Abstract
Aim: Matrix metalloproteinases (MMPs), angiotensin II (AII) and its receptors are implicated in atherosclerotic plaque instability, however the roles of the two receptor subtypes, ATR1 and ATR2, in MMP regulation remain uncertain. In this study, we investigated the effect of ATR1 and ATR2 blockade on the expression and activity of MMP-2, MMP-3 and MMP-9, in human carotid atheroma.
Methods: Atheroma samples (n = 36) were obtained from patients undergoing carotid endarterectomy. The effects of ATR1 (irbesartan), ATR2 (PD123319) and combined ATR1 and ATR2 blockade on the expression and activity of the MMPs and the expression of tissue inhibitors of metalloproteinases (TIMPs) were investigated in explant culture experiments. Paired atheroma samples were incubated with the intervention or media control for 4 days. Protein levels (MMP-2, MMP-3, MMP-9, TIMP-1, TIMP-2, TIMP-4, ATR1 and ATR2) were determined by ELISA. Overall gelatinase activity and specific activation were measured by chromogenic activity assays and zymography, respectively.
Results: ATR1 blockade, but not ATR2 blockade significantly reduced TIMP-1, TIMP-2 and TIMP-4 expression in atheroma supernatant. Combined ATR1 and ATR2 blockade significantly reduced MMP-2, MMP-3 and MMP-9 expression. MMP-2 and MMP-9 relative activation, and overall MMP-9 catalytic capacity were significantly increased by ATR1 blockade.
Conclusions: Our findings suggest that ATR1 blockade reduces TIMP expression and increases gelatinase activity in human carotid atheroma.
Keywords: Matrix metalloproteinase, Gelatinase, Tissue inhibitor of matrix metalloproteinase, Angiotensin II receptor, Atheroma
See editorial vol. 23: 766–768
Introduction
The majority of ischemic events, including stroke, are believed to be preceded by rupture of the fibrous cap lining the atherosclerotic plaque1). Fibrillar interstitial collagen is considered a key stabilising component of the fibrous cap and collagen degradation has been implicated in plaque erosion and rupture2). The matrix metalloproteinases (MMPs) are a family of proteases that have been implicated in promoting plaque rupture due to their capacity to degrade all the components of extracellular matrix (ECM), including fibrillar interstitial collagen, at neutral pH3). It is hypothesised that depletion of ECM components, particularly fibrillar interstitial collagens, from the fibrous cap favours an imbalance towards breakdown over synthesis, leading to cap thinning and rupture4).
However, MMPs have further functions beyond ECM degradation. MMPs are also involved in the proteolysis of a range of non-matrix substrates and cell surface molecules giving them roles in such processes as normal development, tissue morphogenesis and wound repair5) and only being expressed when needed for these processes6). MMPs directly modify cell to matrix or cell to cell interactions as well as activation, release and inactivation of signalling molecules and their cell-surface receptors7, 8). Atherosclerotic plaques removed from asymptomatic patients or those with more stable symptoms are reported to have a thicker fibrous cap with relatively abundant vascular smooth muscle cells (SMCs) within them. The fibrous cap SMCs are believed to produce ECM proteins, such as fibrillar collagen, providing structural support1).
The gelatinase subfamily, comprising, MMP-2 and -9, most efficiently cleave denatured collagens (rather than structural fibrillar collagens) and elastin9) and have been implicated as key players in the proliferation and migration of SMCs from the media to the fibrous cap thereby possibly providing plaque stabilising effects10). MMP-3 mediated activation of MMP-9 has also been implicated in efficient SMC migration and neointima formation in mice11). The development of an unstable plaque phenotype in a murine model with less SMCs relative to macrophages was also favoured by broad spectrum inhibition of MMP activity12).
MMPs are now recognised as having counteracting roles with regards to plaque stability, on the one hand they facilitate intimal thickening (particularly gelatinases and MMP-3), which potentially stabilises plaques, while on the other hand they promote ECM destruction, which potentially destabilises plaques (particularly interstitial collagenases) (Fig. 1a)13).
Fig. 1.
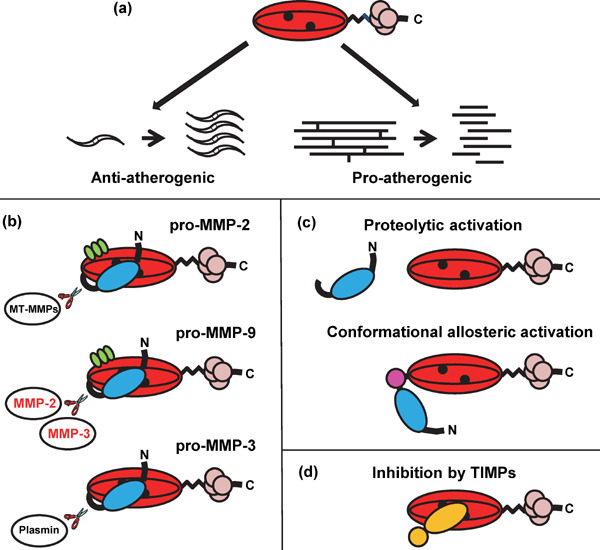
Structure and function of MMPs and role in atherosclerosis. (a) MMPs are potentially involved in SMC migration and proliferation (anti-atherogenic activity) and also collagen and elastin degradation (pro-atherogenic activity) within the atheroma. (b) Domain structure for the MMPs. The archetypal domain format is an N-terminal signal sequence+propeptide (blue) which blocks substrate access to the active site by forming a bond between the Zn2+ ions (black) in the catalytic site and Cys 73 in the propeptide (cysteine “switch”) + catalytic domain (red) + hinged region + 4 hemopexin domains (pink) + tail sequence. MMP-3 has this domain format, whereas MMP-2 and MMP-9 have 3 extra fibronectin type II regions within the catalytic domain (green). Key enzymes that can activate the MMPs are also shown including family members investigated in this study (red text). (c) MMPs are heavily regulated with activation occurring when the regulating Cys-Zn2+ bond is broken exposing the catalytic site. This can be by protease degradation or allosteric means. (d) Inhibition occurs when TIMPs bind to the active site. All the TIMPs are capable of binding to all the MMPs. The overall catalytic capacity of the MMPs is a finely tuned balance between all these factors.
The MMPs are secreted as zymogens (Fig. 1b), these are latent forms activated by removal of the N terminal pro-domain (Fig. 1c). A number of MMP family members can also behave as activating proteases (Fig. 1b)14). MMPs are also inhibited by the tissue inhibitors of metalloproteinases (TIMPs) (Fig. 1d)15). The overall catalytic capacity of the MMPs is a finely tuned balance between expression, activation and inhibition16). MMP-2, MMP-9, MMP-3 (capable of activating MMP-9) and the TIMPs have been reported to be expressed in atherosclerotic plaques. MMP-3 and -9 have been identified in the vulnerable shoulder region of the plaque16).
Angiotensin II (AII) signalling has been implicated in the progression of atherosclerosis17) and associated with gelatinase and TIMP regulation18, 19). Several clinical trials have demonstrated the benefits of AII blockade using anti-hypertensive drugs in reducing acute cardiovascular events, independent of blood pressure reduction20–22). There are at least 2 receptors for AII present in atheroma, angiotensin receptor type 1 (ATR1)23) and 2 (ATR2)24). Signalling via both receptors has been reported to regulate MMP expression with most studies suggesting ATR1 mediates MMP up-regulation18).
ATR1 blockade leads to increased levels of AII both in vivo and in culture25–28). The physiological role of ATR2, which has minimal, restricted expression in healthy adults29), but is present in atheroma24), is only partially understood with suggestions it opposes ATR1 or alternately has a synergistic role with ATR1 blockade30). The impact of increased signalling through ATR2 with ATR1 blockade has not been fully determined31). The underlying regulatory mechanisms behind the pleomorphic beneficial outcomes of ATR1 blockade in atherosclerosis patients are also yet to be elucidated. In this study we aimed to clarify the roles of ATR1 and ATR2 in gelatinase regulation in carotid atheroma using paired human carotid atheroma explant culture28, 32–35) and ATR blockers specific to type 1 or type 2.
Methods
Patients
Patients were recruited from those selected to undergo primary carotid endarterectomy at The Townsville and Mater Hospitals, Queensland, Australia between Aug 2004 and Jan 2013. Inclusion criteria included: 1) verbal and written informed consent; 2) carotid stenosis of ≥ 70% identified on duplex imaging using criteria previously described34); and 3) no previous carotid artery intervention. Ethics approval was provided by the appropriate committees (James Cook University, H2196; Townsville Health Service District, 61/05). Patients with a history of transient ischemic attack (TIA), stroke with good recovery and those with an asymptomatic stenosis were included. Patient details are presented in Supplemental Table 1.
Supplemental Table 1. Patient characteristics.
| Characteristic | ATR1 blockade | ATR2 and ATR1/2 dual blockade | ERK1/2 blockade | ATR and angiotensin peptide correlation |
|---|---|---|---|---|
| Number of patients, n | 6 | 6 | 6 | 18 |
| Number of samples, n | 28 | 24 | 24 | 36 |
| Number of matched pairs, n | 14 | 12 | 12 | |
| Age, y, mean ± SD | 73.8 ± 5.4 | 73.8 ± 5.8 | 68.9 ± 7.6 | 70.5 ± 7.9 |
| Male, n | 5 | 3 | 4 | 12 |
| TIA, n | 3 | 3 | 3 | 8 |
| Stroke, n | 1 | 2 | 1 | 4 |
| Asymptomatic, n | 0 | 2 | 2 | 5 |
| Diabetes, n | 1 | 3 | 1 | 3 |
| Hypertension, n | 6 | 5 | 4 | 17 |
| CHD, n | 5 | 2 | 1 | 5 |
| Non-smoker, n | 1 | 1 | 1 | 1 |
| ARB, n | 0 | 1 | 0 | 7 |
| ACEi, n | 5 | 3 | 2 | 9 |
TIA, Transient Ischaemic Attack; CHD, Coronary Heart Disease; ATR, Angiotensin II receptor; Angiotensin Receptor Blocker, ARB; Angiotensin Converting Enzyme inhibitor, ACEi
Carotid Atheroma Samples
A conventional endarterectomy was performed, developing a deep medial plane and removing the specimen en bloc34). Specimens were placed in chilled culture medium immediately after excision, and transferred chilled to the laboratory for processing. Specimens were dissected aseptically for explant culture28).
Explant Culture
Explant culture was carried out as described previously28, 34). Adjacent biopsies were obtained from endarterectomy samples (at least two macroscopically similar disease matched pairs were obtained from each of the six patients selected). We have previously established that: i) Carotid endarterectomy samples remain viable in culture for at least 8 days, as assessed by histology, immunohistochemistry, cytokine release and measurement of tissue ATP36); ii) Adjacent paired biopsies have similar features of plaque instability in vitro. Tissue factor (TF) concentration and activity were similar in adjacent biopsies with a coefficient of variation for TF activity < 5% (n = 6)34); iii) The paired biopsies responded to ATR1 and angiotensin converting enzyme (ACE)1 blockade in keeping with the in vivo response to these drugs with an increase in AII levels in the culture supernatant28) and a decrease in cytokine expression with ATR1 blockade32). Paired adjacent disease matched biopsies were cultured with media containing an intervention agent and the matched control sample was incubated with media alone. The concentration of interventional agents used were based on circulating concentrations measured in patients receiving therapeutic levels of the drug or published effective in vitro concentrations. Therapeutic circulating concentrations of irbesartan are reported to be between ∼1–10 mmol/L25, 37). Pharmacokinetic studies suggest that the effective inhibitory dose of PD123319 for use in in vitro culture is 0.1–1 µmol/L38). The MKK1/2 inhibitor, PD98059, binds directly to MKK1/2 blocking it's capacity to phosphorylate ERK1/2, consequently blocking ERK1/2 activation39). When used at a concentration of 20 µmol/L, PD98059, abolished AII induced upregulation of C-reactive protein from vascular SMCs40) and expression of interleukin (IL)-6, IL-8, C-C motif ligand (CCL)-2 and C-X-C- motif ligand (CXCL)-5 in cultured human atheroma tissue32). The following interventions were assessed: a) ATR1 blocker (Irbesartan; 2.3 mmol/L; Sigma #I2286); b) ATR2 blocker (PD123319; 1 µmol/L; Sigma #P186); c) Combined ATR1 and ATR2 blocker (Irbesartan; 2.3 mmol/L; Sigma #I2286 and PD123319; 1 µmol/L; Sigma #P186); d) ERK1/2 (PD98059; 20 µmol/L; Sigma #P215).
Primary Vascular Cell Culture
Due to the nature of the carotid endarterectomy procedure the types of vascular cells present in the explant tissue include: endothelial cells (ECs); SMCs; inflammatory cells which have infiltrated the atheroma from the blood; and red blood cells (RBCs) on the tissue surface. A previously developed mixed, healthy, primary vascular cell technique32, 33) was used to assess the impact of ATR blockade on MMP-9 production from healthy vascular cells stimulated by serum, which would not be expected to have ATR2 expression. Irbesartan (0, 0.144, 0.575, 2.3 and 4.6 mmol/L) and PD123319 (0, 0.063, 0.25, 1 and 2 µM) were titrated in the mixed, healthy, primary vascular cell cultures. The blood was collected from four healthy, male volunteers and pooled. Verbal and written, informed consent was received. Ethics approval for these samples was provided by the appropriate committee (James Cook University, H4109).
ELISA
Commercially available ELISA kits were used to measure expression of the gelatinases, MMP-3, the TIMPs (R&D Systems), AII and AIII (USCN) in the explant conditioned supernatant; MMP-9 in the mixed, healthy, primary vascular cell culture supernatant; and activated ERK1-P, activated ERK2-P (R&D Systems), ATR1 and ATR2 (USCN) in the explant tissue, following the manufacturer's instructions. Each of the MMP kits measured latent, active and inhibited forms combined (Fig. 1b–d).
Previously, atheroma supernatant concentrations of AII were assessed using an AII ELISA that cross reacts with AII-A (also called des [Asp1]-[Ala1]-AII. In this form the Asp is swapped to an Ala at position 141)) and AIII28). As AII can potentially be converted to the other peptides by enzymes present in the culture supernatant this ELISA effectively measured a combination of all the AII, AII-A and AIII present in the atheroma culture supernatant28). Individual AII and AIII levels were assessed in the current study with ELISAs specific for each of the individual peptides.
The appropriate conditions for each assay were determined by sample titration and are listed in the Supplemental methods at the end of the manuscript.
Protein Quantification
Following completion of the explant studies atheroma tissue was processed as described previously to determine the total protein content of each cultured sample for normalising purposes28). These extracts were also used in the ATR1, ATR2, ERK1-P and ERK2-P ELISAs, and ERK1/2 Western blots.
Western Blotting
The atheroma tissue proteins (30 µg total protein/10 µL load) were separated under denaturing conditions on a 12% SDS polyacrylamide gel42). The ECL Advance Western Blotting Detection kit (Amersham, #RPN2135) was used for visualisation, as per the manufacturer's instructions. Briefly, the separated proteins were transferred onto PVDF membrane (Biorad, #162-0177) and probed with 0.1 µg/mL rabbit polyclonal antibody against ERK1 and ERK2 (Calbiochem, #442704) and 1/2000 goat anti-rabbit HRP secondary (Dako, #P0448). Bands migrating at ∼42 and 44kD were digitally captured using a Chemidoc XRS (Biorad) and band volume (intensity units*mm2) determined using Quantity One software (Biorad).
Zymography
Explant conditioned supernatant removed from atheroma exposed to interventional agent for 4 days and controls were assessed for relative MMP-2 and MMP-9 activation using gelatin zymography as described previously43). Briefly, 5 µL of supernatant (prepared in 4x non-reducing, SDS loading buffer) was resolved in 9% SDS PAGE with 1 mg/mL gelatin added (200 V, 55 min). The resolved gel was rinsed twice in 2.5% triton-X 100 (15 min) then incubated for 21h in substrate buffer (50 mmol/L Tris pH 8, 5 mmol/L CaCl2) at 37°C. The gel was stained with Coomassie blue (0.1%w/v Coomassie blue R-250, 40%v/v methanol, 10%v/v glacial acetic acid) for 30 min at room temperature and destained with 40% v/v methanol, 10% v/v glacial acetic acid. Band volumes (intensity units*mm2) for the latent (72kD band for MMP-2 and 92kD band for MMP-9) and active gelatinases (62kD band for MMP-2 and 82kD band for MMP-9), were digitally captured and inverted using a Chemidoc XRS (Biorad). Band volume was determined using Quantity One software (Biorad) and assessment of relative gelatinase activation was determined from a ratio of the active band volume (Fig. 1c) to latent band volume (Fig. 1b)43).
Chromogenic Activity Assay
Commercially available activity kits were used to measure the overall catalytic capacity of MMP-2 and -9 in the explant conditioned supernatant following the manufacturer's instructions (Quickzyme). These kits only detect proteolytically cleaved and activated MMPs, which are not complexed to a TIMP (Fig. 1c). The appropriate conditions for each assay were determined by sample titration. Based on these preliminary studies the following volumes and dilutions of conditioned media were assayed: MMP-2 (20 µL supernatant; #QZBmmp2H) and MMP-9 (100 µL supernatant; #QZBmmp9H). As the explant culture system is a closed system, the total amount of active protein was determined for each sample and used in the subsequent analyses.
Data Analysis
MMP-2, -3, -9, TIMP-1, -2, -4, ATR1, ATR2, ERK1-P, ERK2-P and ERK1/2 expression and MMP-2 and -9 relative activation and overall catalytic capacity in the paired explant samples are presented as mean ± SEM for the relative ratios of paired experimental and control samples (n = 14 or 12 pairs)28, 32–35). Differences between experimental and control samples were assessed using Wilcoxon's paired test in SPSS v21 software. Values for experimental samples > 1 implied the intervention enhanced activity or expression while values < 1 implied the intervention reduced activity or expression and values = 1 implied the intervention had no effect. Correlations of expression of various proteins in the explant cultures were assessed using Spearman's rho correlation. A p value of < 0.05 was considered statistically significant. The mean ± SEM protein levels across all the control samples were also presented to demonstrate the inherent heterogeneity between the individual patients.
Results
Effect of ATR Blockers on MMP Expression in Human Carotid Atheroma Supernatant
The expression of MMP-2, -3 and -9 present in the human carotid atheroma supernatant were measured using ELISA. All the MMPs were detectable in the culture supernatant. The mean normalised amount of MMP ± SEM in the control culture supernatants (n = 38) were as follows: MMP-2, 275.5 ± 38.16 ranging from 13.1–862.5 ng/mg; MMP-3, 9.207 ± 1.59 ranging from 0.61–46.50 ng/mg; MMP-9, 77.7 ± 16.0 ranging from 2.7–515.9 ng/mg (Fig. 2a).
Fig. 2.
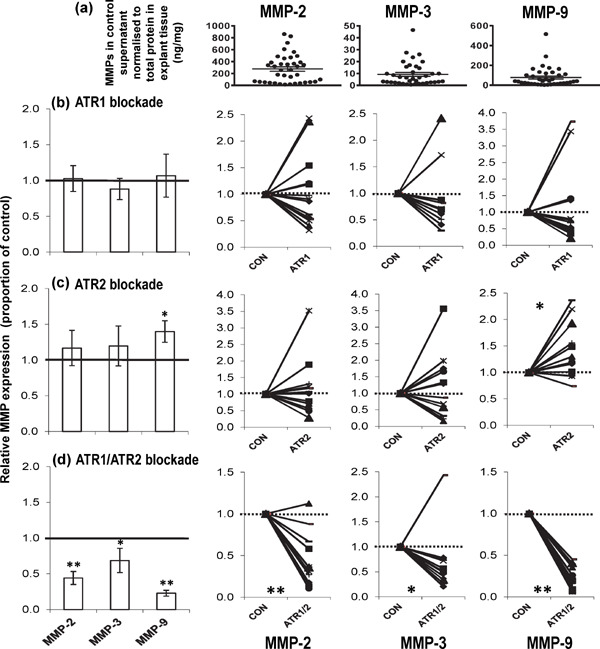
Changes in MMP expression following angiotensin receptor blockade. Mean ± SEM of (a) MMP-2, MMP-3 and MMP-9 detected in the control culture supernatant, normalised to the amount of protein present in the explant tissue (n = 38, control samples from the 3 interventions). Relative expression of MMPs in human carotid atheroma supernatant after 4d culture with and without (b) ATR1 (irbesartan, 2.3 mmol/L, n = 14 pairs); (c) ATR2 (PD123319, 1 µmol/L, n = 12 pairs); and (d) ATR1/ATR2 (irbesartan, 2.3 mmol/L and PD123319, 1 µmol/L, n = 12 pairs) blockade. Shown are the mean values ± SEM of the ratio of enzyme (normalised to total protein) secreted by paired atheroma samples incubated with intervention relative to control. Also shown are the individual matched pairs. Total MMP expression was determined using ELISA. **P < 0.01; *P < 0.05 using Wilcoxon's paired test. (1 = no change, > 1 increased, < 1 decreased with intervention)
Incubation of atheroma samples with irbesartan (ATR1 specific inhibitor, 2.3 mmol/L) for 4d had no significant impact on the expression of any of the MMPs measured in the culture supernatant (P > 0.05; n = 14 pairs) (Fig. 2b). Incubation of the atheroma samples with PD123319 (ATR2 specific inhibitor, 1 µmol/L) for 4d caused a weakly significant upregulation in expression of MMP-9 in the culture supernatant (∼1.4-fold change, P = 0.0186; n = 12 pairs) (Fig. 2c). Interestingly, MMP-2, -3 and -9 expression in the culture supernatant were significantly downregulated by combined incubation of the atheroma sample with irbesartan and PD123319 for 4d (∼0.44-fold change, P = 0.0037; ∼0.53-fold change, P = 0.0342; ∼0.23-fold change, P = 0.0022, respectively; n = 12 pairs) (Fig. 2d).
Effect of ATR Blockers on TIMP Expression in Human Carotid Atheroma Supernatant
The expression of TIMP-1, -2 and -4 present in the human carotid atheroma supernatant were measured by ELISA. All the TIMPs were detectable in the supernatant. The mean normalised amount of TIMP ± SEM in the control supernatants (n = 50) were as follows: TIMP-1, 3682 ± 370 ranging from 100–10718 ng/mg; TIMP-2, 52.9 ± 3.8 ranging from 6.1–110.1 ng/mg; TIMP-4, 248.9 ± 26.0 ranging from 25.9–830.0 pg/mg (Fig. 3a).
Fig. 3.
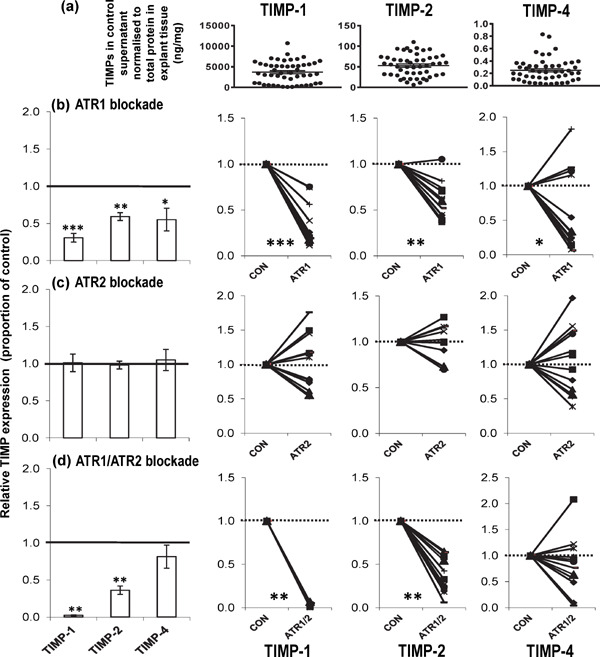
Changes in TIMP expression following angiotensin receptor blockade. Mean ± SEM of (a) TIMP-1, TIMP-2 and TIMP-4 detected in the control culture supernatant, normalised to the amount of protein present in the explant tissue (n = 50, control samples from the 4 interventions). Relative expression of TIMPs in human carotid atheroma supernatant after 4d culture with and without (b) ATR1 (irbesartan, 2.3 mmol/L, n = 14 pairs); (c) ATR2 (PD123319, 1 µmol/L, n = 12 pairs); and (d) ATR1/ATR2 (irbesartan, 2.3 mmol/L and PD123319, 1 µmol/L, n = 12 pairs) blockade. Shown are the mean values ± SEM of the ratio of enzyme (normalised to total protein) secreted by paired atheroma samples incubated with intervention relative to control. Also shown are the individual matched pairs. TIMP expression was determined using ELISA. ***P < 0.001; **P < 0.01; *P < 0.05 using Wilcoxon's paired test. (1 = no change, > 1 increased, < 1 decreased with intervention)
Incubation of the atheroma samples with irbesartan (2.3 mmol/L) for 4d significantly reduced TIMP-1, -2 and -4 expression in the culture supernatant (∼0.31-fold change, P = 0.0010; ∼0.59-fold change, P = 0.0012; ∼0.55-fold change, P = 0.0186, respectively; n = 14 pairs) (Fig. 3b), but not PD123319 (1 µmol/L) incubation (P > 0.05; n = 12 pairs) (Fig. 3c). Similarly, incubation of the atheroma samples with combined irbesartan and PD123319 for 4d significantly reduced the expression of TIMP-1 and -2 in the culture supernatant (∼0.02-fold change, P = 0.0022; ∼0.36-fold change, P = 0.0022, respectively; n = 12 pairs) (Fig. 3d).
Effect of ATR Blockers on Relative Gelatinase Activation in Human Carotid Atheroma Supernatant
The gelatinolytic activity of the latent (uncleaved) and active (cleaved) forms of MMP-2 and MMP-9 were assessed by zymography. Relative activation was determined by dividing the active band volume by the latent band volume. The mean normalised relative gelatinase activation ± SEM in the control supernatants were as follows: MMP-2 relative activation, 0.709 ± 0.084 ranging from 0.035–1.742 INT* mm2/mg (n = 38); MMP-9 relative activation, 0.678 ± 0.094 ranging from 0.023–2.415 INT*mm2/mg (n = 38) (Fig. 4a).
Fig. 4.
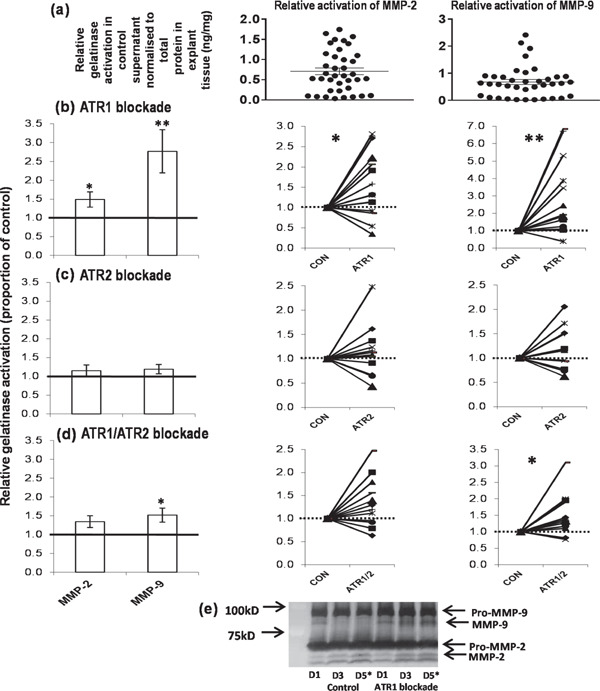
Changes in relative gelatinase activation following angiotensin receptor blockade. Mean ± SEM of (a) relative activated MMP-2 and relative activated MMP-9 detected in the control culture supernatant, normalised to the amount of protein present in the explant tissue (n = 36, control samples from the 3 interventions). Relative activation of MMP-2 and -9 in human carotid atheroma supernatant after 4d culture with and without (b) ATR1 (irbesartan, 2.3 mmol/L, n = 14 pairs), (c) ATR2 (PD123319, 1 µmol/L, n = 12 pairs) and (d) ATR1/ATR2 (irbesartan, 2.3 mmol/L and PD123319, 1 µmol/L, n = 12 pairs) blockade. Shown are the mean values ± SEM of the ratio of cleaved enzyme (Fig. 1c) to pro-enzyme (Fig. 1b) (normalised to total protein) secreted by paired atheroma samples incubated with intervention relative to control. Also shown are the individual matched pairs. (e) Relative levels of gelatinase activation were determined using zymography. The asterisked day 5 samples (D5*) were the ones used in the analyses. NB the day 1 samples (D1) are actually the same for both control and ATR1 blockade as there is an initial 24h “media only” pre-culture step to reduce the impact of any medications the patient may have been taking before implementation of the interventions in culture. This gel also demonstrates that gelatinase expression does not decline in the 5 days of culture as all the control samples are consistent for all three time points. **P < 0.01; *P < 0.05 using Wilcoxon's paired test. (1 = no change, > 1 increased, < 1 decreased with intervention)
Zymography results using culture supernatant revealed that relative activation of MMP-2 and -9 increased significantly in the presence of irbesartan (2.3 mmol/L) (∼1.5-fold change, P = 0.0413; ∼2.8-fold change, P = 0.0029, respectively; n = 14 pairs) after 4d culture (Fig. 4b). Incubation of atheroma samples with PD123319 (1 µmol/L) for 4d did not alter relative gelatinase activation in the culture supernatant (P > 0.05; n = 12 pairs) (Fig. 4c). Combined incubation of the atheroma samples with irbesartan and PD123319 for 4d also weakly enhanced relative MMP-9 activation in the culture supernatant (∼1.5-fold change, P = 0.0121; n = 12 pairs) (Fig. 4d).
Correlation of ATRs with MMPs and TIMPs in Control Human Carotid Atheroma Culture
Tissue expression of ATR1 was significantly correlated to expression of MMP-9 and TIMP-2 in the control atheroma culture supernatant (Spearman's rho = 0.7373, P < 0.0001; Spearman's rho = 0.4280, P = 0.0092, respectively; n = 36) (Table 1). Equally, tissue expression of ATR2 was also correlated to expression of MMP-9 and TIMP-2 in the control atheroma culture supernatant (Spearman's rho = 0.7822, P < 0.0001; Spearman's rho = 0.4561, P = 0.0050, respectively; n = 36) (Table 1).
Table 1. Correlation of MMPs and TIMPs in control explant culture supernatant with ATRs in the control explant tissue.
| ATR1 (n = 36) |
ATR2 (n = 36) |
|||
|---|---|---|---|---|
| Spearman's rho | P value | Spearman's rho | P value | |
| MMP-2 | 0.1139 | 0.5083 | 0.1439 | 0.4025 |
| MMP-3 | 0.0475 | 0.7833 | −0.0893 | 0.6044 |
| MMP-9 | 0.7373**** | < 0.0001 | 0.7822**** | < 0.0001 |
| TIMP-1 | 0.0085 | 0.9629 | 0.1050 | 0.5421 |
| TIMP-2 | 0.4280*** | 0.0092 | 0.4561*** | 0.0050 |
| TIMP-4 | −0.0467 | 0.7867 | −0.1454 | 0.3974 |
Spearman's rho correlations between the MMPs and TIMPs and the two AII receptors were performed using the total amount of each protein in the control sample (n = 36). As the explant culture system is a closed system and the amount of vascular remodelling enzymes present in the culture supernatant are a direct result of the amount of proteins present in the tissue the correlations were determined using the total amount of ATR and enzyme present within each culture well.
P < 0.0001
P < 0.001
P < 0.01
P < 0.05
Scatter curves are presented in Supplemental Figs. 1–2.
Effect of ATR1 Blocker on Overall Gelatinase Catalytic Capacity in Human Carotid Atheroma Supernatant
The concentration of activated, un-complexed MMP-2 and -9 (Fig. 1c) were measured using a chromogenic activity assay. Active MMP-2 and -9 were not consistently detectable in all control culture supernatants. Of the 12 control samples assessed, 9 had detectable active MMP-2 and 8 had detectable active MMP-9. The mean normalised amount of active MMP ± SEM in the control cultures were as follows: catalytically active MMP-2, 0.104 ± 0.033 ranging from 0.020–0.269 ng/mg (n = 9); catalytically active MMP-9, 0.065 ± 0.018 ranging from 0.015–0.152 ng/mg (n = 8) (Fig. 5a). Incubation of atheroma samples with irbesartan (2.3 mmol/L) for 4d, significantly upregulated the catalytic capacity of MMP-9 in the culture supernatant (∼2.6-fold change, P = 0.0499; n = 8 pairs) (Fig. 5b).
Fig. 5.
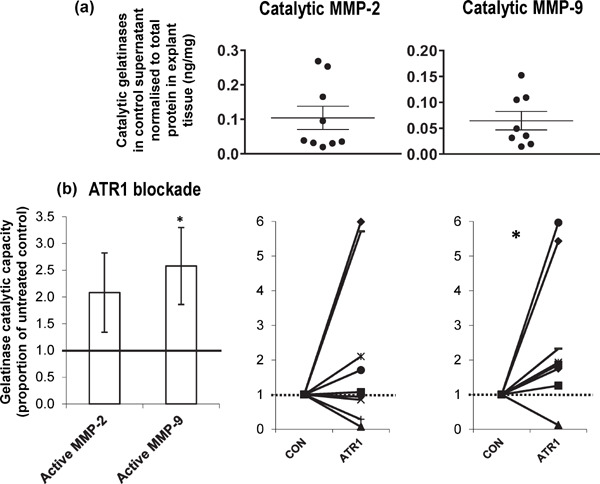
Changes in overall gelatinase catalytic capacity following ATR1 blockade. Mean ± SEM of (a) catalytically active MMP-2 (n = 9) and catalytically active MMP-9 (n = 8) (active MMP-2 and -9 were not detectable in all sample pairs) detected in the control culture supernatant, normalised to the amount of protein present in the explant tissue. Overall catalytic capacity of MMP-2 and -9 in human carotid atheroma supernatant after 4d culture with and without ATR1 blockade (irbesartan, 2.3 mmol/L, n = 9 and 8 pairs, respectively). Shown are the mean values ± SEM of the ratio of active enzyme (normalised to total protein) secreted by paired atheroma samples incubated with intervention relative to control. Also shown are the individual matched pairs. Overall catalytic capacity for each gelatinase was determined by a chromogenic assay, which only detects proteolytically cleaved and activated MMPs that are not complexed to a TIMP (Fig. 1c). *P < 0.05 using Wilcoxon's paired test. (1 = no change, > 1 increased with intervention).
Effect of ATR Blockers on MMP-9 Expression in Mixed, Healthy, Primary Vascular Cells Stimulated by Human Serum in vitro
The level of MMP-9 secreted by healthy, primary vascular cells increased with addition of normal human serum suggesting the serum stimulates the cells to produce MMP-9 (Fig. 6a). Within the mixed culture MMP-9 appears to be predominantly secreted by the cells derived from the blood (Fig. 6a). Titration of irbesartan in the mixed culture (EC+SMC+BC+ serum) downregulated secretion of MMP-9 (Fig. 6b) in a dose dependent manner, with addition of PD123319 having no further impact on MMP-9 expression (Fig. 6c). Titration of PD123319 in the mixed culture had no impact on MMP-9 expression (Fig. 6d).
Fig. 6.

MMP-9 up-regulation in the serum stimulated, mixed, healthy primary vascular cell model. (a) Expression of MMP-9 in the culture supernatant after 4d of culture using various combinations of healthy EC, SMC, BCs and serum. Shown are the mean values from triplicate cultures ± SEM. Expression of MMP-9 in the mixed, healthy primary vascular cell culture (EC, SMC, BC and serum) supernatant after 4d of culture with (b) titrated ATR1 blockade, (b) combined ATR1/ATR2 blockade, and (c) titrated ATR2 blockade. Shown are the mean values from triplicate cultures ± SEM.
Effect of ATR1 Blockade on Levels of ERK Signalling Proteins in Human Carotid Atheroma Tissue
The expression of ERK1/2 present in the human carotid atheroma tissue was measured by western blotting and the levels of ERK1-P and ERK2-P by ELISA. The mean normalised amount of ERK ± SEM in the control tissue samples were as follows: ERK1/2, 6219 ± 1020 ranging from 1857–14980 INT*mm2/mg (n = 14); ERK1-P, 69.9 ± 12.5 ranging from 12.9–129.5 pg/mg (n = 11); ERK2-P, 86.4 ± 15.8 ranging from 12.9–163.9 pg/mg (n = 11) (Fig. 7a).
Fig. 7.
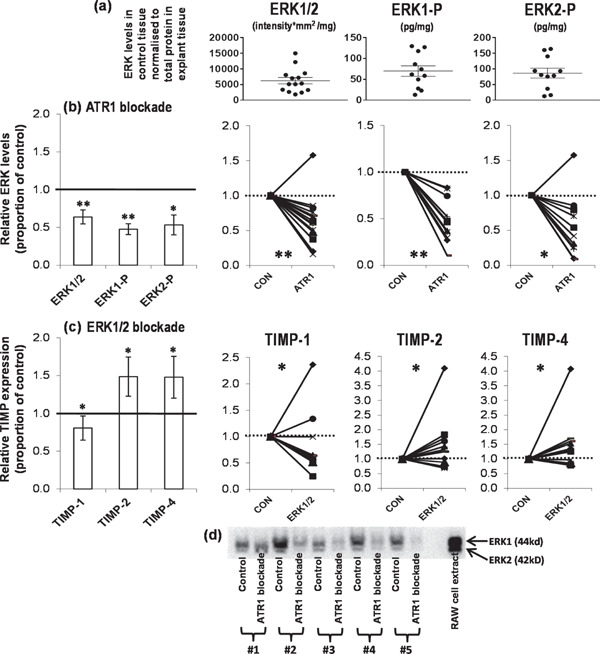
Changes in ERK and TIMP expression following various interventions. Mean ± SEM of (a) ERK1/2, ERK1-P and ERK2-P detected in the control culture tissue, normalised to the amount of total protein present in the explant tissue (n = 14 control samples from the ATR1 intervention). Relative expression of (b) ERKs in human carotid atheroma tissue after 4d culture with and without ATR1 (irbesartan, 2.3 mmol/L, n = 14 pairs) blockade and (c) TIMPs in the human carotid atheroma culture supernatant after 4d culture with and without ERK1/2 activity (PD98059, 20 µmol/L, n = 12 pairs) blockade. Shown are the mean values ± SEM of the ratio of enzyme (normalised to total protein) secreted by paired atheroma samples incubated with intervention relative to control. Also shown are the individual matched pairs. TIMP and activated ERK levels were determined using ELISA. (d) ERK1/2 expression was determined using Western blotting (load 30 µg total protein in 10 µL loading buffer). **P < 0.01; *P < 0.05 using Wilcoxon's paired test. (1 = no change, > 1 increased, < 1 decreased with intervention)
Incubation of the atheroma samples with irbesartan (2.3 mmol/L) for 4d significantly reduced ERK1/2 expression in the cultured tissue (∼0.64-fold change, P = 0.0092; n = 14 pairs). The levels of the activated, phosphorylated forms, ERK1-P and ERK2-P, were also significantly reduced (∼0.48-fold change, P = 0.0033; and ∼0.53-fold change, P = 0.0128, respectively; n = 11 pairs) with irbesartan incubation (Fig. 7b)
Effect of ERK1/2 Activity Blockers on TIMP Expression in Human Carotid Atheroma Supernatant
Incubation of atheroma samples with PD98059 (ERK1/2 specific inhibitor, 20 µmol/L, n = 12 pairs) for 4d significantly reduced expression of TIMP-1 (∼0.81-fold change, P = 0.0499), and increased expression of TIMP-2 (∼1.49-fold change, P = 0.0342) and TIMP-4 (∼1.48-fold change, P = 0.0409) in the culture supernatant (Fig. 7c).
Correlation of Angiotensin Peptides with TIMPs in Control Human Carotid Atheroma Culture
Expression of TIMP-4 was significantly correlated to levels of AII (Spearman's rho = 0.5275, P = 0.0081; n = 24) and expression of TIMP-2 was significantly correlated to levels of AIII and combined AII/AIII/AII-A (Spearman's rho = 0.4340, P = 0.0131, n = 32; Spearman's rho = 0.3953, P = 0.0170, n = 36; respectively) in the control atheroma culture supernatant (Table 2). TIMP-1 expression was not correlated to levels of any of the angiotensin peptides measured in the control atheroma culture supernatant (P > 0.05) (Table 2).
Table 2. Correlation of TIMPs with angiotensin signalling peptides in control explant culture supernatant and TIMPs with ERK tissue proteins in the study examining the effect of ATR1 blockade on human carotid atheroma.
| TIMP-1 |
TIMP-2 |
TIMP-4 |
||||
|---|---|---|---|---|---|---|
| rho | P value | rho | P value | rho | P value | |
| AII in control (n = 22) | −0.2223 | 0.2965 | 0.2553 | 0.2286 | 0.5275** | 0.0081 |
| AIII in control (n = 32) | 0.2304 | 0.2046 | 0.4340* | 0.0131 | 0.2247 | 0.2163 |
| AII/AIII/AII-A in control (n = 36) | 0.0417 | 0.8092 | 0.3953* | 0.0170 | −0.0663 | 0.7009 |
| ERK1/2 in ATR1 blockade (n = 28) | 0.3419 | 0.0750 | 0.6552*** | 0.0002 | 0.5298** | 0.0037 |
Spearman's rho correlations between TIMPs and the angiotensin signalling peptides in the control atheroma culture supernatant and TIMPs and the ERK signalling proteins in the matched atheroma tissue samples incubated with and without irbesartan (2.3 mmol/L). As the explant culture system is a closed system the correlations were determined using the total amounts of proteins present within each culture well.
P < 0.01
P < 0.05
Scatter curves are presented in Supplemental Figs. 3–4.
Correlation of ERK Signalling Proteins with TIMP Expression in the ATR1 Blockade Human Carotid Atheroma Culture
Unlike the heterogeneity study that only used the control samples, this study assessed the correlation between the secreted and cellular proteins in the matched irbesartan cultured (2.3 mmol/L; n = 14 samples) and control samples (n = 14 samples). The aim was to determine if there was a direct association between the reduction in ERK1/2 expression in the tissue and the reduced expression of TIMPs in the supernatant.
Expression of ERK1/2 (n = 28 samples from 14 matched pairs) in the atheroma tissue was correlated to the expression of TIMP-2 and TIMP-4 in the atheroma culture supernatant with ATR1 blockade (Spearman's rho = 0.6552, P = 0.0002; Spearman's rho = 0.5293, P = 0.0037; respectively) (Table 2).
Discussion
In the present study using human atheroma explant culture, we demonstrated for the first time, that ATR1 blockade reduces TIMP expression leading to consequent increased gelatinase activity. We demonstrated that relative gelatinase activation and overall catalytic capacity were increased with ATR1 blockade in cultured human atheroma, suggesting that increased gelatinase activity (these MMPs could potentially stabilise plaques by facilitating intimal thickening10)) in the atheroma may be one of the underlying factors of the pleiotropic beneficial effects of anti-hypertensive AII blocking drugs in reducing the incidence of stroke.
It is widely accepted that AII has a role in atherosclerosis development and progression, impacting on inflammation, vascular remodelling and thrombosis44). As the gelatinases and their regulating TIMPs are involved in vascular remodelling a fundamental aim of this study was to determine the influence of ATR1 and ATR2 signalling on gelatinase regulation within human carotid atherosclerosis in vitro. Several previous studies suggested that signalling through these two receptors leads to opposing outcomes with ATR2 signalling abrogating ATR1 signalling and potentially synergistically contributing to the efficacy of ATR1 antagonists30).
ATR1 blockade alone reduced the expression of TIMP-1, -2, and -4. These results support a prominent role for ATR1 signalling in MMP activity regulation in the atheroma and are in accordance with previous studies which also support a key role for ATR1 in TIMP expression45–47). TIMP-1 expression was virtually abolished with additional inclusion of an ATR2 blocker. Since ATR2 alone blockade had no impact on TIMP-1 expression, this result encourages the authors to speculate that ATR2 inhibition may have had a synergistic effect on the ability of the ATR1 blocker to down-regulate TIMP-1 expression.
Further investigation of the molecular mechanism of TIMP down-regulation with ATR1 blockade (proposed model presented in Fig. 8) suggested the down-regulation of TIMP-1 may be a direct result of down-regulation of the ERK1/2 signalling proteins as blockade of ERK1/2 activity also down-regulated TIMP-1 expression. TIMP-2 and -4 expression were correlated to AIII and AII levels, respectively. This result suggests that ATR1 blockade directly antagonised angiotensin peptide signalling leading to TIMP-2 and -4 expression.
Fig. 8.
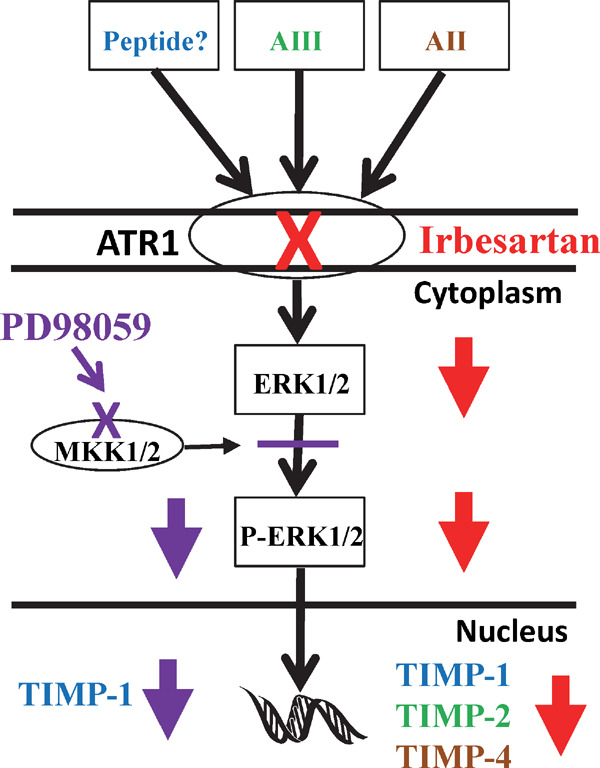
Model of molecular mechanism of TIMP expression with ATR1 blockade. Presented is a simplified pathway by which ATR1 blockade potentially leads to reduced TIMP production. ATR1 has a number of angiotensin peptide agonists that are blocked by nonpeptide diphenylimidazole antagonists, such as irbesartan (red cross). The correlation studies suggest that TIMP-2 expression is dependent on AIII binding to ATR1 and TIMP-4 expression is dependent on AII binding to ATR1. Blockade of ATR1 led to a decrease in expression of the TIMPs and a concomitant decrease in the expression of ERK1/2 in the tissue. Activation of ERK1/2 was blocked by PD98059, which binds to MKK1/2, inactivating the enzymes capacity to phosphorylate ERK1/2 (purple cross). Blockade of ERK1/2 activity also led to a decrease in TIMP-1 expression suggesting that TIMP-1 expression is down-regulated with ATR1 blockade via a decrease in ERK1/2 expression.
ATR2 blockade alone did not have any impact on MMP or TIMP expression, which is in keeping with a number of other studies that have looked at ATR2 blockade in cultured monocytes, rat heart and keratinocytes18, 45, 48). These results suggest that ATR2 does not oppose ATR1 signalling in the atheroma with regards to gelatinase regulation.
Both the gelatinases and MMP-3 (capable of activating MMP-9) showed a reduction in expression when both receptors were blocked, suggesting that AII signalling is involved in their up-regulation in the atheroma, and that either receptor can control expression of these proteins when the other receptor is blocked. In the healthy mixed culture system, where ATR2 would not be expected to be expressed, ATR1 blockade alone was sufficient to down-regulate MMP-9 expression. Correlation studies revealed a very strong association between MMP-9 and expression of both receptors in the atheroma. These results further support a role for both receptors in atheroma MMP-9 regulation.
The activity studies suggested that a drop in TIMP expression led to an increase in MMP catalytic capacity, potentially by a combination of both increased activation and reduced inhibition. In both the ATR1 and dual blockade study where TIMP expression was reduced, gelatinase activation was increased. MMP-2 can be activated by MT-MMPs and MMP-9 can be activated by both MMP-2 and -3 (Fig. 1b). In the combined receptor blockade study where the expression of MMP-2 and -3 was reduced the increased activation of MMP-9 was only ∼1.5-fold. Whereas the MMP-9 increase was ∼2.8-fold in the ATR1 alone blockade study where the expression levels of MMP-2 and -3 did not change. These findings suggest a role for MMP-2 and/or -3 in the cleavage of pro-MMP-9 in atheroma as their expression levels were unchanged with ATR1 blockade and dramatically reduced with dual blockade. Overall MMP-9 catalytic capacity was significantly increased with ATR1 blockade, further confirming that a reduction in TIMP levels led to a consequent increase in MMP activity.
Measuring signalling pathways using explant culture has potential to be more representative of what happens in the patient than cell monocultures or animal models. Using fresh human tissue from an active disease state rather than animals is a major advantage in the application of findings to treatments in humans. However, there is still the inherent in vitro limitation, which is that elements from the in vivo context may be missing in the culture. Previously our group has demonstrated AII signalling in in vitro atheroma culture responds to ATR1 blockade and angiotensin converting enzyme 1 blockade in a manner consistent with the in vivo response to these drugs28, 32).
The other major limitation of this technique is the heterogeneity of the cultured tissue from individual patients which may be inherent to carotid artery disease itself. In order to minimise this effect we have developed a system of assessing paired biopsies from adjacent sites within the atheroma, matching the pairs by macroscopic disease severity. The patients included in these studies are also heterogenous in terms of medical therapies. To minimise the impact of any medications the patient may have been receiving, a 24h preincubation step is included before the intervention study itself. We have previously shown the combination of these techniques minimises the heterogeneity between paired blockade and control samples28, 32–35). We also accept the potential for false discovery when many different outcomes are examined in the same samples.
It is accepted that anti-hypertensive ATR1 blocking drugs are beneficial for cardiovascular patients and effective at reducing the incidence of stroke44). In a study using a rabbit based model designed to represent human atherosclerosis development and treatment, where macrophages infiltrate the intima after SMC accumulation and the anti-hypertensive drugs were administered after development of the plaque, ATR1 blockade led to a reduction in macrophages in the plaque and increased thickness of the fibrous cap with more collagen present. These are all plaque stabilising features. Gelatinase expression in this study was also unchanged with ATR1 blockade49).
Our findings suggest that one of the plaque stabilising effects of ATR1 blockade could be through an increase in gelatinase activity, in particular MMP-9. The functional impact of the MMPs is determined by their overall catalytic capacity, which is a finely tuned balance between expression, activation and inhibition16). This study suggests that ATR1 blockade could potentially lead to generation of more active forms of MMP-2 and -9 with less inhibition from the TIMPs. An increase in overall MMP-9 activity may impart beneficial effects through an enhanced capacity to stimulate SMC proliferation and migration into the fibrous cap10). Elastin degradation is also a feature of atherosclerosis50) and elastin is a recognised substrate of MMP-951), however it is unlikely that an increase in MMP-9 activation would lead to increased elastinolysis in the plaque as our previous study blocking MMP activity suggests MMPs are not involved in elastin degradation in carotid atheroma28). Further studies determining the functional roles of MMPs, such as MMP-9, other than fibrous cap ECM degradation in atherosclerosis are needed.
Conclusion
This study demonstrated ATR1 blockade in atheroma could potentially lead to generation of more active forms of MMP-2 and -9 with less inhibition from the TIMPs. The findings also emphasise that protein expression is only part of the story when elucidating the functional role of MMPs within the atherosclerotic human plaque. Studies using human tissue are potentially important to fully understand the functional impact and mechanism of anti-hypertensive AII blocking drugs in cardiovascular disease.
Acknowledgements
We would like to acknowledge the Queensland Tropical Health Alliance (QTHA) and the Australian Institute of Tropical Health and Medicine (AITHM) for their support of our research. This work was supported by grants from the Australian National Health and Medical Research Council (NHMRC; 1011649, 1003707). JG holds a Practitioner Fellowship from the NHMRC (1019921) and a Senior Clinical Research Fellowship from the Office of Medical research, Queensland Government. The authors would also like to thank Dr Jennifer Elliman for her editorial input.
Disclosure
The authors declare that they have no conflicts of interest to disclose.
Supplemental ELISA Details
The appropriate conditions for each assay were determined by sample titration. Based on these preliminary studies the following volumes and dilutions of conditioned media or tissue lysate were assayed: MMP-2 (75 µL 1/10; #DY902), MMP-3 (70 µL 1/10; #DY513), MMP-9 (15 µL 1/10; #DY911), TIMP-1 (8 µL 1/100; #DY970), TIMP-2 (12 µL 1/10; #DY971), TIMP-4 (100 µL neat; #DY974), AII (50 µL of neat; #CEA005Hu), AIII (50 µL of neat; #CEC312Hu), ERK1-P (100 µL lysate; #DYC1825), ERK2-P (100 µL lysate; #DYC1483), ATR1 (20 µL lysate; #E91658Hu) and ATR2 (5 µL lysate; #E90973Hu). As the explant culture system is a closed system, the total amount of each protein expressed was determined for each sample and used in the subsequent analyses.
Supplemental Fig. 1.
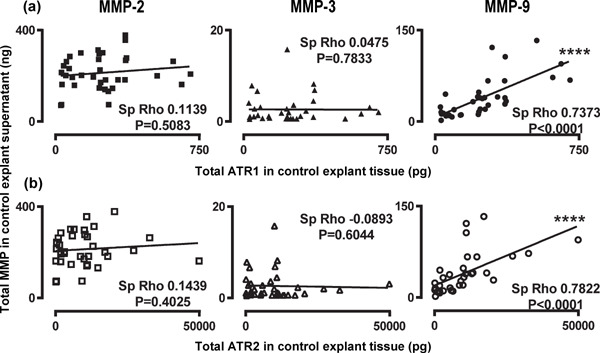
Correlation of MMPs in control explant culture supernatant with ATRs in the control explant tissue. Scatter graphs for Spearman's rho correlations between MMPs and (a) ATR1 and (b) ATR2.
Supplemental Fig. 2.
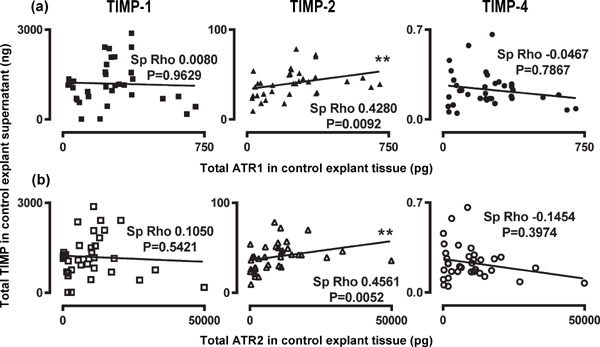
Correlation of TIMPs in control explant culture supernatant with ATRs in the control explant tissue. Scatter graphs for Spearman's rho correlations between TIMPs and (a) ATR1 and (b) ATR2.
Supplemental Fig. 3.
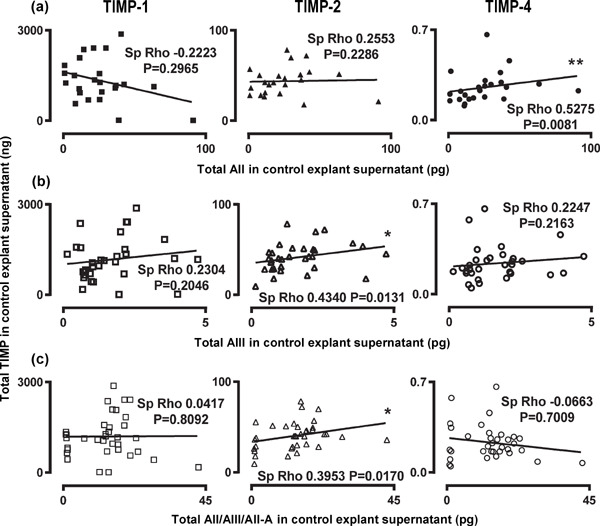
Correlation of TIMPs and angiotensin peptides in control explant culture supernatant. Scatter graphs for Spearman's rho correlations between TIMPs and (a) AII, (b) AIII and (c) AII/AIII/AII-A.
Supplemental Fig. 4.
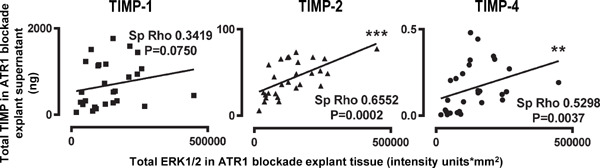
Correlation of TIMPs in matched control and ATR1 blockade sample supernatants with ERK1/2 signalling proteins in the explant tissue. Scatter graphs for Spearman's rho correlations between TIMPs and ERK1/2 signalling proteins.
References
- 1). Golledge J, Greenhalgh RM, Davies AH: The symptomatic carotid plaque. Stroke, 2000; 31: 774-781 [DOI] [PubMed] [Google Scholar]
- 2). Dollery CM, Libby P: Atherosclerosis and proteinase activation. Cardiovasc Res, 2005; 69: 625-635 [DOI] [PubMed] [Google Scholar]
- 3). Sukhova GK, Schonbeck U, Rabkin E, Schoen FJ, Poole AR, Billinghurst RC, Libby P: Evidence for increased Collagenolysis by interstitial Collagenases-1 and -3 in Vulnerable Human Atheromatous Plaques. Circulation, 1999; 99: 2503-2509 [DOI] [PubMed] [Google Scholar]
- 4). Shah PK: Mechanisms of plaque vulnerability and rupture. J Am Coll Cardiol, 2003; 41: 15S-22S [DOI] [PubMed] [Google Scholar]
- 5). Pardo A, Selman M: MMP-1: the elder of the family. Int J Biochem Cell Biol, 2005; 37: 283-288 [DOI] [PubMed] [Google Scholar]
- 6). Rundhaug JE: Matrix Metalloproteinases, Angiogenesis, and Cancer. Clin Cancer Res, 2003; 9: 551-554 [PubMed] [Google Scholar]
- 7). Sternlicht MD, Werb Z: How matrix metalloproteinases regulate cell behaviour. Ann Rev Cell Dev Biol, 2001; 17: 463-516 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8). McCawley LJ, Matrisian LM: Matrix metalloproteinases: they're not just for matrix anymore! Curr Opin Cell Biol, 2001; 13: 534-540 [DOI] [PubMed] [Google Scholar]
- 9). Xu D, Suenaga N, Edelmann MJ, Fridman R, Muschel RJ, Kessler BM: Novel MMP-9 substrates in cancer cells revealed by a label-free quantitative proteomics approach. Mol Cell Prot, 2008; 7: 2215-2228 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10). Newby AC: Matrix metalloproteinases regulate migration, proliferation, and death of vascular smooth muscle cells by degrading matrix and non-matrix substrates. Cardiovasc Res, 2006; 69: 614-624 [DOI] [PubMed] [Google Scholar]
- 11). Johnson JL, Dwivedi A, Somerville M, George SJ, Newby AC: Matrix metalloproteinase (MMP)-3 activates MMP-9 mediated vascular smooth muscle cell migration and neointima formation in mice. Arterioscler Thromb Vasc Biol, 2011; 31: e35-344 [DOI] [PubMed] [Google Scholar]
- 12). Johnson JL, Fritsche-Danielson R, Behrendt M, Westin-Eriksson A, Wennbo H, Herslof M, Elebring M, George SJ, McPheat WL, Jackson CL: Effect of broad-spectrum matrix metalloproteinase inhibition on atherosclerotic plaque stability. Cardiovasc Res, 2006; 71: 586-595 [DOI] [PubMed] [Google Scholar]
- 13). Newby AC: Metalloproteinases promote plaque rupture and myocardial infarction: a persuasive concept waiting for clinical translation. Matrix Biol, 2015; 44-46: 157-166 [DOI] [PubMed] [Google Scholar]
- 14). Cuzner ML, Opdenakker G: Plasminogen activators and matrix metalloproteinases, mediators of extracellular proteolysis in inflammatory demyelination of the central nervous system. J Neuroimmunol, 1999; 94: 1-14 [DOI] [PubMed] [Google Scholar]
- 15). Van Wart HE, Birkedal-Hansen H: The cysteine switch: A principle of regulation of metalloproteinase activity with potential applicability to the entire matrix metalloproteinase gene family. Proc Nat Acad Sci USA, 1990; 87: 5578-5582 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16). Galis ZS, Sukhova GK, Lark MW, Libby P: Increased expression of matrix metalloproteinases and matrix degrading activity in vulnerable regions of human atherosclerotic plaques. J Clin Invest, 1994; 94: 1149-1158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17). Montecucco F, Pende A, Mach F: The renin-angiotensin system modulates inflammatory processes in atherosclerosis: evidence from basic research and clinical studies. Mediators Inflamm, 2009: 752406. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18). Cipollone F, Fazia M, Iezzi A, Pini B, Cuccurullo C, Zucchelli M, de Cesare D, Ucchino S, Spigonardo F, De Luca M, Muraro R, Bei R, Bucci M, Cuccurullo F, Mezzetti A: Blockade of the Angiotensin II Type 1 Receptor Stabilises Atherosclerotic Plaques in Humans by Inhibiting Prostaglandin E2-Dependent Matrix Metalloproteinase Activity. Circulation, 2004; 109: 1482-1488 [DOI] [PubMed] [Google Scholar]
- 19). Yaghooti H, Firoozrai M, Fallah S, Khorramizadeh MR: Angiotensin II differentially induces matrix metalloproteinase-9 and tissue inhibitor of metalloproteinase-1 production and disturbs MMP/TIMP balance. Avicenna J Med Biotech, 2010; 2: 79-85 [PMC free article] [PubMed] [Google Scholar]
- 20). LIFE Study Group: Cardiovascular morbidity and mortality in the Losartan Intervention For Endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. The Lancet, 2002; 359: 995-1003 [DOI] [PubMed] [Google Scholar]
- 21). Second Australian National Blood Pressure Study Group: A Comparison of Outcomes with Angiotensin-Converting-Enzyme Inhibitors and Diuretics for Hypertension in the Elderly. N Eng J Med, 2003; 348: 583-592 [DOI] [PubMed] [Google Scholar]
- 22). The Heart Outcomes Prevention Evaluation Study Investigators: Effects of an Angiotensin-Converting-Enzyme Inhibitor, Ramipril, on Cardiovascular Events in High-Risk Patients. N Eng J Med, 2000; 342: 145-153 [DOI] [PubMed] [Google Scholar]
- 23). Legedz L, Randon J, Sessa C, Baguet JP, Feugier P, Cerutti C, McGregor J, Bricca G: Cathepsin G is associated with atheroma formation in human carotid artery. J Hypertens, 2004; 22: 157-166 [DOI] [PubMed] [Google Scholar]
- 24). Johansson ME, Fagerberg B, Bergstrom G: Angiotensin type 2 receptor is expressed in human atherosclerotic lesions. J Renin Angiotensin Aldosterone Syst, 2008; 9: 17-21 [DOI] [PubMed] [Google Scholar]
- 25). Pool JL, Guthrie RM, Littlejohn TW, 3rd, Raskin P, Shephard AM, Weber MA, Weir MR, Wilson TW, Wright J, Kassler-Taub KB, Reeves RA: Dose-related antihypertensive effects of Irbesartan in patients with mild-tomoderate hypertension. Am J Hypertens, 1998; 11: 462-470 [DOI] [PubMed] [Google Scholar]
- 26). Christen Y, Waeber B, Nussberger J, Porchet M, Borland RM, Lee RJ, Maggon K, Shum L, Timmermans PB, Brunner HR: Oral administration of DuP 753, a specific angiotensin II receptor antagonist, to normal male volunteers. Inhibition of pressor response to exogenous angiotensin 1 and II. Circulation, 1991; 83: 1333-1342 [DOI] [PubMed] [Google Scholar]
- 27). Mazzolai L, Pedrazzini T, Nicoud F, Gabbiani G, Brunner HR, Nussberger J: Increased cardiac angiotensin II levels induce right and left ventricular hypertrophy in normotensive mice. Hypertension, 2000; 35: 985-991 [DOI] [PubMed] [Google Scholar]
- 28). Clancy P, Seto S-W, Koblar SA, Golledge JG: Role of the angiotensin converting enzyme 1/angiotensin II/angiotensin receptor 1 axis in interstitial collagenase expression in human carotid atheroma. Atherosclerosis, 2013; 229: 331-337 [DOI] [PubMed] [Google Scholar]
- 29). Appel GB, Appel AS: Angiotensin II receptor antagonists: role in hypertension, cardiovascular disease, and renoprotection. Prog Cardiovasc Dis, 2004; 47(2): 105-115 [DOI] [PubMed] [Google Scholar]
- 30). Widdop RE, Jones ES, Hannan RE, Gaspari TA: Angiotensin AT2 receptors: cardiovascular hope or hype? Br J Pharmacol, 2003; 140: 809-824 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31). Strauss MH, Hall AS: Angiotensin receptor blockers may increase risk of myocardial infarction: unravelling the ARB-MI paradox. Circulation, 2006; 114: 838-854 [DOI] [PubMed] [Google Scholar]
- 32). Clancy P, Koblar S, Golledge J: Angiotensin receptor 1 blockade reduces secretion of pro-inflammatory cytokines from cultured human carotid atheroma and vascular cells in association with reduced extracellular signal regulated kinase expression and activation. Atherosclerosis, 2014; 236(1): 108-115 [DOI] [PubMed] [Google Scholar]
- 33). Clancy P, Lincz L, Maguire J, McEvoy M, Koblar S, Golledge J: Tenascin-C is increased in atherothrombotic stroke patients and has an anti-inflammatory effect in the human carotid artery. BioFactors, 2014; 40(4): 448-457 [DOI] [PubMed] [Google Scholar]
- 34). Golledge J, Mangan S, Clancy P: Effects of peroxisome proliferator-activated receptor ligands in modulating tissue factor and tissue factor pathway inhibitor in acutely symptomatic carotid atheromas. Stroke, 2007; 38: 1501-1508 [DOI] [PubMed] [Google Scholar]
- 35). Mangan S, Clancy P, Golledge J: Modulation of endothelial cell thrombomodulin by PPAR ligands- variation according to environment. Thromb Res, 2007; 121: 827-834 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36). Golledge J, McCann M, Mangan S, Lam A, Karan M: Osteoprotegerin and osteopontin are expressed at high concentrations within symptomatic carotid atherosclerosis. Stroke, 2004; 35: 1636-1641 [DOI] [PubMed] [Google Scholar]
- 37). Bae SK, Kim MJ, Shim EJ, Cho DY, Shon JH, Liu KH, Kim EY, Shin JG: HPLC determination of irbesartan in human plasma: its application to pharmacokinetic studies. Biomed Chromatogr, 2009; 23: 568-572 [DOI] [PubMed] [Google Scholar]
- 38). Brechler V, Jones PW, Levens NR, de Gasparo M, Bottari SP: Agonistic and antagonistic properties of angiotensin analogs at the AT2 receptor in PC12W cells. Regulatory Peptides, 1993; 44: 207-213 [DOI] [PubMed] [Google Scholar]
- 39). Alessi DR, Cuenda A, Cohen P, Dudley DT, Saltiel AR: PD 098059 Is a Specific Inhibitor of the Activation of Mitogen-activated Protein Kinase Kinase in Vitro and in Vivo. J Biol Chem, 1995; 270: 27489-27494 [DOI] [PubMed] [Google Scholar]
- 40). Peng N, Liu JT, Gao DF, Lin R, Li R: Angiotensin II-induced C-reactive protein generation: Inflammatory role of vascular smooth muscle cells in atherosclerosis. Atherosclerosis, 2007; 193: 292-298 [DOI] [PubMed] [Google Scholar]
- 41). Jankowski V, Vanholder R, van der Giet M, Tölle M, Karadogan S, Gobom J, Furkert J, Oksche A, Krause E, Tran TN, Tepel M, Schuchardt M, Schlüter H, Wiedon A, Beyermann M, Bader M, Todiras M, Zidek W, Jankowski J: Mass-spectrometric identification of a novel angiotensin peptide in human plasma. Arterioscler Thromb Vasc Biol, 2007; 27: 297-302 [DOI] [PubMed] [Google Scholar]
- 42). Laemmli UK: Cleavage of structural proteins during the assembly of the head of the bacteriophage T4. Nature, 1970; 227: 680-685 [DOI] [PubMed] [Google Scholar]
- 43). Morrison CJ, Overall CM: TIMP independence of matrix metalloproteinase (MMP)-2 activation by membrane type 2 (MT2)-MMP is determined by contributions of both the MT2-MMP catalytic and hemopexin C domains. J Biol Chem, 2006; 281: 26528-26539 [DOI] [PubMed] [Google Scholar]
- 44). Cipollone F, Fazia ML, Mezetti A: Role of angiotensin II receptor blockers in atherosclerotic plaque stability. Expert Opin Pharmacother, 2006; 7: 277-285 [DOI] [PubMed] [Google Scholar]
- 45). Yang D, Ma S, Li D, Tang B, Yang Y: Angiotensin II receptor blockade improves matrix metalloproteinases/tissue inhibitor of metalloproteinase-1 balance and restores fibronectin expression in rat infarcted myocardium. Biochem Biophys Res Commun, 2009; 388: 606-611 [DOI] [PubMed] [Google Scholar]
- 46). Varo N, Iraburu MJ, Varela M, Lopez B, Etayo JC, Diez J. Chronic AT1 blockade stimulates extracellular collagen type 1 degradation and reverses myocardial fibrosis in spontaneously hypertensive rats. Hypertension, 2000; 35: 1197-1202 [DOI] [PubMed] [Google Scholar]
- 47). Yoshiji H, Kuriyama S, Yoshii J, Ikenaka Y, Noguchi R, Yanase K, Namisaki T, Yamazaki M, Tsujinoue H, Imazu H, Fukui H: Angiotensin-II induces the tissue inhibitor of metalloproteinases-1 through the protein kinase-C signalling pathway in rat liver fibrosis development. Hepatol Res, 2003; 27: 51-56 [DOI] [PubMed] [Google Scholar]
- 48). Santibanez JF, Kocic J, Fabra A, Cano A, Quintanilla M: Rac1 modulates TGF-beta1-mediated epithelial cell plasticity and MMP-9 production in transformed keratinocytes. FEBS Lett, 2010; 584: 2305-2310 [DOI] [PubMed] [Google Scholar]
- 49). Hotchi J, Hoshiga M, Takeda Y, Yuki T, Fujisaka T, Ishihara T, Hanafusa T: Plaque-stabilising effect of angiotensin-converting enzyme inhibitor and/or angiotensin receptor blockade in a rabbit plaque model. J Atheroscler Thromb, 2013; 20(3): 257-266 [DOI] [PubMed] [Google Scholar]
- 50). Fulop T, Wei SM, Robert L, Jacob MP: Determination of elastin peptides in normal and arteriosclerotic human sera by ELISA. Clin Physiol Biochem, 1990; 8(6): 273-282 [PubMed] [Google Scholar]
- 51). Birkedal-Hansen H, Moore WGI, Bodden MK, Windsor LJ, Birkedal-Hansen B, DeCarlo A, Engler JA: Matrix metalloproteinases: a review. Critical Rev Oral Biol Med, 1993; 4: 197-250 [DOI] [PubMed] [Google Scholar]


