Abstract
The objective of this study was to evaluate the effects of forage legume diets on haematological and biochemical indices of Xhosa goats. Eighteen animals, one-year-old, of average live weight 14.13 ± 0.24 kg were assigned, in a completely randomized design, to three treatment diets. Animals were housed individually and sex was equally represented among treatments with 6 animals per treatment. Weights and blood samples were taken every fortnight. A pair of blood samples (5 ml each) were collected from each goat, via jugular vein puncture and were subjected to either haematological or biochemical analysis. The results show that treatment diets and sex significantly (P < 0.05) affected DMI, total gain, ADG, and feed conversion ratio. Treatment diets and sex had no effects (P > 0.05) on serum total proteins, globulins, urea, creatinine, alanine aminotransferase, alkaline phosphatase (ALP), and gamma- glutamyl transferase concentrations. The diet influenced albumin was (P < 0.05) while sex influenced ALP concentration (P < 0.05). Measured over time, the plasma concentration of urea and ALP significantly increased (P < 0.05) while creatinine and cholesterol concentration decreased (P < 0.05). Although the overall blood biochemical properties remain unchanged among the treatments and between sexes; treatment by sex, treatment by day and sex by day interactions were significant (P < 0.05). The white blood cells, red blood cell, mean corpuscular haemoglobin, mean corpuscular haemoglobin concentration, haemoglobin, haematocrit, and platelet and mean platelet volume levels were similar among treatments and between sexes (P > 0.05). The mean cell volume and red cell distribution width were different among treatments (P < 0.05). Treatment diets significantly (P < 0.05) influenced neutrophil (%) content while sex influenced basophile (%) content. In conclusion forage legumes did not cause toxicity in goats or any pathological condition.
Keywords: Growth, Serum biochemistry, Haematology, Forage legumes, Goats
1. Introduction
The changing climatic conditions in the past years have resulted in persistent droughts, heat waves and shortages in animal feed (FAO, 2014). This has severely affected ruminant animal production leading to a dire need to address feed shortages particularly in small scale farming systems. One of such proposals is the use of forage legumes as protein supplements (Bayble, Melaku, & Prasad, 2007; Gebregiorgis, Negesse, & Nurfeta, 2011) in ruminant diets. Forage legumes provide adequate protein (Washaya et al., 2018) and average energy (McDonald, Edwards, Greenhalgh, & Morgan, 2011) for ruminant livestock production. Lablab and cowpea were chosen for this study because of their high biomass yield (Ayan, Mut, Basaran, Eki Acar, & Asci, 2012; Jingura, Sibanda, & Hamudikuwanda, 2001; Washaya et al., 2018), high digestibility (Maarsdop, & Titterton, 1997) drought tolerance (Jingura et al., 2001; Mahala, Amasiab, Yousif, & Elsadig, 2012) and hay keeping quality. Furthermore, there is little research on their effects on small ruminants particularly, goats in sub Saharan Africa, although there is enough evidence (Nurfeta, 2010; Warghon & Clark, 2004) that they significantly improve ruminant nutrition and reduce feed costs. In an experiment done by Jingura et al. (2001), Lablab ranked high in biomass yield (5.9t/ha), organic matter (889 g/kgDM) and OM digestibility (601 g/kgDM), it was observed to be least in NDF (465.5 g/kgDM), ADF (331.4 g/kgDM), high in P (2.1%) and the authors concluded that it is the most appropriate forage legume for tropical semi-arid regions when compared to Siratro, Silver leaf and Fine stem stylo.
Information on Cowpea production requirements and its potential as animal feed potential is still lacking in Southern Africa except that it's used as human food (Department of Agriculture, Forestry, & Fisheries, 2011). However, Ayan et al. (2012) reported forage yield ranged between 6.03 and 7.94 t ha−1 and CP ranged from 170.2 g kg−1 up to185.2 g kg-1 in various cultivars grown in Turkey. In the same experiment cultivars produced ADF, NDF, magnesium (Mg), P, Ca and K contents which were higher than animal needs. This was earlier indicated by Akinlade et al. (2005) in Nigeria and later by Gebreyowhans and Gebremeskel (2014) in Ethiopia. In another experiment done by Ravhuhali, Ng'ambi, and Norris (2010), it was discovered that Cowpea cultivars had higher in vitro DM, OM and protein digestibility's than buffalo grass hay, with in vitro DM, OM and protein digestibility's ranging from 0.64 to 0.75.
Because animals are exposed to these adverse climatic variants, their voluntary feed intake, protein and energy metabolism and enzyme reaction have been lowered leading to increased organ function (Olafadehan, 2011). In order for animals to survive these climatic environments, they have developed copping strategies which include among other things alteration of renal and liver functions (Olafadehan, 2011; Žubčić, 2001). However noble these adaptive processes might be, they are also bound to put the well-being of animals at risk. Goats by their nature are browsers and raising them in confinement would stimulate this type of adaptive behaviour over time. Stella et al. (2007) reported that goats are world-wide providers of essential meat and dairy products but very little research is performed on them than on cattle and sheep.
The biochemical and haematological variables of blood can be used to monitor and or evaluate health, nutritional and physiological status of ruminants (Al-Eissa et al., 2012). Furthermore, Mohammed, Razzaque, Omar, Albert and Al-Gallaf (2016) reported that blood constituents are markers to determine the efficacy of feed nutrient content and its utilization. Al-Seaf and AlHarbi (2012) observed that biochemical and haematology profiles can also be used to assess the immunity status of goats while they can also be an index of transportation stress (Ambore, Ravikanth, Maini, & Rekhe, 2009). Among other factors, nutrition, stress, reproductive status, age, sex, genetics, management, housing, and other environmental factors have been reported to have profound effects on the haematological and biochemical profiles of small ruminants (Mohammed et al., 2016) Therefore, there is a need to establish the dietary effects of forage legumes fed to goats. Because biochemical and haematological parameters are a clear indication of unobservable animal wellness; the objective of this study was to evaluate the effects of forage legume diets on growth, haematological and biochemical parameters of Xhosa goats.
2. Methodology
The protocols and procedures applied in this study were according to the ethical principles for use of experimental animals as established by the Committee of Ethics on Animal Use of the Society for the Prevention of Cruelty to Animals. The University of Fort Hare Ethics Committee ensured that these requirements were adhered to by approving the study and a certificate was issued (MUP121SWA01)
2.1. Description of the study site
The research was conducted at the University of Fort Hare Research Farm, in the Eastern Cape Province of South Africa. The farm is located at a latitude of 32°46′ S and longitude 26°50′ E at an altitude of 535 metres above sea level. It has a warm temperate climate with an average annual rainfall of about 575 mm received mainly during the summer months of November to March. The maximum temperature is 24.6 °C, minimum temperature (11.1 °C) and average temperature (17.8 °C).
2.2. Study design and animal management
2.2.1. Animal management
Eighteen goats (9 castrate males and 9 females) were used in this experiment. The average age of goats was 12 months at an average weight of 14.2 ± 0.24 kg. Goats were dewormed using niclosamide 20% (Lintex L) and dipping was done using a pour on acaricide (Coopers Redline). Animals on T1 and T3 were injected with a mineral and vitamin complex (Cipla Agrimed) prior to housing and thereafter, every 15 days. Animals were housed individually in metabolic pens measuring 1.5 × 1.0 m and were acclimated to the environment and experimental conditions for two weeks, which was followed by 45 days of growth/feeding trial. Animals were fed two equal portions at 08:00 and 15:00 h daily and the amount on offer was adjusted based on body weight measured every 15 days. Clean water was available to animal's ad-libitum.
2.2.2. Treatment diets
Animals were subjected to three treatments on DM basis of treatment one (T1); 71% Vigna hay, 19% Katambora hay salt (0.5%), molasses (3%), maize (5%) and mineral vitamin premix 1.5%; treatment two (T2); 90% lamb and ewe pellet plus 10% Katambora grass hay; and treatment three (T3); 72% Lablab hay 19% Katambora hay, salt (0.5%), molasses (2%) maize (5%) and mineral vitamin premix 1.5%. Treatment two was the positive control diet. All diets were formulated to contain CP and energy to meet the minimum recommendation for intensive feeding (i.e. 14% CP and 9 MJ ME/ kg DM) and animals were fed at 3% of metabolic body weight.
2.2.3. Blood sampling and collection
Every fifteen days’ goats were restrained for blood collection. A pair of 5 ml blood samples were withdrawn into test tubes via jugular vein puncture; one for haematological analysis and the other for serum chemistry. The sterile test-tubes containing EDTA as anticoagulant were labelled in accordance to the corresponding animal number and then placed in a cooler bag containing ice cubes before being moved to the laboratory within an hour of collection for the determination of haematological parameters. Blood tubes without anticoagulant, were allowed to coagulate in kaylite holders at 37 °C for serum separation, centrifuged at 3500 rpm at 21 °C for 10 min and was stored at −20 °C for biochemical analysis. The haematological estimates including haemoglobin (Hb), haematocrit (PCV), red blood cells (RBC), white blood cells (WBC), mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH), and mean corpuscular haemoglobin concentration (MCHC). RDW, Platelets, MPV were analysed by an auto blood-analyser, (NIHON KOHDEN blood auto analyser; MEK-6450 K; Span Nihon Kohden Diagnostic PVT Ltd., Surat, Gujarat, India). Serum concentration of biochemical metabolites including urea, cholesterol, total protein (TP) and albumin and serum enzymes including alanine aminotransferase (ALT), alkaline phosphatase(ALP) and gamma- glutamyl transferase (GGT) were determined using commercially available kits (Cogent TM of Span Diagnostic Ltd., Surat, Gujarat, India).
2.2.4. Statistical analysis
The effect of diet and sex on the mean concentrations of the blood haematology and metabolites was analyzed using the GLM of SAS (2010) version 9.3. All the resulting data for the blood metabolites was checked for normality using the PROC UNIVARIATE of SAS (2010). A repeated measures analysis of SAS (2010) was used to assess the effect of time on blood metabolites and haematological parameters. The Turkeys HSD method was used to separate means for blood metabolites and haematological parameters and were considered to be significantly different at P < 0.05.
3. Results
3.1. Chemical composition of diets
Three diets were given to growing goats over a period of sixty days and the dietary inclusions are listed in Table 1. The three diets were formulated to provide the same amount of protein (iso nitrogenous) and energy (iso energetic). The basis was the pellet diet which had a CP level of 14% and ME of 8.87Mj/kg. The CP level is above the minimum requirement as indicated by NRC, (2007) of 12.6%.
Table 1.
Chemical composition (% for DM and% DM for others) of treatment diets.
| Nutrient | Diet 1 | Diet 2 | Diet 3 |
|---|---|---|---|
| Katambora /Vigna (1:4) | Pellets | Katambora/Lablab (1:4) | |
| DM% | 92.3 | 89.92 | 92.72 |
| CP% | 13.93 | 13.49 | 13.77 |
| CF% | – | 23.51 | – |
| Fat% | 2.17 | 5.92 | 1.97 |
| Me Mj/kg | 8.60 | 8.87 | 8.90 |
| NE Mcal/kg | – | 0.78 | – |
| NDF% | 35.41 | 32.39 | 37.51 |
DM=dry matter; CP=crude protein; NDF=neutral detergent fibre; CF= crude fibre; ME = metabolizable energy; NE = net energy.
3.2. Feed intake and body weight gain of xhosa goats
After a feeding trial of sixty days, changes in weight were determined and results are shown in Table 2. Treatment diets and sex significantly (P < 0.05) affected DMI, final body weight, total gain, ADG, and feed conversion ratio (FCR). Male animals exhibited higher DMI (P < 0.05) compared to female animals. Animals on T2 showed higher (P < 0.05) DMI of 24 g/d and 6.7 g/d compared to T1 and T3 respectively. Animals under T2 exhibited the highest ADG and total gain with the lowest FCR (P < 0.05). Sex had no significant (P > 0.05) effect on FCR, however male animals significantly (P < 0.05) gained less than females by 0.2kgs.
Table 2.
Feed intake, growth parameters and morphometric data of Xhosa goats under three dietary treatments.
| Variables | Treatment (T) |
SE | Sex (S) |
SE | P values |
|||||
|---|---|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | M | F | T | S | T x S | |||
| DMI(g/d) | 670.8c | 694.8a | 688.1b | 4.612 | 716.1a | 653.0b | 3.766 | 0.001 | 0.001 | 0.026 |
| Growth Parameters (kg) | ||||||||||
| IW | 14.13 | 13.96 | 14.02 | 0.469 | 14.90 | 14.71 | 0.392 | 0.103 | 0.113 | 0.481 |
| FW | 16.81 | 16.34 | 16.57 | 0.444 | 16.90 | 16.31 | 0.374 | 0.684 | 0.439 | 0.024 |
| Total gain | 2.08c | 2.68a | 2.55b | 0.077 | 2.4a | 2.6b | 0.008 | 0.04 | 0.430 | 0.035 |
| ADG (g/d) | 35.0b | 45.0a | 43.0a | 0.002 | 40.0 | 43.0 | 0.001 | 0.003 | 0.44 | 0.030 |
| FCR | 19.16a | 15.44b | 18.11a | 0.155 | 17.2 | 16.65 | 0.023 | 0.030 | 0.200 | 0.320 |
Means with different superscript letters within treatments and sex in the same row, differ significantly (P < 0.05). T1= 71% Vigna hay, 19% Katambora hay salt (0.5%), molasses (3%), maize (5%) and mineral vitamin premix 1.5%; T2= 90% lamb and ewe pellet plus 10% Katambora grass hay; T3= 72% Lablab hay 19% Katambora hay, salt (0.5%), molasses (2%) maize (5%) and mineral vitamin premix 1.5% IW – initial weight, FW = final weight.
3.3. Biochemical indices
The effects of diet, sex and time on biochemical indices was evaluated in this study and results are shown in Table 3, Table 4 and Fig. 1. The urea (mmol/l), TP (g/l), creatinine (µm/l), globulins (g/L) and cholesterol (mmol/l) levels; and activities (IU/L) of ALP, GGT and ALT were similar among treatments and between sexes (Table 3). Feeding forage legume diets to goats had no significant effects (P > 0.05) on serum TP, globulin, urea, creatinine, ALT, ALP and GGT concentrations. However, the serum concentration of albumin was influenced by diet (P < 0.05). Sex of animals did not affect (P > 0.05) serum TP, globulin, urea, creatinine, ALT, and GGT, however sex influenced ALP concentration (P > 0.05). Measured over time, the plasma concentration of urea and ALP significantly increased (P < 0.05) while creatinine and cholesterol concentration decreased (P < 0.05) as shown in Table 4. No changes were observed for TP, albumin and globulins with time. Although the overall serum concentration of urea, TP, globulin, creatinine, ALT, ALP and GGT remained unchanged among the treatments and between sexes; treatment by sex (Table 5), treatment by day (Table 6) and sex by day (Table 7) interactions were significant (P < 0.05). The female animals on T1 exhibited higher (P < 0.05) urea, creatinine, ALP and cholesterol concentrations than contemporaries on other treatments. Urea, creatinine and albumin concentrations were higher (P < 0.05) in male animals on T3. Female animals on T2 showed the lowest creatinine and ALT levels (P < 0.05). Male animals on T1 had the lowest (P < 0.05) cholesterol and urea concentrations but recorded the highest TP. Female animals on T3 had the highest albumin concentration while male animals on T2 showed the least albumin concentration (P < 0.05). On average male animals exhibited higher (P < 0.05) urea, creatinine, ALT and cholesterol concentrations while female animals showed higher (P < 0.05) albumin and ALP plasma concentration. Time × treatment affected (P < 0.05) the urea, creatinine, TP, albumin globulin ALT ALP and cholesterol levels in blood serum of Xhosa goats.
Table 3.
Biochemical attributes of goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing Chloris gayana.
| Treatments (T) |
Sex (S) |
P values |
Merck, 2016 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Plasma parameters | T1 | T2 | T3 | SE | M | F | SE | T | S | |
| Urea (mmol/l) | 4.91 | 5.62 | 4.87 | 0.370 | 5.19 | 5.08 | 0.302 | 0.291 | 0.804 | 5.6- 7.691 |
| Total protein(g/dl) | 56.43 | 57.31 | 56.17 | 1.953 | 57.78 | 55.49 | 1.595 | 0.911 | 0.317 | 64 −78 |
| Creatinine (mg/dl) | 0.571 | 0.565 | 0.631 | 0.302 | 0.598 | 0.579 | 0.246 | 0.236 | 0.609 | 0.5 −1.8 |
| Globulin(g/l) | 41.96 | 43.11 | 40.61 | 1.902 | 43.48 | 40.31 | 1.553 | 0.652 | 0.157 | 35 - 451 |
| Albumin(g/l) | 14.47b | 14.19b | 15.56a | 0.404 | 14.30 | 15.19 | 0.330 | 0.045 | 0.065 | 20 - 401 |
| Cholesterol(mmol/l) | 1.601 | 1.81 | 1.578 | 0.129 | 1.68 | 1.651 | 0.105 | 0.371 | 0.857 | 1.5 – 1.91 |
| ALT(U/l) | 19.92 | 15.78 | 20.78 | 1.852 | 19.78 | 17.87 | 1.453 | 0.121 | 0.366 | 15.3 −52.3 |
| ALP(U/l) | 123.92 | 95.75 | 107.11 | 31.050 | 53.67b | 164.19a | 25.352 | 0.213 | 0.004 | 66 −230 |
| GGT(U/l) | 37.86 | 45.81 | 43.56 | 3.434 | 39.89 | 44.93 | 2.803 | 0.255 | 0.212 | 20 −50 |
Means with different superscript letters within treatments and sex in the same row, differ significantly (P < 0.05). ALT = alanine aminotransferase, ALP = alkaline phosphatase, GGT= gamma- glutamyl transferase. T1 = 71% Vigna hay, 19% Katambora hay, salt (0.5%), molasses (3%) maize (5%) and mineral vitamin premix (1.5%); T2= 90% lamb and ewe pellet plus 10% Katambora grass hay; T3 = 72% Lablab hay 19% Katambora hay, salt (0.5%), molasses (2%) maize (5%) and mineral vitamin premix (1.5%).
Table 4.
Changes in blood biochemical attributes of goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing Chloris gayana.
| Day(D) |
SE | P value | |||
|---|---|---|---|---|---|
| 15 | 30 | 45 | D | ||
| Urea | 4.76b | 4.26b | 6.38a | 0.370 | 0.0007 |
| Creatinine | 69.94a | 56.47b | 50.47b | 3.015 | 0.0002 |
| TP | 58.83 | 53.39 | 57.68 | 1.95 | 0.1304 |
| Albumin | 15.11 | 14.50 | 14.61 | 0.400 | 0.5281 |
| Globulins | 43.72 | 38.89 | 43.07 | 1.902 | 0.1642 |
| ALT | 19.50 | 19.3 | 17.67 | 1.779 | 0.7300 |
| ALP | 61.89c | 90.83b | 174.05a | 0.495 | 0.0495 |
| GGT | 37.44 | 45.81 | 43.97 | 3.434 | 0.2086 |
| Cholesterol | 2.57a | 1.35bc | 1.067c | 0.129 | 0.0001 |
Means with different superscript letters within rows, differ significantly (P < 0.05). TP = total protein, ALT = alanine aminotransferase, ALP = alkaline phosphatase, GGT= gamma- glutamyl transferase.
Fig. 1.
Changes in blood haematological attributes of goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing Chloris gayana.
Table 5.
Interactive effects of diet and sex on goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing Chloris gayana .
| Treatment |
SE | P value | ||||||
|---|---|---|---|---|---|---|---|---|
| 1 | 1 | 2 | 2 | 3 | 3 | |||
| Sex |
||||||||
| F | M | F | M | F | M | |||
| Urea | 5.58ab | 4.24b | 5.42ab | 5.82a | 4.24b | 5.50ab | 0.382 | 0.041 |
| Creatinine | 61.44ab | 52.67ab | 52.17b | 60.78ab | 60.3ab | 65.89a | 4.264 | 0.0385 |
| TP | 54.19 | 58.67 | 57.94 | 56.67 | 54.33 | 52.56 | 3.546 | 0.622 |
| Albumin | 14.94ab | 14.00ab | 15.06a | 13.33b | 15.56a | 15.56a | 0.571 | 0.0310 |
| Globulins | 39.25 | 44.67 | 42.89 | 43.33 | 38.78 | 38.56 | 3.181 | 0.629 |
| ALT | 17.06ab | 22.78a | 15.33b | 16.22ab | 21.22ab | 20.33ab | 2.718 | 0.0424 |
| ALP | 201.50a | 46.33b | 123.28ab | 68.22b | 167.78ab | 46.44b | 46.444 | 0.0401 |
| GGT | 43.38 | 32.33 | 45.83 | 45.78 | 45.56 | 41.56 | 0.524 | 0.521 |
| Cholesterol | 1.95ab | 1.26c | 1.56abc | 2.07a | 1.44bc | 1.71abc | 0.182 | 0.005 |
Means with different superscript letters within treatments and sex in the same row, differ significantly (P < 0.05). TP = total protein, ALT = alanine aminotransferase, ALP = alkaline phosphatase, GGT= gamma- glutamyl transferase. T1 = 71% Vigna hay, 19% Katambora hay, salt (0.5%), molasses (3%), maize (5%) and mineral vitamin premix (1.5%); T2= 90% lamb and ewe pellet plus 10% Katambora grass hay; T3 = 72% Lablab hay 19% Katambora hay, salt (0.5%), molasses (2%) maize (5%) and mineral vitamin premix (1.5%).
Table 6.
Interactive effects of diet and days on goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing Chloris gayana.
| Treatments |
SE | P value | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 1 | 1 | 2 | 2 | 2 | 3 | 3 | 3 | |||
| Days |
|||||||||||
| 15 | 30 | 45 | 15 | 30 | 45 | 15 | 30 | 45 | TD | ||
| Urea | 4.78bd | 3.24d | 6.71a | 4.94abcd | 5.66abc | 6.27ab | 4.567bcd | 3.88cd | 6.17ab | 0.6410 | 0.039 |
| Creatinine | 66.67ab | 54.25bcd | 50.25cd | 70.50a | 50.25cd | 48.67d | 72.67a | 64.17abc | 52.50bcd | 5.222 | 0.022 |
| TP | 63.50a | 50.50bc | 55.29abc | 56.33abc | 55.00abc | 60.58ab | 56.67abc | 46.50 | 57.17abc | 4.343 | 0.046 |
| Albumin | 15.17ab | 14.33ab | 13.92b | 13.83b | 13.83b | 14.92ab | 16.33a | 15.33ab | 15.00ab | 0.6994 | 0.043 |
| Globulins | 48.33a | 36.17bc | 41.38abc | 42.50abc | 41.17abc | 45.67ab | 40.33abc | 33.50c | 42.17abc | 3.896 | 0.041 |
| ALT | 19.17ab | 19.08ab | 21.50ab | 17.50ab | 14.67b | 15.17b | 21.83ab | 24.17a | 16.33ab | 3.0813 | 0.043 |
| ALP | 90.67ab | 118.25ab | 162.83ab | 50.00b | 88.25ab | 149.00ab | 45.00b | 66.00ab | 210.33a | 53.779 | 0.041 |
| GGT | 36.67 | 38.00 | 38.92 | 36.83 | 50.42 | 50.17 | 38.83 | 49.00 | 42.83 | 5.948 | 0.791 |
| Cholesterol | 2.35a | 1.49b | 0.97b | 2.95a | 1.14b | 1.35b | 2.41a | 1.43b | 0.88b | 0.223 | 0.0211 |
Means with different superscript letters within rows, differ significantly (P < 0.05).TP = total protein, ALT = alanine aminotransferase, ALP = alkaline phosphatase, GGT= gamma- glutamyl transferase. T1 = 71% Vigna hay, 19% Katambora hay, salt (0.5%), molasses (3%), maize (5%) and mineral vitamin premix (1.5%); T2= 90% lamb and ewe pellet plus 10% Katambora grass hay; T3 = 72% Lablab hay 19% Katambora hay, salt (0.5%), molasses (2%) maize (5%) and mineral vitamin premix (1.5%).
Table 7.
Interactive effects of Sex and days on goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing Chloris gayana.
| Sex |
SE | P value | ||||||
|---|---|---|---|---|---|---|---|---|
| F |
M |
|||||||
| Days |
||||||||
| 15 | 30 | 45 | 15 | 30 | 45 | SD | ||
| Urea | 4.68bc | 3.92c | 6.64a | 4.85bc | 4.60c | 6.12ab | 0.523 | 0.0530 |
| Creatinine | 62.89b | 57.78bc | 53.28bc | 77.00a | 54.67bc | 47.67c | 4.264 | 0.0539 |
| TP | 60.00 | 50.11 | 56.36 | 57.67 | 51.22 | 59.00 | 3.546 | 0.7740 |
| Albumin | 14.56abc | 15.22ab | 15.78a | 15.67a | 13.78bc | 13.44c | 0.571 | 0.0131 |
| Globulins | 45.44a | 34.89b | 40.58ab | 42.00ab | 39.00ab | 45.56a | 3.181 | 0.0357 |
| ALT | 16.67 | 19.61 | 17.33 | 22.33 | 19.00 | 18.00 | 2.516 | 0.4360 |
| ALP | 84.67b | 133.89b | 274.00a | 39.11b | 47.78b | 74.11b | 43.91 | 0.0204 |
| GTP | 42.67ab | 47.28a | 44.83ab | 32.22b | 44.33ab | 43.11ab | 4.857 | 0.6271 |
| Cholesterol | 2.48a | 1.30b | 1.18b | 2.67a | 1.41b | 0.96b | 0.182 | 0.0493 |
Means with different superscript letters within sex and days in the same row, differ significantly (P < 0.05). TP = total protein, ALT = alanine aminotransferase, ALP = alkaline phosphatase, GGT= gamma- glutamyl transferase.
For all treatments, day 30 recorded lower (P < 0.05) urea, TP and globulin concentration and day 45 showed higher urea TP and ALP levels. There was a general increase in plasma metabolites with time among treatments (P < 0.05) with a general decline in creatinine and cholesterol levels with time for both sexes (P < 0.05) this is however coinciding with a sharp increase in urea from day 30 to day 45 of 2.72 mmol/l; and 1.52 mmol/l for female and male respectively. Female animals exhibited higher ALT and albumin levels over time than male animals (P < 0.05).
3.4. Haematological parameters
The WBC (103/l), RBC, MCH (pg) and MCHC (g/dL), Hb, haematocrit, platelet and MPV levels were similar among treatments and between sexes (Table 8). The MCV and RDW (%) were significant difference amoung treatments (P < 0.05). Goats under T3 showed increased (P < 0.05). MCV compared to T1 and T2. When the goats were fed T1 their RDW level was higher (P < 0.05) in comparison to T2 and T3. Haematological constituents over time are shown in Fig. 1. The WBC and MCV count differed with time (P < 0.05). WBC decreased with time by 8.07 × 10 9/l while MCV increased by 11.83(fl) from day 15 to day 45. There were no significant differences in RBC (x1012/l), HB, haematocrit, MCH (pg) MCHC (g/dl), RDW% platelet (x10 9/l) and MPV (fl) levels with time.
Table 8.
Haematological attributes of goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing Chloris gayana.
| Treatments |
Sex |
P values |
Merck, 2016 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Blood parameter | T1 | T2 | T3 | SE | M | F | SE | T | S | |
| WBC (x109/l) | 11.2 | 12.05 | 15.17 | 1.634 | 13.63 | 11.98 | 1.315 | 0.2066 | 0.384 | 4 −13 |
| RBC(x1012/l) | 16.67 | 18.49 | 16.56 | 1.083 | 16.84 | 17.64 | 0.872 | 0.3690 | 0.525 | 8 −18 |
| Haemoglobin(g/L) | 87.89 | 92.50 | 89.50 | 0.354 | 86.10 | 93.90 | 0.285 | 0.6476 | 0.062 | 80 - 120 |
| Haematocrit (%) | 9.30 | 10.1 | 9.90 | 0.89 | 9.50 | 10.10 | 0.71 | 0.7549 | 0.519 | 10 - 30 |
| MCV(fl) | 54.67ab | 52.52c | 59.22a | 2.806 | 55.66 | 55.28 | 2.258 | 0.0239 | 0.530 | 16 −25 |
| MCH(pg) | 5.59 | 5.17 | 6.16 | 0.546 | 5.35 | 5.92 | 0.439 | 0.4352 | 0.367 | 5.2 - 8 |
| MCHC(g/dl) | 10.56 | 11.09 | 12.41 | 1.970 | 10.36 | 12.34 | 1.586 | 0.7920 | 0.385 | 30 - 36 |
| RDW (%) | 45.84a | 34.31b | 35.46b | 2.527 | 38.95 | 38.11 | 2.035 | 0.0042 | 0.774 | 30 - 451 |
| Platelet (x109/l) | 689.24 | 577.86 | 755.83 | 120.166 | 693.59 | 775.70 | 96.74 | 0.264 | 0.233 | 300 - 600 |
| MPV(fl) | 6.88 | 6.31 | 6.94 | 0.225 | 6.56 | 6.86 | 0.1794 | 0.0927 | 0.247 | 3.5 - 12 |
Means with different superscript letters within treatments and sex in the same row, differ significantly (P < 0.05). MCH= mean cell haemoglobin, MCHC= mean corpuscular haemoglobin concentration, MCV= mean cell volume, MPV= mean platelet volume, RDW= red cell distribution width.
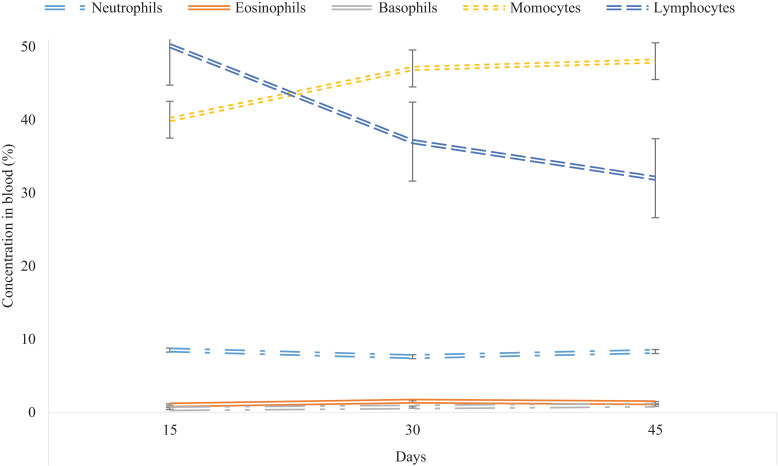
Component WBCs were also evaluated and results are shown in Table 9. Treatment diets influenced (P < 0.05) neutrophil (%) content while sex influenced basophile (%) content. There were no treatment or sex effects on lymphocytes (%), monocytes (%) and eosinophils (%) (P > 0.05). Animals on T2 exhibited higher (P < 0.05) neutrophil content of 10.50% compared to 5.47% and 4.58% for T1 and T3 respectively. Measured over time, the basophil content increased while the lymphocyte content decreased with time (P < 0.05) as shown in Fig. 2.
Table 9.
White blood cell component of goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing Chloris gayana.
| Treatments (T) |
Sex |
P values |
Merck, 2016 | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| WBCs | T1 | T2 | T3 | SE | M | F | SE | T | S | |
| Neutrophils (%) | 5.47ab | 10.50a | 4.58b | 1.910 | 5.026 | 8.67 | 1.555 | 0.0403 | 0.104 | 30 −40 |
| Lymphocytes (%) | 47.24 | 35.72 | 39.54 | 5.396 | 41.02 | 40.65 | 4.3417 | 0.3191 | 0.9520 | 50 −70 |
| Monocytes (%) | 38.62 | 53.20 | 43.29 | 8.869 | 44.57 | 45.50 | 7.1400 | 0.4923 | 0.9275 | 0 - 4 |
| Eosinophils (%) | 1.22 | 1.57 | 1.09 | 0.2878 | 1.24 | 1.34 | 0.2317 | 0.433 | 0.7670 | 1 – 8 |
| Basophils (%) | 0.81 | 0.78 | 0.60 | 0.09 | 0.62b | 0.48a | 0.0694 | 0.1947 | 0.0347 | 0 −1 |
Means with different superscript letters within treatments and sex in the same row, differ significantly (P < 0.05).
Fig. 2.
Changes in blood WBC attributes of goats fed concentrate pellets, Lablab purpureus and Vigna unguiculata supplementing a Chloris gayana basal diet.
4. Discussion
4.1. Feed intake and growth
The daily DMI of goats in this experiment is within the range of 2.6 to 4.0% reported for South African goats by Dzakuma, Risch, Smith, and Blackburn (2004) and Asnakew (2007). Animals showed significantly low forage intake for Vigna compared to Lablab and pellets; thus it affects their growth rate. The differences in total gain among treatments were due to differences in DMI. Results observed for average daily gain in the present study, were within the acceptable range of 29 to 51 g/day reported for Small East African goats (Wallie, Mekasha, Urge, Abebe, & Goetsch, 2012). However, this gain was slightly higher than what was observed by Ameha, Casey, van Niekerk, Azage, and Coertze (2007) and Seid et al. (2012). Growth rates between 36 – 53 g/day (Mengistu, Dahlborn, & Olsson, 2007) and 42–65 g/day (Solomon, Melaku, & Tolera, 2008) have also been reported for goats. These animals used by the mentioned authors were fed concentrate as supplement to a basal hay diet and that is the reason for higher gains compared to the current study. Female animals gained more in the current study, the reason for this is that female animals are known to lay fat faster than males (Louveau & Gondret, 2004) and that castration tend to prolong skeletal growth in male animals, particularly of long bones, accompanied by a retarded increase in bone diameter (Webb, Casey, & Simela, 2016). This explains the difference in total gain in the current study.
4.2. Serum biochemical indices
Blood urea has been shown to be an indirect indicator of protein composition of feed. High serum urea concentrations are associated with greater ruminal degradation of protein with a concurrent increase in ammonia production (Kanyinji, Kumagai, Maeda, Kaneshima, & Yokoi, 2009). Brickell, McGowan, and Wathes (2009) also reported that blood urea nitrogen increases when there is a catabolism of body protein. Furthermore, Lehninger, Nelson, and Cox (2013) observed that high cortisol levels stimulate the breakdown of muscle proteins. This leads to the deamination and release of amino acids in the liver for the purposes of gluconeogenesis. The serum urea levels in the current study are below those reported by Malecky et al. (2017); Wang, Fang, Xing, and Yang (2008). However, they fall within the same range of 3.6 to 9.2 mmol/l reported by Carlos et al. (2015), Kaneko, Harvey and Bruss (2008) and Radostits et al. (2002) for healthy animals. Urea concentration increased with time in the present study, this was also observed by Malecky et al. (2017) and Mani and Chandra (2005). The possible reason for this increase could be the increase in DM intake exhibited by goats with time. Overall serum urea concentration remained unchanged among the treatments and between sexes, however treatment by sex, treatment by day and sex by day interactive effect on urea were different. Blood urea N has been reported to increase in cases of rumen ungradable protein and where very large volumes of amino acids are consumed (Wang et al., 2008). Creatinine is formed in skeletal muscle by the degradation of phosphocreatine to produce energy (Carlos et al., 2015). Serum creatinine concentration has been observed to be proportional to the muscle mass (Carlos et al., 2015) and this explains the higher creatinine content recorded in males compared to females in the present study, although not significant. Increase in creatinine levels are associated with reduction of serum thyroxine levels (Yokus, Cakir, Kanay & Uysal, 2006). A general decline in creatinine levels was observed with time. This would suggest that there was an increase in thyroxine levels hence better energy utilization.
The TP for goats was between 5.5 and 5.8 g/dl. These values are considered lower than the normal range value of 6.0 to 7.0 g/dl (Malecky, Ghadbeigi, Aliarabi, Bahari & Zaboli, 2017; Mohammed et al., 2016; Wang et al., 2008). However, these values fall within a range reported by Carlos et al. (2015) in sheep with a body condition score of 2.5, and that recorded by Opara, Udevi and Okoli (2010) of 5.2 g/dl for West African Dwarf goats. According to Akinrinmade and Akinrinde (2012) goats with a TP value below 4.2 g/dl show rumen compaction, this was therefore not the case in this study. Mohammed et al. (2016) also reported that total protein value in goat serum can go as high as 7.5 g/dl under extensive production systems. Furthermore, they also reported that high serum protein levels are an indication of high intake of grains (Sandabe & Chaudhary, 2000), dehydration or high temperature as a result of kidney failure. Therefore, lower protein values occurred because animals did not consume any considerable amount of grain. Total protein level generally increased with time for treatments two (T2) and three (T3) and this improvement is suggestive of the fact that more amino acids were absorbed from the dietary protein and undegradable methionine, as was also observed by Abdelrahman and Aljumaah (2014).
The albumin content was within the acceptable range, according to Raju et al. (2015). Tripathi, Mondal and Karim (2007) reported that low serum albumin concentration is indicative of liver dysfunction, nonetheless other indicators of liver function; ALT, ALP and GGT were not different among treatments and between sexes, indicating no negative effects of diets on the liver.
The decrease in cholesterol content, with an increase in ALP content over time has been observed by Khalel et al. (2014) and Min et al. (2012), however, Elitok (2012) and Mani and Chandra (2005) observed higher values of ALP within the first 40 days of life of treatment time with an increase cholesterol content over time. A decrease in cholesterol concentration over time could have been caused by the presence of phytochemicals particularly in forages as these have the capacity to reduce the synthesis and absorption of cholesterol (Saxena, Saxena, Nema, Singh & Gupta, 2013). Moreover, reduced blood cholesterol and lipid levels are some of the biological consequences of phenolic acids.
There was also a treatment by sex, treatment by day and sex by day interaction effect on albumin, ALP, ALT cholesterol concentration (Malecky et al., 2017). Globulins were also significantly influenced by both treatment and days. Elitok (2012) also reported that GGT is the first enzyme to increase even at the slightest hepatic sufferance. Results from the current study indicate that animals on T2 recorded higher GGT values compared to T1 and T3; this would suggest that Cowpea and Lablab does not produce toxic substances on degradation to warrant any effect on hepatocyte health. The globulin content decreased with time, this is in agreement with Carlos et al. (2015) in which they concluded that globulins only increase under pathological conditions. This also attests to the fact that treatment diets did not result in any inflammatory response in goats.
4.3. Haematological parameters
The blood haemoglobin (g/dL), hematocrite (L/L), RBC (106/l), WBC (10 9/l) MCH (pg), MCHC (g/dL), MPV, were not influenced by diet or sex. However, these results were within the ranges of 8.0 −13 g/dl, 20–28% and 8–17 × 1012 L, 5.2 – 8pg, 30 – 36 g/dl, and 5.3–9.4fL for Hb, PCV, RBC, WBC, MCH, MCHCC, and MPV respectively, reported for clinically healthy goats (Merck, 2016; Sirois, 1995). The normal RBC values observed in this study indicate the absence of haemolytic anaemia and depression of erythrogenesis (Olafadehan, 2011). Similar haemoglobin concentrations were observed by Olafadehan (2011) and are suggestive of the absence of microcytic hypochromic anaemia which is associated with iron deficiency and improper utilization during the formation of Hb. However, Solaiman et al. (2010) observed higher Hb values when kids were fed diet containing 22.2 g CT/kg DM.
The fact that Lablab only resulted in higher MCV (fL) levels while Cowpea affected RDW (%) levels concur with those reported by Durge, Tripathi, Dutta, Rout and Chaudhary (2014) in which they concluded that concentrate diets did not significantly influence most of the blood metabolites. The lack of treatment effect on MCV, MCH and MCHC values also agree with earlier reports (Solaiman et al., 2010). Measured against time, animals exhibited very high WBC during adaptation period which significantly decreased until day of slaughter. Time did not influence RBC, haemoglobin, MCH, MPV, RDW, MCV, however MCHC significantly increased at day 30 and decreased at day 45.
MCV, MCH and MCHC are diagnostic blood indices indicative of anaemic animals. When they fall within normal ranges (Sirois, 1995), it explicitly shows that the goats are not anaemic (Olafadehan, 2011). This lack of effects on WBC over time, between sexes and/or treatment diets represents no inflammatory effects of forage legumes on growing goats; this was also reported by Malecky et al. (2017). No studies have reported either inflammatory or haemolytic effects of forage legumes. Platelet count was higher than the recommended values of 300–600 by Marck Veterinary. This could have resulted from physiological thrombocytosis which is caused by exercise, stress, or adrenaline (Malecky et al., 2017). In this case the only logical conclusion would be the influence of adrenaline coupled with stress, as goats don't like confinement for long periods. Component WBCs particularly monocytes and lymphocytes are essential for the immune system. Monocytes are precursors of macrophages and lymphocytes are important for humoral and cell-mediated immunity responses (Mahgoub et al., 2008). In general, toxic substances in feed have a suppressive effect on haemopoietic tissues with consequent production of a low WBC count (Mahgoub et al., 2008; Malecky et al., 2017). The results from the current study show no such effects as was also reported by Mahgoub et al. (2008).
5. Conclusion
Lablab and Cowpea can support the same level of growth as commercial pellets. Lablab exhibited a significantly higher DM intake, ADG, total gain and lower FCR than Cowpea. Male goats consumed more feed, but total gain and ADG were low, with a higher FCR than female goats. Forage legumes did not influence biochemical parameters of goats, save for albumin in which Lablab exhibited higher albumin levels. Sex influenced only ALP levels with female animals showing higher ALP values. Urea and ALP increased with time while creatinine and cholesterol decreased until they reached a steady state which is within the generally acceptable levels for goats. Haematological parameters showed healthy goats throughout the study period although the platelet count was higher than the normal range. Animals didn't show any signs of haemolysis, anaemia, or inflammatory response to treatment diets.
Declaration of Competing Interest
All authors declare that there are no present or potential conflicts of interest among the authors and other people or organizations that could inappropriately bias their work.
Acknowledgments
Acknowledgement
This research was financially supported by the South African Government through the GMRDC- NRF fund T 579.
Ethical Statement
None.
References
- Abdelrahman M.M., Aljumaah R.S. Dietary protein level and performance of growing baladi kids. Iranian Journal of Veterinary Research. 2014;15:353–358. [PMC free article] [PubMed] [Google Scholar]
- Akinlade J.A., Smith J.W., Raji A.M., Busari A.A., Adekunle I.O., Adewumi M.K. Effect of two cowpea (Vigna unguiculata) fodder cultivars as supplements on voluntary intake, milk yield and manure production of bunaji cows. Journal of Agriculture and Rural Development in the Tropics and Subtropics. 2005;106(2):105–111. [Google Scholar]
- Akinrinmade J.F.I., Akinrinde A.S. Hematological and serum biochemical indices of west african dwarf goats with foreign body rumen impaction. Nigerian Journal Physiology Science. 2012;27:83–87. [PubMed] [Google Scholar]
- Al-Eissa M.S., Alkahtani S., Al-Farraj S.A., Alarifi S.A., Al-Dahmash B., AlYahya H. Seasonal variation effects on the composition of blood in nubian ibex (Capra nubiana) in saudi arabia. African Journal Biotechnology. 2012;11(5):1283–1286. [Google Scholar]
- Al-Seaf A.M., Al-Harbi K.B. Variability of disease resistance, haematological parameters and lymphocyte proliferation in two goat breeds and their F1 and F2 crosses. International Journal of Food Agriculture and Veterinary Science. 2012;2:47–53. [Google Scholar]
- Ambore B., Ravikanth K., Maini S., Rekhe D.S. Haematological profile and growth performance of goats under transportation stress. Veterinary World. 2009;2(5):195–198. [Google Scholar]
- Ameha S., Casey N.H., van Niekerk W.A., Azage T., Coertze R.J. Growth performance and carcass characteristics of three ethiopian goat breeds fed grainless diets varying in concentrate to roughage ratios. South African Journal of Animal Science. 2007;37:221–232. [Google Scholar]
- Asnakew A.B. Feed intake, live weight gain and carcass yield characteristics of intact hararghe highland male goats fed on different hay to concentrate ratios. East African Journal of Science. 2007;1:45–54. [Google Scholar]
- Ayan I., Mut H., Basaran U., Eki Acar Z., Asci O.O. Forage potential of cowpea (Vigna unguiculata L. WALP) Turkish Journal of Field Crops. 2012;17(2):135–138. [Google Scholar]
- Bayble T., Melaku S., Prasad N.K. Effects of cutting dates on nutritive value of napier (Pennisetum purpureum) grass planted sole and in association with desmodium (Desmodium intortum) or lablab (Lablab purpureus) Livestock Research for Rural Development. 2007;19 http://www.lrrd.org/lrrd19/1/bayb19011.htm Article #11. Retrieved January 30, 2018, from. [Google Scholar]
- Brickell J.S., McGowan M.M., Wathes D.C. Effect of management factors and blood metabolites during the rearing period on growth in dairy heifers on uk farms. Domestic Animal Endocrinology. 2009;36:67–81. doi: 10.1016/j.domaniend.2008.10.005. [DOI] [PubMed] [Google Scholar]
- Carlos M.M.L., Leite J.H.G.M., Chaves D.F., Vale A.M., Façanha D.A.E., Melo M.M. Blood parameters in the morada nova sheep: Influence of age, sex and body condition score. Journal of Animal and Plant Science. 2015;25:950–955. [Google Scholar]
- Directorate Agricultural Information Services. (2011). Department of Agriculture, Forestry and Fisheries. Private Bag X144, Pretoria, 0001 South Africa.
- Durge S.M., Tripathi M.K., Dutta N., Rout P.K., Chaudhary U.B. Intake, nutrient utilization, rumen fermentation, microbial hydrolytic enzymes and haemato-biochemical attributes of lactating goats fed concentrate containing brassica juncea oil meal. Small Ruminant Research. 2014;121:300–307. [Google Scholar]
- Dzakuma J.M., Risch E., Smith C.O., Blackburn H.D. Level of feed intake on performance of two goat genotypes. South African Journal of Animal Science. 2004;34:38–41. [Google Scholar]
- Elitok B. Reference values for haematological and biochemical parameters in saanen goats breeding in afyonkarahisar province. Kocatepe Veterinary Journal. 2012;5:7–11. [Google Scholar]
- FAO . Food and Agriculture Organization of the United Nations. Viale delle Terme di Caracalla; 2014. Understanding the drought impact of el nino on the global agricultural areas: An assessment using FAO's agricultural stress index (ASI) Rome, Italy. [Google Scholar]
- Gebregiorgis F., Negesse T., Nurfeta A. Feed intake and utilization in sheep fed graded levels of dried moringa (Moringa stenopetala) leaf as a supplement to rhodes grass hay. Tropical Animal Health and Production. 2011;41:1–6. doi: 10.1007/s11250-011-9927-9. [DOI] [PubMed] [Google Scholar]
- Gebreyowhans S., Gebremeskel K. Forage production potential and nutritive value of cowpea (Vigna unguiculata) genotypes in the northern lowlands of ethiopia. Journal of Agricultural Research and Development. 2014;5(4):066–071. [Google Scholar]
- Jingura R.M., Sibanda S., Hamudikuwanda H. Yield and nutritive value of tropical forage legumes grown in semi-arid parts of zimbabwe. Tropical Grasslands. 2001;35:168–174. [Google Scholar]
- Kaneko J.J., Harvey J.W., Bruss M.L. 6th ed. Academic Press; Burlington, MA: 2008. Clinical biochemistry of domestic animals. [Google Scholar]
- Kanyinji F., Kumagai H., Maeda T., Kaneshima S., Yokoi D. Effects of supplementary inosine on nutrient digestibility, ruminal fermentation and nitrogen balance in goats fed high amount of concentrate. Animal Feed Science Technology. 2009;152:12–20. [Google Scholar]
- Khalel M.S., Shwerab A.M., Hassan A.A., Yacout M.H., El-Badawi A.Y., Zaki M.S. Nutritional evaluation of moringa oleifera fodder in comparison with trifolium alexandrinum (berseem) and impact of feeding on lactation performance of cows. Life Science Journal. 2014;11:1040–1054. [Google Scholar]
- Lehninger A.L., Nelson D.L., Cox M.M. W.H. Freeman; New York: 2013. Lehninger principles of biochemistry. [Google Scholar]
- Louveau I., Gondret F. Regulation of development and metabolism of adipose tissue by growth hormone and the insulin-like growth factor system. Domestic Animal Endocrinology. 2004;27:241–255. doi: 10.1016/j.domaniend.2004.06.004. [DOI] [PubMed] [Google Scholar]
- Maasdorp B.V., Titterton M. Nutritional improvement of maize silage for dairying: Mixed-crop silages from sole and intercropped legumes and a long-season variety of maize. biomass yield and nutritive value. Animal Feed Science and Technology. 1997;69:241–261. [Google Scholar]
- Mahala A.G., Amasiab S.O., Yousif M.A., Elsadig A. Effect of plant age on dm yield and nutritive value of some leguminous plants (Cyamopsis tetragonoloba, lablab purpureus and clitoria ternatea) International Research Journal of Agricultural Science and Soil Science. 2012;2(12):502–508. [Google Scholar]
- Mahgoub O., Kadim I.T., Tageldin M.H., Al-Marzooqi W.S., Khalaf S.Q., Ali A.A. Clinical profile of sheep fed non-conventional feeds containing phenols and condensed tannins. Small Ruminant Research. 2008;78:115–122. [Google Scholar]
- Malecky M., Ghadbeigi M., Aliarabi H., Bahari A.A., Zaboli K. Effect of replacing alfalfa with processed potato vines on growth performance, ruminal and total tract digestibility and blood metabolites in fattening lambs. Small Ruminant Research. 2017;146:13–22. [Google Scholar]
- Mani V., Chandra P. Effect of feeding -irradiated soybean on growth performance and blood metabolites in barbari kids. Small Ruminant Research. 2005;57:213–219. [Google Scholar]
- McDonald P., Edwards R.A., Greenhalgh J.F.D., Morgan C.A. 7th Ed. Cape Town, South Africa; 2011. Animal nutrion. [Google Scholar]
- Mengistu U., Dahlborn K., Olsson K. Effect of intermittent watering on growth, thermoregulation and behaviour of ethiopian somali goat kids. Small Ruminant Research. 2007;72:214–220. [Google Scholar]
- Merck Veterinary Manual (2016). http://www.merckvetmanual.com/appendixes/reference-guides/hematologic-reference-ranges[accessed 20december 2016].
- Min H.K., Kapoor A., Fuchs M., Mirshahi F., Zhou H., Maher J. Increased hepatic synthesis and dysregulation of cholesterol metabolism is associated with the severity of non-alcoholic fatty liver disease. Cell Metabolism. 2012;15:665–674. doi: 10.1016/j.cmet.2012.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohammed S.A., Razzaque M.A., Omar A.E., Albert S., Al-Gallaf W.M. Biochemical and haematological profile of different breeds of goat maintained under intensive production system. African Journal of Biotechnology. 2016;15:1253–1257. [Google Scholar]
- NRC . Nutrient requirements of small ruminants. National Academy Press; Washington DC, USA: 2007. [Google Scholar]
- Nurfeta A. Feed intake, digestibility, nitrogen utilization, and body weight change of sheep consuming wheat straw supplemented with local agricultural and agro-industrial by-products. Tropical Animal Health Production. 2010;42(5):815–824. doi: 10.1007/s11250-009-9491-8. [DOI] [PubMed] [Google Scholar]
- Olafadehan O.A. Changes in haematological and biochemical diagnostic parameters of red sokoto goats fed tannin-rich pterocarpus erinaceus forage diets. Veterinarski Archives. 2011;81:471–483. [Google Scholar]
- Opara M.N., Udevi N., Okoli I.C. Haematological parameters and blood chemistry of apparently healthy wad goats in owerri, south eastern nigeria. New York Science Journal. 2010;3:68–72. [Google Scholar]
- Radostits O.M., Blood D.C., Gay C.C. Veterinary Medicine: A Textbook of the Diseases of Cattle, Sheep, Pigs, Goats and Horses 8th Ed. Bailliere Tindall; Britain: 2002. [Google Scholar]
- Raju J., Sahoo B., Chandrakar A., Sankar M., Garg A.K., Sharma A.K. Effect of feeding oak leaves (Quercus semecarpifolia vs quercus leucotricophora) on nutrient utilization, growth performance and gastrointestinal nematodes of goats in temperate sub himalayas. Small Ruminant Research. 2015;125:1–9. [Google Scholar]
- Ravhuhali K.E., Ng'ambi J.W., Norris D. Chemical composition and enzymatic in-vitro digestibility of cowpea cultivars and buffalo grass hay grown in limpopo province of south africa. Livestock Research for Rural Development. 2010;22 http://www.lrrd.org/lrrd22/9/ravh22170.htm Article#170.Retrieved December 3, 2017, from. [Google Scholar]
- Sandabe U.K., Chaudhary S.U.R. Effect of environmental temperature on some biochemical values in female sahel goats. Pakistan Veterinary Journal. 2000;20(1):10–12. [Google Scholar]
- SAS. (2010). SAS Institute Inc, Cary, NC , USA.
- Saxena M., Saxena J., Nema R., Singh D., Gupta A. Phytochemistry of medicinal plants. Journal of Pharmacology and Phytochemicals. 2013;1:168–182. [Google Scholar]
- Seid M., Mengistu U., Getachew A., Kassahun A., Girma A., Goetsch A.L. Effects of level of concentrate supplementation on growth performance of arsi-bale and boer×arsi-bale male goats consuming low-quality grass hay. Tropical Animal Health Production. 2012;44:1181–1189. doi: 10.1007/s11250-011-0056-2. [DOI] [PubMed] [Google Scholar]
- Sirois M. Mosby Year Book, Inc; St Louis, Missouri: 1995. Veterinary clinical laboratory procedure. [Google Scholar]
- Solaiman S., Thomas J., Dupre Y., Min B.R., Gurung N., Terrill T.H. Effect of feeding sericea lespedeza (Lespedeza cuneata) on growth performance, blood metabolites, and carcass characteristics of kiko crossbred male kids. Small Ruminant Research. 2010;93:149–156. [Google Scholar]
- Solomon M., Melaku S., Tolera A. Supplementation of cottonseed meal on feed intake, digestibility, and live weight and carcass parameters of sidama goats. Livestock Science. 2008;119:137–144. [Google Scholar]
- Stella A.V., Paratte R., Valnegri L., Cigalino G., Soncini G., Chevaux E. Effect of administration of live saccharomyces cerevisiae on milk production, milk composition, blood metabolites, and faecal flora in early lactating dairy goats. Small Ruminant Research. 2007;67:7–13. [Google Scholar]
- Tripathi M.K., Mondal D., Karim S.A. Growth, haematology, blood constituents and immunological status of lambs fed graded levels of animal feed grade damaged wheat as substitute of maize. Journal of Animal Physiology and Animal Nutrition. 2007;92:75–85. doi: 10.1111/j.1439-0396.2007.00712.x. [DOI] [PubMed] [Google Scholar]
- Waghorn G.C., Clark D.A. Feeding value of pastures for ruminants. New Zeeland Veterinary Journal. 2004;52(6):320–331. doi: 10.1080/00480169.2004.36448. [DOI] [PubMed] [Google Scholar]
- Wallie M., Mekasha Y., Urge M., Abebe G., Goetsch A.L. Effects of form of leftover khat (Catha edulis) on feed intake, digestion, and growth performance of hararghe highland goats. Small Ruminant Research. 2012;102:1–6. [Google Scholar]
- Wang D., Fang J., Xing F., Yang L. Alfalfa as a supplement of dried cornstalk diets: Associative effects on intake, digestibility, nitrogen metabolisation, rumen environment and haematological parameters in sheep. Livestock Science. 2008;113:87–97. [Google Scholar]
- Washaya S., Mupangwa J., Muchenje V. Chemical composition of Lablab purpureus and Vigna unguiculata and their subsequent effects on methane production in goats. South African Journal of Animal Science. 2018;48:445–458. [Google Scholar]
- Webb, E.C., .Casey, N.H., .& Simela, L. (2016). Growth, development and growth manipulation in goats. https://www.researchgate.net/publication/287318757[accessed 06 December 2017].
- Yokus B., Cakir D.U., Kanay T., Uysal E. Effects of seasonal and physiological variations on the serum chemistry, vitamins and thyroid hormone concentrations in sheep. Journal Veterinary Medicine. 2006;53:271–276. doi: 10.1111/j.1439-0442.2006.00831.x. [DOI] [PubMed] [Google Scholar]
- Žubčić D. Some biochemical parameters in the blood of grazing german improved fawn goats from istria, croatia. Veterinary arhives. 2001;71:237–244. [Google Scholar]