Abstract
Introduction
Severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2), which causes the coronavirus disease 2019 (COVID-19), may result in severe complications, multiorgan dysfunction, acute respiratory failure, and death. SARS-CoV-2 is highly contagious and places healthcare workers at significant risk, especially during aerosol-generating procedures, including airway management.
Objective
This narrative review outlines the underlying respiratory pathophysiology of patients with COVID-19 and discusses approaches to airway management in the emergency department (ED) based on current literature.
Discussion
Patients presenting with SARS-CoV-2 infection are at high risk for acute respiratory failure requiring airway management. Among hospitalized patients, 10–20% require intensive care unit admission, and 3–10% require intubation and mechanical ventilation. While providing respiratory support for these patients, proper infection control measures, including adherence to personal protective equipment policies, are necessary to prevent nosocomial transmission to healthcare workers. A structured approach to respiratory failure in these patients includes the use of exogenous oxygen via nasal cannula or non-rebreather, as well as titrated high-flow nasal cannula and non-invasive ventilation. This review offers several guiding principles and resources designed to be adapted in conjunction with local workplace policies for patients requiring respiratory support.
Conclusions
While the fundamental principles of acute respiratory failure management are similar between COVID-19 and non-COVID-19 patients, there are some notable differences, including a focus on provider safety. This review provides an approach to airway management and respiratory support in the patient with COVID-19.
Keywords: COVID-19, SARS-CoV-2, Coronavirus, Airway, Emergency medicine, Intensive care, Intubation
1. Introduction
Coronavirus disease 2019 (COVID-19) caused by severe acute respiratory syndrome-related coronavirus 2 (SARS-CoV-2) has become a pandemic involving millions of people and causing hundreds of thousands deaths worldwide [1]. COVID-19 was first reported within Wuhan, China in December 2019 and has spread rapidly [2]. Patients presenting with COVID-19 are at high risk for acute respiratory failure necessitating advanced airway management [[2], [3], [4]]. Overall, the presence of hypoxic respiratory failure among COVID-19 patients approaches 20% [[5], [6], [7]]. Data from China reported that up to 41% of all patients with COVID-19 required oxygen therapy, 4% to 13% required noninvasive ventilation (NIV), and 2.3% to 12% required intubation and mechanical ventilation [3,5]. Risk factors for developing acute respiratory failure appear to include male gender; age over 60 years; and comorbidities including diabetes, active cancer, and immunocompromising states [5,[8], [9], [10]].
Healthcare workers (HCWs) caring for this population are at high risk of contracting SARS-CoV-2 via large droplets, respiratory secretions, and contact with contaminated surfaces [11]. Airway management is particularly high risk because it involves aerosol-generating procedures [11,12]. Emergency providers must be prepared to manage patients with acute respiratory failure due to SARS-CoV-2. In this review, we provide an overview of the underlying respiratory pathophysiology of SARS-CoV-2, followed by an approach to airway management for suspected or confirmed patients with COVID-19, while maintaining the safety of HCWs and other patients.
2. Methods
This narrative review outlines the underlying respiratory pathophysiology and clinical manifestations of COVID-19 in the adult patient and discusses current approaches to airway management in the ED. A literature review of PubMed and Google Scholar databases was performed from January 1st, 2000 to May 20th, 2020 for articles using the keywords ‘COVID’ OR ‘SARS-CoV-2’ OR ‘coronavirus’ OR ‘SARS’ AND ‘airway management’ for production of this narrative review. Authors included case reports and series, retrospective and prospective studies, systematic reviews and meta-analyses, clinical guidelines, and other narrative reviews. Commentaries and letters were also included. The literature search was restricted to studies published or translated into English. The initial literature search revealed 1555 articles. Authors reviewed all relevant articles and decided which studies to include for the review by consensus, with focus on emergency medicine-relevant articles, including guidelines. When available, systematic reviews and meta-analyses were preferentially selected. These were followed sequentially by randomized controlled trials, prospective studies, retrospective studies, case reports, and other narrative reviews, when alternate data were not available. A total of 75 resources were selected for inclusion in this review. As this is a narrative review, the authors did not pool individual study data.
3. Discussion
3.1. Principles of management
The underlying pathophysiology of COVID-19 can lead to respiratory failure, with some patients developing acute respiratory distress syndrome (ARDS) [8,13]. In patients who deteriorate and require intensive care unit admission, NIV, invasive mechanical ventilation, or extracorporeal membrane oxygenation should be considered as necessary [14]. Respiratory failure due to SARS-CoV-2 and more common respiratory pathogens is similar, but requires slight variations to infection control policies and procedures, which are detailed below [14]. Concerning personal protective equipment (PPE), HCWs should follow the infection control policies at their individual healthcare institutions.
3.2. Supplemental oxygenation
Many guidelines recommend exogenous oxygen administration as an initial therapy in patients with mild hypoxemic respiratory failure due to COVID-19 [[15], [16], [17]]. Supplemental oxygen is recommended if the patient's oxygen saturation (SpO2) is less than 90%, with a target SpO2 of no higher than 96%, based on recommendations from several societies, including the Society of Critical Care Medicine (SCCM) [14,18].
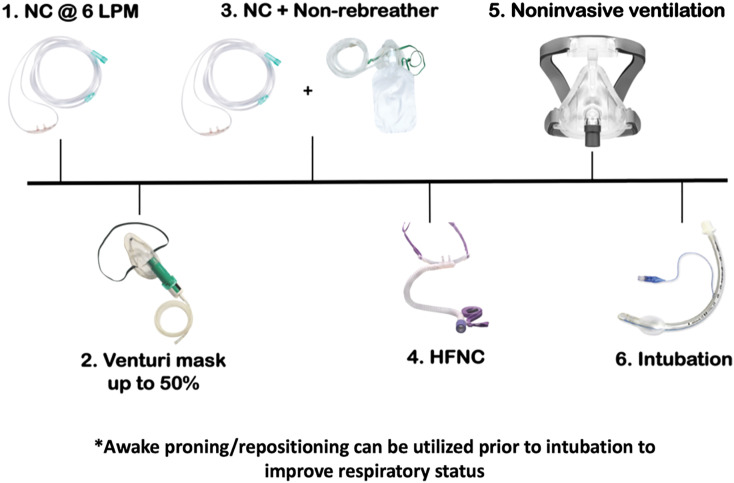
Clinicians should assess patient respiratory status inclusive of mental status and respiratory effort (e.g., work of breathing, respiratory rate) rather than oxygen saturation alone, when determining the need for airway intervention. In patients with mild hypoxemia due to SARS-CoV-2 but no evidence of respiratory failure requiring immediate endotracheal intubation, supplemental oxygen may be provided [4]. A strategy of oxygen escalation therapy may assist patients, in which nasal cannula (NC) can be started at 6 L/min [19]. If the patient does not improve with NC, further steps include Venturi mask up to 50% or non-rebreather mask up to 15 L/min, non-rebreather at 15 L/min in addition to NC at 6 L/min, high flow nasal cannula (HFNC), and then NIV (Fig. 1 ) [20]. Oxygen flow of 6 L/min or greater is considered high-flow oxygen and may cause aerosolization of viral pathogens, although this is controversial [[21], [22], [23]].
Fig. 1.
Oxygen Escalation Strategy. Abbreviations: NC – nasal cannula; LPM – liters per minute; HFNC – high flow nasal cannula; NIPPV – noninvasive positive pressure ventilation; CPAP – continuous positive airway pressure.
Respiratory support of patients with SARS-CoV-2 requires modification in order to minimize viral spread. For instance, a standard surgical mask should be worn over the NC, non-rebreather, or Venturi mask to reduce the risk of droplet spread [4]. For patients requiring higher oxygen delivery via the use of a simple facemask or nonrebreather, an exhalation filter can be attached; however, this strategy has not been thoroughly evaluated in terms of viral transmission [4]. As a result they are not recommended for routine use in patients with SARS-CoV-2 respiratory disease [20]. When necessary, non-rebreather masks are preferred over simple facemasks [19]. Similarly, nebulization of medications via simple facemask should be avoided in this population if possible [4,18,19]. Bronchodilators may be administered by metered-dose inhalers if necessary [4].
HFNC has become more prevalent in the years after the SARS outbreak and has been found to decrease the need for mechanical ventilation in patients with acute hypoxemic respiratory failure and potentially improve 90-day mortality [[24], [25], [26]]. HFNC is an emerging support modality for patients with COVID-19 and has been associated with increased survival in COVID-19 patients when compared to either NIV or invasive mechanical ventilation [9,27,28]. While the risk of bacterial transmission with HFNC is low, the risk of respiratory viral pathogen transmission remains unclear [[29], [30], [31], [32]]. Based on currently available evidence, the WHO states that “HFNC and NIV systems with good interface fitting do not create widespread dispersion of exhaled air and therefore should be associated with [a] low risk of airborne transmission.” [15] The risk of respiratory pathogen transmission when using HFNC is subject to a variety of factors, including the duration of support, maximal flow rate, patient sneezing or coughing, cannula fit, and patient cooperation [20]. Special attention must be paid to the connections between the oxygen tubing and the nasal cannula [19]. Any disruption to this connection may lead to dispersal of SARS-CoV-2 [33]. Some experts have recommended placement of a surgical mask over the HFNC to reduce viral transmission [19,34]. Patients on HFNC should be placed under airborne precautions in a negative-pressure room, if available [15,19].
HFNC provides gas flows between 40 and 60 L/min and may not result in aerosolization when compared to a patient on standard nasal cannula [35]. Many guidelines, including those by Australian and New Zealand Intensive Care Society (ANZICS), the WHO, and the Surviving Sepsis Campaign recommend the use of HFNC in COVID-19 patients presenting with acute hypoxemic respiratory failure unresponsive to conventional oxygen therapy [15,16,18]. Several groups have developed management strategies utilizing HFNC preferentially over NIV [9,36]. It is prudent to avoid HFNC in patients presenting with severe respiratory distress or failure, thoracoabdominal asynchrony, increasing vasopressor support, refractory hypoxemia despite other therapies, or a “clinical trajectory that suggests mechanical ventilation is inevitable” [4,25,37]. NIV may be more effective for other forms of respiratory failure, such as hypercapnic respiratory failure or obstructive airway disease [18]. However, HFNC can increase airway pressures, improve oxygenation, reduce dead space, and reduce a patient's work of breathing and can be utilized in patients with COVID-19 [38].
3.3. Non-invasive ventilation
International guidelines on the use of NIV for COVID-19 patients vary, with many guidelines recommending against the routine use of NIV due to increased risk of virus aerosolization and unproven utility in patients with ARDS [20]. Notably, the SCCM guidelines on the management of critically ill patients with COVID-19 recommend “a trial of NIV with close monitoring and short-interval assessment for worsening of respiratory failure” if HFNC is not available and there is no urgent indication for intubation [18]. Current epidemiological forecasts suggest that the requirements for mechanical ventilation may outpace the current ventilator capacity of many hospitals if NIV is not routinely used [18].
NIV does have limitations. In previous cohorts of patients with acute respiratory failure due to Influenza A, NIV failed in up to 85% of cases, portending a higher mortality compared to patients treated with invasive ventilation [39,40]. In a group of 302 patients with Middle East Respiratory Syndrome (MERS) across 14 Saudi Arabian hospitals, 92% of patients trialed on NIV failed to substantially improve, eventually requiring intubation [41]. A relatively similar failure rate was noted in a cohort of patients with COVID-19 associated respiratory failure in China [9]. There is also concern that NIV may worsen lung injury due to elevated transpulmonary pressures and large tidal volumes [42,43]. NIV use in patients with excessive respiratory efforts may induce substantial intrathoracic negative pressures and self-inflicted lung injury [43]. Additionally, the use of NIV may delay initiation of mechanical ventilation until the patient has no reserve, thereby increasing the risk of inappropriate donning of adequate PPE and transmission to HCWs due to time pressures to establish a definitive airway [4].
Although some centers reported successful management of SARS patients with NIV, there are documented cases of nosocomial transmission between patients in the same hospital [44,45]. This risk of aerosolization and viral transmission is variable, depending on a variety of factors, including the support parameters, model of the machine, and mask type [19,46]. This risk could be diminished by use of appropriate viral exhalation filters on the NIV and cohorting high-risk patients in an appropriate airborne isolation room [19]. However, any significant mask leak may render filtration of viral pathogens incomplete [4]. Despite these limitations, NIV may improve patient respiratory status and is a component of current guidelines [18,19]. NIV should be used in patients with COVID-19 with hypercarbic respiratory failure, refractory hypoxemia despite other therapies (including HFNC), or if HFNC is not available [18].
If utilized for patients with COVID-19, special attention should be paid to the use of viral filters, closed circuit systems, adequate mask seal, use of helmet systems (if available), appropriate PPE use, and appropriate isolation in a negative pressure room [19]. When available, a helmet-based NIV interface may have several advantages over traditional mask-based NIV, including decreased risk of aspiration and environmental contamination [33,47]. In one randomized clinical trial of 83 patients with ARDS, a NIV helmet reduced intubation rates and 90-day mortality compared to traditional NIV facemask [48].
3.4. Patient repositioning
Many clinicians have recommended awake proning or repositioning of patients on supplemental oxygen, HFNC, and NIV. Prone positioning of the patient may improve respiratory status and oxygenation, decreasing the need for endotracheal intubation in early ARDS [49,50]. While proning or repositioning may improve oxygenation, clinicians should be aware that this typically induces a temporary, non-sustainable improvement in oxygenation, and patients may require movement to another position (i.e., left lateral recumbent, right lateral recumbent, sitting upright) to maintain the benefit associated with this technique [49]. Patient comfort is important during proning/repositioning, and maternity cushioning devices may be beneficial. Regardless of oxygenation, these patients remain at risk for deterioration and must be monitored closely.
3.5. Decision to intubate
There are currently no evidence-based guidelines describing when to pursue intubation and mechanical ventilation for patients with SARS-CoV-2 [19]. However, in cases of severe respiratory distress or refractory hypoxemia despite oxygen escalation therapy including NIV, the patient should undergo endotracheal intubation and invasive ventilation [14]. Many patients who develop acute respiratory failure do so with hypoxemia and minimal signs of respiratory distress or tachypnea (so called “silent hypoxemia”), making work of breathing alone a potentially unreliable indicator for failure of NIV [51,52]. Thus, clinicians should consider patient mental status, work of breathing, respiratory rate, and oxygen saturation in their decision to intubate. In a series of 202 COVID-19 patients undergoing tracheal intubation in two hospitals in Wuhan, China, more than 75% of patients were hypoxemic (SaO2 < 90%) before induction [53]. The authors hypothesized that the shortage of available hospital beds during the COVID-19 pandemic, as well as result in delayed recognition respiratory failure severity due to “silent hypoxemia,” may have led to delays in the decision to intubate [53].
The Chinese Society of Anesthesiology Task Force on Airway Management recommends endotracheal intubation for patients showing no improvement in respiratory distress, tachypnea (respiratory rate > 30 breaths per minute), and poor oxygenation (PaO2 to FiO2 ratio ≤ 150 mmHg) after a 2 h trial of HFNC or NIV [8]. However, these recommendations are expert consensus and lack robust supporting evidence [54]. Some have liberalized their criteria, recommending that physicians consider intubation in any patient with respiratory distress (respiratory rate > 30 breaths per minute) or SpO2 less than 93% on room air and a PaO2 to FiO2 ratio less than 300 mmHg [54]. We recommend using a combination of factors in deciding to intubate, including progressively increasing oxygenation requirements despite oxygen escalation therapy, increasing vasopressor support, persistent thoracoabdominal asynchrony, increasing work of breathing, increasing respiratory distress, low ROX index (determined by oxygen saturation as measured by pulse oximetry divided by fraction of inspired oxygen [FiO2] divided by respiratory rate), hypercarbia, and altered mentation [19,25,36,37,55]. However, this must be balanced with potential resource limitations (availability of ventilators or staff, intensive care unit capability), clinical trajectory, and individual patient wishes [19].
3.6. Safety and preparedness
Airway management for patients with suspected or confirmed COVID-19 shares similarities with techniques in non-COVID-19 patients, with some notable exceptions, including an emphasis on staff safety throughout the procedure [56]. The increased risk of transmission of viral pathogens to HCWs with subsequent attempts necessitates the use of familiar and reliable airway techniques to ensure the greatest likelihood of first pass success [23]. Airborne precautions are indicated during the peri-intubation period, as the highest viral load appears in airway secretions of patients with COVID-19 [5,57]. Careful preparation and planning at the institutional level addressing appropriate equipment, staff preparedness, development of airway packs and endotracheal intubation checklists (Table 1 and Fig. 2 ), and availability of PPE are essential [23]. This should be augmented and evaluated using frequent in-situ simulation [23,58].
Table 1.
COVID-19 airway pack contents [33]
| Appropriate PPE for all team members |
| Induction medications: |
| High dose succinylcholine or rocuronium at 1.5–2.0 mg/kg |
| Properly dosed induction agent of choice |
| Post-intubation sedation and analgesia (Bolus and infusion) |
| Bag valve mask with PEEP valve and pressure manometer |
| High efficiency particulate air (HEPA) filter |
| Video laryngoscope tower with screen |
| Video laryngoscope blades (one each of size 3, 4, and hyperangulated) |
| Subglottic drainage tracheal tube (size 7.0 and 8.0) |
| Standard tracheal tube (multiple sizes, including size 7 and 8) and 10 mL syringe |
| Capnography monitoring line |
| Packets of water-soluble gel lubricant |
| Adult Magill forceps |
| Blue Portex swivel connector 15 mm |
| Gum elastic bougie |
| Video laryngoscope tracheal intubation stylet (for hyperangulated blades) |
| Supraglottic airways (multiple sizes) |
| Emergency front of neck airway (FONA) kit: |
| Size 10 scalpel |
| Size 6 cuffed endotracheal tube |
| Gum elastic bougie |
| (Available outside of room) Bronchoscope tower containing: |
| Single use “slim” (size 3.8) disposable bronchoscope |
| 4% lidocaine for airway topicalization |
| Mucosal atomizer |
| *Note: single-use equipment preferred, when available |
Abbreviations: PPE – personal protective equipment; mg/kg – milligrams per kilogram; PEEP – positive end-expiratory pressure; HEPA - High-efficiency particulate air; mL – milliliters; mm – millimeters; FONA – front of neck access.
Fig. 2.
A sample COVID-19 endotracheal intubation checklist.
Several patient, environmental, and team factors affect airway management in the COVID-19 patient. Endotracheal intubation is a high-risk procedure, with 10% of critically ill patients developing severe hypoxemia and 2% experiencing cardiac arrest [59,60]. The first pass success rate for endotracheal intubation among critically ill patients is typically less than 80%, with a significant proportion requiring two or more attempts [59]. These figures are likely to be worse in the critically ill COVID-19 patient due to the use of PPE and the patient's physiological state [23,61]. Patients with COVID-19 may experience myocardial injury which can worsen hemodynamic instability, lead to multiorgan failure, and reduce oxygen reserve [62]. Moreover, fogging of eyewear when using PPE during intubation affects up to 80% of providers, which can make intubation attempts more challenging [23]. Some clinicians have advocated for placement of a clear drape or box over the patients face to minimize aerosolization, but this may affect first pass success [63]. One study evaluating the use of aerosol boxes to protect HCW found reduced first pass success, longer time to intubation, and decreased laryngoscopic grade [64]. PPE may also decrease the clinician's field of vision, lead to reductions in manual dexterity, and interfere with team communication [65,66]. Team communication should use clear, direct language and closed loop communication [23,56]. Cognitive bandwidth and team communication may benefit from an endotracheal intubation checklist (Fig. 2).
Institutions should create a mobile endotracheal intubation pack that is decontaminated after each use (Table 1) [23,56]. This pack should preferentially contain single-use equipment brought to the patient's bedside during the procedure [4,23]. Some institutions may choose to include appropriate PPE for the airway management team. Additional airway equipment may be stored outside the negative pressure room as necessary [4]. All essential medications should be present before the procedure. Rapid sequence induction (RSI) medications should be drawn up and labeled. Depending on the patient's hemodynamic status, push dose vasopressors or a norepinephrine infusion should be readily available [56]. Ensure appropriate post-intubation analgesia, sedation, and paralyzing medications are present [56].
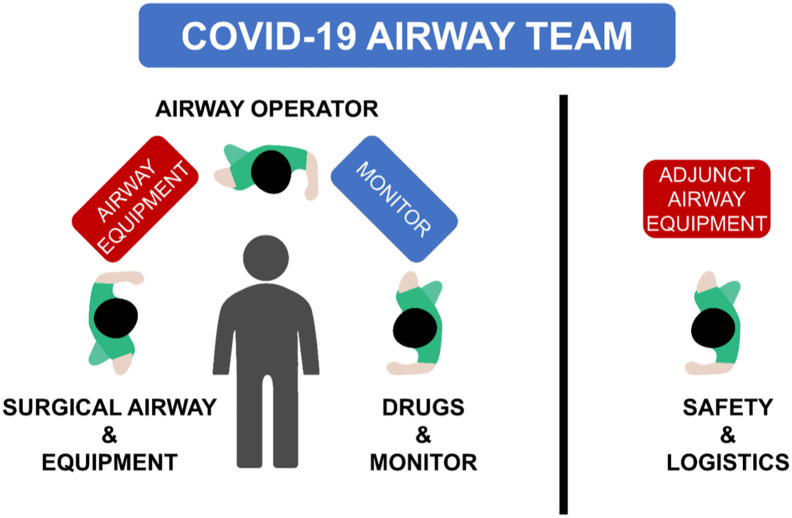
Staff members who are not involved in the procedure should not be present during any aerosol-generating procedure, including intubation [4]. There should be a clear delineation of roles and responsibilities on the team. Three personnel are likely sufficient within the room: an airway operator, an airway assistant, and a healthcare provider to give the medications and monitor the patient (Fig. 3 ) [23]. One to two team members should wait outside the room in PPE ready to enter if the primary team requires help or extra equipment, with an additional team member watching from the outside, ready to summon help rapidly if needed [23]. A designated safety/logistics officer should remain outside the room to observe for strict adherence to team safety and proper donning and doffing of PPE. A single team member may perform more than one role, depending on how many personnel are available.
Fig. 3.
The composition and roles of a COVID-19 airway team. During an intubation procedure, the discrete functional roles can be described as: (1) airway operator; (2) drug administrator and observer of patient's clinical state and monitors (drugs & monitor); (3) surgical airway operator and equipment assistant (surgical airway & equipment); (4) team member to fetch additional equipment or call for help during the procedure, and to observe strict adherence to team safety and donning and doffing of PPE in the peri-procedure period (safety and logistics officer). A single team member may perform more than one role, depending on how many personnel are available.
The choice of airway operator encompasses consideration of the available clinicians' airway expertise, predicted difficulty of intubation, patient factors, and clinician risk factors for poor outcomes if infected with SARS-CoV-2 [23,56]. While little guidance exists to risk stratify HCWs who are exposed to potential aerosol-generating procedures, it is prudent to exclude staff who are over the age of 60 years; pregnant; immunosuppressed; and those with cardiac disease, respiratory diseases, and recent cancer [5,8,23].
3.7. Intubation technique
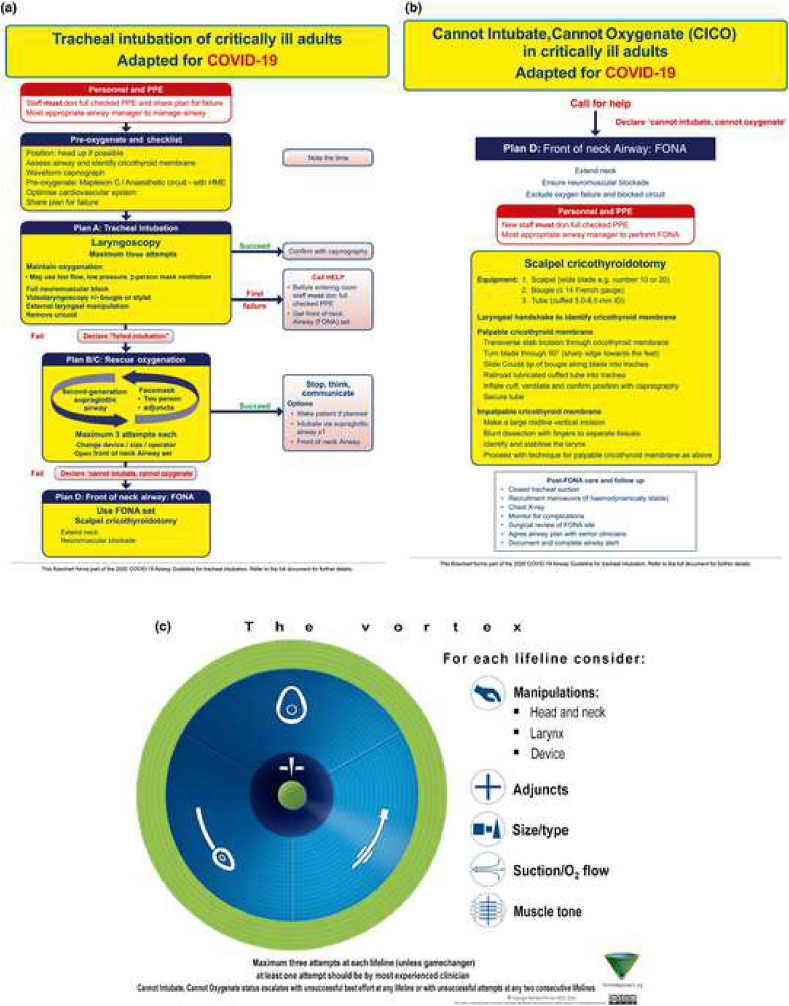
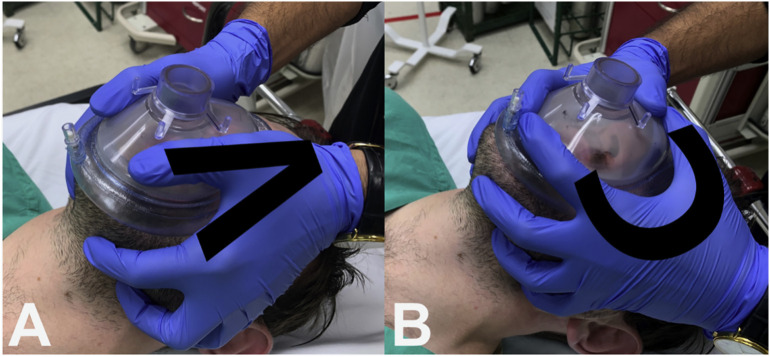
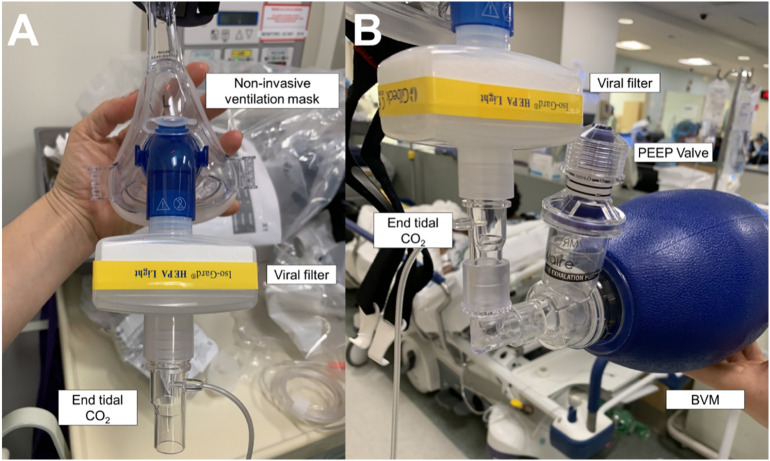
The airway strategy, including preoxygenation strategy, primary plan, rescue plans, and transitions, should be standardized. The basic algorithm for intubation is similar to the Difficult Airway Society (DAS) 2018 guideline for tracheal intubation of the critically ill patient (Fig. 4a and b) or the Vortex approach (Fig. 4c) [23,67]. There is emphasis on appropriate equipment selection, with a focus on closed systems to prevent viral transmission [4,23]. Closed suction systems should be used to minimize aerosolization. Apneic oxygenation should be avoided in these patients. However, preoxygenation may be accomplished with a bag valve mask (BVM) and viral exhalation filter [56,68]. The airway operator should ensure a tight BVM seal with two hands using the “V” grip while applying 10–15 cm of PEEP (Fig. 5 ). This BVM is held passively in order to maintain PEEP, preventing decruitment and hypoxia [56,62]. To improve mask seal and decrease airway operator fatigue, a NIV mask may be used in conjunction with a BVM to preoxygenate the patient (Fig. 6 ). For patients requiring manual ventilation, small tidal volumes are recommended [68]. Elevation of the head of the bed and ramping may be utilized [23].
Fig. 4.
Cognitive aids for use when managing unexpected difficulty when intubating a patient with coronavirus disease 2019. (a) Unexpected difficult tracheal intubation. (b) Cannot intubate, cannot oxygenate. (c) Vortex approach cognitive aid [23,67].
Fig. 5.
A. Two-handed BVM technique with the V-grip hand position. B. The C hand position, which should be avoided during preoxygenation of suspected or confirmed COVID-19 patients.
Fig. 6.
A. Noninvasive ventilation mask preoxygenation setup for suspected or confirmed COVID-19 patients. Note the mask is connected to a viral filter, then end tidal CO2 capnography monitoring line, a bag valve mask with a PEEP valve on the exhalation port, and finally a wall oxygen source (≥15 L/min).
The preferred airway management technique is RSI with the use of a video laryngoscope (VL) by the most appropriate clinician to maximize first pass success [18,23,53]. In a systemic review and meta-analysis of 64 studies, VL reduced the risk of failed intubation with no impact on the rate of first pass success, hypoxia, or time to endotracheal intubation [69]. If possible, the VL setup should include a monitor screen separate from the handle to reduce potential exposure to the patient's upper airway secretions. When available, a standard geometry video laryngoscope should be used in conjunction with a tracheal tube introducer (i.e., bougie), as this has been shown to improve first pass success when compared to a traditional stylet [56,70]. Awake flexible endoscopic intubation should be avoided, as the atomized local anesthesia may induce coughing [68]. When possible, direct laryngoscopy (DL) should be avoided as it places the face of the intubating clinician close to the patient's airway and may increase the risk of exposure [4,18].
RSI is recommended, as inadequate sedation or paralysis can produce coughing during laryngoscopy, generating aerosols [4,56]. A higher dose of sedative, as well as high-dose neuromuscular blockade should be administered during induction [23,56]. Nondepolarizing muscle relaxants such as rocuronium provide an advantage over depolarizing agents due to their extended duration of action, which prevents coughing should attempts at airway management be prolonged [20]. In order to improve ventilator synchrony and decrease aerosolization from inadequate sedation, prepare the patient's post-intubation analgesia and sedation before the procedure [56].
If the initial intubation is unsuccessful and the patient requires oxygenation, a second-generation supraglottic airway device can be used to reduce aerosolization risk [22,56,71]. There is no robust evidence to suggest that supraglottic devices are more effective than BVM in this scenario. However, they are easy to place and have better seal pressure compared to BVM, thus reducing staff exposure [20]. If oxygenation cannot be maintained using a BVM or a supraglottic device, a cricothyroidotomy should be performed [56]. The simplified DAS 2018 guidance should be followed (Fig. 4b). We recommend the scalpel-bougie-tube technique in order to decrease potential aerosolization [20,56]. While attempting an emergency front of neck access (FONA) procedure, application of oxygenation from above is not recommended, as this may cause aerosolization when the cricothyroid membrane is punctured [20]. Suction during the procedure should consist of a closed system with a viral filter [72].
3.8. Post-intubation period
After successful intubation, it is important to avoid ventilation until an appropriate viral filter is in place and the endotracheal tube cuff is inflated [23]. Ensure tight connections between all parts of the ventilator circuit, and avoid unnecessary disconnections whenever possible. However, if the circuit must be disconnected, clamp the endotracheal tube to prevent aerosolization. Endotracheal tube placement must be confirmed with waveform capnography, as PPE may preclude reliable auscultation of breath sounds [4,73]. Providers may also observe bilateral chest rise during assisted breaths, or alternatively, the ventilator waveform [62]. Ultrasonography is a useful adjunct for confirming endotracheal intubation, as it can allow direct confirmation without the requirement for ventilations [74]. A systematic review of 17 studies (n = 1595 patients) reported transtracheal ultrasonography is 98.7% sensitive (95% confidence interval [CI] 97.8% to 99.2%) and 97.1% specific (95% CI 92.4% to 99.0%), with a positive likelihood ratio of 34.4 (95% CI 12.7 to 93.1) and a negative likelihood ratio of 0.01 (95% CI 0.01 to 0.02) [75]. To prevent repeated exposures, a nasogastric tube may be inserted in the immediate post-intubation period [23]. Clinicians may also consider obtaining deep tracheal sputum samples. Ensure proper analgesia and sedation in order to prevent patient coughing and potential transmission of SARS-CoV-2 [56].
Any immediate post-intubation complications should be aggressively investigated and corrected. In a series of 202 COVID-19 patients undergoing tracheal intubation in two hospitals in Wuhan, China, peri-intubation hypotension (arterial blood pressure less than 90/60 mmHg) occurred in 22.3% of patients [53]. Pneumothorax occurred in 5.9% of patients, while 2% suffered peri-intubation cardiac arrest [53]. Peri-intubation hypotension should be managed with intravenous fluids and/or vasopressors, while pneumothorax may be corrected with chest tube drainage. After the immediate post-intubation period, equipment should be disposed of or decontaminated. Individual PPE should be removed under the guidance of a trained observer.
4. Conclusion
Patients with COVID-19 may develop acute respiratory failure and require respiratory support, as well as advanced airway management [4]. Airway management in these patients is a high-risk procedure for HCWs due to aerosolization and viral transmission [11]. The principles of airway management are similar between COVID-19 and non-COVID-19 patients, but with an enhanced focus on HCW safety [56]. A pragmatic approach to respiratory support in this population centers on appropriate infectious precautions (isolation, negative pressure rooms, and PPE), titrated support with exogenous oxygen, HFNC, NIV, and endotracheal intubation.
Declaration of Competing Interest
None.
Acknowledgements
BL, TM, MR, MG, and DH conceived the idea for this manuscript and contributed substantially to the writing and editing of the review. This manuscript did not utilize any grants, and it has not been presented in abstract form. This clinical review has not been published, it is not under consideration for publication elsewhere, its publication is approved by all authors and tacitly or explicitly by the responsible authorities where the work was carried out, and that, if accepted, it will not be published elsewhere in the same form, in English or in any other language, including electronically without the written consent of the copyright-holder. This review does not reflect the views or opinions of the U.S. government, Department of Defense, U.S. Army, U.S. Air Force, or SAUSHEC EM Residency Program.
References
- 1.World Health Organization. Situation Report 89. Geneva, Switzerland 2020.
- 2.Huang C., Wang Y., Li X. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet. 2020;395(10223):497–506. doi: 10.1016/S0140-6736(20)30183-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Guan W.J., Ni Z.Y., Hu Y. Clinical characteristics of coronavirus disease 2019 in China. N Engl J Med. 2020;382:1708–1720. doi: 10.1056/NEJMoa2002032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wax R.S., Christian M.D. Practical recommendations for critical care and anesthesiology teams caring for novel coronavirus (2019-nCoV) patients. Can J Anaesth. 2020;67(5):568–576. doi: 10.1007/s12630-020-01591-x. PMID: 32052373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Wu Z., McGoogan J.M. Characteristics of and important lessons from the coronavirus disease 2019 (COVID-19) outbreak in China: summary of a report of 72314 cases from the Chinese Center for Disease Control and Prevention. JAMA. 2020 doi: 10.1001/jama.2020.2648. [DOI] [PubMed] [Google Scholar]
- 6.Grasselli G., Zangrillo A., Zanella A. Baseline characteristics and outcomes of 1591 patients infected with SARS-CoV-2 admitted to ICUs of the Lombardy region, Italy. JAMA. 2020;323(16):1574–1581. doi: 10.1001/jama.2020.5394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bhatraju P.K., Ghassemieh B.J., Nichols M. Covid-19 in critically ill patients in the Seattle region - case series. N Engl J Med. 2020;382(21):2012–2022. doi: 10.1056/NEJMoa2004500. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zuo M., Huang Y., Ma W. Expert recommendations for tracheal intubation in critically ill patients with noval coronavirus disease 2019. CMSJ. 2020;35(2):105–109. doi: 10.24920/003724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Yang X., Yu Y., Xu J. Clinical course and outcomes of critically ill patients with SARS-CoV-2 pneumonia in Wuhan, China: a single-centered, retrospective, observational study. Lancet Respir Med. 2020;8(5):475–481. doi: 10.1016/s2213-2600(20)30079-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang D., Hu B., Hu C. Clinical characteristics of 138 hospitalized patients with 2019 novel coronavirus–infected pneumonia in Wuhan, China. JAMA. 2020;323(11) doi: 10.1001/jama.2020.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chang Xu H., Rebaza A., Sharma L., Dela Cruz C.S. Protecting health-care workers from subclinical coronavirus infection. Lancet Respir Med. 2020;8(3):e13. doi: 10.1016/S2213-2600(20)30066-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Tran K., Cimon K., Severn M., Pessoa-Silva C.L., Conly J. Aerosol generating procedures and risk of transmission of acute respiratory infections to healthcare workers: a systematic review. PLoS One. 2012;7(4) doi: 10.1371/journal.pone.0035797. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Xu Z., Shi L., Wang Y. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020;8(4):420–422. doi: 10.1016/S2213-2600(20)30076-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Giwa A.L., Desai A., Duca A. Novel 2019 coronavirus SARS-CoV-2 (COVID-19): an updated overview for emergency clinicians. Emerg Med Pract. 2020;22(5):1–28. [PubMed] [Google Scholar]
- 15.World Health Organization. Clinical management of severe acute respiratory infection when novel coronavirus (nCoV) infection is suspected. World Health Organization. https://www.who.int/publications-detail/clinical-management-of-severe-acute-respiratory-infection-when-novel-coronavirus-(ncov)-infection-is-suspected. Published 2020. Accessed Mar 20th, 2020.
- 16.The Australian and New Zealand Intensive Care Society. COVID-19 Guidelines ANZICS. https://www.anzics.com.au/coronavirus-guidelines/. Published 2020. Accessed Mar 20th, 2020.
- 17.Matos R, Chung K. COVID-19 practice management guide: Clinical management of COVID-19. In: Defense Do, ed 2020.
- 18.Alhazzani W., Moller M.H., Arabi Y.M. Surviving Sepsis campaign: guidelines on the management of critically ill adults with coronavirus disease 2019 (COVID-19) Intensive Care Med. 2020:1–34. doi: 10.1007/s00134-020-06022-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Whittle J.S., Pavlov I., Sacchetti A.D., Atwood C., Rosenberg M.S. Respiratory support for adult patients with COVID-19. J Am Coll Emerg Physic Open. 2020;1:95–101. doi: 10.1002/emp2.12071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Brewster D., Chrimes N., Do T. Consensus statement: safe airway society principles of airway management and tracheal intubation specific to the COVID-19 adult patient group. Med J Aust. 2020;212(10):472–481. doi: 10.5694/mja2.50598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Yu I.T., Xie Z.H., Tsoi K.K. Why did outbreaks of severe acute respiratory syndrome occur in some hospital wards but not in others? Clin Infect Dis. 2007;44(8):1017–1025. doi: 10.1086/512819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Cheung J.C., Ho L.T., Cheng J.V., Cham E.Y.K., Lam K.N. Staff safety during emergency airway management for COVID-19 in Hong Kong. Lancet Respir Med. 2020;8(4) doi: 10.1016/S2213-2600(20)30084-9. e19, Epub 2020 Feb 24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cook T.M., El-Boghdadly K., McGuire B., McNarry A.F., Patel A., Higgs A. Consensus guidelines for managing the airway in patients with COVID-19. Anaesthesia. 2020;75(6):785–799. doi: 10.1111/anae.15054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Rochwerg B., Granton D., Wang D.X. High flow nasal cannula compared with conventional oxygen therapy for acute hypoxemic respiratory failure: a systematic review and meta-analysis. Intensive Care Med. 2019;45(5):563–572. doi: 10.1007/s00134-019-05590-5. [DOI] [PubMed] [Google Scholar]
- 25.Rello J., Perez M., Roca O. High-flow nasal therapy in adults with severe acute respiratory infection: a cohort study in patients with 2009 influenza a/H1N1v. J Crit Care. 2012;27(5):434–439. doi: 10.1016/j.jcrc.2012.04.006. [DOI] [PubMed] [Google Scholar]
- 26.Frat J.P., Thille A.W., Mercat A. High-flow oxygen through nasal cannula in acute hypoxemic respiratory failure. N Engl J Med. 2015;372(23):2185–2196. doi: 10.1056/NEJMoa1503326. [DOI] [PubMed] [Google Scholar]
- 27.Wang K., Zhao W., Li J., Shu W., Duan J. The experience of high-flow nasal cannula in hospitalized patients with 2019 novel coronavirus-infected pneumonia in two hospitals of Chongqing, China. Ann Intensive Care. 2020;10(1):37. doi: 10.1186/s13613-020-00653-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Slessarev M., Cheng J., Ondrejicka M., Arntfield R. Patient self-proning with high-flow nasal cannula improves oxygenation in COVID-19 pneumonia. Can J Anesth. 2020:1–3. doi: 10.1007/s12630-020-01661-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Leung C.C.H., Joynt G.M., Gomersall C.D. Comparison of high-flow nasal cannula versus oxygen face mask for environmental bacterial contamination in critically ill pneumonia patients: a randomized controlled crossover trial. J Hosp Infect. 2019;101(1):84–87. doi: 10.1016/j.jhin.2018.10.007. [DOI] [PubMed] [Google Scholar]
- 30.Roberts S., Kabaliuk N., Spence C. Nasal high-flow therapy and dispersion of nasal aerosols in an experimental setting. J Crit Care. 2015;30(4) [Google Scholar]
- 31.Kotoda M., Hishiyama S., Mitsui K. Assessment of the potential for pathogen dispersal during high-flow nasal therapy. J Hosp Infect. 2019;104(4):534–537. doi: 10.1016/j.jhin.2019.11.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Montgomery J.M., Raboud J., Shigayeva A. Risk factors for SARS transmission from patients requiring intubation: a multicentre investigation in Toronto, Canada. PLoS ONE. 2010;5(5) doi: 10.1371/journal.pone.0010717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Hui D.S., Chow B.K., Lo T. Exhaled air dispersion during high-flow nasal cannula therapy versus CPAP via different masks. Eur Respir J. 2019;53(4) doi: 10.1183/13993003.02339-2018. [DOI] [PubMed] [Google Scholar]
- 34.Jhaveri R., Hui D.S., Chow B.K. Exhaled air dispersion during coughing with and without wearing a surgical or N95 mask. PLoS ONE. 2012;7(12) doi: 10.1371/journal.pone.0050845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Mellies U., Goebel C. Optimum insufflation capacity and peak cough flow in neuromuscular disorders. Ann Am Thorac Soc. 2014;11(10):1560–1568. doi: 10.1513/AnnalsATS.201406-264OC. [DOI] [PubMed] [Google Scholar]
- 36.Bouadma L., Lescure F.-X., Lucet J.-C., Yazdanpanah Y., Timsit J.-F. Severe SARS-CoV-2 infections: practical considerations and management strategy for intensivists. Intensive Care Med. 2020;46(4):579–582. doi: 10.1007/s00134-020-05967-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Sztrymf B., Messika J., Bertrand F. Beneficial effects of humidified high flow nasal oxygen in critical care patients: a prospective pilot study. Intensive Care Med. 2011;37(11):1780–1786. doi: 10.1007/s00134-011-2354-6. [DOI] [PubMed] [Google Scholar]
- 38.Lodeserto F.J., Lettich T.M., Rezaie S.R. High-flow nasal cannula: mechanisms of action and adult and pediatric indications. Cureus. 2018;10(11) doi: 10.7759/cureus.3639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kumar A., Zarychanski R., Pinto R. Critically ill patients with 2009 influenza A(H1N1) infection in Canada. JAMA. 2009;302(17):1872–1879. doi: 10.1001/jama.2009.1496. [DOI] [PubMed] [Google Scholar]
- 40.Rodriguez A., Ferri C., Martin-Loeches I. Risk factors for noninvasive ventilation failure in critically ill subjects with confirmed influenza infection. Respir Care. 2017;62(10):1307–1315. doi: 10.4187/respcare.05481. [DOI] [PubMed] [Google Scholar]
- 41.Alraddadi B.M., Qushmaq I., Al-Hameed F.M. Noninvasive ventilation in critically ill patients with the Middle East respiratory syndrome. Influenza Other Respi Viruses. 2019;13(4):382–390. doi: 10.1111/irv.12635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Beitler J.R., Malhotra A., Thompson B.T. Ventilator-induced lung injury. Clin Chest Med. 2016;37(4):633–646. doi: 10.1016/j.ccm.2016.07.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Brochard L., Lefebvre J.C., Cordioli R.L., Akoumianaki E., Richard J.C. Noninvasive ventilation for patients with hypoxemic acute respiratory failure. Semin Respir Crit Care Med. 2014;35(4):492–500. doi: 10.1055/s-0034-1383863. [DOI] [PubMed] [Google Scholar]
- 44.Cheung T.M., Yam L.Y., So L.K. Effectiveness of noninvasive positive pressure ventilation in the treatment of acute respiratory failure in severe acute respiratory syndrome. Chest. 2004;126(3):845–850. doi: 10.1378/chest.126.3.845. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Li Y., Huang X., Yu I.T., Wong T.W., Qian H. Role of air distribution in SARS transmission during the largest nosocomial outbreak in Hong Kong. Indoor Air. 2005;15(2):83–95. doi: 10.1111/j.1600-0668.2004.00317.x. [DOI] [PubMed] [Google Scholar]
- 46.Hui D.S., Chow B.K., Lo T. Exhaled air dispersion during noninvasive ventilation via helmets and a total facemask. Chest. 2015;147(5):1336–1343. doi: 10.1378/chest.14-1934. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cabrini L., Landoni G., Zangrillo A. Minimise nosocomial spread of 2019-nCoV when treating acute respiratory failure. Lancet. 2020;395(10225) doi: 10.1016/S0140-6736(20)30359-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Patel B.K., Wolfe K.S., Pohlman A.S., Hall J.B., Kress J.P. Effect of noninvasive ventilation delivered by helmet vs face mask on the rate of endotracheal intubation in patients with acute respiratory distress syndrome: a randomized clinical trial. JAMA. 2016;315(22):2435–2441. doi: 10.1001/jama.2016.6338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Ding L., Wang L., Ma W., He H. Efficacy and safety of early prone positioning combined with HFNC or NIV in moderate to severe ARDS: a multi-center prospective cohort study. Crit Care. 2020;24(1):28. doi: 10.1186/s13054-020-2738-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Munshi L., Del Sorbo L., Adhikari N.K.J. Prone position for acute respiratory distress syndrome. a systematic review and meta-analysis. Ann Am Thorac Soc. 2017;14(Supplement 4):S280–S288. doi: 10.1513/AnnalsATS.201704-343OT. [DOI] [PubMed] [Google Scholar]
- 51.Xie J., Tong Z., Guan X., Du B., Qiu H., Slutsky A.S. Critical care crisis and some recommendations during the COVID-19 epidemic in China. Intensive Care Med. 2020;46(5):837–840. doi: 10.1007/s00134-020-05979-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Tobin M.J. Basing respiratory Management of Coronavirus on physiological principles. Am J Respir Crit Care Med. 2020;201(11):1319–1320. doi: 10.1164/rccm.202004-1076ED. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Yao W., Wang T., Jiang B. Emergency tracheal intubation in 202 patients with COVID-19 in Wuhan, China: lessons learnt and international expert recommendations. Br J Anaesth. 2020;125(1):e28–e37. doi: 10.1016/j.bja.2020.03.026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Meng L., Qiu H., Wan L. Intubation and ventilation amid the COVID-19 outbreak. Anesthesiology. 2020;132(6):1317–1332. doi: 10.1097/ALN.0000000000003296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Roca O., Caralt B., Messika J. An index combining respiratory rate and oxygenation to predict outcome of nasal high-flow therapy. Am J Respir Crit Care Med. 2019;199(11):1368–1376. doi: 10.1164/rccm.201803-0589OC. [DOI] [PubMed] [Google Scholar]
- 56.Kovacs G., Sowers N., Campbell S., French J., Atkinson P. Just the facts: airway management during the COVID-19 pandemic. CJEM. 2020:1–7. doi: 10.1017/cem.2020.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wang W., Xu Y., Gao R. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA. 2020;323(18):1843–1844. doi: 10.1001/jama.2020.3786. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Abrahamson S.D., Canzian S., Brunet F. Using simulation for training and to change protocol during the outbreak of severe acute respiratory syndrome. Crit Care. 2006;10(1):R3. doi: 10.1186/cc3916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Nolan J.P., Kelly F.E. Airway challenges in critical care. Anaesthesia. 2011;66(Suppl. 2):81–92. doi: 10.1111/j.1365-2044.2011.06937.x. [DOI] [PubMed] [Google Scholar]
- 60.Higgs A., McGrath B.A., Goddard C. Guidelines for the management of tracheal intubation in critically ill adults. Br J Anaesth. 2018;120(2):323–352. doi: 10.1016/j.bja.2017.10.021. [DOI] [PubMed] [Google Scholar]
- 61.Yousif S., Machan J.T., Alaska Y., Suner S. Airway Management in Disaster Response: a manikin study comparing direct and video laryngoscopy for endotracheal intubation by Prehospital providers in level C personal protective equipment. Prehosp Disaster Med. 2017;32(4):352–356. doi: 10.1017/S1049023X17000188. [DOI] [PubMed] [Google Scholar]
- 62.Luo M., Cao S., Wei L. Precautions for intubating patients with COVID-19. Anesthesiology. 2020 doi: 10.1097/ALN.0000000000003288. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Canelli R., Connor C.W., Gonzalez M., Nozari A., Ortega R. Barrier enclosure during endotracheal intubation. N Engl J Med. 2020;382:1957–1958. doi: 10.1056/NEJMc2007589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Begley J.L., Lavery K.E., Nickson C.P., Brewster D.J. The aerosol box for intubation in COVID-19 patients: an in-situ simulation crossover study. Anaesthesia. 2020;75(8):1014–1021. doi: 10.1111/anae.15115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Visentin L.M., Bondy S.J., Schwartz B., Morrison L.J. Use of personal protective equipment during infectious disease outbreak and nonoutbreak conditions: a survey of emergency medical technicians. CJEM. 2009;11(1):44–56. doi: 10.1017/s1481803500010915. [DOI] [PubMed] [Google Scholar]
- 66.Daugherty E.L., Perl T.M., Needham D.M., Rubinson L., Bilderback A., Rand C.S. The use of personal protective equipment for control of influenza among critical care clinicians: a survey study. Crit Care Med. 2009;37(4):1210–1216. doi: 10.1097/CCM.0b013e31819d67b5. [DOI] [PubMed] [Google Scholar]
- 67.Chrimes N. The Vortex approach. http://vortexapproach.org. Published 2016. Accessed 25 Mar, 2020.
- 68.Peng P.W.H., Ho P.-L., Hota S.S. Outbreak of a new coronavirus: what anaesthetists should know. Br J Anaesth. 2020;124(5):497–501. doi: 10.1016/j.bja.2020.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Lewis S.R., Butler A.R., Parker J., Cook T.M., Smith A.F. Videolaryngoscopy versus direct laryngoscopy for adult patients requiring tracheal intubation. Cochrane Database Syst Rev. 2016;11 doi: 10.1002/14651858.CD011136.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Driver B.E., Prekker M.E., Klein L.R. Effect of use of a Bougie vs endotracheal tube and Stylet on first-attempt intubation success among patients with difficult airways undergoing emergency intubation: a randomized clinical trial. JAMA. 2018;319(21):2179–2189. doi: 10.1001/jama.2018.6496. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Keller C., Brimacombe J., Kleinsasser A., Brimacombe L. The Laryngeal Mask Airway ProSeal(TM) as a temporary ventilatory device in grossly and morbidly obese patients before laryngoscope-guided tracheal intubation. Anesth Analg. 2002;94(3):737–740. doi: 10.1097/00000539-200203000-00048. ; table of contents. [DOI] [PubMed] [Google Scholar]
- 72.Tay J.K., Khoo M.L.-C., Loh W.S. Surgical considerations for tracheostomy during the COVID-19 pandemic. JAMA Otolaryngol Head Neck Surg. 2020 doi: 10.1001/jamaoto.2020.0764. [DOI] [PubMed] [Google Scholar]
- 73.White S.J. Auscultation without contamination: a solution for stethoscope use with personal protective equipment. Ann Emerg Med. 2015;65(2):235–236. doi: 10.1016/j.annemergmed.2014.11.021. [DOI] [PubMed] [Google Scholar]
- 74.Gottlieb M., Holladay D., Burns K.M., Nakitende D., Bailitz J. Ultrasound for airway management: an evidence-based review for the emergency clinician. Am J Emerg Med. 2019;38(5):1007–1013. doi: 10.1016/j.ajem.2019.12.019. [DOI] [PubMed] [Google Scholar]
- 75.Gottlieb M., Holladay D., Peksa G.D. Ultrasonography for the confirmation of endotracheal tube intubation: a systematic review and meta-analysis. Ann Emerg Med. 2018;72(6):627–636. doi: 10.1016/j.annemergmed.2018.06.024. [DOI] [PubMed] [Google Scholar]