Abstract
Background
In patients with colorectal liver metastases (CRLM) preoperative imaging may include contrast‐enhanced (ce) MRI and [18F]fluorodeoxyglucose (18F‐FDG) PET–CT. This study assessed trends and variation between hospitals and oncological networks in the use of preoperative imaging in the Netherlands.
Methods
Data for all patients who underwent liver resection for CRLM in the Netherlands between 2014 and 2018 were retrieved from a nationwide auditing database. Multivariable logistic regression analysis was used to assess use of ceMRI, 18F‐FDG PET–CT and combined ceMRI and 18F‐FDG PET–CT, and trends in preoperative imaging and hospital and oncological network variation.
Results
A total of 4510 patients were included, of whom 1562 had ceMRI, 872 had 18F‐FDG PET–CT, and 1293 had combined ceMRI and 18F‐FDG PET–CT. Use of ceMRI increased over time (from 9·6 to 26·2 per cent; P < 0·001), use of 18F‐FDG PET–CT decreased (from 28·6 to 6·0 per cent; P < 0·001), and use of both ceMRI and 18F‐FDG PET–CT 16·9 per cent) remained stable. Unadjusted variation in the use of ceMRI, 18F‐FDG PET–CT, and combined ceMRI and 18F‐FDG PET–CT ranged from 5·6 to 100 per cent between hospitals. After case‐mix correction, hospital and oncological network variation was found for all imaging modalities.
Discussion
Significant variation exists concerning the use of preoperative imaging for CRLM between hospitals and oncological networks in the Netherlands. The use of MRI is increasing, whereas that of 18F‐FDG PET–CT is decreasing.
A nationwide population‐based overview from 2014 to 2018 in the Netherlands showed an increased use of contrast‐enhanced MRI and decreased use of PET–CT. There was significant hospital variation in the use of both MRI and PET–CT.

Wide variation in the use of preoperative MRI and FDG‐PET‐CT
Antecedentes
En pacientes con metástasis hepáticas colorrectales (colorrectal liver metastases, CRLM), los estudios de imagen preoperatorios pueden incluir resonancia magnética con contraste (ce)MRI y 18F‐FDG‐PET‐CT. Este estudio evaluó las tendencias y la variación entre los hospitales y las redes oncológicas en el uso de estudios de imagen preoperatorios en los Países Bajos.
Métodos
Todos los pacientes que se sometieron a una resección hepática por CRLM en los Países Bajos entre 2014 y 2018 fueron seleccionados a partir de una base de datos a nivel nacional auditada. El análisis de regresión logística multivariable se utilizó para evaluar el uso de ceMRI, de 18F‐FDG‐PET‐CT y de ceMRI combinado con 18F‐FDG‐PET‐CT, así como para determinar las tendencias en los estudios de imagen preoperatorios y las variaciones hospitalarias y de la red oncológica.
Resultados
En total, se incluyeron 4.510 pacientes, de los cuales 1.562 se sometieron a ceMRI, 872 a 18F‐FDG‐PET‐CT y 1.293 a ceMRI combinado con 18F‐FDG‐PET‐CT. El uso de ceMRI aumentó con el tiempo del 9,6% al 26,2% (P < 0,001), el uso de 18F‐FDG‐PET‐CT disminuyó (25% a 6,0%, P < 0,001) y el uso de ceMRI y 18F‐FDG‐PET‐ CT (17%) se mantuvo estable. La variación no ajustada entre hospitales en el uso de ceMRI, 18F‐FDG‐PET‐CT y la combinación de ceMRI y 18F‐FDG‐PET‐CT oscilaba del 5% al 10%. Después de la corrección por case‐mix, la variación hospitalaria y de la red oncológica persistía en todas las pruebas de imagen.
Conclusión
En los Países Bajos existe una variación significativa entre hospitales y redes oncológicas respecto al uso de pruebas de imagen preoperatorias para el CRLM. El uso de MRI está aumentando, mientras que el uso de 18F‐FDG‐PET‐CT está disminuyendo.
Introduction
Colorectal liver metastases (CRLM) are the leading indication for liver surgery in the Netherlands, accounting for approximately 1000 liver resections each year1.
Current multidisciplinary management of CRLM by surgeons, interventional radiologists, radiation therapists and oncologists demands detailed preoperative knowledge consisting of anatomical location in relation to vascular structures, number and size of CRLM, and individual patients' risks and preferences2, 3. Increasingly used options include contrast‐enhanced (ce) MRI and [18F]fluorodeoxyglucose (18F‐FDG) PET–CT4, 5, 6. ceMRI has been suggested to have a significant advantage over CT in detecting additional (small) liver metastases, in particular those of subcapsular or peribiliary origin4, 7, 8, 9, 10, 11. The oncological advantage of preoperative 18F‐FDG PET–CT to assess CRLM is doubtful12, although this imaging method seems to have an advantage in identifying extrahepatic metastases of colorectal cancer13. Some authors14, 15 propose using 18F‐FDG PET–CT during follow‐up to assess intrahepatic and extrahepatic metastases. Several European countries have preoperative imaging guidelines that contain advice regarding the use of both ceMRI and 18F‐FDG PET–CT16. Guidelines in the UK17, 18 and Japan19, as well as the European Society for Medical Oncology consensus guideline on metastatic colorectal cancer20, point out that ceMRI and 18F‐FDG PET–CT can be performed in the preoperative work‐up. However, these guidelines indicate that more research is needed to address the added value of preoperative imaging in patients with CRLM.
The Dutch guidelines21 in dicate that, at baseline, CT should be performed to assess the presence of CRLM22. If treatment is considered, ceMRI can be performed to detect lesions smaller than 10 mm. The guideline further states that 18F‐FDG PET–CT should not be performed as part of preoperative work‐up, but is indicated only when extrahepatic metastases are suspected.
The aims of the present study were to provide a population‐based overview of factors associated with the use of different types of preoperative imaging modality, in patients with colorectal liver metastases, to report on trends over the years, and to assess variation between hospitals and oncological networks in the Netherlands.
Methods
This was a population‐based nationwide cohort study performed in the Netherlands with data from the Dutch Hepato‐Biliary Audit (DHBA)23. The Netherlands is a western European country with approximately 17 million inhabitants living on 33 883 square kilometres24. Healthcare is organized in 71 hospitals, including seven university hospitals and one comprehensive cancer centre23, 25. Twenty‐five hospitals perform liver surgery. A national minimum annual centre volume of 20 liver resections and infrastructural requirements (24/7 availability of an interventional radiologist) have led to the centralization of liver surgery26. Hospitals performing liver surgery in the Netherlands have been obliged to register liver resections in the DHBA since 2013. Detailed information on patient and disease characteristics, as well as diagnostic and treatment information, has been collected from 2013 onwards. Information regarding the formation and content of the DHBA has been described previously23. Data verification provided insight into the completeness and accuracy of the DHBA27. During this process, data in the DHBA were compared with those in the Dutch Cancer Registry. The completeness of data retrieved from 2015 was 97 per cent23.
Patient selection
All consecutive patients who underwent liver resection for CRLM between 1 January 2014 and 31 December 2018, and were registered in the DHBA before 22 March 2019, were included in the study. Patients who had ablation of CRLM alone were not included in the study as registration of such patients in the DHBA commenced on 1 January 2018. Patients were considered not eligible for analysis when missing data included date of birth, preoperative imaging modalities used, date of surgery, type of procedure or origin of the tumour for which resection was performed.
No ethical approval was needed as the DHBA is an obligatory audit from the Dutch inspectorate of healthcare and all analyses were performed on an anonymized data set.
Patient groups
In all patients CT of the abdomen and chest was performed as baseline imaging. Patients were divided into four groups for analysis: no additional imaging of the liver; preoperative imaging consisting of CT and ceMRI of the liver; preoperative imaging consisting of CT and 18F‐FDG PET–CT; and preoperative imaging consisting of CT, ceMRI and 18F‐FDG PET–CT.
Variables
Studied variables included patient characteristics (age, sex, ASA fitness grade, co‐morbidity score according to the Charlson Co‐morbidity Index (CCI), liver disease before surgery, previous liver surgery for CRLM and year of surgery), tumour characteristics (number of CRLM, diameter of largest CRLM before treatment on preoperative CT, synchronous or metachronous metastases, presence of extrahepatic metastases, and whether metastases were bilobar), and type of hospital and oncological network where treatment took place. Factors contributing to the use of ceMRI, 18F‐FDG PET–CT, and combined use of ceMRI and 18F‐FDG PET–CT were primary variables for case‐mix correction. Other studied variables and parameters were the use of the different preoperative imaging modalities over the years, and between‐hospital and between‐oncological network variation in the use of preoperative imaging modalities. Both were corrected for case‐mix variables.
All variables concerning tumour characteristics were based on normal preoperative work‐up before surgery, and therefore assessed using preoperative CT before additional imaging was performed. However, as a result of the retrospective nature of this study, these variables might resemble characteristics of the CRLM after ceMRI or 18F‐FDG PET–CT. Sensitivity analyses were performed in all statistical models, which consisted of dropping tumour characteristics.
As described previously28, oncological networks were classified according to treatment collaboration between hospitals, or topographical location if no collaboration network was present ( Fig. S1 , supporting information). An oncological network consists of one or more tertiary referral centres, including one of the seven university hospitals in the Netherlands. All regional hospitals are included in an oncological network, of which a few perform liver surgery. Regional hospitals not performing liver surgery refer patients to either a regional hospital performing liver surgery or tertiary referral centre for the treatment of CRLM, based on agreements in the oncology network. All hospitals in an oncological network have multidisciplinary meetings using video conferencing to discuss patients with CRLM and obtain a patient‐centred treatment plan. If necessary, patients with a high surgical risk as a result of co‐morbidity or need for more complex surgical procedures can be referred to tertiary referral centres28.
Statistical analysis
Baseline characteristics were compared between all groups using the χ2 test or Fisher's exact test as appropriate for categorical variables. Continuous variables were compared using independent two‐samples t test.
Identification of case‐mix factors, defined as non‐modifiable patient and tumour characteristics influencing the use of the different preoperative imaging modalities, was performed. Potential case‐mix factors were entered in univariable and multivariable multilevel logistic regression models, one model for each preoperative imaging modality. A multilevel analysis was used to take into account the changes in hospital policy, as well as unmeasured similarities of patients within the year of surgery. Separate analysis for trends in preoperative imaging over the years was performed using univariable and multivariable logistic regression for each treatment modality. These models were performed using case‐mix variables to correct for confounding factors associated with the use of the specific preoperative treatment modality.
Case‐mix correction was performed using the observed/expected (O/E) ratio, calculated by dividing the observed number of patients who had a preoperative imaging modality by the number of patients expected to receive that modality. The expected number of patients was based on a multivariable multilevel logistic regression model including case‐mix variables, resulting in case mix‐corrected variability in the use of preoperative imaging modalities between hospitals and oncological networks. An O/E ratio of 1 was considered to indicate that a hospital or oncological network performed exactly the expected amount of preoperative imaging. When the O/E ratio was below 1, a hospital or oncological network performed less preoperative imaging than expected. If the O/E ratio was higher than 1, a hospital or network performed more preoperative imaging than expected. On the basis of the model and O/E ratios for all hospitals or oncological networks, 95 per cent confidence intervals were calculated, indicating statistically significant outliers.
For all multivariable analyses, a two‐step method was undertaken. All variables were tested in a univariable model per outcome variable. If a significant association was found (P < 0·100, Wald test), the variable was entered in the multivariable model. Statistical significance was defined as a two‐sided P < 0·050 in the multivariable model. Outcomes were adjusted odds ratios (ORs) and 95 per cent confidence intervals. Multicollinearity was assessed in all multivariable models. This was done by calculation of the variance inflation factor (VIF). A VIF higher than 2·5 was considered to indicate multicollinearity.
All analyses were performed in R version 3.2.2 (R Foundation for Statistical Computing, Vienna, Austria).
Results
During the study inclusion period, 4846 patients underwent surgical liver resection for CRLM. Of these, 336 patients were excluded because of missing information on baseline characteristics, preoperative imaging techniques, postoperative outcomes and postoperative oncological classification. A total of 4510 patients were analysed, of whom 1562 (34·6 per cent) had ceMRI, 872 (19·3 per cent) had 18F‐FDG PET–CT, and 1293 (28·7 per cent) had both ceMRI and 18F‐FDG PET–CT. The remaining 783 patients (17·4 per cent) did not receive any additional imaging apart from CT.
ceMRI or combined ceMRI and 18F‐FDG PET–CT was used more often in patients with a history of liver disease, preoperative chemotherapy, synchronous metastases and a rectal primary tumour. ceMRI was used less often in patients with a greater maximum diameter of the largest liver metastases. If more CRLM were present, ceMRI or combined ceMRI and 18F‐FDG PET–CT was used more often. In patients with extrahepatic metastases 18F‐FDG PET–CT was used more often (Table 1).
Table 1.
Baseline characteristics for preoperative imaging in patients diagnosed with colorectal liver metastases between 2014 and 2018 in the Netherlands
| No additional imaging (n = 783) | MRI (n = 1562) | PET–CT (n = 872) | MRI + PET–CT (n = 1293) | P ‡ | |
|---|---|---|---|---|---|
| Age (years) | 0·038 | ||||
| ≤ 70 | 496 (63·5) | 1001 (64·2) | 520 (59·7) | 867 (67·2) | |
| > 70 | 285 (36·5) | 559 (35·8) | 351 (40·3) | 424 (32·8) | |
| Missing | 2 | 2 | 1 | 2 | |
| Sex | 0·078 | ||||
| M | 468 (59·8) | 1012 (64·8) | 555 (63·6) | 796 (61·6) | |
| F | 315 (40·2) | 550 (35·2) | 317 (36·4) | 497 (38·4) | |
| Charlson Co‐morbidity Index | < 0·001 | ||||
| 0–1 | 593 (76·7) | 1186 (77·1) | 598 (69·0) | 955 (74·7) | |
| ≥ 2 | 180 (23·3) | 352 (22·9) | 269 (31·0) | 324 (25·3) | |
| Missing | 10 | 24 | 5 | 14 | |
| BMI (kg/m2) * | 26·1(4·4) | 26·3(4·3) | 26·1(4·4) | 26·5(4·4) | 0·124§ |
| ASA grade | 0·032 | ||||
| I–II | 606 (77·9) | 1271 (81·6) | 654 (79·3) | 1058 (82·6) | |
| ≥ III | 172 (22·1) | 286 (18·4) | 171 (20·7) | 223 (17·4) | |
| Missing | 5 | 5 | 47 | 12 | |
| Previous liver resection | 0·002 | ||||
| No | 615 (79·8) | 1303 (84·6) | 681 (79·0) | 1063 (82·7) | |
| Yes | 156 (20·2) | 238 (15·4) | 181 (21·0) | 222 (17·3) | |
| Missing | 12 | 21 | 10 | 8 | |
| History of liver disease † | 0·145 | ||||
| No | 758 (98·8) | 1499 (98·1) | 839 (98·5) | 1225 (99·1) | |
| Yes | 9 (1·2) | 29 (1·9) | 13 (1·5) | 11 (0·9) | |
| Missing | 16 | 34 | 20 | 57 | |
| History of preoperative chemotherapy | < 0·001 | ||||
| No | 457 (64·5) | 1004 (70·1) | 581 (75·0) | 800 (68·6) | |
| Yes | 252 (35·5) | 429 (29·9) | 194 (25·0) | 367 (31·4) | |
| Missing | 74 | 129 | 97 | 126 | |
| No. of lesions | < 0·001 | ||||
| 1 | 353 (47·5) | 617 (40·5) | 440 (52·1) | 515 (40·8) | |
| 2 | 153 (20·6) | 339 (22·3) | 199 (23·6) | 260 (20·6) | |
| 3 | 91 (12·2) | 160 (10·5) | 95 (11·3) | 157 (12·5) | |
| 4 | 52 (7·0) | 112 (7·4) | 41 (4·9) | 110 (8·7) | |
| 5 | 28 (3·8) | 81 (5·3) | 24 (2·8) | 57 (4·5) | |
| > 5 | 66 (8·9) | 214 (14·1) | 45 (5·3) | 162 (12·8) | |
| Missing | 40 | 39 | 28 | 32 | |
| Maximum diameter of largest CRLM (mm) | < 0·001 | ||||
| < 20 | 169 (26·2) | 514 (35·8) | 180 (24·7) | 369 (31·3) | |
| 20–34 | 232 (36·0) | 544 (37·9) | 297 (40·8) | 437 (37·1) | |
| 35–54 | 137 (21·3) | 239 (16·7) | 157 (21·6) | 231 (19·6) | |
| ≥ 55 | 106 (16·5) | 137 (9·6) | 94 (12·9) | 141 (12·0) | |
| Missing | 139 | 128 | 144 | 115 | |
| Location of primary tumour | < 0·001 | ||||
| Colon | 527 (67·5) | 974 (62·5) | 614 (70·4) | 793 (61·3) | |
| Rectum | 254 (32·5) | 584 (37·5) | 258 (29·6) | 500 (38·7) | |
| Missing | 2 | 4 | 0 | 0 | |
| Nodal status of primary tumour | 0·109 | ||||
| pN0 | 194 (35·6) | 405 (37·0) | 281 (41·4) | 366 (37·3) | |
| pN1 | 206 (37·8) | 406 (37·1) | 233 (34·3) | 349 (35·5) | |
| pN2 | 145 (26·6) | 284 (25·9) | 165 (24·3) | 267 (27·2) | |
| Unknown | 238 | 467 | 193 | 311 | |
| Type of metastases | < 0·001 | ||||
| Metachronous | 390 (50·4) | 723 (46·6) | 552 (63·9) | 697 (54·6) | |
| Synchronous | 384 (49·6) | 827 (53·4) | 312 (36·1) | 580 (45·4) | |
| Missing | 9 | 12 | 8 | 16 | |
| Extrahepatic disease | < 0·001 | ||||
| No | 628 (91·1) | 1383 (93.8) | 702 (88.5) | 1110 (91.1) | |
| Yes | 61 (8.9) | 92 (6.2) | 91 (11.5) | 109 (8.9) | |
| Missing | 94 | 87 | 79 | 74 | |
| Type of hospital | < 0·001 | ||||
| Regional | 350 (44·7) | 928 (59·4) | 499 (57·2) | 713 (55·1) | |
| Tertiary referral centre | 433 (55·3) | 634 (40·6) | 373 (42·8) | 580 (44·9) | |
| Year of surgery | < 0·001 | ||||
| 2014 | 178 (22·7) | 150 (9·6) | 249 (28·6) | 194 (15·0) | |
| 2015 | 142 (18·1) | 250 (16·0) | 219 (25·1) | 273 (21·1) | |
| 2016 | 155 (19·8) | 340 (21·8) | 224 (25·7) | 289 (22·4) | |
| 2017 | 150 (19·2) | 413 (26·4) | 128 (14·7) | 318 (24·6) | |
| 2018 | 158 (20·2) | 409 (26·2) | 52 (6·0) | 219 (16·9) |
Values in parentheses are percentages unless indicated otherwise;
values are mean(s.d.).
Liver cirrhosis, oesophageal variceal disease, hepatorenal syndrome, liver failure, alcoholic liver disease, toxic liver disease (mild), (chronic) hepatitis or liver fibrosis. CRLM, colorectal liver metastases.
χ2 test or Fisher's exact test, except §independent two‐samples t test.
Factors associated with use of different preoperative imaging modalities
In multivariable multilevel logistic regression analysis, factors positively associated with preoperative use of ceMRI included having an increasing number of CRLM (5 or more tumours versus 1 tumour: adjusted odds ratio (OR) 2·45, 95 per cent c.i. 1·89 to 3·17; P < 0·001), a rectal primary tumour (adjusted OR 1·44, 1·25 to 1·67; P < 0·001) and synchronous metastases (adjusted OR 1·22, 1·05 to 1·41; P = 0·012) (Table 2). Factors negatively associated with preoperative use of ceMRI included high ASA grade (adjusted OR 0·74, 0·62 to 0·88; P = 0·001), history of liver resection (adjusted OR 0·79, 0·66 to 0·94; P = 0·006), maximum diameter of the largest CRLM (less than 20 mm versus 55 mm or more: adjusted OR 0·32, 0·25 to 0·40; P < 0·001), extrahepatic metastases (adjusted OR 0·74, 0·60 to 0·90; P = 0·003) and treatment in a tertiary referral centre (adjusted OR 0·79, 0·66 to 0·89; P < 0·001) (Table 2).
Table 2.
Association model of patient and tumour factors with the use of preoperative contrast‐enhanced MRI in patients with colorectal liver metastases in the Netherlands, 2014–2018
| Univariable analysis* | Multivariable analysis* | ||||
|---|---|---|---|---|---|
| No. of patients (n = 4510) | Odds ratio | P | Adjusted odds ratio | P | |
| Age (years) | 0·015 | 0·632 | |||
| ≤ 50 | 315 | 1·00 (reference) | 1·00 (reference) | ||
| 50–64 | 1543 | 0·93 (0·72, 1·21) | 0·603 | 0·96 (0·50, 1·96) | 0·762 |
| 65–79 | 2331 | 0·81 (0·63, 1·04) | 0·097 | 0·88 (0·71, 1·28) | 0·383 |
| ≥ 80 | 314 | 0·67 (0·48, 0·93) | 0·016 | 0·86 (0·66, 1·17) | 0·418 |
| Missing† | 7 | ||||
| Sex | 0·310 | ||||
| M | 2831 | 1·00 (reference) | |||
| F | 1679 | 0·94 (0·83, 1·06) | |||
| Charlson Co‐morbidity Index | 0·012 | 0·753 | |||
| 0–1 | 3332 | 1·00 (reference) | 1·00 (reference) | ||
| ≥ 2 | 1125 | 0·84 (0·73, 0·96) | 0·98 (0·83, 1·14) | ||
| Missing† | 53 | ||||
| BMI | 1·02 (1·00, 1·04) | 0·023 | 1·02 (1·01, 1·04) | 0·014 | |
| ASA grade | 0·005 | 0·001 | |||
| I–II | 3589 | 1·00 (reference) | 1·00 (reference) | ||
| ≥ III | 852 | 0·80 (0·69, 0·94) | 0·74 (0·62, 0·88) | ||
| Missing† | 69 | ||||
| History of liver disease ‡ | 0·811 | ||||
| No | 4321 | 1·00 (reference) | |||
| Yes | 62 | 1·07 (0·64, 1·83) | |||
| Missing† | 127 | ||||
| History of liver resection | < 0·001 | 0·006 | |||
| No | 3662 | 1·00 (reference) | 1·00 (reference) | ||
| Yes | 797 | 0·75 (0·64, 0·87) | 0·79 (0·66, 0·94) | ||
| Missing† | 51 | ||||
| History of preoperative chemotherapy | 0·708 | ||||
| No | 2842 | 1·00 (reference) | |||
| Yes | 1242 | 1·03 (0·89, 1·18) | |||
| Missing† | 426 | ||||
| No. of CRLM | < 0·001 | < 0·001 | |||
| 1 | 1925 | 1·00 (reference) | 1·00 (reference) | ||
| 2 | 951 | 1·19 (1·02, 1·40) | 0·031 | 1·19 (1·00, 1·42) | 0·051 |
| 3 | 503 | 1·19 (0·98, 1·46) | 0·086 | 1·28 (1·02, 1·60) | 0·047 |
| 4 | 315 | 1·67 (1·30, 2·17) | < 0·001 | 1·71 (1·29, 2·27) | 0·001 |
| 5 | 190 | 1·86 (1·34, 2·61) | < 0·001 | 1·86 (1·29, 2·69) | 0·002 |
| >5 | 487 | 2·37 (1·89, 3·00) | < 0·001 | 2·45 (1·89, 3·17) | < 0·001 |
| Missing† | 139 | ||||
| Maximum diameter of largest CRLM (mm) | < 0·001 | < 0·001 | |||
| < 20 | 1232 | 1·00 (reference) | 1·00 (reference) | ||
| 20–34 | 1510 | 0·73 (0·62, 0·86) | < 0·001 | 0·72 (0·61, 0·87) | < 0·001 |
| 35–54 | 764 | 0·63 (0·52, 0·77) | < 0·001 | 0·66 (0·53, 0·81) | < 0·001 |
| ≥ 55 | 478 | 0·55 (0·44, 0·69) | < 0·001 | 0·56 (0·44, 0·72) | < 0·001 |
| Missing | 526 | 0·34 (0·27, 0·42) | < 0·001 | 0·32 (0·25, 0·40) | < 0·001 |
| Bilobar disease | 0·716 | ||||
| No | 2423 | 1·00 (reference) | |||
| Yes | 2043 | 1·02 (0·91, 1·16) | |||
| Missing† | 44 | ||||
| Location of primary tumour | < 0·001 | < 0·001 | |||
| Colon | 2908 | 1·00 (reference) | 1·00 (reference) | ||
| Rectal | 1596 | 1·37 (1·20, 1·56) | 1·44 (1·25, 1·67) | ||
| Missing† | 6 | ||||
| Nodal stage of primary tumour | 0·607 | ||||
| pN0 | 1246 | 1·00 (reference) | |||
| pN1 | 1194 | 1·06 (0·90, 1·25) | 0·489 | ||
| pN2 | 861 | 1·10 (0·91, 1·31) | 0·323 | ||
| Missing | 1209 | 1·11 (0·94, 1·31) | 0·204 | ||
| Type of metastases | < 0·001 | 0·012 | |||
| Metachronous | 2362 | 1·00 (reference) | 1·00 (reference) | ||
| Synchronous | 2103 | 1·34 (1·19, 1·52) | 1·22 (1·05, 1·41) | ||
| Missing† | 45 | ||||
| Extrahepatic metastases | < 0·001 | 0·003 | |||
| No | 3823 | 1·00 (reference) | 1·00 (reference) | ||
| Yes | 566 | 0·66 (0·56, 0·80) | 0·74 (0·60, 0·90) | ||
| Missing | 121 | ||||
| Type of hospital | < 0·001 | < 0·001 | |||
| Regional | 2490 | 1·00 (reference) | 1·00 (reference) | ||
| Tertiary referral centre§ | 2020 | 0·78 (0·69, 0·88) | 0·79 (0·66, 0·89) | ||
Values in parentheses are 95 per cent confidence intervals.
Multilevel logistic regression model with individuals nested for year of surgery.
Missing values not included in analyses because of relatively small group.
Liver cirrhosis, oesophageal variceal disease, hepatorenal syndrome, liver failure, alcoholic liver disease, toxic liver disease (mild), (chronic) hepatitis or liver fibrosis.
Defined as hospitals with highest expertise on oncological surgery.
In multivariable multilevel logistic regression analysis, factors positively associated with preoperative use of 18F‐FDG PET–CT included higher CCI score (adjusted OR 1·22, 95 per cent c.i. 1·05 to 1·40; P = 0·003), maximum diameter of largest CRLM (less than 20 mm versus 55 mm or more: adjusted OR 1·29, 1·03 to 1·62; P = 0·027) and extrahepatic metastases (adjusted OR 1·45, 1·20 to 1·75; P < 0·001) (Table 3). Factors negatively associated with preoperative use of 18F‐FDG PET CT included only synchronous metastases (adjusted OR 0·66, 0·58 to 0·76; P < 0·001) (Table 3).
Table 3.
Association model of patient and tumour factors with the use of preoperative [18F]fluorodeoxyglucose PET–CT in patients with colorectal liver metastases in the Netherlands, 2014–2018
| Univariable analysis* | Multivariable analysis* | ||||
|---|---|---|---|---|---|
| No. of patients (n = 4510) | Odds ratio | P | Adjusted odds ratio | P | |
| Age (years) | 0·314 | ||||
| ≤ 50 | 315 | 1·00 (reference) | |||
| 50–64 | 1543 | 1·13 (0·88, 1·44) | 0·333 | ||
| 65–79 | 2331 | 1·22 (0·97, 1·55) | 0·096 | ||
| ≥ 80 | 314 | 1·17 (0·86, 1·61) | 0·319 | ||
| Missing† | 7 | ||||
| Sex | 0·622 | ||||
| M | 2831 | 1·00 (reference) | |||
| F | 1679 | 1·03 (0·91, 1·16) | |||
| Charlson Co‐morbidity Index | < 0·001 | 0·003 | |||
| 0–1 | 3332 | 1·00 (reference) | 1·00 (reference) | ||
| ≥ 2 | 1125 | 1·28 (1·12, 1·46) | 1·22 (1·05, 1·40) | ||
| Missing† | 53 | ||||
| BMI | 1·00 (0·99, 1·02) | 0·815 | |||
| ASA grade | 0·444 | ||||
| I–II | 3589 | 1·00 (reference) | |||
| ≥ III | 852 | 0·94 (0·81, 1·10) | |||
| Missing† | 69 | ||||
| History of liver disease ‡ | 0·156 | ||||
| No | 4321 | 1·00 (reference) | |||
| Yes | 62 | 0·69 (0·41, 1·15) | |||
| Missing† | 127 | ||||
| History of liver resection | 0·132 | ||||
| No | 3662 | 1·00 (reference) | |||
| Yes | 797 | 1·12 (0·97, 1·31) | |||
| Missing† | 51 | ||||
| History of preoperative chemotherapy | 0·044 | 0·164 | |||
| No | 2842 | 1·00 (reference) | 1·00 (reference) | ||
| Yes | 1242 | 0·87 (0·77, 1·00) | 0·97 (0·94, 1·32) | ||
| Missing† | 426 | ||||
| No. of CRLM | 0·056 | 0·235 | |||
| 1 | 1925 | 1·00 (reference) | 1·00 (reference) | ||
| 2 | 951 | 0·95 (0·81, 1·11) | 0·498 | 0·89 (0·75, 1·06) | 0·17 |
| 3 | 503 | 1·02 (0·84, 1·24) | 0·845 | 1·04 (0·85, 1·34) | 0·786 |
| 4 | 315 | 0·94 (0·74, 1·19) | 0·582 | 0·96 (0·73, 1·26) | 0·561 |
| 5 | 190 | 0·75 (0·56, 1·02) | 0·067 | 0·80 (0·57, 1·12) | 0·206 |
| >5 | 487 | 0·75 (0·61, 0·92) | 0·005 | 0·81 (0·64, 1·04) | 0·091 |
| Missing† | 139 | ||||
| Maximum diameter of largest CRLM (mm) | 0·060 | 0·018 | |||
| < 20 | 1232 | 1·00 (reference) | 1·00 (reference) | ||
| 20–34 | 1510 | 1·17 (1·02, 1·37) | 0·035 | 1·18 (1·01, 1·39) | 0·034 |
| 35–54 | 764 | 1·28 (1·07, 1·54) | 0·007 | 1·30 (1·08, 1·62) | 0·002 |
| ≥ 55 | 478 | 1·20 (0·97, 1·49) | 0·087 | 1·29 (1·03, 1·62) | 0·027 |
| Missing | 526 | 1·21 (0·98, 1·48) | 0·072 | 1·34 (1·06, 1·68) | 0·009 |
| Bilobar disease | 0·041 | 0·096 | |||
| No | 2423 | 1·00 (reference) | 1·00 (reference) | ||
| Yes | 2043 | 1·13 (1·01, 1·27) | 1·15 (0·97, 1·36) | ||
| Missing† | 44 | ||||
| Location of primary tumour | 0·567 | ||||
| Colon | 2908 | 1·00 (reference) | |||
| Rectal | 1596 | 0·96 (0·85, 1·09) | |||
| Missing† | 6 | ||||
| Nodal stage of primary tumour | < 0·001 | 0·104 | |||
| pN0 | 1246 | 1·00 (reference) | 1·00 (reference) | ||
| pN1 | 1194 | 0·88 (0·75, 1·03) | 0·117 | 0·89 (0·75, 1·05) | 0·184 |
| pN2 | 861 | 0·93 (0·78, 1·11) | 0·429 | 0·96 (0·80, 0·96) | 0·591 |
| Missing | 1209 | 0·66 (0·56, 0·78) | < 0·001 | 0·80 (0·67, 0·96) | 0·024 |
| Type of metastases | < 0·001 | < 0·001 | |||
| Metachronous | 2362 | 1·00 (reference) | 1·00 (reference) | ||
| Synchronous | 2103 | 0·66 (0·58, 0·74) | 0·66 (0·58, 0·76) | ||
| Missing† | 45 | ||||
| Extrahepatic metastases | < 0·001 | < 0·001 | |||
| No | 3823 | 1·00 (reference) | 1·00 (reference) | ||
| Yes | 566 | 1·44 (1·21, 1·73) | 1·45 (1·20, 1·75) | ||
| Missing | 121 | ||||
| Type of hospital | 0·317 | ||||
| Regional | 2490 | 1·00 (reference) | |||
| Tertiary referral centre§ | 2020 | 0·94 (0·84, 1·06) | |||
Values in parentheses are 95 per cent confidence intervals.
Multilevel logistic regression model with individuals nested for year of surgery.
Missing values not included in analyses because of relatively small group.
Liver cirrhosis, oesophageal variceal disease, hepatorenal syndrome, liver failure, alcoholic liver disease, toxic liver disease (mild), (chronic) hepatitis or liver fibrosis.
Defined as hospitals with highest expertise on oncological surgery.
In multivariable multilevel logistic regression analysis, the only factor associated positively with preoperative use of a combination of ceMRI and 18F‐FDG PET–CT was rectal primary tumour (adjusted OR 1·23, 95 per cent c.i. 1·06 to 1·42; P = 0·005) (Table 4). There were no factors associated negatively with the combined use of ceMRI and 18F‐FDG PET–CT.
Table 4.
Association model of patient and tumour factors with the use of preoperative contrast‐enhanced MRI and [18F]fluorodeoxyglucose PET–CT in patients with colorectal liver metastases in the Netherlands, 2014–2018
| Univariable analysis* | Multivariable analysis* | ||||
|---|---|---|---|---|---|
| No. of patients (n = 4510) | Odds ratio | P | Adjusted odds ratio | P | |
| Age (years) | 0·289 | ||||
| ≤ 50 | 315 | 1·00 (reference) | |||
| 50–64 | 1543 | 1·04 (0·80, 1·36) | 0·802 | ||
| 65–79 | 2331 | 0·96 (0·74, 1·24) | 0·730 | ||
| ≥ 80 | 314 | 0·80 (0·56, 1·14) | 0·218 | ||
| Missing† | 7 | ||||
| Sex | |||||
| M | 2831 | 1·00 (reference) | |||
| F | 1679 | 1·07 (0·94, 1·23) | |||
| Charlson Co‐morbidity Index | 0·929 | ||||
| 0–1 | 3332 | 1·00 (reference) | |||
| ≥ 2 | 1125 | 1·01 (0·87, 1·17) | |||
| Missing† | 53 | ||||
| BMI | 1·01 (1·00, 1·03) | 0·091 | 1·01 (0·99, 1·04) | 0·204 | |
| ASA grade | 0·056 | 0·126 | |||
| I–II | 3589 | 1·00 (reference) | 1·00 (reference) | ||
| ≥ III | 852 | 0·85 (0·72, 1·00) | 0·87 (0·73, 1·04) | ||
| Missing† | 69 | ||||
| History of liver disease ‡ | 0·057 | 0·057 | |||
| No | 4321 | 1·00 (reference) | 1·00 (reference) | ||
| Yes | 62 | 0·54 (0·27, 1·01) | 0·51 (0·26, 1·02) | ||
| Missing† | 127 | ||||
| History of liver resection | 0·010 | 0·760 | |||
| No | 3662 | 1·00 (reference) | 1·00 (reference) | ||
| Yes | 797 | 0·75 (0·64, 0·87) | 0·97 (0·81, 1·17) | ||
| Missing† | 51 | ||||
| History of preoperative chemotherapy | 0·324 | ||||
| No | 2842 | 1·00 (reference) | |||
| Yes | 1242 | 1·07 (0·93, 1·24) | |||
| Missing† | 426 | ||||
| No. of CRLM | 0·005 | 0·126 | |||
| 1 | 1925 | 1·00 (reference) | 1·00 (reference) | ||
| 2 | 951 | 1·03 (0·86, 1·23) | 0·738 | 0·93 (0·76, 1·13) | 0·467 |
| 3 | 503 | 1·24 (1·00, 1·54) | 0·051 | 1·21 (0·95, 1·55) | 0·129 |
| 4 | 315 | 1·47 (1·14, 1·89) | 0·002 | 1·28 (0·95, 1·71) | 0·099 |
| 5 | 190 | 1·17 (0·84, 1·62) | 0·341 | 1·06 (0·74, 1·53) | 0·752 |
| >5 | 487 | 1·37 (1·10, 1·69) | 0·004 | 1·22 (0·94, 1·58) | 0·140 |
| Missing† | 139 | ||||
| Maximum diameter of largest CRLM (mm) | 0·005 | 0·024 | |||
| < 20 | 1232 | 1·00 (reference) | 1·00 (reference) | ||
| 20–34 | 1510 | 0·95 (0·81, 1·12) | 0·563 | 0·95 (0·80, 1·14) | 0·615 |
| 35–54 | 764 | 1·01 (0·83, 1·23) | 0·892 | 1·04 (0·85, 1·28) | 0·691 |
| ≥ 55 | 478 | 0·98 (0·78, 1·23) | 0·854 | 0·98 (0·77, 1·26) | 0·897 |
| Missing | 526 | 0·65 (0·51, 0·83) | < 0·001 | 0·65 (0·50, 0·86) | < 0·001 |
| Bilobar disease | 0·007 | 0·107 | |||
| No | 2423 | 1·00 (reference) | 1·00 (reference) | ||
| Yes | 2043 | 1·19 (1·05, 1·36) | 1·16 (0·97, 1·39) | ||
| Missing† | 44 | ||||
| Location of primary tumour | 0·004 | 0·005 | |||
| Colon | 2908 | 1·00 (reference) | 1·00 (reference) | ||
| Rectal | 1596 | 1·22 (1·06, 1·39) | 1·23 (1·06, 1·42) | ||
| Missing† | 6 | ||||
| Nodal status of primary tumour | 0·047 | 0·016 | |||
| pN0 | 1246 | 1·00 (reference) | 1·00 (reference) | ||
| pN1 | 1194 | 0·99 (0·83, 1·18) | 0·837 | 0·93 (0·77, 1·12) | 0·430 |
| pN2 | 861 | 1·08 (0·89, 1·31) | 0·421 | 1·04 (0·86, 1·27) | 0·675 |
| Missing | 1209 | 0·83 (0·70, 0·99) | 0·043 | 0·76 (0·63, 0·93) | 0·006 |
| Type of metastases | 0·155 | ||||
| Metachronous | 2362 | 1·00 (reference) | |||
| Synchronous | 2103 | 0·91 (0·80, 1·04) | |||
| Missing† | 45 | ||||
| Extrahepatic metastases | 0·687 | ||||
| No | 3823 | 1·00 (reference) | |||
| Yes | 566 | 1·04 (0·86, 1·26) | |||
| Missing | 121 | ||||
| Type of hospital | 0·954 | ||||
| Regional | 2490 | 1·00 (reference) | |||
| Tertiary referral centre§ | 2020 | 1·00 (0·88, 1·14) | |||
Values in parentheses are 95 per cent confidence intervals.
Multilevel logistic regression model with individuals nested for year of surgery.
Missing values not included in analyses because of relatively small group.
Liver cirrhosis, oesophageal variceal disease, hepatorenal syndrome, liver failure, alcoholic liver disease, toxic liver disease (mild), (chronic) hepatitis or liver fibrosis.
Defined as hospitals with highest expertise on oncological surgery.
Trends in use of different imaging modalities over the years
In the Netherlands, an increase was observed in the preoperative use of ceMRI, from 9·6 per cent in 2014 to 26·2 per cent in 2018. Univariable and multivariable logistic regression for trend over the years showed that this increase was statistically significant (adjusted OR 4·72, 95 per cent c.i. 3·69 to 6·05; P < 0·001) (Fig. 1; Table S1 , supporting information).
Figure 1.
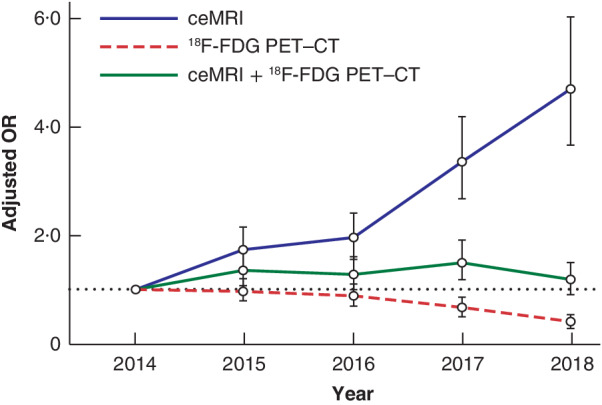
Case mix‐corrected trend analysis using multivariable logistic regression for the use of pretreatment imaging modalities for colorectal liver metastases in the Netherlands, 2014–2018 Adjusted odds ratios (ORs) are shown with 95 per cent confidence intervals. Case‐mix variables for contrast‐enhanced (ce) MRI were age, Charlson Co‐morbidity Index (CCI) score, BMI, ASA grade, history of liver resection, number of colorectal liver metastases (CRLM), maximum diameter of largest CRLM, location of primary tumour, type of metastases, extrahepatic metastases and type of hospital. Case‐mix variables for [18F]fluorodeoxyglucose (18F‐FDG) PET–CT were CCI score, preoperative chemotherapy, number of CRLM, maximum diameter of largest CRLM, bilobar disease, location of primary tumour, nodal status of primary tumour, extrahepatic metastases and type of hospital. Case‐mix variables for ceMRI and 18F‐FDG PET–CT were ASA grade, BMI, history of liver disease, history of liver resection, number of CRLM, maximum diameter of largest CRLM, bilobar disease, location of primary tumour and nodal status of primary tumour.
The use of preoperative 18F‐FDG PET–CT between 2014 and 2016 was stable at around 25 per cent, but use decreased in 2017 (14·7 per cent) and 2018 (6·0 per cent). Univariable and multivariable logistic regression for trend over the years showed that the decreasing trend was statistically significant (adjusted OR 0·42, 95 per cent c.i. 0·29 to 0·54; P < 0·001) (Fig. 1; Table S2 , supporting information).
The use of combined preoperative ceMRI and 18F‐FDG PET–CT was 15·0 per cent in 2014. During 2015 to 2017 this increased to 24·6 per cent, but was only 16.9 per cent in 2018. Univariable and multivariable logistic regression for trend over the years showed concordant results regarding the use of combined preoperative ceMRI and 18F‐FDG PET–CT (Fig. 1; Table S3 , supporting information).
Variation in use of different imaging modalities
Variation between hospitals and oncological networks was present for all preoperative imaging modalities. After case‐mix correction, significant hospital and oncological network variation was still present.
Unadjusted rates for the proportion of patients with CRLM receiving ceMRI in Dutch hospitals ranged between 15.4 and 96.2 per cent (Fig. 2 a). After case‐mix correction, widespread variation was observed in the use of ceMRI in the Netherlands. Seven hospitals performed more and eight hospitals performed less preoperative ceMRI than expected based on their case mix (Fig. 2 b). O/E ratios concerning the use of ceMRI between hospitals ranged from 0·21 to 1·51. In addition, two oncological networks performed more preoperative ceMRI than expected, whereas two other networks performed less preoperative ceMRI than expected, with O/E ratios ranging between 0·75 and 1·23 (Fig. 2 c).
Figure 2.
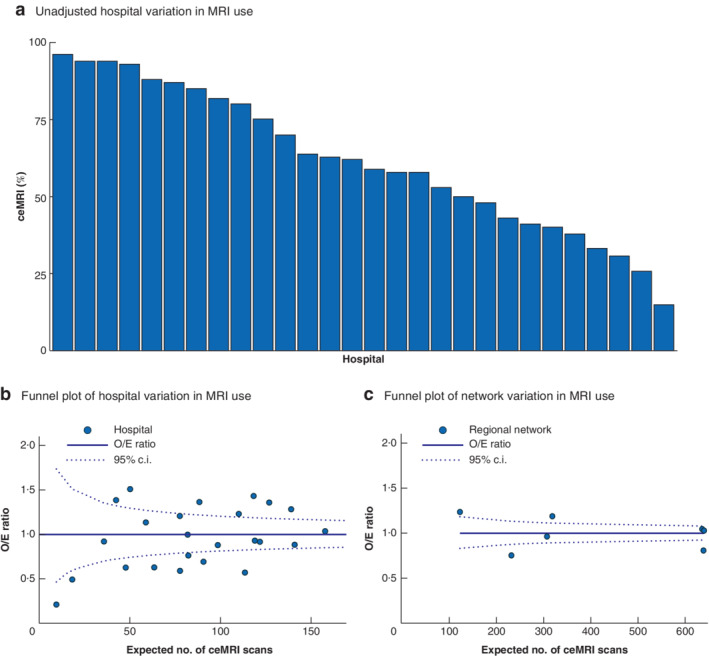
Unadjusted rates of hospital variation and case mix‐corrected funnel plots of between‐hospital and oncological network variation in the use of preoperative contrast‐enhanced MRI in patients with colorectal liver metastases in the Netherlands, 2014–2018 a Unadjusted rates of between‐hospital variation in use of contrast‐enhanced (ce) MRI. b Funnel plot of between‐hospital variation, case mix‐corrected for age, Charlson Co‐morbidity Index (CCI) score, BMI, ASA grade, history of liver resection, number of colorectal liver metastases (CRLM), maximum diameter of largest CRLM, location of primary tumour, type of metastases, extrahepatic metastases and type of hospital. c Funnel plot of oncological network variation, case mix‐corrected for age, CCI score, BMI, ASA grade, history of liver resection, number of CRLM, maximum diameter of largest CRLM, location of primary tumour, type of metastases, extrahepatic metastases and type of hospital. O/E, observed/expected.
Unadjusted rates for the proportion of patients with CRLM receiving 18F‐FDG PET–CT in Dutch hospitals ranged from 10.0 to 100 per cent (Fig. 3 a). After case‐mix correction, widespread variation in the use of 18F‐FDG PET–CT in the Netherlands was observed, with nine hospitals performing more and ten hospitals performing less preoperative 18F‐FDG PET–CT than expected based on their case mix (Fig. 3 b). O/E ratios concerning the use of 18F‐FDG PET–CT between hospitals ranged from 0·24 to 2·20. In addition, three oncological networks performed more preoperative 18F‐FDG PET–CT than expected and three other networks performed less than expected, with O/E ratios ranging between 0·50 and 1·67 (Fig. 3 c).
Figure 3.
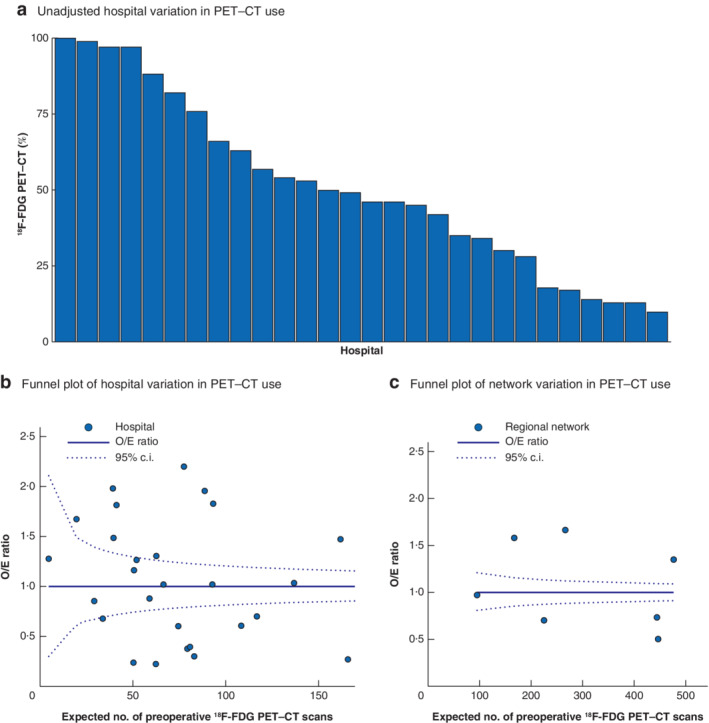
Unadjusted rates of hospital variation and case mix‐corrected funnel plots of between‐hospital and oncological network variation in the use of preoperative [18F]fluorodeoxyglucose PET–CT in patients with colorectal liver metastases in the Netherlands, 2014–2018 a Unadjusted rates of between‐hospital variation in use of [18F]fluorodeoxyglucose (18F‐FGD) PET–CT. b Funnel plot of between‐hospital variation, case mix‐corrected for Charlson Co‐morbidity Index (CCI) score, preoperative chemotherapy, number of colorectal liver metastases (CRLM), maximum diameter of largest CRLM, bilobar disease, location of primary tumour, nodal status of primary tumour, extrahepatic metastases and type of hospital. c Funnel plot of oncological network variation, case mix‐corrected for CCI score, preoperative chemotherapy, number of CRLM, maximum diameter of largest CRLM, bilobar disease, location of primary tumour, nodal status of primary tumour, extrahepatic metastases and type of hospital. O/E, observed/expected.
Unadjusted rates for the proportion of patients with CRLM receiving combined ceMRI and 18F‐FDG PET–CT in Dutch hospitals ranged between 5.6 and 94.9 per cent (Fig. 4 a). After case‐mix correction, widespread variation in the use of these combined imaging modalities was found. Eight hospitals performed preoperative ceMRI and 18F‐FDG PET–CT more often and 11 hospitals performed the combined imaging less often than expected based on their case mix (Fig. 4 b). O/E ratios for the use of ceMRI and 18F‐FDG PET–CT between hospitals ranged from 0·19 to 3·25. In addition, two oncological networks performed preoperative ceMRI and 18F‐FDG PET–CT more often than expected, whereas three other networks performed the combined imaging less often than expected, with O/E ratios ranging between 0·29 and 2·12 (Fig. 4 c).
Figure 4.
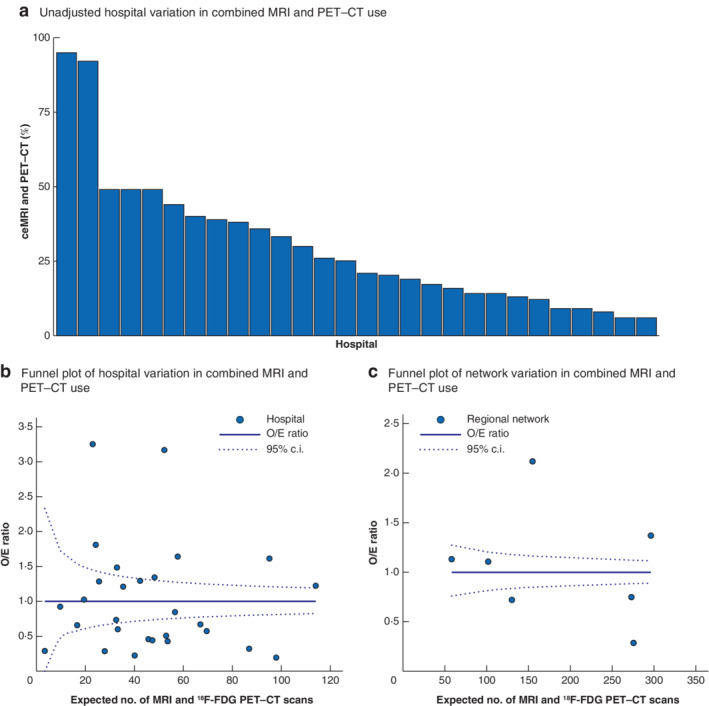
Unadjusted rates of hospital variation and case mix‐corrected funnel plots of between‐hospital and oncological network variation in the preoperative use of combined contrast‐enhanced MRI and [18F]fluorodeoxyglucose PET–CT in patients with colorectal liver metastases in the Netherlands, 2014–2018 a Unadjusted rates of between‐hospital variation in use of combined contrast‐enhanced (ce) MRI and [18F]fluorodeoxyglucose (18F‐FGD) PET–CT. b Funnel plot of between‐hospital variation, case mix‐corrected for ASA grade, BMI, history of liver disease, history of liver resection, number of colorectal liver metastases (CRLM), maximum diameter of CRLM, bilobar disease, location of primary tumour and nodal status of primary tumour. c Funnel plot of oncological network variation, case mix‐corrected for ASA grade, BMI, history of liver disease, history of liver resection, number of CRLM, maximum diameter of largest CRLM, bilobar disease, location of primary tumour and nodal status of primary tumour. O/E, observed/expected.
Multicollinearity was not observed for any of the reported models in this study: the VIF was always below 2·0. Sensitivity analyses, in which tumour characteristics were dropped from the analyses, did not show differences in any of the outcomes.
Discussion
In this nationwide population‐based analysis, ceMRI as preoperative imaging for CRLM was used increasingly in the Netherlands over time, whereas the use of 18F‐FDG PET–CT decreased. The use of combined ceMRI and 18F‐FDG PET–CT remained stable over the years. Use of MRI was associated with smaller diameter of CRLM or more CRLM. Use of 18F‐FDG PET–CT was associated with extrahepatic metastases and larger diameters of CRLM. Notable variation was present regarding the use of preoperative ceMRI, 18F‐FDG PET–CT, and combined ceMRI and 18F‐FDG PET–CT between hospitals and oncological networks in the Netherlands.
Few studies on trends and variation in the use of preoperative imaging have been published in the past. One French study29 showed that use of preoperative liver ceMRI increased from 53 to 80 per cent between 2009 and 2013, and 72 per cent of patients with resectable CRLM had preoperative ceMRI. In a Swedish population‐based study30, only 2 per cent of all patients with colorectal cancer had preoperative ceMRI of the liver. Unfortunately, this study did not report on trends or report a subanalysis of patients with CRLM.
The available evidence is not conclusive regarding the use of additional preoperative imaging modalities, resulting in variability in the use of ceMRI and 18F‐FDG PET–CT. Over the past few years, several studies8, 10, 11 have reported superior per lesion detection with MRI compared with conventional CT in patients with CRLM. An earlier report by Rojas Llimpe and colleagues31 provided insight into the additional value of ceMRI in patients receiving preoperative chemotherapy. Mostly retrospective studies have been performed to assess differences between different types of MRI, such as ceMRI, diffusion‐weighted MRI or gadoxetic acid‐enhanced liver MRI. New insights into the added value of different types of MRI in a prospective setting are needed. For this reason, the multicentre CAMINO trial (https://www.trialregister.nl/trial/8039): Netherlands Trial Register number NL8039 was commenced in the Netherlands in 2019; this trial aims to provide information concerning the clinical additional value of ceMRI in patients with CRLM.
18F‐FDG PET–CT is thought to have lower sensitivity than ceMRI, and is not favoured in the detection of CRLM4, 32. Detection rates are lower in patients who have received preoperative chemotherapy32. One RCT12 investigated the additional value of 18F‐FDG PET–CT in CRLM and concluded that this did not influence survival, whereas several unrandomized studies15, 33, 34 indicated that there could be added value for 18F‐FDG PET–CT in patients with extrahepatic metastases.
Large randomized trials or prospective multicentre studies on the use of ceMRI or 18F‐FDG PET–CT in patients with CRLM have not been conducted, and thus existing guidelines (such as the Dutch guideline) do not provide recommendations on what is needed. The Dutch guideline does not favour either ceMRI or CT in the work‐up before liver resection. It advises using 18F‐FDG PET–CT only in patients with extrahepatic metastases22. In the present study, an increase in the use of ceMRI in the Netherlands was observed, whereas use of 18F‐FDG PET–CT decreased. These trends are probably the result of international publications4, 7, 8, 10, 12, 13, 15, 31, 34 reporting the additional value of these imaging modalities.
Interestingly, ceMRI is thought to provide better insight into tumour burden in patients with a medical history of liver disease, and the literature10, 32 indicates that ceMRI could be useful as preoperative imaging in patients undergoing preoperative chemotherapy or who have had previous liver resection. 18F‐FDG PET–CT might have added value in patients with a higher nodal status of the colorectal primary tumour. However, this was not the case in the present study, as these factors were not associated with the use of either of the imaging modalities in this population‐based cohort. In addition, ceMRI was used less often in tertiary referral centres, whereas there was no difference in the use of 18F‐FDG PET–CT in the different types of hospital.
Variation in the use of imaging in the Netherlands could be explained by the fact that the Dutch guideline allows different approaches35. Notable variation in imaging at both a hospital and oncological network level reflects lack of consensus on both levels. There are several possible reasons for this. First, there is insufficient evidence and guidelines concerning the use of preoperative imaging in patients with CRLM. Second, health economic discussions could influence the use of these imaging modalities, as ceMRI and 18F‐FDG PET–CT are both more expensive than baseline ultrasonography and CT36. As there are considerable differences in the costs of the various imaging modalities, it is important to acknowledge these and to assess the cost‐effectiveness of imaging modalities for CRLM in the future.
Hospital variation is undesirable from a national healthcare perspective. Either unnecessary imaging was performed or different approaches to imaging led to different patient selection for treatment. It would be interesting to explore whether these differences in preoperative imaging lead to differences in treatment selection, and in disease‐free and overall survival. A next step in the audit is to incorporate long‐term follow‐up to investigate these associations further, to ensure that conclusions can be drawn concerning survival data. The authors advocate clear evidence‐based guidelines regarding preoperative imaging for CRLM. This study and the upcoming CAMINO trial can be used to revise the Dutch, and maybe international, guidelines.
The present study has several limitations. First, the disadvantage of the audit data may be accuracy, design and selection of patients. Details including information on the timing of registration of tumour characteristics, multidisciplinary meetings and outcomes of these meetings were missing and could not be retrieved in this retrospective study. The denominator (the sum of patients treated surgically and those treated otherwise) was unclear. Second, it is not mandatory to register open‐and‐close procedures in the DHBA. This makes it difficult to evaluate the impact of the use of preoperative imaging on perioperative outcomes.
The strength of the study is the nationwide collection of data through mandatory participation of all Dutch hospitals performing liver surgery. Because of the nationwide coverage, the results reflect daily clinical practice. It is possible to reflect on how Dutch clinicians use preoperative imaging and to evaluate hospital and oncological network variation.
Trends over the years show increasing use of ceMRI and decreasing use of 18F‐FDG PET–CT for CRLM in the Netherlands. The lack of specific guidelines on preoperative imaging encourages hospital and oncological network variation in the use of ceMRI, 18F‐FDG PET–CT, and combined ceMRI and 18F‐FDG PET–CT. Convincing evidence concerning effective preoperative imaging modalities for CRLM is needed to decrease nationwide variation.
Collaborators
K. Bosscha (Department of Surgery, Jeroen Bosch Hospital, Den Bosch, the Netherlands), E. J. T. Belt (Department of Surgery, Albert Schweitzer Hospital, Dordrecht, the Netherlands), M. Vermaas (Department of Surgery, IJsselland Hospital, Capelle a/d Ijssel, the Netherlands), H. A. Marsman (Department of Surgery, OLVG, Amsterdam, the Netherlands), N. T. van Heek (Department of Surgery, Gelderse Vallei, Ede, the Netherlands), S. J. Oosterling (Department of Surgery, Spaarne Gasthuis, Hoofddorp, the Netherlands), H. Torrenga (Department of Surgery, Deventer Hospital, Deventer, the Netherlands), E. R. Manusama (Department of Surgery, Medical Centre Leeuwarden, Leeuwarden, the Netherlands), I. Somers (Department of Radiology, Meander Medical Centre, Amersfoort, the Netherlands), J. Hagendoorn (Departments of Surgery, University Medical Centre Utrecht, Utrecht, and St Antonius Hospital, Nieuwegein, the Netherlands).
Dutch Hepato‐Biliary Audit Group members: M. T. de Boer (Department of Surgery, University Medical Centre Groningen, Groningen, the Netherlands), R.‐J. Swijnenburg (Department of Surgery, Amsterdam UMC, Cancer Centre Amsterdam, University of Amsterdam, the Netherlands), C. H. C. Dejong (Department of Surgery, Maastricht University Medical Centre, Maastricht, the Netherlands), T. H. van Gulik (Department of Surgery, Amsterdam UMC, Cancer Centre Amsterdam, University of Amsterdam, the Netherlands), F. J. H. Hoogwater (Department of Surgery, University Medical Centre Groningen, Groningen, the Netherlands), I. Q. Molenaar (Department of Surgery, University Medical Centre Utrecht, Utrecht, the Netherlands), O. M. van Delden (Department of Radiology, Amsterdam University Medical Centre, University of Amsterdam, the Netherlands), C. van der Leij (Department of Radiology, Maastricht University Medical Centre, Maastricht, the Netherlands), A. Moelker (Department of Radiology, Erasmus MC Cancer Institute, Rotterdam, the Netherlands), W. Prevoo (Department of Radiology, OLVG, Amsterdam, the Netherlands).
Supporting information
Appendix S1: Supporting information
Acknowledgements
The authors thank all surgeons, interventional radiologists and administrative nurses for data registration in the DHBA database, as well as the Dutch Hepato‐Biliary Audit Group for scientific input.
Disclosure: The authors declare no conflict of interest.
Funding information
No funding
References
- 1. Netherlands Comprehensive Cancer Organization (IKNL). Dutch Cancer Registry; 2018. https://www.iknl.nl/kankersoorten/darmkanker/registratie/incidentie [accessed 27 September 2019]. [Google Scholar]
- 2. Takahashi H, Kahramangil B, Kose E, Berber E. A comparison of microwave thermosphere versus radiofrequency thermal ablation in the treatment of colorectal liver metastases. HPB (Oxford) 2018; 20: 1157–1162. [DOI] [PubMed] [Google Scholar]
- 3. Mitchell D, Puckett Y, Nguyen QN. Literature review of current management of colorectal liver metastasis. Cureus 2019; 11: e3940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Maffione AM, Lopci E, Bluemel C, Giammarile F, Herrmann K, Rubello D. Diagnostic accuracy and impact on management of 18F‐FDG PET and PET/CT in colorectal liver metastasis: a meta‐analysis and systematic review. Eur J Nucl Med Mol Imaging 2015; 42: 152–163. [DOI] [PubMed] [Google Scholar]
- 5. Parikh U, Marcus C, Sarangi R, Taghipour M, Subramaniam RM. FDG PET/CT in pancreatic and hepatobiliary carcinomas: value to patient management and patient outcomes. PET Clin 2015; 10: 327–343. [DOI] [PubMed] [Google Scholar]
- 6. Choi SH, Kim SY, Park SH, Kim KW, Lee JY, Lee SS et al Diagnostic performance of CT, gadoxetate disodium‐enhanced MRI, and PET/CT for the diagnosis of colorectal liver metastasis: systematic review and meta‐analysis. J Magn Reson Imaging 2018; 47: 1237–1250. [DOI] [PubMed] [Google Scholar]
- 7. Schulz A, Viktil E, Godt JC, Johansen CK, Dormagen JB, Holtedahl JE et al Diagnostic performance of CT, MRI and PET/CT in patients with suspected colorectal liver metastases: the superiority of MRI. Acta Radiol 2016; 57: 1040–1048. [DOI] [PubMed] [Google Scholar]
- 8. Sivesgaard K, Larsen LP, Sorensen M, Kramer S, Schlander S, Amanavicius N et al Diagnostic accuracy of CE‐CT, MRI and FDG PET/CT for detecting colorectal cancer liver metastases in patients considered eligible for hepatic resection and/or local ablation. Eur Radiol 2018; 28: 4735–4747. [DOI] [PubMed] [Google Scholar]
- 9. Niekel MC, Bipat S, Stoker J. Diagnostic imaging of colorectal liver metastases with CT, MR imaging, FDG PET, and/or FDG PET/CT: a meta‐analysis of prospective studies including patients who have not previously undergone treatment. Radiology 2010; 257: 674–684. [DOI] [PubMed] [Google Scholar]
- 10. Granata V, Fusco R, de Lutio di Castelguidone E, Avallone A, Palaia R, Delrio P et al Diagnostic performance of gadoxetic acid‐enhanced liver MRI versus multidetector CT in the assessment of colorectal liver metastases compared to hepatic resection. BMC Gastroenterol 2019; 19: 129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Sibinga Mulder BG, Visser K, Feshtali S, Vahrmeijer AL, Swijnenburg RJ, Hartgrink HH et al Gadoxetic acid‐enhanced magnetic resonance imaging significantly influences the clinical course in patients with colorectal liver metastases. BMC Med Imaging 2018; 18: 44. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Moulton CA, Gu CS, Law CH, Tandan VR, Hart R, Quan D et al Effect of PET before liver resection on surgical management for colorectal adenocarcinoma metastases: a randomized clinical trial. JAMA 2014; 311: 1863–1869. [DOI] [PubMed] [Google Scholar]
- 13. Metser U, Halankar J, Langer D, Mohan R, Hussey D, Hadas M et al Effect of chemotherapy on the impact of FDG‐PET/CT in selection of patients for surgical resection of colorectal liver metastases: single center analysis of PET‐CAM randomized trial. Ann Nucl Med 2017; 31: 153–162. [DOI] [PubMed] [Google Scholar]
- 14. Nielsen K, Scheffer HJ, Pieters IC, van Tilborg AA, van Waesberghe JH, Oprea‐Lager DE et al The use of PET–MRI in the follow‐up after radiofrequency‐ and microwave ablation of colorectal liver metastases. BMC Med Imaging 2014; 14: 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15. Lake ES, Wadhwani S, Subar D, Kauser A, Harris C, Chang D et al The influence of FDG PET–CT on the detection of extrahepatic disease in patients being considered for resection of colorectal liver metastasis. Ann R Coll Surg Engl 2014; 96: 211–215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Tamandl D, Ba‐Ssalamah A, Bohm G, Emmanuel K, Forstner R, Függe R et al Austrian consensus guidelines on imaging requirements prior to hepatic surgery and during follow‐up in patients with malignant hepatic lesions. Wien Klin Wochenschr 2018; 130: 665–672. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Garden OJ, Rees M, Poston GJ, Mirza D, Saunders M, Ledermann J et al Guidelines for resection of colorectal cancer liver metastases. Gut 2006; 5: iii1–iii8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Guideline Development Group, NICE. Colorectal Cancer: Diagnosis and Management; 2011. https://www.nice.org.uk/guidance/ng151 [accessed 27 September 2019]. [Google Scholar]
- 19. Hashiguchi Y, Muro K, Saito Y, Ito Y, Ajioka Y, Hamaguchi T et al; Japanese Society for Cancer of the Colon and Rectum. Japanese Society for Cancer of the Colon and Rectum (JSCCR) guidelines 2019 for the treatment of colorectal cancer. Int J Clin Oncol 2019; 25: 1–42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Van Cutsem E, Cervantes A, Adam R, Sobrero A, Van Krieken JH, Aderka D et al ESMO consensus guidelines for the management of patients with metastatic colorectal cancer. Ann Oncol 2016; 27: 1386–1422. [DOI] [PubMed] [Google Scholar]
- 21. Bipat S, van Leeuwen MS, Ijzermans JN, Comans EF, Planting AS, Bossuyt PM et al Evidence‐based guideline on management of colorectal liver metastases in the Netherlands. Neth J Med 2007; 65: 5–14. [PubMed] [Google Scholar]
- 22. Oncoline Dutch Tumour Database . Dutch Guideline – Colorectal Carcinoma; 2014. https://www.oncoline.nl/colorectaalcarcinoom [accessed 27 September 2019].
- 23. van der Werf LR, Kok NFM, Buis CI, Grünhagen DJ, Hoogwater FJH, Swijnenburg RJ et al; Dutch Hepato Biliary Audit Group. Implementation and first results of a mandatory, nationwide audit on liver surgery. HPB (Oxford) 2019; 21; 1400–1410. [DOI] [PubMed] [Google Scholar]
- 24.Statistics Netherlands (CBS). [Geographical and Population Overview of the Netherlands]; 2019. https://www.cbs.nl/nl-nl/maatschappij/bevolking [acccessed 27 September 2019].
- 25.National Institute for Public Health and the Environment (RIVM). [Regional and Academic Hospital Distribution in the Netherlands]; 2019. https://www.volksgezondheidenzorg.info/onderwerp/ziekenhuiszorg/regionaal-internationaal/locaties [accessed 27 September 2019].
- 26.SONCOS. Multidisciplinaire Normering Oncologische Zorg in Nederland; 2019. https://www.soncos.org/wp-content/uploads/2017/10/43SONCOS-normeringsrapport-versie-5.pdf [accessed 27 September 2019].
- 27. van der Werf LR, Voeten SC, van Loe CMM, Karthaus EG, Wouters MWJM, Prins HA. Data verification of nationwide clinical quality registries. BJS Open 2019; 3: 857–864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Elfrink AKE, Kok NFM, van der Werf LR, Krul MF, Marra E, Wouters MWJM et al; Dutch Hepato Biliary Audit Group; Collaborators. Population‐based study on practice variation regarding preoperative systemic chemotherapy in patients with colorectal liver metastases and impact on short‐term outcomes. Eur J Surg Oncol 2020; https://doi:10.1016/j.ejso.2020.03.221 [Epub ahead of print]. [DOI] [PubMed] [Google Scholar]
- 29. Pech L, Cercueil JP, Jooste V, Krause D, Facy O, Bouvier AM. Current use of MRI in patients with liver metastatic colorectal cancer: a population‐based study. Eur J Gastroenterol Hepatol 2017; 29: 1126–1130. [DOI] [PubMed] [Google Scholar]
- 30. Sjovall A, Blomqvist L, Martling A. Pretreatment staging of colon cancer in the Swedish population. Colorectal Dis 2013; 15: 1361–1366. [DOI] [PubMed] [Google Scholar]
- 31. Rojas Llimpe FL, Di Fabio F, Ercolani G, Giampalma E, Cappelli A, Serra C et al Imaging in resectable colorectal liver metastasis patients with or without preoperative chemotherapy: results of the PROMETEO‐01 study. Br J Cancer 2014; 111: 667–673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. van Kessel CS, Buckens CF, van den Bosch MA, van Leeuwen MS, van Hillegersberg R, Verkooijen HM. Preoperative imaging of colorectal liver metastases after neoadjuvant chemotherapy: a meta‐analysis. Ann Surg Oncol 2012; 19: 2805–2813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Patel S, McCall M, Ohinmaa A, Bigam D, Dryden DM. Positron emission tomography/computed tomographic scans compared to computed tomographic scans for detecting colorectal liver metastases: a systematic review. Ann Surg 2011; 253: 666–671. [DOI] [PubMed] [Google Scholar]
- 34. Lopez‐Lopez V, Robles R, Brusadin R, López Conesa A, Torres J, Perez Flores D et al Role of 18F‐FDG PET/CT vs CT‐scan in patients with pulmonary metastases previously operated on for colorectal liver metastases. Br J Radiol 2018; 91: 20170216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35. Bipat S, van Leeuwen MS, Ijzermans JN, Bossuyt PM, Greve JW, Stoker J. Imaging and treatment of patients with colorectal liver metastases in the Netherlands: a survey. Neth J Med 2006; 64: 147–151. [PubMed] [Google Scholar]
- 36. Sistrom CL, McKay NL. Costs, charges, and revenues for hospital diagnostic imaging procedures: differences by modality and hospital characteristics. J Am Coll Radiol 2005; 2: 511–519. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Appendix S1: Supporting information


