Abstract
Pneumonia is a persistent and pervasive disease, the effects of which can be severe. MicroRNA (miR)-127-5p has been utilized as a novel biomarker for the diagnosis of severe pneumonia. The present study aimed to investigate the function of miR-127-5p during severe pneumonia. An in vitro model of severe pneumonia in Ana-1 murine macrophages was established using lipopolysaccharide (LPS). Subsequently, reverse transcription-quantitative PCR and ELISA were performed to detect the mRNA and protein expression levels of interleukin (IL)-1β, IL-6 and tumor necrosis factor (TNF)-α. Western blotting was also performed to measure the activity of AKT and NF-κB. The results indicated that compared with the control group, LPS treatment increased TNF receptor-associated factor 1 (TRAF1) expression levels and reduced miR-127-5p expression levels. Furthermore, the results revealed that the 3'-untranslated region of TRAF1 was targeted by miR-127-5p. miR-127-5p mimic reduced LPS-induced increases in IL-1β, IL-6 and TNF-α expression by targeting TRAF1, which was potentially mediated by inactivation of the AKT and NF-κB signaling pathways. Collectively, the results demonstrated that miR-127-5p may attenuate severe pneumonia by reducing LPS-induced inflammatory cytokine production, and inactivating the AKT and NF-κB signaling pathways by targeting TRAF1.
Keywords: microRNA-127-5p, severe pneumonia, tumor necrosis factor receptor-associated factor 1
Introduction
Pneumonia is a persistent and pervasive disease (1). Despite not usually being fatal, the effects of the disease can be severe; for example, 25% of American pediatric patients (age, 1-6 years) hospitalized for pneumonia were admitted to intensive care units (ICUs), with 33.3% of pediatric ICU patients requiring mechanical ventilation in 2015 (2,3). Therefore, identifying the molecular mechanisms mediating severe pneumonia is important to reduce the disease burden.
MicroRNAs (miRNAs/miRs) are endogenous, evolutionarily conserved, non-coding RNAs that are ~22 nucleotides in length. miRNAs post-transcriptionally regulate gene expression by targeting and binding to the 3'-untranslated region (UTR) of target mRNAs (4-7). In mammals, miRNAs are associated with various diseases, including cancer (8) and viral infections (9), and have been identified as biomarkers for pneumonia (10). Moreover, numerous miRNAs are involved in the progression of pneumonia; miR-302e reduced inflammation during infantile pneumonia via the NF-κB signaling pathway (11), miR-146a-5p regulated lipopolysaccharide (LPS)-induced cell apoptosis and inflammation via CC motif chemokine ligand 5 during acute pneumonia (12), and miR-370-3p modulated LPS-induced cell apoptosis and inflammation via toll like receptor 4 during acute pneumonia (13). It has also been reported that miR-127 may reduce lung inflammation by targeting immunoglobulin (Ig)G Fcγ receptor I (14). Additionally, miR-127-5p expression, which is reduced in bronchoalveolar lavage fluid, may serve as a biomarker for the diagnosis of the disease (15). The present study aimed to investigate the function of miR-127-5p during severe pneumonia, as well as the potential molecules regulated by miR-127-5p, to identify a potential therapeutic target for severe pneumonia.
Materials and methods
Cell culture
Ana-1 murine macrophages were obtained from The Cell Bank of Type Culture Collection of the Chinese Academy of Sciences and cultured in RPMI-1640 medium (Gibco; Thermo Fisher Scientific, Inc.) supplemented with 10% low endotoxin fetal calf serum (Hyclone; GE Healthcare Life Sciences) and 1% penicillin-streptomycin (Gibco; Thermo Fisher Scientific, Inc.) at 37˚C with 95% humidity and 5% CO2.
An in vitro model of pneumonia was induced by treating Ana-1 murine macrophages with 0.1 µg/ml LPS (Sigma-Aldrich; Merck KGaA) at 37˚C for 24 h (16). Subsequently, cells were randomly divided into the following four groups: Control (treated with saline), LPS, and/or LPS + miR-127-5p mimic and/or LPS + miR-127-5p mimic + pcDNA3.1-TRAF1.
Cell transfection
miR-NC mimic (5'-UAGUCUCGGGAGAC UCACUACC-3') and miR-127-5p mimic (5'-UAGUCUCGGG AGACUCGAAGUC-3') were obtained from Guangzhou Ribobio Co., Ltd. miR-NC mimic (200 nM) or miR-127-5p mimic (200 nM) were mixed with Lipofectamine® 2000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.) and incubated for 15 min at room temperature. Subsequently, Ana-1 murine macrophages were seeded (1x106 cells/well) into 6-well plates and the mimic-Lipofectamine mix was added to each well.
TRAF1 was amplified from Ana-1 murine macrophages by PCR at thermocycling conditions of 95˚C for 15 min, followed by 30 cycles of denaturation at 98˚C for 10 sec, annealing at 55˚C for 30 sec and extension at 72˚C for 30 sec using PrimeSTAR Max DNA Polymerase (Takara Bio, Inc.) and then cloned into pcDNA3.1 (Thermo Fisher Scientific, Inc.) to generate pcDNA3.1 TRAF1. A total of 2 µg pcDNA3.1 TRAF1 and pcDNA3.1 (control) were transfected into Ana-1 murine macrophages (1x105) using Lipofectamine® 2000 reagent (Thermo Fisher Scientific, Inc.).
Following incubation for 48 h at 37˚C, transfection efficiency was determined by reverse transcription-quantitative PCR (RT-qPCR). All experiments were performed 48 h post-transfection.
ELISA
Ana-1 murine macrophages (5x105 cells/well) were plated into 24-well plates and incubated at 37˚C with 95% humidity and 5% CO2 overnight. The protein levels of TNF-α (cat. no. BMS607-3; Invitrogen; Thermo Fisher Scientific, Inc.), IL-6 (cat. no. RAB0308; Sigma-Aldrich; Merck KGaA) and IL-1β (cat. no. RAB0274; Sigma-Aldrich; Merck KGaA) in the cell media were assessed using ELISA kits, according to the manufacturer's protocol.
RT-qPCR
Total RNA was extracted from Ana-1 murine macrophages using TRIzol® (Invitrogen; Thermo Fisher Scientific, Inc.) and mirVana kits (Applied Biosystems; Thermo Fisher Scientific, Inc.) for the detection of RNA and miRNA, respectively, according to the manufacturer's protocol. The TaqMan Gene Expression assay and TaqMan MicroRNA Reverse Transcription kits (Applied Biosystems; Thermo Fisher Scientific, Inc.) were used to reverse transcribe RNA(RT temperature protocols: (50˚C for 2 min, 95˚C for 10 min, followed with 95˚C for 15 sec and 60˚C for 1 min for 40 cycles) and miRNA (RT temperature protocols: 16˚C for 30 min, 42˚C for 30 min and 85˚C for 5 min) to cDNA, respectively, according to the manufacturer's protocol. Subsequently, qPCR was performed using a SYBR Green qPCR Master Mix kit (Takara Biotechnology Co., Ltd) the StepOnePlus™ Real-Time PCR system (Applied Biosystems; Thermo Fisher Scientific, Inc.) with the following thermocycling conditions: initial denaturation at 95˚C for 2 min, 35 cycles of 95˚C for 15 sec and 64˚C for 30 sec. The kit and the system were used according to the manufacturer's protocol. The following primer pairs were used for qPCR: miR-127-5p, forward 5'-CT CTTCAAGCTCCAAACCAAAC-3', reverse 5'-GTATCC ACCAGAACCACCAGG-3'; IL-1β, forward 5'-GAAAGC TCTCCACCTAATG-3', reverse 5'-GCCGTCTTTCATTACA CAGG-3'; IL-6, forward 5'-CCAGAGATACAAAGAAATG ATGG-3', reverse 5'-ACTCCAGAAGACCAGAGGAAA-3'; TNF-α, forward 5'-TCTCATCAGTTCTATGGCCC-3', reverse 5'-GGGATGAGACAAGGTACAAC-3'; U6, forward 5'-AT TGGAACGATACAGAGAAGATT 3', reverse 5'-GGAACGCT TCACGAATTTG 3'; and GAPDH, forward 5'-TGATGACA TCAAGAAGGTGGTGAAG-3' and reverse 5'-TCCTTGGA GGCCATGTGGGCCAT-3'. mRNA and miRNA expression levels were quantified using the 2-ΔΔCq method (17). mRNA and miRNA expression levels were normalized to the internal reference genes GAPDH and U6, respectively.
Western blotting
Total protein was extracted from Ana-1 murine macrophages using RIPA buffer (Roche Diagnostics) and protein concentrations were determined using BCA (Beyotime Institute of Biotechnology). Proteins were then separated via 8% SDS-PAGE and transferred to PVDF membranes (EMD Millipore), which were subsequently blocked with 5% non-fat milk at room temperature for 1 h. The membranes were incubated at 4˚C overnight with primary antibodies targeted against the following: GAPDH (cat no. 5174; 1:1,000), TRAF1 (cat no. 4710; 1:1,000), phosphorylated (p)-AKT (cat no. 4060; 1:1,000), AKT (cat no. 4691; 1:1000), p-p65 (cat no. 3033; 1:1,000) and p65 (cat no. 8242; 1:1,000; all, Cell Signaling Technology, Inc.). Following primary antibody incubation, the membranes were incubated with an anti-rabbit horseradish peroxidase-conjugated IgG secondary antibody (cat no. 7047; 1:2,000; Cell Signaling Technology, Inc.) at room temperature for 2 h. Protein bands were visualized using an enhanced chemiluminescence detection system (PerkinElmer, Inc.). Protein expression was semi-quantified using Quantity One software (version 4.62; Bio-Rad Laboratories, Inc.) with GAPDH as the loading control.
Dual luciferase reporter assay
TargetScan (version 7.1; www.targetscan.org/vert_71) was used to predict the binding site between miR-127-5p and the 3'-UTR of TRAF1.
The wild-type (WT) 3'-UTR of TRAF1, containing complementary sequences for the seed sequence of miR-127-5p, was amplified from Ana-1 murine macrophages via PCR with the thermocycling conditions of 95˚C for 15 min, followed by 30 cycles of denaturation at 98˚C for 10 sec, annealing at 55˚C for 30 sec and extension at 72˚C for 30 sec via PrimeSTAR Max DNA Polymerase (Takara Bio, Inc.) and cloned into the psi-CHECK-2 vector (Promega Corporation). The mutant (MUT) 3'-UTR of TRAF1 was constructed using the QuikChange II Site-Directed Mutagenesis kit (Agilent Technologies, Inc.), according to the manufacturer's protocol.
Ana-1 murine macrophages (1x104 cells/well) were seeded into 96-well plates and transfected with psi-CHECK-2-TRAF1-WT-3'UTR (400 ng) or psi-CHECK-2-TRAF1-MUT-3'-UTR (400 ng) and miR-127-5p mimic (50 ng) or miR-NC mimic (50 ng) using Lipofectamine® 2000 reagent (Invitrogen; Thermo Fisher Scientific, Inc.). Following incubation for 48 h at 37˚C, luciferase activities were determined using a Dual-Luciferase assay system (Promega Corporation), according to the manufacturer's protocol. Firefly luciferase activity was normalized to Renilla luciferase activity.
Statistical analysis
Statistical analyses were performed using GraphPad Prism software (version 5.04; GraphPad Software, Inc.). Data are expressed as the mean ± standard error of the mean. Differences between two groups were analyzed using the unpaired Student's t-test. Differences among four groups were analyzed using one-way ANOVA followed by Bonferroni's post hoc test. P<0.05 was considered to indicate a statistically significant difference.
Results
miR-127-5p expression is downregulated during pneumonia
Once the in vitro model of pneumonia was established using LPS, as evidenced by increased inflammatory cytokines (data not shown), the expression of miR-127-5p in Ana-1 murine macrophages was detected by RT-qPCR. The results indicated that miR-127-5p expression was significantly decreased in Ana-1 murine macrophages following LPS exposure compared with the control group (Fig. 1); therefore, LPS-treated Ana-1 murine macrophages were used for subsequent experiments.
Figure 1.
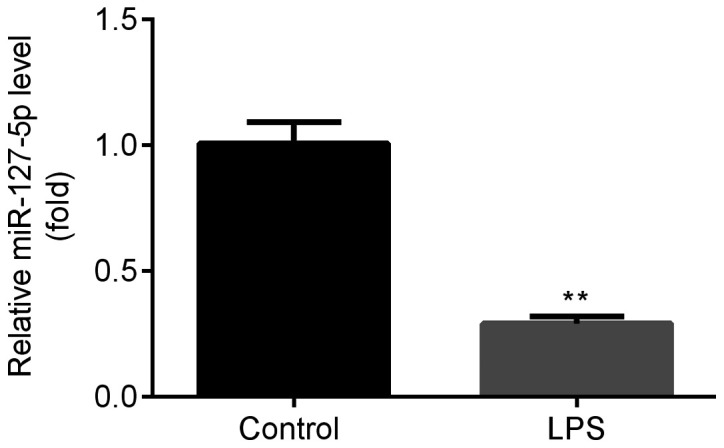
miR-127-5p expression is downregulated during pneumonia. Compared with the control group, miR-127-5p expression was significantly decreased in the LPS (0.1 µg/ml) group. **P<0.01 vs. the control group. miR, microRNA; LPS, lipopolysaccharide.
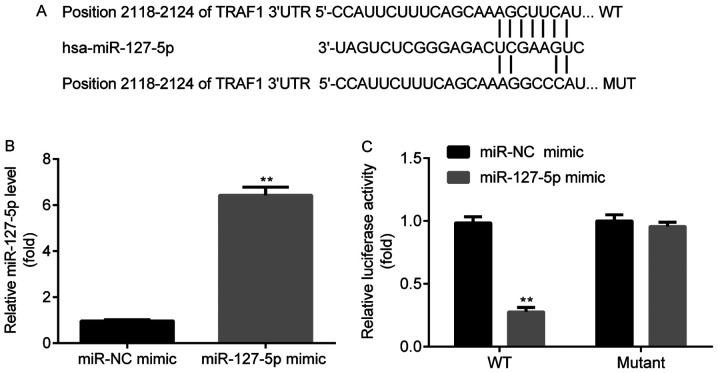
miR-127-5p targets the 3'-UTR of TRAF1
TargetScan indicated that miR-127-5p targeted TRAF1 at the 2,118-2,124 position of the 3'-UTR (Fig. 2A). To determine whether miR-127-5p mimic was successfully transfected into Ana-1 murine macrophages, the expression of miR-127-5p in the miR-NC mimic and miR-127-5p mimic groups was detected by RT-qPCR. miR-127-5p expression was significantly increased in the miR-127-5p mimic group compared with the miR-NC mimic group (Fig. 2B). A dual-luciferase reporter assay was subsequently performed to investigate the interaction between miR-127-5p and the 3'-UTR of TRAF1. Luciferase activity was significantly reduced in the TRAF1-WT-3'UTR + miR-127-5p mimic group compared with the TRAF1-WT-3'UTR + miR-NC mimic group. However, no significant difference in luciferase activity was observed between the TRAF1-MUT-3'UTR + miR-127-5p mimic and TRAF1-MUT-3'UTR + miR-NC mimic groups (Fig. 2C).
Figure 2.
miR-127-5p targets TRAF 3'-UTR. (A) miR-127-5p targets the 2,118-2,124 position of TRAF 3'-UTR. (B) miR-127-5p expression levels were significantly increased in the miR-127-5p mimic group compared with the miR-NC mimic group. (C) Luciferase activity was significantly decreased in the miR-126-5p mimic group compared with the miR-NC mimic group in Ana-1 murine macrophages transfected with TRAF1-WT-3'UTR. **P<0.01 vs. the miR-NC mimic group. 3'-UTR, 3'-untranslated region; miR, microRNA; NC, negative control; TRAF1, tumor necrosis factor receptor-associated factor 1; WT, wild-type; MUT, mutant.
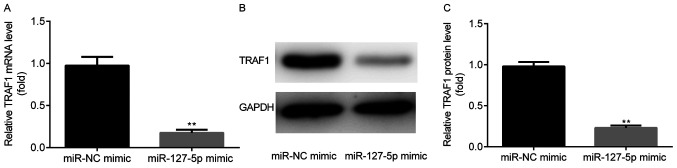
miR-127-5p inhibits TRAF1 expression
To determine whether miR-127-5p mimic could regulate the expression of TRAF1, the mRNA and protein expression levels of TRAF1 were detected using western blotting and RT-qPCR, respectively. The results suggested that the mRNA (Fig. 3A) and protein (Fig. 3B and C) expression levels of TRAF1 were significantly decreased in the miR-127-5p mimic group compared with the miR-NC mimic group.
Figure 3.
miR-127-5p inhibits TRAF1 expression. TRAF1 (A) mRNA and (B and C) protein expression levels were decreased in the miR-127-5p mimic group compared with the miR-NC mimic group. **P<0.01 vs. the miR-NC mimic group. miR, microRNA; NC, negative control; TRAF1, tumor necrosis factor receptor-associated factor 1.
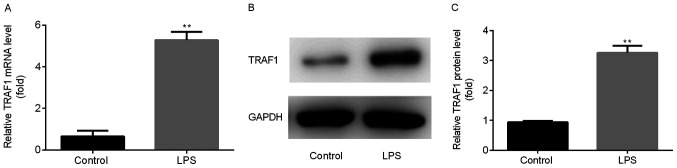
TRAF1 expression is upregulated during pneumonia
Following the establishment of the in vitro LPS-induced pneumonia model, the mRNA and protein expression levels of TRAF1 were detected in Ana-1 murine macrophages using RT-qPCR and western blotting, respectively. TRAF1 mRNA (Fig. 4A) and protein (Fig. 4B and C) expression levels were significantly increased in Ana-1 murine macrophages following LPS treatment compared with the control group.
Figure 4.
TRAF1 expression is upregulated during pneumonia. TRAF1 (A) mRNA and (B and C) protein expression levels were increased in the LPS (0.1 µg/ml) group compared with the control group. **P<0.01 vs. the control group. LPS, lipopolysaccharide; TRAF1, tumor necrosis factor receptor-associated factor 1..
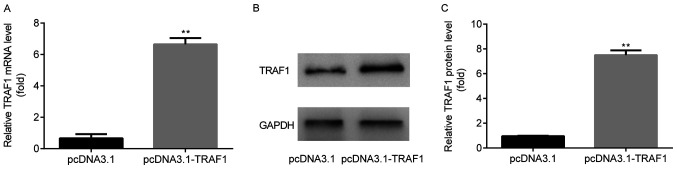
TRAF1 overexpression is induced by pcDNA3.1-TRAF1
TRAF1 mRNA (Fig. 5A) and protein (Fig. 5B and C) expression levels were significantly increased in the pcDNA3.1-TRAF1 group compared with the pcDNA3.1 group.
Figure 5.
TRAF1 overexpression is induced by pcDNA3.1-TRAF1. TRAF1 (A) mRNA and (B and C) protein expression levels were increased in the pcDNA3.1-TRAF1 group compared with the pcDNA3.1 group. **P<0.01 vs. the pcDNA3.1 group. TRAF1, TNF receptor-associated factor 1..
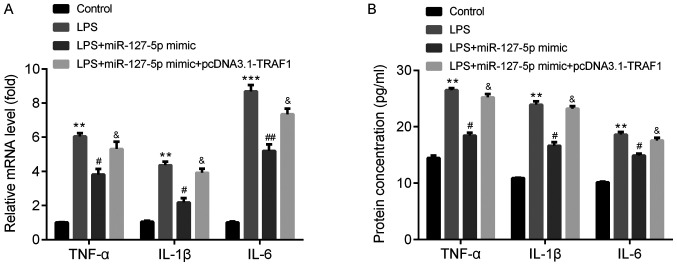
miR-127-5p mimic downregulates TNF-α, IL-1β and IL-6 levels by targeting TRAF1
To determine the function of miR-127-5p during pneumonia, the mRNA and protein expression levels of certain inflammatory cytokines, including TNF-α, IL-1β and IL-6, were detected using RT-qPCR and ELISA, respectively.
The mRNA expression levels of TNF-α, IL-1β and IL-6 were significantly increased in the LPS group compared with the control group. Furthermore, LPS-induced effects on inflammatory cytokine expression were significantly decreased following transfection with the miR-127-5p mimic; however, TRAF1 overexpression reversed miR-127-5p mimic-induced effects (Fig. 6A). The protein levels of TNF-α, IL-1β and IL-6 displayed a similar pattern to the mRNA levels in response to LPS, miR-127-5p mimic and pcDNA3.2-TRAF1 (Fig. 6B).
Figure 6.
miR-127-5p mimic downregulates TNF-α, IL-1β and IL-6 levels by targeting TRAF1. miR-127-5p mimic reversed LPS-induced TNF-α, IL-1β and IL-6 (A) mRNA expression and (B) protein levels, and TRAF1 overexpression inhibited miR-127-5p mimic-induced effects. **P<0.01, ***P<0.001 vs. the control group (miR-NC mimic + pcDNA3.1); #P<0.05 and ##P<0.01 vs. the miR-127-5p mimic group; &P<0.05 vs. the LPS + miR-127-5p mimic group. IL, interleukin; LPS, lipopolysaccharide; miR, microRNA; TNF, tumor necrosis factor; TRAF1, TNF receptor-associated factor 1.
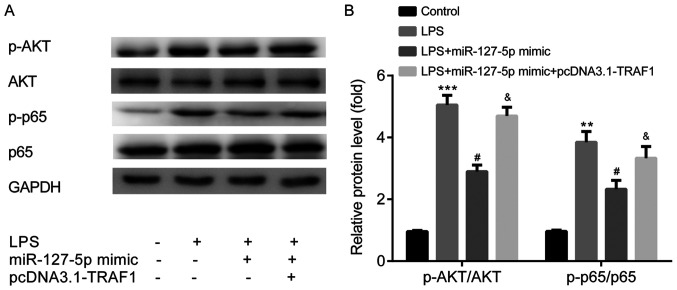
miR-127-5p mimic blocks the AKT/NF-κB signaling pathway by targeting TRAF1
To determine the effect of miR-127-5p on the signaling pathway involved in pneumonia, AKT phosphorylation and NF-κB activity were measured by western blotting. The ratios of p-AKT/AKT and p-p65/p65 expression levels were significantly increased in the LPS group compared with the control group. LPS-induced effects were significantly decreased by transfection with the miR-127-5p mimic; however, treatment with miR-127-5p mimic + pcDNA3.1-TRAF1 did not significantly alter LPS-induced effects (Fig. 7A and B).
Figure 7.
miR-127-5p mimic blocks the AKT/NF-κB signaling pathway by targeting TRAF1. Protein expression levels were (A) determined by western blotting, and (B) the ratios of p-AKT/AKT and p-65/p65 were semi-quantified. **P<0.01, ***P<0.001 vs. the control group (miR-NC mimic + pcDNA3.1); #P<0.05 vs. the miR-127-5p mimic group; &P<0.05 vs. the LPS + miR-127-5p mimic group. LPS, lipopolysaccharide; miR, microRNA; p, phosphorylated; TRAF1, tumor necrosis factor receptor-associated factor 1.
Discussion
Pneumonia is a persistent and pervasive disease (1); therefore, further investigation into the molecules that mediate severe pneumonia is required to identify novel therapeutics for the disease and reduce the disease burden.
miR-127 serves a role in numerous lung diseases. For example, miR-127 inhibits lung inflammation by targeting IgG Fcγ receptor I (14). Additionally, miR-127-5p, which is downregulated in bronchoalveolar lavage fluid, serves as a novel biomarker for the diagnosis of severe pneumonia (15). In the present study, lower miR-127-5p expression levels were observed in the LPS group compared with the control group, indicating the inhibitory effects of miR-127-5p during severe pneumonia. However, to the best of our knowledge, the molecules underlying this effect during severe pneumonia have not been previously reported.
The present study indicated that TRAF1 was targeted by miR-127-5p. TRAF1 is associated with a number of lung diseases, including asthma (18), non-small cell lung cancer (19) and lung inflammation (20). In the present study, higher TRAF1 expression levels were observed in the LPS group compared with the control group. In addition, TRAF1 was targeted by miR-127-5p and its expression was decreased by miR-127-5p mimic, indicating the enhancing effects of TRAF1 during severe pneumonia.
In in vitro monocyte models of pneumonia, LPS increased the levels of TNF-α, IL-1β and IL-6 (21,22). TNF is one of the most studied proinflammatory cytokines that displays numerous immunomodulatory activities (23). TNF is highly active in the lung and is therefore implicated in numerous pulmonary diseases, including asthma, chronic bronchitis and chronic obstructive pulmonary disease (24). Serum TNF-α and IL-6 levels have been associated with early death in patients with community-acquired pneumonia (25). In addition, IL-1β is a biomarker for the severity of community-acquired pneumonia in pediatric patients (26). Consistent with the aforementioned studies (21-26), the present study indicated that miR-127-5p mimic reversed LPS-induced upregulation of TNF-α, IL-1β and IL-6 levels, and TRAF1 overexpression inhibited miR-127-5p mimic-induced effects. Collectively, these results indicated that miR-127-5p inhibited LPS-induced inflammation by targeting TRAF1 during severe pneumonia.
NF-κB activation is a prerequisite for the production of a number of inflammatory cytokines, including TNF-α, IL-1β and IL-6(27), which leads to a more severe inflammatory reaction in stimulated macrophages (28). AKT is an upstream activator of the NF-κB signaling pathway (29). In the present study, miR-127-5p mimic reversed LPS-induced activation of p-AKT and p-p65, which was rescued by TRAF1 overexpression. Collectively, the results indicated that miR-127-5p inhibited LPS-induced AKT/NF-κB activation by targeting TRAF1 during severe pneumonia.
In conclusion, miR-127-5p may attenuate severe pneumonia, by reducing LPS-induced production of TNF-α, IL-1β and IL-6, and inactivating the AKT/NF-κB signaling pathway via TRAF1. Therefore, the present study suggested that TRAF1 may serve as a therapeutic target for severe pneumonia.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
CC, SL, LZ, JW, JC, RY, HL, JL, and ZX carried out the experimentations and data analyses. MC designed, conceived and supervised the research and prepared the manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Mizgerd JP. Respiratory infection and the impact of pulmonary immunity on lung health and disease. Am J Respir Crit Care Med. 2012;186:824–829. doi: 10.1164/rccm.201206-1063PP. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Jain S, Self WH, Wunderink RG, Fakhran S, Balk R, Bramley AM, Reed C, Grijalva CG, Anderson EJ, Courtney DM, et al. CDC EPIC Study Team: Community-Acquired Pneumonia Requiring Hospitalization among U.S Adults. N Engl J Med. 2015;373:415–427. doi: 10.1056/NEJMoa1500245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Jain S, Williams DJ, Arnold SR, Ampofo K, Bramley AM, Reed C, Stockmann C, Anderson EJ, Grijalva CG, Self WH, et al. CDC EPIC Study Team: Community-acquired pneumonia requiring hospitalization among U.S children. N Engl J Med. 2015;372:835–845. doi: 10.1056/NEJMoa1405870. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bartel DP. MicroRNAs: Genomics, biogenesis, mechanism, and function. Cell. 2004;116:281–297. doi: 10.1016/s0092-8674(04)00045-5. [DOI] [PubMed] [Google Scholar]
- 5.Ambros V. The functions of animal microRNAs. Nature. 2004;431:350–355. doi: 10.1038/nature02871. [DOI] [PubMed] [Google Scholar]
- 6.Farh KK, Grimson A, Jan C, Lewis BP, Johnston WK, Lim LP, Burge CB, Bartel DP. The widespread impact of mammalian MicroRNAs on mRNA repression and evolution. Science. 2005;310:1817–1821. doi: 10.1126/science.1121158. [DOI] [PubMed] [Google Scholar]
- 7.He L, Hannon GJ. MicroRNAs: Small RNAs with a big role in gene regulation. Nat Rev Genet. 2004;5:522–531. doi: 10.1038/nrg1379. [DOI] [PubMed] [Google Scholar]
- 8.He L, Thomson JM, Hemann MT, Hernando-Monge E, Mu D, Goodson S, Powers S, Cordon-Cardo C, Lowe SW, Hannon GJ, et al. A microRNA polycistron as a potential human oncogene. Nature. 2005;435:828–833. doi: 10.1038/nature03552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lecellier CH, Dunoyer P, Arar K, Lehmann-Che J, Eyquem S, Himber C, Saïb A, Voinnet O. A cellular microRNA mediates antiviral defense in human cells. Science. 2005;308:557–560. doi: 10.1126/science.1108784. [DOI] [PubMed] [Google Scholar]
- 10.Huang S, Feng C, Zhai YZ, Zhou X, Li B, Wang LL, Chen W, Lv FQ, Li TS. Identification of miRNA biomarkers of pneumonia using RNA-sequencing and bioinformatics analysis. Exp Ther Med. 2017;13:1235–1244. doi: 10.3892/etm.2017.4151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Li S, Cui W, Song Q, Zhou Y, Li J. miRNA-302e attenuates inflammation in infantile pneumonia though the RelA/BRD4/NF-κB signaling pathway. Int J Mol Med. 2019;44:47–56. doi: 10.3892/ijmm.2019.4194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Zhou Z, Zhu Y, Gao G, Zhang Y. Long noncoding RNA SNHG16 targets miR-146a-5p/CCL5 to regulate LPS-induced WI-38 cell apoptosis and inflammation in acute pneumonia. Life Sci. 2019;228:189–197. doi: 10.1016/j.lfs.2019.05.008. [DOI] [PubMed] [Google Scholar]
- 13.Zhang Y, Zhu Y, Gao G, Zhou Z. Knockdown XIST alleviates LPS-induced WI-38 cell apoptosis and inflammation injury via targeting miR-370-3p/TLR4 in acute pneumonia. Cell Biochem Funct. 2019;37:348–358. doi: 10.1002/cbf.3392. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Xie T, Liang J, Liu N, Wang Q, Li Y, Noble PW, Jiang D. MicroRNA-127 inhibits lung inflammation by targeting IgG Fcγ receptor I. J Immunol. 2012;188:2437–2444. doi: 10.4049/jimmunol.1101070. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Wang Z, Liu Q, Wang X, Wang Y, Zhang J, Zhou W, Yang X. Expressions of microRNA-127-5p in bronchoalveolar lavage fluid of patients with severe pneumonia and its diagnostic value. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue. 2017;29:592–595. doi: 10.3760/cma.j.issn.2095-4352.2017.07.004. (In Chinese) [DOI] [PubMed] [Google Scholar]
- 16.Chen Y, Luo G, Yuan J, Wang Y, Yang X, Wang X, Li G, Liu Z, Zhong N. Vitamin C mitigates oxidative stress and tumor necrosis factor-alpha in severe community-acquired pneumonia and LPS-induced macrophages. Mediators Inflamm. 2014;2014(426740) doi: 10.1155/2014/426740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Livak KJ, Schmittgen TD. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods. 2001;25:402–408. doi: 10.1006/meth.2001.1262. [DOI] [PubMed] [Google Scholar]
- 18.Oyoshi MK, Bryce P, Goya S, Pichavant M, Umetsu DT, Oettgen HC, Tsitsikov EN. TNF receptor-associated factor 1 expressed in resident lung cells is required for the development of allergic lung inflammation. J Immunol. 2008;180:1878–1885. doi: 10.4049/jimmunol.180.3.1878. [DOI] [PubMed] [Google Scholar]
- 19.Wen X, Wang B, Feng T, Yuan W, Zhou J, Fang T. TNF receptor-associated factor 1 as a biomarker for assessment of non-small cell lung cancer metastasis and overall survival. Clin Respir J. 2018;12:2197–2203. doi: 10.1111/crj.12789. [DOI] [PubMed] [Google Scholar]
- 20.Oyoshi MK, Barthel R, Tsitsikov EN. TRAF1 regulates recruitment of lymphocytes and, to a lesser extent, neutrophils, myeloid dendritic cells and monocytes to the lung airways following lipopolysaccharide inhalation. Immunology. 2007;120:303–314. doi: 10.1111/j.1365-2567.2006.02499.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Taganov KD, Boldin MP, Chang KJ, Baltimore D. NF-kappaB-dependent induction of microRNA miR-146, an inhibitor targeted to signaling proteins of innate immune responses. Proc Natl Acad Sci USA. 2006;103:12481–12486. doi: 10.1073/pnas.0605298103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liu Z, Yu H, Guo Q. MicroRNA-20a promotes inflammation via the nuclear factor-κB signaling pathway in pediatric pneumonia. Mol Med Rep. 2018;17:612–617. doi: 10.3892/mmr.2017.7899. [DOI] [PubMed] [Google Scholar]
- 23.Locksley RM, Killeen N, Lenardo MJ. The TNF and TNF receptor superfamilies: Integrating mammalian biology. Cell. 2001;104:487–501. doi: 10.1016/s0092-8674(01)00237-9. [DOI] [PubMed] [Google Scholar]
- 24.Mukhopadhyay S, Hoidal JR, Mukherjee TK. Role of TNFalpha in pulmonary pathophysiology. Respir Res. 2006;7(125) doi: 10.1186/1465-9921-7-125. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bacci MR, Leme RC, Zing NP, Murad N, Adami F, Hinnig PF, Feder D, Chagas AC, Fonseca FL. IL-6 and TNF-α serum levels are associated with early death in community-acquired pneumonia patients. Braz J Med Biol Res. 2015;48:427–432. doi: 10.1590/1414-431X20144402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Korkmaz MF, Güzel A, Açıkgöz M, Okuyucu A, Alaçam H. Reliability of Pro-adrenomedullin and Interleukin 1β in predicting severity of community-acquired pneumonia in pediatric patients. Ann Clin Lab Sci. 2018;48:81–89. [PubMed] [Google Scholar]
- 27.Li Y, Reddy MA, Miao F, Shanmugam N, Yee JK, Hawkins D, Ren B, Natarajan R. Role of the histone H3 lysine 4 methyltransferase, SET7/9, in the regulation of NF-kappaB-dependent inflammatory genes. Relevance to diabetes and inflammation. J Biol Chem. 2008;283:26771–26781. doi: 10.1074/jbc.M802800200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Liu SF, Malik AB. NF-κB activation as a pathological mechanism of septic shock and inflammation. Am J Physiol Lung Cell Mol Physiol. 2006;290:L622–L645. doi: 10.1152/ajplung.00477.2005. [DOI] [PubMed] [Google Scholar]
- 29.Venkatesan B, Valente AJ, Prabhu SD, Shanmugam P, Delafontaine P, Chandrasekar B. EMMPRIN activates multiple transcription factors in cardiomyocytes, and induces interleukin-18 expression via Rac1-dependent PI3K/Akt/IKK/NF-kappaB andMKK7/JNK/AP-1 signaling. J Mol Cell Cardiol. 2010;49:655–663. doi: 10.1016/j.yjmcc.2010.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.