Abstract
Human papilloma virus (HPV)-positive oropharyngeal squamous cell carcinomas (OPSCC) tend to have good outcomes, however a subset does not share this favourable prognosis. The aim of this paper is to investigate the utility of tumour cell anaplasia and multinucleation as prognostic markers in oropharyngeal squamous cell carcinoma. Retrospective review of 104 patients with OPSCC or squamous cell carcinoma of unknown primary site (SCCUP) who underwent primary resection and/or lymph node dissection. Slides of both primary and nodal metastatic disease were assessed for the presence of anaplasia and multinucleation. 53 patients were HPV-positive. Anaplasia was more frequent in males (p = 0.005), smokers (p = 0.003), and HPV-negative disease (p = 0.04). HPV status and > 10 pack-year smoking history were independent predictors of recurrence-free survival (RFS) and disease-specific survival (DSS). Neither anaplasia, nor multinucleation, at the primary site or in cervical metastases, had any significant impact on RFS or DSS. We did not find either anaplasia or multinucleation to have any significant prognostic impact in OPSCC. However, given the small number of adverse events in the HPV-positive cohort, we may have lacked sufficient power to detect significance in what was the subgroup of primary interest. Our study highlights the challenge of identifying markers of poor prognosis in HPV-positive OPSCC.
Electronic supplementary material
The online version of this article (10.1007/s12105-019-01081-7) contains supplementary material, which is available to authorized users.
Keywords: Oropharynx, Squamous cell carcinoma, Human papilloma virus, Anaplasia, Multinucleation
Introduction
Recent decades have seen a marked increase in the incidence of oropharyngeal squamous cell carcinoma (OPSCC) in Western countries, which appears to be driven by an upsurge in the incidence of HPV-related OPSCC. HPV-positive OPSCC is associated with better treatment response and improved survival outcomes compared with HPV-negative cases [1–11]. Consequently, there is growing interest in de-escalation treatment strategies for this patient cohort. Numerous de-intensification clinical trials are ongoing, the ultimate aim of which is avoidance of unnecessary radiotherapy and/or chemotherapy related morbidity [12].
On the other hand, there is a subset (approximately 15–20%) of patients with HPV-positive OPSCC who have unfavourable outcomes [1]. Such patients may not be suitable for de-escalated treatment. Smoking has been associated with a higher risk of recurrence in this group [1, 11, 13, 14]. However, other traditional prognostic markers used in HPV-negative disease such as extracapsular extension (ECS) have not been found to be clearly prognostic in HPV-positive disease [15–18].
Histological grade has been considered as a possible prognostic marker in SCC, but assignation of grade in OPSCC has proven problematic. The traditional 3 tier grading system for SCC of well, moderately and poorly differentiated with keratinization being a key feature of better differentiated cases is not applicable to HPV-related disease. The characteristic non-keratinizing morphology of HPV-positive OPSCC has in the past generally been classified as poorly differentiated, contrary to the overall favourable prognosis of this tumour. More recently it had been proposed that given the close morphological resemblance between non-keratinizing OPSCC and the crypt epithelium from which it arises, that this tumour type is in fact best regarded as well differentiated. Subsequently the recommendation has emerged that grading of HPV-positive OPSCC be abandoned altogether, a view now endorsed by the WHO [19].
An alternative approach to histological grading of HPV-positive OPSCC based on tumour cell anaplasia and multinucleation has recently been proposed by Lewis et al. and shown in their cohort to be prognostically significant [20]. If these findings can be reproduced in other cohorts, they may indicate clinically important histological markers which could eventually be used in selecting patients with OPSCC for more aggressive treatment. With this in mind, the aim of the present study was to evaluate the potential utility of anaplasia and multinucleation as grading parameters in our series of patients with OPSCC and SCC of unknown primary site (SCCUP).
Methods
This study was a retrospective review of medical charts and histological slides of patients with newly diagnosed OPSCC or cervical squamous cell carcinoma of unknown primary site (SCCUP) who underwent primary surgical resection and/or neck dissection (ND) at our institution between the years 1998 and 2016 inclusive. Ethical approval was obtained a priori from the Cork Clinical Research Ethics Committee. Cases were identified from a database of all head and neck cancer patients at our hospital. Inclusion criteria were (1) newly diagnosed primary OPSCC or SCCUP patients managed with upfront surgery, (2) availability of original histology slides for review and (3) availability of blocks for p16 immunohistochemistry in cases not previously tested. Cases of SCCUP were included provided the pattern of metastasis was consistent with that of a primary mucosal SCC with involvement of upper deep cervical (level II/III) nodes. Exclusion criteria were (1) recurrence of primary tumour after previous non-surgical treatment, (2) delayed neck dissection not performed simultaneously with surgical resection of the primary, (3) inadequate patient follow-up, and (4) in case of SCCUP, previous history of skin SCC, or pattern of metastasis atypical for oropharyngeal primary site (involvement of parotid, posterior triangle, or submandibular triangle nodes). Histological data were extracted from pathology reports. Missing information was populated by re-review of pathology slides.
Tumour cell anaplasia and multinucleation were defined using the qualitative criteria proposed by Lewis et al. [20] and were assessed in both the primary and nodal metastatic disease. Anaplasia was defined as any × 400 magnification field with ≥ 3 nuclei with diameters equal to or wider than 5 lymphocyte nuclei (~ 25 μm) (Fig. 1). Multinucleation was defined as any × 400 magnification field with ≥ 3 tumour cells having multiple nuclei (Fig. 2). In addition, again using the criteria of Lewis et al. both parameters when present were quantified as focal, multifocal or diffuse. All tumour slides from all cases were scored independently by two pathologists (LF, CH) with discrepancies resolved by consensus agreement following re-review at a multiheaded microscope.
Fig. 1.
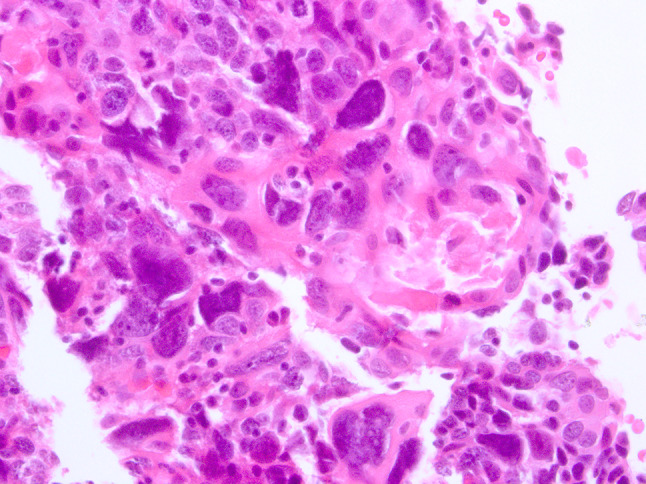
Anaplasia characterized by tumour nuclei with diameters equal to or wider than 5 lymphocyte nuclei (~ 25 μm)
Fig. 2.
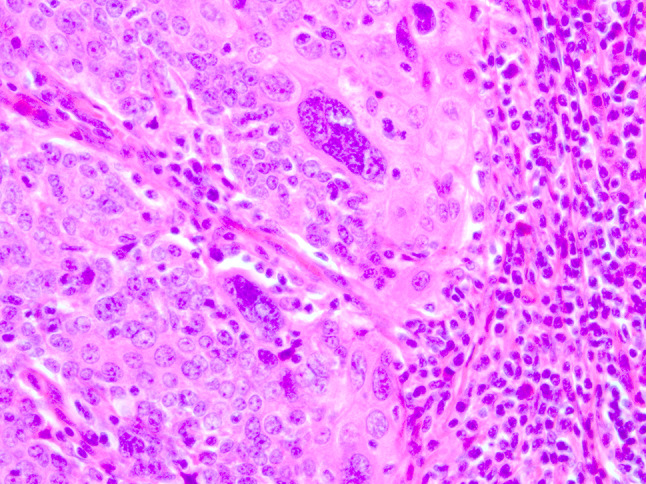
Tumour cell multinucleation
HPV status was assigned based on a combination of tumour morphology (keratinizing vs. non-keratinizing), p16 immunohistochemistry and additional HPV testing in selected cases. Morphology was determined by independent review of all cases by two pathologists (LF, CH). Histological features characteristic of keratinizing SCC included growth as irregular nests and cords with associated stromal desmoplasia, abundant keratinizing cytoplasm imparting a pink appearance on low power, keratin pearls, and prominent intercellular bridges. In contrast features characteristic of non-keratinizing SCC included growth as circumscribed nests and sheets of cells without associated desmoplasia, a blue appearance on low power due to a combination of high nuclear-cytoplasmic ratio, tumour infiltrating lymphocytes and lack of keratin, prominent tumour necrosis and lack of intercellular bridges [21, 22]. Disagreements were again resolved at a multiheaded microscope by consensus.
All cases underwent p16 immunohistochemical staining. For older cases in which p16 staining had not been done at the time of diagnosis paraffin blocks were retrieved and p16 immunohistochemistry performed on 3 µm sections. Antibody clone p16 CINtec® E6H4; Iso-type—IgG2a was used on the Ventana Benchmark Ultra platform, utilizing the Ventana Optiview detection system (Ventana Medical Systems, Tucson, AZ). Antigen retrieval employed solution CC1 (Tris/Borate/EDTA buffer pH 8.05–8.5) at 95 °C for 24 min. p16-positive cervical squamous carcinoma samples served as positive controls. Cases were evaluated by a pathologist (LF) and medical scientist (RW) with strong, diffuse, nuclear and cytoplasmic staining in greater than 70% of carcinoma cells scored as p16-positive [23].
Further HPV testing was performed in cases in which the histological subtype and the p16 status by immunohistochemistry were discordant (i.e. p16-positive keratinizing cases and p16-negative non-keratinizing cases) in accordance with former College of American Pathologist (CAP) guidelines [23]. For HPV detection the SPF10 primer set which targets a 65 bp region of the HPV L1 open reading frame and enables the amplification of at least 57 genital HPV types was used. The SPF10 PCR was performed in a final reaction volume of 20 µl containing 1 µl of the isolated DNA, 1 × PCR buffer II (Applied Biosystems), 1 mM MgCl2 (Applied Biosystems), 200 nM of deoxynucleoside triphosphate (dNTP) (Roche Applied Science), 300 nM of each of the SPF10 primers (Invitrogen), 1U of AmpliTaq Gold® (Applied Biosystems) and 10.4 µl of deionized water. The amplification conditions were 5 min at 95 °C for AmpliTaq Gold® activation, followed by 40 cycles of 30 s at 95 °C for denaturation, 45 s at 52 °C for annealing and 45 s at 72 °C for extension. The resulting amplicons were analysed using the FlashGel™ system (Lonza). Cases that were positive for HPV by SPF10 PCR were subsequently genotyped specifically for HPV 16 using an in-house developed HPV16 TaqMan PCR assay.
Statistical analysis was performed using XLSTAT version 2015.1.03 (Addinsoft). Recurrence-free survival (RFS) was defined by time period between date of treatment and first evidence of any disease recurrence. Disease-specific survival (DSS) was defined by time period between date of treatment and death, in cases with recurrent or persistent cancer. Continuous normally distributed data was compared using a Student’s t test. A Fisher exact test was used on 2 × 2 contingency tables. Survival analyses were performed using the Kaplan–Meier method, with p-values calculated using the log-rank test. Multivariate analysis was performed using backwards Cox proportional hazards modelling.
Results
During the study period, 106 patients with OPSCC or SCCUP underwent surgical resection of the primary tumour and/or neck disease, and analysis of slides for anaplasia and multinucleation. Two of these were subsequently excluded due to inadequate patient follow-up. Thus, the final study cohort comprised of 104 patients (81 with OPSCC and 23 with SCCUP). Sixty-four patients underwent resection of primary OPSCC, and 94 underwent neck dissection. Analysis of primary tumours for anaplasia and multinucleation was performed on 62 cases. Among patients undergoing neck dissection, 54 were performed simultaneous with primary tumour excision and 40 were performed as stand-alone procedures in patients with SCCUP (22) or undergoing non-surgical treatment of the primary OPSCC (18). Eight patients had pathologically negative necks leaving 86 patients with nodal metastases included in the analysis.
Clinicopathological and demographic features of the study population are shown in Table 1. Fifty-three patients were HPV-positive, and 51 HPV-negative. Overall 85 of 104 cases were concordant on morphology and p16 staining (34 keratinizing and p16-negative, 51 non-keratinizing and p16-positive). HPV PCR testing was performed on 18 of the 19 discordant cases. In all 18 cases, the final HPV status was congruent with the p16 status. The 1 discordant case which did not undergo further testing was p16-negative and was therefore classified as HPV-negative for the purposes of statistical analysis.
Table 1.
Clinicopathological and demographic features of study population
| Sex | |
| Male | 87 |
| Female | 17 |
| Primary site | |
| Tonsil | 51 |
| Base of tongue | 18 |
| Soft palate | 8 |
| Pharyngeal wall | 4 |
| Unknown | 23 |
| HPV status | |
| p16-positive | 53 |
| p16-negative | 51 |
| Subtype | |
| Keratinizing | 36 |
| Non-keratinizing | 68 |
| Smoking status | |
| Never | 20 |
| < 10 pack years | 8 |
| > 10 pack years | 86 |
| Unknown | 1 |
| Treatment group | |
| Primary resection only | 10 |
| Primary resection + neck dissection | 53 |
| Neck dissection only (non-surgical treatment of primary) | 18 |
| Neck dissection for unknown primary | 22 |
| T-classification (n = 62)* AJCC 8th edition | |
| T1 | 38 |
| T2 | 18 |
| T3 | 5 |
| T4 | 1 |
| N-classification AJCC 8h edition, p16-positive (n = 51) | |
| N0 | 3 |
| N1 | 37 |
| N2 | 11 |
| N-classification AJCC 8h edition, p16-negative (n = 43) | |
| N0 | 5 |
| N1 | 0 |
| N2a | 2 |
| N2b | 3 |
| N2c | 0 |
| N3b | 33 |
| Anaplasia in primary tumour (n = 62)* | |
| Yes | 37 |
| No | 25 |
| Anaplasia in metastasis (n = 86)** | |
| Yes | 54 |
| No | 32 |
| Multinucleation in primary tumour | |
| Yes | 26 |
| No | 36 |
| Multinucleation in metastasis | |
| Yes | 44 |
| No | 42 |
*T-stage of 62 patients with primary tumours undergoing analysis of anaplasia and multinucleation
**Information on 86 patients with cervical metastases, excluding 8 patients with pN0 necks
Anaplasia was found in the primary tumours of 37 (60%) patients, and within cervical metastases of 54 (63%) patients. Multinucleation was present in the primary tumours of 26 (42%) patients, and within cervical metastases of 44 (51%) patients. Overall, 69 (66%) patients had anaplasia in either the primary or metastatic tumour, and 52 (50%) patients had multinucleation in either the primary or metastatic tumour. In patients with both primary and nodal tumour for assessment the presence/absence of anaplasia and multinucleation was concordant at both sites in 78% and 82% of cases respectively. However, when present the extent of anaplasia and multinucleation (focal vs. multifocal/diffuse) showed a high frequency of discordance (63.6% and 55.6% respectively). For this reason and due to small numbers in each subgroup further subanalysis was not performed (see Supplementary Table 1—Online Resource 1). Clinicopathological features according to presence of anaplasia or multinucleation are shown in Table 2. There was a higher frequency of both anaplasia (p = 0.005) and multinucleation (p = 0.03) among males. There was also a higher incidence of anaplasia, but not multinucleation, among smokers (p = 0.003) and HPV-negative patients (p = 0.04).
Table 2.
Correlations between anaplasia and multinucleation and clinical and pathological data
| Variable | Anaplasia present (n = 69) | Anaplasia absent (n = 35) | p value | Multinucleation present (n = 52) | Multinucleation absent (n = 52) | p value | |
|---|---|---|---|---|---|---|---|
| Sex | Male | 63 | 24 | 0.005 | 48 | 39 | 0.03 |
| Female | 6 | 11 | 4 | 13 | |||
| Smoking status | Never | 10 | 10 | 0.003a | 11 | 9 | 0.83 |
| < 10pkyr | 3 | 5 | 2 | 6 | |||
| > 10pkyr | 56 | 20 | 39 | 37 | |||
| Pathological nodal status (n = 94) | N0 | 115 | 83 | 0.71 | 92 | 6 | 0.27 |
| N+ | 59 | 2827 | 45 | 41 | |||
| Morphology | Non-keratinizing | 42 | 26 | 0.19 | 34 | 34 | > 0.99 |
| Keratinizing | 27 | 9 | 18 | 18 | |||
| p16/HPV status | Positive | 30 | 23 | 0.04 | 29 | 24 | 0.43 |
| Negative | 39 | 12 | 23 | 28 | |||
ap-value given for comparison between never/< 10 pack year smokers versus > 10 pack year smokers
Mean and median follow up for the entire cohort were 62.9 and 54 months, respectively. 36 patients developed recurrence, and 31 died from cancer. Among surviving patients, 50/57 were followed for at least 3 years.
The impact of clinicopathological variables on recurrence free survival (RFS) and disease specific survival (DSS) is shown in Table 3 for patients undergoing excision of primary tumour, and in Table 4 for patients undergoing excision of cervical metastases. In Table 5 impact for clinicopathological variables is shown for patients with cancers of known primary site undergoing neck dissection.
Table 3.
Impact of clinical and pathological factors on survival outcomes for patients undergoing primary tumour excision (n = 62)
| Variable | 3-year RFS (%) | p-value | 3-year DSS (%) | p-value |
|---|---|---|---|---|
| p-16 status | ||||
| p-16 positive | 93.8 | < 0.0001 | 97.0 | < 0.0001 |
| p-16 negative | 47.6 | 51.3 | ||
| Anaplasia | ||||
| Anaplasia present | 67.5 | 0.25 | 70.6 | 0.16 |
| Anaplasia absent | 83.2 | 87.4 | ||
| Multinucleation | ||||
| Multinucleation present | 79.8 | 0.72 | 79.8 | 0.99 |
| Multinucleation absent | 70.2 | 76.1 | ||
| Smoking status | ||||
| < 10 pack years | 88.2 | 0.03 | 100 | 0.005 |
| > 10 pack years or never | 68.4 | 67.7 | ||
| T-classification | ||||
| T1 | 83.0 | 0.04 | 85.5 | 0.04 |
| T2–4 | 59.8 | 64.7 | ||
RFS recurrence-free survival, DSS disease specific survival
Table 4.
Impact of clinical and pathological factors on survival outcomes for patients undergoing neck dissection (n = 86)
| Variable | 3-year RFS (%) | p-value | 3-year DSS | p-value |
|---|---|---|---|---|
| p-16 status | ||||
| p-16 positive | 91.5 | < 0.0001 | 95.7 | < 0.0001 |
| p-16 negative | 37.7 | 32.0 | ||
| Anaplasia | ||||
| Anaplasia present | 65.4 | 0.77 | 64.8 | 0.47 |
| Anaplasia absent | 73.9 | 74.0 | ||
| Multinucleation | ||||
| Multinucleation present | 69.9 | 0.99 | 68.7 | 0.93 |
| Multinucleation absent | 67.9 | 67.9 | ||
| Smoking status | ||||
| < 10 pack years | 95.8 | < 0.0001 | 100 | < 0.0001 |
| > 10 pack years or never | 57.4 | 55.7 | ||
| Extracapsular spread | ||||
| ECS absent | 89.1 | 0.006 | 92.4 | 0.001 |
| ECS present | 58.5 | 56.7 | ||
| Number of positive nodes | ||||
| > 4 nodes positive | 58.9 | 0.37 | 56.3 | 0.10 |
| ≤ 4 nodes positive | 73.0 | 74.2 | ||
RFS recurrence-free survival, DSS disease specific survival
Table 5.
Impact of clinical and pathological factors on survival outcomes for patients undergoing neck dissection with known primary tumour site (n = 64)
| Variable | 3-year RFS (%) | p-value | 3-year DSS (%) | p-value |
|---|---|---|---|---|
| p-16 status | ||||
| p-16 positive | 94.7 | < 0.0001 | 94.6 | < 0.0001 |
| p-16 negative | 36.9 | 32.9 | ||
| Anaplasia | ||||
| Anaplasia present | 70.0 | 0.68 | 64.8 | 0.29 |
| Anaplasia absent | 80.4 | 80.0 | ||
| Multinucleation | ||||
| Multinucleation present | 69.0 | 0.51 | 65.0 | 0.43 |
| Multinucleation absent | 75.6 | 75.2 | ||
| Smoking status | ||||
| < 10 pack years | 100 | 0.001 | 100 | 0.001 |
| > 10 pack years or never | 60.8 | 58.1 | ||
| Extracapsular spread | ||||
| ECS absent | 90.2 | 0.03 | 90.2 | 0.01 |
| ECS present | 63.5 | 60.8 | ||
| Number of positive nodes | ||||
| > 4 nodes positive | 59.5 | 0.31 | 55.9 | 0.09 |
| ≤ 4 nodes positive | 78.3 | 77.9 | ||
RFS recurrence-free survival, DSS disease specific survival
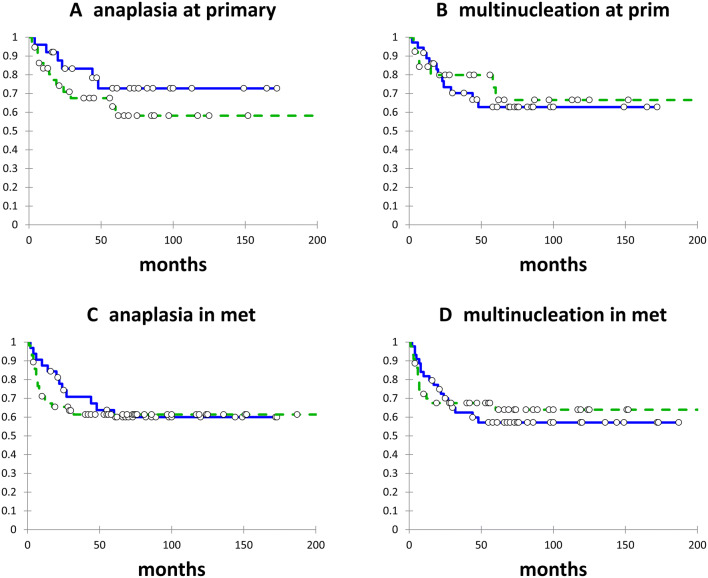
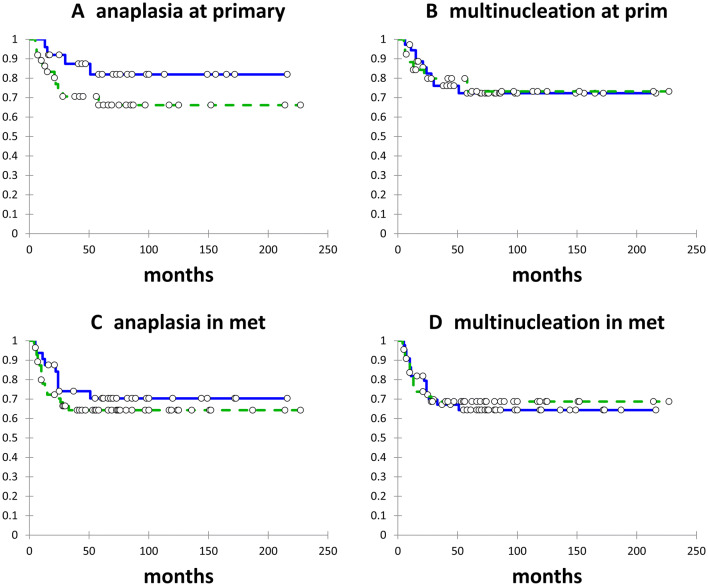
Among patients undergoing primary tumour excision, HPV-negative status, > 10 pack year smoking history, and T2–4 classification had significant adverse impact on both RFS and DSS. On multivariate analysis, only HPV-negative status was significant for RFS (p < 0.0001) and DSS (p = 0.02). Neither anaplasia nor multinucleation within the primary tumour had any significant impact on either RFS (Fig. 3a, b) or on DSS (Fig. 4a, b).
Fig. 3.
Recurrence-free survival (RFS) according to anaplasia at primary site (a); multinucleation at primary site (b); anaplasia in metastases (c); and multinucleation in metastases (d). No anaplasia/multinucleation = solid line; anaplasia/multinucleation = dashed line
Fig. 4.
Disease specific survival (DSS) according to anaplasia at primary site (a); multinucleation at primary site (b); anaplasia in metastases (c); and multinucleation in metastases (d). No anaplasia/multinucleation = solid line; anaplasia/multinucleation = dashed line
Among 86 pN+ patients undergoing excision of cervical metastases, HPV-negative status, > 10 pack year smoking history, and extracapsular spread (ECS) were significant adverse factors for both RFS and DSS. On multivariate analysis, only HPV-negative status remained significant (p < 0.0001 for both RFS and DSS). Neither anaplasia nor multinucleation within the cervical metastases had any significant impact on either RFS (Fig. 3c, d) or on DSS (Fig. 4c, d).
When we repeated the analysis on pN+ cases with known primary tumour site undergoing neck dissection, the results were very similar to those found for all pN+ cases (Table 5). Once again, only HPV-status was significant on multivariate analysis (p < 0.0001 for both RFS and DSS).
We also analyzed the impact of anaplasia and multinucleation on the HPV-positive and HPV-negative cohorts separately. Thirty-four HPV-positive patients underwent primary OPSCC excision, while 48 underwent excision of cervical metastatic disease. Twenty-nine HPV-negative patients underwent primary tumour excision, with 38 undergoing excision of nodal metastases. Neither anaplasia nor multinucleation had any impact on RFS or DSS in any of these cohorts (Supplementary Tables 2 and 3—Online Resource 2 and Online Resource 3 respectively).
Discussion
Five-year survival rates amongst patients with HPV-positive tumours range from approximately 75 to 80%. Thus, the vast majority of patients have excellent oncological outcomes, and much of the current research emphasis is on techniques to de-escalate treatment in selected patients in a bid to reduce treatment related morbidity [12, 24]. However, there remains a significant minority that does not share the improved prognosis of the overall group [1]. There are currently no established molecular markers that correlate with survival in this patient population. Traditional predictors of poor outcome in HPV-negative OPSCC such as positive surgical margins and extracapsular spread of nodal metastatic disease do not carry the same significance in HPV-positive cases [15–18, 25–28]. Advanced T stage and in some, but not all studies, N stage, have been shown to predict for reduced survival in HPV-positive OPSCC [1, 4, 9, 11, 29]. Smoking is also known to compromise outcome [1, 11, 13, 14] and more recently low levels of tumour infiltrating lymphocytes have been associated with worse survival [30, 31]. However, other robust prognosticators, which can easily be incorporated into routine clinical practice will be required to accurately identify patients who may benefit from adjuvant therapy.
Tumour cell anaplasia and multinucleation have been proposed as having potential utility as grading parameters in HPV-driven disease. In a study of 149 surgically resected OPSCCs by Lewis et al. the presence of anaplasia and multinucleation was assessed in both the primary tumour and nodal metastatic disease. They demonstrated that these ‘grading’ parameters were independent predictors of recurrence and poor survival in the overall cohort and also within the p16-positive group [20].
In the present study, we wished to investigate whether presence of anaplasia and multinucleation among patients in our cohort had prognostic significance. We used the same qualitative criteria as those proposed by Lewis et al. with slides of all cases reviewed by two independent pathologists, and discordances resolved at multiheaded microscope by consensus. Among our series, the incidence of anaplasia was somewhat higher than in Lewis’s study (67% vs. 43%), and the incidence of multinucleation roughly similar (52% vs. 48%). It is probable that the higher incidence of anaplasia in our series was related to the higher proportion of HPV-negative patients (49% vs. 14%) and smokers (81% vs. 64%, including “ever smokers”). Both of these factors were found in our series to be associated with higher incidence of anaplasia. Lewis et al. also demonstrated significant correlation between presence of anaplasia and HPV negativity and a borderline correlation for smoking adding further weight to this conclusion [20].
In contrast to the findings of Lewis et al. we did not find the presence of either anaplasia or multinucleation to be predictive of disease recurrence or poor survival in OPSCC. However, given the small number of events among HPV-positive patients (there were only seven cancer recurrences, and two deaths due to cancer), it is possible that our study was underpowered to detect any significant effect for these variables in the HPV-positive population. In contrast, the study of Lewis et al. contained a significantly larger number of HPV-positive patients (128 vs. 55), although a small number of HPV-negative cases (26 vs. 53).
Jing et al. evaluated the significance of anaplasia in OPSCC. Their study cohort comprised of 129 patients who underwent primary surgical resection of whom 91.5% had HPV positive disease. Frequent monster tumour nuclei (p = 0.03) and high tumour pleomorphism (p = 0.0001) both correlated significantly with worse overall survival (published in abstract form) [32]. However, we are not the only group to fail to demonstrate an association between anaplasia and multinucleation in OPSCC and patient outcome. In a large cohort of 390 OPSCC patients of whom 342 were p16-positive and with a minimum of 5 years follow-up, Thompson et al. found no correlation between either anaplasia or multinucleation and survival (published in abstract form) [33].
There are some limitations to our study. Firstly, our patient cohort was heterogenous, including patients undergoing simultaneous surgical resection of the primary OPSCC and neck, surgical resection of the primary site alone, as well as patients with OPSCC and SCCUP undergoing up-front neck dissection, with the primary site treated non-surgically. The reason for this was in order to achieve as large a cohort as possible of patients with adequate follow-up, treated during an era when most patients were undergoing non-surgical treatment of OPSCC. However, we believe that the potential impact of this heterogeneity was minimized through our separate analyses of anaplasia and multinucleation in the primary tumour (which included only patients undergoing primary resection), and in cervical metastases (which included only patients undergoing ND). A second potential limitation was our approach to HPV testing in SCCUP. In the present study, we adhered to current CAP guidelines, which state that p16 staining alone is adequate in metastatic SCC to level II and III neck nodes of unknown primary with concordant histology. However, metastatic SCC to other neck levels requires confirmatory HPV testing [34]. These latter cases of metastases to other neck levels were excluded from our study. Due to resource limitations, confirmatory HPV testing was performed only in cases with discordance between morphology and p16 immunohistochemistry. However, the CAP method has been criticized based on the relatively high incidence of p16-positivity (up to 29.5%) in metastatic skin SCC [35], as well as the somewhat moderate specificity of p16 immunohistochemistry for transcriptionally active HPV (approximately 80%) [36]. A third limitation is the possibility of inter-observer variation in the interpretation of study parameters of anaplasia and multinucleation. This was limited by using the exact criteria described by Lewis et al. and having two independent pathologists review the slides. Finally, because of the small number of events in our HPV-positive group, our study may have remained too underpowered to detect any significant prognostic impact for the study variables.
In conclusion, we did not find anaplasia or multinucleation to be significant predictors of outcome in OPSCC in this study, although because of the excellent prognosis and low number of events in the HPV-positive group, our study may have been underpowered to detect same in this cohort. Further adequately powered studies are needed to investigate possible markers of poor prognosis in HPV-positive OPSCC to facilitate appropriate selection of patients for treatment de-escalation versus adjuvant therapy.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Funding
Funding for the p16 staining was provided by the Head and Neck Oncology Fund, South Infirmary Victoria University Hospital. The funding source had no input into the study design, collection or analysis of data, interpretation, writing of the manuscript, or decision to submit the article for publication.
Compliance with Ethical Standards
Conflict of interest
No conflict of interest to disclose
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Ang KK, Harris J, Wheeler R, et al. Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med. 2010;363:24–35. doi: 10.1056/NEJMoa0912217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Dayyani F, Etzel CJ, Liu M, et al. Meta-analysis of the impact of human papillomavirus (HPV) on cancer risk and overall survival in head and neck squamous cell carcinomas (HNSCC) Head Neck Oncol. 2010;2:15. doi: 10.1186/1758-3284-2-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Fakhry C, Westra WH, Li S, et al. Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J Natl Cancer Inst. 2008;100:261–269. doi: 10.1093/jnci/djn011. [DOI] [PubMed] [Google Scholar]
- 4.Fischer CA, Zlobec I, Green E, et al. Is the improved prognosis of p16 positive oropharyngeal squamous cell carcinoma dependent of the treatment modality? Int J Cancer. 2010;126:1256–1262. doi: 10.1002/ijc.24842. [DOI] [PubMed] [Google Scholar]
- 5.Hoffman M, Gorogh T, Gottschlich S, et al. Human papillomavirus in head and neck cancer: 8 year-survival-analysis of 73 patients. Cancer Lett. 2005;218:199–206. doi: 10.1016/j.canlet.2004.09.027. [DOI] [PubMed] [Google Scholar]
- 6.Hong AM, Dobbins TA, Lee CS, et al. Human papillomavirus predicts outcome in oropharyngeal cancer in patients treated primarily with surgery or radiation therapy. Br J Cancer. 2010;103:1510–1517. doi: 10.1038/sj.bjc.6605944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Posner MR, Lorch JH, Goloubeva O, et al. Survival and human papillomavirus in oropharynx cancer in TAX 324: a subset analysis from an international phase III trial. Ann Oncol. 2011;22:1071–1077. doi: 10.1093/annonc/mdr006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ragin CC, Taioli E. Survival of squamous cell carcinoma of the head and neck in relation to human papillomavirus infection: review and meta-analysis. Int J Cancer. 2007;121:1813–1820. doi: 10.1002/ijc.22851. [DOI] [PubMed] [Google Scholar]
- 9.Rich JT, Milov S, Lewis JS, Jr, et al. Transoral laser microsurgery (TLM) +⁄- adjuvant therapy for advanced stage oropharyngeal cancer: outcomes and prognostic factors. Laryngoscope. 2009;119:1709–1719. doi: 10.1002/lary.20552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Rischin D, Young RJ, Fisher R, et al. Prognostic significance of p16INK4A and human papillomavirus in patients with oropharyngeal cancer treated on TROG 02.02 phase III trial. J Clin Oncol. 2010;28:4142–4148. doi: 10.1200/JCO.2010.29.2904. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.O’Sullivan B, Huang SH, Siu LL, et al. Deintensification candidate subgroups in human papillomavirus-related oropharyngeal cancer according to minimal risk of distant metastasis. J Clin Oncol. 2013;31:543–550. doi: 10.1200/JCO.2012.44.0164. [DOI] [PubMed] [Google Scholar]
- 12.Masterson L, Moualed D, Liu ZW, et al. De-escalation treatment protocols for human papillomavirus-associated oropharyngeal squamous cell carcinoma: a systematic review and meta-analysis of current clinical trials. Eur J Cancer. 2014;50:2636–2648. doi: 10.1016/j.ejca.2014.07.001. [DOI] [PubMed] [Google Scholar]
- 13.Hafkamp HC, Manni JJ, Haesevoets A, et al. Marked differences in survival rate between smokers and nonsmokers with HPV 16-associated tonsillar carcinomas. Int J Cancer. 2008;122:2656–2664. doi: 10.1002/ijc.23458. [DOI] [PubMed] [Google Scholar]
- 14.Lindquist D, Romanitan M, Hammarstedt L, et al. Human papillomavirus is a favourable prognostic factor in tonsillar cancer and its oncogenic role is supported by the expression of E6 and E7. Mol Oncol. 2007;1:350–355. doi: 10.1016/j.molonc.2007.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Iyer NG, Dogan S, Palmer F, et al. Detailed analysis of clinicopathologic factors demonstrate distinct difference in outcome and prognostic factors between surgically treated HPV-positive and negative oropharyngeal cancer. Ann Surg Oncol. 2015;22:4411–4421. doi: 10.1245/s10434-015-4525-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kharytaniuk N, Molony P, Boyle S, et al. Association of extracapsular spread with survival according to human papillomavirus status in oropharynx squamous cell carcinoma and carcinoma of unknown primary site. JAMA Otolaryngol Head Neck Surg. 2016;142:683–690. doi: 10.1001/jamaoto.2016.0882. [DOI] [PubMed] [Google Scholar]
- 17.Lewis JS, Jr, Carpenter DH, Thorstad WL, et al. Extracapsular extension is a poor predictor of disease recurrence in surgically treated oropharyngeal squamous cell carcinoma. Mod Pathol. 2011;24:1413–1420. doi: 10.1038/modpathol.2011.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Maxwell JH, Ferris RL, Gooding W, et al. Extracapsular spread in head and neck carcinoma: impact of site and human papillomavirus status. Cancer. 2013;119:3302–3308. doi: 10.1002/cncr.28169. [DOI] [PubMed] [Google Scholar]
- 19.Chan JKC, Slootweg P. Introduction. In: El-Naggar AK, Chan JKC, Grandis JR, Takata T, Slootweg PJ, editors. WHO Classification of Head and Neck Tumours. Geneva: WHO Press; 2017. pp. 64–65. [Google Scholar]
- 20.Lewis JS, Jr, Scantlebury JB, Luo J, et al. Tumour cell anaplasia and multinucleation are predictors of disease recurrence in oropharyngeal squamous cell carcinoma, including among just the human papillomavirus-related cancers. Am J Surg Pathol. 2012;36:1036–1046. doi: 10.1097/PAS.0b013e3182583678. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.El-Mofty SK, Lu DW. Prevalence of human papillomavirus type 16 DNA in squamous cell carcinoma of the palatine tonsil, and not the oral cavity, in young patients: a distinct clinicopathologic and molecular disease entity. Am J Surg Pathol. 2003;27:1463–1470. doi: 10.1097/00000478-200311000-00010. [DOI] [PubMed] [Google Scholar]
- 22.El-Mofty SK, Patil S. Human papillomavirus (HPV)-related oropharyngeal nonkeratinizing squamous cell carcinoma: characterization of a distinct phenotype. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;101:339–345. doi: 10.1016/j.tripleo.2005.08.001. [DOI] [PubMed] [Google Scholar]
- 23.Carlson DL, Barnes L, Chan J, et al. Protocol for the examination of specimens from patients with carcinomas of the pharynx. http://www.cap.org/apps/docs/committees/cancer/cancer_protocols/ 2012/Pharynx_12protocol.pdf.
- 24.Chen AM, Felix C, Wang PC, et al. Reduced-dose radiotherapy for human papillomavirus-associated squamous-cell carcinoma of the oropharynx: a single-arm, phase 2 study. Lancet Oncol. 2017;18:803–811. doi: 10.1016/S1470-2045(17)30246-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Haughey BH, Sinha P. Prognostic factors and survival unique to surgically treated p16 + oropharyngeal cancer. Laryngoscope. 2012;122(Suppl 2):S13–S33. doi: 10.1002/lary.23493. [DOI] [PubMed] [Google Scholar]
- 26.Kaczmar JM, Tan KS, Heitjan DF, et al. HPV-related oropharyngeal cancer: risk factors for treatment failure in patients managed with primary transoral robotic surgery. Head Neck. 2016;38:59–65. doi: 10.1002/hed.23850. [DOI] [PubMed] [Google Scholar]
- 27.Molony P, Kharytaniuk N, Boyle S, et al. Impact of positive margins on outcomes of oropharyngeal squamous cell carcinoma according to p16 status. Head Neck. 2017;39:1680–1688. doi: 10.1002/hed.24824. [DOI] [PubMed] [Google Scholar]
- 28.O’Sullivan B, Lydiatt WM, Haughey BH, Brandwein-Gensler MS, Glastonbury CM, Shah JP. HPV mediated (p16 +) oropharyngeal cancer. In: Amin MB, editor. AJCC Cancer Staging Manual. 8. New York: Springer; 2017. [Google Scholar]
- 29.Lewis JS, Jr, Thorstad WL, Chernock RD, et al. p16 positive oropharyngeal squamous cell carcinoma:an entity with a favorable prognosis regardless of tumor HPV status. Am J Surg Pathol. 2010;34:1088–1096. doi: 10.1097/PAS.0b013e3181e84652. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Nordfors C, Grun N, Tertipis N, et al. CD8 + and CD4 + tumour infiltrating lymphocytes in relation to human papillomavirus status and clinical outcome in tonsillar and base of tongue squamous cell carcinoma. Eur J Cancer. 2013;49:2522–2530. doi: 10.1016/j.ejca.2013.03.019. [DOI] [PubMed] [Google Scholar]
- 31.Ward MJ, Thirdborough SM, Mellows T, et al. Tumour-infiltrating lymphocytes predict for outcome in HPV-positive oropharyngeal cancer. Br J Cancer. 2014;110:489–500. doi: 10.1038/bjc.2013.639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Jing J, Varier I, Dong J, et al. Prognostic significance of histologic features in HPV-associated tonsillar squamous cell carcinoma. Mod Pathol. 2016;29(Suppl 2):323A–324A. [Google Scholar]
- 33.Thompson LD, Herrera H, Iganej S, et al. Histologically significant features in 390 oropharyngeal squamous cell carcinomas. Mod Pathol. 2018;31(Suppl 2):488A. [Google Scholar]
- 34.Lewis JS, Jr, Beadle B, Bishop JA, et al. Human papillomavirus testing in head and neck carcinomas: guideline from the College of American Pathologists. Arch Pathol Lab Med. 2018;142:559–597. doi: 10.5858/arpa.2017-0286-CP. [DOI] [PubMed] [Google Scholar]
- 35.Satgunaseelan L, Chia N, Suh H, et al. p16 expression in cutaneous squamous cell carcinoma of the head and neck is not associated with integration of high risk HPV DNA or prognosis. Pathology. 2017;49:494–498. doi: 10.1016/j.pathol.2017.04.002. [DOI] [PubMed] [Google Scholar]
- 36.Smeets SJ, Hessselink AT, Speel EJ, et al. A novel algorithm for reliable detection of human papilloma virus in paraffin embedded head and neck cancer specimens. Int J Cancer. 2007;121:2465–2472. doi: 10.1002/ijc.22980. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.