Abstract
Patient: Gender, 26-year-old
Final Diagnosis: COVID-19
Symptoms: Cough • fever
Medication:—
Clinical Procedure: —
Specialty: Hematology
Objective:
Rare co-existance of disease or pathology
Background:
Beta-hemoglobinopathies and glucose-6-phosphate dehydrogenase (G6PD) deficiency are genetic disorders that cause hemolytic anemia when exposed to oxidative stress. Their co-existence is, however, not proven to enhance the severity of anemia.
Case Report:
We report the case of a young man with no known co-morbidities, who came with fever and cough and was diagnosed with COVID-19 pneumonia. He was found to have hemoglobin D thalassemia and G6PD deficiency during further evaluation. Hydroxychloroquine therapy started initially, was discontinued after 3 doses once the G6PD deficiency was diagnosed. His hospital course showed a mild drop in hemoglobin with evidence of hemolysis on peripheral smear. However, the hemoglobin improved without any need for transfusion.
Conclusions:
Hydroxychloroquine therapy can induce hemolytic crises in patients with underlying G6PD deficiency or hemoglobinopathies and should be avoided or closely monitored. Immediate intervention to stop hydroxychloroquine after 3 doses saved our patient from a major hemolytic crisis. The significance of this case report is that it is the first report that outlines the clinic course of COVID-19 pneumonia in a patient with underlying hemoglobin D disease and G6PD deficiency.
MeSH Keywords: COVID-19, Glucosephosphate Dehydrogenase Deficiency, Hemoglobinopathies, Hydroxychloroquine, SARS Virus
Background
Beta-hemoglobinopathies and glucose-6-phosphate dehydrogenase (G6PD) deficiency are genetic disorders that cause hemolytic anemia when exposed to oxidative stress. Their coexistence is, however, not proven to enhance the severity of anemia [1,2]. Hemoglobin D results from the substitution of glutamic acid by glutamine in the beta chain of hemoglobin. Hemoglobin D disease can occur in four different forms, namely heterozygous hemoglobin D trait, hemoglobin D thalassemia, hemoglobin SD disease, and homozygous hemoglobin D disease [3]. G6PD deficiency, an x-linked hereditary genetic defect due to mutations in the G6PD gene, is the most common human enzyme defect. The highest prevalence of G6PD deficiency is seen in the Arabian Peninsula and tropical Africa [4]. G6PD enzyme catalyzes the conversion of nicotinamide ade-nine dinucleotide phosphate (NADP) into nicotinamide adenine dinucleotide phosphate hydrogen (NADPH), which protects cells from oxidative damage. A hemolytic crisis is triggered by oxidative stress caused by food (fava beans), drugs, or infections [5]. There is no evidence showing that SARS-CoV-2 can induce a hemolytic crisis. However, there is experimental data that shows oxidative stress in host cells is an important factor in human coronavirus 229E infectivity. So, G6PD deficiency enhances HCoV 229E infection [6]. This is the first reported case of COVID-19 in a patient with simultaneous hemoglobin D thalassemia and G6PD deficiency.
Case Report
A 26-year-old Pakistani male working as a military officer in Qatar, without any known comorbidities, presented to our fever clinic with 3 days of sore throat, rhinitis, and headache, and 2 days of nonproductive cough and low-grade fever. It was associated with generalized body ache, but the review of systems was otherwise unremarkable. There was no shortness of breath or change in bowel or bladder habits. He was recently exposed to a colleague who was diagnosed with pneumonia due to SARS-CoV-2. He denied any episodes of cola-colored urine. There was no significant family history of medical conditions or consanguineous marriages. General examination showed that he was febrile (39.6°C) and had tachycardia (115 beats per minute) with normal blood pressure and saturation in room air. He was not in any distress, and there was no use of accessory muscles for breathing. There were diffuse bilateral coarse crackles on auscultation of the chest. Other systems were unremarkable on examination. Initial laboratory assessment showed a normal white cell count (7700/µL), microcytic hypochromic anemia with increased red cell distribution width (RDW=17.7%), elevated urea (9.2 mmol/L), creatinine (162 µmol/L), and C-reactive protein (34 mg/L) (Table 1).
Table 1.
Results of important laboratory tests on day 1, day 10, and day 21.
| Detail | Result | Normal range | ||
|---|---|---|---|---|
| Day 1 | Day 12 | Day 21 | ||
| Complete blood count | ||||
| White blood cells (×103/µL) | 8.4 | 4.4 | 9.5 | 4–10 |
| Red blood cells (×106/µL) | 6.2 | 5.2 | 6.1 | 4.5–5.5 |
| Platelets (×103/µL) | 252 | 325 | 475 | 150–400 |
| Hemoglobin (gm/dL) | 12.2 | 9.5 | 11.6 | 13.0–17.0 |
| Hematocrit (%) | 37.4 | 30.3 | 36.8 | 40.0–50.0 |
| Mean corpuscular volume (fL) | 57.6 | 58.8 | 60.3 | 83.0–101.0 |
| Mean corpuscular hemoglobin (pg) | 19.0 | 18.4 | 19 | 27.0–32.0 |
| Mean corpuscular hemoglobin concentration (gm/dL) | 32.6 | 31.6 | 31.5 | 31.5–34.4 |
| RDW (%) | 17.7 | 15.8 | 18.4 | 11.6–14.5 |
| Reticulocytes (%) | 0.7 | 0.8 | 0.7 | 0.5–2.5 |
| Comprehensive metabolic panel | ||||
| Urea (mmol/L) | 9.2 | 5.8 | 5.2 | 2.8–8.1 |
| Creatinine (µmol/L) | 162 | 98 | 92 | 62–106 |
| Total bilirubin (µmol/L) | 13.7 | 12 | 14.7 | 3.4–20.5 |
| Alkaline phosphatase (U/L) | 69.4 | 46 | 69.7 | 40–150 |
| Alanine amino transferase (U/L) | 23 | 164 | 115.7 | 0–55 |
| Aspartate amino transferase (U/L) | 23 | 76 | 38 | 5–34 |
| C-reactive protein (mg/L) | 34 | 13.2 | 3 | 0–5 |
RDW – red cell distribution width.
The polymerase chain reaction (PCR) test for COVID-19 from nasopharyngeal and throat swab was positive. Combined nasopharyngeal and oropharyngeal swabs (Copan Diagnostics Inc., Italy) were collected and placed in Universal Transport Medium (UTM). Aliquots of UTM were either extracted on the QIAsymphony platform (Qiagen, USA) and tested by reverse transcriptase polymerase chain reaction (RT-PCR) with the Thermo Fisher TaqPath™ COVID-19 RT-PCR Kit (Thermo Fisher, USA), targeting the S, N,and ORF-1a/b genes, or loaded directly on to a Roche Cobas® 6800 and assayed with the Cobas® SARS-CoV-2 Test (Roche, Switzerland) targeting the ORF-1a/b and E-gene regions of SARS-CoV-2.
Initial chest x-ray showed bilateral accentuation of vascular markings more in the lower zone. He was started on the treatment for COVID-19 associated pneumonia with oseltamivir 150 mg oral twice daily, lopinavir/ritonavir 400 mg/100 mg oral twice daily, azithromycin 500 mg intravenous once daily, hydroxychloroquine 400 mg twice daily followed by once daily and ceftriaxone 2 g once daily, as per hospital protocol. Monitoring of QTc interval in electrocardiogram (ECG) is a part of our hospital protocol before initiation and then daily during treatment of COVID-19 patients with hydroxychloroquine and azithromycin (Figure 1). Hemoglobin electrophoresis pattern was most consistent with hemoglobin D disease (Table 2). Urea and creatinine levels improved over the next 1 week with adequate hydration, suggesting that it was most likely pre-renal due to dehydration. Vitamin B12 levels were low, and he was started on oral supplementation. Screening of glucose-6-phosphate dehydrogenase (G6PD) level was done as a part of the protocol while initiating treatment with hydroxychloroquine, and it was found to be deficient at <9 mU/109 red blood cells (RBCs). Hydroxychloroquine was stopped after 3 doses, with the finding of G6PD deficiency. Azithromycin, oseltamivir, and lopinavir/ritonavir were continued at the same dose, while ceftriaxone was escalated to piperacillin-tazobactam considering the progression of previously seen bilateral multiple pneumonic consolidations on chest x-ray. The changes in the chest x-ray from presentation until day 17 is shown in Figure 2A–2D). Serum aspartate transaminase (AST) and ala-nine aminotransferase (ALT) levels were normal before admission. There was a transient rise in liver transaminases from day 7 to 14. His hemoglobin level was found to drop from 12.2 to 9.1 gm/dL over the next 10 days. Hemolysis workup showed normal bilirubin and normal haptoglobin but elevated lactate dehydrogenase (LDH) of 300 U/L, normal range: 135–225 U/L. Peripheral smear showed moderate hypochromic microcytic anemia with anisocytosis, spherocytes, schistocytes (1.2%), fragmented RBCs, few ovalocytes, and few teardrop cells. Reticulocyte count was 0.7% on admission, 0.8% on day 10 and 0.7% on day 18. Urine dipstick and microscopy were normal, and there was no evidence of hemoglobinuria or hematuria. His clinical condition worsened, starting from day 3 of admission.
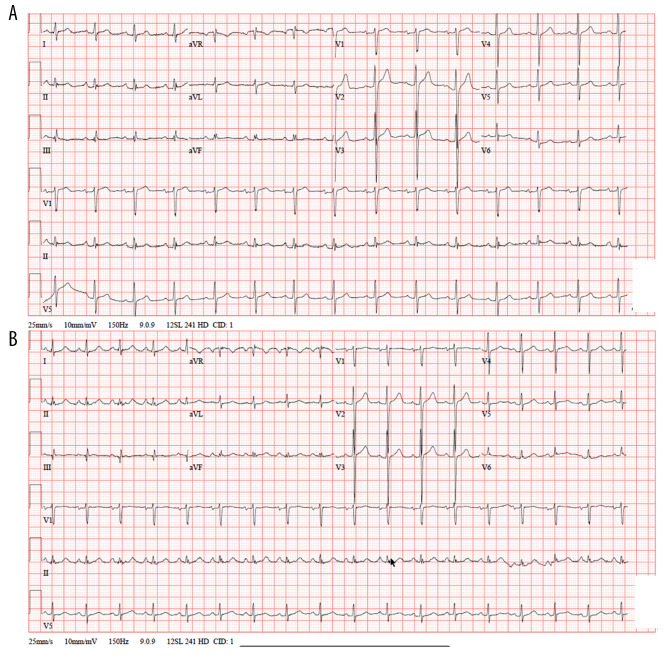
Figure 1.
(A) Electrocardiogram (ECG) on admission showing a QTc interval of 394 milliseconds before initiation of hydroxychloroquineazithromycin therapy. (B) ECG on day 12 showing a gradual prolongation of QTc interval to 433 milliseconds.
Table 2.
Results of anemia workup including hemoglobin electrophoresis.
| Detail | Result | Normal range |
|---|---|---|
| Iron studies | ||
| Iron (µmol/L) | 3 | 6–35 |
| TIBC (µmol/L) | 60 | 45–80 |
| Transferrin (gm/L) | 2.4 | 2.0–3.6 |
| Iron saturation (%) | 5 | 15–45 |
| Ferritin (µg/L) | 450.0 | 38.0–270.0 |
| B12 and folate | ||
| Folate (nmol/L) | 35 | 10–70 |
| Vitamin B12 (pmol/L) | 134.0 | 145.0–596.0 |
| Hemoglobin electrophoresis | ||
| Hgb A (%) | 0.0 | 95.8–98.0 |
| Hgb A2 (%) | 1.9 | 2.0–3.3 |
| Hgb F (%) | 1.9 | 0.0–0.9 |
| Hgb S (%) | 0.0 | |
| Hgb D (%) | 96.2 | |
| Sickle test | Negative |
TIBC – total iron-binding capacity; Hgb – hemoglobin.
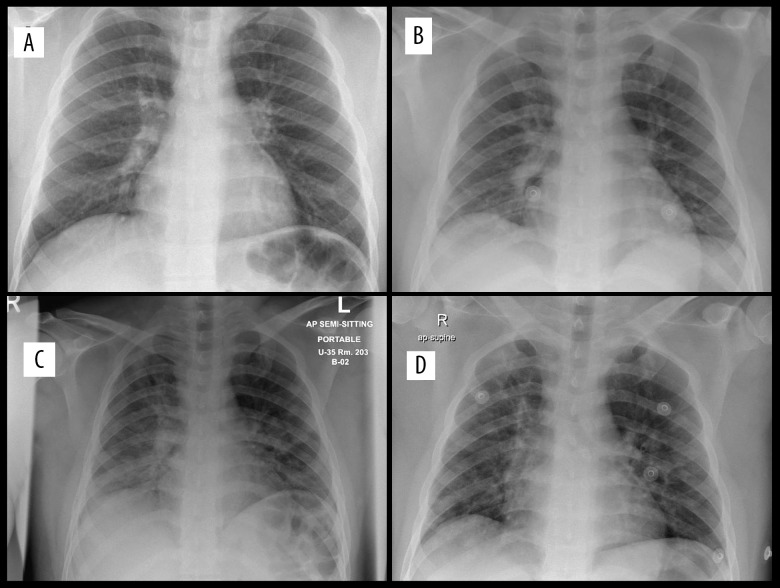
Figure 2.
(A) Day 1. Bilateral accentuation of vascular markings of the lung fields, more seen on the lower zones. No pleural effusion. No pneumothorax. Early features suggestive of viral pneumonia. (B) Day 4. More prominent hilar shadows on the right. Accentuated prominent broncho-vascular markings, thick pulmonary reticulations, and peri-bronchial thickening with some confluence at the perihilar and both lung bases with basal interstitial infiltrates predominantly. (C) Day 9. Progression of the previously seen bilateral multiple pneumonic consolidation. (D) Day 17. Mild Improvement in lower zone infiltrates.
Oxygen requirement increased progressively from 2 liters via nasal cannula on day 3 to 10 liters via a non-rebreather mask on day 11. The same medications were continued except escalating ceftriaxone to piperacillin-tazobactam. He continued to spike fever till day 8. After day 12, his oxygen requirements started improving, and he was back to baseline by day 15. He received 9 days of azithromycin, 2 days of hydroxychloroquine, 3 days of ceftriaxone, 5 days of lopinavir/ritonavir, 10 days of oseltamivir, and 10 days of piperacillin-tazobactam. He also received intravenous methylprednisolone 40 mg twice daily for 5 days from day 10 to day 14 (Figure 3). COVID-19 PCR repeated on day 14 and day 21 were positive, but he was clinically free after day 14. Hemoglobin was stable after day 12, and improved to his baseline by day 22, without any need for transfusion. COVID-19 PCR finally turned negative on day 28.
Figure 3.
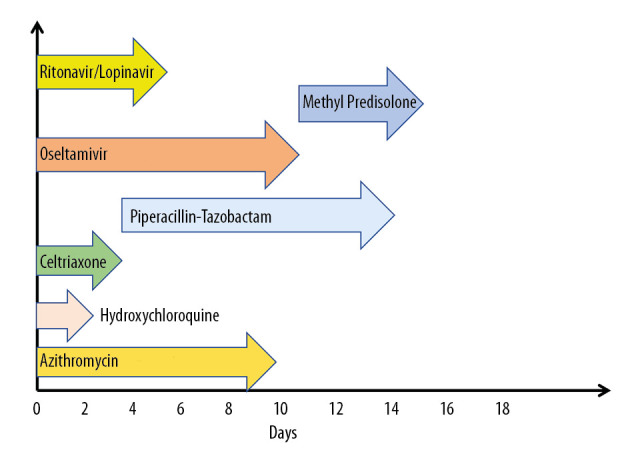
Flowchart summarizing the treatments received.
Discussion
Patients with chronic morbidities are likely to be severely affected by SARS-CoV-2 infection, but no data is available regarding hemoglobinopathies. There is limited data about the clinical course of individuals with thalassemia who have contracted COVID-19. A recent study from Italy involving 11 patients with thalassemia syndromes (10 transfusion dependent and 1 non-transfusion dependent), infected with SARS-CoV-2, did not show increased severity of COVID-19 in thalassemia syndromes. Neither death nor severe SARS or signs of cytokine storm was observed in these 11 patients. The clinical course ranged from 10 to 29 days, and 2 of them received hydroxychloroquine without a hemolytic crisis [7]. In our patient also, the clinical course was lengthy and complicated.
The co-existence of hemoglobin D disease with G6PD deficiency has seldom been reported. Our patient had a baseline microcytic hypochromic anemia, which worsened during the period of hospitalization. His anemia was influenced by multiple factors such as underlying hemoglobinopathy, B12 deficiency, possible iron deficiency, acute illness, and acute hemolysis. An RDW of 17.7 was more in favor of iron deficiency anemia (IDA). However, a Red Cell Distribution Width Index (RDWI) of 181 and Mentzer Index of 9.2 were suggestive of thalassemia. RDWI is a reliable and useful index for differentiation between IDA and beta-thalassemia trait, as compared to RDW [8]. Iron studies were not conclusive for a diagnosis of iron deficiency. Even though the serum iron level and iron saturations were low, serum transferrin and total iron-binding capacity (TIBC) were normal. Serum ferritin was high but was not reliable as it is often elevated in acute inflammatory states. However, at this point, we cannot totally rule out the possibility of an underlying iron deficiency in addition to thalassemia and it is mandatory to repeat the iron studies after the patient is out of acute infection. The pattern of anemia could also be influenced by the fact that he had vitamin B12 deficiency and possibly a component of chronic hemolysis. The drop in our patient’s hemoglobin was multifactorial. A mild drop in hemoglobin levels could be seen in acute illness [9]. Hemolysis was evidenced by the elevation of LDH and the presence of characteristic cells in the peripheral smear. The period of hemolysis could not be co-related with a significant rise in reticulocyte count. The reason could be that a) acute viral infections cause transient aplasia due to direct and indirect damage to the hematopoietic stem and progenitor cells (HSPCs) and the surrounding tissue [10], b) the hemolysis was mild and transient, c) the patient had underlying B12 deficiency, and d) possible iron deficiency.
The fact that our patient did not develop hemolytic crisis despite these comorbidities supports the general consideration that SARS-CoV-1, SARS-CoV-2, or MERS-CoV do not act as a trigger for G6PD deficiency hemolytic crisis. There is a report of severe hemolytic crisis in a patient with G6PD deficiency, initiated by severe COVID-19 infection and hydroxychloroquine use, but it is not clear whether the hemolytic crisis was induced by the infection or by the drug [10]. A recent large study in rheumatology patients evaluating G6PD deficiency with concurrent use of hydroxychloroquine recommended against the routine measurement of G6PD levels or withholding hydroxychloroquine therapy among African American patients with G6PD deficiency [11]. Azithromycin and hydroxychloroquine are part of the protocol of most countries to treat COVID-19 infections, and there is upcoming evidence supporting their use [12]. In our hospital, testing for G6PD deficiency is mandatory before initiation of hydroxychloroquine therapy in patients with COVID-19 infection. There is limited availability of data regarding the impact of COVID-19 on the quality of life of patients with thalassemia. Children with thalassemia major require periodic blood transfusions. Present data do not suggest the risk of transfusion transmission of SARS-CoV-2. However, there is a general understanding to defer blood donation for 21 days after possible exposure to a confirmed case and for 28 days after symptom resolution in a positive case [13,14].
Conclusions
Considering the high prevalence of G6PD deficiency in the Arabian Peninsula, and the fact that there is no exclusive data regarding the association between SARS-CoV-2, G6PD deficiency, and hemolytic crisis, we recommend screening COVID-19 patients for G6PD deficiency, especially before initiation of hydroxychloroquine therapy. The significance of this case report is that it is the first report that outlines the clinic course of COVID-19 pneumonia in a patient with underlying hemoglobin D disease and G6PD deficiency.
Acknowledgments
The authors would like to acknowledge Internal Medicine Residency Program of Hamad Medical Corporation for scientific support.
Footnotes
Department and Institution where work was done
Department of Internal Medicine, Hamad General Hospital, Hamad Medical Corporation, Doha, Qatar.
References:
- 1.Pornprasert S, Phanthong S. Anemia in patients with coinherited thalassemia and glucose-6-phosphate dehydrogenase deficiency. Hemoglobin. 2013;37(6):536–43. doi: 10.3109/03630269.2013.819558. [DOI] [PubMed] [Google Scholar]
- 2.Fibach E, Rachmilewitz E. The role of oxidative stress in hemolytic anemia. Curr Mol Med. 2008;8(7):609–19. doi: 10.2174/156652408786241384. [DOI] [PubMed] [Google Scholar]
- 3.Torres Lde S, Okumura JV, Silva DG, et al. Hemoglobin D-Punjab: Origin, distribution and laboratory diagnosis. Rev Bras Hematol Hemoter. 2015;37(2):120–26. doi: 10.1016/j.bjhh.2015.02.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Howes RE, Piel FB, Patil AP, et al. G6PD deficiency prevalence and estimates of affected populations in malaria endemic countries: A geostatistical model-based map. PLoS Med. 2012;9(11):e1001339. doi: 10.1371/journal.pmed.1001339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Cappellini MD, Fiorelli G. Glucose-6-phosphate dehydrogenase deficiency. Lancet Lond Engl. 2008;371(9606):64–74. doi: 10.1016/S0140-6736(08)60073-2. [DOI] [PubMed] [Google Scholar]
- 6.Wu Y-H, Tseng C-P, Cheng M-L, et al. Glucose-6-phosphate dehydrogenase deficiency enhances human coronavirus 229E infection. J Infect Dis. 2008;197(6):812–16. doi: 10.1086/528377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Motta I, Amicis De Migone M, et al. SARS-CoV-2 infection in beta thalassemia: Preliminary data from the Italian experience. Am J Hematol. 2020 doi: 10.1002/ajh.25840. [Online ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Jameel T, Baig M, Ahmed I, et al. Differentiation of beta thalassemia trait from iron deficiency anemia by hematological indices. Pak J Med Sci. 2017;33(3):665–69. doi: 10.12669/pjms.333.12098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Viana MB. Anemia and infection: A complex relationship. Rev Bras Hematol Hemoter. 2011;33(2):90–92. doi: 10.5581/1516-8484.20110024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Beauverd Y, Adam Y, Assouline B, Samii K. COVID-19 infection and treatment with hydroxychloroquine cause severe haemolysis crisis in a patient with glucose-6-phosphate dehydrogenase deficiency. Eur J Haematol. 2020 doi: 10.1111/ejh.13432. [Online ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Mohammad S, Clowse MEB, Eudy AM, Criscione-Schreiber LG. Examination of hydroxychloroquine use and hemolytic anemia in G6PDH-deficient patients. Arthritis Care Res. 2018;70(3):481–85. doi: 10.1002/acr.23296. [DOI] [PubMed] [Google Scholar]
- 12.Gautret P, Lagier J-C, Parola P, et al. Hydroxychloroquine and azithromycin as a treatment of COVID-19: Results of an open-label non-randomized clinical trial. Int J Antimicrob Agents. 2020 doi: 10.1016/j.ijantimicag.2020.105949. [Online ahead of print] [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yadav U, Pal R. Challenging times for children with transfusion-dependent thalassemia amid the COVID-19 pandemic. Indian Pediatr. 2020;57(5):478. doi: 10.1007/s13312-020-1831-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Chang L, Yan Y, Wang L. Coronavirus disease 2019: Coronaviruses and blood safety. Transfus Med Rev. 2020;34(2):75–80. doi: 10.1016/j.tmrv.2020.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]