Abstract
Nine independent Gram-negative bacterial strains were isolated from rectal swabs or stool samples of immunocompromised patients from two different wards of a university hospital. All isolates were phylogenetically analysed based on their 16S rRNA gene sequence, housekeeping gene recN, multilocus sequence analysis of concatenated partial fusA, leuS, pyrG and rpoB sequences, and by whole genome sequencing data. The analysed strains of the new species cluster together and form a separate branch with Citrobacter werkmanii NBRC105721T as the most closely related species. An average nucleotide identity value of 95.9–96% and computation of digital DNA–DNA hybridization values separate the new species from all other type strains of the genus Citrobacter . Biochemical characteristics further delimit the isolates from closely related Citrobacter type strains. As a result of the described data, a new Citrobacter species is introduced, for which the name Citrobacter cronae sp. nov. is proposed. The type strain is Tue2-1T with a G+C DNA content of 52.2 mol%.
Isolation and ecology
Citrobacter species are Gram-negative micro-organisms frequently encountered in the environment, but also from the intestinal tract of humans [1–3]. They were also reported as opportunistic pathogens causing wound infections, abscesses, severe forms of meningitis, endocarditis or bloodstream infections [4–8].
In clinical microbiology laboratories, Citrobacter species represent up to 6 % of all isolated Enterobacteriaceae from clinical specimens [4]. As they can have chromosomal AmpC β-lactamases [9] as well as plasmid encoded carbapenemases [10], many antibiotics are ineffective increasing the intricacy of treatment [11]. To date, 15 Citrobacter species are published in the literature [12]. In the present study, nine independent clinical isolates (Tue2-1T, Tue2-3 and Tue2-5–Tue2-11) were investigated that originated from nine patients of two different wards with underlying haematological conditions. Isolates were collected and stored from rectal swabs or stool samples over a 3-year period (2012–2015), but even more isolates were obtained since 2016. Three of the strains (Tue2-1T, Tue2-3, Tue2-5) harbouring metallo-β-lactamase (MBL) enzymes were already characterized by comparative genomics using next generation sequencing, but could not be unambiguously identified to the species level by standard routine methods [13]. We first considered that the studied isolates belong to the species Citrobacter werkmanii . However, experimental evidence suggested that the three isolates belong to a new Citrobacter species, which will be characterized here. In order to gain further evidence for our new hypothesis we additionally characterized and sequenced six more isolates (Tue2-6–Tue2-11).
Methods
MALDI-TOF AXIMA system assurance (bioMérieux; Saramis database version 4.09) and the Microflex LT instrument (Bruker Daltonics; MBT IVD Library.5627) were performed on all isolates, but failed to unambiguously identify the strains. Additionally, biochemically based identification using the API 20 E System (bioMérieux; apiweb) and the vitek GN ID card (bioMérieux) was applied. Bacterial DNA was extracted from cultures grown on Columbia agar with 5 % sheep blood (Becton Dickinson) using the Genomic-tip 100/G system (Qiagen) following the manufacturer’s instructions. For whole genome sequencing, libraries were prepared using the TruSeq DNA LT Sample Prep Kit (Illumina) with 24 different barcodes using standard protocols as described previously [13, 14]. Barcoded libraries were analysed by the Agilent 2100 Bioanalyzer (Agilent Technologies) or QIAxcel Advanced Instrument (Qiagen) and quantified by Real-Time (RT)-PCR. Normalized libraries were pooled and sequenced with v3 chemistry (2×300 bp) or with v2 chemistry (2×250 bp) on the MiSeq platform (Illumina). Assembly of genome sequences was performed using SPAdes (version 3.7.0) [15] with default settings.
For phylogenetic analysis of the isolates, publically available whole genome sequencing (WGS) data from Citrobacter type strains were included in the analysis (Table S1). ProgressiveMauve (version 2.3.1) [16] was run to conduct a full alignment of 23 genomes using default settings and prophage regions were investigated and excluded using phaster (phaster.ca) [17]. Maximum-likelihood phylogenetic trees of 23 whole genome-sequences were reconstructed by applying IQ-Tree with 1000 bootstrap replicates. Alignments of 16S rRNA gene sequences downloaded from EZ-Taxon [18 ], concatenated partial fusA (protein synthesis elongation factor-G), leuS (leucine tRNA synthetase), pyrG (CTP synthetase) and rpoB (β-subunit of RNA polymerase) [19] as well as recN sequences (DNA repair) [2, 20] used for recent description of new Citrobacter species extracted from available WGS data were done by clustal_w (BioEdit version 7.2.5) followed by phylogenetic treeing with RAxML and the GTR model in conjunction with GAMMA rates [21]. Trees were visualized using FigTree (version 1.4.3). The average nucleotide identity (ANI) was assessed by JSpecies (version 1.2) [22, 23] based on blast +2.2.29 (ANIb). The Genome-to-Genome Distance Calculator (GGDC 2.1) using the recommended Formula 2 was applied for in silico genome comparison and computation of digital DNA–DNA hybridization (dDDH) values [24]. Employing the online multi-locus sequence typing (MLST) service of the Center for Genomic Epidemiology (https://cge.cbs.dtu.dk/services/MLST/; version 2.0), MLST sequence types were obtained from assembled sequences based on the MLST scheme for C. freundii [25].
Phylogeny
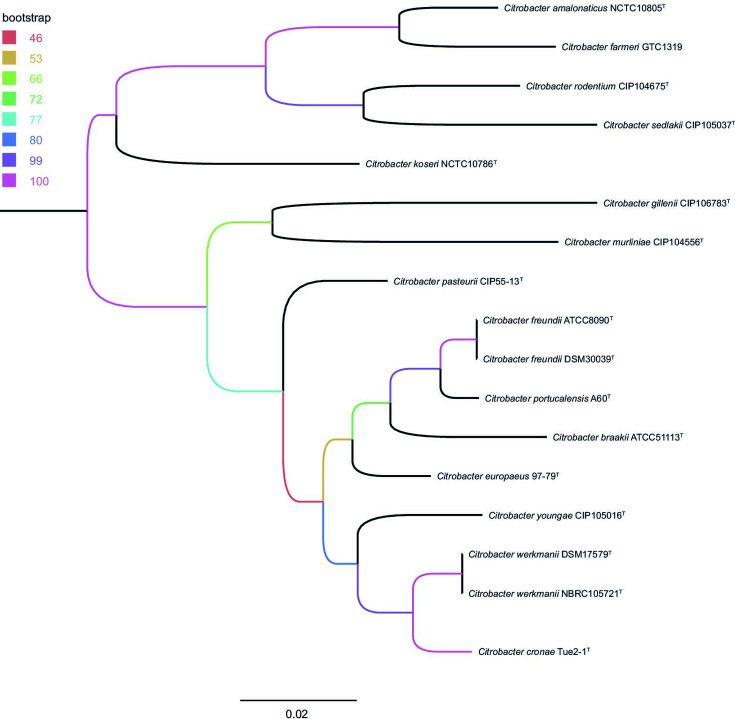
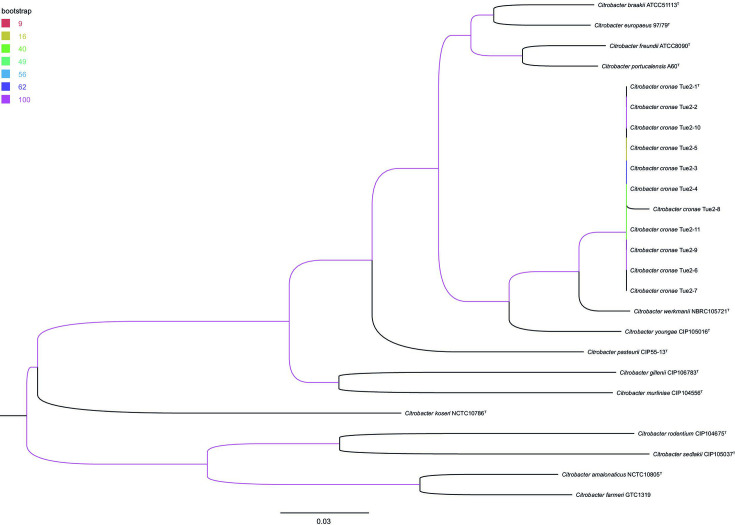
MALDI-TOF using Bruker and bioMérieux systems as well as analysing the biochemical characteristics of these strains with API 20E and VITEK2 system (bioMérieux) did not allow for unambiguous identification of all nine study isolates on the species level. Dissecting the MLST type showed the same result for all nine strains isolated over the 3-year collection period. Their phylogenetic relationship to other Citrobacter type strains was assessed by analysing the 16S rRNA, the recN gene, the concatenated partial fusA, leuS, pyrG and rpoB genes as well as by WGS. 16S rRNA gene-based phylogeny represented a distinct branch of all isolates of the new Citrobacter species clustering together in group I including the formerly published species Citrobacter freundii , Citrobacter youngae , Citrobacter braakii , Citrobacter werkmanii , Citrobacter gillenii and Citrobacter murliniae [26] as well as Citrobacter pasteurii [19] and the recently described C. europaeus [2] and C. portucalensis [20] (Fig. S1, available in the online version of this article). Regarding the limited resolution of 16S rRNA genes in discrimination of Citrobacter species [19, 27], the closest similarity in 16S rRNA gene comparison was found to C. freundii (99.73 %). Phylogenetic analysis based on the recN gene (Fig. S2) as well as MLSA of concatenated partial fusA, leuS, pyrG and rpoB (Fig. 1) extracted from WGS data of type strains confirmed 16S rRNA gene-based clustering of all nine isolates in a separate branch The maximum-likelihood tree generated using WGS data enabled further distinction of the new Citrobacter species from other type strains of the genus including the most closely related species C. werkmanii strain NBRC 105721T (Fig. 2).
Fig. 1.
Multilocus sequence analysis of concatenated partial fusA, leuS, pyrG and rpoB gene sequences extracted from whole genome data of the study isolates (Citrobacter cronae Tue2-1T, Tue2-3, Tue2-5 – Tue2-11) and available genome data of Citrobacter type strains. The scale bar represents the expected number of changes per site. Bootstrap values [%] are colour-coded for all nodes (based on 1000 replicates). The tree was rooted at midpoint.
Fig. 2.
Maximum-likelihood phylogeny based on whole genome sequencing data of the study isolates (Citrobacter cronae Tue2-1T, Tue2-3, Tue2-5 – Tue2-11) and available genome data of Citrobacter type strains. The scale bar represents the expected number of changes per site. Bootstrap values (%) are colour-coded for all nodes (based on 1000 replicates). The tree was rooted at midpoint.
MALDI-TOF using Bruker and bioMérieux systems as well as analysing the biochemical characteristics of these strains with API 20E and VITEK2 system (bioMérieux) did not allow for unambiguous identification of all nine study isolates on the species level. Dissecting the MLST type showed the same result for all nine strains isolated over the 3-year collection period. Their phylogenetic relationship to other Citrobacter type strains was assessed by analysing the 16S rRNA, the recN gene, the concatenated partial fusA, leuS, pyrG and rpoB genes as well as by WGS. 16S rRNA gene-based phylogeny represented a distinct branch of all isolates of the new Citrobacter species clustering together in group I including the formerly published species Citrobacter freundii , Citrobacter youngae , Citrobacter braakii , Citrobacter werkmanii , Citrobacter gillenii and Citrobacter murliniae [26] as well as the recently described C. europaeus [2] and C. portucalensis [20] (Fig. S1, available in the online version of this article). Regarding the limited resolution of 16S rRNA genes in discrimination of Citrobacter species [19, 27], the closest similarity in 16S rRNA gene comparison was found to C. freundii (99.73 %). Phylogenetic analysis based on the recN gene (Fig. S2) as well as MLSA of concatenated partial fusA, leuS, pyrG and rpoB (Fig. 1) extracted from WGS data of type strains confirmed 16S rRNA gene-based clustering of all nine isolates in a separate branch The maximum-likelihood tree generated using WGS data enabled further distinction of the new Citrobacter species from other type strains of the genus including the most closely related species C. werkmanii strain NBRC 105721T (Fig. 2).
Genome features
Species definition can also be based on ANI value [22, 28]. Therefore the new Citrobacter isolates were compared to all Citrobacter type strains with available WGS data (Table S2b). The closest relationship of the new Citrobacter species was found with C. werkmanii NBRC105721T (95.92 %), slightly below the proposed cut-off value of 96 % for the assignment of a new species [29]. In comparison, ANI values between all nine analysed isolates (Tue2-1, Tue2-3, Tue2-5– 2–11) were above 99.5 %, demonstrating their close relationship (Table S2a).
As described recently, dDDH can be used for delineation of a new bacterial species using WGS data [29]. As illustrated in Table S2b, dDDH values were calculated for all available Citrobacter type strains in relation to Tue2-1T. The lowest intergenomic distance of our nine analysed Citrobacter species isolates was found to C. werkmanii NBRC105721T with a dDDH value of 70 %, exactly the cutoff proposed for bacterial species delineation [22, 28]. The dDDH values between any pair of the new Citrobacter species isolates were above 99.1 %.
Physiology
Biochemical characteristics were analysed by the API 20E and VITEK2 systems and results are listed in Table 1 for all nine study isolates and closely related type strains of the genus Citrobacter [2, 19, 20, 30]. The data show that all nine C. cronae isolates are able to catabolize amygdalin distinguishing the novel species from all other closely related Citrobacter species tested. In addition all C. cronae isolates are able to catabolize cellobiose, which is not the case for C. werkmanii DSM17579T. No enzymatic activity for phosphatase could be found for C. cronae, whereas C. werkmanii DSM17579T was positive for phosphatase. Moreover due to some variable characteristics not found to be diverse in different C. werkmanii isolates [19, 30], C. cronae can be separated biochemically. Taken together, phylogenetic analysis based on 16S rRNA gene, recN, the concatenated partial genes fusA, leuS, pyrG and rpoB and WGS data, calculation of genome relatedness by ANI and dDDH as well as biochemical properties classifies Citrobacter Tue2-1T, 2-3 and 2-5–2-11 as representing a new species within the genus Citrobacter for which we propose the name Citrobacter cronae sp.nov., with Tue2-1T as type strain.
Table 1.
Biochemical characteristics of all Citrobacter cronae study isolates and closely related Citrobacter type strains
Strains: 1, Tue2-1T–Tue2_11; 2, Citrobacter werkmanii DSM17579T; 3, Citrobacter werkmanii ; 4, Citrobacter youngae ; 5, Citrobacter pasteurii ; 6, Citrobacter portucalensis A60T; 7, Citrobacter freundii ; 8, Citrobacter europaeus 97/79T; 8, Citrobacter braakii. +, Positive result; –, negative result; v, variable result; na, not available. For variable reactions of Citrobacter cronae isolates (n=9), values in parentheses represent percentage of positive strains.
|
Characteristics |
1 |
2 |
3 |
4 |
5 |
6 |
7 |
8 |
9 |
|---|---|---|---|---|---|---|---|---|---|
|
Amygdalin |
+ |
– |
– |
– |
– |
– |
– |
– |
– |
|
Cellobiose |
+ |
– |
v |
v |
+ |
+ |
v |
+ |
v |
|
Catalase |
+ |
+ |
v |
v |
– |
– |
+ |
+ |
+ |
|
Phosphatase |
– |
+ |
na |
na |
+ |
na |
na |
na |
na |
|
α-Glucosidase |
– |
– |
+ |
v |
– |
na |
v |
na |
v |
|
Indole |
– |
– |
– |
– |
– |
v |
– |
– |
v |
|
Melibiose |
– |
– |
– |
v |
– |
+ |
+ |
+ |
v |
|
β-Glucosidase |
v (66.6) |
– |
– |
v |
+ |
na |
v |
na |
– |
|
Adonitol |
v (44.4) |
– |
– |
– |
– |
– |
– |
– |
– |
|
H2S |
v (44.4) |
+ |
+ |
+ |
+ |
+ |
+ |
+ |
v |
|
Malonate |
v (88.8) |
+ |
+ |
– |
– |
na |
– |
na |
+ |
|
Ornithine |
v (44.4) |
– |
– |
v |
– |
na |
– |
na |
v |
|
Sucrose |
v (77.7) |
– |
– |
– |
v |
+ |
+ |
– |
– |
|
5-Ketogluconate |
v (66.6) |
– |
– |
+ |
+ |
+ |
+ |
+ |
+ |
|
Data obtained from |
This study |
This study |
[19, 30] |
[19, 30] |
[19] |
[20] |
[19, 30] |
[2] |
[19, 30] |
Description of Citrobacter cronae sp. nov.
Citrobacter cronae [cro'nae. N.L. gen. n. cronae, pertaining to he crona (the landmark building of the university hospital; acronym for Surgery, Radiology, Orthopedics, Neurology, Anesthesiology) clinics, Tuebingen, Germany].
Citrobacter cronae is a Gram-stain-negative, oxidase-negative, catalase-positive (delayed), facultative anaerobic, rod-shaped bacterium. It is able to ferment the following carbohydrates and derivatives: d-mannitol, sorbitol, d-mannose, cellobiose, trehalose and amygdalin. The strains cannot utilize aesculin, inositol and melibiose and are negative for acetoin- and indole production. Variable reactions are observed for fermentation of d-glucose l-rhamnose, arabinose, maltose, malonate, d-adonitol, citrate, potassium 5-keto-gluconate, sucrose and d-tagatose. Citrobacter cronae is variable for urease and production of H2S.
Semi-quantitative analysis of enzymatic activities of all strains demonstrate positive reactions for l-pyrrolidonyl-arylamidase and β-galactosidase, but negative reactions for α-glucosidase, phosphatase, lipase, lysine decarboxylase, tryptophan deaminase, gelatinase, α-galactosidase, Glu-Gly-Arg-arylamidase, β-N-acetyl-galactosaminidase, β-xylosidase, β-alanin-arylamidase-pNA and β-glucoronidase. Variable enzymatic reactions are seen for N-acetyl-β-glucosaminidase, ornithine decarboxylase, arginine dihydrolase, tyrosin arylamidase and β-glucosidase.
The type strain of Citrobacter cronae is Tue2-1T, which was isolated from a rectal swab of a patient hospitalized at University Hospital Tuebingen, Tuebingen, Germany. The G+C DNA content of the type strain is 52.2 mol%. The accession numbers for the WGS and 16S rRNA gene of strain Tue2-1T are VOSQ00000000 and MN548424, respectively. The culture certificate accession numbers are CCUG 73860T from the CCUG, Göteborg, Sweden, and DSM 110040 from the DSMZ, Braunschweig, Germany.
Supplementary Data
Funding information
This work was partly funded by the TÜFF program, Medical Faculty, University Tuebingen (2243-0-0 to S. P.).
Acknowledgements
We thank Nadine Hoffmann, Baris Bader and the team of diagnostic technicians for supporting the project with perfect technical assistance. Also thanks to Sophia Wolf and Jan Liese for assisting with BioNumerics.
Conflicts of interest
The authors declare that there are no conflicts of interest.
Ethical statement
The study was conducted in accordance with the local ethics committee from the medical faculty of the university clinics at Tübingen, Germany (407/2013R).
Footnotes
Abbreviations: ANI, average nucleotide identity; dDDH, digital DNA-DNA hybridization; MLSA, multilocus sequence analysis; MLST, multilocus sequence typing.
The GenBank accession numbers of the whole genome sequencing data for Citrobacter cronae Tue2-1T are VOSQ00000000 and MN548424 for 16S rRNA sequence, respectively.
Two supplementary tables and two supplementary figures are available with the online version of this article.
References
- 1.Arens S, Verbist L. Differentiation and susceptibility of Citrobacter isolates from patients in a university hospital. Clin Microbiol Infect. 1997;3:53–57. doi: 10.1111/j.1469-0691.1997.tb00251.x. [DOI] [PubMed] [Google Scholar]
- 2.Ribeiro TG, Clermont D, Branquinho R, Machado E, Peixe L, et al. Citrobacter europaeus sp. nov., isolated from water and human faecal samples. Int J Syst Evol Microbiol. 2017;67:170–173. doi: 10.1099/ijsem.0.001606. [DOI] [PubMed] [Google Scholar]
- 3.Borenshtein D, Schauer DB. The Genus Citrobacter. In: Dworkin M, Falkow S, Rosenberg E, Schleifer K-H, Stackebrandt E, editors. The Prokaryotes: Volume 6: Proteobacteria: Gamma Subclass. New York, NY: Springer New York; 2006. pp. 90–98. [Google Scholar]
- 4.Lipsky BA, Hook EW, Smith AA, Plorde JJ. Citrobacter infections in humans: experience at the Seattle Veterans administration medical center and a review of the literature. Rev Infect Dis. 1980;2:746–760. doi: 10.1093/clinids/2.5.746. [DOI] [PubMed] [Google Scholar]
- 5.Hodges GR, Degener CE, Barnes WG. Clinical significance of citrobacter isolates. Am J Clin Pathol. 1978;70:37–40. doi: 10.1093/ajcp/70.1.37. [DOI] [PubMed] [Google Scholar]
- 6.Vaz Marecos C, Ferreira M, Ferreira MM, Barroso MR, Sepsis BMR. Sepsis, meningitis and cerebral abscesses caused by Citrobacter koseri . BMJ Case Rep. 2012;2012:bcr1020114941. doi: 10.1136/bcr.10.2011.4941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Samonis G, Karageorgopoulos DE, Kofteridis DP, Matthaiou DK, Sidiropoulou V, et al. Citrobacter infections in a general hospital: characteristics and outcomes. Eur J Clin Microbiol Infect Dis. 2009;28:61–68. doi: 10.1007/s10096-008-0598-z. [DOI] [PubMed] [Google Scholar]
- 8.Ribeiro CD, Davis P, Jones DM. Citrobacter koseri meningitis in a special care baby unit. J Clin Pathol. 1976;29:1094–1096. doi: 10.1136/jcp.29.12.1094. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Porres-Osante N, Sáenz Y, Somalo S, Torres C. Characterization of Beta-lactamases in Faecal Enterobacteriaceae recovered from healthy humans in Spain: focusing on AmpC Polymorphisms. Microb Ecol. 2015;70:132–140. doi: 10.1007/s00248-014-0544-9. [DOI] [PubMed] [Google Scholar]
- 10.Peter S, Wolz C, Kaase M, Marschal M, Schulte B, et al. Emergence of Citrobacter freundii carrying IMP-8 metallo-β-lactamase in Germany. New Microbes New Infect. 2014;2:42–45. doi: 10.1002/nmi2.36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lavigne J-P, Defez C, Bouziges N, Mahamat A, Sotto A. Clinical and molecular epidemiology of multidrug-resistant Citrobacter spp. infections in a French university hospital. Eur J Clin Microbiol Infect Dis. 2007;26:439–441. doi: 10.1007/s10096-007-0315-3. [DOI] [PubMed] [Google Scholar]
- 12.Euzéby JP. List of bacterial names with standing in nomenclature: a folder available on the Internet. Int J Syst Bacteriol. 1997;47:590–592. doi: 10.1099/00207713-47-2-590. [DOI] [PubMed] [Google Scholar]
- 13.Peter S, Bezdan D, Oberhettinger P, Vogel W, Dörfel D, et al. Whole-Genome sequencing enabling the detection of a colistin-resistant hypermutating Citrobacter werkmanii strain harbouring a novel metallo-β-lactamase VIM-48. Int J Antimicrob Agents. 2018;51:867–874. doi: 10.1016/j.ijantimicag.2018.01.015. [DOI] [PubMed] [Google Scholar]
- 14.Liese J, Schüle L, Oberhettinger P, Tschörner L, Nguyen T, et al. Expansion of vancomycin-Resistant Enterococcus faecium in an academic tertiary Hospital in Southwest Germany: a large-scale whole-genome-based outbreak Investigation. Antimicrob Agents Chemother. 2019;63 doi: 10.1128/AAC.01978-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Nurk S, Bankevich A, Antipov D, Gurevich AA, Korobeynikov A, et al. Assembling single-cell genomes and mini-metagenomes from chimeric MDA products. J Comput Biol. 2013;20:714–737. doi: 10.1089/cmb.2013.0084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Darling AE, Mau B, Perna NT. progressiveMauve: multiple genome alignment with gene gain, loss and rearrangement. PLoS One. 2010;5:e11147. doi: 10.1371/journal.pone.0011147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Arndt D, Grant JR, Marcu A, Sajed T, Pon A, et al. PHASTER: a better, faster version of the PHAST phage search tool. Nucleic Acids Res. 2016;44:W16–W21. doi: 10.1093/nar/gkw387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Yoon S-H, Ha S-M, Kwon S, Lim J, Kim Y, et al. Introducing EzBioCloud: a taxonomically United database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol. 2017;67:1613–1617. doi: 10.1099/ijsem.0.001755. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Clermont D, Motreff L, Passet V, Fernandez J-C, Bizet C, et al. Multilocus sequence analysis of the genus Citrobacter and description of Citrobacter pasteurii sp. nov. Int J Syst Evol Microbiol. 2015;65:1486–1490. doi: 10.1099/ijs.0.000122. [DOI] [PubMed] [Google Scholar]
- 20.Ribeiro TG, Gonçalves BR, da Silva MS, Novais Ângela, Machado E, et al. Citrobacter portucalensis sp. nov., isolated from an aquatic sample. Int J Syst Evol Microbiol. 2017;67:3513–3517. doi: 10.1099/ijsem.0.002154. [DOI] [PubMed] [Google Scholar]
- 21.Stamatakis A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 2014;30:1312–1313. doi: 10.1093/bioinformatics/btu033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Richter M, Rosselló-Móra R. Shifting the genomic gold standard for the prokaryotic species definition. Proc Natl Acad Sci U S A. 2009;106:19126–19131. doi: 10.1073/pnas.0906412106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Richter M, Rosselló-Móra R, Oliver Glöckner F, Peplies J. JSpeciesWS: a web server for prokaryotic species circumscription based on pairwise genome comparison. Bioinformatics. 2016;32:929–931. doi: 10.1093/bioinformatics/btv681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Meier-Kolthoff JP, Auch AF, Klenk H-P, Göker M. Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics. 2013;14:60. doi: 10.1186/1471-2105-14-60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Larsen MV, Cosentino S, Rasmussen S, Friis C, Hasman H, et al. Multilocus sequence typing of total-genome-sequenced bacteria. J Clin Microbiol. 2012;50:1355–1361. doi: 10.1128/JCM.06094-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Warren JR, Farmer JJ, Dewhirst FE, Birkhead K, Zembower T, et al. Outbreak of nosocomial infections due to extended-spectrum beta-lactamase-producing strains of enteric group 137, a new member of the family Enterobacteriaceae closely related to Citrobacter farmeri and Citrobacter amalonaticus . J Clin Microbiol. 2000;38:3946–3952. doi: 10.1128/JCM.38.11.3946-3952.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Naum M, Brown EW, Mason-Gamer RJ. Is 16S rDNA a reliable phylogenetic marker to characterize relationships below the family level in the enterobacteriaceae? J Mol Evol. 2008;66:630–642. doi: 10.1007/s00239-008-9115-3. [DOI] [PubMed] [Google Scholar]
- 28.Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P, et al. DNA-DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol. 2007;57:81–91. doi: 10.1099/ijs.0.64483-0. [DOI] [PubMed] [Google Scholar]
- 29.Chun J, Oren A, Ventosa A, Christensen H, Arahal DR, et al. Proposed minimal standards for the use of genome data for the taxonomy of prokaryotes. Int J Syst Evol Microbiol. 2018;68:461–466. doi: 10.1099/ijsem.0.002516. [DOI] [PubMed] [Google Scholar]
- 30.Brenner DJ, Grimont PA, Steigerwalt AG, Fanning GR, Ageron E, et al. Classification of citrobacteria by DNA hybridization: designation of Citrobacter farmeri sp. nov., Citrobacter youngae sp. nov., Citrobacter braakii sp. nov., Citrobacter werkmanii sp. nov., Citrobacter sedlakii sp. nov., and three unnamed Citrobacter genomospecies. Int J Syst Bacteriol. 1993;43:645–658. doi: 10.1099/00207713-43-4-645. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.