Abstract
Since the widespread of acute respiratory syndrome infection caused by Coronavirus-19 unenhanced computed tomography (CT) was considered an useful imaging tool commonly used in early diagnosis and monitoring of patients with complicated COVID-19 pneumonia.
Many typical imaging features of this disease were described such as bilateral multilobar ground-glass opacification (GGO) with a prevalent peripheral or posterior distribution, mainly in the lower lobes, and sometimes consolidative opacities superimposed on GGO. As less common findings were mentioned septal thickening, bronchiectasis, pleural thickening, and subpleural involvement.
After 3 months from the onset of COVID-19 pneumonia some studies published the evolution of imaging features of COVID-19 pneumonia such as an increase of GGOs and a progressive transformation of GGO into multifocal consolidative opacities, septal thickening, and development of a crazy-paving pattern.
As far as we know bronchiectasis were described only as a possible aspecific imaging feature of COVID-19 pneumonia and no studies reporting the onset or evolution of bronchiectasis during imaging follow-up in patients with COVID-19 have been published. Here we describe two cases of rapid evolution of bronchiectasis documented at CT in patients with COVID-19 pneumonia.
Further studies are necessary to determine predisposing factors to the onset of bronchiectasis and to evaluate clinical correlation with respiratory distress. Radiologists should always consider bronchial features when they report CT scans of patients with COVID-19 pneumonia.
Keywords: COVID-19, bronchiectasis, Chest CT, follow-up
Background
Since the initial outbreak of Coronavirus disease-19 (COVID-19) from Hubei province in China in late December 2019, acute respiratory syndrome infection has spread across China and to several countries around the world during the first 3 months of 2020 [1]. Symptoms are variable and nonspecific and include dry cough, fever, fatigue, myalgias, dyspnea, anosmia, and ageusia. Less commonly encountered symptoms include productive cough, pleuritic chest pain, hemoptysis, and diarrhea [2]. Real-time reverse transcription polymerase chain reaction (rRT-PCR) test is the current gold standard for confirming infection, performed using nasal or pharyngeal swab specimens or induced sputum. Routine unenhanced chest computed tomography (CT) is useful in early diagnosis of COVID-19 infection, in monitoring disease progression, coinfection, or disease stability [3]. Since chest CT was introduced as a diagnostic tool for COVID-19 pneumonia, many typical features of this disease were described such as bilateral multilobar ground-glass opacification (GGO) with a prevalent peripheral or posterior distribution, mainly in the lower lobes, and sometimes consolidative opacities superimposed on GGOs could be found. The less common imaging findings include septal thickening, bronchiectasis, pleural thickening, and subpleural involvement [4], [5], [6], [7], [8].
Some studies described the evolution of imaging features of COVID-19 pneumonia: an increase in GGOs and a progressive transformation of GGO into multifocal consolidative opacities, septal thickening and development of a crazy-paving pattern [9]. To our best knowledge, no studies reporting the onset or evolution of bronchiectasis during follow-up in patients with COVID-19 have been published. We describe two cases of rapid evolution of bronchiectasis documented at CT in patients with COVID-19 pneumonia.
Case 1 presentation
A 68-year-old woman with no significant comorbidities was admitted to the emergency department after 10 days with fever, sore throat and diarrhea secondary to COVID-19 infection confirmed with rRT-PCR test. She was discharged from the hospital with supportive therapy. A week later she returned to the emergency department with worsening dyspnea and body temperature >38.5°C. Her vital signs were blood pressure 114/65 mm Hg and heart rate 53 bpm. Chest X-ray showed bilateral shaded patchy opacities. Blood test revealed lymphocytes 900/mm3 and C-reactive protein 110 mg/L. Arterial blood gas analysis showed pH 7.55, pO2 39 mm Hg, pCO2 28 mm Hg, HCO3− 24.5 mmol/L and arterial oxygen saturation (SaO2) of 81.6%. She was hospitalized in the Internal Medicine Department. On clinical examination: blood pressure 110/60 mm Hg, heart rate 48 bpm rhythmics, respiratory rate 32/min, and SpO2, as measured by a pulse oximeter was 98% while breathing in a respiratory mask with reservoir bag of 8 L/min. Chest auscultation revealed fine crackles on both pulmonary bases. The electrocardiogram showed sinus bradycardia with QTc 416 ms. Therapy with 400 mg per day of hydroxychloroquine and lopinavir/ritonavir at a dose of 400/100 mg was begun.
On day 1 after admission a dosage of the D-dimer was 2472 µg/L (and remained stable during the next days) whereas fibrinogen dropped down (from 7.28 g/L to 4.93 in 2 days). Prothrombin time and activated partial thromboplastin time remained stable (1.02 and 0.9, respectively).
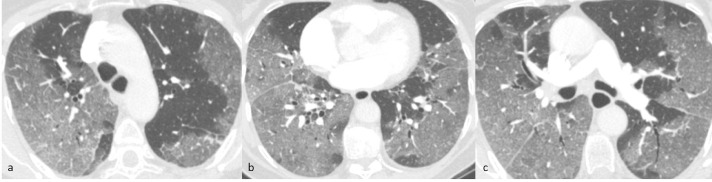
During hospitalization, oxygen saturation further worsened, and it was necessary to introduce pronation and morphine and to increase O2-therapy. On day 3 from admission, D-dimer remained raised, therefore CT pulmonary angiography (CTPA) was performed. On the CTPA exam, no filling defects were detected in the pulmonary arteries, but peripheral patchy ground-glass opacities involving all pulmonary lobes were described. Segmental and subsegmental bronchi had normal caliber (Fig. 1).
Fig. 1.
Axial CTPA of case 1 (lung window) 3 days after admission shows multiple bilateral peripheral patchy ground-glass opacities with ill-defined margins and mainly subpleural distribution involving all the lobes, associated with initial interlobular septal thickening (a-c). There are no parenchymal nodules, enlarged mediastinal lymph nodes or pleural effusion. The caliber and walls of segmental and subsegmental bronchi are regular.
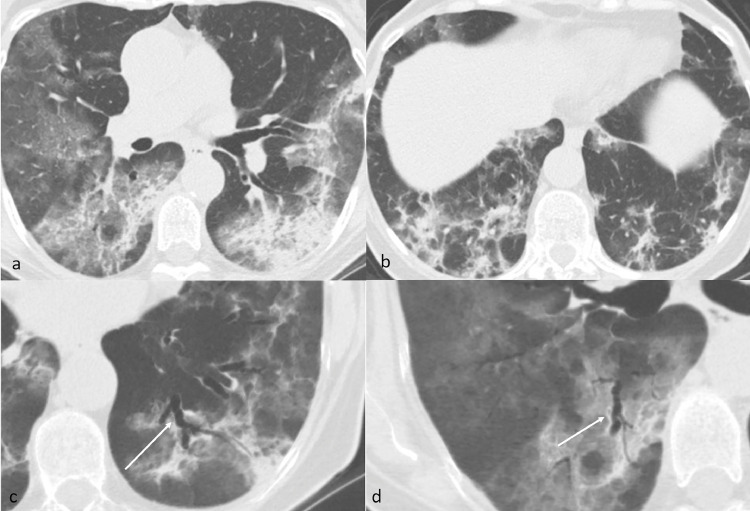
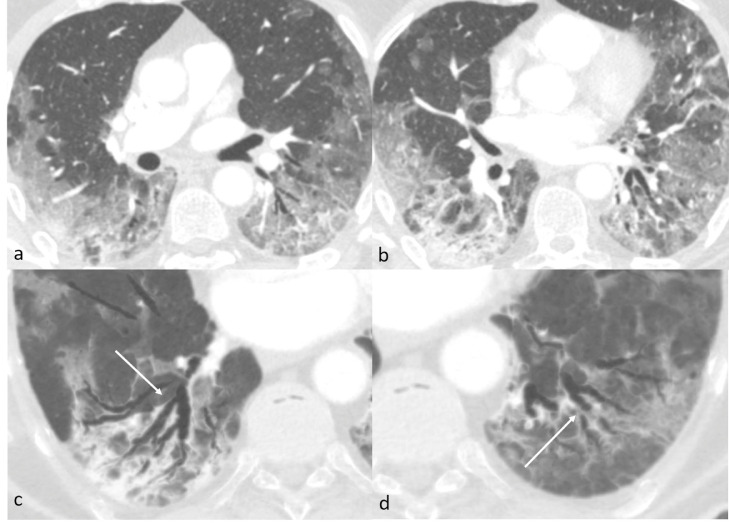
On the next week, the saturation got worst, on Chest X-ray the opacities were diffuse and increased compared to the previous exam. SpO2 was 95% using a mask with reservoir bag at 12 L of O2/min and exactly 6 days after the first CTPA an unenhanced chest CT was performed. CT showed a progression of viral pneumonia, with an increase in number and extension of multiple patchy GGOs with inter- and intralobular septal thickening characterized by peripheral distribution in all lobes, with the transformation of GGOs into consolidative opacifications on posterior-basal segments of both lower lobes (Fig. 2).
Fig. 2.
Axial Unenhanced CT of case 1 (lung window) 9 days after admission shows progression of viral pneumonia with diffuse peripheral predominant patchy ground-glass opacities, subsegmental crazy paving and initial subpleural consolidation (a and b). Axial MinIP reformat (c and d) shows enlarged caliber and irregular walls of segmental and subsegmental bronchi next to the areas of parenchymal consolidation of the apical segment of the left (c) and right (d) lower lobes (arrows).
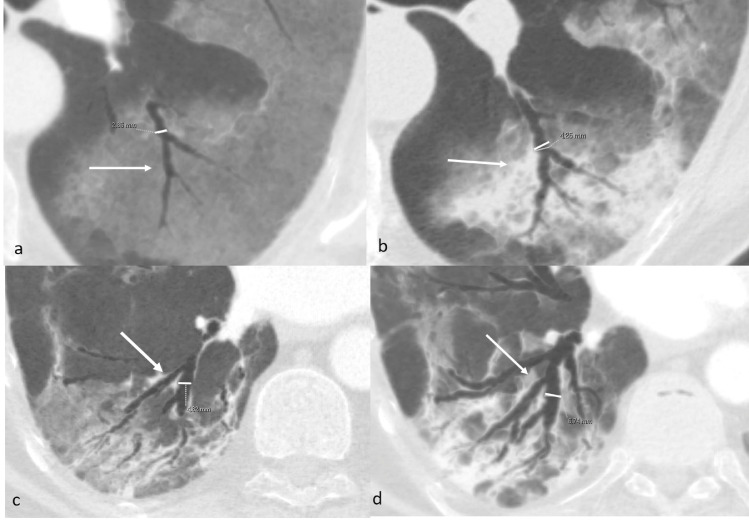
The segmental bronchi in both lungs, with a significant predominance next to the consolidative opacifications in the lower lobes, showed an enlarged caliber and irregular walls, becoming bronchiectasic (Fig. 2c and d). The ratio between the caliber of the bronchus and the corresponding artery increased, nearly doubling (from 0.61 in the first exam to 1.18 in the second exam evaluating the same segmental bronchus) in an interval time between the two exams of only 6 days (Fig. 3).
Fig. 5.
Axial MinIP reformats of case 1 (a and b) and case 2 (c and d). Measure of caliber of the apical segmental bronchus of the left lower lobe on the first (a, arrow) and second exam (b, arrow) in case 1 and of the posterobasal segmental bronchus of the right lower lobe on the first (c, arrow) and second exam (d, arrow) in case 2. The enlargement of the bronchi and evolution into bronchiectasis is well visible and occurred respectively after 6 and 5 days of hospitalization.
On the other hand, comparing the two multidetector CT exams, no variation in the caliber of the segmental and subsegmental branches of pulmonary arteries was documented.
After hospital discharge the patient has reported an improvement of SaO2 in ambient air 95% (despite 95% by using a mask with reservoir bag at hospital), but she still complained about dyspnea during mild exercise.
Case 2 presentation
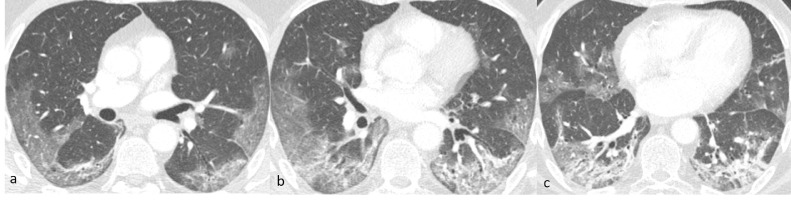
A 67-year-old man with high blood pressure developed influenza-like symptoms after direct contact with a person with COVID-19 infection. After 10 days with fever, headache, dry cough, and two syncopal episodes he presented to the emergency department. COVID-19 infection was confirmed with an rRT-PCR test. Chest X-ray detected bilateral patchy opacities. Blood test results included white blood cells 4540/ mm3, lymphocytes 77%, and C-reactive protein 23 mg/L. Arterial blood gas analysis revealed pH 7.5, pO2 54 mm Hg, pCO2 32 mm Hg, and SaO2 95%. Electrocardiogram did not show any rhythm alteration with QTc 419 ms. He was admitted to the Infectious Diseases Department. Vital signs were blood pressure 120/75 mm Hg, heart rate 82 bpm, SpO2 of 94% without O2-therapy. The body temperature was 36.7°C. Arterial blood gas analysis was repeated: pH 7.48, pO2 65 mm Hg, pCO2 34 mm Hg. Blood tests showed lymphocytes 630/mm3, C-reactive protein 24 mg/L and D-dimer 684 µg/L. On day 3 after admission, SpO2 worsened and it was necessary to add morphine and modify O2-therapy from nasal cannula 4 L/min to respiratory mask 10 L/min. SpO2 went from 87% to 94% with PaO2/FiO2 112 mm Hg. D-dimer was 3937 µg/L. CTPA was performed: no filling defects were detected in the pulmonary arteries, but multiple bilateral peripheral and with mainly subpleural distribution patchy GGOs were documented, together with initial parenchymal distortion and some areas of consolidation at the posterior-basal segment of both lower lobes. Segmental and subsegmental bronchi caliber was at the upper limits of standard Fig. 4. The following day, the saturation worsened with PaO2/FiO2 87 mm Hg, and the patient was admitted to the Intensive Care Unit for 2 days because he needed noninvasive positive pressure ventilation. A week after admission, lower-limb compression ultrasonography detected deep venous thrombosis in the venous tract distal to the left popliteal vein. Furthermore, D-dimer was 1661 µg/L (from 500 µg/L) and CTPA was repeated. CTPA detected a filling defect in a subsegmental pulmonary artery in the lower left lobe.
Fig. 3.
Axial CT of case 2 (lung window) 3 days after admission shows multiple bilateral peripheral and with mainly subpleural distribution patchy ground-glass opacities (a-c) with ill-defined margins, involving all the lobes with visible interlobular septal thickening within the lesions and initial parenchymal distortion with some areas of consolidation in the posterobasal segments of the lower lobes (b and c). There are no parenchymal nodules, enlarged mediastinal lymph nodes or pleural effusion. The caliber and walls of segmental and subsegmental bronchi are regular.
Progression of viral pneumonia with increased interlobular septal thickening and larger areas of consolidation inside the ground-glass opacities were visible Fig. 5.
Fig. 4.
Axial Unenhanced CT of case 2 (lung window) 8 days after admission shows progression of viral pneumonia with increase of the interlobular septal thickening and areas of consolidation inside the ground-glass opacities (a and b). Axial MinIP reformat (c and d) shows enlarged caliber and irregular walls of segmental and subsegmental bronchi next to the areas of parenchymal consolidations in the posterobasal segment of the right (c) and left (d) lower lobes (arrows).
Also in this case, the segmental bronchi in both lungs, with a significant predominance next to the consolidative opacifications in the lower lobes, showed an enlarged caliber and irregular walls, becoming bronchiectasic Fig. 5c and d. The ratio between the caliber of the bronchus and the corresponding artery increased (from 0.7 in the first exam to 1.12 in the second exam evaluating the same segmental bronchus) in an interval time between the two exams of only 5 days Fig. 3.
Similarly to the previous case, comparing the two CT exams, no variation in the caliber of the segmental and subsegmental branches of pulmonary arteries was documented. After 2 months from discharge, the SaO2 of the patient in ambient air was 98%-99%. He had still dyspnea, but only during mild exercise.
Discussion
In the literature, several studies are reporting parenchymal findings of COVID-19 pneumonia. CT findings of COVID-19 pulmonary infection are bilateral, peripheral, and basal predominant GGO, crazy-paving and consolidation, often with an extensive geographical distribution. Multiple areas of GGOs and consolidation occur in the same patient and have round morphology or a reversed halo sign [4], [5], [6], [7]. Few studies have reported short-term CT follow-up patients with COVID-19 infection [6,9]. Pan et al described the temporal evolution of 21 patients with mild pneumonia without severe respiratory symptoms [9]. Other studies report airway changes and bronchiectasis [8]: Fang describe 2 cases of COVID-19 pneumonia with bronchiectasis [10]. Hanley in a postmortem CT study on 101 bodies observed traction bronchiectasis in 53% of cases, the changes being more likely bilateral and involving the lower lobes [11].
As far as we know, bronchiectasis were described as a possible imaging feature of COVID-19 pneumonia, but no published studies are reporting the onset and evolution of bronchiectasis during the short follow-up in a patient with this disease.
Bronchiectasis is defined at CT as a dilated bronchial lumen relative to the adjacent pulmonary artery (with a ratio B/A superior to 0.7), lack of bronchial tapering, or identification of bronchi within 1 cm of the pleural surface [12].
We present two cases of patients with severe respiratory symptoms occurring in a few days and who underwent two CT exams over 6 and 5 days, respectively. In both the patients, the first exam did not show any bronchiectasis (Fig. 1, Fig. 3). Only a few days later, in both patients, the second CT scan showed bilateral lower lobes bronchiectases of segmental and subsegmental bronchi with a visible increase in the caliber of the bronchi and the ratio between the caliber of the bronchi and the corresponding segmental artery (from 0.61 in the first exam to 1.18 in the second exam evaluating the same segmental bronchus for case 1 and from 0.7 to 1.12 for case 2) (Fig. 1, Fig. 5, Fig. 4) associated to peripheral parenchymal consolidation (Fig. 2, Fig. 4).
Unfortunately to date we do not know if bronchiectasis are transient or permanent because none of them underwent a control CT exam yet.
Although in the literature, a definite time course of onset of bronchiectasis in respiratory disease is not described, bronchial dilatation is always expression of a chronic respiratory disease with a fibrotic parenchymal change [13]. The rapid onset of bronchiectasis in COVID patients could be a sign of a rapid and progressive pulmonary fibrotic process triggered by virus infection.
Conclusions
We report two cases of bronchiectasis in the lower lobes developing in a few days in two patients with severe respiratory symptoms without significant comorbidities. Further studies about CT follow-up in COVID-19 patients with severe pneumonia are necessary to determine predisposing factors to the development of bronchial dilatation and to evaluate if there is a clinical correlation with respiratory distress and rapid development of bilateral bronchial ectasia. Follow-up CT could suggest if the onset of bronchiectasis during COVID-19 pneumonia could be related to an evolution to a fibrosing lung pattern.
Footnotes
During the COVID-19 pandemic, physicians were not allowed to introduce or remove from patients' rooms any piece of paper or pen. The patient gave oral consent to the utilization of her clinical and imaging data. The consent was annotated in the clinical diary.
References
- 1.Zhu N, Zhang D, Wang W, Li X, Yang B, Song J. A novel coronavirus from patients with pneumonia in China, 2019. N Engl J Med. 2020;382:727–733. doi: 10.1056/NEJMoa2001017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Huang C, Wang Y, Li X, Ren L, Zhao J, Hu Y. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395:497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nasir MU, Roberts J, Muller NL, Macri F, Mohammed MF, Akhlaghpoor S. The role of emergency radiology in COVID-19: from preparedness to diagnosis. Can Assoc Radiol J. 2020;71(3):293–300. doi: 10.1177/0846537120916419. [DOI] [PubMed] [Google Scholar]
- 4.Kanne JP, Little BP, Chung JH, Elicker BM, Ketai LH. Essentials for eadiologists on COVID-19: an update— Radiology Scientific Expert Panel. Radiology. 2020 doi: 10.1148/radiol.2020200527. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bernheim A, Mei X, Huang M, Yang Y, Fayad ZA, Zhang N. Chest CT findings in coronavirus disease-19 (COVID-19): relationship to duration of infection. Radiology. 2020 doi: 10.1148/radiol.2020200463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Hosseiny M, Kooraki S, Gholamrezanezhad A, Reddy S, Myers L. Radiology perspective of coronavirus disease 2019 (COVID-19): lessons from severe acute respiratory syndrome and middle east respiratory syndrome. Am J Roentgenol. 2020;214:1078–1082. doi: 10.2214/AJR.20.22969. [DOI] [PubMed] [Google Scholar]
- 7.Caruso D, Zerunian M, Polici M, Pucciarelli F, Polidori T, Rucci C. Chest CT features of COVID-19 in Rome, Italy. Radiology. 2020 doi: 10.1148/radiol.2020201237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ye Z, Zhang Y, Wang Y, Huang Z, Song B. Chest CT manifestations of new coronavirus disease 2019 (COVID-19): a pictorial review. Eur Radiol. 2020 doi: 10.1007/s00330-020-06801-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Pan F, Ye T, Sun P, Gui S, Liang B, Li L. Time course of lung changes on chest CT during recovery from 2019 novel coronavirus (COVID-19) pneumonia. Radiology. 2020 doi: 10.1148/radiol.2020200370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fang Y, Zhang H, Xu Y, Xie J, Pang P, Ji W. CT manifestations of two cases of 2019 novel coronavirus (2019-NCoV) pneumonia. Radiology. 2020 doi: 10.1148/radiol.2020200280. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hanley B, Lucas SB, Youd E, Swift B, Osborn M. Autopsy in suspected COVID-19 cases. J Clin Pathol. 2020 doi: 10.1136/jclinpath-2020-206522. [DOI] [PubMed] [Google Scholar]
- 12.Hansell DM, Bankier AA, MacMahon H, McLoud TC, Müller NL, Remy J. Fleischner society: glossary of terms for thoracic imaging. Radiology. 2008 doi: 10.1148/radiol.2462070712. [DOI] [PubMed] [Google Scholar]
- 13.McShane PJ, Naureckas ET, Tino G, Strek ME. Non–cystic fibrosis bronchiectasis. Am J Respir Crit Care Med. 2013;188(6):647–656. doi: 10.1164/rccm.201303-0411CI. [DOI] [PubMed] [Google Scholar]