Dear Editor,
The severity of acute hypoxemic respiratory failure (AHRF) in Coronavirus Disease 2019 (COVID-19) correlates poorly with lung weight and lung mechanics, leading to the proposal of phenotypes that may be associated with similar degree of hypoxaemia but different lung volume, weight, and compliance [1]. The alteration of the pulmonary vascular tone and immune thrombosis of the alveolar capillaries [2] may account for these pathophysiological characteristics and for the high physiological dead space observed in these patients.
To describe estimated indices of physiological dead space—and their association with respiratory mechanics, severity of hypoxaemia, biomarkers, and outcomes—we performed a retrospective analysis of adult patients with COVID-19 respiratory failure requiring mechanical ventilation in four medical Intensive Care Units (ICU) within Guy’s and St Thomas’ NHS Trust—London, UK (Ethics reference: 10,796).
We used the recorded values at the time of worst PaO2/FiO2 observed on the day of critical care admission. Continuous variables were compared using Mann–Whitney U test. This cohort included 213 patients (73% males), mean (95%CI) age 56 (54–57) years, and PaO2/FiO2 128 (121–135) mmHg. When subdivided in four groups based on cut-off PaO2/FiO2 of 150 mmHg and compliance of 40 mL/cmH2O; 72% (n = 154) had PaO2/FiO2 < 150 mmHg, of these, 112 (73%, or 53% of the overall cohort) had compliance < 40 mL/cmH2O (eTable 1). The mean (95%CI) estimated physiological dead space fraction [3] was high in the entire cohort at 0.53 (0.51–0.56).
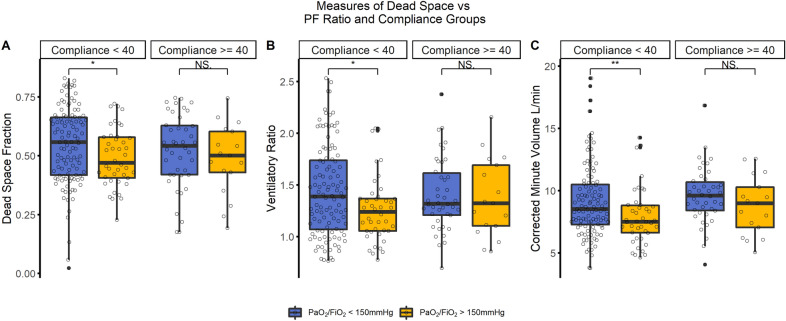
ICU outcome was available for 193 patients, where estimated physiological dead space fraction was higher in non-survivors [median (IQR), 0.57 (0.46–0.65) vs. 0.5 (0.4–0.64); p = 0.03]. All estimates of physiological dead space increased with the degree of hypoxaemia, but not with the reduction in lung compliance (Fig. 1). Compared with patients with PaO2/FiO2 > 150 mmHg, patients with PaO2/FiO2 < 150 mmHg had higher estimated physiological dead space fraction [0.55 (0.52–0.57) vs. 0.5 (0.47–0.53); p = 0.036) (Fig. 1A), mean (95%CI) corrected minute volume [4] [9.3(8.8–9.7) vs. 8.2 (7.6–8.8) L/min; p = 0.004] (Fig. 1B), and ventilatory ratio [5] [1.43 (1.37–1.5) vs. 1.29 (1.2–1.38); p = 0.001] (Fig. 1C). Although patients with compliance < 40 mL/cmH2O had a higher corrected minute volume [4] [9.4 (8.8–10) vs. 8.8 (8.4–9.3) L/min; p = 0.023] (Fig. 1C), there was no difference in the two compliance groups in estimated physiological dead space fraction [0.55 (0.48–0.56) vs. 0.54 (0.52–0.56); p = 0.72] (Fig. 1A) and ventilatory ratio [5] 1.4 (1.3–1.5) vs. 1.4 (1.33–1.46); p = 0.76) (Fig. 1B).
Fig. 1.
Distributions of dead space indices in the groups of compliance and PaO2/FiO2. (A): Physiological dead space fraction; (B): ventilatory ratio; (C): corrected minute ventilation
Physiological dead space correlated with hypoxaemia, but it was dissociated from alterations in lung mechanics in COVID-19 ventilated patients. Immuno-thrombosis is a mechanism that may explain both the increase in physiological dead space and hypoxaemia. Interestingly, we found that the highest median (IQR) levels of D-Dimers [2.1 (1–7) mg/L]; ferritin [1,627 (914–2555) ug/L], CRP [219.5 (147.25–324) mg/L], and troponin [23 (12–62) ng/L] were seen in patients with PaO2/FiO2 < 150 mmHg, but relatively preserved compliance (> 40 mL/cmH2O). However, these values were statistically similar to the ones recorded in the other groups.
In conclusion, these data suggest that increased physiological dead space is a characteristic of patients with COVID-19 AHRF, with no relation with compliance of the respiratory system. Given that both PaO2/FiO2 and physiological dead space are worse in non-survivors, it is unclear if dead space is independently associated with mortality or if its effect on outcome is mediated through hypoxaemia.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Funding
S–H is supported by the National Institute for Health Research Clinician Scientist Award (CS-2016-16-011). FF is supported by the National Institute of Health Research i4i (NIHR200681). The views expressed in this publication are those of the authors and not necessarily those of the NHS, the National Institute for Health Research or the Department of Health and Social Care.
Compliance with ethical standards
Conflicts of interest
The authors declare no conflict of interest.
Footnotes
Luigi Camporota and Manu Shankar-Hari are to be considered joint last (senior) authors.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Gattinoni L, Chiumello D, Caironi P, Busana M, Romitti F, Brazzi L, Camporota L. COVID-19 pneumonia: different respiratory treatments for different phenotypes? Intensive Care Med. 2020;46:1099–1102. doi: 10.1007/s00134-020-06033-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ackermann M, Verleden SE, Kuehnel M, Haverich A, Welte T, Laenger F, Vanstapel A, Werlein C, Stark H, Tzankov A, Li WW, Li VW, Mentzer SJ, Jonigk D. Pulmonary vascular endothelialitis, thrombosis, and angiogenesis in Covid-19. N Engl J Med. 2020;2:19–57. doi: 10.1056/NEJMoa2015432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beitler JR, Thompson BT, Matthay MA, Talmor D, Liu KD, Zhuo H, Hayden D, Spragg RG, Malhotra A. Estimating dead-space fraction for secondary analyses of acute respiratory distress syndrome clinical trials. Crit Care Med. 2015;43:1026–1035. doi: 10.1097/CCM.0000000000000921. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wexler HR, Lok P. A simple formula for adjusting arterial carbon dioxide tension. Can Anaesth Soc J. 1981;28:370–372. doi: 10.1007/BF03007805. [DOI] [PubMed] [Google Scholar]
- 5.Sinha P, Fauvel NJ, Singh S, Soni N. Ventilatory ratio: a simple bedside measure of ventilation. Br J Anaesth. 2009;102:692–697. doi: 10.1093/bja/aep054. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.