Abstract
Variation in stress responses between individuals are linked to factors ranging from stress coping styles to sensitivity of neurotransmitter systems. Many anxiolytic compounds (e.g. ethanol) can increase stressor engagement through modulation of neurotransmitter systems and are used to investigate stress response mechanisms. There are two alternative suites of correlated behavioral and physiological responses to stressors (stress coping styles) that differ in exploration tendencies: proactive and reactive stress coping styles. By chronically treating individuals differing in stress coping style with ethanol, a GABA-acting drug, we assessed the role of the GABAergic system on the behavioral stress response. Specifically, we investigated resulting changes in stress-related behavior (i.e. exploratory behavior) and whole-brain GABAA receptor subunits (gabra1, gabra2, gabrd, & gabrg2) in response to a novelty stressor. We found that ethanol-treated proactive individuals showed lower stress-related behaviors than their reactive counterparts. Proactive individuals showed significantly higher expression of gabra1, gabra2, and gabrg2 compared to reactive individuals and ethanol treatment resulted in upregulation of gabra1 and gabrg2 in both stress coping styles. These results suggest that impacts of ethanol on stress-related behaviors vary by stress coping style and that expression of select GABAA receptor subunits may be one of the underlying mechanisms.
Subject terms: Genetics of the nervous system, Ion channels in the nervous system, Molecular neuroscience
Introduction
While an organism’s stress response is essential to its survival, not all conspecifics exhibit similar responses and often differ both behaviorally and physiologically1–5. Upon perception of a stressor the nervous system simultaneously stimulates the sympathetic adrenal medullary and the hypothalamic–pituitary–adrenal pathways within mammals that rapidly lead to changes in behavior and the endocrine system. An individual’s behavioral and physiological stress responses are often correlated and are consistent across contexts. Throughout many taxa there exists two alternative correlated suites of behavioral and physiological responses to stressors known as the proactive and reactive stress coping styles2,3,5–7. In response to novelty, proactive individuals actively engage stressors and characteristically exhibit a lower whole-body cortisol response compared to reactive individuals2,3,5,8–11. Additionally, proactive and reactive individuals differ in expression of key neurotransmitter receptors related to stress and anxiety, such as serotonin, dopamine, and GABA (γ-amino butyric acid) receptors2,3,12,13. Drugs designed to target such systems are often employed to study a neurotransmitter’s influence on stress-related behaviors14–16. Therefore, pharmaceuticals can be used to investigate underlying differences in the molecular mechanisms between stress coping styles. Specifically, measuring different molecular responses to behavioral-altering anxiolytics or anxiogenics can provide insight on the underlying mechanisms of these individual differences and ultimately stress and anxiety.
Dysregulation of the GABAergic, the serotoninergic, and the glutamatergic systems often contribute to a disproportional behavioral stress response14,17, which, if sustained over an extended period of time, can be classified as an anxiety disorder18,19. GABAergic system dysfunction is thought to contribute to the underlying etiology of anxiety-related disorders20,21. GABAA receptor (GABAAR) agonists, such as ethanol, allow for positive modulation of the GABAergic system to produce an anxiolytic response, while antagonists result in an anxiogenic response in rodents (Rattus norvegicus, Mus musculus) and zebrafish (Danio rerio) 16,17,22–31. The GABAAR itself is a pentamer composed of any combination of the α-, β-, γ-, δ-, ε-, and θ-subunits and each has their own respective variants (α1–α6, β1–β3, γ1–γ3, ρ1–ρ3, δ, ε, θ)32. GABA-acting drugs influence the expression of the protein subunits that make up the receptor subtype32,33. For example, rodents exposed to GABAA agonists show an increase in expression of the α1-, α2-, and δ-subunits of the GABAAR, while expression of the γ2-subunit decreases34–37. Studies utilizing zebrafish similarly show that ethanol administration produces anxiolytic behavioral effects14,23,30,31,38,39. In zebrafish, there are baseline differences in mRNA expression of both GABAA and GABAB receptors between the two stress coping styles13. How GABA-acting drugs differentially influence both behavior and physiology between them, however, is not understood.
Zebrafish are widely used to understand the effects of pharmaceuticals on stress and anxiety-related behaviors and physiology due to their conserved behavioral, neuroanatomical, pharmacological, and transcriptional stress responses with mammals and other species14–16,31,40–44. Many studies have examined the anxiogenic and anxiolytic impacts of pharmaceuticals and developed a variety of behavioral assays to measure stress and anxiety14,16,41,45–48. For example one assay used to quantify stress-related behaviors is the Novel Tank Diving Test (NTDT), which measures a subject’s level of vertical exploration as a behavioral proxy for stress10,31,45,49,50. In the NTDT there is an inverse relationship between stress levels and depth preference10,31,45,49,50. Several studies have also validated the NTDT as a model to study anxiolytic compounds (e.g. GABA acting drugs)51–53. Many studies have focused on acute effects of ethanol but relatively less is known on effects of chronic treatment14,30,49,54–56.
Of note both wild and laboratory strains of zebrafish show the proactive and reactive stress coping styles5,6. These coping styles in zebrafish display differences in genetic backgrounds, behavior and neuroendocrine responses to stressors that are consistent with what has been documented in birds and mammals14,57,58. Using artificial selection, we previously generated two lines of zebrafish (low stationary behavior, LSB; high stationary behavior, HSB) that show consistency with the proactive and reactive stress coping styles. More specifically, the LSB and HSB lines show consistent differences in stress-related behaviors across multiple behavioral assays, morphology and escape performance, whole-brain transcriptome profiles, cognitive performances, and endocrine responses characteristic of the proactive and reactive stress coping styles, respectively5–7,10,11,59–63. Only recently are studies beginning to demonstrate the roles of synaptic plasticity and neurotransmitter system regulation in facilitating the display of alternative stress coping styles in zebrafish5,7,13,59,62,64. However, the differential impact of GABA-acting drugs (e.g. ethanol) on behavior and the GABA system between stress coping styles is just beginning to be explored55,56.
In this study, we assessed the effects of ethanol treatment on stress-related behavior and GABAAR subunit gene expression in two zebrafish lines selectively bred to display the proactive and reactive stress coping styles. Specifically, we quantified exploratory behavior using the NTDT and expression of four genes encoding for the α1-, α2-, δ-, and γ2-subunits of the GABAAR (gabra1, gabra2, gadrd, and gabrg2, respectively65. These particular subunits were chosen as they are found in relatively high abundance in the GABAAR32,35, and previous studies in other species suggest the expression of these subunits is altered by GABA-acting drugs34–37. We hypothesized that chronic ethanol treatment will reduce stress-related behaviors in both lines of zebrafish with a greater anxiolytic response for the reactive line. Additionally, based on previous literature we predicted to see an increase in mRNA expression of α1-, α2-, δ-subunits and decrease expression of the γ2-subunit for both lines but the magnitude of the effect would be greater in the reactive line34–37. Understanding how a GABAAR agonist impacts GABA neurotransmission between the two coping styles will give insight into one mechanism that may explain differences in their stress and anxiety-related behavioral responses.
Results
Identifying an ethanol treatment duration and concentration that produces an anxiolytic effect across lines
To find a biologically relevant dose and treatment length applicable to both HSB and LSB fish, we tested durations from 7 days (0.25%, 0.4%, 0.5%, 0.75%, 1%, 1.15%, 1.25%, and 1.5% ethanol), 10 days (0.5% ethanol), and 14 days (0.5% and 0.75% ethanol) (Fig. 1, Supplementary Table S1). There were significant main effects of 0.75% ethanol concentration on time spent in the top half of the tank for both the HSB and LSB lines at the 14-day duration [HSB: Wald χ2(2) = 12.338, p = 4.43 × 10–4; LSB: Wald χ2(2) = 8.707, p = 0.003]. Examination of simple main effects revealed fish treated with 0.75% ethanol concentration showed an increase in time spent in the top half of the tank compared to 0.0% concentration for both the HSB and LSB line (HSB: p = 1.70 × 10–5; LSB: p = 0.003) with no drug-impaired locomotion. Therefore, we selected the 0.75% ethanol for two weeks as the treatment regime for this study. Full model results are presented in Supplementary Table S1.
Figure 1.
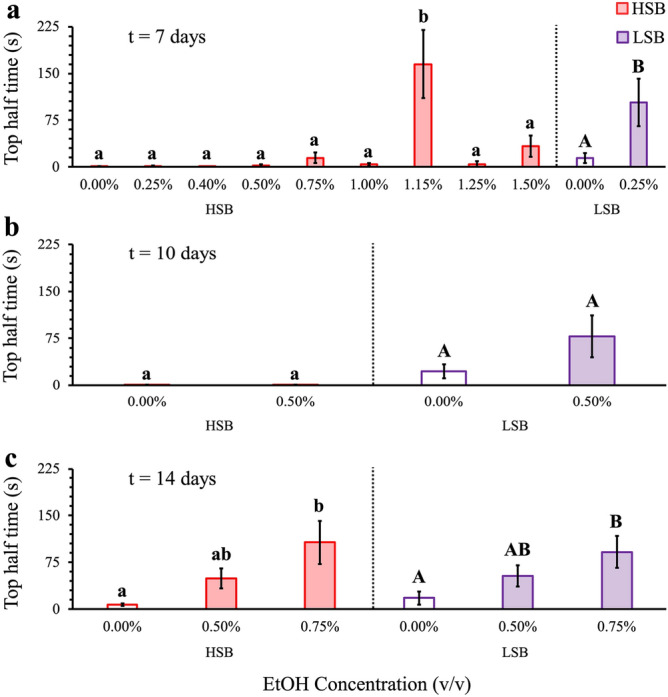
Dose response analysis of ethanol concentration on time spent in the top half of the tank during NTDT. Measured time spent in the top half of the tank after (a) 7, (b) 10, or (c) 14 days of treatment. Control groups are represented by unfilled in bars, while ethanol-treated groups are represented by filled bars. HSB and LSB are red and purple, respectively. Data shown are mean ± 1 SEM. Individual differences within the HSB line are indicated by lower case letters, while differences within the LSB line are indicated by upper case letters.
Ethanol-treatment increases exploration for both lines
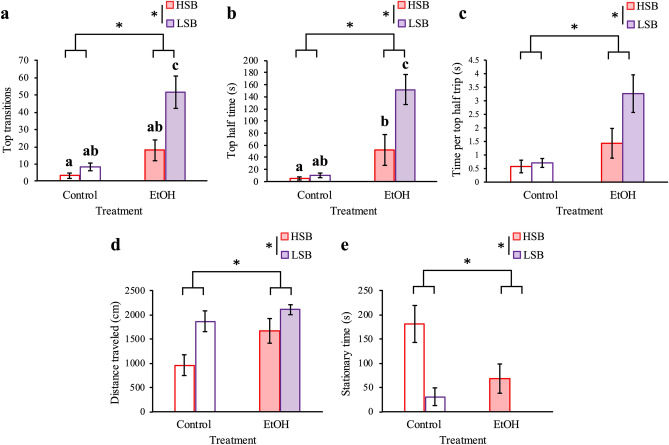
There were significant main effects of line (HSB, LSB) on number of top transitions [Wald χ2(1) = 12.579, p = 3.90 × 10–4], time spent in the top half of the tank [Wald χ2(1) = 10.215, p = 0.001], and time per trip to top half [Wald χ2(1) = 5.045, p = 0.025]. LSB fish transitioned to the top half of the tank (p = 3.90 × 10–4; Fig. 2a), spent more time in the top half of the tank (p = 0.001; Fig. 2b; Supplementary Fig. S1), and spent longer time per trip to the top half (p = 0.025; Fig. 2c) than HSB fish. There were also significant main effects of treatment (ethanol, control) on top transitions [Wald χ2(1) = 28.054, p = 1.18 × 10–7], time spent in the top half of the tank [Wald χ2(1) = 32.659, p = 1.10 × 10–8], and time per trip to top half [Wald χ2(1) = 15.227, p = 9.53 × 10–5]. Ethanol-treated fish transitioned to the top half of the tank (p = 1.18 × 10–7), spent significantly more time in the top half of the tank (p = 1.10 × 10–8), and spent longer time per trip to the top half of the tank (p = 9.53 × 10–5) than control fish. There was a significant line by treatment interaction effect for transitions to the top half of the tank [Wald χ2(1) = 6.788, p = 0.009] and time spent in the top half of the tank [Wald χ2(1) = 8.182, p = 0.004]. There was a trend for a line by treatment interaction effect for time per trip to top half [Wald χ2(1) = 3.784, p = 0.052]. Ethanol-treated LSB fish exhibited the most top transitions compared to the control HSB (p = 2.85 × 10–10), control LSB (p = 1.75 × 10–8), and ethanol-treated HSB fish (p = 2.12 × 10–5). This pattern was also found for time spent in the top half with ethanol-treated LSB exhibiting the most time spent in the top half of the tank compared to control HSB (p = 2.09 × 10–10), control LSB (p = 9.63 × 10–10), and ethanol-treated HSB fish (p = 2.84 × 10–5). The LSB ethanol-treated fish averaged more time per trip to the top half of the tank than the control HSB (p = 1.16 × 10–5), control LSB (p = 3.05 × 10–5), and ethanol-treated HSB fish (p = 0.004). Full model results are presented in Supplementary Table S2.
Figure 2.
Differentiated ethanol treatment effect on stress-related behaviors between lines with no effect on locomotion. We measured top transitions (a), time in top half of the tank (b), average time spent in top half per trip (c), distance traveled (d), and stationary time (e) for each treatment group. Control groups are represented by unfilled in bars, while ethanol-treated groups are represented by filled bars. HSB and LSB are red and purple, respectively. Data shown are mean ± 1 SEM. Significant line and treatment differences are indicated by an asterisk (p ≤ 0.05), while differences between groups are indicated by different lower-case letters. The number of subjects tested in each group are as follows: 17 HSB control, 17 LSB control, 15 HSB EtOH, 16 LSB EtOH.
No impaired locomotion from ethanol-treatment for both lines
There were significant line effects for total distance swam [Wald χ2(1) = 11.378, p = 0.001] and stationary time [Wald χ2(1) = 18.173, p = 2.02 × 10–5]. LSB fish swam a significantly farther distance (p = 0.001; Fig. 2d) and spent significantly less time stationary (p = 2.02 × 10–5; Fig. 2e) than HSB fish. We also found significant treatment effects for total distance swam [Wald χ2(1) = 5.729, p = 0.016] and stationary time [Wald χ2(1) = 7.831, p = 0.005]. Ethanol-treated fish traveled farther (p = 0.016) and spent less time stationary (p = 0.005) than control fish. There were no significant line by treatment interaction effects for total distance traveled [Wald χ2(1) = 1.391, p = 0.238] or stationary time [Wald χ2(1) = 2.639, p = 0.104]. Full model results are presented in Supplementary Table S2.
When examining changes in locomotion within a trial there was a significant effect of trial time for both lines on distanced traveled [HSB: Wald χ2(1) = 23.359, p = 2.88 × 10–4; LSB: Wald χ2(1) = 45.354, p = 1.23 × 10–8] and transitions to the top half. [HSB: Wald χ2(1) = 14.059, p = 0.015; LSB: Wald χ2(1) = 14.000, p = 0.016; Supplementary Fig. S2]. Transitions increased as the trial progressed, which suggests that habituation occurred to the testing chamber during the trial. There were no significant main effects of trial time on time in top half [HSB: Wald χ2(1) = 8.564, p = 0.128; LSB: Wald χ2(1) = 1.158, p = 0.949], time per trip to the top half [HSB: Wald χ2(1) = 4.469, p = 0.484; LSB: Wald χ2(1) = 5.112, p = 0.402], or stationary time [HSB: Wald χ2(1) = 2.675, p = 0.750; LSB: Wald χ2(1) = 7.083, p = 0.215] for either line (Supplementary Fig. S2). The only significant treatment by time interaction effects were seen on number of top transitions [Wald χ2(1) = 12.211, p = 0.032] and time per trip to top half [Wald χ2(1) = 13.785, p = 0.017] in the LSB line. Full model results are presented in Supplementary Table S3.
Ethanol-treatment increases expression of gabra1 and gabrg2
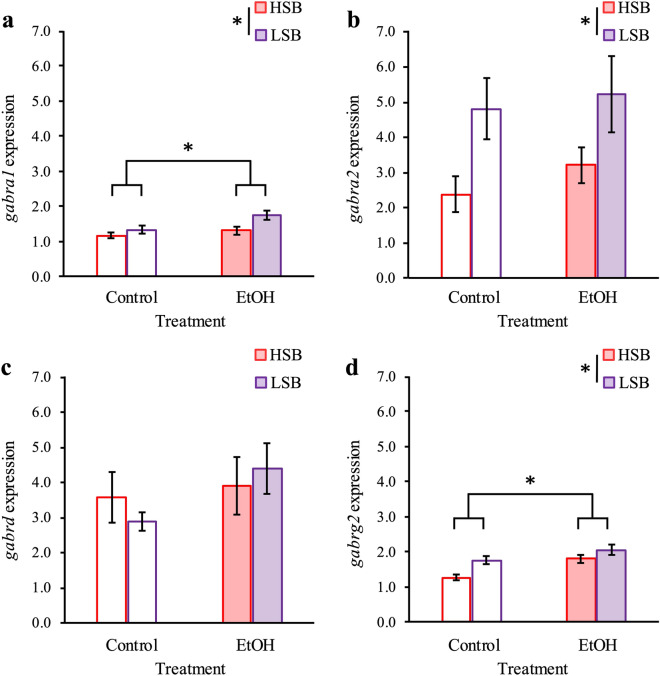
We found significant main effects of line on expression of gabra1 [Wald χ2(1) = 7.310, p = 0.007; Fig. 3a], gabra2 [Wald χ2(1) = 8.235, p = 0.004; Fig. 3b], and gabrg2 [Wald χ2(1) = 5.929, p = 0.015; Fig. 3d], but not gabrd [Wald χ2(1) = 0.023, p = 0.880; Fig. 3c]. The LSB fish showed higher expression of the gabra1 (p = 0.007), gabra2 (p = 0.004), and gabrg2 (p = 0.015) than the HSB fish. There were significant main effects of treatment on expression of gabra1 [Wald χ2(1) = 6.507, p = 0.011] and gabrg2 [Wald χ2(1) = 7.220, p = 0.007] but not gabra2 [Wald χ2(1) = 0.648, p = 0.421] or gabrd [Wald χ2(1) = 2.042, p = 0.153]. Ethanol-treated fish showed greater expression of gabra1 (p = 0.011) and gabrg2 (p = 0.007) than control fish. There were no significant line by treatment interaction effects for any of the four genes of interest [gabra1: Wald χ2(1) = 1.339, p = 0.247; gabra2: Wald χ2(1) = 0.073, p = 0.787; gabrd: Wald χ2(1) = 0.832, p = 0.362; gabrg2: Wald χ2(1) = 0.659, p = 0.417]. Full model results are presented in Supplementary Table S4.
Figure 3.
Effect of line and treatment on GABAA receptor subunits. Normalized expression of gabra1 (a), gabra2 (b), gabrd (c), and gabrg2 (d) for each treatment group following treatment. Control groups are represented by unfilled in bars, while ethanol-treated groups are represented by filled bars. HSB and LSB are red and purple, respectively. Data shown are mean ± 1 SEM. Significant differences are indicated by an asterisk (p ≤ 0.05). The number of subjects tested in each group are as follows: 17 HSB control, 18 LSB control, 17 HSB EtOH, 17 LSB EtOH.
Discussion
GABAAR agonists, such as ethanol, produce an anxiolytic response across many taxa14,17,22,23,29–31,66,67. Through the use of these stress-reducing compounds, we can investigate the role of the GABAergic system in facilitating the expression of a stress coping style. In this study, we assessed both the behavioral and molecular responses of ethanol treatment between proactive (LSB) and reactive (HSB) lines of zebrafish. We found that while chronic ethanol treatment decreased stress-related behaviors in both lines, ethanol treatment had a greater anxiolytic effect on the LSB line. The differences in stress-related behavior are linked to differential GABAAR subunit expression between the lines (α1-, α2-, and γ2-subunits) or in response to ethanol treatment (α1-, and γ2-subunits). The results suggest molecular differences in the GABAergic neurotransmitter system contribute to the variation in stress-related behaviors between the two stress coping styles.
The anxiolytic behavioral response to ethanol in zebrafish is well documented14,23,31,38,39, but the effect of an individual’s stress coping style on the response to GABAAR agonists has only been investigated recently55,56. We predicted that treatment with a GABAAR agonist would have a greater anxiolytic effect on both stress-related behaviors and GABAAR subunit expression in the reactive stress coping style than the proactive stress coping style. As expected, we found that both the LSB (proactive) and HSB (reactive) lines significantly increased their exploration and locomotion with chronic ethanol treatment. There are at least two alternative interpretations of the behavioral changes due to ethanol treatment: anxiolytic or anxiogenic and hyperactive effect. Increased distance traveled and number of transitions to the top half are predicted to occur with both stress-induced hyperactivity and stress reduction (e.g. more motivation to explore). When considering time duration in the top half, if an anxiolytic and hyperactive effect occurred, then we would have expected decreased time in the top and shorter time per trip in the top relative to controls. However, ethanol treated fish spent significantly more time in the top half and longer time in the top half per trip, which suggests an anxiolytic effect of ethanol treatment. These behavioral displays are also consistent with other studies that administered anxiolytic and stress-reducing pharmaceuticals14,30,31,39,68,69. Furthermore, we and others have shown that zebrafish that spend more time in the upper half of the water column also showed significantly lower cortisol levels10,45,70. Altogether our results in conjunction with prior studies suggest ethanol treatment resulted in an anxiolytic effect rather than a hyperactive and anxiogenic effect.
Surprisingly, the proactive individuals showed a greater anxiolytic response than the reactive individuals. To our knowledge, only a couple of other studies have accounted for stress coping style when examining the anxiolytic effects of ethanol in zebrafish55,56. In these studies, acute ethanol treatment (60 min) resulted in a greater anxiolytic effect (fish spent more time in an area of the tank furthest from conspecifics, swan faster, and traveled further from the bottom half of the tank) on reactive fish, while proactive fish increased their stress-related behaviors55,56. We speculate the opposing observations between our studies could be due to differences in treatment length (60 min vs. 2 weeks), social stress buffering (social vs. isolation), and assignment of stress coping style (behavioral screen vs. selectively bred lines). Regardless, ethanol is known to have an anxiolytic effect and the behavioral results from the prior and current studies suggest that an individual’s stress coping style can modulate the magnitude of the effect.
We found that the LSB line of zebrafish showed the greatest increase in transitions to, time spent in the top half of the tank, and average top half trip time during the NTDT compared to the HSB line (Fig. 2a–c). This line-specific response can be seen in other zebrafish studies and also in rodents5,6,10,55,56,68,69,71–74. Laboratory lines of zebrafish require a higher concentration of ethanol to match exploratory behavior of wild-caught lines, while wild-caught lines exhibit abolishment of shoaling behavior at higher concentrations of ethanol68,69,72. Rodents selectively bred to exhibit diverging novelty-seeking behaviors show differing levels of responsiveness to ethanol71,73,74. Maintaining laboratory and selectively bred lines of animals simultaneously results in line-specific genetic backgrounds. For example, the HSB and LSB zebrafish lines used here show distinct whole-brain transcriptome profiles13,63, and divergent novelty-seeking rodent lines differ in neuropeptide gene expression relating to the dopaminergic system71,74. This suggests an individual’s behavioral response can be influenced by its genetic profile and underlying expression of neurotransmitters. Altogether our results show that differences in molecular mechanisms can contribute to the alternative behavioral stress-response between stress coping styles.
It is possible that the higher expression of α1-, α2-, and γ2-subunits GABAAR subunits we observed in this study in the proactive (LSB) zebrafish facilitated a greater anxiolytic response to ethanol treatment than in the reactive (HSB) zebrafish (Fig. 3a, b, d). In rodents, removal of the α2-subunit results in the abolishment of the anxiolytic effect for both ethanol and other benzodiazepines75,76, suggesting this is a critical subunit needed for ethanol’s anxiolytic effect. We hypothesize that higher expression of these subunits in our proactive line may allow for greater sensitivity of GABAAR ligands leading to a greater anxiolytic response.
In addition to being differentially expressed between the two lines, expression of the α1-, and γ2-subunits increased as a result of ethanol treatment (Fig. 3a, d). These results are consistent with previous studies in rodents where α1-subunit increased expression with ethanol treatment34–37, suggesting that ethanol-induced modulation of this subunit may be a conserved response across taxa. Prior studies examining the change in the γ2-subunit expression to ethanol treatment show conflicting information77–79. While our results are consistent with studies showing lower expression of this particular subunit decreases stress-related behaviors, other studies have shown increased expression similarly leading to a reduction in stress-related behaviors. It has been hypothesized that the γ2-subunit increases the overall responsiveness of the GABA neurotransmitter system79,80. Our results are consistent with this hypothesis as the proactive line showed higher expression of the γ2-subunit and had a greater change in the anxiolytic behavioral response from a GABAAR agonist (ethanol). Interestingly, knockouts of either the α1- or γ2-subunits do not abolish ethanol’s anxiolytic effect. Both wild type and α1-subunit knockout rodents display an anxiolytic response to GABAAR agonists, but rodents with the knockout display a greater decrease in anxiety-related behaviors, such as time spent in the open and number of open arm entries in the elevated plus maze81–83. Results of previous studies assessing γ2-subunit knockouts on stress-related behaviors are inconsistent. Some studies found partial knockout of this receptor subtype decreases exploratory behavior in an open field test (i.e. increasing anxiety)77,78, while a more recent study found complete knockout of the subunit in dopaminergic neurons increases exploratory behavior79. While removal of the α1- or γ2-subunits alters behavior in the rodent animal model, the anxiolytic effect of GABAAR agonist is still present regardless of the presence in the GABAAR. This suggests that the α1- and γ2-subunits are sufficient but not necessary for the anxiolytic response and their increased expression in the current study may have facilitated the reduction of stress-related behavioral displays in both lines.
Of note, we did not observe any significant line by treatment interaction effects on expression of any of the examined GABAAR subunits. It is possible that by looking at whole-brain expression levels, we masked brain-region specific responses that may have shown interaction effects. Published data suggest that within broad brain divisions implicated in regulating stress (e.g. telencephalon, diencephalon), α1-, α2-, and γ2-subunits of GABAAR showed similar expression across individuals in the telencephalon and olfactory bulbs, but were more variable within the diencephalon84–86. While studies have demonstrated differences in GABAergic neurons and GABAAR neurons across broad divisions of the zebrafish brain84–86, to our knowledge no study has mapped and quantified GABAAR distribution at the resolution of individual brain nuclei in zebrafish. Future studies comparing GABAAR expression within the network of brain regions regulating stress and anxiety-like behavior between alternative stress coping styles in zebrafish are needed. As the GABAergic system can be differentially modulated depending on length (acute vs chronic) of ethanol exposure67,72,87, we also cannot rule out the possibility that our results may change with acute ethanol exposure. Another possibility is that other GABAAR subunits that were not examined in this study could influence stress-related behaviors between stress coping styles. In rodents ethanol treatment alters expression of α4-, α5-, and γ1-subunits34,88,89. Finally, it is possible that the GABAergic system does not play a significant role in the differentiated anxiolytic behavioral effects of chronic ethanol exposure between stress coping styles in zebrafish. Rather, the anxiolytic effects could be mediated by another neurotransmitter system such as the dopaminergic or serotoninergic system. Prior studies in fish and rodents have documented that administration of ethanol and other anxiolytic compounds alter several neurotransmitter systems in addition to the target system60,90–95. Of note, a prior study showed that the proactive (LSB) line showed higher baseline expression of the dopamine receptor D2 compared to the reactive (HSB) line13. Given this receptor’s role in ethanol-induced activation of the mesolimbic dopaminergic reward pathway of the brain and drug-seeking and novelty exploration behaviors96–98, we speculate that the differences in the magnitude of the anxiolytic effects of chronic ethanol on behavior between the two stress coping style lines involve the dopaminergic system. Future studies are needed to assess the extent of ethanol effects on neurotransmitter systems beyond the GABAA system between the two stress coping styles.
Conclusions
In this study, we showed significant main effects of line on anxiety-related behaviors and GABAAR subunit expressions where individuals with the proactive (LSB line) stress coping style had lower anxiety-related behaviors and higher expression of the α1, α2, and γ2-subunits relative to reactive (HSB line) individuals. This demonstrates that variation in behavioral responses to a novelty stressor may be explained by differences in the GABAergic system (e.g. GABAAR subunit expression) between the two stress coping styles. Intriguingly, we observed a significant line by ethanol treatment interaction effects on stress and anxiety-related behaviors. Chronic ethanol treatment had a surprisingly greater anxiolytic effect on proactive individuals, which suggests that ethanol alters the underlying neuromolecular mechanisms in a coping style-specific manner. However, the lack of an interaction effect between line and treatment on any of the four measured GABAAR subunits leads us to speculate that the differences in the magnitude of effect between the lines induced by chronic ethanol treatment may be mediated by other GABAAR subunits or a neurotransmitter system other than the GABAergic system. More broadly, this study shows that differences in stress and anxiety-related behaviors between the proactive and reactive stress coping styles are due in part to differences in the GABAergic system, but any coping-style specific anxiolytic behavioral effects of chronic ethanol exposure likely involve other neurotransmitter systems.
Methods
Subjects
In this study, we used the high-stationary behavior (HSB; reactive line) and low-stationary behavior (LSB; proactive line) lines of zebrafish (Danio rerio). These two lines exhibit differences in stress-related behaviors across multiple behavioral assays, learning and memory, glucocorticoid responses, neurotranscriptome profiles, and morphology consistent with the reactive and proactive stress coping styles5,6,10,11,59,60,62,63. Therefore, we consider any fish from the HSB or LSB lines to have the reactive or proactive stress coping style, respectively. Lines were generated starting from a wild-caught population from Gaighata in West Bengal, India and were maintained through a bidirectional selective breeding paradigm on behavioral stress response to a novelty stressor5. The subjects used in this study underwent 11 generations of this breeding paradigm. Both lines were sexually mature (12–15 months post-fertilization) when testing began. Prior to testing, fish were housed in 40-L mixed-sex tanks all on the same custom-built recirculating system with solid filtration. Fish were kept at a water temperature of 27 °C, on a 14:10 L/D cycle and fed twice daily with Tetramin Tropical Flakes (Tetra, USA). All procedures and experiments were approved by the Institutional Animal Care and Use Committee of the University of Nebraska at Omaha/University of Nebraska Medical Center (17-070-09-FC). All methods were performed in accordance with the relevant guidelines and regulations.
Pharmacological manipulation
Using a modified protocol for chronic ethanol administration in zebrafish31, groups of six fish were housed in a 3-L trapezoidal tank (15.2 height × 27.9 top × 22.5 bottom × 11.4 cm width; Pentair Aquatic Ecosystems) throughout the treatment period. The tank contained either 2-L of 0.75% ethanol (v/v; Sigma-Aldrich) or 2-L of system water as a control over the span of 14 days. Every two days we replaced the entire water in each tank with fresh ethanol or system water. At the end of 14 days, a group of fish was used for either behavioral testing or for quantification of whole-brain GABAAR subunit mRNA expression. For behavioral testing we used three groups of six fish for each treatment group, where fish were randomly selected individuals from each of the HSB and LSB lines (36 total fish; n = 18 for each treatment group). We used a separate set of tanks to treat a different set of 36 individuals from each line (n = 18 for each treatment group) for quantification of GABAAR subunit expression. Some fish died during the 14-day treatment period resulting in final sample sizes of 32 individuals from the HSB (n = 15 treated, 17 control; female = 13, male = 19) line and 33 from the LSB (n = 16 treated, 17 control; female = 14, male = 19) that were behaviorally tested. A total of 34 individuals from the HSB (n = 17 treated, 17 control; female = 15, male = 19) line and 35 from the LSB (n = 17 treated, 18 control; female = 18, male = 17) were used for GABAAR subunit quantification. In total, ten fish were lost during the treatment period, and we observed no consistent pattern in the timing of mortality. Deceased fish were removed within one day.
To identify a biologically relevant ethanol dose, we conducted a pilot dose–response study. We chronically administered ethanol of varying concentrations and durations to both lines followed by a behavioral stress assay (Novel Tank Diving Test) to measure stress and anxiety-related behaviors. Ethanol treatment began at 0.25% v/v over a period of seven days. We increased both the concentration and duration until an anxiolytic effect was observed in both lines of zebrafish without drug-impaired locomotion (i.e. significant change in depth preference without a significant difference or decrease in distance traveled and stationary time relative to control fish). Due to drug-impaired locomotion at higher ethanol concentrations in the 7-day group and lack of anxiolytic effects at lower concentrations, 0.5% ethanol was the chosen starting point for both the 10- and 14-day groups. Similarly, the 0.5% was the only tested concentration for the 10-day group due to lack of anxiolytic effect. We used total distance traveled and total stationary time during the trials as proxies for locomotion to ensure the chosen concentration of ethanol was not impairing the fish’s ability to swim.
Behavioral testing
Following the 14th day of treatment, fish designated for behavioral testing were exposed to a novelty stressor by placing them into the Novel Tank Diving Test (NTDT) assay following established procedures5,10,31,60,99,100. In brief, fish were netted from their treatment tanks and individually placed in a clear 3-L trapezoidal tank (15.2 height × 27.9 top × 22.5 bottom × 11.4 cm width; Pentair Aquatic Ecosystems) filled with 2-L of system water. We video-recorded the fish for six minutes and quantified behaviors using an automated tracking software (Noldus Ethovision XT Version 14, Wageningen, Netherlands) as previously described5,6,10,13. In brief, we used the software to virtually partition the tank into top and bottom halves to measure the number of transitions to the top portion of the tank, time spent in the top portion of the tank (s), total distance traveled (cm), and stationary time (s). We used the entirety of the 6-min trial for analysis. The subject was considered stationary if it was moving less than 0.5 cm/s. Reduced transitions to and time spent in the top half of the tank are indicators of heightened stress and anxiety5,31,99. Stationary time and distance traveled were used as proxies for locomotor activity to assess whether or not ethanol treatment impaired general locomotor activity. Testing occurred between 2–11 h after light onset with control and ethanol-treated group testing being randomly distributed. We digitally measured standard length of each fish following completion of the trial.
Quantification of GABAAR subunit expression
We quantified whole-brain expression of four genes that encode for GABAAR subunits (gabra1, gabra2, gadrd, and gabrg2 with no known paralogs; Supplementary Table S5), and one endogenous reference gene (ef1a) using quantitative reverse transcriptase PCR (qRT-PCR) following established protocols13,60,63. In brief, whole brains were homogenized with 50–100 µL of zirconium oxide beads (Bullet Blender, Next Advanced) in Tri Reagent (Sigma-Aldrich). Then, we extracted RNA and removed genomic DNA using column filtration (PureLink RNA Mini Kit, Ambion). We subsequently synthesized cDNA using both random hexamers and oligo(dT)20 primers (SuperScript IV First-Strand Synthesis System for qRT-PCR (Invitrogen). Finally, we purified the cDNA using Amicon Ultracentrifugal filters (Millipore). We carried out all protocols according to each manufacturers’ protocol.
We ran the qRT-PCR on QuantStudio 7 Flex Real-Time PCR System (Applied Biosystems) using SYBR green detection chemistry (PowerUp SYBR Green Master Mix, Applied Biosystems). For gabrg2 and ef1a, we used primer sequences from previously published studies84,101. For the remaining genes, we designed primers using Primer-Blast102 with chosen primers either spanning exon-exon junctions or with the amplicon spanning exons where the intron region was over one kilobase (Supplementary Table S5). Primer concentrations were 5 pmol for all genes. Reaction parameters for all genes were as follows: 2 min at 50 °C, 2 min at 95 °C, followed by 40 cycles at 95 °C for 15 s then 60 °C for 1 min. We ran each sample in triplicate. Primers for all genes showed high specificity as evidenced by (1) PCR reaction resulting in a single band on gel electrophoresis, (2) sanger sequencing of PCR amplicon aligned with target gene after using NCBI BLAST and (3) observing a single peak on melt curve analysis following qRT-PCR. We quantified expression using the relative standard curve method and normalized expression to an endogenous reference gene (ef1a). ef1a expression is stable across sex, tissue types, age, and chemical treatment in zebrafish101. For each fish we calculated normalized expression by dividing quantity of gene interest by quantity of ef1a. We also checked the validity of ef1a as an endogenous reference by comparing its expression between ethanol-treated and control individuals. After normalizing ef1a expression by total cDNA input, there was no significant difference in ef1a expression between treated and controlled fish [t(55) = 1.297, p = 0.393].
Statistical analysis
Six of our nine endpoint measurements were not normally distributed. Five of those six endpoints were still not normally distributed after a log transformation. Thus, we used a generalized linear model (GLZM) applying the identity link function in SPSS (Version 26) to investigate changes in behaviors and normalized gene expressions. The significance value was set at α = 0.05. Line (HSB, LSB), sex (male, female) and treatment group (0.75% ethanol, control) were used as between-subject variables. All interaction terms were included in the GLZM model. As the relationship between body size and locomotion is well documented59,103–105, we included standard length as a covariate. Since we did not find a significant main effect of sex on behavior [top transitions: Wald χ2(1) = 2.385, p = 0.123; top time: Wald χ2(1) = 0.852, p = 0.356; average top trip: Wald χ2(1) = 0.179, p = 0.672; distance: Wald χ2(1) = 0.682, p = 0.409; and stationary time: Wald χ2(1) = 0.092, p = 0.762] or gene expression [gabra1: Wald χ2(1) = 0.036, p = 0.850; gabra2: Wald χ2(1) = 0.382, p = 0.536; gadrd: Wald χ2(1) = 1.942, p = 0.163; gabrg2: Wald χ2(1) = 1.426, p = 0.232], we just used line and treatment group as the only between-subject variables. Since there is only one model for each endpoint, use of goodness of fit criteria to select a model is not applicable. To assess the direction of effects, we investigated the simple main effects within each GLZM. We applied a Benjamini–Hochberg correction to all simple main effect investigations to account for multiple comparisons106. To analyze changes in behavior across the trial, we divided the 6-min trial into 1-min bins and ran a repeated-measures generalized estimating equation (GEE) for each behavior with treatment and time (6, 1-min blocks) as factors. We ran separate GEEs for each line and applied an identity link function.
Supplementary information
Acknowledgements
We are grateful to D. Revers, S. Roundtree, A. Park, and N. Mohamed for zebrafish husbandry. We thank M. Baker, K. Cullen, R. Patterson, and other members of the Wong lab for helpful discussions. This study was supported by the UNO Fund for Undergraduate Scholarly Experiences to A.C.G. along with the National Institutes of Health (R15MH113074), Nebraska EPSCoR First Award (OIA-1557417), Nebraska Research Initiative, and UNO start-up funds to RYW.
Author contributions
A.C.G. and R.Y.W. conceived the study, conducted statistical analyses, and wrote the manuscript. A.C.G. conducted the behavioral testing, brain extraction, gene expression quantification, and data collection.
Data availability
All data generated or analyzed during this study are included in this published article and its Supplementary Information files.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Alexander C. Goodman, Email: alexander.c.goodman@gmail.com
Ryan Y. Wong, Email: rwong@unomaha.edu
Supplementary information
is available for this paper at 10.1038/s41598-020-69980-2.
References
- 1.Demin KA, et al. The role of intraspecies variation in fish neurobehavioral and neuropharmacological phenotypes in aquatic models. Aquat. Toxicol. 2019;210:44–55. doi: 10.1016/j.aquatox.2019.02.015. [DOI] [PubMed] [Google Scholar]
- 2.Koolhaas JM, de Boer SF, Coppens CM, Buwalda B. Neuroendocrinology of coping styles: towards understanding the biology of individual variation. Front. Neuroendocrinol. 2010;31:307–321. doi: 10.1016/j.yfrne.2010.04.001. [DOI] [PubMed] [Google Scholar]
- 3.Øverli Ø, et al. Evolutionary background for stress-coping styles: relationships between physiological, behavioral, and cognitive traits in non-mammalian vertebrates. Neurosci. Biobehav. Rev. 2007;31:396–412. doi: 10.1016/j.neubiorev.2006.10.006. [DOI] [PubMed] [Google Scholar]
- 4.Slater PB. Individual differences in animal behavior. In: Bateson PPG, Klopfer PH, editors. Perspectives in Ethology. New York: Springer; 1981. pp. 35–49. [Google Scholar]
- 5.Wong RY, et al. Comparing behavioral responses across multiple assays of stress and anxiety in zebrafish (Danio rerio) Behaviour. 2012;149:1205–1240. [Google Scholar]
- 6.Baker MR, Goodman AC, Santo JB, Wong RY. Repeatability and reliability of exploratory behavior in proactive and reactive zebrafish, Danio rerio. Sci. Rep. 2018;8:12114. doi: 10.1038/s41598-018-30630-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Baker MR, Hofmann HA, Wong RY. Neurogenomics of behavioural plasticity in socioecological contexts. eLS. 2017 doi: 10.1002/9780470015902.a0026839. [DOI] [Google Scholar]
- 8.Schjolden J, Backstrom T, Pulman KG, Pottinger TG, Winberg S. Divergence in behavioural responses to stress in two strains of rainbow trout (Oncorhynchus mykiss) with contrasting stress responsiveness. Horm. Behav. 2005;48:537–544. doi: 10.1016/j.yhbeh.2005.04.008. [DOI] [PubMed] [Google Scholar]
- 9.Evans MR, Roberts ML, Buchanan KL, Goldsmith AR. Heritability of corticosterone response and changes in life history traits during selection in the zebra finch. J. Evol. Biol. 2006;19:343–352. doi: 10.1111/j.1420-9101.2005.01034.x. [DOI] [PubMed] [Google Scholar]
- 10.Wong RY, French J, Russ JB. Differences in stress reactivity between zebrafish with alternative stress coping styles. R. Soc. Open Sci. 2019 doi: 10.1098/rsos.181797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Johnson ZV, et al. Exploratory behaviour is associated with microhabitat and evolutionary radiation in Lake Malawi cichlids. Anim. Behav. 2020;160:121–134. [Google Scholar]
- 12.Koolhaas JM, de Boer SF, Buwalda B, van Reenen K. Individual variation in coping with stress: a multidimensional approach of ultimate and proximate mechanisms. Brain. Behav. Evol. 2007;70:218–226. doi: 10.1159/000105485. [DOI] [PubMed] [Google Scholar]
- 13.Wong RY, Lamm MS, Godwin J. Characterizing the neurotranscriptomic states in alternative stress coping styles. BMC Genom. 2015;16:1–11. doi: 10.1186/s12864-015-1626-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Stewart A, et al. Pharmacological modulation of anxiety-like phenotypes in adult zebrafish behavioral models. Prog. Neuro-Psychopharmacol. Biol. Psych. 2011;35:1421–1431. doi: 10.1016/j.pnpbp.2010.11.035. [DOI] [PubMed] [Google Scholar]
- 15.Kalueff AV, et al. Zebrafish neurobehavioral phenomics for aquatic neuropharmacology and toxicology research. Aquat. Toxicol. 2016;170:297–309. doi: 10.1016/j.aquatox.2015.08.007. [DOI] [PubMed] [Google Scholar]
- 16.Stewart A, et al. Modeling anxiety using adult zebrafish: a conceptual review. Neuropharmacology. 2012;62:135–143. doi: 10.1016/j.neuropharm.2011.07.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Durant C, Christmas D, Nutt D. The pharmacology of anxiety. In: Stein MB, Steckler T, editors. Behavioral Neurobiology of Anxiety and Its Treatment. Berlin: Springer; 2009. [Google Scholar]
- 18.American Psychiatric Association. Diagnostic and statistical manual of mental disorders. (2013).
- 19.Craske MG, Stein MB. Anxiety. Lancet. 2016;388:3048–3059. doi: 10.1016/S0140-6736(16)30381-6. [DOI] [PubMed] [Google Scholar]
- 20.Lydiard RB. The role of GABA in anxiety disorders. J. Clin. Psych. 2003;64:21–27. [PubMed] [Google Scholar]
- 21.Nemeroff CB. The role of GABA in the pathophysiology and treatment of anxiety disorders. Psychopharmacol. Bull. 2003;37:133–146. [PubMed] [Google Scholar]
- 22.Koen N, Stein DJ. Pharmacotherapy of anxiety disorders: a critical review. Dialogues Clin. Neurosci. 2011;13:423–437. doi: 10.31887/DCNS.2011.13.4/nkoen. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Magno LDP, Fontes A, Gonçalves BMN, Gouveia A. Pharmacological study of the light/dark preference test in zebrafish (Danio rerio): waterborne administration. Pharmacol. Biochem. Behav. 2015;135:169–176. doi: 10.1016/j.pbb.2015.05.014. [DOI] [PubMed] [Google Scholar]
- 24.Blanchard RJ, Magee L, Veniegas R, Blanchard DC. Alcohol and anxiety: ethopharmacological approaches. Prog. Neuro-Psychopharmacol. Biol. Psych. 1993;17:171–182. doi: 10.1016/0278-5846(93)90041-p. [DOI] [PubMed] [Google Scholar]
- 25.Kliethermes CL, Cronise K, Crabbe JC. Anxiety-like behavior in mice in two apparatuses during withdrawal from chronic ethanol vapor inhalation. Alcohol. Clin. Exp. Res. 2004;28:1012–1019. doi: 10.1097/01.alc.0000131976.40428.8f. [DOI] [PubMed] [Google Scholar]
- 26.Mueller T, Vernier P, Wullimann MF. The adult central nervous cholinergic system of a neurogenetic model animal, the zebrafish Danio rerio. Brain Res. 2004;1011:156–169. doi: 10.1016/j.brainres.2004.02.073. [DOI] [PubMed] [Google Scholar]
- 27.Kliethermes CL. Anxiety-like behaviors following chronic ethanol exposure. Neurosci. Biobehav. Rev. 2005;28:837–850. doi: 10.1016/j.neubiorev.2004.11.001. [DOI] [PubMed] [Google Scholar]
- 28.Panula P, et al. Modulatory neurotransmitter systems and behavior: towards zebrafish models of neurodegenerative diseases. Zebrafish. 2006;3:235–247. doi: 10.1089/zeb.2006.3.235. [DOI] [PubMed] [Google Scholar]
- 29.Kalueff AV, Nutt DJ. Role of GABA in anxiety and depression. Depress. Anxiety. 2007;24:495–517. doi: 10.1002/da.20262. [DOI] [PubMed] [Google Scholar]
- 30.Bencan Z, Sledge D, Levin ED. Buspirone, chlordiazepoxide and diazepam effects in a zebrafish model of anxiety. Pharmacol. Biochem. Behav. 2009;94:75–80. doi: 10.1016/j.pbb.2009.07.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Egan RJ, et al. Understanding behavioral and physiological phenotypes of stress and anxiety in zebrafish. Behav. Brain Res. 2009;205:38–44. doi: 10.1016/j.bbr.2009.06.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Rudolph U, Crestani F, Möhler H. GABAA receptor subtypes: dissecting their pharmacological functions. Trends Pharmacol. Sci. 2001;22:188–194. doi: 10.1016/s0165-6147(00)01646-1. [DOI] [PubMed] [Google Scholar]
- 33.von Blankenfeld G, et al. Differential benzodiazepine pharmacology of mammalian recombinant GABAA receptors. Neurosci. Lett. 1990;115:269–273. doi: 10.1016/0304-3940(90)90467-n. [DOI] [PubMed] [Google Scholar]
- 34.Devaud LL, Fritschy J-M, Sieghart W, Morrow AL. Bidirectional alterations of GABAA receptor subunit peptide levels in rat cortex during chronic ethanol consumption and withdrawal. J. Neurochem. 2002;69:126–130. doi: 10.1046/j.1471-4159.1997.69010126.x. [DOI] [PubMed] [Google Scholar]
- 35.Wafford KA. GABAA receptor subtypes: any clues to the mechanism of benzodiazepine dependence? Curr. Opin. Pharmacol. 2005;5:47–52. doi: 10.1016/j.coph.2004.08.006. [DOI] [PubMed] [Google Scholar]
- 36.Francesca PF, et al. Neurosteroids, GABAA receptors, and ethanol dependence. Psychopharmacology. 2006;186:267–280. doi: 10.1007/s00213-005-0126-0. [DOI] [PubMed] [Google Scholar]
- 37.Rudolph U, Knoflach F. Beyond classical benzodiazepines: novel therapeutic potential of GABAA receptor subtypes. Nat. Rev. Drug Discov. 2011;10:685–697. doi: 10.1038/nrd3502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Blaser R, Gerlai R. Behavioral phenotyping in zebrafish: comparison of three behavioral quantification methods. Behav. Res. Methods. 2006;38:456–469. doi: 10.3758/bf03192800. [DOI] [PubMed] [Google Scholar]
- 39.Sackerman J, et al. Zebrafish behavior in novel environments: effects of acute exposure to anxiolytic compounds and choice of Danio rerio line. Int. J. Comp. Psychol. 2010;23:43–61. [PMC free article] [PubMed] [Google Scholar]
- 40.Gerlai R. Using zebrafish to unravel the genetics of complex brain disorders. In: Cryan JF, Reif A, editors. Behavioral Neurogenetics. Berlin: Springer; 2011. pp. 3–24. [DOI] [PubMed] [Google Scholar]
- 41.Stewart AM, Braubach O, Spitsbergen J, Gerlai R, Kalueff AV. Zebrafish models for translational neuroscience research: from tank to bedside. Trends Neurosci. 2014;37:264–278. doi: 10.1016/j.tins.2014.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Grunwald DJ, Eisen JS. Headwaters of the zebrafish—emergence of a new model vertebrate. Nat. Rev. Genet. 2002;3:717–724. doi: 10.1038/nrg892. [DOI] [PubMed] [Google Scholar]
- 43.Key B, Devine CA. Zebrafish as an experimental model: strategies for developmental and molecular neurobiology studies. Methods Cell Sci. 2003;25:1–6. doi: 10.1023/B:MICS.0000006849.98007.03. [DOI] [PubMed] [Google Scholar]
- 44.Rinkwitz S, Mourrain P, Becker TS. Zebrafish: An integrative system for neurogenomics and neurosciences. Prog. Neurobiol. 2011;93:231–243. doi: 10.1016/j.pneurobio.2010.11.003. [DOI] [PubMed] [Google Scholar]
- 45.Cachat J, et al. Measuring behavioral and endocrine responses to novelty stress in adult zebrafish. Nat. Protoc. 2010;5:1786–1799. doi: 10.1038/nprot.2010.140. [DOI] [PubMed] [Google Scholar]
- 46.Cachat JM, et al. Modeling stress and anxiety in zebrafish. In: Kalueff AV, Cachat JM, et al., editors. Zebrafish Models in Neurobehavioral Research. Totowa: Humana Press; 2011. pp. 73–88. [Google Scholar]
- 47.Ibrahim M, et al. Anxiolytic effects of diphenyl diselenide on adult zebrafish in a novelty paradigm. Prog. Neuro-Psychopharmacol. Biol. Psych. 2014;54:187–194. doi: 10.1016/j.pnpbp.2014.06.002. [DOI] [PubMed] [Google Scholar]
- 48.Rosemberg DB, et al. Behavioral effects of taurine pretreatment in zebrafish acutely exposed to ethanol. Neuropharmacology. 2012;63:613–623. doi: 10.1016/j.neuropharm.2012.05.009. [DOI] [PubMed] [Google Scholar]
- 49.Levin ED, Bencan Z, Cerutti DT. Anxiolytic effects of nicotine in zebrafish. Physiol. Behav. 2007;90:54–58. doi: 10.1016/j.physbeh.2006.08.026. [DOI] [PubMed] [Google Scholar]
- 50.Maximino C, et al. A comparison of the light/dark and novel tank tests in zebrafish. Behaviour. 2012;149:1099–1123. [Google Scholar]
- 51.Wong K, et al. Analyzing habituation responses to novelty in zebrafish (Danio rerio) Behav. Brain Res. 2010;208:450–457. doi: 10.1016/j.bbr.2009.12.023. [DOI] [PubMed] [Google Scholar]
- 52.Tran S, Gerlai R. The novel tank test: handling stress and the context specific psychopharmacology of anxiety. Curr. Psychopharmacol. 2016;5:169–179. [Google Scholar]
- 53.Tran S, Gerlai R. Time-course of behavioural changes induced by ethanol in zebrafish (Danio rerio) Behav. Brain Res. 2013;252:204–213. doi: 10.1016/j.bbr.2013.05.065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Gebauer DL, et al. Effects of anxiolytics in zebrafish: similarities and differences between benzodiazepines, buspirone and ethanol. Pharmacol. Biochem. Behav. 2011;99:480–486. doi: 10.1016/j.pbb.2011.04.021. [DOI] [PubMed] [Google Scholar]
- 55.Araujo-Silva H, Pinheiro-da-Silva J, Silva PF, Luchiari AC. Individual differences in response to alcohol exposure in zebrafish (Danio rerio) PLoS ONE. 2018;13:e0198856. doi: 10.1371/journal.pone.0198856. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Araujo-Silva H, Leite-Ferreira ME, Luchiari AC. Behavioral screening of alcohol effects and individual differences in zebrafish (Danio rerio) Alcohol Alcohol. 2020 doi: 10.1093/alcalc/agaa046. [DOI] [PubMed] [Google Scholar]
- 57.Levin ED. Zebrafish assessment of cognitive improvement and anxiolysis: filling the gap between in vitro and rodent models for drug development. Rev. Neurosci. 2011;22:75–84. doi: 10.1515/RNS.2011.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Steenbergen PJ, Richardson MK, Champagne DL. The use of the zebrafish model in stress research. Prog. Neuro-Psychopharmacol. Biol. Psych. 2011;35:1432–1451. doi: 10.1016/j.pnpbp.2010.10.010. [DOI] [PubMed] [Google Scholar]
- 59.Kern EMA, et al. Correlated evolution of personality, morphology and performance HHS Public Access. Anim Behav. 2016;117:79–86. doi: 10.1016/j.anbehav.2016.04.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Wong RY, Oxendine SE, Godwin J. Behavioral and neurogenomic transcriptome changes in wild-derived zebrafish with fluoxetine treatment. BMC Genom. 2013;14:348. doi: 10.1186/1471-2164-14-348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Wong RY, McLeod MM, Godwin J. Limited sex-biased neural gene expression patterns across strains in zebrafish (Danio rerio) BMC Genom. 2014;15:905. doi: 10.1186/1471-2164-15-905. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Baker MR, Wong RY. Contextual fear learning and memory differ between stress coping styles in zebrafish. Sci. Rep. 2019;9:9935. doi: 10.1038/s41598-019-46319-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wong RY, Godwin J. Neurotranscriptome profiles of multiple zebrafish strains. Genom. Data. 2015;5:206–209. doi: 10.1016/j.gdata.2015.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Thörnqvist P-O, McCarrick S, Ericsson M, Roman E, Winberg S. Bold zebrafish (Danio rerio) express higher levels of delta opioid and dopamine D2 receptors in the brain compared to shy fish. Behav. Brain Res. 2019;359:927–934. doi: 10.1016/j.bbr.2018.06.017. [DOI] [PubMed] [Google Scholar]
- 65.Horzmann K, Freeman J. Zebrafish get connected: investigating neurotransmission targets and alterations in chemical toxicity. Toxics. 2016;4:19. doi: 10.3390/toxics4030019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Prut L, Belzung C. The open field as a paradigm to measure the effects of drugs on anxiety-like behaviors: a review. Eur. J. Pharmacol. 2003;463:3–33. doi: 10.1016/s0014-2999(03)01272-x. [DOI] [PubMed] [Google Scholar]
- 67.Förstera B, Castro PA, Moraga-Cid G, Aguayo LG. Potentiation of gamma aminobutyric acid receptors (GABAAR) by ethanol: how are inhibitory receptors affected? Front. Cell. Neurosci. 2016;10:114. doi: 10.3389/fncel.2016.00114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Gerlai R, Ahmad F, Prajapati S. Differences in acute alcohol-induced behavioral responses among zebrafish populations. Alcohol. Clin. Exp. Res. 2008;32:1763–1773. doi: 10.1111/j.1530-0277.2008.00761.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Dlugos CA, Rabin RA. Ethanol effects on three strains of zebrafish: model system for genetic investigations. Pharmacol. Biochem. Behav. 2003;74:471–480. doi: 10.1016/s0091-3057(02)01026-2. [DOI] [PubMed] [Google Scholar]
- 70.Pittman J, Hylton A. Behavioral, endocrine, and neuronal alterations in zebrafish (Danio rerio) following sub-chronic coadministration of fluoxetine and ketamine. Pharmacol. Biochem. Behav. 2015;139:158–162. doi: 10.1016/j.pbb.2015.08.014. [DOI] [PubMed] [Google Scholar]
- 71.Moreno M, et al. Impulsivity characterization in the roman high- and low-avoidance rat strains: behavioral and neurochemical differences. Neuropsychopharmacology. 2010;35:1198–1208. doi: 10.1038/npp.2009.224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Gerlai R, Chatterjee D, Pereira T, Sawashima T, Krishnannair R. Acute and chronic alcohol dose: population differences in behavior and neurochemistry of zebrafish. Genes Brain Behav. 2009;8:586–599. doi: 10.1111/j.1601-183X.2009.00488.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Driscoll P, et al. Genetic selection and differential stress responses the roman lines/strains of rats. Ann. N. Y. Acad. Sci. 1998;851:501–510. doi: 10.1111/j.1749-6632.1998.tb09029.x. [DOI] [PubMed] [Google Scholar]
- 74.Guitart-Masip M, et al. Reduced ethanol response in the alcohol-preferring RHA rats and neuropeptide mRNAs in relevant structures. Eur. J. Neurosci. 2006;23:531–540. doi: 10.1111/j.1460-9568.2005.04556.x. [DOI] [PubMed] [Google Scholar]
- 75.Löw K, et al. Molecular and neuronal substrate for the selective attenuation of anxiety. Science. 2000;290:131–134. doi: 10.1126/science.290.5489.131. [DOI] [PubMed] [Google Scholar]
- 76.Skórzewska A, et al. The effect of chronic administration of corticosterone on anxiety- and depression-like behavior and the expression of GABA-A receptor alpha-2 subunits in brain structures of low- and high-anxiety rats. Horm. Behav. 2014;65:6–13. doi: 10.1016/j.yhbeh.2013.10.011. [DOI] [PubMed] [Google Scholar]
- 77.Cagetti E, Liang J, Spigelman I, Olsen RW. Withdrawal from chronic intermittent ethanol treatment changes subunit composition, reduces synaptic function, and decreases behavioral responses to positive allosteric modulators of GABA A receptors. Mol. Pharmacol. 2003;63:53–64. doi: 10.1124/mol.63.1.53. [DOI] [PubMed] [Google Scholar]
- 78.Chandra D, Korpi ER, Miralles CP, De Blas AL, Homanics GE. GABA A receptor γ2 subunit knockdown mice have enhanced anxiety-like behavior but unaltered hypnotic response to benzodiazepines. BMC Neurosci. 2005;6:30. doi: 10.1186/1471-2202-6-30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Stojakovic, A. et al. Several behavioral traits relevant for alcoholism are controlled by ɣ2 subunit containing GABA A receptors on dopamine neurons in mice (2018). [DOI] [PMC free article] [PubMed]
- 80.Schweizer C, et al. The γ2 subunit of GABAA receptors is required for maintenance of receptors at mature synapses. Mol. Cell. Neurosci. 2003;24:442–450. doi: 10.1016/s1044-7431(03)00202-1. [DOI] [PubMed] [Google Scholar]
- 81.Kralic JE, et al. GABAA receptor alpha-1 subunit deletion alters receptor subtype assembly, pharmacological and behavioral responses to benzodiazepines and zolpidem. Neuropharmacology. 2002;43:685–694. doi: 10.1016/s0028-3908(02)00174-0. [DOI] [PubMed] [Google Scholar]
- 82.Kralic K, et al. Deletion of GABAA receptor α1 subunit-containing receptors alters responses to ethanol and other anesthetics. J. Pharmacol. Exp. Ther. 2003;305:600–607. doi: 10.1124/jpet.102.048124. [DOI] [PubMed] [Google Scholar]
- 83.Blednov YA, et al. GABAA receptor α1 and β2 subunit null mutant mice: behavioral responses to ethanol. J. Pharmacol. Exp. Ther. 2003;305:854–863. doi: 10.1124/jpet.103.049478. [DOI] [PubMed] [Google Scholar]
- 84.Cocco A, et al. Characterization of the γ-aminobutyric acid signaling system in the zebrafish (Danio rerio Hamilton) central nervous system by reverse transcription-quantitative polymerase chain reaction. Neuroscience. 2017;343:300–321. doi: 10.1016/j.neuroscience.2016.07.018. [DOI] [PubMed] [Google Scholar]
- 85.Kim Y-J, Nam R-H, Yoo YM, Lee C-J. Identification and functional evidence of GABAergic neurons in parts of the brain of adult zebrafish (Danio rerio) Neurosci. Lett. 2004;355:29–32. doi: 10.1016/j.neulet.2003.10.024. [DOI] [PubMed] [Google Scholar]
- 86.Monesson-Olson B, et al. Expression of the eight GABA A receptor α subunits in the developing zebrafish central nervous system. PLoS ONE. 2018;13:e0196083. doi: 10.1371/journal.pone.0196083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Abrahao KP, Salinas AG, Lovinger DM. Alcohol and the brain: neuronal molecular targets, synapses, and circuits. Neuron. 2017;96:1223–1238. doi: 10.1016/j.neuron.2017.10.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Holt RA, Bateson AN, Martin IL. Chronic treatment with diazepam or abecarnil differentially affects the expression of GABAA receptor subunit mRNAs in the rat cortex. Neuropharmacology. 1996;35:1457–1463. doi: 10.1016/s0028-3908(96)00064-0. [DOI] [PubMed] [Google Scholar]
- 89.Mahmoudi M, Kang M-H, Tillakaratne N, Tobin AJ, Olsen RW. Chronic intermittent ethanol treatment in rats increases GABAA receptor α4-subunit expression: possible relevance to alcohol dependence. J. Neurochem. 2002;68:2485–2492. doi: 10.1046/j.1471-4159.1997.68062485.x. [DOI] [PubMed] [Google Scholar]
- 90.Chatterjee D, Gerlai R. High precision liquid chromatography analysis of dopaminergic and serotoninergic responses to acute alcohol exposure in zebrafish. Behav. Brain Res. 2009;200:208–213. doi: 10.1016/j.bbr.2009.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Pan Y, Kaiguo M, Razak Z, Westwood JT, Gerlai R. Chronic alcohol exposure induced gene expression changes in the zebrafish brain. Behav. Brain Res. 2011;216:66–76. doi: 10.1016/j.bbr.2010.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Benton CS, et al. Evaluating genetic markers and neurobiochemical analytes for fluoxetine response using a panel of mouse inbred strains. Psychopharmacology. 2012;221:297–315. doi: 10.1007/s00213-011-2574-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Huang GJ, et al. Neurogenomic evidence for a shared mechanism of the antidepressant effects of exercise and chronic fluoxetine in mice. PLoS ONE. 2012;7:e35901. doi: 10.1371/journal.pone.0035901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Lee JH, et al. Gene expression profile analysis of genes in rat hippocampus from antidepressant treated rats using DNA microarray. BMC Neurosci. 2010;11:152. doi: 10.1186/1471-2202-11-152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Mennigen JA, et al. Effects of fluoxetine on the reproductive axis of female goldfish (Carassius auratus) Physiol. Genom. 2008;35:273–282. doi: 10.1152/physiolgenomics.90263.2008. [DOI] [PubMed] [Google Scholar]
- 96.Blum K, et al. Global evidence for the key role of the dopamine D2 receptor gene (DRD2) and DRD2 receptors in alcoholism. J. Neurogenet. Res. 2017;1:1001. [Google Scholar]
- 97.Dobbs LK, Lemos JC, Alvarez VA. Restructuring of basal ganglia circuitry and associated behaviors triggered by low striatal D2 receptor expression: implications for substance use disorders. Genes Brain Behav. 2017;16:56–70. doi: 10.1111/gbb.12361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Volkow ND, et al. Neurochemical and metabolic effects of acute and chronic alcohol in the human brain: studies with positron emission tomography. Neuropharmacology. 2017;122:175–188. doi: 10.1016/j.neuropharm.2017.01.012. [DOI] [PubMed] [Google Scholar]
- 99.Blaser RE, Rosemberg DB. Measures of anxiety in zebrafish (Danio rerio): dissociation of black/white preference and novel tank test. PLoS ONE. 2012;7:e36931. doi: 10.1371/journal.pone.0036931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Rosemberg DB, et al. Differences in spatio-temporal behavior of zebrafish in the open tank paradigm after a short-period confinement into dark and bright environments. PLoS ONE. 2011;6:e19397. doi: 10.1371/journal.pone.0019397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Mccurley AT, Callard GV. Characterization of housekeeping genes in zebrafish: male-female differences and effects of tissue type, developmental stage and chemical treatment. BMC Mol. Biol. 2008;9:102. doi: 10.1186/1471-2199-9-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Ye J, et al. Primer-BLAST: a tool to design target-specific primers for polymerase chain reaction. BMC Bioinform. 2012;13:134. doi: 10.1186/1471-2105-13-134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Brown C, Braithwaite VA. Size matters: a test of boldness in eight populations of the poeciliid Brachyraphis episcopi. Anim. Behav. 2004;68:1325–1329. [Google Scholar]
- 104.Harris S, Ramnarine IW, Smith HG, Pettersson LB. Picking personalities apart: estimating the influence of predation, sex and body size on boldness in the guppy Poecilia reticulata. Oikos. 2010;119:1711–1718. [Google Scholar]
- 105.Roy T, Bhat A. Population, sex and body size: determinants of behavioural variations and behavioural correlations among wild zebrafish Danio rerio. R. Soc. Open Sci. 2018;5:170978. doi: 10.1098/rsos.170978. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Benjamini Y, Drai D, Elmer G, Kafkafi N, Golani I. Controlling the false discovery rate in behavior genetics research. Behav. Brain Res. 2001;125:279–284. doi: 10.1016/s0166-4328(01)00297-2. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analyzed during this study are included in this published article and its Supplementary Information files.