Abstract
Globally, a staggering 310 million major surgeries are performed each year; around 40 to 50 million in USA and 20 million in Europe. It is estimated that 1–4% of these patients will die, up to 15% will have serious postoperative morbidity, and 5–15% will be readmitted within 30 days. An annual global mortality of around 8 million patients places major surgery comparable with the leading causes of death from cardiovascular disease and stroke, cancer and injury. If surgical complications were classified as a pandemic, like HIV/AIDS or coronavirus (COVID-19), developed countries would work together and devise an immediate action plan and allocate resources to address it. Seeking to reduce preventable deaths and post-surgical complications would save billions of dollars in healthcare costs. Part of the global problem resides in differences in institutional practice patterns in high- and low-income countries, and part from a lack of effective perioperative drug therapies to protect the patient from surgical stress. We briefly review the history of surgical stress and provide a path forward from a systems-based approach. Key to progress is recognizing that the anesthetized brain is still physiologically ‘awake’ and responsive to the sterile stressors of surgery. New intravenous drug therapies are urgently required after anesthesia and before the first incision to prevent the brain from switching to sympathetic overdrive and activating secondary injury progression such as hyperinflammation, coagulopathy, immune activation and metabolic dysfunction. A systems-based approach targeting central nervous system-mitochondrial coupling may help drive research to improve outcomes following major surgery in civilian and military medicine.
Keywords: Surgery, Perioperative, Mortality, Morbidity, Trauma, Global
Our findings suggest that surgery now occurs at a tremendous volume worldwide, in settings both rich and poor. … A public-health strategy for surgical care is paramount.
Weiser and colleagues (2008) [1]p143
1. A global healthcare crisis and growing
Mortality during surgery is relatively rare, however, 30-day postoperative mortality is not. Around 234 million surgical operations were performed globally in 2004, and this number increased by 25%–310 million in 2012 [2]. Of these, 40 to 50 million were performed in the USA [3,4] and 20 million in Europe [5]. Estimates of early postoperative mortality rate are 1–4% depending upon country and institution, with the lowest in developed countries and highest in low-income countries [[6], [7], [8], [9], [10]]. These percentages translate to an average global mortality rate of nearly 8 million deaths per year, which is ∼50% lower than cardiovascular disease, and on par with cancer and total injuries [11] (Table 1 ). Global postoperative mortality rate represents up to 14% of the deaths worldwide in 2018. Similar trends are found in the USA where the postoperative mortality rate is 1.3% (Table 2 ). Notwithstanding the difficulty of capturing all major operations from worldwide data within defined parameters [1,2], the healthcare implications are alarming, and support Farmer's characterization, made over a decade ago, that major surgery is “the neglected step-child of global health” [12]. Further, the problem is increasing by ∼400,000 every year, which by 2030, could be as high as 12 million deaths per year from global surgery.
Table 1.
Estimated Annual Global Mortality from Major Surgery compared to Cardiovascular Disease and Stroke, Cancer and Injury.
| Deaths per year (million) | Year | Percent Total Deaths |
Reference | |
|---|---|---|---|---|
| Total All-Aged Deaths | 56.9 | 2017 | 10 | |
| Cardiovascular and Stroke | 17.9 | 2016 | 31% | WHO |
| Cancer | 9.6 | 2018 | 17% | WHO |
| Injury | 5.8 | 2014 | 10% | WHO |
| Major Surgery (1–4% of 310M) | ∼8.0a | See Text | 14% | See Text |
Average mortality. World Health Organization (WHO) data from website.
Table 2.
Estimated annual mortality in the USA from major non-cardiac surgery, cardiovascular disease and stroke, cancer and injury.
| Deaths per year | Year | Percent Total Deaths |
Reference | |
|---|---|---|---|---|
| Total All-Aged Deaths | 2,839,205 | 2018 | 9 | |
| Cardiovascular and Stroke | 793,840 | 2017 | 28% | CDC |
| Cancer | 599,108 | 2017 | 21% | CDC |
| Injury | 169,936 | 2017 | 6% | CDC |
| Major Surgery (inpatient) (1.32% of 50M) |
660,000 | 2006 | 23% | 4, 8 |
Data from Centers for Disease Control (CDC) website.
Equally challenging is surgical morbidity. In 2013, Anderson and colleagues reviewed 16,424 surgical patients from 8 developed countries and reported 14.4% of patients experienced adverse events and over one-third of these were preventable [13]. Other studies have reported over 50% of adverse events were potentially preventable [[13], [14], [15]]. This is a huge window of opportunity to reduce surgical morbidity. Leape and colleagues evaluated nearly 500,000 surgeries in the USA and reported a 30-day readmission rate of 5.7%, with over 30% attributable to surgical site infections [16]. In another study, Dimick and Ghaferi assessed nearly 60,000 patients from 112 US Department of Veterans Affairs hospitals, and found an overall 30-day readmission rate of 12%, twice the number found in Leape's study [17]. Pearse and colleagues conducted another large 7-day cohort study on 46,539 non-cardiac surgery patients from 498 hospitals in 28 European countries, and found that 4% of patients died before hospital discharge, and 8% required critical care with a median length stay of 1–2 days [7]. More recently, an African Surgical Outcomes Study provided further insight into the perioperative problem across countries and institutions. The 2018 study of Biccard and colleagues involving 11,422 in-patient surgeries from twenty-five African countries reported a 10% mortality rate, with 20% of patients developing only one perioperative complication [18]. They concluded in these 247 hospitals that: “Despite a low-risk profile and few postoperative complications, patients in Africa were twice as likely to die after surgery when compared with the global average for postoperative deaths” [18].
Global estimates indicate that on average 8 million patients die every year from major surgery, and up to twice this number experience postoperative complications. Given that 50% of these outcomes are potentially preventable, new management programs and drug therapies could save up to 500 lives every hour and prevent 1000 complications. While not a disease entity, major surgery needs to find a home within the global healthcare system, and a designated Commission needs to be formed to provide sufficient and sustainable resources to address the problem.
2. Surgical stress defined
As the patient goes to the operating room and anesthesia is induced, trauma is suffered and convalescence begins.
Francis D. Moore [19] p291
Surgical stress can be defined as an acute response to one or more breaches of the body's barrier functions from sterile injury (incision, excision, manipulation and pain), pathogen invasion (gut bacterial translocation or postoperative wound infection) and/or anesthesia. After induction of anesthesia and the first incision, the stress response begins with an increase in sympathetic discharge [[20], [21], [22], [23]] which, if not contained, has multiple effects on whole body homeostasis; it increases inflammation [[24], [25], [26], [27]], alters coagulation [[28], [29], [30]], modifies immune competency and T cell mobilization through a β2-adrenergically-mediated process [24,[31], [32], [33]], increases susceptibility to infection [31,34], and reduces tissue oxygenation [23,[35], [36], [37], [38]]. This wave of secondary injury increases the probability of multiple organ dysfunction by altering multiple homeostatic circuits linking brain, heart, lung, kidney, liver, gut and muscle functions [39] (see Fig. 1 ). Reducing surgical stress may reduce the wave of secondary injury, thereby improving patient outcomes.
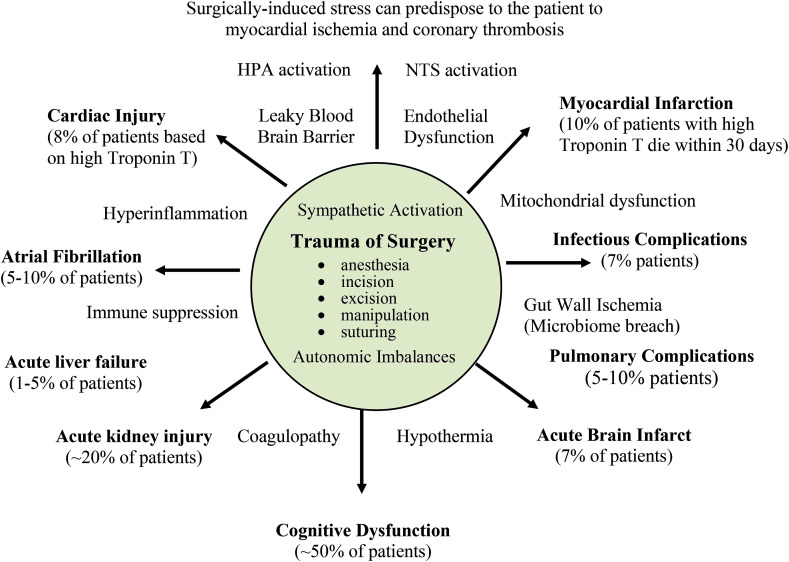
Fig. 1.
Perioperative Complications after Non-Cardiac Major Surgery
Major surgery and anesthesia are associated with stress-induced activation of the sympathetic nervous system, hemodynamic compromise, hyperinflammation, coagulopathy, immune dysfunction, metabolic imbalances and hypothermia. Large population studies indicate that ∼8% of adult patients will suffer heart ischemia/injury, and 10% of these will die within 30 days [118,119]. Surgical stress also leads to perioperative complications involving brain [120,121], kidney [122], lung [123], liver [124], and possibly the gut microbiome [97]. Atrial fibrillation [125] and infections [126] are also significant complications following major surgery. It is estimated that around 50% of these complications are potentially preventable [[13], [14], [15]].
The extent of a patient's stress response is influenced by the type and duration of surgery, and is affected by age, gender, ethnicity, health status, medication profile, number of prior surgeries, and other factors [23,[40], [41], [42]]. Open surgical procedures generally produce a greater stress response than less invasive laparoscopic or robotic interventions [21,39,43]. Non-cardiac major surgical procedures include a median sternotomy, thoracotomy, laparotomy, abdominal hysterectomy and orthopedic hip or knee replacement and open bone fractures [44]. Importantly, the trauma of surgery per se is not the only contributor to secondary injury after major surgery, as it depends upon many factors such as intraoperative bleeding, coagulopathy, cardiac compromise, metabolic dysregulation and organ dysfunction, which appear to be closely intertwined with the patient's physiological reserve and stress response.
3. The questions few people are asking
A major goal of any drug therapy for the management of surgical stress is to prevent or restore homeostatic imbalances in the body and avoid uncontrolled, overexpressed and injurious processes. Notwithstanding the clinical importance of high quality epidemiological data, ongoing studies examining perioperative protection using drugs such as: 1) statins, 2) beta-adrenergic blockers, 3) calcium-channel blockers, 4) angiotensin-converting enzyme (ACE) inhibitors, and 5) aspirin [42,[45], [46], [47], [48]] and optimising fluid therapies [49], it appears there has been much less focus over the past decade on understanding the underlying physiological reasons for poor surgical outcomes. It is time to revisit history and ask two outstanding questions:
-
•
How does the body respond to the trauma of surgery?
-
•
How can surgical stress be reduced?
After presenting a brief history of the CNS-controlled stress-response, we break the system into its component parts, and suggest possible ways to blunt the process before it switches into overdrive and secondary injury progression.
4. Brief history of the stress response
The organism which with the aid of increased adrenal secretion can best muster its energies, can best call forth sugar to supply the labouring muscles, can best lessen fatigue, and can best send blood to the parts essential in the run or the fight for life, is most likely to survive. Such, according to the view here propounded, is the function of the adrenal medulla at times of great emergency.
Walter B. Cannon (1914) [50] p372
Stress research began in the mid-nineteenth century with Pfluger's concept of “steady-state” (1877), Claude Bernard's concept of “milieu intérieur” (1878), and Richet's “stability of the organism” (1900) [42,51]. These physiological concepts of how the body responds to stress were further developed by Walter B. Cannon, Hans Selye and David Cuthbertson. Cannon viewed stress as a challenge to the body's homeostasis or dynamic steady-state where he suggested a role for an activated sympathetic nervous system with adrenal secretions to increase cardiac output and mobilize energy stores, leading to his concept of ‘fight-or-flight’ [52,53]. Seyle similarly viewed stress as a “non-specific strain” on the body that led to irregularities in normal body functions [54]. He also distinguished acute stress from chronic stress, terming the latter ‘general adaptation syndrome’. He identified three major stages of stress as alarm (fight-or-flight), resistance (body's response to recover), and exhaustion (chronic burnout, depression, anxiety) [54]. Selye believed the alarm type of stress was different from emotional arousal or nervous tension, even though the underlying response of nervous tension was similar to a human undergoing surgical anesthesia [55]. Seyle's insight led to the separation of surgical stress from psychogenic stress.
Around the same time, David Cuthbertson was characterizing the complex “stress response” in trauma patients [56]. He divided the body's stress response into two quantifiable events: 1) An early “ebb” phase which began 2 h post-injury and lasted 2–3 days, and was associated with glucose intolerance and a downregulation of cardiac function, tissue perfusion and metabolic rate, and 2) a second “flow” phase lasting days and weeks, which was characterized by an upregulation of metabolism, cardiac function, higher blood glucose, and muscle wasting [56,57]. The studies of Cannon, Seyle and Cuthbertson (and many others) were ground-breaking and instrumental to the discovery in the early 1950s of the neuroendocrine hypothalamic–pituitary-adrenal (HPA) axis, and its multiple functions in maintaining whole body homeostasis [[58], [59], [60]].
A key point to understand about the stress response is that it has evolved over millions of years to increase, not to decrease, survival; it is self-limiting, and restorative [33,42]. However, when an insult or the trauma exceeds the body's internal tolerances, the CNS and downstream effector responses can switch to overdrive and promote secondary injury, slower healing, and in some cases, death.
5. Stress-free surgery
Traumatic impulses are not excluded by ether anaesthesia from that part of the brain that is apparently asleep.
George W. Crile (1913) [61] p7
Before physiologists were studying the stress response in a more systematic manner, neurosurgeon George Crile, in the early 1900s, was already underway with his own clinical research. He was studying whether or not “anesthetised” cells were influenced by surgical stress in his patients. To this end, Crile was continually modifying his operative technique to improve recovery, including lightly anesthetizing the patient with mask inhalation of nitrous oxide and oxygen, and infiltrating all tissues with a dilute local anesthetic procaine before the first incision [62,63]. Crile recognized that general anesthesia was insufficient to reduce the patient's stress response, and so he proposed the word anoci-association (harmful stimuli) to describe the ‘stressors’ of surgery. Crile called his surgical strategy “stress-free anesthesia” [61]. Harvey Cushing extended Crile's ideas to improve postoperative recovery by the use of regional blocks before removing ether anesthesia [64]. Cushing also confirmed Crile's observation that surgical shock could be prevented by the careful monitoring of blood pressure and avoiding the anoci ‘stressors’ associated with the trauma of surgery [64,65]. Today, these stressors include damage-associated molecular patterns (DAMPS) and activation of multiple afferent pathways from peripheral and visceral injured sites to higher centers [42]. These century-old ideas and practices of Crile and Cushing form the basis of modern-day anesthesiology. Unfortunately, Crile's ideas on surgical stress appear to have been lost to the archives, and it is refreshing and energizing to re-read his century-old Lancet paper [61].
6. Activation of the HPA axis and brainstem in response to tissue injury
Since the seminal work of Ludwig and his colleagues, scientists have strived to identify the brain regions that are critical for steady-state and reflex control of the cardiovascular system in health and disease.
S.M. Barman (2020) [66] pR194
Surgical stress is associated with activation of both the central HPA axis and the brainstem's nucleus tractus solitarius (NTS) [66,67]. These central systems regulate cardiovascular, endocrine, thermoregulatory, inflammatory, immune, endothelial and metabolic functions [60,66,68]. Stress-induced activation of paraventricular nucleus (PVN) in the hypothalamus produces corticotrophin-releasing hormone (CRH) and vasopressin (VPR), which stimulates the release of ACTH from the pituitary into the circulation, and activates the adrenal cortex to synthesize and release glucocorticoids (e.g. cortisol). Cortisol exerts its effects via glucocorticoid receptors that are located on most organs of the body, including the brain itself as part of negative feedback circuits. At the cellular level, cortisol alters cellular signalling and the transcription of a wide variety of genes involved in inflammation, immune function, mitochondrial metabolism, adipose tissue and cognition. Cortisol also modulates cardiac contractile function, and sensitizes and potentiates the effects of catecholamines, and other vasoactivators, on vascular smooth muscle cells by suppressing the production of vasodilators, such as nitric oxide, through glucocorticoid alpha- and beta-receptor activation [69]. As mentioned, high plasma cortisol levels also exert negative feedback inhibition on PVN neurons to reduce its own production as a self-protective mechanism.
After surgical stress, an overactive HPA axis can contribute to excess glucocorticoid levels, which can result in pathological states, including cardiac dysfunction [68,69]. During surgical trauma, plasma inflammatory cytokines IL-1, IL-6 and TNF-α stimulate the HPA axis leading to the release of cortisol and catecholamines and systematization of the stress response [22,70]. In 2018, Prete and colleagues reviewed 71 studies and reported increased serum cortisol in 2953 surgical patients [71], with levels correlating with surgery complexity and anesthetic variables. Prete further emphasized that 95% of serum cortisol binds to cortisol-binding globulin, and that this binding decreases with the complexity of surgery [71]. In addition to HPA-axis neuroendocrine control of whole-body homeostasis, PVN neurones form part of neuronal pathways and sympathetic flow to the heart, kidney and other autonomic end organs [72].
The second control system interacting with the HPA axis is the NTS via neurons that innervate the PVN [68]. These include noradrenergic (as well as adrenergic) neurons that send direct projections from the NTS to CRH neurons, and control HPA axis responses to acute systemic (but not psychogenic) stressors, and parasympathetic systems [66]. The NTS is also rich in glucocorticoid receptors which are responsive to an overexpression of cortisol during stress. In addition to interacting with the HPA axis, the NTS network dominates cardiovascular control by continually receiving afferent signals from arterial baroreceptors, which control sympathetic-parasympathetic outflows of different frequencies to the heart and organs of body. The NTS, like the HPA axis, regulates blood pressure, cardio-respiratory function, inflammation, immune function and metabolism [42,[72], [73], [74], [75]]. Surgical stress is associated with a shift towards a higher sympathetic (‘fight or flight’) to parasympathetic (‘rest and digest’) flow ratio, and associated circuits [42,76]. Sympatho-vagal imbalances can lead to cardiac pump dysfunction and arrhythmias, immune dysfunction, hyperinflammation and organ dysfunction. Some surgical patients may already have comorbidities associated with increased sympathetic outflow and impaired parasympathetic responsiveness [77,78] making them particularly vulnerable to surgical stress. The discharges of sympathetic nerves with cardiovascular targets contain a mixture of rhythmic components with frequencies ranging from ∼0.05 Hz to 10 Hz [66,79]. Gebber and colleagues proposed that the 10-Hz rhythm in sympathetic nerve discharge served as a “carrier” frequency for complex cardiovascular response patterns [80], and possible changes in this frequency deserves further investigation after surgical stress.
7. A working hypothesis to reduce surgical stress
It should be remembered always that the patient who has been in shock and resuscitated, and then operated upon, is in a precarious state. His nervous system has been disturbed not only by the original trauma, but also by the low nutrient flow of blood, and by the surgical procedures incidental to operation
Walter B. Cannon (1923) Quoted from Traumatic Shock [81] p192
In formulating a working hypothesis to reduce surgical morbidity and mortality, Cannon's insight into traumatic injury being a system's perturbation beginning with the CNS cannot be overstated [29,42]. New ways are required to prevent the CNS from entering into ‘overdrive’ after receiving damage signals from the surgical incision, excision, cauterizing, manipulation, suturing and anesthesia. If an intravenous (IV) therapy can commence before the first incision, it may lead to reduced sympathetic discharge and perioperative complications. Targeting areas of the sympathetically-activated HPA axis and NTS in the patient's anesthetized brain that is still physiologically ‘awake’ may improve cardiac function, arterial compliance, endothelial function, tissue perfusion and energy production. Shifting the autonomic balance by inhibiting sympathetic and upregulating parasympathetic outflows is a key component to the hypothesis because activation of parasympathetic pathways exerts anti-inflammatory actions [78] and reduces the potential for ‘downstream’ secondary injury progression. We have proposed how this might work in a Systems Hypothesis of Trauma (SHOT) which has three pillars of protection [29] and apply it to surgical stress (Fig. 2 ).
-
1.
CNS-Cardiovascular Coupling (Central Controller)
-
2.
Endothelial Glycocalyx Health (Systems Integrator)
-
3.
Mitochondrial Integrity (Energy Production)
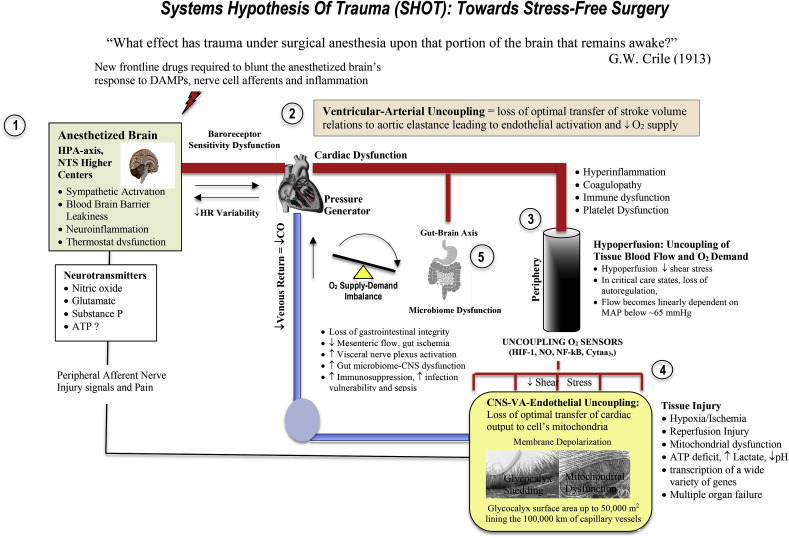
Fig. 2.
A Systems-Based Approach to the Trauma of Surgery
Surgical stress triggers a myriad of neuroendocrine, inflammatory, metabolic, and transcriptional perturbations that predispose the patient to further injury. The early drivers of surgical stress are sterile local injury, firing of injured peripheral and visceral afferent nerves which activate the brain's emergency response (see text). During and after major surgery, this system can switch to overdrive and exceed the body's normal tolerances of protection. This can lead to secondary injury, slower healing, poor outcomes, and in some cases, death. From a systems-based perspective surgical stress is levelled at: 1) the CNS, 2) left ventricular-arterial coupling, 3) microvascular blood flow distribution 4) the vascular endothelium/glycocalyx, and 5) the gut microbiome. A hypoperfused vascular endothelium can lead to glycocalyx shedding, systemic hyperinflammation, coagulopathy, platelet dysfunction, immune dysfunction, loss of vascular tone, reduced O2 supply and mitochondrial dysfunction. The hypothesis is that if central and local control of cardiac output and ventricular-arterial coupling can be maintained, the endothelium will not activate and tissue O2 delivery will not be compromised. A potential target therefore is to prevent the “awake’ anesthetised brain from switching into overdrive. This may be achieved by shifting the CNS's autonomic balance from a sympathetically-driven system to a more parasympathetic anti-inflammatory one and improve perioperative outcomes. Modified after Dobson [34].
If imbalances can be prevented, surgical complications may be reduced (Fig. 2). The reason for targeting the CNS first is because it is the central controller of all downstream pathways that are activated after the first incision. Current practice of targeting single-nodal downstream changes after major surgery in a treat-as-you-go approach leads to what US surgeon William C. Shoemaker considered: “an uncoordinated and sometimes contradictory therapeutic outcome” [36]. This may be one reason why few advances have been made in reducing surgical mortality and morbidity, and a by-product of decades of highly reductionist thinking and research. Reductionism is important to break a system into its constituent parts but it does not do away with the system. New therapies may be more successful if they target upstream controllers of the stress response and target the system.
Loss of CNS-Cardiovascular Coupling
Cardiovascular complications are the leading cause of death within 30 days after non-cardiac surgery.
Devereaux and Sessler (2015) [82] p2267
Suppressing CNS neurohormonal outflows before surgical stress is key to protection of cardiovascular function and secondary injury progression. A parameter that has received little attention is ventricular-arterial (VA) coupling, which links the CNS to cardiac output, endothelial health and mitochondrial energy production. VA coupling is defined as the ratio of left-ventricular elastance to arterial elastance, and can be derived from routine echocardiography [[83], [84], [85], [86], [87], [88]]. It is a measure of left ventricular systolic performance to pump blood into the arterial tree, and the ability of arterial conduits to accept that blood. In critical care states [89], it is consistently reported to be a reliable and effective measure of cardiovascular performance in maintaining tissue oxygen supply [86,90,91]. When the ratio is close to unity, the efficiency of the system is considered optimal. If the ratio is excessively high or low, the heart as a pump and vascular load become uncoupled with adverse downstream clinical outcomes [86,89,92].
While there are few or no studies measuring VA coupling ratios in non-cardiac surgery patients, an altered ratio would be expected to reduce blood flows to the major organs and promote hyperinflammation, coagulopathy and immune dysfunction [39,42]. Infectious complications may also arise from reduced blood flow to the gut wall and leakiness with bacteria, or their active metabolic products (lipopolysaccharides, cytokines, neuropeptides, and protein messengers to passing across the ischemic wall into the circulation, and further exacerbating inflammation, coagulopathy, immunosuppression and limiting tissue O2 supply [93]. After major surgery, the patient's gut microbiome has been reported to change in composition, and is linked to the CNS dysfunction through vagal afferents and HPA axis modulation [77,[93], [94], [95], [96]], and other complications [97]. This is further exacerbated by the use of postoperative antibiotics. More studies are required on VA coupling in patients experiencing secondary complications after major surgery.
Endothelial glycocalyx shedding
There is increasing recognition that targeting the endothelium and maximizing inherent physiological function in the perioperative period may improve postoperative outcomes.
Riedel and colleagues (2013) [98]p151
The endothelial-glycocalyx forms a nexus between the blood circulation and tissues and covers an enormous surface area (up to 50,000 m2). By maintaining different cell morphologies over this vast surface area the endothelial-glycocalyx plays a key integrator role in coupling CNS-Cardiovascular function to oxygen transport and energy production [29]. Impaired VA coupling can lead to hypoperfusion and activate the endothelium. An activated endothelium can lead to shedding of its luminal ‘fuzzy layer’, the glycocalyx, leading to widespread inflammation, coagulopathy, platelet dysfunction, altered immune trafficking and loss of vascular tone [99,100].
Endothelial dysfunction and glycocalyx shedding is triggered by tissue trauma, hypoperfusion, ischemia-reperfusion injury, increased shear-stress, hypovolemia, hyperglycemia, oxidative stress, hyperinflammation and coagulopathy [101,102] (Fig. 2). In the perioperative setting, this can lead to further myocardial injury and potentially increase 30-day mortality [102] (see Fig. 1). Release of high levels of syndecan-1, hyaluronan, and heparan sulphate levels, and other endothelial injury markers (thrombomodulin, annexin-II, von Willebrand factor), are also correlated with poor outcomes [103]. Glycocalyx shedding, however, is reversible in a time frame that is dependent upon the type and severity of the operation and surgical trauma [[104], [105], [106]]. Considering its strategic location and integrating functions, drug therapies that protect the endothelial-glycocalyx may improve clinical outcomes following major surgery. Although currently there are no specific perioperative treatments to protect the endothelium, a number of promising strategies are being developed using different drugs [107] and liposomal nanocarriers [103].
Loss of mitochondrial function
Mitochondrial abnormalities and oxidant stress strongly suggest that mitochondrial ROS production may be a major determinant of the lymphocyte apoptosis and immune suppression that follow surgery and general anesthesia.
Delogu and colleagues (2001) [35].
Surgical stress is triggered from cell injury to the CNS and then back again. Surgical-induced perioperative ischemia can lead to mitochondrial abnormalities and oxidant stress in all peripheral tissues, immune cells and platelets [35]. The most susceptible tissues to ischemia-reperfusion injury are the more aerobic tissues such as brain, heart, lung and kidney. Impaired myocardial mitochondrial function, apoptosis and integrity are particularly challenging because they can lead to contractile dysfunction, ventricular arrhythmias and possibly death [108]. Mitochondria are believed of bacterial origin and located at the terminal step of oxygen transfer from the lung to cell metabolism [37,38]. They are the cell's oxygen consumers and ATP generators [29]. Mitochondrial dysfunction occurs after periods of hypoperfusion, endothelial activation and tissue hypoxia/ischemia, which is accompanied by a shift in redox state (cytosolic NAD+/NADH ratio), increased lactate production, decreased proton pumping across the inner membrane, collapse of membrane potential, opening of the permeability transition pore, Ca2+ loading, loss of cytochrome C, and release of apoptotic-cascade inducing factors [29,38]. Tissue mitochondrial fragments also contribute to surgical stress by releasing DAMPS which can activate the immune system, worsen coagulopathy and cause further tissue damage via proteolytic degradation and oxidative stress [109]. Mitochondrial DAMPS can also be released by activated platelets, which can act as powerful pro-inflammatory agents and contribute to secondary injury progression [110]. Collectively, mitochondrial dysfunction is associated with HPA axis overdrive, coagulopathy, inflammation, immunosuppression, platelet dysfunction, organ dysfunction and, if unchecked, multiple organ failure [111].
8. Future research and a possible path forward
In current practice, little emphasis is placed on the amelioration of stress-induced changes in endocrine/metabolic/cardiovascular functions–mainly because measures to counteract the response (e.g. insulin treatment of stress induced hyperglycaemia, perioperative beta-blockade) have spectacularly failed in reducing surgical morbidity or mortality.
P.G. Berthelsen (2015) [112] p247
Currently, no drug therapy exists that targets the CNS-cardiovascular-endothelial-mitochondrial system to reduce surgical stress. While varying benefits have been reported from the use of statins, beta-adrenergic blockers, calcium-channel blockers, angiotensin-converting enzyme (ACE) inhibitors, or aspirin [42,[45], [46], [47], [48]], the high rates of postoperative complications continue to increase globally as the patient population ages and present with more complex comorbidities. A path forward may identifying drug therapies that support a CNS-controlled high flow, hypotensive, vasodilatory state that maintains endothelial patency and tissue O2 supply. We have been working on such a drug (adenosine, lidocaine and Mg2+, ALM) in severe trauma states, and preclinical studies show some promise in protecting the CNS, decreasing the sympathetic/parasympathetic outlows, maintaining a VA coupling ratio close to unity, correcting early coagulopathy, suppressing inflammation and immune dysfunction, and lowering of energy demand [51,92,[113], [114], [115]]. Studies carried out by US Army Institute of Surgical Research have also shown that ALM therapy protects against endothelial glycocalyx shedding with 97% rapid restoration after hemorrhagic shock [107]. We also have shown that the ALM therapy in a rat surgical laparotomy model reduced proinflammatory IL-6, increased anti-inflammatory IL-10, lowered serum lactate and decreased the release of neutrophils [116]. Despite showing some promise for surgical stress, we appreciate the long road ahead given the high failure rate of translating new drugs into humans (>95%) [51], and of those having received FDA approval, ∼30% have displayed post-market adverse events [117].
12. Concluding remarks
There are many unanswered questions on the best management strategies and drug therapies to prevent and manage perioperative complications after major surgery. A key starting point is to recognize that the anesthetized brain is still physiologically ‘awake’ and responsive to the sterile injury stressors of surgery. A systems-based approach to restore sympathetic/parasympathetic balance and targeting CNS-Mitochondrial coupling may help drive development of novel therapeutics to reduce secondary injury progression and improve outcomes.
Provenance and peer review
Not commissioned, externally peer-reviewed.
Ethical approval
This is a Perspective and Not Applicable.
Sources of funding
This work was supported by USSOCOM, IACUC protocol A2118 and A2296, USAMRMC proposal SO150053 under Award No. W81XWH-USSOCOM-BAA-15-1. The opinions, interpretations, conclusions are those of the author and are not necessarily endorsed by the US Department of Defense.
Author contribution
The author is the sole contributor to the design, implementation, literature analysis and writing of the manuscript.
Guarantor
The author accepts full responsibility for the work.
CRediT authorship contribution statement
Geoffrey P. Dobson: Writing - original draft.
Declaration of competing interest
Geoffrey Dobson is the sole inventor of the ALM concept for cardioplegia, organ preservation, surgery, infection and trauma, and has no financial interests from any entity or organization.
Acknowledgements
The author would like to thank the JCU research team Drs Hayley Letson, Jodie Morris and Erik Biros for many discussions on the topic of surgical trauma. I would particularly like to thank the US Department of Defense for their continued support of our work, and the College of Medicine and Dentistry at James Cook University.
References
- 1.Weiser T.G., Regenbogen S.E., Thompson K.D., Haynes A.B., Lipsitz S.R., Berry W.R., Gawande A.A. An estimation of the global volume of surgery: a modelling strategy based on available data. Lancet. 2008;372(9633):139–144. doi: 10.1016/S0140-6736(08)60878-8. [DOI] [PubMed] [Google Scholar]
- 2.Weiser T.G., Haynes A.B., Molina G., Lipsitz S.R., Esquivel M.M., Uribe-Leitz T., Fu R., Azad T., Chao T.E., Berry W.R., et al. Estimate of the global volume of surgery in 2012: an assessment supporting improved health outcomes. Lancet. 2015:385. doi: 10.1016/S0140-6736(15)60806-6. [DOI] [PubMed] [Google Scholar]
- 3.Maggio P.M., Taheri P.A. Perioperative issues: myocardial ischemia and protection--beta-blockade. Surg. Clin. 2005;85(6):1091–1102. doi: 10.1016/j.suc.2005.09.016. [DOI] [PubMed] [Google Scholar]
- 4.Patel A.Y., Eagle K.A. Cardiac risk of noncardiac surgery. J. Am. Coll. Cardiol. 2015;66(19):2140–2148. doi: 10.1016/j.jacc.2015.09.026. [DOI] [PubMed] [Google Scholar]
- 5.Kristensen S.D., Knuuti J., Saraste A., Anker S., Bøtker H.E., Hert S.D., Ford I., Gonzalez-Juanatey J.R., Gorenek B., Heyndrickx G.R., et al. 2014 ESC/ESA Guidelines on non-cardiac surgery: cardiovascular assessment and management. Eur. Heart J. 2014;35:2383–2431. doi: 10.1093/eurheartj/ehu282. Sept 14 (35) [DOI] [PubMed] [Google Scholar]
- 6.Devereaux P.J., Chan M.T., Alonso-Coello P. Association between postoperative troponin levels and 30-day mortality among patients undergoing noncardiac surgery. J. Am. Med. Assoc. 2012;307(21):2295–2304. doi: 10.1001/jama.2012.5502. al. e. [DOI] [PubMed] [Google Scholar]
- 7.Pearse R.M., Moreno R.P., Bauer P., Pelosi P., Metnitz P., Spies C., Vallet B., Vincent J.L., Hoeft A., Rhodes A. Mortality after surgery in Europe: a 7 day cohort study. Lancet. 2012;380(9847):1059–1065. doi: 10.1016/S0140-6736(12)61148-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Semel M.E., Lipsitz S.R., Funk L.M., Bader A.M., Weiser T.G., Gawande A.A. Rates and patterns of death after surgery in the United States, 1996 and 2006. Surgery. 2012;151(2):171–182. doi: 10.1016/j.surg.2011.07.021. [DOI] [PubMed] [Google Scholar]
- 9.Xu J., Murphy S.L., Kochanek K.D., Arias E. Mortality in the United States, 2018. NCHS Data Brief. 2020;355:1–8. [PubMed] [Google Scholar]
- 10.Eyob B., Boeck M.A., FaSiOen P., Cawich S., Kluger M.D. Ensuring safe surgical care across resource settings via surgical outcomes data & quality improvement initiatives. Int. J. Surg. 2019;72S:27–32. doi: 10.1016/j.ijsu.2019.07.036. [DOI] [PubMed] [Google Scholar]
- 11.Roth G.A., Abate D., Abate K.H., Abay S.M., Abbafati C., Abbasi N., Abbastabar H., Abd-Allah F., Abdela J., Abdelalim A., et al. Global, regional, and national age-sex-specific mortality for 282 causes of death in 195 countries and territories, 1980–2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet. 2018;392(10159):1736–1788. doi: 10.1016/S0140-6736(18)32203-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Farmer P.E., Kim J.Y. Surgery and global health: a view from beyond the OR. World J. Surg. 2008;32:533–536. doi: 10.1007/s00268-008-9525-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Anderson O., Davis R., Hanna G.B., Vincent C.A. Surgical adverse events: a systematic review. Am. J. Surg. 2013;206(2):253–262. doi: 10.1016/j.amjsurg.2012.11.009. [DOI] [PubMed] [Google Scholar]
- 14.Healey M.A., Shackford S.R., Osler T.M., Rogers F.B., Burns E. Complications in surgical patients. Arch. Surg. 2002;137(5):611–618. doi: 10.1001/archsurg.137.5.611. [DOI] [PubMed] [Google Scholar]
- 15.Neale G., Woloshynowych M., Vincent C. Exploring the causes of adverse events in NHS hospital practice. J. R. Soc. Med. 2001;94:322–330. doi: 10.1177/014107680109400702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Leape L.L. Hospital readmissions following surgery. Turning complications into 'treasures'. J. Am. Med. Assoc. 2015;313(5):467–468. doi: 10.1001/jama.2014.18666. [DOI] [PubMed] [Google Scholar]
- 17.Dimick J.B., Ghaferi A.A. Hospital readmission as a quality measure in surgery. J. Am. Med. Assoc. 2015;313(5):512–513. doi: 10.1001/jama.2014.14179. [DOI] [PubMed] [Google Scholar]
- 18.Biccard B.M., Madiba T.E., Kluyts H.-L., Munlemvo D.M., Madzimbamuto F.D., Basenero A., Gordon C.S., Youssouf C., Rakotoarison S.R., Gobin V., et al. Perioperative patient outcomes in the African Surgical Outcomes Study: a 7-day prospective observational cohort study. Lancet. 2018;391(10130):1589–1598. doi: 10.1016/S0140-6736(18)30001-1. [DOI] [PubMed] [Google Scholar]
- 19.Moore F.D. Bodily changes in surgical convelescence. 1.The normal sequence-observations and interpretations. Ann. Surg. 1953;137(3):289–315. doi: 10.1097/00000658-195303000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Hall G.M. The anesthetic modification of the endocrine and metabolic response to surgery. Ann. R. Coll. Surg. Engl. 1985;67:25–29. [PMC free article] [PubMed] [Google Scholar]
- 21.Desborough J.P. The stress response to trauma and surgery. Br. J. Anaesth. 2000;85(1):109–117. doi: 10.1093/bja/85.1.109. [DOI] [PubMed] [Google Scholar]
- 22.Burton D., Nicholson G., Hall G. Endocrine and metabolic response to surgery. Continuing Ed Anesthesia Crit Care Pain. 2004;4(5):144–147. [Google Scholar]
- 23.Giannoudis P.V., Dinopoulos H., Chalidis B., Hall G.M. Surgical stress response. Injury. 2006;37(Suppl 5):S3–S9. doi: 10.1016/S0020-1383(07)70005-0. [DOI] [PubMed] [Google Scholar]
- 24.Menger M.D., Vollmar B. Surgical trauma: hyperinflammation versus immunosuppression? Langenbeck's Arch. Surg. 2004;389(6):475–484. doi: 10.1007/s00423-004-0472-0. [DOI] [PubMed] [Google Scholar]
- 25.Kohl B.A., Deutschman C.S. The inflammatory response to surgery and trauma. Curr. Opin. Crit. Care. 2006;12(4):325–332. doi: 10.1097/01.ccx.0000235210.85073.fc. [DOI] [PubMed] [Google Scholar]
- 26.Lin E., Calvano S.E., Lowry S.F. Inflammatory cytokines and cell response in surgery. Surgery. 2000;127(2):117–126. doi: 10.1067/msy.2000.101584. [DOI] [PubMed] [Google Scholar]
- 27.Lin E., Gletsu-Miller N. Surgical stress induces an amplified inflammatory response in patients with type 2 diabetes. ISRN Obesity. 2013;2013:1–5. doi: 10.1155/2013/910586. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Levy J.H., Goodnough L.T. How I use fibrinogen replacement therapy in acquired bleeding. Blood. 2015;125(9):1387–1393. doi: 10.1182/blood-2014-08-552000. [DOI] [PubMed] [Google Scholar]
- 29.Dobson G.P., Morris J.L., Davenport L.M., Letson H.L. Traumatic-induced coagulopathy as a systems failure: a new window into hemostasis. Semin. Thromb. Hemost. 2020;46(2):199–214. doi: 10.1055/s-0039-1701018. [DOI] [PubMed] [Google Scholar]
- 30.Curry N., Brohi K. Surgery in traumatic injury and perioperative considerations. Semin. Thromb. Hemost. 2020;46:73–82. doi: 10.1055/s-0039-1697932. [DOI] [PubMed] [Google Scholar]
- 31.Marik P.E., Flemmer M. The immune response to surgery and trauma: implications for treatment. J Trauma Acute Care Surg. 2012;73(4):801–808. doi: 10.1097/TA.0b013e318265cf87. [DOI] [PubMed] [Google Scholar]
- 32.Elenkov I.J., Chrousos G.P. Stress system – organization, physiology and immunoregulation. Neuroimmunomodulation. 2006;13:257–267. doi: 10.1159/000104853. [DOI] [PubMed] [Google Scholar]
- 33.Dragoş D., Tănăsescu M.D. The effect of stress on the defense systems. J Med Life. 2010;3(1):10–18. [PMC free article] [PubMed] [Google Scholar]
- 34.Anderson D.J., Chen L.F., Sexton D.J., Kaye K.S. Complex surgical site infections and the devilish details of risk adjustment: important implications for public reporting. Infect. Control Hosp. Epidemiol. 2008;29(10):941–946. doi: 10.1086/591457. [DOI] [PubMed] [Google Scholar]
- 35.Delogu G., Moretti S., Famularo G., Santini G., Antonucci A., Marandola M., Signore L. Mitochondrial perturbations and oxidant stress in lymphocytes from patients undergoing surgery and general anesthesia. Arch. Surg. 2001;136(10):1190–1196. doi: 10.1001/archsurg.136.10.1190. [DOI] [PubMed] [Google Scholar]
- 36.Shoemaker W.C., Beez M. Pathophysiology, monitoring, and therapy of shock with organ failure. Appl. Cardiopulm. Pathophysiol. 2010;14:5–15. [Google Scholar]
- 37.Picard M., McManus M.J., Gray J.D., Nasca C., Moffat C., Kopinski P.K., Seifert E.L., McEwen B.S., Wallace D.C. Mitochondrial functions modulate neuroendocrine, metabolic, inflammatory, and transcriptional responses to acute psychological stress. Proc. Natl. Acad. Sci. U. S. A. 2015;112(48):E6614–E6623. doi: 10.1073/pnas.1515733112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Thurairajah K., Briggs G.D., Balogh Z.J. The source of cell-free mitochondrial DNA in trauma and potential therapeutic strategies. Eur. J. Trauma Emerg. Surg. 2018;44(3):325–334. doi: 10.1007/s00068-018-0954-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Choileain N.N., Redmond H.P. Cell response to surgery. Arch. Surg. 2006;141(11) doi: 10.1001/archsurg.141.11.1132. [DOI] [PubMed] [Google Scholar]
- 40.Li L., Messina J.L. Acute insulin resistance following surgery. Trends Endocrinol. Metabol. 2009;20(9):429–435. doi: 10.1016/j.tem.2009.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Abdelmalak B.B., Bonilla A.M., Yang D., Chowdary H.T., Gottlieb A., Lyden S.P., Sessler D.I. The hyperglycemic response to major noncardiac surgery and the added effect of steroid administration in patients with and without diabetes. Anesth. Analg. 2013;116(5):1116–1122. doi: 10.1213/ANE.0b013e318288416d. [DOI] [PubMed] [Google Scholar]
- 42.Dobson G.P. Addressing the global burden of trauma in major surgery. Front Surg. 2015;2(Sept):43. doi: 10.3389/fsurg.2015.00043. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ueda A., Turner P.M.G. Stress response to laparoscopic liver resection. HPB (Oxford) 2004;6(4):247–252. doi: 10.1080/13651820410023987. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Oka Y., Murata A., Nishijima J., Yasuda T., Hiraoka N., Ohmachi Y., Kitagawa K., Yasuda T., Toda H., Tanaka N., et al. Circulating interleukin 6 as a useful marker for predicting postoperative complications. Cytokine. 1992;4(4):298–304. doi: 10.1016/1043-4666(92)90070-8. [DOI] [PubMed] [Google Scholar]
- 45.Weinberg R.L., Eagle K.A. Perioperative statin use in noncardiac surgery: who and when? JAMA Intern Med. 2017;177(2):242–243. doi: 10.1001/jamainternmed.2016.8037. [DOI] [PubMed] [Google Scholar]
- 46.Blessberger H., Kammler J., Steinwender C. Perioperative use of β-blockers in cardiac and noncardiac surgery. J. Am. Med. Assoc. 2015;313(20):2070–2071. doi: 10.1001/jama.2015.1883. [DOI] [PubMed] [Google Scholar]
- 47.Hollmann C., Fernandes N.L., Biccard B.M. A systematic review of outcomes associated with withholding or continuing angiotensin-converting enzyme inhibitors and angiotensin receptor blockers before noncardiac surgery. Anesth. Analg. 2018;127(3):678–687. doi: 10.1213/ANE.0000000000002837. [DOI] [PubMed] [Google Scholar]
- 48.Devereaux P.J., Mrkobrada M., Sessler D.I., Leslie K., Alonso-Coello P., Kurz A., Villar J.C., Sigamani A., Biccard B.M., Meyhoff C.S., et al. Aspirin in patients undergoing noncardiac surgery. N. Engl. J. Med. 2014;370(16):1494–1503. doi: 10.1056/NEJMoa1401105. [DOI] [PubMed] [Google Scholar]
- 49.Miller T.E., Myles P.S. Perioperative fluid therapy for major surgery. Anesthesiology. 2019;130(5):825–832. doi: 10.1097/ALN.0000000000002603. [DOI] [PubMed] [Google Scholar]
- 50.Cannon W.B. The emergency function of the adrenal medulla in pain and the major emotions. Am. J. Physiol. 1914;33(3):356–372. [Google Scholar]
- 51.Dobson G.P., Letson H.L. Adenosine, lidocaine and Mg2+ (ALM): from cardiac surgery to combat casualty care: teaching old drugs new tricks. J Trauma and Acute Care Surgery. 2016;80(1):135–145. doi: 10.1097/TA.0000000000000881. [DOI] [PubMed] [Google Scholar]
- 52.Cannon W.B. Organisation for physiological homeostasis. Physiol. Rev. 1929;9(3):399–431. [Google Scholar]
- 53.Cannon W.B. W.W. Norton; 1932. The Wisdom of the Body. [Google Scholar]
- 54.Selye H. The evolution of the stress concept. Am. Sci. 1973;61(6):692–699. [PubMed] [Google Scholar]
- 55.Fink G. Eighty years of stress. Nature. 2016;539:175–176. doi: 10.1038/nature20473. [DOI] [PubMed] [Google Scholar]
- 56.Cuthbertson D.P. Symposium on 'surgery and nutrition'. Historical approach. Proc. Nutr. Soc. 1980;39(2):101–105. doi: 10.1079/pns19800017. [DOI] [PubMed] [Google Scholar]
- 57.Goldstein D.S., Kopin I.J. Evolution of concepts of stress. Stress. 2007;10:109–120. doi: 10.1080/10253890701288935. [DOI] [PubMed] [Google Scholar]
- 58.Harris G.W. The hypothalamus and endocrine glands. Br. Med. Bull. 1950;6:345–350. doi: 10.1093/oxfordjournals.bmb.a073628. [DOI] [PubMed] [Google Scholar]
- 59.O'Connor T.M., O'Halloran D.J., Shanahan F. The stress response and the hypothalamic-pituitary-adrenal axis: from molecule to melancholia. Q. J. Med. 2000;93(6):323–333. doi: 10.1093/qjmed/93.6.323. [DOI] [PubMed] [Google Scholar]
- 60.Smith S.M., Vale W.W. The role of the hypothalamic-pituitary-adrenal axis in neuroendocrine responses to stress. Dialogues Clin. Neurosci. 2006;8(4):383–395. doi: 10.31887/DCNS.2006.8.4/ssmith. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Crile G.W. The kinetic theory of shock and its prevention through anoci-association {shockless operation) Lancet. 1913;185:7–16. [Google Scholar]
- 62.Crile G. Nitrous oxide anaesthesia and a note on anoci-association, a new principle in operative surgery. Surg. Gynecol. Obstet. 1911;13:170–173. [Google Scholar]
- 63.Nathoo N., Lautzenheiser F.K., Barnett G.H. George W. Crile, Ohio's first neurosurgeon, and his relationship with Harvey Cushing. J. Neurosurg. 2005;103(2):378–386. doi: 10.3171/jns.2005.103.2.0378. [DOI] [PubMed] [Google Scholar]
- 64.Cushing H. On the avoidance of shock in major amputations by cocainization of large nerve-trunks preliminary to their division. Ann. Surg. 1902;36:321–345. doi: 10.1097/00000658-190209000-00001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Soto-Ruiz K.M., Varon J. Resuscitation great. George W. Crile: a visionary mind in resuscitation. Resuscitation. 2009;80(1):6–8. doi: 10.1016/j.resuscitation.2008.09.008. [DOI] [PubMed] [Google Scholar]
- 66.Barman S.M. 2019 Ludwig Lecture: rhythms in sympathetic nerve activity are a key to understanding neural control of the cardiovascular system. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2020;318:R191–R205. doi: 10.1152/ajpregu.00298.2019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Charkoudian N., Wallin B.G. Sympathetic neural activity to the cardiovascular system: integrator of systemic physiology and interindividual characteristics. Comp. Physiol. 2014;4:825–850. doi: 10.1002/cphy.c130038. [DOI] [PubMed] [Google Scholar]
- 68.Herman J.P. Regulation of hypothalamo-pituitary-adrenocortical responses to stressors by the nucleus of the solitary tract/dorsal vagal complex. Cell. Mol. Neurobiol. 2018;38:25–35. doi: 10.1007/s10571-017-0543-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Burford N.G., Webster N.A., Cruz-Topete D. Hypothalamic-pituitary-adrenal Axis modulation of glucocorticoids in the cardiovascular system. Int. J. Mol. Sci. 2017;18:2150. doi: 10.3390/ijms18102150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Turnbull A.V., Rivier C. Regulation of the HPA axis by cytokines. Brain Behav. Immun. 1995;9(4):253–275. doi: 10.1006/brbi.1995.1026. [DOI] [PubMed] [Google Scholar]
- 71.Prete A., Yan Q., Al-Tarrah K., Akturk H.K., Prokop L.J., Alahdab F., Foster M.A., Lord J.M., Karavitaki N., Wass J.A., et al. The cortisol stress response induced by surgery: a systematic review and meta-analysis. Clin. Endocrinol. 2018;89(5):554–567. doi: 10.1111/cen.13820. [DOI] [PubMed] [Google Scholar]
- 72.Coote J.H. Landmarks in understanding the central nervous controlof the cardiovascular system. Exp. Physiol. 2007;92(1):3–18. doi: 10.1113/expphysiol.2006.035378. [DOI] [PubMed] [Google Scholar]
- 73.Thayer J.F. Vagal tone and the inflammatory reflex. Cleve. Clin. J. Med. 2009;76(Suppl 2):S23–S26. doi: 10.3949/ccjm.76.s2.05. [DOI] [PubMed] [Google Scholar]
- 74.Tracey K.J. Reflex control of immunity. Nat. Rev. Immunol. 2009;9:418–428. doi: 10.1038/nri2566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Grill H.J., Hayes M.R. The nucleus tractus solitarius: a portal for visceral afferent signal processing, energy status assessment and integration of their combined effects on food intake. Int. J. Obes. 2009;33(Suppl 1):S11–S15. doi: 10.1038/ijo.2009.10. [DOI] [PubMed] [Google Scholar]
- 76.Pongratz G., Straub R.H. The sympathetic nervous response in inflammation. Arthritis Res. Ther. 2014;16:504. doi: 10.1186/s13075-014-0504-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Morris J.L., Letson H.L., Gillman R., Hazratwala K., Wilkinson M., McEwen P., Dobson G.P. The CNS theory of osteoarthritis: opportunities beyond the joint. Semin. Arthritis Rheum. 2019;49:331–336. doi: 10.1016/j.semarthrit.2019.03.008. [DOI] [PubMed] [Google Scholar]
- 78.Olshansky B. Vagus nerve modulation of inflammation: cardiovascular implications. Trends Cardiovasc. Med. 2016;26:1–11. doi: 10.1016/j.tcm.2015.03.016. [DOI] [PubMed] [Google Scholar]
- 79.Coote J.H. The sympathetic innervation of the heart: important new insights. Auton. Neurosci. 2016;199:17–23. doi: 10.1016/j.autneu.2016.08.014. [DOI] [PubMed] [Google Scholar]
- 80.Gebber G.L., Zhong S., Barman S.M. The functional significance of the 10-Hz sympathetic rhythm: a hypothesis. Clin. Exp. Hypertens. 1995;17(1):181–195. doi: 10.3109/10641969509087064. [DOI] [PubMed] [Google Scholar]
- 81.Cannon W.B. D. Appleton and Co.; New York: 1923. Traumatic Shock. [Google Scholar]
- 82.Devereaux P., Sessler D.I. Cardiac complications in patients undergoing major noncardiac surgery. N. Engl. J. Med. 2015;373:2258–2269. doi: 10.1056/NEJMra1502824. [DOI] [PubMed] [Google Scholar]
- 83.Suga H., Goto Y., Kawaguchi O., Hata K., Takasago T., Saeki T.W., Taylor T.W. In: Burkhoff D., Schaefer J., Schaffner K., Yue D.T., editors. vol. 88. Springer-Verlag; New York: 1993. Ventricular perspective on efficiency; pp. 43–65. (Myocardial Optimization and Efficiency, Evolutionary Aspects and Philosophy of Science Considerations). Suppl.2. [Google Scholar]
- 84.London G.M. Role of arterial wall properties in the pathogenesis of systolic hypertension. Am. J. Hypertens. 2005;18(1 Pt 2):19S–22S. doi: 10.1016/j.amjhyper.2004.10.001. [DOI] [PubMed] [Google Scholar]
- 85.Kass D.A. Ventricular arterial stiffening: integrating the pathophysiology. Hypertension. 2005;46(1):185–193. doi: 10.1161/01.HYP.0000168053.34306.d4. [DOI] [PubMed] [Google Scholar]
- 86.Guarracino F., Baldassarri R., Pinsky M.R. Ventriculo-arterial decoupling in acutely altered hemodynamic states. Crit. Care. 2013;17(2):213–220. doi: 10.1186/cc12522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Cholley B., Le Gall A. Ventriculo-arterial coupling: the comeback? J. Thorac. Dis. 2016;8(9):2287–2289. doi: 10.21037/jtd.2016.08.34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Dobson G.P., Arsyad A., Letson H.L. The adenosine hypothesis revisited: a possible role for arterial compliance and its implications to coronary perfusion. Front. Physiol. 2017;8(October) doi: 10.3389/fphys.2017.00824. Article 824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Onorati F., Santini F., Dandale R., Ucci G., Pechlivanidis K., Menon T., Chiominto B., Mazzucco A., Faggian G. Polarizing" microplegia improves cardiac cycle efficiency after CABG for unstable angina. Int. J. Cardiol. 2013;167(6):2739–2746. doi: 10.1016/j.ijcard.2012.06.099. [DOI] [PubMed] [Google Scholar]
- 90.Ky B., French B., May Khan A., Plappert T., Wang A., Chirinos J.A., Fang J.C., Sweitzer N.K., Borlaug B.A., Kass D.A., et al. Ventricular-arterial coupling, remodeling, and prognosis in chronic heart failure. J. Am. Coll. Cardiol. 2013;62(13):1165–1172. doi: 10.1016/j.jacc.2013.03.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Axell R.G., Messer S.J., White P.A., McCabe C., Priest A., Statopoulou T., Drozdzynska M., Viscasillas J., Hinchy E.C., Hampton-Till J., et al. Ventriculo-arterial coupling detects occult RV dysfunction in chronic thromboembolic pulmonary vascular disease. Phys. Rep. 2017;5(7) doi: 10.14814/phy2.13227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Granfeldt A., Letson H.L., Hyldebrandt J.A., Wang E.R., Salcedo P.A., Nielson T.K., Tonnesen E., Vinten-Johansen J., Dobson G.P. Small-Volume 7.5% NaCl adenosine, lidocaine and Mg2+ has multiple benefits during hypotensive and blood resuscitation in the pig following severe blood loss: rat to Pig Translation. Crit. Care Med. 2014;42(5):e329–e344. doi: 10.1097/CCM.0000000000000225. [DOI] [PubMed] [Google Scholar]
- 93.Mayer Ea K.T., Gupta A. Gut/brain axis and the microbiota. J. Clin. Invest. 2015;125(3):926–938. doi: 10.1172/JCI76304. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Tillisch K. The effects of gut microbiota on CNS function in humans. Gut Microb. 2014;5(3):404–410. doi: 10.4161/gmic.29232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Dobson G.P., Letson H.L., Biros E., Morris J.L. Specific pathogen-free (SPF) animal status as a variable in biomedical research: have we come full circle? EBioMedicine (Lancet) 2019;41(March):42–43. doi: 10.1016/j.ebiom.2019.02.038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Letson H.L., Morris J.L., Biros E., Dobson G.P. Conventional and specific-pathogen free rats respond differently to anesthesia and surgical trauma. Sci. Rep. 2019;9(1):9399. doi: 10.1038/s41598-019-45871-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Lederer A.K., Pisarski P., Kousoulas L., Fichtner-Feigl S., Hess C., Huber R. Postoperative changes of the microbiome: are surgical complications related to the gut flora? A systematic review. BMC Surg. 2017;17(1):125. doi: 10.1186/s12893-017-0325-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Riedel B., Rafat N., Browne K., Burbury K., Schier R. Perioperative implications of vascular endothelial dysfunction: current understanding of this critical sensor-effector organ. Current Anesthesiology Reports. 2013;3(3):151–161. [Google Scholar]
- 99.Aird W.C. Spatial and temporal dynamics of the endothelium. J. Thromb. Haemostasis. 2005;3(7):1392–1406. doi: 10.1111/j.1538-7836.2005.01328.x. [DOI] [PubMed] [Google Scholar]
- 100.Huang M.L., Godula K. Nanoscale materials for probing the biological functions of the glycocalyx. Glycobiology. 2016;26(8):797–803. doi: 10.1093/glycob/cww022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Astapenko D., Benes J., Pouska J., Lehmann C., Islam S., Cerny V. Endothelial glycocalyx in acute care surgery - what anaesthesiologists need to know for clinical practice. BMC Anesthesiol. 2019;19(1):238. doi: 10.1186/s12871-019-0896-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Ekeloef S., Larsen M.H., Schou-Pedersen A.M., Lykkesfeldt J., Rosenberg J., Gogenur I. Endothelial dysfunction in the early postoperative period after major colon cancer surgery. Br. J. Anaesth. 2017;118(2):200–206. doi: 10.1093/bja/aew410. [DOI] [PubMed] [Google Scholar]
- 103.Song J.W., Goligorsky M.S. Perioperative implication of the endothelial glycocalyx. Korean J Anesthesiol. 2018;71(2):92–102. doi: 10.4097/kjae.2018.71.2.92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Luft J.H. The structure and properties of the cell surface coat. Int. Rev. Cytol. 1976;45:291–382. doi: 10.1016/s0074-7696(08)60081-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zeng Y., Tarbell J.M. The adaptive remodeling of endothelial glycocalyx in response to fluid shear stress. PloS One. 2014;9(1) doi: 10.1371/journal.pone.0086249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Naumann D.N., Hazeldine J., Davies D.J., Bishop J., Midwinter M.J., Belli A., Harrison P., Lord J.M. Endotheliopathy of trauma is an on-scene phenomenon, and is associated with multiple organ dysfunction syndrome: a prospective observational study. Shock. 2018;49(4):420–428. doi: 10.1097/SHK.0000000000000999. [DOI] [PubMed] [Google Scholar]
- 107.Torres Filho I.P., Torres L.N., Salgado C., Dubick M.A. Novel adjunct drugs reverse endothelial glycocalyx damage after hemorrhagic shock in rats. Shock. 2017;48(5):583–589. doi: 10.1097/SHK.0000000000000895. [DOI] [PubMed] [Google Scholar]
- 108.Cherry A.D. Mitochondrial dysfunction in cardiac surgery. Anesthesiol. Clin. 2019;37(4):769–785. doi: 10.1016/j.anclin.2019.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Cap A.P., Hunt B. Acute traumatic coagulopathy. Curr. Opin. Crit. Care. 2014;20(6):638–645. doi: 10.1097/MCC.0000000000000158. [DOI] [PubMed] [Google Scholar]
- 110.Boudreau L.H., Duchez A.C., Cloutier N., Soulet D., Martin N., Bollinger J., Pare A., Rousseau M., Naika G.S., Levesque T., et al. Platelets release mitochondria serving as substrate for bactericidal group IIA-secreted phospholipase A2 to promote inflammation. Blood. 2014;124(14):2173–2183. doi: 10.1182/blood-2014-05-573543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Hauser C.J., Otterbein L.E. Danger signals from mitochondrial DAMPS in trauma and post-injury sepsis. Eur. J. Trauma Emerg. Surg. 2018;44(3):317–324. doi: 10.1007/s00068-018-0963-2. [DOI] [PubMed] [Google Scholar]
- 112.Berthelsen P.G. On the quest for stress-free surgical operations. Acta Anaesthesiol. Scand. 2015;59 doi: 10.1111/aas.12444. 246–24. [DOI] [PubMed] [Google Scholar]
- 113.Letson H.L., Dobson G.P. 3.0% NaCl Adenosine, Lidocaine, Mg2+ (ALM) bolus and 4 hours ‘drip’ infusion reduces non-compressible hemorrhage by 60% in a rat model. J Trauma Acute Care Surg. 2017;82(6):1063–1072. doi: 10.1097/TA.0000000000001454. [DOI] [PubMed] [Google Scholar]
- 114.Letson H.L., Morris J.L., Biros E., Dobson G.P. ALM fluid therapy leads to 72 hr survival after hemorrhagic shock: a model for studying differential gene expression and extending biological time. J Trauma Acute Care Surg. 2019;87(3):606–613. doi: 10.1097/TA.0000000000002397. [DOI] [PubMed] [Google Scholar]
- 115.Letson H.L., Granfeldt A., Jensen T.H., Mattson T.H., Dobson G.P. ALM supports a high flow, hypotensive, vasodilatory state with improved O2 delivery and cerebral protection in a pig model of non-compressible hemorrhage. J. Surg. Res. 2020;253(Sept):127–138. doi: 10.1016/j.jss.2020.03.048. [DOI] [PubMed] [Google Scholar]
- 116.Davenport L., Letson H.L., Dobson G.P. Immune-inflammatory activation after a single laparotomy in a rat model: effect of adenosine, lidocaine and Mg2+ infusion to dampen the stress response. Innate Immun. 2017;23(5):482–494. doi: 10.1177/1753425917718921. [DOI] [PubMed] [Google Scholar]
- 117.Downing N.S., Shah N.D., Aminawung J.A., Pease A.M., Zeitoun J.D., Krumholz H.M., Ross J.S. Postmarket safety events among novel therapeutics approved by the US food and drug administration between 2001 and 2010. J. Am. Med. Assoc. 2017;317(18):1854–1863. doi: 10.1001/jama.2017.5150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Botto F., Alonso-Coello P., Chan M.T., Villar J.C., Xavier D., Srinathan S. Myocardial injury after noncardiac surgery: a large, international, prospective cohort study establishing diagnostic criteria, characteristics, predictors, and 30-day outcomes. Anesthesiology. 2014;120(3):564–578. doi: 10.1097/ALN.0000000000000113. [DOI] [PubMed] [Google Scholar]
- 119.Sessler D.I., Devereaux P. Perioperative troponin screening. Anesth. Analg. 2016;123(2):359–360. doi: 10.1213/ANE.0000000000001450. [DOI] [PubMed] [Google Scholar]
- 120.Price C.C., Garvan C.W., Monk T.G. Type and severity of cognitive decline in older adults after noncardiac surgery. Anesthesiology. 2008;108(1):8–17. doi: 10.1097/01.anes.0000296072.02527.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Mrkobrada M., Chan M.T.V., Cowan D., Campbell D., Wang C.Y., Torres D., Malaga G., Sanders R.D., Sharma M., Brown C., et al. Perioperative covert stroke in patients undergoing non-cardiac surgery (NeuroVISION): a prospective cohort study. Lancet. 2019;394(10203):1022–1029. doi: 10.1016/S0140-6736(19)31795-7. [DOI] [PubMed] [Google Scholar]
- 122.Vaara S.T., Bellomo R. Postoperative renal dysfunction after noncardiac surgery. Curr. Opin. Crit. Care. 2017;23(5):440–446. doi: 10.1097/MCC.0000000000000439. [DOI] [PubMed] [Google Scholar]
- 123.Kelkar K.V. Post-operative pulmonary complications after non-cardiothoracic surgery. Indian J. Anaesth. 2015;59(9):599–605. doi: 10.4103/0019-5049.165857. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Kheterpal S., Tremper K.K., Englesbe M.J., O'Reilly M., Shanks A.M., Fetterman D.M., Rosenberg A.L., Swartz R.D. Predictors of postoperative acute renal failure after noncardiac surgery in patients with previously normal renal function. Anesthesiology. 2007;107:892–902. doi: 10.1097/01.anes.0000290588.29668.38. [DOI] [PubMed] [Google Scholar]
- 125.Danelich I.M., Lose J.M., Wright S.S., Asirvatham S.J., Ballinger B.A., Larson D.W., Lovely J.K. Practical management of postoperative atrial fibrillation after noncardiac surgery. J. Am. Coll. Surg. 2014;219(4):831–841. doi: 10.1016/j.jamcollsurg.2014.02.038. [DOI] [PubMed] [Google Scholar]
- 126.Torrance H.D., Pearse R.M., O'Dwyer M.J. Does major surgery induce immune suppression and increase the risk of postoperative infection? Curr. Opin. Anaesthesiol. 2016;29(3):376–383. doi: 10.1097/ACO.0000000000000331. [DOI] [PubMed] [Google Scholar]