Abstract
Colorectal cancer (CRC) is the third and second most common type of cancer diagnosed in males and females, respectively, and is the fourth leading cause of cancer-associated mortality worldwide. Liver metastasis is the primary cause of mortality in patients with CRC, and therefore requires therapeutic focus. Regulatory T cells (Tregs) and hepatic stellate cells (HSCs) are potentially involved in regulating the immune response during liver metastasis. The aim of the present study was to evaluate the influence of CD4+ forkhead box p3 (Foxp3)+ Tregs and the HGF/c-Met signaling pathway in the liver metastasis of CRC. A model of the latter was established using Balb/c mice via splenic injection of human CRC cells (CT-26 line). The mice were monitored for 3 weeks after being injected, and the spleens and livers were removed on day 22 for further analysis. Moreover, the single-cell suspensions were labeled with CD4 and Foxp3 antibodies, and were analyzed using flow cytometry. Expression levels of α-smooth muscle actin (SMA), hepatocyte growth factor (HGF) and hepatocyte growth factor receptor (c-Met) were analyzed using immunohistochemistry. Mice injected with CT-26 cells exhibited signs of illness and significant weight loss, compared with the control mice (P=0.013), and they also developed liver metastases, at an average of 20.5 tumors per mouse. Pathological evaluation using hematoxylin and eosin staining confirmed the tumors as liver metastases of CRC. The numbers of CD4+ T cells were significantly decreased in the spleen (P<0.001) and liver (P=0.003) of tumor-bearing mice, while the proportions of CD4+FOXP3+ Tregs increased significantly in the spleen (P<0.001) and liver (P=0.026) compared with that in the controls. Additionally, α-SMA, HGF and c-Met levels increased significantly during metastatic growth in the liver. In conclusion, CD4+FOXP3+ Treg levels increased and the HGF/c-Met pathway was upregulated during the liver metastasis of CRC in mice, indicating the presence of potential therapeutic targets.
Keywords: liver metastasis, colorectal cancer, CD4+ T cells, CD4+FOXP3+ Treg, hepatic stellate cells
Introduction
Colorectal cancer (CRC) is the third and second most commonly diagnosed type of cancer in males and females, respectively, and is the fourth leading cause of cancer-associated mortalities worldwide (1). Approximately 30% of patients with CRC have synchronous liver metastases at the time of diagnosis (2,3), and nearly 50% of patients acquire metachronous liver metastases following radical resection of the primary lesion (4). Liver metastasis represents the most common cause of mortality among patients with CRC (5). Therefore, in order to improve the therapeutic outcome of patients with CRC, it is imperative to target liver metastases.
The liver influences numerous immunological processes and houses immune cells that serve important roles in local and systemic host defense pathways (6). Regulatory T cells (Tregs) are a key factor in antitumor immune responses, and tumor infiltration of Tregs is associated with attenuated antitumor T-cell responses (7). Moreover, it has previously been revealed that there is a decreased number of CD4+ and CD8+ T cells, but an increase in the number of CD4+CD25+ Tregs, relative to the total CD4+ T cell number in metastatic liver tissues (8).
Hepatic stellate cells (HSCs), also known as Ito cells or lipocytes, are vitamin A-storing cells located in the Disse space between hepatocytes and sinusoidal endothelial cells, and serve a major role in the tumorigenesis of hepatocellular carcinoma (9,10). HSC activation is characterized by an elevation of α-smooth muscle actin (α-SMA) levels (11) and the secretion of hepatocyte growth factor (HGF), which suppresses the immune response and promotes cancer cell proliferation and migration by binding to hepatocyte growth factor receptor (c-Met) (12–14). Inhibition of the HGF/c-Met signaling pathway may therefore represent a novel therapeutic approach to the treatment of CRC (15,16).
In the present study, a reproducible liver metastases model was established by injecting colon carcinoma cells into the spleen of immunocompetent mice and evaluating changes in the number of CD4+ forkhead box p3 (Foxp3)+ Tregs and the HGF/c-Met pathway during liver metastases.
Materials and methods
Mice
Female Balb/c mice aged 6–8 weeks old with an average weight of 20.52±0.83 g were obtained from the Laboratory Animal Center of Sun Yat-sen University (Guangzhou, China). The mice were housed in a special pathogen-free facility with free access to drinking water and a pellet-based diet, and were quarantined for 7 days prior to surgery. The mice were kept in a room with controlled temperature (22±1°C) and humidity (50-70%), and a 12-h light/dark cycle. All animal experiments were approved by the Ethical Committee of Sun Yat-Sen University and were conducted in accordance with guidelines of the Institutional Animal Care and Use Committee of Sun Yat-Sen University and the Committee for Animal Experiments of Sun Yat-Sen University.
On day 1, 20 mice were randomly assigned into experimental and control groups, with 10 mice in each group. Mice were anesthetized using an intraperitoneal injection of 4% chloral hydrate (400 mg/kg). To verify anesthesia, it was ensured that the mice were unresponsive when pinching the skin or passively opening the eyes. After local disinfection of the skin, an abdominal incision was made along the left subcostal margin, and 1×105 CT-26 cells in 50 µl phosphate-buffered saline (PBS) were injected into the spleen of the mice in the experimental group using a 32-G needle. The control mice received the same volume of sterile PBS. The abdominal wall was closed using a two-layer technique and non-absorbable sutures. The mice were monitored, with body weight and general health indicators being recorded every 3 days over a period of 3 weeks. No mouse exhibited signs of peritonitis in the present study following the use of chloral hydrate. If a mouse could not ambulate or was unable to intake food or water independently during the experiment, it would be euthanized ahead of schedule; however, no mouse was euthanized or found dead ahead of schedule in the present study. The mice were sacrificed on day 22 using an intraperitoneal injection of 4% chloral hydrate (400 mg/kg), followed by cervical dislocation. To verify death, breath and heartbeat were observed. Briefly, the mice exhibited no abdominal or thoracic respiration and no observable throbbing in the precordium. After verification of death, the liver and spleen were removed, and the number of macroscopic metastatic nodules in the liver was counted. Liver tissues were fixed for 24 h at room temperature using 10% neutral buffered formalin for subsequent paraffin embedding, or stored in RNA stabilization solution at −80°C (RNA later; Ambion; Thermo Fisher Scientific, Inc.) for further analysis.
Cell lines
The undifferentiated murine colon adenocarcinoma cell line, CT-26, was obtained from the Chinese Academy of Science Cell Bank (Shanghai, China) and cultured in RPMI-1640 medium with 10% fetal calf serum and antibiotics (1% penicillin and streptomycin) (all Gibco; Thermo Fisher Scientific, Inc.) at 37°C in a humidified incubator (Thermo Fisher Scientific, Inc.) with 5% CO2. The cultures were routinely tested once a week for mycoplasma contamination using MycoProbe (R&D Systems, Inc.), and the medium was regularly replaced every 2–3 days. The cells were harvested using 0.25% trypsin (Gibco; Thermo Fisher Scientific, Inc.) for 2 min at room temperature.
Flow cytometry
The spleen and liver were homogenized by trituration (300 × g at 4°C for 10 min) and suspended in PBS, and single-cell suspensions were prepared. After RBC lysis (cat. no. 555899; BD Biosciences) with 0.17 M ammonium chloride at 4°C for 5 min, the cells were washed in PBS and counted using the cellometer Auto T4 (BD Biosciences). Aliquots of 1×106 cells were stained using anti CD4-FITC antibody (1:10; cat. no. 561833; BD Biosciences) and/or anti Foxp3-PE-Cy5 antibody (1:10; cat. no. 563101; BD Biosciences) at 4°C for 30 min, according to the manufacturer's instructions. The stained cells were added into a FACS canto flow cytometer (BD Biosciences), and the T-cell subsets were analyzed in samples of 1×104 cells each using Cell Quest software (v5.1; BD Pharmingen). Samples from all mice were analyzed using flow cytometry, which was repeated three times.
Histopathological evaluation and immunohistochemistry
Tissues were fixed with 10% formalin at room temperature for 24 h and embedded in 10% paraffin for 24 h, cut into 4-µm thick sections and placed on salinized glass slides. Hematoxylin and eosin staining was performed as per the standard protocol. For immunohistochemistry, the sections were deparaffinized with dimethylbenzene and rehydrated through an alcohol gradient, followed by antigen retrieval in a hot sodium citrate buffer at 96–98°C for 15 min. After quenching the endogenous peroxidase activity with 0.3% hydrogen peroxide solution and blocking using 5% goat serum (cat. no. 16210064; Gibco; Thermo Fisher Scientific, Inc.) at room temperature for 30 min, the sections were incubated overnight with anti-α-SMA (1:1,000; cat. no. ab124964), anti-HGF (1:1,000; cat. no; ab83760) and anti-c-Met (1:1,000; cat. no. ab51067) primary antibodies (all Abcam) at 4°C. The sections were then incubated with a Qdot 655-conjugated goat anti-chicken IgY (H+L) secondary antibody (1:50; cat. no. Q14421MP; Invitrogen; Thermo Fisher Scientific, Inc.) at room temperature for 60 min, washed and stained with diaminobenzidine in an Envision System (Dako), followed by counterstaining with hematoxylin at room temperature for 2 min. Slides were observed under an inverted phase contrast light microscope (magnification, ×200; Leica Microsystems GmbH).
Statistical analysis
Sample size in each group was determined according to the study by Syed Khaja et al (17) and the power test using G power (t-test, difference between two independent means). Briefly, the mean and standard deviation in each group was used to calculate the effect size d, and the power was >90% for all tests using G power software. All data are expressed as mean ± standard deviation (n=3). The weight of the mice was compared using a repeated-measures analysis of variance and the expression of T cell markers was compared using an unpaired Student's t-test in SPSS v21 software (IBM Corp.). Two-sided P-values of <0.05 were considered to indicate a statistically significant difference.
Results
Survival and general status of the mice
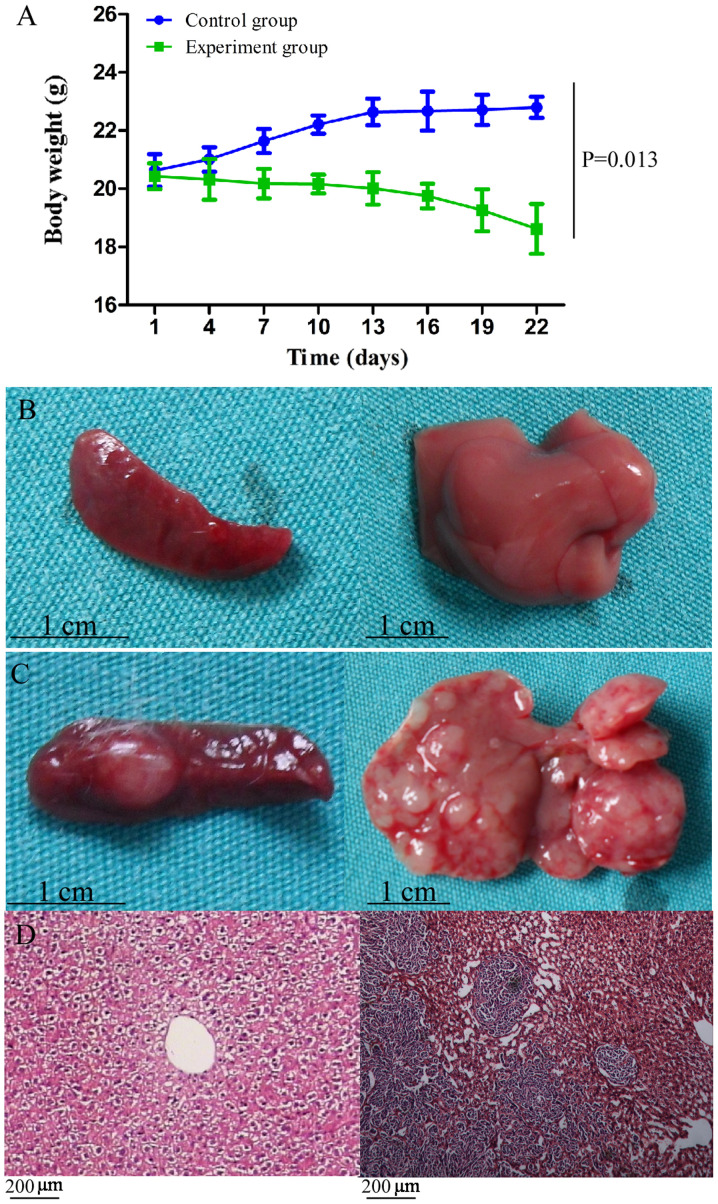
All tumor-bearing and control mice survived until the end of the experiment. However, unlike the control mice, the tumor-bearing mice exhibited signs of illness, such as reduced activity, slow response, lackluster fur, loss of appetite, emaciation and a distended abdomen, which were accompanied by significant loss in body weight (P=0.013; Fig. 1A). The maximum percentage of body weight loss was 8.86%.
Figure 1.
General status and observation of metastases. (A) Body weight loss was observed in mice in the experimental group and the dynamic curve of body weight was significantly different compared with that of the control group (P=0.013). (B) Spleen (left) and liver (right) of the mice in the control group, without neoplasm. (C) Spleen (left) and liver (right) of the mice in the experimental group, with macroscopic tumors bulging onto the surface. (D) Pathological evaluation by hematoxylin and eosin staining of the normal liver and liver neoplasm, confirming the tumors as liver metastases of colon carcinoma.
Successful induction of liver metastases of CT-26 cells
At 3 weeks post-tumor cell injection, the mice were sacrificed. The liver and spleen of the control mice was soft and smooth without any neoplasms (Fig. 1B). By contrast, both organs in the CT-26-injected mice had a rough and uneven surface, while macroscopic tumors were also observed (Fig. 1C). An average of 20.5 metastatic nodules was observed on the liver of the CT-26-injected mice, and pathological evaluation using hematoxylin and eosin staining confirmed the nodules as liver metastases of colon carcinoma (Fig. 1D).
CD4+ T cells and CD4+FOXP3+ Treg populations are influenced by liver metastases
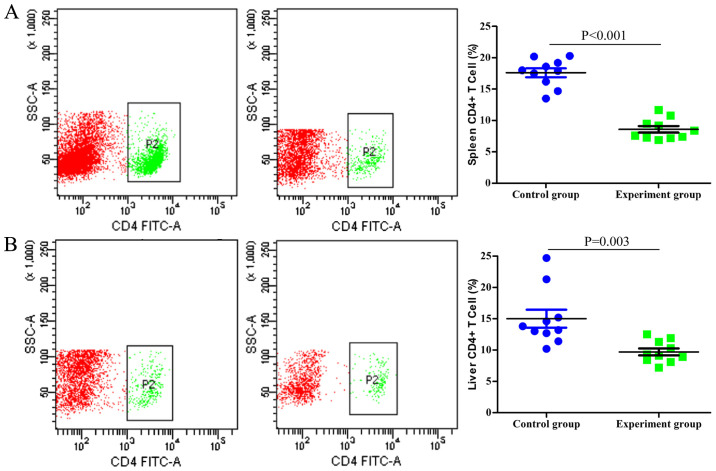
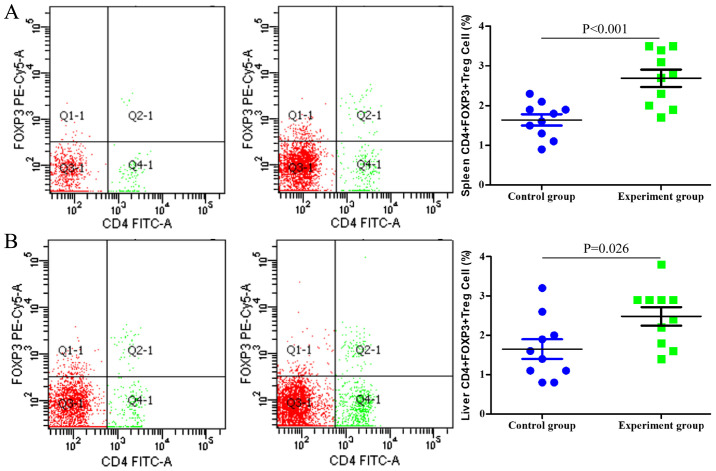
Single-cell suspensions of the spleen and liver were prepared and analyzed using flow cytometry. As indicated in Fig. 2, the numbers of CD4+ T cells in the spleen and liver of the tumor-bearing mice were significantly lower compared with those in the control group (P<0.001 and P=0.003, respectively). By contrast, the proportion of CD4+FOXP3+ Tregs among the entire CD4+ T cell population was significantly higher in the tumor-bearing group compared with that in the control group (P<0.001 and P=0.026 in the spleen and liver, respectively; Fig. 3).
Figure 2.
Analysis of CD4+ T cells using flow cytometry in (A) the spleen and (B) the liver. There were significantly fewer CD4+ T cells in the experimental groups (middle panels) compared with those in the control groups (left panels) (P<0.001 and P=0.003, respectively). P2 represents CD4+ T cells. SSC-A, side scatter-area.
Figure 3.
Analysis of CD4+FOXP3+ Tregs by flow cytometry in (A) the spleen and (B) the liver. The proportion of CD4+FOXP3+ Tregs in the whole CD4+ T cell population was significantly higher in the experimental groups (middle panels) compared with that in the control groups (left panels) (P<0.001 and P=0.026, respectively). FOXP3, forkhead box p3.
HGF/c-Met signaling pathway is upregulated in liver metastases
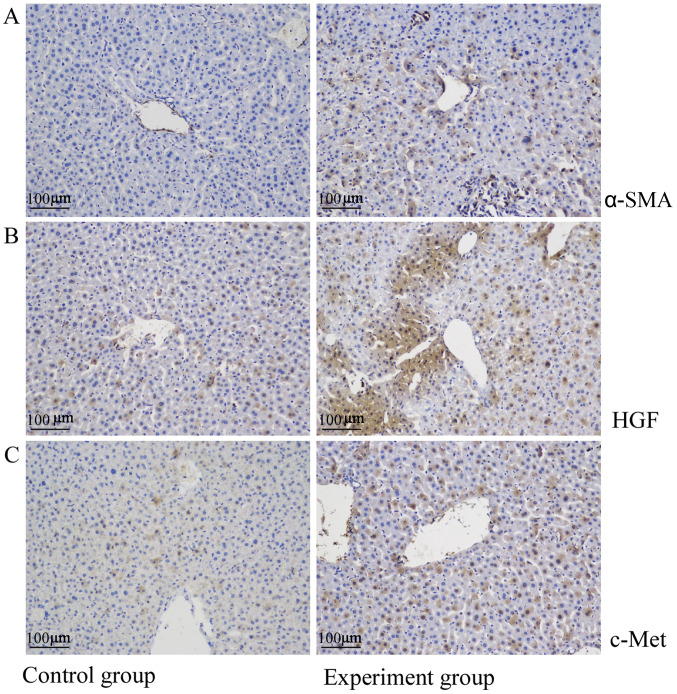
The liver tissues of the control mice were negative for α-SMA and expressed only low levels of HGF and c-Met. By contrast, α-SMA, HGF and c-Met levels were significantly upregulated in the liver metastatic nodules (Fig. 4).
Figure 4.
Upregulation of the HGF/c-Met signaling pathway in liver metastasis. In the control group, (A) α-SMA was not expressed while (B) HGF and (C) c-Met were expressed at a low level. By contrast, α-SMA, HGF and c-Met were highly expressed in the experimental group. α-SMA, α smooth muscle actin, HGF, hepatocyte growth factor; c-Met, hepatocyte growth factor receptor.
Discussion
Liver metastasis is the primary cause of mortality in patients with CRC. In the present study, a liver metastasis model of colon carcinoma in mice was established via an injection of CT-26 cells into the spleen (8,18), in order to evaluate changes in CD4+Foxp3+ Treg numbers and HGF/c-Met expression levels. All mice developed metastatic growth in the liver and the survival rate was 100% despite a lethargic appearance and loss of appetite. Macroscopic tumors were observed on the liver surface, with an average of 20.5 metastatic nodules per mouse.
T cells serve an important role during tumorigenesis, with cytotoxic T cells eliminating cancer cells presenting major histocompatibility complex class I molecules, while CD4+Foxp3+ Tregs promote tumor progression via suppression of the antitumor immune response (19,20). Tumor tissues are often infiltrated by Tregs, and their depletion has been revealed to augment the antitumor immune response in mice (21,22). Tregs elicit an immunosuppressive effect by inhibiting CD4+ and CD8+ effector T cells via T cell receptor engagement and a cell contact-dependent mechanism (23,24). In our previous study, the numbers of CD4+ and CD8+ T cells were shown to decrease in liver metastases (8), a fact which was supported in the present study, along with an increase in the proportion of CD4+Foxp3+ Tregs in the spleen and liver. A higher proportion of Tregs may inhibit cytotoxic T cells and promote metastasis of cancer cells from the spleen to the liver.
The HGF/c-Met signaling pathway serves a key role in metastasis (25) and influences the proliferation, survival and invasive potential of colorectal, gastric, gastro-esophageal and gastro-esophageal junction cancer (26). In addition, HGF and c-Met are upregulated in a number of cancer types, including CRC (27), and are associated with tumor recurrence and an unfavorable patient prognosis (28). Consistent with these findings, HGF/c-Met expression was revealed to be significantly higher in liver metastatic tissues in the current study, indicating that the downregulation of HGF/c-Met expression may suppress tumor cell invasion; however, further studies are required to confirm this finding.
The role and potential mechanism of CD4+FOXP3+ Tregs have also been investigated in colitis-associated colon and colorectal cancer. Notably, Olguín et al (29) discovered that the percentage of CD4+FOXP3+ Tregs was increased in colitis-associated colon cancer. Another study by Syed Khaja et al (17) revealed that numbers of CD4+FOXP3+ Tregs were increased in colorectal cancer tissues. Although the present study only obtained results and conclusions similar to those described in previous papers, to the best of our knowledge, there is no previously published study on this topic for the liver metastasis of colorectal cancer. The results of the present study should therefore supplement the existing knowledge in the field of CD4+FOXP3+ Tregs and colorectal cancer.
In conclusion, liver metastasis of CRC is associated with an increase in the number of CD4+FOXP3+ Tregs and upregulation of the HGF/c-Met signaling pathway, indicating the presence of potential novel therapeutic targets for liver metastasis.
Acknowledgements
Not applicable.
Glossary
Abbreviations
- CRC
colorectal cancer
- Treg
regulatory T cell
- HSC
hepatic stellate cell
- α-SMA
α-smooth muscle actin
- HGF
hepatocyte growth factor
Funding
The present study was supported by a Medical Science Research Grant from the Health Department of Guangdong Province (grant no. A2018007).
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
XH and ZXC contributed equally to this work. XH, PL and YW conceived and designed the study. XH, ZXC and NZ performed the animal experiments and drafted the manuscript. CZ and XL performed the cell experiments. JY and ZPC contributed to the analysis and interpretation of data. All authors read and approved the final manuscript.
Ethics approval and consent to participate
All animal experiments were approved by the Ethical Committee of Sun Yat-Sen University, and were conducted in accordance with guidelines of the Institutional Animal Care and Use Committee of Sun Yat-Sen University, and the Committee for Animal Experiments.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Bengmark S, Hafstrom L. The natural history of primary and secondary malignant tumors of the liver. I. The prognosis for patients with hepatic metastases from colonic and rectal carcinoma by laparotomy. Cancer Am Cancer Soc. 1969;23:198–202. doi: 10.1002/1097-0142(196901)23:1<198::aid-cncr2820230126>3.0.co;2-j. [DOI] [PubMed] [Google Scholar]
- 3.Manfredi S, Lepage C, Hatem C, Coatmeur O, Faivre J, Bouvier AM. Epidemiology and management of liver metastases from colorectal cancer. Ann Surg. 2006;244:254–259. doi: 10.1097/01.sla.0000217629.94941.cf. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Konopke R, Roth J, Volk A, Pistorius S, Folprecht G, Zöphel K, Schuetze C, Laniado M, Saeger HD, Kersting S. Colorectal liver metastases: An update on palliative treatment options. J Gastrointestin Liver Dis. 2012;21:83–91. [PubMed] [Google Scholar]
- 5.Lykoudis PM, O'Reilly D, Nastos K, Fusai G. Systematic review of surgical management of synchronous colorectal liver metastases. Br J Surg. 2014;101:605–612. doi: 10.1002/bjs.9449. [DOI] [PubMed] [Google Scholar]
- 6.Robinson MW, Harmon C, O'Farrelly C. Liver immunology and its role in inflammation and homeostasis. Cell Mol Immunol. 2016;13:267–276. doi: 10.1038/cmi.2016.3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Nishikawa H, Sakaguchi S. Regulatory T cells in cancer immunotherapy. Curr Opin Immunol. 2014;27:1–7. doi: 10.1016/j.coi.2013.12.005. [DOI] [PubMed] [Google Scholar]
- 8.Huang X, Zou Y, Lian L, Wu X, He X, He X, Wu X, Huang Y, Lan P. Changes of T cells and cytokines TGF-β1 and IL-10 in mice during liver metastasis of colon carcinoma: Implications for liver anti-tumor immunity. J Gastrointest Surg. 2013;17:1283–1291. doi: 10.1007/s11605-013-2194-5. [DOI] [PubMed] [Google Scholar]
- 9.Bárcena C, Stefanovic M, Tutusaus A, Martinez-Nieto GA, Martinez L, García-Ruiz C, de Mingo A, Caballeria J, Fernandez-Checa JC, Marí M, Morales A. Angiogenin secretion from hepatoma cells activates hepatic stellate cells to amplify a self-sustained cycle promoting liver cancer. Sci Rep. 2015;5:7916. doi: 10.1038/srep07916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Gupta G, Khadem F, Uzonna JE. Role of hepatic stellate cell (HSC)-derived cytokines in hepatic inflammation and immunity. Cytokine. 2019;124:154542. doi: 10.1016/j.cyto.2018.09.004. [DOI] [PubMed] [Google Scholar]
- 11.Lu DH, Guo XY, Qin SY, Luo W, Huang XL, Chen M, Wang JX, Ma SJ, Yang XW, Jiang HX. Interleukin-22 ameliorates liver fibrogenesis by attenuating hepatic stellate cell activation and downregulating the levels of inflammatory cytokines. World J Gastroenterol. 2015;21:1531–1545. doi: 10.3748/wjg.v21.i5.1531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Weiskirchen R, Tacke F. Cellular and molecular functions of hepatic stellate cells in inflammatory responses and liver immunology. Hepatobiliary Surg Nutr. 2014;3:344–363. doi: 10.3978/j.issn.2304-3881.2014.11.03. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Patel MB, Pothula SP, Xu Z, Lee AK, Goldstein D, Pirola RC, Apte MV, Wilson JS. The role of the hepatocyte growth factor/c-MET pathway in pancreatic stellate cell-endothelial cell interactions: Antiangiogenic implications in pancreatic cancer. Carcinogenesis. 2014;35:1891–1900. doi: 10.1093/carcin/bgu122. [DOI] [PubMed] [Google Scholar]
- 14.Cai W, Rook SL, Jiang ZY, Takahara N, Aiello LP. Mechanisms of hepatocyte growth factor-induced retinal endothelial cell migration and growth. Invest Ophthalmol Vis Sci. 2000;41:1885–1893. [PubMed] [Google Scholar]
- 15.Pothula SP, Xu Z, Goldstein D, Biankin AV, Pirola RC, Wilson JS, Apte MV. Hepatocyte growth factor inhibition: A novel therapeutic approach in pancreatic cancer. Br J Cancer. 2016;114:269–280. doi: 10.1038/bjc.2015.478. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Parr C, Jiang WG. Expression of hepatocyte growth factor/scatter factor, its activator, inhibitors and the c-Met receptor in human cancer cells. Int J Oncol. 2001;19:857–863. [PubMed] [Google Scholar]
- 17.Syed Khaja A, Toor SM, El SH, Ali BR, Elkord E. Intratumoral FoxP3+Helios+ regulatory T cells upregulating immunosuppressive molecules are expanded in human colorectal cancer. Front Immunol. 2017;8:619. doi: 10.3389/fimmu.2017.00619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Liu HY, Huang ZL, Yang GH, Lu WQ, Yu NR. Inhibitory effect of modified citrus pectin on liver metastases in a mouse colon cancer model. World J Gastroenterol. 2008;14:7386–7391. doi: 10.3748/wjg.14.7386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Takeuchi Y, Nishikawa H. Roles of regulatory T cells in cancer immunity. Int Immunol. 2016;28:401–409. doi: 10.1093/intimm/dxw025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Fontenot JD, Gavin MA, Rudensky AY. Foxp3 programs the development and function of CD4+CD25+ regulatory T cells. Nat Immunol. 2003;4:330–336. doi: 10.1038/ni904. [DOI] [PubMed] [Google Scholar]
- 21.Shimizu J, Yamazaki S, Sakaguchi S. Induction of tumor immunity by removing CD25+CD4+ T cells: A common basis between tumor immunity and autoimmunity. J Immunol. 1999;163:5211–5218. [PubMed] [Google Scholar]
- 22.Onizuka S, Tawara I, Shimizu J, Sakaguchi S, Fujita T, Nakayama E. Tumor rejection by in vivo administration of anti-CD25 (interleukin-2 receptor alpha) monoclonal antibody. Cancer Res. 1999;59:3128–3133. [PubMed] [Google Scholar]
- 23.Almeida AR, Ciernik IF, Sallusto F, Lanzavecchia A. CD4+ CD25+ Treg regulate the contribution of CD8+ T-cell subsets in repopulation of the lymphopenic environment. Eur J Immunol. 2010;40:3478–3488. doi: 10.1002/eji.201040600. [DOI] [PubMed] [Google Scholar]
- 24.Zanin-Zhorov A, Ding Y, Kumari S, Attur M, Hippen KL, Brown M, Blazar BR, Abramson SB, Lafaille JJ, Dustin ML. Protein kinase C-theta mediates negative feedback on regulatory T cell function. Science. 2010;328:372–376. doi: 10.1126/science.1186068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Arlt F, Stein U. Colon cancer metastasis: MACC1 and Met as metastatic pacemakers. Int J Biochem Cell Biol. 2009;41:2356–2359. doi: 10.1016/j.biocel.2009.08.001. [DOI] [PubMed] [Google Scholar]
- 26.Bradley CA, Salto-Tellez M, Laurent-Puig P, Bardelli A, Rolfo C, Tabernero J, Khawaja HA, Lawler M, Johnston PG, Van Schaeybroeck S, MErCuRIC consortium Targeting c-MET in gastrointestinal tumours: Rationale, opportunities and challenges. Nat Rev Clin Oncol. 2017;14:562–576. doi: 10.1038/nrclinonc.2017.40. [DOI] [PubMed] [Google Scholar]
- 27.Shojaei F, Simmons BH, Lee JH, Lappin PB, Christensen JG. HGF/c-Met pathway is one of the mediators of sunitinib-induced tumor cell type-dependent metastasis. Cancer Lett. 2012;320:48–55. doi: 10.1016/j.canlet.2012.01.026. [DOI] [PubMed] [Google Scholar]
- 28.Osada S, Matsui S, Komori S, Yamada J, Sanada Y, Ihawa A, Tanaka Y, Tokuyama Y, Okumura N, Nonaka K, et al. Effect of hepatocyte growth factor on progression of liver metastasis in colorectal cancer. Hepatogastroenterology. 2010;57:76–80. [PubMed] [Google Scholar]
- 29.Olguín JE, Medina-Andrade I, Molina E, Vázquez A, Pacheco-Fernández T, Saavedra R, Pérez-Plasencia C, Chirino YI, Vaca-Paniagua F, Arias-Romero LE, et al. Early and partial reduction in CD4+Foxp3+ regulatory T cells during Colitis-associated colon cancer induces CD4+ and CD8+ T cell activation inhibiting tumorigenesis. J Cancer. 2018;9:239–249. doi: 10.7150/jca.21336. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.