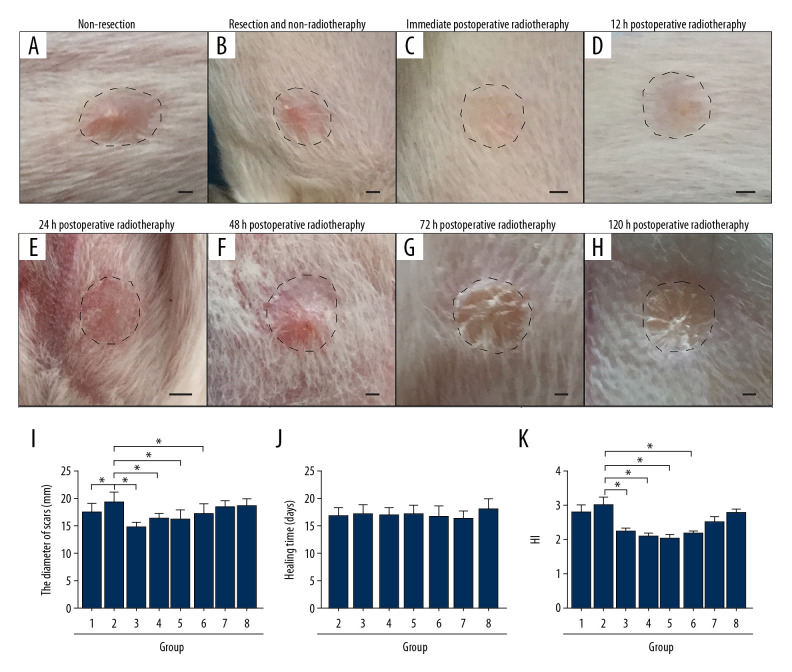
In the hypertrophic scar model of the rabbit ear, the hypertrophic scar was significantly raised above the surrounding normal skin. The skin thickness was two to three times the normal skin, and the skin color was bright red (Figure 1A). Compared with the non-resection group, the size of scars in the resection and non-radiotherapy group was increased (P<0.05) (Figure 1A, 1B, 1I). There was no significant difference in the mean healing time between the resection and non-radiotherapy group and the postoperative radiotherapy groups (P>0.05) (Figure 1J). However, the size of scars in the early (immediately, 12 h, 24 h, and 48 h) postoperative radiotherapy groups (P<0.05) (Figure 1B–1E, 1I), but not the late (72 h and 120 h) postoperative radiotherapy groups (P>0.05) (Figure 1G–1I), was significantly reduced when compared with the resection and non-radiotherapy group.
Figure 1.
In the rabbit model, early radiotherapy significantly reduced the severity of the hypertrophic scar. (A) The non-resection group, without surgery and radiotherapy, shows the visible convex surface of the hypertrophic scar at 30 days after surgery. (B) The resection and radiotherapy group shows the visible convex surface of the hypertrophic scar. (C–F) In the early postoperative radiotherapy groups, the hypertrophic scar surface was flat, and the skin color was normal. (G, H) In the late postoperative radiotherapy groups, a convex hypertrophic scar remained. (I) Quantification of the diameter of the hypertrophic scar. (J) Quantification of the healing time of the hypertrophic scar. (K) Quantification of the hypertrophic scar index (HI). Group 1, the non-resection group. Group 2, the resection and non-radiotherapy group. Group 3, the immediate postoperative radiotherapy group. Group 4, the 12-hour postoperative radiotherapy group. Group 5, the 24-hour postoperative radiotherapy group. Group 6, the 48-hour postoperative radiotherapy group. Group 7, the 72-hour postoperative radiotherapy group. Group 8, the 120-hour postoperative radiotherapy group. Scale bar = 50 μm.
These findings in the rabbit model showed that surgery alone did not cure the hypertrophic scars, but early postoperative radiotherapy reduced the size. The HI showed these findings. Compared with the resection and non-radiotherapy group, there was no significant difference in the HI of the non-resection group and the late postoperative radiotherapy groups (P>0.05) (Figure 1K). However, the HI of the early postoperative radiotherapy groups was significantly reduced (P<0.05) (Figure 1K), and there was no significant difference among the early postoperative radiotherapy groups (P>0.05) (Figure 1K). Both the resection and non-radiotherapy group and the postoperative radiotherapy groups had a similar incidence of infection (P>0.05) (Table 1).
Early radiotherapy had significant effects on the function of fibroblasts
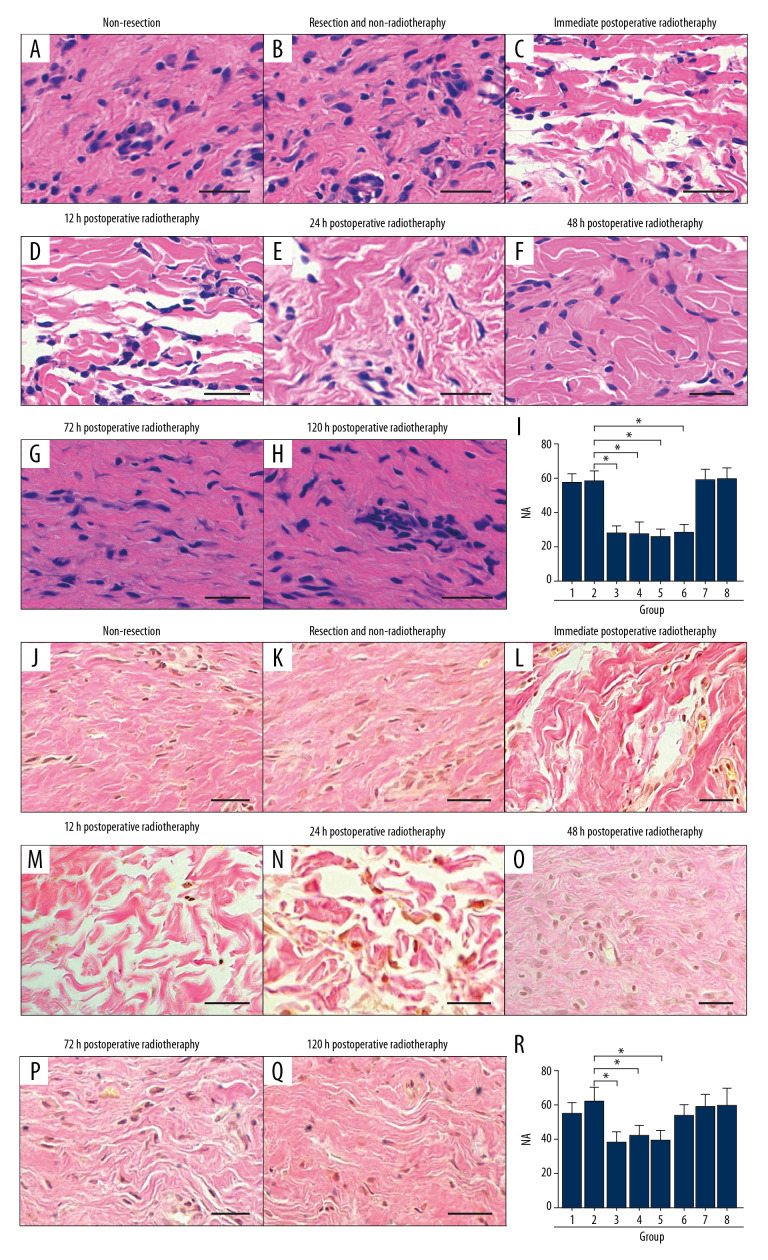
There were a larger number of darkly stained fibroblasts and the extracellular matrix (ECM) in the non-resection group, the resection group, the non-radiotherapy group, and the late postoperative radiotherapy groups (P>0.05) (Figure 2A, 2B, 2G–2I). The NA of the early postoperative radiotherapy groups were significantly reduced (P<0.05) (Figure 2C–2F, 2I), with reduced and regularly arranged fibroblasts and reduced ECM. Meanwhile, there was no significant difference in NA between the early postoperative radiotherapy groups (P>0.05) (Figure 2I).
Figure 2.
(A–R) In the rabbit model, early radiotherapy significantly reduced the number of fibroblasts in hypertrophic scar. (A, B) The non-resection group and the resection and non-radiotherapy group showed a large number of fibroblasts in the hypertrophic scar. (C–F) In the early postoperative radiotherapy groups, fibroblasts were significantly reduced. (G, H) The late postoperative radiotherapy groups showed a large number of fibroblasts. (I) The quantification of fibroblast numerical area density (NA). (J, K) In the non-resection group and the resection and non-radiotherapy group, a large number of dense collagen fibers can be seen. (O–Q) Collagen fibers were denser in the late postoperative radiotherapy groups than in the early postoperative radiotherapy groups, except the 48-hour postoperative radiotherapy group. (R) The quantification of collagen fiber area density (AA). Scale bar=50 μm.
Early postoperative radiotherapy resulted in regularly arranged fibroblasts that were reduced in number
The collagen fibers in the non-resection group were denser and disordered, and the AA was the same as the resection and non-radiotherapy group and the late postoperative radiotherapy groups (P>0.05) (Figure 2J, 2K, 2P–2R). Similar findings for the HI, NI, and AA were found for the early postoperative radiotherapy groups. However, the findings in the 48-hour postoperative radiotherapy group were significantly reduced when compared with the other groups (P<0.05) (Figure 2L–2O, 2R). Also, there was no significant difference in AA between the early postoperative radiotherapy groups, except for 48-hour postoperative radiotherapy group (P>0.05) (Figure 2R), which showed by that the collagen fibers were tightly structured, regular in their arrangement and almost parallel to the epidermis.
Discussion
Hypertrophic scar is associated with increased proliferation of fibroblasts and increased deposition of collagen fibers. This study investigated the association between the timing of postoperative radiotherapy and the effects on hypertrophic scar in a rabbit model of hypertrophic scar following surgical resection with or without postoperative radiotherapy. The rabbit ear skin was observed after treatment, and the hypertrophic scar index (HI), fibroblast numerical area density (NA), and collagen fiber area density (AA) were determined.
Following surgical scar resection alone, the recurrence rate of hypertrophic scar is high and occurs in between 45–100% of cases [3,4], and the scar area will be larger after recurrence than before surgery, which is a finding supported by the present study (Figure 1I). The effect of radiotherapy alone is also not ideal, because mature fibroblasts and collagen fibers, which compose hypertrophic scars, hypertrophic scar is not sensitive to radiation, and radiotherapy can only alleviate symptoms.
In the early postoperative period, naïve fibroblasts and immature and disorganized collagen are present in granulation tissue, which is more sensitive to radiation. Therefore, radiation therapy at an early stage following the surgical resection of hypertrophic scar is more likely to affect the balance between collagen synthesis and degradation [1,2].
In 2006, Slemp and Kirschner found that surgery combined with radiotherapy significantly reduced the recurrence rate of hypertrophic scar by 50% when compared with surgery alone [18]. The findings from the present study demonstrated that compared with the non-resection group and the resection and non-radiotherapy group, the size of the hypertrophic scar, the HI, NA, and AA were all significantly lower only in immediate, 12-hour, and 24-hour postoperative radiotherapy groups (Figures 1K, 2I, and 2R). Also, there was no significant difference in the HI, NA, and AA between immediate, 12-hour, and 24-hour postoperative radiotherapy groups (Figures 1K, 2I, 2R).
The findings from this preliminary study in a rabbit model of hypertrophic scar support that the role of radiotherapy should be investigated within 24 hours after surgery for hypertrophic scar. This finding is supported by previous in vitro studies that have reported adjuvant radiotherapy within the first 24 hours to inhibit hypertrophic scar and keloid after surgery [13,19].
However, other studies have shown that early postoperative radiotherapy may increase the risk of postoperative infection and wound dehiscence [8,13,20]. Therefore, it may be recommended that radiotherapy is commenced between 24–48 hours and 5–10 days after surgery [8,13,20]. The findings from the present study did not show that radiotherapy increased postoperative infection (Table 1). This finding may be because radiotherapy results in small blood vessel occlusion and reduced levels of inflammatory mediators, which not only has a hemostatic effect but also prevents wound infection [1,21,22].
Conclusions
This study aimed to investigate the association between the timing of radiotherapy and the effect on hypertrophic scar following surgery in a rabbit model. The findings showed that surgical resection combined with radiotherapy resulted in an optimal effect within 24 hours after surgery.
Acknowledgments
The China Medical University Review Board approved this study.
Footnotes
Conflict of interest
None.
Source of support: This study was supported by the National Natural Science Foundation of China (NSFC) (Grant No. 51872332, for Dr. Shu Guo)
References
- 1.Ogawa R, Akaishi S. Endothelial dysfunction may play a key role in keloid and hypertrophic scar pathogenesis – Keloids and hypertrophic scars may be vascular disorders. Med Hypotheses. 2016;96:51–60. doi: 10.1016/j.mehy.2016.09.024. [DOI] [PubMed] [Google Scholar]
- 2.Ogawa R. Keloid and hypertrophic scars Are the result of chronic inflammation in the reticular dermis. Int J Mol Sci. 2017;18(3):606. doi: 10.3390/ijms18030606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Berman B, Perez OA, Konda S, et al. A review of the biologic effects, clinical efficacy, and safety of silicone elastomer sheeting for hypertrophic and keloid scar treatment and management. Dermatol Surg. 2007;33(11):1291–30. doi: 10.1111/j.1524-4725.2007.33280.x. [DOI] [PubMed] [Google Scholar]
- 4.Kal HB, Veen RE. Biologically effective doses of postoperative radiotherapy in the prevention of keloids. Dose-effect relationship. Strahlenther Onkol. 2005;181(11):717–23. doi: 10.1007/s00066-005-1407-6. [DOI] [PubMed] [Google Scholar]
- 5.Mankowski P, Kanevsky J, Tomlinson J, et al. Optimizing radiotherapy for keloids: A meta-analysis systematic review comparing recurrence rates between different radiation modalities. Ann Plast Surg. 2017;78(4):403–11. doi: 10.1097/SAP.0000000000000989. [DOI] [PubMed] [Google Scholar]
- 6.Leventhal D, Furr M, Reiter D. Treatment of keloids and hypertrophic scars: A meta-analysis and review of the literature. Arch Facial Plast Surg. 2006;8(6):362–68. doi: 10.1001/archfaci.8.6.362. [DOI] [PubMed] [Google Scholar]
- 7.Mustoe TA, Cooter RD, Gold MH, et al. International clinical recommendations on scar management. Plast Reconstr Surg. 2002;110(2):560–571. doi: 10.1097/00006534-200208000-00031. [DOI] [PubMed] [Google Scholar]
- 8.Akita S, Akino K, Yakabe A, et al. Combined surgical excision and radiation therapy for keloid treatment. J Craniofac Surg. 2007;18(5):1164–69. doi: 10.1097/scs.0b013e3180de62a1. [DOI] [PubMed] [Google Scholar]
- 9.Ogawa R, Mitsuhashi K, Hyakusoku H, et al. Postoperative electron-beam irradiation therapy for keloids and hypertrophic scars: Retrospective study of 147 cases followed for more than 18 months. Plast Reconstr Surg. 2003;111(2):547–53. doi: 10.1097/01.PRS.0000040466.55214.35. [DOI] [PubMed] [Google Scholar]
- 10.Zhang W, Cheng X, Li H, et al. Evaluation of the therapeutic effect of micro-plasma radio frequency on hypertrophic scars in rabbit ears. Lasers Med Sci. 2018;33(9):1961–68. doi: 10.1007/s10103-018-2562-1. [DOI] [PubMed] [Google Scholar]
- 11.Ramirez-Fort MK, Meier B, Feily A, et al. Adjuvant irradiation to prevent keloidal fibroproliferative growth should be standard of care. Br J Dermatol. 2017;177(6):e327–28. doi: 10.1111/bjd.15667. [DOI] [PubMed] [Google Scholar]
- 12.Ji J, Tian Y, Zhu YQ, et al. Ionizing irradiation inhibits keloid fibroblast cell proliferation and induces premature cellular senescence. J Dermatol. 2015;42(1):56–63. doi: 10.1111/1346-8138.12702. [DOI] [PubMed] [Google Scholar]
- 13.Dinh Q, Veness M, Richards S. Role of adjuvant radiotherapy in recurrent earlobe keloids. Australas J Dermatol. 2004;45(3):162–66. doi: 10.1111/j.1440-0960.2004.00079.x. [DOI] [PubMed] [Google Scholar]
- 14.Ogawa R, Tosa M, Dohi T, et al. Surgical excision and postoperative radiotherapy for keloids. Scars Burn Heal. 2019;5:2059513119891113. doi: 10.1177/2059513119891113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Liu CL, Yuan ZY. Retrospective study of immediate postoperative electron radiotherapy for therapy-resistant earlobe keloids. Arch Dermatol Res. 2019;311(6):469–75. doi: 10.1007/s00403-019-01922-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Lyu A, Xu E, Wang Q. A retrospective analysis of surgical resection of large ear keloids. Australas J Dermatol. 2019;60(1):29–32. doi: 10.1111/ajd.12872. [DOI] [PubMed] [Google Scholar]
- 17.Zhang W, Cheng X, Li H, et al. Evaluation of the therapeutic effect of micro-plasma radio frequency on hypertrophic scars in rabbit ears. Lasers Med Sci. 2018;33(9):1961–68. doi: 10.1007/s10103-018-2562-1. [DOI] [PubMed] [Google Scholar]
- 18.Slemp AE, Kirschner RE. Keloids and scars: A review of keloids and scars, their pathogenesis ‘risk factors’ and management. Curr Opin Pediatr. 2006;18(4):396–402. doi: 10.1097/01.mop.0000236389.41462.ef. [DOI] [PubMed] [Google Scholar]
- 19.De Lorenzi F, Tielemans HJ, van der Hulst RR, et al. Is the treatment of keloid sears still a challenge in 2006? Ann Plast Surg. 2007;58(2):186–92. doi: 10.1097/01.sap.0000237761.52586.f9. [DOI] [PubMed] [Google Scholar]
- 20.Recalcati S, Caccialanza M, Piccinno R. Postoperative radiotherapy of auricular keloids: A 26-year experience. J Dermatolog Treat. 2011;22(1):38–42. doi: 10.3109/09546630903460278. [DOI] [PubMed] [Google Scholar]
- 21.Levitt WM. Radiotherapy in the prevention and treatment of hypertrophic scars. Br J Plast Surg. 1951;4(2):104–12. doi: 10.1016/s0007-1226(51)80015-8. [DOI] [PubMed] [Google Scholar]
- 22.Ogawa R. Keloid and hypertrophic scars are the result of chronic inflammation in the reticular dermis. Int J Mol Sci. 2017;18(3) doi: 10.3390/ijms18030606. pii: E606. [DOI] [PMC free article] [PubMed] [Google Scholar]