Abstract
Purpose
Extended contact lens (CL) wear predisposes the wearer to Pseudomonas aeruginosa infection of the cornea, but the mechanism involved remains incompletely understood. The purpose of this study was to investigate the role of the stress hormone norepinephrine (NE) in the pathogenesis of CL-induced P. aeruginosa keratitis.
Methods
A total 195 adult C57BL/6 mice were used in this study. Corneal NE content was measured after 48 hours of sterile CL wear in mice. The effect of NE on P. aeruginosa adhesion and biofilm formation on the CL surface was examined in vitro. Moreover, mouse eyes were covered with P. aeruginosa-contaminated CLs, and either 500-µM NE was topically applied or the eyes were subconjunctivally injected with 100 µg of N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP-4) to deplete local NE. Clinical scores, neutrophil infiltration, proinflammatory cytokine levels, and bacterial load on the corneas and CLs were evaluated.
Results
Corneal NE content was elevated with extended CL wear in mice. In vitro, NE promoted the adhesion and biofilm formation of P. aeruginosa on the CL surface. In mice, topical application of NE aggravated P. aeruginosa infection, accompanied with increased clinical scores, neutrophil infiltration, proinflammatory cytokine expression, and bacterial burden on the corneas and CLs. However, pre-depletion of local NE with DSP-4 significantly alleviated the severity of P. aeruginosa keratitis.
Conclusions
Extended CL wear elevates corneal NE content, which promotes the pathogenesis of CL-induced P. aeruginosa keratitis in mice. Targeting NE may provide a potential strategy for the treatment of CL-related corneal infection caused by P. aeruginosa.
Keywords: contact lens, norepinephrine, Pseudomonas aeruginosa, corneal infection
Contact lenses (CLs) have become ubiquitous medical devices used for the correction of vision disorders in myopic patients; however, the incidence of microbial keratitis has increased due to the extended use of CLs and failure to comply with routine lens care.1–4 It has been reported that the risk of microbial keratitis increased 10- to 15-fold with extended CL wearing.5 The incidence has been estimated to vary from 1.2 to 25.4 per 10,000 contact lens wearers annually, varying with lens types and wearing time.6 Among the various CL-related microbial corneal infections, Pseudomonas aeruginosa is the leading cause; such infections develop very rapidly and may lead to corneal perforation or even vision loss.1,7–9
Catecholamines are a group of neurotransmitters that include epinephrine, norepinephrine (NE), and dopamine, which are released during stress response. In the eye, NE can be detected in the tears, aqueous humor, and cornea, although the origin remains incompletely known. It has been reported that NE secretion increases in hypoxic conditions of cultured pheochromocytoma cells,10 whereas hypoxia commonly occurs during prolonged CL wearing.3,11 More importantly, NE can stimulate the in vitro growth of several Gram-negative and Gram-positive bacteria, including P. aeruginosa.12–14 Mechanistically, studies have confirmed that quorum sensing may mediate bacterial responses to the stress hormone and affect bacterial virulence in the process of infection.15
Considering the risk of extended CL wearing and a potential relationship between NE and bacterial infection, we hypothesized that prolonged CL wear may elevate local NE secretion and subsequently increase susceptibility to P. aeruginosa infections in the cornea. This study aimed to explore the effects of extended CL wearing on corneal NE levels and the role of NE in the occurrence of CL-related P. aeruginosa keratitis.
Materials and Methods
Contact Lens Preparation
Hilafilcon B soft CLs (Bausch & Lomb, Rochester, NY, USA) were punched using a 3.5-mm-diameter trephine and trimmed using a microkeratome to fit mouse eyes. The prepared lenses were washed three times with sterile normal saline before use.
In Vitro Co-Culture of Bacteria and Contact Lenses
P. aeruginosa (ATCC 19660) was cultured in Luria–Bertani (LB) broth and diluted before use. P. aeruginosa adhesion to and biofilm formation on the CLs was evaluated as previously described.16,17 Briefly, the CL was inoculated with 10 CFU P. aeruginosa and incubated with 500-µM NE (Grand Pharma, Wuhan, China) or PBS in serum-containing SAPI media for 48 hours at 37°C. Subsequently, each contact lens (n = 6 per group) was collected to evaluate the number of viable bacteria and biofilm formation.
Mouse Model of Contact Lens-Induced P. aeruginosa Keratitis
The 195 adult C57BL/6 mice (Vital River Laboratory Animal Technology Co., Ltd, Beijing, China) used in this study were maintained in the animal facility of the Shandong Eye Institute. All animal experiments were approved by the ethics committee of the Shandong Eye Institute and carried out in accordance with the guidelines of the ARVO Statement for the Use of Laboratory Animals in Ophthalmic and Vision Research. Each experiment was repeated three times. Only the right eye of each mouse was used.
For the mouse model of CL wearing, two sterile lenses were placed onto the mouse eye in a stacked fashion. The eyelid was then sutured in a half-open position to prevent the CL from falling out and to keep the eye half open.18 The sham control mice received only the half-eyelid suture and no CL, and normal mice without a suture or CL served as control group (n = 5 per group). After 48 hours, the mouse corneas were harvested for the detection of NE.
For the mouse model of CL-related corneal infection, the induction method was modified as previously described.19 In brief, 106 CFU/ml of bacteria were inoculated onto the CL surface and incubated for 24 hours at 37°C.19 Each mouse cornea was blotted 20 times with single-ply tissue paper (Kimwipes; Kimberly-Clark, Irving, TX, USA) before a contaminated CL was applied. Exogenous NE (500 µM) or PBS was topically applied six times within 24 hours of the CL placement (n = 15 per group), or each cornea was subconjunctivally injected with selective neurotoxin N-(2-chloroethyl)-N-ethyl-2-bromobenzylamine (DSP-4, 20 mg/ml; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) to deplete local NE or normal saline as vehicle control at 12 hours before the CL placement (n = 10 per group).20 After 48 hours, six worn CLs were randomly collected for each group to evaluate the number of viable bacteria (n = 3 per group) and biofilm formation (n = 3 per group). Mouse eyes were observed by slit-lamp microscopy to grade disease severity according to the established scale.21
Quantification of Bacterial Load on Mouse Corneas and Contact Lenses
In vitro cultured CLs, mouse infected corneas, and worn CLs were harvested, washed, and homogenized in 500 µl normal sterile saline. After homogenization, the samples were replated onto LB agar in triplicate after serial 1:10 dilution. The viable bacteria were counted after overnight incubation at 37°C.22
Evaluation of Biofilm Formation on Contact Lenses
CLs cultured in vitro or worn by the mice were collected, washed, and fixed in 2.5% glutaraldehyde for 1 hour. The samples were stained with 50 mg/mL fluorescein isothiocyanate-conjugated concanavalin A (FITC-ConA) for 20 minutes and 5-µg/mL propidium iodide (PI; Sigma-Aldrich, St. Louis, MO, USA) for 15 minutes, as previously described, with a slight modification.23 The biofilm formation was observed using a confocal laser scanning microscope (Carl Zeiss Meditec, Oberkochen, Germany). FITC-ConA bound to the polysaccharide of the biofilm to emit green fluorescence, and the PI bound to the DNA of dead bacteria to emit red fluorescence. Finally, three-dimensional images were reconstructed with Imaris software (Bitplane, South Windsor, CT, USA).
Immunofluorescence Staining
Mouse eyes were embedded in Tissue-Tek O.C.T. compound (Sakura Finetek, Alphen aan den Rijn, the Netherlands) and cut into sections 7 µm thick. The samples were stained with the rat anti-mouse Ly6G antibody (Abcam, Cambridge, UK) overnight at 4°C and then subsequently incubated in FITC-conjugated goat anti-rat IgG (Invitrogen, Thermo Fisher Scientific, Waltham, MA, USA) for 2 hours, as previously described.24 All slides were examined using fluorescence microscopy after counterstaining with 4′,6-diamidino-2-phenylindole (Vector Laboratories, Burlingame, CA, USA).
ELISA
Mouse corneas were homogenized in 100 µL of PBS with a TissueLyser (QIAGEN, Hilden, Germany). All samples were centrifuged, and an aliquot of each homogenized supernatant was assayed in triplicate following the manufacturer's instructions, including NE (Cloud Clone, Wuhan, China); myeloperoxidase (MPO), IL-1β, and IL-6 (Meilian, Shanghai, China); and TNF-α (Proteintech, Wuhan, China).
Statistical Analysis
Statistical analysis was performed using Student's t-test and one-way ANOVA with GraphPad Prism 5.0 (GraphPad Software, La Jolla, CA, USA). All of the data in this study were obtained from at least three independent experiments and are expressed as mean ± standard error of the mean (SEM). P < 0.05 was considered statistically significant.
Results
Extended Contact Lens Wear Elevated Corneal Norepinephrine Content
The mouse corneas were collected after 48 hours of sterile CL wearing for NE measurement. Five corneas were pooled for each sample. As shown in Figure 1, NE content in mouse corneas with CLs was increased by 2.2-fold when compared with either the normal control mice (P < 0.001) or the sham control mice with only half-eyelid sutures (P < 0.001). The NE content showed no significant change in the mice with only half-eyelid sutures when compared with the normal control mice (P = 0.465). The results suggest that corneal NE content was elevated with prolonged wearing of CLs, but not with the half-eyelid sutures only.
Figure 1.
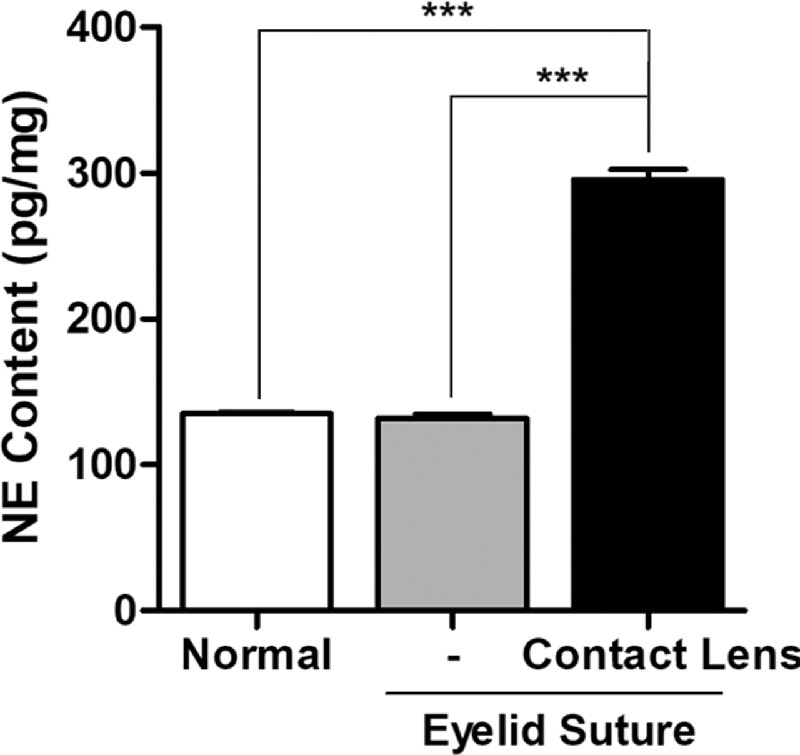
Extended contact lens wearing caused elevated levels of corneal norepinephrine. Shown is the measurement of corneal NE with ELISA after 48 hours of half-eyelid suture with or without contact lens wear in mice (***P < 0.001, n = 5).
Norepinephrine Enhanced P. aeruginosa Adhesion and Biofilm Formation on the Contact Lens Surface In Vitro
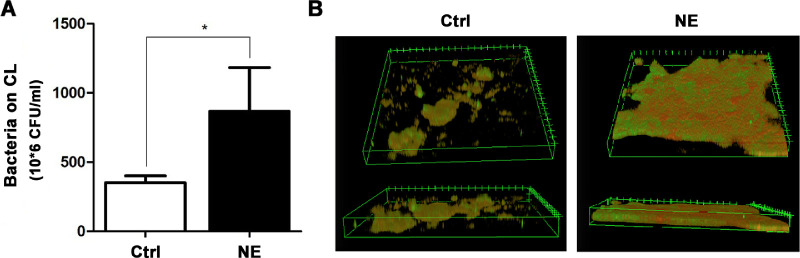
To determine the direct effects of NE on bacterial adhesion and biofilm formation, P. aeruginosa was inoculated in vitro onto the surface of CLs, which were treated for 48 hours with either 500-µM NE or PBS as a vehicle control. The adhered bacteria were recovered and quantified using plating and colony counting. The biofilm formation was monitored by confocal microscopy after treatment with FITC-ConA or PI. Three CLs were pooled for each sample. The results showed that NE increased the number of adhered bacteria on the CLs nearly twofold when compared with the PBS control (Fig. 2A). Moreover, the density and architecture of the formed biofilms were markedly enhanced, with extensive coverage of extracellular polysaccharide across the surface of the clumped bacteria (Fig. 2B).
Figure 2.
Norepinephrine promoted P. aeruginosa adhesion and biofilm formation on contact lenses. P. aeruginosa was inoculated on the contact lenses and treated with 500-µM NE or PBS as vehicle control for 48 hours. (A) Quantification of the adhered viable bacteria with direct plating and colony counting (*P = 0.048, n = 3). (B) Representative images of biofilm formation on the contact lens after the staining with FITC-ConA (green) and propidium iodide (red).
Norepinephrine Increased the Severity of Keratitis Induced by Contact Lenses Contaminated with P. aeruginosa
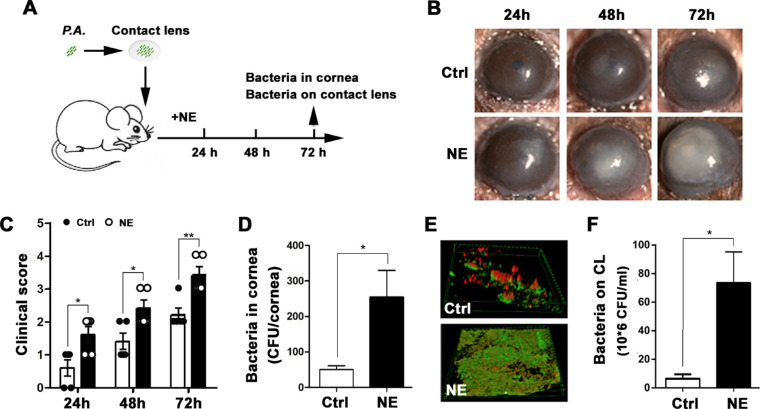
Mouse eyes were covered with CLs contaminated with P. aeruginosa as shown in Figure 3A. The mice were treated with topically applied 500-µM NE or PBS six times within 24 hours of CL placement. Slit-lamp microscopic examination showed obvious corneal opacification after 24 hours which spread to the entire cornea at 72 hours in the mice who received topical NE (Fig. 3B). In contrast, the control mice exhibited a slower progression, as indicated by the clinical scores (Fig. 3C). The bacterial burden of infected corneas after 48 hours showed a greater than fivefold increase in the NE-treated mice compared with the control mice (Fig. 3D). Moreover, the biofilm on CLs removed from NE-treated eyes covered a larger area and had fewer dead bacteria (red fluorescence with PI staining) compared with the control group (Fig. 3E), a finding that was further confirmed by direct plating and colony counting of the recovered bacteria (Fig. 3F). This suggests that NE promotes the progression of CL-induced P. aeruginosa keratitis and enhances its severity.
Figure 3.
Norepinephrine accelerated P. aeruginosa infection in the cornea during contact lens wear. (A) Schematic diagram of P. aeruginosa infection by the contaminated contact lens. Briefly, after preincubation with 103 CFU/ml P. aeruginosa for 24 hours, the contact lens was placed onto a mouse eye followed by the topical application of NE or PBS as vehicle control. (B) Representative images of slit-lamp microscopy and (C) clinical scores after 24, 48, and 72 hours of contact lens wear (*P = 0.020, 0.020, and 0.005, respectively; n = 5). (D) Quantification of corneal bacterial burden with direct plating and colony counting (*P = 0.019, n = 5). (E) Representative images of biofilm formation on the contact lens after the staining with FITC-ConA (green) and propidium iodide (red). (F) Quantification of the adhered viable bacteria on the contact lens with direct plating and colony counting (*P = 0.036, n = 3).
Norepinephrine Aggravated Corneal Inflammatory Responses
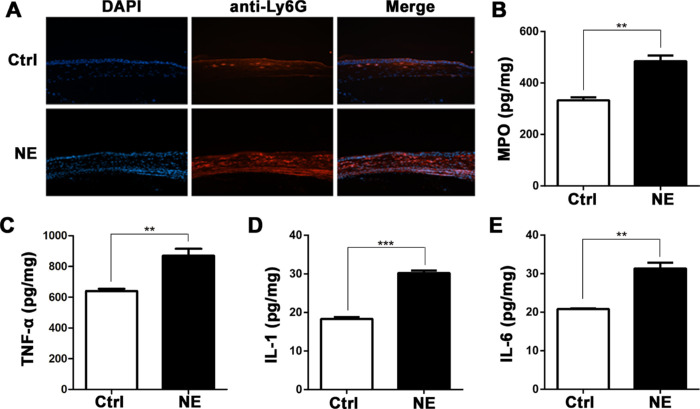
Neutrophil infiltration and proinflammatory factor expression were measured at 24 hours after CL placement and NE application. Immunofluorescence staining with anti-Ly6G antibody disclosed that neutrophils infiltrated into the full thickness of the corneal stroma, compared with anterior stromal infiltration in the vehicle control corneas (Fig. 4A). The enhanced neutrophil infiltration was accompanied by increased MPO activity (Fig. 4B) and levels of proinflammatory factors such as TNF-α, IL-1β, and IL-6 (Figs. 4C–4E) in the NE-treated mouse corneas.
Figure 4.
Norepinephrine aggravated corneal inflammation of P. aeruginosa-induced keratitis. Mouse corneas were collected after 24 hours of contact lens wear with topical NE application. (A) Representative immunofluorescence staining with anti-Ly6G antibody. (B–E) Quantification of MPO content and TNF-α, IL-1β, and IL-6 protein levels (**P < 0.005, ***P < 0.001; n = 5).
Local Norepinephrine Depletion Alleviated the Severity of P. aeruginosa Keratitis
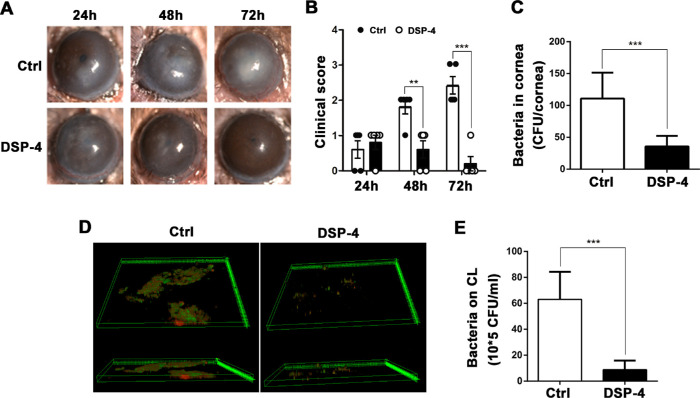
Based on the observed effect of NE on P. aeruginosa keratitis, we further investigated whether or not local NE depletion could alleviate the disease severity in mice. The mice were subconjunctivally injected with the selective neurotoxin DSP-4 or normal saline before placement of contaminated CLs. After 12 hours of DSP-4 pretreatment, the corneal NE content decreased about 38% as compared with the control mice (108 pg/mg vs. 165 pg/mg). Slit-lamp microscopy examination showed aggravated corneal opacification in the control mice, whereas mouse corneas pretreated with DSP-4 showed gradual recovery of transparency within 72 hours after CL placement (Fig. 5A). Similarly, the clinical scores confirmed that DSP-4 pretreatment significantly alleviated the severity from 48 to 72 hours after CL placement (Fig. 5B). Moreover, the corneal bacterial burden showed a 70% reduction in the DSP-4-treated mice as compared with the controls after 48 hours (Fig. 5C). The biofilms on the CLs removed from the DSP-4-treated eyes exhibited smaller coverage areas and fewer viable bacteria compared with the control group (Fig. 5D), as confirmed by direct plating and colony counting of recovered bacteria (Fig. 5E). These findings indicated that local depletion of NE with DSP-4 alleviated the severity of CL-induced P. aeruginosa infection in the cornea.
Figure 5.
Local norepinephrine depletion reduced the severity of P. aeruginosa keratitis. Mice were injected subconjunctivally with DSP-4 12 hours before contaminated contact lens wear. (A) Representative images of slit-lamp microscopy, and (B) clinical scores after 24, 48, and 72 hours of contact lens wear. (C) Quantification of corneal bacterial burden with direct plating and colony counting (***P < 0.001, n = 5). (D) Representative images of biofilm formation on the contact lens. (E) Quantification of the adhered viable bacteria on the contact lens with direct plating and colony counting (***P < 0.001, n = 3).
Discussion
CL-induced microbial corneal infections, especially P. aeruginosa keratitis, remain a challenge in clinical practice. The exact mechanism of host response to susceptibility to P. aeruginosa infection is still unclear. In the present study, we confirmed significant elevation of corneal NE content in mouse eyes after wearing CLs for as long as 48 hours. In turn, elevated NE promoted bacterial growth and biofilm formation on the CLs in vitro. More importantly, contaminated CLs aggravated the severity of P. aeruginosa infection in mice. Conversely, local depletion of NE alleviated and even reversed the progression and severity of keratitis. These findings reveal a novel mechanism of CL-induced P. aeruginosa keratitis, which could provide a potential target for the clinical prevention and management of such infections.
Overnight use of CLs has been reported to be the predominant risk factor for microbial keratitis.2,25 Previous studies focused on the impact of hypoxia and hypercapnia on conjunctival flora or homeostasis of the ocular surface. In this study, we found that corneal NE content was elevated over twofold after 48 hours of CL wear, which may be caused by either hypoxia, similar to the response of pheochromocytoma cells under hypoxic conditions,10 or by microlesions of the ocular surface, which are commonly detected in patients with overnight wearing of CLs.
The pathogenesis of P. aeruginosa keratitis relies on the microbe–host interactions, where P. aeruginosa can sense the host response signal, thereby enhancing virulence and affecting the occurrence, process, and outcome of infections.26–28 Among various signals, the stress hormone NE could facilitate the growth, virulence, motility, and attachment of P. aeruginosa through the las quorum-sensing pathway.15 Our study confirmed that exogenous NE aggravated the severity of CL-related P. aeruginosa keratitis, and local depletion of NE with DSP-4 alleviated the phenotype in mice. Furthermore, the direct effects of NE on P. aeruginosa growth and biofilm formation on the CLs were confirmed through in vitro experiments, which indicated that commensal P. aeruginosa in the conjunctiva could be activated through the elevated NE level caused by the extended wearing of CLs, contributing to the onset of microbial keratitis. The elevation of host NE content may serve as an important link between extended CL wearing and P. aeruginosa susceptibility.
In our study, however, the phenomenon of extended CL wear promoting corneal NE secretion and P. aeruginosa infection was only demonstrated in the corneas of mice. NE concentration has also been studied in corneas of other species. The concentration of NE in human corneas has been measured as 9.78 ± 3.75 pmol/g in the epithelium, 2.95 ± 1.02 pmol/g in the stroma, and 15.22 ± 4.99 pmol/g in the endothelium, whereas in rabbit corneas the measurements were 238.96 ± 54.59 pmol/g, 48.17 ± 7.80 pmol/g, and 525.36 ± 71.57 pmol/g, respectively.29 There is still a way to go before being able to apply the current findings to actual clinical practice. Tears are the only accessible sample from humans after extended wearing of CLs, but the inevitable increase of tear secretion during collection will dilute the concentration of NE. Therefore, further study should be performed on the clinical relevance of corneal NE content and microbial infection.
In summary, our results showed that opportunistic pathogenic P. aeruginosa utilized the host hormone NE to trigger the onset of keratitis in a mouse model with extended CL wearing. Targeting the elevated NE may be a potential strategy for preventing CL-induced P. aeruginosa infection in the cornea.
Acknowledgments
The authors thank Bining Zhang and Ping Lin for their helpful revision of this manuscript.
Supported by grants from the National Natural Science Foundation of China (81770904 and 81900831), Academic Promotion Program and Innovation Project of Shandong First Medical University (2019ZL001 and 2019RC008), and Qingdao Science and Technology Project (19-6-1-24-nsh).
Disclosure: J. Li, None; X. Ma, None; L. Zhao, None; Y. Li, None; Q. Zhou, None; X. Du, None
References
- 1. Jin H, Parker WT, Law NW, et al.. Evolving risk factors and antibiotic sensitivity patterns for microbial keratitis at a large county hospital. Br J Ophthalmol. 2017; 101: 1483–1487. [DOI] [PubMed] [Google Scholar]
- 2. Cope JR, Collier, A, Nethercut H, Jones JM, Yates K, Yoder JS. Risk behaviors for contact lens-related eye infections among adults and adolescents – United States, 2016. MMWR Morb Mortal Wkly Rep. 2017; 66: 841–845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Lin MC, Polse KA. Hypoxia, overnight wear, and tear stagnation effects on the corneal epithelium: data and proposed model. Eye Contact Lens. 2007; 33: 378–381. [DOI] [PubMed] [Google Scholar]
- 4. Stapleton F, Edwards K, Keay L, et al.. Risk factors for moderate and severe microbial keratitis in daily wear contact lens users. Ophthalmology. 2012; 119: 1516–1521. [DOI] [PubMed] [Google Scholar]
- 5. Schein OD, Poggio EC. Ulcerative keratitis in contact lens wearers incidence and risk factors. Cornea. 1990; 9 (suppl 1): S55–S58. [DOI] [PubMed] [Google Scholar]
- 6. Stapleton F, Keay L, Edwards K, et al.. The incidence of contact lens-related microbial keratitis in Australia. Ophthalmology. 2008; 115: 1655–1662. [DOI] [PubMed] [Google Scholar]
- 7. Fleiszig SMJ, Kroken AR, Nieto V, et al.. Contact lens-related corneal infection: intrinsic resistance and its compromise [published online ahead of print November 20, 2019]. Prog Retin Eye Res. 10.1016/j.preteyeres.2019.100804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Kolar SSN, Luca V, Baidouri H, Mannino G, McDermott AM, Mangoni ML. Esculentin-1a (1-21) NH2: a frog skin-derived peptide for microbial keratitis. Cell Mol Life Sci. 2015; 72: 617–627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Ibrahim YW, Boase DL, Cree IA. Epidemiologic characteristics, predisposing factors and microbiological profile of infectious corneal ulcers: the Portsmouth corneal ulcer study. Br J Ophthalmol. 2009; 93: 1319–1324. [DOI] [PubMed] [Google Scholar]
- 10. Bechmann N, Poser I, Seifert V, et al.. Impact of extrinsic and intrinsic hypoxia on catecholamine biosynthesis in absence or presence of HIF2α in pheochromocytoma cells. Cancers (Basel). 2019; 11: 594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Lim CHL, Stapleton F, Mehta JS. Review of contact lens-related complications. Eye Contact Lens. 2018; 44 (suppl 2): S1–S10. [DOI] [PubMed] [Google Scholar]
- 12. Sarkodie EK, Zhou S, Baidoo SA, Chu W. Influences of stress hormones on microbial infections. Microb Pathog. 2019; 131: 270–276. [DOI] [PubMed] [Google Scholar]
- 13. Sandrini SM, Shergill R, Woodward J, et al.. Elucidation of the mechanism by which catecholamine stress hormones liberate iron from the innate immune defense proteins transferrin and lactoferrin. J Bacteriol. 2010; 192: 587–594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Freestone PP, Hirst RA, Sandrini SM, et al.. Pseudomonas aeruginosa-catecholamine inotrope interactions: a contributory factor in the development of ventilator associated pneumonia? Chest. 2012; 142: 1200–1210. [DOI] [PubMed] [Google Scholar]
- 15. Hegde M, Wood TK, Jayaraman A. The neuroendocrine hormone norepinephrine increases Pseudomonas aeruginosa PA14 virulence through the las quorum-sensing pathway. Appl Microbiol Biotechnol. 2009; 84: 763–776. [DOI] [PubMed] [Google Scholar]
- 16. Lyte M, Ernst S. Catecholamine induced growth of gram negative bacteria. Life Sci. 1992; 50: 203–212. [DOI] [PubMed] [Google Scholar]
- 17. Butrus SI, Klotz SA, Misra RP. The adherence of pseudomonas aeruginosa to soft contact lenses. Ophthalmology. 1987; 94: 1310–1314. [DOI] [PubMed] [Google Scholar]
- 18. Chen P, Yin H, Wang Y, Wang Y, Xie L. Inhibition of VEGF expression and corneal neovascularization by shRNA targeting HIF-1α in a mouse model of closed eye contact lens wear. Mol Vis. 2012; 18: 864–873. [PMC free article] [PubMed] [Google Scholar]
- 19. Tam C, Mun JJ, Evans DJ, Fleiszig SM. The impact of inoculation parameters on the pathogenesis of contact lens-related infectious keratitis. Invest Ophthalmol Vis Sci. 2010; 51: 3100–3106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Scullion GA, Kendall DA, Sunter D, Marsden CA, Pardon MC. Central noradrenergic depletion by DSP-4 prevents stress-induced memory impairments in the object recognition task. Neuroscience. 2009; 164: 415–423. [DOI] [PubMed] [Google Scholar]
- 21. Hazlett LD, Moon MM, Strejc M, Berk RS. Evidence for N-acetylmannosamine as an ocular receptor for P. aeruginosa adherence to scarified cornea. Invest Ophthalmol Vis Sci. 1987; 28: 1978–1985. [PubMed] [Google Scholar]
- 22. Robertson DM, Parks QM, Young RL, et al.. Disruption of contact lens-associated Pseudomonas aeruginosa biofilms formed in the presence of neutrophils. Invest Ophthalmol Vis Sci. 2011; 52: 2844–2850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Pakkulnan R, Anutrakunchai C, Kanthawong S, Taweechaisupapong S, Chareonsudjai P, Chareonsudjai S. Extracellular DNA facilitates bacterial adhesion during Burkholderia pseudomallei biofilm formation. PLoS One. 2019; 14: e0213288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Xiao T, Tongsong W, Songmei Z, et al.. PEDF reduces the severity of herpetic simplex keratitis in mice. Invest Ophthalmol Vis Sci. 2018; 59: 2923–2931. [DOI] [PubMed] [Google Scholar]
- 25. Landers JA, Crompton JL. Microbial keratitis associated with overnight wear of silicone hydrogel contact lenses. Med J Aust. 2006; 185: 177–178. [DOI] [PubMed] [Google Scholar]
- 26. Wu L. Recognition of host immune activation by Pseudomonas aeruginosa. Science. 2005; 309: 774–777. [DOI] [PubMed] [Google Scholar]
- 27. Babrowski T, Romanowski K, Fink D, et al.. The intestinal environment of surgical injury transforms Pseudomonas aeruginosa into a discrete hypervirulent morphotype capable of causing lethal peritonitis. Surgery. 2013; 153: 36–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Thanabalasuriar A, Scott BNV, Peiseler M, et al.. Neutrophil extracellular traps confine Pseudomonas aeruginosa ocular biofilms and restrict brain invasion. Cell Host Microbe. 2019; 25: 526–536.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Luis F, Catarina J, Fatima F, et al.. Regulation of corneal noradrenaline release and topography of sympathetic innervation: functional implications for adrenergic mechanisms in the human cornea. Exp Eye Res. 2018; 174: 121–132. [DOI] [PubMed] [Google Scholar]