Abstract
C-X-C motif chemokine ligand 17 (CXCL17) is a mucous chemokine and its expression is highly correlated with that of G protein-coupled receptor 35 (GPR35), which has been confirmed as its receptor and named C-X-C motif chemokine receptor 8 (CXCR8). CXCL17 is upregulated in several types of cancer. However, the biological role of this chemokine in colon cancer remains unknown. In the present study, the expression levels of CXCL17 and CXCR8 were examined using immunohistochemistry in 101 colon cancer tissues and 79 healthy tumour-adjacent tissues. CXCL17 and CXCR8 expression levels were increased in the colon cancer samples compared with tumour-adjacent samples. Patients with high CXCL17 expression had longer overall survival (OS) compared with patients with low expression of CXCL17 (log-rank test; P=0.027). However, CXCR8 expression, but not CXCL17, was an independent prognostic factor for OS in patients with colon cancer. The expression of CXCR8 correlated positively with that of CXCL17 in colon cancer samples (ρ=0.295; P=0.003). Furthermore, the combined high expression of CXCL17 and CXCR8 was a significant independent prognostic factor for OS in patients with colon cancer (P=0.001). In subgroups with a TNM stage of I–II, the patients with combined high expression of CXCL17 and CXCR8 had a longer survival compared with those without combined high expression (P=0.001). However, this difference was not observed in subgroups with a TNM stage of III–IV. Collectively, these findings suggest that CXCL17/CXCR8 signalling may be involved in colon cancer and contribute to improved patient outcomes.
Keywords: CXCL17, CXCR8, GPR35, colon cancer, immunohistochemistry
Introduction
Chemokines are a large family of cytokines serving important roles in the immune system, such as the regulation of cell trafficking to inflammatory sites (1). By regulating the activity of cells in homeostasis and signal transduction, chemokines are involved in numerous physiological and pathological processes such as angiogenesis, embryonic development, wound healing, organ sclerosis, tumour growth and autoimmune disease (2,3).
Chemokine (C-X-C motif) ligand 17 (CXCL17) was first identified in 2006 and is also known as vascular endothelial growth factor-correlated chemokine-1 or dendritic cell and monocyte chemokine-like protein (4,5). CXCL17 is widely expressed in mucosal tissues and is considered a mucous chemokine (6,7). Moreover, this chemokine has potential anti-inflammatory and antibacterial effects and is related to the homeostasis of the intramucosal environment (6,8). Matsui et al (8) reported that the expression of CXCL17 was common in colon and breast carcinoma cell lines, some gastric cell lines, non-small cell lung cancer and pancreatic carcinoma cell lines, but not in melanoma cell lines. However, in vivo, the significance of CXCL17 expression in different types of tumour is inconsistent. Previous studies involving cancer types, such as endometrial cancer (9), hepatocellular cancer (10), lung cancer (8) and colon cancer (11), suggested that CXCL17 was upregulated in tumour tissue, which was associated with oncogenicity and poor prognosis. However, CXCL17 expression has also been reported to be downregulated in pancreatic cancer (12) and gastric cancer (13).
Maravillas-Montero et al (14) suggested that the levels of orphan receptor G-coupled protein receptor 35 (GPR35) in mucous membranes correlated with the expression of CXCL17. The same study also confirmed that GPR35 was the receptor for CXCL17, and this was renamed CXCR8 (14). The relationship between both proteins and the associated and signalling axis have since been studied in tumours. For instance, Guo et al (15) reported that both CXCL17 and CXCR8 were highly expressed in breast cancer tissues and cell lines, but no correlation was observed between the expression of the two proteins. However, high expression of CXCR8 correlated with histological grade and high expression of Ki67 (15). CXCL17 is associated with shorter overall survival (OS) and represents an independent predictive factor of poor prognosis. In addition, the same study also identified how the CXCL17/CXCR8 signalling axis may play a role in activating the ERK pathway and contribute to the proliferation and migration of breast cancer cells (15). Khandelwal et al (16) examined the role of CXCL17 and CXCR8 in the pathogenesis of cutaneous squamous cell carcinoma (CSCC) and revealed that these were significantly expressed in CSCC cell lines. Moreover, stimulation with CXCL17 significantly induced CSCC cell proliferation and migration. Therefore, CXCL17/CXCR8 signalling in CSCC could represent a potentially novel target for the treatment of non-melanoma skin cancer (16).
As CXCL17 is a mucous chemokine that is highly expressed in several tumour types, this chemokine may serve an important role in the pathophysiological processes of colon cancer. However, the biological role of CXCL17 in the onset and progression of colon cancer remains unknown. Therefore, the aim of the present study was to examine the significance of both CXCL17 and its receptor in colon cancer by investigating their expression patterns in Chinese patients. The association of these two proteins with clinicopathological parameters and survival rates was also investigated. Furthermore, CXCL17 and CXCR8 co-expression in colon cancer tissue was assessed in order to determine the prognostic value of the combined high expression of both proteins.
Materials and methods
Clinical samples and microarray
A commercial tissue microarray (TMA) was obtained from The National Human Genetic Resources Sharing Service Platform (Shanghai Outdo Biotech Co. Ltd.). The TMA samples were from 101 patients with sporadic colon cancer who had undergone surgery and 79 healthy tumour-adjacent samples were included for comparative analysis. The colon tissues outside the tumour loci were selected as healthy tumour-adjacent samples that exhibited no tumour texture histologically. Clinicopathological data included patient age (43–91 years old), sex (male:female, 50:61), tumour type, lymph node metastasis, histological grade and TNM stage (17). The survival data were obtained from the supplier of the TMA by follow-up via telephone consultation. The ‘event’ for the purposes of survival analysis was defined as colon cancer-related mortality. Follow-up information was available for 101 patients with colon cancer. The average follow-up period was 61.96±3.72 months (range, 2–97 months; between September 2007 and July 2015). During the follow-up, mortality occurred in 52 cases. OS was calculated as the time between the first day of diagnosis and colon cancer related mortality or the last known follow-up. The median OS was 89.0 months. All patients provided specimens with written informed consent and approval from the Ethics Committee in The Taizhou Hospital of Zhejiang Province was obtained in this study.
Data on CXCL17 and GPR35 mRNA expression from TCGA
Data on CXCL17 and GPR35 mRNA expression levels in colon cancer tissues from American patients including survival information were obtained from The Cancer Genome Atlas (TCGA) (https://portal.gdc.cancer.gov) in March 2020 (Data on ‘CXCR8’ as the key word was not available from TCGA). The results without follow-up were excluded and the means of the repeated detection outcomes were used for statistical analysis.
Immunohistochemistry (IHC) staining
The sections (4 µm thick) wereobtained from 10% formalin-fixed for 24 h and paraffin-embedded TMA blocks and used for IHC. The sections were washed in xylene to remove the paraffin and rehydrated in a graded alcohol series of 100, 100, 90, 80 and 70%, followed by PBS washing. Antigen retrieval was achieved by heating sections in EDTA antigen retrieval solution for 20 min at 99°C. Endogenous peroxidase activity was blocked using 3% H2O2/methanol for 20 min at 37°C. After blocking non-specific protein binding with 10% normal goat serum (Boster Biological Technology co.ltd) for 30 min at 37°C, the sections were incubated with primary antibodies against CXCL17 (1:50; cat. no. 422208; R&D Systems, Inc.) and CXCR8 (1:750; cat. no. DF4973; Affinity Biosciences), respectively, overnight at 4°C. After leaving at room temperature for 30 min and rinsing three times in PBS, the sections were incubated with secondary antibody (ready to use, Dako K8002, Agilent Technologies, Inc.) for 30 min at 37°C. The 3,3′-diaminobenzidine substrate was applied to the sections, which were then counterstained for 1 min at room temperature with haematoxylin, dehydrated and mounted. Negative controls were run in parallel and PBS was used instead of primary antibody.
Evaluation of IHC
Immunostaining was evaluated under a light microscope (magnification, ×200) by two pathologists blinded to the experimental conditions and clinicopathological information. Immunostaining was evaluated using a semi-quantitative scoring system, according to the percentage of stained cells (0, <10%; 1, 10–29%; 2, 30–49%; 3, 50–74%; 4, ≥75%.) and the intensity of immunoreactivity (1, weak; 2, moderate; 3, strong staining). The final immunostaining score was determined by multiplying the intensity score with the score for the percentage of positively stained cells, which ranged between 0–12 (18). Agreement on the final scores between the two pathologists was ~95%. In cases where different scores were obtained, the IHC scoring was repeated by both pathologists until the same score was achieved.
Receiver operating curve (ROC) analysis was performed to determine an optimal cut-off value for the final immunostaining scores of CXCL17 and CXCR8 expression. Because of a large difference between the expression of CXCL17 and CXCR8, the expression levels of each were graded by different criteria. The area under the curve (AUC) with the largest area for depth of invasion had the greatest Youden's index at 5 of the final scores for CXCL17. Therefore, low and high expression levels of CXCL17 were defined by a final score of <6 and ≥6, respectively (data not shown). Moreover, the AUC with the largest area for histological grade had the largest Youden's index at 7 of the final scores for CXCR8. Thus, low and high expression levels of CXCR8 were defined by a final score of ≤6 and >6, respectively (data not shown).
Statistical analysis
Analyses were performed with SPSS 16.0 (SPSS, Inc.). Spearman correlation analysis and Pearson's χ2 test were used to analyse the association between CXCL17 and CXCR8 expression and clinicopathological parameters, as well as the association between CXCL17 and CXCR8 expression in colon cancer tissue. Survival probabilities were estimated using the Kaplan-Meier method and the log rank test. Univariate and multivariate Cox proportional hazards regression models were applied to assess the association between potential confounding variables and prognosis. In order to avoid missing important information, multivariate analysis included variables that had P<0.1 in univariate analysis. Survival analysis was conducted on the data from TCGA by Kaplan-Meier method and log rank test based on both medians and means of mRNA expression. P<0.05 was considered to indicate a statistically significant difference.
Results
CXCL17 and CXCR8 expression in colon cancer
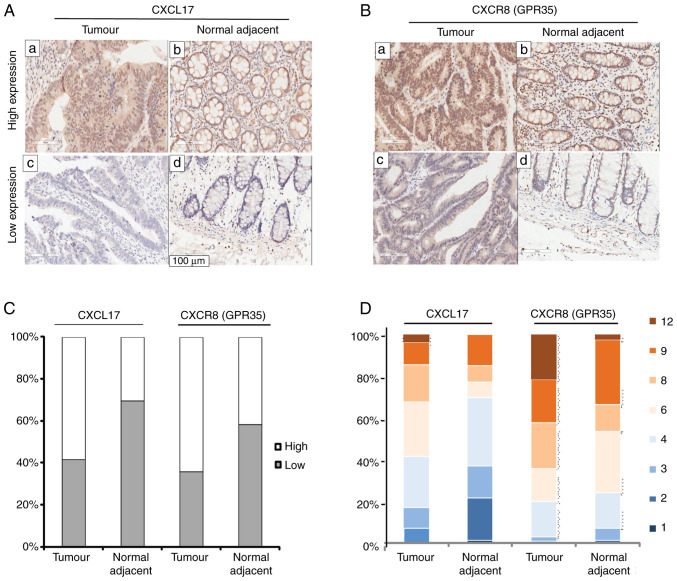
In order to investigate the expression levels of CXCL17 and CXCR8 in colon cancer tissues, IHC staining for CXCL17 and CXCR8 was performed on a commercial colon cancer TMA with complete pathological and survival information. CXCL17 expression was mainly detected in the cytoplasm of colon cancer cells, but also in the nuclei and occasionally in the cell membrane. Immunoreactivity for CXCR8 was focused in the nuclei and cytoplasm of colon cancer cells compared with the healthy tumour-adjacent tissues, high expression levels of both CXCL17 (P=0.003) and CXCR8 (P=0.001) were more common in colon cancer tissues (Fig. 1C). Furthermore, high expression of CXCL17 was observed in 58.4% (59/101) of colon cancer tissues and in 30.4% (24/79) of healthy tumour-adjacent tissues. High expression levels of CXCR8 reached 66.4% (65/101) in colon cancer tissues and 41.8% (33/79) in healthy tumour-adjacent tissues. Fig. 1A and B showed high and low expression of CXCL17 and CXCR8 in colon cancer and healthy tumour-adjacent tissues, respectively. Fig. 1D shows the percentage of the final staining scores of the two proteins in colon cancer and healthy tumour-adjacent tissues. In general, the immunostaining of CXCR8 was much stronger than that of CXCL17.
Figure 1.
CXCL17 and CXCR8 expression levels in colon cancer and healthy tumour-adjacent samples. Representative micrographs of high expression (a and b) and low expression (c and d) of CXCL17 (A) and CXCR8 (B) in colon cancer samples and healthy tumour-adjacent samples. Magnification, ×200. (C) Stacked bar charts representing the proportions of high and low-expression samples in colon cancer and healthy tumour-adjacent samples. (D) Stacked bar charts representing the proportions of each immunostaining final score in different colours for CXCL17 and CXCR8 in colon cancer and healthy tumour-adjacent samples. Each colour represents each corresponding immunostaining final score. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8.
Association between the expression levels of CXCL17 and CXCR8 and the clinicopathological characteristics of colon cancer cases
The relationship between CXCL17 and CXCR8 expression levels and the clinicopathological parameters of colon cancer cases was then assessed using Pearson's χ2 test (Table I). No significant associations were identified between the expression of CXCL17 and CXCR8 and patient age, sex, tumour type, lymph nodes metastasis status, histological grade and TNM stage.
Table I.
Association between the expression of CXCL17 and CXCR8 and the clinicopathological characteristics of colon cancer cases.
| CXCL17 | CXCR8 | |||||||
|---|---|---|---|---|---|---|---|---|
| Clinicopathological characteristic | Low, n=42 | High, n=59 | P-valuea | χ2 a | Low, n=36 | High, n=65 | P-valuea | χ2 a |
| Age, years | ||||||||
| ≤67 | 23 | 26 | 0.289 | 1.123 | 17 | 32 | 0.847 | 0.037 |
| >67 | 19 | 33 | 19 | 33 | ||||
| Sex | ||||||||
| Male | 17 | 33 | 0.126 | 2.345 | 15 | 35 | 0.241 | 1.375 |
| Female | 25 | 26 | 21 | 30 | ||||
| Tumour typeb | ||||||||
| TA | 21 | 29 | 0.971 | 0.059 | 19 | 31 | 0.327 | 2.233 |
| A | 15 | 23 | 15 | 23 | ||||
| CC | 5 | 7 | 2 | 10 | ||||
| Node metastasis, n | ||||||||
| 0 | 19 | 38 | 0.404 | 1.812 | 26 | 35 | 0.101 | 4.579 |
| 1-3 | 13 | 17 | 6 | 24 | ||||
| >3 | 6 | 4 | 4 | 6 | ||||
| Histological grade | ||||||||
| 1 | 6 | 16 | 0.300 | 2.405 | 10 | 12 | 0.112 | 4.377 |
| 2 | 27 | 33 | 23 | 37 | ||||
| 3 | 9 | 10 | 3 | 16 | ||||
| Depth of invasionb | ||||||||
| T1-2 | 1 | 5 | 0.067 | 5.414 | 2 | 4 | 0.518 | 1.316 |
| T3 | 29 | 46 | 25 | 50 | ||||
| T4 | 12 | 7 | 9 | 10 | ||||
| TNM stage | ||||||||
| I–II | 23 | 38 | 0.329 | 0.954 | 26 | 35 | 0.071 | 3.271 |
| III–IV | 19 | 21 | 10 | 30 | ||||
P-value and χ2 value were obtained using a χ2 test.
There was 1 case lack of tumour type and depth of invasion, respectively. TA, tubular adenoma; A, adenoma; CC, colloid carcinoma; CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8.
Association between the expression of CXCL17 and CXCR8 and OS of patients with colon cancer
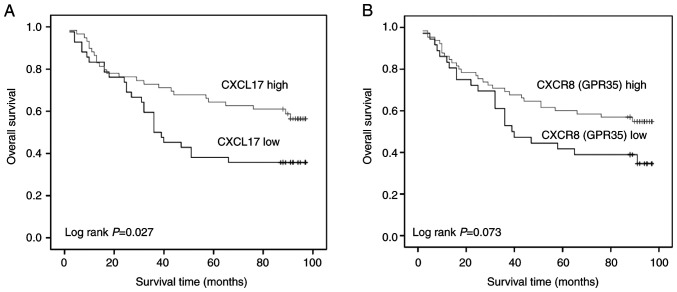
The association of CXCL17 and CXCR8 expression levels with OS in patients with colon cancer was evaluated using Kaplan-Meier analysis. Patients with a higher CXCL17 expression levels had longer OS (log rank; P=0.027). A similar trend was observed for CXCR8; however, when compared with patients with lower CXCR8 expression, no significant differences were observed (log rank; P=0.073; Fig. 2).
Figure 2.
Association between the expression of CXCL17 and CXCR8 and overall survival of patients with colon cancer. Prognostic value of (A) CXCL17 and (B) CXCR8 in colon cancer. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8.
A univariate Cox regression model was used to determine the influence of each clinicopathological variable on OS (Table II). Univariate analysis indicated that lymph node metastasis and TNM stage were significantly associated with prognosis in patients with colon cancer (RR=2.618; 95% CI, 1.514–4.527; P=0.001). However, although there appeared to be a relationship between histological grade and prognosis in patients with colon cancer, the differences in risk ratio (RR) were not statistically significant (RR=2.017; 95% CI, 0.949–4.290; P=0.068). In particular, high expression of CXCL17 reduced the RR in patients with colon cancer (RR=0.545; 95% CI, 0.316–0.942; P=0.030). While high expression of CXCR8 had a similar trend to that of CXCL17, there was no statistical significance (RR=0.610; 95% CI, 0.353–1.057; P=0.078).
Table II.
Cox regression univariate analysis of clinicopathological characteristics, CXCL17 and CXCR8 expression and overall survival in colon cancer.
| Clinicopathological characteristic | n | RR (95% CI) | P-value |
|---|---|---|---|
| Age (≤67 vs. >67 years) | 101 | 1.199 (0.695–2.070) | 0.514 |
| Sex (male vs. female) | 101 | 1.070 (0.621–1.843) | 0.809 |
| Tumour type (TA/A vs. CC) | 100 | 1.490 (0.862–2.577) | 0.153 |
| Node metastasis (yes vs. no) | 101 | 2.618 (1.514–4.527) | 0.001 |
| Histological grade (1 vs. 2/3) | 101 | 2.017 (0.949–4.290) | 0.068 |
| Depth of invasion (T1/2/3 vs. T4) | 100 | 1.488 (0.778–2.842) | 0.230 |
| TNM stage (I/II vs. III/IV) | 101 | 2.618 (1.514–4.527) | 0.001 |
| CXCL17 (low vs. high) | 101 | 0.545 (0.316–0.942) | 0.030 |
| CXCR8 (low vs. high) | 101 | 0.610 (0.353–1.057) | 0.078 |
TA, tubular adenoma; A, adenoma; CC, colloid carcinoma; CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8; RR, relative risk.
Multivariate Cox regression analysis demonstrated that TNM stage were independent prognostic factors for OS in patients with colon cancer (RR=2.793; 95% CI, 1.590–4.906; P<0.001), while histological grade and high CXCL17 expression were not. Moreover, CXCR8 expression was also an independent prognostic factor for OS (Table III); however, no statistical significance was identified using Cox regression univariate analysis (Table II).
Table III.
Cox regression multivariate analysis of CXCL17 and CXCR8 expression and overall survival in colon cancer.
| Clinicopathological characteristic | n | RR (95% CI) | P-value |
|---|---|---|---|
| TNM stage (I/II vs. III/IV) | 101 | 2.793 (1.590–4.906) | <0.001 |
| Histological grade (1 vs. 2/3) | 101 | 1.804 (0.833–3.905) | 0.135 |
| CXC17 (low vs. high) | 101 | 0.723 (0.401–1.302) | 0.280 |
| CXCR8 (low vs. high) | 101 | 0.543 (0.298–0.987) | 0.045 |
Node metastasis was not analysed as it was linearly associated with TNM stage. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8; RR, relative risk.
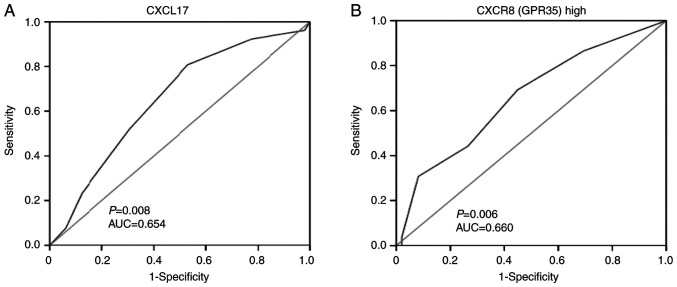
ROC analysis was performed to evaluate the prognostic significance of the expression of the two proteins in surgically treated patients with colon cancer. According to the analysed results by SPSS, the AUC for CXCL17 was 0.654, and CXCL17 expression at a score of 7 had the largest sum of sensitivity and specificity (Youden's index). Sensitivity reached 0.808 and specificity was 0.469. In addition, the AUC for CXCR8 was 0.660, and CXCR8 expression at a score of 8.5 had the greatest Youden's index with, sensitivity at 0.692 and specificity at 0.551 (Fig. 3).
Figure 3.
Receiver operating characteristic curves used to evaluate the prognostic value of CXCL17 and CXCR8 expression in colon cancer. (A) showed ROC curve for CXCL17; (B) showed ROC curve for CXCR8; Sensitivity and specificity are represented, and the AUC and associated P-value are indicated. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8; AUC, area under the curve.
CXCL17 expression is positively associated with the expression of CXCR8 and CXCL17/CXCR8 co-expression is an independent prognostic factor in colon cancer
The association between CXCL17 and CXCR8 expression in colon cancer tissues was assessed using Spearman rank correlation analysis. The expression of CXCR8 was positively correlated with the expression of CXCL17 in colon cancer samples (Ρ=0.295; P=0.003, χ2=11.317), but not in healthy tumour-adjacent samples (Table IV and Fig. 4).
Table IV.
Correlation between CXCL17 and CXCR8 expression in colon cancer samples and healthy tumour-adjacent samples.
| CXCR8, n (%) | |||||||
|---|---|---|---|---|---|---|---|
| Gene | Tissue | Low | High | ρa | P-valueb | χ2b | |
| CXCL17, n (%) | Tumour | Low | 22 (52.4) | 20 (47.6) | 0.295 | 0.003 | 8.781 |
| High | 14 (23.7) | 45 (76.3) | |||||
| Tumour-adjacent | Low | 32 (58.2) | 23 (41.8) | −0.001 | 0.990 | <0.001 | |
| High | 14 (58.3) | 10 (41.7) | |||||
ρ value was obtained using Spearman correlation analysis.
P-value and χ2 value were obtained with a χ2 test. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8.
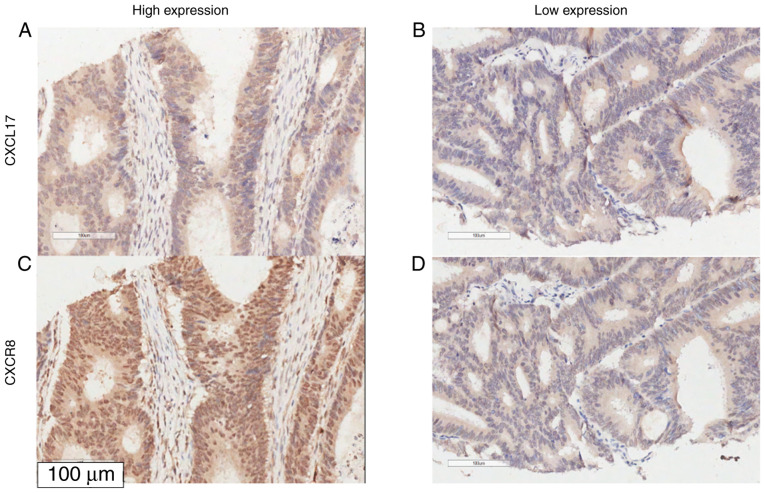
Figure 4.
Representative micrographs of combined high expression and low expression of CXCL17 and CXCR8 in colon cancer samples. (A) High expression of CXCL17; (B) low expression of CXCL17; (C) showed high expression of CXCR8 and (D) showed low expression of CXCR8. Magnification, ×200. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8.
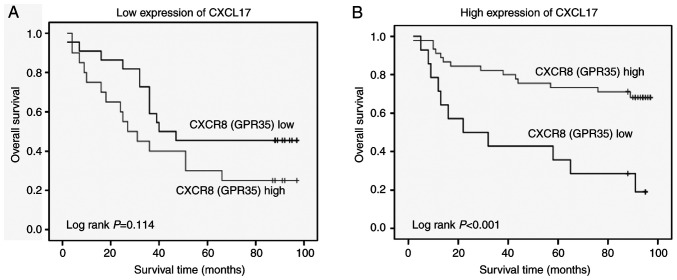
Based on the positive association between CXCL17 and CXCR8 expression and their high expression levels in colon cancer tissues, the association between different expression levels of CXCR8 and OS was investigated in patients with colon cancer stratified according to their CXCL17 expression. A longer OS was observed in patients with a high CXCR8 expression in the high CXCL17 expression group compared with those with low expression of CXCR8 (P<0.001). However, there was no significant association in low CXCR17 expression group (P=0.114; Fig. 5).
Figure 5.
Kaplan-Meier survival curves demonstrating the association between CXCR8 expression and overall survival in samples stratified according to CXCL17 expression. (A) Low CXCL17 expression. N=42. (B) High CXCL17 expression. N=59. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8.
To evaluate the prognostic value of combined high expression levels of CXCL17 and CXCR8, one low expression or both low expression of two proteins was referred to as ‘non-combined expression’. However, both high expression of two proteins was referred to as ‘combined expression’. The association between combined expression and clinicopathological characteristics in colon cancer cases was evaluated accordingly. However, the results only showed a small negative association between the depth of tumor invasion and combined expressions of CXCL17 and CXCR8, where P=−0.240 and P=0.018 by Spearman's correlation analysis (data not shown), but P=0.055 using the Pearson χ2 test (data not shown). However, multivariate Cox regression analysis suggested that the combined expression of CXCL17 and CXCR8 was a significant, independent prognostic factor for OS in patients with colon cancer (P=0.001; Table V).
Table V.
Cox regression multivariate analysis of CXCL17 and CXCR8 co-expression and overall survival in colon cancer.
| Clinicopathological characteristic | n | RR (95% CI) | P-value |
|---|---|---|---|
| TNM stage (I/II vs. III/IV) | 101 | 2.375 (1.367–4.127) | 0.002 |
| Histological grade (1 vs. 2/3) | 101 | 1.769 (0.826–3.789) | 0.142 |
| Expression of CXCL17 and CXCR8 (non-combined vs. combined) | 101 | 0.350 (0.189–0.650) | 0.001 |
Non-combined expression: One low expression or both low expression of two proteins; Combined expression: Both high expression of two proteins; Node metastasis was not analysed as it was linearly associated with TNM stage. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8; RR, relative risk.
The association between combined CXCL17/CXCR8 expression and OS was then evaluated in subgroups of patients with colon cancer stratified according to TNM stage. Patients with combined high expression of CXCL17 and CXCR8 presented a longer OS, compared with those without combined high expression in the subgroups with a TNM stage of I–II (P=0.001). However, this was not observed in patients with a TNM stage of III–IV (Table VI).
Table VI.
Association between combined expression of CXCL17 and CXCR8 and survival status in subgroups of patients with colon cancer stratified by TNM stage.
| Expression of CXCL17 and CXCR8, n (%) | |||||
|---|---|---|---|---|---|
| TNM stage | Outcome | Non-combined | Combined | P-valuea | χ2 |
| I–II | Dead | 19 (59.4) | 5 (17.2) | 0.001 | 11.317 |
| Alive | 13 (40.6) | 24 (82.8) | |||
| III–IV | Dead | 19 (79.2) | 9 (56.2) | 0.121 | 2.401 |
| Alive | 5 (20.8) | 7 (43.8) | |||
P-value and χ2 value were obtained with a χ2 test. Non-combined expression means low expression for one or both proteins. Combined expression means high expression of both proteins. CXCL17, C-X-C motif chemokine ligand 17; CXCR8, C-X-C motif chemokine receptor 8; TNM, tumour-node-metastasis.
The mRNA expression of CXCL17 and CXCR8 (GPR35) were not detected in 101 colon cancer samples and 79 healthy tumour-adjacent samples. However, in the present study, data on CXCL17 and GPR35 mRNA expression levels in colon cancer tissues from American patients were obtained from TCGA. Of the 432 patients included in the final data, mortality occurred in 87 patients. Kaplan-Meier analysis indicated no significant associations between the mRNA expression levels of CXCL17 and GPR35 and OS in patients with colon cancer; although patients with high mRNA expression levels of CXCL17 and GPR35 tended to have a longer OS time compared with those with low expression of CXCL17 and GPR35 (Fig. S1).
Discussion
In the present study, the expression levels of CXCL17 and CXCR8 were higher in colon cancer tissues compared with healthy tumour-adjacent tissues. This finding was consistent with previous studies on CXCL17 in colon cancer (8), as well as CXCR8 in breast cancer (15) and lung cancer (19). Recent studies performed at the mRNA level suggested that both CXCL17 (20) and CXCR8 (21) were ectopically expressed in primary colon cancer tissues and regional positive lymph nodes. High expression levels of CXCR8 variants 2/3 and CXCL17 mRNA in regional metastatic lymph nodes are indicators of poor prognosis in patients with colon cancer, based on the analysis of survival data. Moreover, the prognostic value of CXCR8 and CXCL17 is enhanced when combined with carcinoembryonic antigen mRNA expression. However, these studies investigated CXCL17 and CXCR8 at the mRNA level in regional lymph nodes in European patients with colon cancer (20). In the present study, data on CXCL17 and CXCR8 mRNA expression levels in colon cancer tissues from American patients were obtained from TCGA in March 2020. Although Kaplan-Meier analysis indicated no significant associations between the mRNA expression levels of CXCL17 and CXCR8 and OS in patients with colon cancer; patients with high mRNA expression levels of CXCL17 and GPR35 (CXCR8) tended to have a longer OS compared with those with low expression of CXCL17 and GPR35, which had a similar tendency to the current study of proteins expression. Ohlsson et al (11) reported that CXCL17 was also highly expressed in primary tumours compared with healthy colon tissues by IHC; however, further findings related to the clinical significance of CXCL17 and its association with survival were not reported, which could be attributed to the small cohort size (11).
To the best of the authors' knowledge, the present study was the first to investigate CXCL17 and CXCR8 co-expression using TMA from a relatively large number of cases of colon cancer, including healthy tumour-adjacent tissues. Although the association observed between the expression levels of CXCL17 and CXCR8 and the clinicopathological features was not significant in the current study, the results suggested that patients with high expression of either CXCL17 or CXCR8 presented longer survival times compared with patients with low expression of either of the two genes. In previous studies (11,15,22), high expression of CXCL17 was also mostly observed in cancer tissues, such as breast cancer, and the prognosis of patients with breast cancer with high expression of CXCL17 was poor (15). The prognostic value of high expression levels of CXCL17 in hepatocellular carcinoma (HCC) has been reported to be similar to that in breast cancer (15,23). A recent study revealed that CXCL17 in HCC mediates the invasion of malignant cells (22). In addition, high GPR35 expression correlates with drug resistance in lung cancer (19). Compared with studies on other malignancies, high CXCL17 and CXCR8 expression in the present study was associated with different outcomes, as patients with high expression had an improved prognosis. Moreover, increased CXCL17 expression in different tissues and organs can largely vary under distinct pathophysiological conditions (4–6), suggesting that this chemokine serves a specific biological role in specific tissues.
Further univariate and multivariate COX regression analyses indicated that lymph node metastasis and clinical staging were independent prognostic factors in colon cancer. However, the expression of CXCL17 was not a statistically significant independent prognostic factor when using multivariate analysis. In addition, CXCR8, which had a slightly higher P-value compared with the statistical significance threshold in univariate analysis, was a strong independent prognostic factor in the multivariate analysis. This trend should be further investigated using studies that involve a larger number of cases. A possible reason why CXCR8 was a strong independent prognostic factor when using multivariate analysis is that CXCL17 exhibits its biological effects via interaction with CXCR8.
The association between the CXCL17 and CXCR8 expression levels and the potential significance of their combined high expression was also examined. The expression levels of the two proteins were associated in cancer tissues, but there was no significant association between combined high CXCL17/CXCR8 expression and clinicopathological variables. Nonetheless, patients with combined high expression of the two proteins presented longer survival times compared with those with non-combined expression. However, the present data indicated that the expression of the two proteins did not both increase or decrease simultaneously, therefore there was not a totally dependent relationship between the two proteins. Prior to the identification of CXCR8 as a receptor of CXCL17, endogenous ligands of CXCR8 have been reported to include kynurenic acid, lodoxamide (14,24,25) and lysophosphatidic acid (26). Walczak et al (25) demonstrated that kynurenic acid synthesis increased in healthy and cancer colorectal cells. Recent studies have also hypothesised whether CXCR8 is the receptor of CXCL17. It has been reported that THP-1 cells pre-treated with a CXCR8 antagonist have no significant effects on migration to the gradient CXCL17, and the migration of THP-1 cells stimulated by CXCL17 was not dependent on CXCR8 (27,28). However, the present results suggested a close functional association between CXCL17 and CXCR8 in colon cancer, which is consistent with previous studies supporting their ligand-receptor relationship (14–16).
The present study indicated that the CXCL17/CXCR8 axis may have a protective biological role in colon cancer, and it is possible that CXCL17 promotes the antitumour immunity in patients with colon cancer. However, it has been reported that CXCL17-responding myeloid-derived cells cause a notable enhancement of xenograft tumour formation (8). Hiraoka et al (12) injected CMS5a fibrosarcoma cells and CT26 colon cancer cells stably expressing CXCL17 and/or intercellular adhesion molecule 2 (ICAM2) into BALB/c mice, which resulted in slower tumour growth compared with mice injected with wild-type cells; indeed, tumours failed to develop in the former group. Moreover, Hiraoka et al (12) concluded that tumour growth was inhibited by immune surveillance by cytotoxic T cell-mediated cytolysis in the context of CXCL17 and ICAM2 during the early stages of pancreatic carcinogenesis. However, the antitumor immune reaction changed from an immune response to immune tolerance between the stages of intraductal papillary mucinous adenoma and intraductal papillary mucinous carcinoma (12). In line with these findings, the present study demonstrated that the combined high expression of CXCL17 and CXCR8 in patients with early TNM staging presented longer survival times compared with those without combined high expressions of CXCL17 and CXCR8. However, this was not the case in patients with late TNM staging. These findings support an antitumour role for CXCL17/CXCR8 signalling in the early stages of colon cancer.
In conclusion, the results of the present study indicated that CXCL17/CXCR8 signalling is involved in colon cancer. While the underlying biological mechanism is yet to be elucidated, these findings suggested that CXCL17 and CXCR8 may serve as useful biomarkers for improved prognosis of colon cancer. However, further experimental validation is required to verify the protective effect of CXCL17/CXCR8 signalling in colon cancer.
Supplementary Material
Acknowledgements
Not applicable.
Glossary
Abbreviations
- CXCL17
C-X-C motif chemokine ligand 17
- CXCR8
C-X-C motif chemokine receptor 8
- GPR35
G-coupled protein receptor 35
- OS
overall survival
- RR
relative risk
Funding
The present study was supported by Liaoning Science and Technology Projects (grant no. 2014022040).
Availability of data and materials
All data generated or analysed during this study are included in this published article.
Authors' contributions
XL and HY contributed to the conception and design of the study. HY, YL and XB performed IHC investigation, statistical analysis and prepared figures and tables. XB, ZY and YL interpreted the results. XL, HY and ZY drafted and revised the manuscript. All authors read and approved the final manuscript.
Ethics approval and consent to participate
The commercial tissue microarray specimens were collected from Taizhou Hospital by Shanghai Outdo Biotech Co., Ltd. All patients provided specimens with written informed consent and approval from the Ethics Committee in The Taizhou Hospital of Zhejiang Province was obtained in this study.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Sokol CL, Luster AD. The chemokine system in innate immunity. Cold Spring Harb Perspect Biol. 2015;7:a016303. doi: 10.1101/cshperspect.a016303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Rotondi M, Chiovato L, Romagnani S, Serio M, Romagnani P. Role of chemokines in endocrine autoimmune diseases. Endocr Rev. 2007;28:492–520. doi: 10.1210/er.2006-0044. [DOI] [PubMed] [Google Scholar]
- 3.Raman D, Baugher PJ, Thu YM, Richmond A. Role of chemokines in tumor growth. Cancer Lett. 2007;256:137–165. doi: 10.1016/j.canlet.2007.05.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Weinstein EJ, Head R, Griggs DW, Sun D, Evans RJ, Swearingen ML, Westlin MM, Mazzarella R. VCC-1, a novel chemokine, promotes tumor growth. Biochem Biophys Res Commun. 2006;350:74–81. doi: 10.1016/j.bbrc.2006.08.194. [DOI] [PubMed] [Google Scholar]
- 5.Pisabarro MT, Leung B, Kwong M, Corpuz R, Frantz GD, Chiang N, Vandlen R, Diehl LJ, Skelton N, Kim HS, et al. Cutting edge: Novel human dendritic cell- and monocyte-attracting chemokine-like protein identified by fold recognition methods. J Immunol. 2006;176:2069–2073. doi: 10.4049/jimmunol.176.4.2069. [DOI] [PubMed] [Google Scholar]
- 6.Burkhardt AM, Tai KP, Flores-Guiterrez JP, Vilches-Cisneros N, Kamdar K, Barbosa-Quintana O, Valle-Rios R, Hevezi P, Zuñiga J, Selman M, et al. CXCL17 is a mucosal chemokine elevated in idiopathic pulmonary fibrosis that exhibits broad antimicrobial activity. J Immunol. 2012;188:6399–6406. doi: 10.4049/jimmunol.1102903. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Hernández-Ruiz M, Zlotnik A. Mucosal chemokines. J Interferon Cytokine Res. 2017;37:62–70. doi: 10.1089/jir.2016.0076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Matsui A, Yokoo H, Negishi Y, Endo-Takahashi Y, Chun NA, Kadouchi I, Suzuki R, Maruyama K, Aramaki Y, Semba K, et al. CXCL17 expression by tumor cells recruits CD11b+Gr1 high F4/80-cells and promotes tumor progression. PLoS One. 2012;7:e44080. doi: 10.1371/journal.pone.0044080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Saghir FS, Rose IM, Dali AZ, Shamsuddin Z, Jamal AR, Mokhtar NM. Gene expression profiling and cancer-related pathways in type I endometrial carcinoma. Int J Gynecol Cancer. 2010;20:724–731. doi: 10.1111/IGC.0b013e3181e1c14c. [DOI] [PubMed] [Google Scholar]
- 10.Mu X, Chen Y, Wang S, Huang X, Pan H, Li M. Overexpression of VCC-1 gene in human hepatocellular carcinoma cells promotes cell proliferation and invasion. Acta Biochim Biophysica Sinica. 2009;41:631–637. doi: 10.1093/abbs/gmp051. [DOI] [PubMed] [Google Scholar]
- 11.Ohlsson L, Hammarström ML, Lindmark G, Hammarstrom S, Sitohy B. Ectopic expression of the chemokine CXCL17 in colon cancer cells. Br J Cancer. 2016;114:697–703. doi: 10.1038/bjc.2016.4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hiraoka N, Yamazaki-Itoh R, Ino Y, Mizuguchi Y, Yamada T, Hirohashi S, Kanai Y. CXCL17 and ICAM2 are associated with a potential anti-tumor immune response in early intraepithelial stages of human pancreatic carcinogenesis. Gastroenterology. 2011;140:310–321. doi: 10.1053/j.gastro.2010.10.009. [DOI] [PubMed] [Google Scholar]
- 13.Mao Y, Zhao Q, Yin S, Ding X, Wang H. Genome-wide expression profiling and bioinformatics analysis of deregulated genes in human gastric cancer tissue after gastroscopy. Asia Pac J Clin Oncol. 2018;14:e29–e36. doi: 10.1111/ajco.12688. [DOI] [PubMed] [Google Scholar]
- 14.Maravillas-Montero JL, Burkhardt AM, Hevezi PA, Carnevale CD, Smit MJ, Zlotnik A. Cutting edge: GPR35/CXCR8 is the receptor of the mucosal chemokine CXCL17. J Immunol. 2015;194:29–33. doi: 10.4049/jimmunol.1401704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Guo YJ, Zhou YJ, Yang XL, Shao ZM, Ou ZL. The role and clinical significance of the CXCL17-CXCR8 (GPR35) axis in breast cancer. Biochem Biophys Res Commun. 2017;493:1159–1167. doi: 10.1016/j.bbrc.2017.09.113. [DOI] [PubMed] [Google Scholar]
- 16.Khandelwal AR, Alam MM, Moore-Medlin T, Savage HA, Nathan CAO. Role of the CXCL17-CXCR8 (GPR35) axis in cutaneous squamous cell carcinoma. Cancer Res. 2019:79. [Google Scholar]
- 17.Lan YT, Yang SH, Chang SC, Liang WY, Li AF, Wang HS, Jiang JK, Chen WS, Lin TC, Lin JK. Analysis of the seventh edition of american joint committee on colon cancer staging. Int J Colorectal Dis. 2012;27:657–663. doi: 10.1007/s00384-011-1366-6. [DOI] [PubMed] [Google Scholar]
- 18.Liu X, Xiao Q, Bai X, Yu Z, Sun M, Zhao H, Mi X, Wang E, Yao W, Jin F, et al. Activation of STAT3 is involved in malignancy mediated by CXCL12-CXCR4 signaling in human breast cancer. Oncol Rep. 2014;32:2760–2768. doi: 10.3892/or.2014.3536. [DOI] [PubMed] [Google Scholar]
- 19.Wang W, Han T, Tong W, Zhao J, Qiu X. Overexpression of GPR35 confers drug resistance in NSCLC cells by β-arrestin/Akt signaling. Onco Targets Ther. 2018;11:6249–6257. doi: 10.2147/OTT.S175606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Rashad Y, Olsson L, Israelsson A, Öberg Å, Lindmark G, Hammarström ML, Hammarström S, Sitohy B. Lymph node CXCL17 messenger RNA: A new prognostic biomarker for colon cancer. Tumour Biol. 2018;40:1010428318799251. doi: 10.1177/1010428318799251. [DOI] [PubMed] [Google Scholar]
- 21.Ali H, AbdelMageed M, Olsson L, Israelsson A, Lindmark G, Hammarström ML, Hammarström S, Sitohy B. Utility of G protein-coupled receptor 35 expression for predicting outcome in colon cancer. Tumour Biol. 2019;41:1010428319858885. doi: 10.1177/1010428319858885. [DOI] [PubMed] [Google Scholar]
- 22.Wang L, Li H, Zhen Z, Ma X, Yu W, Zeng H, Li L. CXCL17 promotes cell metastasis and inhibits autophagy via the LKB1-AMPK pathway in hepatocellular carcinoma. Gene. 2019;690:129–136. doi: 10.1016/j.gene.2018.12.043. [DOI] [PubMed] [Google Scholar]
- 23.Li L, Yan J, Xu J, Liu CQ, Zhen ZJ, Chen HW, Ji Y, Wu ZP, Hu JY, Zheng L, Lau WY. CXCL17 expression predicts poor prognosis and correlates with adverse immune infiltration in hepatocellular carcinoma. PLoS One. 2014;9:e110064. doi: 10.1371/journal.pone.0110064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang X, Deavers M, Patenia R, Bassett RL, Jr, Mueller P, Ma Q, Wang E, Freedman RS. Monocyte/macrophage and T-cell infiltrates in peritoneum of patients with ovarian cancer or benign pelvic disease. J Transl Med. 2006;4:30. doi: 10.1186/1479-5876-4-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Walczak K, Dabrowski W, Langner E, Zgrajka W, Piłat J, Kocki T, Rzeski W, Turski WA. Kynurenic acid synthesis and kynurenine aminotransferases expression in colon derived normal and cancer cells. Scand J Gastroenterol. 2011;46:903–912. doi: 10.3109/00365521.2011.579159. [DOI] [PubMed] [Google Scholar]
- 26.Oka S, Ota R, Shima M, Yamashita A, Sugiura T. GPR35 is a novel lysophosphatidic acid receptor. Biochem Biophys Res Commun. 2010;395:232–237. doi: 10.1016/j.bbrc.2010.03.169. [DOI] [PubMed] [Google Scholar]
- 27.Binti Mohd Amir NAS, Mackenzie AE, Jenkins L, Boustani K, Hillier MC, Tsuchiya T, Milligan G, Pease JE. Evidence for the Existence of a CXCL17 receptor distinct from GPR35. J Immunol. 2018;201:714–724. doi: 10.4049/jimmunol.1700884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Park SJ, Lee SJ, Nam SY, Im DS. GPR35 mediates lodoxamide-induced migration inhibitory response but not CXCL17-induced migration stimulatory response in THP-1 cells; is GPR35 a receptor for CXCL17? Br J Pharmacol. 2018;175:154–161. doi: 10.1111/bph.14082. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analysed during this study are included in this published article.