On December 31, 2019, the China Country Office of the World Health Organization was informed of 44 cases of pneumonia of unknown etiology that had been detected in Wuhan City, Hubei Province, China, which have since been recognized as due to SARS-CoV-2 (severe acute respiratory syndrome coronavirus 2). The outcome of these infections is the current global pandemic. Also, because of our interdependent and highly mobile global population, we have all, to a certain extent, been affected, either personally or economically, or both. The rapid spread of the coronavirus disease 2019 (COVID-19) pandemic led to many countries and healthcare organizations being caught unprepared. However, the nations that had been initially severely affected by COVID-19 now appear to be beyond the peak of the infection curve, with the virus reproductive rate decreasing (ie, reproduction number < 1). However, the collateral damage to cancer diagnostic and treatment services has been severe, and cancer outcomes are likely to be adversely affected, potentially for many years. Colorectal cancer (CRC) is the third-leading global cause of cancer-related deaths, accounting for 880,792 deaths (9.2%), with an estimate of 1,849,518 new cases (10.2%) in 2018.1 To avoid a future crisis of avoidable cancer deaths, the diagnosis and treatment pathways for cancer must be maintained at a near normal throughput, with urgent attention to the backlog of patients we can expect.2 To date, most endoscopy units have severely restricted their activity except for time-critical diagnostic and therapeutic procedures. For patients already waiting to undergo diagnostic tests, especially those with worrying symptoms, this delay has been of great concern to both patients and doctors.
To begin providing endoscopy services again, practice statements from recognized societies have provided thorough guides on how to implement and resume endoscopy services. However, a strategy is also needed to manage the backlog of diagnostic and therapeutic endoscopic procedures for patients requiring colorectal testing.3, 4, 5
Traditionally, colonoscopy has been the first-line diagnostic test and reference standard for the investigation of colorectal symptoms, including for patients with minor and low-risk symptoms. However, the low threshold for performing colonoscopy led to low diagnostic yields of significant disease, despite the high endoscopic workload. In a review of 1116 cases during a 2-year period, the correlation of lower gastrointestinal symptoms with colonoscopy showed that only 14.5% of those patients had had clinically significant disease and only 1.7% had had CRC.6 To clear the backlog of colonoscopies resulting from the lockdown during the COVID-19 pandemic, a new paradigm for diagnostic testing is required. This new paradigm must combine patient demographics, patient symptoms, and noninvasive testing to prioritize colonoscopy for patients at a high risk of cancer and avoid unnecessary colonoscopies for those with a low cancer risk. Several noninvasive tests are now available, which could allow us to triage referrals into high- and low-risk categories and to reprioritize those patients already on a waiting list for colonoscopy. These tests include the fecal immunochemical test (FIT), the multitarget stool DNA test (FIT-DNA), fecal calprotectin, the methylated Septin9 (mSEPT9) blood test, computed tomography-guided colonoscopy (CTC), and colon capsule endoscopy (CCE).
The FIT has superseded the use of the fecal occult blood test. This is because the FIT is specific for human hemoglobin, is easier for patients to perform, has greater sensitivity, and has fewer false-positive and false-negative rates compared with the fecal occult blood test.7 , 8 In a study of 1000 symptomatic patients who had also undergone a FIT with a threshold of ≥ 10 μg of hemoglobin/1 g, 742 patients (74%) had had negative FIT results.9 A total of 48 CRC cases and 7 FIT-negative cancer cases were diagnosed. Of the latter, 1 was a lymphoma and 6 were cecal adenocarcinoma, with 5 of the latter 6 patients anemic. The specificity for FIT in that study was 86.9% (95% confidence interval 96%) and the negative predictive value (NPV) was 99.05%.9 The positive predictive value (PPV) for iron deficiency anemia (IDA) was 34% compared with 18% for “other symptoms.” Although a negative FIT result has a NPV of 99.05%, it is not as sensitive for right-sided CRCs. However, when combined with the presence of iron deficiency anemia, only 1 case of lymphoma and 1 case of CRC would have been missed.9
In another UK study, 612 patients who had been urgently referred because of symptoms of suspected CRC via the 2-week-wait referral pathway had also undergone a FIT with a cutoff of ≥ 10 μg of hemoglobin/1 g.10 Of these 612 patients, 477 (77.9%) had had negative FIT results, with 5 false-negative FIT results. In this cohort, 35 cases of cancer and 3 cases of high-grade dysplasia had been diagnosed. Thus, the sensitivity, specificity, NPV, and PPV of FIT for the diagnosis of CRC was 86.84%, 82.23%, 98.95%, and 22.96%, respectively.10 Of the 5 patients with negative FIT results for cancer, 60% had had anemia, 40% had had a mass, and 80% had lost weight. Therefore, combining these “safety-net” high-risk symptoms with FIT would have detected virtually all the cases of cancer and still significantly reduced the colonoscopy workload. Therefore, for patients worried about their symptoms in the COVID-19 era, during which services have been severely limited and doctors have been unable to provide a timely review, a negative FIT result with “safety-netting” could provide considerable reassurance.
For patients with rectal bleeding, the FIT is not appropriate, and the mSEPT9 test might be a better option to rule out CRC with reasonable accuracy. In a recent systematic review and meta-analysis of the diagnostic test accuracy of the mSEPT9 test for the detection of CRC based on 19 studies, the pooled estimates for mSEPT9 to detect CRC showed a sensitivity of 69%, specificity of 92%, PPV of 2.6%, and NPV of 99.9% in an average-risk population (0.3% CRC prevalence) and a PPV of 9.5% and an NPV of 99.6% in a high-risk population (1.2% CRC prevalence).11 , 12 Overall, the mSEPT9 test has had lower sensitivity and specificity compared with the FIT. Therefore, from a purely performance-based comparison, mSEPT9 should not be considered a replacement test for FIT but complementary. Furthermore, it is also more expensive at ∼£100 (∼US$125) compared with a cost for FIT of £6 (∼US$8). In addition, the mSEPT9 test is not widely available outside of the United States, although it has been approved by the US Preventative Services Task Force as a first-line CRC screening test.13
The FIT, FIT-DNA, and mSEPT9 test alone are not sensitive enough to exclude the presence of precancerous colonic lesions such as advanced adenoma or polyps because of the lower sensitivity, which has ranged from 5% to 42%.10 , 11 , 14 However, patients with potential symptoms of CRC could also be offered the FIT combined with the FIT-DNA if they do not have rectal bleeding. This test combines the FIT with testing for altered DNA biomarkers in cells that have been shed into the stool. The FIT-DNA performance has shown increased sensitivity for detecting advanced precancerous lesions compared with the use of FIT alone when screening an average-risk CRC population. Its specificity is lower than that of FIT alone. Thus, it will have a greater number of false-positive results and a greater likelihood of requiring follow-up colonoscopy and an associated adverse event per screening test.13 , 15 Furthermore, the FIT-DNA is not readily available outside the United States.
CTC is also a good diagnostic test in the first line of investigations for symptomatic colorectal patients. In a study from the United Kingdom, CTC was offered to all patients aged > 60 years with a change in bowel habits and IDA.16 During a 12-month study period, 1792 straight-to-test CTCs were performed. The CRC detection rate was 4.9%, and the polyp detection rate was 13.5%. The investigators concluded that the results were comparable to those from colonoscopy in terms of diagnostic accuracy and similar to those from CTC in reported multicenter trials.16 CTC also has the advantage of detecting extracolonic pathologic features.
In the future, CCE might also play a role in evaluating patients. The SCOTCAP (Scottish Capsule Project) trial, which had evaluated 455 symptomatic and surveillance patients with CCE ended in March 2020, and the results are pending.17 , 18
No single test, including colonoscopy, has shown 100% sensitivity or specificity for diagnosing or excluding CRC. In a retrospective, single-center study of postcolonoscopy CRCs diagnosed within 6 to 36 months, the ratio of interval cancers to all new CRCs from 2006 to 2015 was 6% to 11.5%.19 The reasons included (1) a missed diagnosis; (2) incomplete resection of advanced adenomas; and (3) neoplasms that grew more quickly than the average sporadic cases of CRC.20, 21, 22 Missed diagnoses are likely in the first 36 months after colonoscopy on the basis that the interval is too soon for a de novo cancer to have arisen.23 In the study by Strum and Boland,19 a “difficult examination” was often associated with the occurrence of interval cancer and might be a risk factor for this problem. In a recent review of the quality of CRC screening with colonoscopy, interval CRC most commonly occurred within 24 months among patients who had had only 1 to 2 small adenomas found on the initial colonoscopy. Interval CRC appeared to arise from flat, large (1-cm) adenomas or serrated polyps in the right side of the colon.24 Therefore, if the bowel preparation was not adequate, colonoscopy should be repeated.24 Also, increasing the withdrawal time to 8 to 10 minutes will increase the adenoma detection rate.24
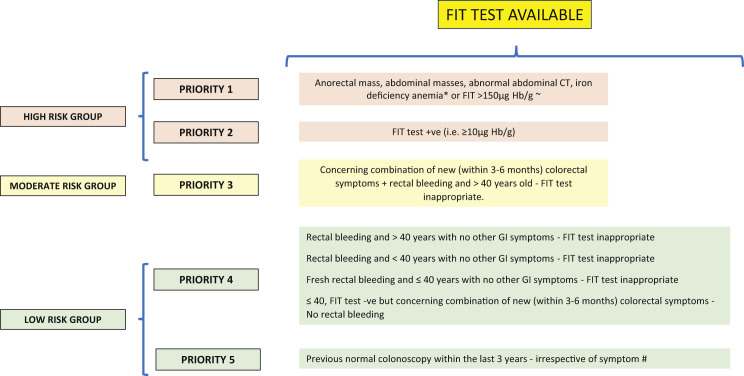
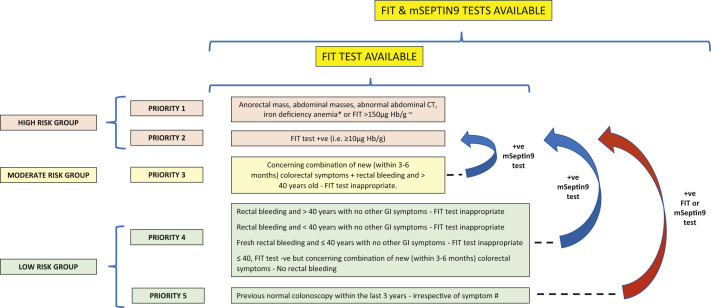
Because it has been generally accepted that CRCs will typically grow for many months and years before presentation, one might expect that short delays in diagnosis and surgery would not have negative effects on patient outcomes. To the best of our knowledge, no reported studies have analyzed the delay in diagnosis on survival outcomes. However, studies that have analyzed the effect of a delay in treatment from the diagnosis of CRC have shown that a deferral of surgery for ≤ 12 weeks will be unlikely to have any effect on the survival outcomes of patients with CRC.25, 26, 27 With the markedly reduced capacity of our endoscopy services during the COVID-19 pandemic, we need to consider radical solutions for the use of colonoscopy as the first diagnostic test for patients with colorectal symptoms to identify patients with CRC early and prevent poor survival outcomes. Therefore, for patients already included in diagnostic pathways and waiting to undergo colonoscopy, it will be essential to triage these patients into different risk categories for cancer. Both the FIT and mSEPT9 test have proven reasonably good accuracy when combined with “safety netting” for excluding CRC and could be used to stratify patients into high- and low-risk groups, with the high-risk group prioritized for diagnostic colonoscopy.9 , 13 , 28 Thus, the first-line investigation should be the FIT because of its greater sensitivity and specificity and lower cost compared with the mSEPT9 test, which should be reserved for those patients with rectal bleeding for whom the FIT is not appropriate. Both the FIT and the mSEPT9 test have a high NPV and are, therefore, good tests to use to “rule out” cancer. However, they do not have 100% sensitivity, and a few cancer cases could be missed (ie, false-negative results). Thus, it will be important to have some “safety netting” in place using patient symptoms and blood test results (eg, weight loss, a mass, IDA). Figures 1 and 2 both show a method of prioritization of symptomatic patients for colonoscopy using several factors during the COVID-19 pandemic. The ideal and most cost-effective scenario will be the availability of FIT and mSEPT9 blood test results to assist in risk stratification and prioritization of patients. These tests are relatively inexpensive, and their results could help manage endoscopy demands until the burden of the COVID-19 pandemic has subsided and endoscopy services can return to normal, which is not likely to be for many months, if not years, unless an effective vaccine becomes available. During the COVID-19 endoscopic recovery period, the use of noninvasive tests such as the FIT and mSEPT9 test could reduce the need for colonoscopy by ≤ 80%. This has huge cost-saving implications. Furthermore, the use of colonoscopy will also be likely to result in a greater yield of CRC if the results from noninvasive tests have been used to approve referrals compared with the 1.7% rate of CRC in the previously cited study by Ismail et al,6 which had stratified patients using clinical symptoms only. However, rationing one’s service to make it more efficient should not be at the sacrifice of the small numbers of missed CRCs, and efforts are required to minimize this. Our recommendation would be to use FIT, with or without the mSEPT9 test, and “safety netting” to risk-stratify patients for the presence or absence of significant pathologic features and rule out cancer because the latter patients would require no further investigations. Those patients with positive FIT results or with “high-risk” symptoms, such as weight loss, an abdominal mass, or IDA, should undergo urgent investigation, irrespective of their FIT or mSEPT9 test results. Such a protocol would avoid missing the small number of cancers from the use of the noninvasive tests alone (ie, false-negative results). A later clinical review of patients with a moderate or low risk of CRC should be performed after FIT negative results (if appropriate) or mSEPT9 negative results. If symptoms persist despite an appropriate initial clinical working diagnosis and treatment, and a clinical concern for CRC exists, colonoscopy could be performed at that stage. If access to colonoscopy or CTC is still limited, another option would be to repeat the FIT, if appropriate, or the mSEPT9 test. Although not supported by clinical investigatory evidence, a second negative test result is likely to be more reassuring than just 1 test result. Nevertheless, the importance of assessing each clinical case based on its own merits and not relying on just the FIT or mSEPT9 test results cannot be overstated.
Figure 1.
Method of Prioritization of Symptomatic Patients for Colonoscopy During Coronavirus Disease 2019 Pandemic Using Several Factors and for Situations in Which Only the Fecal Immunochemical Test (FIT) Is Available
∗Patients with iron deficiency anemia will also require gastroscopy. ∼Based on unpublished data from the NICE FIT study (2020) of 9822 patients; positive predictive value for FIT for 2-week-wait patients at a threshold of > 150 μg Hb/1 g was 37%. #We Would Recommend FIT if No History of Rectal Bleeding, Otherwise the methylated Septin9 (mSEPT9) Test Should Be Used. +ve, positive; -ve, negative; CT, computed tomography; GI, gastrointestinal; Hb, hemoglobin.
Figure 2.
Method of Prioritization of Symptomatic Patients for Colonoscopy During Coronavirus Disease 2019 Pandemic Using Several Factors and for Situations in Which Both the Fecal Immunochemical Test (FIT) and the methylated Septin9 (mSEPT9) Test Is Available
∗Patients with iron deficiency anemia will also require gastroscopy. ∼Based on unpublished data from the NICE FIT study (2020) of 9822 patients; positive predictive value for FIT for 2-week-wait patients at a threshold of > 150 μg Hb/1 g was 37%. #We Would Recommend FIT if No History of Rectal Bleeding, Otherwise the mSeptin9 Test Should Be Used. +ve, positive; -ve, negative; CT, computed tomography; GI, gastrointestinal; Hb, hemoglobin.
In conclusion, the COVID-19 pandemic has severely restricted our ability to provide timely diagnostic endoscopic tests to rule out CRC in symptomatic patients. Because of the infection control measures, the endoscopy waiting lists could still be incapacitated for several months because of COVID-19. In the present report, we have discussed strategic opportunities to drastically reimagine endoscopy services using noninvasive tests and a “safety netting” approach to prioritize the use of colonoscopy. Ultimately, what could transpire from an overdue rethinking of the use of endoscopy services could be more efficient use in the future of our colonoscopy services after COVID-19.
Disclosure
The authors declare that they have no competing interests.
Acknowledgments
The authors thank Alison Walker for English language proof reading.
References
- 1.39-All-cancers-fact-sheet.pdf. https://gco.iarc.fr/today/data/factsheets/cancers/39-All-cancers-fact-sheet.pdf Available at: Accessed May 25, 2020.
- 2.Sud A., Jones M.E., Broggio J. Collateral damage: the impact on cancer outcomes of the COVID-19 pandemic. Ann Oncol. 2020;31:1065–1074. doi: 10.1016/j.annonc.2020.05.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Chiu P.W.Y., Ng S.C., Inoue H. Practice of endoscopy during COVID-19 pandemic: position statements of the Asian Pacific Society for Digestive Endoscopy (APSDE-COVID statements) Gut. 2020;69:991–996. doi: 10.1136/gutjnl-2020-321185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Hennessy B., Vicari J., Bernstein B. Guidance for resuming GI endoscopy and practice operations after the COVID-19 pandemic. Gastrointest Endosc. 2020;92:743–747.e1. doi: 10.1016/j.gie.2020.05.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.The British Society of Gastroenterology BSG Guidance on Recommencing GI Endoscopy in the Deceleration & Early Recovery Phases of the COVID-19 Pandemic. Published April 30, 2020. https://www.bsg.org.uk/covid-19-advice/bsg-guidance-on-recommencing-gi-endoscopy-in-the-deceleration-early-recovery-phases-of-the-covid-19-pandemic/ Available at: Accessed May 23, 2020. [DOI] [PMC free article] [PubMed]
- 6.Ismail M.S., Aoko O., Sihag S. Lower gastrointestinal symptoms and symptoms-based triaging systems are poor predictors of clinical significant disease on colonoscopy. BMJ Open Gastroenterol. 2020;7:e000221. doi: 10.1136/bmjgast-2018-000221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Chambers J.A., Callander A.S., Grangeret R., O’Carroll R.E. Attitudes towards the faecal occult blood test (FOBT) versus the faecal immunochemical test (FIT) for colorectal cancer screening: perceived ease of completion and disgust. BMC Cancer. 2016;16:96. doi: 10.1186/s12885-016-2133-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Mousavinezhad M., Majdzadeh R., Akbari Sari A., Delavari A., Mohtasham F. The effectiveness of FOBT vs. FIT: a meta-analysis on colorectal cancer screening test. Med J Islam Repub Iran. 2016;30:366. [PMC free article] [PubMed] [Google Scholar]
- 9.Cunin L, Khan AA, Ibrahim M, Lango A, Klimovskij M, Harshen R. FIT negative cancers: a right-sided problem? Implications for screening and whether iron deficiency anaemia has a role to play [e-pub ahead of print]. Surgeon. https://doi.org/10.1016/j.surge.2020.02.003, accessed September 5, 2020. [DOI] [PubMed]
- 10.Farrugia A., Widlak M., Evans C., Smith S.C., Arasaradnam R. Faecal immunochemical testing (FIT) in symptomatic patients: what are we missing? Frontline Gastroenterol. 2019;11:28–33. doi: 10.1136/flgastro-2018-101174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hariharan R., Jenkins M. Utility of the methylated SEPT9 test for the early detection of colorectal cancer: a systematic review and meta-analysis of diagnostic test accuracy. BMJ Open Gastroenterol. 2020;7:e000355. doi: 10.1136/bmjgast-2019-000355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Potter N.T., Hurban P., White M.N. Validation of a real-time PCR-based qualitative assay for the detection of methylated SEPT9 DNA in human plasma. Clin Chem. 2014;60:1183–1191. doi: 10.1373/clinchem.2013.221044. [DOI] [PubMed] [Google Scholar]
- 13.Bosch L.J.W., Melotte V., Mongera S. Multitarget stool DNA test performance in an average-risk colorectal cancer screening population. Am J Gastroenterol. 2019;114:1909–1918. doi: 10.14309/ajg.0000000000000445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Carethers J.M. Fecal DNA testing for colorectal cancer screening. Annu Rev Med. 2020;71:59–69. doi: 10.1146/annurev-med-103018-123125. [DOI] [PubMed] [Google Scholar]
- 15.Imperiale T.F., Ransohoff D.F., Itzkowitz S.H. Multitarget stool DNA testing for colorectal-cancer screening. N Engl J Med. 2014;370:1287–1297. doi: 10.1056/NEJMoa1311194. [DOI] [PubMed] [Google Scholar]
- 16.Stephenson J.A., Pancholi J., Ivan C.V. Straight-to-test faecal tagging CT colonography for exclusion of colon cancer in symptomatic patients under the English 2-week-wait cancer investigation pathway: a service review. Clin Radiol. 2018;73:836.e1–836.e7. doi: 10.1016/j.crad.2018.05.013. [DOI] [PubMed] [Google Scholar]
- 17.MacLeod C., Wilson P., Watson A. Colon capsule endoscopy: an innovative method for detecting colorectal pathology during the COVID-19 pandemic? Colorectal Dis. 2020;22:621–624. doi: 10.1111/codi.15134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Pecere S., Senore C., Hassan C. Accuracy of colon capsule endoscopy for advanced neoplasia. Gastrointest Endosc. 2020;91:406–414.e1. doi: 10.1016/j.gie.2019.09.041. [DOI] [PubMed] [Google Scholar]
- 19.Strum WB, Boland CR. Interval colorectal cancer 2006-2015: novel observations [e-pub ahead of print]. Dig Dis Scihttps://doi.org/10.1007/s10620-020-06242-1, accessed September 5, 2020. [DOI] [PubMed]
- 20.Sawhney M.S., Farrar W.D., Gudiseva S. Microsatellite instability in interval colon cancers. Gastroenterology. 2006;131:1700–1705. doi: 10.1053/j.gastro.2006.10.022. [DOI] [PubMed] [Google Scholar]
- 21.Nishihara R., Wu K., Lochhead P. Long-term colorectal-cancer incidence and mortality after lower endoscopy. N Engl J Med. 2013;369:1095–1105. doi: 10.1056/NEJMoa1301969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stoffel E.M., Erichsen R., Frøslev T. Clinical and molecular characteristics of post-colonoscopy colorectal cancer: a population-based study. Gastroenterology. 2016;151:870–878.e3. doi: 10.1053/j.gastro.2016.07.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Fuccio L., Rex D., Ponchon T. New and recurrent colorectal cancers after resection: a systematic review and meta-analysis of endoscopic surveillance studies. Gastroenterology. 2019;156:1309–1323.e3. doi: 10.1053/j.gastro.2018.12.006. [DOI] [PubMed] [Google Scholar]
- 24.Schoenfeld P. Quality in colorectal cancer screening with colonoscopy. Gastrointest Endosc Clin N Am. 2020;30:541–551. doi: 10.1016/j.giec.2020.02.014. [DOI] [PubMed] [Google Scholar]
- 25.Bagaria S.P., Heckman M.G., Diehl N.N., Parker A., Wasif N. Delay to colectomy and survival for patients diagnosed with colon cancer. J Invest Surg. 2019;32:350–357. doi: 10.1080/08941939.2017.1421732. [DOI] [PubMed] [Google Scholar]
- 26.Hangaard Hansen C., Gögenur M., Tvilling Madsen M., Gögenur I. The effect of time from diagnosis to surgery on oncological outcomes in patients undergoing surgery for colon cancer: a systematic review. Eur J Surg Oncol. 2018;44:1479–1485. doi: 10.1016/j.ejso.2018.06.015. [DOI] [PubMed] [Google Scholar]
- 27.Turaga KK, Girotra S. Are we harming cancer patients by delaying their cancer surgery during the COVID-19 pandemic? [e-pub ahead of print]. Ann Surghttps://doi.org/10.1097/SLA.0000000000003967, accessed September 5, 2020. [DOI] [PMC free article] [PubMed]
- 28.Lee J.K., Liles E.G., Bent S., Levin T.R., Corley D.A. Accuracy of fecal immunochemical tests for colorectal cancer: systematic review and meta-analysis. Ann Intern Med. 2014;160:171. doi: 10.7326/M13-1484. [DOI] [PMC free article] [PubMed] [Google Scholar]