Abstract
Podocyte injury serves an important role during the progression of diabetic nephropathy (DN), and lycopene (Lyc) may display a potential protective effect against DN progression. The effects of Lyc on high glucose (HG)-induced podocyte apoptosis and the underlying mechanisms are not completely understood; therefore, the present study aimed to investigate the effects of Lyc on HG-induced MPC5 podocyte apoptosis and the underlying mechanism. In the present study, MPC5 podocytes were exposed to HG and different doses of Lyc. MPC5 podocyte viability and apoptosis were assessed by performing the MTT assay and flow cytometry, respectively. To explore the effects of Lyc on the PI3K/AKT signaling pathway and autophagy, LY294002 (LY) and 3-methyladenine (3-MA) were used as PI3K and autophagy inhibitors, respectively. The expression levels of nephrin, podocin, apoptosis-related proteins (Bax, Bcl-2 and cleaved caspase-3), autophagy-related proteins [Beclin-1 and microtubule associated protein 1 light chain 3 (LC3)II/LC3I] and certain key proteins involved in the PI3K/AKT signaling pathway were measured via western blotting. The results suggested that Lyc reversed the inhibitory effect of HG on cell viability, and the protein expression levels of nephrin and podocin, as well as the promoting effect of HG on MPC5 podocyte apoptosis. In addition, under HG conditions, Lyc upregulated the phosphorylation levels of PI3K and AKT, and reduced HG- and LY-mediated MPC5 podocyte apoptosis. Moreover, Lyc further increased HG-induced protein expression levels of Beclin-1 and LC3II/LC3I, and attenuated LY-mediated inhibition of HG-induced MPC5 podocyte autophagy. In addition, the effects of Lyc on HG-mediated MPC5 podocyte apoptosis were alleviated by 3-MA. Therefore, the present study suggested that Lyc may protect against HG-induced MPC5 podocyte apoptosis by promoting autophagy activity via activation of the PI3K/AKT signaling pathway.
Keywords: MPC5 podocytes, apoptosis, autophagy, lycopene, PI3K/AKT signaling pathway
Introduction
Long-term hyperglycemia, which causes diabetic nephropathy (DN), is a frequent and principal cause of death and morbidity among patients with diabetes (1). DN is one of the most common diabetic complications that often occurs in patients with type 1 and 2 diabetes (2), and is also major risk factor for chronic kidney diseases (3). Moreover, patients with DN are highly likely to develop end-stage renal disease, which ultimately requires renal replacement (4); therefore, the current treatments, such as intensive blood pressure regulation focused on blockage of the renin-angiotensin system, and the regulation of hyperglycaemia, dyslipidaemia and albuminuria, are not effective (5).
Previous studies have demonstrated that factors, such as mesangial extracellular matrix deposition, thickening of the basement membrane, glomerular pericytes, and decreased numbers of podocytes and microvascular alterations, are closely related to renal dysfunction in DN (6-9). Terminally differentiated podocytes are specialized glomerular visceral endothelial cells that exert critical effects on the formation of the glomerular filtration barrier and inhibition of urinary protein loss (10). Damages to the glomerular filtration barrier increase the glomerular filtration rate and albuminuria (11). Podocytes damage contribute to proteinuria and the development of glomerulosclerosis (11). Collectively, the aforementioned findings suggested that protecting against podocyte injury may serve as a potential therapeutic strategy for DN progression. In addition, podocyte injury includes loss of podocytes in the glomerulus (12), which is attributed to hyperglycemia-induced apoptosis, and directly leads to proteinuria and glomerular sclerosis (13). Therefore, the potential therapeutic application of mitigating podocyte apoptosis for the progression of DN requires further investigation.
Lycopene (Lyc), a nutraceutical, is a natural pigment that is rich in tomatoes and other plants (14). Lyc not only displays remarkable active oxygen scavenging abilities and antioxidation activities against quenching singlet oxygen and lipid peroxidation, but also displays anticancer, anti-inflammatory and antiapoptotic effects (15-17). Moreover, Li et al (18) demonstrated that Lyc can improve DN progression in diabetic model rats, and Ni et al (19) reported that Lyc can enhance autophagy and attenuate apoptosis to protect against high glucose (HG)-induced podocyte injury.
Although the effects of Lyc on podocyte injury and apoptosis have attracted increasing attention, the exact mechanism underlying how Lyc exerts its protective effect on HG-induced podocyte apoptosis is not completely understood. Therefore, the present study explored the protective effect of Lyc on HG-induced MPC5 podocyte apoptosis and the underlying mechanism.
Materials and method
Cell culture
Conditionally immortalized mouse podocytes (MPC5) were purchased from American Tissue Culture Collection. MPC5 podocytes were cultured and induced for cell proliferation and differentiation as previously described (20). MPC5 podocytes were cultured in RPMI 1640 medium (Beijing Solarbio Science & Technology Co., Ltd.) containing 10% FBS (Sigma-Aldrich; Merck KGaA) and 10 U/ml recombinant mouse interferon-γ (IFNγ; Peprotech, Inc.) at 33˚C with 5% CO2 and 95% relative humidity.
To stimulate cell differentiation, MPC5 podocytes were subcultured in RPMI-1640 containing 10% FBS without mouse IFNγ for 10-14 days at 37˚C with 5% CO2 to reach 80-90% confluence. Prepared MPC5 podocytes were used for subsequent experiments.
Cell viability assay
The viability of differentiated MPC5 podocytes was determined using an MTT assay (Sigma-Aldrich; Merck KGaA), according to the manufacturer's protocol. MPC5 podocytes were seeded (1x104 cells/well) into 96-well plates and incubated with RPMI-1640 supplemented with 10% FBS for 24 h at 37˚C. As previously described (21), MPC5 podocytes were divided into seven groups: i) Normal group (NG; 5.5 mM glucose); ii) hypertonic group [HP; 5.5 mM glucose and 19.5 mM mannitol (Sigma-Aldrich; Merck KGaA)]; iii) HG (25 mM glucose); iv) HG and low-concentration Lyc treatment group [HG + L-Lyc; 25 mM glucose + 3.125 mM Lyc (Sigma-Aldrich; Merck KGaA)]; v) HG and high-concentration Lyc treatment group (HG + H-Lyc; 25 mM glucose + 12.5 mM Lyc); vi) low-concentration Lyc treatment group (L-Lyc; 3.125 mM Lyc); and vii) high-concentration Lyc treatment group (H-Lyc; 12.5 mM Lyc). All groups were treated at 37˚C for 48 h. Subsequently, 20 µl MTT solution (5 mM) was added to each well and incubated for another 4 h at 37˚C. The absorbance of each well was measured at a wavelength of 570 nm using a microplate reader. The cell viability in individual groups of cells was calculated as the optical density (OD) value of experiments/the OD values of control cells.
Western blotting
MPC5 podocyte protein expression was assessed via western blotting as previously described (11). Briefly, MPC5 podocytes were washed twice with PBS. Subsequently, total protein was extracted from MPC5 podocytes using RIPA buffer (Thermo Fisher Scientific, Inc.) with a complete protease inhibitor cocktail (Roche Diagnostics GmbH) on ice for 30 min. Subsequently, the supernatants were collected by centrifugation at 12,000 x g for 10 min at 4˚C and total protein was quantified using the BCA protein assay kit (Beijing Solarbio Science & Technology Co., Ltd.).
Subsequently, protein (30 µg/lane) was separated via 12% SDS-PAGE and transferred onto PVDF membranes (EMD Millipore), which were then blocked with 5% non-fat milk in TBST (0.1% Tween-20) for 1 h at room temperature. The membranes were incubated at 4˚C overnight with the following primary antibodies: Anti-nephrin (rabbit; 1:400; cat. no. ab58968; Abcam), anti-podocin (rabbit; 1:1,300; cat. no. ab50339; Abcam), anti-Bcl-2 (rabbit; 1:1,000; cat. no. ab59348; Abcam), anti-Bax (rabbit; 1:1,000; cat. no. ab32503; Abcam), anti-cleaved (C) caspase-3 (rabbit; 1:500; cat. no. ab2302; Abcam), anti-phosphorylated (p)-PI3K (rabbit; 1:1,000; cat. no. 4228; Cell Signaling Technology, Inc.), anti-PI3K (rabbit; 1:1,000; cat. no. 4292; Cell Signaling Technology, Inc.), anti-p-AKT (rabbit; 1:1,000; cat. no. 9271; Cell Signaling Technology, Inc.), anti-AKT (rabbit; 1:1,000; cat. no. 9272; Cell Signaling Technology, Inc.), anti-Beclin-1 (rabbit; 1:200; cat. no. ab62557; Abcam), anti-microtubule associated protein 1 light chain 3 (LC3)I (rabbit; 1:1,000; cat. no. 12741; Cell Signaling Technology, Inc.), anti-LC3II (rabbit; 1:1,000; cat. no. 12741; Cell Signaling Technology, Inc.) and anti-GAPDH (rabbit; 1:10,000; cat. no. ab181602; Abcam). Following primary incubation, the membranes were incubated with a horseradish peroxidase-conjugated goat anti-rabbit secondary antibody (1:2,000; cat. no. ab205718; Abcam) at room temperature for 2 h. Subsequently, the membranes were washed three times with TBST [50 mM Tris-HCl (pH 8.0); 150 mM NaCl; 0.05% Tween-20] for 10 min. Protein bands were visualized using an ECL detection kit (Promega Corporation). Protein expression levels were quantified using ImageJ software (version 1.8.0; National Institute of Health) with GAPDH as the loading control.
Flow cytometry analysis
MPC5 podocytes (1x105 cells/well) were cultured in 24-well plates and FBS-starved for 24 h at 37˚C. To investigate the effects of Lyc on HG-induced MPC5 podocyte injury, cells were then divided into five groups: i) NG; ii) HG; iii) HG + L-Lyc; iv) HG + H-Lyc; and v) H-Lyc. Podocytes were treated with the aforementioned treatment for 48 h at 37˚C. MPC5 podocytes (1x105 cells/well) were cultured in 24-well plates and FBS-starved for 24 h at 37˚C. To investigate the effects of Lyc on HG-induced MPC5 podocyte injury, and the PI3K/AKT pathway, cells were divided into five groups: i) NG; ii) HG; iii) HG + H-Lyc; iv) HG + LY; and v) HG + H-Lyc + LY. Podocytes were treated with the normal glucose, high glucose, high glucose with H-Lyc, high glucose with the PI3K inhibitor 20 µM LY (cat. no. S1105; Selleck Chemicals) or 20 µM LY for 48 h at 37˚C, respectively. MPC5 podocytes (1x105 cells/well) were cultured in 24-well plates and FBS-starved for 24 h at 37˚C. To investigate whether Lyc has an effect on HG-induced MPC5 podocyte injury through autophagy, the cells were divided into five groups: i) NG; ii) HG; iii) HG + H-Lyc; iv) HG + 3-MA; and v) HG + H-Lyc + 3-MA. MPC5 podocytes in the HG + 3-MA and HG + H-Lyc + 3-MA groups were pretreated with 5 mM 3-MA (cat. no. M9281; Sigma-Aldrich; Merck KGaA), an inhibitor of autophagy initiation, for 2 h at 37˚C, then treated with high glucose or H-Lyc additively for 48 h at 37˚C. The cells of NG, HG and HG + H-Lyc groups were treated with normal glucose, high glucose and high glucose + H-Lyc for 48 h at 37˚C, respectively. Subsequently, treated MPC5 podocytes were collected in PBS and stained using the Annexin V-FITC Apoptosis Detection kit (cat. no. CA1020; Beijing Solarbio Science & Technology Co., Ltd.). After washing, MPC5 podocyte apoptosis was determined by flow cytometry using the Accuri™ C6 flow cytometer (BD Biosciences) and Cell Quest software (version 3.3; BD Biosciences). The apoptotic rate was calculated as follows: Apoptotic rate=early + late apoptosis.
Statistical analysis
Data are presented as the mean ± SD. Comparisons among multiple groups were conducted using one-way ANOVA followed by Tukey's post hoc test. P<0.05 was considered to indicate a statistically significant difference. Statistical analyses were performed using SPSS software (version 17.0; SPSS, Inc.). All experiments were performed in triplicate.
Results
Lyc mitigates HG-induced MPC5 podocyte injury
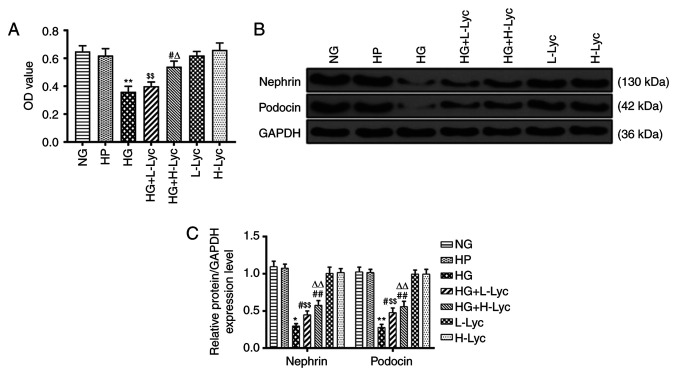
Cell viability was not significantly different between the NG and HP groups, which indicated that HP had a limited effect on MPC5 podocyte viability (Fig. 1A; P>0.05). However, cell viability in the HG group was significantly reduced compared with the NG group (Fig. 1A; P<0.01). Compared with the HG group, cell viability in the HG + L-Lyc group was not significantly increased (Fig. 1A; P>0.05), whereas cell viability was significantly increased in the HG + H-Lyc group (Fig. 1A; P<0.01), which indicated that Lyc may increase MPC5 podocyte viability under HG conditions.
Figure 1.
Lyc protects against HG-induced MPC5 podocyte injury. MPC5 podocytes were divided into the following groups: i) NG (5.5 mM glucose); ii) HP (5.5 mM glucose + 19.5 mM mannitol); iii) HG (25 mM glucose); iv) HG + L-Lyc (25 mM glucose + 3.125 mM Lyc); v) HG + H-Lyc (25 mM glucose + 12.5 mM Lyc); vi) L-Lyc (3.125 mM Lyc); and vii) H-Lyc (12.5 mM Lyc). (A) Effect of Lyc on MPC5 podocyte cell viability. Nephrin and podocin protein expression levels were (B) determined by western blotting and (C) semi-quantified. *P<0.05 and **P<0.01 vs. NG; #P<0.05 and ##P<0.01 vs. HG; $$P<0.01 vs. L-Lyc; ∆P<0.05 and ∆∆P<0.01 vs. H-Lyc. Lyc, lycopene; NG, normal glucose; HP, hypertonic; HG, high glucose; L-Lyc, low-concentration lycopene; H-Lyc, high-concentration lycopene; OD, optical density.
Compared with the NG group, the relative protein expression levels of nephrin and podocin were significantly reduced in the HG group (Fig. 1B and C; P<0.05 and P<0.01, respectively); however, L-Lyc or H-Lyc significantly reversed HG-mediated effects on nephrin and podocin expression (Fig. 1B and C; P<0.05 and P<0.01, respectively).
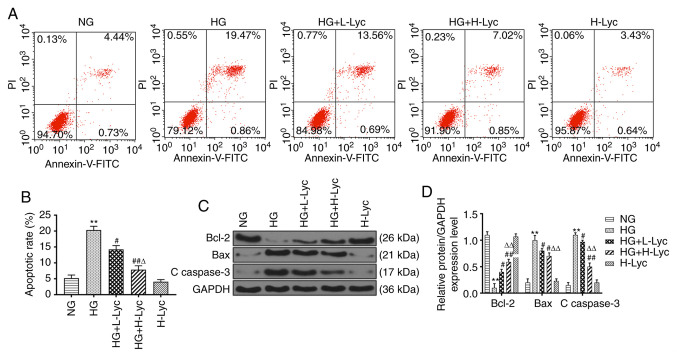
Lyc attenuates HG-induced MPC5 podocyte apoptosis
The effect of HG and Lyc on MPC5 podocyte apoptosis was assessed by flow cytometry (Fig. 2A and B). The HG group displayed a significantly increased rate of apoptosis compared with the NG group (P<0.01), which was significantly reduced by the addition of L-Lyc or H-Lyc (Fig. 2A and B; P<0.05 and P<0.01, respectively). Subsequently, the expression levels of apoptosis-related proteins (Bcl-2, Bax and C caspase-3) in the different groups were measured. Compared with the NG group, the protein expression levels of Bax and C caspase-3 were significantly increased (Fig. 2C and D; P<0.01), whereas the protein expression levels of Bcl-2 were significantly decreased in the HG group (Fig. 2C and D; P<0.01). However, the presence of L-Lyc or H-Lyc significantly reversed HG-mediated effects on the expression of apoptosis-related proteins (Fig. 2C and D; all P<0.05).
Figure 2.
Lyc protects against HG-induced MPC5 podocyte apoptosis. MPC5 podocytes were divided into the following groups: i) NG (5.5 mM glucose); ii) HG (25 mM glucose); iii) HG + L-Lyc (25 mM glucose + 3.125 mM Lyc); iv) HG + H-Lyc (25 mM glucose + 12.5 mM Lyc); and v) H-Lyc (12.5 mM Lyc). The rate of MPC5 podocyte apoptosis was (A) determined by flow cytometry and (B) quantified. The protein expression levels of antiapoptotic (Bcl-2) and proapoptotic (Bax and C caspase-3) proteins were (C) determined by western blotting and (D) semi-quantified. **P<0.01 vs. NG; #P<0.05 and ##P<0.01 vs. HG; ∆P<0.05 and ∆∆P<0.01 vs. H-Lyc. Lyc, lycopene; NG, normal glucose; HG, high glucose; L-Lyc, low-concentration lycopene; H-Lyc, high-concentration lycopene; C caspase-3, cleaved caspase-3; PI, propidium iodide.
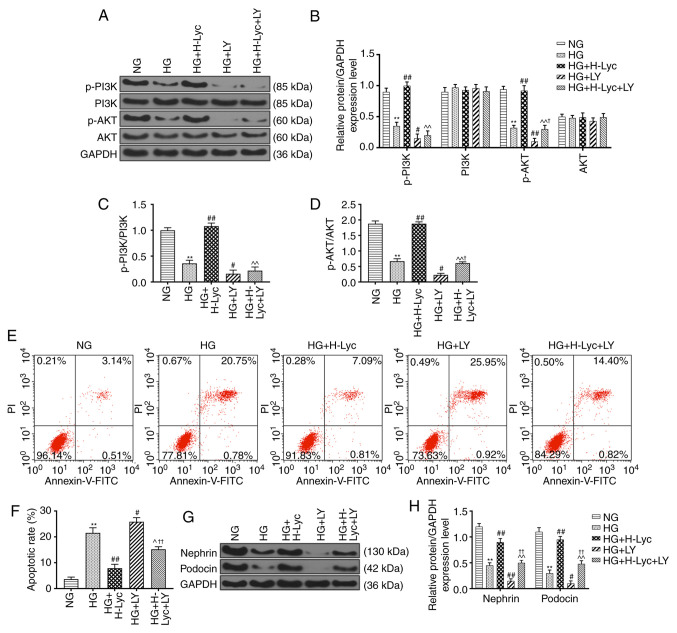
Lyc attenuates HG-induced MPC5 podocyte apoptosis via activation of the PI3K/AKT signaling pathway
In order to investigate the mechanism underlying Lyc-mediated inhibition of HG-induced MPC5 podocyte apoptosis, the expression levels of PI3K/AKT signaling pathway-related proteins were measured via western blotting. Compared with the NG group, the protein expression levels of p-PI3K and p-AKT were significantly downregulated in the HG group (Fig. 3A-D; P<0.01). By contrast, compared with the HG group, the protein expression levels of p-PI3K and p-AKT were significantly upregulated in the HG + H-Lyc group (Fig. 3A-D; P<0.01), but significantly downregulated in the HG + LY group (Fig. 3A-D; P<0.05 and P<0.01, respectively). Moreover, compared with the HG + H-Lyc group, the expression levels of p-PI3K and p-AKT were significantly downregulated in the HG + H-Lyc + LY group (Fig. 3A-D; P<0.01). In addition, HG-mediated downregulation of p-PI3K and p-AKT expression levels was enhanced by LY, with the HG + LY group displaying significantly decreased p-PI3K and p-AKT expression levels compared with the HG group (Fig. 3A-D; P<0.05 and P<0.01, respectively). However, under HG conditions, Lyc increased the expression levels of p-PI3K and p-AKT in MPC5 podocytes but LY inhibited Lyc-mediated upregulation (Fig. 3A-D).
Figure 3.
Lyc enhances PI3K/AKT activation and inhibits HG-induced MPC5 podocyte apoptosis. MPC5 podocytes were divided into the following groups: i) NG (5.5 mM glucose); ii) HG (25 mM glucose); iii) HG + H-Lyc (25 mM glucose + 12.5 mM Lyc); iv) HG + LY (25 mM glucose + 20 µM LY); and v) HG + H-Lyc + LY (25 mM glucose + 12.5 mM Lyc + 20 µM LY). Protein expression levels of p-PI3K, PI3K, p-AKT and AKT were (A) determined by western blotting and (B) quantified. The ratios of (C) p-PI3K/PI3K and (D) p-AKT/AKT. The rate of apoptosis was (E) determined by flow cytometry and (F) quantified. Protein expression levels of nephrin and podocin were (G) determined by western blotting and (H) quantified. **P<0.01 vs. NG; #P<0.05 and ##P<0.01 vs. HG; ^P<0.05 and ^^P<0.01 vs. HG + H-Lyc; †P<0.05 and ††P<0.01 vs. HG + LY. Lyc, lycopene; NG, normal glucose; HG, high glucose; H-Lyc, high-concentration lycopene; LY, LY294002; p, phosphorylated; PI, propidium iodide.
Flow cytometry analysis indicated that the HG group displayed significantly increased rates of MPC5 podocyte apoptosis compared with those in the NG group, whereas the HG + H-Lyc + LY group displayed a significantly reduced rate of MPC5 podocyte apoptosis compared with that in the HG + LY group (Fig. 3E and F; P<0.01). HG + H-Lyc + LY group exhibited significantly increased rates of MPC5 podocyte apoptosis compared with those in the HG + H-Lyc group (Fig. 3E and F; P<0.05). In addition the expression levels of nephrin and podocin were significantly reduced in the HG group compared with the NG group. Compared with those in the HG group, the expression levels of nephrin and podocin were significantly increased in the HG + H-Lyc group (Fig. 3G and H; P<0.01), but were significantly reduced in the HG + LY group compared with HG group (Fig. 3G and H; P<0.05, respectively). However, the HG + H-Lyc + LY group displayed significantly increased nephrin and podocin expression levels compared with the HG + LY group (Fig. 3G and H; P<0.01).
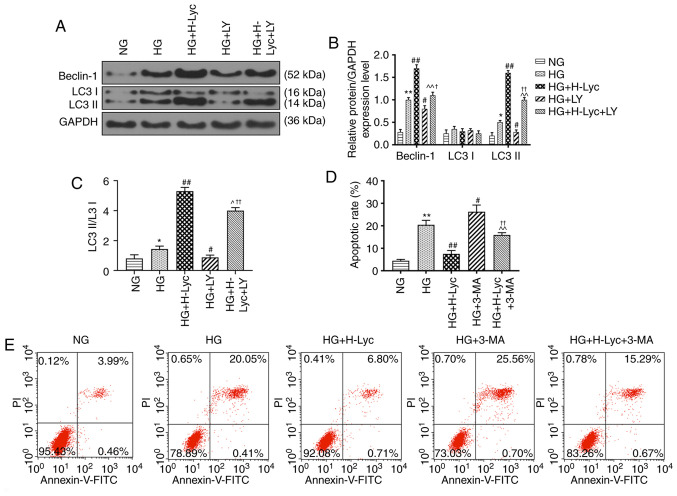
Lyc mitigates HG-induced MPC5 podocyte apoptosis by promoting autophagy
The expression levels of autophagy-related proteins were measured via western blotting. The protein expression levels of LC3II/LC3I and Beclin-1 were significantly upregulated in the HG group compared with the NG group (Fig. 4A-C; P<0.05 and P<0.01, respectively). Compared with the HG group, the protein expression levels of LC3II/LC3I and Beclin-1 were significantly increased in the HG + H-Lyc group (P<0.01), but significantly decreased in the HG + LY group (P<0.05). Moreover, the effects of LY on Beclin-1 and LC3II/LC3I expression levels were significantly inhibited in the presence of Lyc under HG conditions (Fig. 4A-C; P<0.05 and P<0.01, respectively).
Figure 4.
Lyc inhibits HG-induced MPC5 podocyte apoptosis by promoting autophagy via the PI3K/AKT signaling pathway. MPC5 podocytes were divided into the following groups: i) NG (5.5 mM glucose); ii) HG (25 mM glucose); iii) HG + H-Lyc (25 mM glucose + 12.5 mM Lyc); iv) HG + LY (25 mM glucose + 20 µM LY); and v) HG + H-Lyc + LY (25 mM glucose + 12.5 mM Lyc + 20 µM LY). Protein expression levels of Beclin-1, LC3I and LC3II were (A) determined by western blotting and (B) quantified. (C) The ratio of LC3II/LC3I. *P<0.05 and **P<0.01 vs. NG; #P<0.05 and ##P<0.01 vs. HG; ^P<0.05 and ^^P<0.01 vs. HG + H-Lyc; †P<0.05 and ††P<0.01 vs. HG + LY. MPC5 podocytes were divided into the following groups: i) NG (5.5 mM glucose); ii) HG (25 mM glucose); iii) HG + H-Lyc (25 mM glucose + 12.5 mM Lyc); iv) HG + 3-MA (25 mM glucose + 5 mM 3-MA); and v) HG + H-Lyc + 3-MA (25 mM glucose + 12.5 mM Lyc + 5 mM 3-MA). (D) The rate of apoptosis was determined by flow cytometry. (E) Representative scatter plots. **P<0.01 vs. NG; #P<0.05 and ##P<0.01 vs. HG; ^^P<0.01 vs. HG + H-Lyc; ††P<0.01 vs. HG + 3-MA. Lyc, lycopene; NG, normal glucose; HG, high glucose; H-Lyc, high-concentration lycopene; LY, LY294002; LC3, microtubule associated protein 1 light chain 3; 3-MA, 3-methyladenine; PI, propidium iodide.
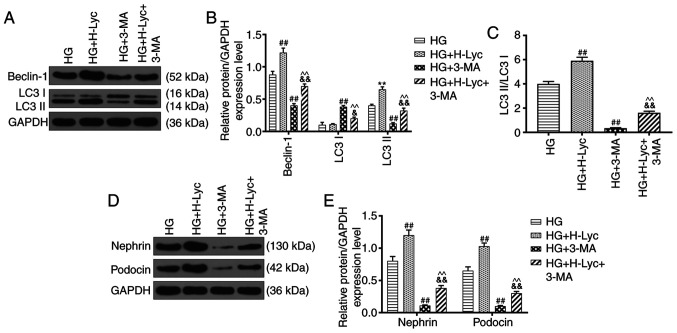
Autophagy is associated with cell apoptosis (22), so whether 3-methyladenine (3-MA), an autophagy inhibitor, altered the protective effect of Lyc on HG-induced MPC5 podocyte apoptosis was investigated. The HG + 3-MA group displayed significantly increased rates of MPC5 podocyte apoptosis compared with the HG group (Fig. 4D and E; P<0.05). Pretreament with 3-MA inhibited the protective effect of Lyc on HG-induced MPC5 podocyte apoptosis (Fig. 4D and E; P<0.01). In addition, compared with the HG group, the protein expression levels of LC3II/LC3I and Beclin-1 were significantly increased in the HG + H-Lyc group, but significantly reduced in the HG + 3-MA group (Fig. 5A-C; P<0.01). Moreover, 3-MA partially reversed H-Lyc-mediated effects on autophagy-related protein expression under HG conditions (Fig. 5A-C; P<0.01). Furthermore, compared with the HG group, the HG + H-Lyc group displayed significantly increased podocin and nephrin expression levels (P<0.01), whereas the HG + 3-MA group displayed significantly decreased expression levels of podocin and nephrin (Fig. 5D-E; P<0.01). In addition, 3-MA weakened H-Lyc-induced podocin and nephrin expression (Fig. 5D-E; P<0.01).
Figure 5.
Lyc promotes the expression of nephrin, podocin and autophagy-related proteins in MPC5 podocytes. MPC5 podocytes were divided into the following groups: i) HG (25 mM glucose); ii) HG + H-Lyc (25 mM glucose + 12.5 mM Lyc); iii) HG + 3-MA (25 mM glucose + 5 mM 3-MA); and HG + H-Lyc + 3-MA (25 mM glucose + 12.5 mM Lyc + 5 mM 3-MA). Protein expression levels of Beclin-1, LC3II and LC3I were (A) determined by western blotting and (B) semi-quantified. (C) The ratio of LC3BII/LC3BI. Protein expression levels of nephrin and podocin were (D) determined by western blotting and (E) semi-quantified. ##P<0.01 vs. HG; ^^P<0.01 vs. HG + H-Lyc; &P<0.05 and &&P<0.01 vs. HG + 3-MA. Lyc, lycopene; HG, high glucose; H-Lyc, high-concentration lycopene; 3-MA, 3-methyladenine; LC3, microtubule associated protein 1 light chain 3.
Therefore, the results suggested that HG and Lyc significantly enhanced MPC5 podocyte autophagy, which may be crucial for the protective effects of Lyc against HG-induced MPC5 podocyte apoptosis.
Discussion
The function of a podocyte depends on the cytoskeleton and certain marker proteins of podocytes, such as nephrin and podocin (23). Nephrin and podocin proteins function as glomerular filtration barriers, and their expression affects the permeability of the glomerular matrix membrane, which in turn affects kidney function (24,25). Abnormal expression of nephrin and podocin is a marker of podocyte injury (26). Consistent with previous study (21), the present study demonstrated that Lyc significantly reversed the inhibitory effects of HG on cell viability, and nephrin and podocin expression in podocytes.
Podocyte loss is one of the most important pathological alterations that occurs in DN (27). As a key event during DN progression, HG-induced apoptosis is often accompanied by podocyte loss, which is an early feature of DN and is highly predictive of the disease progression (28-30). Apoptosis-defined programmed cell death is also caused by external killers in certain circumstances (31). For example, irradiation or drugs used for cancer chemotherapy results in DNA damage in some cells, which can lead to apoptotic death (32). In the present study, the results indicated that HG promoted MPC5 podocyte apoptosis, and Lyc mitigated MPC5 podocyte apoptosis under HG conditions. Apoptosis can be regulated via the PI3K/AKT signaling pathways (33) and PI3K/AKT can be activated by a number of different cellular stimuli or toxic insults (34,35). Previous studies have reported that Lyc can regulate PI3K/AKT signaling pathways in prostate cancer and human mesenchymal stem cells (36,37). To further explore the mechanism underlying the effects of Lyc on HG-induced MPC5 podocyte apoptosis, a PI3K inhibitor was used. Lyc activated the PI3K/AKT signaling pathway, which inhibited HG-mediated MPC5 podocyte apoptosis. Similarly, a previous study reported that Lyc reduced tert-Butyl hydroperoxide-induced cell apoptosis, which is possibly related to activation of the PI3K/AKT signaling pathway (38). However, Chan et al (39) reported that Lyc can inhibit the PI3K/AKT signaling pathway during platelet-derived growth factor-BB-induced retinal pigment epithelial cell migration. In addition, the results of the present study suggested that Lyc treatment had no significant effect on cell viability or the expression of nephrin and podocin in MPC5 podocytes compared with the NG and HG groups; however, under HG conditions, Lyc treatment promoted MPC5 podocyte cell viability, and nephrin and podocin protein expression. Li et al (40) reported that Lyc treatment had no significant effect on ameloblast apoptosis, whereas Lyc attenuated fluoride-induced ameloblasts apoptosis. Collectively, the aforementioned findings indicated that Lyc may serve a role under certain pathological conditions.
As the inhibition of autophagy is highly associated with triggering cell apoptosis, and both apoptosis and autophagy are involved in cell growth, differentiation, and death (41), whether autophagy regulated the protective effect of Lyc on HG-induced MPC5 podocyte apoptosis was investigated. Autophagy is a natural process related to numerous human diseases, which can protect the human body from cell injury and death (42). Therefore, autophagic activity of podocytes serves a protective role in renal injury and could delay the progression of podocytopathies (43). Cell apoptosis and autophagy are complex regulatory processes that involve a number of upstream regulatory signaling pathways, including the PI3K/AKT signaling pathway (44). A previous study demonstrated that enhancing autophagy protects against palmitic acid-mediated podocyte apoptosis (45), and progranulin alleviates HG-induced human podocyte injury by regulating CAMKK/AMPK-mediated autophagy (46). Therefore, promoting autophagy could attenuate HG-induced podocyte apoptosis (47). Moreover, resveratrol protected against podocyte apoptosis by activating autophagy in DN model mice (48). Consistently, another study reported that Lyc protected against apoptosis by increasing autophagy in hypoxia/reoxygenation-induced H9C2 myocardioblast cells (49). However, Kobayashi et al (50) reported that suppression of autophagy exerted protective effects against HG-mediated cardiomyocyte injury. In the present study, the results also indicated that the HG group displayed increased apoptosis compared with the NG group, and promotion of autophagy promotes apoptosis, which is a stress phenomenon (51).
In the present study, 3-MA, an autophagy inhibitor, was used to explore the mechanism underlying the effect of Lyc on autophagy. The results indicated that promoting autophagy by Lyc attenuated HG-mediated MPC5 podocyte apoptosis, which was associated with activation of the PI3K/AKT signaling pathway. It has been reported that Lyc relieved HG-mediated podocyte injury by promoting MPC5 podocyte autophagy, which may involve the PI3K/AKT signaling pathway (21). Similarly, a previous study reported that notoginsenoside R1 attenuated glucose-mediated podocyte injury by reducing apoptosis and promoting autophagy via activation of the PI3K/AKT/mTOR signaling pathway (11). However, in colorectal cancer cells, autophagy and apoptosis were promoted by Grape seed procyanidin B2 via regulation of the PI3K/AKT signaling pathway (44).
In conclusion, the present study indicated that Lyc protected against HG-induced MPC5 podocyte injury and attenuated HG-induced MPC5 podocyte apoptosis by promoting autophagy via activation of the PI3K/AKT signaling pathway. The present study demonstrated a potential mechanism underlying the therapeutic effect of Lyc on DN, which may serve as a potential therapeutic target for DN progression.
Acknowledgements
Not applicable.
Funding
No funding was received.
Availability of data and materials
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Authors' contributions
QW substantially contributed to the conception and design of the study. RL, ZX and CH acquired, analyzed and interpreted the data. QW drafted the manuscript and critically revised it for important intellectual content. All authors read and approved the final manuscript.
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Jin Y, Liu S, Ma Q, Xiao D, Chen L. Berberine enhances the AMPK activation and autophagy and mitigates high glucose-induced apoptosis of mouse podocytes. Eur J Pharmacol. 2017;794:106–114. doi: 10.1016/j.ejphar.2016.11.037. [DOI] [PubMed] [Google Scholar]
- 2.Hazman Ö, Bozkurt MF. Anti-inflammatory and antioxidative activities of safranal in the reduction of renal dysfunction and damage that occur in diabetic nephropathy. Inflammation. 2015;38:1537–1545. doi: 10.1007/s10753-015-0128-y. [DOI] [PubMed] [Google Scholar]
- 3.Aso Y. Cardiovascular disease in patients with diabetic nephropathy. Curr Mol Med. 2008;8:533–543. doi: 10.2174/156652408785747960. [DOI] [PubMed] [Google Scholar]
- 4.Lee JH, Kim D, Oh YS, Jun HS. Lysophosphatidic acid signaling in diabetic nephropathy. Int J Mol Sci. 2019;20(2850) doi: 10.3390/ijms20112850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vervoort G. Treatment Goals in Diabetic Nephropathy. Pathophysiology and Clinical. Aspects. In: Diabetic Nephropathy. Roelofs J and Vogt L (eds). Springer, Cham, pp435-450, 2019. [Google Scholar]
- 6.Najafian B, Alpers CE, Fogo AB. Pathology of human diabetic nephropathy. Contrib Nephrol. 2011;170:36–47. doi: 10.1159/000324942. [DOI] [PubMed] [Google Scholar]
- 7.Ziyadeh FN, Hoffman BB, Han DC, Iglesias-De La, Cruz MC, Hong SW, Isono M, Chen S, McGowan TA, Sharma K. Long-term prevention of renal insufficiency, excess matrix gene expression, and glomerular mesangial matrix expansion by treatment with monoclonal antitransforming growth factor-beta antibody in db/db diabetic mice. Proc Natl Acad Sci USA. 2000;97:8015–8020. doi: 10.1073/pnas.120055097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Yu SM, Bonventre JV. Acute kidney injury and progression of diabetic kidney disease. Adv Chronic Kidney Dis. 2018;25:166–180. doi: 10.1053/j.ackd.2017.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Tharaux PL, Huber TB. How is proteinuric diabetic nephropathy caused by disturbed proteostasis and autophagy in podocytes? Diabetes. 2016;65:539–541. doi: 10.2337/dbi15-0026. [DOI] [PubMed] [Google Scholar]
- 10.Marusugi K, Nakano K, Sasaki H, Kimura J, Yanobu-Takanashi R, Okamura T, Sasaki N. Functional validation of tensin2 SH2-PTB domain by CRISPR/Cas9-mediated genome editing. J Vet Med Sci. 2016;78:1413–1420. doi: 10.1292/jvms.16-0205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Huang G, Zou B, Lv J, Li T, Huai G, Xiang S, Lu S, Luo H, Zhang Y, Jin Y, Wang Y. Notoginsenoside R1 attenuates glucose-induced podocyte injury via the inhibition of apoptosis and the activation of autophagy through the PI3K/Akt/mTOR signaling pathway. Int J Mol Med. 2017;39:559–568. doi: 10.3892/ijmm.2017.2864. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Barisoni L, Schnaper HW, Kopp JB. Advances in the biology and genetics of the podocytopathies: Implications for diagnosis and therapy. Arch Pathol Lab Med. 2009;133:201–216. doi: 10.1043/1543-2165-133.2.201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shankland SJ. The podocyte's response to injury: Role in proteinuria and glomerulosclerosis. Kidney Int. 2006;69:2131–2147. doi: 10.1038/sj.ki.5000410. [DOI] [PubMed] [Google Scholar]
- 14.Agarwal S, Rao AV. Tomato lycopene and its role in human health and chronic diseases. CMAJ. 2000;163:739–744. [PMC free article] [PubMed] [Google Scholar]
- 15.Fan S, Sun JB, Li R, Song X, Li J. Lycopene protects myocardial ischemia injury through anti-apoptosis and anti-oxidative stress. Eur Rev Med Pharmacol Sci. 2019;23:3096–3104. doi: 10.26355/eurrev_201904_17593. [DOI] [PubMed] [Google Scholar]
- 16.Sahin K, Orhan C, Sahin N, Kucuk O. Anticancer Properties of Lycopene. Springer, Cham, 2019. [Google Scholar]
- 17.Kawata A, Murakami Y, Suzuki S, Fujisawa S. Anti-inflammatory activity of β-Carotene, Lycopene and Tri-n-butylborane, a scavenger of reactive oxygen species. In Vivo. 2018;32:255–264. doi: 10.21873/invivo.11232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li W, Wang G, Lu X, Jiang Y, Xu L, Zhao X. Lycopene ameliorates renal function in rats with streptozotocin-induced diabetes. Int J Clin Exp Pathol. 2014;7:5008–5015. [PMC free article] [PubMed] [Google Scholar]
- 19.Ni L, Yang XL, Qiao H, Lin J. Effects of lycopene on high glucose-induced autophagy and apoptosis of podocytes. Clin Nephrol. 2018;18:48–53. (In Chinese) [Google Scholar]
- 20.Liu WT, Peng FF, Li HY, Chen XW, Gong WQ, Chen WJ, Chen YH, Li PL, Li ST, Xu ZZ, Long HB. Metadherin facilitates podocyte apoptosis in diabetic nephropathy. Cell Death Dis. 2016;7(e2477) doi: 10.1038/cddis.2016.335. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ni L, Qiao H, Lin J, Chang-xuan L. Lycopene relieves the damage of mouse podocytes induced by high glucose and its mechanism. Southeast Univ (Med Sci Edi) 2018;37:229–234. (In Chinese) [Google Scholar]
- 22.Wang K. Autophagy and apoptosis in liver injury. Cell Cycle. 2015;14:1631–1642. doi: 10.1080/15384101.2015.1038685. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gao F, He X, Liang S, Liu S, Liu H, He Q, Chen L, Jiang H, Zhang Y. Quercetin ameliorates podocyte injury via inhibition of oxidative stress and the TGF-β1/Smad pathway in DN rats. RSC Adv. 2018;8:35413–35421. doi: 10.1039/c8ra07935h. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wei M, Li Z, Xiao L, Yang Z. Effects of ROS-relative NF-KB signaling on high glucose-induced TLR4 and MCP-1 expression in podocyte injury. Mol Immunol. 2015;68:261–271. doi: 10.1016/j.molimm.2015.09.002. [DOI] [PubMed] [Google Scholar]
- 25.Gong Q, He LL, Wang ML, Ouyang H, Gao HW, Feng YL, Yang SL, Du LJ, Li J, Luo YY. Anemoside B4 protects rat kidney from adenine-induced injury by attenuating inflammation and fibrosis and enhancing podocin and nephrin expression. Evid Based Complement Alternat Med. 2019;2019(8031039) doi: 10.1155/2019/8031039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Tesch GH. Review: Serum and urine biomarkers of kidney disease: A pathophysiological perspective. Nephrology (Carlton) 2010;15:609–616. doi: 10.1111/j.1440-1797.2010.01361.x. [DOI] [PubMed] [Google Scholar]
- 27.Szrejder M, Piwkowska A. AMPK signalling: Implications for podocyte biology in diabetic nephropathy. Biol Cell. 2019;111:109–120. doi: 10.1111/boc.201800077. [DOI] [PubMed] [Google Scholar]
- 28.Reidy K, Kang HM, Hostetter T, Susztak K. Molecular mechanisms of diabetic kidney disease. J Clin Invest. 2014;124:2333–2340. doi: 10.1172/JCI72271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Li W, Wang Q, Du M, Ma X, Wu L, Guo F, Zhao S, Huang F, Wang H, Qin G. Effects of overexpressing FoxO1 on apoptosis in glomeruli of diabetic mice and in podocytes cultured in high glucose medium. Biochem Biophys Res Commun. 2016;478:612–617. doi: 10.1016/j.bbrc.2016.07.115. [DOI] [PubMed] [Google Scholar]
- 30.Saurus P, Kuusela S, Dumont V, Lehtonen E, Fogarty CL, Lassenius MI, Forsblom C, Lehto M, Saleem MA, Groop PH, Lehtonen S. Cyclin-dependent kinase 2 protects podocytes from apoptosis. Sci Rep. 2016;6(21664) doi: 10.1038/srep21664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Nagata S. Apoptosis by death factor. Cell. 1997;88:355–365. doi: 10.1016/s0092-8674(00)81874-7. [DOI] [PubMed] [Google Scholar]
- 32.Fulda S. Therapeutic exploitation of necroptosis for cancer therapy. Semin Cell Dev Biol. 2014;35:51–56. doi: 10.1016/j.semcdb.2014.07.002. [DOI] [PubMed] [Google Scholar]
- 33.Dey JH, Bianchi F, Voshol J, Bonenfant D, Oakeley EJ, Hynes NE. Targeting fibroblast growth factor receptors blocks PI3K/AKT signaling, induces apoptosis, and impairs mammary tumor outgrowth and metastasis. Cancer Res. 2016;70:4151–4162. doi: 10.1158/0008-5472.CAN-09-4479. [DOI] [PubMed] [Google Scholar]
- 34.Song H, Fu Y, Wan D, Xia W, Lyu F, Liu L, Shen L. Mytoxin B and myrothecine A induce apoptosis in human hepatocarcinoma cell line SMMC-7721 via PI3K/Akt signaling pathway. Molecules. 2019;24(2291) doi: 10.3390/molecules24122291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Porta C, Figlin RA. Phosphatidylinositol-3-Kinase/Akt signaling pathway and kidney cancer, and the therapeutic potential of phosphatidylinositol-3-Kinase/Akt inhibitors. J Urol. 2009;182:2569–2577. doi: 10.1016/j.juro.2009.08.085. [DOI] [PubMed] [Google Scholar]
- 36.Chen J, O'Donoghue A, Deng YF, Zhang B, Kent F, O'Hare T. The effect of lycopene on the PI3K/Akt signalling pathway in prostate cancer. Anticancer Agents Med Chem. 2014;14:800–805. doi: 10.2174/1871520614666140521121317. [DOI] [PubMed] [Google Scholar]
- 37.Ji YK, Lee JS, Han YS, Lee JH, Bae I, Yoon YM, Kwon SM, Lee SH. Pretreatment with lycopene attenuates oxidative Stress-Induced apoptosis in human mesenchymal stem cells. Biomol Ther (Seoul) 2015;23:517–524. doi: 10.4062/biomolther.2015.085. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Huang C, Wen C, Yang M, Gan D, Fan C, Li A, Li Q, Zhao J, Zhu L, Lu D. Lycopene protects against t-BHP-induced neuronal oxidative damage and apoptosis via activation of the PI3K/Akt pathway. Mol Biol Rep. 2019;46:3387–3397. doi: 10.1007/s11033-019-04801-y. [DOI] [PubMed] [Google Scholar]
- 39.Chan CM, Fang JY, Lin HH, Yang CY, Hung CF. Lycopene inhibits PDGF-BB-induced retinal pigment epithelial cell migration by suppression of PI3K/Akt and MAPK pathways. Biochem Biophys Res Commun. 2009;388:172–176. doi: 10.1016/j.bbrc.2009.07.155. [DOI] [PubMed] [Google Scholar]
- 40.Li W, Jiang B, Cao X, Xie Y, Huang T. Protective effect of lycopene on fluoride-induced ameloblasts apoptosis and dental fluorosis through oxidative stress-mediated Caspase pathways. Chem Biol Interact. 2017;261:27–34. doi: 10.1016/j.cbi.2016.11.021. [DOI] [PubMed] [Google Scholar]
- 41.Fiandalo MV, Kyprianou N. Caspase control: Protagonists of cancer cell apoptosis. Exp Oncol. 2012;34:165–175. [PMC free article] [PubMed] [Google Scholar]
- 42.Murrow L, Debnath J. Autophagy as a stress-response and quality-control mechanism: Implications for cell injury and human disease. Annu Rev Pathol. 2013;8:105–137. doi: 10.1146/annurev-pathol-020712-163918. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Zeng C, Fan Y, Wu J, Shi S, Chen Z, Zhong Y, Zhang C, Zen K, Liu Z. Podocyte autophagic activity plays a protective role in renal injury and delays the progression of podocytopathies. J Pathol. 2014;234:203–213. doi: 10.1002/path.4382. [DOI] [PubMed] [Google Scholar]
- 44.Zhang R, Yu Q, Lu W, Shen J, Zhou D, Wang Y, Gao S, Wang Z. Grape seed procyanidin B2 promotes the autophagy and apoptosis in colorectal cancer cells via regulating PI3K/Akt signaling pathway. Onco Targets Ther. 2019;12:4109–4118. doi: 10.2147/OTT.S195615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Jiang XS, Chen XM, Wan JM, Gui HB, Ruan XZ, Du XG. Autophagy protects against palmitic acid-induced apoptosis in podocytes in vitro. Sci Rep. 2017;7(42764) doi: 10.1038/srep42764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Zhou D, Zhou M, Wang Z, Fu Y, Jia M, Wang X, Liu M, Zhang Y, Sun Y, Zhou Y, et al. Progranulin alleviates podocyte injury via regulating CAMKK/AMPK-mediated autophagy under diabetic conditions. J Mol Med (Berl) 2019;97:1507–1520. doi: 10.1007/s00109-019-01828-3. [DOI] [PubMed] [Google Scholar]
- 47.Dong C, Zheng H, Huang S, You N, Xu J, Ye X, Zhu Q, Feng Y, You Q, Miao H, et al. Heme oxygenase-1 enhances autophagy in podocytes as a protective mechanism against high glucose-induced apoptosis. Exp Cell Res. 2015;337:146–159. doi: 10.1016/j.yexcr.2015.04.005. [DOI] [PubMed] [Google Scholar]
- 48.Huang SS, Ding DF, Chen S, Dong CL, Ye XL, Yuan YG, Feng YM, You N, Xu JR, Miao H, et al. Resveratrol protects podocytes against apoptosis via stimulation of autophagy in a mouse model of diabetic nephropathy. Sci Rep. 2017;7(45692) doi: 10.1038/srep45692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Chen F, Sun ZW, Ye LF, Fu GS, Mou Y, Hu SJ. Lycopene protects against apoptosis in hypoxia/reoxygenation-induced H9C2 myocardioblast cells through increased autophagy. Mol Med Rep. 2015;11:1358–1365. doi: 10.3892/mmr.2014.2771. [DOI] [PubMed] [Google Scholar]
- 50.Kobayashi S, Xu X, Chen K, Liang Q. Suppression of autophagy is protective in high glucose-induced cardiomyocyte injury. Autophagy. 2012;8:577–592. doi: 10.4161/auto.18980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Chiarelli R, Martino C, Agnello M, Bosco L, Roccheri MC. Autophagy as a defense strategy against stress: Focus on paracentrotus lividus sea urchin embryos exposed to cadmium. Cell Stress Chaperones. 2016;21:19–27. doi: 10.1007/s12192-015-0639-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.