Abstract
The number of eye diseases treated with intravitreal injections is increasing. Obviously, an injection of fluid into the eye results in an increase of intraocular pressure (IOP), the main risk factor for glaucoma. However, the effect of these repeated IOP increases on the eye is unclear. Therefore, we performed a systematic review with meta-analyses. PubMed, Embase and Clinical Trials Registries were searched for articles investigating the relationship between intravitreal injections (anti-vascular endothelial growth factor [anti-VEGF] or steroids) and either IOP, retinal nerve fiber layer (RNFL)-thickness and glaucoma. Multiple meta-analyses were performed, combining data on intravitreal injection of anti-VEGF medication and dexamethasone implants. A total of 74 articles were eligible for meta-analyses. The short-term effect of an intravitreal injection of anti-VEGF showed a statistically significant increase in IOP. One day after injection of anti-VEGF, however, IOP was significantly lower than baseline. The long-term time-intervals showed no significant difference in IOP. After intravitreal injection of a dexamethasone implant, IOP was significantly higher than baseline 1 month post-injection. RNFL-thickness was significantly reduced 6 and 12 months post-injection of anti-VEGF, as well as at end of follow up. Caution is advised when using intravitreal medication, especially when treating patients with advanced glaucoma; in these cases, prophylactic IOP-lowering medication may be considered.
Subject terms: Ocular hypertension, Glaucoma, Retinal diseases
Introduction
The intravitreal administration of medication through injections has revolutionized the treatment for several retinal disorders. One of the first descriptions of an intravitreal injection was in 1911 when Ohm reported a method to inject air into the eye for retinal tamponade1,2. Almost half a century later, Sorsby and Ungar treated an endophthalmitis case with intravitreal administration of Penicillin3. Nowadays, besides antibiotics several types of intravitreal medications are available such as anti-vascular endothelial growth factor (anti-VEGF; e.g.bevacizumab, ranibizumab, and aflibercept). Since the introduction of anti-VEGF in 2004, the indications of anti-VEGF for retinal disorders have been increasing continuously4–6. Choroidal neovascularization is a leading cause of vision loss and can occur in a variety of retinopathies [e.g. diabetic retinopathy, age-related macular degeneration (AMD), and retinal vein occlusion]7,8. Currently approximately 196 million people suffer worldwide from AMD, an estimated 16.4 million suffer from some form of retinal vein occlusion, and 93 million people from diabetic retinopathy9–11. As a result of both population growth and increased lifespan globally, these retinopathies are only expected to become even more prevalent. For example, it is estimated that the number of people suffering from AMD will have risen to 288 million by 204010. The process of angiogenesis associated with these conditions is heavily mediated by VEGF12. Blocking the actions of VEGF has shown to be an effective way of blocking neovascularization, substantially limiting the vision loss associated with these retinopathies13. As a result routine injection of anti-VEGF has been implemented on an immense scale. More recently intravitreal steroids (e.g. dexamethasone implants) have also been introduced.
In general, these injections are considered relatively safe, nevertheless, long-term use of the injections can increase the chance of ocular complications14,15. Obviously, if a fluid is injected into the eye the intraocular pressure (IOP) should increase. Therefore immediately after the administration of an intravitreal injection, acute fluctuations of the IOP will be present; which are related to the procedure and usually last a maximum of a few hours14,15. Recently however, several studies have reported that the administration of multiple intravitreal injections with anti-VEGF is associated with an increased risk for sustained IOP elevation, especially in patients with pre-existing glaucoma16. An increase in IOP is a major risk factor for thinning of the retinal nerve fiber layer (RNFL)17. Chronic suppression of VEGF may cause harmful downstream effects on the RNFL. Furthermore, research has shown that the post-injection IOP fluctuations can also be damaging to the RNFL18. Steroids are also notorious for their steroid-induced increase in IOP19.
Although a lot of research has been done on this topic, the results are not consistent. It is clearly stated in multiple research what the short-term effects are; yet the long-term results have not been definitive 20. A recently published report on the effect of anti-VEGF on IOP and glaucoma has shown that already much research has been done on this topic, nonetheless the results have not been concisely quantified in a meta-analysis20.
Therefore, we performed a systematic review and meta-analyses to address the conflicting results regarding the short- and long-term effect of intravitreal injections on IOP, RNFL-thickness, and glaucoma. Using data from both observational and interventional studies, we calculated the pooled effect size for anti-VEGF and dexamethasone both for short- and long-term outcomes. Finally, we reported the current level of evidence for a potentially negative effect of intravitreal injections on IOP, RNFL-thickness, and glaucoma.
Methods
This systematic review was performed according to the Preferred Reporting Items for Systematic review and Meta-Analyses (PRISMA) and the Meta-analysis Of Observational Studies in Epidemiology (MOOSE) guidelines21,22.
Eligibility criteria for considering studies for this review
We only included observational or interventional quantitative studies, which reported on the relationship between intravitreal injection of anti-VEGF (bevacizumab, ranibizumab, and aflibercept) or steroids (dexamethasone implant) vs. either IOP, RNFL-thickness, or glaucoma. With the goal of maximizing generalizability we did not exclude studies based on the indication(s) for treatment. In contrast to the eligibility criteria for IOP, the studies which reported mean RNFL-thickness for a patient population which received multiple types of injections, were also included. Unfortunately, our literature search yielded no studies in which glaucoma served as the outcome.
Search methods for identifying studies
Two researchers (VAdeV and FLB) independently conducted literature searches in the PubMed, and Embase databases and Clinical Trial Registries (clinicaltrialsregister.eu; clinicaltrials.gov) on August 7th23,24, 2019. The following search strategy was performed to search for articles investigating the relationship between intravitreal injections and IOP: “(intravitreal bevacizumab AND intraocular pressure) OR (intravitreal ranibizumab AND intraocular pressure) OR (intravitreal aflibercept AND intraocular pressure) OR (intravitreal dexamethasone AND intraocular pressure)”. Similarly, a search was performed for studies investigating intravitreal injection and RNFL-thickness: “(intravitreal bevacizumab AND retinal nerve fiber layer) OR (intravitreal ranibizumab AND retinal nerve fiber layer) OR (intravitreal aflibercept AND retinal nerve fiber layer) OR (intravitreal dexamethasone AND retinal nerve fiber layer) OR (intravitreal AND retinal fiber layer)”. Finally, we performed a search for studies on the relationship between intravitreal injections and glaucoma: “(intravitreal injections) AND (glaucoma) NOT (neovascular glaucoma) NOT (surgery)”. We only included studies with an available abstract, with human participants, and studies of which a full text was available in either English or German.
Study selection
From the initial search, titles and abstracts were scanned independently by two authors for eligible articles, in which the relationship between intravitreal injection and IOP or RNFL-thickness, respectively, was investigated, without any other restricting selection criteria. Next, the full text of the eligible studies was used to select additional studies from the reference lists in a similar fashion. Finally, the results were compared, and discrepancies were discussed. If the two researchers could not reach consensus, a third researcher was involved to clarify.
Data collection and risk of bias assessment
For each study we extracted the name of the authors, title, year of publication, study design, sample size, method of assessing outcome(s), indication for injection, injected medication (exposure), means with corresponding standard deviations for both a post-injection and control group, and type of control (i.e., the same eye pre-injection, the contralateral eye, or healthy volunteers). To assess the methodological quality and risk of bias within individual studies the Newcastle–Ottawa Scale (NOS) for assessing the quality of comparative non-randomized studies was used. For randomized studies we used the Cochrane risk-of-bias Tool for Randomized Trials (RoB2). Two authors assessed the retrieved studies independently, after which any discrepancies were discussed. Where these two authors could not reach consensus, a third would give resolution. The certainty of evidence was determined by using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) approach.
Data synthesis and analysis
As the number of studies reporting an effect estimate of intravitreal injections on glaucoma was zero, a meta-analysis could not be performed. Therefore, we focused on the most important quantitative measures for treatment (IOP) and diagnosis (RNFL) of glaucoma. The primary outcome in the IOP-analyses and RNFL-thickness analyses was the difference in IOP and RNFL-thickness, respectively, between pre- (baseline) and post-injection (follow-up). For both primary outcomes we performed meta-analyses to calculate the weighted mean difference (WMD) with corresponding 95% confidence interval (CI) for each of the four intravitreal medications (e.g. bevacizumab, ranibizumab, aflibercept, or dexamethasone implant), and for all anti-VEGF medications combined, for all time intervals with at least 3 studies reporting IOP or RNFL. The specified time-intervals were: within 1 min after injection, 5 min after injection, 10 min, 15 min, 30 min, 1 h, 1 day, 1 week, 2 weeks, 1 month, 2 month, 3 months, 4 months, 6 months, and 12 months after injection. We also performed a meta-analysis for the RNFL-thickness at the end of follow-up. If a meaningful meta-analysis could not be conducted for one of the intravitreal medications, for example, in case of an insufficient number of studies, we reported the individual results of these studies. The threshold for statistical significance was identified as a P value of 0.05 or less.
Heterogeneity of the meta-analyses was measured by calculating I225. In case of low heterogeneity (I2 < 50%), a fixed effects model was used in our meta-analyses. In case of high heterogeneity (I2 > 50%) a random effects model was used.
Statistical analyses were performed using Review Manager 5.3 for Windows (Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration, 2014) and SPSS (IBM, Statistical Package for Social Sciences, Release 24.0.0.1).
Results
Across all performed searches a total of 4,024 articles were found, of which 77 were included in our review and 74 were included in our meta-analyses.
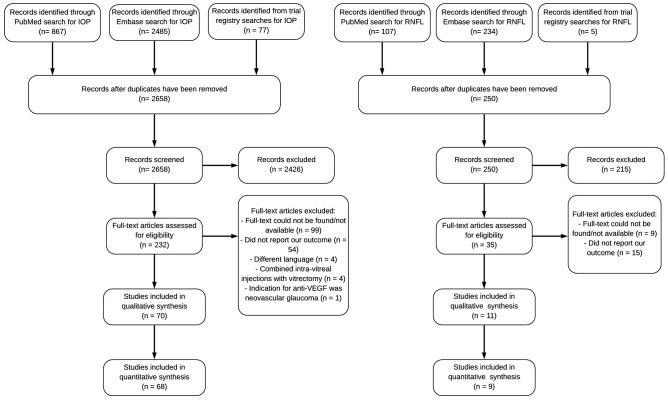
The literature search for studies investigating IOP yielded a total of 70 eligible articles26–95, of which 68 were eligible for meta-analyses (Fig. 1; Supplementary Table S1). The literature search for studies investigating RNFL-thickness yielded a total of 11 eligible articles58,86,96–103, of which 9 were eligible for meta-analyses (Fig. 1; Supplementary Table S1). Overall, the methodological quality within individual studies was good (Supplementary Tables S2, S3).
Figure 1.
Flow diagram (according to PRISMA) showing the selection process for inclusion of studies from our searches on the relationship between intravitreal injection and IOP or RNFL-thickness. IOP intraocular pressure, RNFL retinal nerve fiber layer, PRISMA Preferred Reporting Items for Systematic Reviews and Meta-Analyses.
The literature search for studies investigating the relationship between intravitreal injections and glaucoma yielded 260 articles from Pubmed and 448 articles from Embase. After scanning the articles and reading the full texts of potentially suitable articles, no studies were deemed eligible for inclusion.
Intravitreal injection of anti-VEGF and IOP
For the meta-analysis of bevacizumab, a total of 27 studies (1,488 eyes), investigating IOP after intravitreal injection were identified for meta-analysis (Supplementary Table S1; Supplementary Fig. S1). The WMD [95% CI] for IOP (in mmHg) was 23.88 [22.41, 25.34] immediately after injection and declined to 2.48 [1.24, 3.72] 30 min post-injection (Supplementary Fig. S2). On the day after injection the WMD of IOP (in mmHg) was significantly lower (− 1.19 [− 2.00, − 0.37]) compared to baseline, and no significant difference in IOP was found for any later time-interval. For ranibizumab, 14 studies (1,057 eyes) were included for the meta-analysis, which reported on IOP after intravitreal injection (Supplementary Table S1, Supplementary Fig. S3). The WMD [95% CI] in IOP (in mmHg) was 25.95 [21.46, 30.45] immediately after injection and declined to 2.61 [1.76, 3.46] 30 min post-injection (Supplementary Fig. S4). Although the IOP showed a downwards trend on the day after injection with an WMD of − 0.17 [− 0.59, 0.26], this difference was not statistically significant (P = 0.44). The WMD of IOP was also not significantly different than baseline after the later time-intervals. For aflibercept, 4 studies (133 eyes) were found (Supplementary Table S1). The minimum threshold of 3 studies or more was not reached for a single time-interval. Therefore, a meaningful meta-analysis could not be performed on the relationship between aflibercept intravitreal injections and post-injection IOP. Studies reporting on aflibercept were, however, included in our meta-analysis on IOP after intravitreal injection of any anti-VEGF (i.e., all anti-VEGF medications combined).
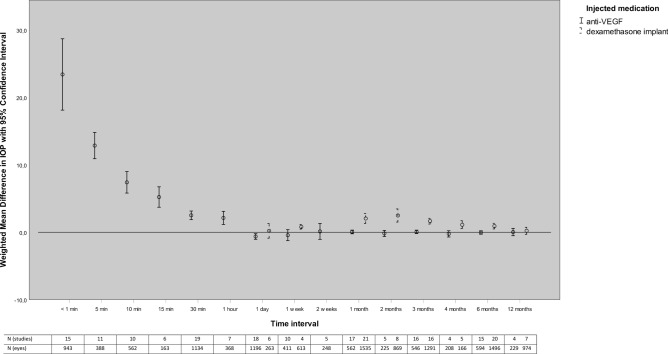
A total of 46 articles (2,872 eyes) were included for the meta-analyses on the relationship between the intravitreal injection of any anti-VEGF and post-injection IOP. The meta-analyses were performed for 15 different time intervals, ranging from the IOP immediately after injection (< 1 min) to the IOP after 12 months (Fig. 2; Supplementary Fig. S5). On the day of injection, we found for all included time-intervals a statistically significant increase in IOP. The WMD [95% CI] for IOP (in mmHg) was 23.41 [18.12, 28.70] immediately after injection and declined to 2.51 [1.87, 3.15] after 30 min. On the day after injection of anti-VEGF, however, the WMD [95% CI] for IOP (in mmHg) was significantly lower at − 0.63 [− 1.04, − 0.22]. IOP normalized after one week to 0.12 [− 1.05, 1.29] and no significant difference in IOP compared to baseline was found for all subsequent intervals. Heterogeneity (I2) was higher than 50% for all our short-term time-intervals up to and including IOP at 2 weeks post-injection. For IOP at 1-month post-injection and all later time-intervals (2, 3, 4, 6, and 12 months post-injection) heterogeneity was below 50%.
Figure 2.
Weighted mean difference in IOP during follow-up after intravitreal injection of anti-VEGF and Dexamethasone implant. IOP intraocular pressure, VEGF vascular endothelial growth factor.
Intravitreal injection of a dexamethasone implant and IOP
A total of 26 articles (1,744 eyes) on intravitreal injection of a dexamethasone implant and IOP were identified for meta-analysis (Fig. 2; Supplementary Fig. S6). None of the identified studies reported on the change in IOP within 1 day after injection. No statistically significant difference was found between the IOP 1 day after injection and IOP at baseline (P = 0.70). IOP was significantly increased at all later time-intervals, except of at end of follow-up (12 months: P = 0.11). The WMD [95% CI] for IOP (in mmHg) was 0.80 [0.47, 1.14] at 1 week, 2.04 [1.31, 2.78] at 1 month, 2.49 [1.51, 3.46] at 2 months, 1.65 [1.22, 2.07] at 3 months, 1.11 [0.53, 1.70] at 4 months, and 0.91 [0.49, 1.33] at 6 months after intravitreal injection. Heterogeneity (I2) was higher than 50% for all specified time-intervals, except for 1 week (I2 = 0%) and 12 months post-injection (I2 = 38%).
Intravitreal injection of anti-VEGF or dexamethasone implant and RNFL-thickness
Our literature search yielded 11 articles reporting on the relationship between anti-VEGF or dexamethasone implants and the average RNFL-thickness.
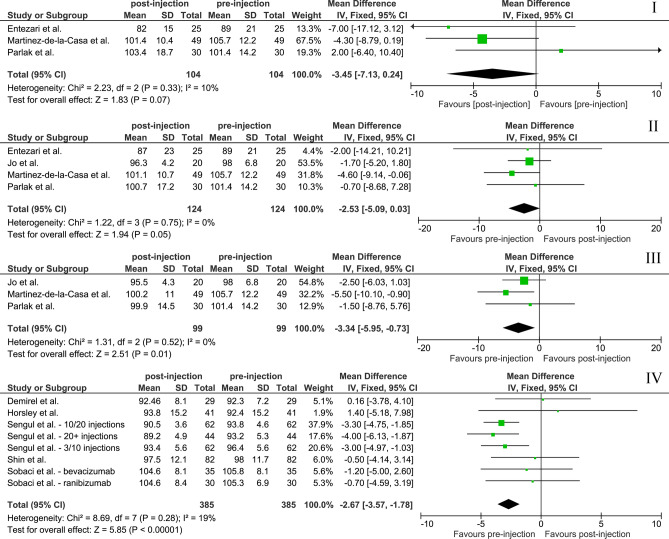
Five studies reported on RNFL-thickness at end of follow-up and were eligible for meta-analyses (Supplementary Table S1). Mean ± standard deviation follow-up for studies included in the meta-analysis was 23.40 ± 9.08 months. A total of 385 eyes were included in this meta-analysis. The WMD [95% CI] was − 2.67 [− 3.57, − 1.78] µm (Fig. 3).
Figure 3.
Meta‐analyses for the association of anti-VEGF and RNFL-thickness. Black diamonds indicate the overall WMD. The size of the gray box is inversely proportional to the variance. Horizontal lines indicate 95% CI. The vertical line in each panel shows the value for no effect (WMD = 0). RNFL retinal nerve fiber layer, WMD weighted mean difference, I 3 months post-injection, II 6 months, III 12 months, IV at end of follow-up.
Six studies investigated RNFL-thickness at a specific point in time during follow-up. Bulut et al. found that 6 months after dexamethasone implant the average RNFL-thickness had been reduced from 104.1 ± 11.1 µm to 101.1 ± 10.4 µm (P < 0.001)37. Four of the studies (124 eyes) were included in our meta-analysis for anti-VEGF. The WMD [95% CI] for RNFL-thickness (in µm) at 3, 6, and 12 months was − 3.45 [− 7.13, 0.24], − 2.53 [− 5.09, 0.03], and − 3.34 [− 5.95, − 0.73], respectively. RNFL-thickness was only significantly reduced at 12 months (P = 0.01; Fig. 3). Heterogeneity (I2) was low between 0 and 19% for the specified time-intervals.
Risk of bias and overall certainty of evidence
The mean ± standard deviation Newcastle–Ottawa sum score for all observational studies included in this review was 8.65 ± 0.66 (Supplementary Table S2). For observational studies investigating IOP and RNFL-thickness the score was 8.64 ± 0.69 and 8.77 ± 0.43, respectively. The Cochrane risk-of-bias for randomized studies tool showed a high risk of bias for all interventional studies (Supplementary Table S3). As per the GRADE approach, the evidence of this meta-analysis warrants a low level of certainty (Supplementary Table S4).
Discussion
Summary of findings
In this study we summarized the available scientific literature on the effect of anti-VEGF intravitreal injections and dexamethasone implant injections on the most important quantifiable measures for glaucoma (IOP and RNFL-thickness). After intravitreal injection of anti-VEGF, the IOP was significantly increased for all measured time-intervals on the day of injection. IOP was slightly decreased on the day after injection. IOP did not differ significantly from baseline during any later follow-up measurements. After the injection of a dexamethasone implant, IOP was increased for all long-term time-intervals until the 12-month endpoint.
Substantially less evidence was available on the relationship between intravitreal injections and RNFL-thickness. For our meta-analysis pooling all anti-VEGF results at end of follow-up we found a significant reduction in RNFL-thickness at 12 months and at end of follow-up. However, with a RNFL-thickness reduction of only 2 to 3 µm after an average follow-up of 23 months, the clinical relevance of these findings is unclear.
Relationship with literature
As mentioned before, the IOP showed a slight decrease the day immediately after injection, according to research this is most likely due to ciliary body inflammation as a result of the sharp IOP rise after the intravitreal injection76.
Although the literature on alternative corticosteroids is still sparse, some studies have shown that triamcinolone acetonide intravitreal injections result in similar rates of ocular hypertension compared to dexamethasone implant104,105. Similarly Vaz-Pereira et al. could not find a significant difference in IOP after switching from dexamethasone to fluocinolone acetonide106. More research will have to be done in order to be able to more clearly distinguish the pros and cons of the different corticosteroid injections available.
There are several known risk factors for glaucoma such as older age, a family history of glaucoma, and myopia107–109. It has been suggested that the recurrent transient rising IOP after intravitreal injection could also be correlated with an increased incidence of glaucoma. A recent report by the American Academy of Ophthalmology reviews this effect and found mixed results, with 6 studies reporting an incidence between 4 and 15% of sustained IOP elevation and 7 studies reporting no long-term change in IOP20. By performing meta-analyses, we have come to a more definitive answer: IOP normalizes within days after intravitreal injection of anti-VEGF, but after injection of a dexamethasone implant IOP remains elevated. Recurrent injections of anti-VEGF reduce RNFL-thickness; however, this reduction does not appear to be clinically relevant. Moreover, the studies used different optical coherence tomography (OCT)-devices of which the results may not be interchangeable110. However, within the studies baseline and follow-up RNFL were measured with the same OCT-device.
Several studies have shown that the post-intravitreal injection rise in IOP can be prevented by using glaucoma medication such as brimonidine/timolol and acetazolamide. For example, administering patients 500 mg oral acetazolamide 60–90 min before injection resulted in a significant reduction in IOP at 30 min post-injection of 5.5 mmHg74. Another study evaluated the effects of oral acetazolamide and topical brimonidine tartrate. In this study the IOP of pretreated eyes returned to baseline within 10 min, whereas eyes that were not pretreated remained elevated at 30 min post-injection111. A fixed combination of brimonidine/timolol twice a day on the day before and the day of the intravitreal injection was also an effective prophylaxis to reduce the acute IOP-spikes of the post-injection period112. At 5 min post-injection the mean IOP was significantly higher in the control group when compared with the pretreatment group. The mean IOP normalized at 15 min for the pretreatment group, whereas this was only the case after 30 min for the control group112. Additionally, several studies have shown that prophylactic anterior chamber paracentesis is a safe and effective option for preventing IOP spikes after an intravitreal injection87,113,114. Most notably, Enders et al. found a significant RNFL-loss in patients who had not undergone anterior chamber paracentesis prior to their intravitreal injections, but could not find a significant reduction in RNFL-thickness in those who did115. We can therefore conclude that there are several effective options for IOP prophylaxis. It has been suggested that the acute IOP changing after intravitreal injections could be causing perfusion-related injury to ocular structures, potentially resulting in glaucomatous damage to the macula and optic nerve116. This mechanism may be similar to the IOP-increase seen during intraocular surgery, which may result in a sudden loss of vision, also known as “wipe-out”117,118. During cataract surgery, which takes around 7 min, IOP increases up to 30–40 mmHg several times. This is one of the main reasons that ophthalmologists are very reluctant to operate on eyes with advanced glaucoma (visual field < 10°). A similar rise in IOP is seen after intravitreal injections, which therefore might have a similar effect of visual field loss. Scientific evidence to support this hypothesis is nonetheless lacking119.
IOP monitoring and prophylaxis should therefore be considered before giving intravitreal injections, especially in patients with advanced glaucoma or patients at risk for glaucoma who will receive a dexamethasone implant.
Strengths and weakness
The strengths of this study include a high quality and number of reviewed studies. By comparing the post-injection IOP or RNFL-thickness with the pre-injection IOP or RNFL-thickness of the same eye, the chance of confounding factors influencing the outcome was minimalized. Furthermore, we performed a large amount of meta-analyses to combine all available evidence in a concise and clear manner. By looking at both IOP and RNFL-thickness, we attempted to establish both the development of IOP over time after an intravitreal injection, and the long-term effect of these IOP elevations. Finally, by including studies regardless of the indication for an intravitreal injection, our results are generalizable, and its implications can be applied to a large number of patients. For example, most patients treated with anti-VEGF are treated with different types of anti-VEGF during their life depending on the effect of the anti-VEGF on the indicated disease.
Nevertheless, some limitations have to be considered. Most notably, the heterogeneity was high for some of our IOP related meta-analyses and especially for our short-term time-intervals. This is possibly due to the volatile and unpredictable nature of the immediate IOP rise after injection. Alternatively, the variability in IOP-measuring methods, frequency of injection, and the usage of IOP-lowering agents could have an effect on the heterogeneity of this review. During a previous study a greater IOP elevation post-injection was seen, if the interval between injections was less than 8 weeks and the frequency of injections in eyes with a sustained IOP elevation was significantly higher than in eyes without sustained IOP elevation120. Another factor that could contribute to the heterogeneity is the type of anti-VEGF medication used. We included studies evaluating the effect of either bevacizumab, ranibizumab or aflibercept. According to previous research, a significant IOP increase can be found when using bevacizumab and ranibizumab and not with aflibercept121–124. We did not exclude studies based on the use of anesthetic method for the intravitreal injection. One study has shown that decompressing the eye with lidocaine soaked cotton swabs applied with moderate pressure, compared to lidocaine gel applied without pressure, reduces the post-injection IOP spike52. A last factor that could explain the heterogeneity is that some studies did not exclude patients with a history of glaucoma. Several studies have shown a relationship between a history of glaucoma and higher IOP elevations after injection122,125,126.
Secondly, this systematic review is based primarily on observational studies, with only a view RCT’s included. Some unknown confounding or predisposing factors may have been missed; a threat inherent to the observational nature of these studies.
Thirdly, although the injection of a dexamethasone implant did seem to result in a sustained rise in IOP, we were not able to evaluate the long-term effects of this elevation on RNFL-thickness, due to a lack of available literature on this topic.
Conclusion and recommendation
IOP rises sharply immediately after intravitreal injection of anti-VEGF. This appears to be a transient rise, with the IOP normalizing within 1–7 days. However, after the injection of a dexamethasone implant, IOP will remain elevated for several months, before finally normalizing 1-year post-injection. Caution is therefore advised with the use of this form of intravitreal medication. Very little evidence is available on the long-term effects on RNFL-thickness. Some diminishment in RNFL-thickness seems to occur after intravitreal injection of anti-VEGF, but the clinical relevance of this finding remains unclear. In particular, the relationship between the intravitreal injection of a dexamethasone implant and RNFL-thickness requires further research. The current results suggest that the effect of intravitreal injection on glaucoma is not of clinical significance, however, caution should be taken when treating patients with advanced glaucoma. In these cases, prophylactic IOP-lowering medication or anterior chamber paracentesis should be considered. Future studies with visual field progression or glaucoma as an outcome itself are warranted.
Supplementary information
Author contributions
V.A.d.V. and W.D.R. wrote the main manuscript text and analyzed the data. V.A.d.V. and F.L.B. performed the systematic review and prepared the figures and tables. W.D.R. conceived and designed this study. All authors reviewed the manuscript. All authors read and approved the final manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-020-70269-7.
References
- 1.Ohm J. Über die Behandlung der Netzhautablösung durch operative Entleerung der subretinalen Flüssigkeit und Einspritzung von Luft in den Glaskörper. Albrecht Graefes Arch. Ophthalmol. 1911;79:442–450. [Google Scholar]
- 2.Ranibizumab (Lucentis) for macular degeneration. Med. Lett. Drugs Therap.48, 85–86 (2006). [PubMed]
- 3.Sorsby A, Ungar J. The control of experimental infections of the anterior chamber and of the vitreous by subconjunctival and retrobulbar injections of crystalline penicillin in doses of 1,000,000 units. Br. J. Ophthalmol. 1948;32:873–878. doi: 10.1136/bjo.32.12.873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Gragoudas ES, et al. Pegaptanib for neovascular age-related macular degeneration. N. Engl. J. Med. 2004;351:2805–2816. doi: 10.1056/NEJMoa042760. [DOI] [PubMed] [Google Scholar]
- 5.Michels S, Rosenfeld PJ, Puliafito CA, Marcus EN, Venkatraman AS. Systemic bevacizumab (Avastin) therapy for neovascular age-related macular degeneration twelve-week results of an uncontrolled open-label clinical study. Ophthalmology. 2005;112:1035–1047. doi: 10.1016/j.ophtha.2005.02.007. [DOI] [PubMed] [Google Scholar]
- 6.Rosenfeld PJ, et al. Ranibizumab for neovascular age-related macular degeneration. N. Engl. J. Med. 2006;355:1419–1431. doi: 10.1056/NEJMoa054481. [DOI] [PubMed] [Google Scholar]
- 7.Finger RP, Fimmers R, Holz FG, Scholl HP. Incidence of blindness and severe visual impairment in Germany: Projections for 2030. Invest. Ophthalmol. Vis. Sci. 2011;52:4381–4389. doi: 10.1167/iovs.10-6987. [DOI] [PubMed] [Google Scholar]
- 8.Bunce C, Xing W, Wormald R. Causes of blind and partial sight certifications in England and Wales: April 2007–March 2008. Eye (Lond) 2010;24:1692–1699. doi: 10.1038/eye.2010.122. [DOI] [PubMed] [Google Scholar]
- 9.Rogers S, et al. The prevalence of retinal vein occlusion: Pooled data from population studies from the United States, Europe, Asia, and Australia. Ophthalmology. 2010;117:313 e31–319 e311. doi: 10.1016/j.ophtha.2009.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wong WL, et al. Global prevalence of age-related macular degeneration and disease burden projection for 2020 and 2040: A systematic review and meta-analysis. Lancet Glob. Health. 2014;2:e106–116. doi: 10.1016/S2214-109X(13)70145-1. [DOI] [PubMed] [Google Scholar]
- 11.Yau JW, et al. Global prevalence and major risk factors of diabetic retinopathy. Diabetes Care. 2012;35:556–564. doi: 10.2337/dc11-1909. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Campochiaro PA. Molecular pathogenesis of retinal and choroidal vascular diseases. Prog. Retin. Eye Res. 2015;49:67–81. doi: 10.1016/j.preteyeres.2015.06.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Solomon SD, Lindsley K, Vedula SS, Krzystolik MG, Hawkins BS. Anti-vascular endothelial growth factor for neovascular age-related macular degeneration. Cochrane Database Syst. Rev. 2019;3:005139. doi: 10.1002/14651858.CD005139.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Bakri SJ, et al. Immediate intraocular pressure changes following intravitreal injections of triamcinolone, pegaptanib, and bevacizumab. Eye (Lond) 2009;23:181–185. doi: 10.1038/sj.eye.6702938. [DOI] [PubMed] [Google Scholar]
- 15.Falavarjani KG, Nguyen QD. Adverse events and complications associated with intravitreal injection of anti-VEGF agents: A review of literature. Eye (Lond.) 2013;27:787–794. doi: 10.1038/eye.2013.107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Good TJ, Kimura AE, Mandava N, Kahook MY. Sustained elevation of intraocular pressure after intravitreal injections of anti-VEGF agents. Br. J. Ophthalmol. 2011;95:1111–1114. doi: 10.1136/bjo.2010.180729. [DOI] [PubMed] [Google Scholar]
- 17.Medeiros FA, Alencar LM, Zangwill LM, Sample PA, Weinreb RN. The Relationship between intraocular pressure and progressive retinal nerve fiber layer loss in glaucoma. Ophthalmology. 2009;116(1125–1133):e1121–1123. doi: 10.1016/j.ophtha.2008.12.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Shin HJ, Kim SN, Chung H, Kim TE, Kim HC. intravitreal anti-vascular endothelial growth factor therapy and retinal nerve fiber layer loss in eyes with age-related macular degeneration: A meta-analysis. Invest. Ophthalmol. Vis. Sci. 2016;57:1798–1806. doi: 10.1167/iovs.15-18404. [DOI] [PubMed] [Google Scholar]
- 19.Gordon DM, et al. The use of ACTH and cortisone in ophthalmology. Am. J. Ophthalmol. 1951;34:1675–1686. doi: 10.1016/0002-9394(51)90032-3. [DOI] [PubMed] [Google Scholar]
- 20.Hoguet A, et al. The effect of anti-vascular endothelial growth factor agents on intraocular pressure and glaucoma: A report by the american academy of ophthalmology. Ophthalmology. 2019;126:611–622. doi: 10.1016/j.ophtha.2018.11.019. [DOI] [PubMed] [Google Scholar]
- 21.Liberati A, et al. The PRISMA statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. J. Clin. Epidemiol. 2009;62:e1–34. doi: 10.1016/j.jclinepi.2009.06.006. [DOI] [PubMed] [Google Scholar]
- 22.DF Stroup et al. Meta-analysis of observational studies in epidemiology: A proposal for reporting. meta-analysis of observational studies in epidemiology (MOOSE) group. JAMA. 2000;283:2008–2012. doi: 10.1001/jama.283.15.2008. [DOI] [PubMed] [Google Scholar]
- 23.European Medicines Agency. EU Clinical Trials Register version 2.2. https://www.clinicaltrialsregister.eu/.
- 24.U.S. National Library of Medicine. https://clinicaltrials.gov/ct2/home. [DOI] [PubMed]
- 25.Higgins JP, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta-analyses. BMJ. 2003;327:557–560. doi: 10.1136/bmj.327.7414.557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Alagoz N, et al. Immediate intraocular pressure changes following intravitreal dexamethasone implant. J. Ocul. Pharmacol. Ther. 2016;32:44–49. doi: 10.1089/jop.2015.0087. [DOI] [PubMed] [Google Scholar]
- 27.Alba-Linero C, et al. Long-term intravitreal dexamethasone implant outcomes in uveitis. Ocul. Immunol. Inflamm. 2019;20:1–10. doi: 10.1080/09273948.2019.1578380. [DOI] [PubMed] [Google Scholar]
- 28.Ali M, Khan FA, Zafar S, Akhtar F. Short and long term intraocular pressure changes after intravitreal injections of triamcinolone acetonide and bevacizumab in diabetic macular edema. Rawal. Med. J. 2014;39:443–445. [Google Scholar]
- 29.Al-khersan H, Hariprasad SM, Singh SR, Chhablani J. Long-term outcomes after intravitreal dexamethasone treatment in steroid responders. Acta Diabetol. 2019;56:675–680. doi: 10.1007/s00592-019-01299-5. [DOI] [PubMed] [Google Scholar]
- 30.Arikan G, Osman Saatci A, Hakan Oner F. Immediate intraocular pressure rise after intravitreal injection of ranibizumab and two doses of triamcinolone acetonide. Int. J. Ophthalmol. 2011;4:402–405. doi: 10.3980/j.issn.2222-3959.2011.04.16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Arikan Yorgun M, Toklu Y, Mutlu M, Uysal BS, Cakmak HB. Efficacy of single-dose dexamethasone implantation in patients with persistent diabetic macular edema. Int. Ophthalmol. 2016;36:531–539. doi: 10.1007/s10792-015-0155-6. [DOI] [PubMed] [Google Scholar]
- 32.Arrascue S, et al. Effects of the dexamethasone intravitreal implant (Ozurdex) on intraocular pressure for the treatment of macular edema. Rev. Mex. Oftalmol. 2018;92:201–206, 233–238. [Google Scholar]
- 33.Ayar O, et al. The effect of dexamethasone intravitreal implant on retinal nerve fiber layer in patients diagnosed with branch retinal vein occlusion. Curr. Eye Res. 2017;42:1287–1292. doi: 10.1080/02713683.2017.1313430. [DOI] [PubMed] [Google Scholar]
- 34.Bansal P, Gupta V, Gupta A, Dogra MR, Ram J. Efficacy of Ozurdex implant in recalcitrant diabetic macular edema—a single-center experience. Int. Ophthalmol. 2016;36:207–216. doi: 10.1007/s10792-015-0103-5. [DOI] [PubMed] [Google Scholar]
- 35.Bellocq D, et al. Effectiveness and safety of dexamethasone implants for post-surgical macular oedema including Irvine–Gass syndrome: The EPISODIC study. Br. J. Ophthalmol. 2015;99:979–983. doi: 10.1136/bjophthalmol-2014-306159. [DOI] [PubMed] [Google Scholar]
- 36.Bellocq D, et al. Effectiveness and safety of dexamethasone implants for postsurgical macular oedema including Irvine–Gass syndrome: The EPISODIC-2 study. Br. J. Ophthalmol. 2017;101:333–341. doi: 10.1136/bjophthalmol-2016-308544. [DOI] [PubMed] [Google Scholar]
- 37.Bulut MN, et al. Evaluation of peripapillary nerve fiber layer after dexamethasone implantation (ozurdex) in branch retinal vein occlusions. J. Ophthalmol. 2016;2016:2050796. doi: 10.1155/2016/2050796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Cacciamani A, et al. Intravitreal injection of bevacizumab: Changes in intraocular pressure related to ocular axial length. Jpn. J. Ophthalmol. 2013;57:63–67. doi: 10.1007/s10384-012-0194-8. [DOI] [PubMed] [Google Scholar]
- 39.Choi W, et al. Intraocular pressure change after injection of intravitreal dexamethasone (Ozurdex) implant in Korean patients. Br. J. Ophthalmol. 2018;20:20. doi: 10.1136/bjophthalmol-2018-312958. [DOI] [PubMed] [Google Scholar]
- 40.Coskun E, et al. Intravitreal dexamethasone implant for treatment of refractory behcet posterior uveitis: One-year follow-up results. Ocul. Immunol. Inflamm. 2015;23:437–443. doi: 10.3109/09273948.2015.1042167. [DOI] [PubMed] [Google Scholar]
- 41.Cunha RB, et al. Safety and feasibility of a novel 25-gauge biodegradable implant of dexamethasone for treatment of macular edema associated with retinal vein occlusion: A phase I clinical trial. Retin. Cases Brief. Rep. 2018;12:50–58. doi: 10.1097/ICB.0000000000000413. [DOI] [PubMed] [Google Scholar]
- 42.Demirel S, Yanik O, Batioglu F, Ozmert E. Intraocular pressure changes related to intravitreal injections of ranibizumab: Analysis of pseudophakia and glaucoma subgroup. Int. Ophthalmol. 2015;35:541–547. doi: 10.1007/s10792-014-9981-1. [DOI] [PubMed] [Google Scholar]
- 43.Ding X, et al. Prospective study of intravitreal triamcinolone acetonide versus bevacizumab for macular edema secondary to central retinal vein occlusion. Retina. 2011;31:838–845. doi: 10.1097/IAE.0b013e3181f4420d. [DOI] [PubMed] [Google Scholar]
- 44.El Chehab H, Agard E, Russo A, Boujnah Y, Dot C. Intraocular pressure spikes after aflibercept intravitreal injections. Ophthalmologica. 2016;236:43–47. doi: 10.1159/000446878. [DOI] [PubMed] [Google Scholar]
- 45.Faghihi H, Yahyapour H, Mahmoudzadeh R, Faghihi S. Comparison of intravitreal bevacizumab and intravitreal diclofenac in the treatment of diabetic macular edema: A 6-month follow-up. Med. Hypothesis Discov. Innov. Ophthalmol. 2017;6:67–75. [PMC free article] [PubMed] [Google Scholar]
- 46.Falkenstein IA, Cheng L, Freeman WR. Changes of intraocular pressure after intravitreal injection of bevacizumab (avastin) Retina. 2007;27:1044–1047. doi: 10.1097/IAE.0b013e3180592ba6. [DOI] [PubMed] [Google Scholar]
- 47.Frere A, et al. Single dexamethasone intravitreal implant in the treatment of noninfectious uveitis. J. Ocul. Pharmacol. Ther. 2017;33:290–297. doi: 10.1089/jop.2016.0139. [DOI] [PubMed] [Google Scholar]
- 48.Fuest M, Kotliar K, Walter P, Plange N. Monitoring intraocular pressure changes after intravitreal Ranibizumab injection using rebound tonometry. Ophthalm. Physiol. Opt. 2014;34:438–444. doi: 10.1111/opo.12134. [DOI] [PubMed] [Google Scholar]
- 49.Gado AS, Macky TA. Dexamethasone intravitreous implant versus bevacizumab for central retinal vein occlusion-related macular oedema: A prospective randomized comparison. Clin. Exp. Ophthalmol. 2014;42:650–655. doi: 10.1111/ceo.12311. [DOI] [PubMed] [Google Scholar]
- 50.Gismondi M, Salati C, Salvetat ML, Zeppieri M, Brusini P. Short-term effect of intravitreal injection of Ranibizumab (Lucentis) on intraocular pressure. J. Glaucoma. 2009;18:658–661. doi: 10.1097/IJG.0b013e31819c4893. [DOI] [PubMed] [Google Scholar]
- 51.Goktas A, Goktas S, Atas M, Demircan S, Yurtsever Y. Short-term impact of intravitreal ranibizumab injection on axial ocular dimension and intraocular pressure. Cutan. Ocul. Toxicol. 2013;32:23–26. doi: 10.3109/15569527.2012.696569. [DOI] [PubMed] [Google Scholar]
- 52.Gregori NZ, et al. Ocular decompression with cotton swabs lowers intraocular pressure elevation after intravitreal injection. J. Glaucoma. 2014;23:508–512. doi: 10.1097/IJG.0b013e318294865c. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Guler M, et al. Short-term effects of intravitreal bevacizumab on cornea and anterior chamber. Curr. Eye Res. 2014;39:989–993. doi: 10.3109/02713683.2014.888452. [DOI] [PubMed] [Google Scholar]
- 54.Higashiyama T, et al. Prospective comparisons of intravitreal injections of triamcinolone acetonide and bevacizumab for macular oedema due to branch retinal vein occlusion. Acta Ophthalmol. 2013;91:318–324. doi: 10.1111/j.1755-3768.2011.02298.x. [DOI] [PubMed] [Google Scholar]
- 55.Hohn F, Mirshahi A. Impact of injection techniques on intraocular pressure (IOP) increase after intravitreal ranibizumab application. Graefes Arch. Clin. Exp. Ophthalmol. 2010;248:1371–1375. doi: 10.1007/s00417-010-1382-1. [DOI] [PubMed] [Google Scholar]
- 56.Hollands H, et al. Short-term intraocular pressure changes after intravitreal injection of bevacizumab. Can. J. Ophthalmol. 2007;42:807–811. doi: 10.3129/i07-172. [DOI] [PubMed] [Google Scholar]
- 57.Jaffar S, Tayyab A, Matin ZI, Masrur A, Naqaish R. Effect of intra vitreal injection of bevacizumab on intra-ocular pressure. J. Ayub Med. Coll. Abbottabad. 2016;28:360–363. [PubMed] [Google Scholar]
- 58.Jo YJ, Kim WJ, Shin IH, Kim JY. Longitudinal changes in retinal nerve fiber layer thickness after intravitreal anti-vascular endothelial growth factor therapy. Korean J. Ophthalmol. 2016;30:114–120. doi: 10.3341/kjo.2016.30.2.114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Kaldirim HE, Yazgan S. A comparison of three different intravitreal treatment modalities of macular edema due to branch retinal vein occlusion. Int. Ophthalmol. 2018;38:1549–1558. doi: 10.1007/s10792-017-0618-z. [DOI] [PubMed] [Google Scholar]
- 60.Karakurt Y, et al. The effects of intravitreal ranibizumab, aflibercept or dexamethasone implant injections on intraocular pressure changes. Med. Sci. Monit. 2018;24:9019–9025. doi: 10.12659/MSM.910923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kernt M, Neubauer AS, Kampik A. Intravitreal bevacizumab (Avastin) treatment is safe in terms of intraocular and blood pressure. Acta. Ophthalmol. Scand. 2007;85:119–120. doi: 10.1111/j.1600-0420.2006.00819.x. [DOI] [PubMed] [Google Scholar]
- 62.Kim D, Nam WH, Kim HK, Yi K. Does intravitreal injections of bevacizumab for age-related macular degeneration affect long-term intraocular pressure? J. Glaucoma. 2014;23:446–448. doi: 10.1097/IJG.0b013e3182946505. [DOI] [PubMed] [Google Scholar]
- 63.Kim GN, et al. Effect of dorzolamide/timolol or brinzolamide/timolol prophylaxis on intravitreal anti-VEGF injection-induced intraocular hypertension. Semin. Ophthalmol. 2013;28:61–67. doi: 10.3109/08820538.2012.754479. [DOI] [PubMed] [Google Scholar]
- 64.Kim JY, Park SP. Comparison between intravitreal bevacizumab and triamcinolone for macular edema secondary to branch retinal vein occlusion. Korean J. Ophthalmol. 2009;23:259–265. doi: 10.3341/kjo.2009.23.4.259. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Lee K, Yang H, Lim H, Lew HM. A prospective study of blood pressure and intraocular pressure changes in hypertensive and nonhypertensive patients after intravitreal bevacizumab injection. Retina. 2009;29:1409–1417. doi: 10.1097/IAE.0b013e3181b21056. [DOI] [PubMed] [Google Scholar]
- 66.Lee JW, et al. Short-term changes of intraocular pressure and ocular perfusion pressure after intravitreal injection of bevacizumab or ranibizumab. BMC Ophthalmol. 2016;16:69. doi: 10.1186/s12886-016-0255-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lemos V, et al. Intraocular pressure in eyes receiving intravitreal antivascular endothelial growth factor injections. Ophthalmologica. 2015;233:162–168. doi: 10.1159/000369478. [DOI] [PubMed] [Google Scholar]
- 68.Lemos-Reis R, Moreira-Goncalves N, Melo AB, Carneiro AM, Falcao-Reis FM. Immediate effect of intravitreal injection of bevacizumab on intraocular pressure. Clin. Ophthalmol. 2014;8:1383–1388. doi: 10.2147/OPTH.S64721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lim HB, Kim MS, Jo YJ, Kim JY. Short-term visual acuity and intraocular pressure changes and their correlation after anti-vascular endothelial growth factor injection. Ophthalmologica. 2016;236:36–42. doi: 10.1159/000445038. [DOI] [PubMed] [Google Scholar]
- 70.Marey HM, Ellakwa AF. Intravitreal bevacizumab alone or combined with triamcinolone acetonide as the primary treatment for diabetic macular edema. Clin. Ophthalmol. 2011;5:1011–1016. doi: 10.2147/OPTH.S22103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Mazzarella S, et al. Effect of intravitreal injection of dexamethasone 0.7 mg (Ozurdex(R)) on intraocular pressure in patients with macular edema. Ophthalm. Res. 2015;54:143–149. doi: 10.1159/000438759. [DOI] [PubMed] [Google Scholar]
- 72.Mazzulla DA, Hariprasad SM, Jager RD, Mieler WF. Short-term intraocular pressure trends after intravitreal injection of bevacizumab (avastin) Retina Cases Brief. Rep. 2008;2:234–235. doi: 10.1097/ICB.0b013e31815e9409. [DOI] [PubMed] [Google Scholar]
- 73.Mishra S, Gupta A, Maggon R. Intravitreal dexamethasone v/s intravitreal triamcinolone in central retinal vein occlusion. Ophthalmologica. 2014;232:88. [Google Scholar]
- 74.Murray CD, Wood D, Allgar V, Walters G, Gale RP. Short-term intraocular pressure trends following intravitreal ranibizumab injections for neovascular age-related macular degeneration—the role of oral acetazolamide in protecting glaucoma patients. Eye (Lond) 2014;28:1218–1222. doi: 10.1038/eye.2014.180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Nalcaci S, Akkin C, Afrashi F. Dexamethasone implant in patients with diabetic macular edema resistant to anti-VEGF therapy. Turk. J. Ophthalmol. 2019;49:73–77. doi: 10.4274/tjo.galenos.2018.84665. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Omay E, Elgin U, Sen E, Yilmazbas P. The early effects of intravitreal anti vascular endothelial growth factor agents on intraocular pressure and central corneal thickness. Int. Ophthalmol. 2016;36:665–670. doi: 10.1007/s10792-016-0171-1. [DOI] [PubMed] [Google Scholar]
- 77.Özata K, Atum M, Çelik E, Doğan E, Alagöz G. Efficacy of intravitreal dexamethasone implant in persistent diabetic macular edema after primary treatment with intravitreal ranibizumab. J. Curr. Ophthalmol. 2019;20:20. doi: 10.1016/j.joco.2019.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Ozkan S, Fatih H, Erkan C, Birol T. Intravitreal bevacizumab versus bevacizumab and 1 mg triamcinolone acetonide in eyes with bilateral diabetic macular edema. Int. Eye Sci. 2019;19:1–8. [Google Scholar]
- 79.Özkaya A, et al. Comparing the effects of three different intravitreal injection techniques on vitreous reflux and intraocular pressure. J. Ocul. Pharmacol. Ther. 2013;29:325–329. doi: 10.1089/jop.2012.0144. [DOI] [PubMed] [Google Scholar]
- 80.Park J, Lee M. Short-term effects and safety of an acute increase of intraocular pressure after intravitreal bevacizumab injection on corneal endothelial cells. BMC Ophthalmol. 2018;18:17. doi: 10.1186/s12886-018-0682-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Pohlmann D, et al. Dexamethasone inserts in noninfectious uveitis: A single-center experience. Ophthalmology. 2018;125:1088–1099. doi: 10.1016/j.ophtha.2017.12.038. [DOI] [PubMed] [Google Scholar]
- 82.Ramezani A, et al. Three intravitreal bevacizumab versus two intravitreal triamcinolone injections in recent-onset branch retinal vein occlusion. Graefes Arch. Clin. Exp. Ophthalmol. 2012;250:1149–1160. doi: 10.1007/s00417-012-1941-8. [DOI] [PubMed] [Google Scholar]
- 83.Rebolleda G, et al. Optic nerve head biomechanic and IOP changes before and after the injection of aflibercept for neovascular age-related macular degeneration. Invest. Ophthalmol. Vis. Sci. 2016;57:5688–5695. doi: 10.1167/iovs.16-20111. [DOI] [PubMed] [Google Scholar]
- 84.Rensch F, Jonas JB, Spandau UH. Early intravitreal bevacizumab for non-ischaemic central retinal vein occlusion. Acta Ophthalmol. 2009;87:77–81. doi: 10.1111/j.1755-3768.2008.01313.x. [DOI] [PubMed] [Google Scholar]
- 85.Ryder SJ, Iannetta D, Bhaleeya SD, Kiss S. Efficacy and tolerability of bilateral sustained-release dexamethasone intravitreal implants for the treatment of noninfectious posterior uveitis and macular edema secondary to retinal vein occlusion. Clin. Ophthalmol. 2015;9:1109–1116. doi: 10.2147/OPTH.S84207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Sengul EA, et al. Retinal nerve fiber layer thickness changes in age-related macular degeneration treated with multiple intravitreal ranibizumab. J. Ocul. Pharmacol. Ther. 2016;32:665–670. doi: 10.1089/jop.2016.0014. [DOI] [PubMed] [Google Scholar]
- 87.Soheilian M, Karimi S, Montahae T, Nikkhah H, Mosavi SA. Effects of intravitreal injection of bevacizumab with or without anterior chamber paracentesis on intraocular pressure and peripapillary retinal nerve fiber layer thickness: A prospective study. Graefes Arch. Clin. Exp. Ophthalmol. 2017;255:1705–1712. doi: 10.1007/s00417-017-3702-1. [DOI] [PubMed] [Google Scholar]
- 88.Song JH, Lee JJ, Lee SJ. Comparison of the short-term effects of intravitreal triamcinolone acetonide and bevacizumab injection for diabetic macular edema. Korean J. Ophthalmol. 2011;25:156–160. doi: 10.3341/kjo.2011.25.3.156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Song S, Yu XB, Dai H. Effect of prophylactic intraocular pressure-lowering medication (brinzolamide) on intraocular pressure after ranibizumab intravitreal injection: A case–control study. Indian J. Ophthalmol. 2016;64:762–766. doi: 10.4103/0301-4738.195006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Tas M, et al. Single injection of triamcinolone versus three repeated injections of bevacizumab for treatment of diabetic macular edema. Int. Ophthalmol. 2013;33:375–380. doi: 10.1007/s10792-012-9709-z. [DOI] [PubMed] [Google Scholar]
- 91.Totan Y, Guler E, Guragac FB. Dexamethasone intravitreal implant for chronic diabetic macular edema resistant to intravitreal bevacizumab treatment. Curr. Eye Res. 2016;41:107–113. doi: 10.3109/02713683.2014.1002048. [DOI] [PubMed] [Google Scholar]
- 92.Tsai MJ, Hsieh YT, Peng YJ. Comparison between intravitreal bevacizumab and posterior sub-tenon injection of triamcinolone acetonide in macular edema secondary to retinal vein occlusion. Clin. Ophthalmol. 2018;12:1229–1235. doi: 10.2147/OPTH.S170562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Unsal E, Eltutar K, Sultan P, Erkul SO, Osmanbasoglu OA. Efficacy and safety of intravitreal dexamethasone implants for treatment of refractory diabetic macular edema. Korean J. Ophthalmol. 2017;31:115–122. doi: 10.3341/kjo.2017.31.2.115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Yilmaz I, et al. Ocular pulse amplitude and retinal vessel caliber changes after intravitreal dexamethasone implant. Middle East Afr. J. Ophthalmol. 2016;23:250–252. doi: 10.4103/0974-9233.186112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Zarranz-Ventura J, et al. Multicenter study of intravitreal dexamethasone implant in noninfectious uveitis: Indications, outcomes, and reinjection frequency. Am J. Ophthalmol. 2014;158:1136–1145 e1135. doi: 10.1016/j.ajo.2014.09.003. [DOI] [PubMed] [Google Scholar]
- 96.Demirel S, Batioglu F, Ozmert E, Erenler F. The effect of multiple injections of ranibizumab on retinal nerve fiber layer thickness in patients with age-related macular degeneration. Curr. Eye Res. 2015;40:87–92. doi: 10.3109/02713683.2014.917190. [DOI] [PubMed] [Google Scholar]
- 97.Horsley MB, Mandava N, Maycotte MA, Kahook MY. Retinal nerve fiber layer thickness in patients receiving chronic anti-vascular endothelial growth factor therapy. Am J. Ophthalmol. 2010;150:558–561 e551. doi: 10.1016/j.ajo.2010.04.029. [DOI] [PubMed] [Google Scholar]
- 98.Hwang DJ, Lee EJ, Lee SY, Park KH, Woo SJ. Effect of diabetic macular edema on peripapillary retinal nerve fiber layer thickness profiles. Invest. Ophthalmol. Vis. Sci. 2014;55:4213–4219. doi: 10.1167/iovs.13-13776. [DOI] [PubMed] [Google Scholar]
- 99.Martinez-de-la-Casa JM, et al. Retinal nerve fiber layer thickness changes in patients with age-related macular degeneration treated with intravitreal ranibizumab. Invest. Ophthalmol. Vis. Sci. 2012;53:6214–6218. doi: 10.1167/iovs.12-9875. [DOI] [PubMed] [Google Scholar]
- 100.Parlak M, Oner FH, Saatci AO. The long-term effect of intravitreal ranibizumab on retinal nerve fiber layer thickness in exudative age-related macular degeneration. Int. Ophthalmol. 2015;35:473–480. doi: 10.1007/s10792-014-9972-2. [DOI] [PubMed] [Google Scholar]
- 101.Shin HJ, Shin KC, Chung H, Kim HC. Change of retinal nerve fiber layer thickness in various retinal diseases treated with multiple intravitreal antivascular endothelial growth factor. Invest. Ophthalmol. Vis. Sci. 2014;55:2403–2411. doi: 10.1167/iovs.13-13769. [DOI] [PubMed] [Google Scholar]
- 102.Sobaci G, Gungor R, Ozge G. Effects of multiple intravitreal anti-VEGF injections on retinal nerve fiber layer and intraocular pressure: A comparative clinical study. Int. J. Ophthalmol. 2013;6:211–215. doi: 10.3980/j.issn.2222-3959.2013.02.20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Entezari M, Ramezani A, Yaseri M. Changes in retinal nerve fiber layer thickness after two intravitreal bevacizumab injections for wet type age-related macular degeneration. J. Ophthalm. Vis. Res. 2014;9:449–452. doi: 10.4103/2008-322X.150815. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Kuley B, et al. Ocular hypertension following intravitreal injection of 0.7 mg dexamethasone implant versus 2 mg triamcinolone. Semin. Ophthalmol. 2020;35:141–146. doi: 10.1080/08820538.2020.1758161. [DOI] [PubMed] [Google Scholar]
- 105.Thorne JE, et al. Periocular triamcinolone vs intravitreal triamcinolone vs intravitreal dexamethasone implant for the treatment of uveitic macular edema: The periocular vs intravitreal corticosteroids for uveitic macular edema (POINT) trial. Ophthalmology. 2019;126:283–295. doi: 10.1016/j.ophtha.2018.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Vaz-Pereira S, et al. The outcomes of switching from short- to long-term intravitreal corticosteroid implant therapy in patients with diabetic macular edema. Ophthalm. Res. 2020;63:114–121. doi: 10.1159/000503036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Le A, Mukesh BN, McCarty CA, Taylor HR. Risk factors associated with the incidence of open-angle glaucoma: The visual impairment project. Invest. Ophthalmol. Vis. Sci. 2003;44:3783–3789. doi: 10.1167/iovs.03-0077. [DOI] [PubMed] [Google Scholar]
- 108.Suzuki Y, et al. Risk factors for open-angle glaucoma in a Japanese population: The Tajimi Study. Ophthalmology. 2006;113:1613–1617. doi: 10.1016/j.ophtha.2006.03.059. [DOI] [PubMed] [Google Scholar]
- 109.European Glaucoma Prevention Study, G et al. Predictive factors for open-angle glaucoma among patients with ocular hypertension in the European Glaucoma Prevention Study. Ophthalmology. 2007;114:3–9. doi: 10.1016/j.ophtha.2006.05.075. [DOI] [PubMed] [Google Scholar]
- 110.Li XX, et al. A quantitative comparison of five optical coherence tomography angiography systems in clinical performance. Int. J. Ophthalmol. 2018;11:1784–1795. doi: 10.18240/ijo.2018.11.09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Katayama BY, et al. Comparison of acetazolamide, brimonidine, and anterior chamber paracentesis for ocular hypertension control after initial intravitreal bevacizumab injection: A randomized clinical trial. J. Glaucoma. 2014;23:461–463. doi: 10.1097/IJG.0b013e3182948476. [DOI] [PubMed] [Google Scholar]
- 112.Theoulakis PE, et al. Effect of brimonidine/timolol fixed combination on preventing the short-term intraocular pressure increase after intravitreal injection of ranibizumab. Klin. Monbl. Augenheilkd. 2010;227:280–284. doi: 10.1055/s-0029-1245201. [DOI] [PubMed] [Google Scholar]
- 113.Ichiyama Y, et al. Anterior chamber paracentesis might prevent sustained intraocular pressure elevation after intravitreal injections of ranibizumab for age-related macular degeneration. Ophthalm. Res. 2014;52:234–238. doi: 10.1159/000365253. [DOI] [PubMed] [Google Scholar]
- 114.Saxena S, et al. Anterior chamber paracentesis during intravitreal injections in observational trials: Effectiveness and safety and effects. Int. J. Retina Vitreous. 2019;5:8. doi: 10.1186/s40942-019-0157-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Enders P, et al. Retinal nerve fiber loss in anti-VEGF therapy for age-related macular degeneration can be decreased by anterior chamber paracentesis. Ophthalmologica. 2017;237:111–118. doi: 10.1159/000457907. [DOI] [PubMed] [Google Scholar]
- 116.Barash A, Chui TYP, Garcia P, Rosen RB. Acute macular and peripapillary angiographic changes with intravitreal injections. Retina. 2019;20:20. doi: 10.1097/IAE.0000000000002433. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Topouzis F, et al. Risk of sudden visual loss following filtration surgery in end-stage glaucoma. Am. J. Ophthalmol. 2005;140:661–666. doi: 10.1016/j.ajo.2005.04.016. [DOI] [PubMed] [Google Scholar]
- 118.Francis BA, et al. Vision loss and recovery after trabeculectomy: Risk and associated risk factors. Arch. Ophthalmol. 2011;129:1011–1017. doi: 10.1001/archophthalmol.2011.182. [DOI] [PubMed] [Google Scholar]
- 119.Moster MR, Moster ML. Wipe-out: A complication of glaucoma surgery or just a blast from the past? Am. J. Ophthalmol. 2005;140:705–706. doi: 10.1016/j.ajo.2005.05.024. [DOI] [PubMed] [Google Scholar]
- 120.Mathalone N, et al. Sustained elevation of intraocular pressure after intravitreal injections of bevacizumab in eyes with neovascular age-related macular degeneration. Graefes Arch. Clin. Exp. Ophthalmol. 2012;250:1435–1440. doi: 10.1007/s00417-012-1981-0. [DOI] [PubMed] [Google Scholar]
- 121.Al-Abdullah AA, Nowilaty SR, Asghar N, Al-Kharashi AS, Ghazi NG. Intraocular pressure trends after intravitreal injections of anti-vascular endothelial growth factor agents for diabetic macular edema. Retina. 2015;35:440–448. doi: 10.1097/IAE.0000000000000371. [DOI] [PubMed] [Google Scholar]
- 122.Atchison EA, et al. The Real-world effect of intravitreous anti-vascular endothelial growth factor drugs on intraocular pressure: An analysis using the IRIS Registry. Ophthalmology. 2018;125:676–682. doi: 10.1016/j.ophtha.2017.11.027. [DOI] [PubMed] [Google Scholar]
- 123.Rusu IM, et al. Intraocular pressure in patients with neovascular age-related macular degeneration switched to aflibercept injection after previous anti-vascular endothelial growth factor treatments. Retina. 2014;34:2161–2166. doi: 10.1097/IAE.0000000000000264. [DOI] [PubMed] [Google Scholar]
- 124.Wehrli SJ, et al. A lack of delayed intraocular pressure elevation in patients treated with intravitreal injection of bevacizumab and ranibizumab. Retina. 2012;32:1295–1301. doi: 10.1097/IAE.0b013e31823f0c95. [DOI] [PubMed] [Google Scholar]
- 125.Kim MJ, Choi DW, Choi SH, Heo JS, Sung JY. Surgical treatment of solid pseudopapillary neoplasms of the pancreas and risk factors for malignancy. Br. J. Surg. 2014;101:1266–1271. doi: 10.1002/bjs.9577. [DOI] [PubMed] [Google Scholar]
- 126.Boyer DS, Goldbaum M, Leys AM, Starita C, Group, V. I. S. I. O. N. S. Effect of pegaptanib sodium 0*3 mg intravitreal injections (Macugen) in intraocular pressure: Posthoc analysis from VISION study. Br. J. Ophthalmol. 2014;98:1543–1546. doi: 10.1136/bjophthalmol-2013-304075. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.