Abstract
Immunotherapies that harness the immune system to kill cancer cells have showed significant therapeutic efficacy in many human malignancies. A growing number of studies have highlighted the relevance of neoantigens in recognizing cancer cells by intrinsic T cells. Cancer neoantigens are a direct consequence of somatic mutations presenting on the surface of individual cancer cells. Neoantigens are fully cancer-specific and exempt from central tolerance. In addition, neoantigens are important targets for checkpoint blockade therapy. Recently, technological innovations have made neoantigen discovery possible in a variety of malignancies, thus providing an impetus to develop novel immunotherapies that selectively enhance T cell reactivity for the destruction of cancer cells while leaving normal tissues unharmed. In this review, we aim to introduce the methods of the identification of neoantigens, the mutational patterns of human cancers, related clinical trials, neoantigen burden and sensitivity to immune checkpoint blockade. Moreover, we focus on relevant challenges of targeting neoantigens for cancer treatment.
Keywords: neoantigens, cancer immunotherapy, clinical trial, checkpoint blockade, targeted cancer
Introduction
The occurrence rate of cancer is increasing rapidly (Torre et al., 2015). New methods such as immunotherapy have become an optimal choice in cancer treatment, along with chemotherapy, radiation and surgery (Restifo et al., 2012; Hamid et al., 2013; Robert et al., 2015a). The foundation of cancer immunology was based on tumor transplantation studies in syngeneic mice (Prehn and Main, 1957; Old and Boyse, 1964). Concurrently, clinicians observed that in gastric carcinoma patients, tumor-infiltration lymphocytes (TILs) were correlated with longer post-operative survival (Black et al., 1954). Recently, it has demonstrated that autologous T cells can show profound tumoricidal activity and immunotherapies based on T cells are effective in multiple human malignancies (Rosenberg, 2011; Rosenberg et al., 2011). In particular, treatment of patients with checkpoint blockade has shown a significant effect on patient survival (Hodi et al., 2010; Brahmer et al., 2012; Topalian et al., 2012; Sharma and Allison, 2015). These data provide clear evidence that endogenous T cells can recognize antigenic determinants—epitopes that present on the tumor cell surface from the major histocompatibility complexes (MHCs). Such epitopes may be originated from three classes of antigens: (1) viral antigens that are encoded by viral open reading frames (ORF) in virus-infected tumor cells (Walboomers et al., 1999; Gillison et al., 2000; Feng et al., 2008), such as hepatitis B virus (HBV) (Merlo et al., 2010), Epstein-Barr virus (EBV) (Lin et al., 2002), and human papilloma virus (HPV) (Morrow et al., 2013); (2) tumor-associated antigens (TAAs) that expression levels are very low in some normal tissues but are overexpressed in malignant cells, including oncofetal antigens, cancer testis antigens (CTA), overexpressed oncogenic proteins and selected differentiation antigens (Caballero and Chen, 2009; Melero et al., 2014; Ward et al., 2016), for example NY-ESO-1 (D’Angelo et al., 2018) and CD19 on B cell malignancies (Sabatino et al., 2016); and (3) neoantigens, which are immunogenic products of somatic mutations that are fully specific to tumors. In the late 1980s, Boon and colleagues were among the first to report that aberrant peptides derived from tumor mutations were able to elicit a tumor specific T cell response in a mouse model (De Plaen et al., 1988; Lurquin et al., 1989). A few years later, it was also observed in human tumors that somatic mutations were a source of neoantigens recognized by T cells (Coulie et al., 1995; Monach et al., 1995; Wolfel et al., 1995). Recently, studies have demonstrated that neoantigens are able to recognize cancer cells by intrinsic T cells (Lennerz et al., 2005; Castle et al., 2012; Matsushita et al., 2012; Kvistborg et al., 2014; Rajasagi et al., 2014; Wick et al., 2014; Chan et al., 2015; Cohen et al., 2015; Rizvi et al., 2015). Neoantigens are fully tumor-specific and bypass central tolerance (Heemskerk et al., 2013), and thus they are not expected to induce autoimmune toxicity and they are a potential target for cancer immunotherapy. With the recent development of cancer genomics (Garraway and Lander, 2013; Vogelstein et al., 2013) and high-throughput immunologic screening (Lin et al., 2008; Zhang et al., 2011), the goal of the analysis of neoantigens based on individual patients has become achievable, which makes neoantigen-directed immunotherapy highly attractive. In this review, we aim to introduce the framework for the identification and prioritization of neoantigens, the mutational patterns of cancer, neoantigen-related trials, mutational burden and sensitivity to immune checkpoint blockade. More importantly, we highlight the relevant challenges of targeting neoantigens for cancer treatment.
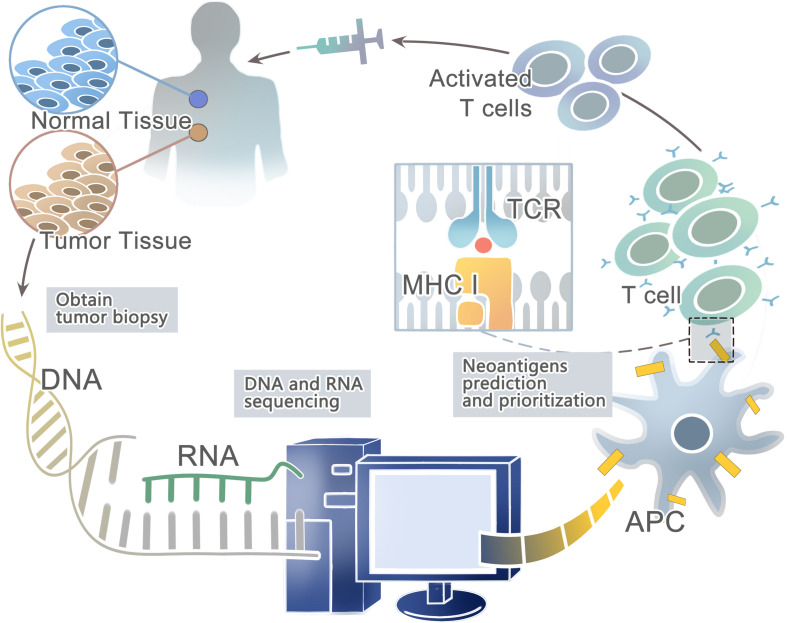
Neoantigen Identification
Advances in next-generation sequencing (NGS) (Margulies et al., 2005; Shendure et al., 2005; Bentley et al., 2008; Wheeler et al., 2008; Drmanac et al., 2010) have allowed for the rapid and relatively inexpensive exhaustive sequencing of genomic changes across tens of thousands of human cancers (Li et al., 2011; Stratton, 2011; Gubin et al., 2015; Lu and Robbins, 2016). In conjunction, the innovation of high-throughput immunologic screening techniques has promoted the detection and isolation of neoantigens that can evoke specific T cell responses (Hadrup et al., 2009; Bentzen and Marquard, 2016; Bentzen and Hadrup, 2017) (Figure 1). In the human genome, only 1% codes for known expressed genes (the exomes) among the approximately 3 billion nucleotides (Choi et al., 2009; Ng et al., 2009). Therefore, it can significantly reduce time and costs to sequence only functional exomes. Large projects such as The Cancer Genome Atlas (TCGA) [Cancer Genome Atlas (TCGA) Research Network, 2008; Garraway and Lander, 2013] and The International Cancer Genome Consortium (ICGC) (Hudson et al., 2010) have identified cancer genomes across multiple tumor types. However, whole-exome sequencing (WES) provides limited information on non-coding regions of cancer genomes, including untranslated regions (UTRs), promoters, enhancers, introns, regulatory elements and diverse non-coding RNAs (ncRNAs) as well as unannotated regions (Garraway and Lander, 2013). In contrast, whole-genome sequencing (WGS) may be able to detect these events (Meyerson et al., 2010). Second-generation sequencing of the transcriptome (RNA-seq) is another powerful approach — as cDNA may derive from mRNA, total RNA or other RNAs, such as microRNAs and lncRNA (Morrissy et al., 2009). Recent studies have suggested that tumors harbor more abundant alternative splicing events than paired normal tissues by comprehensive analysis of WES with RNA-seq data and proteomic data, which is a potential source to generate tumor-specific neoantigens (Kahles et al., 2018; Park et al., 2018). In fact, a few studies have developed peptide arrays representing all possible frameshift peptides and detected the antibody responses to the frameshift peptides. This might be a useful method for cancer neoantigen screening (Zhang et al., 2018; Shen et al., 2019). In conclusion, somatic DNA mutations are usually computed from WGS, WES or RNA-seq data from comparisons between tumor and normal sequences (Ding et al., 2014; Tran et al., 2015).
FIGURE 1.
A framework for the identification and prioritization of neoantigens from computational analysis for an individual cancer sample.
Then the patient-specific NGS data from WGS, WES, or RNA-seq can be used to predict HLA types with computational tools such as Optiptype (Szolek et al., 2014) and Polysolver (polymorphic loci resolver) (Shukla et al., 2015). First, reads are selected from the NGS data that potentially derived from the HLA region; second, they are aligned to a full-length genomic library of all known HLA alleles (Robinson et al., 2013); and third, all the best-scoring alignments of each read are kept for further study. After predicting HLA types, computational algorithms such as NetMHC (Andreatta and Nielsen, 2016), NetMHCpan (Rammensee et al., 1999; Nielsen et al., 2007; Nielsen and Andreatta, 2016; Jurtz and Paul, 2017), and MHCflurry (O’Donnell et al., 2018) trained on large in vitro experimental datasets can be used to prioritize candidate neoantigens that bind to the predicted HLA types with high affinity. For example, Neopepsee and pVAC-Seq are representative analysis pipelines for tumor somatic mutations (Hundal et al., 2016; Kim et al., 2018). However, these algorithms are not a good predictor of actual HLA presentation (Bassani-Sternberg et al., 2016; Abelin et al., 2017), with only <5% of predicted peptides found on tumor cell surface (Yadav et al., 2014; Bassani-Sternberg et al., 2015). On the other hand, these prediction strategies do not consider proximal variants that can alter peptide sequences and affect neoantigen binding predictions (Hundal and Kiwala, 2019). Recently, a new prediction model-EDGE based on tumor HLA peptide mass spectrometry (MS) datasets has increased the positive predictive value up to nine-fold. However, it still does not incorporate TCR binding or predict HLA class II binding epitopes (Bulik-Sullivan et al., 2018).
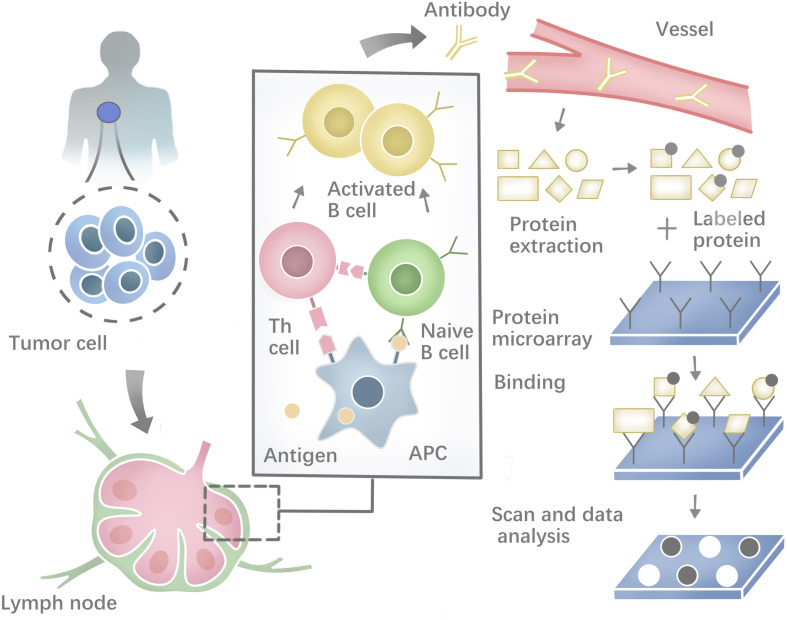
Besides, a range of methodologies can be used to identify autoantibodies against tumor neoantigens based on B-cell response. The costimulatory molecules from CD4+ helper T cells and the neoepitopes presented on the surface of antigen-presenting cells (APCs) could activate the Naïve B cells in lymphoid organs. Most activated B cells will then differentiate into plasma cells to produce antibodies against tumor neoantigens (Zaenker et al., 2016). Protein microarrays are time-effective high-throughput tools (Figure 2). Sandwich immunoassays in the miniaturized system could successfully identify tumor antigens in sera samples extracted from patients (Pollard et al., 2007; Yang et al., 2013). First described by O’Farrell (1975), another tool named Serologic Proteome analysis (SERPA) or 2-D western blots, consists of the isoelectric focusing (IEF) gel run in the first dimension and SDS-PAGE gel run in the second dimension. SERPA separates the proteins in the gel by their isoelectric point (IP) and molecular mass and then transfers the proteins from the gel to a carrier membrane to screen antibodies. Finally, the antigenic protein spots can be identified by MS (Tjalsma et al., 2008). This approach has been used to identify antigens in different tumor types (Dai et al., 2017; Belousov et al., 2019). Serological analysis of recombinant cDNA expression libraries (SEREX), which combines serological analysis with antigen cloning techniques, is a widely used technique to explore tumors’ antigen repertoire. SEREX first construct a cDNA library from cancer cell lines or fresh tumor samples, then screen the cDNA library with autologous sera of cancer patients, and finally sequence the immune-reactive clones. Despite the laborious process, SEREX have identified a variety of tumor antigens including CTAs, differentiation antigens, mutational antigens, splice-variant antigens and over-expressed antigens (Chen et al., 1997; Scanlan et al., 1998; Türeci et al., 1998a, b; Brass et al., 1999; Jäger et al., 1999). Furthermore, other methods such as Multiple Affinity Protein Profiling (MAPPing) and nanoplasmonic biosensor have also been developed to identify tumor antigens (Lee et al., 2015; Jo et al., 2016).
FIGURE 2.
Profiling the humoral immune response and the process of protein microarray to capture autoantibodies against antigens.
Mutational Patterns in Human Cancers
Somatic mutations presenting in cancer cells but not in a patient’s germ line are the primary cause of cancers (Weir et al., 2004). The cancer genome alterations include single base substitutions, insertion-deletions (so-called indels), rearrangements (inversions, translocations, duplications, and deletions) (Stratton et al., 2009; Alexandrov et al., 2013; Barrick and Lenski, 2013; Vogelstein et al., 2013; Helleday et al., 2014), but also new DNA sequences acquired from exogenous sources, notably those of viruses such as HBV, HPV, and EBV (Talbot and Crawford, 2004). Somatic mutations can be classified as driver and passenger mutations, and driver mutations, such as BRAF, KRAS, EGFR, IDH1, and PIK3CA, can provide a selective growth advantage and promote cancer development while passenger mutations do not (Haber and Settleman, 2007). Even though cancer has a mere handful of driver mutations, they are still attractive targets for immunotherapy when they are shared between different cancers and individuals. The rates of mutations vary among tumors and cancer types. Some cancer types, such as medulloblastomas, carcinoids, acute leukemias, and testicular germ cell tumors, generally carry relatively few mutations (Greenman et al., 2007; Parsons et al., 2011). However, lung cancers and melanomas occasionally have more than 100,000 mutations (Ding et al., 2008; Kan et al., 2010; Pleasance et al., 2010a), most likely because of overwhelming exposure to mutagenic carcinogen such as ultraviolet (UV) light (Trucco et al., 2018) and tobacco carcinogens (Pleasance et al., 2010b). Except for exogenous mutagenic exposure, endogenous mutational processes, such as mismatch repair deficiency in some colorectal cancers (Pena-Diaz et al., 2017) or upregulation of APOBEC cytosine deaminases, can also contribute to mutation burden (Alexandrov et al., 2013; Helleday et al., 2014). More information about the somatic mutations in human cancer can be found in COSMIC, the Catalog Of Somatic Mutations In Cancer1 (Forbes et al., 2015). One can even explore how cancer mutations impact the structure and function of more than 8,000 human proteins in COSMIC-3D (No authors, 2017a). Non-synonymous somatic mutations, which can alter amino acid coding sequences, are the main cause of neo-epitopes (Wood et al., 2007). Here, we summarize the number of non-synonymous somatic mutations in different cancer types (Table 1). For example, Sjoblom et al. (2006) identified 1,307 somatic mutations among 13,023 genes in 11 breast and 11 colorectal cancers (83% were missense mutations, 6% were nonsense, and the remainder were insertions, deletions, duplications, and changes in non-coding regions). Neoantigens generated from tumor somatic DNA mutations can be identified by the approach described in the first section.
TABLE 1.
Non-synonymous somatic mutations in different cancers.
| Cancer types | Non-synonymous somatic mutations | References |
| Acute myeloid leukemia | 10, n = 1 | Ley et al., 2008 |
| Breast cancer | Average of 84, n = 11 | Sjoblom et al., 2006; Wood et al., 2007 |
| Glioblastoma | Average of 36, n = 21 | Parsons et al., 2008 |
| Pancreatic cancer | Average of 48, n = 24 | Jones et al., 2008 |
| Hepatocellular cancer | 63, n = 1 | Totoki et al., 2011 |
| Colon cancer | Average of 76, n = 11 | Sjoblom et al., 2006; Segal et al., 2008 |
| Melanoma | Average of 201, n = 14 | Wei et al., 2011 |
| Lung cancer | More than 300, n = 1 | Lee et al., 2010 |
Except for somatic non-synonymous protein-altering mutations, tumor neoantigens can be generated from alternative splicing variations. Pre-mRNA splicing is a biological process that contains the removal of introns and the ligation of exons to form mature RNA products (Sharp, 1994). However, the splice site and exons are alternative, which means a single gene can produce numerous mRNA isoforms (Nilsen and Graveley, 2010). And genomic variants in splicing regulatory sequences can disrupt splicing and produce aberrant mRNA and protein products (Pagani and Baralle, 2004). Recently, studies have suggested that tumors harbor more abundant alternative splicing events than paired normal tissues by comprehensive analysis of WES with RNA-seq data and proteomic data. For example, Kahles et al. (2018) performed a comprehensive analysis of alternative splicing across 32 tumor types from 8,705 patients and they found that tumors have on average 20% more alternative splicing events than normal tissues. They mainly focused on five types of alternative splicing events including exon skipping, mutually exclusive exons, intron retention, as well as alternative 3′ and alternative 5′ splice site changes. What’s more, they found that tumors harbored more exon–exon junctions termed neojunctions than normal tissues. Besides, they confirmed neojunctions-derived peptides from the Clinical Proteomic Tumor Analysis Consortium (CPTAC) and predicted the underlying target, MHC-I. The evidence showed that neojunctions-derived peptides were potential neoantigens for cancer immunotherapy. Multiple computational methods and databases have been developed to identify alternative splicing events from RNA-seq data, such as SplAdder and CancerSplicingQTL2 (Kahles et al., 2016; Tian et al., 2019). Smart and Margolis (2018) developed a computational strategy to identify neoepitopes generated from intron retention events in tumor transcriptomes and confirmed that these neoepitopes were processed and presented on MHC-I. Their results suggested that RNA-derived neoantigens were promising candidates for a personalized cancer vaccine (Smart and Margolis, 2018). Another mechanism to promote proteome diversity is RNA editing, which will influence RNA metabolism and function. The most prevalent form of RNA editing is the deamination of adenosine (A) to inosine (I) which may be a source for neoantigen production (Peng et al., 2018). What’s more, databases of A-to-I RNA editing events in humans have been developed, such as REDIportal3 and RADAR4 (Ramaswami and Li, 2014; Picardi et al., 2017). Recently, Zhang and Fritsche (2018) demonstrated that epitopes derived from RNA editing were presented by HLA molecules and capable of inducing immune responses.
Neoantigen Clinical Trial
Promising results of neoantigen vaccines from preclinical studies have raised significant interest in clinical development (Castle et al., 2012; Gubin et al., 2014; Kreiter et al., 2015; Zhang et al., 2017). Recently, the clinical response in patients with advanced melanoma who received neoantigen vaccines treatment is quite encouraging in several phase I clinical trials (Yadav et al., 2014; Ott et al., 2017; Sahin et al., 2017). The main platforms for neoantigen vaccine are synthetic long peptide (SLP) vaccine, DNA vaccine, RNA vaccine, and dendritic cell (DC) vaccine. In addition, adoptive T cell therapy (ACT) targeting neo-epitopes has shown significant efficacy. Currently, there is an increasing number of clinical trials on neoantigen vaccines in a variety of cancers (Tables 2, 3).
TABLE 2.
Ongoing clinical trials targeting cancer neoantigens.
| ClinicalTrial.gov identifier | Phase | Enrollment status | Sample size | Vaccine | Cancer type | Primary endpoint |
| NCT03645148 | I | Recruiting | 20 | Biological: iNeo-Vac-P01 Other: GM-CSF | Pancreatic cancer | (1) Objective response rate; (2) Number of participants experiencing clinical and laboratory adverse events |
| NCT02950766 | I | Recruiting | 20 | Biological: NeoVax | Kidney cancer | Number of participants with dose-limiting toxicity experienced within 49 days of treatment initiation as assessed by CTCAE v4.0 |
| NCT03558945 | I | Recruiting | 60 | Biological: Personalized neoantigen vaccine | Pancreatic cancer | Overall time: the time between operation and the death of patients |
| NCT03662815 | I | Active, not recruiting | 30 | Biological: iNeo-Vac-P01 Other: GM-CSF | AMST | (1) Objective response rate; (2) Number of participants experiencing clinical and laboratory adverse events |
| NCT03122106 | I | Active, not recruiting | 15 | Biological: Personalized neoantigen DNA vaccine | Pancreatic cancer | Safety of neoantigen DNA vaccine as measured by the number of subjects experiencing each type of adverse event |
| NCT03532217 | I | Recruiting | 20 | Biological: Neoantigen DNA vaccine Other: PROSTVAC-V PROSTVAC-F | Metastatic hormone-sensitive prostate cancer | (1) Safety and tolerability of regimen as defined by incidence of adverse events; (2) Immune response as measured by tetramers; (3) Immune response as measured by genomic studies; (4) Immune response as measured by flow cytometry; (5) Safety and tolerability of regimen as defined by incidence of dose-limiting toxicities |
| NCT02632019 | I, II | Unknown status | 40 | Biological: Dendritic cell-precision T cell for neo-antigen | Advanced biliary tract malignant tumor | Overall survival |
| NCT03199040 | I | Recruiting | 24 | Biological: Neoantigen DNA vaccine | Triple negative BC | Safety of neoantigen DNA vaccines given alone or in combination with Durvalumab as measured by number of adverse events experienced by patient |
| NCT03639714 | I, II | Recruiting | 214 | Biological: GRT-C901 GRT-R902 | NSCLC; CC; Gastroesophageal; Adenocarcinoma; Urothelial Carcinoma | (1) Incidence of adverse events, serious adverse events, and dose-limiting toxicities; (2) Objective response rate in phase 2 using RECIST v1.1; (3) Identify the recommended Phase 2 dose of GRT-C901 and GRT-R902 |
| NCT03606967 | II | Not yet recruiting | 70 | Biological: Personalized synthetic long peptide vaccine Others: Poly ICLC | Anatomic stage IV BC | Progression-free survival |
| NCT03715985 | I | Recruiting | 25 | Biological: EVAX-01-CAF09b | Malignant melanoma metastatic; NSCLC metastatic; Kidney cancer metastatic | Number and type of reported adverse events |
| NCT03412877 | II | Recruiting | 210 | Biological: Individual patient TCR-transduced PBL | Glioblastoma; NSCLC; Ovarian Cancer; BC; Gastrointestinal/Genitourinary cancer | Response rate |
| NCT03171220 | I, II | Recruiting | 40 | Biological: Neoantigen reactive T cells (NRTs) Other: SHR-1210 | AMST | Number of participants with adverse events |
| NCT03359239 | I | Recruiting | 15 | Biological: PGV001 Other: Poly ICLC | Urothelial/Bladder cancer; Nos | (1) Number of neo-antigens; (2) Number of peptides synthesized; (3) Vaccine Production time; (4) Proportion of consent to tissue acquisition phase; (5) Proportion of subjects eligible for the treatment phase; (6) Number of toxicities |
| NCT03658785 | I, II | Not yet recruiting | 40 | Biological: TIL | RC; MC; Solid tumor | Objective response rate |
| NCT03674073 | I | Recruiting | 24 | Biological: Neoantigen vaccines | HCC | Safety of neoantigen-based DC vaccine as measured by the number of subjects experiencing each type of adverse event according to the National Cancer Institute Common Terminology Criteria for Adverse Events v4.0. |
| NCT01970358 | I | Active, not recruiting | 20 | Biological: Peptides Other: Poly-ICLC | Melanoma | (1) Number of participants experiencing clinical and laboratory adverse events; (2) Number of participants for whom sequencing and analysis leads to identification of at least 10 actionable peptides to initiate vaccine production |
| NCT02287428 | I | Active, not recruiting | 46 | Biological: Personalized neoantigen vaccine | Glioblastoma | (1) Cohorts 1, 1a, 1b, and 1c: Number of participants with adverse events as a measure of safety and tolerability; (2) Cohorts 1d: Number of participants with adverse events as a measure of safety and tolerability; (3) Cohort 1: Number of participants with at least 10 actionable peptides as a measure of study feasibility; (4) Cohort 1: Number of participants who are clinically able to initiate post-RT vaccine therapy within 12 weeks or less from date of surgery |
| NCT03361852 | I | Not yet recruiting | 20 | Biological: NeoVax | Follicular lymphoma | Feasibility of Neovax following 4 weekly doses of Rituximab |
| NCT03219450 | I | Not yet recruiting | 10 | Biological: NeoVax | Lymphocytic leukemia | (1) The proportion of all enrolled patients for whom sequencing and analysis leads to identification of at least 7 actionable peptides to initiate vaccine production; (2) The proportion for whom the time from sample collection to vaccine availability is less than 12 weeks; (3) The number of patients with treatment-limiting toxicities |
All the relevant information of clinical trials was registered on the official website (clinicaltrials.gov). AMST (advanced malignant solid tumor), BC (breast cancer), NSCLC (non-small cell lung cancer), CC (colorectal cancer), HCC (hepatocellular carcinoma), RC (recurrent cancer), MC (metastatic cancer).
TABLE 3.
Completed clinical trials targeting cancer neoantigens.
| ClinicalTrial.gov identifier | Vaccine | Phase | Cancer type | Mechanism | Primary outcome | Adverse events | References |
| NCT00683670 | DC vaccine | I | Melanoma | Autologous DC vaccines directed at tumor amino acid substitutions | Vaccination broadened the antigenic breadth and clonal diversity of anti-tumor immunity | – | Carreno et al., 2015 |
| NCT01970358 | Peptide vaccine | I | Melanoma | Vaccine targeting up to 20 predicted personal tumor neoantigens | Four of six vaccinated patients experienced no recurrence and another two relapse patients experienced CR to anti- PD-1 therapy | Fatigue, rash, injection site reactions, mild flu-like symptoms | Ott et al., 2017 |
| NCT01174121 | TIL vaccine | II | Epithelial cancer | Mutated ERBB2IP-specific CD4+ T cell response | The patient experienced disease stabilization for about 13 months after cell infusion | – | Tran et al., 2014 |
| NCT00204607 | RNA vaccine | I/II | Malignant melanoma | Protamine-stabilized mRNA vaccine coding for Melan-A, Tyrosinase, gp100, Mage-A1, Mage-A3, and Survivin | Vaccine-directed T cells increased, while the frequency of immunosuppressive cells decreased. No adverse events more than grade II were observed | Injection site reactions, fatigue and flu-like symptoms | Weide et al., 2009 |
| NCT02287428 | Peptide vaccine | I | Glioblastoma | Vaccines contained up to 20 long peptides that were divided into pools of 3–5 peptides admixed with poly-ICLC | Median PFS and OS were 7.6 and 16.8 months, respectively | Chills, dizziness, fatigue, flushing, headache, myalgia, nausea and injection site reaction | Keskin et al., 2019 |
| NCT02035956 | RNA vaccine | I | Melanoma | RNA vaccine encoding shared tumor-associated self-antigens | Two patients had a vaccine-related OR among the five patients with metastatic disease, and the other eight patients mostly experienced prolonged DFS | – | Sahin et al., 2017 |
| NCT01174121 | TIL vaccine | I | Metastatic colorectal cancer | TIL vaccine that specifically targeted KRAS G12D | The objective regression of six lung metastases was observed, while one metastatic lesions had progressed | - | Tran et al., 2016 |
DC (dendritic cell), TIL (tumor-infiltrating lymphocyte), CR (complete responses), PFS (progression-free survival), OS (overall survival), OR (objective response), DFS (disease-free survival).
SLP Vaccine
The peptide vaccine is fast and flexible, and it is simple to make an individual cocktail for patients. Antigenic peptide has been widely developed in vaccination due to its strengths including low cost, low toxicity and direct function as pivotal T cell epitope (Li et al., 2014; Kumai et al., 2017). Ott et al. (2017) developed an immunogenic personal neoantigen vaccine (NCT01970358). In this phase I clinical study, 6 patients with resected high-risk melanoma (stage IIIB/C and IVM1a/b) received personalized long peptide vaccines targeting up to 20 neoantigens per patient after sequencing and prioritizing HLA class I prediction (toll-like receptor 3 [TLR3] and melanoma differentiation-associated protein 5 [MDA-5] poly-ICLC were also co-administered as adjuvants). The vaccination activated both CD8+ and CD4+ T cells responses against tumor. Treatment-related adverse events were fatigue, rash, injection site reactions and mild flu-like symptoms, with no major autoimmune toxicity. Encouragingly, four of these six vaccinated patients experienced no recurrence at 25 months after vaccination, and another two relapse patients with metastatic disease later experienced complete responses to anti- PD-1 therapy (Ott et al., 2017). However, the therapeutic efficacy of SLP vaccine is limited by inefficient delivery to desired lymphoid organs as it could rapidly diffuse into the peripheral blood vessels due to its small molecular size (Irvine et al., 2015; Zhu and Zhang, 2017).
DNA Vaccine
DNA vaccine is stable, safe in handling, cost-efficient and easy to manufacture (Prazeres and Monteiro, 2014). Importantly, DNA vaccine can activate both CD4+ and CD8+ T cell response, as well as innate immune response due to the recognition of double stranded DNA structure by cytosolic sensors (Tang and Pietersz, 2009; Ori et al., 2017). Newer DNA vaccines have shown efficacy in the clinic (Trimble et al., 2015; Tebas et al., 2017; Aggarwal et al., 2019). Recently, one group utilized a DNA vaccine platform to target tumor neoantigens in a mouse model. They chose TC1, LLC, and ID8 as tumor models that hardly respond to immune-checkpoint blockade alone. After sequencing these tumors and identifying neoantigens, they designed 12 epitopes per plasmid and these synthetic neoantigen DNA vaccines (SNDVs) were tested in vivo. Not surprisingly, it showed robust T cell immunity. Intriguingly, a larger proportion of CD8+ T cell responses was generated during the SNDVs treatment (25% CD4+ and 75% CD8+ T cell responses) compared with other platforms such as SLP neoantigen vaccines (60% CD4+ and 16% CD8+ T cell responses). Although the epitopes were selected in silico for high MHC class I binding affinity, SLP and RNA neoantigen vaccines generated a higher proportion of MHC class II–restricted CD4+ T cells (Kreiter et al., 2015; Martin et al., 2016; Ott et al., 2017; Sahin et al., 2017). However, DNA vaccine shows poor immunogenicity in human trials (Jorritsma et al., 2016).
RNA Vaccine
The advantages of RNA vaccine include low risk of insertional mutagenesis, direct translation into the cytoplasm and simple and inexpensive manufacturing procedure (Sahin et al., 2014). Kreiter et al. (2015) developed synthetic poly-neo-epitope messenger RNA vaccines after exome sequencing and bioinformatic prioritization in three independent murine tumor models. This vaccination induced complete rejection of established tumors and reshaped the tumor microenvironment (Kreiter et al., 2015). As RNA is the genetic material in many viruses, the human immune system tends to be on high alert for it, which gives an RNA vaccine a unique advantage. “It is its own adjuvant,” Sahin says (Sahin et al., 2017), so he implemented the RNA-based poly-neo-epitope vaccines in 13 patients with stage III-IV melanoma (NCT02035956). Two patients had a vaccine-related objective response among the five patients with metastatic disease, and the other eight patients who had no detectable disease mostly experienced prolonged disease-free survival. Two-thirds of vaccination developed de novo in addition to pre-existing immunity. Weide et al. (2009) also showed that direct injection of protamine-protected mRNA vaccine is feasible and safe; it can also increase the T cell response and decrease immunosuppressive cells in metastatic melanoma patients (NCT00204607). However, the translational efficiency of RNA vaccine remains challenging as only a small portion of administered mRNA can be captured and presented by APCs. Therefore, Sahin and his group administered the RNA vaccine directly into lymph nodes through ultrasound-guided percutaneous injection, noted as intranodal injection (i.n.).
DC Vaccine
Dendritic cells have a key role in presenting antigens to the immune system. DCs are often recognized as the most potent APCs, which are capable of acquiring and processing antigens for presentation to T cells and expressing high levels of costimulatory molecules (Sabado et al., 2017). Therefore, vaccination based on DCs is a promising platform for neoantigen vaccine. Carreno et al. (2015) were the first to report autologous DC vaccines directed at tumor amino acid substitutions (AAS) in three melanoma patients (NCT00683670). They filtered the candidate HLA-A∗ 02: 01 epitopes containing mutations residues after whole exome sequencing and HLA binding prediction and evaluated the MHC-epitope binding using mass spectrometry. Then they filtered precursors of DCs from patients’ bloodstream, matured them and exposed them to synthetic epitopes. The peptide-loaded DCs were then returned to the patients by intravenous infusion. It increased the breadth and diversity of anti-tumor immunity after receiving the DC neoantigen vaccine (Carreno et al., 2015). However, DC vaccine is laborious, costly and need highly skilled technicians for manufacturing (Chen et al., 2016).
Adoptive T Cell Therapy (ACT)
T cell therapy targeting driver mutations is quite attractive, since they are not only specific and biologically important, but also shared between different patients (McGranahan et al., 2015). Currently, KRAS mutations are hot-spot driver mutations and the most frequent KRAS mutant is KRAS G12D that is expressed in ∼45% of pancreatic adenocarcinomas (Bryant et al., 2014) and ∼13% of colorectal cancers (Vaughn et al., 2011). Rosenberg et al. (2016) administered cytotoxic T cells targeting mutant KRAS G12D into a patient with metastatic colorectal cancer (NCT01174121). After whole-exome and transcriptome sequencing of three resected lung lesions, they found that CD8+ T cells in TILs specifically recognized mutant KRAS G12D. Then they selected and expanded TILs that were reactive to the mutant KRAS G12D. The patient received a single infusion of 1.48 × 1011 TILs, which contained 1.11 × 1011 HLA-C∗08:02–restricted CD8+ T cells that specifically targeted KRAS G12D. The objective regression of all seven lung metastases was observed at the first follow-up visit. However, one of these metastatic lesions had progressed when evaluating after 9 months of therapy. The loss of the chromosome 6 haplotype encoding the HLA-C∗08:02 class I MHC molecule resulted in progression after resecting this lesion and sequencing, which provides direct evidence of tumor immune evasion (Tran et al., 2016). Furthermore, the group used this approach to demonstrate that CD4+ T helper 1 (TH1) cells in TILs recognized a mutation in erbb2 interacting protein (ERBB2IP) in a patient with metastatic cholangiocarcinoma. This patient was treated with mutation-reactive TH1 cells twice and experienced tumor regression (NCT01174121) (Tran et al., 2014).
Neoantigen Load Associates With Immune Checkpoint Inhibitors
Antibodies targeting two immune checkpoints, PD-1 and CTLA-4, represent the greatest success of cancer immunotherapy, which can elicit durable antitumor responses in a wide range of malignancies (Hodi et al., 2010; Hamid et al., 2013; Chen and Han, 2015; Postow et al., 2015; Robert et al., 2015a; Zou et al., 2016). The treatment of immune checkpoint inhibitors has improved OS and PFS in many different cancers (Robert et al., 2011; Topalian et al., 2014; Borghaei et al., 2015; Brahmer et al., 2015; Garon et al., 2015; Larkin et al., 2015; Robert et al., 2015b; Sundar et al., 2015; George et al., 2016; Tomita et al., 2017). Accumulating evidence suggests that cancers with higher mutation burden are associated with more survival benefits from both anti- PD-1 and anti- CTLA-4 therapy (Table 4) (Hamid et al., 2013; Asaoka et al., 2015; Rizvi et al., 2015; Andor and Graham, 2016; Erratum for the Report “Genomic correlates of response to CTLA-4 blockade in metastatic melanoma” by Van Allen et al., 2016; Hugo et al., 2016; Matsushita et al., 2016; McGranahan et al., 2016; Morris et al., 2016; Rosenberg et al., 2016). Mutations may increase the possibility of generating immunogenic neoantigens, which facilitate the recognition of cancer cells as foreign (Schumacher and Schreiber, 2015; Riaz and Havel, 2016). Studies in melanoma patients have demonstrated that neoantigen-specific CD8+ and CD4+ T cells in TILs responded to checkpoint blockade therapy (Kvistborg et al., 2014; Lu et al., 2014; Tran et al., 2014; Linnemann et al., 2015), which provides testimony for the hypothesis. Furthermore, neoantigen loss may contribute to acquired resistance through tumor cell elimination or chromosomal deletions during immune checkpoint blockade therapy (Anagnostou et al., 2017); other mechanisms include upregulation of alternate immune checkpoints (Koyama et al., 2016), loss of HLA haplotypes (Maeurer et al., 1996), or somatic mutations in HLA or JAK1/JAK2 genes (Shukla et al., 2015; Garcia-Diaz et al., 2017). However, a proof-of-concept by Nicholas in which cytotoxic chemotherapy–induced subclonal neoantigens were enriched in certain poor responders to immune checkpoint inhibitors was presented (McGranahan et al., 2016). Additionally, gliomas that recurred after treatment with the DNA alkylating agent temozolomide were identified to carry numbers of mutations (Hunter et al., 2006; Cahill et al., 2007), which had a higher mutation burden generated from chemotherapy, but had less clinical benefit. As a result, the association between response to immune checkpoint blockade and neoantigen burden is not linear and clear (Le et al., 2015). Matsushita et al. (2017) demonstrated that the number of neoantigens per missense mutation (neoAg frequency) was an independent predictive factor for PFS in ovarian clear cell carcinoma (OCCC), and the low neoAg frequencies were correlated with increased PFS. High mutation and neoantigen load negatively influenced PFS in multiple myeloma (MM) patients, which had a lower mutational burden (Miller et al., 2017). Luksza et al. (2017) developed a neoantigen fitness model that could describe the evolutionary dynamics of cancer cells and predict tumor response to checkpoint blockade immunotherapy. Further studies are needed regarding the relationship between neoantigen load and checkpoint blockade immunotherapy.
TABLE 4.
Trials defining a TMB threshold for ICB benefit.
| Cancer type | Agents | Methods | Threshold defined | RR | PFS | References |
| NSCLC | Pembrolizumab | WES | 200 mut | 59% versus 12% | NR versus 3.4 months | Rizvi et al., 2015 |
| NSCLC | Anti-PD-1 and anti-PD-L1 | MSKCC and NGS | 7.4 mut/Mb | 38.6% versus 25% | – | Rizvi et al., 2018 |
| NSCLC | Nivolumab and ipilimumab | WES | 158 mut | 51% versus 13% | 17.1 versus 3.7 months | Hellmann et al., 2018a |
| SCLC | Nivolumab and ipilimumab | WES | 248 mut | 46.2% versus 21.3% | 7.8 versus 1.4 months | Hellmann et al., 2018b |
| NSCLC | Nivolumab | WES | >243 mut | 47%versus 23% | HR 0.62 | Carbone et al., 2017 |
| NSCLC | Nivolumab and ipilimumab | FM and NGS | >10 mut/Mb | 45.3% versus 24.6% | 7.1 versus 3.2 months | Hellmann et al., 2018c |
| Melanoma | Anti-CTLA-4 | WES | 100 mut | – | – | Snyder et al., 2014 |
| Melanoma | Nivolumab | WES | 100 mut | – | – | Riaz et al., 2017 |
| Various cancers | Various immunotherapies | FM and NGS | 20 mut/Mb | 58% versus 20% | 12.8 versus 3.3 months | Goodman et al., 2017 |
| UC | Atezolizumab | FM and NGS | 16 mut/Mb | – | – | Balar et al., 2017 |
| UC | Atezolizumab | FM and NGS | >9.65 mut/Mb | – | – | Powles et al., 2018 |
| UC | Nivolumab | WES | ≥170 versus < 85 mut | 31.9% versus 10.9% | 3 versus 2 months | Galsky et al., 2020 |
RR (relative risk), PFS (progression-free survival), NSCLC (non-small-cell lung cancer), SCLC (small-cell lung cancer), UC (urothelial cancer), PD-1 (programmed cell death protein 1), PD-L1 (programmed cell death-ligand 1), CTLA-4 (cytotoxic T-lymphocyte-associated protein 4), WES (whole exome sequencing), NGS (next generation sequencing), FM (foundation medicine), mut (mutation), HR (hazard ratio).
High mutation burden is associated with survival benefits from ICB therapy, but autoantibodies may also predict the efficacy of immune checkpoint inhibitors (ICIs) (De Moel et al., 2019). However, recent studies have demonstrated that autoantibodies correlate with immune checkpoint therapy-induced toxicities (Da Gama et al., 2018; De Moel et al., 2019; Tahir et al., 2019). Since the mechanism of ICB therapy is to mediate non-specific suppression of T cells by negative costimulatory signals, it may break the balance of autoimmunity and lead to the activation of autoreactive B cells that produce autoantibodies (Pardoll, 2012; Oh et al., 2017). As we know, ICB therapy can cause immune-related adverse events (irAEs), referring to the release of distinctive toxicities including pneumonitis, dermatitis, hepatitis, colitis, and hypophysitis (Hodi et al., 2010; Weber et al., 2012; Gao et al., 2015). A recent study found that CD21lo B cells and plasmablasts increased in patients following ICB treatment, and these changes in B cells preceded and correlated with the frequency and timing of irAEs (Das et al., 2018). Besides, one phase I clinical trial including advanced metastatic melanoma patients who received BCG and ipilimumab treatment was suspended due to the occurrence of irAEs. Researchers found profound increases in the repertoire of autoantibodies directed against both selves- and cancer antigens at time points preceded the development of symptomatic toxicity (NCT01838200) (Da Gama et al., 2018). Furthermore, Tahir et al. (2019) also found that anti-GNAL and anti-ITM2B autoantibodies correlated with the development of ICI–related hypophysitis and that anti-CD74 autoantibodies were associated with ICB–induced pneumonitis development. They also tested additional patient samples by enzyme-linked immunosorbent assay to verify these findings (Tahir et al., 2019). These data suggest that autoantibodies under ICB treatment may serve as a predictive biomarker for irAEs.
Challenges
The Challenges With MHC–Peptide-Binding Prediction
Current neoantigen identification techniques are still time consuming and labourious (Tran et al., 2015; Gros et al., 2016). The predictors of immunogenicity are immature (Calis et al., 2013). In addition, since cytotoxic CD8+ T cells are the main killer of cancer cells, the available computational tools can only predict neo-epitopes that bind to MHC class I molecules presented on CD8+ T cells (Nielsen and Andreatta, 2016). Humans have approximately 5,000 alleles encoding MHC class I molecules, with expression of up to six MHC class I molecules (No authors, 2017b) and computational tools cannot predict them all. Intriguingly, even though the epitopes are selected for high MHC class I binding affinity, the neoantigen vaccine trials showed a higher proportion of MHC class II–restricted, CD4+ T cells (Kreiter et al., 2015; Ott et al., 2017). Furthermore, studies demonstrated that CD4+ T cells also recognize a higher number of neo-epitopes than was previously known and can confer potent antitumor activity (Tran et al., 2014; Kreiter et al., 2015). However, it will be more difficult to develop predictive algorithms for MHC class II molecules (Nielsen et al., 2010). First, MHC class II molecules are heterodimers of alpha and beta peptides encoded by four different loci, with three of them being highly polymorphic in the human genome (Robinson et al., 2003). Second, the MHC class II binding groove is open on both ends, presenting longer sequences of amino acids (11–20 amino acids or even longer) than MHC class I molecules (8–11 amino acids) (Babbitt et al., 1985; Bjorkman et al., 1987). Recently, Andreatta et al. (2015) described the method for the quantitative prediction of peptide binding affinity of MHC class II molecules of known sequence.
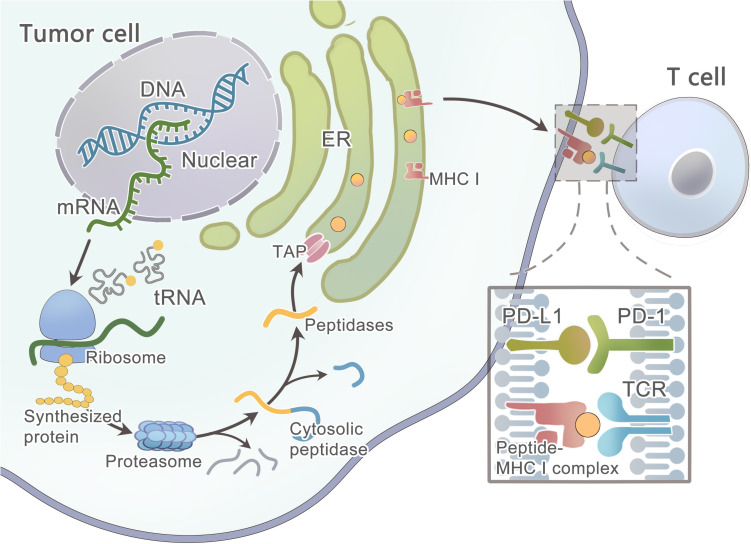
Primarily, the protein that contains the mutated residue is processed by the proteasome (Figure 3) (Nielsen et al., 2005) —a catalytic complex in the cytosol that can cleave the amino acid (AA) sequence to peptides ranging from 3 to 22 AA in length (Kisselev et al., 1999; Rock et al., 2002; Murata et al., 2009). A fraction of the peptides is further trimmed by aminopeptidases and endopeptidases in the cytosol and the endoplasmic reticulum(ER) (Beninga et al., 1998; York et al., 2002; Yan et al., 2006; Rock et al., 2010). Then, this peptide will be transported into the ER lumen by the TAP1/TAP2 transporter to assemble with MHC class I molecules (Peters et al., 2003; Larsen et al., 2007). Finally, the peptide-MHC class I complex can be presented on the cell surface. However, these computational tools hardly consider the endogenous processing and transport of peptides before HLA binding, which results in a high false-positive rate. As studied by Robbins and colleagues, 229 tumor-specific neo-epitopes were predicted in three melanoma patients, but only 11 (4.8%) of these neo-epitopes elicited a T cell response (Robbins et al., 2013). Neo-epitopes can also be produced by an altered MHC class I processing machinery in cancer cells (Van Hall et al., 2006; Seidel et al., 2012; Van Der Burg et al., 2016), which results in a high false-negative rate. Recently, Abelin et al. (2017) developed a new method of liquid chromatography-tandem mass spectrometry (LC–MS/MS) analysis of HLA-associated peptides which takes into account the endogenous processing of peptides, they also developed a new predictor with better algorithm since it is trained on peptide affinity data.
FIGURE 3.
Schematic diagram illustrates the steps involved in tumor neoantigen processing and presentation on MHC class I molecules.
However, some patients may have no expression or aberrated expression of MHC molecules as immune evasion mechanisms (Marincola et al., 2000; Garrido et al., 2010; Leone et al., 2013; Textor et al., 2017). The loss of MHC class I can be detected in the early stage of some cancers (Vermeulen et al., 2005; Van Esch et al., 2014). As studied by Cabrera et al. (2003), β2m mutations and LMP7/TAP2 downregulation are the two main mechanisms in colorectal cancer that are responsible for the loss of MHC surface expression, and thus T cells fail to recognize cancer cells during an immune response. In some tumors, the MAPK pathway may regulate MHC I presentation (Mimura et al., 2013; Bradley et al., 2015; Ebert et al., 2016). After the mutated peptide and MHC class I complex presenting on the tumor cell surface, T cell recognition can occur only when TCRs that have the ability to recognize the mutant epitope exist within the T cell repertoire. Luckily, prior data showed that the T cell repertoire had a diversity of ∼2.5 × 107 (Arstila et al., 1999), and a single TCR was able to recognize up to 106 different MHC/peptide complexes, and thus the immune system could recognize ∼1012 possible foreign epitopes (Mason, 1998), which means the immune system has a strong recognition ability to distinguish even minor variations in MHC/peptide complexes. A study has shown that only certain types of mutations can be missed, such as conservative substitutions at other positions or alterations at the N-terminal peptide residue (Kessels et al., 2004).
Tumor Heterogeneity
Genomic instability and mutational processes can result in extensive tumor heterogeneity in each patient (Gerlinger et al., 2012). First, spontaneous mutations occur during the stages of tumor progression. Second, tumor microenvironments such as T cells can mediate neoantigen immunoediting (Verdegaal et al., 2016) or neoantigen loss. Third, metastatic lesions can involve the distal outgrowth of tumor cells originated from a subclone of the primary tumor; although there is little heterogeneity in driver mutations, there is still considerable epigenetic reprogramming between primary and metastatic tumors as studied in pancreatic ductal adenocarcinoma (PDAC) (Alderton, 2017). Therefore, a single site tumor biopsy may not adequately capture the total number of antigen clones present in the tumors (Sankin et al., 2014). It is well established that intratumor heterogeneity (ITH) correlates with the response of cancer patients to treatments with targeted therapies (Diaz et al., 2012; Shi et al., 2014; Landau et al., 2015). This is only tumor heterogeneity for each individual patient. For different patients and different cancers, the neo-epitopes were rarely shared except for driver mutations, which account for a fraction of mutations. Charoentong et al. (2017) analyzed more than 8,000 patients comprising 20 solid cancers from the TCGA (results available at https://tcia.at/). The pan-cancer analysis showed that the fraction of neo-epitopes generated from driver genes was 7.6%. Only 24 of 911,548 unique predicted neo-epitopes were common in more than 5% of patients (Charoentong et al., 2017). Therefore, neoantigen immunotherapy will probably need to be fully personalized for each patient, and this will be the next generation of precision medicine.
Tumor Suppression Environment
Cancer immunotherapy can offer limited clinical benefit without mitigating the immunosuppressive microenvironment of tumors. It has been increasingly recognized that tumors develop a specialized niche termed the tumor microenvironment (TME) in which tumor cells are protected from therapeutic interventions (Quail and Joyce, 2013). This niche includes fibroblasts (Kalluri and Zeisberg, 2006; Chen and Song, 2018), myeloid suppressor cells (MDSCs) (Goedegebuure et al., 2011; Kumar et al., 2016), regulatory T (Treg) cells (Van Der Burg et al., 2007; Bonertz et al., 2009), tumor-associated macrophages (TAM) (Qian and Pollard, 2010; Panni et al., 2013), lymphocytes, the extracellular matrix (ECM) and abnormal blood and lymphatic vessels (Jain, 2013). For example, high stromal density can limit T cell access to tumor cells and delivery of cytotoxic agents that provide a barrier (Provenzano et al., 2012; Feig et al., 2013; Ozdemir et al., 2014). Cancer cells can also express ligands for inhibitory receptors on T cells and secrete a multitude of chemokines and cytokines to affect antitumor immunity (Walker et al., 2003; Thomas and Massague, 2005). The vaccine strategies can successfully increase the frequency and activity of T cells, but they fail to guarantee that these T cells can exert their function within the tumors. The most important reason is the immune escape mechanisms in cancers, and thus proper co-treatment during vaccination is needed (Arens et al., 2013; Van Der Burg et al., 2016). Other than ICIs, there are multiple inhibitors targeting tumor immunosuppressive factors, including IDO1 inhibitors (Spranger et al., 2014), MEK inhibitors (Ebert et al., 2016), colony-stimulating factor-1 receptor (CSF1R) and chemokine (C-C motif) receptor 2 (CCR2) inhibitors (Mitchem et al., 2013), tumor extracellular matrix and stromal inhibitors (Provenzano et al., 2012), adenosine signaling through the adenosine A2a receptor (A2aR) (Leone et al., 2015), and other metabolic signaling pathways (Pardoll, 2015). In addition, combining such TME modulators with neoantigen-specific therapy may augment antitumor immunity (Melief et al., 2015). As studied by Zhu et al. (2014) CSF1R blockade could significantly improve the efficacy of PD-1 or CTLA-4 antagonists on tumor regressions. Furthermore, among the large number of predicted epitopes, only a minority can be recognized by autologous T cells (Robbins et al., 2013; Van Rooij et al., 2013; Rizvi et al., 2015); therefore, one group has proved that T cells redirected with T cell repertoires of healthy blood donors can efficiently recognize cancer epitopes that are neglected by a patient’s autologous T cells (Stronen et al., 2016).
Conclusion and Perspectives
Personal neoantigen vaccines can elicite strong T cell responses, which not only expand existing neoantigen-specific T cell populations, but also induce a new proportion of specific T cells in cancer patients (Ott et al., 2017). Hence, the identification of neoantigens is of utmost importance to improve cancer immunotherapy and broaden its efficacy to a larger number of patients. Khodadoust et al. (2017) discovered that immunoglobulin neoantigens in human mantle-cell lymphomas and CD4+ T cells specific for the neoantigens could mediate killing of autologous lymphoma cells. Keskin et al. (2019) also demonstrated that a strategy using multi-epitope, personalized neoantigen vaccination is feasible not only for high-risk melanoma (Rizvi et al., 2015; Ott et al., 2017; Sahin et al., 2017) but also for glioblastoma (Keskin et al., 2019), which has a relatively low mutation load. Furthermore, Balachandran et al. (2017) identified MUC16 as immunogenic hotspots in long-term survivors of pancreatic ductal adenocarcinoma, which is a presumed poorly immunogenic and checkpoint blockade-refractory tumor. These results are encouraging, but efforts are needed to tackle those challenges, and we will be very likely to witness these exciting developments in the near future.
Author Contributions
X-JH and XM was a major writer of the manuscript and designed the figures, tables. XW and YW developed the structure of the article, reviewed and edited the manuscript. LY and YP researched the appropriate references and reviewed the manuscript. All the authors read and approved the final manuscript.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding. This work was supported by the National Key Research and Development Program of China under (Grant Numbers 2016YFC1303502 and 2016YFA0201402).
References
- Abelin J. G., Keskin D. B., Sarkizova S., Hartigan C. R., Zhang W., Sidney J., et al. (2017). Mass spectrometry profiling of HLA-associated peptidomes in mono-allelic cells enables more accurate epitope prediction. Immunity 46 315–326. 10.1016/j.immuni.2017.02.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aggarwal C., Cohen R. B., Morrow M. P., Kraynyak K. A., Sylvester A. J., Knoblock D. M., et al. (2019). Immunotherapy targeting HPV16/18 generates potent immune responses in HPV-associated head and neck cancer. Clin. Cancer Res. 25 110–124. 10.1158/1078-0432.ccr-18-1763 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Alderton G. K. (2017). Tumour evolution: epigenetic and genetic heterogeneity in metastasis. Nat. Rev. Cancer 17:141. 10.1038/nrc.2017.11 [DOI] [PubMed] [Google Scholar]
- Alexandrov L. B., Nik-Zainal S., Wedge D. C., Aparicio S. A., Behjati S., Biankin A. V., et al. (2013). Signatures of mutational processes in human cancer. Nature 500 415–421. 10.1038/nature12477 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anagnostou V., Smith K. N., Forde P. M., Niknafs N., Bhattacharya R., White J., et al. (2017). Evolution of neoantigen landscape during immune checkpoint blockade in non-small cell lung cancer. Cancer Discov. 7 264–276. 10.1158/2159-8290.cd-16-0828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andor N., Graham T. A. (2016). Pan-cancer analysis of the extent and consequences of intratumor heterogeneity. Nat. Med. 22 105–113. 10.1038/nm.3984 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreatta M., Karosiene E., Rasmussen M., Stryhn A., Buus S., Nielsen M. (2015). Accurate pan-specific prediction of peptide-MHC class II binding affinity with improved binding core identification. Immunogenetics 67 641–650. 10.1007/s00251-015-0873-y [DOI] [PMC free article] [PubMed] [Google Scholar]
- Andreatta M., Nielsen M. (2016). Gapped sequence alignment using artificial neural networks: application to the MHC class I system. Bioinformatics 32 511–517. 10.1093/bioinformatics/btv639 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Arens R., Van Hall T., Van Der Burg S. H., Ossendorp F., Melief C. J. (2013). Prospects of combinatorial synthetic peptide vaccine-based immunotherapy against cancer. Semin Immunol. 25 182–190. 10.1016/j.smim.2013.04.008 [DOI] [PubMed] [Google Scholar]
- Arstila T. P., Casrouge A., Baron V., Even J., Kanellopoulos J., Kourilsky P. (1999). A direct estimate of the human alphabeta T cell receptor diversity. Science 286 958–961. 10.1126/science.286.5441.958 [DOI] [PubMed] [Google Scholar]
- Asaoka Y., Ijichi H., Koike K. (2015). PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. 373:1979 10.1056/NEJMc1510353 [DOI] [PubMed] [Google Scholar]
- Babbitt B. P., Allen P. M., Matsueda G., Haber E., Unanue E. R. (1985). Binding of immunogenic peptides to Ia histocompatibility molecules. Nature 317 359–361. 10.1038/317359a0 [DOI] [PubMed] [Google Scholar]
- Balachandran V. P., Luksza M., Zhao J. N., Makarov V., Moral J. A., Remark R., et al. (2017). Identification of unique neoantigen qualities in long-term survivors of pancreatic cancer. Nature 551 512–516. 10.1038/nature24462 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Balar A. V., Galsky M. D., Rosenberg J. E., Powles T., Petrylak D. P., Bellmunt J., et al. (2017). Atezolizumab as first-line treatment in cisplatin-ineligible patients with locally advanced and metastatic urothelial carcinoma: a single-arm, multicentre, phase 2 trial. Lancet 389 67–76. 10.1016/s0140-6736(16)32455-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barrick J. E., Lenski R. E. (2013). Genome dynamics during experimental evolution. Nat. Rev. Genet. 14 827–839. 10.1038/nrg3564 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassani-Sternberg M., Braunlein E., Klar R., Engleitner T., Sinitcyn P., Audehm S., et al. (2016). Direct identification of clinically relevant neoepitopes presented on native human melanoma tissue by mass spectrometry. Nat. Commun. 7:13404. 10.1038/ncomms13404 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bassani-Sternberg M., Pletscher-Frankild S., Jensen L. J., Mann M. (2015). Mass spectrometry of human leukocyte antigen class I peptidomes reveals strong effects of protein abundance and turnover on antigen presentation. Mol. Cell Proteomics 14 658–673. 10.1074/mcp.M114.042812 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Belousov P. V., Afanasyeva M. A., Gubernatorova E. O., Bogolyubova A. V., Uvarova A. N., Putlyaeva L. V., et al. (2019). Multi-dimensional immunoproteomics coupled with in vitro recapitulation of oncogenic NRAS(Q61R) identifies diagnostically relevant autoantibody biomarkers in thyroid neoplasia. Cancer Lett. 467 96–106. 10.1016/j.canlet.2019.07.013 [DOI] [PubMed] [Google Scholar]
- Beninga J., Rock K. L., Goldberg A. L. (1998). Interferon-gamma can stimulate post-proteasomal trimming of the N terminus of an antigenic peptide by inducing leucine aminopeptidase. J. Biol. Chem. 273 18734–18742. 10.1074/jbc.273.30.18734 [DOI] [PubMed] [Google Scholar]
- Bentley D. R., Balasubramanian S., Swerdlow H. P., Smith G. P., Milton J., Brown C. G., et al. (2008). Accurate whole human genome sequencing using reversible terminator chemistry. Nature 456 53–59. 10.1038/nature07517 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bentzen A. K., Hadrup S. R. (2017). Evolution of MHC-based technologies used for detection of antigen-responsive T cells. Cancer Immunol. Immunother. 66 657–666. 10.1007/s00262-017-1971-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bentzen A. K., Marquard A. M. (2016). Large-scale detection of antigen-specific T cells using peptide-MHC-I multimers labeled with DNA barcodes. Nat. Biotechnol. 34 1037–1045. 10.1038/nbt.3662 [DOI] [PubMed] [Google Scholar]
- Bjorkman P. J., Saper M. A., Samraoui B., Bennett W. S., Strominger J. L., Wiley D. C. (1987). Structure of the human class I histocompatibility antigen. HLA-A2. Nature 329 506–512. 10.1038/329506a0 [DOI] [PubMed] [Google Scholar]
- Black M. M., Opler S. R., Speer F. D. (1954). Microscopic structure of gastric carcinomas and their regional lymph nodes in relation to survival. Surg. Gynecol. Obstet. 98 725–734. [PubMed] [Google Scholar]
- Bonertz A., Weitz J., Pietsch D. H., Rahbari N. N., Schlude C., Ge Y., et al. (2009). Antigen-specific Tregs control T cell responses against a limited repertoire of tumor antigens in patients with colorectal carcinoma. J. Clin. Invest. 119 3311–3321. 10.1172/jci39608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borghaei H., Paz-Ares L., Horn L., Spigel D. R., Steins M., Ready N. E., et al. (2015). Nivolumab versus Docetaxel in advanced nonsquamous non-small-cell lung cancer. N. Engl. J. Med. 373 1627–1639. 10.1056/NEJMoa1507643 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bradley S. D., Chen Z., Melendez B., Talukder A., Khalili J. S., Rodriguez-Cruz T., et al. (2015). BRAFV600E Co-opts a conserved MHC class I internalization pathway to diminish antigen presentation and CD8+ T-cell recognition of melanoma. Cancer Immunol. Res. 3 602–609. 10.1158/2326-6066.cir-15-0030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brahmer J., Reckamp K. L., Baas P., Crino L., Eberhardt W. E., Poddubskaya E., et al. (2015). Nivolumab versus docetaxel in advanced squamous-cell non-small-cell lung cancer. N. Engl. J. Med. 373 123–135. 10.1056/NEJMoa1504627 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brahmer J. R., Tykodi S. S., Chow L. Q., Hwu W. J., Topalian S. L., Hwu P., et al. (2012). Safety and activity of anti-PD-L1 antibody in patients with advanced cancer. N. Engl. J. Med. 366 2455–2465. 10.1056/NEJMoa1200694 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brass N., Rácz A., Bauer C., Heckel D., Sybrecht G., Meese E. (1999). Role of amplified genes in the production of autoantibodies. Blood 93 2158–2166. 10.1182/blood.v93.7.2158.407a34_2158_2166 [DOI] [PubMed] [Google Scholar]
- Bryant K. L., Mancias J. D., Kimmelman A. C., Der C. J. (2014). KRAS: feeding pancreatic cancer proliferation. Trends Biochem. Sci. 39 91–100. 10.1016/j.tibs.2013.12.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulik-Sullivan B., Busby J., Palmer C. D., Davis M. J., Murphy T., Clark A., et al. (2018). Deep learning using tumor HLA peptide mass spectrometry datasets improves neoantigen identification. Nat. Biotechnol. 18:4313. 10.1038/nbt.4313 [DOI] [PubMed] [Google Scholar]
- Caballero O. L., Chen Y. T. (2009). Cancer/testis (CT) antigens: potential targets for immunotherapy. Cancer Sci. 100 2014–2021. 10.1111/j.1349-7006.2009.01303.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cabrera C. M., Jimenez P., Cabrera T., Esparza C., Ruiz-Cabello F., Garrido F. (2003). Total loss of MHC class I in colorectal tumors can be explained by two molecular pathways: beta2-microglobulin inactivation in MSI-positive tumors and LMP7/TAP2 downregulation in MSI-negative tumors. Tissue Antigens 61 211–219. 10.1034/j.1399-0039.2003.00020.x [DOI] [PubMed] [Google Scholar]
- Cahill D. P., Levine K. K., Betensky R. A., Codd P. J., Romany C. A., Reavie L. B., et al. (2007). Loss of the mismatch repair protein MSH6 in human glioblastomas is associated with tumor progression during temozolomide treatment. Clin. Cancer Res. 13 2038–2045. 10.1158/1078-0432.ccr-06-2149 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Calis J. J., Maybeno M., Greenbaum J. A., Weiskopf D., De Silva A. D., Sette A., et al. (2013). Properties of MHC class I presented peptides that enhance immunogenicity. PLoS Comput. Biol. 9:e1003266. 10.1371/journal.pcbi.1003266 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cancer Genome Atlas (TCGA) Research Network (2008). Comprehensive genomic characterization defines human glioblastoma genes and core pathways. Nature 455 1061–1068. 10.1038/nature07385 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carbone D. P., Reck M., Paz-Ares L., Creelan B., Horn L., Steins M., et al. (2017). First-line nivolumab in stage IV or recurrent non-small-cell lung cancer. N. Engl. J. Med 376 2415–2426. 10.1056/NEJMoa1613493 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carreno B. M., Magrini V., Becker-Hapak M., Kaabinejadian S., Hundal J., Petti A. A., et al. (2015). Cancer immunotherapy. A dendritic cell vaccine increases the breadth and diversity of melanoma neoantigen-specific T cells. Science 348 803–808. 10.1126/science.aaa3828 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Castle J. C., Kreiter S., Diekmann J., Lower M., van de Roemer N., De Graaf J., et al. (2012). Exploiting the mutanome for tumor vaccination. Cancer Res. 72 1081–1091. 10.1158/0008-5472.can-11-3722 [DOI] [PubMed] [Google Scholar]
- Chan T. A., Wolchok J. D., Snyder A. (2015). Genetic basis for clinical response to CTLA-4 blockade in melanoma. N. Engl. J. Med. 373:1984. 10.1056/NEJMc1508163 [DOI] [PubMed] [Google Scholar]
- Charoentong P., Finotello F., Angelova M., Mayer C., Efremova M., Rieder D., et al. (2017). Pan-cancer immunogenomic analyses reveal genotype-immunophenotype relationships and predictors of response to checkpoint blockade. Cell Rep. 18 248–262. 10.1016/j.celrep.2016.12.019 [DOI] [PubMed] [Google Scholar]
- Chen L., Han X. (2015). Anti-PD-1/PD-L1 therapy of human cancer: past, present, and future. J. Clin. Invest. 125 3384–3391. 10.1172/jci80011 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen P., Liu X., Sun Y., Zhou P., Wang Y., Zhang Y. (2016). Dendritic cell targeted vaccines: recent progresses and challenges. Hum. Vaccin. Immunother. 12 612–622. 10.1080/21645515.2015.1105415 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen X., Song E. (2018). Turning foes to friends: targeting cancer-associated fibroblasts. Nat. Rev. Drug Discovery 18 99–115. 10.1038/s41573-018-0004-1 [DOI] [PubMed] [Google Scholar]
- Chen Y. T., Scanlan M. J., Sahin U., Türeci O., Gure A. O., Tsang S., et al. (1997). A testicular antigen aberrantly expressed in human cancers detected by autologous antibody screening. Proc. Natl. Acad. Sci. U.S.A. 94 1914–1918. 10.1073/pnas.94.5.1914 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Choi M., Scholl U. I., Ji W., Liu T., Tikhonova I. R., Zumbo P., et al. (2009). Genetic diagnosis by whole exome capture and massively parallel DNA sequencing. Proc. Natl. Acad. Sci. U.S.A. 106 19096–19101. 10.1073/pnas.0910672106 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen C. J., Gartner J. J., Horovitz-Fried M., Shamalov K., Trebska-McGowan K., Bliskovsky V. V., et al. (2015). Isolation of neoantigen-specific T cells from tumor and peripheral lymphocytes. J. Clin. Invest. 125 3981–3991. 10.1172/jci82416 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coulie P. G., Lehmann F., Lethe B., Herman J., Lurquin C., Andrawiss M., et al. (1995). A mutated intron sequence codes for an antigenic peptide recognized by cytolytic T lymphocytes on a human melanoma. Proc. Natl. Acad. Sci. U.S.A. 92 7976–7980. 10.1073/pnas.92.17.7976 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Da Gama D. J., Parakh S., Andrews M. C., Woods K., Pasam A., et al. (2018). Autoantibodies may predict immune-related toxicity: results from a phase I study of intralesional bacillus calmette-guérin followed by ipilimumab in patients with advanced metastatic melanoma. Front. Immunol. 9:411. 10.3389/fimmu.2018.00411 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dai L., Li J., Tsay J. J., Yie T. A., Munger J. S., Pass H., et al. (2017). Identification of autoantibodies to ECH1 and HNRNPA2B1 as potential biomarkers in the early detection of lung cancer. Oncoimmunology 6:e1310359. 10.1080/2162402x.2017.1310359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- D’Angelo S. P., Melchiori L., Merchant M. S., Bernstein D., Glod J., Kaplan R., et al. (2018). Antitumor activity associated with prolonged persistence of adoptively transferred NY-ESO-1 (c259)T cells in synovial sarcoma. Cancer Discov. 8 944–957. 10.1158/2159-8290.cd-17-1417 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Das R., Bar N., Ferreira M., Newman A. M., Zhang L., Bailur J. K., et al. (2018). Early B cell changes predict autoimmunity following combination immune checkpoint blockade. J. Clin. Invest. 128 715–720. 10.1172/jci96798 [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Moel E. C., Rozeman E. A., Kapiteijn E. H., Verdegaal E. M. E., Grummels A., Bakker J. A. (2019). Autoantibody development under treatment with immune-checkpoint inhibitors. Cancer Immunol. Res. 7 6–11. 10.1158/2326-6066.cir-18-0245 [DOI] [PubMed] [Google Scholar]
- De Plaen E., Lurquin C., Van Pel A., Mariame B., Szikora J. P., Wolfel T., et al. (1988). Immunogenic (tum-) variants of mouse tumor P815: cloning of the gene of tum- antigen P91A and identification of the tum- mutation. Proc. Natl. Acad. Sci. U.S.A. 85 2274–2278. 10.1073/pnas.85.7.2274 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Diaz L. A., Jr., Williams R. T., Wu J., Kinde I., Hecht J. R., Berlin J., et al. (2012). The molecular evolution of acquired resistance to targeted EGFR blockade in colorectal cancers. Nature 486 537–540. 10.1038/nature11219 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding L., Getz G., Wheeler D. A., Mardis E. R., McLellan M. D., Cibulskis K., et al. (2008). Somatic mutations affect key pathways in lung adenocarcinoma. Nature 455 1069–1075. 10.1038/nature07423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ding L., Wendl M. C., McMichael J. F., Raphael B. J. (2014). Expanding the computational toolbox for mining cancer genomes. Nat. Rev. Genet. 15 556–570. 10.1038/nrg3767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Drmanac R., Sparks A. B., Callow M. J., Halpern A. L., Burns N. L., Kermani B. G., et al. (2010). Human genome sequencing using unchained base reads on self-assembling DNA nanoarrays. Science 327 78–81. 10.1126/science.1181498 [DOI] [PubMed] [Google Scholar]
- Ebert P. J. R., Cheung J., Yang Y., McNamara E., Hong R., Moskalenko M., et al. (2016). MAP kinase inhibition promotes T cell and anti-tumor activity in combination with pd-l1 checkpoint blockade. Immunity 44 609–621. 10.1016/j.immuni.2016.01.024 [DOI] [PubMed] [Google Scholar]
- Feig C., Jones J. O., Kraman M., Wells R. J., Deonarine A., Chan D. S., et al. (2013). Targeting CXCL12 from FAP-expressing carcinoma-associated fibroblasts synergizes with anti-PD-L1 immunotherapy in pancreatic cancer. Proc. Natl. Acad. Sci. U.S.A. 110 20212–20217. 10.1073/pnas.1320318110 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng H., Shuda M., Chang Y., Moore P. S. (2008). Clonal integration of a polyomavirus in human Merkel cell carcinoma. Science 319 1096–1100. 10.1126/science.1152586 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Forbes S. A., Beare D., Gunasekaran P., Leung K., Bindal N., Boutselakis H., et al. (2015). COSMIC: exploring the world’s knowledge of somatic mutations in human cancer. Nucleic Acids Res. 43 D805–D811. 10.1093/nar/gku1075 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galsky M., Saci A., Szabo P. M., Han G. C., Grossfeld G. D., Collette S., et al. (2020). Nivolumab in patients with advanced platinum-resistant urothelial carcinoma: efficacy, safety, and biomarker analyses with extended follow-up from checkmate 275. Clin. Cancer Res. [Epub ahead of print]. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gao J., He Q., Subudhi S., Aparicio A., Zurita-Saavedra A., Lee D. H., et al. (2015). Review of immune-related adverse events in prostate cancer patients treated with ipilimumab: MD Anderson experience. Oncogene 34 5411–5417. 10.1038/onc.2015.5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia-Diaz A., Shin D. S., Moreno B. H., Saco J., Escuin-Ordinas H., Rodriguez G. A., et al. (2017). Interferon receptor signaling pathways regulating PD-L1 and PD-L2 expression. Cell Rep. 19 1189–1201. 10.1016/j.celrep.2017.04.031 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garon E. B., Rizvi N. A., Hui R., Leighl N., Balmanoukian A. S., Eder J. P., et al. (2015). Pembrolizumab for the treatment of non-small-cell lung cancer. N. Engl. J. Med. 372 2018–2028. 10.1056/NEJMoa1501824 [DOI] [PubMed] [Google Scholar]
- Garraway L. A., Lander E. S. (2013). Lessons from the cancer genome. Cell 153 17–37. 10.1016/j.cell.2013.03.002 [DOI] [PubMed] [Google Scholar]
- Garrido F., Cabrera T., Aptsiauri N. (2010). “Hard” and “soft” lesions underlying the HLA class I alterations in cancer cells: implications for immunotherapy. Int. J. Cancer 127 249–256. 10.1002/ijc.25270 [DOI] [PubMed] [Google Scholar]
- George S., Motzer R. J., Hammers H. J., Redman B. G., Kuzel T. M., Tykodi S. S., et al. (2016). Safety and efficacy of nivolumab in patients with metastatic renal cell carcinoma treated beyond progression: a subgroup analysis of a randomized clinical trial. JAMA Oncol. 2 1179–1186. 10.1001/jamaoncol.2016.0775 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerlinger M., Rowan A. J., Horswell S., Math M., Larkin J., Endesfelder D., et al. (2012). Intratumor heterogeneity and branched evolution revealed by multiregion sequencing. N. Engl. J. Med. 366 883–892. 10.1056/NEJMoa1113205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gillison M. L., Koch W. M., Capone R. B., Spafford M., Westra W. H., Wu L., et al. (2000). Evidence for a causal association between human papillomavirus and a subset of head and neck cancers. J. Natl. Cancer Inst. 92 709–720. 10.1093/jnci/92.9.709 [DOI] [PubMed] [Google Scholar]
- Goedegebuure P., Mitchem J. B., Porembka M. R., Tan M. C., Belt B. A., Wang-Gillam A., et al. (2011). Myeloid-derived suppressor cells: general characteristics and relevance to clinical management of pancreatic cancer. Curr. Cancer Drug Targets 11 734–751. 10.2174/156800911796191024 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman A. M., Kato S., Bazhenova L., Patel S. P., Frampton G. M., Miller V., et al. (2017). Tumor mutational burden as an independent predictor of response to immunotherapy in diverse cancers. Mol. Cancer Ther. 16 2598–2608. 10.1158/1535-7163.mct-17-0386 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Greenman C., Stephens P., Smith R., Dalgliesh G. L., Hunter C., Bignell G., et al. (2007). Patterns of somatic mutation in human cancer genomes. Nature 446 153–158. 10.1038/nature05610 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gros A., Parkhurst M. R., Tran E., Pasetto A., Robbins P. F., Ilyas S., et al. (2016). Prospective identification of neoantigen-specific lymphocytes in the peripheral blood of melanoma patients. Nat. Med. 22 433–438. 10.1038/nm.4051 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gubin M. M., Artyomov M. N., Mardis E. R., Schreiber R. D. (2015). Tumor neoantigens: building a framework for personalized cancer immunotherapy. J. Clin. Invest. 125 3413–3421. 10.1172/jci80008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gubin M. M., Zhang X., Schuster H., Caron E., Ward J. P., Noguchi T., et al. (2014). Checkpoint blockade cancer immunotherapy targets tumour-specific mutant antigens. Nature 515 577–581. 10.1038/nature13988 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haber D. A., Settleman J. (2007). Cancer: drivers and passengers. Nature 446 145–146. 10.1038/446145a [DOI] [PubMed] [Google Scholar]
- Hadrup S. R., Bakker A. H., Shu C. J., Andersen R. S., van Veluw J., Hombrink P., et al. (2009). Parallel detection of antigen-specific T-cell responses by multidimensional encoding of MHC multimers. Nat. Methods 6 520–526. 10.1038/nmeth.1345 [DOI] [PubMed] [Google Scholar]
- Hamid O., Robert C., Daud A., Hodi F. S., Hwu W. J., Kefford R., et al. (2013). Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N. Engl. J. Med. 369 134–144. 10.1056/NEJMoa1305133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heemskerk B., Kvistborg P., Schumacher T. N. (2013). The cancer antigenome. Embo J. 32 194–203. 10.1038/emboj.2012.333 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Helleday T., Eshtad S., Nik-Zainal S. (2014). Mechanisms underlying mutational signatures in human cancers. Nat. Rev. Genet. 15 585–598. 10.1038/nrg3729 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellmann M. D., Callahan M. K., Awad M. M., Calvo E., Ascierto P. A., Atmaca A., et al. (2018a). Tumor mutational burden and efficacy of nivolumab monotherapy and in combination with ipilimumab in small-cell lung cancer. Cancer Cell 33 853–861.e854. 10.1016/j.ccell.2018.04.001853-861.e854 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellmann M. D., Ciuleanu T. E., Pluzanski A., Lee J. S., Otterson G. A., Audigier-Valette C., et al. (2018b). Nivolumab plus ipilimumab in lung cancer with a high tumor mutational burden. N. Engl. J. Med. 378 2093–2104. 10.1056/NEJMoa1801946 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hellmann M. D., Nathanson T., Rizvi H., Creelan B. C., Sanchez-Vega F., Ahuja A., et al. (2018c). Genomic features of response to combination immunotherapy in patients with advanced non-small-cell lung cancer. Cancer Cell 33 843–852.e844. 10.1016/j.ccell.2018.03.018843-852.e844 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hodi F. S., O’Day S. J., McDermott D. F., Weber R. W., Sosman J. A., Haanen J. B., et al. (2010). Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 363 711–723. 10.1056/NEJMoa1003466 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hudson T. J., Anderson W., Artez A., Barker A. D., Bell C., Bernabe R. R., et al. (2010). International network of cancer genome projects. Nature 464 993–998. 10.1038/nature08987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hugo W., Zaretsky J. M., Sun L., Song C., Moreno B. H., Hu-Lieskovan S., et al. (2016). Genomic and transcriptomic features of response to Anti-PD-1 therapy in metastatic melanoma. Cell 165 35–44. 10.1016/j.cell.2016.02.065 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hundal J., Carreno B. M., Petti A. A., Linette G. P., Griffith O. L., Mardis E. R., et al. (2016). pVAC-Seq: a genome-guided in silico approach to identifying tumor neoantigens. Genome Med. 8 11. 10.1186/s13073-016-0264-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hundal J., Kiwala S. (2019). Accounting for proximal variants improves neoantigen prediction. Nat. Genet. 51 175–179. 10.1038/s41588-018-0283-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hunter C., Smith R., Cahill D. P., Stephens P., Stevens C., Teague J., et al. (2006). A hypermutation phenotype and somatic MSH6 mutations in recurrent human malignant gliomas after alkylator chemotherapy. Cancer Res. 66 3987–3991. 10.1158/0008-5472.can-06-0127 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irvine D. J., Hanson M. C., Rakhra K., Tokatlian T. (2015). Synthetic Nanoparticles for Vaccines and Immunotherapy. Chem. Rev. 115 11109–11146. 10.1021/acs.chemrev.5b00109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jäger D., Stockert E., Scanlan M. J., Güre A. O., Jäger E., Knuth A., et al. (1999). Cancer-testis antigens and ING1 tumor suppressor gene product are breast cancer antigens: characterization of tissue-specific ING1 transcripts and a homologue gene. Cancer Res. 59 6197–6204. [PubMed] [Google Scholar]
- Jain R. K. (2013). Normalizing tumor microenvironment to treat cancer: bench to bedside to biomarkers. J. Clin. Oncol. 31 2205–2218. 10.1200/jco.2012.46.3653 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jo N. R., Lee K. J., Shin Y. B. (2016). Enzyme-coupled nanoplasmonic biosensing of cancer markers in human serum. Biosens. Bioelectron. 81 324–333. 10.1016/j.bios.2016.03.009 [DOI] [PubMed] [Google Scholar]
- Jones S., Zhang X., Parsons D. W., Lin J. C., Leary R. J., Angenendt P., et al. (2008). Core signaling pathways in human pancreatic cancers revealed by global genomic analyses. Science 321 1801–1806. 10.1126/science.1164368 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jorritsma S. H. T., Gowans E. J., Grubor-Bauk B., Wijesundara D. K. (2016). Delivery methods to increase cellular uptake and immunogenicity of DNA vaccines. Vaccine 34 5488–5494. 10.1016/j.vaccine.2016.09.062 [DOI] [PubMed] [Google Scholar]
- Jurtz V., Paul S. (2017). NetMHCpan-4.0: improved peptide-MHC class I interaction predictions integrating eluted ligand and peptide binding affinity data. J. Immunol. 199 3360–3368. 10.4049/jimmunol.1700893 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahles A., Lehmann K. V., Toussaint N. C., Hüser M., Stark S. G., Sachsenberg T., et al. (2018). Comprehensive analysis of alternative splicing across tumors from 8,705 patients. Cancer Cell 34 211–224.e216. 10.1016/j.ccell.2018.07.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kahles A., Ong C. S., Zhong Y., Rätsch G. (2016). SplAdder: identification, quantification and testing of alternative splicing events from RNA-Seq data. Bioinformatics 32 1840–1847. 10.1093/bioinformatics/btw076 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalluri R., Zeisberg M. (2006). Fibroblasts in cancer. Nat. Rev. Cancer 6 392–401. 10.1038/nrc1877 [DOI] [PubMed] [Google Scholar]
- Kan Z., Jaiswal B. S., Stinson J., Janakiraman V., Bhatt D., Stern H. M., et al. (2010). Diverse somatic mutation patterns and pathway alterations in human cancers. Nature 466 869–873. 10.1038/nature09208 [DOI] [PubMed] [Google Scholar]
- Keskin D. B., Anandappa A. J., Sun J., Tirosh I., Mathewson N. D., Li S., et al. (2019). Neoantigen vaccine generates intratumoral T cell responses in phase Ib glioblastoma trial. Nature 565 234–239. 10.1038/s41586-018-0792-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kessels H. W., de Visser K. E., Tirion F. H., Coccoris M., Kruisbeek A. M., Schumacher T. N. (2004). The impact of self-tolerance on the polyclonal CD8+ T cell repertoire. J. Immunol. 172 2324–2331. 10.4049/jimmunol.172.4.2324 [DOI] [PubMed] [Google Scholar]
- Khodadoust M. S., Olsson N., Wagar L. E., Haabeth O. A., Chen B., Swaminathan K., et al. (2017). Antigen presentation profiling reveals recognition of lymphoma immunoglobulin neoantigens. Nature 543 723–727. 10.1038/nature21433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim S., Kim H. S., Kim E., Lee M. G., Shin E. C., Paik S., et al. (2018). Neopepsee: accurate genome-level prediction of neoantigens by harnessing sequence and amino acid immunogenicity information. Ann. Oncol. 29 1030–1036. 10.1093/annonc/mdy022 [DOI] [PubMed] [Google Scholar]
- Kisselev A. F., Akopian T. N., Woo K. M., Goldberg A. L. (1999). The sizes of peptides generated from protein by mammalian 26 and 20 S proteasomes. Implications for understanding the degradative mechanism and antigen presentation. J. Biol. Chem. 274 3363–3371. 10.1074/jbc.274.6.3363 [DOI] [PubMed] [Google Scholar]
- Koyama S., Akbay E. A., Li Y. Y., Herter-Sprie G. S., Buczkowski K. A., Richards W. G., et al. (2016). Adaptive resistance to therapeutic PD-1 blockade is associated with upregulation of alternative immune checkpoints. Nat. Commun. 7:10501. 10.1038/ncomms10501 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kreiter S., Vormehr M., van de Roemer N., Diken M., Lower M., Diekmann J., et al. (2015). Mutant MHC class II epitopes drive therapeutic immune responses to cancer. Nature 520 692–696. 10.1038/nature14426 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumai T., Kobayashi H., Harabuchi Y., Celis E. (2017). Peptide vaccines in cancer-old concept revisited. Curr. Opin. Immunol. 45 1–7. 10.1016/j.coi.2016.11.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kumar V., Patel S., Tcyganov E., Gabrilovich D. I. (2016). The nature of myeloid-derived suppressor cells in the tumor microenvironment. Trends Immunol. 37 208–220. 10.1016/j.it.2016.01.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kvistborg P., Philips D., Kelderman S., Hageman L., Ottensmeier C., Joseph-Pietras D., et al. (2014). Anti-CTLA-4 therapy broadens the melanoma-reactive CD8+ T cell response. Sci. Transl. Med. 6:254ra128. 10.1126/scitranslmed.3008918 [DOI] [PubMed] [Google Scholar]
- Landau D. A., Tausch E., Taylor-Weiner A. N., Stewart C., Reiter J. G., Bahlo J., et al. (2015). Mutations driving CLL and their evolution in progression and relapse. Nature 526 525–530. 10.1038/nature15395 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Larkin J., Hodi F. S., Wolchok J. D. (2015). Combined nivolumab and ipilimumab or monotherapy in untreated melanoma. N. Engl. J. Med. 373 1270–1271. 10.1056/NEJMc1509660 [DOI] [PubMed] [Google Scholar]
- Larsen M. V., Lundegaard C., Lamberth K., Buus S., Lund O., Nielsen M. (2007). Large-scale validation of methods for cytotoxic T-lymphocyte epitope prediction. BMC Bioinformatics 8:424. 10.1186/1471-2105-8-424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Le D. T., Uram J. N., Wang H., Bartlett B. R., Kemberling H., Eyring A. D., et al. (2015). PD-1 blockade in tumors with mismatch-repair deficiency. N. Engl. J. Med. 372 2509–2520. 10.1056/NEJMoa1500596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee J. U., Nguyen A. H., Sim S. J. (2015). A nanoplasmonic biosensor for label-free multiplex detection of cancer biomarkers. Biosens. Bioelectron. 74 341–346. 10.1016/j.bios.2015.06.059 [DOI] [PubMed] [Google Scholar]
- Lee W., Jiang Z., Liu J., Haverty P. M., Guan Y., Stinson J., et al. (2010). The mutation spectrum revealed by paired genome sequences from a lung cancer patient. Nature 465 473–477. 10.1038/nature09004 [DOI] [PubMed] [Google Scholar]
- Lennerz V., Fatho M., Gentilini C., Frye R. A., Lifke A., Ferel D., et al. (2005). The response of autologous T cells to a human melanoma is dominated by mutated neoantigens. Proc. Natl. Acad. Sci. U.S.A. 102 16013–16018. 10.1073/pnas.0500090102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leone P., Shin E. C., Perosa F., Vacca A., Dammacco F., Racanelli V. (2013). MHC class I antigen processing and presenting machinery: organization, function, and defects in tumor cells. J. Natl. Cancer Inst. 105 1172–1187. 10.1093/jnci/djt184 [DOI] [PubMed] [Google Scholar]
- Leone R. D., Lo Y. C., Powell J. D. (2015). A2aR antagonists: Next generation checkpoint blockade for cancer immunotherapy. Comput. Struct. Biotechnol. J. 13 265–272. 10.1016/j.csbj.2015.03.008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ley T. J., Mardis E. R., Ding L., Fulton B., McLellan M. D., Chen K., et al. (2008). DNA sequencing of a cytogenetically normal acute myeloid leukaemia genome. Nature 456 66–72. 10.1038/nature07485 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L., Goedegebuure P., Mardis E. R., Ellis M. J., Zhang X., Herndon J. M., et al. (2011). Cancer genome sequencing and its implications for personalized cancer vaccines. Cancers (Basel) 3 4191–4211. 10.3390/cancers3044191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W., Joshi M. D., Singhania S., Ramsey K. H., Murthy A. K. (2014). Peptide vaccine: progress and challenges. Vaccines (Basel) 2 515–536. 10.3390/vaccines2030515 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lin C. L., Lo W. F., Lee T. H., Ren Y., Hwang S. L., Cheng Y. F., et al. (2002). Immunization with Epstein-Barr Virus (EBV) peptide-pulsed dendritic cells induces functional CD8+ T-cell immunity and may lead to tumor regression in patients with EBV-positive nasopharyngeal carcinoma. Cancer Res. 62 6952–6958. [PubMed] [Google Scholar]
- Lin H. H., Ray S., Tongchusak S., Reinherz E. L., Brusic V. (2008). Evaluation of MHC class I peptide binding prediction servers: applications for vaccine research. BMC Immunol. 9:8. 10.1186/1471-2172-9-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Linnemann C., van Buuren M. M., Bies L., Verdegaal E. M., Schotte R., Calis J. J., et al. (2015). High-throughput epitope discovery reveals frequent recognition of neo-antigens by CD4+ T cells in human melanoma. Nat. Med. 21 81–85. 10.1038/nm.3773 [DOI] [PubMed] [Google Scholar]
- Lu Y. C., Robbins P. F. (2016). Cancer immunotherapy targeting neoantigens. Semin Immunol. 28 22–27. 10.1016/j.smim.2015.11.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lu Y. C., Yao X., Crystal J. S., Li Y. F., El-Gamil M., Gross C., et al. (2014). Efficient identification of mutated cancer antigens recognized by T cells associated with durable tumor regressions. Clin. Cancer Res. 20 3401–3410. 10.1158/1078-0432.ccr-14-0433 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luksza M., Riaz N., Makarov V., Balachandran V. P., Hellmann M. D., Solovyov A., et al. (2017). A neoantigen fitness model predicts tumour response to checkpoint blockade immunotherapy. Nature 551 517–520. 10.1038/nature24473 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lurquin C., Van Pel A., Mariame B., De Plaen E., Szikora J. P., Janssens C., et al. (1989). Structure of the gene of tum- transplantation antigen P91A: the mutated exon encodes a peptide recognized with Ld by cytolytic T cells. Cell 58 293–303. 10.1016/0092-8674(89)90844-1 [DOI] [PubMed] [Google Scholar]
- Maeurer M. J., Gollin S. M., Storkus W. J., Swaney W., Karbach J., Martin D., et al. (1996). Tumor escape from immune recognition: loss of HLA-A2 melanoma cell surface expression is associated with a complex rearrangement of the short arm of chromosome 6. Clin. Cancer Res. 2 641–652. [PubMed] [Google Scholar]
- Margulies M., Egholm M., Altman W. E., Attiya S., Bader J. S., Bemben L. A., et al. (2005). Genome sequencing in microfabricated high-density picolitre reactors. Nature 437 376–380. 10.1038/nature03959 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marincola F. M., Jaffee E. M., Hicklin D. J., Ferrone S. (2000). Escape of human solid tumors from T-cell recognition: molecular mechanisms and functional significance. Adv. Immunol. 74 181–273. 10.1016/s0065-2776(08)60911-6 [DOI] [PubMed] [Google Scholar]
- Martin S. D., Brown S. D., Wick D. A., Nielsen J. S., Kroeger D. R., Twumasi-Boateng K., et al. (2016). Low mutation burden in ovarian cancer may limit the utility of neoantigen-targeted vaccines. PLoS One 11:e0155189. 10.1371/journal.pone.0155189 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason D. (1998). A very high level of crossreactivity is an essential feature of the T-cell receptor. Immunol. Today 19 395–404. 10.1016/s0167-5699(98)01299-7 [DOI] [PubMed] [Google Scholar]
- Matsushita H., Hasegawa K., Oda K., Yamamoto S., Nishijima A., Imai Y., et al. (2017). The frequency of neoantigens per somatic mutation rather than overall mutational load or number of predicted neoantigens per se is a prognostic factor in ovarian clear cell carcinoma. Oncoimmunology 6:e1338996. 10.1080/2162402x.2017.1338996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsushita H., Sato Y., Karasaki T., Nakagawa T., Kume H., Ogawa S., et al. (2016). Neoantigen load, antigen presentation machinery, and immune signatures determine prognosis in clear cell renal cell carcinoma. Cancer Immunol. Res. 4 463–471. 10.1158/2326-6066.cir-15-0225 [DOI] [PubMed] [Google Scholar]
- Matsushita H., Vesely M. D., Koboldt D. C., Rickert C. G., Uppaluri R., Magrini V. J., et al. (2012). Cancer exome analysis reveals a T-cell-dependent mechanism of cancer immunoediting. Nature 482 400–404. 10.1038/nature10755 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGranahan N., Favero F., de Bruin E. C., Birkbak N. J., Szallasi Z., Swanton C. (2015). Clonal status of actionable driver events and the timing of mutational processes in cancer evolution. Sci. Transl. Med. 7:283ra254. 10.1126/scitranslmed.aaa1408 [DOI] [PMC free article] [PubMed] [Google Scholar]
- McGranahan N., Furness A. J., Rosenthal R., Ramskov S., Lyngaa R., Saini S. K., et al. (2016). Clonal neoantigens elicit T cell immunoreactivity and sensitivity to immune checkpoint blockade. Science 351 1463–1469. 10.1126/science.aaf1490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Melero I., Gaudernack G., Gerritsen W., Huber C., Parmiani G., Scholl S., et al. (2014). Therapeutic vaccines for cancer: an overview of clinical trials. Nat. Rev. Clin. Oncol. 11 509–524. 10.1038/nrclinonc.2014.111 [DOI] [PubMed] [Google Scholar]
- Melief C. J., van Hall T., Arens R., Ossendorp F., Van Der Burg S. H. (2015). Therapeutic cancer vaccines. J. Clin. Invest. 125 3401–3412. 10.1172/jci80009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merlo A., Turrini R., Dolcetti R., Martorelli D., Muraro E., Comoli P., et al. (2010). The interplay between Epstein-Barr virus and the immune system: a rationale for adoptive cell therapy of EBV-related disorders. Haematologica 95 1769–1777. 10.3324/haematol.2010.023689 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyerson M., Gabriel S., Getz G. (2010). Advances in understanding cancer genomes through second-generation sequencing. Nat. Rev. Genet. 11 685–696. 10.1038/nrg2841 [DOI] [PubMed] [Google Scholar]
- Miller A., Asmann Y., Cattaneo L., Braggio E., Keats J., Auclair D., et al. (2017). High somatic mutation and neoantigen burden are correlated with decreased progression-free survival in multiple myeloma. Blood Cancer J. 7:e612. 10.1038/bcj.2017.94 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mimura K., Shiraishi K., Mueller A., Izawa S., Kua L. F., So J., et al. (2013). The MAPK pathway is a predominant regulator of HLA-A expression in esophageal and gastric cancer. J. Immunol. 191 6261–6272. 10.4049/jimmunol.1301597 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mitchem J. B., Brennan D. J., Knolhoff B. L., Belt B. A., Zhu Y., Sanford D. E., et al. (2013). Targeting tumor-infiltrating macrophages decreases tumor-initiating cells, relieves immunosuppression, and improves chemotherapeutic responses. Cancer Res. 73 1128–1141. 10.1158/0008-5472.can-12-2731 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Monach P. A., Meredith S. C., Siegel C. T., Schreiber H. (1995). A unique tumor antigen produced by a single amino acid substitution. Immunity 2 45–59. 10.1016/1074-7613(95)90078-0 [DOI] [PubMed] [Google Scholar]
- Morris L. G., Riaz N., Desrichard A., Senbabaoglu Y., Hakimi A. A., Makarov V., et al. (2016). Pan-cancer analysis of intratumor heterogeneity as a prognostic determinant of survival. Oncotarget 7 10051–10063. 10.18632/oncotarget.7067 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrissy A. S., Morin R. D., Delaney A., Zeng T., McDonald H., Jones S., et al. (2009). Next-generation tag sequencing for cancer gene expression profiling. Genome Res. 19 1825–1835. 10.1101/gr.094482.109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Morrow M. P., Yan J., Sardesai N. Y. (2013). Human papillomavirus therapeutic vaccines: targeting viral antigens as immunotherapy for precancerous disease and cancer. Expert Rev. Vaccines 12 271–283. 10.1586/erv.13.23 [DOI] [PubMed] [Google Scholar]
- Murata S., Yashiroda H., Tanaka K. (2009). Molecular mechanisms of proteasome assembly. Nat. Rev. Mol. Cell Biol. 10 104–115. 10.1038/nrm2630 [DOI] [PubMed] [Google Scholar]
- Ng S. B., Turner E. H., Robertson P. D., Flygare S. D., Bigham A. W., Lee C., et al. (2009). Targeted capture and massively parallel sequencing of 12 human exomes. Nature 461 272–276. 10.1038/nature08250 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen M., Andreatta M. (2016). NetMHCpan-3.0; improved prediction of binding to MHC class I molecules integrating information from multiple receptor and peptide length datasets. Genome Med. 8:33. 10.1186/s13073-016-0288-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen M., Lund O., Buus S., Lundegaard C. (2010). MHC class II epitope predictive algorithms. Immunology 130 319–328. 10.1111/j.1365-2567.2010.03268.x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen M., Lundegaard C., Blicher T., Lamberth K., Harndahl M., Justesen S., et al. (2007). NetMHCpan, a method for quantitative predictions of peptide binding to any HLA-A and -B locus protein of known sequence. PLoS One 2:e796. 10.1371/journal.pone.0000796 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nielsen M., Lundegaard C., Lund O., Kesmir C. (2005). The role of the proteasome in generating cytotoxic T-cell epitopes: insights obtained from improved predictions of proteasomal cleavage. Immunogenetics 57 33–41. 10.1007/s00251-005-0781-7 [DOI] [PubMed] [Google Scholar]
- Nilsen T. W., Graveley B. R. (2010). Expansion of the eukaryotic proteome by alternative splicing. Nature 463 457–463. 10.1038/nature08909 [DOI] [PMC free article] [PubMed] [Google Scholar]
- No authors (2017a). Modeling cancer mutations in 3-D. Cancer Discov. 7 787–788. 10.1158/2159-8290.cd-nb2017-091 [DOI] [PubMed] [Google Scholar]
- No authors (2017b). The problem with neoantigen prediction. Nat. Biotechnol. 35:97. 10.1038/nbt.3800 [DOI] [PubMed] [Google Scholar]
- O’Donnell T. J., Rubinsteyn A., Bonsack M., Riemer A. B., Laserson U., Hammerbacher J. (2018). MHCflurry: open-source class I MHC binding affinity prediction. Cell Syst. 7 129–132.e124. 10.1016/j.cels.2018.05.014129-132.e124 [DOI] [PubMed] [Google Scholar]
- O’Farrell P. H. (1975). High resolution two-dimensional electrophoresis of proteins. J. Biol. Chem. 250 4007–4021. [PMC free article] [PubMed] [Google Scholar]
- Oh D. Y., Cham J., Zhang L., Fong G., Kwek S. S., Klinger M., et al. (2017). Immune toxicities elicted by CTLA-4 blockade in cancer patients are associated with early diversification of the T-cell repertoire. Cancer Res. 77 1322–1330. 10.1158/0008-5472.can-16-2324 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Old L. J., Boyse E. A. (1964). IMMUNOLOGY OF EXPERIMENTAL TUMORS. Annu. Rev. Med. 15 167–186. 10.1146/annurev.me.15.020164.001123 [DOI] [PubMed] [Google Scholar]
- Ori D., Murase M., Kawai T. (2017). Cytosolic nucleic acid sensors and innate immune regulation. Int. Rev. Immunol. 36 74–88. 10.1080/08830185.2017.1298749 [DOI] [PubMed] [Google Scholar]
- Ott P. A., Hu Z., Keskin D. B., Shukla S. A., Sun J., Bozym D. J., et al. (2017). An immunogenic personal neoantigen vaccine for patients with melanoma. Nature 547 217–221. 10.1038/nature22991 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ozdemir B. C., Pentcheva-Hoang T., Carstens J. L., Zheng X., Wu C. C., Simpson T. R., et al. (2014). Depletion of carcinoma-associated fibroblasts and fibrosis induces immunosuppression and accelerates pancreas cancer with reduced survival. Cancer Cell 25 719–734. 10.1016/j.ccr.2014.04.005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pagani F., Baralle F. E. (2004). Genomic variants in exons and introns: identifying the splicing spoilers. Nat. Rev. Genet. 5 389–396. 10.1038/nrg1327 [DOI] [PubMed] [Google Scholar]
- Panni R. Z., Linehan D. C., DeNardo D. G. (2013). Targeting tumor-infiltrating macrophages to combat cancer. Immunotherapy 5 1075–1087. 10.2217/imt.13.102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardoll D. (2015). Cancer and the immune system: basic concepts and targets for intervention. Semin. Oncol. 42 523–538. 10.1053/j.seminoncol.2015.05.003 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardoll D. M. (2012). The blockade of immune checkpoints in cancer immunotherapy. Nat. Rev. Cancer 12 252–264. 10.1038/nrc3239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Park E., Pan Z., Zhang Z., Lin L., Xing Y. (2018). The expanding landscape of alternative splicing variation in human populations. Am. J. Hum. Genet. 102 11–26. 10.1016/j.ajhg.2017.11.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons D. W., Jones S., Zhang X., Lin J. C., Leary R. J., Angenendt P., et al. (2008). An integrated genomic analysis of human glioblastoma multiforme. Science 321 1807–1812. 10.1126/science.1164382 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parsons D. W., Li M., Zhang X., Jones S., Leary R. J., Lin J. C., et al. (2011). The genetic landscape of the childhood cancer medulloblastoma. Science 331 435–439. 10.1126/science.1198056 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pena-Diaz J., Bregenhorn S., Ghodgaonkar M., Follonier C., Artola-Boran M., Castor D., et al. (2017). Noncanonical mismatch repair as a source of genomic instability in human cells. Mol Cell 67:162. 10.1016/j.molcel.2017.06.026 [DOI] [PubMed] [Google Scholar]
- Peng X., Xu X., Wang Y., Hawke D. H., Yu S., Han L., et al. (2018). A-to-I RNA editing contributes to proteomic diversity in cancer. Cancer Cell 33:817–828.e817. 10.1016/j.ccell.2018.03.026817-828.e817 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters B., Bulik S., Tampe R., Van Endert P. M., Holzhutter H. G. (2003). Identifying MHC class I epitopes by predicting the TAP transport efficiency of epitope precursors. J. Immunol. 171 1741–1749. 10.4049/jimmunol.171.4.1741 [DOI] [PubMed] [Google Scholar]
- Picardi E., D’Erchia A. M., Lo G. C., Pesole G. (2017). REDIportal: a comprehensive database of A-to-I RNA editing events in humans. Nucleic Acids Res. 45 D750–D757. 10.1093/nar/gkw767 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pleasance E. D., Cheetham R. K., Stephens P. J., McBride D. J., Humphray S. J., Greenman C. D., et al. (2010a). A comprehensive catalogue of somatic mutations from a human cancer genome. Nature 463 191–196. 10.1038/nature08658 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pleasance E. D., Stephens P. J., O’Meara S., McBride D. J., Meynert A., Jones D., et al. (2010b). A small-cell lung cancer genome with complex signatures of tobacco exposure. Nature 463 184–190. 10.1038/nature08629 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollard H. B., Srivastava M., Eidelman O., Jozwik C., Rothwell S. W., Mueller G. P., et al. (2007). Protein microarray platforms for clinical proteomics. Proteomics Clin. Appl. 1 934–952. 10.1002/prca.200700154 [DOI] [PubMed] [Google Scholar]
- Postow M. A., Chesney J., Pavlick A. C., Robert C., Grossmann K., McDermott D., et al. (2015). Nivolumab and ipilimumab versus ipilimumab in untreated melanoma. N. Engl. J. Med. 372 2006–2017. 10.1056/NEJMoa1414428 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Powles T., Durán I., van der Heijden M. S., Loriot Y., Vogelzang N. J., De Giorgi U., et al. (2018). Atezolizumab versus chemotherapy in patients with platinum-treated locally advanced or metastatic urothelial carcinoma (IMvigor211): a multicentre, open-label, phase 3 randomised controlled trial. Lancet 391 748–757. 10.1016/s0140-6736(17)33297-x [DOI] [PubMed] [Google Scholar]
- Prazeres D. M. F., Monteiro G. A. (2014). Plasmid Biopharmaceuticals. Microbiol. Spectr. 2:1128. 10.1128/microbiolspec.PLAS-0022-2014 [DOI] [PubMed] [Google Scholar]
- Prehn R. T., Main J. M. (1957). Immunity to methylcholanthrene-induced sarcomas. J. Natl. Cancer Inst. 18 769–778. [PubMed] [Google Scholar]
- Provenzano P. P., Cuevas C., Chang A. E., Goel V. K., Von Hoff D. D., Hingorani S. R. (2012). Enzymatic targeting of the stroma ablates physical barriers to treatment of pancreatic ductal adenocarcinoma. Cancer Cell 21 418–429. 10.1016/j.ccr.2012.01.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qian B. Z., Pollard J. W. (2010). Macrophage diversity enhances tumor progression and metastasis. Cell 141 39–51. 10.1016/j.cell.2010.03.014 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Quail D. F., Joyce J. A. (2013). Microenvironmental regulation of tumor progression and metastasis. Nat. Med. 19 1423–1437. 10.1038/nm.3394 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rajasagi M., Shukla S. A., Fritsch E. F., Keskin D. B., DeLuca D., Carmona E., et al. (2014). Systematic identification of personal tumor-specific neoantigens in chronic lymphocytic leukemia. Blood 124 453–462. 10.1182/blood-2014-04-567933 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ramaswami G., Li J. B. (2014). RADAR: a rigorously annotated database of A-to-I RNA editing. Nucleic Acids Res. 42 D109–D113. 10.1093/nar/gkt996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rammensee H., Bachmann J., Emmerich N. P., Bachor O. A., Stevanovic S. (1999). SYFPEITHI: database for MHC ligands and peptide motifs. Immunogenetics 50 213–219. 10.1007/s002510050595 [DOI] [PubMed] [Google Scholar]
- Restifo N. P., Dudley M. E., Rosenberg S. A. (2012). Adoptive immunotherapy for cancer: harnessing the T cell response. Nat. Rev. Immunol. 12 269–281. 10.1038/nri3191 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riaz N., Havel J. J. (2016). Recurrent SERPINB3 and SERPINB4 mutations in patients who respond to anti-CTLA4 immunotherapy. Nat. Genet. 48 1327–1329. 10.1038/ng.3677 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Riaz N., Havel J. J., Makarov V., Desrichard A., Urba W. J., Sims J. S., et al. (2017). Tumor and microenvironment evolution during immunotherapy with nivolumab. Cell 171 934–949.e916. 10.1016/j.cell.2017.09.028934-949.e916 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rizvi H., Sanchez-Vega F., La K., Chatila W., Jonsson P., Halpenny D., et al. (2018). Molecular determinants of response to anti-programmed cell death (PD)-1 and anti-programmed death-ligand 1 (PD-L1) blockade in patients with non-small-cell lung cancer profiled with targeted next-generation sequencing. J. Clin. Oncol. 36 633–641. 10.1200/jco.2017.75.3384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rizvi N. A., Hellmann M. D., Snyder A., Kvistborg P., Makarov V., Havel J. J., et al. (2015). Cancer immunology. Mutational landscape determines sensitivity to PD-1 blockade in non-small cell lung cancer. Science 348 124–128. 10.1126/science.aaa1348 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robbins P. F., Lu Y. C., El-Gamil M., Li Y. F., Gross C., Gartner J., et al. (2013). Mining exomic sequencing data to identify mutated antigens recognized by adoptively transferred tumor-reactive T cells. Nat. Med. 19 747–752. 10.1038/nm.3161 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robert C., Long G. V., Brady B., Dutriaux C., Maio M., Mortier L., et al. (2015a). Nivolumab in previously untreated melanoma without BRAF mutation. N. Engl. J. Med. 372 320–330. 10.1056/NEJMoa1412082 [DOI] [PubMed] [Google Scholar]
- Robert C., Schachter J., Long G. V., Arance A., Grob J. J., Mortier L., et al. (2015b). Pembrolizumab versus ipilimumab in advanced melanoma. N. Engl. J. Med. 372 2521–2532. 10.1056/NEJMoa1503093 [DOI] [PubMed] [Google Scholar]
- Robert C., Thomas L., Bondarenko I., O’Day S., Weber J., Garbe C., et al. (2011). Ipilimumab plus dacarbazine for previously untreated metastatic melanoma. N. Engl. J. Med. 364 2517–2526. 10.1056/NEJMoa1104621 [DOI] [PubMed] [Google Scholar]
- Robinson J., Halliwell J. A., McWilliam H., Lopez R., Parham P., Marsh S. G. (2013). The IMGT/HLA database. Nucleic Acids Res. 41 D1222–D1227. 10.1093/nar/gks949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robinson J., Waller M. J., Parham P., de Groot N., Bontrop R., Kennedy L. J., et al. (2003). IMGT/HLA and IMGT/MHC: sequence databases for the study of the major histocompatibility complex. Nucleic Acids Res. 31 311–314. 10.1093/nar/gkg070 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rock K. L., Farfan-Arribas D. J., Shen L. (2010). Proteases in MHC class I presentation and cross-presentation. J. Immunol. 184 9–15. 10.4049/jimmunol.0903399 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rock K. L., York I. A., Saric T., Goldberg A. L. (2002). Protein degradation and the generation of MHC class I-presented peptides. Adv. Immunol. 80 1–70. 10.1016/s0065-2776(02)80012-8 [DOI] [PubMed] [Google Scholar]
- Rosenberg J. E., Hoffman-Censits J., Powles T., van der Heijden M. S., Balar A. V., Necchi A., et al. (2016). Atezolizumab in patients with locally advanced and metastatic urothelial carcinoma who have progressed following treatment with platinum-based chemotherapy: a single-arm, multicentre, phase 2 trial. Lancet 387 1909–1920. 10.1016/s0140-6736(16)00561-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg S. A. (2011). Cell transfer immunotherapy for metastatic solid cancer–what clinicians need to know. Nat. Rev. Clin. Oncol. 8 577–585. 10.1038/nrclinonc.2011.116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rosenberg S. A., Yang J. C., Sherry R. M., Kammula U. S., Hughes M. S., Phan G. Q., et al. (2011). Durable complete responses in heavily pretreated patients with metastatic melanoma using T-cell transfer immunotherapy. Clin. Cancer Res. 17 4550–4557. 10.1158/1078-0432.ccr-11-0116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabado R. L., Balan S., Bhardwaj N. (2017). Dendritic cell-based immunotherapy. Cell Res. 27 74–95. 10.1038/cr.2016.157 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabatino M., Hu J., Sommariva M., Gautam S., Fellowes V., Hocker J. D., et al. (2016). Generation of clinical-grade CD19-specific CAR-modified CD8+ memory stem cells for the treatment of human B-cell malignancies. Blood 128 519–528. 10.1182/blood-2015-11-683847 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sahin U., Derhovanessian E., Miller M., Kloke B. P., Simon P., Löwer M., et al. (2017). Personalized RNA mutanome vaccines mobilize poly-specific therapeutic immunity against cancer. Nature 547 222–226. 10.1038/nature23003 [DOI] [PubMed] [Google Scholar]
- Sahin U., Karikó K., Türeci Ö. (2014). mRNA-based therapeutics–developing a new class of drugs. Nat. Rev. Drug Discov. 13 759–780. 10.1038/nrd4278 [DOI] [PubMed] [Google Scholar]
- Sankin A., Hakimi A. A., Mikkilineni N., Ostrovnaya I., Silk M. T., Liang Y., et al. (2014). The impact of genetic heterogeneity on biomarker development in kidney cancer assessed by multiregional sampling. Cancer Med. 3 1485–1492. 10.1002/cam4.293 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Scanlan M. J., Chen Y. T., Williamson B., Gure A. O., Stockert E., Gordan J. D., et al. (1998). Characterization of human colon cancer antigens recognized by autologous antibodies. Int. J. Cancer 76 652–658. [DOI] [PubMed] [Google Scholar]
- Schumacher T. N., Schreiber R. D. (2015). Neoantigens in cancer immunotherapy. Science 348 69–74. 10.1126/science.aaa4971 [DOI] [PubMed] [Google Scholar]
- Segal N. H., Parsons D. W., Peggs K. S., Velculescu V., Kinzler K. W., Vogelstein B., et al. (2008). Epitope landscape in breast and colorectal cancer. Cancer Res. 68 889–892. 10.1158/0008-5472.can-07-3095 [DOI] [PubMed] [Google Scholar]
- Seidel U. J., Oliveira C. C., Lampen M. H., Hall T. (2012). A novel category of antigens enabling CTL immunity to tumor escape variants: Cinderella antigens. Cancer Immunol. Immunother. 61 119–125. 10.1007/s00262-011-1160-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma P., Allison J. P. (2015). The future of immune checkpoint therapy. Science 348 56–61. 10.1126/science.aaa8172 [DOI] [PubMed] [Google Scholar]
- Sharp P. A. (1994). Split genes and RNA splicing. Cell 77 805–815. 10.1016/0092-8674(94)90130-9 [DOI] [PubMed] [Google Scholar]
- Shen L., Zhang J., Lee H. (2019). RNA transcription and splicing errors as a source of cancer frameshift neoantigens for vaccines. Sci. Rep. 9:14184. 10.1038/s41598-019-50738-4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shendure J., Porreca G. J., Reppas N. B., Lin X., McCutcheon J. P., Rosenbaum A. M., et al. (2005). Accurate multiplex polony sequencing of an evolved bacterial genome. Science 309 1728–1732. 10.1126/science.1117389 [DOI] [PubMed] [Google Scholar]
- Shi H., Hugo W., Kong X., Hong A., Koya R. C., Moriceau G., et al. (2014). Acquired resistance and clonal evolution in melanoma during BRAF inhibitor therapy. Cancer Discov. 4 80–93. 10.1158/2159-8290.cd-13-0642 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shukla S. A., Rooney M. S., Rajasagi M., Tiao G., Dixon P. M., Lawrence M. S., et al. (2015). Comprehensive analysis of cancer-associated somatic mutations in class I HLA genes. Nat. Biotechnol. 33 1152–1158. 10.1038/nbt.3344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sjoblom T., Jones S., Wood L. D., Parsons D. W., Lin J., Barber T. D., et al. (2006). The consensus coding sequences of human breast and colorectal cancers. Science 314 268–274. 10.1126/science.1133427 [DOI] [PubMed] [Google Scholar]
- Smart A. C., Margolis C. A. (2018). Intron retention is a source of neoepitopes in cancer. Nat. Biotechnol. 36 1056–1058. 10.1038/nbt.4239 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Snyder A., Makarov V., Merghoub T., Yuan J., Zaretsky J. M., Desrichard A., et al. (2014). Genetic basis for clinical response to CTLA-4 blockade in melanoma. N. Engl. J. Med. 371 2189–2199. 10.1056/NEJMoa1406498 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spranger S., Koblish H. K., Horton B., Scherle P. A., Newton R., Gajewski T. F. (2014). Mechanism of tumor rejection with doublets of CTLA-4, PD-1/PD-L1, or IDO blockade involves restored IL-2 production and proliferation of CD8(+) T cells directly within the tumor microenvironment. J. Immunother. Cancer. 2:3. 10.1186/2051-1426-2-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stratton M. R. (2011). Exploring the genomes of cancer cells: progress and promise. Science 331 1553–1558. 10.1126/science.1204040 [DOI] [PubMed] [Google Scholar]
- Stratton M. R., Campbell P. J., Futreal P. A. (2009). The cancer genome. Nature 458 719–724. 10.1038/nature07943 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stronen E., Toebes M., Kelderman S., van Buuren M. M., Yang W., van Rooij N., et al. (2016). Targeting of cancer neoantigens with donor-derived T cell receptor repertoires. Science 352 1337–1341. 10.1126/science.aaf2288 [DOI] [PubMed] [Google Scholar]
- Sundar R., Cho B. C., Brahmer J. R., Soo R. A. (2015). Nivolumab in NSCLC: latest evidence and clinical potential. Ther. Adv. Med. Oncol. 7 85–96. 10.1177/1758834014567470 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Szolek A., Schubert B., Mohr C., Sturm M., Feldhahn M., Kohlbacher O. (2014). OptiType: precision HLA typing from next-generation sequencing data. Bioinformatics 30 3310–3316. 10.1093/bioinformatics/btu548 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tahir S. A., Gao J., Miura Y., Blando J., Tidwell R. S. S., Zhao H., et al. (2019). Autoimmune antibodies correlate with immune checkpoint therapy-induced toxicities. Proc. Natl. Acad. Sci U.S.A. 116 22246–22251. 10.1073/pnas.1908079116 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Talbot S. J., Crawford D. H. (2004). Viruses and tumours–an update. Eur. J. Cancer 40 1998–2005. 10.1016/j.ejca.2003.11.039 [DOI] [PubMed] [Google Scholar]
- Tang C. K., Pietersz G. A. (2009). Intracellular detection and immune signaling pathways of DNA vaccines. Expert. Rev. Vaccines 8 1161–1170. 10.1586/erv.09.79 [DOI] [PubMed] [Google Scholar]
- Tebas P., Roberts C. C., Muthumani K., Reuschel E. L., Kudchodkar S. B., Zaidi F. I., et al. (2017). Safety and immunogenicity of an anti-zika virus DNA vaccine–preliminary report. N. Engl. J. Med. 4:1056. 10.1056/NEJMoa1708120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Textor A., Schmidt K., Kloetzel P. M., Weissbrich B., Perez C., Charo J., et al. (2017). Correction: Preventing tumor escape by targeting a post-proteasomal trimming independent epitope. J. Exp. Med. 214:567. 10.1084/jem.2016063601122017c [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomas D. A., Massague J. (2005). TGF-beta directly targets cytotoxic T cell functions during tumor evasion of immune surveillance. Cancer Cell 8 369–380. 10.1016/j.ccr.2005.10.012 [DOI] [PubMed] [Google Scholar]
- Tian J., Wang Z., Mei S., Yang N., Yang Y., Ke J., et al. (2019). CancerSplicingQTL: a database for genome-wide identification of splicing QTLs in human cancer. Nucleic Acids Res. 47 D909–D916. 10.1093/nar/gky954 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tjalsma H., Schaeps R. M., Swinkels D. W. (2008). Immunoproteomics: from biomarker discovery to diagnostic applications. Proteomics Clin. Appl. 2 167–180. 10.1002/prca.200780012 [DOI] [PubMed] [Google Scholar]
- Tomita Y., Fukasawa S., Shinohara N., Kitamura H., Oya M., Eto M., et al. (2017). Nivolumab versus everolimus in advanced renal cell carcinoma: Japanese subgroup analysis from the CheckMate 025 study. Jpn. J. Clin. Oncol. 47 639–646. 10.1093/jjco/hyx049 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topalian S. L., Hodi F. S., Brahmer J. R., Gettinger S. N., Smith D. C., McDermott D. F., et al. (2012). Safety, activity, and immune correlates of anti-PD-1 antibody in cancer. N. Engl. J. Med. 366 2443–2454. 10.1056/NEJMoa1200690 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Topalian S. L., Sznol M., McDermott D. F., Kluger H. M., Carvajal R. D., Sharfman W. H., et al. (2014). Survival, durable tumor remission, and long-term safety in patients with advanced melanoma receiving nivolumab. J. Clin. Oncol. 32 1020–1030. 10.1200/jco.2013.53.0105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Torre L. A., Bray F., Siegel R. L., Ferlay J., Lortet-Tieulent J., Jemal A. (2015). Global cancer statistics, 2012. CA Cancer J. Clin. 65 87–108. 10.3322/caac.21262 [DOI] [PubMed] [Google Scholar]
- Totoki Y., Tatsuno K., Yamamoto S., Arai Y., Hosoda F., Ishikawa S., et al. (2011). High-resolution characterization of a hepatocellular carcinoma genome. Nat. Genet. 43 464–469. 10.1038/ng.804 [DOI] [PubMed] [Google Scholar]
- Tran E., Ahmadzadeh M., Lu Y. C., Gros A., Turcotte S., Robbins P. F., et al. (2015). Immunogenicity of somatic mutations in human gastrointestinal cancers. Science 350 1387–1390. 10.1126/science.aad1253 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran E., Robbins P. F., Lu Y. C., Prickett T. D., Gartner J. J., Jia L., et al. (2016). T-cell transfer therapy targeting mutant KRAS in cancer. N. Engl. J. Med. 375 2255–2262. 10.1056/NEJMoa1609279 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tran E., Turcotte S., Gros A., Robbins P. F., Lu Y. C., Dudley M. E., et al. (2014). Cancer immunotherapy based on mutation-specific CD4+ T cells in a patient with epithelial cancer. Science 344 641–645. 10.1126/science.1251102 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trimble C. L., Morrow M. P., Kraynyak K. A., Shen X., Dallas M., Yan J., et al. (2015). Safety, efficacy, and immunogenicity of VGX-3100, a therapeutic synthetic DNA vaccine targeting human papillomavirus 16 and 18 E6 and E7 proteins for cervical intraepithelial neoplasia 2/3: a randomised, double-blind, placebo-controlled phase 2b trial. Lancet 386 2078–2088. 10.1016/s0140-6736(15)00239-1 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trucco L. D., Mundra P. A., Hogan K., Garcia-Martinez P., Viros A., Mandal A. K., et al. (2018). Ultraviolet radiation-induced DNA damage is prognostic for outcome in melanoma. Nat. Med. 25 221–224. 10.1038/s41591-018-0265-6 [DOI] [PubMed] [Google Scholar]
- Türeci O., Sahin U., Vollmar E., Siemer S., Göttert E., Seitz G., et al. (1998a). Human carbonic anhydrase XII: cDNA cloning, expression, and chromosomal localization of a carbonic anhydrase gene that is overexpressed in some renal cell cancers. Proc. Natl. Acad. Sci. U.S.A. 95 7608–7613. 10.1073/pnas.95.13.7608 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Türeci O., Sahin U., Zwick C., Koslowski M., Seitz G., Pfreundschuh M. (1998b). Identification of a meiosis-specific protein as a member of the class of cancer/testis antigens. Proc. Natl. Acad. Sci. U.S.A. 95 5211–5216. 10.1073/pnas.95.9.5211 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Allen E. M., Miao D., Schilling B., Shukla S. A., Blank C., Zimmer L., et al. (2016). Erratum for the Report Genomic correlates of response to CTLA-4 blockade in metastatic melanoma. Science 352:6283. 10.1126/science.aaf8264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Der Burg S. H., Arens R., Ossendorp F., van Hall T., Melief C. J. (2016). Vaccines for established cancer: overcoming the challenges posed by immune evasion. Nat. Rev. Cancer 16 219–233. 10.1038/nrc.2016.16 [DOI] [PubMed] [Google Scholar]
- Van Der Burg S. H., Piersma S. J., de Jong A., van der Hulst J. M., Kwappenberg K. M., van den Hende M., et al. (2007). Association of cervical cancer with the presence of CD4+ regulatory T cells specific for human papillomavirus antigens. Proc. Natl. Acad. Sci. U.S.A. 104 12087–12092. 10.1073/pnas.0704672104 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Esch E. M., Tummers B., Baartmans V., Osse E. M., Ter Haar N., Trietsch M. D., et al. (2014). Alterations in classical and nonclassical HLA expression in recurrent and progressive HPV-induced usual vulvar intraepithelial neoplasia and implications for immunotherapy. Int. J. Cancer 135 830–842. 10.1002/ijc.28713 [DOI] [PubMed] [Google Scholar]
- Van Hall T., Wolpert E. Z., van Veelen P., Laban S., van der Veer M., Roseboom M., et al. (2006). Selective cytotoxic T-lymphocyte targeting of tumor immune escape variants. Nat. Med. 12 417–424. 10.1038/nm1381 [DOI] [PubMed] [Google Scholar]
- Van Rooij N., van Buuren M. M., Philips D., Velds A., Toebes M., Heemskerk B., et al. (2013). Tumor exome analysis reveals neoantigen-specific T-cell reactivity in an ipilimumab-responsive melanoma. J. Clin. Oncol. 31 e439–e442. 10.1200/jco.2012.47.7521 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vaughn C. P., Zobell S. D., Furtado L. V., Baker C. L., Samowitz W. S. (2011). Frequency of KRAS, BRAF, and NRAS mutations in colorectal cancer. Genes Chromosomes Cancer 50 307–312. 10.1002/gcc.20854 [DOI] [PubMed] [Google Scholar]
- Verdegaal E. M., de Miranda N. F., Visser M., Harryvan T., van Buuren M. M., Andersen R. S., et al. (2016). Neoantigen landscape dynamics during human melanoma-T cell interactions. Nature 536 91–95. 10.1038/nature18945 [DOI] [PubMed] [Google Scholar]
- Vermeulen C. F., Jordanova E. S., Zomerdijk-Nooijen Y. A., Ter Haar N. T., Peters A. A., Fleuren G. J. (2005). Frequent HLA class I loss is an early event in cervical carcinogenesis. Hum. Immunol. 66 1167–1173. 10.1016/j.humimm.2005.10.011 [DOI] [PubMed] [Google Scholar]
- Vogelstein B., Papadopoulos N., Velculescu V. E., Zhou S., Diaz L. A., Jr., Kinzler K. W. (2013). Cancer genome landscapes. Science 339 1546–1558. 10.1126/science.1235122 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walboomers J. M., Jacobs M. V., Manos M. M., Bosch F. X., Kummer J. A., Shah K. V., et al. (1999). Human papillomavirus is a necessary cause of invasive cervical cancer worldwide. J. Pathol. 189 12–19. [DOI] [PubMed] [Google Scholar]
- Walker M. R., Kasprowicz D. J., Gersuk V. H., Benard A., Van Landeghen M., Buckner J. H., et al. (2003). Induction of FoxP3 and acquisition of T regulatory activity by stimulated human CD4+CD25- T cells. J. Clin. Invest. 112 1437–1443. 10.1172/jci19441 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward J. P., Gubin M. M., Schreiber R. D. (2016). The role of neoantigens in naturally occurring and therapeutically induced immune responses to cancer. Adv. Immunol. 130 25–74. 10.1016/bs.ai.2016.01.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weber J. S., Kähler K. C., Hauschild A. (2012). Management of immune-related adverse events and kinetics of response with ipilimumab. J. Clin. Oncol. 30 2691–2697. 10.1200/jco.2012.41.6750 [DOI] [PubMed] [Google Scholar]
- Wei X., Walia V., Lin J. C., Teer J. K., Prickett T. D., Gartner J., et al. (2011). Exome sequencing identifies GRIN2A as frequently mutated in melanoma. Nat. Genet. 43 442–446. 10.1038/ng.810 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weide B., Pascolo S., Scheel B., Derhovanessian E., Pflugfelder A., Eigentler T. K., et al. (2009). Direct injection of protamine-protected mRNA: results of a phase 1/2 vaccination trial in metastatic melanoma patients. J. Immunother. 32 498–507. 10.1097/CJI.0b013e3181a00068 [DOI] [PubMed] [Google Scholar]
- Weir B., Zhao X., Meyerson M. (2004). Somatic alterations in the human cancer genome. Cancer Cell 6 433–438. 10.1016/j.ccr.2004.11.004 [DOI] [PubMed] [Google Scholar]
- Wheeler D. A., Srinivasan M., Egholm M., Shen Y., Chen L., McGuire A., et al. (2008). The complete genome of an individual by massively parallel DNA sequencing. Nature 452 872–876. 10.1038/nature06884 [DOI] [PubMed] [Google Scholar]
- Wick D. A., Webb J. R., Nielsen J. S., Martin S. D., Kroeger D. R., Milne K., et al. (2014). Surveillance of the tumor mutanome by T cells during progression from primary to recurrent ovarian cancer. Clin. Cancer Res. 20 1125–1134. 10.1158/1078-0432.ccr-13-2147 [DOI] [PubMed] [Google Scholar]
- Wolfel T., Hauer M., Schneider J., Serrano M., Wolfel C., Klehmann-Hieb E., et al. (1995). A p16INK4a-insensitive CDK4 mutant targeted by cytolytic T lymphocytes in a human melanoma. Science 269 1281–1284. 10.1126/science.7652577 [DOI] [PubMed] [Google Scholar]
- Wood L. D., Parsons D. W., Jones S., Lin J., Sjoblom T., Leary R. J., et al. (2007). The genomic landscapes of human breast and colorectal cancers. Science 318 1108–1113. 10.1126/science.1145720 [DOI] [PubMed] [Google Scholar]
- Yadav M., Jhunjhunwala S., Phung Q. T., Lupardus P., Tanguay J., Bumbaca S., et al. (2014). Predicting immunogenic tumour mutations by combining mass spectrometry and exome sequencing. Nature 515 572–576. 10.1038/nature14001 [DOI] [PubMed] [Google Scholar]
- Yan J., Parekh V. V., Mendez-Fernandez Y., Olivares-Villagomez D., Dragovic S., Hill T., et al. (2006). In vivo role of ER-associated peptidase activity in tailoring peptides for presentation by MHC class Ia and class Ib molecules. J. Exp. Med. 203 647–659. 10.1084/jem.20052271 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yang Z., Chevolot Y., Géhin T., Solassol J., Mange A., Souteyrand E., et al. (2013). Improvement of protein immobilization for the elaboration of tumor-associated antigen microarrays: application to the sensitive and specific detection of tumor markers from breast cancer sera. Biosens. Bioelectron. 40 385–392. 10.1016/j.bios.2012.08.019 [DOI] [PubMed] [Google Scholar]
- York I. A., Chang S. C., Saric T., Keys J. A., Favreau J. M., Goldberg A. L., et al. (2002). The ER aminopeptidase ERAP1 enhances or limits antigen presentation by trimming epitopes to 8-9 residues. Nat. Immunol. 3 1177–1184. 10.1038/ni860 [DOI] [PubMed] [Google Scholar]
- Zaenker P., Gray E. S., Ziman M. R. (2016). Autoantibody production in cancer–the humoral immune response toward autologous antigens in cancer patients. Autoimmun. Rev. 15 477–483. 10.1016/j.autrev.2016.01.017 [DOI] [PubMed] [Google Scholar]
- Zhang G. L., Ansari H. R., Bradley P., Cawley G. C., Hertz T., Hu X., et al. (2011). Machine learning competition in immunology–Prediction of HLA class I binding peptides. J. Immunol. Methods 374 1–4. 10.1016/j.jim.2011.09.010 [DOI] [PubMed] [Google Scholar]
- Zhang J., Shen L., Johnston S. A. (2018). Using frameshift peptide arrays for cancer neo-antigens screening. Sci. Rep. 8:17366. 10.1038/s41598-018-35673-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang M., Fritsche J. (2018). RNA editing derived epitopes function as cancer antigens to elicit immune responses. Nat. Commun. 9:3919. 10.1038/s41467-018-06405-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X., Kim S., Hundal J., Herndon J. M., Li S., Petti A. A., et al. (2017). Breast cancer neoantigens can induce CD8(+) T-cell responses and antitumor immunity. Cancer Immunol. Res. 5 516–523. 10.1158/2326-6066.cir-16-0264 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhu G., Zhang F. (2017). Efficient nanovaccine delivery in cancer immunotherapy. ACS Nano. 11 2387–2392. 10.1021/acsnano.7b00978 [DOI] [PubMed] [Google Scholar]
- Zhu Y., Knolhoff B. L., Meyer M. A., Nywening T. M., West B. L., Luo J., et al. (2014). CSF1/CSF1R blockade reprograms tumor-infiltrating macrophages and improves response to T-cell checkpoint immunotherapy in pancreatic cancer models. Cancer Res. 74 5057–5069. 10.1158/0008-5472.can-13-3723 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou W., Wolchok J. D., Chen L. (2016). PD-L1 (B7-H1) and PD-1 pathway blockade for cancer therapy: Mechanisms, response biomarkers, and combinations. Sci. Transl. Med. 8:328rv324. 10.1126/scitranslmed.aad7118 [DOI] [PMC free article] [PubMed] [Google Scholar]