Abstract
Background
Tumor cells can die via immunogenic cell death pathway, in which damage-associated molecular pattern molecules (DAMPs) are released from the cells. These molecules activate cells involved in the immune response. Both innate and adaptive immune response can be activated, causing a destruction of the remaining infected cells. Activation of immune response is also an important component of tumor treatment with electrochemotherapy (ECT) and irreversible electroporation (IRE). We thus explored, if and when specific DAMPs are released as a consequence of electroporation in vitro.
Materials and methods
In this in vitro study, 100 μs long electric pulses were applied to a suspension of Chinese hamster ovary cells. The release of DAMPs – specifically: adenosine triphosphate (ATP), calreticulin, nucleic acids and uric acid was investigated at different time points after exposing the cells to electric pulses of different amplitudes. The release of DAMPs was statistically correlated with cell permeabilization and cell survival, e.g. reversible and irreversible electroporation.
Results
In general, the release of DAMPs increases with increasing pulse amplitude. Concentration of DAMPs depend on the time interval between exposure of the cells to pulses and the analysis. Concentrations of most DAMPs correlate strongly with cell death. However, we detected no uric acid in the investigated samples.
Conclusions
Release of DAMPs can serve as a marker for prediction of cell death. Since the stability of certain DAMPs is time dependent, this should be considered when designing protocols for detecting DAMPs after electric pulse treatment.
Key words: electroporation, pulsed electric field treatment, damage-associated molecular pattern molecules, immunogenic cell death, electrochemotherapy
Introduction
Electroporation or pulsed electric field (PEF) treatment can cause changes in membrane permeability, which allows molecules, that are otherwise membrane impermeable, to cross the plasma membrane. In reversible electroporation the damage to cell membrane is repaired, enabling the cell to reestablish its metabolism and survive. This type of electroporation is used in multiple therapies. Electrochemotherapy (ECT) is one of such widely used therapies in which the increased cell membrane permeability enables chemotherapeutic drug to enter the cell and thus potentiates the cytotoxicity of the drug.1, 2 In irreversible electroporation (IRE) the damage to the cells however is too severe for the cells to recover which leads to cell death. While the cells are destroyed, the integrity of tissue like vessels, nerves and extracellular matrix remains preserved3, 4, making this therapy very appealing for ablation of tumor and other tissues, otherwise unsuitable for surgical removal or thermal ablation such as radiofrequency ablation or cryo-ablation.5, 6
In ECT eight square 100 μs electrical pulses, with an amplitude of 100-1000 V are usually used to induce a reversible membrane permeabilization. For IRE, more pulses (80-100 pulses) at higher amplitude (up to 3000 V) are required, to overwhelm the reparative capacity of the cells which leads to cell death.7 From morphological, biochemical, and functional perspectives, different cell death pathways/types can be activated.8 Historically, based on morphological changes, three different forms of cell death were defined: apoptosis (cell shrinkage, chromatin condensation, formation of apoptotic bodies); autophagy (cytoplasmic vacuolization); and necrosis (loss of plasma membrane integrity).8, 9 Such classification is still employed, but in newer classification based on genetic, biochemical, pharmacological and functional differences, cell death is either accidental (uncontrollable death caused by disassembly of the plasma membrane) or regulated (activation of signal transduction). Depending on signaling pathways different types of regulated cell death are being characterized, e.g. intrinsic and extrinsic apoptosis, necroptosis, ferroptosis, pyroptosis, immunogenic cell death, lysosome-dependent cell death, mitochondrial permeably transition driven necrosis and many others gathered and described by Galluzi et al..10 In electroporation studies, cell death has been most extensively explored in the range of nanosecond pulse treatment, where the majority of studies confirmed cell death by apoptosis (intrinsic and extrinsic) and only few studies indicated necrosis.11, 12 Both pathways were confirmed also in microsecond pulse treatment.13, 14, 15, 16, 17, 18, 19 Nevertheless, in recent studies new cell death types were also detected like pyroptosis20, necroptosis20, 21 and immunogenic cell death.22, 23, 24, 25, 26, 27, 28, 29
In IRE 18,30, 31, 32, 33, 34 and ECT with either bleomycin or cisplatin26,35, 36, 37 used for cancer treatment, involvement and importance of host immune response was demonstrated, counteracting tumor escape mechanisms.29,38, 39 After these therapies, dying tumor cells can release specific molecules, which are being recognized by the cells of immune system. These molecules can activate the innate and adaptive immune response, leading to the destruction of the remaining tumor cells in the body40 and inducing long-lasting protective antitumor immunity.41 Some studies even suggest that immunogenic effect of IRE is more pronounced than in other ablation therapies like radiofrequency ablation31 and cryoablation.32 Evidence suggests that administration of immune-stimulating molecules can even enhance the local effectiveness of ECT35 and IRE29,42, 43, 44 allowing simultaneous treatment of distant tumors.
Our immune system consists of two complementary and closely collaborative systems, an innate (non-specific) and an adaptive (antigen-specific) system. Activation of immune system is essential for our survival, as it distinguishes and eliminates potentially harmful molecules, even the ones that derive from the host/our own tissues. Well known are the pathogen-associated molecules (PAMPs), which are present on microbes and are being recognized by cells of the innate immune system when they bind to pattern recognition receptors (PRRs). The same pathways are activated by the host’s damage-associated molecular pattern molecules (DAMPs), which act as endogenous damage signal in case of cell death or response to stress, leading to inflammatory response.45, 46, 47 Release of DAMPs characterizes immunogenic cell death (ICD). Most of DAMPs are normally located intracellularly48, where under normal physiological conditions have an important intracellular role. When a cell is damaged or dies, DAMPs are actively or passively exposed or released to extracellular space.49, 50, 51 The release of DAMPs is often accompanied by cytokines, chemokines and other inflammatory mediators.52 In extracellular space DAMPs have a completely different function, as they are being recognized by pattern recognition receptors (PRRs), such as TRLs, NOD-like, PRLs and RAGE receptors on immune cells.50, 53 Binding of DAMPs to these receptors stimulates innate immune response through promoting the release of pro-inflammatory mediators and recruiting immune cells (dendritic cells, macrophages, T cells and neutrophils). Usually, the exposure of different DAMPs depends on endoplasmic reticulum stress, followed by reactive oxygen species (ROS) production.41 Release of DAMPs correlates with the degree of trauma.54 Some DAMPs can even be involved in tissue repair pathway.55, 56, 57 It depends on DAMPs and their triggered pathways, together with cytokines and growth factor to determine, if mild acute inflammation and wound healing 56, 58 or severe inflammation and fibrosis will follow.59
Electroporation causes an increase in membrane permeability and allows molecules, for which the membrane is usually impermeable, including DAMPs, to cross it. ATP, one of the main DAMPs, was even used as an indicator of cell membrane permeabilization in the first electroporation studies.60 In recent years reports on electroporation studies have started to emerge investigating the immunogenic cell death caused by electroporation. Studies detected DAMPs, like ATP, high-mobility group box 1 protein (HMGB1) release and calreticulin externalization, as they are the gold standard for predicting the ICD in cancer cells.41 So far mostly single (or a small subset of) DAMPs were studied. Most studies involved nanosecond pulse treatment22, 23, 24, 25, whereas studies using microsecond26, 29, millisecond28 and H-FIRE pulse treatments27 are even more scarce. For now, different DAMPs were investigated at different intervals after electroporation ranging from 30 min to 72 hours, using different types of cancer cells.
Because in both ECT and IRE the immune system response is essential for successful and complete tumor eradication, we decided to explore if and when specific DAMPs are released in response to electroporation in vitro. The experiments were performed using 100 μs long pulses, as they are most commonly used in ECT treatment and in IRE for soft tissue ablation.
Materials and methods
Cell preparation
Chinese hamster ovary (CHO-K1) from European Collection of Authenticated Cell Cultures were grown in culture flasks (TPP, Switzerland) filled with HAM F-12 growth medium (PAA, Austria) at 37°C with a humidified 5% CO2. The growth medium was enriched with 10% fetal bovine serum (FBS) (Sigma-Aldrich, Germany), L-glutamine (StemCell, Canada) and antibiotics penicillin/ streptomycin (PAA, Austria) and gentamycin (Sigma-Aldrich, Germany). At 70% confluency, cells were detached with trypsin solution (10x trypsin-EDTA (PAA, Austria) 1:9 diluted in Hank’s basal salt solution (StemCell, Canada), which was inactivated after 3 minutes by the growth medium. After 5 minutes of centrifugation at 180 g and 22°C supernatant was removed. Cell were mixed with the growth medium to obtain cell density at 2x106 cells/ml.
Electric pulse generation
Laboratory prototype pulse generator (University of Ljubljana), based on H-bridge digital amplifier with 1 kV MOSFETs (DE275-102N06A, IXYS, USA), described in 61 was used. Eight 100 μs long monopolar electric pulses with repetition frequency 1 Hz and amplitude of 0–600 V (0-3 kV/cm; voltage to distance ratio) with increments of 100 V (and additional increments in the permeabilization assay) were applied between stainless steel 304 plate electrodes (d = 2 mm). Oscilloscope HDO6104A-MS, differential probe HVD3206A and the current probe CP031A, all from LeCroy, USA, were used to monitor the delivered pulses, i.e. voltage and current. The delivered voltage was approximately 10-15% lower than the value set on the pulse generator and the current was in the range of 3-21 A. When pulses with high amplitudes were applied (Figure 1), current decreased slightly during the pulse, presumingly due to electrochemistry at electrode-electrolyte interface reducing the available interface area for ion exchange between the metal electrode and the electrolyte and possibly also due to ion depletion at the said interface.
Figure 1.
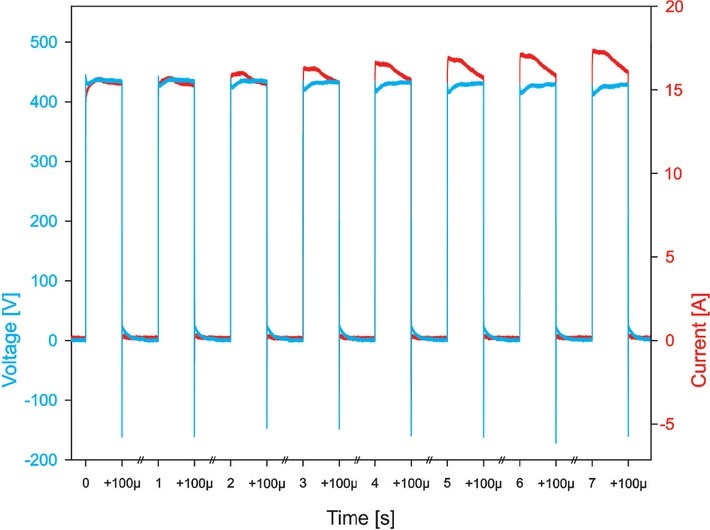
Application of 500 V pulses. Blue line shows voltage and red line shows current. Due to sequencing, all eight pulses are in one picture, separated by //.
Results
First, the permeabilization and the survival curves were obtained to determine experimental points for the studies on release of DAMPs. Permeabilization and survival curves are presented in all figures showing the concentration of various DAMPs to visualize how the presence of DAMPs is related to changes in permeabilization and cell viability. In figures the permeabilization and survival curves are shown only at pulse amplitudes tested for the presence of DAMPs; in steps of 50 V in the range of pulses where changes in permeabilization occur and in steps of 100 V above 200 V.
The concentration of ATP in supernatant was first measured with fluorescent method 30 minutes and 24 hours after electroporation (Figure 2). At 30 minutes the concentration of ATP in supernatant was detected at 200 V. However, statistical difference between the control and the treatment groups (obtained by one-way analysis of variance [ANOVA] followed by the post-hoc test) was only detected at 500 V and above. The concentration of ATP in supernatant grew with increasing pulse amplitude, which after 24 hours led to decreased cell viability; e.g. correlation between the cell survival and ATP concentration in supernatant detected after 30 min is quite strong and negative; R = -0.864. Also, weak correlation between cell permeabilization and ATP concentration in supernatant (R = 0.594) confirms, that ATP presence in supernatant is more strongly correlated with the irreversible than the reversible electroporation. It may indicate that strong ATP release from cells leads to cell death.
Figure 2.
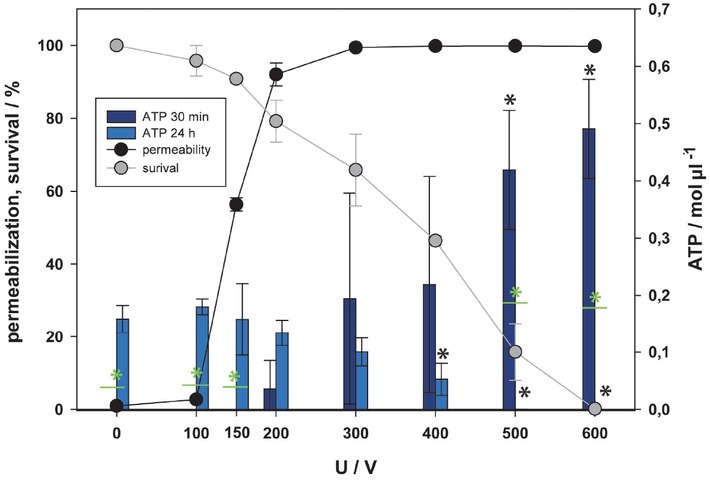
Release of adenosine triphosphate (ATP) as a function of electric pulse amplitude determined by fluorescent assay. Two-time points after electroporation were assessed. Permeabilization and survival curves are also presented. Black and green asterisks (*) indicate statistically significant differences between the samples at different voltages and the corresponding control at 0 V (one-way analysis of variance [ANOVA] followed by Holm-Sidak post-hoc test, (p < 0.05) and within the pair of samples at different voltages (t-test, p < 0.05), respectively).
After 24 hours (Figure 2) the lowest ATP concentration was achieved at the pulse amplitude resulting in death of most cells (500, 600 V). The concentration of ATP at these points is statistically different to the results obtained 30 minutes after pulse treatment. After 24 hours the concentration of ATP had decreased with the lower viability, but statistical differences between the control and the treatment groups were present from 400 to 600 V. At 24 hours there is a positive statistical correlation between the cell survival and concentration of ATP in supernatant (R = 0.888), which is stronger than the correlation to permeabilization (R = -0.695).
Since no ATP was detected in supernatant within the range of reversible electroporation after 30 minutes using the fluorescent method (Figure 2), we also used a more sensitive luminescent method (Figure 3). Furthermore, since ATP analysis showed that 24 h after treatment ATP is not detected in all samples, we were also interested in how fast ATP was degraded. Scuderi et at. showed complete resealing of plasma membrane 10 minutes after pulse treatment using 8 x 100 μs pulses.63 Thus, another time point for ATP measurement was chosen, i.e. 15 minutes after (Figure 3). With more sensitive luminescent detection assay, ATP was detected in supernatant already at 100 V, however statistically significant difference to control was only detected at 300 V and higher. These results are more reliable due to higher assay sensitivity however even with this method statistically significant amount of ATP in supernatant is detected in the range of irreversible electroporation, as increased electric field/voltage kills more cells more ATP is present in the extracellular space. This is also confirmed by a strong correlation between the survival and the amount of ATP in supernatant, R = -0.947 for 15 min and R = -0.964 for 30 min, and much weaker correlation between the permeabilization and the amount of ATP (R = 0.704 for 15 min and R = 0.728 for 30 min). In our results, only one significant difference was found in detected ATP amount between 15 and 30 minutes after pulse treatment at 500 V. Since this difference was not detected for all the experimental points (voltages), we believe that ATP in extracellular space is not degraded in this first 30 minutes after pulse treatment.
Figure 3.
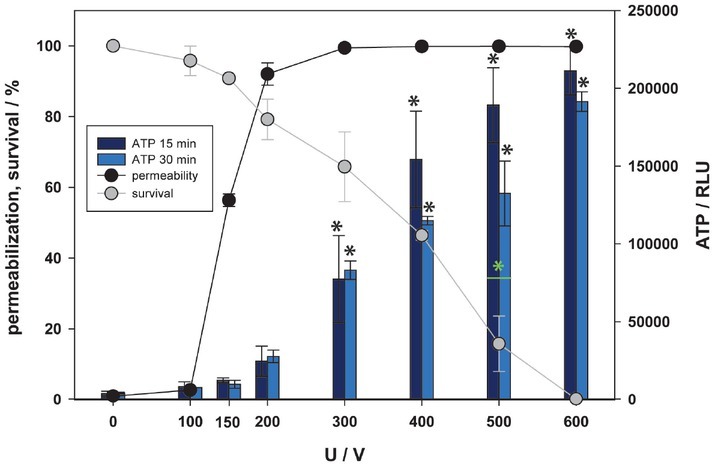
Release of adenosine triphosphate (ATP), as a function of electric pulse amplitude determined by luminescence assay. Two-time points after electroporation were assessed. Permeabilization and survival curves are also presented. Black and green asterisks (*) indicate statistically significant differences between the samples at different voltages and the corresponding control at 0 V (one-way analysis of variance [ANOVA] followed by Holm-Sidak post-hoc test, (p < 0.05) and within the pair of samples at different voltages (t-test, p < 0.05), respectively).
Calreticulin (CRT) is an endoplasmic reticulum protein which needs to be transferred to the outer leaflet of the plasma membrane in order to act as a DAMP. Externalization of calreticulin to outer membrane in an active process involving also its transport across the cell. Due to this active and time demanding process the externalization of calreticulin was investigated 4 and 24 hours (also used in previous studies22, 23, 24, 25,28) after pulse treatment (Figure 4) on viable cells (determined by propidium iodide [PI] staining). Calreticulin was first detected at 300 V and its fluorescence increased with increasing voltage of pulses. Furthermore, the lowest viability at 600 V with < 5% of viable cell has the strongest signal of calreticulin after 4 and 24 hours. This could indicate the amount of externalized calreticulin per viable cell increases with the level of stress (amplitude of applied electric pulses). Furthermore, analysis shows a strong correlation between survival determined by MTS test and externalization of calreticulin, as survival decreased, the detection of calreticulin increased (R = -0.801 for 4h and R = -0.946 for 24h) and weak correlation between permeabilization and externalization of calreticulin was observed (R = 0.535 for 4h and R = 0.556 for 24h). Since calreticulin was detected only in viable cells (determined by PI) additional information on viability was obtained, and results were normalized to control (0 V) for each investigated time point separately. Except for the results at 300 V, no statistically significant difference between 4 and 24 hours was detected at any other experimental point, suggesting that calreticulin can be detected 4 hours after pulse treatment and that expression of the protein remains stable for the next 20 hours.
Figure 4.
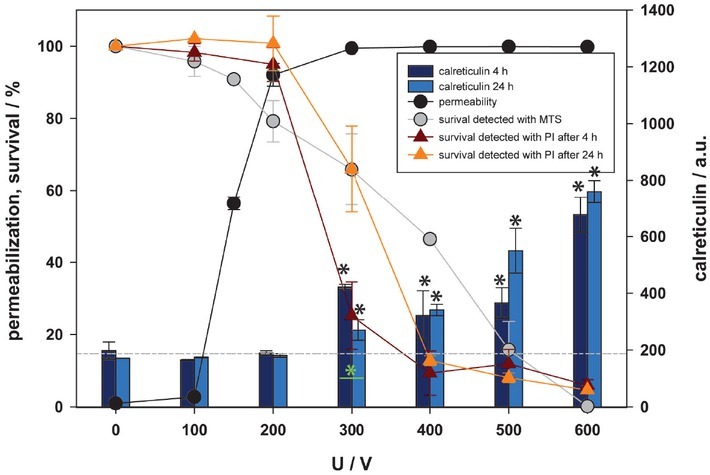
Externalization of calreticulin as a function of electric pulse amplitude. Two-time points after electroporation were assessed. Permeabilization and survival (MTS) curves are also presented. Survival detected by propidium iodide (PI) protocol is normalized to control and presented with red (4 hours after pulse treatment) and orange (24 hours after pulse treatment) line. Approximate baseline of calreticulin is presented with -----. Black and green asterisks (*) indicate statistically significant differences between the samples at different voltages and the corresponding control at 0 V (one-way analysis of variance [ANOVA] followed by Holm-Sidak posthoc test, (p < 0.05) and within the pair of samples at different voltages (t-test, p < 0.05), respectively).
Until now, nucleic acids (in the role of DAMPs) have not been investigated in relation to electroporation. Most of RNA (except fresh transcripted mRNA) is located in cytoplasm, while DNA is located in the cell nucleus. The concentration of RNA and DNA in supernatant has been detected 15, 30 minutes and 24 hours after electroporation like in ATP assay (Figure 5 and 6). Concentration of DNA/ RNA started to rise from 400 V up (Figure 5 and 6). This happened at the same pulse amplitudes where after 24 hours cell viability was affected, indicating the amount of nucleic acid occurs in the range of cell death, i.e. irreversible electroporation. Exposure of cells to higher pulse amplitudes caused higher release of nucleic acids, which after 24 h resulted in lower cell viability. This is confirmed also by a strong negative correlation between survival and release of RNA (R = -0.909 for 15 min, R = -0.909 for 30 min, R = -0.919 for 24 h) and weak correlation between permeabilization and release of RNA (R = 0.584 for 15 min, R = 0.696 for 30 min, R is not significant for 24 h), respectively. Similarly, for DNA, a strong correlation between survival and release of DNA (R = -0.935 for 15 min, R = -0.919 for 30 min, R = -0.928 for 24 h) and a weak correlation between permeabilization and release of DNA (R = 0.571 for 15 min, R = 0.689 for 30 min, R is not significant for 24 h) was found. This correlation may indicate that loss of nucleic acids results in cell death.
Figure 5.
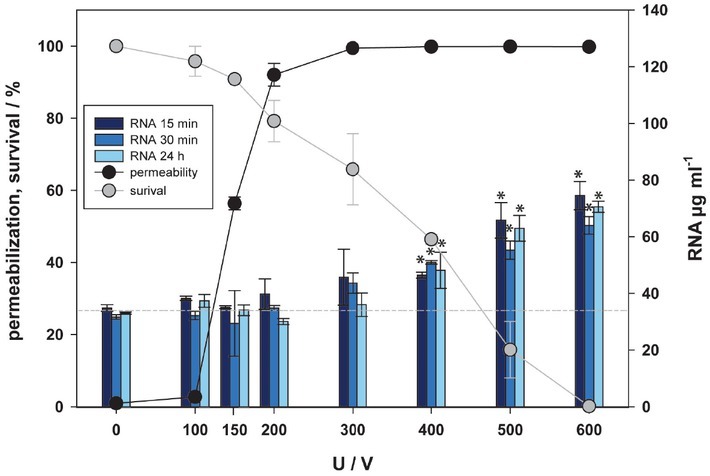
Release of RNA as a function of electric pulse amplitude. Three-time points after electroporation were assessed. Permeabilization and survival curves are also presented. Approximate baseline of RNA is presented with -----. Black and green asterisks (*) indicate statistically significant differences between the samples at different voltages and the corresponding control at 0 V (one-way analysis of variance [ANOVA] followed by Holm-Sidak post-hoc test, (p < 0.05) and within the pair of samples at different voltages (t-test, p < 0.05), respectively).
Figure 6.
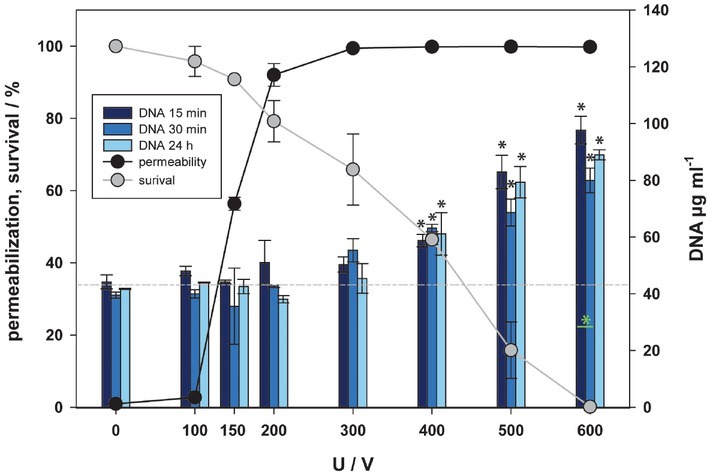
Release of DNA as a function of electric pulse amplitude. Three-time points after electroporation were assessed. Permeabilization and survival curves are also presented. Approximate baseline of DNA is presented with -----. Black and green asterisks (*) indicate statistically significant differences between the samples at different voltages and the corresponding control at 0 V (one-way analysis of variance [ANOVA] followed by Holm-Sidak post-hoc test, (p < 0.05) and within the pair of samples at different voltages (t-test, p < 0.05) respectively.
Comparison of the concentration of RNA detected in supernatant 15, 30 minutes and 24 hours after pulse treatment did not show any significant differences. Comparison of the concentration of DNA detected in supernatant 15, 30 minutes and 24 hours after pulse treatment showed only significant differences between 15 and 30 minutes at 600 V, yet interestingly this was not the case between 15/30 minutes and 24 hours after pulse treatment. According to our results, the released nucleic acids in vitro are stable and are not degraded within 24 hours after pulse treatment.
We also tried to detect uric acid, another well know DAMP molecule. The release of uric acid in supernatant was analyzed 24 hours after pulse treatment (Figure 7). In our experiments we were however unable to detect any uric acid in supernatant after pulse treatment. Initial experiments were also performed after 30 minutes, but results were the same (data not shown).
Figure 7.
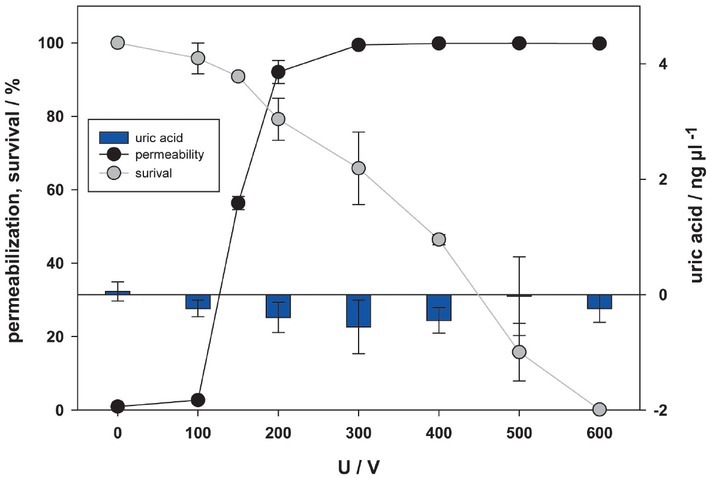
Release of uric acid as a function of electric pulse amplitude. Amount of uric acid was analysed 24 hours after pulse treatment in supernatant. Permeabilization and survival curves are also presented. No statistical difference was detected.
Discussion
Besides the induced membrane permeabilization, followed by cell death, activation of the immune response seems to be an important component in effectiveness of ECT26,35, 36, 37 and IRE18,30, 31, 32, 33, 34 treatment in vivo. Activation of the immune system can be triggered by a special type of cell death, called immunogenic cell death (ICD) in which DAMPs are the key mediators.64 Presence of DAMPs after pulse treatment has been detected in different types of cancer cells and normal tissues.22, 23, 24, 25, 26, 27, 28, 29 However, only one study was performed with 100 us pulses, which are predominantly used in ECT and IRE.26 The authors investigated release of ATP, calreticulin and HMGB1 due to electroporation pulses alone, bleomycin alone and combination of electroporation pulses and bleomycin. Their study demonstrated the release of ATP and calreticulin after pulse treatment, but how this correlates to reversible and/or irreversible electroporation remained elusive. This question is addressed in our study, were release/ detection of different DAMPs was correlated with permeabilization and survival curve in vitro.
Detected amounts of DAMPs were correlated to cell membrane permeabilization determined by PI assay immediately after pulse treatment and cell survival, analyzed 24 hours after treatment by MTS test, i.e. to reversible and irreversible electroporation, respectively. It is generally believed that membrane permeabilization and cell survival after pulse treatment are causally related, i.e. in IRE cell death occurs due to membrane permeabilization and loss of cell homeostasis (in this study correlation coefficient between the two is -0.680) and in ECT increased accumulation of drug leads to increased cell cytotoxicity.
In ECT and IRE it was also demonstrated that the immune response plays an important role in achieving therapeutic effect.33, 34, 35, 36 We have therefore determined different DAMPs at different times after exposing the cells to electric pulses and determined whether the concentrations of extracellular DAMPs were better correlated to cell death or to membrane permeabilization. While activation inflammatory response and activation of immune system is desired in cancer therapies, on the other hand it can be a wanted or an unwanted effect in gene therapy.68, 69 In DNA vaccination therapies, changed permeability of cell membrane enhances the introduction of DNA vaccine inside of cell and the presence of DAMPs additionally activates inflammatory response, which leads to enhanced production of antibodies, thus enhancing the efficiency of vaccination.65, 66, 67 Nevertheless, in most cases of gene therapy, the immune response in unwanted, as it may destroy the transfected cells and prevent transgenic protein expression.68, 69 By now many DAMPs have been identified and the number is still increasing. Most known are the HMGB1, nucleic acids, proteins like heat-shock proteins, S100 and calreticulin, purine metabolites like ATP and uric acid and saccharides. A list of know DAMPs and their receptors is given by Roh and Sohn.48
ATP is a well-known molecule in biology and biochemistry for being a universal energy source in the cell and necessary for multiple cell processes and cell metabolism. Interestingly, first studies of electroporation and increase in plasma membrane permeability involved adenosine triphosphate ATP detection in electroporation buffer.70 The released ATP is also considered a DAMP. In our study two types of ATP detection methods with different sensitivities were used.71, 72 In a previous study performed by Calvet et al.26 using the same pulses as in our study, the release of ATP was detected 30 minutes after the treatment. We were able to confirm their observations with the fluorescence and the luminescence method (Figures 2 and 3 respectively). Furthermore, the investigation of the effects at different pulse amplitudes showed that the release of ATP increases with increasing amplitude. This was expected, as ATP release was previously used as a permeability marker after electroporation.70 However, in our results statistical differences between the control and the treatment groups were not detected in the range where the permeabilization curve is ascending, but was detected only in the range of pulse amplitudes at which all cells were already permeabilized and many were dead. Since ANOVA analysis is less sensitive when large amount of samples with big differences between them are analyzed, additional ANOVA was performed, taking into account only the results from 0 to 300 V (e.g. where permeabilization changes from 0 to 100%). Now additional analysis showed that statistical differences between control and the treatment groups are present at 200 V and above in luminescent method, suggesting ATP release as possible membrane permeabilization detection method. In the fluorescence method, this statistical difference was obtained only at 300 V. Such difference in analysis and also a bigger ratio of ATP between the control and the treatment groups indicates that the luminescence method is more sensitive method than the fluorescence method. Taken into consideration ATP release at all investigated voltages (also the one leading to cell death after 24 hours) the release of ATP is more strongly correlated to cell death/irreversible electroporation (R = 0.888) than permeability/reversible electroporation (R = -0.695).
24 hours after pulse treatment (Figure 2) the highest amount of ATP was detected in the supernatant of control sample and the amount of ATP decreased with increasing pulse amplitude. This can be explained by homeostasis of ATP in living cells. In a living homeostatic cell most of the ATP is located intracellularly, however in considerably lower concentration ATP is also present in extracellular space.73 When cells are damaged, considerable release of ATP molecule affects ATP pumps, causing depletion of intracellular K+ and accumulation of intracellular NA+ and Ca2+ and leading to cell death.74 A previous study showed that electroporation pulses cause ATP depletion, which in 24 results in lower viability, presumingly by affecting Ca2+-ATPase.75 In our study the effect on survival was also confirmed by very strong positive correlation between survival/irreversible electroporation and amount of ATP detected in supernatant (R = 0.888). Nevertheless, we need to consider, that some of the ATP detected in supernatant could be from the cells damaged due to cell handling during experiment. In extracellular space ATP is degraded by nucleotides like CD39 and CD37, which convert ATP through ADP and AMP to adenosine73, which explains why ATP was detected 30 minutes at very high voltages (500, 600 V), but was no longer detected after 24 hours (Figure 2). This can also explain why Calvet et al.26, was unable to detect ATP 30 hours after pulse treatment alone, however it does not explain, why ATP was still detected when bleomycin alone or in combination with electroporation pulses was used. How fast ATP degrades in extracellular space, remains unknown. Our results do not indicate that ATP degrades within the first 30 minutes after pulse treatment, since no difference between 15 and 30 minutes after pulse treatment was detected. In a different study76 the results for ATP 4 hours after pulse treatment was lower in samples exposed to pulse treatment than in the control. If this is taken into consideration together with our results, then ATP degradation in vitro occurs somewhere between 30 minutes and 4 hours after pulse treatment.
Calreticulin was another molecule of interest in our study. This highly conserved protein has major functions in lumen of the endoplasmic reticulum (ER). It is involved in correct folding of proteins that are produced in endoplasmic reticulum77 and in regulation of calcium metabolism, as it affects Ca2+ capacity of the ER stores.78 In the early phase of cell death, activated ER stress leads to translocation of calreticulin to cells surface trough ER-Golgi pathway or lysosome exocytosis.79, 80 Calreticulin, as DAMP, was investigated previously in electroporation studies.22, 23, 24, 25, 26,28 In Calvets’s study26, which used the same pulses as in our study (eight 100 μs pulses), calreticulin was determined 30 hours after treatment using different treatments. Calreticulin was detected on the plasma membrane after electroporation pulses alone or in combination with bleomycin (ECT), yet no externalization was detected in cells threated with bleomycin alone. Since only calreticulin, exposed on the cell surface acts as a DAMP, only viable cells (determined by PI) were taken into analysis. The presence of calreticulin on the cell surface was previously detected already 4 hours after electroporation with millisecond pulses28, thus we assumed 4 hours is sufficient time for calreticulin to transfer to cells surface. Additionally, calreticulin was detected also 24 after pulse treatment. In our study calreticulin was investigated 4 and 24 hours after treatment (Figure 4), which is after the resealing of cell membrane.63 Even though calreticulin was detected on the surface of live cells, it was detected only in the range of irreversible electroporation. Additionally, calreticulin detection increased with decreasing cell viability (less viable cells), implicating that bigger stress or in this case pulse amplitude causes more calreticulin molecules to be externalized to cell surface. Nevertheless, since it is believed that externalization of calreticulin occurs in early phase of cell death79, 80, it is possible that cell determined as viable would die within next hours.
In comparison to ATP, calreticulin is more stable. Only at 300 V the difference between 4 and 24 hours was statistically significant. Stability of externalized calreticulin was previously also confirmed in another in vitro study.24 Nevertheless, in vivo study shows expression is the strongest between four and six hours, and diminishes 24 h after the treatment.28
So far studies investigating DAMPs, released by the electroporation treatment, included ATP, calreticulin and HMGB1. In addition to ATP and calreticulin we also included other known DAMPs in our study, namely nucleic acids and uric acid, which so far have not been investigated as DAMPs after electroporation. Inside the cells nucleic acids are the source of genetic information. As DAMPs in extracellular space nucleic acids bind to TLR receptors. Bound DNA can even attract HMGB1 (a non-histone nuclear protein, which can be actively or passively released into extracellular space, where it acts as a DAMP81) and together they form complexes stimulating dendritic cells to produce type 1 interferon (non-specific immune response), which can lead to anti-DNA autoantibody production (specific immune response).82 Nucleic acids (RNA and DNA) can be detected in supernatant already within minutes after pulse treatment (Figure 5,6). Nevertheless, we need to consider – based on the control, 0 V in Figures 5 and 6, that some of the nucleic acids detected in supernatant could be from the cells damaged due to cell handling during experiment. Since RNA is more abundantly present in cells than DNA83, 84, the same was expected to be the case in the supernatant after pulse treatment. However, the amount of detected DNA in our samples was bigger than that of RNA. Since RNA is more prone to degradation than DNA84, it is possible that some of the RNA was destroyed during the process of analysis. Nevertheless, the amount of released nucleic acids increases with increasing voltage of electric pulses to which the cells were exposed. Our results indicate that the release of nucleic acids (RNA and DNA) occurs in the range of irreversible electroporation; i.e. pulse amplitudes that lead to cell death as determined by MTS test at 24 h post treatment. This was confirmed also by very strong negative correlation between the cell survival and the release for RNA and DNA.
Uric acid is a product of purine metabolism within the cell, like degradation of nucleic acids, and is released from injured and dying cells.85 A molecule that is soluble inside the cell, accumulates in extracellular space, where it is transformed in insoluble crystal of monosodium urate, stimulating the maturation of dendritic cells and T-cell response.85, 86 Here, the presence of uric acid after electroporation was investigated for the first time. Presence of uric acid in supernatant was investigated 24 hours after electroporation treatment (Figure 7). We expected uric acid to show a similar behavior in pulse parameter dependency as other DAMPs. However, we did not detect uric acid in supernatant after pulse treatment. Standard curve was obtained, therefore Uric Acid Assay Kit worked. Maybe uric acid production did not happen or uric acid was still inside of cells and not yet in supernatant as predicted. Furthermore, we found no existing data on CHO cells and uric acid in the literature, so maybe formation of uric acid in ovarian cells does not occur.
With respect to the results obtained, detection of DAMPs and its correlation to cell membrane permeabilization and cell survival seems to be more complex than initially thought. Even a DAMP like ATP, which can be released due to electroporation alone is better correlated to cell survival than membrane permeabilization (Tables 1, 2). A recent study performed by Ringel-Scaia et al.27, in which multiple signaling pathways were analyzed, showed that the cell and cell population is a dynamic system which changes with time. Two hours after pulse treatment RNA analyses showed activation of immunosuppressive pathway, cell injury and apoptosis. With time these genes became less pronounces and after 24 hours change in gene expression indicated proinflammatory response, cell repair and necrosis/pyroptosis. This explains changes in DAMPs detection hours after pulse treatment, including the presence and absence of different DAMPs and its correlation with cell survival. Since statistical correlations between DAMP release and cell survival is much stronger than with membrane permeabilization, involvement of immune system in IRE can be explained. However, activation of immune system was demonstrated also in ECT treatments, were reversible electroporation is used. How can that be, if correlation between membrane permeabilization and released DAMPs is weak or does not even exist? Modeling87 and in vivo experiments88 show that application of nominally reversible electroporation pulses such as those used for ECT of tumors still causes some cell death by means of irreversible electroporation in tissue close to the electrodes, due to inhomogeneous electric field distribution, which can thus lead to the release of DAMPs and activation of the immune system.
Table 1.
Correlation (R) between survival and release of damage-associated molecular pattern molecules (DAMPs) after pulse treatment. Investigated time points for each molecule are presented in the bottom row. Correlation was evaluated with Pearson correlation coefficient and survival was analyzed via MTS assay 24 hours after pulse treatment
| R vs. survival (MTS) | |||||
|---|---|---|---|---|---|
| PI | -0.680 | ||||
| ATP | -0.947 (L) | -0.964 (L)/-0.864 (F) | 0.888 (F) | ||
| DNA | -0.935 | -0.919 | -0.928 | ||
| RNA | -0.909 | -0.909 | -0.919 | ||
| CRT | -0.801 | -0.946 | |||
| uric acid | NS | ||||
| time after points EP | 3 min | 15 min | 30 min | 4 h | 24 h |
ATP = adenosine triphosphate; CRT = calreticulin; (F) = fluorescence assay; (L) = luminescence assay; NS = no statistical significance; PI = propidium iodide
Table 2.
Correlation (R) between permeabilization and release of damage-associated molecular pattern molecules (DAMPs) after pulse treatment. Investigated time points for each molecule are present in the bottom row. Correlation was evaluated with Pearson correlation coefficient and permeabilization was analyzed by propidium iodide (PI) assay 3 minutes after pulse treatment
| R vs permeabilization (PI) | |||||
|---|---|---|---|---|---|
| MTS | -0.680 | ||||
| ATP | 0.704 | 0.728 (L)/0.594 (F) | -0.695 (F) | ||
| DNA | 0.571 | 0.689 | NS | ||
| RNA | 0.584 | 0.696 | NS | ||
| CRT | 0.535 | 0.556 | |||
| uric acid | NS | ||||
| time after points EP | 3 min | 15 min | 30 min | 4 h | 24 h |
ATP = adenosine triphosphate; CRT = calreticulin; (F) = fluorescence assay; (L) = luminescence assay; NS = no statistical significance
The aim of this study was to explore, if and when specific DAMPs are released as a consequence of electroporation and if the release of DAMPs can be correlated to reversible and/or irreversible electroporation. Even though detection of certain DAMPs remains uncertain, others show strong correlation to cell survival/irreversible electroporation and much weaker correlation to membrane permeabilization/reversible electroporation. Release of DAMPs could perhaps serve as a predictor of cell death. In addition, it may indicates that the stability of certain DAMPs is questionable and thus their presence and detectability is time dependent. This needs to be taken into consideration when designing protocols to detect DAMPs after electroporation treatment. Finally, to obtain a better insight of DAMP release with respect to electroporation treatment other cell types including also cancer cell types should be investigated.
Acknowledgements
Authors would like to thank L. Vukanović and D. Hodžić for their help in the cell culture laboratory and Dr. Matej Reberšek for help with pulse recording and image production. The research was supported by Medtronic and the Slovenian Research Agency (research core funding No. IP-0510, P2-0249 and grant to young researcher Tamara Polajžer).
Disclosure
No potential conflicts of interest were disclosed.
References
- 1.Orlowski S, Belehradek J, Paoletti C, Mir LM. Transient electropermeabilization of cells in culture. Increase of the cytotoxicity of anticancer drugs. J Biochem Pharmacol Res. 1988;3:4727–33. doi: 10.1016/0006-2952(88)90344-9. [DOI] [PubMed] [Google Scholar]
- 2.Mir LM. Bases and rationale of the electrochemotherapy. EJC Suppl. 2006;4:38–44. doi: 0.1016/j.ejcsup.2006.08.005. [Google Scholar]
- 3.Scheffer HJ, Nielsen K, De Jong MC, Van Tilborg AJM, Vieveen JM, Bouwman A. Irreversible electroporation for nonthermal tumor ablation in the clinical setting: a systematic review of safety and efficacy. J Vasc Interv Radiol. 2014;25:997–1011. doi: 10.1016/j.jvir.2014.01.028. et al. [DOI] [PubMed] [Google Scholar]
- 4.Phillips M, Maor E, Rubinsky B. Nonthermal irreversible electroporation for tissue decellularization. J Biomech Eng. 2010;132:091003. doi: 10.1115/1.4001882. [DOI] [PubMed] [Google Scholar]
- 5.Davalos RV, Mir LM, Rubinsky B. Tissue ablation with irreversible electroporation. Ann Biomed Eng. 2005;3:223–31. doi: 10.1007/s10439-005-8981-8. [DOI] [PubMed] [Google Scholar]
- 6.Chen X, Ren Z, Zhu T, Zhang X, Peng Z, Xie H. Electric ablation with irreversible electroporation (IRE) in vital hepatic structures and follow-up investigation. Sci Rep. 2015;5:16233. doi: 10.1038/srep16233. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jiang C, Davalos RV, Bischof JC. A review of basic to clinical studies of irreversible electroporation therapy. IEEE Trans Biomed Eng. 2015;62:4–20. doi: 10.1109/TBME.2014.2367543. [DOI] [PubMed] [Google Scholar]
- 8.Galluzzi L, Maiuri MC, Vitale I, Zischka H, Castedo M, Zitvogel L. Cell death modalities: classification and pathophysiological implications. Cell Death Differ. 2007;14:1237–43. doi: 10.1038/sj.cdd.4402148. et al. [DOI] [PubMed] [Google Scholar]
- 9.Schweichel JU, Merker HJ. The morphology of various types of cell death in prenatal tissues. Exp Teratol. 1973;7:253–66. doi: 10.1002/tera.1420070306. [DOI] [PubMed] [Google Scholar]
- 10.Galluzzi L, Vitale I, Aaronson SA, Abrams JM, Adam D, Agostinis P. Molecular mechanisms of cell death: recommendations of the nomenclature committee on cell death 2018. Cell Death Differ. 2018;25:486–541. doi: 10.1038/s41418-017-0012-4. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Batista Napotnik T, Rebersek M, Vernier PT, Mali B, Miklavcic D. Effects of high voltage nanosecond electric pulses on eukaryotic cells (in vitro): a systematic review. Bioelectrochemistry. 2016;110:1–12. doi: 10.1016/j.bioelechem.2016.02.011. [DOI] [PubMed] [Google Scholar]
- 12.Beebe SJ. Miklavčič D. Handbook of electroporation. Heidelberg: Springer International Publishing; 2017. Regulated and apoptotic cell death after nanosecond electroporation; pp. 511–28. editor. p. –. [DOI] [Google Scholar]
- 13.Chai W, Zhang W, Wei Z, Xu Y, Shi J, Luo X. Irreversible electroporation of the uterine cervix in a rabbit model. Biomed Microdevices. 2017;19:103. doi: 10.1007/s10544-017-0248-2. et al. [DOI] [PubMed] [Google Scholar]
- 14.Kim HB, Sung CK, Baik KY, Moon KW, Kim HS, Yi JH. Changes of apoptosis in tumor tissues with time after irreversible electroporation. Biochem Biophys Res Commun. 2013;435:651–6. doi: 10.1016/j.bbrc.2013.05.039. et al. [DOI] [PubMed] [Google Scholar]
- 15.Lee EW, Loh CT, Kee ST. Imaging guided percutaneous irreversible electroporation: Ultrasound and immunohistological correlation. Technol Cancer Res Treat. 2007;6:287–93. doi: 10.1177/153303460700600404. [DOI] [PubMed] [Google Scholar]
- 16.Lee EW, Wong D, Tafti BA, Prieto V, Totonchy M, Hilton J. Irreversible electroporation in eradication of rabbit VX2 liver tumor. J Vasc Interv Radiol. 2012;23:833–40. doi: 10.1016/j.jvir.2012.02.017. et al. [DOI] [PubMed] [Google Scholar]
- 17.Zhang Z, Li W, Procissi D, Tyler P, Omary RA, Larson AC. Rapid dramatic alterations to the tumor microstructure in pancreatic cancer following irreversible electroporation ablation. Nanomedicine. 2014;9:1181–92. doi: 10.2217/nnm.13.72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.José A, Sobrevals L, Ivorra A, Fillat C. Irreversible electroporation shows efficacy against pancreatic carcinoma without systemic toxicity in mouse models. Cancer Lett. 2012;317:16–23. doi: 10.1016/j.canlet.2011.11.004. [DOI] [PubMed] [Google Scholar]
- 19.Al-Sakere B, André F, Bernat C, Connault E, Opolon P, Davalos RV. Tumor ablation with irreversible electroporation. PLoS One. 2007;2:e1135. doi: 10.1371/journal.pone.0001135. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zhang Y, Lyu C, Liu Y, Lv Y, Chang TT, Rubinsky B. Molecular and histological study on the effects of non-thermal irreversible electroporation on the liver. Biochem Biophys Res Commun. 2018;500:665–70. doi: 10.1016/j.bbrc.2018.04.132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.López-Alonso B, Hernáez A, Sarnago H, Naval A, Güemes A, Junquera C. Histopathological and ultrastructural changes after electroporation in pig liver using parallel-plate electrodes and high-performance generator. Sci Rep. 2019;9:2467. doi: 10.1038/s41598-019-39433-6. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Nuccitelli R, Berridge JC, Mallon Z, Kreis M, Athos B, Nuccitelli P. Nanoelectroablation of murine tumors triggers a cd8-dependent inhibition of secondary tumor growth. PLoS One. 2015;10:e0134364. doi: 10.1371/journal.pone.0134364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nuccitelli R, McDaniel A, Anand S, Cha J, Mallon Z, Berridge J. Nanopulse stimulation is a physical modality that can trigger immunogenic tumor cell death. J Immunother Cancer. 2017;5:32. doi: 10.1186/s40425-017-0234-5. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Guo S, Jing Y, Burcus NI, Lassiter BP, Tanaz R, Heller R. Nano-pulse stimulation induces potent immune responses, eradicating local breast cancer while reducing distant metastases. Int J Cancer. 2018;142:629–40. doi: 10.1002/ijc.31071. et al. [DOI] [PubMed] [Google Scholar]
- 25.Rossi A, Pakhomova ON, Mollica PA, Casciola M, Mangalanathan U, Pakhomov AG. Nanosecond pulsed electric fields induce endoplasmic reticulum stress accompanied by immunogenic cell death in murine models of lymphoma and colorectal cancer. Cancers. 2019;11:2034. doi: 10.3390/cancers11122034. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Calvet CY, Famin D, André FM, Mir LM. Electrochemotherapy with bleomycin induces hallmarks of immunogenic cell death in murine colon cancer cells. Oncoimmunology. 2014;3:e28131. doi: 10.4161/onci.28131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ringel-Scaia VM, Beitel-White N, Lorenzo MF, Brock RM, Huie KE, Coutermarsh-Ott S. High-frequency irreversible electroporation is an effective tumor ablation strategy that induces immunologic cell death and promotes systemic anti-tumor immunity. EBioMedicine. 2019;44:112–25. doi: 10.1016/j.ebiom.2019.05.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schultheis K, Smith TRF, Kiosses WB, Kraynyak KA, Wong A, Oh J. Delineating the cellular mechanisms associated with skin electroporation. Hum Gene Ther Methods. 2018;29:177–88. doi: 10.1089/hgtb.2017.105. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhao J, Wen X, Tian L, Li T, Xu C, Wen X. Irreversible electroporation reverses resistance to immune checkpoint blockade in pancreatic cancer. Nat Commun. 2019;10:1–14. doi: 10.1038/s41467-019-08782-1. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Vogl TJ, Wissniowski TT, Naguib NNN, Hammerstingl RM, Mack MG, Münch S. Activation of tumor-specific T lymphocytes after laser-induced thermotherapy in patients with colorectal liver metastases. Cancer Immunol Immunother. 2019;58:1557–63. doi: 10.1007/s00262-009-0663-1. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bulvik BE, Rozenblum N, Gourevich S, Ahmed M, Andriyanov AV, Galun E. Irreversible electroporation versus radiofrequency ablation: a comparison of local and systemic effects in a small-animal model. Radiology. 2016;280:413–24. doi: 10.1148/radiol.2015151166. et al. [DOI] [PubMed] [Google Scholar]
- 32.White SB, Zhang Z, Chen J, Gogineni VR, Larson AC. Early immunologic response of irreversible electroporation versus cryoablation in a rodent model of pancreatic cancer. J Vasc Interv Radiol. 2018;29:1764–9. doi: 10.1016/j.jvir.2018.07.009. [DOI] [PubMed] [Google Scholar]
- 33.Scheffer HJ, Stam AGM, Geboers B, Vroomen LGPH, Ruarus A, de Bruijn B. Irreversible electroporation of locally advanced pancreatic cancer transiently alleviates immune suppression and creates a window for antitumor T cell activation. Oncoimmunology. 2019;8:1652532. doi: 10.1080/2162402X.2019.1652532. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pandit H, Hong YK, Li Y, Rostas J, Pulliam Z, Li P. Evaluating the regulatory immunomodulation effect of irreversible electroporation (ire) in pancreatic adenocarcinoma. Ann Surg Oncol. 2019;26:800–6. doi: 10.1245/s10434-018-07144-3. et al. [DOI] [PubMed] [Google Scholar]
- 35.Calvet CY, Mir LM. The promising alliance of anti-cancer electrochemotherapy with immunotherapy. Cancer Metastasis Rev. 2016;35:165–77. doi: 10.1007/s10555-016-9615-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Sersa G, Teissie J, Cemazar M, Signori E, Kamensek U, Marshall G. Electrochemotherapy of tumors as in situ vaccination boosted by immunogene electrotransfer. Cancer Immunol Immunother. 2015;64:1315–27. doi: 10.1007/s00262-015-1724-2. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Serša G, Miklavcic D, Cemazar M, Belehradek J, Jarm T, Mir LM. Electrochemotherapy with CDDP on LPB sarcoma: comparison of the anti- tumor effectiveness in immunocompetent and immunodeficient mice. Bioelectrochem Bioenerg. 1997;43:279–83. doi: 10.1016/S0302-4598(96)05194-X. [DOI] [Google Scholar]
- 38.Gerlini G, Tun-Kyi A, Dudli C, Burg G, Pimpinelli N, Nestle FO. Metastatic melanoma secreted IL-10 down-regulates CD1 molecules on dendritic cells in metastatic tumor lesions. Am J Pathol. 2004;165:1853–63. doi: 10.1016/S0002-9440(10)63238-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Gerlini G, Di Gennaro P, Mariotti G, Urso C, Chiarugi A, Pimpinelli N. Indoleamine 2,3-dioxygenase cells correspond to the BDCA2 plasmacytoid dendritic cells in human melanoma sentinel nodes. J Investig Dermatol. 2010;130:898–901. doi: 10.1038/jid.2009.307. et al. [DOI] [PubMed] [Google Scholar]
- 40.Geboers B, Scheffer HJ, Graybill PM, Ruarus AH, Nieuwenhuizen S, Puijk RS. High-voltage electrical pulses in oncology: irreversible electroporation, electrochemotherapy, gene electrotransfer, electrofusion, and electroimmunotherapy. Radiology. 2020;295:192190. doi: 10.1148/radiol.2020192190. et al. [DOI] [PubMed] [Google Scholar]
- 41.Zhou J, Wang G, Chen Y, Wang H, Hua Y, Cai Z. Immunogenic cell death in cancer therapy: Present and emerging inducers. J Cell Mol Med. 2019;23:4854–65. doi: 10.1111/jcmm.14356. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Alnaggar M, Lin M, Mesmar A, Liang S, Qaid A, Xu K. Allogenic natural killer cell immunotherapy combined with irreversible electroporation for stage iv hepatocellular carcinoma: survival outcome. Cell Physiol Biochem. 2018;48:1882–93. doi: 10.1159/000492509. et al. [DOI] [PubMed] [Google Scholar]
- 43.Yang Y, Qin Z, Du D, Wu Y, Qiu S, Mu F. Safety and short-term efficacy of irreversible electroporation and allogenic natural killer cell immunotherapy combination in the treatment of patients with unresectable primary liver cancer. Cardiovasc Intervent Radiol. 2019;42:48–59. doi: 10.1007/s00270-018-2069-y. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Lin M, Liang S, Wang X, Liang Y, Zhang M, Chen J. Percutaneous irreversible electroporation combined with allogeneic natural killer cell immunotherapy for patients with unresectable (Stage III/IV) pancreatic cancer: a promising treatment. J Cancer Res Clin Oncol. 2017;143:2607–18. doi: 10.1007/s00432-017-2513-4. [DOI] [PubMed] [Google Scholar]
- 45.Diercks GFH, Kluin PM. Jonkman FM. Autoimmune bullous diseases. Heidelberg: Springer International Publishing; 2016. Basic principles of the immune system and autoimmunity; pp. 3–12. editor. p. –. [DOI] [Google Scholar]
- 46.Kellie S, Al-Mansour Z. Skwarczynski M, Toth I. Micro- and nanotechnology in vaccine development. Elsevier Inc; 2017. Overview of the immune system; pp. 63–81. p. –. [DOI] [Google Scholar]
- 47.Chaplin DD. Overview of the immune response. J Allergy Clin Immunol. 2010;125:S3. doi: 10.1016/j.jaci.2009.12.980. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Roh JS, Sohn DH. Damage-associated molecular patterns in inflammatory diseases. Immune Netw. 2018;18:e27. doi: 10.4110/in.2018.18.e27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Obeid M, Tesniere A, Ghiringhelli F, Fimia GM, Apetoh L, Perfettini JL. Calreticulin exposure dictates the immunogenicity of cancer cell death. Nat Med. 2007;13:54–61. doi: 10.1038/nm1523. et al. [DOI] [PubMed] [Google Scholar]
- 50.Kato J, Svensson CI. Role of extracellular damage-associated molecular pattern molecules (DAMPs) as mediators of persistent pain. Prog Mol Biol Transl Sci. 2015;131:251–79. doi: 10.1016/bs.pmbts.2014.11.014. [DOI] [PubMed] [Google Scholar]
- 51.Bianchi ME. DAMPs, PAMPs and alarmins: all we need to know about danger. J Leukoc Biol. 2007;81:1–5. doi: 10.1189/jlb.0306164. [DOI] [PubMed] [Google Scholar]
- 52.Chan JK, Roth J, Oppenheim JJ, Tracey KJ, Vogl T, Feldmann M. Science in medicine Alarmins : awaiting a clinical response. J Clin Invest. 2012;122:2711–9. doi: 10.1172/JCI62423.tification. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Rock KL, Lai JJ, Kono H. Innate and adaptive immune responses to cell death. Immunol Rev. 2011;243:191–205. doi: 10.1111/j.1600-065X.2011.01040.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Stoecklein VM, Osuka A, Lederer JA. Trauma equals danger - damage control by the immune system. J Leukoc Biol. 2012;92:539–51. doi: 10.1189/jlb.0212072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wynn TA, Ramalingam TR. Mechanisms of fibrosis: therapeutic translation for fibrotic disease. Nat Med. 2012;18:1028–40. doi: 10.1038/nm.2807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Straino S, Di Carlo A, Mangoni A, De Mori R, Guerra L, Maurelli R. High-mobility group box 1 protein in human and murine skin: involvement in wound healing. J Invest Dermatol. 2008;128:1545–53. doi: 10.1038/sj.jid.5701212. [DOI] [PubMed] [Google Scholar]
- 57.Yang S, Xu L, Yang T, Wang F. High-mobility group box-1 and its role in angiogenesis. J Leukoc Biol. 2014;95:563–74. doi: 10.1189/jlb.0713412. [DOI] [PubMed] [Google Scholar]
- 58.Zampell JC, Yan A, Avraham T, Andrade V, Malliaris S, Aschen S. Temporal and spatial patterns of endogenous danger signal expression after wound healing and in response to lymphedema. Am J Physiol Cell Physiol. 2011;300:1107–21. doi: 10.1152/ajpcell.00378.2010. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Duffield JS, Lupher M, Thannickal VJ, Wynn TA. Host responses in tissue repair and fibrosis. Annu Rev Pathol. 2013;8:241–76. doi: 10.1146/annurevpathol-020712-163930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Rols MP, Teissié J. Electropermeabilization of mammalian cells. Quantitative analysis of the phenomenon. Biophys J. 1990;58:1089–98. doi: 10.1016/S0006-3495(90)82451-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Sweeney DC, Reberšek M, Dermol J, Rems L, Miklavčič D, Davalos RV. Quantification of cell membrane permeability induced by monopolar and high-frequency bipolar bursts of electrical pulses. Biochim Biophys Acta Biomembr. 2016;1858:2689–98. doi: 10.1016/j.bbamem.2016.06.024. [DOI] [PubMed] [Google Scholar]
- 62.Batista Napotnik T, Miklavčič D. In vitro electroporation detection methods – an overview. Bioelectrochemistry. 2018;120:166–82. doi: 10.1016/j.bioelechem.2017.12.005. [DOI] [PubMed] [Google Scholar]
- 63.Scuderi M, Rebersek M, Miklavcic D, Dermol-Cerne J. The use of high-frequency short bipolar pulses in cisplatin electrochemotherapy in vitro. Radiol Oncol. 2019;53:194–205. doi: 10.2478/raon-2019-0025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.O’Brien MA, Power DG, Clover AJP, Bird B, Soden DM, Forde PF. Local tumour ablative therapies: opportunities for maximising immune engagement and activation. Biochim Biophys Acta. 2014;184:510–23. doi: 10.1016/j.bbcan.2014.09.005. [DOI] [PubMed] [Google Scholar]
- 65.Babiuk S, Baca-Estrada ME, Foldvari M, Middleton DM, Rabussay D, Widera G. Increased gene expression and inflammatory cell infiltration caused by electroporation are both important for improving the efficacy of DNA vaccines. J Biotechnol. 2004;110:1–10. doi: 10.1016/j.jbiotec.2004.01.015. et al. [DOI] [PubMed] [Google Scholar]
- 66.Roos AK, Moreno S, Leder C, Pavlenko M, King A, Pisa P. Enhancement of cellular immune response to a prostate cancer DNA vaccine by intradermal electroporation. Mol Ther. 2006;13:320–7. doi: 10.1016/j.ymthe.2005.08.005. [DOI] [PubMed] [Google Scholar]
- 67.Chiarella P, Massi E, De Robertis M, Sibilio A, Parrella P, Fazio VM. Electroporation of skeletal muscle induces danger signal release and antigen-presenting cell recruitment independently of DNA vaccine administration. Expert Opin Biol Ther. 2008;8:1645–57. doi: 10.1517/14712598.8.11.1645. et al. [DOI] [PubMed] [Google Scholar]
- 68.Bessis N, Garcia Cozar FJ, Boissier MC. Immune responses to gene therapy vectors: influence on vector function and effector mechanisms. Gene Ther. 2004;11(Suppl 1):S10–7. doi: 10.1038/sj.gt.3302364. [DOI] [PubMed] [Google Scholar]
- 69.Shirley JL, de Jong YP, Terhorst C, Herzog RW. Immune responses to viral gene therapy vectors. Mol Ther. 2020;28:709–22. doi: 10.1016/j.ymthe.2020.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Rols MP, Teissié J. Electropermeabilization of mammalian cells. Quantitative analysis of the phenomenon. Biophys J. 1990;58:1089–98. doi: 10.1016/S0006-3495(90)82451-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Fan F, Wood KV. Bioluminescent assays for high-throughput screening. Assay Drug Dev Technol. 2007;5:127–36. doi: 10.1089/adt.2006.053. [DOI] [PubMed] [Google Scholar]
- 72.Wood KV. The bioluminescence advantage. https://worldwide.promega.com/resources/pubhub/enotes/the-bioluminescence-advantage/ [cited 2020 May 12]. Available at.
- 73.Falzoni S, Donvito G, Di Virgilio F. Detecting adenosine triphosphate in the pericellular space. Interface Focus. 2013;3:2012. doi: 10.1089/adt.2006.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Wang XQ, Xiao AY, Sheline C, Hyrc K, Yang A, Goldberg MP. Apoptotic insults impair Na+, K+-ATPase activity as a mechanism of neuronal death mediated by concurrent ATP deficiency and oxidant stress. J Cell Sci. 2003;116:2099–110. doi: 10.1242/jcs.00420. et al. [DOI] [PubMed] [Google Scholar]
- 75.Hansen EL, Sozer EB, Romeo S, Frandsen SK, Vernier PT, Gehl J. Dose-dependent ATP depletion and cancer cell death following calcium electroporation, relative effect of calcium concentration and electriamount strength. PLoS One. 2015;10:e0122973. doi: 10.1371/journal.pone.0122973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Ashdown CP, Johns SC, Aminov E, Unanian M, Connacher W, Friend J. Pulsed low-frequency magnetic fields induce tumor membrane disruption and altered cell viability. Biophys J. 2020;118:1552–63. doi: 10.1016/j.bpj.2020.02.013. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Krause KH, Michalak M. Calreticulin. Cell. 1997;88:439–43. doi: 10.1016/s0092-8674(00)81884-x. [DOI] [PubMed] [Google Scholar]
- 78.Gelebart P, Opas M, Michalak M. Calreticulin, a Ca2+-binding chaperone of the endoplasmic reticulum. Int J Biochem Cell Biol. 2005;37:260–6. doi: 10.1016/j.biocel.2004.02.030. [DOI] [PubMed] [Google Scholar]
- 79.Panaretakis T, Kepp O, Brockmeier U, Tesniere A, Bjorklund AC, Chapman DC. Mechanisms of pre-apoptotic calreticulin exposure in immunogenic cell death. Embo J. 2009;28:578–90. doi: 10.1038/emboj.2009.1. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Kranz P, Neumann F, Wolf A, Classen F, Pompsch M, Ocklenburg T. PDI is an essential redox-sensitive activator of PERK during the unfolded protein response (UPR) Cell Death Dis. 2017;8:e2986. doi: 10.1038/cddis.2017.369. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Hou W, Zhang Q, Yan Z, Chen R, Zeh HJ, Kang R. Strange attractors: DAMPs and autophagy link tumor cell death and immunity. Cell Death Dis. 2013;4:e966. doi: 10.1038/cddis.2013.493. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Pisetsky DS. The origin and properties of extracellular DNA: from PAMP to DAMP. Clin Immunolog. 2012;144:32–40. doi: 10.1016/j.clim.2012.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Shinohara K, Toné S, Ejima T, Ohigashi T, Ito A. Quantitative distribution of DNA, RNA, histone and proteins other than histone in mammalian cells, nuclei and a chromosome at high resolution observed by scanning transmission soft x-ray microscopy (stxm) Cells. 2019;8:164. doi: 10.3390/cells8020164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Mackenzie RJ. DNA vs. RNA – 5 key differences and comparison. Technology Networks. https://www.technologynet-works.com/genomics/lists/what-are-the-key-differences-between-dna-and-rna-296719 [cited 2020 Jun 9]. Available at.
- 85.Shi Y, Evans JE, Rock KL. Molecular identification of a danger signal that alerts the immune system to dying cells. Nature. 2003;425:516–21. doi: 10.1038/nature01991. [DOI] [PubMed] [Google Scholar]
- 86.Shi Y, Galusha SA, Rock KL. Cutting Edge: elimination of an endogenous adjuvant reduces the activation of cd8 t lymphocytes to transplanted cells and in an autoimmune diabetes model. J Immunol. 2006;176:3905–8. doi: 10.4049/jimmunol.176.7.3905. [DOI] [PubMed] [Google Scholar]
- 87.Miklavcic D, Semrov D, Mekid H, Mir LM. A validated model of in vivo electric field distribution in tissues for electrochemotherapy and fo: DNA electrotransfer for gene therapy. Biochim Biophys Acta Gen Subj. 2000;1523:73–83. doi: 10.1016/S0304-4165(00)00101-X. [DOI] [PubMed] [Google Scholar]
- 88.Zmuc J, Gasljevic G, Sersa G, Edhemovic I, Boc N, Seliskar A. Large liver blood vessels and bile ducts are not damaged by electrochemotherapy with bleomycin in pigs. Sci Rep. 2019;9:3649. doi: 10.1038/s41598-019-40395-y. et al. [DOI] [PMC free article] [PubMed] [Google Scholar]


